Abstract
Parathyroid Hormone related Protein (PTHrP) is a critical regulator of mammary gland morphogenesis in the mouse embryo. Loss of PTHrP, or its receptor, PTHR1, results in arrested mammary buds at day 15 of embryonic development (E15). In contrast, overexpression of PTHrP converts the ventral epidermis into hairless nipple skin. PTHrP signaling appears to be critical for mammary mesenchyme specification, which in turn maintains mammary epithelial identity, directs bud outgrowth, disrupts the male mammary rudiment and specifies the formation of the nipple. In the embryonic mammary bud, PTHrP exerts its effects on morphogenesis, in part, through epithelial-stromal crosstalk mediated by Wnt and BMP signaling. Recently, PTHLH has been identified as a strong candidate for a novel breast cancer susceptibility locus, although PTHrP’s role in breast cancer has not been clearly defined. The effects of PTHrP on the growth of the embryonic mammary rudiment and its invasion into the dermis may, in turn, have connections to the role of PTHrP in breast cancer.
Keywords: PTHrP, mammary, development, embryonic, breast, Wnt, Bmp
Introduction
Parathyroid hormone-related protein (PTHrP) derives its name from structural and functional similarities with parathyroid hormone (PTH) (1). Both PTH and PTHrP are derived from the same ancestral gene and are highly homologous in their amino-terminal domains (2). As a result, both PTH and PTHrP bind and signal through the same G-protein coupled receptor, known as the Type 1 PTH/PTHrP receptor (PTHR1) (3). While PTH is secreted almost exclusively by the parathyroid glands, PTHrP is expressed widely. During normal embryonic and postnatal development, PTHrP acts as a local autocrine/paracrine/intracrine growth factor in many tissues including bone, cartilage and the mammary gland (4). Unlike PTH, which acts as a classical peptide hormone, PTHrP does not normally circulate. The exceptions to this rule include lactation and patients with cancer, including breast cancer. During lactation, circulating PTHrP mobilizes skeletal calcium stores to be delivered to the breast for milk production. In some patients with cancer, PTHrP, secreted by malignant cells, mobilizes skeletal calcium to cause humoral hypercalcemia of malignancy (1, 5, 6).
Expression of PTHrP in the embryonic mammary gland
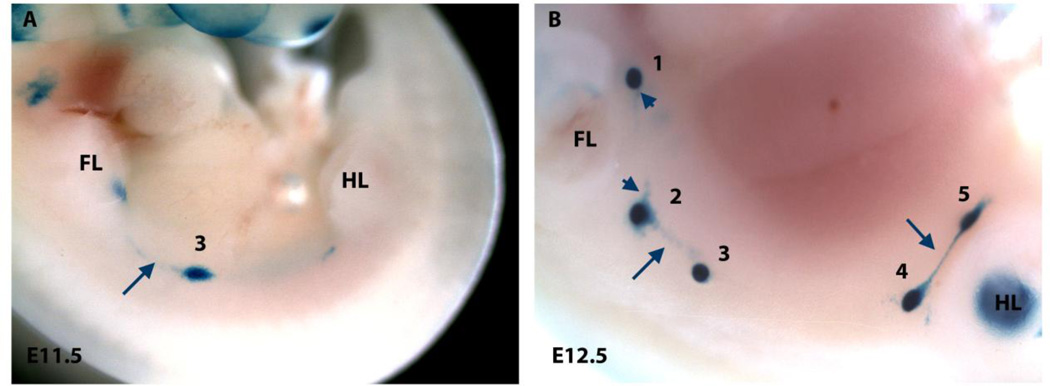
Endogenous PTHrP expression in the mammary gland has been most recently profiled using genetically modified mice in which the lacZ gene was knocked into the PTHrP locus (PTHrPlacZ/+). In these mice, blue X-gal (4-chloro-5-bromo-3-indoyl-D-galactopyranoside) staining resulting from β-galactosidase activity reflects the activity of the Pthlh promoter (7). Using these mice, X-gal staining was observed in the emerging mammary placodes beginning on embryonic day 11.5 (E11.5) (Fig. 1A). Staining extended outward on one side of each placode along a “tail” of adjacent epithelial cells, and faint staining was observed between developing placodes within the mammary line (8) (Fig. 1B). This pattern suggests that PTHrP gene (Pthlh) expression is first activated within the cells of the mammary line, perhaps as these cells coalesce into the developing epithelial placodes.
Figure 1. Expression of PTHrP-lacZ in the mammary placodes.
X-gal staining is seen in the mammary placode (A) and in cells of the mammary line (A, arrow). At E12.5, staining is observed in all five placodes (B) and along a “tail” of cells extending outwards from the placodes (arrowheads). Staining is also observed in the cells between the mammary placodes (B, arrows). FL= forelimb bud, HL= hindlimb bud.
In subsequent stages, PTHrP-lacZ activity was restricted to the epithelial cells of the mammary bud (E12.5), the elongated mammary sprout (E16.5) and the epithelial cells of the rudimentary ductal tree at birth. PTHrP-lacZ activity was found to be completely absent in the adjacent mammary mesenchyme (7–9). These data agree with previous assessments of Pthlh expression in the embryonic mammary gland using in situ hybridization (10, 11). In contrast to the epithelial pattern of Pthlh expression, the Pthr1 gene is expressed exclusively in the mesenchyme. Pthr1 expression is observed generally within the dermal mesenchyme as early as E12 and is maintained in the mammary mesenchyme at later stages such as E15, E18 (10, 12, 13). While the above data have been generated in mouse embryos, similar patterns of epithelial PTHLH expression and mesenchymal PTHR1 expression have also been documented in the embryonic breast buds of human fetuses (14, 15).
The factors that induce PTHrP and PTHR1 expression in the embryonic mammary gland have not been studied extensively. However, we have observed that PTHrP-lacZ expression is maintained in Lef1−/− mutants, suggesting that Wnt signaling, via Lef1, does not regulate Pthlh expression (M.H and J.W, unpublished observations). Recent studies have shown that Pthlh is transcriptionally upregulated by Eda signaling and that mammary buds from K14-Eda mice exhibit higher PTHLH gene expression compared to their wild-type (WT) counterparts (16). In addition, these same investigators demonstrated that Pthlh gene expression is reduced in mammary buds from IκBαΔN mice that fail to activate NF-κB. Therefore, it appears that Eda-Edar signaling acting through the NF-κB pathway contributes to the activation of PTHrP expression in the embryonic mammary bud (16). The identity of the other pathways that interact with NF-κB to regulate PTHrP in these structures remains an open question for future investigation.
Role of PTHrP signaling in mammary mesenchyme specification
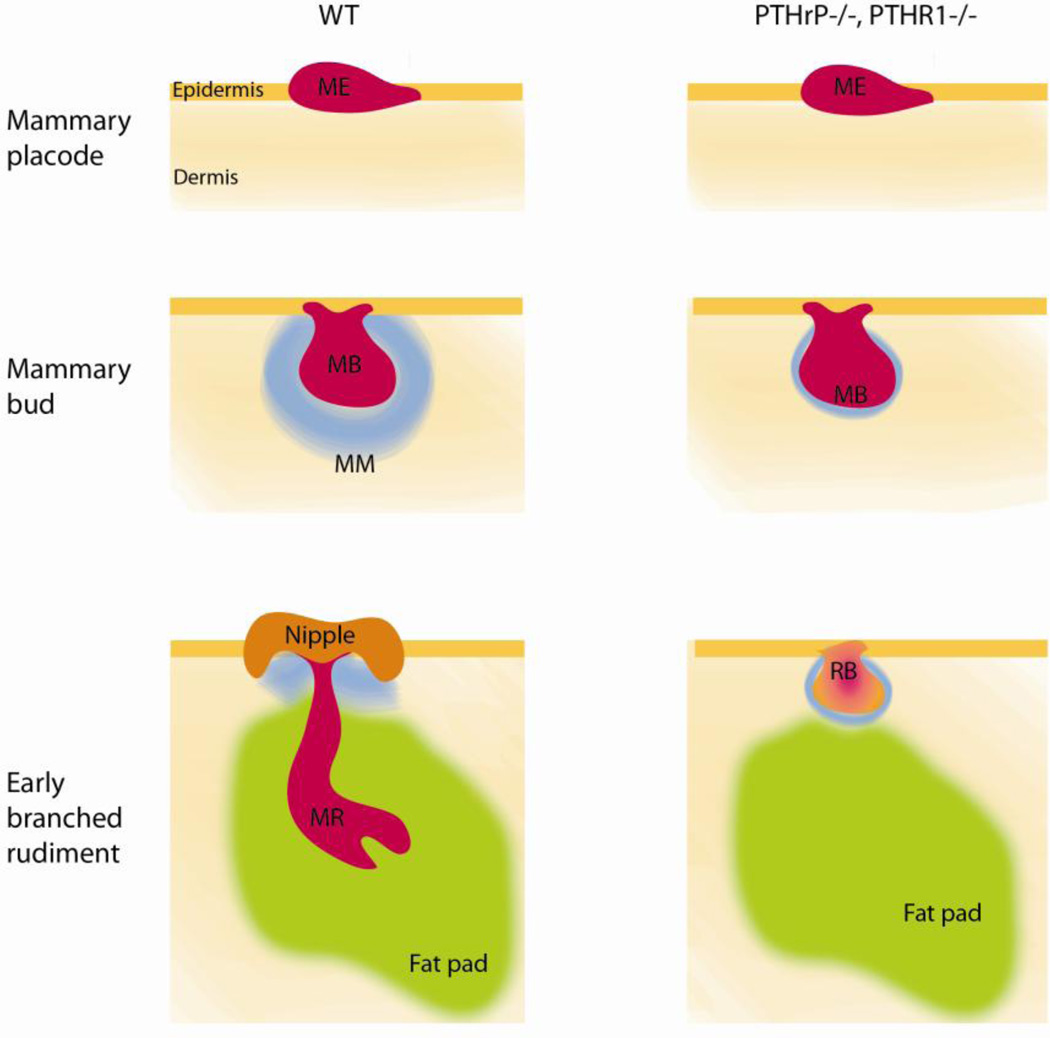
Despite the observation that PTHrP expression appears very early during embryonic mammary development, PTHrP signaling is not required for the formation of the mammary line or the mammary placodes. All five pairs of placodes form normally in appropriate positions in PTHrP−/− and PTH1R−/− embryos. However, a few days later, mammary development is severely compromised when either the Pthlh or the Pthr1 genes are disrupted (Fig. 2). The mammary buds from PTHrP−/− and PTHR1−/− embryos have fewer layers of dense mammary mesenchyme as early as E13 (17). Furthermore, the expression of many molecular markers characteristic of the mammary mesenchyme, such as Lef1, β-catenin, tenascin C, cadherin 11, the androgen receptor and the estrogen receptor is severely reduced or completely abolished, suggesting that the mammary mesenchyme is not properly specified by E15 in the absence of PTHrP signaling (12, 17). Transgenic overexpression of PTHrP in basal keratinocytes causes the ectopic induction of molecular markers of the mammary mesenchyme in the dermal mesenchyme, at least on the ventral surface of embryos (18, 19). The effects of PTHrP on the mesenchyme are an example of epithelial induction of mesenchymal differentiation, since loss of either Pthlh or Pthr1 produces similar defects in mammary mesenchyme differentiation and, as noted previously, PTHrP is made by epithelial cells and its receptor is expressed in the mesenchyme. These data suggest that PTHrP acts as a signal that converts the general dermal mesenchyme into the specialized, dense mammary mesenchyme as epithelial cells within the mammary placodes are invaginating into the dermis to form the mammary buds.
Figure 2. Schematic representation of the consequences of PTHrP/PTHR1 deficiency.
Mammary epithelium (pink, ME) is specified in WT, PTHrP−/− and PTHR1−/− embryos. Loss of PTHrP and PTHR1 results in reduced mammary mesenchyme (blue, MM) that does not support outgrowth of the mammary rudiment (MR) or formation of the nipple. Instead, the mammary buds revert to an epidermal fate (reverting bud, RB).
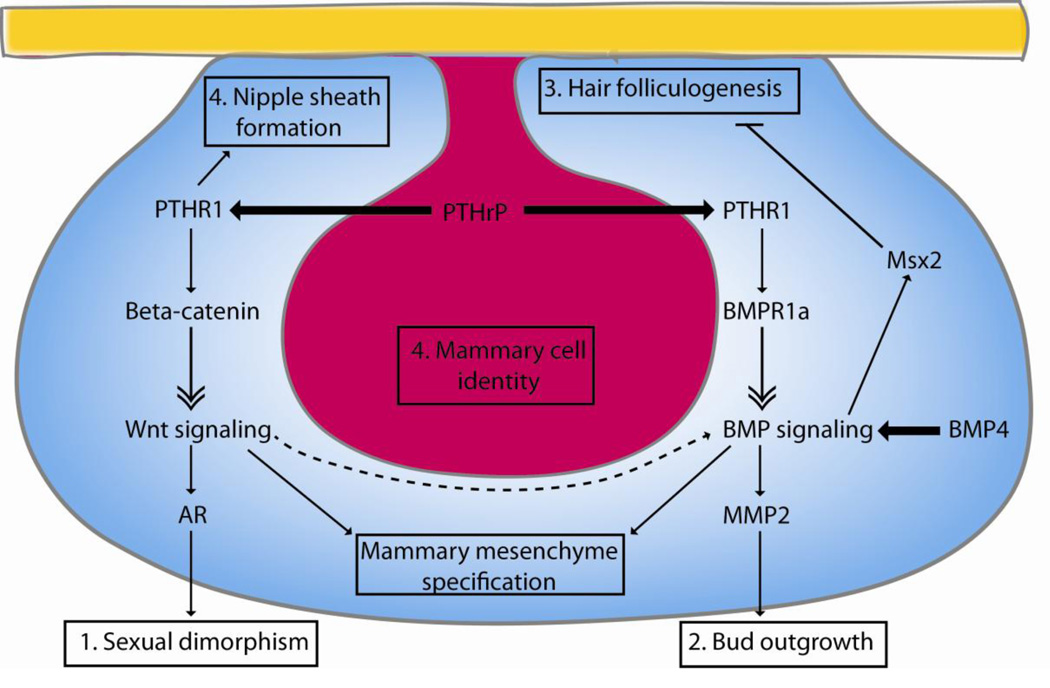
As noted in the previous paragraph, overexpression of PTHrP in basal keratinocytes in K14-PTHrP transgenic mice converts the dermis into mammary mesenchyme, but this occurs only on the ventral aspect of the embryo despite equal levels of PTHrP overexpression in the dorsal skin (18, 19). This appears to be explained, in part, by the expression of BMP4 in the ventral, but not the lateral or the dorsal dermis, during the period in which the mammary buds are forming. In fact, experiments have demonstrated that PTHrP signaling interacts with BMPR1a signaling within mesenchymal cells in order to specify proper differentiation of the mammary mesenchyme (20). PTHrP also augments mesenchymal Wnt signaling. Recent experiments have demonstrated that Wnt activation in the mesenchymal, but not epithelial, cells of the mammary bud is completely dependent on PTHrP signaling (17). This is likely due, in part, to the upregulation of Wnt 11 and R-spondin1 expression in mesenchymal cells in response to PTHrP. Furthermore, tissue-specific deletion of β-catenin in the overall dermal mesenchyme, including the developing mammary mesenchyme phenocopies the effects of loss of PTHrP on the expression of mammary mesenchyme markers and prevents the formation of ectopic mammary mesenchyme in response to PTHrP overexpression in K14-PTHrP mice. Moreover, loss of β-catenin inhibits the proliferation of mammary mesenchyme cells (17, 21). As a result of these findings, our working hypothesis is that focal expression of PTHrP in the developing mammary bud activates mesenchymal Wnt signaling. It also allows mammary mesenchyme cells to respond to BMP4 already found in the ventral dermis. In turn, the coordinated activation of BMP and Wnt signaling downstream of PTHrP is critical to proper expansion and specification of the mammary-specific mesenchyme around the epithelial bud (Fig. 3).
Figure 3. Downstream signaling engaged by PTHrP during embryonic mammary development.
PTHrP is secreted from the mammary epithelium (pink) and acts on the mammary mesenchyme (blue) to activate signaling via β-catenin. This pathway directs sexual dimorphism, in part via regulation of AR expression, and possibly regulates the response to BMP signaling. PTHrP signaling, via PTHR1, also upregulates BMPR1a and sensitizes mammary mesenchymal cells to BMP4, possibly upregulating BMP signaling in the mammary mesenchyme. This pathway is required for bud outgrowth, via MMP2 and for inhibiting hair folliculogenesis in the nipple, via Msx2. PTHrP signaling also maintains mammary epithelial identity via signaling in the mammary mesenchyme, although the exact mechanism remains unknown.
PTHrP maintains mammary cell fate via the mesenchyme
The failure of proper mammary mesenchyme development in PTHrP−/− and PTH1R−/− embryos has consequences for mammary epithelial development. Loss of PTHrP signaling causes the arrest of mammary development at the bud stage and the subsequent reversion of mammary epithelial cells to an epidermal fate. In WT mammary rudiments at E15-E18, all mammary epithelial cells express the basal keratinocyte marker, keratin 14 (K14), but not the suprabasal markers, keratin1, keratin10, or involucrin. However, when either the PTHLH or PTHR1 genes are disrupted, cells in the center of the epithelial bud acquire the expression of suprabasal keratinocytes and keratin 14 expression becomes restricted only to the peripheral cells, by E18, similar to its expression in the basal epidermis (19). Essentially, the mammary bud comes to resemble a simple invagination of skin and fails to give rise to a proper ductal system. Consistent with this observation, microarray analyses of mammary buds at E15 from PTHrP−/− and WT embryos have shown that loss of PTHLH results in downregulation of many genes whose expression distinguishes mammary buds from skin (i.e genes that are normally overexpressed in the mammary buds compared to the developing skin and hair follicles) (22). These genes include epithelial keratin genes (Keratin 4, 6a, 8, 13, 18, 19), transcription factors (Sim2, Sox4, Id1, Tbx3) and growth factor signaling genes (Tachykinin1, Sostdc1, Rspo1), and are likely to point towards important signaling pathways that contribute to mammary epithelial cell specification and maintenance (22).
PTHrP signaling regulates sexual dimorphism
There is a well-described pattern of sexual dimorphism to the development of the embryonic mammary gland in mice. In the developing mammary bud, the epithelial cells induce the expression of the androgen receptor (AR) within the mammary mesenchyme (21, 23). Beginning on day 13 of development, the fetal testes release androgens into the circulation, whose action on the AR in the mammary mesenchyme becomes evident around E14 as it promotes the condensation of the mesenchymal cells around the stalk that connects the mammary bud to the overlying epidermis (24–29). This results in severing of the stalk and the apoptosis of the mesenchymal cells and some of the mammary epithelial cells as well (23). As a result, mammary ducts and nipples do not develop in male mice of most strains (21, 23, 30). We have observed that PTHrP secreted by the epithelium is critical for the destruction of the stalk. In PTHrP−/− and PTH1R−/− embryos, androgen receptors are not expressed in the mesenchyme. Consequently, sexual dimorphism does not occur in these mutants and mammary buds in males retain a stalk like in females (31). Furthermore, we have recently shown that the deletion of β-catenin from the mammary mesenchyme is also associated with a reduction in mesenchymal AR expression and a failure of the androgen-mediated destruction of the mammary bud (17). Therefore, upregulation of mesenchymal Wnt signaling is critical to PTHrP’s ability to orchestrate the sexual dimorphism of the embryonic gland (Fig. 3).
PTHrP signaling promotes mammary bud outgrowth
PTHrP signaling is also required for proper outgrowth of the initial duct system in the embryonic mammary gland. In WT embryos the mammary bud sprouts away from the epidermis, downward towards the developing mammary fat pad around E15-E16 (32). In the absence of Pthlh or Pthr1, the outgrowth of the mammary sprout is arrested and no ductal tree forms despite the presence of the mammary fat pad (12). Outgrowth of the mammary sprouts involves the activation of mesenchymal BMP signaling downstream of PTHrP. In the absence of PTHrP, SMAD phosphorylation is reduced in the mammary mesenchyme, and sprouting of PTHrP−/− mammary buds can be rescued by the addition of BMP4 in mammary bud cultures ex vivo (20). Hens et al. also showed that PTHrP and BMP4 synergize to upregulate matrix metalloprotease 2 (MMP2) and that inhibition of MMP2 impairs mammary sprouting. Taken together, these studies suggest that PTHrP activates BMP signaling to induce MMP2, which facilitates mammary sprouting by remodeling the matrix (22) (Fig. 3). However, disruption of the MMP2 gene does not recapitulate the failure of bud outgrowth as seen in PTHRP−/− or PTHR1−/− embryos, demonstrating that there must be other redundant factors that also mediate the effects of PTHrP signaling on the initial phase of epithelial outgrowth.
PTHrP signaling directs nipple development
In addition to regulating mammary epithelial cell specification and outgrowth, the mammary mesenchyme also communicates with the keratinocytes around the proximal part of the mammary sprout to direct the formation of the nipple sheath. The nipple consists of circular invagination of the basal epidermis as well as a pattern of keratinocyte differentiation that produces a hairless, thickened and hyperkeratotic epidermis onto which the mammary duct opens (33, 34). Loss of PTHrP signaling leads to the failure of nipple development, while conversely, overexpression of PTHrP in keratinocytes recapitulates many of the properties of nipple skin including the suppression of hair follicle development, thickening of the epidermis, and hyperkeratosis characterized by the expanded expression of keratin 1, keratin 14 and fillagrin in the ventral skin (19). The formation of ectopic nipple-like skin in K14-PTHrP mice correlates with the ectopic expression of mammary mesenchymal markers throughout the top layers of the dermal mesenchyme, demonstrating that the epithelial effects of PTHrP overexpression are indirect and are mediated through PTHrP’s actions on mesenchymal cells. PTHrP induces Msx2 expression in mesenchymal cells in vitro and in vivo and loss of Msx2 rescues hair follicle formation in the ventral skin of K14- PTHrP mice. Therefore, PTHrP-induced Msx2 expression in the mammary mesenchyme is critical for the inhibition of hair follicle development in the nipple region (20) (Fig. 3). In addition, loss of Lef1 reverts some aspects of the ventral nipple-skin of K14-PTHrP mice, including epidermal thickness, but does not revert the phenotype completely. It is likely that PTHrP-induced activation of Wnt and Msx2-regulated genes in the mesenchyme is necessary for the secretion of some mesenchymal factor(s) that orchestrates nipple skin development and that a critical threshold of BMP signaling restricts ectopic nipple development to the ventral surface.
Conclusion
In conclusion, PTHrP signals via the PTHR1 to direct the formation of the mammary mesenchyme. Specification of the mammary mesenchyme by PTHrP is a critical step in mammary morphogenesis and is required for sexual dimorphism, sprouting of the mammary bud, maintenance of mammary epithelial fate and formation of the nipple. The effects of PTHrP signaling on these cells are likely mediated by the coordinated actions of multiple downstream pathways including Wnt and BMP signaling. Further investigation of the detailed mechanisms by which PTHrP regulates embryonic mammary gland development is of interest for several reasons. First, it can contribute specifically to a better understanding of embryonic mammary morphogenesis and mammary epithelial and mesenchymal specification as well as serving as a model system in which to study how mesenchymal cells direct epithelial morphogenesis more generally. Second, there is growing awareness of the role of tumor stroma in the regulation of breast cancer progression and metastases. There are many parallels between mammary sprouting and the metastatic cascade. In addition, it has been suggested that the embryonic mammary mesenchyme is similar to tumor-associated stroma (30, 35). Therefore, a detailed understanding of the specification of the mammary mesenchyme and its role in regulating mammary bud outgrowth might shed light on breast tumor progression and/or metastasis. Finally, PTHrP, itself, has been associated with tumor progression in animal models and human patients (36, 37). PTHrP and PTHR1 are often overexpressed in breast cancer and PTHR1 is overexpressed in breast cancer stroma (38–42). Furthermore, the recent iCOGs studies have implicated both the PTHLH and the PTHR1 loci as breast cancer susceptibility genes (43, 44). It is possible that the growth promoting effects of PTHrP on the embryonic mammary epithelium are recapitulated during breast cancer progression. Therefore, understanding how PTHrP regulates normal mammary development is important for understanding how PTHrP and/or the PTHR1 may contribute to breast cancer growth.
Acknowledgements
JW is supported by National Institutes of Health (NIH) grants R01 DK55501, R01 CA153702 and R01 DK077565. MH is supported by the Idaho INBRE Program [P20 RR016454 and P20GM103408] and by a postdoctoral fellowship from the Department of Defense Breast Cancer Research program (W81XWH-09-1-0580). We would like to thank Dr. Kata Granic for providing images of PTHrP-lacZ embryos.
Abbreviations
- PTHrP
Parathyroid hormone related protein
- K14
Keratin 14
- PTHR1
Parathyroid Hormone Receptor
- WT
Wild-type
- AR
Androgen receptor
- ER
Estrogen receptor
- BMP
Bone Morphogenic Protein
- MMP
Matrix metalloprotease
References
- 1.Broadus AE, Mangin M, Ikeda K, Insogna KL, Weir EC, Burtis WJ, et al. Humoral hypercalcemia of cancer. Identification of a novel parathyroid hormone-like peptide. N. Engl J Med. 1988;319(9):556–563. doi: 10.1056/NEJM198809013190906. [DOI] [PubMed] [Google Scholar]
- 2.Philbrick WM, Wysolmerski JJ, Galbraith S, Holt E, Orloff JJ, Yang KH, et al. Defining the roles of parathyroid hormone-related protein in normal physiology. Physiol Rev. 1996;76(1):127–173. doi: 10.1152/physrev.1996.76.1.127. [DOI] [PubMed] [Google Scholar]
- 3.Jüppner H, Abou-Samra AB, Freeman M, Kong XF, Schipani E, Richards J, et al. A G protein-linked receptor for parathyroid hormone and parathyroid hormone-related peptide. Science. 1991;254(5034):1024–1026. doi: 10.1126/science.1658941. [DOI] [PubMed] [Google Scholar]
- 4.McCauley LK, Martin TJ. Twenty-five years of PTHrP progress: from cancer hormone to multifunctional cytokine. J Bone Miner Res. 2012;27(6):1231–1239. doi: 10.1002/jbmr.1617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wysolmerski JJ. Interactions between breast, bone, and brain regulate mineral and skeletal metabolism during lactation. Ann N Y Acad Sci. 2010;1192:161–169. doi: 10.1111/j.1749-6632.2009.05249.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wysolmerski JJ. Parathyroid hormone-related protein: an update. J Clin Endocrinol Metab. 2012;97(9):2947–2956. doi: 10.1210/jc.2012-2142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chen X, Macica CM, Dreyer BE, Hammond VE, Hens JR, Philbrick WM, et al. Initial characterization of PTH-related protein gene-driven lacZ expression in the mouse. J Bone Miner Res. 2006;21(1):113–123. doi: 10.1359/JBMR.051005. [DOI] [PubMed] [Google Scholar]
- 8.Boras-Granic K, Vanhouten J, Hiremath M, Wysolmerski J. Parathyroid hormone-related protein is not required for normal ductal or alveolar development in the post-natal mammary gland. PLoS One. 2011;6(11):e27278. doi: 10.1371/journal.pone.0027278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wansbury O, Mackay A, Kogata N, Mitsopoulos C, Kendrick H, Davidson K, et al. Transcriptome analysis of embryonic mammary cells reveals insights into mammary lineage establishment. Breast Cancer Res. 2011;13(4):R79. doi: 10.1186/bcr2928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dunbar ME, Young P, Zhang JP, McCaughern-Carucci J, Lanske B, Orloff JJ, et al. Stromal cells are critical targets in the regulation of mammary ductal morphogenesis by parathyroid hormone-related protein. Dev Biol. 1998;203(1):75–89. doi: 10.1006/dbio.1998.9029. [DOI] [PubMed] [Google Scholar]
- 11.Dunbar ME, Dann PR, Robinson GW, Hennighausen L, Zhang JP, Wysolmerski JJ. Parathyroid hormone-related protein signaling is necessary for sexual dimorphism during embryonic mammary development. Development. 1999;126(16):3485–3493. doi: 10.1242/dev.126.16.3485. [DOI] [PubMed] [Google Scholar]
- 12.Wysolmerski JJ, Philbrick WM, Dunbar ME, Lanske B, Kronenberg H, Broadus AE. Rescue of the parathyroid hormone-related protein knockout mouse demonstrates that parathyroid hormone-related protein is essential for mammary gland development. Development. 1998;125(7):1285–1294. doi: 10.1242/dev.125.7.1285. [DOI] [PubMed] [Google Scholar]
- 13.Cho YM, Woodard GL, Dunbar M, Gocken T, Jimenez JA, et al. Hair-Cycle-Dependent Catagen Transition. Journal of Investigative Dermatology. 2003;120(5):715–727. doi: 10.1046/j.1523-1747.2003.12147.x. [DOI] [PubMed] [Google Scholar]
- 14.Cormier S, Delezoide AL, Silve C. Expression patterns of parathyroid hormonerelated peptide (PTHrP) and parathyroid hormone receptor type 1 (PTHR1) during human development are suggestive of roles specific for each gene that are not mediated through the PTHrP/PTHR1 paracrine signaling pathway. Gene Expr Patterns. 2003;3(1):59–63. doi: 10.1016/s1567-133x(02)00074-1. [DOI] [PubMed] [Google Scholar]
- 15.Wysolmerski JJ, Cormier S, Philbrick WM, Dann P, Zhang JP, Roume J, et al. Absence of functional type 1 parathyroid hormone (PTH)/PTH-related protein receptors in humans is associated with abnormal breast development and tooth impaction. J Clin Endocrinol Metab. 2001;86(4):1788–1794. doi: 10.1210/jcem.86.4.7404. [DOI] [PubMed] [Google Scholar]
- 16.Voutilainen M, Lindfors PH, Lefebvre S, Ahtiainen L, Fliniaux I, Rysti E, et al. Ectodysplasin regulates hormone-independent mammary ductal morphogenesis via NF-kappaB. Proc Natl Acad Sci U S A. 2012;109:5744–5749. doi: 10.1073/pnas.1110627109. United States. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hiremath M, Dann P, Fischer J, Butterworth D, Boras-Granic K, Hens J, et al. Parathyroid hormone-related protein activates Wnt signaling to specify the embryonic mammary mesenchyme. Development. 2012;139(22):4239–4249. doi: 10.1242/dev.080671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wysolmerski JJ, Broadus AE, Zhou J, Fuchs E, Milstone LM, Philbrick WM. Overexpression of parathyroid hormone-related protein in the skin of transgenic mice interferes with hair follicle development. Proc Natl Acad Sci U S A. 1994;91(3):1133–1137. doi: 10.1073/pnas.91.3.1133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Foley J, Dann P, Hong J, Cosgrove J, Dreyer B, Rimm D, et al. Parathyroid hormone-related protein maintains mammary epithelial fate and triggers nipple skin differentiation during embryonic breast development. Development. 2001;128(4):513–525. doi: 10.1242/dev.128.4.513. [DOI] [PubMed] [Google Scholar]
- 20.Hens JR, Dann P, Zhang JP, Harris S, Robinson GW, Wysolmerski J. BMP4 and PTHrP interact to stimulate ductal outgrowth during embryonic mammary development and to inhibit hair follicle induction. Development. 2007;134(6):1221–1230. doi: 10.1242/dev.000182. [DOI] [PubMed] [Google Scholar]
- 21.Heuberger B, Fitzka I, Wasner G, Kratochwil K. Induction of androgen receptor formation by epithelium-mesenchyme interaction in embryonic mouse mammary gland. Proc Natl Acad Sci U S A. 1982;79(9):2957–2961. doi: 10.1073/pnas.79.9.2957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hens J, Dann P, Hiremath M, Pan TC, Chodosh L, Wysolmerski J. Analysis of gene expression in PTHrP−/− mammary buds supports a role for BMP signaling and MMP2 in the initiation of ductal morphogenesis. Dev Dyn. 2009;238(11):2713–2724. doi: 10.1002/dvdy.22097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sakakura T, Kusano I, Kusakabe M, Inaguma Y, Nishizuka Y. Biology of mammary fat pad in fetal mouse: capacity to support development of various fetal epithelia in vivo. Development. 1987;100(3):421–430. doi: 10.1242/dev.100.3.421. [DOI] [PubMed] [Google Scholar]
- 24.Hoshino k. Development and function of mammary glands of mice prenatally exposed to testosterone propionate. Endocrinology. 1965;76:789–794. doi: 10.1210/endo-76-4-789. [DOI] [PubMed] [Google Scholar]
- 25.Neumann F, Berswordt-Wallrabe RVO, Elger W, Steinbeck H, Hahn JD, Kramer M. Aspects of androgen-dependent events as studied by antiandrogens. Recent Prog Horm Res. 1970;26:337–410. doi: 10.1016/b978-0-12-571126-5.50013-3. [DOI] [PubMed] [Google Scholar]
- 26.Kratochwil K. Development and loss of androgen responsiveness in the embryonic rudiment of the mouse mammary gland. Dev Biol. 1977;61(2):358–365. doi: 10.1016/0012-1606(77)90305-0. [DOI] [PubMed] [Google Scholar]
- 27.Kratochwil K, Schwartz P. Tissue interaction in androgen response of embryonic mammary rudiment of mouse: identification of target tissue for testosterone. Proc Natl Acad Sci U S A. 1976;73(11):4041–4044. doi: 10.1073/pnas.73.11.4041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kratochwil K. In vitro analysis of the hormonal basis for the sexual dimorphism in the embryonic development of the mouse mammary gland. J Embryol Exp Morphol. 1971;25(1):141–153. [PubMed] [Google Scholar]
- 29.Gondos B. Development and differentiation of the testis and male reproductive tract. In: Steinberger A, Steinberger B, editors. Testicular Development, Structure and Function. New York, NY: Raven Press; 1980. pp. 3–20. [Google Scholar]
- 30.Mackie EJ, Chiquet-Ehrismann R, Pearson CA, Inaguma Y, Taya K, Kawarada Y, et al. Tenascin is a stromal marker for epithelial malignancy in the mammary gland. Proc Natl Acad Sci U S A. 1987;84(13):4621–4625. doi: 10.1073/pnas.84.13.4621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dunbar ME, Wysolmerski JJ. Parathyroid hormone-related protein: a developmental regulatory molecule necessary for mammary gland development. J Mammary Gland Biol Neoplasia. 1999;4(1):21–34. doi: 10.1023/a:1018700502518. [DOI] [PubMed] [Google Scholar]
- 32.Veltmaat JM, Mailleux AA, Thiery JP, Bellusci S. Mouse embryonic mammogenesis as a model for the molecular regulation of pattern formation. Differentiation. 2003;71(1):1–17. doi: 10.1046/j.1432-0436.2003.700601.x. [DOI] [PubMed] [Google Scholar]
- 33.Toyoshima Y, Ohsako S, Nagano R, Matsumoto M, Hidaka S, Nishinakagawa H. Histological changes in mouse nipple tissue during the reproductive cycle. J Vet Med Sci. 1998;60(4):405–411. doi: 10.1292/jvms.60.405. [DOI] [PubMed] [Google Scholar]
- 34.Montagna W. Histology and cytochemistry of human skin. XXXV. The nipple and areola. Br J Dermatol. 1970;83(Suppl):2–13. doi: 10.1111/j.1365-2133.1970.tb12859.x. [DOI] [PubMed] [Google Scholar]
- 35.Inaguma Y, Kusakabe M, Mackie EJ, Pearson CA, Chiquet-Ehrismann R, Sakakura T. Epithelial induction of stromal tenascin in the mouse mammary gland: from embryogenesis to carcinogenesis. Dev Biol. 1988;128(2):245–255. doi: 10.1016/0012-1606(88)90288-6. [DOI] [PubMed] [Google Scholar]
- 36.Ghoussaini M, Fletcher O, Michailidou K, Turnbull C, Schmidt MK, Dicks E, et al. Genome-wide association analysis identifies three new breast cancer susceptibility loci. Nat Genet. 2012;44(3):312–318. doi: 10.1038/ng.1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kremer R, Li J, Camirand A, Karaplis AC. Parathyroid hormone related protein (PTHrP) in tumor progression. Adv Exp Med Biol. 2011;720:145–160. doi: 10.1007/978-1-4614-0254-1_12. [DOI] [PubMed] [Google Scholar]
- 38.Henderson MA, Danks JA, Slavin JL, Byrnes GB, Choong PF, Spillane JB, et al. Parathyroid hormone-related protein localization in breast cancers predict improved prognosis. Cancer Res. 2006;66(4):2250–2256. doi: 10.1158/0008-5472.CAN-05-2814. [DOI] [PubMed] [Google Scholar]
- 39.Henderson M, Danks J, Moseley J, Slavin J, Harris T, McKinlay M, et al. Parathyroid hormone-related protein production by breast cancers, improved survival, and reduced bone metastases. J Natl Cancer Inst. 2001;93(3):234–237. doi: 10.1093/jnci/93.3.234. [DOI] [PubMed] [Google Scholar]
- 40.Southby J, Kissin MW, Danks JA, Hayman JA, Moseley JM, Henderson MA, et al. Immunohistochemical localization of parathyroid hormone-related protein in human breast cancer. Cancer Res. 1990;50(23):7710–7716. [PubMed] [Google Scholar]
- 41.Powell GJ, Southby J, Danks JA, Stillwell RG, Hayman JA, Henderson MA, et al. Localization of parathyroid hormone-related protein in breast cancer metastases: increased incidence in bone compared with other sites. Cancer Res. 1991;51(11):3059–3061. [PubMed] [Google Scholar]
- 42.Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, et al. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia. 2004;6(1):1–6. doi: 10.1016/s1476-5586(04)80047-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Garcia-Closas M, Couch FJ, Lindstrom S, Michailidou K, Schmidt MK, Brook MN, et al. Genome-wide association studies identify four ER negative-specific breast cancer risk loci. Nat Genet. 2013;45(4):392–398. doi: 10.1038/ng.2561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Michailidou K, Hall P, Gonzalez-Neira A, Ghoussaini M, Dennis J, Milne RL, et al. Large-scale genotyping identifies 41 new loci associated with breast cancer risk. Nat Genet. 2013;45(4):353–361. doi: 10.1038/ng.2563. [DOI] [PMC free article] [PubMed] [Google Scholar]