Abstract
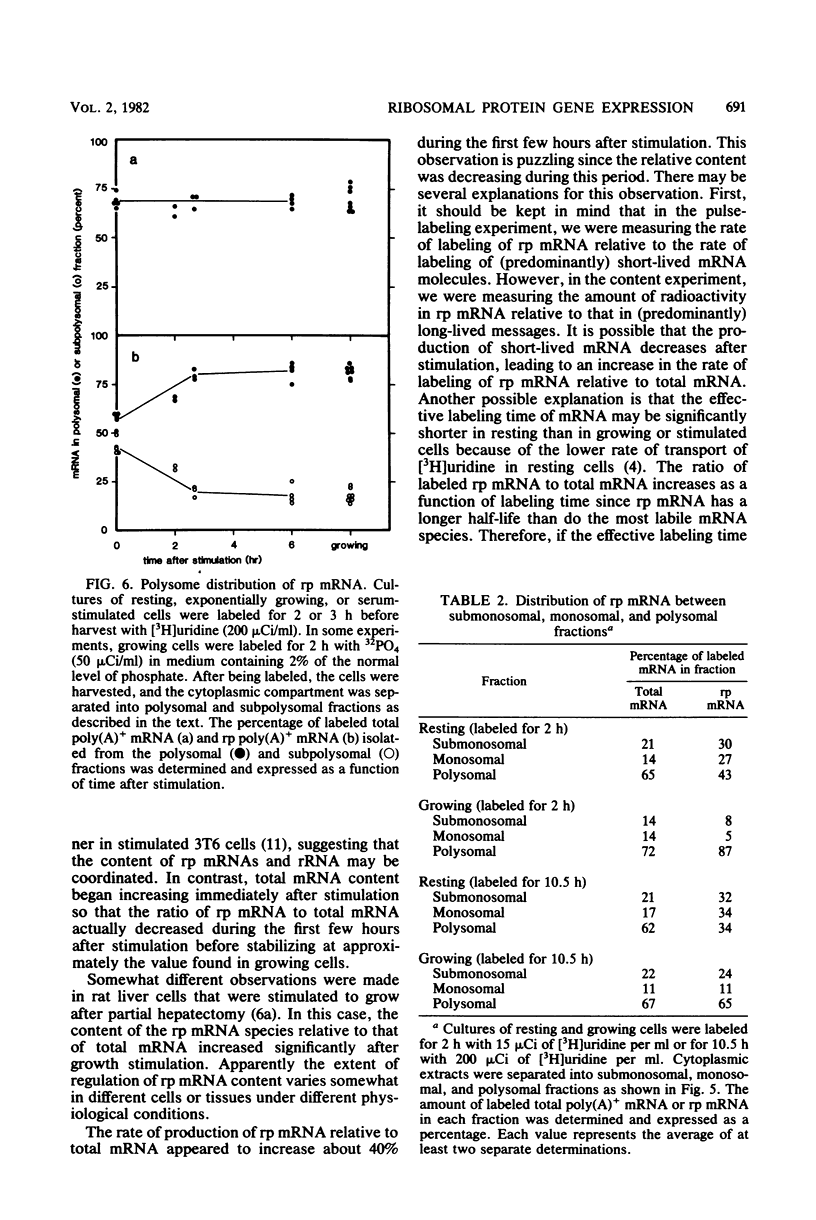
When resting (G0) mouse 3T6 fibroblasts are serum stimulated to reenter the cell cycle, the rates of synthesis of rRNA and ribosomal proteins increase, resulting in an increase in ribosome content beginning about 6 h after stimulation. In this study, we monitored the content, metabolism, and translation of ribosomal protein mRNA (rp mRNA) in resting, exponentially growing, and serum-stimulated 3T6 cells. Cloned cDNAs for seven rp mRNAs were used in DNA-excess filter hybridization studies to assay rp mRNA. We found that about 85% of rp mRNA is polyadenylated under all growth conditions. The rate of labeling of rp mRNA relative to total polyadenylated mRNA changed very little after stimulation. The half-life of rp mRNA was about 11 h in resting cells and about 8 h in exponentially growing cells, values which are similar to the half-lives of total mRNA in resting and growing cells (about 9 h). The content of rp mRNA relative to total mRNA was about the same in resting and growing 3T6 cells. Furthermore, the total amount of rp mRNA did not begin to increase until about 6 h after stimulation. Since an increase in rp mRNA content did not appear to be responsible for the increase in ribosomal protein synthesis, we determined the efficiency of translation of rp mRNA under different conditions. We found that about 85% of pulse-labeled rp mRNA was associated with polysomes in exponentially growing cells. In resting cells, however, only about half was associated with polysomes, and about 30% was found in the monosomal fraction. The distribution shifted to that found in growing cells within 3 h after serum stimulation. Similar results were obtained when cells were labeled for 10.5 h. About 70% of total polyadenylated mRNA was in the polysome fraction in all growth states regardless of labeling time, indicating that the shift in mRNA distribution was species specific. These results indicate that the content and metabolism of rp mRNA do not change significantly after growth stimulation. The rate of ribosomal protein synthesis appears to be controlled during the resting-growing transition by an alteration of the efficiency of translation of rp mRNA, possibly at the level of protein synthesis initiation.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunningham D. D., Pardee A. B. Transport changes rapidly initiated by serum addition to "contact inhibited" 3T3 cells. Proc Natl Acad Sci U S A. 1969 Nov;64(3):1049–1056. doi: 10.1073/pnas.64.3.1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Eustachio P., Meyuhas O., Ruddle F., Perry R. P. Chromosomal distribution of ribosomal protein genes in the mouse. Cell. 1981 May;24(2):307–312. doi: 10.1016/0092-8674(81)90320-2. [DOI] [PubMed] [Google Scholar]
- DePhilip R. M., Rudert W. A., Lieberman I. Preferential stimulation of ribosomal protein synthesis by insulin and in the absence of ribosomal and messenger ribonucleic acid formation. Biochemistry. 1980 Apr 15;19(8):1662–1669. doi: 10.1021/bi00549a022. [DOI] [PubMed] [Google Scholar]
- Faliks D., Meyuhas O. Coordinate regulation of ribosomal protein mRNA level in regenerating rat liver. Study with the corresponding mouse cloned cDNAs. Nucleic Acids Res. 1982 Feb 11;10(3):789–801. doi: 10.1093/nar/10.3.789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fallon A. M., Jinks C. S., Strycharz G. D., Nomura M. Regulation of ribosomal protein synthesis in Escherichia coli by selective mRNA inactivation. Proc Natl Acad Sci U S A. 1979 Jul;76(7):3411–3415. doi: 10.1073/pnas.76.7.3411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gorenstein C., Warner J. R. Coordinate regulation of the synthesis of eukaryotic ribosomal proteins. Proc Natl Acad Sci U S A. 1976 May;73(5):1547–1551. doi: 10.1073/pnas.73.5.1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hendrickson S. L., Wu J. S., Johnson L. F. Cell cycle regulation of dihydrofolate reductase mRNA metabolism in mouse fibroblasts. Proc Natl Acad Sci U S A. 1980 Sep;77(9):5140–5144. doi: 10.1073/pnas.77.9.5140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kafatos F. C., Jones C. W., Efstratiadis A. Determination of nucleic acid sequence homologies and relative concentrations by a dot hybridization procedure. Nucleic Acids Res. 1979 Nov 24;7(6):1541–1552. doi: 10.1093/nar/7.6.1541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lodish H. F. Model for the regulation of mRNA translation applied to haemoglobin synthesis. Nature. 1974 Oct 4;251(5474):385–388. doi: 10.1038/251385a0. [DOI] [PubMed] [Google Scholar]
- Mauck J. C., Green H. Regulation of RNA synthesis in fibroblasts during transition from resting to growing state. Proc Natl Acad Sci U S A. 1973 Oct;70(10):2819–2822. doi: 10.1073/pnas.70.10.2819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyuhas O., Perry R. P. Construction and identification of cDNA clones for mouse ribosomal proteins: application for the study of r-protein gene expression. Gene. 1980 Jul;10(2):113–129. doi: 10.1016/0378-1119(80)90129-8. [DOI] [PubMed] [Google Scholar]
- Monk R. J., Meyuhas O., Perry R. P. Mammals have multiple genes for individual ribosomal proteins. Cell. 1981 May;24(2):301–306. doi: 10.1016/0092-8674(81)90319-6. [DOI] [PubMed] [Google Scholar]
- Nabeshima Y. I., Ogata K. Stimulation of the synthesis of ribosomal proteins in regenerating rat liver with special reference to the increase in the amounts of effective mRNAs for ribosomal proteins. Eur J Biochem. 1980 Jun;107(2):323–329. doi: 10.1111/j.1432-1033.1980.tb06032.x. [DOI] [PubMed] [Google Scholar]
- Nomura M., Morgan E. A. Genetics of bacterial ribosomes. Annu Rev Genet. 1977;11:297–347. doi: 10.1146/annurev.ge.11.120177.001501. [DOI] [PubMed] [Google Scholar]
- Nomura M., Yates J. L., Dean D., Post L. E. Feedback regulation of ribosomal protein gene expression in Escherichia coli: structural homology of ribosomal RNA and ribosomal protein MRNA. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7084–7088. doi: 10.1073/pnas.77.12.7084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pratt R. E., Johnson L. F. Absence of control of poly(A)(+) messenger RNA translation in growth-stimulated mouse 3T6 fibroblasts. Biochim Biophys Acta. 1980 Jul 29;608(2):332–343. doi: 10.1016/0005-2787(80)90179-3. [DOI] [PubMed] [Google Scholar]
- TODARO G. J., GREEN H. Quantitative studies of the growth of mouse embryo cells in culture and their development into established lines. J Cell Biol. 1963 May;17:299–313. doi: 10.1083/jcb.17.2.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wool I. G. The structure and function of eukaryotic ribosomes. Annu Rev Biochem. 1979;48:719–754. doi: 10.1146/annurev.bi.48.070179.003443. [DOI] [PubMed] [Google Scholar]
- Yates J. L., Nomura M. Feedback regulation of ribosomal protein synthesis in E. coli: localization of the mRNA target sites for repressor action of ribosomal protein L1. Cell. 1981 Apr;24(1):243–249. doi: 10.1016/0092-8674(81)90520-1. [DOI] [PubMed] [Google Scholar]
- Zengel J. M., Mueckl D., Lindahl L. Protein L4 of the E. coli ribosome regulates an eleven gene r protein operon. Cell. 1980 Sep;21(2):523–535. doi: 10.1016/0092-8674(80)90490-0. [DOI] [PubMed] [Google Scholar]