Abstract
Background
The emergence and transmission of HIV-1 drug resistance (HIVDR) has raised concerns after rapid global antiretroviral therapy (ART) scale-up. There are limited data on the epidemiology of primary HIVDR in resource-limited settings in Asia. We aimed to determine the prevalence and compare the distribution of HIVDR in a cohort of ART-naïve Asian patients with recent and chronic HIV-1 infection.
Methods
Multicenter prospective study was conducted in ART-naïve patients between 2007 and 2010. Resistance-associated mutations (RAMs) were assessed using the World Health Organization 2009 list for surveillance of primary HIVDR.
Results
A total of 458 patients with recent and 1,340 patients with chronic HIV-1 infection were included in the analysis. The overall prevalence of primary HIVDR was 4.6%. Recently infected patients had a higher prevalence of primary HIVDR (6.1% vs. 4.0%, p = 0.065) and frequencies of RAMs to protease inhibitors (PIs; 3.9% vs. 1.0%, p<0.001). Among those with recent infection, the most common RAMs to nucleoside reverse transcriptase inhibitors (NRTIs) were M184I/V and T215D/E/F/I/S/Y (1.1%), to non-NRTIs was Y181C (1.3%), and to PIs was M46I (1.5%). Of patients with chronic infection, T215D/E/F/I/S/Y (0.8%; NRTI), Y181C (0.5%; non-NRTI), and M46I (0.4%; PI) were the most common RAMs. K70R (p = 0.016) and M46I (p = 0.026) were found more frequently among recently infected patients. In multivariate logistic regression analysis in patients with chronic infection, heterosexual contact as a risk factor for HIV-1 infection was less likely to be associated with primary HIVDR compared to other risk categories (odds ratio 0.34, 95% confidence interval 0.20–0.59, p<0.001).
Conclusions
The prevalence of primary HIVDR was higher among patients with recent than chronic HIV-1 infection in our cohort, but of borderline statistical significance. Chronically infected patients with non-heterosexual risks for HIV were more likely to have primary HIVDR.
Introduction
Highly active antiretroviral therapy (ART) has significantly improved the prognosis of HIV-1-infected patients and prolonged survival worldwide [1]–[3]. Since 2004, the number of people receiving therapy has increased substantially, and exceeded 5 million people in low- and middle-income countries in 2010 [3]. In 2009, UNAIDS reported a 30% increase in the number of people receiving treatment in a single year [3].
The epidemic within the Asia-Pacific region includes largely concentrated epidemics that vary by transmission risk factors. For example, heterosexual transmission is the dominant risk factor for transmission in Thailand, male-to-male sex is the primary risk factor in the Philippines, and injection drug use is the main driver of the epidemic in Malaysia and Indonesia, but Hong Kong has multiple primary epidemic drivers. The main HIV subtype within Southeast Asia is circulating recombinant factor 01, type AE (CRF01_AE). ART was available in high-income countries in the region at similar times to Western countries, including use of protease inhibitors (PI). National programs in resource-limited settings were not scaled up until the mid-2000s, and continue to primarily use non-nucleoside reverse transcriptase inhibitors (NNRTI).
However, now that low- and middle-income countries in the region are increasing their ART coverage [4], [5], there has been an emerging challenge of HIV-1 drug resistance (HIVDR) and first-line treatment failure. Primary HIVDR, pre-existing resistance in those who have not received ART [6], [7], is increasing in settings where ART has been widely available for longer periods of time due to a greater likelihood of acquired resistance-associated mutations (RAMs) in the pool of transmissible virus [5], [8], [9]. The transmission of drug-resistant virus is a growing concern, and has been associated with increased mortality, morbidity, and medical expenditures because of compromising the effectiveness of first-line ART regimens [4], [10], [11].
The reported prevalence of primary HIVDR varies from approximately 1.1% to 21% in the United States, Europe, and Africa [5], [11]–[14]. There are limited data on the epidemiology of primary HIVDR in resource-limited settings in Asia, and pre-ART resistance testing is not routinely performed owing to high cost and limited laboratory infrastructure. To assess the extent of HIVDR in Asia, surveillance of primary HIVDR and monitoring of the development HIVDR in patients taking ART have been conducted through the TREAT Asia Studies to Evaluate Resistance-Surveillance (TASER-S) and the TREAT Asia Studies to Evaluate Resistance-Monitoring (TASER-M) programs [15]. The primary objective of TASER-S is to assess the prevalence of primary HIVDR in ART-naïve, recently HIV-1-infected patients. The primary objectives of TASER-M are to evaluate the prevalence and incidence of emerging HIVDR in ART-naïve HIV-1-infected patients initiating first-line ART and those who are switching from first-line ART to second-line ART after treatment failure.
We aimed to compare the prevalence of primary HIVDR and the distribution of frequencies of RAMs in these cohorts; and to determine factors associated with primary HIVDR. Knowing this epidemiological data can inform healthcare providers and national policy makers in Asia on the emerging issue of primary HIVDR.
Patients and Methods
Four clinical research sites participated in the surveillance study TASER-S [Thailand (N = 2), Hong Kong (N = 1), and Philippines (N = 1)], and 11 sites participated in the monitoring study TASER-M [Thailand (N = 5), Malaysia (N = 3), Hong Kong (N = 1), Philippines (N = 1), and Indonesia (N = 1)]. ART-naïve HIV-1-infected patients enrolled in these cohorts from 2007 to 2010 were included in this study. All patients provided written informed consent to participate and have their data stored in both the site-level and centralized study databases for the purposes of research.
Recent HIV-1 infection was defined according to the World Health Organization (WHO) 2008 criteria for HIVDR threshold surveys [16]. Briefly, the criteria include 1) laboratory confirmation of HIV-1 infection, 2) evidence of recent infection (i.e., positive BED assay or previous negative HIV-1 test in the past year), or 3) has an indeterminate or negative HIV-1 test with detectable HIV-1 RNA or positive p24 antigen. Only the Philippines site use BED assay for the diagnosis of recent HIV infection. Chronic HIV-1 infection was defined as meeting local or national criteria for ART initiation in treatment-naïve patients. Data were collected on age, sex, ethnicity, HIV-1 exposure, the United States Center for Disease Control and Prevention (CDC) disease stage classification, hepatitis B virus (HBV) and hepatitis C virus (HCV) co-infection status, CD4 cell count, HIV-1 RNA level, and HIV-1 subtype. However, the CDC category and hepatitis co-infection status were collected from the patients in TASER-M cohort only.
Genotypic HIV-1 drug resistance testing was performed locally with externally quality controlled in-house or commercial assays, on samples collected within six months prior to ART initiation. Laboratories were required to participate in the TREAT Asia Quality Assurance Scheme (TAQAS), an external assessment program to build genotyping capacity conducted through the National Serologic Reference Laboratory in Australia [17]. RAMs to the major three drug classes of nucleoside reverse transcriptase inhibitors (NRTIs), non-nucleoside reverse transcriptase inhibitors (NNRTIs), and protease inhibitors (PIs) were assessed using the WHO 2009 list of mutations for surveillance of primary HIVDR [18]. Subtype was determined based on reverse transcriptase and protease sequences submitted for drug resistance interpretation using the Stanford University HIV Drug Resistance Database (version 6.0.11). All sequences were submitted to GenBank and the accession numbers are the following; KC791222–KC791422, KC810320–KC810570, KC810571–KC810821, KC867560–KC867647, KC856944–KC857104, KC857105–KC857265, KC921394–KC921484, KC921485–KC921766, KC921767–KC921992, KC961260, KC962512–KC962549, KC970855–KC970880, KC970881–KC970981, KC970982–KC971006, KC971007, KC971008, KC971009–KC971042, KC994162–KC994341, KC994342–KC994450, KC994451, KC994452, KF059614–KF059717, KF059718–KF059833.
Statistical analysis
Baseline characteristics, prevalence of primary HIVDR, and RAM frequencies were compared between groups using the Wilcoxon Rank Sum test for continuous variables and Chi-square test or Fisher's exact test for categorical variables. Factors associated with primary HIVDR were assessed using logistic regression analysis. Variables that were selected by univariate analyses at p-value <0.1, as well as those considered a priori as possible associated factors on the basis of prior research were included in the final multivariate model using a forward stepwise selection process. Statistical analyses were performed using Stata statistical software version 10.0 (Stata Corporation, College Station, TX, 2007).
Results
A total of 458 patients with recent and 1,340 patients with chronic HIV-1 infection were included in the analysis. Their baseline characteristics are shown in Table 1. Patients with recent HIV-1 infection were younger (median 23 vs. 36 years old, p<0.001), mostly male (91.9% vs. 65.9%, p<0.001), and less likely to report heterosexual HIV-1 exposure (13.1% vs. 72.2%, p<0.001). They had a lower proportion of infection with CRF01_AE virus (68.8% vs. 78.7%, p<0.001) and higher CD4 cell counts (median 349 vs. 104 cells/mm3, p<0.001).
Table 1. Baseline characteristics of 1,798 HIV-infected antiretroviral therapy-naïve patients stratified by duration of HIV infection.
| Characteristics | Duration of HIV infection | p-value* | |
| Recent (N = 458) | Chronic (N = 1,340) | ||
| Age groups, years (%) | <0.001 | ||
| ≤20 | 86 (18.8) | 12 (0.9) | |
| 21–30 | 308 (67.2) | 295 (22.0) | |
| 31–40 | 45 (9.8) | 552 (41.2) | |
| 41–50 | 15 (3.3) | 333 (24.8) | |
| ≥51 | 4 (0.9) | 148 (11.0) | |
| Gender, (%) | <0.001 | ||
| Male | 421 (91.9) | 883 (65.9) | |
| Female | 37 (8.1) | 457 (34.1) | |
| Ethnicity, (%) | <0.001 | ||
| Thai | 293 (64.0) | 922 (68.8) | |
| Chinese | 67 (14.7) | 252 (18.8) | |
| Filipino | 84 (18.3) | 40 (3.0) | |
| Other | 14 (3.1) | 126 (9.4) | |
| HIV exposure, (%) | <0.001 | ||
| Heterosexual contact | 60 (13.1) | 965 (72.2) | |
| Homosexual contact | 369 (80.6) | 268 (20.0) | |
| Injecting drug use | 1 (0.2) | 51 (3.8) | |
| Other/unknown | 28 (6.1) | 56 (4.2) | |
| CD4 group, (%) | <0.001 | ||
| ≤50 | 13 (2.8) | 423 (31.6) | |
| 51–100 | 6 (1.3) | 218 (16.3) | |
| 101–200 | 24 (5.2) | 333 (24.8) | |
| ≥201 | 263 (57.4) | 328 (24.5) | |
| Missing | 152 (33.2) | 38 (2.8) | |
| HIV RNA group, copies/mL (%) | <0.001 | ||
| ≤50,000 | 195 (42.6) | 379 (28.3) | |
| 50,001–250,000 | 120 (26.2) | 636 (47.5) | |
| ≥250, 001 | 60 (13.1) | 314 (23.4) | |
| Missing | 83 (18.1) | 11 (0.8) | |
| Subtype, (%) | <0.001 | ||
| CRF01_AE | 315 (68.8) | 1,055 (78.7) | |
| B | 104 (22.7) | 211 (15.7) | |
| Other, non B | 39 (8.5) | 74 (5.5) | |
| Baseline CDC category, (%) | |||
| A | 641 (47.8) | ||
| B | 220 (16.4) | ||
| C | 479 (35.8) | ||
| Missing | 458 (100.0) | 0 | |
| HBsAg, (%) | |||
| Negative | 1,062 (79.2) | ||
| Positive | 131 (9.8) | ||
| Not tested (missing) | 458 (100.0) | 147 (11.0) | |
| Anti-HCV, (%) | |||
| Negative | 968 (72.2) | ||
| Positive | 84 (6.3) | ||
| Not tested (missing) | 458 (100.0) | 288 (21.5) | |
Abbreviations: HBsAg – hepatitis B surface antigen, anti-HCV – antibody to hepatitis C virus.
p-value calculated for categorical data that did not include missing values, using Chi-square test or Fisher's exact test.
The crude combined prevalence of primary HIVDR in both cohorts was 4.6% (82 of 1,798 patients). Of these, 28 (6.1%) patients with recent HIV-1 infection and 54 (4.0%) patients with chronic HIV-1 infection had primary HIVDR (p = 0.065). Overall, the frequency of RAMs was 4.1% for NRTIs, 2.3% for NNRTIs, and 1.8% for PIs. Recently infected patients had higher frequencies of RAMs to NRTIs (5.2% vs. 3.6%, p = 0.138), NNRTIs (2.8% vs. 2.2%, p = 0.410), and PIs (3.9% vs. 1.0%, p<0.001).
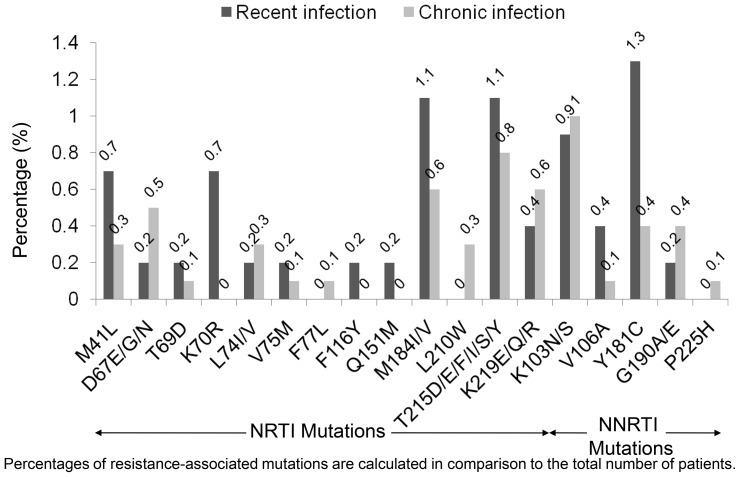
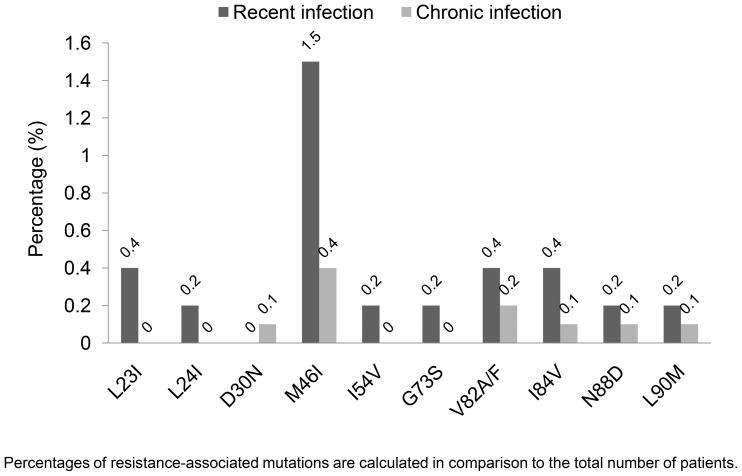
The distributions of RAMs among all HIV-1-infected patients are shown in Figures 1 and 2. In patients with recent HIV-1 infection, M184I/V and T215D/E/F/I/S/Y were the most common RAMs to NRTIs (1.1% each); Y181C was the most common RAM to NNRTIs (1.3%), and M46I was the most common RAM to PIs (1.5%). In patients with chronic HIV-1 infection, T215D/E/F/I/S/Y (0.8%), Y181C (0.5%), and M46I (0.4%) were the most common RAMs to the corresponding drug classes. Higher frequencies of K70R (p = 0.016) and M46I (p = 0.026) were present among patients with recent HIV-1 infection; there were no statistically significant differences in the frequencies of other RAMs.
Figure 1. Distribution of reverse transcriptase resistance-associated mutations among 1,798 antiretroviral-naive HIV-infected patients.
Abbreviations: NRTI; nucleoside reverse transcriptase inhibitor, NNRTI; non-nucleoside reverse transcriptase inhibitor.
Figure 2. Distribution of protease resistance-associated mutations among 1,798 antiretroviral-naive HIV-infected patients.
Patients who had primary HIVDR were less likely to report heterosexual sex as their main risk factor for HIV infection (34.2% vs. 58.1%, p<0.001) or be infected with CRF01_AE virus (65.8% vs. 76.7%, p = 0.024). In the unadjusted model of recent infection, no significant factor associated with primary HIVDR was indentified. Among patients with chronic infection, those with heterosexual HIV risk exposure were less likely to have primary HIVDR compared to those within other risk categories [odds ratio (OR) 0.34, 95% confidence interval (CI) 0.20–0.59, p<0.001; Table 2]. In multivariate logistic regression among patients with chronic infection, patients with heterosexual HIV risk exposure also were less likely to have primary HIVDR (OR 0.34, 95% CI 0.20–0.59, p<0.001).
Table 2. Factors associated with primary HIV drug resistance by duration of HIV infection - univariate logistic regression.
| Variables | Recent HIV infection | Chronic HIV infection | ||||
| Odds ratio | 95% Confidence interval | p-value* | Odds ratio | 95% Confidence interval | p-value* | |
| Age groups, years | 0.054 | 0.112 | ||||
| ≤30 | 1 | 1 | ||||
| 31–40 | 3.64 | 1.44–9.20 | 0.006 | 0.63 | 0.33–1.21 | 0.168 |
| 41–50 | 1.41 | 0.18–11.29 | 0.746 | 0.55 | 0.25–1.18 | 0.125 |
| ≥51 | 6.58 | 0.65–66.25 | 0.110 | 0.44 | 0.15–1.34 | 0.151 |
| Gender | ||||||
| Female | 1 | 1 | ||||
| Male | 2.47 | 0.32–18.69 | 0.382 | 1.24 | 0.68–2.25 | 0.480 |
| Ethnicity | ||||||
| Non Thai | 1 | 1 | ||||
| Thai | 0.74 | 0.34–1.60 | 0.439 | 0.76 | 0.43–1.34 | 0.345 |
| Exposure | ||||||
| Non-heterosexual | 1 | 1 | ||||
| Heterosexual contact | 0.49 | 0.11–2.13 | 0.344 | 0.34 | 0.20–0.59 | <0.001 |
| CDC category * | 0.589 | |||||
| A | 1 | |||||
| B | 1.21 | 0.57–2.57 | 0.623 | |||
| C | 0.85 | 0.46–1.57 | 0.613 | |||
| Subtype | ||||||
| Non CRF01_AE | 1 | 1 | ||||
| CRF01_AE | 0.68 | 0.31–1.50 | 0.344 | 0.57 | 0.32–1.03 | 0.064 |
| CD4, cells/mm3 | 0.394 | 0.824 | ||||
| ≤200 | 1 | 1 | ||||
| ≥201 | 2.54 | 0.33–19.74 | 0.373 | 0.86 | 0.49–1.66 | 0.663 |
| Missing | 3.06 | 0.45–28.50 | 0.225 | 0.62 | 0.08–4.59 | 0.636 |
| HIV RNA, copies/mL | 0.560 | 0.508 | ||||
| ≤50,000 | 1 | 1 | ||||
| 50,001–250,000 | 0.28 | 0.06–1.30 | 0.105 | 0.75 | 0.40–1.41 | 0.377 |
| ≥250,001 | 1.86 | 0.66–5.26 | 0.243 | 0.80 | 0.38–1.68 | 0.551 |
| Missing | 2.03 | 0.81–5.11 | 0.131 | 2.00 | 0.24–16.53 | 0.518 |
| HBsAg serostatus * | 0.226 | |||||
| Negative | 1 | |||||
| Positive | 1.88 | 0.89–3.98 | 0.096 | |||
| Missing | 0.90 | 0.35–2.32 | 0.827 | |||
| Anti-HCV serostatus * | 0.473 | |||||
| Negative | 1 | |||||
| Positive | 1.10 | 0.38–3.15 | 0.856 | |||
| Missing | 0.63 | 0.29–1.36 | 0.238 | |||
Abbreviations: HBsAg – hepatitis B surface antigen, anti-HCV – antibody to hepatitis C virus.
p-value is for test for trend (excluding missing values) for age groups, CDC category, and HIV RNA.
Discussion
To our knowledge, this is the largest cohort study of primary and pre-ART HIVDR among Asian HIV-1-infected patients. The present study showed an overall prevalence of primary HIVDR of almost 5%, which is categorized as low prevalence by WHO criteria. Patients with recent HIV-1 infection had a higher prevalence of primary HIVDR compared to those with chronic HIV-1 infection (6.1% vs. 4.0%), although of borderline statistical significance, and had a higher frequency of RAMs to PIs. This may be explained by both virologic and epidemiologic factors. Resistance testing at the time of HIV-1 transmission is more likely to reveal resistance, as the dominant genetic pattern may revert to wild-type over time and be missed by standard resistance testing in the absence of therapy [19], [20].
Previous reports of primary HIVDR among Asian patients ranged from 0% to 13.8% [8], [9], [21]–[24]. Results have varied by study methodology, route of HIV-1 transmission, duration of HIV-1 infection, HIV-1 subtype, pattern of local ART regimen use, and the reference list of RAMs used to assess the presence of relevant HIVDR mutations [4], [25], [26]. The impact of the reference list is seen when comparing these results to a preliminary report of the same cohort that used International AIDS Society (IAS)-USA 2009 criteria and reported 13.8% overall prevalence of primary HIVDR in a subset of 700 patients with chronic HIV-1 infection [23]. The overall prevalence of HIVDR increased to 10.4% when using the IAS-USA criteria in the present study.
In this study, the prevalence of primary HIVDR in patients with recent infection was higher than that found in previous surveys of primary HIVDR in this region [8], [21], [22], [27]. However, the recent study in Thailand showed that primary resistance among men who have sex with men was approximately 7% [28]. The frequencies of RAMs to NRTIs and NNRTIs found in this study were higher than those to PIs. This pattern of primary HIVDR is comparable to reports from African countries and in other settings where NNRTI-based regimens are more commonly used in the scale-up of ART [5], [29]. There were no statistically significant differences in the frequencies of RAMs to NRTIs and NNRTIs between patients with recent and chronic HIV-1 infection, but RAMs to PIs where significantly more frequent in patients with recent HIV-1 infection, which may reflect the increasing use of PIs as part of second-line regimens.
The mutation selected by lamivudine, M184I/V, and the thymidine analogue mutation of T215D/E/F/I/S/Y were the most common RAMs in patients with recent HIV-1 infection and T215D/E/F/I/S/Y were the most common RAMs in those with chronic HIV-1 infection. T215D/E/I/S confers an increased risk of virologic failure for ART-naïve patients started on regimens including zidovudine or stavudine [30], [31]. The most common RAM to NNRTIs found in this study was Y181C, which is selected by nevirapine or efavirenz [32] and the most widely used “anchor” drugs in low- and middle-income countries. The most common regimen which has been widely prescribed in Asia before 2007 is NNRTI-based regimen, such as stavudine/lamivudine/nevirapine and zidovudine/lamivudine/nevirapine.
We also noted higher frequencies of K70R and M46I in patients with recent HIV-1 infection. Previous studies also have shown that K70R has been one of the most common RAMs observed among ART-naïve patients, and is consistent with the widespread use of zidovudine and stavudine in the region [33], [34]. Its higher frequency reflects the longer periods of antiretroviral drugs use in the study settings [35], [36]. M46I was the most common RAMs to PIs found in this study, in agreement with reports from Europe and South Africa [37], [38]. M46I was identified as a relevant RAM for surveillance of primary HIVDR because it reduces the susceptibility to several PIs even in the absence of other surveillance drug-resistance mutations [39].
Our results raise concerns with regards to the risk of treatment failure among Asian patients. Primary HIVDR is clearly associated with the risk of early virologic failure [10], [11]. Although the prevalence of HIVDR in the chronically infected cohort was below 5%, considered in the low range by the WHO criteria, those with non-heterosexual contact as a risk factor for HIV-1 infection were more likely to have primary HIVDR. This finding may guide the selection of ART-naïve patients who would more likely benefit from resistance testing prior to ART initiation in Asia, where routine primary HIVDR screening is not feasible. Further study to determine the cost-effectiveness of routine primary HIVDR testing prior to ART initiation in specific risk groups in Asia is needed.
Our study may not be directly generalizable across the region. Participating sites were largely in urban referral centers and our findings should be considered in the context of the specific characteristics of the study populations and commonly used ART regimens in the study sites. We have described these data as being in Asian patients, but recognize the intra-regional and inter-ethnic variations across the participating study sites. There were difference of clinical characteristics between patients with recent HIV-1 infection and ones with chronic HIV-1 infection. The patients were enrolled separately from each two cohorts. The difference of primary HIVDR between these two patient groups identified might be associated with the sampling bias. In addition, we appreciate that the BED assay is not considered a reliable indicator of recent infection; however, it is a convenient assay. In general, there were few patients for whom the BED test was used as the primary inclusion criteria. In our study, 84 the Filipino patients (18.3% of patients with recent HIV infection) met inclusion criteria based on their BED results. Unfortunately, we do not have CD4 cell count information and it is possible that some Filipino patients may be misclassified by BED to recent HIV infection if they have CD4 cell count <100 cells/mm3. Furthermore, as a limited number of variables were available in the TASER-S cohort, the potential effects of some common demographic characteristics could not be assessed. The small number of patients with primary HIVDR limited the power of some of our statistical analysis, notably with regards to patients with recent HIV-1 infection.
Conclusions
The overall prevalence of primary HIVDR in Asian HIV-1-infected patients in these cohorts is approximately 5%. The prevalence is somewhat higher among patients with recent HIV-1 infection. Patients with chronic infection with non-heterosexual HIV-1 exposures were at higher risk of having primary HIVDR. Ongoing national and regional HIVDR evaluation is needed to identify higher risk populations who may benefit from targeted pre-ART HIVDR testing in settings with limited resources.
Acknowledgments
The TREAT Asia Studies to Evaluate Resistance (TASER) is an initiative of TREAT Asia, a program of amfAR, The Foundation for AIDS Research, with major support provided by the Dutch Ministry of Foreign Affairs through a partnership with Stichting Aids Fonds, and with additional support from amfAR and the National Institute of Allergy and Infectious Diseases (NIAID) of the U.S. National Institutes of Health (NIH) and the National Cancer Institute (NCI) as part of the International Epidemiologic Databases to Evaluate AIDS (IeDEA) (grant no. U01AI069907). Queen Elizabeth Hospital and the Integrated Treatment Centre are supported by the Hong Kong Council for AIDS Trust Fund. The Kirby Institute is funded by the Australian Government Department of Health and Ageing, and is affiliated with the Faculty of Medicine, The University of New South Wales. The content of this publication is solely the responsibility of the authors and does not necessarily represent the official views of any of the institutions mentioned above.
The members of the TASER study are:
• Li PCK and Lee MP, Queen Elizabeth Hospital and Wong KH, Integrated Treatment Centre, Hong Kong, China;
• Kumarasamy N and Saghayam S, YRG CARE Medical Centre, Chennai, India;
• Pujari S and Joshi K, Institute of Infectious Diseases, Pune, India;
• Merati TP† and Yuliana F, Faculty of Medicine, Udayana University & Sanglah Hospital, Bali, Indonesia;
• Kamarulzaman A and Ong LY, University of Malaya, Kuala Lumpur, Malaysia;
• Lee C KC and Sim B HL, Hospital Sungai Buloh, Kuala Lumpur, Malaysia;
• Mustafa M and Nordin N, Hospital Raja Perempuan Zainab II, Kota Bharu, Malaysia;
• Ditangco R‡ and Bantique RO, Research Institute for Tropical Medicine, Manila, Philippines;
• Chen YMA, Chen YJ and Lin YT, AIDS Prevention and Research Centre, National Yang-Ming University, Taipei, Taiwan;
• Kantipong P and Kambua P, Chiangrai Prachanukroh Hospital, Chiang Rai, Thailand;
• Phanuphak P and Sirivichayakul S, HIV-NAT/Thai Red Cross AIDS Research Centre, Bangkok, Thailand;
• Ratanasuwan W and Sriondee R, Faculty of Medicine Siriraj Hospital, Mahidol University, Bangkok, Thailand;
• Sirisanthana T and Praparattanapan J, Research Institute for Health Sciences, Chiang Mai University, Chiang Mai, Thailand;
• Sungkanuparph S, Kiertiburanakul S, and Chumla L, Faculty of Medicine Ramathibodi Hospital, Mahidol University, Bangkok, Thailand;
• Kantor R, Brown University, Rhode Island, U.S.A.;
• Sohn AH, Durier N and Singtoroj T, TREAT Asia, amfAR - The Foundation for AIDS Research, Bangkok, Thailand;
• Cooper DA, Law MG, Jiamsakul A and Boettiger DC, The Kirby Institute, University of New South Wales, Sydney, Australia.
† Steering Committee Chair, ‡ Co-Chair
Funding Statement
The TREAT Asia Studies to Evaluate Resistance is an initiative of TREAT Asia, a program of amfAR, The Foundation for AIDS Research, with major support provided by the Dutch Ministry of Foreign Affairs through a partnership with Stichting Aids Fonds, and with additional support from amfAR and the National Institute of Allergy and Infectious Diseases (NIAID) of the U.S. National Institutes of Health (NIH) and the National Cancer Institute (NCI) as part of the International Epidemiologic Databases to Evaluate AIDS (IeDEA) (grant no. U01AI069907). Queen Elizabeth Hospital and the Integrated Treatment Centre are supported by the Hong Kong Council for AIDS Trust Fund. The Kirby Institute is funded by the Australian Government Department of Health and Ageing, and is affiliated with the Faculty of Medicine, The University of New South Wales. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Johansson KA, Robberstad B, Norheim OF (2010) Further benefits by early start of HIV treatment in low income countries: survival estimates of early versus deferred antiretroviral therapy. AIDS Res Ther 7: 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Jongwutiwes U, Sungkanuparph S, Kiertiburanakul S (2008) Comparison of clinical features and survival between cryptococcosis in human immunodeficiency virus (HIV)-positive and HIV-negative patients. Jpn J Infect Dis 61: 111–115. [PubMed] [Google Scholar]
- 3.Joint United Nations Programme on HIV/AIDS and World Health Organization: Global report (2010) UNAIDS report on the global AIDS epidemic 2010. WHO Library Cataloguing-in-Publication. Available: http://www.unaids.org/globalreport/documents/20101123_GlobalReport_full_en.pdf. Assessed 25 September 2012.
- 4. Geretti AM (2007) Epidemiology of antiretroviral drug resistance in drug naive persons. Curr Opin Infect Dis 20: 22–32. [DOI] [PubMed] [Google Scholar]
- 5. Hamers RL, Wallis CL, Kityo C (2011) HIV-1 drug resistance in antiretroviral-naive individuals in sub-Saharan Africa after rollout of antiretroviral therapy: a multicentre observational study. Lancet Infect Dis 11: 750–759. [DOI] [PubMed] [Google Scholar]
- 6.Division of HIV/AIDS Epidemiology and Surveillance, National HIV and Retrovirology Laboratories, Centre for Infectious Disease Prevention and Control, Public Health Agency of Canada, Health Canada: HIV-1 strain and primary drug resistance in Canada (2006) Available: http://www.phacaspc.gc.ca/publicat/hiv1-vih1/pdf/hiv-1-strain-01-e.pdf. Assessed 25 September 2012.
- 7.World Health Organization: Monitoring the emergence of antiretroviral resistance. Report of a WHO consultation organized in collaboration with Istituto di Sanità and the International AIDS Society, Rome, Italy (2000) Available: http://apps.who.int/medicinedocs/documents/s16347e/s16347e.pdf. Assessed 25 September 2012.
- 8. Apisarnthanarak A, Jirayasethpong T, Sa-nguansilp C, Thongprapai H, Kittihanukul C, et al. (2008) Antiretroviral drug resistance among antiretroviral-naive persons with recent HIV infection in Thailand. HIV Med 9: 322–325. [DOI] [PubMed] [Google Scholar]
- 9. Apisarnthanarak A, Mundy LM (2008) Antiretroviral drug resistance among antiretroviral-naive individuals with HIV infection of unknown duration in Thailand. Clin Infect Dis 46: 1630–1631. [DOI] [PubMed] [Google Scholar]
- 10. Gagliani LH, Alkmim Maia WT, Sa-Filho D (2011) The association between primary antiretroviral resistance and HAART virologic failure in a developing set. AIDS Res Hum Retroviruses 27: 251–256. [DOI] [PubMed] [Google Scholar]
- 11. Little SJ, Holte S, Routy JP, Daar ES, Markowitz M, et al. (2002) Antiretroviral-drug resistance among patients recently infected with HIV. N Engl J Med 347: 385–394. [DOI] [PubMed] [Google Scholar]
- 12. Charpentier C, Bellecave P, Cisse M, Mamadou S, Diakite M, et al. (2011) High prevalence of antiretroviral drug resistance among HIV-1-untreated patients in Guinea-Conakry and in Niger. Antivir Ther 16: 429–433. [DOI] [PubMed] [Google Scholar]
- 13. Grant RM, Hecht FM, Warmerdam M, Liu L, Liegler T, et al. (2002) Time trends in primary HIV-1 drug resistance among recently infected persons. JAMA 288: 181–188. [DOI] [PubMed] [Google Scholar]
- 14. Vergne L, Diagbouga S, Kouanfack C, Aghokeng A, Butel C, et al. (2006) HIV-1 drug-resistance mutations among newly diagnosed patients before scaling-up programmes in Burkina Faso and Cameroon. Antivir Ther 11: 575–579. [PubMed] [Google Scholar]
- 15. Hamers RL, Oyomopito R, Kityo C, Phanuphak P, Siwale M, et al. (2012) The PharmAccess African (PASER-M) and the TREAT Asia (TASER-M) Monitoring Studies to Evaluate Resistance–HIV drug resistance in sub-Saharan Africa and the Asia-Pacific. Int J Epidemiol 41: 43–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Bennett DE, Bertagnolio S, Sutherland D, Gilks CF (2008) The World Health Organization's global strategy for prevention and assessment of HIV drug resistance. Antivir Ther 13: 1–13. [PubMed] [Google Scholar]
- 17. Land S, Cunningham P, Zhou J, Frost K, Katzenstein D, et al. (2009) TREAT Asia Quality Assessment Scheme (TAQAS) to standardize the outcome of HIV genotypic resistance testing in a group of Asian laboratories. J Virol Methods 159: 185–193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Bennett DE, Camacho RJ, Otelea D, Kuritzkes DR, Fleury H, et al. (2009) Drug resistance mutations for surveillance of transmitted HIV-1 drug-resistance: 2009 update. PLoS One 4: e4724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infec ted adults and adolescents. DHHS. March 27, 2012 (2012) Available at: http://www.aidsinfo.nih.gov/contentfiles/lvguidelines/adultandadolescentgl.pdf. Assessed 25 September 2012.
- 20. Kuritzkes DR (2004) Preventing and managing antiretroviral drug resistance. AIDS Patient Care STDS 18: 259–273. [DOI] [PubMed] [Google Scholar]
- 21. Nguyen HT, Duc NB, Shrivastava R, Tran TH, Nguyen TA, et al. (2008) HIV drug resistance threshold survey using specimens from voluntary counselling and testing sites in Hanoi, Vietnam. Antivir Ther 13: 115–121. [PubMed] [Google Scholar]
- 22. Sirivichayakul S, Phanuphak P, Pankam T, O-Charoen R, Sutherland D, et al. (2008) HIV drug resistance transmission threshold survey in Bangkok, Thailand. Antivir Ther 13: 109–113. [PubMed] [Google Scholar]
- 23. Sungkanuparph S, Oyomopito R, Sirivichayakul S, Sirisanthana T, Li PC, et al. (2011) HIV-1 drug resistance mutations among antiretroviral-naive HIV-1-infected patients in Asia: results from the TREAT Asia Studies to Evaluate Resistance-Monitoring Study. Clin Infect Dis 52: 1053–1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Zhao B, Han X, Dai D, Liu J, Ding H, et al. (2011) New trends of primary drug resistance among HIV type 1-infected men who have sex with men in Liaoning province, China. AIDS Res Hum Retroviruses 27: 1047–1053. [DOI] [PubMed] [Google Scholar]
- 25. Kiertiburanakul S, Sungkanuparph S (2009) Emerging of HIV drug resistance: epidemiology, diagnosis, treatment and prevention. Curr HIV Res 7: 273–278. [DOI] [PubMed] [Google Scholar]
- 26. Sungkanuparph S, Kiertiburanakul S, Sukasem C, Chantratita W (2012) Discrepancies Between WHO 2009 and IASUSA 2009 Lists for Determining the Rate of Transmitted HIV-1 Drug Resistance: A Prospective Study. J Acquir Immune Defic Syndr 59: e3–5. [DOI] [PubMed] [Google Scholar]
- 27. Lee CC, Sun YJ, Barkham T, Leo YS (2009) Primary drug resistance and transmission analysis of HIV-1 in acute and recent drug-naive seroconverters in Singapore. HIV Med 10: 370–377. [DOI] [PubMed] [Google Scholar]
- 28.Sirivichayakul S, DeLong A, Wongkunya R, Mekprasan S, Sohn AH, et al.. Increasing HIV drug resistance among recently infected treatment-naïve MSM in Thailand: results from three years annual surveillance. In program and abstract of the 6th IAS Conference on HIV Pathogenesis, Treatment and Prevention (Rome), July 17–20, 2011 (2011) [abstract MOPE053] [Google Scholar]
- 29. Haidara A, Chamberland A, Sylla M, Aboubacrine SA, Cissé M, et al. (2010) High level of primary drug resistance in Mali. HIV Med 11: 404–411. [DOI] [PubMed] [Google Scholar]
- 30. Riva C, Violin M, Cozzi-Lepri A, Aboubacrine SA, Cissé M, et al. (2002) Transmitted virus with substitutions at position 215 and risk of virological failure in antiretroviral-naive patients starting highly active antiretroviral therapy. Antivir Ther 7: S103–104. [Google Scholar]
- 31. Violin M, Cozzi-Lepri A, Velleca R, Vincenti A, D'Elia S, et al. (2004) Risk of failure in patients with 215 HIV-1 revertants starting their first thymidine analog-containing highly active antiretroviral therapy. AIDS 18: 227–235. [DOI] [PubMed] [Google Scholar]
- 32. Johnson VA, Brun-Vezinet F, Clotet B, Günthard HF, Kuritzkes DR, et al. (2010) Update of the drug resistance mutations in HIV-1: December 2010. Top HIV Med 18: 156–163. [PubMed] [Google Scholar]
- 33. Eiros JM, Labayru C, Hernández B, Ortega M, Bachiller P, et al. (2002) Prevalence of genotypic resistance in untreated HIV patients in Spain. Eur J Clin Microbiol Infect Dis 21: 310–313. [DOI] [PubMed] [Google Scholar]
- 34. Ammaranond P, Cunningham P, Oelrichs R, Suzuki K, Harris C, et al. (2003) Rates of transmission of antiretroviral drug resistant strains of HIV-1. J Clin Virol 26: 153–161. [DOI] [PubMed] [Google Scholar]
- 35. Larder BA, Kemp SD (1989) Multiple mutations in HIV-1 reverse transcriptase confer high-level resistance to zidovudine (AZT). Science 246: 1155–1158. [DOI] [PubMed] [Google Scholar]
- 36. Picard V, Angelini E, Maillard A, Race E, Clavel F, et al. (2001) Comparison of genotypic and phenotypic resistance patterns of human immunodeficiency virus type 1 isolates from patients treated with stavudine and didanosine or zidovudine and lamivudine. J Infect Dis 184: 781–784. [DOI] [PubMed] [Google Scholar]
- 37. Maphalala G, Okello V, Mndzebele S, Gwebu P, Mulima N, et al. (2008) Surveillance of transmitted HIV drug resistance in the Manzini-Mbabane corridor, Swaziland, in 2006. Antivir Ther 13 suppl 2: 95–100. [PubMed] [Google Scholar]
- 38. Inocencio LA, Pereira AA, Sucupira MC, Fernandez JC, Jorge CP, et al. (2009) Brazilian Network for HIV Drug Resistance Surveillance: a survey of individuals recently diagnosed with HIV. J Int AIDS Soc 12: 20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Rhee SY, Liu T, Ravela J, Gonzales MJ, Shafer RW (2004) Distribution of human immunodeficiency virus type 1 protease and reverse transcriptase mutation patterns in 4,183 persons undergoing genotypic resistance testing. Antimicrob Agents Chemother 48: 3122–3126. [DOI] [PMC free article] [PubMed] [Google Scholar]