Abstract
The mucosal immune system mediates contact between the host, and the trillions of microbes that symbiotically colonize the gastrointestinal tract. Failure to tolerate the antigens within this “extended self” can result in inflammatory bowel disease (IBD). Within the adaptive immune system, the most significant cells modulating this interaction are Foxp3+ regulatory T (Treg) cells. Treg cells can be divided into two primary subsets: “natural” Treg (nTreg) cells, and “adaptive” or “induced” Treg (iTreg). Recent research suggests that these subsets serve to play both independent and synergistic roles in mucosal tolerance. Studies from both mouse models and human patients suggest defects in Treg cells can play distinct causative roles in IBD. Numerous genetic, microbial, nutritional, and environmental factors that associate with IBD may also affect Treg cells. In this review we summarize the development and function of Treg cells, and how their regulatory mechanisms may fail, leading to a loss of mucosal tolerance. We discuss both animal models and studies of IBD patients suggesting Treg cell involvement in IBD, and consider how Treg cells may be used in future therapies.
Keywords: induced regulatory T cell, natural regulatory T cell, colitis, Crohn’s disease, Foxp3
Introduction
The gastrointestinal tract is the largest lymphoid organ in the body. This complex network consists of specialized epithelial cells, lymphatic vessels, dendritic cells, lymphocytes, and secondary lymphoid structures. The gut immune system mediates contact between the host and the microbes that symbiotically colonize this mucosal surface (termed the microbiota), while protecting the host against pathological invasion. Exposure to nutritional, microbial, and host antigens in the intestine creates an “extended self” that varies dynamically in this complex environment. Thus, the division between “self” and “non-self” is often indistinct, and multiple regulatory mechanisms have evolved to restrain misdirected immune responses. Failure of these regulatory pathways and/or imbalances in the composition of the microbiota can result in pathologic inflammatory processes such as inflammatory bowel disease (IBD), which primarily consists of ulcerative colitis (UC) and Crohn’s disease (CD).
Numerous tissues and specialized cells are involved in maintaining mucosal tolerance in the intestine. Paneth cells at the base of the intestinal crypts of the small intestine produce antimicrobial peptides and lectins, limiting direct contact between the epithelia and microbiota.1 M cells of the follicle-associated epithelium translocate luminal antigens across the epithelium into the dome region of Peyer’s patches (PP) and isolated lymphoid follicles (ILF) for uptake and processing by dendritic cells (DCs).2 Recent data suggests that a subset of goblet cells in the small intestine may also deliver antigens to DCs.3 The DCs within the gut lamina propria and secondary lymphoid tissues encounter and process the antigen. Alternatively, DC within the lamina propria can sample luminal antigens directly by extending dendrites between tight junctions of the epithelia, into the lumen. After encountering antigen, DCs migrate to T cell zones in the PP or mesenteric lymph node (mLN) to present antigen and activate effector T cells. Activated CD4 effector T cells can stimulate B cells, resulting in IgA-producing plasma cells. IgA plasma cells and activated effector T cells then populate the lamina propria, resulting in a low-grade inflammation that appears to be the normal state of the intestine.2
IBD appears to be a primarily T cell-mediated disease. Indeed, CD4 T cells are responsible for orchestrating the excessive inflammation involved in IBD.4, 5 However, T cells also appear to play the largest role in establishing and maintaining immunological tolerance by suppressing excessive or abnormal immune responses. Numerous subtypes of T cells appear to display immunosuppressive function in the gastrointestinal tract. Classical TCRαβ T cells such as CD8+ CTLs and CD4+ Tr1 cells exhibit immune suppression in the gut.6, 7 In addition, more atypical CD8αα and TCRγδ+ T cells have been shown to exhibit protection against mouse models of colitis.6, 7 Other innate-like T lymphocytes with invariant TCRs such as iNKT and mucosal-associated invariant T (MAIT) cells may also play immunoregulatory roles in the GI tract. Nonetheless, the most significant of the immunosuppressive T lymphocytes are TCRαβ CD4 + regulatory T (Treg) cells that are characterized by the expression of the transcription factor Foxp3.8 This review will focus on these CD4+ Foxp3+ Treg cells.
Global failure of Treg cell development via Foxp3 deficiency results in the lethal multi-system autoimmune disease immunodysregulation polyendocrinopathy enteropathy X-linked syndrome (IPEX) that underscores the essential nature of Treg cells in immune regulation. The most commonly affected organ in IPEX is the intestine, highlighting the essential role for Treg cells in the gastrointestinal tract. In addition to IPEX, other genetic deficiencies suggest the importance of Treg in IBD. Mutations in WASP, CD25, and IL-10 all lead to abnormal Treg cell numbers and/or function, and also increase an individual’s risk for IBD.9 Although these genetic defects suggest a role for Treg cells in human IBD, much of the mechanistic evidence has come from mouse models of disease. Mouse models of IBD can be separated into 4 relatively distinct categories: 1) chemically induced barrier disruption models, 2) spontaneous models due to genetic deficiency, 3) overexpression of inflammatory mediators, and 4) lymphopenic T cell transfer models (Table 1).
Table 1.
Treg cell involvement in commonly used mouse models of IBD
| Category of IBD Model |
Model Name | Role for Regulatory T cells |
References |
|---|---|---|---|
| 1) Barrier disruption | DSS (chemical-induced) | - Absence of Treg or TGF-β1 signaling in Treg cells increases severity | 20,217 |
| TNBS/DNBS/Oxazolone (hapten-induced) | - Ethanol in absence of TNBS leads to resistance | 218 | |
| - B. fragilis PSA increases iTreg cells leading to resistance | 146 | ||
| - Treg cells transiently decrease then home to intestine during remission | 219 | ||
| 2) Genetic deficiency | IL-10 deficiency | - Treg-produced IL-10 is necessary to prevent intestinal inflammation | 75, 76, 77 |
| SAMP/YitFc (PPARG mutation) | - Treg cells from these mice are defective in suppressing colitis upon transfer | 16, 220 | |
| Mdr1 deficiency | - mutation leads to decreased in vivo and in vitro differentiation of iTreg | 17 | |
| 3) Overexpression of inflammatory mediators | TNFΔARE (TNF-α overexpression) | - increased differentiation of iTreg attenuates disease | 221 |
| STAT4 overexpression | - unknown, Treg cells hypothesized to be normal | 21 | |
| 4) Lymphopenic T cell transfer | Naïve Tconv transfer into SCID/ RAG−/− mice | - co-transfer of nTreg cells prevents disease | 19 |
| - nTreg + in vivo or in vitro iT reg cells treat disease | 38 | ||
Commonly used chemically induced barrier disruption models include dextran sulfate sodium (DSS) and hapten-induced colitis via 2,4,6-trinitrobenzene sulfonic acid (TNBS) and oxazolone. These chemicals disrupt the epithelium, allowing microbial and immunogenic molecules to penetrate and initiate an inflammatory response.10, 11 Spontaneous genetic models include IL-10 deficient mice,12 defective PPARG expression in SAMP1/YitFc mice,13 and Mdr1 deficiency.14 Numerous other genetic deficiencies also lead to intestinal inflammation, but IL10, PPARG, and MDR1 are particularly notable in that they play roles in Treg cell number and/or function and are also human IBD susceptibility loci.11, 15–17 Other mouse models utilize gene overexpression to model intestinal inflammation. Such models include TNF-α overexpression in TNFΔARE mice and STAT4 overexpression.11
Perhaps the most compelling data on the role of Treg cells in IBD has come from the T cell transfer model of colitis. In this model, naïve CD4+ T cells, depleted of Treg cells, (typically CD45RBhi) are adoptively transferred into mice lacking B and T lymphocytes (SCID or RAG-deficiency). These effector T cells proliferate and become activated in response to bacterial antigens in the intestine, resulting in inflammation and colitis. Disease can be both prevented and treated via elimination of the microbiota or co-transfer of Treg cells.18, 19 This model offers the distinct advantage that the pathogenic and regulatory T cell subsets can be genetically targeted independently. Thus, several important inflammatory and suppressive mechanisms have been identified through transfer of Treg or effector T cells (Teff ) from mutant mice.6
Numerous studies utilizing mouse models support a role for Treg cells in IBD. In barrier models Treg cells localize to the intestine and mLN in acute disease, and an absence of Treg cells exacerbates disease.6, 20 A mild breech of the intestinal barrier via ethanol in the absence of TNBS does not result in colitis, suggesting regulatory responses predominate.6 Many genetic models of spontaneous IBD involve genes affecting Treg function, and in treatment models Treg cells home to the intestine to resolve inflammation.6, 21 Two subsets of Treg cells have been described, “natural” Treg (nTreg) cells and “induced” or “adaptive” Treg (iTreg) cells. Both nTreg and iTreg subsets are characterized by the expression of Foxp3, and Foxp3 expression is necessary for their overall fate and function.8, 22 The nTreg and iTreg subsets are largely distinguished by their developmental origin and appear to play non-redundant roles enforcing gastrointestinal tolerance.
Natural regulatory T cells
“Natural” Treg (nTreg) cells arise as a discrete and largely stable lineage originating in the thymus. Foxp3+ cells are first detectable in a small fraction of CD4+ CD8+ double positive thymocytes, and are subsequently more frequent in CD4+ single positive thymocytes.8 The nTreg subset exhibits a TCR repertoire that is distinct from Foxp3− conventional T cells (Tconv) and from iTreg cells. Data shows that the TCRs of nTreg cells may have increased affinities for self-peptides.23–26 Moreover, mutations in MHC and TCR signaling suggest that a strong TCR signal is required for nTreg development. TCR signaling activates the NF-κB pathway, and several conditional mutations in NF-κB members show nTreg defects.27 NF-κB family member c-Rel binds directly to the conserved non-coding sequence 3 (CNS3) region of the Foxp3 promoter, providing a link between TCR signaling and Foxp3 expression.28
TCR-ligand affinity alone does not determine Foxp3 expression.28 nTreg cells also require IL-2 for their development and maintenance. In fact, regulatory T cells were first identified by their elevated expression of the high-affinity IL-2 receptor CD25 (IL-2Rα). Mice lacking IL-2 signaling via antibody neutralization or genetic deficiency of IL-2 or IL-2 receptors show nTreg deficiencies and spontaneous autoimmune disease including IBD.29–32 CD25 deficiency in human patients also results in an IPEX-like disease, supporting the importance of IL-2 signaling in nTreg development.33 Downstream signaling from IL-2 receptors is mediated through STAT5, and STAT5 binds to the conserved non-coding sequence 2 (CNS2) enhancer region of the Foxp3 gene. This suggests that IL-2 signaling directly promotes the initial expression and/or maintenance of Foxp3 to support nTreg development.27
In addition to IL-2 signaling, normal Treg development in the thymus requires autophagy. Transplant of autophagy-deficient thymi from Atg−/− mice results in abnormal T cell selection and spontaneous colitis.34 This suggests that a subset of intestinal auto-antigens may require autophagy for proper presentation and deletion in the thymus. In fact, autophagy genes are a commonly identified class of IBD susceptibility alleles. Thus, there may be a role for autophagy defects in clinical disease, though Treg specificity and function in IBD patients with autophagy mutations has not been investigated.
Adaptive or induced regulatory T cells
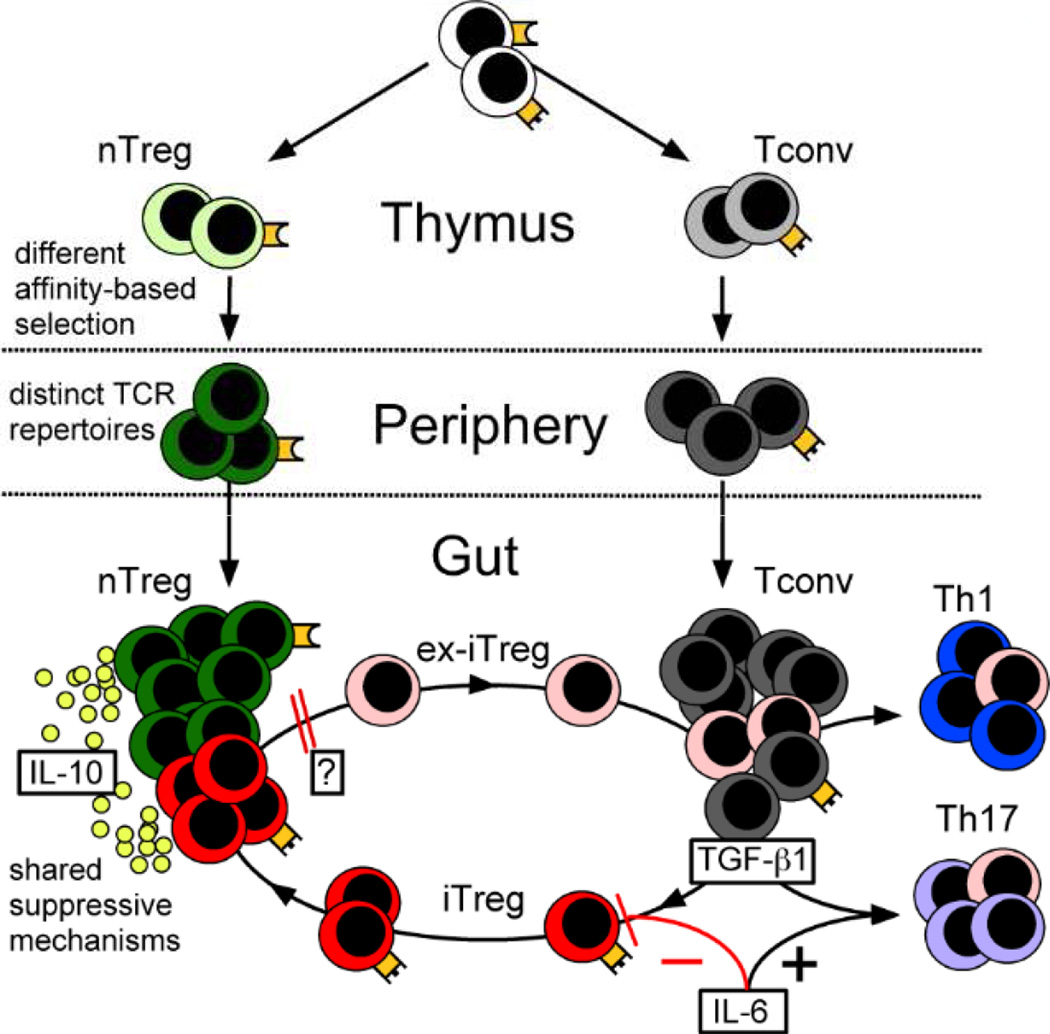
Whereas nTreg cells are defined by Foxp3 expression that originates during thymic T cell development, “adaptive” or “induced” Treg (iTreg) cells are generated during the course of an immune response from naive Tconv cells in the periphery. In healthy mice, the majority of peripheral Foxp3+ cells in the spleen and lymph nodes are thymically-established nTreg cells. In contrast, the iTreg subset of Treg cells make up a large fraction of T cells in the lamina propria (LP) and gut-associated lymphoid tissue (GALT) of the intestine.34, 35 The formation of iTreg cells involves the peripheral activation of Tconv cells in the presence of TGF-β1 (Figure 1). This conversion can also be performed in culture, resulting in Foxp3+ in vitro iTreg cells.36 These in vitro iTreg cells may serve as cell-based therapies for autoimmune diseases such as IBD since they can be derived in large numbers from the patient’s own cells, and can potentially be enriched for relevant antigen specificities. Indeed, clinical trials utilizing in vitro-derived iTreg cells as a new therapy are currently under way.37
Figure 1.
Model of Treg cell development. The nTreg (green) and Tconv (grey) cell populations develop as separate lineages in the thymus, based in part on differences in the threshold for affinity-based selection. The result is a peripheral population of nTreg and Tconv cells with distinct TCR (orange) repertoires. In the gut and at other environmental interfaces, TGF-β1 induces Tconv cells to become iTreg cells (red) or Th17 cells (purple). The peripheral Treg cell pool is therefore comprised of both iTreg and nTreg cells that share suppressive mechanisms such as IL-10 production. Increased levels of IL-6 in the local environment promote production of Th17 cells while blocking iTreg cell formation. In general, iTreg cells are unstable and may lose Foxp3 expression (ex-iTreg cells), although certain factors may increase iTreg stability. The ex-iTreg cells (pink) are available to become Th1 (blue) or Th17 cells, or to reacquire Foxp3 expression and cycle back into the iTreg pool. Importantly, nTreg and iTreg cells act synergistically and are non-redundant, a feature based largely on their different TCR repertoires.
In vivo, antigen exposure and/or lymphopenia can result in Foxp3 expression in up to 15% of antigen experienced Tconv cells.38 These in vivo iTreg cells appear to be particularly prevalent at mucosal surfaces where tolerance must be expanded to antigens within the colonizing microbiota.27, 39 In addition to the microbiota, ingested food contains a plethora of antigens. Immune responses to these dietary molecules can be actively suppressed by inducing oral tolerance. Antigen feeding experiments identified a TGF-β1-producing CD4+ T cell population in the LP and GALT coined “Th3” that was important in oral tolerance.40 These studies were performed prior to the discovery of Foxp3, and subsequent studies have shown that most “Th3” cells express Foxp3 and are de facto iTreg cells. However, a small fraction of CD4+ TGF-β1+ Foxp3− Th3 cells may yet exist.41
Similarly to in vitro iTreg, the peripheral induction of Foxp3 in vivo appears to be dependent upon TCR activation in the presence of TGF-β1.36, 42, 43 TGF-βR signaling leads to phosphorylation of SMAD molecules and the binding of SMADs to the CNS1 enhancer element in the Foxp3 locus.44 This association together with the binding of other transcription factors leads to the TGF-β1-mediated activation of Foxp3 transcription, which drives differentiation of the iTreg lineage.28, 45 The vitamin A metabolite retinoic acid (RA) further increases Foxp3 induction.46
In vivo induction of Foxp3 seems to be particularly important in the gut. In fact, the GI tract is a TGF-β1 rich environment, LP and GALT dendritic cells (DCs) produce RA, and RA drives gut tropism in Treg cells.6, 47 Additionally, APCs derived from the intestine, including CD103+ DCs and CD11bhi CD11c− macrophages preferentially promote iTreg development in comparison to spleen or peripheral lymph node derived APCs.6, 27, 39 These data strongly support a role for induction of iTreg and iTreg-mediated suppression in the gastrointestinal environment.
Distinguishing nTreg and iTreg cells
There are few mechanisms for differentiating nTreg and iTreg cells. Most molecules showing increased expression on Treg cells (including CD25, CTLA4, and GITR) appear to be targets of Foxp3 transcriptional activation, and are thus expressed similarly between iTreg and nTreg cells. Also, both nTreg and iTreg cells suppress immune responses using similar antigen-dependent and antigen-independent mechanisms.2 Other cell types can also express ostensible Treg markers, further confusing the discrimination between Treg subsets.48, 49
Helios is one molecule that has been suggested to differentiate nTreg and iTreg cells. Helios is a transcription factor that is expressed at elevated levels in nTreg but not iTreg cells.50 However, genetic deficiency of Helios does not appear to affect Treg differentiation and function.51 Subsequent studies have suggested that Helios is a marker of activation and proliferation that can be expressed on both Treg and Tconv cells.52 Furthermore, in some studies only a small fraction of in vivo-derived iTreg cells lacked Helios expression24 and both in vitro and in vivo-derived iTreg could express Helios in response to APC stimuli.53
Neuropilin-1 (Nrp-1) is another marker that may differentiate nTreg and iTreg cells. Recent studies suggest that Nrp-1 is limitied to the nTreg subset.54, 55 The iTreg cell subset expresses only low levels of Nrp-1 after in vitro conversion via TGF-β1. In vivo iTreg cells differentiated via oral or intravenous antigen delivery, exposure to a peripherally-expressed antigen, or lymphopenia-induced proliferation also lack Nrp-1 expression. Interestingly, Nrp-1 expression differences are largely maintained, even when cells are activated.54, 55 Conversely, iTreg cells differentiated under inflammatory conditions in the lung or CNS appear to gain Nrp-1 expression.54 Thus, it will be interesting to examine Nrp-1 expression in mouse models of IBD. Unfortunately, Nrp-1 does not appear to be expressed by human PBMCs, so the utility of this marker for clinical studies may be limited.56
Differences in the methylation status of the Foxp3 genetic locus are a more universally accepted difference between the nTreg and iTreg subsets. Thymus-derived nTreg cells have demethylated CpG motifs in the Treg cell-specific demethylation region (TSDR) of conserved noncoding sequence 2 (CNS2) in Foxp3, whereas the TSDR of iTreg cells is only partially demethylated.57 Heritable and stable Foxp3 expression are linked to TSDR demethylation. The factors that influence the demethylation status of the TSDR in iTreg cells are unknown and the level of demethylation varies depending on the nature of the iTreg differentiation.24, 58 Unfortunately, detection of Foxp3 TSDR methylation must be performed on the genomic DNA of large pools of cells, and thus cannot differentiate nTreg and iTreg cells on a per cell basis as with surface markers.
Since markers that differentiate nTreg and iTreg are controversial, current studies often identify all peripheral Foxp3+ T cells as “nTreg”. This practice complicates the precise determination of nTreg and iTreg cell identity and function, because the peripheral Foxp3+ T cell pool undoubtedly contains some fraction of Treg cells that induced Foxp3 after leaving the thymus. Thus, the population of cells identified as “nTreg cells” are likely contaminated with in vivo-derived iTreg cells. The fraction of iTreg in a typical peripheral Treg pool has been estimated by tracking Foxp3− Tconv cells that upregulate Foxp3 expression in vivo. In these approaches, congenically marked Foxp3− Tconv cells are transferred into lymphopenic, lymphoreplete, or Foxp3-deficient hosts. Using these transfer approaches, the overall fraction of iTreg cells in the peripheral Treg pool appears to be 4–15%, depending on the system used. As noted above, higher frequencies of iTreg cells are generally present in the mLN and GALT than the peripheral lymph nodes and spleen, supporting a role for iTreg cells at mucosal surfaces.24, 38, 59
The size of the iTreg pool was also estimated in Carma-1 deficient mice, which lack thymically-derived nTreg cells but develop iTreg cells in the periphery.34 The overall frequency of Foxp3+ cells in the periphery of Carma-1−/− is 3–4% of that in wild-type mice. Since Carma-1−/− mice only have iTreg cells yet have a higher threshold for TCR activation, 3–4% likely represents the low end of the iTreg contribution to the peripheral Treg pool, in agreement with transfer studies. Additionally, compared to the spleen, Foxp3+ cells in these mice are twice as frequent in the mLN and ten times as frequent in the LP, further supporting an increased frequency and role for iTreg development at these sites.34 Thus, when considering most studies, it appears that the typical contribution of the iTreg subset to the peripheral Treg pool ranges from 3 to 15% and is highly context-dependent. Consistently, the contribution of iTreg cells increases greatly in the intestine, favoring a significant role for iTreg cells in the control of IBD.
Due to the lack of a specific surface marker, it is currently difficult to identify human in vivo-derived iTreg cells, although it is reasonable to assume that the iTreg cell subset exists. Human T cells share a capacity for the in vitro differentiation of iTreg cells and have similarly increased numbers and frequency of Treg cells in mucosal locations. One important distiction between mice and humans is that activated non-regulatory human Tconv cells transiently express FOXP3 at low levels. Accordingly, human FOXP3+ T cell populations are subdivided as a resting/naive Treg cell subset that is CD25hiCD45RA+FOXP3hi, an effector Treg cell subset that is CD25hiCD45RA−FOXP3hi, and a non-regulatory cytokine-secreting FOXP3+ subset that is CD25hiCD45RA−FOXP3lo.60 Additional markers such as low expression of CD127, and increased expression of CD39, MHCII, GITR, and CTLA-4 further distinguish human Treg cells.61
Mechanisms of Treg suppression in IBD
Treg cells utilize numerous effector mechanisms to suppress immune responses, targeting both the innate and adaptive arms of the immune system. Interestingly, no one effector mechanism or pathway has been shown to be both necessary and sufficient for all Treg-mediated suppressive activities. There is a great deal of functional redundancy between Treg suppressive pathways, which results in compensatory mechanisms in targeted Treg cells and the need for a very strong inflammatory stimulus to differentiate phenotypes. Also, early deletion of genes involved in Treg function often leads to systemic immune issues before intestinal involvement can be detected.6 Studies of IBD are particularly sensitive to slight disparities in the colonizing microbiota between mouse colonies and differing and mouse strains, leading to inconsistent results. Despite these difficulties, several effector mechanisms for Treg-mediated suppression of IBD have been described (Table 2). In general they can be divided into two categories 1) immunoregulation through consumption or production of soluble mediators (primarily cytokines) and 2) immunoregulation via direct interaction with APCs or Teff cells.
Table 2.
Treg cell suppressive mechanisms in IBD
| Regulatory Mechanism |
Utilized by | Role in IBD | References | |||
|---|---|---|---|---|---|---|
| nTreg | iTreg | Mouse Models | Human Patients | |||
| Soluble Mediators | TGF-β1 | Yes | Yes | Yes | Yes | 42, 222 |
| IL-10 | Yes | Yes | Yes | Yes | 12, 73–75,117 | |
| IL-35 | Yes | No | Yes | Unknown | 79, 223 | |
| Pericellular adenosine (CD39/CD73) | Yes | Unknown | Yes | Unknown | 82–85 | |
| Sink for IL-2 (CD25hi) | Yes | Yes | Yes | Yes | 22, 33, 36, 83, 87 | |
| Cell-Cell Interaction | Cytotoxicity (perforin/granzymes) | Yes | Yes | Unknown | Unknown | 89, 90 |
| CTLA-4 | Yes | Yes | Yes | Yes | 95, 98, 224, 225 | |
| LAG-3 | Yes | Unknown | Yes | Unknown | 99, 226, 227 | |
| PD-1/PD-L1 | Yes | Yes | Yes | Unknown | 100 | |
| Nrp-1 | Yes | No | Unknown | Unknown | 54, 55 | |
| Galectins | Yes | Unknown | Yes | Yes | 228–230 | |
| GITR | Yes | Yes | Yes | Unknown | 231, 241 | |
| OX40 | Yes | Yes | Yes | Unknown | 232, 233 | |
Among the cytokine-based approaches, TGF-β1 and IL-10 are the best studied. TGF-β1 is a pleiotropic cytokine produced by many immune and non-immune cells. TGF-β1 controls cell proliferation and differentiation in numerous tissues throughout the body. In particular, TGF-β1 plays an important role in the extra-thymic induction of Foxp3 in iTreg cells. In addition, Treg cells themselves produce large amounts of TGF-β1. Global deficiency of TGF-β1 results in a lethal multi-organ lymphoproliferative disease similar to Foxp3 deficiency. Since TGF-β1 deficient mice die at weaning, analysis of gut-specific effects of TGF-β1 difficult.
The intestine appears to be a particularly TGF-β1 -rich environment and apoptotic cells stimulate TGF-β1 release.62 The high cell turnover and/or acute epithelial damage in the intestine may contribute to the secretion of TGF-β1 to prevent excessive immune responses in the chronically immune-stimulated intestinal environment. However, much of the TGF-β1 in the intestine is present in an inactive form that must be activated by integrins. Mice deficient in αv or β8 TGF-β1-activating integrins in APC populations develop spontaneous colitis, suggesting DCs and perhaps other APCs must activate TGF-β1 in the GALT.6, 34 This activated TGF-β1 likely has direct effects on T lymphocytes since depletion of T cells prolongs the survival of TGF-β1 deficient mice.63–65 Further, T cell-specific deletion of TGF-β-receptor II results in a similar autoimmune phenotype to global deficiency of TGF-β1.66, 67 In the T cell transfer model of colitis, a TGF-β1 blocking antibody eliminates the ability of a Treg-containing CD45RBlow pool to prevent colitis.68 In a similar transfer model, transferred T cells expressing an endogenous inhibitor of TGF-β1, Smad7, were unable to be suppressed by co-transferred Treg.42 This suggests that Treg-produced TGF-β1 has direct immunosuppressive effects on colitogenic Teff cells, and/or that TGF-β1-driven differentiation of iTreg is necessary for prevention of intestinal inflammation.
IL-10 is a potent anti-inflammatory cytokine produced by both non-immune and immune cells including Treg cells. IL-10 has particular significance in IBD because IL-10 and IL-10R2 deficient mice do not develop lethal systemic autoimmunity, as seen in TGF-β1 deficient mice, but instead develop colitis.12, 69 Established intestinal inflammation in mice can be ameliorated via treatment with recombinant IL-10 protein, IL-10 expresing transgenic T cells, or intestinal bacteria engineered to produce IL-10.70–72 Significantly, the IL10 gene is a susceptibility locus for human ulcerative colitis, and patients with mutations in the IL-10 receptor develop IBD at an earlier age and with higher penetrance than IPEX patients.73, 74 Treg cell-specific deletion of IL-10 results in a similar, though less severe intestinal inflammation than global IL-10 knockout mice.75 In the T cell transfer model of colitis, Treg-produced IL-10 is particularly necessary in the presence of triggering microorganisms such as Helicobacter hepaticus.76, 77 These results suggest that IL-10 production by both Treg and non-Treg cells is important in maintenance of tolerance in the GI tract, depending on the inflammatory context.
Tr1 cells are one other potential source for IL-10 in the intestine. Tr1 cells produce high levels of IL-10 and require IL-10 for their differentiation. Tr1 cells seem to share other suppressive pathways with Treg cells, but Tr1 cells are characterized by their lack of Foxp3 expression.6 In the colon LP, Foxp3+ Treg cells are the predominant IL-10 producing T cells. However, in the small intestine, the majority of IL-10-producing intraepithelial lymphocytes (IEL) do not express Foxp3, suggesting a potential role for Tr1 cells.78 Since Tr1 cells lack a specific marker, little is known about this population. However, the prevalence of Foxp3− IL-10-producing T cells in the intestine, and less severe disease in Treg cell-specific IL-10 knockout mice suggests that Tr1 cells may also contribute to IL-10-mediated suppression.
Another immunosuppressive cytokine secreted by Treg cells is IL-35. IL-35 is a newly discovered heterodimeric cytokine consisting of subunits IL12p35 and Ebi3. IL-35 is preferentially produced by Treg cells and suppresses T cell proliferation in vitro. In the T cell transfer model of colitis Treg cells deficient in either IL12p35 or Ebi3 showed decreased ability to ameliorate established disease.79 In addition, mice deficient for Ebi3 were more susceptible to EAE, and both Ebi3−/− and IL-12p35−/− mice showed increased airway hyperresponsiveness, suggesting Treg-produced IL-35 may play roles in numerous tissues.80, 81
Pericellular adenosine is another soluble immunoregulatory molecule utilized by Treg cells. Both mouse and human Treg cells express surface bound enzymes such as CD39 and CD73 that convert proinflammatory extracellular ATP into the immunosuppressive nucleoside adenosine. Adenosine produced by Treg cells binds adenosine receptors such as A2A on DCs and Teff cells to decrease their activation and function.82, 83 Pericellular adenosine appears to play a critical role in intestinal immune tolerance. In the T cell transfer model of colitis, wild-type Treg cells could not suppress disease induced by A2A-deficient colitogenic T cells.84 Treatment with A2A agonists also ameliorated a spontaneous mouse model of ileitis.85
In addition to producing cytokines, Treg cells can also cause immunosuppression through cytokine deprivation. Treg cells express elevated levels of numerous cytokine receptors including the high affinity IL-2 receptor CD25.8, 22, 86 This increased expression of cytokine receptors is hypothesized to allow Treg cells to outcompete Teff cells for survival signals, causing Teff cell apoptosis. In the T cell transfer model of IBD, transferred Treg cells increase apoptosis in colitogenic Teff cells. In vitro provision of exogenous IL-2 or other common gamma chain cytokines decreases Teff cell apoptosis in Treg/ Teff co-cultures.87 The Bcl-2 family member Bim signals apoptosis in response to cytokine deprivation. Teff cells deficient in Bim are resistant to Treg-driven apoptosis both in vitro and in the T cell transfer model of colitis, suggesting Teff cells are undergoing apoptosis due to an absence of cytokine-derived survival signals.87 But, a role for cytokine consumption by Treg cells in vivo is difficult to prove. Since Treg cells could utilize other mechanisms to initiate termination of the immune response, survival cytokines would be expected to subsequently decrease as a result of the decreasing immune response.6
Treg cells also directly interact with other immune cells to mediate immune suppression. The increased apoptosis of Teff cells via Treg cells may not only be due to cytokine deprivation, but may also be due to direct cytotoxic effects of Treg cells on Teff cells. The direct killing of numerous cell populations including CD4+ Teff, CD8+ CTL, and APCs by Treg cells via perforins and granzymes is well established in vitro.88–90 Treg-mediated killing of APCs is a particularly intriguing effector mechanism since this regulatory approach could yield antigen specificity through the TCR/MHCII interactions of these two cell types. The in vivo evidence for cytotoxicity of Treg cells is more limited. In a mouse model of transplantation tolerance, granzyme B expression by Treg cells and granzyme targeting in Teff cells was important for maintenance of the graft.91 In addition, Treg cells were shown to control CD8+ T cell responses to lung infection in a granzyme B-dependent manner.92 The extent to which cytotoxicity plays a role in Treg cell suppressive function in IBD is currently unknown. Still, it is interesting to note that the increased apoptosis of effector populations through both Treg -mediated cytokine deprivation and cytotoxicity could result in increased TGF-β1 production, further amplifying the regulatory response.
Treg cells also express numerous surface receptors that confer suppressive activity. Cytotoxic T lymphocyte antigen 4 (CTLA-4) is foremost among these receptors. CTLA-4 is a high-affinity inhibitory receptor related to CD28. CTLA-4 has greater affinity for its B7 ligands than CD28. This increased affinity seems to increase the duration of time Treg cells engage with APCs, and modulate the APC function and maturation. In fact, both in vitro and intravital imaging suggests that these stable Treg-APC contacts limit access of Tconv cells to APCs, thus limiting Teff cell activation.93, 94 In the T cell transfer model of colitis, CTLA-4 blockade limits protection by Treg cells. Also, CTLA-4 deficient nTreg cells can prevent colitis induced by wild-type naïve T cells, but not disease induced by CTLA-4-deficient T cells.95 This suggests a role for CTLA-4 on nTreg and also Tconv and/or in vivo iTreg cells. Treg cell-specific CTLA-4 deficiency leads to a lethal autoimmune disorder. However, this lethality occurs much later than in Foxp3 deficiency, suggesting other Treg cell effector mechanisms can temporarily compensate for the absence of CTLA-4.96
CTLA-4 also appears to play a particularly important immunoregulatory role in the human intestine. Anti-CTLA-4 (ipilimumab or tremelimumab) treatment increases the immune response to some cancers by presumably decreasing Treg cell function. However, data from over 4000 patients shows that these treatments can result in potentially lethal colitis.34, 97 In addition, treatment with CTLA4-Ig (abatacept) has shown negative effects including new-onset UC in arthritis patients, and exacerbation of symptoms in CD. These results suggest that CTLA-4 may not only block B7 molecules to promote regulation, but that CTLA-4 signaling may be required for Treg cell function in the gut.98
Treg cells also express several other receptors that may play a role in immunoregulation. Lymphocyte Activation Gene-3 (LAG-3) is a homologue of CD4 that binds with high affinity to MHCII on the surface of APCs. This binding inhibits DC maturation and leads to more tolerogenic DCs.99 Treg cells are one of few cell types that express both programmed death (PD) receptors and ligands. Expression of PD-L1 and PD-1 may promote the differentiation of iTreg cells. PD-L1 and PD-1 expressed by Treg cells may also promote stable contacts between Treg cells and APCs, and modulate APC function, similar to CTLA-4 and LAG-3.100 These Treg-APC contacts may be maintained and enhanced via Treg cell expression of Nrp-1. Treg cells preferentially expresses additional receptors such as galectins, GITR, and OX40, but their contribution to Treg effector function is relatively unknown.101
Interestingly, many of these receptors have the potential to interact with both APC and Tconv cells to regulate immune responses. Several studies suggest Treg cell suppression may be more important in the lymph nodes than the site of inflammation. In the T cell transfer model of colitis, deletions of gut-homing integrins such as CD103 (αE) shows that migration to the intestine is necessary only in the APC population for Treg to mediate suppression. In addition, Treg cells must express lymph node homing receptors to suppress colitis. This data argues that in the T cell transfer model of colitis Treg cells mediate most of their suppressor function through interactions with gut-homing DCs in the lymph node.102–105
Independent and synergistic roles of nTreg and iTreg cells in mucosal tolerance
It is becoming clear in mouse models of IBD that both nTreg and iTreg cells are necessary for protection and perhaps also treatment of disease.38 However, how the nTreg vs iTreg cell responses are calibrated is currently unknown. TCR repertoire and Treg differentiation studies suggest that the dual requirement for nTreg and iTreg cells may be due to differing TCR specificities between the two subsets of Treg cells. In particular, it seems that nTreg cells are necessary to confer tolerance to self antigens, whereas iTreg cells are induced at mucosal surfaces to confer tolerance to antigens in food and to the microbiota that makeup the “extended self”. Interestingly, IBD patients have increased immunoreactivity to both self and microbial antigens suggesting important roles for both nTreg and iTreg subsets and their corresponding specificities in human patients as well.106, 107
The iTreg subset develops after peripheral activation of Tconv cells in the presence of TGF-β1. However, when levels of the pro-inflammatory cytokine IL-6 are also high, TCR and TGF-β1 signaling results in upregulation of RORγt and development of pro-inflammatory Th17 cells that express the cytokine IL-17 (Figure 1). Thus, iTreg and Th17 cells both arise from Tconv cells in the presence of TGF-β1. In fact, iTreg and Th17 cells may share a common Foxp3+/RORγt+ precursor.108 Under normal conditions, Th17 cells recruit neutrophils to combat infection by fungi and extracellular bacteria. However, these cells are also associated with numerous autoimmune diseases, including IBD. In fact, increased levels of characteristic Th17 cytokines are found in biopsies from IBD patients.48 The role of Th17 cells in IBD is unclear though, as several studies have shown them to be both pathogenic and protective.39 Mouse studies have disagreed, in particular, on the role of IL-17. Disease can be exacerbated, ameliorated, or unaffected by the absence of IL-17 depending on the method of depletion and whether the IL-17A or IL-17F isoform was targeted.48
In addition to IL-17, Th17 cells produce IL-21 and IL-22. IL-21 appears to be largely pro-inflammatory, and promotes further amplification of additional Th17 cells. IL-21 expression is upregulated in lesions of IBD patients.109 In contrast to the other Th17 cytokines, IL-22 appears to be protective in IBD. IL-22 promotes intestinal barrier integrity and prevent microbes from breeching the epithelium. IL-22 increases cell proliferation and survival of the epithelium, upregulates production of antimicrobial peptides (AMP) such as defensins, and stimulates mucus production.48
The Th17 cell phenotype is maintained through binding of IL-23 to Th17-expressed IL-23R. IL-23 appears to be necessary for the development of IBD in several mouse models, suggesting a pathogenic role for Th17 cells.77, 110–112 In contrast, one study suggests that IL-23 deficient mice have increased susceptibility to colitis induced by TNBS barrier disruption, which agrees with the role of Th17-produced IL-22 in barrier maintenence.113 Genome-wide association studies identified IL-23R as one of the genes most strongly associated with both UC and CD in human patients.5 One allele of IL-23R (R381Q) is protective for IBD. This mutation results in decreased IL-23R signaling and thus decreased numbers of Th17 cells.114 Since this allele confers about three-fold protection from CD, it appears that Th17 cells may play largely pathogenic roles in human IBD.5
Therefore, although iTreg and Th17 cells may share receptor specificities, developmental precursors, and differentiation signals, they have differing impacts on immune responses. In fact, the increased methylation of the Foxp3 locus of iTreg cells versus that of nTreg cells suggests that the iTreg subset may serve as a more flexible lineage, able to adapt to the antigenic or inflammatory environment. Determining whether iTreg cells can downregulate Foxp3 expression, becoming so-called “ex-iTreg”, and revert to alternate fates such as Th17 is an active area of research. It is likely that control of the iTreg/Th17 balance and the stability of iTreg are key immune regulatory checkpoints.115, 116 Thus, iTreg/Th17 conversion may control regulatory versus inflammatory responses to extended self and foreign antigens, while nTreg cells control responses to self-antigens in the intestine (Figure 1).
A dual requirement for both nTreg and iTreg cells in mucosal tolerance has been supported through several studies. In the T cell transfer model of colitis, disease is much more severe when induced with Tconv cells that have an inability to become in vivo-derived iTreg cells.38 Mice with established colitis can be successfully treated with the transfer of nTreg cells, but only if the colitogenic Tconv population can induce Foxp3. In mice lacking in vivo iTreg cells, co-transfer of nTreg cells and in vitro iTreg can cure disease, suggesting a requisite role for these cells in reversing IBD.38 Further studies demonstrated differences in TCR repertoire between the subsets and suggested the primary role for iTreg cells is to expand the antigen diversity of immunoregulatory responses.24 Though nTreg and iTreg cells appear to utilize similar effector mechanisms, iTreg cells may serve as a flexible lineage able to expand immune regulation to antigens not expressed during thymic nTreg development.117 The ability of iTreg cells to aid in peripheral tolerance appears to be particularly important at mucosal interfaces, where the majority of iTreg cell specificities are to antigens derived from the microbiota.118
Effects of the microbiota on Treg cells
The microbiota have co-evoved with the host to aid in host metabolism, and also play significant roles in host protection and immunity. The microbiota provide signals leading to the maintenance of the epithelium, and stimulate production of antimicrobial peptides and lectins, thus preventing breech of the initial immune barrier.27, 119 The microbiota also directly interact with the innate immune system and aid in adaptive immunity by promoting the development of Peyer’s patches and isolated lymphoid follicles in the gut, as evidenced by GF mice lacking these structures.120 In turn, the host immune system limits microbial access to the epithelium via the production of antimicrobial products and the secretion of mucus and IgA.121
IBD patients exhibit abnormal shifts in the taxa of their microbiota. It is not known whether this abnormal bacterial growth is a direct cause of IBD, or a side effect of increased inflammation in the intestine. Nonetheless, probiotics, antibiotics, and fecal diversion have all shown some beneficial effects in IBD.39, 122, 123 Thus, it seems increasingly likely that in IBD there are abnormalities in both the microbiota and the immune response, and that these dual abnormalities may be related.
The Treg cell compartment seems to be particularly sensitive to changes in the microbiota. TGF-β1 and IL-10 expression by Treg cells is necessary for their suppressive capabilities, but only in the presence of an activating microbiota.34 Treg cells from GF mice are generally less suppressive, and express less Foxp3 than Treg cells from mice under normal conditions.124–126 Though, one study has demonstrated significant IBD suppression mediated by a Treg-containing population from GF mice.127 The presence of colonizing microbes may also affect the prevalence of Treg cells in the GALT. Treg cells numbers are decreased in the colons of GF mice, but are increased in the small intestine.27 Further, Treg cell numbers in the small intestine LP increase in response to decreasing bacterial load via vancomycin treatment.128 Consistent with these findings, TLR9-deficient mice have increased numbers of Treg cells in the small intestine, suggesting that inflammatory signals from bacterial DNA plays a role in inhibiting iTreg cell differentiation or Treg cell proliferation.129
Mouse studies of IBD have identified Helicobacter hepaticus and segmented filamentous bacteria (SFB) as two particularly pro-inflammatory members of the intestinal microbiota. Helicobacter hepaticus is a normal member of the microbiota in many mouse facilities. Helicobacter hepaticus and a related species Helicobacter bilus normally do not cause pathology, but can trigger inflammation in susceptible hosts.6, 130 In the T cell transfer model of colitis, disease severity was greatly increased in the presence of Helicobacter hepaticus.131 Further, mice with a Treg-specific deletion of IL-10 do not normally present any spontaneous phenotype. When infected with Helicobacter, these mice develop spontaneous colitis.75 Though Helicobacter hepaticus appears to drive inflammation, Treg cells from animals infected with Helicobacter hepaticus appear to be more suppressive than Treg cells from uninfected animals.132 Given the numerous effects of Helicobacter spp. on Treg cells and IBD, it seems likely that the variable presence of Helicobacter in mouse colonies may be one potential explanation for inconsistencies in IBD studies between institutions.
In mice, segmented filamentous bacteria (Candidatus arthromitus) also play an important role in intestinal immune responses. SFB are able to penetrate the mucus layer and directly contact the epithelium, making them particularly equipped to impact host immune responses.121 SFB are approximately 25 times more prevalent in C57BL/6 mice from the vendor Taconic Farms than in C57BL/6 mice from Jackson Labs, providing sources for relatively SFB sufficient and deficient mice.133 In the T cell transfer model of colitis, the addition of SFB to a specific pathogen free bacterial cocktail led to severe colitis.134 Further studies determined that SFB likely promote inflammation by driving Th17 cell differentiation in the small intestine LP.133, 135, 136 Monocolonization of GF animals with SFB also leads to IgA production and increased numbers of IEL in the gut.135, 137 SFB induction of Th17 cells appears to be dependent on SFB production of serum amyloid A and ATP, which act on LP DCs, that in turn drive Th17 differentiation.128, 138
The impact of SFB on human IBD is unknown however, since this bacterium is rarely found in the human microbiota. This may be due to prevention of SFB colonization by specific human defensins. Indeed, transgenic expression of human defensin 5 in mice leads to a lack of SFB colonization and a subsequent elimination of Th17 cells in the intestinal LP.136 One report suggests that in addition to the induction of Th17 cells, SFB may also play a role in differentiation of iTreg cells.135 However, others found SFB did not increase Treg cells, suggesting the source of SFB may play a role.133
Though Treg cell numbers generally increase when overall bacterial load is decreased, several studies demonstrated that specific bacterial taxa may drive peripheral Foxp3 induction and/or Treg cell proliferation. In fact, the colon Treg TCR repertoire was shown to differ from the Treg repertoire at other peripheral sites, and these TCR sequences were specific for members of the microbiota. Further, these TCRs were colitogenic when expressed in the absence of Foxp3. These results demonstrate a role for peripheral differentiation of iTreg cells specific for the bacterial microbiota and provide exciting support for the hypothesized role of iTreg cells in broadening tolerance to the “extended self” of commensal microorganisms.118
Specific bacterial species may play roles in iTreg differentiation. Though GF mice lack Treg cells in the colonic LP, the colonization of GF mice with a distinct combination of 46 strains of Clostridium spp. led to normal development of Treg cells in the gut 139. These Clostridium-colonized mice had greater levels of intestinal TGF-β1 and increased levels of CTLA4 and IL-10 expression in the intestinal Treg pool. Faecalibacterium prausnitzii is one species included in the 46 Clostridium spp.139 F. prausnitzii has been shown to induce IL-10 production and reduce severity of colitis in mouse models. Interestingly, this bacterium is decreased in the intestines of IBD patients and is associated with greater risk of disease recurrence in post-surgical Crohn’s disease patients.140
Numerous lactic acid bacteria and Bifidobacteria are used as probiotics, and several strains have shown effects on Treg cells. Lactobacillus acidophilus, Lactobacillus rhamnosus, and Lactobacillus reuteri have all been shown to modulate inflammation in mouse models of disease.39 Lactobacillus reuteri were specifically shown to increase the frequency of Treg cells in the mLN and spleen of host animals.141, 142 Bifidobacterium infantis increase the frequency and number of Treg cells in the spleen and PP and decrease intestinal inflammation induced by Salmonella typhimurium infection.143 Consuming B. infantis also increased the percent of Foxp3+ Treg cells in the peripheral blood of human subjects.144 VSL#3 is a probiotic mixture of numerous Lactobacillus and Bifidobacteria designed for the management of IBD. Treatment of mice with VSL#3 increases the production of IL-10 and number of TGF-β1-producing T cells, though it is unknown whether these cells are Foxp3+. VSL#3 treatment also decreases the severity of a chemically-induced model of colitis.145
Generally, the presence or absence of bacterial taxa only correlates with effects on the Treg cell pool. Administered microbes may exert their effects by modifying other members of the microbiota, and not through direct interaction with the immune system. However, Bacteroides fragilis exhibits a distinct mechanism for direct modification of host immune development. Monocolonization of GF mice with B. fragilis leads to increased differentiation of IL-10-producing iTreg cells.146 This induction is completely dependent upon expression of a single B. fragilis molecule, polysaccharide A (PSA). Further, this PSA-dependent iTreg cell development requires TLR2 signaling, suggesting B. fragilis modifies the host APC to drive iTreg cell differentiation. B. fragilis-produced PSA inhibits Th17 induction and promotes iTreg development, thereby protecting the host from both chemically-induced and H. hepaticus-driven colitis.146, 147 Conversely, enterotoxigenic strains of B. fragilis, which naturally lack PSA expression, play a role in inducing Th17 cells and promote intestinal inflammation.148
Numerous bacterial strains can affect the host Treg cell compartment. Many of these bacteria are promising candidates for probiotic or antibiotic treatments for IBD. However, most of these taxa, including Lactobacillus, Bifidobacterium, Helicobacter, F. prausnitzii, and B. fragilis are of low or variable abundance in the human microbiota, and SFB are largely absent in humans.39, 121 Further, probiotic and antibiotic-driven changes in the microbiota often do not endure once treatment is discontinued. Therefore, additional studies will be necessary to identify the role indigenous bacteria play on the Treg cell lineage, and how these interactions may promote or prevent IBD.
Failure of regulatory mechanisms in IBD
Since IBD involces a failure in immune regulation, it seems likley that IBD is caused in part by defects in Treg cells. These defects could be Treg cell-intrinsic, resulting from inherently defective Treg cells in the patient. Alternatively, the causes of defective Treg cells could be Treg cell-extrinsic, and a function of the abnormal intestinal environment and cytokine milieu in IBD patients. These Treg-intrinsic or extrinsic defects can lead to insufficient Treg cell suppression of IBD in at least 4 ways: 1) deficient Treg cell numbers, 2) defective Treg cell function, 3) unstable Treg cell phenotype, and 4) pathogenic T cell resistance.149
The majority of what we know about the mechanisms of Treg involvement in IBD comes from mouse models. Mouse models allow better identification and tracking of bona fide Treg cells and easier analysis of the intestinal milieu and sites of inflammation. Complete Treg cell deficiency, such as that in scurfy or other Foxp3 mutant mice, leads to fatal autoimmunity that includes intestinal inflammation, underscoring deficient Treg number as a potential mechanism for IBD.8, 150 In T cell transfer models of colitis the colitogenic T cell population must be depleted of Treg cells in order to cause disease.19 Further, injection of both Treg cell subsets into these mice can prevent or cure IBD in a dose-dependent fashion. 38 Mutations in other potentially Treg cell-intrinsic genes such as CD25, IL-2, CD28, and TGF-β1 also lead to decreased numbers of Treg cells, though IL-2 and TGF-β1 expression by other cell types likely contributes.2, 149 Other Treg cell-extrinsic factors also play a role in mouse models. Cytokines such as IL-6, IL-21, IL-27, and IL-23 may suppress differentiation of iTreg, suggesting a role for the intestinal milieu on Treg cell number.6
Treg cell function may also be affected by the same cytokines that lead to changes in Treg cell number. In addition, other Treg cell-extrinisic cytokines such as TNF, IL-4, IL-12, IL-7, and IL-15 may limit Treg function.149 Several molecules have been shown to play Treg cell-intrinsic roles in Treg suppressive function. Mutations in Treg surface receptors CTLA-4, CD39, LAG-3, and Fas have been shown to limit Treg cell suppression.149 In addition, Treg cells deficient in the soluble factors TGF-β1, IL-10, and IL-35 have decreased function (Table 2).149
Mouse Treg cells have also shown variable stability. IL-6 exposure drives a fraction of Treg cells into the Th17 lineage in vitro.151 In the T cell transfer model of colitis, transfer of Treg cells into an IL-10 deficient host led to a loss of Foxp3 expression and conversion of Treg cells into Th1 pathogenic effector cells.152 Adoptive transfer of Treg cells into T cell deficient hosts led to the loss of Foxp3 expression and conversion of Treg cells into T follicular helper cells in the intestinal PP.153 Other lineage-tracing studies showed that Treg cells that lost Foxp3 expression converted to Th17 cells in the PP, but became more Th1-like in the spleen and mLN. Further, these studies showed that in a mouse model of diabetes Treg cells that lost Foxp3 expression could be pathogenic upon re-transfer.154 In the transfer model of colitis, the transfer of in vitro-derived iTreg cells as treatment resulted in 85% of transferred cells losing Foxp3 expression, becoming ex-iTreg cells with pathogenic potential.117 Conversely, another study utilizing an inducible cell-tracking system found Treg to be rather stable.155 The differences in these studies may reflect the differences in iTreg cell vs nTreg cell involvement in each system. In general, these studies have shown the iTreg cell lineage to be more plastic and able to become pathogenic ex-iTreg, whereas the nTreg cell lineage demonstrates greater stability (Figure 1).
Mouse models have shown that even in the presence of functional Treg cells, pathogenic effector cells can be resistant to Treg-mediated suppression. In addition to affecting Treg cell function, the cytokines IL-2, IL-4, IL-7, and IL-15 can drive proliferation of effector T cells in the presence of Treg cells in vitro.156 Also, agonistic stimulation of TNF receptor family members OX40 or 4-1BB on conventional T cells leads to effector T cells that are resistant to Treg suppression in vitro.157, 158 For OX40, this action has been demonstrated in vivo in the T cell transfer model of colitis.158
Human studies of IBD have a distinct advantage over other autoimmune diseases such as multiple sclerosis and lupus because tissue samples from the site of inflammation can be removed and studied. However, since human activated effector T cells also express Foxp3, histological analysis of Treg cells in IBD patients can be difficult. Nonetheless, studies with human patients have been able to support regulatory mechanisms first discovered in mouse models. Research in human patients has similarly investigated Treg cell number, Treg cell function, Treg cell stability, and effector cell resistance to Treg as potential causes of IBD.
Numerous studies have investigated Treg cell number in human IBD patients. In general, these reports observe greater numbers of Foxp3+ cells in the intestine of patients, particularly in active inflammatory lesions.77, 159–164 Numbers of Treg cells in the peripheral blood are less consistent than the intestine. However, most studies identify a decreased frequency of Treg cells in the peripheral blood of IBD patients. This decrease is intensified as Treg cell numbers increase in the intestine during active disease, suggesting a potential redistribution of these cells in response to active inflammation.77, 162–165 Interestingly, the increase in Treg cells in the intestine does not appear to be specific to autoimmune disease, as similar or greater numbers of Treg cells are seen in other inflammatory conditions such as diverticulitis and enteritis. 77, 162
Treg cells at the site of inflammation fail to protect against IBD despite being present in increased numbers. This suggests that Treg function may be compromised in these patients. To investigate this point, several groups removed Treg cells from IBD patients and tested their suppressive function in vitro. These studies showed uniformly that Treg cells from the peripheral blood, mLN, and LP of IBD patients were all equally suppressive to Treg cells from normal controls.159, 161–163, 166–168 Thus, Treg cells from IBD patients appear to be present in sufficient numbers and function normally ex vivo. These results suggest that there may not be an intrinsic defect of Treg cells in IBD, but an extrinsic effect of the intestinal milieu on Treg cells. In fact, numerous IBD susceptibility genes such as autophagy genes, NOD2, and IL23R, could play a role in creating an abnormal intestinal microenvironment that limits Treg cell function in vivo. Previous studies in mouse models suggest that cytokines within an inflammatory microenvironment may also promote Treg cell instability.169 Similar results have been seen for human Treg cells. Human Treg cells from peripheral blood can convert to Th17 cells when stimulated in vitro in the presence of IL-2. This conversion is increased by inflammatory cytokines IL-1β IL-23, and IL-21 or APCs activated by microbial stimuli.170
Effector T cell hyporesponsiveness to suppression is another Treg cell-extrinsic factor that may play a role in human IBD. Two studies investigated Teff responsiveness and found no difference between IBD patients and controls.159, 167 Other groups found Teff cells from patients to be resistant to Treg suppression. These studies suggested that Teff cells from IBD patients overexpress SMAD7, thus making them resistant to TGF-β1 secreted by Treg cells.42, 171 Further, Treg cells may suppress disease by driving Teff apoptosis, and Teff cells in IBD patients are generally resistant to apoptosis.172, 173 Given the data suggesting a potential role for the intestinal milieu, it is important to note that these studies of Teff responsiveness are performed on T cells outside of the inflammatory intestinal niche.
The potential for Treg cells in IBD therapies
Since Treg cells play a requisite role in preventing autoimmunity, these cells may be ideal targets for developing new treatments for IBD. There are currently over 50 products being tested for use in the treatment of IBD. These include small molecules, siRNA, peptides, vaccines, and numerous monoclonal antibodies and cell-based treatments.98 Several of these treatments center on Treg cells. However, additional research has suggested several other potential mechanisms for Treg-based therapies. In order to find new therapies that rely on Treg cells researchers must characterize Treg cells during the disease process, and determine the mechanisms of impaired Treg cell suppression in IBD. This knowledge will aid in developing treatments that will increase Treg cell number, function, and stability.149 Developing treatments involving Treg cells is particularly challenging for IBD since the precise role for Treg cells in IBD patients has not been fully elucidated.
Traditional treatments for IBD appear to have effects on the Treg cell compartment. Standard treatment of UC patients with aminosalicylates or glucocorticoids led to increased peripheral blood frequencies of CD4+CD45RO+CD25+ T cells, a population enriched in Treg cells.164 In CD however, treatment with azathioprine or mercaptopurine led to decreased frequencies of Treg cells in the peripheral blood.163 Treatment with monoclonal antibodies to TNF-α (infliximab) was shown to increase Treg cells in the peripheral blood and intestinal LP by two separate groups.174, 175 However, another group did not see any difference in Treg cell frequency upon infliximab treatment.176 These classically used IBD treatments have broad effects, and a disadvantage of these approaches is down-modulation of both innate and adaptive immunity and increased risk for opportunistic infection.48 Therefore, there is a need to increase the specificity of IBD treatments, with Treg cells as one particular area of interest.
IBD treatments focused on Treg cells fall into two categories: cell-based treatments that involve the transfer of in vitro expanded or stimulated Treg cells into patients, or pharmacological approaches that attempt to influence Treg cells in vivo. The small number of Treg cells that can be obtained from a patient’s blood limits cell-based therapies. Thus numerous approaches are being investigated to expand and activate a patient’s own peripheral blood Treg cells for re-transfer. Such approaches have been shown to increase the number of Treg cells up to 13000 fold in 3–4 weeks. Yet, a fraction of these cells lose Foxp3 expression, indicating instability of the Treg cells or outgrowth of Tconv cells in this population.61 Another potential source for Treg cell-based treatments is the in vitro differentiation of iTreg cells from Tconv precursors via TCR stimulation and TGF-β1. This approach can yields an average of 240 × 109 iTreg cells in approximately 2 weeks in culture.177
Both in vitro expansion of nTreg and differentiation of iTreg are limited by several factors. These Treg cells may still have relatively few antigen specificities or the incorrect antigen specificities for proper suppression. Tetramer-based approaches could lead to expansion of relevant Treg cell specificites, but rely on knowledge of the target antigens for disease.178 Further, transferred Treg cells may harbor potentially harmful specificities if they do not remain stably suppressive.117, 154 Addition of rapamycin to cultures leads to increased numbers of Treg cells for both in vitro nTreg expansion and iTreg differentiation.177, 179 Further, rapamycin reduces the outgrowth of contaminating effector T cells, and increases Treg stability.180 Another limitation of cell-based approaches is that transferred Treg cells may not localize to the inflamed target tissue. Treg cells could be engineered with receptors such as tissue-specific integrins, driving a Treg cell to home the intestine. However, for these approaches to work in human patients gene therapy would have to be utilized, which raises safety concerns.
Mouse studies have utilized Treg cells that become activated by a bystander Ag in the target tissue to increase Treg localization.181
One recent phase 1/2a clinical study demonstrated both safety and efficacy of ovalbumin-specific Treg cells in a small CD patient population.182 Autologous Treg cells were expanded in vitro, cloned by limiting dilution, and selected for IL-10 production in response to ovalbumin. Since ovalbumin is a common food antigen, ovalbumin specificity was used as a bystander antigen in an attempt to drive gut homing and local immune regulation in the intestine. Five weeks post-treatment 8/20 patients showed significant clinical improvement, with 6/8 showing improvement at one dose. However, the Treg subset being used in these studies is indeterminate since they express numerous molecules associated with Tr1 cells, yet are activated in vitro and can also express Foxp3. 182
Cell-based treatments may also utilize populations other than Treg cells. Transfer of tolerogenic DCs could drive iTreg differentiation in vivo.183 Mesenchymal stem cells can produce IL-10 and TGF-β1, which could lead to increased iTreg differentiation or overall Treg function.98, 184 Transfer of mesenchymal stem cells ameliorated a mouse model of chemically induced colitis and more than doubled the frequency of Treg cells in the mLN of treated mice.185
Recent studies underscore the need for both nTreg and iTreg cells for complete tolerance.24 Thus, clinical studies may need to utilize transfer of both nTreg and iTreg cells. Alternatively, nTreg cells could be transferred in addition to Tconv cells that cannot become pathogenic, or nTreg cells in combination with another cell population to drive in vivo iTreg differentiation. One can also imagine utilizing either nTreg or iTreg cells for different clinical outcomes.35 For example, an inflammatory response against the extended self may benefit more from iTreg cell specificities, whereas an autoimmune response may be better treated via nTreg cells. However, as noted Treg cell numbers are not generally decreased in human patients, and Treg cells from these patients are generally suppressive ex vivo. Thus, Treg cells in IBD patients may not be intrinsically flawed, but may fail to suppress disease due to local factors affecting Treg cell function. Thus, other therapeutic approaches may be necessary to increase Treg cell number or function in situ.
Probiotics are one approach to increase Treg cell responses specifically in the intestine. Since IBD patients often display an abnormal microbiome, reshaping of the intestinal microbiota has been suggested as a potential treatment for disease. In fact, complete microbiota transplant, also known as fecal bacteriotherapy, has been used to treat Clostridium difficile-associated colitis, and has shown promise in some cases of IBD.123, 186, 187 More commonly, specific species or of bacteria are utilized as probiotics. Many of these treatments were found to increase Treg cell numbers and function in both humans and mice, likely through local effects on gut-associated DCs.144, 188–190 Probiotic strains can also be genetically engineered to better modify the inflammatory intestinal milieu.191 Lactococcus lactis engineered to secrete IL-10 transiently colonized mice and decreased two models of colitis.72 A phase I clinical trial demonstrated safety of a similar approach in human patients, but efficacy has not been determined.192
Vitamins A and D may also affect Treg cells and serve as potential therapeutic agents for IBD. The vitamin A metabolite RA appears to increase Treg cell induction and homing in the intestine, and RA treatment ameliorated mouse models of IBD.193 Further, RA treatment increases Foxp3 expression in human intestinal biopsies, suggesting potential therapeutic applications for vitamin A metabolites in IBD.193 Vitamin D is a sunlight-dependent precursor to the hormone calcitriol. Vitamin D metabolites and analogues have been variably shown to stimulate mouse and human Treg cell and Teff differentiation and function.194–196 Interestingly, IBD risk increases significantly with increasing latitude, suggesting a role for vitamin D as an environmental risk factor in IBD.197 Also, higher predicted serum levels of vitamin D reduce the risk for IBD, and the vitamin D receptor is a potential susceptibility allele for UC and CD.198, 199 Numerous mouse studies also support a protective role for vitamin D in IBD.200 However, few intervention studies treating IBD patients with vitamin D have been performed. A recent meta-analysis suggested a potential benefit to vitamin D supplementation of IBD patients, but underscored the need for randomized placebo-control studies.201
In atopic disease allergen-specific hyposensitization is a common approach for inducing tolerance to an antigen. This involves repeated parenteral or mucosal vaccination with the antigen at increasing doses, and has been shown to be safe and decrease hypersensitivity. Significantly, this approach seems to rely on the differentiation or activation of antigen-specific Treg cells.7 These approaches have been used for food allergy, suggesting this approach is effective for intestinal antigens.202 Hyposensitization may be difficult to apply to IBD since the specific antigens for this hypersensitivity are largely unknown. Nevertheless, a recent study in a mouse model showed promising results. Oral administration of components of one specific species of the microbiota led to increased Treg cell number and frequency in the mLN. Further, this treatment prevented a chemically induced model of colitis, and attenuated established disease.203
As mentioned, the classical treatments of IBD may obtain some of their beneficial results via effects on Treg cells. However numerous other pharmacologic agents are also used to treat IBD, and often have notable effects on Treg cells. In mouse models, histone deacetlylase inhibitors increased Treg cell number and function, and decreased chemically induced colitis.204, 205 Other small molecules such as siRNA and nanoparticles are currently being used to target intestinal tissue, and may also have future applications to increase Treg cell number and/or function in the gut.98
Monoclonal antibodies are the largest class of pharmaceuticals in development for treatment of IBD. Anti-p40 (briakinumab, ustekinumab) limits Th17 and Th1 responses in IBD patients by blocking the cytokines IL-12 and IL-23.206–208 Anti-p40 may also have beneficial effects on Treg cells since IL23Rko mice have increased Treg cell numbers, particularly in the GI tract.209, 210 Anti-CD3 has shown beneficial effects in mouse models of autoimmune disease, leading to deletion and anergy of pathogenic T cells, while promoting Treg cell responses.211 However, one anti-CD3 monoclonal antibody (visilizumab) recently failed to show efficacy after parenteral injection, despite promising pilot studies.212 Clinical studies treating IBD patients with an oral anti-CD3 antibody (muromonab) are currently underway.213
Other integrin-specific monoclonal antibodies are being used to limit pathogenic T cell access to intestine. Natalizumab, anti-α4 integrin (CD49d) has been approved for use in IBD in the U.S. However, unexpected complications arose with this drug due to α4 integrin’s role in lymphocyte homing to the CNS.214 Thus, newer studies are utilizing vedolizumab to target the α4β7 integrin dimer, which appears to control homing only to the intestine.215, 216 One concern with monoclonal antibodies targeting integrins it that they may also limit Treg cell access to the site of inflammation. However, any constrains on Treg access to the intestine maybe counteracted by the blockade of pathogenic T cells and the ability of Treg cells to suppress autoimmune responses in the mLN and other peripheral lymphoid tissues.
In general, the development of potential treatments for IBD is outpacing the ability to test these approaches in human patients. Clinical studies are often statistically limited by the ability of a small number of willing patients to differentiate relatively small effects on disease progression.98 Future studies may benefit greatly from personalized medicine approaches to stratify treatment groups in a clinical study. Genetic screens and serum biomarker analysis may identify the patients most likely to respond to a Treg-based therapy. Examination of a patient’s microbiota, and other non-genetic factors such as nutritional and vitamin status may also increase the success of treatments. Exciting new treatments from IBD will likely result from combining the increased ability to develop novel therapies with new technologies to identify the patients most likely to respond. A combination of genetics, microbiota, and other non-genetic/environmental influences are thought to contribute to IBD risk. Importantly, Treg cells interact with, and can be affected by all three of these contributing factors. It therefore seems highly likely that future treatments for IBD will include therapies targeting and capitalizing on the potent immunomodulatory effects of Treg cells.
Acknowledgments
We thank Dominic Co, Bhaskar Gurram, and Nita Salzman for critical reading of the manuscript. We apologize to the many researchers whose work we could not highlight due to space constraints.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Supported by NIH F32 AI092977 (CGM), a Senior Research Award #2858 from the Crohn’s and Colitis Foundation of America (CBW), NIH R01 AI073731 and R01 AI085090 (CBW), and the D.B. and Marjorie Reinhart Family Foundation (CBW).
References
- 1.Salzman NH. Paneth cell defensins and the regulation of the microbiome: détente at mucosal surfaces. Gut Microbes. 2010;1:401–406. doi: 10.4161/gmic.1.6.14076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.MacDonald TT, Monteleone I, Fantini MC, et al. Regulation of homeostasis and inflammation in the intestine. Gastroenterology. 2011;140:1768–1775. doi: 10.1053/j.gastro.2011.02.047. [DOI] [PubMed] [Google Scholar]
- 3.McDole JR, Wheeler LW, McDonald KG, et al. Goblet cells deliver luminal antigen to CD103+ dendritic cells in the small intestine. Nature. 2012;483:345–349. doi: 10.1038/nature10863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brand S. Crohn's disease: Th1, Th17 or both? The change of a paradigm: new immunological and genetic insights implicate Th17 cells in the pathogenesis of Crohn's disease. Gut. 2009;58:1152–1167. doi: 10.1136/gut.2008.163667. [DOI] [PubMed] [Google Scholar]
- 5.Cho JH. The genetics and immunopathogenesis of inflammatory bowel disease. Nat Rev Immunol. 2008;8:458–466. doi: 10.1038/nri2340. [DOI] [PubMed] [Google Scholar]
- 6.Izcue A, Coombes JL, Powrie F. Regulatory lymphocytes and intestinal inflammation. Annu Rev Immunol. 2009;27:313–338. doi: 10.1146/annurev.immunol.021908.132657. [DOI] [PubMed] [Google Scholar]
- 7.Saurer L, Mueller C. T cell-mediated immunoregulation in the gastrointestinal tract. Allergy. 2009;64:505–519. doi: 10.1111/j.1398-9995.2009.01965.x. [DOI] [PubMed] [Google Scholar]
- 8.Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nature Immunology. 2003;4:330–336. doi: 10.1038/ni904. [DOI] [PubMed] [Google Scholar]
- 9.Boden EK, Snapper SB. Regulatory T cells in inflammatory bowel disease. Curr Opin Gastroenterol. 2008;24:733–741. doi: 10.1097/mog.0b013e328311f26e. [DOI] [PubMed] [Google Scholar]
- 10.Maxwell JR, Brown WA, Smith CL, et al. Methods of inducing inflammatory bowel disease in mice. Curr Protoc Pharmacol. 2009 doi: 10.1002/0471141755.ph0558s47. Chapter 5. [DOI] [PubMed] [Google Scholar]
- 11.Wirtz S, Neurath MF. Mouse models of inflammatory bowel disease. Advanced Drug Delivery Reviews. 2007;59:1073–1083. doi: 10.1016/j.addr.2007.07.003. [DOI] [PubMed] [Google Scholar]
- 12.Kühn R, Löhler J, Rennick D, et al. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263–274. doi: 10.1016/0092-8674(93)80068-p. [DOI] [PubMed] [Google Scholar]
- 13.Sugawara K, Olson TS, Moskaluk CA, et al. Linkage to peroxisome proliferator-activated receptor-γ in SAMP1/YitFc mice and in human Crohn’s disease. Gastroenterology. 2005;128:351–360. doi: 10.1053/j.gastro.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 14.Panwala CM, Jones JC, Viney JL. A novel model of inflammatory bowel disease: mice deficient for the multiple drug resistance gene, mdr1a, spontaneously develop colitis. J Immunol. 1998;161:5733–5744. [PubMed] [Google Scholar]
- 15.Chaudhry A, Samstein RM, Treuting P, et al. Interleukin-10 signaling in regulatory T cells is required for suppression of Th17 cell-mediated inflammation. Immunity. 2011;34:566–578. doi: 10.1016/j.immuni.2011.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hontecillas R, Bassaganya-Riera J. Peroxisome Proliferator-Activated Receptor γ Is Required for Regulatory CD4+ T Cell-Mediated Protection against Colitis. J Immunol. 2007;178:2940–2949. doi: 10.4049/jimmunol.178.5.2940. [DOI] [PubMed] [Google Scholar]
- 17.Tanner SM, Staley EM, Lorenz RG. Altered generation of induced regulatory T cells in the FVB.mdr1a|[minus]|/|[minus]| mouse model of colitis. Mucosal Immunology. 2012 doi: 10.1038/mi.2012.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Morrissey PJ, Charrier K. Induction of wasting disease in SCID mice by the transfer of normal CD4+/CD45RBhi T cells and the regulation of this autoreactivity by CD4+/CD45RBlo T cells. Research in immunology. 1994;145:357–362. doi: 10.1016/s0923-2494(94)80200-9. [DOI] [PubMed] [Google Scholar]
- 19.Powrie F, Leach MW, Mauze S, et al. Phenotypically distinct subsets of CD4+ T cells induce or protect from chronic intestinal inflammation in C. B-17 scid mice. Int Immunol. 1993;5:1461–1471. doi: 10.1093/intimm/5.11.1461. [DOI] [PubMed] [Google Scholar]
- 20.Boehm F, Martin M, Kesselring R, et al. Deletion of Foxp3+ regulatory T cells in genetically targeted mice supports development of intestinal inflammation. BMC Gastroenterol. 2012:12. doi: 10.1186/1471-230X-12-97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Elson CO, Cong Y, McCracken VJ, et al. Experimental models of inflammatory bowel disease reveal innate, adaptive, and regulatory mechanisms of host dialogue with the microbiota. Immunological Reviews. 2005;206:260–276. doi: 10.1111/j.0105-2896.2005.00291.x. [DOI] [PubMed] [Google Scholar]
- 22.Hori S, Nomura T, Sakaguchi S. Control of Regulatory T Cell Development by the Transcription Factor Foxp3. Science. 2003;299:1057–1061. [PubMed] [Google Scholar]
- 23.Feuerer M, Hill JA, Kretschmer K, et al. Genomic definition of multiple ex vivo regulatory T cell subphenotypes. Proc Natl Acad Sci USA. 2010;107:5919–5924. doi: 10.1073/pnas.1002006107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Haribhai D, Williams JB, Jia S, et al. A requisite role for induced regulatory T cells in tolerance based on expanding antigen receptor diversity. Immunity. 2011;35:109–122. doi: 10.1016/j.immuni.2011.03.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Josefowicz SZ, Rudensky A. Control of regulatory T cell lineage commitment and maintenance. Immunity. 2009;30:616–625. doi: 10.1016/j.immuni.2009.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Relland LM, Williams JB, Relland GN, et al. The TCR Repertoires of Regulatory and Conventional T Cells Specific for the Same Foreign Antigen Are Distinct. J Immunol. 2012 doi: 10.4049/jimmunol.1102646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tanoue T, Honda K. Induction of Treg cells in the mouse colonic mucosa: a central mechanism to maintain host-microbiota homeostasis. Semin Immunol. 2012;24:50–57. doi: 10.1016/j.smim.2011.11.009. [DOI] [PubMed] [Google Scholar]
- 28.Zheng Y, Josefowicz S, Chaudhry A, et al. Role of conserved non-coding DNA elements in the Foxp3 gene in regulatory T-cell fate. Nature. 2010;463:808–812. doi: 10.1038/nature08750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Almeida ARM, Legrand N, Papiernik M, et al. Homeostasis of peripheral CD4+ T cells: IL-2R alpha and IL-2 shape a population of regulatory cells that controls CD4+ T cell numbers. J Immunol. 2002;169:4850–4860. doi: 10.4049/jimmunol.169.9.4850. [DOI] [PubMed] [Google Scholar]
- 30.Malek TR, Yu A, Vincek V, et al. CD4 regulatory Tcells prevent lethal autoimmunity in IL-2Rbeta-deficient miceImplications for the nonredundant function of IL-2. Immunity. 2002;17:167–178. doi: 10.1016/s1074-7613(02)00367-9. [DOI] [PubMed] [Google Scholar]
- 31.Papiernik M, de Moraes ML, Pontoux C, et al. Regulatory CD4 T cells: expression of IL-2R alpha chain, resistance to clonal deletion and IL-2 dependency. Int Immunol. 1998;10:371–378. doi: 10.1093/intimm/10.4.371. [DOI] [PubMed] [Google Scholar]
- 32.Setoguchi R, Hori S, Takahashi T, et al. Homeostatic maintenance of natural Foxp3+ CD25+ CD4+ regulatory T cells by interleukin (IL)-2 and induction of autoimmune disease by IL-2 neutralization. J Exp Med. 2005;201:723–735. doi: 10.1084/jem.20041982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Caudy AA, Reddy ST, Chatila T, et al. CD25 deficiency causes an immune dysregulation, polyendocrinopathy, enteropathy, X-linked-like syndrome, and defective IL-10 expression from CD4 lymphocytes. J Allergy Clin Immunol. 2007;119:482–487. doi: 10.1016/j.jaci.2006.10.007. [DOI] [PubMed] [Google Scholar]
- 34.Barnes MJ, Powrie F. Regulatory T cells reinforce intestinal homeostasis. Immunity. 2009;31:401–411. doi: 10.1016/j.immuni.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 35.Bilate AB, Lafaille JJ. It takes two to tango. Immunity. 2011;35:6–8. doi: 10.1016/j.immuni.2011.07.003. [DOI] [PubMed] [Google Scholar]
- 36.Chen W, Jin W, Hardegen N, et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J Exp Med. 2003;198:1875–1886. doi: 10.1084/jem.20030152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Riley JL, June CH, Blazar BR. Human T regulatory cell therapy: take a billion or so and call me in the morning. Immunity. 2009;30:656–665. doi: 10.1016/j.immuni.2009.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Haribhai D, Lin W, Edwards B, et al. A central role for induced regulatory T cells in tolerance induction in experimental colitis. J Immunol. 2009;182:3461–3468. doi: 10.4049/jimmunol.0802535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Atarashi K, Umesaki Y, Honda K. Microbiotal influence on T cell subset development. Semin Immunol. 2011;23:146–153. doi: 10.1016/j.smim.2011.01.010. [DOI] [PubMed] [Google Scholar]
- 40.Weiner HL. Induction and mechanism of action of transforming growth factor-beta-secreting Th3 regulatory cells. Immunological reviews. 2001;182:207–214. doi: 10.1034/j.1600-065x.2001.1820117.x. [DOI] [PubMed] [Google Scholar]
- 41.Carrier Y, Yuan J, Kuchroo VK, et al. Th3 cells in peripheral tolerance II TGF-beta-transgenic Th3 cells rescue IL-2-deficient mice from autoimmunity. J Immunol. 2007;178:172–178. doi: 10.4049/jimmunol.178.1.172. [DOI] [PubMed] [Google Scholar]
- 42.Fantini MC, Rizzo A, Fina D, et al. Smad7 controls resistance of colitogenic T cells to regulatory T cell-mediated suppression. Gastroenterology. 2009;136:1308–1316. doi: 10.1053/j.gastro.2008.12.053. e1301–1303. [DOI] [PubMed] [Google Scholar]
- 43.Zheng SG, Gray JD, Ohtsuka K, et al. Generation ex vivo of TGF-beta-producing regulatory T cells from CD4+CD25− precursors. J Immunol. 2002;169:4183–4189. doi: 10.4049/jimmunol.169.8.4183. [DOI] [PubMed] [Google Scholar]
- 44.Tone Y, Furuuchi K, Kojima Y, et al. Smad3 and NFAT cooperate to induce Foxp3 expression through its enhancer. Nature immunology. 2008;9:194–202. doi: 10.1038/ni1549. [DOI] [PubMed] [Google Scholar]
- 45.Josefowicz SZ, Niec RE, Kim HY, et al. Extrathymically generated regulatory T cells control mucosal TH2 inflammation. Nature. 2012;482:395–399. doi: 10.1038/nature10772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Mucida D, Park Y, Kim G, et al. Reciprocal TH17 and regulatory T cell differentiation mediated by retinoic acid. Science. 2007;317:256–260. doi: 10.1126/science.1145697. [DOI] [PubMed] [Google Scholar]
- 47.Benson MJ, Pino-Lagos K, Rosemblatt M, et al. All-trans retinoic acid mediates enhanced T reg cell growth, differentiation, and gut homing in the face of high levels of co-stimulation. J Exp Med. 2007;204:1765–1774. doi: 10.1084/jem.20070719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zenewicz LA, Antov A, Flavell RA. CD4 T-cell differentiation and inflammatory bowel disease. Trends Mol Med. 2009;15:199–207. doi: 10.1016/j.molmed.2009.03.002. [DOI] [PubMed] [Google Scholar]
- 49.Zheng Y, Josefowicz SZ, Kas A, et al. Genome-wide analysis of Foxp3 target genes in developing and mature regulatory T cells. Nature. 2007;445:936–940. doi: 10.1038/nature05563. [DOI] [PubMed] [Google Scholar]
- 50.Thornton AM, Korty PE, Tran DQ, et al. Expression of Helios, an Ikaros transcription factor family member, differentiates thymic-derived from peripherally induced Foxp3+ T regulatory cells. J Immunol. 2010;184:3433–3441. doi: 10.4049/jimmunol.0904028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cai Q, Dierich A, Oulad-Abdelghani M, et al. Helios deficiency has minimal impact on T cell development and function. J Immunol. 2009;183:2303–2311. doi: 10.4049/jimmunol.0901407. [DOI] [PubMed] [Google Scholar]
- 52.Akimova T, Beier UH, Wang L, et al. Helios expression is a marker of T cell activation and proliferation. PLoS ONE. 2011:6. doi: 10.1371/journal.pone.0024226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gottschalk RA, Corse E, Allison JP. Expression of Helios in peripherally induced Foxp3+ regulatory T cells. J Immunol. 2012;188:976–980. doi: 10.4049/jimmunol.1102964. [DOI] [PubMed] [Google Scholar]
- 54.Weiss JM, Bilate AM, Gobert M, et al. Neuropilin 1 is expressed on thymus-derived natural regulatory T cells, but not mucosa-generated induced Foxp3+ T reg cells. J Exp Med. 2012;209:1723–1742. doi: 10.1084/jem.20120914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yadav M, Louvet C, Davini D, et al. Neuropilin-1 distinguishes natural and inducible regulatory T cells among regulatory T cell subsets in vivo. J Exp Med. :2012. doi: 10.1084/jem.20120822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Milpied P, Renand A, Bruneau J, et al. Neuropilin-1 is not a marker of human Foxp3+ Treg. European journal of immunology. 2009;39:1466–1471. doi: 10.1002/eji.200839040. [DOI] [PubMed] [Google Scholar]
- 57.Janson PCJ, Winerdal ME, Marits P, et al. FOXP3 promoter demethylation reveals the committed Treg population in humans. PLoS ONE. 2008:3. doi: 10.1371/journal.pone.0001612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Polansky JK, Kretschmer K, Freyer J, et al. DNA methylation controls Foxp3 gene expression. European journal of immunology. 2008;38:1654–1663. doi: 10.1002/eji.200838105. [DOI] [PubMed] [Google Scholar]
- 59.Lathrop SK, Santacruz NA, Pham D, et al. Antigen-specific peripheral shaping of the natural regulatory T cell population. J Exp Med. 2008;205:3105–3117. doi: 10.1084/jem.20081359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Miyara M, Yoshioka Y, Kitoh A, et al. Functional delineation and differentiation dynamics of human CD4+ T cells expressing the FoxP3 transcription factor. Immunity. 2009;30:899–911. doi: 10.1016/j.immuni.2009.03.019. [DOI] [PubMed] [Google Scholar]
- 61.Schmetterer KG, Neunkirchner A, Pickl WF. Naturally occurring regulatory T cells: markers, mechanisms, and manipulation. FASEB J. 2012;26:2253–2276. doi: 10.1096/fj.11-193672. [DOI] [PubMed] [Google Scholar]
- 62.Chen W, Frank ME, Jin W, et al. TGF-beta released by apoptotic T cells contributes to an immunosuppressive milieu. Immunity. 2001;14:715–725. doi: 10.1016/s1074-7613(01)00147-9. [DOI] [PubMed] [Google Scholar]
- 63.Diebold RJ, Eis MJ, Yin M, et al. Early-onset multifocal inflammation in the transforming growth factor beta 1-null mouse is lymphocyte mediated. Proc Natl Acad Sci USA. 1995;92:12215–12219. doi: 10.1073/pnas.92.26.12215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kobayashi S, Yoshida K, Ward JM, et al. Beta 2-microglobulin-deficient background ameliorates lethal phenotype of the TGF-beta 1 null mouse. J Immunol. 1999;163:4013–4019. [PubMed] [Google Scholar]
- 65.Letterio JJ, Geiser AG, Kulkarni AB, et al. Autoimmunity associated with TGF-beta1-deficiency in mice is dependent on MHC class II antigen expression. J Clin Invest. 1996;98:2109–2119. doi: 10.1172/JCI119017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Li MO, Sanjabi S, Flavell RA. Transforming growth factor-beta controls development, homeostasis, and tolerance of T cells by regulatory T cell-dependent and -independent mechanisms. Immunity. 2006;25:455–471. doi: 10.1016/j.immuni.2006.07.011. [DOI] [PubMed] [Google Scholar]
- 67.Marie JC, Liggitt D, Rudensky AY. Cellular mechanisms of fatal early-onset autoimmunity in mice with the T cell-specific targeting of transforming growth factor-beta receptor. Immunity. 2006;25:441–454. doi: 10.1016/j.immuni.2006.07.012. [DOI] [PubMed] [Google Scholar]
- 68.Powrie F, Carlino J, Leach MW, et al. A critical role for transforming growth factor-beta but not interleukin 4 in the suppression of T helper type 1-mediated colitis by CD45RB(low) CD4+ T cells. J Exp Med. 1996;183:2669–2674. doi: 10.1084/jem.183.6.2669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Spencer SD, Di Marco F, Hooley J, et al. The orphan receptor CRF2-4 is an essential subunit of the interleukin 10 receptor. J Exp Med. 1998;187:571–578. doi: 10.1084/jem.187.4.571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hagenbaugh A, Sharma S, Dubinett SM, et al. Altered immune responses in interleukin 10 transgenic mice. J Exp Med. 1997;185:2101–2110. doi: 10.1084/jem.185.12.2101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Powrie F, Leach MW, Mauze S, et al. Inhibition of Th1 responses prevents inflammatory bowel disease in scid mice reconstituted with CD45RBhi CD4+ T cells. Immunity. 1994;1:553–562. doi: 10.1016/1074-7613(94)90045-0. [DOI] [PubMed] [Google Scholar]
- 72.Steidler L, Hans W, Schotte L, et al. Treatment of murine colitis by Lactococcus lactis secreting interleukin-10. Science. 2000;289:1352–1355. doi: 10.1126/science.289.5483.1352. [DOI] [PubMed] [Google Scholar]
- 73.Franke A, Balschun T, Karlsen TH, et al. Sequence variants in IL10, ARPC2 and multiple other loci contribute to ulcerative colitis susceptibility. Nat Genet. 2008;40:1319–1323. doi: 10.1038/ng.221. [DOI] [PubMed] [Google Scholar]
- 74.Glocker E-O, Kotlarz D, Boztug K, et al. Inflammatory bowel disease and mutations affecting the interleukin-10 receptor. N Engl J Med. 2009;361:2033–2045. doi: 10.1056/NEJMoa0907206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Rubtsov YP, Rasmussen JP, Chi EY, et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity. 2008;28:546–558. doi: 10.1016/j.immuni.2008.02.017. [DOI] [PubMed] [Google Scholar]
- 76.Maloy KJ, Salaun L, Cahill R, et al. CD4+CD25+ T(R) cells suppress innate immune pathology through cytokine-dependent mechanisms. J Exp Med. 2003;197:111–119. doi: 10.1084/jem.20021345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Uhlig HH, Coombes J, Mottet C, et al. Characterization of Foxp3+CD4+CD25+ and IL-10-secreting CD4+CD25+ T cells during cure of colitis. J Immunol. 2006;177:5852–5860. doi: 10.4049/jimmunol.177.9.5852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Maynard CL, Harrington LE, Janowski KM, et al. Regulatory T cells expressing interleukin 10 develop from Foxp3+ and Foxp3|[minus]| precursor cells in the absence of interleukin 10. Nature Immunology. 2007;8:931–941. doi: 10.1038/ni1504. [DOI] [PubMed] [Google Scholar]
- 79.Collison LW, Workman CJ, Kuo TT, et al. The inhibitory cytokine IL-35 contributes to regulatory T-cell function. Nature. 2007;450:566–569. doi: 10.1038/nature06306. [DOI] [PubMed] [Google Scholar]
- 80.Liu J-Q, Liu Z, Zhang X, et al. Increased Th17 and regulatory T cell responses in EBV-induced gene 3-deficient mice lead to marginally enhanced development of autoimmune encephalomyelitis. J Immunol. 2012;188:3099–3106. doi: 10.4049/jimmunol.1100106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Whitehead GS, Wilson RH, Nakano K, et al. IL-35 production by inducible costimulator (ICOS)-positive regulatory T cells reverses established IL-17-dependent allergic airways disease. J Allergy Clin Immunol. 2012;129:207–215. doi: 10.1016/j.jaci.2011.08.009. e201-205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Borsellino G, Kleinewietfeld M, Di Mitri D, et al. Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood. 2007;110:1225–1232. doi: 10.1182/blood-2006-12-064527. [DOI] [PubMed] [Google Scholar]
- 83.Deaglio S, Dwyer KM, Gao W, et al. Adenosine generation catalyzed by CD39 and CD73 expressed on regulatory T cells mediates immune suppression. J Exp Med. 2007;204:1257–1265. doi: 10.1084/jem.20062512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Naganuma M, Wiznerowicz EB, Lappas CM, et al. Cutting edge: Critical role for A2A adenosine receptors in the T cell-mediated regulation of colitis. J Immunol. 2006;177:2765–2769. doi: 10.4049/jimmunol.177.5.2765. [DOI] [PubMed] [Google Scholar]
- 85.Odashima M, Bamias G, Rivera-Nieves J, et al. Activation of A2A adenosine receptor attenuates intestinal inflammation in animal models of inflammatory bowel disease. Gastroenterology. 2005;129:26–33. doi: 10.1053/j.gastro.2005.05.032. [DOI] [PubMed] [Google Scholar]
- 86.Thornton AM, Shevach EM. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J Exp Med. 1998;188:287–296. doi: 10.1084/jem.188.2.287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Pandiyan P, Zheng L, Ishihara S, et al. CD4+CD25+Foxp3+ regulatory T cells induce cytokine deprivation-mediated apoptosis of effector CD4+ T cells. Nature immunology. 2007;8:1353–1362. doi: 10.1038/ni1536. [DOI] [PubMed] [Google Scholar]
- 88.Gondek DC, Lu L-F, Quezada SA, et al. Cutting edge: contact-mediated suppression by CD4+CD25+ regulatory cells involves a granzyme B-dependent, perforin-independent mechanism. J Immunol. 2005;174:1783–1786. doi: 10.4049/jimmunol.174.4.1783. [DOI] [PubMed] [Google Scholar]
- 89.Grossman WJ, Verbsky JW, Barchet W, et al. Human T regulatory cells can use the perforin pathway to cause autologous target cell death. Immunity. 2004;21:589–601. doi: 10.1016/j.immuni.2004.09.002. [DOI] [PubMed] [Google Scholar]
- 90.Zhao D-M, Thornton AM, DiPaolo RJ, et al. Activated CD4+CD25+ T cells selectively kill B lymphocytes. Blood. 2006;107:3925–3932. doi: 10.1182/blood-2005-11-4502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gondek DC, Devries V, Nowak EC, et al. Transplantation survival is maintained by granzyme B+ regulatory cells and adaptive regulatory T cells. J Immunol. 2008;181:4752–4760. doi: 10.4049/jimmunol.181.7.4752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Salti SM, Hammelev EM, Grewal JL, et al. Granzyme B regulates antiviral CD8+ T cell responses. J Immunol. 2011;187:6301–6309. doi: 10.4049/jimmunol.1100891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Onishi Y, Fehervari Z, Yamaguchi T, et al. Foxp3+ natural regulatory T cells preferentially form aggregates on dendritic cells in vitro and actively inhibit their maturation. Proc Natl Acad Sci USA. 2008;105:10113–10118. doi: 10.1073/pnas.0711106105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Tang Q, Krummel MF. Imaging the function of regulatory T cells in vivo. Curr Opin Immunol. 2006;18:496–502. doi: 10.1016/j.coi.2006.05.007. [DOI] [PubMed] [Google Scholar]
- 95.Read S, Greenwald R, Izcue A, et al. Blockade of CTLA-4 on CD4+CD25+ regulatory T cells abrogates their function in vivo. J Immunol. 2006;177:4376–4383. doi: 10.4049/jimmunol.177.7.4376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wing K, Onishi Y, Prieto-Martin P, et al. CTLA-4 control over Foxp3+ regulatory T cell function. Science. 2008;322:271–275. doi: 10.1126/science.1160062. [DOI] [PubMed] [Google Scholar]
- 97.Peggs KS, Quezada SA, Allison JP. Cancer immunotherapy: co-stimulatory agonists and co-inhibitory antagonists. Clin Exp Immunol. 2009;157:9–19. doi: 10.1111/j.1365-2249.2009.03912.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Plevy SE, Targan SR. Future therapeutic approaches for inflammatory bowel diseases. Gastroenterology. 2011;140:1838–1846. doi: 10.1053/j.gastro.2011.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Liang B, Workman C, Lee J, et al. Regulatory T cells inhibit dendritic cells by lymphocyte activation gene-3 engagement of MHC class II. J Immunol. 2008;180:5916–5926. doi: 10.4049/jimmunol.180.9.5916. [DOI] [PubMed] [Google Scholar]
- 100.Francisco LM, Sage PT, Sharpe AH. The PD-1 pathway in tolerance and autoimmunity. Immunological reviews. 2010;236:219–242. doi: 10.1111/j.1600-065X.2010.00923.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Shevach EM. Mechanisms of foxp3+ T regulatory cell-mediated suppression. Immunity. 2009;30:636–645. doi: 10.1016/j.immuni.2009.04.010. [DOI] [PubMed] [Google Scholar]
- 102.Annacker O, Coombes JL, Malmstrom V, et al. Essential role for CD103 in the T cell-mediated regulation of experimental colitis. J Exp Med. 2005;202:1051–1061. doi: 10.1084/jem.20040662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Johansson-Lindbom B, Agace WW. Generation of gut-homing T cells and their localization to the small intestinal mucosa. Immunological reviews. 2007;215:226–242. doi: 10.1111/j.1600-065X.2006.00482.x. [DOI] [PubMed] [Google Scholar]
- 104.Schneider MA, Meingassner JG, Lipp M, et al. CCR7 is required for the in vivo function of CD4+ CD25+ regulatory T cells. J Exp Med. 2007;204:735–745. doi: 10.1084/jem.20061405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Yuan Q, Bromley SK, Means TK, et al. CCR4-dependent regulatory T cell function in inflammatory bowel disease. J Exp Med. 2007;204:1327–1334. doi: 10.1084/jem.20062076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lodes MJ, Cong Y, Elson CO, et al. Bacterial flagellin is a dominant antigen in Crohn disease. J Clin Invest. 2004;113:1296–1306. doi: 10.1172/JCI20295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Vermeulen N, de Béeck KO, Vermeire S, et al. Identification of a novel autoantigen in inflammatory bowel disease by protein microarray. Inflamm Bowel Dis. 2011;17:1291–1300. doi: 10.1002/ibd.21508. [DOI] [PubMed] [Google Scholar]
- 108.Zhou L, Lopes JE, Chong MMW, et al. TGF-beta-induced Foxp3 inhibits T(H)17 cell differentiation by antagonizing RORgammat function. Nature. 2008;453:236–240. doi: 10.1038/nature06878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Monteleone G, Monteleone I, Fina D, et al. Interleukin-21 enhances T-helper cell type I signaling and interferon-gamma production in Crohn's disease. Gastroenterology. 2005;128:687–694. doi: 10.1053/j.gastro.2004.12.042. [DOI] [PubMed] [Google Scholar]
- 110.Hue S, Ahern P, Buonocore S, et al. Interleukin-23 drives innate and T cell-mediated intestinal inflammation. J Exp Med. 2006;203:2473–2483. doi: 10.1084/jem.20061099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kullberg MC, Jankovic D, Feng CG, et al. IL-23 plays a key role in Helicobacter hepaticus-induced T cell-dependent colitis. J Exp Med. 2006;203:2485–2494. doi: 10.1084/jem.20061082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Yen D, Cheung J, Scheerens H, et al. IL-23 is essential for T cell-mediated colitis and promotes inflammation via IL-17 and IL-6. J Clin Invest. 2006;116:1310–1316. doi: 10.1172/JCI21404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Becker C, Dornhoff H, Neufert C, et al. Cutting edge: IL-23 cross-regulates IL-12 production in T cell-dependent experimental colitis. J Immunol. 2006;177:2760–2764. doi: 10.4049/jimmunol.177.5.2760. [DOI] [PubMed] [Google Scholar]
- 114.Sarin R, Wu X, Abraham C. Inflammatory disease protective R381Q IL23 receptor polymorphism results in decreased primary CD4+ and CD8+ human T-cell functional responses. Proc Natl Acad Sci USA. 2011;108:9560–9565. doi: 10.1073/pnas.1017854108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Curotto de Lafaille MA, Lafaille JJ. Natural and adaptive foxp3+ regulatory T cells: more of the same or a division of labor? Immunity. 2009;30:626–635. doi: 10.1016/j.immuni.2009.05.002. [DOI] [PubMed] [Google Scholar]
- 116.Eisenstein EM, Williams CB. The T(reg)/Th17 cell balance: a new paradigm for autoimmunity. Pediatr Res. 2009;65:26R–31R. doi: 10.1203/PDR.0b013e31819e76c7. [DOI] [PubMed] [Google Scholar]
- 117.Schmitt EG, Haribhai D, Williams JB, et al. IL-10 produced by induced regulatory T cells (iTregs) controls colitis and pathogenic ex-iTregs during immunotherapy. J Immunol. 2012:189. doi: 10.4049/jimmunol.1200936. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Lathrop SK, Bloom SM, Rao SM, et al. Peripheral education of the immune system by colonic commensal microbiota. Nature. 2011;478:250–254. doi: 10.1038/nature10434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Bevins CL, Salzman NH. The potter's wheel: the host's role in sculpting its microbiota. Cell Mol Life Sci. 2011;68:3675–3685. doi: 10.1007/s00018-011-0830-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Bouskra D, Brézillon C, Bérard M, et al. Lymphoid tissue genesis induced by commensals through NOD1 regulates intestinal homeostasis. Nature. 2008;456:507–510. doi: 10.1038/nature07450. [DOI] [PubMed] [Google Scholar]
- 121.Salzman NH. Microbiota-immune system interaction: an uneasy alliance. Curr Opin Microbiol. 2011;14:99–105. doi: 10.1016/j.mib.2010.09.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Blumberg R, Powrie F. Microbiota, disease, and back to health: a metastable journey. Sci Transl Med. 2012:4. doi: 10.1126/scitranslmed.3004184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Kahn SA, Gorawara-Bhat R, Rubin DT. Fecal bacteriotherapy for ulcerative colitis: patients are ready, are we? Inflamm Bowel Dis. 2012;18:676–684. doi: 10.1002/ibd.21775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Ostman S, Rask C, Wold AE, et al. Impaired regulatory T cell function in germ-free mice. European journal of immunology. 2006;36:2336–2346. doi: 10.1002/eji.200535244. [DOI] [PubMed] [Google Scholar]
- 125.Singh B, Read S, Asseman C, et al. Control of intestinal inflammation by regulatory T cells. Immunological reviews. 2001;182:190–200. doi: 10.1034/j.1600-065x.2001.1820115.x. [DOI] [PubMed] [Google Scholar]
- 126.Strauch UG, Obermeier F, Grunwald N, et al. Influence of intestinal bacteria on induction of regulatory T cells: lessons from a transfer model of colitis. Gut. 2005;54:1546–1552. doi: 10.1136/gut.2004.059451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Annacker O, Burlen-Defranoux O, Pimenta-Araujo R, et al. Regulatory CD4 T cells control the size of the peripheral activated/memory CD4 T cell compartment. J Immunol. 2000;164:3573–3580. doi: 10.4049/jimmunol.164.7.3573. [DOI] [PubMed] [Google Scholar]
- 128.Ivanov II, Frutos RdL, Manel N, et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe. 2008;4:337–349. doi: 10.1016/j.chom.2008.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Hall JA, Bouladoux N, Sun CM, et al. Commensal DNA limits regulatory T cell conversion and is a natural adjuvant of intestinal immune responses. Immunity. 2008;29:637–649. doi: 10.1016/j.immuni.2008.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Shomer NH, Dangler CA, Schrenzel MD, et al. Helicobacter bilis-induced inflammatory bowel disease in scid mice with defined flora. Infect Immun. 1997;65:4858–4864. doi: 10.1128/iai.65.11.4858-4864.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Cahill RJ, Foltz CJ, Fox JG, et al. Inflammatory bowel disease: an immunity-mediated condition triggered by bacterial infection with Helicobacter hepaticus. Infect Immun. 1997;65:3126–3131. doi: 10.1128/iai.65.8.3126-3131.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Kullberg MC, Jankovic D, Gorelick PL, et al. Bacteria-triggered CD4(+) T regulatory cells suppress Helicobacter hepaticus-induced colitis. J Exp Med. 2002;196:505–515. doi: 10.1084/jem.20020556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ivanov II, Atarashi K, Manel N, et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell. 2009;139:485–498. doi: 10.1016/j.cell.2009.09.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Stepankova R, Powrie F, Kofronova O, et al. Segmented filamentous bacteria in a defined bacterial cocktail induce intestinal inflammation in SCID mice reconstituted with CD45RBhigh CD4+ T cells. Inflamm Bowel Dis. 2007;13:1202–1211. doi: 10.1002/ibd.20221. [DOI] [PubMed] [Google Scholar]
- 135.Gaboriau-Routhiau V, Rakotobe S, Lécuyer E, et al. The key role of segmented filamentous bacteria in the coordinated maturation of gut helper T cell responses. Immunity. 2009;31:677–689. doi: 10.1016/j.immuni.2009.08.020. [DOI] [PubMed] [Google Scholar]
- 136.Salzman NH, Hung K, Haribhai D, et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nature immunology. 2010;11:76–83. doi: 10.1038/ni.1825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Umesaki Y, Setoyama H, Matsumoto S, et al. Differential roles of segmented filamentous bacteria and clostridia in development of the intestinal immune system. Infect Immun. 1999;67:3504–3511. doi: 10.1128/iai.67.7.3504-3511.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Atarashi K, Nishimura J, Shima T, et al. ATP drives lamina propria T(H)17 cell differentiation. Nature. 2008;455:808–812. doi: 10.1038/nature07240. [DOI] [PubMed] [Google Scholar]
- 139.Atarashi K, Tanoue T, Shima T, et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science. 2011;331:337–341. doi: 10.1126/science.1198469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Sokol H, Pigneur B, Watterlot L, et al. Faecalibacterium prausnitzii is an anti-inflammatory commensal bacterium identified by gut microbiota analysis of Crohn disease patients. Proc Natl Acad Sci USA. 2008;105:16731–16736. doi: 10.1073/pnas.0804812105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Karimi K, Inman MD, Bienenstock J, et al. Lactobacillus reuteri-induced regulatory T cells protect against an allergic airway response in mice. Am J Respir Crit Care Med. 2009;179:186–193. doi: 10.1164/rccm.200806-951OC. [DOI] [PubMed] [Google Scholar]
- 142.Livingston M, Loach D, Wilson M, et al. Gut commensal Lactobacillus reuteri 100-23 stimulates an immunoregulatory response. Immunol Cell Biol. 2010;88:99–102. doi: 10.1038/icb.2009.71. [DOI] [PubMed] [Google Scholar]
- 143.O'Mahony C, Scully P, O'Mahony D, et al. Commensal-induced regulatory T cells mediate protection against pathogen-stimulated NF-kappaB activation. PLoS Pathog. 2008:4. doi: 10.1371/journal.ppat.1000112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Konieczna P, Groeger D, Ziegler M, et al. Bifidobacterium infantis 35624 administration induces Foxp3 T regulatory cells in human peripheral blood: potential role for myeloid and plasmacytoid dendritic cells. Gut. 2012;61:354–366. doi: 10.1136/gutjnl-2011-300936. [DOI] [PubMed] [Google Scholar]
- 145.Di Giacinto C, Marinaro M, Sanchez M, et al. Probiotics ameliorate recurrent Th1-mediated murine colitis by inducing IL-10 and IL-10-dependent TGF-beta-bearing regulatory cells. J Immunol. 2005;174:3237–3246. doi: 10.4049/jimmunol.174.6.3237. [DOI] [PubMed] [Google Scholar]
- 146.Round JL, Mazmanian SK. Inducible Foxp3+ regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc Natl Acad Sci USA. 2010;107:12204–12209. doi: 10.1073/pnas.0909122107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Mazmanian SK, Round JL, Kasper DL. A microbial symbiosis factor prevents intestinal inflammatory disease. Nature. 2008;453:620–625. doi: 10.1038/nature07008. [DOI] [PubMed] [Google Scholar]
- 148.Wu S, Rhee K-J, Albesiano E, et al. A human colonic commensal promotes colon tumorigenesis via activation of T helper type 17 T cell responses. Nat Med. 2009;15:1016–1022. doi: 10.1038/nm.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Buckner JH. Mechanisms of impaired regulation by CD4(+)CD25(+)FOXP3(+) regulatory T cells in human autoimmune diseases. Nat Rev Immunol. 2010;10:849–859. doi: 10.1038/nri2889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Lin W, Truong N, Grossman WJ, et al. Allergic dysregulation and hyperimmunoglobulinemia E in Foxp3 mutant mice. J Allergy Clin Immunol. 2005;116:1106–1115. doi: 10.1016/j.jaci.2005.08.046. [DOI] [PubMed] [Google Scholar]
- 151.Xu L, Kitani A, Fuss I, et al. Cutting edge: regulatory T cells induce CD4+CD25-Foxp3− T cells or are self-induced to become Th17 cells in the absence of exogenous TGF-beta. J Immunol. 2007;178:6725–6729. doi: 10.4049/jimmunol.178.11.6725. [DOI] [PubMed] [Google Scholar]
- 152.Murai M, Turovskaya O, Kim G, et al. Interleukin 10 acts on regulatory T cells to maintain expression of the transcription factor Foxp3 and suppressive function in mice with colitis. Nature immunology. 2009;10:1178–1184. doi: 10.1038/ni.1791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Tsuji M, Komatsu N, Kawamoto S, et al. Preferential generation of follicular B helper T cells from Foxp3+ T cells in gut Peyer's patches. Science. 2009;323:1488–1492. doi: 10.1126/science.1169152. [DOI] [PubMed] [Google Scholar]
- 154.Zhou X, Bailey-Bucktrout SL, Jeker LT, et al. Instability of the transcription factor Foxp3 leads to the generation of pathogenic memory T cells in vivo. Nature immunology. 2009;10:1000–1007. doi: 10.1038/ni.1774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Rubtsov YP, Niec RE, Josefowicz S, et al. Stability of the regulatory T cell lineage in vivo. Science. 2010;329:1667–1671. doi: 10.1126/science.1191996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Yates J, Rovis F, Mitchell P, et al. The maintenance of human CD4+CD25+ regulatory T cell function: IL-2, IL-4, IL-7 and IL-15 preserve optimal suppressive potency in vitro. Int Immunol. 2007;19:785–799. doi: 10.1093/intimm/dxm047. [DOI] [PubMed] [Google Scholar]
- 157.Choi BK, Bae JS, Choi EM, et al. 4-1BB-dependent inhibition of immunosuppression by activated CD4+CD25+ T cells. J Leukoc Biol. 2004;75:785–791. doi: 10.1189/jlb.1003491. [DOI] [PubMed] [Google Scholar]
- 158.Takeda I, Ine S, Killeen N, et al. Distinct roles for the OX40-OX40 ligand interaction in regulatory and nonregulatory T cells. J Immunol. 2004;172:3580–3589. doi: 10.4049/jimmunol.172.6.3580. [DOI] [PubMed] [Google Scholar]
- 159.Holmén N, Lundgren A, Lundin S, et al. Functional CD4+CD25high regulatory T cells are enriched in the colonic mucosa of patients with active ulcerative colitis and increase with disease activity. Inflamm Bowel Dis. 2006;12:447–456. doi: 10.1097/00054725-200606000-00003. [DOI] [PubMed] [Google Scholar]
- 160.Hölttä V, Klemetti P, Sipponen T, et al. IL-23/IL-17 immunity as a hallmark of Crohn's disease. Inflamm Bowel Dis. 2008;14:1175–1184. doi: 10.1002/ibd.20475. [DOI] [PubMed] [Google Scholar]
- 161.Makita S, Kanai T, Oshima S, et al. CD4+CD25bright T cells in human intestinal lamina propria as regulatory cells. J Immunol. 2004;173:3119–3130. doi: 10.4049/jimmunol.173.5.3119. [DOI] [PubMed] [Google Scholar]
- 162.Maul J, Loddenkemper C, Mundt P, et al. Peripheral and intestinal regulatory CD4+ CD25(high) T cells in inflammatory bowel disease. Gastroenterology. 2005;128:1868–1878. doi: 10.1053/j.gastro.2005.03.043. [DOI] [PubMed] [Google Scholar]
- 163.Saruta M, Yu QT, Fleshner PR, et al. Characterization of FOXP3+CD4+ regulatory T cells in Crohn's disease. Clin Immunol. 2007;125:281–290. doi: 10.1016/j.clim.2007.08.003. [DOI] [PubMed] [Google Scholar]
- 164.Takahashi M, Nakamura K, Honda K, et al. An inverse correlation of human peripheral blood regulatory T cell frequency with the disease activity of ulcerative colitis. Dig Dis Sci. 2006;51:677–686. doi: 10.1007/s10620-006-3191-2. [DOI] [PubMed] [Google Scholar]
- 165.Eastaff-Leung N, Mabarrack N, Barbour A, et al. Foxp3+ regulatory T cells, Th17 effector cells, and cytokine environment in inflammatory bowel disease. J Clin Immunol. 2010;30:80–89. doi: 10.1007/s10875-009-9345-1. [DOI] [PubMed] [Google Scholar]
- 166.Kelsen J, Agnholt J, Hoffmann HJ, et al. FoxP3(+)CD4(+)CD25(+) T cells with regulatory properties can be cultured from colonic mucosa of patients with Crohn's disease. Clin Exp Immunol. 2005;141:549–557. doi: 10.1111/j.1365-2249.2005.02876.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Rahman MK, Midtling EH, Svingen PA, et al. The pathogen recognition receptor NOD2 regulates human FOXP3+ T cell survival. J Immunol. 2010;184:7247–7256. doi: 10.4049/jimmunol.0901479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Yu QT, Saruta M, Avanesyan A, et al. Expression and functional characterization of FOXP3+ CD4+ regulatory T cells in ulcerative colitis. Inflamm Bowel Dis. 2007;13:191–199. doi: 10.1002/ibd.20053. [DOI] [PubMed] [Google Scholar]
- 169.Hardenberg G, Steiner TS, Levings MK. Environmental influences on T regulatory cells in inflammatory bowel disease. Semin Immunol. 2011;23:130–138. doi: 10.1016/j.smim.2011.01.012. [DOI] [PubMed] [Google Scholar]
- 170.Koenen HJPM, Smeets RL, Vink PM, et al. Human CD25highFoxp3pos regulatory T cells differentiate into IL-17-producing cells. Blood. 2008;112:2340–2352. doi: 10.1182/blood-2008-01-133967. [DOI] [PubMed] [Google Scholar]
- 171.Monteleone G, Kumberova A, Croft NM, et al. Blocking Smad7 restores TGF-beta1 signaling in chronic inflammatory bowel disease. J Clin Invest. 2001;108:601–609. doi: 10.1172/JCI12821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Monteleone I, Monteleone G, Fina D, et al. A functional role of flip in conferring resistance of Crohn's disease lamina propria lymphocytes to FAS-mediated apoptosis. Gastroenterology. 2006;130:389–397. doi: 10.1053/j.gastro.2005.10.021. [DOI] [PubMed] [Google Scholar]
- 173.Neurath MF, Finotto S, Fuss I, et al. Regulation of T-cell apoptosis in inflammatory bowel disease: to die or not to die, that is the mucosal question. Trends Immunol. 2001;22:21–26. doi: 10.1016/s1471-4906(00)01798-1. [DOI] [PubMed] [Google Scholar]
- 174.Li Z, Arijs I, De Hertogh G, et al. Reciprocal changes of Foxp3 expression in blood and intestinal mucosa in IBD patients responding to infliximab. Inflamm Bowel Dis. 2010;16:1299–1310. doi: 10.1002/ibd.21229. [DOI] [PubMed] [Google Scholar]
- 175.Ricciardelli I, Lindley KJ, Londei M, et al. Anti tumour necrosis-alpha therapy increases the number of FOXP3 regulatory T cells in children affected by Crohn's disease. Immunology. 2008;125:178–183. doi: 10.1111/j.1365-2567.2008.02839.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Hvas CL, Kelsen J, Agnholt J, et al. Discrete changes in circulating regulatory T cells during infliximab treatment of Crohn's disease. Autoimmunity. 2010;43:325–333. doi: 10.3109/08916930903509064. [DOI] [PubMed] [Google Scholar]
- 177.Hippen KL, Merkel SC, Schirm DK, et al. Generation and large-scale expansion of human inducible regulatory T cells that suppress graft-versus-host disease. Am J Transplant. 2011;11:1148–1157. doi: 10.1111/j.1600-6143.2011.03558.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Jiang S, Camara N, Lombardi G, et al. Induction of allopeptide-specific human CD4+CD25+ regulatory T cells ex vivo. Blood. 2003;102:2180–2186. doi: 10.1182/blood-2003-04-1164. [DOI] [PubMed] [Google Scholar]
- 179.Battaglia M, Stabilini A, Migliavacca B, et al. Rapamycin promotes expansion of functional CD4+CD25+FOXP3+ regulatory T cells of both healthy subjects and type 1 diabetic patients. J Immunol. 2006;177:8338–8347. doi: 10.4049/jimmunol.177.12.8338. [DOI] [PubMed] [Google Scholar]
- 180.Tresoldi E, Dell’Albani I, Stabilini A, et al. Stability of human rapamycin-expanded CD4+CD25+ T regulatory cells. Haematologica. 2011;96:1357–1365. doi: 10.3324/haematol.2011.041483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Elinav E, Waks T, Eshhar Z. Redirection of regulatory T cells with predetermined specificity for the treatment of experimental colitis in mice. Gastroenterology. 2008;134:2014–2024. doi: 10.1053/j.gastro.2008.02.060. [DOI] [PubMed] [Google Scholar]
- 182.Desreumaux P, Foussat A, Allez M, et al. Safety and efficacy of antigen-specific regulatory T-cell therapy for patients with refractory Crohn's disease. Gastroenterology. 2012;143:1207–1217. doi: 10.1053/j.gastro.2012.07.116. e1202. [DOI] [PubMed] [Google Scholar]
- 183.Thomson AW, Robbins PD. Tolerogenic dendritic cells for autoimmune disease and transplantation. Ann Rheum Dis. 2008;67(Suppl 3:iii):90–96. doi: 10.1136/ard.2008.099176. [DOI] [PubMed] [Google Scholar]
- 184.Salazar KD, Lankford SM, Brody AR. Mesenchymal stem cells produce Wnt isoforms and TGF-β1 that mediate proliferation and procollagen expression by lung fibroblasts. Am J Physiol Lung Cell Mol Physiol. 2009;297:L1002–L1011. doi: 10.1152/ajplung.90347.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.González MA, Gonzalez-Rey E, Rico L, et al. Adipose-derived mesenchymal stem cells alleviate experimental colitis by inhibiting inflammatory and autoimmune responses. Gastroenterology. 2009;136:978–989. doi: 10.1053/j.gastro.2008.11.041. [DOI] [PubMed] [Google Scholar]
- 186.Bakken JS. Fecal bacteriotherapy for recurrent Clostridium difficile infection. Anaerobe. 2009;15:285–289. doi: 10.1016/j.anaerobe.2009.09.007. [DOI] [PubMed] [Google Scholar]
- 187.Borody TJ, Warren EF, Leis S, et al. Treatment of ulcerative colitis using fecal bacteriotherapy. J Clin Gastroenterol. 2003;37:42–47. doi: 10.1097/00004836-200307000-00012. [DOI] [PubMed] [Google Scholar]
- 188.de Roock S, van Elk M, van Dijk MEA, et al. Lactic acid bacteria differ in their ability to induce functional regulatory T cells in humans. Clin Exp Allergy. 2010;40:103–110. doi: 10.1111/j.1365-2222.2009.03344.x. [DOI] [PubMed] [Google Scholar]
- 189.Kwon H-K, Lee C-G, So J-S, et al. Generation of regulatory dendritic cells and CD4+Foxp3+ T cells by probiotics administration suppresses immune disorders. Proc Natl Acad Sci USA. 2010;107:2159–2164. doi: 10.1073/pnas.0904055107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Pronio A, Montesani C, Butteroni C, et al. Probiotic administration in patients with ileal pouch-anal anastomosis for ulcerative colitis is associated with expansion of mucosal regulatory cells. Inflamm Bowel Dis. 2008;14:662–668. doi: 10.1002/ibd.20369. [DOI] [PubMed] [Google Scholar]
- 191.Wells JM, Mercenier A. Mucosal delivery of therapeutic and prophylactic molecules using lactic acid bacteria. Nature Reviews Microbiology. 2008;6:349–362. doi: 10.1038/nrmicro1840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Braat H, Rottiers P, Hommes DW, et al. A phase I trial with transgenic bacteria expressing interleukin-10 in Crohn's disease. Clin Gastroenterol Hepatol. 2006;4:754–759. doi: 10.1016/j.cgh.2006.03.028. [DOI] [PubMed] [Google Scholar]
- 193.Bai A, Lu N, Guo Y, et al. All-trans retinoic acid down-regulates inflammatory responses by shifting the Treg/Th17 profile in human ulcerative and murine colitis. J Leukoc Biol. 2009;86:959–969. doi: 10.1189/jlb.0109006. [DOI] [PubMed] [Google Scholar]
- 194.Hewison M. Vitamin D and immune function: an overview. Proc Nutr Soc. 2012;71:50–61. doi: 10.1017/S0029665111001650. [DOI] [PubMed] [Google Scholar]
- 195.Mayne CG, Spanier JA, Relland LM, et al. 1,25-Dihydroxyvitamin D3 acts directly on the T lymphocyte vitamin D receptor to inhibit experimental autoimmune encephalomyelitis. European journal of immunology. 2011;41:822–832. doi: 10.1002/eji.201040632. [DOI] [PubMed] [Google Scholar]
- 196.Morales-Tirado V, Wichlan DG, Leimig TE, et al. 1α,25-dihydroxyvitamin D3 (vitamin D3) catalyzes suppressive activity on human natural regulatory T cells, uniquely modulates cell cycle progression, and augments FOXP3. Clin Immunol. 2011;138:212–221. doi: 10.1016/j.clim.2010.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Khalili H, Huang ES, Ananthakrishnan AN, et al. Geographical variation and incidence of inflammatory bowel disease among US women. Gut. 2012 doi: 10.1136/gutjnl-2011-301574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Dresner-Pollak R, Ackerman Z, Eliakim R, et al. The BsmI vitamin D receptor gene polymorphism is associated with ulcerative colitis in Jewish Ashkenazi patients. Genet Test. 2004;8:417–420. doi: 10.1089/gte.2004.8.417. [DOI] [PubMed] [Google Scholar]
- 199.Simmons JD, Mullighan C, Welsh KI, et al. Vitamin D receptor gene polymorphism: association with Crohn's disease susceptibility. Gut. 2000;47:211–214. doi: 10.1136/gut.47.2.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Bruce D, Cantorna MT. Chapter 96 - Vitamin D and Inflammatory Bowel Disease. Vitamin D (Third Edition) San Diego: Academic Press; 2011. pp. 1879–1889. [Google Scholar]
- 201.Nicholson I, Dalzell AM, El-Matary W. Vitamin D as a therapy for colitis: A systematic review. Journal of Crohn's and Colitis. 2012;6:405–411. doi: 10.1016/j.crohns.2012.01.007. [DOI] [PubMed] [Google Scholar]
- 202.Niggemann B, Staden U, Rolinck-Werninghaus C, et al. Specific oral tolerance induction in food allergy. Allergy. 2006;61:808–811. doi: 10.1111/j.1398-9995.2006.01066.x. [DOI] [PubMed] [Google Scholar]
- 203.Kverka M, Zakostelska Z, Klimesova K, et al. Oral administration of Parabacteroides distasonis antigens attenuates experimental murine colitis through modulation of immunity and microbiota composition. Clin Exp Immunol. 2011;163:250–259. doi: 10.1111/j.1365-2249.2010.04286.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Glauben R, Batra A, Fedke I, et al. Histone hyperacetylation is associated with amelioration of experimental colitis in mice. J Immunol. 2006;176:5015–5022. doi: 10.4049/jimmunol.176.8.5015. [DOI] [PubMed] [Google Scholar]
- 205.Tao R, de Zoeten EF, Ozkaynak E, et al. Deacetylase inhibition promotes the generation and function of regulatory T cells. Nat Med. 2007;13:1299–1307. doi: 10.1038/nm1652. [DOI] [PubMed] [Google Scholar]
- 206.Ding C, Xu J, Li J. ABT-874, a fully human monoclonal anti-IL-12/IL-23 antibody for the potential treatment of autoimmune diseases. Curr Opin Investig Drugs. 2008;9:515–522. [PubMed] [Google Scholar]
- 207.Fuss IJ, Becker C, Yang Z, et al. Both IL-12p70 and IL-23 are synthesized during active Crohn's disease and are down-regulated by treatment with anti-IL-12 p40 monoclonal antibody. Inflamm Bowel Dis. 2006;12:9–15. doi: 10.1097/01.mib.0000194183.92671.b6. [DOI] [PubMed] [Google Scholar]
- 208.Sandborn WJ, Feagan BG, Fedorak RN, et al. A randomized trial of Ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with moderate-to-severe Crohn's disease. Gastroenterology. 2008;135:1130–1141. doi: 10.1053/j.gastro.2008.07.014. [DOI] [PubMed] [Google Scholar]
- 209.Ahern PP, Schiering C, Buonocore S, et al. Interleukin-23 Drives Intestinal Inflammation through Direct Activity on T Cells. Immunity. 2010;33:279–288. doi: 10.1016/j.immuni.2010.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Izcue A, Hue S, Buonocore S, et al. Interleukin-23 Restrains Regulatory T Cell Activity to Drive T Cell-Dependent Colitis. Immunity. 2008;28:559–570. doi: 10.1016/j.immuni.2008.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Chatenoud L, Bluestone JA. CD3-specific antibodies: a portal to the treatment of autoimmunity. Nat Rev Immunol. 2007;7:622–632. doi: 10.1038/nri2134. [DOI] [PubMed] [Google Scholar]
- 212.Sandborn WJ, Colombel JF, Frankel M, et al. Anti-CD3 antibody visilizumab is not effective in patients with intravenous corticosteroid-refractory ulcerative colitis. Gut. 2010;59:1485–1492. doi: 10.1136/gut.2009.205443. [DOI] [PubMed] [Google Scholar]
- 213.Massachusetts General Hospital; Brigham and Women's Hospital. ClinicalTrials.gov [Internet] Bethesda (MD): National Library of Medicine (US); Oral OKT3 for the Treatment of Active Ulcerative Colitis. 2000- [cited 2012 Oct 28]. Available from: http://www.clinicaltrials.gov/ct2/show/NCT01287195? NLM Identifier: NCT01287195. [Google Scholar]
- 214.Stüve O, Gold R, Chan A, et al. alpha4-Integrin antagonism with natalizumab: effects and adverse effects. J Neurol. 2008;255(Suppl 6):58–65. doi: 10.1007/s00415-008-6011-0. [DOI] [PubMed] [Google Scholar]
- 215.Feagan BG, Greenberg GR, Wild G, et al. Treatment of active Crohn's disease with MLN0002, a humanized antibody to the alpha4beta7 integrin. Clin Gastroenterol Hepatol. 2008;6:1370–1377. doi: 10.1016/j.cgh.2008.06.007. [DOI] [PubMed] [Google Scholar]
- 216.Feagan BG, Greenberg GR, Wild G, et al. Treatment of ulcerative colitis with a humanized antibody to the alpha4beta7 integrin. N Engl J Med. 2005;352:2499–2507. doi: 10.1056/NEJMoa042982. [DOI] [PubMed] [Google Scholar]
- 217.Huber S, Schramm C, Lehr HA, et al. Cutting Edge: TGF-β Signaling Is Required for the In Vivo Expansion and Immunosuppressive Capacity of Regulatory CD4+CD25+ T Cells. J Immunol. 2004;173:6526–6531. doi: 10.4049/jimmunol.173.11.6526. [DOI] [PubMed] [Google Scholar]
- 218.Boirivant M, Amendola A, Butera A, et al. A Transient Breach in the Epithelial Barrier Leads to Regulatory T-Cell Generation and Resistance to Experimental Colitis. Gastroenterology. 2008;135:1612–1623. doi: 10.1053/j.gastro.2008.07.028. e1615. [DOI] [PubMed] [Google Scholar]
- 219.Reardon C, Wang A, McKay DM. Transient local depletion of Foxp3+ regulatory T cells during recovery from colitis via Fas/Fas ligand-induced death. J Immunol. 2008;180:8316–8326. doi: 10.4049/jimmunol.180.12.8316. [DOI] [PubMed] [Google Scholar]
- 220.Ishikawa D, Okazawa A, Corridoni D, et al. Tregs are dysfunctional in vivo in a spontaneous murine model of Crohn's disease. Mucosal Immunology. 2012 doi: 10.1038/mi.2012.67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Collins CB, Aherne CM, McNamee EN, et al. Flt3 ligand expands CD103+ dendritic cells and FoxP3+ T regulatory cells, and attenuates Crohn's-like murine ileitis. Gut. 2012;61:1154–1162. doi: 10.1136/gutjnl-2011-300820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Ebert EC, Panja A, Das KM, et al. Patients with inflammatory bowel disease may have a transforming growth factor-β-, interleukin (IL)-2- or IL-10-deficient state induced by intrinsic neutralizing antibodies. Clinical & Experimental Immunology. 2009;155:65–71. doi: 10.1111/j.1365-2249.2008.03802.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Vignali DAA, Kuchroo VK. IL-12 family cytokines: immunological playmakers. Nature Immunology. 2012;13:722–728. doi: 10.1038/ni.2366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Wing K, Yamaguchi T, Sakaguchi S. Cell-autonomous and -non-autonomous roles of CTLA-4 in immune regulation. Trends Immunol. 2011;32:428–433. doi: 10.1016/j.it.2011.06.002. [DOI] [PubMed] [Google Scholar]
- 225.Zheng SG, Wang JH, Stohl W, et al. TGF-beta requires CTLA-4 early after T cell activation to induce FoxP3 and generate adaptive CD4+CD25+ regulatory cells. J Immunol. 2006;176:3321–3329. doi: 10.4049/jimmunol.176.6.3321. [DOI] [PubMed] [Google Scholar]
- 226.Huang C-T, Workman CJ, Flies D, et al. Role of LAG-3 in Regulatory T Cells. Immunity. 2004;21:503–513. doi: 10.1016/j.immuni.2004.08.010. [DOI] [PubMed] [Google Scholar]
- 227.Okamura T, Fujio K, Shibuya M, et al. CD4+CD25-LAG3+ regulatory T cells controlled by the transcription factor Egr-2. Proc Natl Acad Sci USA. 2009;106:13974–13979. doi: 10.1073/pnas.0906872106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Garín MI, Chu C-C, Golshayan D, et al. Galectin-1: a key effector of regulation mediated by CD4+CD25+ T cells. Blood. 2007;109:2058–2065. doi: 10.1182/blood-2006-04-016451. [DOI] [PubMed] [Google Scholar]
- 229.Hokama A, Mizoguchi E, Mizoguchi A. Roles of galectins in inflammatory bowel disease. World J Gastroenterol. 2008;14:5133–5137. doi: 10.3748/wjg.14.5133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Kubach J, Lutter P, Bopp T, et al. Human CD4+CD25+ regulatory T cells: proteome analysis identifies galectin-10 as a novel marker essential for their anergy and suppressive function. Blood. 2007;110:1550–1558. doi: 10.1182/blood-2007-01-069229. [DOI] [PubMed] [Google Scholar]
- 231.Santucci L, Agostini M, Bruscoli S, et al. GITR modulates innate and adaptive mucosal immunity during the development of experimental colitis in mice. Gut. 2007;56:52–60. doi: 10.1136/gut.2006.091181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Croft M, So T, Duan W, et al. The Significance of OX40 and OX40L to T cell Biology and Immune Disease. Immunological Reviews. 2009;229:173–191. doi: 10.1111/j.1600-065X.2009.00766.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Griseri T, Asquith M, Thompson C, et al. OX40 is required for regulatory T cell-mediated control of colitis. J Exp Med. 2010;207:699–709. doi: 10.1084/jem.20091618. [DOI] [PMC free article] [PubMed] [Google Scholar]