Abstract
Universally viewed as the sentinels and messengers of the immune system and traditionally referred to as professional antigen presenting cells, dendritic cells (DC) play a fundamental role in antitumor immunity. DC are uniquely equipped with the ability to acquire, process and present tumor-derived antigens to T lymphocytes. They can drive the differentiation of naïve T cells into activated tumor-specific effector lymphocytes. DC also dictate the type and regulate the strength and duration of T cell responses. In addition, they contribute to NK and NKT cell antitumoral function and to B cell-mediated immunity. Besides this cardinal role as orchestrators of innate and adaptive immune responses, many studies have provided evidence that DC can also function as direct cytotoxic effectors against tumors. This less conventional aspect of DC lifestyle has however raised controversy as it relates to the origin of these cells and the induction, regulation and mechanisms underlying their tumoricidal activity. The possible impact of the cytotoxic function of DC on their antigen presenting capability has also been the focus of intensive research. In this review, we examine these questions and discuss the biological significance of this non-traditionalproperty and possible strategies to exploit DC killing potential in cancer immunotherapy.
Keywords: Cytotoxicdendritic cells, tumor immunity, dendritic cell-based cancer vaccines
I-INTRODUCTION
Since their original discovery by Steinman et al.,1 DC have been the focus of extensive research to understand their ontology and functions in the regulation of immune responses. It soon became clear that DC are endowed with the unique ability to function as professional antigen presenting cells (APC) critical for the development of adaptive immunity.2, 3 This primordial characteristic has been the foundation for the development of DC-based vaccines. In the realm of cancer immunotherapy, the prospect of exploiting DC to stimulate protective antitumor immunity has instigated interesting studies in animals and human. This body of research demonstrates some promise but also limitations, underscoring the need for further advance in the field.4–12
DC encompass multiple heterogeneous cell subsets defined by a specific phenotype, anatomic localization and specialized functions dictated by their cytokine production profile.2, 3, 13, 14 Strategically distributed in the body, DC are the sentinels of the immune system constantly surveying their environment for the presence of invading pathogens and sensing for tissue damages. They constantly acquire antigens by phagocytosis or pinocytosis and migrate to lymphoid organs where they initiate specific T cell responses. The endocytosed antigenic proteins, delivered into the antigen processing machinery of DC, are converted into small peptides that are associated with MHC Class I or Class II molecules and exported at the DC surface for presentation to CD8+ or CD4+ T lymphocytes, respectively. To efficiently drive the clonal proliferation and activation of CD4+ T helper (Th) or CD8+ cytotoxic T (CTL) cells, DC need to express lymphocyte co-stimulatory molecules such as CD80, CD86, CD40, OX40L, or ICOSL and produce inflammatory cytokines. Typically, T cell fate decisions are directed by the nature of the DC subset, the level of maturation and activation of the DC and the type of cytokines they produce. The type of T lymphocyte response that eventually develops (for instance Th-1, Th-2, Th-17 or Th-9) is dependent on the DC type.2, 3, 14–16 DC are also capable of presenting glycolipids to NKT cells on CD1d molecule and of controlling the activation of NK and B cells.17–20 Importantly, DC also play an essential role in the mechanisms of self-tolerance, thereby preventing autoimmunity. In absence of maturation and activation signals, DC lacking co-stimulatory molecules can indeed present self-antigens acquired from apoptotic cells to potentially auto-reactive T lymphocytes, resulting in T cell deletion or anergy.21–23 These immature, “tolerogenic” DC can additionally participate in the generation of regulatory T lymphocytes (Treg) or IL-10-secreting Tr-1 cells with suppressor activity.24–30
The notion that arising malignant cells can be recognized and eliminated by the immune system before they develop into clinically apparent tumors, a process known as tumor immunosurveillance, was initially formulated by Burnet and Thomas.31, 32 This seminal concept, now supported by compelling experimental evidence, has undergone substantial evolution and refinements leading to its integration into a much broader process, called cancer immunoediting, extensively discussed by Schreiber et al.32–38 Cancer immunoediting more accurately depicts the complex cross-talks between the immune system and tumors and their outcome as it takes into account the observation that immune cells can not only eliminate tumor cells, but they can also select for immune-resistant cancer escape variants.33, 34, 39 This process typically results from three phases, referred to as the 3 “Es” of cancer immunoediting: elimination (the equivalent of tumor immunosurveillance), equilibrium and escape. DC, by virtue of their APC and immune regulatory functions, can intervene at least at two of these stages. During the first phase, the local inflammation generated around neoplastic lesions by modification of the surrounding tissues leads to the recruitment of innate immune cells such as neutrophils, macrophages, NKT or γδT cells which in turn promote the attraction of immature DC through chemokine production. A main initial source of tumor antigens available for uptake by these immature DC is provided by the death of a fraction of the tumor cells induced by innate immune cells or the cytokines they produced, such as IFN-γ, or resulting from the anti-angiogenic effects of the these molecules.32–34, 39 The ingested tumor-derived material is processed into peptides and presented to T lymphocytes on MHC Class I or Class II molecules after homing of the DC to the draining lymphoid tissues. Tumor-specific CTL expanded and activated by DC express specific chemokine receptors leading to their migration to the tumor site where they participate to cancer cell elimination. This CD8+ response is further facilitated by DC-activated tumor-specific CD4+ Th lymphocytes. DC and Th cells also support the activation of NK, NKT and macrophages. When successful, the elimination phase represents the complete cancer immunoediting process. However, some malignant cells may acquire additional mutations which confer them with increased resistance to immune detection and elimination.39–44 During the equilibrium phase, the immune system exerts a selective pressure on developing tumors leading to the elimination of the most sensitive cancer cells but also, as a corollary, to the selection of these resistant tumor cell variants, eventually leading to the escape phase. At this last stage, uncontrolled tumors commonly exhibit the ability to actively suppress the immune system through the production of immunosuppressive factors or cells as discussed in section III. Some of these mechanisms of tumor-induced suppression result in the negative modulation of DC function leading to the generation of tolerogenic DC. These phenotypically and functionally impaired DC are capable of inducing the anergy or deletion of tumor-specific T lymphocytes and have been involved in immunosuppressive Treg expansion.39, 45–47 Thus, although DC play an essential role as orchestrators of the cooperative interactions between the multiple cellular actors involved in the immune attack against tumors during the first phase of cancer immunoediting, these cells can also contribute to the last escape stage by participating to the mechanism of tumor immune tolerance.
Recent studies have lent support to the notion that, besides their role as potent inducers and controllers of immune responses, DC may also be endowed with a direct tumoricidal function. This less conventional characteristic of DC has however received limited attention and has raised many questions as it relates to the actual ontology of so-called “killer DC”, the acquisition of their cytotoxic activity and the underlying mechanism(s) of tumor cell killing. We here review these aspects, further discuss the influence of DC tumoricidal potential on their APC function and their ability to promote or inhibit anti-cancer immunity. We speculate on the integration of this non-traditional property into the broad concept of cancer immunoediting. Possible options to harness and implement DC tumor killing activity in cancer immunotherapeutic strategies are evaluated.
II-PRINCIPAL ASPECTS OF DC CYTOTOXIC FUNCTION
DC endowed with cytotoxic activity have commonly been referred to as “killer DC” (KDC) although these cells do not constitute a separate, dedicated subset. Rather, multiple heterogeneous subpopulations, naturally occurring in vivo (native) or generated in vitro from specific precursors have been described.48–114 A similar degree of diversity and plasticity has been observed as it relates to the modalities of induction of KDC cytotoxic function and to the effector mechanisms underlying tumor cell killing. Because the different subsets of KDC identified in mouse, rat and human have been extensively reviewed previously,59, 61–63, 70, 72 (Table I) this section will only summarize the main findings and the principal characteristics of these cells.
Table I.
KDC subsets involved in tumor cell killing
| Subset | Mechanism | Induction | Target | Reference |
|---|---|---|---|---|
| Human | ||||
| Blood CD11c+ mDC | TRAIL | IFN-γ, IFN-α | Jurkat, OVCAR3, PC-3, WM793, not normal cells | 51 |
| TNF-α | IL-15, IFN-γ, LPS | Various breast cancer cell lines | 53 | |
| Perforin/Granzyme | TLR-7 agonist | K562 | 57 | |
| Monocyte derived | TRAIL | dsRNA, Type I IFN, CD40L | MDA231 | 67 |
| Type I IFN | Jurkat | 166 | ||
| IFN-β | HL60, Reh | 110 | ||
| Not FasL | Spontaneous | Various hematopoietic tumor cells | 167 | |
| Not TRAIL, FasL | Spontaneous | HPV+ keratinocytes | 168 | |
| NO | LPS | HT29 | 71 | |
| Blood pDC | TRAIL | TLR-7 agonist | Jurkat | 57 |
| HIV, TLR-7 agonist | SupT1 | 107 | ||
| Virus and TLR- 7/TLR-9 agonists | A549, MEL | 50 | ||
| CD34+ derived | TRAIL | IFN-β | HL60, Reh | 110 |
| Cord Blood CD14+ derived | TRAIL | LPS, IFN-γ | HL60, Daudi, Jurkat, not normal cells | 55 |
| Blood CD4+HLADR+Lin− | TRAIL, TNF-α, FasL, LT-α1β2 | Spontaneous | Various tumor cell lines | 52, 108 |
| Blood slanDC (M-DC8+) | TNF-α | IFN-γ | Various tumor cell lines, not normal cells | 65 |
| Mouse | ||||
| Bone marrow derived | NO | Spontaneous | MCA205, MC38, TS/A | 56 |
| IFN-γ | 4T1, B16 | 68 | ||
| LPS, Pam3Cys-SK4 | B16F10 | 69 | ||
| FasL | Spontaneous | A20, EG7, EL4 | 79 | |
| TNF-α | IL-12, IL-18 | CMS4, MethA (sarcomas) | 80 | |
| Rat | ||||
| Bone marrow-derived | NO | NKG2D Ligand (anti-NKRP2) | AK-5 | |
| LPS, IFN-γ | PROb | 54 | ||
| Spleen CD103+ | TNF-α | NKG2D Ligand (anti-NKRP2) | AK-5 | 84 |
| No role of FasL, TRAIL, TNF-α | Spontaneous | Various tumor cell lines | 58, 76, 77 | |
A. Identification of mouse and rat KDC
Studies in mouse and rat were the first to highlight the possibility that DC can function as cytotoxic effector cells. An initial report indicated that a mouse splenic DC subpopulation characterized as CD11c+CD8α+ induced apoptosis of T lymphocytes by a FasL-dependent mechanisms.73 Additional studies later demonstrated the capability of native mouse DC such as Langerhans cells to mediate killing of tumor cells or to play a direct cytotoxic role in the control of bacterial infection.74, 75 The notion that native rat DC are endowed with potent tumoricidal activities was initially tackled in a report demonstrating that a subset of splenic DC expressing CD103 spontaneously up-regulated NKR-P1 during overnight culture and kill the NK-sensitive YAC-1 cells by a Ca2+ dependent mechanism.48 In line with this study, KDC were further characterized in the secondary lymphoid tissues as CD4−CD11b+NKp46−CD103+CD200+MHCII+ and determined to be distinct from the NK cell lineage.58, 76, 77 The induction of tumor cell apoptosis was exclusively mediated by immature DC that did not require exogenous activation and depended on a direct cell to cell contact.58 The exact mechanism of cytotoxicity was not elucidated but a possible role for the perforin-granzyme B system or the death receptor family members FasL, TRAIL, or TNF-α was excluded.77 These studies also demonstrated that immediately after killing of tumor cells KDC were capable of phagocytosing the resulting apoptotic bodies.58, 63 Following maturation these DC became potent APC while exhibiting reduced cytotoxic activity. These early studies sparked a series of investigations focusing on the ability of different DC subpopulations to operate as cytotoxic effectors against cancer cells and on the mechanismsunderlying this property.
Multiple studies have further documented that DC generated in vitro, essentially from bone marrow cells cultured with GM-CSF and IL-4, can exhibit tumoricidal activity, but considerably variable results were obtained in terms of cytotoxic mechanisms.56, 78–82, 115 A role for the death receptor ligands FasL, TRAIL, or TNF-α, or the involvement of NO have been described. We have demonstrated that in rat and mouse, the cytotoxic activity of DC generated from bone marrow was not mediated by perforin/granzyme B, FasL, TNF-α, or TRAIL, but depended on peroxynitrites, the main metabolite of NO.54, 68, 69, 72 In these studies, the killing ability of DC generated from iNOS or gp91 knock-out mice was significantly reduced.68, 69 Independent groups have also reported on the essential role of NO in both mouse 56 and rat 83 bone marrow-derived DC-mediated apoptosis of tumor cells. Huang et al. demonstrated that preconditioning tumor cells with a NO donor, promoted the down-regulation of the anti-apoptotic protein survivin, and sensitized lymphomas to Fas-L-dependent DC-mediated killing.79 With regard to the mode of induction of DC cytotoxic function, some groups have suggested that DC killing potential may be spontaneous in rat 58, 76 and mouse.56, 79 By contrast, we and other groups have demonstrated that non-stimulated immature DC are not capable of killing tumor cells but that their cytotoxic activity can be induced upon activation with specific stimuli such as the TLR4 ligand LPS, the TLR2 agonist Pam3Cys-SK4,69 or IFN-γ.68, 69 This discrepancy may be partly explained by the specific DC subset studied and theculture conditions utilized.
An important, yet not fully addressed question in the field relates to the molecular regulation of DC killing activity. Recently, in an effort to unravel the regulatory signaling pathways involved in the regulation of mouse bone marrow-derived DC cytotoxic function we have partly deconstructed the molecular machinery controlling iNOS in DC generated with GM-CSF and IL-4 (IL-4 DC) or IL-15 (IL-15 DC). We demonstrated that LPS-induced iNOS expression required the activation of the transcription factors NF-κB and ISGF3 only in IL-15 DC and that distinct signaling pathways such as AP-1 may be required in IL-4 DC. IFN-γ induced iNOS expression in IL-4 DC by a STAT-1 dependent mechanism. However in IL-15 DC, the cooperation of PIAS-1 and STAT-3 prevented iNOS expression in response to IFN-γ stimulation (Hanke et al, submitted manuscript). These results further underline that depending on the considered DC subset, the mechanisms underlying the cytotoxicity function of these cells are highly variable and are likely to depend on the genetic program specifically activated in a given DC subtype. In addition, differences in DC culture conditions (composition, type of cytokines) and mode of isolation may influence the signaling pathways controlling the expression of defined effector cytotoxic molecules, partly explaining the divergent results reported in the current literature.
The proof of principle that rodent KDC can effectively impair tumor growth in vivo and promote the survival of tumor bearing animals has been provided in different studies. For instance, splenic rat KDC allowed to kill an osteosarcoma cell line and to acquire tumor cell debris in vitro promoted tumor regression when administered to rats bearing the same osteoscarcoma.76 This KDC vaccine was found to be ineffective in CD8+ T cell-depleted animals, suggesting that KDC were capable of efficiently cross-presenting tumor antigens obtained from target cells to specific CTL.76 Other reports have found that NKG2D agonist antibodies promoted the tumoricidal function of rat CD103+ DC in vitro and hindered tumor progression in vivo.84 However, the specific contribution of DC cytotoxic activity was not clearly determined. In another study, bone marrow-derived DC genetically modified to express IL-12 and IL-18 acquired tumoricidal activity and were capable of impairing the growth of an established sarcoma in a CD4+ and CD8+ T cell-dependent manner when injected into the tumor beds.80 The direct contribution of pDC in the elimination of tumor cells following treatment with imiquimod has additionally been recently outlined in a mouse melanoma model.116
Special mention should be made of interferon-producing killer dendritic cells (IKDC).49, 85–103 Originally characterized in the mouse by a B220+CD11c+NK1.1+CD49b+ phenotype with a variable MHC Class II expression profile, these cells were described as hybrids exhibiting characteristics of both NK cells and plasmacytoid DC. IKDC were reported to exhibit potent anti-tumoral effects through TRAIL and perforin/granzyme-dependent mechanisms. It was proposed that when appropriately stimulated IKDC could first function as NK cells by virtue of their cytotoxic property and then following contact with tumor cells could up-regulate MHC Class II and behave as APC endowed with the ability to activate naive CD8+ and CD4+ T lymphocytes. In addition, it was documented that these cells could produce both IFN-γ and IFN-α.90, 98, 100–103 The notion that this hybrid NK-DC cell type unifying killing and APC functions belongs to the DC lineage has however been subjected to intensive debate and more recent studies have eventually provided evidence that IKDC merely originate from NK progenitors.85, 91, 104–106 Importantly, the APC potential of these cells has also been questioned.85, 91 IKDC were consequently redefined as B220+ NK cells and were considered as a subset of activated NK. However, a recent study has again challenged this notion, proposing that B220+NK cells, alias IKDC, may bean intermediate in NK development which therefore may more accurately be refer to as “pre-mNK”.106
B. Human KDC
Numerous independent studies have documented the cytotoxic activity of different subsets of native and in vitrogenerated human DC. Similar to the results obtained in rodents, the tumoricidal function of human KDC can be triggered by a variety of agents. The mechanisms of cancer cell killing primarily involved the death receptor ligands, perforin/granzyme B and NO. The requirement for TRAIL in the cytotoxic activity of native plasmacytoid DC (pDC) activated through TLR-7 or -9 and of blood CD11c+ DC stimulated with IFN-α or IFN-γ has been documented.50, 51, 107 Recent studies have further highlighted the possibility that human pDC producting type I interferons may also function as tumor cell killers.117 Immature CD4+HLA-DR+Lin− DC have also been shown to trigger apoptosis of different cancer cells, but not normal cells, by four different TNF family member ligands (TRAIL, TNF, FasL, and Lymphotoxin (LT)-α1β2).52, 108 Another human DC subset, termed slanDC (characterized by the 6-sulfo LacNAc modification of PSGL-1 and identified using the mAb M-DC8) have also been reported to kill different tumor cell lines by a TNF-α-dependent mechanism.65, 109 Furthermore, a plethora of studies have demonstrated that monocyte-derived DC generated ex-vivo with GM-CSF and IL-4 or IFN-α or with different cytokine combinations can kill cancer cells by TNF-α, TRAIL, FasL, peroxynitrites or granzyme B acting alone or in cooperation.52, 53, 64, 67, 71, 110, 111 In these studies, immature non-activated DC were the primary cytotoxic effectors and their killing activity was reduced or not modified following activation with CD40L, IFN-α, -β, or -γ, or LPS 52, 108, 112, 113 or was induced or enhanced with LPS,71, 114, 118 IFN-α,64 IFN-β,110 IFN-γ,114 CD40L,67, 118 or double stranded DNA.67 We have recently reported that LPS-activated DC generated from human CD14+ blood monocytes killed cancer cells by a peroxynitrite-dependent mechanism.71 Additional DC subsets, such as CD34+-derived DC,110 or cord blood monocyte-derived DC,55 were further described for their capability to eliminate cancer cells by TRAIL-dependent mechanisms following stimulation with IFN-β,110 IFN-γ or LPS.55 Of considerable importance, in the vast majority of these studies, the killing activity of DC was selectively directed against malignant cells while non-cancerous cells were spared. The mechanistic basis explaining normal cell resistance to DC killing activity has not been determined and is still being explored.
In a clinically relevant study, Stary et al. demonstrated that administration of patients with basal cell carcinoma with the TLR7/8 agonist imiquimod induced the recruitment to the tumor site of both CD123+HLA-DR+ pDC and CD11C+HLA-DR+ mDC.57 These two DC subtypes exhibited direct tumor killing activity. However, mDC were detected only at the tumor periphery and expressed perforin and granzyme B, while pDC were identified primarily within the tumor beds and expressed TRAIL.57 Further analysis demonstrated that purified mDC and pDC killed perforin-sensitive K562 and TRAIL-sensitive Jurkat cells, respectively, after stimulationwith TLR7/8 agonists.57
A substantial number of independent studies have thus provided experimental support for the existence, in vivo of different subsets of DC endowed with direct cytotoxic activities and for the possibility to generate such KDC ex vivo. However, the physiological significance of native DC cytotoxic activity as it relates to cancer development is still an open question.
III- KDC IN TUMORIGENESIS AND CANCER IMMUNITY: DR. JEKYLL OR MR HYDE?
Why highly specialized APC such as DC have acquired an effector cytotoxic activity during the process of evolution is still unclear. It has been proposed that immature non-activated KDC may participate in the mechanisms of peripheral self-tolerance by virtue of their ability to kill self-reactive T lymphocytes under specific conditions.63, 73, 119–121 In addition, as molecules associated with bacteria and viruses, such as LPS, TLR-7, TLR-9 or type I IFN can trigger their cytotoxic activity, KDC have been proposed to play a dual role in the control of infection. Indeed, they may eliminate infected cells and present the acquired antigens to specific CTL thus triggering protective immunity, but they may also mediate apoptosis of activated T cells, thereby contributing to pathogen escape from immune responses.50, 63, 67, 107, 122–124 In this section we discuss the implications of these versatile KDC in the development of tumors.
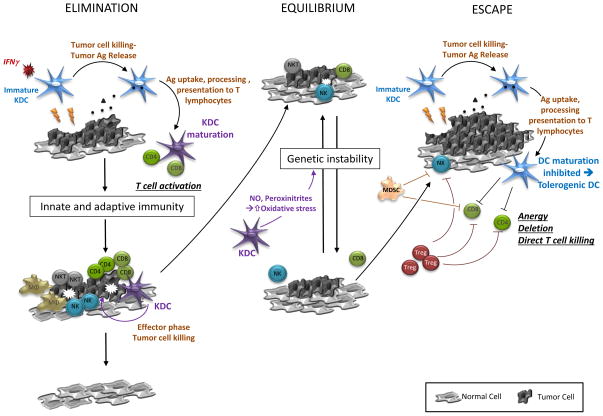
As outlined previously, DC have been integrated in the cancer immunoediting model based on their function as initiators and regulators of immune responses. However, how the killing activity of DC may fit in this immunoediting picture has yet to be formally addressed. We envision that KDC cytotoxic function may contribute to each of the 3 phases and may either foster tumor elimination or promote tumor escape depending on the activation status of the cells (Figure 1). Firstly, KDC may play an essential role during the different steps of the Elimination stage. Together with neutrophils, macrophages, NKT or γδT cells, KDC may participate in the initial killing of tumor cells, which provides a source of antigens and promotes the recruitment of additional inflammatory cells to the tumor site. It is noteworthy that IFN-γ, a primordial cytokine produced during the elimination phase, can also trigger the cytotoxic function of DC. Conceivably, the tumoricidal function of DC may thus be induced as these cells arrive in the tumor beds. Importantly, the direct killing of cancer cells by KDC allows for the rapid uptake of immediately available cancer-derived antigens in the form of debris or apoptotic bodies before their clearance by macrophages or neutrophils, thereby promoting the acquisition of tumor-derived material in a more efficient manner. Whether KDC may induce an immunogenic type of cancer cell death which may enhance antigen uptake and processing still needs to be delineated as discussed in section IV hereafter. The possible release of “danger signals” by dying tumor cells may further promote the recruitment and activation of innate immune cells and augment the production of pro-inflammatory cytokines such as IFN-γ. Following killing of cancer cells DC can switch function to their traditional role as messengers and present processed tumor-derived antigens to CD4+ or CD8+ T lymphocytes leading to the activation of adaptive immunity. Finally, KDC by participating, together with CTL, Th and NK cells, in the direct elimination of tumor cells may contribute to increase the diversity and thereby the efficacy of the immune cytotoxic effector mechanisms resulting ultimately in the elimination of neoplastic foci. The tumoricidal activity of DC may thus be of considerable significance for a successful tumor immunosurveillance process. Secondly, it is possible that during the equilibrium phase and later during the escape stage, the cytotoxic activity of DC may be subverted by cancer cells to escape immune detection and elimination. Indeed, immature KDC that have killed tumor cells, but that are blocked in their maturation by cytokines and factors present in the tumor environment may present acquired tumor antigens in absence of co-stimulation or pro-inflammatory cytokine secretion, leading to apoptosis or anergy of T lymphocytes or to the generation of immunosuppressive Treg. In this case, the tumoricidal property of DC, used to efficiently gather antigens, may promote their tolerogenic function. The possibility that KDC directly mediate killing of CD4+ or CD8+ T cells may also blunt anti-tumor responses. As the maturation status of DC dictates the outcome of the response (anti-cancer immunity versus immune suppression), it would therefore be essential to define the conditions under which tumor cell killing by KDC in vivo results in their full maturation or conversely in the inhibition of their maturation. Such conditions may be partly driven by the type of tumor cell death (immunogenic or not) inflicted by KDC and therefore may depend on the nature of the cytotoxic mechanism (TRAIL, peroxynitrites for instance) triggered in a defined tumor-infiltrating DC subset. Thirdly, in instances where KDC cytotoxic function is associated with the production of peroxynitrites, these cells may serve as a source of reactive nitrogen intermediates (RNI) and reactive oxygen species (ROS) which are known to induce DNA damage.125 It is therefore possible that KDC may enhance the genetic instability that contributes to the emergence of new tumor escape variants during the equilibrium phase. Conceptually, the direct tumor killing activity of DC may thus have important implications at all steps of cancer immunoediting and a transition of KDC from “Dr. Jekyll” to “Mr. Hyde” may occur during the equilibrium phase.
Fig. 1. The multifaceted role of KDC in cancer immunoediting.
The cytotoxic function of KDC may promote the release and subsequent acquisition of tumor-derived antigens during the first steps of the elimination process. After maturation, KDC can switch function and present processed cancer antigens to tumor-specific CD4+ or CD8+ leading to their clonal expansion and activation. KDC may also contribute to the equilibrium phase by producing RNI and ROS which may exacerbate the mutational potential of malignant cells and foster the emergence of new populations of immune escape variants. During the escape stage, immature KDC may take advantage of their killing activity to gather cancer antigens. These KDC, impaired in their maturation by the immunosuppressive environment may trigger the anergy or apoptosis of T lymphocytes or promote the generation of suppressive Treg.
A possible additional role of KDC in cancer has yet to be considered: the implication of these cells in inflammation-promoting tumorigenesis. Chronic inflammation resulting from bacterial or viral infection, exposure to pollutants or obesity has been reported to increase the risk of cancer by different mechanisms.125, 126 During many of these inflammatory conditions DC are recruited and activated. In theory, KDC endowed with the capability of producing ROS and RNI could participate in tumor initiation as these molecules can induce DNA damage and inactivate mismatch repair enzymes therefore enhancing mutagenesis and driving normal cells along the tumorigenic path.127 Although in some cases such as chronic infections, DC cytotoxic property can be triggered,50, 107, 122–124 the actual presence of these cells and the possibility that they may enhance oxidative stress needs to be determined in many chronic inflammatory conditions. In addition, whether the levels of RO and RNI produced by KDC, a relatively sparse population in vivo, are sufficientto actually impact target cellsis questionable.
The specific contribution of DC cytotoxic function in cancer immunoediting and carcinogenesis is complex and difficult to analyzeexperimentally giventhe heterogene ous nature of KDC and the diversity of their mechanisms of toxicity. Comprehensive studies are therefore still needed to address this question. One possibility to address such issues may consist in the evaluation of tumor occurrence and development in conditional knock-out animals. Specifically inhibiting the expression of different cytotoxic molecules in different DC subsets in vivo may provide important information. For example, it would be interesting to determine the susceptibility of animals with iNOS, TRAIL or Fas-L deficiency in the CD11c compartment to develop spontaneous chemically-induced tumors. Similar studies maybe performed in chronic inflammation models.
The cytotoxic function of KDC may thus contribute to the intertwined pro- and anti-tumoral roles of these multifaceted cells but more systematic studies are needed to provide definitive experimental proof of the physiological role of these cellsduring cancer development.
IV- HARNESSING THE TUMOR KILLING ACTIVITY OF KDC: PROMISES, LIMITATIONSAND CURRENT QUESTIONS
The possibility of harnessingthe ability of DC to function as professional APC capable of orchestrating adaptive and innate immunity has led to the development and implementation of DC-based vaccines in cancer immunotherapy strategies. However, the initial evidence that protective anti-tumor immunity can be successfully generated by vaccination with tumor antigen-loaded DC has been undermined by the limited clinical responses observed in cancer patients, dampening enthusiasm for this approach.128–136 The prospect of exploiting the non-conventional tumoricidal activity of these cells may therefore represent a novel step toward the development of improved DC-based cancer immunotherapies. To this end, different applications for KDC in cancer immunotherapy can be envisioned.
One approach may consist in the administration of in vitro generated KDC generated in vitrothat are allowed to kill tumor cells, capture and process the derived antigensin culture. As outlined in the previous section the mode of acquisition of cancer-derived antigens by KDC may be of particular efficacy. In line with this notion, further studies are required to determine whether KDC may promote a more immunogenic type of tumor cell death which may enhance antigen uptake, processing and presentation. It is indeed possible that DC-mediated killing of cancer cells is associated with the generation of signals expressed at the surface of dying tumor cells or secreted, such as calreticulin, HMGB1, uric acid or HSP 137–141 that may enhance DC maturation, cytokine production and ability to induce T lymphocyte activation. It will also be important to determine if KDC-mediated tumor cell death may stimulate DC endocytosis capabilities or activate the antigen presenting molecular machinery, and to define the underlying mechanisms. How conventional DC fed with dead tumor cells isolated from a KDC-cancer cell co-culture compare to the same DC loaded with apoptotic or necrotic cancer cells in term of ability to induce tumor-specific T lymphocyte activation may provide some answers to this question. Of clinical relevance, it will be importantto determine the potential advantage of using KDC (purified after killing of cancer cells in vitro) as cancer vaccines to treat tumor-bearing hosts over conventional DC (loaded with different approaches such as peptides, RNA, DNA, tumor-derived exosomes or dead tumor cells). The possibility to control the activation status of the DC in vitro using cytokines or TLR ligands represents an important advantage of this approach as it avoids the generation of immature tolerogenic DC. A second strategy may entail the systemic or intra-/peri-tumoral injection of unloaded KDC generated in vitro. Encouraging results of this approach have been reported.142 Additional approaches may consist in promoting the tumoricidal activity of DC in vivo and/or the recruitment of these KDC to the tumor beds. The study by Stary et al using the TLR-7 ligand Imiquinod has provided the framework for such therapies.57 Of importance, the efficacy of the strategies outlined above may be augmented by promoting simultaneously the tumoricidal activities of multiple KDC subsets, capable of inducing tumor cell killing by different mechanisms. Lastly, the artificial generation of killer DC by genetic manipulation (induction of TRAIL, FasL or iNOS expression in conventional DC) may also be considered. The engineered KDC may then be directly injected into the tumor beds or administered after killing of tumor cells in vitro.
From a therapeutic and clinical perspective, the primary interest in KDC relies on the multitasking aspect of these cells unifying killer and messenger functions. In the design of KDC-based therapies, the cytotoxic activity of these cells is likely to be more relevant for the acquisition of tumor antigens than for the actual direct control of tumor growth in vivo. Supporting this idea, some studies have indicated that the protective anti-tumoral effects of KDC in vivo ultimately require the induction of CD4+ and CD8+ T lymphocytes.80 Therefore, it is essential that KDC can act not only as powerful cytotoxic effectors but can switch function to efficient antigen presenting cells capable of priming T lymphocytes. This importantnotion that a same population of cells can unify killing and APC activities is still an area of contention and the true antigen-presenting ability of KDC has been intensively debated. We have demonstrated that following killing in vitro of B16 melanoma cells expressing the model antigen ovalbumin (OVA), KDC were capable of activating lymphocytes from OT-I (recognizing H-2Kb/OVA257–264 complexes) or OT-II (recognizing I-Ad/OVA323–339) mice.68, 69 Importantly, these KDC were significantly more efficient at inducing T cell activation than non-cytotoxic DC from iNOS−/− mice. (Hanke et al., submitted manuscript) and 68 Most KDC expressed both iNOS and H-2Kb/OVA257–264 suggesting that the same cells that processed OVA peptides were capable of producing the cytotoxic molecule NO and therefore were equipped with killing abilities. In addition, KDC injected into B16-OVA tumor beds and re-isolated from the draining lymph nodes were more potent at promoting OT-I and OT-II proliferation than their non-killer counterparts. (Hanke et al., submitted manuscript) and 68 These results therefore established a link between the tumoricidal activity of DC and their competence at presenting antigens from the cancer cells they have killed.
The peculiar ability of some KDC subsets to selectively kill malignant cells while sparing normal cells further reinforces the therapeutic interest in KDC-based therapy as limited side-effects associated with the administration of these cells are expected. The molecular bases explaining this relative specificity have not been delineated. KDC may express inhibitory receptors recognizing defined ligands on normal cells, which may lead to the suppression of their cytotoxic activity. Alternatively, KDC may also recognize, through specific receptors such as NKG2D, specific molecules induced at the tumor cell surface during the process of transformation and leading to the induction of their killing potential. Determining how KDC may distinguish tumor from non-malignant cells remains one of the many challenges in the KDC field.
As underlined in section I, cancer cells can alter the development of anti-tumor immunity by a plethora of mechanisms leading to the establishment of a suppressive environment. It is now well-accepted that this immuno-inhibitory tumor environment represents a major obstacle for cancer immunotherapy strategies including DC-based vaccines. A number of molecules, including TGF-β, IL-10, IL-13, VEGF, IDO, or PGE2, produced by tumor or stromal cells may exert inhibitory effects on immune system.13, 143–149 These tumor-derived factors impair DC differentiation and promote the accumulation of immature tolerogenic DC, as well as myeloid-derived suppressor cells (MDSC) 47, 147, 150–154 or CD4+CD25+FoxP3+ Treg induced by tumors.29, 155–160 Multiple studies in humans and in animal models have demonstrated that associating DC-based therapy with Treg or MDSC elimination or inactivation strategies, and more generally with approaches aimed at overcoming tumor-induced tolerance may enhance the clinical efficiency of DC based cancer vaccines.161–163 For instance chemotherapeutic drugs such as cyclophosphamide foster adoptive immunotherapy of established tumor through the elimination/inactivation of immunosuppressive Treg.156, 164, 165 We have reported that in an established lymphoma model, the efficiency of DC pulsed with total tumor cell lysates is significantly enhanced by imatinib mesylate, a chemotherapeutic drug used to treat BCR-ABL+ leukemia.160 Therefore, to reach optimal therapeutic efficacy, KDC-based vaccines may need to be combined with strategies to lessen tumor-induced tolerance.
V-SUMMARY/CONCLUSION
Significant progress in our understanding of DC immunobiology has been the driving force behind the design, optimization and implementation of these cells as vaccines in cancer immunotherapy. However, the modest clinical efficacy of this approach has eroded initial enthusiasm. Arguably, the establishment of tumor-induced immune tolerance represents one of the major challenges encountered in the field, especially in patients with terminal stage disease. The unexpected potential of DC to mediate an effector cytotoxic function has revived the attractiveness of these cells in cancer therapy. Although formal experimental evidences are still awaited, we propose to integrate KDC as a multitasking cellular actor in the cancer immunediting concept with an essential role at different stages of this process. Remaining areas of contentions still persist in the field, which warrant additional investigations. The possibility to generate and manipulate KDC in vitro may open new possibilities for the design of more potent anti-cancer vaccines, which may be exploited to improve immunotherapies for cancer patients. The future of KDC-based vaccines may likely consist in their implementation in combination regimens, such as chemoimmunotherapy, designed to concomitantly promote tumor cell killing, trigger adaptive immune responses and eliminate or avert the suppressive effects of tumor-associated factors or immune inhibitory cells.
Acknowledgments
The authors are supported in part by the NIH grant R01 CA104926, the Cancer Biology Training Grant T32CA009213 (DA), the AZ Cancer Center Support Grant CA023074, the Tee Up for Tots and PANDA Funds.
ABBREVIATIONS
- APC
antigen presenting cells
- CTL
cytotoxic T lymphocytes
- DC
dendritic cells
- FasL
fas-ligand
- γδT
gamma/delta T cells
- GM-CSF
granulocyte macrophage colony stimulating factor
- HMGB1
high mobility group box -1
- HSP
heat shock proteins
- IFN-γ
interferon-gamma
- iNOS
inducible nitric oxide synthase
- ISGF3
interferon-stimulated gene factor 3
- KDC
killer dendritic cells
- LPS
lipopolysaccharide
- mDC
myeloid DC
- MDSC
myeloid-derived-suppressor cells
- MHC
major histocompatibility complex
- NF-κB
nuclear factor κB
- NK
natural killer cells
- NKR-P1
natural killer cell receptor protein 1
- NKT
natural killer T cells
- NO
nitric oxide
- OVA
ovalbumin
- pDC
plasmacytoid DC
- RNI
reactive nitrogen intermediates
- ROS
reactive oxygen species
- STAT
signal transducer and activator of transcription
- Th
T helper lymphocyte
- TLR-
toll-like receptor
- Treg
regulatory T lymphocytes
- TNF-α
tumor necrosis factor alpha
- TRAIL
TNF-related apoptosis-inducing ligand
References
- 1.Steinman RM, Cohn ZA. Identification of a novel cell type in peripheral lymphoid organs of mice. I. Morphology, quantitation, tissue distribution. J Exp Med. 1973;137(5):1142–62. doi: 10.1084/jem.137.5.1142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Banchereau J, Steinman RM. Dendritic cells and the control of immunity. Nature. 1998;392(6673):245–52. doi: 10.1038/32588. [DOI] [PubMed] [Google Scholar]
- 3.Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, Pulendran B, Palucka K. Immunobiology of dendritic cells. Annu Rev Immunol. 2000;18:767–811. doi: 10.1146/annurev.immunol.18.1.767. [DOI] [PubMed] [Google Scholar]
- 4.Gilboa E, Nair SK, Lyerly HK. Immunotherapy of cancer with dendritic-cell-based vaccines. Cancer Immunol Immunother. 1998;46(2):82–7. doi: 10.1007/s002620050465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nestle FO, Farkas A, Conrad C. Dendritic-cell-based therapeutic vaccination against cancer. Curr Opin Immunol. 2005;17(2):163–9. doi: 10.1016/j.coi.2005.02.003. [DOI] [PubMed] [Google Scholar]
- 6.Palucka AK, Laupeze B, Aspord C, Saito H, Jego G, Fay J, Paczesny S, Pascual V, Banchereau J. Immunotherapy via dendritic cells. Adv Exp Med Biol. 2005;560:105–14. doi: 10.1007/0-387-24180-9_14. [DOI] [PubMed] [Google Scholar]
- 7.Mayordomo JI, Zorina T, Storkus WJ, Zitvogel L, Celluzzi C, Falo LD, Melief CJ, Ildstad ST, Kast WM, Deleo AB, et al. Bone marrow-derived dendritic cells pulsed with synthetic tumour peptides elicit protective and therapeutic antitumour immunity. Nat Med. 1995;1(12):1297–302. doi: 10.1038/nm1295-1297. [DOI] [PubMed] [Google Scholar]
- 8.Nagaraj S, Ziske C, Strehl J, Messmer D, Sauerbruch T, Schmidt-Wolf IG. Dendritic cells pulsed with alpha-galactosylceramide induce anti-tumor immunity against pancreatic cancer in vivo. Int Immunol. 2006;18(8):1279–83. doi: 10.1093/intimm/dxl059. [DOI] [PubMed] [Google Scholar]
- 9.Shimizu J, Suda T, Yoshioka T, Kosugi A, Fujiwara H, Hamaoka T. Induction of tumor-specific in vivo protective immunity by immunization with tumor antigen-pulsed antigen-presenting cells. J Immunol. 1989;142(3):1053–9. [PubMed] [Google Scholar]
- 10.Paczesny S, Ueno H, Fay J, Banchereau J, Palucka AK. Dendritic cells as vectors for immunotherapy of cancer. Semin Cancer Biol. 2003;13(6):439–47. doi: 10.1016/j.semcancer.2003.09.008. [DOI] [PubMed] [Google Scholar]
- 11.Figdor CG, de Vries IJ, Lesterhuis WJ, Melief CJ. Dendritic cell immunotherapy: mapping the way. Nat Med. 2004;10(5):475–80. doi: 10.1038/nm1039. [DOI] [PubMed] [Google Scholar]
- 12.Steinman RM, Banchereau J. Taking dendritic cells into medicine. Nature. 2007;449(7161):419–26. doi: 10.1038/nature06175. [DOI] [PubMed] [Google Scholar]
- 13.Ueno H, Klechevsky E, Morita R, Aspord C, Cao T, Matsui T, Di Pucchio T, Connolly J, Fay JW, Pascual V, Palucka AK, Banchereau J. Dendritic cell subsets in health and disease. Immunol Rev. 2007;219:118–42. doi: 10.1111/j.1600-065X.2007.00551.x. [DOI] [PubMed] [Google Scholar]
- 14.Dubsky P, Ueno H, Piqueras B, Connolly J, Banchereau J, Palucka AK. Human dendritic cell subsets for vaccination. J Clin Immunol. 2005;25(6):551–72. doi: 10.1007/s10875-005-8216-7. [DOI] [PubMed] [Google Scholar]
- 15.Pulendran B, Smith JL, Caspary G, Brasel K, Pettit D, Maraskovsky E, Maliszewski CR. Distinct dendritic cell subsets differentially regulate the class of immune response in vivo. Proc Natl Acad Sci U S A. 1999;96(3):1036–41. doi: 10.1073/pnas.96.3.1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kimura A, Naka T, Kishimoto T. IL-6-dependent and -independent pathways in the development of interleukin 17-producing T helper cells. Proc Natl Acad Sci U S A. 2007;104(29):12099–104. doi: 10.1073/pnas.0705268104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jego G, Palucka AK, Blanck JP, Chalouni C, Pascual V, Banchereau J. Plasmacytoid dendritic cells induce plasma cell differentiation through type I interferon and interleukin 6. Immunity. 2003;19(2):225–34. doi: 10.1016/s1074-7613(03)00208-5. [DOI] [PubMed] [Google Scholar]
- 18.Batista FD, Harwood NE. The who, how and where of antigen presentation to B cells. Nat Rev Immunol. 2009;9(1):15–27. doi: 10.1038/nri2454. [DOI] [PubMed] [Google Scholar]
- 19.Walzer T, Dalod M, Robbins SH, Zitvogel L, Vivier E. Natural-killer cells and dendritic cells: “l’union fait la force”. Blood. 2005;106(7):2252–8. doi: 10.1182/blood-2005-03-1154. [DOI] [PubMed] [Google Scholar]
- 20.Fujii S, Shimizu K, Hemmi H, Steinman RM. Innate Valpha14(+) natural killer T cells mature dendritic cells, leading to strong adaptive immunity. Immunol Rev. 2007;220:183–98. doi: 10.1111/j.1600-065X.2007.00561.x. [DOI] [PubMed] [Google Scholar]
- 21.Steinman RM, Hawiger D, Liu K, Bonifaz L, Bonnyay D, Mahnke K, Iyoda T, Ravetch J, Dhodapkar M, Inaba K, Nussenzweig M. Dendritic cell function in vivo during the steady state: a role in peripheral tolerance. Ann N Y Acad Sci. 2003;987:15–25. doi: 10.1111/j.1749-6632.2003.tb06029.x. [DOI] [PubMed] [Google Scholar]
- 22.Tuettenberg A, Huter E, Hubo M, Horn J, Knop J, Grimbacher B, Kroczek RA, Stoll S, Jonuleit H. The role of ICOS in directing T cell responses: ICOS-dependent induction of T cell anergy by tolerogenic dendritic cells. J Immunol. 2009;182(6):3349–56. doi: 10.4049/jimmunol.0802733. [DOI] [PubMed] [Google Scholar]
- 23.Rutella S, Danese S, Leone G. Tolerogenic dendritic cells: cytokine modulation comes of age. Blood. 2006;108(5):1435–40. doi: 10.1182/blood-2006-03-006403. [DOI] [PubMed] [Google Scholar]
- 24.Banerjee DK, Dhodapkar MV, Matayeva E, Steinman RM, Dhodapkar KM. Expansion of FOXP3high regulatory T cells by human dendritic cells (DCs) in vitro and after injection of cytokine-matured DCs in myeloma patients. Blood. 2006;108(8):2655–61. doi: 10.1182/blood-2006-03-011353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Belkaid Y, Oldenhove G. Tuning microenvironments: induction of regulatory T cells by dendritic cells. Immunity. 2008;29(3):362–71. doi: 10.1016/j.immuni.2008.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roncarolo MG, Gregori S, Battaglia M, Bacchetta R, Fleischhauer K, Levings MK. Interleukin-10-secreting type 1 regulatory T cells in rodents and humans. Immunol Rev. 2006;212:28–50. doi: 10.1111/j.0105-2896.2006.00420.x. [DOI] [PubMed] [Google Scholar]
- 27.Hubert P, Jacobs N, Caberg JH, Boniver J, Delvenne P. The cross-talk between dendritic and regulatory T cells: good or evil? J Leukoc Biol. 2007;82(4):781–94. doi: 10.1189/jlb.1106694. [DOI] [PubMed] [Google Scholar]
- 28.Jonuleit H, Schmitt E, Schuler G, Knop J, Enk AH. Induction of interleukin 10-producing, nonproliferating CD4(+) T cells with regulatory properties by repetitive stimulation with allogeneic immature human dendritic cells. J Exp Med. 2000;192(9):1213–22. doi: 10.1084/jem.192.9.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Janikashvili N, Bonnotte B, Katsanis E, Larmonier N. The dendritic cell-regulatory T lymphocyte crosstalk contributes to tumor-induced tolerance. Clinical & developmental immunology. 2011;2011:430394. doi: 10.1155/2011/430394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kushwah R, Wu J, Oliver JR, Jiang G, Zhang J, Siminovitch KA, Hu J. Uptake of apoptotic DC converts immature DC into tolerogenic DC that induce differentiation of Foxp3+ Treg. Eur J Immunol. 2010;40(4):1022–35. doi: 10.1002/eji.200939782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Burnet FM. The concept of immunological surveillance. Progress in experimental tumor research. 1970;13:1–27. doi: 10.1159/000386035. [DOI] [PubMed] [Google Scholar]
- 32.Dunn GP, Old LJ, Schreiber RD. The three Es of cancer immunoediting. Annu Rev Immunol. 2004;22:329–60. doi: 10.1146/annurev.immunol.22.012703.104803. [DOI] [PubMed] [Google Scholar]
- 33.Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD. Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol. 2002;3(11):991–8. doi: 10.1038/ni1102-991. [DOI] [PubMed] [Google Scholar]
- 34.Dunn GP, Old LJ, Schreiber RD. The immunobiology of cancer immunosurveillance and immunoediting. Immunity. 2004;21(2):137–48. doi: 10.1016/j.immuni.2004.07.017. [DOI] [PubMed] [Google Scholar]
- 35.Pure E, Allison JP, Schreiber RD. Breaking down the barriers to cancer immunotherapy. Nat Immunol. 2005;6(12):1207–10. doi: 10.1038/ni1205-1207. [DOI] [PubMed] [Google Scholar]
- 36.Bui JD, Schreiber RD. Cancer immunosurveillance, immunoediting and inflammation: independent or interdependent processes? Curr Opin Immunol. 2007;19(2):203–8. doi: 10.1016/j.coi.2007.02.001. [DOI] [PubMed] [Google Scholar]
- 37.Dunn GP, Bruce AT, Sheehan KC, Shankaran V, Uppaluri R, Bui JD, Diamond MS, Koebel CM, Arthur C, White JM, Schreiber RD. A critical function for type I interferons in cancer immunoediting. Nat Immunol. 2005;6(7):722–9. doi: 10.1038/ni1213. [DOI] [PubMed] [Google Scholar]
- 38.Dunn GP, Ikeda H, Bruce AT, Koebel C, Uppaluri R, Bui J, Chan R, Diamond M, White JM, Sheehan KC, Schreiber RD. Interferon-gamma and cancer immunoediting. Immunol Res. 2005;32(1–3):231–45. doi: 10.1385/ir:32:1-3:231. [DOI] [PubMed] [Google Scholar]
- 39.Zitvogel L, Tesniere A, Kroemer G. Cancer despite immunosurveillance: immunoselection and immunosubversion. Nat Rev Immunol. 2006;6(10):715–27. doi: 10.1038/nri1936. [DOI] [PubMed] [Google Scholar]
- 40.Staveley-O’Carroll K, Sotomayor E, Montgomery J, Borrello I, Hwang L, Fein S, Pardoll D, Levitsky H. Induction of antigen-specific T cell anergy: An early event in the course of tumor progression. Proc Natl Acad Sci U S A. 1998;95(3):1178–83. doi: 10.1073/pnas.95.3.1178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Smyth MJ, Godfrey DI, Trapani JA. A fresh look at tumor immunosurveillance and immunotherapy. Nat Immunol. 2001;2(4):293–9. doi: 10.1038/86297. [DOI] [PubMed] [Google Scholar]
- 42.Feinberg MB, Silvestri G. T(S) cells and immune tolerance induction: a regulatory renaissance? Nat Immunol. 2002;3(3):215–7. doi: 10.1038/ni0302-215. [DOI] [PubMed] [Google Scholar]
- 43.Pardoll D. Does the immune system see tumors as foreign or self? Annu Rev Immunol. 2003;21:807–39. doi: 10.1146/annurev.immunol.21.120601.141135. [DOI] [PubMed] [Google Scholar]
- 44.Khong HT, Restifo NP. Natural selection of tumor variants in the generation of “tumor escape” phenotypes. Nat Immunol. 2002;3(11):999–1005. doi: 10.1038/ni1102-999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lu B, Finn OJ. T-cell death and cancer immune tolerance. Cell Death Differ. 2008;15(1):70–9. doi: 10.1038/sj.cdd.4402274. [DOI] [PubMed] [Google Scholar]
- 46.Vicari AP, Caux C, Trinchieri G. Tumour escape from immune surveillance through dendritic cell inactivation. Semin Cancer Biol. 2002;12(1):33–42. doi: 10.1006/scbi.2001.0400. [DOI] [PubMed] [Google Scholar]
- 47.Rabinovich GA, Gabrilovich D, Sotomayor EM. Immunosuppressive strategies that are mediated by tumor cells. Annu Rev Immunol. 2007;25:267–96. doi: 10.1146/annurev.immunol.25.022106.141609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Josien R, Heslan M, Soulillou JP, Cuturi MC. Rat spleen dendritic cells express natural killer cell receptor protein 1 (NKR-P1) and have cytotoxic activity to select targets via a Ca2+-dependent mechanism. J Exp Med. 1997;186(3):467–72. doi: 10.1084/jem.186.3.467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chan CW, Crafton E, Fan HN, Flook J, Yoshimura K, Skarica M, Brockstedt D, Dubensky TW, Stins MF, Lanier LL, Pardoll DM, Housseau F. Interferon-producing killer dendritic cells provide a link between innate and adaptive immunity. Nat Med. 2006;12(2):207–13. doi: 10.1038/nm1352. [DOI] [PubMed] [Google Scholar]
- 50.Chaperot L, Blum A, Manches O, Lui G, Angel J, Molens JP, Plumas J. Virus or TLR agonists induce TRAIL-mediated cytotoxic activity of plasmacytoid dendritic cells. J Immunol. 2006;176(1):248–55. doi: 10.4049/jimmunol.176.1.248. [DOI] [PubMed] [Google Scholar]
- 51.Fanger NA, Maliszewski CR, Schooley K, Griffith TS. Human dendritic cells mediate cellular apoptosis via tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) J Exp Med. 1999;190(8):1155–64. doi: 10.1084/jem.190.8.1155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lu G, Janjic BM, Janjic J, Whiteside TL, Storkus WJ, Vujanovic NL. Innate direct anticancer effector function of human immature dendritic cells. II. Role of TNF, lymphotoxin-alpha(1)beta(2), Fas ligand, and TNF-related apoptosis-inducing ligand. J Immunol. 2002;168(4):1831–9. doi: 10.4049/jimmunol.168.4.1831. [DOI] [PubMed] [Google Scholar]
- 53.Manna PP, Mohanakumar T. Human dendritic cell mediated cytotoxicity against breast carcinoma cells in vitro. J Leukoc Biol. 2002;72(2):312–20. [PubMed] [Google Scholar]
- 54.Nicolas A, Cathelin D, Larmonier N, Fraszczak J, Puig PE, Bouchot A, Bateman A, Solary E, Bonnotte B. Dendritic cells trigger tumor cell death by a nitric oxide-dependent mechanism. J Immunol. 2007;179(2):812–8. doi: 10.4049/jimmunol.179.2.812. [DOI] [PubMed] [Google Scholar]
- 55.Shi J, Ikeda K, Fujii N, Kondo E, Shinagawa K, Ishimaru F, Kaneda K, Tanimoto M, Li X, Pu Q. Activated human umbilical cord blood dendritic cells kill tumor cells without damaging normal hematological progenitor cells. Cancer Sci. 2005;96(2):127–33. doi: 10.1111/j.1349-7006.2005.00017.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Shimamura H, Cumberland R, Hiroishi K, Watkins SC, Lotze MT, Baar J. Murine dendritic cell-induced tumor apoptosis is partially mediated by nitric oxide. J Immunother. 2002;25(3):226–34. doi: 10.1097/00002371-200205000-00005. [DOI] [PubMed] [Google Scholar]
- 57.Stary G, Bangert C, Tauber M, Strohal R, Kopp T, Stingl G. Tumoricidal activity of TLR7/8-activated inflammatory dendritic cells. J Exp Med. 2007;204(6):1441–51. doi: 10.1084/jem.20070021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Trinite B, Chauvin C, Peche H, Voisine C, Heslan M, Josien R. Immature CD4- CD103+ rat dendritic cells induce rapid caspase-independent apoptosis-like cell death in various tumor and nontumor cells and phagocytose their victims. J Immunol. 2005;175(4):2408–17. doi: 10.4049/jimmunol.175.4.2408. [DOI] [PubMed] [Google Scholar]
- 59.Bonmort M, Dalod M, Mignot G, Ullrich E, Chaput N, Zitvogel L. Killer dendritic cells: IKDC and the others. Curr Opin Immunol. 2008;20(5):558–65. doi: 10.1016/j.coi.2008.04.006. [DOI] [PubMed] [Google Scholar]
- 60.Ullrich E, Chaput N, Zitvogel L. Killer dendritic cells and their potential role in immunotherapy. Hormone and metabolic research = Hormon- und Stoffwechselforschung = Hormones et metabolisme. 2008;40(2):75–81. doi: 10.1055/s-2007-1022554. [DOI] [PubMed] [Google Scholar]
- 61.Wesa AK, Storkus WJ. Killer dendritic cells: mechanisms of action and therapeutic implications for cancer. Cell Death Differ. 2008;15(1):51–7. doi: 10.1038/sj.cdd.4402243. [DOI] [PubMed] [Google Scholar]
- 62.Chan CW, Housseau F. The ‘kiss of death’ by dendritic cells to cancer cells. Cell Death Differ. 2008;15(1):58–69. doi: 10.1038/sj.cdd.4402235. [DOI] [PubMed] [Google Scholar]
- 63.Chauvin C, Josien R. Dendritic cells as killers: mechanistic aspects and potential roles. J Immunol. 2008;181(1):11–6. doi: 10.4049/jimmunol.181.1.11. [DOI] [PubMed] [Google Scholar]
- 64.Griffith TS, Wiley SR, Kubin MZ, Sedger LM, Maliszewski CR, Fanger NA. Monocyte-mediated tumoricidal activity via the tumor necrosis factor-related cytokine, TRAIL. J Exp Med. 1999;189(8):1343–54. doi: 10.1084/jem.189.8.1343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Schmitz M, Zhao S, Deuse Y, Schakel K, Wehner R, Wohner H, Holig K, Wienforth F, Kiessling A, Bornhauser M, Temme A, Rieger MA, Weigle B, Bachmann M, Rieber EP. Tumoricidal potential of native blood dendritic cells: direct tumor cell killing and activation of NK cell-mediated cytotoxicity. J Immunol. 2005;174(7):4127–34. doi: 10.4049/jimmunol.174.7.4127. [DOI] [PubMed] [Google Scholar]
- 66.Yang R, Xu D, Zhang A, Gruber A. Immature dendritic cells kill ovarian carcinoma cells by a FAS/FASL pathway, enabling them to sensitize tumor-specific CTLs. Int J Cancer. 2001;94(3):407–13. doi: 10.1002/ijc.1484. [DOI] [PubMed] [Google Scholar]
- 67.Vidalain PO, Azocar O, Yagita H, Rabourdin-Combe C, Servet-Delprat C. Cytotoxic activity of human dendritic cells is differentially regulated by double-stranded RNA and CD40 ligand. J Immunol. 2001;167(7):3765–72. doi: 10.4049/jimmunol.167.7.3765. [DOI] [PubMed] [Google Scholar]
- 68.LaCasse CJ, Janikashvili N, Larmonier CB, Alizadeh D, Hanke N, Kartchner J, Situ E, Centuori S, Har-Noy M, Bonnotte B, Katsanis E, Larmonier N. Th-1 lymphocytes induce dendritic cell tumor killing activity by an IFN-gamma-dependent mechanism. J Immunol. 2011;187(12):6310–7. doi: 10.4049/jimmunol.1101812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Fraszczak J, Trad M, Janikashvili N, Cathelin D, Lakomy D, Granci V, Morizot A, Audia S, Micheau O, Lagrost L, Katsanis E, Solary E, Larmonier N, Bonnotte B. Peroxynitrite-dependent killing of cancer cells and presentation of released tumor antigens by activated dendritic cells. J Immunol. 2010;184(4):1876–84. doi: 10.4049/jimmunol.0900831. [DOI] [PubMed] [Google Scholar]
- 70.Larmonier N, Fraszczak J, Lakomy D, Bonnotte B, Katsanis E. Killer dendritic cells and their potential for cancer immunotherapy. Cancer Immunol Immunother. 2010;59(1):1–11. doi: 10.1007/s00262-009-0736-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lakomy D, Janikashvili N, Fraszczak J, Trad M, Audia S, Samson M, Ciudad M, Vinit J, Vergely C, Caillot D, Foucher P, Lagrost L, Chouaib S, Katsanis E, Larmonier N, Bonnotte B. Cytotoxic dendritic cells generated from cancer patients. J Immunol. 2011;187(5):2775–82. doi: 10.4049/jimmunol.1004146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Larmonier N, Bonnotte B, Katsanis E. Cytotoxic and antigen presenting functions of T helper-1-activated dendritic cells. Oncoimmunology. 2012;1(4):566–8. doi: 10.4161/onci.19370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Suss G, Shortman K. A subclass of dendritic cells kills CD4 T cells via Fas/Fas-ligand-induced apoptosis. J Exp Med. 1996;183(4):1789–96. doi: 10.1084/jem.183.4.1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Shibaki A, Katz SI. Activation through CD40 ligation induces functional Fas ligand expression by Langerhans cells. Eur J Immunol. 2001;31(10):3006–15. doi: 10.1002/1521-4141(2001010)31:10<3006::aid-immu3006>3.0.co;2-l. [DOI] [PubMed] [Google Scholar]
- 75.Serbina NV, Salazar-Mather TP, Biron CA, Kuziel WA, Pamer EG. TNF/iNOS-producing dendritic cells mediate innate immune defense against bacterial infection. Immunity. 2003;19(1):59–70. doi: 10.1016/s1074-7613(03)00171-7. [DOI] [PubMed] [Google Scholar]
- 76.Chauvin C, Philippeau JM, Hemont C, Hubert FX, Wittrant Y, Lamoureux F, Trinite B, Heymann D, Redini F, Josien R. Killer dendritic cells link innate and adaptive immunity against established osteosarcoma in rats. Cancer Res. 2008;68(22):9433–40. doi: 10.1158/0008-5472.CAN-08-0104. [DOI] [PubMed] [Google Scholar]
- 77.Trinite B, Voisine C, Yagita H, Josien R. A subset of cytolytic dendritic cells in rat. J Immunol. 2000;165(8):4202–8. doi: 10.4049/jimmunol.165.8.4202. [DOI] [PubMed] [Google Scholar]
- 78.Lu L, Qian S, Hershberger PA, Rudert WA, Lynch DH, Thomson AW. Fas ligand (CD95L) and B7 expression on dendritic cells provide counter-regulatory signals for T cell survival and proliferation. J Immunol. 1997;158(12):5676–84. [PubMed] [Google Scholar]
- 79.Huang J, Tatsumi T, Pizzoferrato E, Vujanovic N, Storkus WJ. Nitric oxide sensitizes tumor cells to dendritic cell-mediated apoptosis, uptake, and cross-presentation. Cancer Res. 2005;65(18):8461–70. doi: 10.1158/0008-5472.CAN-05-0654. [DOI] [PubMed] [Google Scholar]
- 80.Tatsumi T, Huang J, Gooding WE, Gambotto A, Robbins PD, Vujanovic NL, Alber SM, Watkins SC, Okada H, Storkus WJ. Intratumoral delivery of dendritic cells engineered to secrete both interleukin (IL)-12 and IL-18 effectively treats local and distant disease in association with broadly reactive Tc1-type immunity. Cancer Res. 2003;63(19):6378–86. [PubMed] [Google Scholar]
- 81.Yu Y, Liu S, Wang W, Song W, Zhang M, Zhang W, Qin Z, Cao X. Involvement of tumour necrosis factor-alpha-related apoptosis-inducing ligand in enhanced cytotoxicity of lipopolysaccharide-stimulated dendritic cells to activated T cells. Immunology. 2002;106(3):308–15. doi: 10.1046/j.1365-2567.2002.01431.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lu L, Qian S, Starzl TE, Lynch DH, Thomson AW. Blocking of the B7-CD28 pathway increases the capacity of FasL+ (CD95L+) dendritic cells to kill alloactivated T cells. Adv Exp Med Biol. 1997;417:275–82. doi: 10.1007/978-1-4757-9966-8_45. [DOI] [PubMed] [Google Scholar]
- 83.Srivastava RM, Varalakshmi C, Khar A. Cross-linking a mAb to NKR-P2/NKG2D on dendritic cells induces their activation and maturation leading to enhanced anti-tumor immune response. Int Immunol. 2007;19(5):591–607. doi: 10.1093/intimm/dxm024. [DOI] [PubMed] [Google Scholar]
- 84.Alli R, Savithri B, Das S, Varalakshmi C, Rangaraj N, Khar A. Involvement of NKR-P2/NKG2D in DC-mediated killing of tumor targets: indicative of a common, innate, target-recognition paradigm? Eur J Immunol. 2004;34(4):1119–26. doi: 10.1002/eji.200324793. [DOI] [PubMed] [Google Scholar]
- 85.Caminschi I, Ahmet F, Heger K, Brady J, Nutt SL, Vremec D, Pietersz S, Lahoud MH, Schofield L, Hansen DS, O’Keeffe M, Smyth MJ, Bedoui S, Davey GM, Villadangos JA, Heath WR, Shortman K. Putative IKDCs are functionally and developmentally similar to natural killer cells, but not to dendritic cells. J Exp Med. 2007;204(11):2579–90. doi: 10.1084/jem.20071351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Shortman K, Villadangos JA. Is it a DC, is it an NK? No, it’s an IKDC. Nat Med. 2006;12(2):167–8. doi: 10.1038/nm0206-167. [DOI] [PubMed] [Google Scholar]
- 87.Vremec D, O’Keeffe M, Hochrein H, Fuchsberger M, Caminschi I, Lahoud M, Shortman K. Production of interferons by dendritic cells, plasmacytoid cells, natural killer cells, and interferon-producing killer dendritic cells. Blood. 2007;109(3):1165–73. doi: 10.1182/blood-2006-05-015354. [DOI] [PubMed] [Google Scholar]
- 88.Homann D, Jahreis A, Wolfe T, Hughes A, Coon B, van Stipdonk MJ, Prilliman KR, Schoenberger SP, von Herrath MG. CD40L blockade prevents autoimmune diabetes by induction of bitypic NK/DC regulatory cells. Immunity. 2002;16(3):403–15. doi: 10.1016/s1074-7613(02)00290-x. [DOI] [PubMed] [Google Scholar]
- 89.Mignot G, Ullrich E, Bonmort M, Menard C, Apetoh L, Taieb J, Bosisio D, Sozzani S, Ferrantini M, Schmitz J, Mack M, Ryffel B, Bulfone-Paus S, Zitvogel L, Chaput N. The critical role of IL-15 in the antitumor effects mediated by the combination therapy imatinib and IL-2. J Immunol. 2008;180(10):6477–83. doi: 10.4049/jimmunol.180.10.6477. [DOI] [PubMed] [Google Scholar]
- 90.Taieb J, Chaput N, Menard C, Apetoh L, Ullrich E, Bonmort M, Pequignot M, Casares N, Terme M, Flament C, Opolon P, Lecluse Y, Metivier D, Tomasello E, Vivier E, Ghiringhelli F, Martin F, Klatzmann D, Poynard T, Tursz T, Raposo G, Yagita H, Ryffel B, Kroemer G, Zitvogel L. A novel dendritic cell subset involved in tumor immunosurveillance. Nat Med. 2006;12(2):214–9. doi: 10.1038/nm1356. [DOI] [PubMed] [Google Scholar]
- 91.Vosshenrich CA, Lesjean-Pottier S, Hasan M, Richard-Le Goff O, Corcuff E, Mandelboim O, Di Santo JP. CD11cloB220+ interferon-producing killer dendritic cells are activated natural killer cells. J Exp Med. 2007;204(11):2569–78. doi: 10.1084/jem.20071451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Guimont-Desrochers F, Cappello ZJ, Chagnon M, McDuffie M, Lesage S. Cutting edge: genetic characterization of IFN-producing killer dendritic cells. J Immunol. 2009;182(9):5193–7. doi: 10.4049/jimmunol.0803969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Pillarisetty VG, Katz SC, Bleier JI, Shah AB, Dematteo RP. Natural killer dendritic cells have both antigen presenting and lytic function and in response to CpG produce IFN-gamma via autocrine IL-12. J Immunol. 2005;174(5):2612–8. doi: 10.4049/jimmunol.174.5.2612. [DOI] [PubMed] [Google Scholar]
- 94.Chaudhry UI, Kingham TP, Plitas G, Katz SC, Raab JR, DeMatteo RP. Combined stimulation with interleukin-18 and CpG induces murine natural killer dendritic cells to produce IFN-gamma and inhibit tumor growth. Cancer Res. 2006;66(21):10497–504. doi: 10.1158/0008-5472.CAN-06-1908. [DOI] [PubMed] [Google Scholar]
- 95.Chaudhry UI, Katz SC, Kingham TP, Pillarisetty VG, Raab JR, Shah AB, DeMatteo RP. In vivo overexpression of Flt3 ligand expands and activates murine spleen natural killer dendritic cells. Faseb J. 2006;20(7):982–4. doi: 10.1096/fj.05-5411fje. [DOI] [PubMed] [Google Scholar]
- 96.Chaudhry UI, Plitas G, Burt BM, Kingham TP, Raab JR, DeMatteo RP. NK dendritic cells expanded in IL-15 exhibit antitumor responses in vivo. J Immunol. 2007;179(7):4654–60. doi: 10.4049/jimmunol.179.7.4654. [DOI] [PubMed] [Google Scholar]
- 97.Plitas G, Chaudhry UI, Kingham TP, Raab JR, DeMatteo RP. NK dendritic cells are innate immune responders to Listeria monocytogenes infection. J Immunol. 2007;178(7):4411–6. doi: 10.4049/jimmunol.178.7.4411. [DOI] [PubMed] [Google Scholar]
- 98.Ullrich E, Bonmort M, Mignot G, Jacobs B, Bosisio D, Sozzani S, Jalil A, Louache F, Bulanova E, Geissman F, Ryffel B, Chaput N, Bulfone-Paus S, Zitvogel L. Trans-presentation of IL-15 dictates IFN-producing killer dendritic cells effector functions. J Immunol. 2008;180(12):7887–97. doi: 10.4049/jimmunol.180.12.7887. [DOI] [PubMed] [Google Scholar]
- 99.Zitvogel L, Mignot G, Bonmort M, Ullrich E, Chaput N. IKDC: killer dendritic cells or antigen-presenting NK cells? Medecine sciences: M/S. 2008;24(5):525–8. doi: 10.1051/medsci/2008245525. [DOI] [PubMed] [Google Scholar]
- 100.Bonmort M, Ullrich E, Mignot G, Jacobs B, Chaput N, Zitvogel L. Interferon-gamma is produced by another player of innate immune responses: the interferon-producing killer dendritic cell (IKDC) Biochimie. 2007;89(6–7):872–7. doi: 10.1016/j.biochi.2007.04.014. [DOI] [PubMed] [Google Scholar]
- 101.Ullrich E, Bonmort M, Mignot G, Chaput N, Taieb J, Menard C, Viaud S, Tursz T, Kroemer G, Zitvogel L. Therapy-induced tumor immunosurveillance involves IFN-producing killer dendritic cells. Cancer Res. 2007;67(3):851–3. doi: 10.1158/0008-5472.CAN-06-3766. [DOI] [PubMed] [Google Scholar]
- 102.Himoudi N, Nabarro S, Buddle J, Eddaoudi A, Thrasher AJ, Anderson J. Bone marrow-derived IFN-producing killer dendritic cells account for the tumoricidal activity of unpulsed dendritic cells. J Immunol. 2008;181(9):6654–63. doi: 10.4049/jimmunol.181.9.6654. [DOI] [PubMed] [Google Scholar]
- 103.Jiang Q, Wei H, Tian Z. IFN-producing killer dendritic cells contribute to the inhibitory effect of poly I:C on the progression of murine melanoma. J Immunother. 2008;31(6):555–62. doi: 10.1097/CJI.0b013e31817d8e75. [DOI] [PubMed] [Google Scholar]
- 104.Blasius AL, Barchet W, Cella M, Colonna M. Development and function of murine B220+CD11c+NK1.1+ cells identify them as a subset of NK cells. J Exp Med. 2007;204(11):2561–8. doi: 10.1084/jem.20070991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zitvogel L, Housseau F. IKDCs or B220+ NK cells are pre-mNK cells. Blood. 2012;119(19):4345–6. doi: 10.1182/blood-2012-03-415026. [DOI] [PubMed] [Google Scholar]
- 106.Guimont-Desrochers F, Boucher G, Dong Z, Dupuis M, Veillette A, Lesage S. Redefining interferon-producing killer dendritic cells as a novel intermediate in NK-cell differentiation. Blood. 2012;119(19):4349–57. doi: 10.1182/blood-2011-11-395954. [DOI] [PubMed] [Google Scholar]
- 107.Hardy AW, Graham DR, Shearer GM, Herbeuval JP. HIV turns plasmacytoid dendritic cells (pDC) into TRAIL-expressing killer pDC and down-regulates HIV coreceptors by Toll-like receptor 7-induced IFN-alpha. Proc Natl Acad Sci U S A. 2007;104(44):17453–8. doi: 10.1073/pnas.0707244104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Janjic BM, Lu G, Pimenov A, Whiteside TL, Storkus WJ, Vujanovic NL. Innate direct anticancer effector function of human immature dendritic cells. I. Involvement of an apoptosis-inducing pathway. J Immunol. 2002;168(4):1823–30. doi: 10.4049/jimmunol.168.4.1823. [DOI] [PubMed] [Google Scholar]
- 109.Schakel K, von Kietzell M, Hansel A, Ebling A, Schulze L, Haase M, Semmler C, Sarfati M, Barclay AN, Randolph GJ, Meurer M, Rieber EP. Human 6-sulfo LacNAc-expressing dendritic cells are principal producers of early interleukin-12 and are controlled by erythrocytes. Immunity. 2006;24(6):767–77. doi: 10.1016/j.immuni.2006.03.020. [DOI] [PubMed] [Google Scholar]
- 110.Liu S, Yu Y, Zhang M, Wang W, Cao X. The involvement of TNF-alpha-related apoptosis-inducing ligand in the enhanced cytotoxicity of IFN-beta-stimulated human dendritic cells totumor cells. J Immunol. 2001;166(9):5407–15. doi: 10.4049/jimmunol.166.9.5407. [DOI] [PubMed] [Google Scholar]
- 111.Korthals M, Safaian N, Kronenwett R, Maihofer D, Schott M, Papewalis C, Diaz Blanco E, Winter M, Czibere A, Haas R, Kobbe G, Fenk R. Monocyte derived dendritic cells generated by IFN-alpha acquire mature dendritic and natural killer cell properties as shown by gene expression analysis. Journal of translational medicine. 2007;5:46. doi: 10.1186/1479-5876-5-46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Vanderheyde N, Vandenabeele P, Goldman M, Willems F. Distinct mechanisms are involved in tumoristatic and tumoricidal activities of monocyte-derived dendritic cells. Immunol Lett. 2004;91(2–3):99–101. doi: 10.1016/j.imlet.2003.11.011. [DOI] [PubMed] [Google Scholar]
- 113.Vanderheyde N, Aksoy E, Amraoui Z, Vandenabeele P, Goldman M, Willems F. Tumoricidal activity of monocyte-derived dendritic cells: evidence for a caspase-8-dependent, Fas-associated death domain-independent mechanism. J Immunol. 2001;167(7):3565–9. doi: 10.4049/jimmunol.167.7.3565. [DOI] [PubMed] [Google Scholar]
- 114.Chapoval AI, Tamada K, Chen L. In vitro growth inhibition of a broad spectrum of tumor cell lines by activated human dendritic cells. Blood. 2000;95(7):2346–51. [PubMed] [Google Scholar]
- 115.Munich S, Sobo-Vujanovic A, Buchser WJ, Beer-Stolz D, Vujanovic NL. Dendritic cell exosomes directly kill tumor cells and activate natural killer cells via TNF superfamily ligands. Oncoimmunology. 2012;1(7):1074–83. doi: 10.4161/onci.20897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Drobits B, Holcmann M, Amberg N, Swiecki M, Grundtner R, Hammer M, Colonna M, Sibilia M. Imiquimod clears tumors in mice independent of adaptive immunity by converting pDCs into tumor-killing effector cells. J Clin Invest. 2012;122(2):575–85. doi: 10.1172/JCI61034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Tel J, Smits EL, Anguille S, Joshi RN, Figdor CG, de Vries IJ. Human plasmacytoid dendritic cells are equipped with antigen-presenting and tumoricidal capacities. Blood. 2012;120(19):3936–44. doi: 10.1182/blood-2012-06-435941. [DOI] [PubMed] [Google Scholar]
- 118.Joo HG, Fleming TP, Tanaka Y, Dunn TJ, Linehan DC, Goedegebuure PS, Eberlein TJ. Human dendritic cells induce tumor-specific apoptosis by soluble factors. Int J Cancer. 2002;102(1):20–8. doi: 10.1002/ijc.10656. [DOI] [PubMed] [Google Scholar]
- 119.Huang FP, Platt N, Wykes M, Major JR, Powell TJ, Jenkins CD, MacPherson GG. A discrete subpopulation of dendritic cells transports apoptotic intestinal epithelial cells to T cell areas of mesenteric lymph nodes. J Exp Med. 2000;191(3):435–44. doi: 10.1084/jem.191.3.435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Aiello S, Noris M, Piccinini G, Tomasoni S, Casiraghi F, Bonazzola S, Mister M, Sayegh MH, Remuzzi G. Thymic dendritic cells express inducible nitric oxide synthase and generate nitric oxide in response to self-and alloantigens. J Immunol. 2000;164(9):4649–58. doi: 10.4049/jimmunol.164.9.4649. [DOI] [PubMed] [Google Scholar]
- 121.Legge KL, Braciale TJ. Lymph node dendritic cells control CD8+ T cell responses through regulated FasL expression. Immunity. 2005;23(6):649–59. doi: 10.1016/j.immuni.2005.11.006. [DOI] [PubMed] [Google Scholar]
- 122.Beaulieu S, Lafontaine M, Richer M, Courchesne I, Cohen EA, Bergeron D. Characterization of the cytotoxic factor(s) released from thymic dendritic cells upon human immunodeficiency virus type 1 infection. Virology. 1998;241(2):285–97. doi: 10.1006/viro.1997.8977. [DOI] [PubMed] [Google Scholar]
- 123.Bergeron D, Beaulieu S, Lafontaine M, Courchesne I, Cohen EA. Infection of human thymic dendritic cells with HIV-1 induces the release of a cytotoxic factor(s) Adv Exp Med Biol. 1997;417:433–8. doi: 10.1007/978-1-4757-9966-8_71. [DOI] [PubMed] [Google Scholar]
- 124.Beaulieu S, Kessous A, Landry D, Montplaisir S, Bergeron D, Cohen EA. In vitro characterization of purified human thymic dendritic cells infected with human immunodeficiency virus type 1. Virology. 1996;222(1):214–26. doi: 10.1006/viro.1996.0412. [DOI] [PubMed] [Google Scholar]
- 125.Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell. 2010;140(6):883–99. doi: 10.1016/j.cell.2010.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Karin M, Greten FR. NF-kappaB: linking inflammation and immunity to cancer development and progression. Nat Rev Immunol. 2005;5(10):749–59. doi: 10.1038/nri1703. [DOI] [PubMed] [Google Scholar]
- 127.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144(5):646–74. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 128.Palucka K, Ueno H, Zurawski G, Fay J, Banchereau J. Building on dendritic cell subsets to improve cancer vaccines. Current opinion in immunology. 2010;22(2):258–63. doi: 10.1016/j.coi.2010.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Schuler G, Schuler-Thurner B, Steinman RM. The use of dendritic cells in cancer immunotherapy. Current opinion in immunology. 2003;15(2):138–47. doi: 10.1016/s0952-7915(03)00015-3. [DOI] [PubMed] [Google Scholar]
- 130.Banchereau J, Klechevsky E, Schmitt N, Morita R, Palucka K, Ueno H. Harnessing human dendritic cell subsets to design novel vaccines. Annals of the New York Academy of Sciences. 2009;1174:24–32. doi: 10.1111/j.1749-6632.2009.04999.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Hernando JJ, Park TW, Kubler K, Offergeld R, Schlebusch H, Bauknecht T. Vaccination with autologous tumour antigen-pulsed dendritic cells in advanced gynaecological malignancies: clinical and immunological evaluation of a phase I trial. Cancer Immunol Immunother. 2002;51(1):45–52. doi: 10.1007/s00262-001-0255-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Holtl L, Rieser C, Papesh C, Ramoner R, Herold M, Klocker H, Radmayr C, Stenzl A, Bartsch G, Thurnher M. Cellular and humoral immune responses in patients with metastatic renal cell carcinoma after vaccination with antigen pulsed dendritic cells. J Urol. 1999;161(3):777–82. [PubMed] [Google Scholar]
- 133.Oosterwijk-Wakka JC, Tiemessen DM, Bleumer I, de Vries IJ, Jongmans W, Adema GJ, Debruyne FM, de Mulder PH, Oosterwijk E, Mulders PF. Vaccination of patients with metastatic renal cell carcinoma with autologous dendritic cells pulsed with autologous tumor antigens in combination with interleukin-2: a phase 1 study. J Immunother. 2002;25(6):500–8. doi: 10.1097/00002371-200211000-00006. [DOI] [PubMed] [Google Scholar]
- 134.Schott M, Seissler J, Feldkamp J, von Schilling C, Scherbaum WA. Dendritic cell immunotherapy induces antitumour response in parathyroid carcinoma and neuroendocrine pancreas carcinoma. Hormone and metabolic research = Hormon- und Stoffwechselforschung = Hormones et metabolisme. 1999;31(12):662–4. doi: 10.1055/s-2007-978817. [DOI] [PubMed] [Google Scholar]
- 135.Cranmer LD, Trevor KT, Hersh EM. Clinical applications of dendritic cell vaccination in the treatment of cancer. Cancer Immunol Immunother. 2004;53(4):275–306. doi: 10.1007/s00262-003-0432-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Lesterhuis WJ, de Vries IJ, Adema GJ, Punt CJ. Dendritic cell-based vaccines in cancer immunotherapy: an update on clinical and immunological results. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2004;15(Suppl 4):iv145–51. doi: 10.1093/annonc/mdh919. [DOI] [PubMed] [Google Scholar]
- 137.Tesniere A, Apetoh L, Ghiringhelli F, Joza N, Panaretakis T, Kepp O, Schlemmer F, Zitvogel L, Kroemer G. Immunogenic cancer cell death: a key-lock paradigm. Curr Opin Immunol. 2008;20(5):504–11. doi: 10.1016/j.coi.2008.05.007. [DOI] [PubMed] [Google Scholar]
- 138.Green DR, Ferguson T, Zitvogel L, Kroemer G. Immunogenic and tolerogenic cell death. Nat Rev Immunol. 2009;9(5):353–63. doi: 10.1038/nri2545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Gallucci S, Matzinger P. Danger signals: SOS to the immune system. Curr Opin Immunol. 2001;13(1):114–9. doi: 10.1016/s0952-7915(00)00191-6. [DOI] [PubMed] [Google Scholar]
- 140.Matzinger P. Tolerance, danger, and the extended family. Annu Rev Immunol. 1994;12:991–1045. doi: 10.1146/annurev.iy.12.040194.005015. [DOI] [PubMed] [Google Scholar]
- 141.Todryk SM, Melcher AA, Dalgleish AG, Vile RG. Heat shock proteins refine the danger theory. Immunology. 2000;99(3):334–7. doi: 10.1046/j.1365-2567.2000.00002.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Triozzi PL, Khurram R, Aldrich WA, Walker MJ, Kim JA, Jaynes S. Intratumoral injection of dendritic cells derived in vitro in patients with metastatic cancer. Cancer. 2000;89(12):2646–54. doi: 10.1002/1097-0142(20001215)89:12<2646::aid-cncr18>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 143.Cheng F, Wang HW, Cuenca A, Huang M, Ghansah T, Brayer J, Kerr WG, Takeda K, Akira S, Schoenberger SP, Yu H, Jove R, Sotomayor EM. A critical role for Stat3 signaling in immune tolerance. Immunity. 2003;19(3):425–36. doi: 10.1016/s1074-7613(03)00232-2. [DOI] [PubMed] [Google Scholar]
- 144.Bromberg JF, Wrzeszczynska MH, Devgan G, Zhao Y, Pestell RG, Albanese C, Darnell JE., Jr Stat3 as an oncogene. Cell. 1999;98(3):295–303. doi: 10.1016/s0092-8674(00)81959-5. [DOI] [PubMed] [Google Scholar]
- 145.Wang T, Niu G, Kortylewski M, Burdelya L, Shain K, Zhang S, Bhattacharya R, Gabrilovich D, Heller R, Coppola D, Dalton W, Jove R, Pardoll D, Yu H. Regulation of the innate and adaptive immune responses by Stat-3 signaling in tumor cells. Nat Med. 2004;10(1):48–54. doi: 10.1038/nm976. [DOI] [PubMed] [Google Scholar]
- 146.Kortylewski M, Kujawski M, Wang T, Wei S, Zhang S, Pilon-Thomas S, Niu G, Kay H, Mule J, Kerr WG, Jove R, Pardoll D, Yu H. Inhibiting Stat3 signaling in the hematopoietic system elicits multicomponent antitumor immunity. Nat Med. 2005;11(12):1314–21. doi: 10.1038/nm1325. [DOI] [PubMed] [Google Scholar]
- 147.Gabrilovich D. Mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat Rev Immunol. 2004;4(12):941–52. doi: 10.1038/nri1498. [DOI] [PubMed] [Google Scholar]
- 148.Burdelya L, Kujawski M, Niu G, Zhong B, Wang T, Zhang S, Kortylewski M, Shain K, Kay H, Djeu J, Dalton W, Pardoll D, Wei S, Yu H. Stat3 activity in melanoma cells affects migration of immune effector cells and nitric oxide-mediated antitumor effects. J Immunol. 2005;174(7):3925–31. doi: 10.4049/jimmunol.174.7.3925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Evel-Kabler K, Song XT, Aldrich M, Huang XF, Chen SY. SOCS1 restricts dendritic cells’ ability to break self tolerance and induce antitumor immunity by regulating IL-12 production and signaling. J Clin Invest. 2006;116(1):90–100. doi: 10.1172/JCI26169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Melief CJ. Mini-review: Regulation of cytotoxic T lymphocyte responses by dendritic cells: peaceful coexistence of cross-priming and direct priming? Eur J Immunol. 2003;33(10):2645–54. doi: 10.1002/eji.200324341. [DOI] [PubMed] [Google Scholar]
- 151.Vermi W, Bonecchi R, Facchetti F, Bianchi D, Sozzani S, Festa S, Berenzi A, Cella M, Colonna M. Recruitment of immature plasmacytoid dendritic cells (plasmacytoid monocytes) and myeloid dendritic cells in primary cutaneous melanomas. J Pathol. 2003;200(2):255–68. doi: 10.1002/path.1344. [DOI] [PubMed] [Google Scholar]
- 152.Salio M, Cella M, Vermi W, Facchetti F, Palmowski MJ, Smith CL, Shepherd D, Colonna M, Cerundolo V. Plasmacytoid dendritic cells prime IFN-gamma-secreting melanoma-specific CD8 lymphocytes and are found in primary melanoma lesions. Eur J Immunol. 2003;33(4):1052–62. doi: 10.1002/eji.200323676. [DOI] [PubMed] [Google Scholar]
- 153.Munn DH, Sharma MD, Hou D, Baban B, Lee JR, Antonia SJ, Messina JL, Chandler P, Koni PA, Mellor AL. Expression of indoleamine 2,3-dioxygenase by plasmacytoid dendritic cells in tumor-draining lymph nodes. J Clin Invest. 2004;114(2):280–90. doi: 10.1172/JCI21583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Zhang M, Tang H, Guo Z, An H, Zhu X, Song W, Guo J, Huang X, Chen T, Wang J, Cao X. Splenic stroma drives mature dendritic cells to differentiate into regulatory dendritic cells. Nat Immunol. 2004;5(11):1124–33. doi: 10.1038/ni1130. [DOI] [PubMed] [Google Scholar]
- 155.Sakaguchi S. Regulatory T cells: key controllers of immunologic self-tolerance. Cell. 2000;101(5):455–8. doi: 10.1016/s0092-8674(00)80856-9. [DOI] [PubMed] [Google Scholar]
- 156.Ghiringhelli F, Larmonier N, Schmitt E, Parcellier A, Cathelin D, Garrido C, Chauffert B, Solary E, Bonnotte B, Martin F. CD4+CD25+ regulatory T cells suppress tumor immunity but are sensitive to cyclophosphamide which allows immunotherapy of established tumors to be curative. Eur J Immunol. 2004;34(2):336–44. doi: 10.1002/eji.200324181. [DOI] [PubMed] [Google Scholar]
- 157.Liyanage UK, Moore TT, Joo HG, Tanaka Y, Herrmann V, Doherty G, Drebin JA, Strasberg SM, Eberlein TJ, Goedegebuure PS, Linehan DC. Prevalence of regulatory T cells is increased in peripheral blood and tumor microenvironment of patients with pancreas or breast adenocarcinoma. J Immunol. 2002;169(5):2756–61. doi: 10.4049/jimmunol.169.5.2756. [DOI] [PubMed] [Google Scholar]
- 158.Viguier M, Lemaitre F, Verola O, Cho MS, Gorochov G, Dubertret L, Bachelez H, Kourilsky P, Ferradini L. Foxp3 expressing CD4+CD25(high) regulatory T cells are overrepresented in human metastatic melanoma lymph nodes and inhibit the function of infiltrating T cells. J Immunol. 2004;173(2):1444–53. doi: 10.4049/jimmunol.173.2.1444. [DOI] [PubMed] [Google Scholar]
- 159.Larmonier N, Marron M, Zeng Y, Cantrell J, Romanoski A, Sepassi M, Thompson S, Chen X, Andreansky S, Katsanis E. Tumor-derived CD4(+)CD25 (+) regulatory T cell suppression of dendritic cell function involves TGF-beta and IL-10. Cancer Immunol Immunother. 2007;56(1):48–59. doi: 10.1007/s00262-006-0160-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Larmonier N, Janikashvili N, LaCasse CJ, Larmonier CB, Cantrell J, Situ E, Lundeen T, Bonnotte B, Katsanis E. Imatinib mesylate inhibits CD4+ CD25+ regulatory T cell activity and enhances active immunotherapy against BCR-ABL-tumors. J Immunol. 2008;181(10):6955–63. doi: 10.4049/jimmunol.181.10.6955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Yang YA, Dukhanina O, Tang B, Mamura M, Letterio JJ, MacGregor J, Patel SC, Khozin S, Liu ZY, Green J, Anver MR, Merlino G, Wakefield LM. Lifetime exposure to a soluble TGF-beta antagonist protects mice against metastasis without adverse side effects. J Clin Invest. 2002;109(12):1607–15. doi: 10.1172/JCI15333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Muraoka RS, Dumont N, Ritter CA, Dugger TC, Brantley DM, Chen J, Easterly E, Roebuck LR, Ryan S, Gotwals PJ, Koteliansky V, Arteaga CL. Blockade of TGF-beta inhibits mammary tumor cell viability, migration, and metastases. J Clin Invest. 2002;109(12):1551–9. doi: 10.1172/JCI15234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Uhl M, Aulwurm S, Wischhusen J, Weiler M, Ma JY, Almirez R, Mangadu R, Liu YW, Platten M, Herrlinger U, Murphy A, Wong DH, Wick W, Higgins LS, Weller M. SD-208, a novel transforming growth factor beta receptor I kinase inhibitor, inhibits growth and invasiveness and enhances immunogenicity of murine and human glioma cells in vitro and in vivo. Cancer Res. 2004;64(21):7954–61. doi: 10.1158/0008-5472.CAN-04-1013. [DOI] [PubMed] [Google Scholar]
- 164.North RJ. Cyclophosphamide-facilitated adoptive immunotherapy of an established tumor depends on elimination of tumor-induced suppressor T cells. J Exp Med. 1982;155(4):1063–74. doi: 10.1084/jem.155.4.1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Larmonier N, Cantrell J, Lacasse C, Li G, Janikashvili N, Situ E, Sepassi M, Andreansky S, Katsanis E. Chaperone-rich tumor cell lysate-mediated activation of antigen-presenting cells resists regulatory T cell suppression. J Leukoc Biol. 2008;83(4):1049–59. doi: 10.1189/jlb.0907635. [DOI] [PubMed] [Google Scholar]
- 166.Santini SM, Lapenta C, Logozzi M, Parlato S, Spada M, Di Pucchio T, Belardelli F. Type I interferon as a powerful adjuvant for monocyte-derived dendritic cell development and activity in vitro and in Hu-PBL-SCID mice. J ExpMed. 2000;191(10):1777–88. doi: 10.1084/jem.191.10.1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Ayres FM, Narita M, Takahashi M, Yano T, Liu A, Toba K, Furukawa T, Aizawa Y. Human dendritic cells mediate anti-tumor activity against hematopoietic tumor cells without direct contact and Fas/FasL killing pathway. Oncol Rep. 2004;11(5):1017–23. [PubMed] [Google Scholar]
- 168.Hubert P, Giannini SL, Vanderplasschen A, Franzen-Detrooz E, Jacobs N, Boniver J, Delvenne P. Dendritic cells induce the death of human papillomavirus-transformed keratinocytes. FASEB J. 2001;15(13):2521–3. doi: 10.1096/fj.00-0872fje. [DOI] [PubMed] [Google Scholar]