Abstract
Objective
To investigate potential functional variants in FTO and SH2B1 genes among Chinese children with obesity.
Methods
Sanger sequencing of PCR products of all FTO and SH2B1 exons and their flanking regions were performed in 338 Chinese Han children with obesity and 221 age- and sex-matched lean controls.
Results
A total of seven and five rare non-synonymous variants were identified in FTO and SH2B1, respectively. The overall frequencies of FTO and SH2B1 rare non-synonymous variants were similar in obese and lean children (2.37% and 0.90% vs. 1.81% and 1.36%, P>0.05). However, four out of the seven variants in FTO were novel and all were unique to obese children (p>0.05). None of the novel variants was consistently being predicted to be deleterious. Four out of five variants in SH2B1 were novel and one was unique to obese children (p>0.05). One variant (L293R) that was consistently being predicted as deleterious in SH2B1 gene was unique to lean control. While rare missense mutations were more frequently detected in girls from obesity as well as lean control than boys, the difference was not statistically significant. In addition, it's shown that the prevalence of rare missense mutations of FTO as well as SH2B1 was similar across different ethnic groups.
Conclusion
The rare missense mutations of FTO and SH2B1 did not confer risks of obesity in Chinese Han children in our cohort.
Introduction
Obesity is a major public health problem leading to increased mortality and morbidity [1], and obesity in childhood is strongly associated with increased incidence of premature death [2], [3].The prevalence of overweight and obesity in children has been increasing in the world [6], as well as in Asia [4], [5]. In Beijing and Shanghai, the prevalence of obesity increased by more than 3-fold from 1985 to 2000, up to 27% in boys and 16% in girls [7].The increasing prevalence of obesity is contributed by the excessive calorie intake and diminished physical activity in the modern environment. However, considerable evidence from familial segregation and twin studies suggest a significant genetic contribution to adiposity [8], [9]. Genetic factors are believed to modulate the impact of the affluent environment on each individual.
Genome-wide association studies (GWAS) have revealed that the single nucleotide polymorphisms (SNPs) in the first intron of fat mass- and obesity-associated gene (FTO) are significantly associated with BMI and increased risk for obesity [10], [11] and it is the strongest adiposity locus identified so far [12], [13].
SNPs in human SH2B adapter protein 1 gene (SH2B1) are also been shown to be associated with leptin resistance and obesity[14]–[17]. In addition, chromosomal deletions that eliminate the SH2B1 gene are also associated with severe obesity and insulin resistance in human [18], [19].Genetic deletion of SH2B1 results in severe leptin resistance, insulin resistance, obesity, and type 2 diabetes in mice [20], [21],and neuron-specific restoration of SH2B1 reverses the obesity and type 2 diabetes phenotypes in SH2B1 null mice [22]. Collectively, these findings strongly suggest that the SH2B1 gene has a conserved role in the control of both body weight and metabolism homeostasis in rodents and humans.
In an effort to identifying potential functional variants in FTO gene, Meyre, et al [23] sequenced the coding regions of the gene in 1433 severe obese and 1433 lean Caucasian individuals. As a result, almost an equal number of non-synonymous variants were detected in obese and lean individuals, with more unique variants to the lean. Enzymatic activity analysis further suggested that loss-of-function variants are compatible with lean and did not impose increased risk for obesity. By deep resequencing using next generation sequencing (NGS) technology in 524 severely obese and 527 lean Swedish children, Almén et al, showed little evidence of functional variants in coding region of the FTO gene. They concluded that the first intron is the only region within the FTO gene associated with obesity [24].Similarly, Bochukova et al. sequenced the SH2B1 gene in 500 Caucasian patients from the Genetics of Obesity Study (GOOS) but did not identify any coding or splice site mutations [18].Since all previous studies were undertaken mainly in Caucasian population, we aim to investigate the prevalence of rare missense mutations of FTO and SH2B1 in Chinese children with early-onset obesity.
Materials and Methods
Ethics Statement
The study was approved by the Ethics Committee of Shanghai Children’s Medical Center, Shanghai Jiao Tong University School of Medicine. Informed written consents were obtained from all children's parents or guardians before blood sampling and DNA analyses.
Subjects
Three hundred and thirty-eight Chinese children with BMI >97% percentile were included in this study. A total of 221 lean children with <50th percentile adjusted for sex and age were selected as control from Shanghai Children’s Sleep Project [25]. All study subjects were Han Chinese, and the anthropological characteristics of obese and lean children were summarized in Table 1.
Table 1. The anthropological characteristics of children with early-onset obesity and lean control.
| Obesity | Control | p value | |
| N (boys/girls) | 338(202/136) | 221(127/94) | >0.05 |
| Age | 9.62±2.75 | 9.97±2.67 | >0.05 |
| BMI Z-score | 3.38±1.45 | −0.41±0.42 | <0.01 |
Data are means±SD unless otherwise indicated.
Sanger Sequencing
We screened the exonic and flanking regions of the FTO and SH2B1 by Sanger sequencing. PCR primers (Table S1 & S2) were designed with Primer3 tool (http: //frodo.wi.mit.edu/primer3/) to contain exonic sequences and their flanking regions of 20 bp. PCR amplifications were inspected for single band of expected sizes on agarose gels before purification with Agencourt AMPure on Biomek NX (Beckman Coulter, USA). Sequencing was achieved using the automated ABI Prism 3730xlDNA Sequencer in combination with the Big Dye Terminator Cycle Sequencing Ready Reaction Kit 3.1 (Applied Biosystems, USA), and purification of sequencing reaction was performed with Agencourt CleanSEQ on Biomek NX(Beckman Coulter, USA). Sequences were assembled and analyzed with MutationSurveyor software (SoftGenetics, USA).
Assessment of Functional Impacts of Non-synonymous Mutations
We assessed the functional impacts of FTO and SH2B1 non-synonymous mutations by three software, i.e., Protein Variation Effect Analyzer (PROVEAN), Sorts Intolerant From Tolerant (SIFT) and Polymorphism Phenotyping v2 (Polyphen-2). The PROVEAN cutoff value for deleterious was set at −2.5 as instructed [26]. A default setup was used for SIFT and Polyphen-2 functional prediction.
Statistics
The prevalence of FTO and SH2B1 rare missense mutations between obese and lean subjects and between our cohort and literature data or public database were compared with Fisher's exact test or Chi-square test. Student's t-test and Mann-Whitney test were used to compare the means of age and BMI Z-score between obese and lean children, respectively. Statistics analysis was performed with SPSS17.0 software. A P value of <0.05 was considered significant throughout this study.
Results
Prevalence of FTO Non-synonymous Mutations
A total of seven rare (frequency <1% in the present cohort) non-synonymous variants FTO were identified in obese and lean children (Table 2). Among them, 4 of the 7 rare missense mutations were novel (not previously reported in 1000 genome, NHLBI GO Exome Sequencing Project (ESP), and the Human Gene Mutation Database (HGMD)). The prevalence of rare non-synonymous mutations in obese children was similar to that in lean control (2.37% vs. 1.81%, P>0.05).
Table 2. Prevalence of rare non-synonymous mutations in FTO in obese and lean children.
| Variants | Obesity(n = 338) | Control(n = 221) |
| L91P | 1 | 0 |
| E129K | 1 | 0 |
| D144N | 3 | 2 |
| Y185C | 1 | 0 |
| H290R | 1 | 0 |
| D348N | 0 | 2 |
| S482L | 1 | 0 |
| Prevalence of mutation carriers | 2.37% | 1.81% |
| P value | >0.05 | |
Rare non-synonymous mutations denote variations with frequency<0.01.
We found that a subset of the rare non-synonymous mutations was unique to each group. Five (L91P, E129K, Y185C, H290R, S482L) variants were identified uniquely in the obese children, whereas only one (D348N) were identified uniquely in the lean control. D144N was present in both obese and lean children. All rare missense mutations were identified in heterozygous pattern. Thus, there was no obvious enrichment of non-synonymous mutations in lean or obese individuals.
Interestingly, we observed a higher frequency of FTO rare non-synonymous mutations in girls of obesity as well as of lean control in our cohort. However, the difference didn't reach a statistical significance (Table 3).
Table 3. Difference on prevalence of FTO rare non-synonymous mutation between boys and girls with obesity.
| Boys | Girls | P value | |
| Obesity | 1.48%(3/202) | 3.68%(5/136) | >0.05 |
| Control | 0.79%(1/127) | 3.19%(3/94) | >0.05 |
Prevalence of SH2B1 Non-synonymous Mutations
A total of 5 rare non-synonymous variants were identified in SH2B1 in this cohort (Table 4). The prevalence of rare missense mutations in obese and lean children were 0.90% and 1.36%, respectively (P>0.05). Two (V209I and M465T) non-synonymous mutations were identified uniquely in obese children, whereas 3 (G131S, L293R, and W649G) were identified uniquely in lean control. One rare mutation, M465T, was identified in two obese children. All mutations were present in heterozygous pattern, and no nonsense variants were reported in the studied populations.
Table 4. Prevalence of SH2B1 rare non-synonymous mutations in obese and lean children.
| Variations | Obesity(n = 334) | Control(n = 221) |
| G131S | 0 | 1 |
| L293R | 0 | 1 |
| W649G | 0 | 1 |
| V209I | 1 | 0 |
| M465T | 2 | 0 |
| Prevalence of mutation carriers | 0.90% | 1.36% |
| P value | >0.05 | |
Prevalence of FTO and SH2B1 Rare Non-synonymous Mutations in Multiple Ethnicities
To explore the possible difference on the prevalence of FTO and SH2B1 rare non-synonymous mutations in different ethnicities, we compared our results with Meyre's [23] data of French and English children with obesity and coding variants data from Exome Variant Server, NHLBI GO Exome Sequencing Project (ESP), Seattle, WA (URL: http: //evs.gs.washington.edu/EVS/). The results showed that the prevalence of rare non-synonymous mutations of FTO in Chinese obese children (2.37%, 8/338) was comparable to the results of Meyer's study [23] (1.32%, 9/680), European American (2.23%, 96/4300), and African American (1.64%, 36/2198) from ESP. Similarly, the prevalence of SH2B1 rare missense mutations didn't differ significantly between Chinese obese children (0.90%, 3/334) and European American (1.79%, 77/4300) as well as African American (2.50%, 55/2197) from ESP, though there's a marginal difference between the latter two populations (P = 0.054) (Table 5).
Table 5. Prevalence of FTO and SH2B1 rare non-synonymous mutations in different populations.
| Ourcohort | Meyre’s[22] | EA_ESP | AA_ESP | ||
| Population | CHC | FC/EC | EA | AA | |
| FTO | N | 338 | 680 | 4300 | 2198 |
| Rare missense | 8 | 9 | 96 | 36 | |
| Prevalence (%) | 2.37 | 1.32 | 2.23 | 1.64 | |
| SH2B1 | N | 334 | − | 4300 | 2197 |
| Rare missense | 3 | − | 77 | 55 | |
| Prevalence (%) | 0.90 | − | 1.79 | 2.50 |
ESP: NHLBI Exome Sequencing Project; CHC: Chinese Han children; FC/EC: French children/English children from Meyre's cohort [22]; EA: European American; AA: African American.
Functional Impacts of FTO and SH2B1 Non-synonymous Mutations
Among FTO rare non-synonymous mutations, L91P and S482L were predicted deleterious, and all SH2B1 non-synonymous mutations but L293R were predicted neutral with the three tool software (Table 6). The functional impacts of non-synonymous mutations of FTO and SH2B1 did not differ between obese and lean children (P>0.05).
Table 6. Functional impacts of FTO and SH2B1 rare non-synonymous mutations.
| Gene | Variants | PROVEAN | SIFT | Polyphen-2 |
| FTO | L91P | Deleterious | Tolerated | Probably damaging |
| E129K | Neutral | Tolerated | Benign | |
| D144N | Neutral | Tolerated | Possibly damaging | |
| Y185C | Neutral | Tolerated | Benign | |
| H290R | Neutral | Tolerated | Benign | |
| D348N | Neutral | Tolerated | Benign | |
| S482L | Neutral | Damaging | Possibly damaging | |
| SH2B1 | G131S | Neutral | Tolerated | Benign |
| L293R | Deleterious | Damaging | Probably damaging | |
| V209I | Neutral | Tolerated | Benign | |
| M465T | Neutral | Tolerated | Benign | |
| W649G | Neutral | Damaging | Benign |
PROVEAN: Protein Variation Effect Analyzer; SIFT: Sorts Intolerant From Tolerant; Polyphen-2: Polymorphism Phenotyping v2.
Discussion
Recent findings in Fto-null mice support the notion that Fto itself has an important influence on energy balance. Fto-null mice are small, are lean, have an increased metabolic rate, and are hyperphagic, whereas Fto −/− mice are resistant to diet-induced obesity [27]. In contrary, over-expression in mice resulted in increased energy intake and increased adiposity in a dose-dependent manner [28], in agreement with the FTO dose-dependent differences in adiposity seen in humans [29]. Taken together, these data suggest that increased FTO expression results in increased food intake, leading to increased adiposity. Thus, a gain-of-function effect is suggested for the pathogenic role of FTO in human obesity.
Given that information, it is reasonable to speculate that function-impairing mutations in FTO might be more common in lean rather than obese subjects.
However, our results and data from obese children of Caucasians by Meyre, et al [23] and of African Ancestry by Deliard et al [30] clearly demonstrate that the rare non-synonymous mutations are not enriched in lean control.
FTO protein is an AlkB-like DNA/RNA demethylase with a strong preference for 3-methylthymidine (3-meT) in single-stranded DNA or 3-methyluracil (3-meU) in single-stranded RNA[30]–[34]. More recently, Jia et al [35] demonstrated that FTO could efficiently demethylate N6-methyladenosine (m6A) at neutral pH in vitro and that the amount of m6A in cellular mRNA is affected by the oxidation activity of FTO in vivo, concluding that m6A acts as the natural substrate for FTO in physiological condition. FTO is consisted of an amino-terminal AlkB-like domain (NTD) and a carboxyl-terminal domain (CTD) with a novel fold. The interaction between these two domains is required for FTO catalytic activity [36].FTO possesses an extra loop (residues 213–224) that it has an important role in FTO selection against double-stranded nucleic acids [36].H231, D233, H307, and residues within the extra loop are highly conserved among FTO proteins from different species. Biochemical assay shows that mutations at F114, C392, R96, and E234 abolish or greatly reduce the catalytic activity of FTO [36]. Boissel, et al [31] found a consanguineous Israeli-Arab family in which nine siblings were homozygous for the R316Q mutation in FTO, which resulted in a diminished catalytic activity. All homozygous carriers were severely growth retarded, had multiple congenital malformations, and died in infancy, and heterozygous parents of these children had no obesity or overweight phenotype. Similarly, R322 is also essential for FTO catalytic activity, and heterozygous mutations that severely impaired enzymatic activity of FTO were found in both lean and obese individuals with no other obvious major clinical phenotypes [23]. Furthermore, the R316Q mutation abolished 80% of the wild-type activity toward m6A demethylation in vitro, while H231A D233A and R316Q R322Q double mutant FTO proteins completely lost m6A-demethylation activity, suggesting that m6A in nuclear RNA is the physiological substrate of FTO and that the function of FTO likely affects the processing of pre-mRNA, other nuclear RNAs, or both [35]. Two non-synonymous mutations (L91P and S482L), that are solely detected in obese children, are within the so-called substrate recognition lid and CTD of FTO protein, respectively (Figure 1.), and predicted to compromise FTO catalytic activity. Meyer et al [23] also tested the enzymatic activity of several other variants representing the different regions of FTO (P5L,V94I, I148R, M223V, E234D, A241T, A405V, I492V, and V493F), and found none of these variants, all of which are located in less conserved positions, had any significant impact on enzymatic activity. The observed activity of FTO is exceedingly low compared to those of the other AlkB-family proteins [37], it's reasonable to speculate that with M6A as a substrate, the functional impacts of the mutant FTO proteins including L91P and S482L identified in this study could be more accurately assessed.
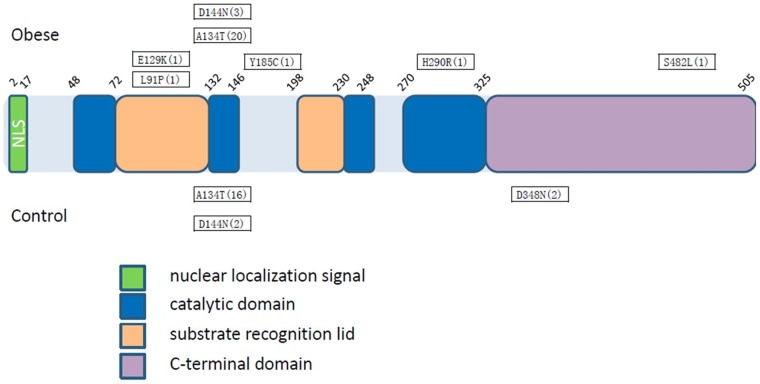
Figure 1. Distribution of rare non-synonymous variants along FTO functional domains.
FTO protein is consisted of a N-terminal domain (NTD, residue 32–326) and a C-terminal domain (CTD, residue 327–505). Different non-synonymous mutations with frequency in parenthesis were presented both for obese children and control population.
As heterozygous deletion of SH2B1 in mice leads to obesity on a high-fat diet [22], Bochukova et al [18] suggested that SH2B1 gene haploinsufficiency may be a plausible mechanism underlying the phenotype seen in humans. However, Bochukova et al. sequenced the SH2B1 gene in 500 patients from the Genetics of Obesity Study (GOOS) but did not identify any coding or splice site mutations [18]. Alike to FTO, a similar prevalence of rare missense mutations of SH2B1 is revealed in Chinese obese children and lean control. Among non-synonymous mutations of SH2B1 detected in this cohort, only one (L293R) is predicted deleterious to protein function by tool software. L293 locates in the pleckstrin homology (PH) domain (residues 249–378) of SH2B1 protein, which is thought to mediate binding to inactive JAK2, whereas the SH2 domain (residues 521–625) near the C terminus is critical to binding to active, phosphorylated JAK2 [38]. Though SH2B1 point mutations may significantly impair the growth hormone (GH)/nerve growth factor (NGF)-mediated signaling in vitro, all mutants except F344LfsX20 did not impair leptin signaling [39]. It's plausible to speculate that loss-of-function mutation of SH2B1 may not significantly involved in pathogenesis of Chinese children obesity.
In conclusion, rare non-synonymous mutations of FTO and SH2B1 are equally detected in obese and lean Chinese children. The rare missense mutations of FTO and SH2B1 do not confer risks of obesity in Chinese Han children in our cohort. Novel and non-deleterious missense variants detected in FTO unique to obese individuals are of significant interest to reveal their potential functional impact on FTO protein.
Supporting Information
PCR primers for amplification of FTO coding regions.
(DOCX)
PCR primers for amplification of SH2B1 coding regions.
(DOCX)
Acknowledgments
We thank the support from the patient and the family.
Funding Statement
This work was supported by Project HOPE "The Abbott Fund Institute of Nutrition Science (AFINS-HOPE-2013-05,to YGY)”, the other is Shanghai Jiaotong University Cross Biomedical Engineering (YG-2011-MS28, to YGY). This study is further supported by grant from the National Natural Science Foundation of China (No.81000346, to YGY), and grant from Shanghai Science & Technology Commission Major Project (No. 11dz1950300, to YGY). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Guh DP, Zhang W, Bansback N, Amarsi Z, Birmingham CL, et al. (2009) The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health 9: 88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Bjorge T, Engeland A, Tverdal A, Smith GD (2008) Body mass index in adolescence in relation to cause-specific mortality: a follow-up of 230,000 Norwegian adolescents. Am J Epidemiol 168: 30–37. [DOI] [PubMed] [Google Scholar]
- 3. Franks PW, Hanson RL, Knowler WC, Sievers ML, Bennett PH, et al. (2010) Childhood obesity, other cardiovascular risk factors, and premature death. N Engl J Med 362: 485–493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Yoon KH, Lee JH, Kim JW, Cho JH, Choi YH, et al. (2006) Epidemic obesity and type 2 diabetes in Asia Lancet. 368: 1681–1688. [DOI] [PubMed] [Google Scholar]
- 5. Chen CM (2008) Overview of obesity in Mainland China. Obes Rev Suppl 1: 9 14–21. [DOI] [PubMed] [Google Scholar]
- 6. de Onis M, Blossner M, Borghi E (2010) Global prevalence and trends of overweight andobesity among preschool children. Am J Clin Nutr 92: 1257–1264. [DOI] [PubMed] [Google Scholar]
- 7. Ji CY (2008) The prevalence of childhood overweight/obesity and the epidemic changes in 1985–2000 for Chinese school-age children and adolescents. Obes Rev 9 (Suppl 1)78–81. [DOI] [PubMed] [Google Scholar]
- 8. Loos RJ (2003) BouchardC (2003) Obesity–is it a genetic disorder?. J Intern Med 254: 401–425. [DOI] [PubMed] [Google Scholar]
- 9. Wardle J, Carnell S, Haworth CM, Plomin R (2008) Evidence for a strong genetic influence on childhood adiposity despite the force of the obesogenic environment. Am JClin Nutr 87: 398–404. [DOI] [PubMed] [Google Scholar]
- 10. Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, et al. (2007) A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 316: 889–894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Dina C, Meyre D, Gallina S, Durand E, Körner A, et al. (2007) Variation in FTO contributes to childhood obesity and severe adult obesity. Nat Genet 39: 724–726. [DOI] [PubMed] [Google Scholar]
- 12. Hinney A, Nguyen TT, Scherag A, Friedel S, Brönner G, et al. (2007) Genome wide association (GWA) study for early onset extreme obesity supports the role of fat mass and obesity associated gene (FTO) variants. PLoS ONE 2: e1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Scuteri A, Sanna S, Chen WM, Uda M, Albai G, et al. (2007) Genome-wide association scan shows genetic variants in the FTO gene are associated with obesity-related traits. PLoS Genet 3: e115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Willer CJ, Speliotes EK, Loos RJ, Li S, Lindgren CM, et al. (2009) Six new loci associated with body mass index highlight a neuronal influence on body weight regulation. Nat Genet 41: 25–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Thorleifsson G, Walters GB, Gudbjartsson DF, Steinthorsdottir V, Sulem P, et al. (2009) Genome-wide association yields new sequence variants at seven loci that associate with measures of obesity. Nat Genet 41: 18–24. [DOI] [PubMed] [Google Scholar]
- 16. Renström F, Payne F, Nordström A, Brito EC, Rolandsson O, et al. (2009) Replication and extension of genome-wide association study results for obesity in 4923 adults from northern Sweden. Hum Mol Genet 18: 1489–1496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bauer F, Elbers CC, Adan RA, Loos RJ, Onland-Moret NC, et al. (2009) Obesity genes identified in genome-wide association studies are associated with adiposity measures and potentially with nutrient-specific food preference. Am J Clin Nutr 90: 951–959. [DOI] [PubMed] [Google Scholar]
- 18. Bochukova EG, Huang N, Keogh J, Henning E, Purmann C, et al. (2010) Large, rare chromosomal deletions associated with severe early-onset obesity. Nature 463: 666–670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Walters RG, Jacquemont S, Valsesia A, de Smith AJ, Martinet D, et al. (2010) A new highly penetrant form of obesity due to deletions on chromosome16p11.2. Nature 463: 671–675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Duan C, Yang H, White MF, Rui L (2004) Disruption of the SH2-B gene causes age-dependent insulin resistance and glucose intolerance. Mol Cell Biol 24: 7435–7443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ren D, Li M, Duan C, Rui L (2005) Identification of SH2-B as a key regulator of leptin sensitivity, energy balance, and body weight in mice. Cell Metab 2: 95–104. [DOI] [PubMed] [Google Scholar]
- 22. Ren D, Zhou Y, Morris D, Li M, Li Z, et al. (2007) Neuronal SH2B1 is essential for controlling energy and glucose homeostasis. J Clin Invest 117: 397–406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Meyre D, Proulx K, Kawagoe-Takaki H, Vatin V, Gutiérrez-Aguilar R, et al. (2010) Prevalence of loss-of-function FTO mutations in lean and obese individuals. Diabetes 59: 311–318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sällman Almén M, Rask-Andersen M, Jacobsson JA, Ameur A, Kalnina I, et al.. (2012) Determination of the obesity-associated gene variants within the entire FTO gene by ultra-deep targeted sequencing in obese and lean children. Int J Obes. doi: 10.1038/ijo.2012.57. [Epub ahead of print] [DOI] [PMC free article] [PubMed]
- 25. J Wang, H Mei, W Chen, Y Jiang, W Sun, et al. (2011) Study of eight GWAS-identified common variants for association with obesity-related indices in Chinese children at puberty. Intl J Obesity 2011: 1–6. [DOI] [PubMed] [Google Scholar]
- 26. Choi Y, Sims GE, Murphy S, Miller JR, Chan AP (2012) Predicting the Functional Effect of Amino Acid Substitutions and Indels. PLoS ONE 7(10): e46688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Fischer J, Koch L, Emmerling C, Vierkotten J, Peters T, et al. (2009) Inactivation of the Fto gene protects from obesity. Nature 458: 894–898. [DOI] [PubMed] [Google Scholar]
- 28. Church C, Moir L, McMurray F, Girard C, Banks GT, et al. (2010) Over-expression of Fto leads to increased food intake and results in obesity. Nat Genet 42: 1086–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Frayling TM, Timpson NJ, Weedon MN, Zeggini E, Freathy RM, et al. (2007) A common variant in the FTO gene is associated with body mass index and predisposes to childhood and adult obesity. Science 316: 889–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Deliard S, Panossian S, Mentch FD, Kim CE, Hou C, et al. (2013) The Missense Variation Landscape of FTO, MC4R, and TMEM18 in Obese Children of African Ancestry. Obesity (Silver Spring) 21: 159–163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Boissel S, Reish O, Proulx K, Kawagoe-Takaki H, Sedgwick B, et al. (2009) Loss-of-function mutation in the dioxygenase-encoding FTO gene causes severe growth retardation and multiple malformations. Am J Hum Genet 85: 106–111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Gerken T, Girard CA, Tung YC, Webby CJ, Saudek V, et al. (2007) The obesity-associated FTO gene encodes a 2-oxoglutaratedependentnucleic acid demethylase. Science 318: 1469–1472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Sanchez-Pulido L, Andrade-Navarro MA (2007) The FTO (fat mass and obesity associated) gene codes for a novel member of the non-heme dioxygenase superfamily. BMC Biochem 8: 23–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Jia G, Yang CG, Yang S, Jian X, Yi C, et al. (2008) Oxidative demethylation of 3-methylthymine and 3-methyluracil in single-stranded DNA and RNA by mouse and human FTO. FEBS Lett 582: 3313–3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Jia G, Fu Y, Zhao X, Dai Q, Zheng G, et al. (2011) N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat Chem Biol 7: 885–887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Han Z, Niu T, Chang J, Lei X, Zhao M, et al. (2010) Crystal structure of the FTO protein reveals basis for its substrate specificity. Nature 464: 1205–9. [DOI] [PubMed] [Google Scholar]
- 37. Lee DH, Jin SG, Cai S, Chen Y, Pfeifer GP, et al. (2005) Repair of methylation damage in DNA and RNA by mammalian AlkB homologues. J Biol Chem 280: 39448–39459. [DOI] [PubMed] [Google Scholar]
- 38. Li Z, Zhou Y, Carter-Su C, Myers Jr MG, Rui L (2007) SH2B1 enhances leptin signaling by both Janus kinase 2 Tyr813 phosphorylation-dependent and -independent mechanisms. Mol Endocrinol 21: 2270–2281. [DOI] [PubMed] [Google Scholar]
- 39. Doche ME, Bochukova EG, Su HW, Pearce LR, Keogh JM, et al. (2012) Human SH2B1 mutations are associated with maladaptive behaviors and obesity. J Clin Invest 122: 4732–4736. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
PCR primers for amplification of FTO coding regions.
(DOCX)
PCR primers for amplification of SH2B1 coding regions.
(DOCX)