Abstract
Anti-tumour necrosis factor (TNF) monoclonal antibody (mAb) (infliximab, IFX) has been shown to be highly effective in the management of Crohn's disease (CD). Herein we investigated the potential role of IFX in inducing clinical remission and regulating interleukin (IL)-21 expression and T helper type 17 (Th17) cell infiltration in the intestinal mucosa of CD patients. Twenty-six CD patients were treated with IFX at weeks 0, 2 and 6. Clinical response, mucosal healing, serum C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) were evaluated at week 10 after IFX administration. Expression of IL-21, IL-17A and retinoic acid-related orphan receptor C (RORC) in intestinal mucosa were analysed by quantitative real-time polymerase chain reaction (PCR) and immunohistochemistry. Peripheral blood and lamina propria CD4+ T cells were stimulated with anti-CD3 and anti-CD28 mAbs in the presence of IFX. Cytokine profiles and RORC were determined with enzyme-linked immunosorbent assay (ELISA) and real-time PCR. IL-21 and Th17 cells were found to be expressed highly in inflamed mucosa of active CD patients compared with healthy controls. Ten weeks after IFX infusion, CD activity index, ESR, CRP and intestinal mucosal healing were improved markedly in CD patients, and IL-21 expression and Th17 cell infiltration were decreased significantly compared with those before IFX therapy. In-vitro study demonstrated that IFX treatment could suppress IL-21, IL-17A and RORC expression in cultured CD biopsies. Moreover, IFX was also observed to down-regulate markedly IL-17A, IL-21 and RORC expression by CD CD4+ T cells. IFX is highly effective in inducing clinical remission and promoting intestinal mucosal healing in CD patients through down-regulation of IL-21 expression and Th17 cell infiltration in intestinal mucosa.
Keywords: Crohn's disease, infliximab, interleukin-21, mucosal healing, Th17 cells
Introduction
Crohn's disease (CD) is a chronic intestinal disorder, which is characterized by a patchy, segmental and typically transmural inflammation in any part of the gastrointestinal tract 1. Although the specific aetiology of CD is still not completely understood, it is well established that chronic inflammation is primarily the consequence of a dysregulation of innate and adaptive immune responses to commensal bacteria in intestinal mucosa leading to an immunological imbalance, characterized by an excessive production of proinflammatory cytokines such as tumour necrosis factor (TNF), interleukin (IL)-21 and IL-23 2–4. Accumulating evidence has shown that CD bears all the stigmata of an exaggerated T helper type 1 (Th1) immune response, characterized by increased interferon (IFN)-γ and IL-12 2–4. However, in past years published data have highlighted the critical role of the Th17 subset in the pathogenesis of CD, as well as the signature cytokine IL-17A, in the development of intestinal mucosal damage 5. Th17 cells are a recently discovered addition to the Th cell ensemble, characterized by the expression of a cytokine family (IL-17A, IL-17F, IL-21, IL-22 and IL-26) and transcription factor retinoic acid-related orphan receptor C (RORC) 6. This cell population subset is plastic, and can be converted into Th1-type cells if they receive appropriate stimuli such as IL-12, although IL-23 was identified originally as a factor necessary for expanding and maintaining the Th17 cell response 7. Previous work has demonstrated that Th17 cells and their associated cytokines (e.g. IL-17A, IL-21, IL-23) are expressed highly in the inflamed mucosa of patients with inflammatory bowel disease (IBD); genome-wide association studies have indicated that IL-23 receptor (IL-23R) and five additional genes involved in Th17 differentiation [IL-12B, Janus kinase 2 (JAK-2), signal transducer and activator of transcription 3 (STAT-3), chemokine receptor 6 (CCR )-6 and TNF-SF15] are associated with susceptibility to CD and also partly to ulcerative colitis 8. Taken together, both Th1 and Th17 cells are important mediators of inflammation in the pathogenesis of CD.
IL-21 is a member of the IL-2 family of cytokines, synthesized mainly by a range of differentiated CD4+ Th cells, including Th1, Th2 and Th17 cells 9, and mediates its biological effects through interaction with an orphan receptor, IL-21R 10. IL-21 plays an important role in the control of innate and adaptive immune responses. It exhibits a pleiotropic capacity to regulate T cell differentiation and function, enhances clonal expansion of antigen-activated naive CD4+ and CD8+ T cells, induces the genes encoding IL-12R, IL-18R, IFN-γ, IL-2R and the Th1-associated transcription factor T-bet in activated memory T cells, drives the differentiation of B cells into memory cells and immunoglobulin (Ig)-secreting plasma cells and promotes the activity of natural killer (NK) cells 11. Moreover, IL-21 enhances T follicular helper cell and Th17 cell differentiation, down-regulates the induction of T regulatory cells and controls the activity of non-immune cells, including epithelial cells and stromal cells 9–11. Evidence has shown that IL-21 is associated with the development of several autoimmune diseases such as rheumatoid arthritis, systemic lupus erythematosus and coeliac disease 11,12. In recent years, IL-21 has been found to be involved in the pathogenesis of human IBD 13,14. Our recent work has also demonstrated that IL-21R-positive cells were increased significantly in the inflamed mucosa of IBD patients. Importantly, IL-21 could enhance the IBD NK cell cytotoxic response, trigger IBD T cells to produce proinflammatory cytokines and induce IBD CD4+ T cells to differentiate into Th17 cells 15. These data indicate that IL-21 plays an important role in the pathogenesis of IBD, and neutralizing IL-21 may have a therapeutic potential in the management of IBD.
Infliximab (IFX) is an anti-TNF monoclonal antibody (mAb) that binds soluble and cell-surface TNF with high affinity and specificity and which has offered a novel approach for the treatment of active CD, with very encouraging results in terms of efficacy and safety 16. However, the anti-inflammatory action of IFX is not understood fully, and it is supposed that several mechanisms contribute to the clinical efficacy of its CD therapy, including triggering apoptosis in monocytes and in lamina propria (LP) T cells 17, restoring epithelial barrier function 18, inducing intestinal fibroblasts motility 19 and down-regulating proinflammatory production in intestinal mucosa 20.
In this study, we evaluated the efficacy of IFX therapy on clinical remission and intestinal mucosal healing in Chinese CD patients, and investigated its role in regulating IL-21 expression and Th17 cell infiltration in intestinal mucosa. We found that IFX administration could induce clinical remission and promote intestinal mucosal healing. Importantly, IFX could down-regulate IL-21 expression and Th17 cell infiltration markedly in inflamed mucosa of CD patients, and suppress proinflammatory cytokine production (e.g. TNF, IFN-γ, IL-17A, IL-21) significantly by CD CD4+ T cells and Th17 differentiation.
Materials and methods
Patients and sample collection
Twenty-six patients with active CD (17 male and nine female, age 20−46 years, disease duration 3−46 months) were recruited at the Department of Gastroenterology, the Shanghai Tenth People's Hospital of Tongji University from June 2009 to October 2012. The diagnosis of CD was based on conventional clinical features, radiological and endoscopic features, and finally confirmed by histological examination of ileal and colonic biopsies. None received any immunosuppressants (i.e. azathioprine, 6-mercaptopurine, methotrexate) and targeted biological therapy (e.g. IFX) at initial study. These CD patents were treated with anti-TNF mAb (IFX; Cilag AG, Schaffhausen, Switzerland) intravenously at a dose of 5 mg/kg at weeks 0, 2 and 6, according to the manufacturer's instructions, and infusions were delivered over a 2-h period. The patients were monitored weekly during follow-up; serum samples were collected at week 10 after the first infusion. Clinical response or remission was evaluated at weeks 0 and 10 after the first IFX infusion using the Crohn's disease activity index (CDAI), endoscopic findings and routine biological analysis, e.g. serum C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) 21. Response to treatment was defined as a decrease in CDAI from baseline of ≥ 100 points and remission as an absolute value ≤ 150. Non-response was defined as no improvement and an increase of CDAI score within 10 weeks following administration. Endoscopic biopsies were taken at sites of active inflammation adjacent to ulcerations before and 10 weeks after IFX therapy. The endoscopic assessment of disease activities for mucosal inflammation was performed using the simple endoscopic score for Crohn's disease (SES-CD) 22. Endoscopic biopsy samples from 18 healthy volunteers paired for sex and age (11 male and seven female, age 21–49 years) were also taken for comparison. In addition, ethylenediamine tetraacetic acid (EDTA) anti-coagulated blood samples (approximately 20 ml) were obtained from these patients and healthy controls after overnight fasting using a vacuum tube for isolation of peripheral blood mononuclear cells (PBMC). This study was approved by the Institutional Review Board for Clinical Research of the Shanghai Tenth People's Hospital. Written informed consent was also obtained from all subjects before study.
Reagents
RPMI-1640 medium, fetal bovine serum (FBS), penicillin (100 U/ml) and streptomycin (100 μg/ml), L-gentamycin and 2-mercaptoethanol (ME) were all purchased from HyClone (Logan, UT, USA). An untouched human CD4+ T cells isolation kit was purchased from Dynal Biotechnology (Oslo, Norway); an RNeasy Kit was purchased from Invitrogen Life Technologies (Grand Island, NY, USA); and a SYBR PrimeScript RT reagent kit was purchased from TaKaRa (Dalian, China). Envision flex peroxidase-blocking reagent was purchased from Dako (Glostrup, Denmark). Rabbit anti-human IL-21 polyclonal antibody was purchased from Novus Biologicals (Littleton, CO, USA), anti-IL-17A mAb (clone eBio64CAP17, mouse IgG1) was purchased from eBioscience (San Diego, CA, USA) and horseradish peroxidase (HRP)-conjugated rabbit anti-mouse IgG and goat anti-mouse IgG were purchased from Jackson Immunoresearch Laboratories (West Grove, PA, USA). Anti-CD3 (clone UCHT1, mouse IgG1), anti-CD28 (clone CD28·2, mouse IgG1) and enzyme-linked immunosorbent assay (ELISA) kits for TNF, IFN-γ, IL-17A, IL-4, IL-10, IL-13, IL-17A and IL-21 were purchased from eBioscience (San Diego, CA, USA).
Immunohistochemistry
To determine the localization of IL-21- and IL-17A-positive cells in intestinal mucosa, immunohistochemistry was performed on 5-μm-thick sections from fresh-frozen intestinal mucosal tissues. Sections were air-dried overnight, fixed in acetone for 10 min and rinsed in phosphate-buffered saline (PBS) for 5 min. After incubation with Envision flex peroxidase-blocking reagent for 10 min, these sections were incubated with goat anti-human IL-21 polyclonal antibody (dilution 1:100) or anti-human IL-17A mAb (dilution 1:150) at 4°C overnight. After washing, the sections were incubated for 60 min with HRP-conjugated goat anti-rabbit or goat anti-mouse IgG (dilution 1:400) at room temperature. The colour reaction was developed with 3,3′-diaminobenzidine and the sections were counterstained with haematoxylin. As negative controls, sections were treated with PBS instead of primary antibody. To determine the proportion of positive cells, five fields of intestinal mucosa were selected randomly at high power (× 400). The positive cells and total stromal cells were counted in the LP. The proportion of positive cells was calculated as follows: [(positive cells)/(total cell number)] × 100 23–25.
Quantitative real-time polymerase chain reaction (PCR)
Total RNA was extracted from the fresh-frozen biopsies using the RNeasy Kit, according to the manufacturer's instructions. Total RNA quantity and quality isolated from each sample was assessed using a NanoVue spectrophotometer (GE Healthcare, Piscataway, NJ, USA), with a 260/280 ratio of > 1·8 and 28S/18S ratio of > 1·4 for the majority of the samples. The cDNA was synthesized with a SYBR PrimeScript reverse transcription (RT) reagent kit, according to the manufacturer's instructions. Quantitative real-time PCR was performed in the ABI prism 7900 HT sequence detector (Applied Biosystems, Foster City, CA, USA) using SYBR green methodology. β-actin was used as the endogenous reference gene. All primers were synthesized and purchased from ShengGong BioTeck (Shanghai, China) and used according to standard methodologies. All PCR reactions were run in triplicate and performed with 40 cycles using the following conditions: 95°C for 1 min, followed by 40 cycles at 95°C for 15 s and 60°C for 30 s. The relative target gene expression levels were calculated as a ratio relative to the β-actin reference mRNA. Quantitative real-time PCR analysis was carried out using the 2-△△Ct method 15,23,24.
Culture of intestinal biopsies
Freshly obtained inflamed biopsies from CD patients during endoscopic examination were cultured (two biopsy samples/well) in 1 ml RPMI-1640 medium containing 10% fetal bovine serum, and supplemented with penicillin (100 U/ml) and streptomycin (100 mg/ml) in the presence of IFX or control human IgG (HIg; Sigma Aldrich, St Louis, MO, USA), both at a dose of 50 μg/ml at 37°C in 5% CO2 humidified air for 24 h. The cultured biopsies were then harvested, followed by extraction of total RNA, and levels of IL-17A, IL-21 and RORC mRNA were analysed by quantitative real-time PCR.
Isolation and culture of CD4+ T cells
PBMC and LPMC were isolated as described previously 15,23, and peripheral blood (PB)- or LP-CD4+ T cells were then purified by negative selection using the Dynal untouched human CD4+ T cells isolation kit (Dynal AS; Invitrogen). Cell purity was evaluated routinely by flow cytometry and ranged from 95 to 99%. Viable cells were determined by 0·1% Trypan blue exclusion (viability ≥ 95%) and counted with a haemocytometer. CD4+ T cells were resuspended in RPMI-1640 medium, supplemented with 10% heat-inactivated FBS, penicillin (100 U/ml) and streptomycin (100 μg/ml), 2 mM gentamycin and 50 μM 2-ME.
Purified PB- or LP-CD4+ T cells (1 × 106/ml) from IBD patients and controls were incubated with anti-CD3 (5 μg/ml) and anti-CD28 (2 μg/ml) in the absence or presence of IFX (50 ng/ml) and IL-21R/Fc (20 μg/ml). After 48 h of culture, supernatants were harvested and analysed for TNF, IFN-γ, IL-17A and IL-21 production by sandwich enzyme-linked immunosorbent assay (ELISA) kits according to our previous reports 15,23. The sensitivity of each assay was 10 pg/ml. Cultured cells were then collected followed by extraction of total RNA; mRNA levels were analysed for evaluation of TNF, IFN-γ, IL-17A, IL-21 and RORC, as described previously 15,23–25.
Statistical analyses
All data are expressed as mean ± standard error of the mean (s.e.m.). Statistical analysis was performed using spss statistics version 13·0 (SPSS, Chicago, IL, USA). Differences between groups were compared using the t-test for two independent samples. A value of P < 0·05 was considered statistically significant.
Results
IFX induces clinical remission and promotes intestinal mucosal healing
Twenty-six patients with active CD were recruited and treated with IFX as indicated in Materials and methods at weeks 0, 2 and 6; the CDAI and endoscopic scores were evaluated at week 10 after IFX administration. As shown in Fig. 1, the CDAI scores were observed to be decreased significantly at week 10 after IFX treatment compared with those at the beginning of treatment (112 ± 31 versus 213 ± 34, P < 0·005). The levels of serum ESR and CRP were also found to be decreased markedly from 46·8 ± 6·8 mm/h to 23·6 ± 8·4 mm/h (P < 0·05) and from 52·1 ± 12·5 mg/l to 10·3 ± 4·4 mg/l (P < 0·005), respectively. Moreover, SES-CD was also performed and demonstrated a marked improvement at week 10 after IFX therapy compared with that the beginning of therapy (7 ± 2 versus 12 ± 3, P < 0·05). Of all 26 CD patients, 12 patients achieved ulcer disappearance (46·2%), seven showed a decreased number of intestinal mucosal ulcer (26·9%), four had a smaller area of ulcer (15·4%) and three showed no response. Taken together, our data indicate that IFX therapy could induce clinical remission and promote intestinal mucosal healing.
Figure 1.
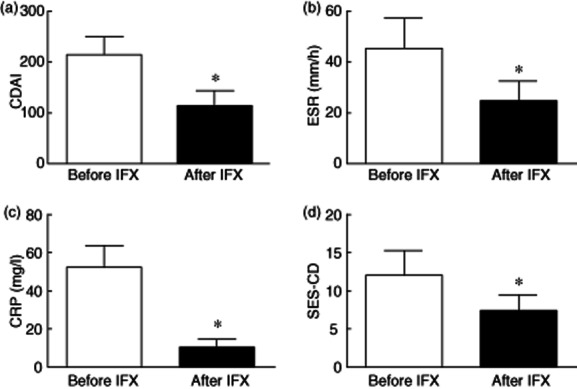
Infliximab (IFX) therapy induces clinical remission and promotes intestinal mucosal healing in Crohn's disease (CD) patients. Twenty-six patients with active CD were treated with IFX at weeks 0, 2 and 6; the CD activity index (CDAI), erythrocyte sedimentation rate (ESR) C-reactive protein (CRP) and endoscopic scores were evaluated before and at week 10 after IFX administration. *P < 0·05 versus values before IFX therapy.
IFX administration down-regulates IL-21 and Th17 cell infiltration in inflamed mucosa of CD patients
Because our previous work has demonstrated that potential role of IL-21 is involved in the induction of mucosal immune response and Th17 cell differentiation in the pathogenesis of IBD 15, we further analysed IL-21-positive cells and Th17 cells in the inflamed mucosa of CD patients at transcriptional and translational levels. First, we analysed the mRNA levels of IL-21, TNF, IFN-γ, IL-17A and RORC in intestinal mucosa from 26 active CD patients before and week 10 after IFX therapy, as well as 16 healthy controls by quantitative real-time PCR. Figure 2 shows that the levels of IL-21, TNF, IFN-γ, IL-17A and RORC mRNA were increased significantly in inflamed ileum/colon of CD patients in contrast to those in healthy controls (P < 0·05), consistent with our previous data showing that proinflammatory cytokines (e.g. IL-21, TNF, IFN-γ) and Th17 cells play an important role in the pathogenesis of CD 15. Interestingly, IFX therapy was demonstrated to down-regulate IL-21, TNF, IFN-γ, IL-17A and RORC mRNA expression significantly in the intestinal mucosa of CD patients (P < 0·05).
Figure 2.
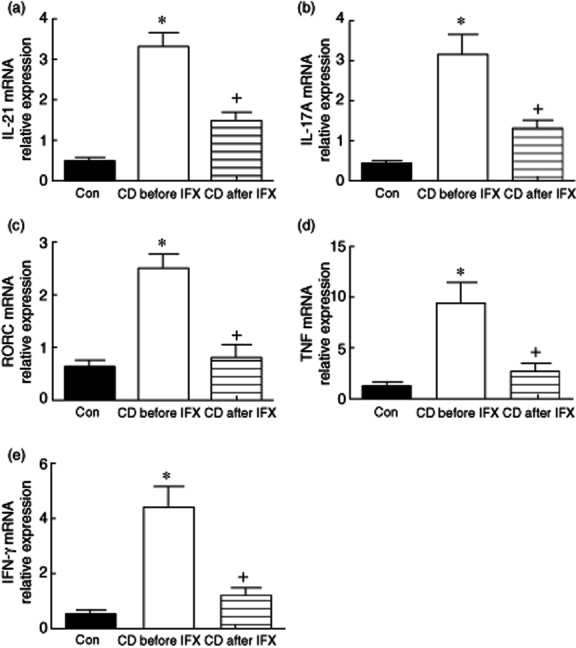
Infliximab (IFX) down-regulates interleukin (IL)-21 and T helper type 17 (Th17) cell infiltration in the inflamed mucosa of Crohn's disease (CD) patients. Intestinal mucosal biopsies were taken from healthy controls (n = 16) and CD patients (n = 26) before and 10 weeks after IFX treatment. Total RNA was extracted and analysed for IL-21, tumour necrosis factor (TNF), interferon (IFN)-γ, IL-17A and retinoic orphan receptor C (RORC) mRNA by quantitative real-time polymerase chain reaction (PCR). *P < 0·005 versus healthy controls; +P < 0·05 versus values before IFX therapy.
Moreover, immunohistological analysis demonstrated the in-situ expression of numerous IL-21+ cells with strong cytoplasmic staining in the LP of inflamed mucosa of active CD patients (Fig. 3a). Intestinal epithelial cells showed negative expression of IL-21 in all sections. After treatment with IFX, the proportion of IL-21+ cells in the LP of inflamed mucosa from CD patients was found to be decreased markedly compared with that before IFX treatment (9·4% ± 2·8% versus 26·7% ± 6·8%, P < 0·005) (Fig. 3b). These results indicate that IFX could suppress IL-21 expression in the intestinal mucosa of CD patients.
Figure 3.
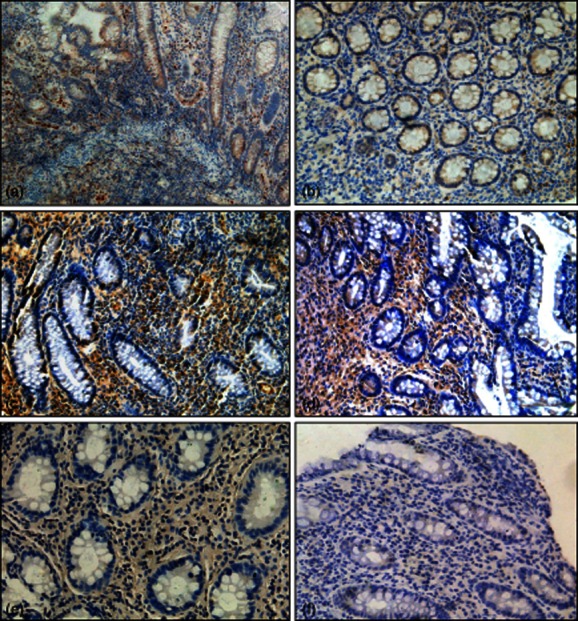
Infliximab (IFX) decreases interleukin (IL)-21- and IL-17A-positive cell infiltration in the inflamed mucosa of Crohn's disease (CD) patients. Representative frozen sections were obtained from inflamed mucosa of CD patients before (a,c) and at week 10 after IFX treatment (b,d), and stained for IL-21 (a,b) and IL-17A (c,d) by immunohistochemistry. (e,f) The CD sections were stained with a control isotype antibody for IL-21 or IL-17A, respectively. Original magnification × 200.
Additionally, we investigated the role of IFX administration in the inhibition of IL-17-expressing cell infiltration in inflamed mucosa of CD patients before and after IFX treatment using immunostaining. As shown in Fig. 3c,d, the levels of IL-17+ cells were decreased markedly after IFX treatment compared with those at the beginning of IFX administration (10·4% ± 2·5% versus 31·5% ± 5·1%, P < 0·005). These data indicate that anti-TNF treatment can also inhibit intestinal mucosal inflammation effectively in CD patients through down-regulation of Th17 cell infiltration.
IFX treatment down-regulates IL-21 and Th17 cell infiltration in intestinal mucosal biopsies of CD patients in vitro
To investigate whether anti-TNF mAb treatment could down-regulate expression of IL-17A and Th17 cell differentiation in intestinal mucosa in vitro, freshly inflamed biopsies were obtained from 10 CD patients during endoscopic examination, cultured (two biopsy samples/well) in 1 ml RPMI-1640 medium containing 10% FBS and supplemented with penicillin (100 U/ml) and streptomycin (100 mg/ml) in the presence of IFX or control HIg (both at 50 μg/ml) at 37°C in 5% CO2 humidified air for 24 h. Total RNA was then extracted and analysed for IL-21, IL-17A and RORC mRNA expression by quantitative real-time PCR. Figure 4 demonstrates that IFX treatment could reduce markedly the expression of IL-21, IL-17A and RORC mRNA in intestinal biopsies. These data confirm further that anti-TNF mAb could suppress IL-21+ and Th17 cell infiltration in cultured biopsies.
Figure 4.
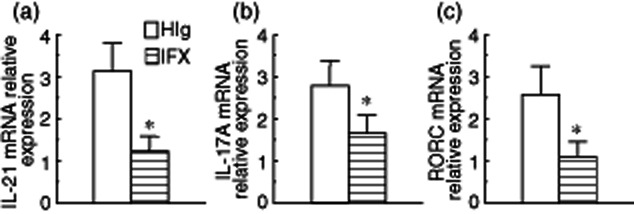
Infliximab (IFX) treatment decreases interleukin (IL)-21 expression and T helper type 17 (Th17) cell infiltration in intestinal mucosal biopsies from Crohn's disease (CD) patients in vitro. Inflamed intestinal biopsies were obtained from 10 patients with active CD during endoscopic examination, and cultured (two biopsy samples/well) in the presence of IFX or control human immunoglobulin (IgG (HIg) (both at 50 μg/ml) at 37°C in 5% CO2 humidified air for 24 h. Total RNA was then extracted and analysed for IL-21, IL-17A and retinoic orphan receptor C (RORC) by quantitative real-time PCR. Gene expression was normalized to β-actin mRNA levels in each sample. *P < 0·05 versus values before IFX therapy.
IFX suppresses CD CD4+ T cell differentiation into Th17 cells and IL-21 expression
To investigate further the potential role of IFX in the regulation of CD CD4+ T cell differentiation, PB- or LP-CD4+ T cells were isolated from CD patients and healthy controls and stimulated with anti-CD3 (5 μg/ml) plus anti-CD28 (2 μg/ml) in the absence or presence of IFX (50 μg/ml) and IL-21R/Fc (20 μg/ml). After 48 h of culture, supernatants were harvested and assayed for IFN-γ, TNF, IL-17A and IL-21. Table 1 demonstrates that the production of IFN-γ, TNF, IL-17A and IL-21 was significantly higher in the patients' cultures than in the cultures from controls when stimulated with anti-CD3 and anti-CD28 (P < 0·05). Interestingly, the inclusion of IFX resulted in down-regulating markedly expression of IFN-γ, TNF, IL-17A and IL-21 by CD CD4+ T cells (P < 0·05). In contrast, low levels of IL-4, IL-5 and IL-13 production were observed in the supernatants by CD4+ T cells under stimulation with anti-CD3 and anti-CD28 from all groups (data not shown).
Table 1.
Infliximab (IFX) down-regulates Crohn's disease (CD) CD4+ T cell proinflammatory cytokine production (pg/ml)
| n | IFN-γ | TNF | IL-17A | IL-21 | ||
|---|---|---|---|---|---|---|
| PB CD4+ | ||||||
| Con | 8 | αCD3 + αCD28 | 1462 ± 256 | 167 ± 42 | 476 ± 114 | 421 ± 121 |
| αCD3 + αCD28 + IFX | 1024 ± 149* | 15 ± 2·5 | 314 ± 78* | 348 ± 117 | ||
| αCD3 + αCD28 + IFX + IL-21R/Fc | 892 ± 210+ | 14 ± 3·4 | 216 ± 58+ | n.d. | ||
| CD | 9 | αCD3 +αCD28 | 3246 ± 452# | 412 ± 68# | 718 ± 149# | 689 ± 175# |
| αCD3 + αCD28 + IFX | 1847 ± 315* | 13 ± 3·5 | 458 ± 108* | 386 ± 102* | ||
| αCD3 + αCD28 + IFX + IL-21R/Fc | 1143 ± 165+ | 12 ± 3·1 | 345 ± 78+ | n.d. | ||
| LP CD4+ | ||||||
| Con | 5 | αCD3 + αCD28 | 1843 ± 387 | 215 ± 52 | 561 ± 172 | 568 ± 147 |
| αCD3 + αCD28 + IFX | 1258 ± 318* | 12 ± 3·3 | 378 ± 106* | 383 ± 96* | ||
| αCD3 + αCD28 + IFX + IL-21R/Fc | 788 ± 215+ | 11 ± 2·6 | 254 ± 62+ | n.d. | ||
| CD | 5 | αCD3 + αCD28 | 4524 ± 548# | 589 ± 117# | 1325 ± 319# | 1018 ± 253# |
| αCD3 + αCD28 + IFX | 2889 ± 414* | 11 ± 2·8 | 745 ± 142* | 585 ± 113* | ||
| αCD3 + αCD28 + IFX + IL-21R/Fc | 1752 ± 325+ | 13 ± 3·9 | 517 ± 72+ | n.d. | ||
P < 0·05 versus data under anti-CD3 + anti-CD28 stimulatory conditions in the same group.
P < 0·05 versus data under anti-CD3 + anti-CD28 + IFX stimulatory conditions in the same group.
P < 0·05 versus controls under the same stimulatory conditions. Peripheral blood (PB) or lamina propria (LP) CD4+ T cells from CD patients and healthy controls (Con) as indicated were stimulated with anti-CD3 and anti-CD28 monoclonal antibodies (mAbs) in the presence of IFX ± IL-21R/Fc. After 48 h of culture, supernatants were harvested and assayed for interferon (IFN)-γ, tumour necrosis factor (TNF), interleukin (IL)-17A and IL-21 by enzyme-linked immunosorbent assay (ELISA); n.d.: not done.
To clarify further whether IFX could regulate CD CD4+ T cell differentiation, the cultured CD4+ T cells as indicated above were also collected after 48 h of culture, followed by extraction of total RNA, and the mRNA levels were analysed for evaluation of Th17 differentiation and IL-21 expression. As shown in Fig. 5, the mRNA levels of IL-21, IL-17A and RORC were increased markedly in CD CD4+ T cells when stimulated with anti-CD3 and anti-CD28. Interestingly, the presence of IFX could prohibit Th17 cell differentiation markedly and reduce the expression of IL-17A and IL-21. Thus, our data suggest that IFX plays an important immunoregulatory role in the treatment of CD patients.
Figure 5.
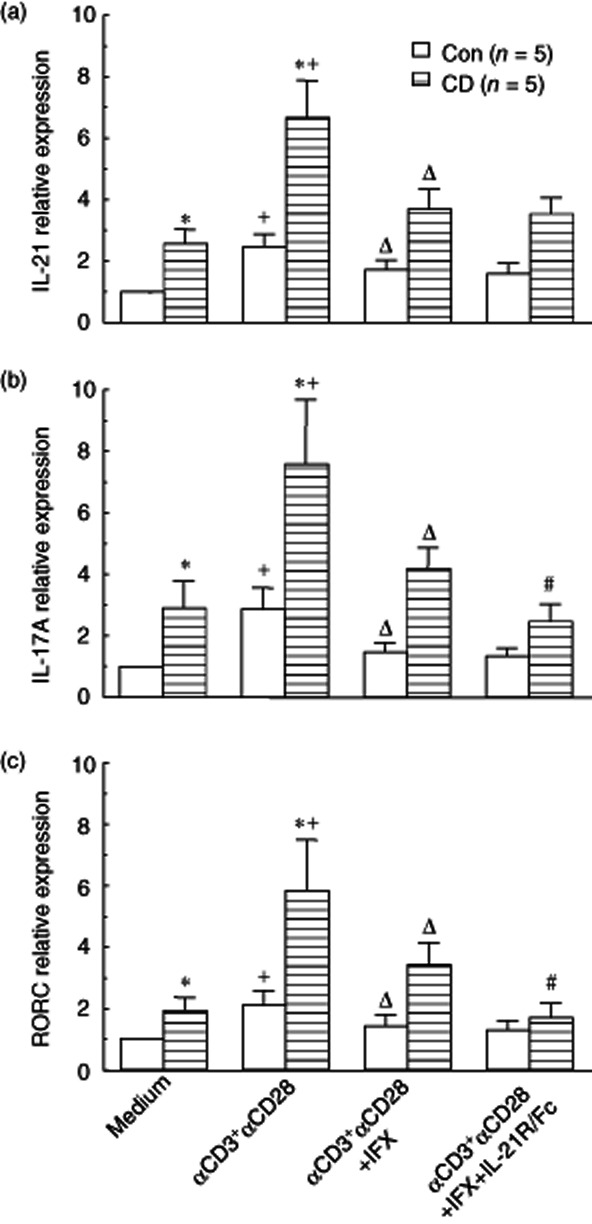
Infliximab (IFX) suppresses Crohn's disease (CD) lamina propria (LP) CD4+ T cell interleukin (IL)-21 and IL-17A mRNA expression and T helper type 17 (Th17) cell differentiation. Purified LP-CD4+ T cells (1 × 106/ml) from CD patients and healthy controls (Con) were stimulated with anti-CD3 (5 μg/ml) and anti-CD28 (2 μg/ml) in the absence or presence of IFX (50 μg/ml) and IL-21R/Fc (20 μg/ml). After 48 h of culture, the cultured LP CD4+ T cells were harvested, followed by extraction of total RNA, and levels of IL-21 (a), IL-17A (b) and Th17 cell transcription factor retinoic orphan receptor C (RORC) (c) were then analysed by quantitative real-time PCR. Gene expression was normalized to β-actin mRNA levels in each sample. Data represent average fold increases or decreases over baseline levels in healthy controls cultured in medium alone (defined arbitrarily as 1·0). *P < 0·005 versus healthy controls under the same stimulatory conditions. +P < 0·05 versus data from the same group cultured in medium alone. ΔP < 0·05 versus data from the same group stimulated with anti-CD3 and anti-CD28 mAbs. #P < 0·05 versus data from the same group stimulated with anti-CD3 and anti-CD28 monoclonal antibodies (mAbs) in the presence of IFX.
As we and others have reported that IL-21 is overexpressed in inflamed mucosa of IBD and induces IBD CD4+ T cell activation to differentiate into Th17 cells 13,15, we asked whether blockage of endogenous IL-21 with recombinant human IL-21R/Fc fusion protein could influence Th17 cell differentiation. To this end, PB- or LP-CD4+ T cells were isolated from CD patients and healthy controls and stimulated with coated anti-CD3 and anti-CD28 in the absence or presence of IFX and IL-21R/Fc for 48 h. Levels of mRNA for IL-17A and RORC were analysed by quantitative real-time PCR. As shown Fig. 5, neutralizing endogenous IL-21 production by activated CD4+ T cells with IL-21R/Fc fusion protein was demonstrated to decrease further the expression of IL-17A and RORC mRNA. These data suggest that IFX could down-regulate Th17 cell infiltration partially through blockage of the IL-21 signalling pathway.
Discussion
The current study has described the efficacy of IFX in inducing clinical remission and promoting mucosal healing in Chinese CD patients. Importantly, our data have proved that IFX could suppress markedly CD CD4+ T cell proinflammatory cytokine production (e.g. IL-17A, IL-21) and Th17 cell infiltration in intestinal mucosa. This work has highlighted the critical role of IFX in the management of human CD patients, and expanded our knowledge of its immunoregulatory role in the control of mucosal inflammation. To date, the precise aetiology of CD remains unknown, but it is well established that there is an infiltration of inflammatory cells focally in the bowel, leading to the production of proinflammatory cytokines and propagation of the immune response 1–3. With the recent discovery of the Th17 subset and Th17-associated cytokines, understanding of CD has expanded rapidly because these cells and cytokines have a critical role in the pathogenesis of CD 5–7. Therefore, Th17-associated cytokines (e.g. IL-17A, IL-21, IL-23) may represent a promising target in the treatment of human CD.
Apart from its direct TNF neutralizing activity, the anti-inflammatory mechanism of IFX is not understood completely, but accumulating evidence has suggested that one of its anti-inflammatory mechanisms may be related to inhibition of the production of proinflammatory cytokines and the infiltration of T cells in intestinal mucosa 26,27. In the present study, our data have shown that two proinflammatory cytokines, IL-17A and IL-21, which were found to be increased significantly in inflamed mucosa of CD patients compared with healthy controls, were down-regulated markedly in those patients with the improvement of routine clinical parameters and endoscopic scores after IFX infusion. Previous data have demonstrated the association of IL-17A and IL-21 with the pathogenesis of various autoimmune diseases, including CD. Fina et al. 28 reported that IL-21-deficient mice are largely protected against intestinal inflammation in both dextran sodium sulphate (DSS)- and 2,4,6-trinitrobenzenesulphonic acid (TNBS)-induced colitis, which was associated with a marked decrease in Th17-related molecules, such as IL-17A and IL-17F. Our previous studies have also shown that IL-21 could promote LP-CD4+ T cells to differentiate into Th17 cells in CD patients 15. Thus, the current results revealed a novel anti-inflammatory activity of IFX by the reduction of IL-21 expression in the inflamed mucosa of CD patients, and the possibility that the cascade by which TNF may induce IL-21 production actually occurs in vivo. A similar effect of IFX was also reported in the treatment of rheumatoid arthritis patients, showing that the serum level of IL-23 was decreased significantly in those patients after IFX infusion and correlated with ESR, CRP and the disease activity scores of 28 joints (DAS28) 29.
Evidence has shown that Th17 cells are identified as a potential subset of Th cells related to the development of autoimmunity and inflammatory responses such as CD, and that IL-21-driven intestinal pathology is associated with increased Th17 cell differentiation 30,31. IL-17-producing RORC+ CD4+ T cells have been found to be constitutively present in the LP in mice 32 and highly infiltrated in the inflamed mucosa of CD patients 33. A previous report comparing the ability of Th1 and Th17 cells to induce colitis in mice has proved that Th17 cells are significantly more pathogenic than their Th1 counterparts 34. Recent work 35 has demonstrated that IL-17 knock-out mice with TNBS-induced colitis had significantly lower neutrophil infiltration and inflammatory macroscopic scores in inflamed colon compared to wild-type mice. The treatment of mice with anti-IL-17A neutralizing antibody confirmed a specific role for IL-17A in TNBS-induced acute colitis, suggesting that IL-17 signalling plays a critical role in the local inflammation of TNBS-induced colitis. In the current study, we observed that the mRNA levels of IL-17A and RORC were decreased markedly after IFX treatment in the inflamed mucosa of CD patients, indicating that IFX could inhibit the infiltration of Th17 cells in these patients. Our results are consistent with a previous study 36 showing that IFX could suppress the differentiation of naive CD4+ T cells isolated from the peripheral blood into Th17 cells in vitro, characterized by the reduction of IL-17A secretion in the culture supernatants. Moreover, another study confirmed further that IFX in combination with methotrexate treatment could decrease the percentage of Th17 cells in peripheral blood mononuclear cells and the concentration of IL-17A in the supernatants from patients with rheumatoid arthritis 37. Taken together, these data suggest that Th17 cells play a critical role in the pathogenesis of intestinal mucosal inflammation and that IFX plays a profound anti-inflammatory role in modulating Th17 cell differentiation in several autoimmunities, particularly in CD.
Our study has revealed that IFX could improve clinical remission and promote intestinal mucosal healing in active CD patients. One important anti-inflammatory mechanism may contribute to down-regulating the expression of proinflammatory cytokines (e.g. IL-17A, IL-21, IL-23) and inhibiting Th17 cell infiltration in the inflamed mucosa of CD patients. This work has shed some light on understanding more clearly the immunoregulatory role of IFX in the control of human IBD. In addition to specific mechanisms, as described previously 17–20, IFX not only suppresses mucosal CD4+ T cell activation directly to differentiate into Th17 cells, but also down-regulates IL-21 expression by mucosal CD4+ T cells, which contributes further to the synergistic role in the inhibition of Th17 cell differentiation.
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (30971358; 81061120521), and Shanghai Science and Technology Commission (12XD1404000).
Disclosure
The authors do not have any conflicts of interest.
Supporting information
Additional Supporting Information may be found in the online version of this article at the publisher's web-site:
Fig. S1. Infliximab (IFX) suppresses Crohn's disease (CD) peripheral blood (PB) CD4+ T cell interleukin (IL)-21 and IL-17A mRNA expression and T helper type 17 (Th17) cell differentiation. Purified PB CD4+ T cells (1 × 106/ml) from CD patients and healthy controls were stimulated with anti-CD3 (5 μg/ml) and anti-CD28 (2 μg/ml) in the absence or presence of IFX (50 μg/ml) and IL-21R/Fc (20 μg/ml). After 48 h of culture the cultured PB CD4+ T cells were harvested, followed by extraction of total RNA, and levels of IL-21 (A), IL-17 (B) and Th17 cell transcription factor retinoic orphan receptor C (RORC) (C) were then analysed by quantitative real-time polymerase chain reaction (PCR). Gene expression was normalized to β-actin mRNA levels in each sample. Data represent average fold increases or decreases over baseline levels in healthy controls cultured in medium alone (defined arbitrarily as 1·0). *P < 0·005 versus healthy controls under the same stimulatory conditions. +P < 0·05 versus data from the same group cultured in medium alone. ΔP < 0·05 versus data from the same group stimulated with anti-CD3 and anti-CD28 monoclonal antibodies (mAbs). #P < 0·05 versus data from the same group stimulated with anti-CD3 and anti-CD28 mAbs in the presence of IFX.
References
- 1.Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009;361:2066–2078. doi: 10.1056/NEJMra0804647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xavier RJ, Podolsky DK. Unravelling the pathogenesis of inflammatory bowel disease. Nature. 2007;448:427–434. doi: 10.1038/nature06005. [DOI] [PubMed] [Google Scholar]
- 3.Di Sabatino A, Biancheri P, Rovedatti L, et al. New pathogenic paradigms in inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:368–371. doi: 10.1002/ibd.21735. [DOI] [PubMed] [Google Scholar]
- 4.Bouma G, Strober W. The immunological and genetic basis of inflammatory bowel disease. Nat Rev Immunol. 2003;3:521–533. doi: 10.1038/nri1132. [DOI] [PubMed] [Google Scholar]
- 5.Liu ZJ, Yadav PK, Su JL, et al. Potential role of Th17 cells in the pathogenesis of inflammatory bowel disease. World J Gastroenterol. 2009;15:5784–5788. doi: 10.3748/wjg.15.5784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Maddur MS, Miossec P, Kaveri SV, et al. Th17 cells: biology, pathogenesis of autoimmune and inflammatory diseases, and therapeutic strategies. Am J Pathol. 2012;181:8–18. doi: 10.1016/j.ajpath.2012.03.044. [DOI] [PubMed] [Google Scholar]
- 7.Ghoreschi K, Laurence A, Yang XP, et al. T helper 17 cell heterogeneity and pathogenicity in autoimmune disease. Trends Immunol. 2011;32:395–401. doi: 10.1016/j.it.2011.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Van Limbergen J, Wilson DC, Satsangi J. The genetics of Crohn's disease. Annu Rev Genomics Hum Genet. 2009;10:89–116. doi: 10.1146/annurev-genom-082908-150013. [DOI] [PubMed] [Google Scholar]
- 9.Spolski R, Leonard WJ. Interleukin-21: basic biology and implications for cancer and autoimmunity. Annu Rev Immunol. 2008;26:57–79. doi: 10.1146/annurev.immunol.26.021607.090316. [DOI] [PubMed] [Google Scholar]
- 10.Leonard WJ, Zeng R, Spolski R. Interleukin 21: a cytokine/cytokine receptor system that has come of age. J Leukoc Biol. 2008;84:348–356. doi: 10.1189/jlb.0308149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Monteleone G, Pallone F, MacDonald TT. Interleukin-21: a critical regulator of the balance between effector and regulatory T-cell responses. Trends Immunol. 2008;29:290–294. doi: 10.1016/j.it.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 12.Monteleone G, Pallone F, MacDonald TT. Interleukin-21 (IL-21)-mediated pathways in T cell-mediated disease. Cytokine Growth Factor Rev. 2009;20:185–191. doi: 10.1016/j.cytogfr.2009.02.002. [DOI] [PubMed] [Google Scholar]
- 13.Pallone F, Fina D, Caruso R, et al. Role of IL-21 in inflammatory bowel disease. Exp Rev Clin Immunol. 2010;6:537–541. doi: 10.1586/eci.10.44. [DOI] [PubMed] [Google Scholar]
- 14.MacDonald TT, Biancheri P, Sarra M, et al. What's the next best cytokine target in IBD? Inflamm Bowel Dis. 2012;18:2180–2189. doi: 10.1002/ibd.22967. [DOI] [PubMed] [Google Scholar]
- 15.Liu Z, Yang L, Cui Y, et al. Il-21 enhances NK cell activation and cytolytic activity and induces Th17 cell differentiation in inflammatory bowel disease. Inflamm Bowel Dis. 2009;15:1133–1134. doi: 10.1002/ibd.20923. [DOI] [PubMed] [Google Scholar]
- 16.Danese S, Colombel JF, Reinisch W, et al. Review article: infliximab for Crohn's disease treatment – shifting therapeutic strategies after 10 years of clinical experience. Aliment Pharmacol Ther. 2011;33:857–869. doi: 10.1111/j.1365-2036.2011.04598.x. [DOI] [PubMed] [Google Scholar]
- 17.ten Hove T, van Montfrans C, Peppelenbosch MP, et al. Infliximab treatment induces apoptosis of lamina propria T lymphocytes in Crohn's disease. Gut. 2002;50:206–211. doi: 10.1136/gut.50.2.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Suenaert P, Bulteel V, Lemmens L, et al. Anti-tumor necrosis factor treatment restores the gut barrier in Crohn's disease. Am J Gastroenterol. 2002;97:2000–2004. doi: 10.1111/j.1572-0241.2002.05914.x. [DOI] [PubMed] [Google Scholar]
- 19.Di Sabatino A, Pender SL, Jackson CL, et al. Functional modulation of Crohn's disease myofibroblasts by anti-tumor necrosis factor antibodies. Gastroenterology. 2007;133:137–149. doi: 10.1053/j.gastro.2007.04.069. [DOI] [PubMed] [Google Scholar]
- 20.Agnholt J, Kaltoft K. Infliximab downregulates interferon-gamma production in activated gut T-lymphocytes from patients with Crohn's disease. Cytokine. 2001;15:212–222. doi: 10.1006/cyto.2001.0919. [DOI] [PubMed] [Google Scholar]
- 21.Jürgens M, Mahachie John JM, Cleynen I, et al. Levels of C-reactive protein are associated with response to infliximab therapy in patients with Crohn's disease. Clin Gastroenterol Hepatol. 2011;9:421–427. doi: 10.1016/j.cgh.2011.02.008. [DOI] [PubMed] [Google Scholar]
- 22.Daperno M, D'Haens G, Van Assche G, et al. Development and validation of a new, simplified endoscopic activity score for Crohn's disease: the SES-CD. Gastrointest Endosc. 2004;60:505–512. doi: 10.1016/s0016-5107(04)01878-4. [DOI] [PubMed] [Google Scholar]
- 23.Liu Z, Yadav PK, Xu X, et al. The increased expression of IL-23 in inflammatory bowel disease promotes intraepithelial and lamina propria lymphocyte inflammatory responses and cytotoxicity. J Leukoc Biol. 2011;89:597–606. doi: 10.1189/jlb.0810456. [DOI] [PubMed] [Google Scholar]
- 24.Liu Z, Feng BS, Yang SB, et al. Interleukin (IL)-23 suppresses IL-10 in the intestine of inflammatory bowel disease. J Biol Chem. 2012;287:3591–3597. doi: 10.1074/jbc.M111.304949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Liu Z, Chen X, Wang X, et al. Chemokine CXCL11 links microbial stimuli to intestinal inflammation. Clin Exp Immunol. 2011;164:396–406. doi: 10.1111/j.1365-2249.2011.04382.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Di Sabatino A, Ciccocioppo R, Cinque B, et al. Defective mucosal T cell death is sustainably reverted by infliximab in a caspase dependent pathway in Crohn's disease. Gut. 2004;53:70–77. doi: 10.1136/gut.53.1.70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Van den Brande JM, Braat H, van den Brink GR, et al. Infliximab but not etanercept induces apoptosis in lamina propria T-lymphocytes from patients with Crohn's disease. Gastroenterology. 2003;124:1774–1785. doi: 10.1016/s0016-5085(03)00382-2. [DOI] [PubMed] [Google Scholar]
- 28.Fina D, Sarra M, Fantini MC, et al. Regulation of gut inflammation and Th17 cell response by interleukin-21. Gastroenterology. 2008;134:1038–1048. doi: 10.1053/j.gastro.2008.01.041. [DOI] [PubMed] [Google Scholar]
- 29.Kageyama Y, Kobayashi H, Kato N. Infliximab treatment reduces the serum levels of interleukin-23 in patients with rheumatoid arthritis. Mod Rheumatol. 2009;19:657–662. doi: 10.1007/s10165-009-0217-6. [DOI] [PubMed] [Google Scholar]
- 30.Korn T, Bettelli E, Gao W, et al. IL-21 initiates an alternative pathway to induce proinflammatory T(H)17 cells. Nature. 2007;448:484–487. doi: 10.1038/nature05970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Feng T, Qin H, Wang L, et al. Th17 cells induce colitis and promote Th1 cell responses through IL-17 induction of innate IL-12 and IL-23 production. J Immunol. 2011;186:6313–6318. doi: 10.4049/jimmunol.1001454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ivanov II, McKenzie BS, Zhou L, et al. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell. 2006;126:1121–1133. doi: 10.1016/j.cell.2006.07.035. [DOI] [PubMed] [Google Scholar]
- 33.Annunziato F, Cosmi L, Santarlasci V, et al. Phenotypic and functional features of human Th17 cells. J Exp Med. 2007;204:1849–1861. doi: 10.1084/jem.20070663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Elson CO, Cong Y, Weaver CT, et al. Monoclonal anti-interleukin 23 reverses active colitis in a T cell-mediated model in mice. Gastroenterology. 2007;132:2359–2370. doi: 10.1053/j.gastro.2007.03.104. [DOI] [PubMed] [Google Scholar]
- 35.Jin Y, Lin Y, Lin L, et al. IL-17/IFN-γ interactions regulate intestinal inflammation in TNBS-induced acute colitis. J Interferon Cytokine Res. 2012;32:548–556. doi: 10.1089/jir.2012.0030. [DOI] [PubMed] [Google Scholar]
- 36.Kanayama K, Nakamura K, Ogino H, et al. Th1 responses are more susceptible to infliximab-mediated immunosuppression than Th17 responses. Dig Dis Sci. 2011;56:3525–3533. doi: 10.1007/s10620-011-1780-1. [DOI] [PubMed] [Google Scholar]
- 37.Shen H, Xia L, Lu J, et al. Infliximab reduces the frequency of interleukin 17-producing cells and the amounts of interleukin 17 in patients with rheumatoid arthritis. J Investig Med. 2010;58:905–908. doi: 10.231/JIM.0b013e3181eb9895. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


