Abstract
Objective: To investigate the effect of hyperbaric oxygen therapy (HBOT) on the iNOS mRNA-iNOS-NO signaling pathway and neurofunction protected in a rat spinal cord injury model. Methods: A total of 36 Sprague-Dawley rats were randomly divided into 3 groups: control group (n=12), SCI group (n=12) and SCI + HBOT group (n=12). SCI + HBOT group In the SCI group and SCI + HBOT groups, SCI was performed on rats. In the SCI + HBOT group, rats with SCI underwent HBO treatment 30 min after SCI for 24 sessions. After HBO therapy, measurement of motor evoked potential (MEP), Basso, Beattie, Bresnahan (BBB) scoring and pathological examination were done. RT-PCR and immunohistochemistry were employed to detect the mRNA and protein expression of iNOS, respectively. Diazo colorimetry was performed to detect the serum NO content. Results: The mRNA and protein expression of iNOS in the spinal cord and the serum NO content were markedly increased in the SCI group as compared to the control group (P<0.05). However, the mRNA and protein expression of iNOS and the serum NO content were dramatically reduced in the SCI + HBOT group as compared to the SCI group (P<0.05). Conclusion: HBO therapy can promote the neuroprotection following SCI, which may be related to the effect of HBO on the iNOS mRNA-iNOS-NO signaling pathway.
Keywords: Hyperbaric oxygen, spinal cord injury, rat, nitric oxide, inducible nitric oxide synthase
Introduction
Spinal cord injury (SCI) is a common trauma, including not only the primary injury but the secondary injury due to the production of numerous factors following trauma. The mechanisms of injury and protected are still poorly understood. Among numerous factors, nitric oxide (NO) plays an important role in the development of secondary injury [1]. Following SCI, the excessive production of NO is closely related to the increased expression of inducible nitric oxide synthase (iNOS) [2]. The trauma or ischemia induced transcription of iNOS (iNOS mRNA) and subsequent protein expression of iNOS attribute to the excessive generation of NO which may result in inflammation and edema of spinal cord and aggravate the injury to neurons surrounding the injury region [3]. The iNOS mRNA-iNOS-NO signaling pathway is closely associated with SCI and to regulate this signaling pathway may attenuate the SCI or even control the development of SCI.
Hyperbaric oxygen (HBO) is often used in the treatment of SCI. Studies have confirmed that HBOT can attenuate the ischemia and hypoxia of affected tissues and subsequently induce the local inflammation [4], which is helpful for the protection of neurofunction. Currently, a lot of studies focus on the therapeutic efficacy of HBO in SCI, and the mechanism underlying the effect of HBO on neuroprotection following SCI is less addressed. In the present study, SCI was performed on rats which then underwent HBOT. The neurofunction, pathology of spinal cord, mRNA and protein expression of iNOS and NO content in the spinal cord were investigated aiming to elucidate the role of iNOS mRNA-iNOS-NO signaling pathway in the promotion of neuroprotection following HBOT in SCI rats.
Materials and methods
Materials
A total of 36 healthy adult Sprague-Dawley rats weighing 180 ± 10 g were purchased from the Animal Center of Guangdong (male:female=1:1). Oxygen chamber for animals (Ningbo Hyperbaric oxygen Chamber Corporation), Allen’s weight (intensity: 10×10 g/cm2), wi72289 evoked potential detector (Beijing Dongxiyi Science and Technology Corporation), GeneAmp 9600 thermal cycler (Perkin Elmer, USA) and light microscope (Olympus, Japan) were used in the present study.
Grouping
Random number table was employed to divide 36 rats into control group (n=12), SCI group (n=12) and SCI + HBOT group (n=12).
Establishment of the SCI model
The SCI was performed on the T8~T10 of rats in the SCI group and SCI + HBOT group according to the Allen’s method [5] and the attack intensity was 10×10 g/cm2.
General treatment
In the SCI group, rats with SCI did not undergo HBO therapy. In the SCI + HBOT group, rats with SCI were treated with HBO at 30 min after SCI once daily for consecutive 24 days.
HBOT
The chamber was flushed with pure oxygen according to previously described [6] and then the pressure was increased at a rate of 0.2 MPa (2 ATA) per 20 min. The oxygen concentration in the chamber was maintained at 80~85%, and treatment last for 80 min. Decompression was performed at a uniform rate within 20 min. HBO therapy was done once daily for consecutive 24 days.
Observations
After establishment of animal model, the first time motor evoked potential (MEP) was measured in each rat. After HBO therapy for 24 days, the Basso, Beattie, Bresnahan (BBB) locomotor rating and the second time detection of MEP were done. RT-PCR, diazo colorimetry and nitrate reductase method were employed to measure the iNOS mRNA expression, iNOS activity and serum NO content, respectively. The spinal cord was also collected for pathological examination.
Statistical analysis
Statistical analysis was done with SPSS version 11.0 for windows. Quantitative data were expressed as mean ± standard deviation (X̅ ±S). Normality test and homogeneity of variance test were done first. Once data met the normal distribution and had homogeneity of variance, analysis of variance was employed for analysis. Once data had heterogeneity of variance, rank sum test was performed. Further Q test and Dunnett test were performed on statistical significance was presented. α was set at 0.05 or 0.01.
Results
BBB score in different groups (X̅ ± S)
The BBB score in different groups is shown in Figure 1. As shown in Figure 1, there was significant difference in the BBB score between control group (21.00 ± 0.00 points) and SCI group (4.372 ± 0.523 points) (P<0.05), control group and SCI + HBOT group (6.638 ± 0.692 points) (P<0.05). In addition, the BBB score in the SCI + HBOT group (6.638 ± 0.292 points) was markedly higher than that in the SCI group (4.372 ± 0.223 points) (P<0.05).
Figure 1.

BBB score in different groups.
MEP in different groups (X̅ ± S)
The potential and amplitude in MEP in different groups are shown in Table 1. As hsown in Table 1, the potential and amplitude in the control group (38.65 ± 0.47 ms; 0.43 ± 0.02 mV) were markedly higher than those in the SCI group (14.33 ± 1.26 ms; 0.23 ± 0.03 mV) and SCI + HBOT group (19.71 ± 1.33 ms; 0.31 ± 0.02 mV). Moreover, the potential and amplitude in the SCI + HBOT group (19.71 ± 1.33 ms; 0.31 ± 0.02 mV) were significantly increased as compared to the SCI group (14.33 ± 1.26 ms; 0.23 ± 0.03 mV).
Table 1.
Potential and amplitude in MEP in different groups ( X̅ ± S)
| Group | The first time MEP | The second time MEP | ||
|---|---|---|---|---|
|
| ||||
| Potential (ms) | amplitude (mV) | Potential (ms) | amplitude (mV) | |
| Normal control | 38.64 ± 0.54 | 0.43 ± 0.04 | 38.65 ± 0.47 | 0.43 ± 0.02 |
| SCI | 13.33 ± 1.21 | 0.22 ± 0.03 | 14.33 ± 1.26 | 0.23 ± 0.03 |
| SCI + HBO | 13.42 ± 1.18 | 0.21 ± 0.02 | 19.71 ± 1.33 | 0.31 ± 0.02 |
Pathology of spinal cord in different groups
In the control group (Figure 2A), the gray matter and white matter had complete structure, the neurons and glial cells were evenly distributed, microvessels had normal morphology, the nerve fibers were distributed regularly, Nissl bodies were present and the intercellular substance was even.
Figure 2.
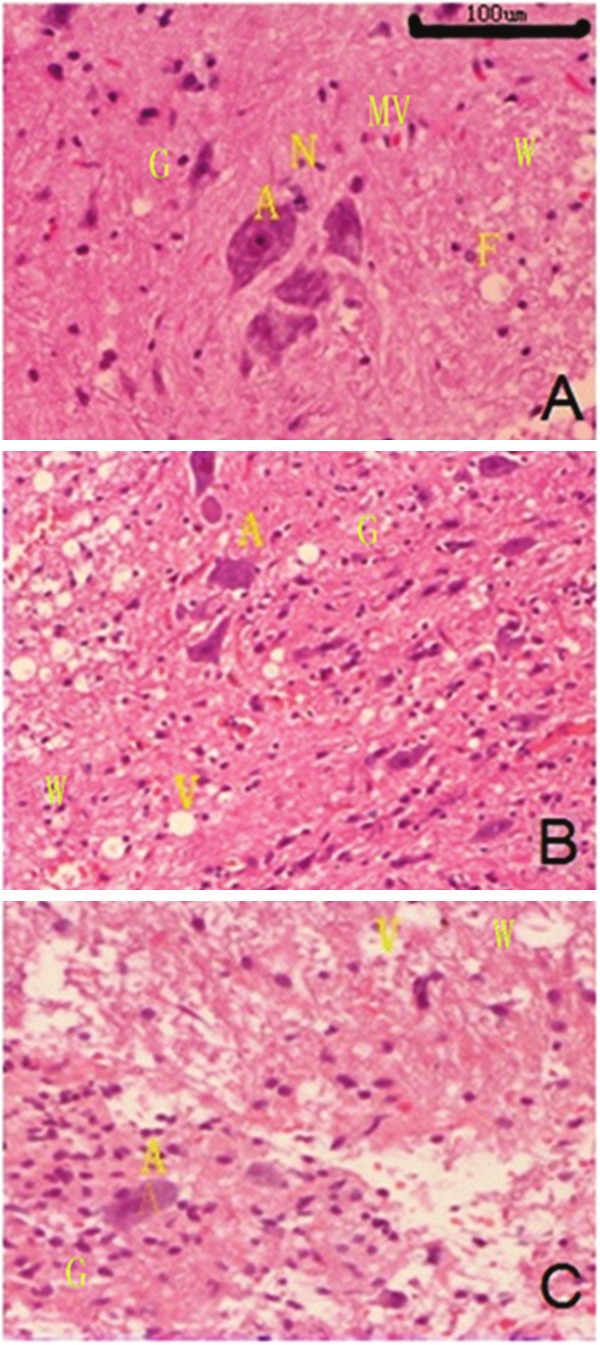
Pathological examination of spinal cord in different groups (HE staining; ×100). A: Astrocytes; V: Vacuolation; N: Nissl bodies; MG: Microglias; MV: Microvessels; G: Gray Matter; W: White Matter.
In the SCI group (Figure 2B), the tissue structure was disordered, the white matter and gray matter had no clear borderline, the neurons and microvessels reduced, the nerve fibers were interrupted by scar tissues and glial scar and vacuolation were also noted.
In the SCI + HBOT group (Figure 2C), the white matter and gray matter in the corresponding spinal cord had relatively clear tissue structure as compared to the SCI group, vacuolation was present, Nissl bodies reduced and the intercellular substance was uneven.
iNOS mRNA expression in spinal cord of different groups (X̅ ± S)
As shown in Figure 3, the mRNA expression of iNOS in the control group (0.009 ± 0.001) was markedly lower than in the SCI group (1.921 ± 0.020 ) and SCI + HBOT group (1.311 ± 0.092 ) (P<0.01). When compared with the SCI group (1.921 ± 0.020), the iNOS mRNA expression was significantly reduced in the SCI + HBOT group (1.311 ± 0.092) (P<0.05).
Figure 3.

iNOS mRNA expression in spinal cord of different groups.
Protein expression of iNOS in spinal cord of different groups (X̅ ± S)
As shown in Figure 4, the iNOS protein expression in the control group (0.003 ± 0.001 U/g) was markedly lower than the SCI group (1.925 ± 0.382 U/g) and SCI + HBOT group (1.183 ± 0.323 U/g) (P<0.01). Furthermore, significant difference in the iNOS protein expression was found between the SCI + HBOT group (1.183 ± 0.323 U/g) and SCI group (1.925 ± 0.382 U/g) (P<0.01).
Figure 4.

Protein expression of iNOS in spinal cord of different groups.
Serum NO content of different groups
As shown in Figure 5, there was significant difference in the serum NO content between control group (22.710 ± 2.631 mmol/L) and SCI + HBOT group (35.472 ± 3.316 mmol/L) (P<0.05). In addition, the serum NO content in the SCI group (43.530 ± 3.078 mmol/L) was significantly higher than that in the control group (22.710 ± 2.631 mmol/L) and SCI + HBOT group (35.472 ± 3.316 mmol/L) (P<0.01).
Figure 5.
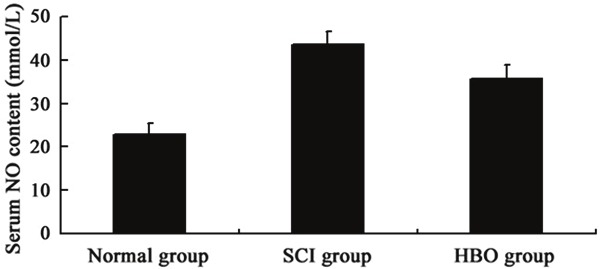
Serum NO content in different groups.
Discussion
NO is an important signaling molecule in mammalians and plays crucial roles in the physiological and pathological processes during the metabolism. NO is active in nature and has short half-life. Thus, the majority of studies related to NO focuses on the NOS. NO is biosynthesized endogenously from L-arginine, oxygen and NADPH by various nitric oxide synthase (NOS) enzymes. Thus, the bioeffects of NO vary among different amounts and sites of NO production. To date, three NOSs (cNOS, iNOS and eNOS) have been identified in humans. iNOS is a key NOS in the production of NO during the inflammation and post-injury recovery. The iNOS expression experiences transcription and translation. Under normal conditions, the iNOS mRNA is not highly replicated and transcribed. Following injury, the iNOS mRNA expression increases gradually and reaches a peak level 24 later [7]. The transcription of iNOS mRNA leads to synthesis of a large amount of iNOS protein and then the production of NO eliciting a series of pathological effects [8]. Excessive production of NO is an important cause of injury aggravation [9] and can induce the Wallerian degeneration of axons and even the apoptosis of cells distant to the injured site [9]. In addition, there is evidence showing that NO of appropriate amount may exert protective effect on the ischemia and hypoxia of nervous tissues via maintaining the vascular activity and permeability [10]. However, in the acute injury including SCI, excessive production and accumulation of NO can mediate the neurotoxicity of excitatory amino acids in which NO may react with superoxide anion (O-2) forming highly toxic peroxynitrite (OONO-) and hydroxyl radical (OH-). The later two reactive oxygen species may induce massive lipid peroxidation and tyrosine nitration of proteins, which further cause damage to neurons at the injured site [11]. Thus, to control or reduce the production of NO, prevent the neuronal injury and preserve the residual structure and function of the spinal cord have become the focuses in future studies.
HBO is often applied in the early treatment of traumas achieving special efficacy. In recent years, studies have demonstrated that HBO can improve the neurofunction and daily activity following SCI and the therapeutic effectiveness is evident [12]. HBO can increase the oxygen content in the blood, elevate the oxygen diffusion distance, dilate arterioles, improve the local microcirculation, and attenuate the edema at the injured site and the ischemia/hypoxia of surrounding spinal cord, which attribute to the protection of neurons at the injured site and reverse of secondary injury to surrounding neurons [13,14]. Recent studies reveal that HBOT can prolong the time window of the treatment of SCI in the acute phase [15,16], which may be attributed to the regulation of tumor necrosis factor receptor 1 (TNF-R1), hypoxia-inducible factor (HIF), neuronal nitric oxide synthase (NOS1), superoxide dismutase 3 (SOD3) and vascular endothelial growth factor (VEGF) by HBO [17].
In the present study, the BBB score, MEP, pathology of spinal cord, mRNA and protein expression of iNOS and serum NO content were determined in rats in the control group, SCI group and SCI + HBOT group. Results showed the BBB score and MEP in the SCI + HBOT group were markedly higher than those in the SCI group, and the vacuolization, scar formation and structural injury in the SCI + HBOT group were also dramatically improved as compared to the SCI group. These findings demonstrated that early treatment with HBO could promote the recovery of motor function and transduction function of the spinal cord, reduce the secondary injury to the spinal cord following SCI. These finally attribute to the improvement of neurofunction in SCI rats following HBOT.
In addition, the mRNA and protein expression of iNOS in the spinal cord and the serum NO content in the SCI group were markedly higher than those in the control group, which indicated that the secondary injury following SCI was related to the iNOS mRNA-iNOS-NO signaling pathway. The potential pathological changes may be explained as follows: the ischemia/hypoxia of spinal cord may activate iNOS mRNA-iNOS-NO signaling pathway, which increases the NO production at the injured site and subsequently leads to the oxygenation of neurons in the spinal cord. These finally result in death or apoptosis of neurons in the spinal cord. At the same time, excessive production of NO may increase the local vascular permeability and then aggravate the edema of local spinal cord, which deteriorate the ischemia/hypoxia of the spinal cord and further activate the iNOS mRNA-iNOS-NO signaling pathway. This forms a vicious cycle and may lead to the irreversibility of damage to the neurofunction and the spread of damage to the surrounding tissues.
Following HBOT, the mRNA and protein expression of iNOS were markedly reduced and the serum NO content significantly increased, which may explain the mechanism underlying the promotive effect of HBO on the neuroprotection following SCI as follows: early HBOT following SCI can down-regulate the mRNA and protein expression of iNOS in the spinal cord, which then reduces the NO production at the injured site. This avoids the death and apoptosis of neurons in the spinal cord and further development of secondary injury. At the same time, HBO can significantly elevate the oxygen supply, which attenuates the damage to the neurons in the spinal cord to a certain extent exerting protective effect on the neuroprotection following SCI.
Taken together, in the present study, the mRNA and protein expression of iNOS in the spinal cord and the serum NO content were determined in the SCI rats following HBOT, and the pathology of spinal cord, MEP and score of motor function were also detected. Our results revealed the SCI was related to the excessive production of NO, and early HBOT could effectively protect the neurons in the spinal cord and promote the recovery of neurofunction, which may be attributed to the regulation of iNOS mRNA-iNOS-NO signaling pathway by HBO and the subsequent reduction of NO production in the spinal cord.
Acknowledgement
This study was supported by the grant from Science and Technology Program of Guangdong Province (2010B031600264).
References
- 1.Hall ED, Springer JE. Neuroprotection and acute spinal cord injury: a reappraisal. NeuroRx. 2004;1:80–100. doi: 10.1602/neurorx.1.1.80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Casha S, Yu WR, Fehlings MG. FAS deficiency reduces apoptosis, spares axons and improves function after spinal cord injury. Exp Neurol. 2005;196:390–400. doi: 10.1016/j.expneurol.2005.08.020. [DOI] [PubMed] [Google Scholar]
- 3.Dayan K, Keser A, Konyalioglu S, Erturk M, Aydin F, Sengul G, Dagci T. The effect of hyperbaric oxygen on neuroregeneration following acute thoracic spinal cord injury. Life Sci. 2012;90:360–364. doi: 10.1016/j.lfs.2011.12.005. [DOI] [PubMed] [Google Scholar]
- 4.Atochin DN, Demchenko IT, Astern J, Boso AE, Piantadosi CA, Huang PL. Contributions of endothelial and neuronal nitric oxide synthases to cerebrovascular responses to hyperoxia. J Cereb Blood Flow Metab. 2003;23:1219–1226. doi: 10.1097/01.WCB.0000089601.87125.E4. [DOI] [PubMed] [Google Scholar]
- 5.Allen AR. Surgery of experimental lesion of spinal cord equivalent to crush injury of fracture dislocation of spinal column: A preliminary report. JAMA. 1911;57:878–880. [Google Scholar]
- 6.Li F, Fang L, Huang S, Yang Z, Nandi J, Thomas S, Chen C, Camporesi E. Hyperbaric oxygenation therapy alleviates chronic constrictive injury-induced neuropathic pain and reduces tumor necrosis factor-alpha production. Anesth Analg. 2011;113:626–633. doi: 10.1213/ANE.0b013e31821f9544. [DOI] [PubMed] [Google Scholar]
- 7.Metzen E, Zhou J, Jelkmann W, Fandrey J, Brune B. Nitric oxide impairs normoxic degradation of HIF-1alpha by inhibition of prolyl hydroxylases. Mol Biol Cell. 2003;14:3470–3481. doi: 10.1091/mbc.E02-12-0791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kimura S, Hosaka N, Yuge I, Yamazaki A, Suda K, Taneichi H, Denda H, Endo N. Cerebrospinal fluid concentrations of nitric oxide metabolites in spinal cord injury. Spine (Phila Pa 1976) 2009;34:E645–652. doi: 10.1097/BRS.0b013e3181abda1d. [DOI] [PubMed] [Google Scholar]
- 9.Vaziri ND, Lee YS, Lin CY, Lin VW, Sindhu RK. NAD(P)H oxidase, superoxide dismutase, catalase, glutathione peroxidase and nitric oxide synthase expression in subacute spinal cord injury. Brain Res. 2004;995:76–83. doi: 10.1016/j.brainres.2003.09.056. [DOI] [PubMed] [Google Scholar]
- 10.Osuka K, Watanabe Y, Usuda N, Atsuzawa K, Aoshima C, Yamauchi K, Takayasu M, Yoshida J. Phosphorylation of neuronal nitric oxide synthase at Ser847 in the nucleus intermediolateralis after spinal cord injury in mice. Neuroscience. 2007;145:241–247. doi: 10.1016/j.neuroscience.2006.10.034. [DOI] [PubMed] [Google Scholar]
- 11.Sezen SF, Lagoda G, Burnett AL. Neuronal nitric oxide signaling regulates erection recovery after cavernous nerve injury. J Urol. 2012;187:757–763. doi: 10.1016/j.juro.2011.09.146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang H, Chen HQ, Gu J, Yu RH. Comparative study of hyperbaric oxygen therapy and conventional drug treatment on spinal cord injury at different therapeutic windows. Sci Res essays. 2011;6:1117–1122. [Google Scholar]
- 13.Garcia E, Silva-Garcia R, Mestre H, Flores N, Martinon S, Calderon-Aranda ES, Ibarra A. Immunization with A91 peptide or copolymer-1 reduces the production of nitric oxide and inducible nitric oxide synthase gene expression after spinal cord injury. J Neurosci Res. 2012;90:656–663. doi: 10.1002/jnr.22771. [DOI] [PubMed] [Google Scholar]
- 14.Gonzalez SL, Labombarda F, Deniselle MC, Mougel A, Guennoun R, Schumacher M, De Nicola AF. Progesterone neuroprotection in spinal cord trauma involves up-regulation of brain-derived neurotrophic factor in motoneurons. J Steroid Biochem Mol Biol. 2005;94:143–149. doi: 10.1016/j.jsbmb.2005.01.016. [DOI] [PubMed] [Google Scholar]
- 15.Huang L, Mehta MP, Nanda A, Zhang JH. The role of multiple hyperbaric oxygenation in expanding therapeutic windows after acute spinal cord injury in rats. J Neurosurg. 2003;99:198–205. doi: 10.3171/spi.2003.99.2.0198. [DOI] [PubMed] [Google Scholar]
- 16.Yu Y, Matsuyama Y, Yanase M, Ito S, Adachi K, Satake K, Ishiguro N, Kiuchi K. Effects of hyperbaric oxygen on GDNF expression and apoptosis in spinal cord injury. Neuroreport. 2004;15:2369–2373. doi: 10.1097/00001756-200410250-00014. [DOI] [PubMed] [Google Scholar]
- 17.Han C, Lin L, Zhang W, Zhang L, Lv S, Sun Q, Tao H, Zhang JH, Sun X. Hyperbaric oxygen preconditioning alleviates myocardial ischemic injury in rats. Exp Biol Med (Maywood) 2008;233:1448–1453. doi: 10.3181/0801-RM-8. [DOI] [PubMed] [Google Scholar]


