Abstract
Lysophosphatidic acid (LPA) is a lipid mediator that acts in paracrine systems via interaction with a subset of G protein-coupled receptors (GPCRs). LPA promotes cell growth and differentiation, and has been shown to be implicated in a variety of developmental and pathophysiological processes. At least 6 LPA GPCRs have been identified to date: LPA1–LPA6. Several studies have suggested that local production of LPA by tissues and cells contributes to paracrine regulation, and a complex interplay between LPA and its receptors, LPA1 and LPA4, is believed to be involved in the regulation of bone cell activity. In particular, LPA1may activate both osteoblasts and osteoclasts. However, its role has not as yet been examined with regard to the overall status of bone in vivo. We attempted to clarify this role by defining the bone phenotype of LPA1(−/−) mice. These mice demonstrated significant bone defects and low bone mass, indicating that LPA1 plays an important role in osteogenesis. The LPA1(−/−) mice also presented growth and sternal and costal abnormalities, which highlights the specific roles of LPA1 during bone development. Microcomputed tomography and histological analysis demonstrated osteoporosis in the trabecular and cortical bone of LPA1(−/−) mice. Finally, bone marrow mesenchymal progenitors from these mice displayed decreased osteoblastic differentiation. These results suggest that LPA1 strongly influences bone development both qualitatively and quantitatively and that, in vivo, its absence results in decreased osteogenesis with no clear modification of osteoclasis. They open perspectives for a better understanding of the role of the LPA/LPA1 paracrine pathway in bone pathophysiology.
Keywords: Lysophosphatidic acid, LPA1, Bone, Osteoblast, Mesenchymal stem cell, Osteogenesis
Introduction
The identification of the mechanisms that promote skeletal growth and bone formation has significant implications in pathophysiology and medicine. Building up adequate bone mass, potentially influenced by genetic factors during development, is essential for a healthy skeleton and subsequent minimal fracture incidence throughout life [1]. With the help of animal models, there is increasing evidence that local molecular factors strongly influence the building up of bone mass through induction and regulation of osteoblast proliferation and differentiation. Lysophosphatidic acid (LPA) is a potent lipid mediator that acts in paracrine systems via interaction with a subset of G protein-coupled receptors (GPCRs). Based on the recently revised international nomenclature, 6 GPCRs have been identified to date for LPA, LPA1–LPA6 [2]. LPA may be present in the systemic circulation in micromolar concentrations, numerous studies having demonstrated that the local production of LPA by tissues and cells highly contributes to paracrine regulation [3–5]. LPA promotes cell growth, motility and differentiation, and has been shown to be implicated in a variety of developmental and pathophysiological processes [6–8]. An increasing interest is attached to the possibility of targeting LPA or LPA receptors for medical purposes [9].
A complex interplay between LPA and its receptors is believed to be involved in the regulation of osteoblast differentiation and bone formation. Thus, LPA induces proliferation of osteoblasts through a pathway that involves Gi proteins and cytosolic calcium [10–12] as well as osteoclast activation [13]. Lysophosphatidic acid synergistically co-operates with calcitriol to promote maturation of the human osteosarcoma cell line, MG63 [14]. In addition, this study demonstrated the dependence on Gi to differentiate mature osteoblasts under LPA. Such an effect of LPA on mature osteoblasts is in agreement with its described role in promoting dendrite outgrowth of the osteocyte-like cell line, MLO-Y4 [15,16]. LPA induces changes in the cytoskeleton and stimulates the migration of MC3T3-E1 osteoblastic cells [17]. It has also been shown to induce membrane blebbing in mouse primary osteoblasts [18]. Moreover, activation of the P2X7 receptor leads to LPA production and increases mineralization [19].
As recently demonstrated, LPA4 inhibits osteoblastic differentiation of stem cells and LPA4-deficient mice have increased bone mass [20]. LPA1 knock-out mice (LPA1(−/−)) exhibit impaired suckling behavior and neurological abnormalities [21]. Nevertheless, the role of the LPA1 receptor has to this day not been extensively examined with regard to the in vivo bone status.
A key point is that, given the complex actions of LPA1 on osteogenesis as well as osteoclasis, and the negative role played by LPA4 on bone mass, the real role of LPA1 remains elusive. We have attempted to clarify the role of the LPA1 receptor by defining the bone phenotype of LPA1(−/−) mice. This study is to our knowledge the first to document the potential role of LPA1 in vivo in bone mass and bone development. These mice were studied by microcomputed tomography (μCT) and histological analysis. Finally, bone marrow mesenchymal progenitors from LPA1(−/−) mice were tested for proliferation rate and osteoblastic differentiation. The overall results clearly suggest a positive role of LPA1 in bone development and bone formation, with LPA1(−/−) mice presenting decreased bone mass and specific skeletal abnormalities. This may open future perspectives targeting LPA receptors for the control of bone mass.
Materials and methods
Animals and reagents
For this study, the LPA1(−/−) mice initially generated by Contos et al. [21]were transferred and maintained in a C57BL/6 background. All data in this study are derived from this strain. Mice were genotyped using PCR analysis of tail or ear DNA to identify homozygous WT (wild-type) or LPA1(−/−) mice as previously described [21]. All experiments were performed in accordance with the principles and guidelines established by the French Institute of Medical Research, INSERM, employing the principles and procedures dictated by the highest standards of humane animal care. Growth evaluation was also performed with the initial 129SvJ/C57BL6 and newly generated knock-out mouse strain. Body length (crown-rump distance) was measured at 1, 2 and 4 weeks of age, and femur and tibia lengths were measured at 2 and 4 weeks.
All reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA), unless otherwise indicated.
Whole mount alizarin red and alcian blue staining
Whole mount skeletal staining was conducted as described previously, with slight modifications [22]. Briefly, mice were anesthetized, eviscerated, skinned, and stained in alcian blue (AB) solution (0.02% AB (w/v), 70% ethanol (v/v), 30% acetic acid (v/v)) for 1 to 2 days at room temperature. Samples were washed through a descending ethanol series, i.e.: 100%, 100%, 95%, 70%, 40%, and 15% ethanol/distilled water (v/v), 1 h each, rinsed twice in distilled water and immerged in distilled water for 1 h. Sample were then treated with 1% (w/v) trypsin, 1 g trypsin digesting 250 g of casein substrate (Invitrogen Molecular Probes, Carlsbad, CA, USA) in 140 mM NaCl solution for 4 h. After rinsing with 140 mM NaCl solution, samples were transferred in 1% (w/v) KOH solution containing 0.1% (w/v) alizarin red S (AR) dye for 24 h at room temperature. Finally, samples were washed through a graded series of 1% (w/v) KOH/glycerol 3/1, 1/1, and 1/3 (v/v), 24 h each, visualized and stored in 100% (v/v) glycerol.
Radiological and histological analysis
Plain radiographs were taken using a soft X-ray apparatus (Softex CMB-2, Softex, Kanagawa, Japan). For histological analysis, some skeletons were fixed in 70° ethanol/distilled water (v/v), embedded in metacrylate and stained with alizarin red according to standard procedures.
High-resolution microcomputed tomography
Femurs and vertebrae of 4-week-old mice were scanned with a high resolution μCT prototype (VivaCT40, Scanco Medical AG, Brüttisellen, Switzerland) as described by Kohlbrenner et al. [23]. At a 3D level, the following calculations were made as previously published [24]: relative bone volume over total bone volume (BV/TV), bone surface over bone volume (BS/BV), trabecular number (Tb.N), trabecular thickness (Tb.Th), trabecular separation (Tb.Sp), and bone mineral density (BMDtrab). Connectivity density (Conn.D) and structure model index (SMI) were calculated without assuming a constant model, as previously described [24]. SMI estimates the plate–rod characteristics of a structure; its value is 0 for an ideal plate and 3 for an ideal rod, with intermediate values reflecting the volume ratio between rods and plates. The geometric degree of anisotropy (DA) is defined as the ratio between the maximal and minimal radius of the mean intercept length (MIL) ellipsoid. DA reduction is correlated with a more isotropic bone structure.
To analyze the femoral cortex, cross-sectional slices were chosen and the following parameters were calculated: cortical thickness (Ct.Th), cortical area (Ct.Ar), marrow area (Ma.Ar), cross-sectional total area (T.Ar), and cortical bone mineral density (BMDcort).
Cell cultures
mBMSC (murine bone marrow stromal cells) were obtained from the bone marrow of femurs and tibias of WT and LPA1(−/−) mice essentially as described elsewhere [25]. Cells were maintained in modified Eagle medium alpha (αMEM) with 10% (v/v) fetal calf serum (FCS) before use. For proliferation assay, cells were seeded at 20000 cells per well in 12-well plates and cultured for up to 10 days in a medium consisting of αMEM supplemented with 10% (v/v) FCS. The cells were harvested 2 days after seeding (D0), and later at D3, D7 and D10. For mineralization assays, cells were cultured for up to 14 days in DMEM with 10% FCS (v/v), 100 μM L-ascorbate 2-phosphate as an osteogenic medium (OM) with 10 mM inorganic phosphate essentially as described elsewhere [25].
DNA assay
Cell layers were washed in PBS, scraped with 0.1% (v/v) NP40 and sonicated. The DNA content was measured using the Picogreen® assay according to the manufacturer’s instructions (Invitrogen Molecular Probes, Carlsbad, CA, USA).
Mineralization assay
Calcium deposits from WT and LPA1(−/−) cultured cells were stained with 40 mM AR solution, pH=4.2, as described previously [26]. Briefly, at days 7 and 14 the medium was removed and wells rinsed twice with phosphate buffer saline (PBS). Cells were fixed in 70% ethanol/distilled water (v/v) for 1 h at 4 °C. AR solution was added to each well for 5 min and rinsed 7 times in order to remove non-specific staining.
Detection of serum biological markers
In order to evaluate their nutritional and mineral status, WT and LPA1(−/−) mice were bled by retro-orbital puncture at 4 weeks. Blood samples were allowed to clot for 30 min and centrifuged for 10 min at 3000 rpm. Levels of serum glucose, iron, albumin, vitamin D, calcium, magnesium, total proteins, phosphorus and CTX-I were measured using conventional enzymatic methods with an Olympus AU400 biochemistry auto-analyzer.
Real time RT-PCR
After removing the bone marrow and grinding the bones, total RNA from WT and LPA1(−/−) radius and humerus was isolated using Trisol solution and quantified using an Agilent Ribogreen assay (Invitrogen Molecular Probes, Carlsbad, CA, USA) as described [27]. RNA was reverse transcribed to cDNA using the SuperArray RT2 First Strand kit (Tebu-bio Laboratories, Le Perray en Yvelines, France) according to the manufacturer’s instructions. Amplification of the cDNA and detection of the target PCR product were conducted in an ABI Prism 7000 sequence detection system (Applied Biosystems, Carlsbad, CA, USA), using the SuperArray Custom RT2 Profiler PCR Array (Tebu-bio Laboratories, Le Perray en Yvelines, France) according to the manufacturer’s instructions. The targets measured included: Col 1, osteocalcin (OC), osterix (OSX), PTHR1 (parathyroid hormone receptor 1), dentin mineral protein 1 (DMP1) and alkaline phosphatase (ALP). RANK (receptor activator of nuclear factor κB), RANKL (RANK ligand), osteoprotegerin (OPG), LPA2, LPA3, LPA4 and autotaxin (ATX) primers were purchased from SABiosciences (Frederick, MD, USA). In order to detect LPA1 expression, forward 5′-ACTGTTAGCACGTGGCTCCT and reverse 3′-GTTGAAAATGGCCCAGAAGA primers were designed with the Primer3 software tool (Biomatters Ltd, Auckland, New Zealand). The RT-PCR results were analyzed using sequence detection software from Applied Biosystems (Carlsbad, CA, USA), and the relative amount of target gene transcript was normalized to the amount of HPRT mRNA control transcript. The data represent results of RNA analyses from 4 different independent experiments obtained with 10 WT and LPA1(−/−) mice. The data shown represent the relative mRNA levels calculated as 2−ΔCt×106 where ΔCt=Ctgene of interest−CtHsp90.
Quantification of LPA
After 12 h of fasting, 4-week-old WT and LPA1(−/−) mice were bled by retro-orbital puncture and the citrated plasma was carefully centrifuged in order to prevent platelet activation and clotting. LPA was butanol-extracted from mouse plasma or bone marrow and quantified using a radioenzymatic assay as described previously [28]. In short, in the presence of [14C] oleoyl-CoA, recombinant rat LPA acyl-transferase selectively catalyzes the transformation of LPA into [14C] phosphatidic acid. Products of the reaction were separated by one-dimensional thin-layer chromatography and autoradiographed. In these conditions, minimal detection of LPA was 0.2 pmol.
Infrared spectroscopy
Infrared spectra were recorded from KBr pellets on a Nicolet Thermo Avatar 320 FTIR spectrometer between 400 and 4000 cm−1. After removing the bone marrow, each sample of tibia and femur cortical bone tissue was ground into 100 mg of IR-grade KBr prior to pelletization. For each spectrum, intensities of the bands between 1670 and 1500 cm−1 (which represent vibrations of the collagenous matrix) were compared to those at 1034, 607, and 572 cm−1 (corresponding to the phosphate vibrations of the apatite phase) in order to determine the degree of mineralization of the bone tissue [29,30].
Statistical analysis
Results are expressed as means±(SD). Groups were compared using Mann–Whitney analyses as appropriate. A p value <0.05 was considered statistically significant.
Results
Skeletal development is altered in LPA1(−/−) mice
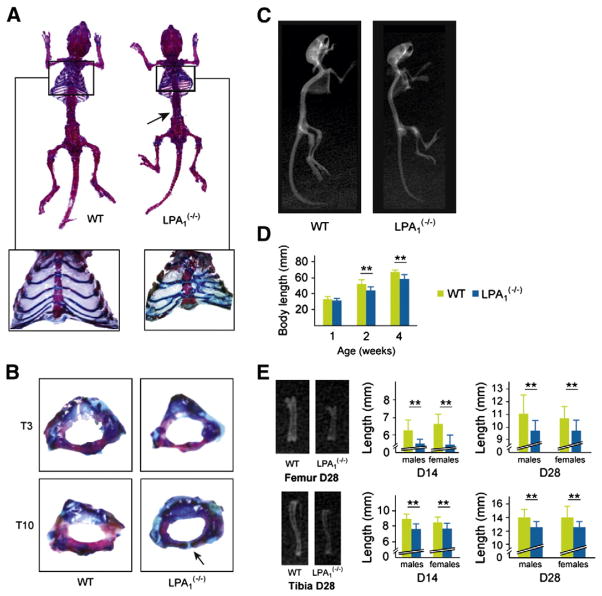
Skeletal preparations of 4-week-old mice stained with alcian blue and alizarin red showed homogeneous dwarfism in LPA1(−/−) mice, as previously described [21] (Fig. 1A). LPA1(−/−) mice also displayed previously described cranial deformities including shorter snouts and more widely spaced eyes compared with control siblings. Strikingly, 100% of the LPA1(−/−) mice analyzed also displayed rib cage deformity. All mice presented several sterno-distal rib fusions (Fig. 1A, close-up). However, the abnormal patterning was not specific to a single pair of ribs and sometimes affected two consecutive costal elements. In addition, the sternebrae, or segments of the sternum, of LPA1(−/−) mice were shorter, crooked and thicker. Some were even triangular, and the number (normally 7) was typically reduced to 6. The sternum thus appeared sinusoidal (Fig. 1A, close-up), suggesting that the LPA1 receptor plays a role in the development of sternal cartilage and bone. However, the 1st and 2nd sterno-distal ribs were unaffected. The number and shape of ribs were normal in the proximal and vertebro-distal parts of the dorsal segment. Taken together, these observations suggest that the ventral mesenchymal and costo-vertebral connections are altered due to the absence of LPA1. Dorsal ossification of the ribs does not appear to be affected.
Fig. 1.
Gross phenotype of LPA1(−/−) mice. Alizarin red S (AR) and alcian blue (AB) staining and X-ray analysis of LPA1(−/−) and WT littermates. (A) Skeletal staining of 4-week-old LPA1(−/−) and WT mice showing homogeneous dwarfism in LPA1(−/−) mice (n=10). These mice also show increased AB staining suggesting an alteration in the bone mineralization process. Close-up AR/AB staining of LPA1(−/−) and WT mice rib cages demonstrates multiple sterno-distal rib fusions and sternebrae abnormalities. Arrow: AB staining of intervertebral discs and vertebral extremities. (B) At D14, LPA1(−/−) thoracic vertebrae are smaller and not fully mineralized (arrow) when compared with WT littermates. (C) Profile whole-body X-ray analysis of the skeleton of 4-week-old LPA1(−/−) and WT mice showing homogeneous dwarfism and less mineralization in LPA1(−/−) vertebrae and long bones. (D) Growth curves of 1-, 2-, and 4-week-old LPA1(−/−) mice (black columns) and WT mice (white columns) (n=10, 15 and 10, respectively). (E) Representative X-ray analysis of 4-week-old LPA1(−/−) and WT mice femurs (upper left) and tibias (lower left) showing size and mineralization differences. Femur lengths of 2- and 4-week-old male and female LPA1(−/−) mice (blue columns) and WT mice (green columns) (n=6) (upper middle and right). Tibia lengths of 2- and 4-week-old male and female LPA1(−/−) mice (black columns) and WT mice (white columns) (n=6) (lower middle and right). LPA1(−/−) femurs and tibias are smaller and less mineralized in the cortical and trabecular areas of the bone than WT littermates. Values are means (SD). Significant statistical differences between groups: **p<0.01, LPA1(−/−) vs. WT by the Mann–Whitney test.
Most interestingly, we observed delayed vertebral calcification and closure in the thoracic spine of LPA1(−/−) mice at 2 weeks of age (Fig. 1B, arrow). Complete ossification was however achieved 2 weeks later. All vertebral extremities remained thicker in the 4-week-old LPA1(−/−) mice (blue stain, Fig. 1A, arrow), also indicating growth plate immaturity.
Thus, the overall phenotype of LPA1(−/−) mice indicates that LPA1 plays a role in the processes of skeletal development, especially those involving sternocostal and vertebral elements.
LPA1(−/−) mice exhibit abnormal growth
LPA1(−/−) mice were described as having short snouts and small heads in the original study [21]. Radiographic analysis of 4-week-old LPA1(−/−) mice confirmed this craniofacial phenotype (Fig. 1C). In addition, the short stature of LPA1(−/−) mice was due to growth defects in the limbs as well as the vertebrae (Figs. 1C–E). Growth retardation was moderate at 1 week of age, the pups having a crown-rump length approximately 95% that of WT pups (31.6 (±3.63) vs. 33.2 (±5.63) mm, respectively) (Fig. 1D). Dwarfism became more apparent as the LPA1(−/−) mice grew. The difference in crown-rump lengths between LPA1(−/−) mice and their WT littermates reached 15.7% at 2 weeks after birth (44.6 (±4.45) vs. 52.9 (±6.12), p<0.001), and 13.1% at 4 weeks (58.8 (±12.4) vs. 67.7 (±5.06), p<0.004) (Fig. 1D). Anthropometric analysis using soft X-rays showed decreased length of a number of skeletal components, particularly limb bones, in LPA1(−/−) mice (Fig. 1E). The femoral and tibial longitudinal lengths were decreased by 20% (6.55 mm(±0.46) vs. 5.47 (±0.36), p<0.001) and by 16% (8.85 mm (±0.59) vs. 7.65 (±0.52), p<0.001), respectively, in 2-week-old LPA1(−/−) mice (Fig. 1E). The difference remained significant at 4 weeks of age (Fig. 1E). No differences in lengths were observed between males and females at any stage. Lower mineral content was also apparent in the femur and tibia of LPA1(−/−) mice at 4 weeks, especially in cortical bone (Figs. 1E, left). The profile X-ray analysis of the LPA1(−/−) mice suggested decreased mineralization of the upper part of the spine (Fig. 1C).
Altogether, the skeletal abnormalities of the ribs, limbs and vertebrae, and the lower mineral content observed were highly suggestive of altered ossification in LPA1(−/−) mice.
LPA1(−/−) mice display decreased vertebral and femoral trabecular bone
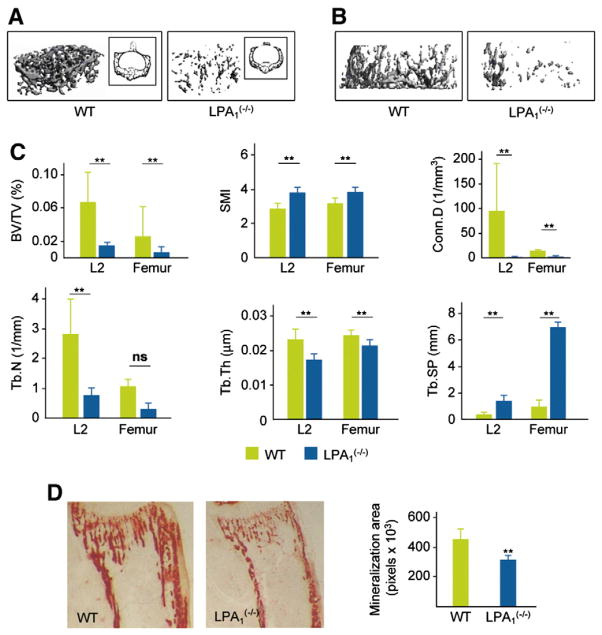
The bone phenotype of LPA1(−/−) mice was further characterized by μCT analysis at 4 weeks of age. The findings of examination of vertebral and femoral microarchitecture are illustrated in Fig. 2. Measurements demonstrated a dramatically decreased bone volume over total volume (BV/TV) value in the trabecular bone of the vertebrae and femurs (81%, p=0.008 and 77%, p=0.009, respectively) (Figs. 2A, B, and C). This was essentially due to a reduction in trabecular number and thickness. Decreased connectivity and increased trabecular spacing were also observed (Fig. 2C). Similar changes were observed in the proximal femurs of LPA1(−/−) mice, with a 62% reduction of BV/TV (p=0.05) and a 58% decrease in trabecular number (p=0.05) (Fig. 2C). SMI was higher in LPA1(−/−) mice, indicative of a rod-like structure. Altogether, these results demonstrated significantly decreased bone mass in the vertebral and femoral trabecular bone of LPA1(−/−) mice. Alizarin red quantification of mineralized tissue in the proximal tibia also demonstrated a 30% decrease in LPA1(−/−) mice (n=6, p<0.002) (Fig. 2D).
Fig. 2.
Altered vertebral and femoral trabecular bone of LPA1(−/−) mice. (A) μCT visualization of L2 vertebra in 4-week-old LPA1(−/−) mice and WT littermates (n=6). (B) μCT visualization of trabecular area of the femur in 4-week-old LPA1(−/−) mice and WT littermates (n=6). (C)High resolution μCT parameters were measured in LPA1−/− and WT mice (n=6). Values analyzed in L2 vertebrae and the trabecular area of the femur were BV/TV, bone volume/total volume; Tb.Th, trabecular thickness; Tb.N, trabecular number; Tb.Sp, trabecular spacing; Conn.D, connectivity density; SMI, structure model index. (D) Alizarin red (AR) staining of the proximal extremity of the tibia in 2-week-old LPA1(−/−) mice and WT littermates. Mineralization area was quantified in pixels, LPA1(−/−) (blue columns) and WT (green columns) (n=4). Values are means (SD). Significant statistical differences between groups: *p<0.05, **p<0.01 for LPA1(−/−) vs. WT by the Mann–Whitney test.
The cortical bone of LPA1(−/−) mice also showed a 32% reduction in thickness (Figs. 3Aand B). Cortical area (Ct.Ar) was significantly decreased by 37%, with marrow area (Ma.Ar) remaining unchanged (Fig. 3B).
Fig. 3.

Altered femoral cortical bone of LPA1(−/−) mice. (A) μCT visualization of the cortical region of the femur in 4-week-old LPA1(−/−) mice and WT littermates (n=6). (B) High resolution μCT parameters were measured in femurs of six 4-week-old LPA1 −/− and WT mice as described in Materials and methods. Values analyzed were Ct.Th, cortical thickness; Ct.Ar, cortical area; Ma.Ar, marrow area; T.Ar, cross-sectional total area. Values are means (SD). Significant statistical differences between groups: *p<0.05, **p<0.001 for LPA1(−/−) vs. WT by the Mann–Whitney test.
The overall result of μCT studies demonstrated decreased bone mass in LPA1(−/−) mice in both trabecular and cortical bone.
Material bone mineral density is not altered in LPA1(−/−) mice
Altered bone density as observed by X-ray analysis may be variably due to defective mineralization (osteomalacia) and/or decreased bone mass (osteoporosis). The degree of mineralization of a given volume of bone (material density)[31] can, in first approach, be appreciated by μCT analysis. As shown in Fig. 4B, the material bone mineral density measured by μCT was not modified in the femoral cortex and vertebral trabecular bone of LPA1(−/−) mice.
Fig. 4.
Bone mineral density and mineral and nutritional status in LPA1(−/−) mice. (A) μCT mineral density is conserved in LPA1(−/−) mice. High resolution μCT parameters were measured in LPA1 −/− and WT mice (n=6). Vertebral and femoral trabecular bone mineral density, BMDtrab, and femoral cortical bone mineral density, BMDcort, were analyzed. (B) Nutritional and mineral status of LPA1(−/−) mice. Values of serum glucose (mmol/l), albumin and total proteins (g/l), 25 OH vitamin D (ng/ml), iron (mg/l). These values were similar in the LPA1(−/−) mice and WT littermates (n=10). Values of total calcium, phosphate, and magnesium (mmol/l). Values were similar in the LPA1(−/−) mice and WT littermates (n=10). (C) Infrared spectra of the cortical area of femurs and tibiae did not show any clear difference between 4-week-old LPA1(−/−) (dotted line) and WT mice (black line) (n=8). (D) Ratio of the absorbance at 1034 and 1660 cm−1, which reflects mineralization status (ratio of the mineral to organic phases) showed no differences between LPA1(−/−) and WT mice (n=8). Values are means (SD). Significant statistical differences between groups: *p<0.05, **p<0.001 of LPA1(−/−) vs. WT mice by the Mann–Whitney test.
In order to confirm this result, we measured the mineral content of LPA1(−/−) mice by infrared spectroscopy (Figs. 4C and D). Comparison of the relative intensities of the vibration bands produced by the collagenous phase with those of the mineral phase showed that mineralization of the cortical bone was similar in LPA1(−/−) and WT mice (Fig. 4D). These observations were in agreement with the μCT data, suggesting decreased bone content (osteoporosis) with unchanged mineralization in LPA1(−/−) mice. These data precluded the hypothesis that osteomalacia alone could be responsible for the alteration of the bone tissue of LPA1(−/−) mice.
Since decreased bone mass and/or mineralization may be secondary to alteration of nutritional status as well as calcium, phosphate, and vitamin D intake, we also investigated nutritional and mineral parameters in 4-week-old LPA1(−/−) mice. Glucose, albumin, iron, and total protein plasma levels were similar in LPA1(−/−) and WT mice, suggesting that the overall nutritional status of the surviving mice was not different from their WT littermates (Fig. 4A, upper). Moreover, no differences between LPA1(−/−) and WT mice were observed (Fig. 4B, lower), ruling out disturbance of mineral homeostasis as a factor responsible for alteration of the skeletal phenotype.
Expression of osteoblast differentiation markers is decreased in LPA1(−/−) mice
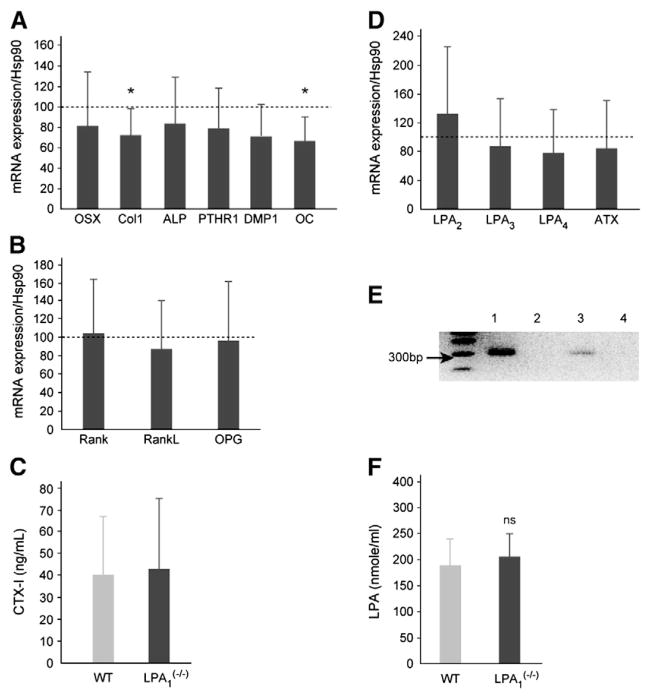
In order to better evaluate the impact of LPA1 receptor deletion on the transcription of osteoblastic differentiation genes, the RNA levels of several bonemarkers were quantified by RT-PCR in the radius and femur (Fig. 5A). Most of the bone formation markers analyzed were decreased. The early indicator of osteoblast differentiation, collagen 1, was significantly decreased by 28% (p=0.04). Osteocalcin levels were also significantly decreased by 35% (p=0.02). Other markers of osteoblast differentiation, i.e. osterix, PTHR1 and DMP1, were also decreased in the bone of LPA1(−/−) mice, by 20%, 22% and30%, respectively, but this did not reach significance (Fig. 5A). Interestingly, the mRNA level of RUNX2 (runt-related transcription factor 2), a transcription factor required for initial mesenchymal stem cell differentiation toward the osteoblastic lineage and acting upstream of osterix, was unchanged in LPA1(−/−) mice compared with their WT littermates (data not shown).
Fig. 5.
Expression of bone markers and LPA receptors in LPA1(−/−) mice. (A) Real time RT-PCR of bone markers in long bones of LPA1(−/−) mice: RNAs were extracted from the humerus and radius of 4-week-old LPA1(−/−) mice and WT littermates (n=9). Expression of collagen 1a (Col 1), alkaline phosphatase (ALP), osterix (OST), osteocalcin (OC), PTH receptor 1 (PTHR1) and dentin matrix protein 1 (DMP1) was tested. Dashed line represents the level normalized to 100% of values observed in WT mice. Columns represent the level of gene expression in LPA1(−/−) mice. (B) Expression of receptor activator of nuclear factor κB (RANK), receptor activator of nuclear factor κB-ligand (RANKL) and osteoprotegerin (OPG) was tested. Dashed line represents the level normalized to 100% of values observed in WT mice. Columns represent the level of gene expression in LPA1(−/−) mice. (C) Values of serum CTX-I (ng/mL), reflecting collagen I degradation, were similar in LPA1(−/−) mice and WT littermates (n=7). (D) Expression of LPA2, LPA3, LPA4 and autotaxin (ATX) was assessed by real time RT-PCR. Dashed line represents the level normalized to 100% of values observed in the WT. Columns represent the level of gene expression in LPA1(−/−) mice. (E) PCR detection of LPA1 receptor in WT and LPA1(−/−) mice. Lanes 1 and 2 represent the genomic detection by PCR of LPA1 receptor in DNA of WT and LPA1(−/−) mice, respectively. Lanes 3 and 4 represent the detection by RT-PCR of LPA1 receptor in bones of WT mice of LPA1(−/−) mice respectively.(F) LPA concentration in plasma from LPA1(−/−) and WT mice. LPA concentration was evaluated in fasting mice as described in Materials and methods. Values are not significantly different between LPA1(−/−) and WT mice. Values are means (SD). Significant statistical differences between groups: *p<0.05 of LPA1(−/−) vs. WT by the Mann–Whitney test.
Taken together, these data demonstrate that some markers of osteoblastic differentiation, collagen 1 and osteocalcin, are significantly less expressed in LPA1(−/−) mouse bones. All markers demonstrated a tendency to decrease, suggesting that the overall osteoblastic differentiation process is altered in LPA1(−/−) mice.
In order to document the level of remodeling, we also investigated the expression of RANK, RANKL and OPG in WT and LPA1(−/−) mouse bones. The mRNA level of these genes was similar in LPA1(−/−) mice and WT littermates (Fig. 5B), indicating that osteoclasis, at least in long bones at this stage of development, is unaffected by the loss of the LPA1 receptor and is therefore not responsible for the altered bone mass. In keeping with these observations, the serum level of CTX-I, a marker of collagen 1 degradation, was unchanged in LPA1(−/−) mice compared with WT littermates (Fig. 5C).
Other LPA receptors, especially LPA4, may influence bone differentiation and bone mass [20]. Expression of LPA receptors was therefore also evaluated by RT-PCR in femurs and tibias of LPA1(−/−) mice. Expression of autotaxin (ATX), a major enzyme involved in LPA production, was also quantified. Expression of LPA2 LPA3 and LPA4 and ATX was similar in LPA1(−/−) mice and WT littermates (Fig. 5D). As a control, Fig. 5E demonstrates that, as expected, LPA1 is expressed in bones of WT mice but not in the LPA LPA1(−/−) strain. LPA concentration in plasma did not differ between LPA1(−/−) mice and WT littermates (209 (±67) vs 189 (±70) pmol/ml, n=4, NS, in LPA1(−/−) mice and WT littermates respectively (Fig. 5F).
Absence of LPA1 therefore seems specifically to influence in vivo bone formation, with no significant effect on bone resorption. In addition, the bone phenotype of LPA1(−/−) mice does not seem due to a modified expression of other LPA receptors or a variation of LPA production.
LPA1(−/−) mBMSC exhibit impaired proliferation and differentiation processes in vitro
To further investigate whether the impaired bone formation is the result of defective osteoblast proliferation and differentiation, we cultured murine bone marrow mesenchymal stem cells (mBMSC) from LPA1(−/−) and WT mice. The proliferation rate of mBMSC was tested in basal medium with 10% FCS for 3, 7 and 10 days (Fig. 6A). After decreasing at D3, the DNA content of WT mBMSC increased significantly at D7 and D10. The DNA content was dramatically decreased in LPA1(−/−) mBMSC (Fig. 6A).
Fig. 6.

LPA1(−/−) mBMSC display decreased proliferation and mineralization. (A) WT and LPA1(−/−) mBMSC were seeded and cultured for 2 days (D0) in medium with 10% FCS (see Materials and methods) and tested at D0, D3, D7 and D10, and DNA content was measured using the Picogreen® assay. (B) mBMSC were cultured in osteogenic medium for 7, 10, or 14 days and stained with alizarin red S solution to evaluate mineralization. Lower mineralization at D7 and D10 was observed in LPA1(−/−) mBMSC cultures. Values are means (SD). Significant statistical differences between groups: ##p<0.001 ofWTD0 vs. D7 or D10; *p<0.05, **p<0.001 of LPA1(−/−) vs. WT by the Mann–Whitney test.
The osteoblastic differentiation of WT and LPA1(−/−) mBMSC was also tested in osteogenic medium. Alizarin red S staining showed highly decreased mineralization in LPA1(−/−) cultured mBMSC (Fig. 6B).
Discussion
Several in vitro studies have so far demonstrated that LPA is involved both in osteoblastic differentiation [10–12,14,15,18,19] and in osteoclast activity [13]. LPA interacts with a set of G-protein-coupled receptors, such as LPA1 and LPA4, which are expressed in bone cells. While LPA4, likely associated to increase of cAMP [9], inhibits osteogenic differentiation [20], so far available in vitro data strongly suggests that LPA1 has a possible role in promoting bone formation. The current model in fact supports the hypothesis of LPA production by osteoblasts, possibly promoting both osteoblastic differentiation and osteoclasis [13]. Therefore, the absence of LPA1 may theoretically contribute to either increase or decrease the overall bone mass. The results presented here demonstrate that LPA1(−/−) mice display bone abnormalities and osteoporosis, suggesting a prominent role of LPA1 in osteogenesis.
The bone developmental abnormalities observed in LPA1(−/−) mice affected the ribs and vertebrae. Attachment of the ribs to the sternum was abnormal, with fusion of the ribs observed in all LPA1(−/−) mice. Fused ribs are observed in a variety of conditions, for example disruption of the transcription factor Hoxa-9which results in fusion of the first and second ribs [32,33], and more recent data have described multiple developmental factors involved in rib formation. In particular, Hand2 overexpression resumes some of the abnormalities observed in LPA1 mice [34]. Nevertheless, absence of the bone morphogenetic proteins BMP-4 and BMP-7 is also responsible for abnormal costo-sternal connections [35]. There is to our knowledge no published data indicating that LPA1 interferes with such processes and, so far, no identified disease clearly resumes the abnormalities observed in LPA1(−/−) mice.
LPA1(−/−) mice also demonstrated low trabecular and cortical bone mass. These mice had low trabecular bone volume and decreased trabecular number and thickness, both vertebral and femoral trabecular bones being affected. The altered SMI was also indicative of a potentially fragile network. Femoral cortical thickness was also significantly decreased in LPA1(−/−) mice. They were therefore severely affected by a global bone defect, suggestive of osteoporosis. Notably, LPA1(+/−) animals were fully exempt from any abnormal bone phenotype, and no significant differences between male and females were observed. Indeed, the growth phenotype, affecting vertebrae and limbs, is suggestive of abnormal endochondral ossification. However cortical thickness was also decreased, suggesting an overall alteration of osteogenesis in the absence of LPA1. In accordance with this finding, the decreased expression of early and late markers of osteoblastic differentiation suggests that LPA1 is significantly involved in bone formation.
The findings obtained with cultured mBMSC are consistent with the data observed in vivo. The LPA1(−/−) mBMSC demonstrated a highly decreased proliferation rate, in keeping with the effect of LPA1(−/−) as a growth factor in osteoblastic models [11,12]. The decreased mineralization of LPA1(−/−) mBMSC suggests that their decreased proliferation also impairs their further mineralization. These results, which show the specific impact of the absence of LPA1 in bone progenitors, are consistent with previous reports obtained in osteoblastic cell lines [14,18,19,36].
Interestingly, as demonstrated by μCT and infrared analysis, the bone mineral deficiency observed in LPA1(−/−) mice seems related to true osteoporosis without a mineralization deficiency as such. The mineral density measured by μCT in both trabecular and cortical bone was not significantly decreased in LPA1(−/−) mice. The ratio of mineralized to organic phase measured by infrared analysis, as a reflect of the material bone mineral density, was even slightly higher in the bones of LPA1(−/−) mice, which may be indicative of a compensatory mechanism to osteoporosis or alteration of mineralization kinetics as observed in osteogenesis imperfecta [37,38]. In parallel, the major parameters involved in the overall control of bone mass, including calcium, phosphate, and vitamin D, were not modified in LPA1(−/−) mice, nor were nutrition parameters.
Of particular interest is the surprising interplay of the different LPA receptors that are thought to act during osteogenesis and are potentially involved in the regulation of bone mass. Recent studies and our data suggest that LPA1 and LPA4 receptors display completely opposite effects on the development of bone mass, with LPA4 exerting a negative effect [20] and LPA1 a positive one. LPA1 has been described as activating several intracellular cascades, namely Gi, Gq and G12/13 pathways [6]. The Gi pathway is usually associated with an increased proliferation rate and seems involved in osteoblastic proliferation under LPA [11,12]. Nevertheless, overexpression of the Gi pathway has also been recently associated with decreased osteogenesis [39]. Therefore, it can be hypothesized that LPA1 participation in tuning of the Gi pathway is necessary to optimize osteoblastic proliferation and differentiation and eventually osteogenesis.
The effect of LPA1 on bone resorption has been documented in the bone metastasis process [40], and a role of the LPA1/Gi pathway in osteoclast activation has recently been demonstrated [13]. Here, the overall effect in vivo of LPA1 was to decrease bone mass and, supposedly, to lower the osteogenesis rate.
Indeed, the potential effect of LPA on osteogenesis or osteoclasis may depend on the respective expression of LPA receptors by bone cells and the concentration of LPA in the tissue. In this view, the potency of specific molecular species of LPA towards LPA1 and LPA4 is variable [9]. In parallel, distinct mechanisms may be involved in the control of the paracrine secretion of LPA [3–5], several data supporting the hypothesis of LPA production in bone tissue itself [19,40]. Osteoblasts secrete LPA through activation of phospholipase C and phospholipase A2 pathways [19]. We also observed that human MSC also produce significant levels of secreted LPA (personal unpublished results). In addition, platelets may contribute to significant increase of the local concentration of LPA in bone tissues [5,40]. In this view, we found no indication in our study of a modification of either the level of circulating LPA or the expression of autaxin, the main enzyme involved in LPA production, in LPA1(−/−) mice. In parallel, the expression of other LPA receptors was not modified in the bone tissue of LPA1(−/−) mice, suggesting that their bone phenotype, and osteoporosis in particular, can be attributed to the absence of LPA1 alone.
Conclusion
In summary, the results of our study help to clarify the role of LPA1 in vivo. LPA1(−/−) mice display defects in bone formation with osteoporosis as consequence, as well as specific developmental abnormalities, indicating that the LPA1 receptor is significantly involved in osteogenesis. Notably, the active emergence of therapeutics involving lysophospholipids and their receptors could include bone as a future target of LPA receptor modulation.
Acknowledgments
We thank N. Laroche, of INSERM U890 and Saint-Etienne University, for technical assistance in μCT analysis. We thank E. Gouze of INSERM UMR 1043 for critical discussion. We also thank A. Tridon of the animal research facility and C. Carriven, A. Bros, M. Nieto and L. Micheletti for their technical support. We thank J.S. Saulnier-Blache for providing us with recombinant rat LPA acyl-transferase.
Footnotes
Funding sources: This work was supported by grants from the French Program of Hospital Clinical Research (PHRC, AOL 0304602), from the Conseil Régional de Midi-Pyrénées (Cellular and Gene Therapy Program) and from the Etablissement Français du Sang (EFS) (Program 2003.02). J. Chun was supported by grants from the NIH: MH051699 and NS048478. S. Laurencin-Dalicieux is the recipient of a grant provided by Pfizer France. R. Oreffo is supported by the Biotechnology and Biological Sciences Research Council. J.P. Salles was supported by a grant provided by Pfizer France (convention 08766A10).
Conflict of interest. All authors have no conflict of interest.
Contributor Information
Isabelle Gennero, Email: gennero.i@chu-toulouse.fr.
Sara Laurencin-Dalicieux, Email: sara.laurencin@inserm.fr.
Françoise Conte-Auriol, Email: crc@chu-toulouse.fr.
Fabienne Briand-Mésange, Email: fabienne.briand-mesange@inserm.fr.
Danielle Laurencin, Email: danielle.laurencin@univ-montp2.fr.
Jackie Rue, Email: rue@cict.fr.
Nicolas Beton, Email: nicolas.beton@inserm.fr.
Nicole Malet, Email: nicole.malet@inserm.fr.
Marianne Mus, Email: marianne.mus@inserm.fr.
Akira Tokumura, Email: tokomura@ph.tokushima-u.ac.jp.
Philippe Bourin, Email: philippe.bourin@efs.sante.fr.
Laurence Vico, Email: vico@univ-st-etienne.fr.
Gérard Brunel, Email: brunel@cict.fr.
Richard O. C. Oreffo, Email: roco@soton.ac.uk.
Jerold Chun, Email: jchun@scripps.edu.
Jean Pierre Salles, Email: salles.jp@chu-toulouse.fr.
References
- 1.Ralston SH, Uitterlinden AG. Genetics of osteoporosis. Endocr Rev. 2010;31:629–62. doi: 10.1210/er.2009-0044. [DOI] [PubMed] [Google Scholar]
- 2.Chun J, Hla T, Lynch KR, Spiegel S, Moolenaar WH International Union of Basic and Clinical Pharmacology. LXXVIII. Lysophospholipid receptor nomenclature. Pharmacol Rev. 2010;62:579–87. doi: 10.1124/pr.110.003111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fourcade O, Simon MF, Viode C, Rugani N, Leballe F, Ragab A, et al. Secretory phospholipase A2 generates the novel lipid mediator lysophosphatidic acid in membrane microvesicles shed from activated cells. Cell. 1995;80:919–27. doi: 10.1016/0092-8674(95)90295-3. [DOI] [PubMed] [Google Scholar]
- 4.Gaits F, Fourcade O, Le Balle F, Gueguen G, Gaige B, Gassama-Diagne A, et al. Lysophosphatidic acid as a phospholipid mediator: pathways of synthesis. FEBS Lett. 1997;410:54–8. doi: 10.1016/s0014-5793(97)00411-0. [DOI] [PubMed] [Google Scholar]
- 5.Ferry G, Tellier E, Try A, Gres S, Naime I, Simon MF, et al. Autotaxin is released from adipocytes, catalyzes lysophosphatidic acid synthesis, and activates preadipocyte proliferation. Up-regulated expression with adipocyte differentiation and obesity. J Biol Chem. 2003;278:18162–9. doi: 10.1074/jbc.M301158200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Noguchi K, Herr D, Mutoh T, Chun J. Lysophosphatidic acid (LPA) and its receptors. Curr Opin Pharmacol. 2009;9:15–23. doi: 10.1016/j.coph.2008.11.010. [DOI] [PubMed] [Google Scholar]
- 7.Choi J, Herr D, Noguchi K, Yung Y, Lee C, Mutoh T, et al. LPA receptors: subtypes and biological actions. Annu Rev Pharmacol Toxicol. 2010;50:157–86. doi: 10.1146/annurev.pharmtox.010909.105753. [DOI] [PubMed] [Google Scholar]
- 8.Lin ME, Herr DR, Chun J. Lysophosphatidic acid (LPA) receptors: signaling properties and disease relevance. Prostaglandins Other Lipid Mediat. 2010;91:130–8. doi: 10.1016/j.prostaglandins.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tigyi G. Aiming drug discovery at lysophosphatidic acid targets. Br J Pharmacol. 2010;161:241–70. doi: 10.1111/j.1476-5381.2010.00815.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Caverzasio J, Palmer G, Suzuki A, Bonjour JP. Evidence for the involvement of two pathways in activation of extracellular signal-regulated kinase (Erk) and cell proliferation by Gi and Gq protein-coupled receptors in osteoblast-like cells. J Bone Miner Res. 2000;15:1697–706. doi: 10.1359/jbmr.2000.15.9.1697. [DOI] [PubMed] [Google Scholar]
- 11.Grey A, Banovic T, Naot D, Hill B, Callon K, Reid I, et al. Lysophosphatidic acid is an osteoblast mitogen whose proliferative actions involve G(i) proteins and protein kinase C, but not P42/44 mitogen-activated protein kinases. Endocrinology. 2001;142:1098–106. doi: 10.1210/endo.142.3.8011. [DOI] [PubMed] [Google Scholar]
- 12.Ahmed I, Gesty-Palmer D, Drezner MK, Luttrell LM. Transactivation of the epidermal growth factor receptor mediates parathyroid hormone and prostaglandin F2 alpha-stimulated mitogen-activated protein kinase activation in cultured transgenic murine osteoblasts. Mol Endocrinol. 2003;17:1607–21. doi: 10.1210/me.2002-0040. [DOI] [PubMed] [Google Scholar]
- 13.Lapierre DM, Tanabe N, Pereverzev A, Spencer M, Shugg RPP, Dixon SJ, et al. Lysophosphatidic acid signals through multiple receptors in osteoclasts to elevate cytosolic calcium concentration, evoke retraction, and promote cell survival. J Biol Chem. 2010;285:25792–801. doi: 10.1074/jbc.M110.109322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gidley J, Openshaw S, Pring ET, Sale S, Mansell JP. Lysophosphatidic acid cooperates with 1alpha,25(OH)2D3 in stimulating human MG63 osteoblast maturation. Prostaglandins Other Lipid Mediat. 2006;80:46–61. doi: 10.1016/j.prostaglandins.2006.04.001. [DOI] [PubMed] [Google Scholar]
- 15.Karagiosis SA, Karin NJ. Lysophosphatidic acid induces osteocyte dendrite outgrowth. Biochem Biophys Res Commun. 2007;357:194–9. doi: 10.1016/j.bbrc.2007.03.121. [DOI] [PubMed] [Google Scholar]
- 16.Waters KM, Jacobs JM, Gritsenko MA, Karin NJ. Regulation of gene expression and subcellular protein distribution in MLO-Y4 osteocytic cells by lysophosphatidic acid: relevance to dendrite outgrowth. Bone. 2011;48:1328–35. doi: 10.1016/j.bone.2011.02.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Masiello LM, Fotos JS, Galileo DS, Karin NJ. Lysophosphatidic acid induces chemotaxis in MC3T3-E1 osteoblastic cells. Bone. 2006;39:72–82. doi: 10.1016/j.bone.2005.12.013. [DOI] [PubMed] [Google Scholar]
- 18.Panupinthu N, Zhao L, Possmayer F, Ke HZ, Sims SM, Dixon SJ. P2X7 nucleotide receptors mediate blebbing in osteoblasts through a pathway involving lysophosphatidic acid. J Biol Chem. 2007;282:3403–12. doi: 10.1074/jbc.M605620200. [DOI] [PubMed] [Google Scholar]
- 19.Panupinthu N, Rogers JT, Zhao L, Solano-Flores LP, Possmayer F, Sims SM, et al. P2X7 receptors on osteoblasts couple to production of lysophosphatidic acid: a signaling axis promoting osteogenesis. J Cell Biol. 2008;181:859–71. doi: 10.1083/jcb.200708037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu YB, Kharode Y, Bodine PV, Yaworsky PJ, Robinson JA, Billiard J. LPA induces osteoblast differentiation through interplay of two receptors: LPA1 and LPA4. J Cell Biochem. 2010;109:794–800. doi: 10.1002/jcb.22471. [DOI] [PubMed] [Google Scholar]
- 21.Contos JJ, Fukushima N, Weiner JA, Kaushal D, Chun J. Requirement for the lpA1 lysophosphatidic acid receptor gene in normal suckling behavior. Proc Natl Acad Sci U S A. 2000;97:13384–9. doi: 10.1073/pnas.97.24.13384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dingerkus G, Uhler LD. Enzyme clearing of alcian blue stained whole small vertebrates for demonstration of cartilage. Stain Technol. 1977;52:229–32. doi: 10.3109/10520297709116780. [DOI] [PubMed] [Google Scholar]
- 23.Kohlbrenner A, Koller B, Hammerle S, Ruegsegger P. In vivo micro tomography. Adv Exp Med Biol. 2001;496:213–24. doi: 10.1007/978-1-4615-0651-5_20. [DOI] [PubMed] [Google Scholar]
- 24.David V, Laroche N, Boudignon B, Lafage-Proust MH, Alexandre C, Ruegsegger P, et al. Noninvasive in vivo monitoring of bone architecture alterations in hindlimb-unloaded female rats using novel three-dimensional microcomputed tomography. J Bone Miner Res. 2003;18:1622–31. doi: 10.1359/jbmr.2003.18.9.1622. [DOI] [PubMed] [Google Scholar]
- 25.Oreffo RO, Kusec V, Romberg S, Triffitt JT. Human bone marrow osteoprogenitors express estrogen receptor-alpha and bone morphogenetic proteins 2 and 4 mRNA during osteoblastic differentiation. J Cell Biochem. 1999;75:382–92. doi: 10.1002/(sici)1097-4644(19991201)75:3<382::aid-jcb4>3.3.co;2-e. [DOI] [PubMed] [Google Scholar]
- 26.Stanford CM, Jacobson PA, Eanes DE, Lembke LA, Midura RJ. Rapidly forming apatitic mineral in osteoblastic cell line (UMR 10601 BSP) J Biol Chem. 1995;270:9420–8. doi: 10.1074/jbc.270.16.9420. [DOI] [PubMed] [Google Scholar]
- 27.Thèves C, Keyser-Tracqui C, Crubézy E, Salles JP, Ludes B, Telmon N. Detection and quantification of the age-related point mutation A189G in the human mitochondrial DNA. Forensic Sci. 2006;51:865–73. doi: 10.1111/j.1556-4029.2006.00163.x. [DOI] [PubMed] [Google Scholar]
- 28.Saulnier-Blache JS, Girard A, Simon MF, Lafontan M, Valet P. A simple and highly sensitive radioenzymatic assay for lysophosphatidic acid quantification. J Lipid Res. 2000;41:1947–51. [PMC free article] [PubMed] [Google Scholar]
- 29.Chang MC, Tanaka J. FT-IR study for hydroxyapatite/collagen nanocomposite cross-linked by glutaraldehyde. Biomaterials. 2002;23:4811–8. doi: 10.1016/s0142-9612(02)00232-6. [DOI] [PubMed] [Google Scholar]
- 30.Boskey AL, Goldberg M, Ashok K, Santiago G. Infrared imaging microscopy of bone: illustrations from a mouse model of Fabry disease. Biochim Biophys Acta. 2006;1758:942–7. doi: 10.1016/j.bbamem.2006.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rauch F, Schoenau E. Changes in bone density during childhood and adolescence: an approach based onbone’s biological organization. J Bone Miner Res. 2001;16:597–604. doi: 10.1359/jbmr.2001.16.4.597. [DOI] [PubMed] [Google Scholar]
- 32.Chen F, Capecchi MR. Targeted mutations in hoxa-9 and hoxb-9 reveal synergistic interactions. Dev Biol. 1997;181:186–96. doi: 10.1006/dbio.1996.8440. [DOI] [PubMed] [Google Scholar]
- 33.McIntyre DC, Rakshit S, Yallowitz AR, Loken L, Jeannotte L, Capecchi MR, et al. Hox patterning of the vertebrate rib cage. Development. 2007;134:2981–9. doi: 10.1242/dev.007567. [DOI] [PubMed] [Google Scholar]
- 34.Abe M, Michikami I, Fukushi T, Abe A, Maeda Y, Ooshima T, et al. Hand2 regulates chondrogenesis in vitro and in vivo. Bone. 2010;46:1359–68. doi: 10.1016/j.bone.2009.11.022. [DOI] [PubMed] [Google Scholar]
- 35.Katagiri T, Boorla S, Frendo JL, Hogan BL, Karsenty G. Skeletal abnormalities in doubly heterozygous Bmp4 and Bmp7 mice. Dev Genet. 1998;22:340–8. doi: 10.1002/(SICI)1520-6408(1998)22:4<340::AID-DVG4>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 36.Grey A, Xu X, Hill B, Watson M, Callon K, Reid IR, et al. Osteoblastic cells express phospholipid receptors and phosphatases and proliferate in response to sphingosine-1-phosphate. Calcif Tissue Int. 2004;74:542–50. doi: 10.1007/s00223-003-0155-9. [DOI] [PubMed] [Google Scholar]
- 37.Fratzl P, Paris O, Klaushofer K, Landis WJ. Bone mineralization in an osteogenesis imperfecta mouse model studied by small-angle x-ray scattering. J Clin Invest. 1996;97:396–402. doi: 10.1172/JCI118428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Roschger P, Fratzl-Zelman N, Misof BM, Glorieux FH, Klaushofer K, Rauch F. Evidence that abnormal high bone mineralization in growing children with osteogenesis imperfecta is not associated with specific collagen mutations. Calcif Tissue Int. 2008;82:263–70. doi: 10.1007/s00223-008-9113-x. [DOI] [PubMed] [Google Scholar]
- 39.Yadav VK, Ryu JH, Suda N, Tanaka KF, Gingrich JA, Schutz G, et al. Lrp5 controls bone formation by inhibiting serotonin synthesis in the duodenum. Cell. 2008;135:825–37. doi: 10.1016/j.cell.2008.09.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Boucharaba A, Serre CM, Gres S, Saulnier-Blache JS, Bordet JC, Guglielmi J, et al. Platelet-derived lysophosphatidic acid supports the progression of osteolytic bone metastases in breast cancer. J Clin Invest. 2004;114:1714–25. doi: 10.1172/JCI22123. [DOI] [PMC free article] [PubMed] [Google Scholar]