Abstract
Objective To describe our experience of cerebrospinal fluid (CSF) rhinorrhea management.
Design Retrospective.
Setting Charing Cross Hospital, London, a tertiary referral center.
Participants Fifty-four patients with CSF rhinorrhea managed from 2003 to 2011.
Main outcome measures Surgical technique; Recurrence.
Results Etiologically, 36 were spontaneous and 18 traumatic. Eight patients with spontaneous and two with traumatic leaks had previous failed repairs in other units. Success rates after first and second surgery were 93% and 100%, respectively. Mean follow-up was 21 months. Four patients, all of spontaneous etiology, had recurrences; three of these underwent successful second repair with three layered technique, and the fourth had complete cessation of the leak after gastric bypass surgery and subsequent weight reduction. Adaptation of anatomic three-layered repair since then averted any further failure in the following 7 years. Mean body mass index was 34.0 kg/m2 in spontaneous and 27.8 kg/m2 in traumatic cases (p < 0.05). Fifty percent of spontaneous leaks were from the cribriform plate, 22% sphenoid, 14% ethmoid, and 14% frontal sinus. In the traumatic CSF leak group: 33.3% were from the cribriform plate, 33.3% sphenoid, 22.2% ethmoid, and 11.1% frontal.
Conclusion Endoscopic CSF fistula closure is a safe and effective operation. All sites of leak can be accessed endoscopically. We recommend the use of an anatomic three-layered closure in difficult cases.
Keywords: cerebrospinal fluid, rhinorrhea, endoscopic, skull base, spontaneous leaks
Introduction
Cerebrospinal fluid (CSF) rhinorrhea results from a breakdown in the integrity of structures separating the subarachnoid space and nasal cavity, namely the subarachnoid space and dura mater, the bony skull base and periostea alongside the upper aerodigestive tract mucosa.1
CSF rhinorrhea can be classified etiologically as traumatic or nontraumatic. Traumatic CSF rhinorrhea can be further categorized as iatrogenic, with endoscopic sinus surgery being the most common cause.2 Nontraumatic CSF rhinorrhea is subdivided into congenital or idiopathic; this condition can also be considered based on whether the underlying CSF pressure is normal or elevated.3
The most common symptom of a CSF fistula is clear, unilateral rhinorrhea, which is classically exacerbated by stooping or performing a Valsalva maneuver and can be associated with headaches, particularly on standing, due to orthostatic CSF hypotension. The index of clinical suspicion should be raised in a setting of trauma, paranasal or cranial surgery, or in middle-aged obese females, who are at increased risk of spontaneous CSF leaks.4 Less commonly, patients may present with neurological deficits caused by ascending intracranial infections.2 Rhinological examination of these patients is important in excluding primary nasal pathology causing rhinorrhea and though often normal, can occasionally elucidate the site of leak or an encephalocele.
The aim of surgery is complete and durable separation of the subarachnoid space from the nasal cavity, which, if left untreated, has up to a 20% risk of ascending intracranial infections.5 In addition, this restores normal CSF circulation, maintaining brain buoyancy and preventing CSF hypotension.6 This study evaluates management and outcomes of CSF rhinorrhea in a tertiary referral center over an 8-year period.
Methods
A cohort of 54 patients, attending for management of CSF rhinorrhea from 2003 to 2011, was identified from the senior author's Charing Cross Hospital patient database. Case notes, imaging studies, and biochemical tests were reviewed.
The London Research Ethics Committee confirmed that ethical approval was not required for the study.
Statistical analysis was performed using GraphPad Prism v5.0 (GraphPad Software, Inc., La Jolla, California, USA).
Preoperative Work-up
Nasal secretions were tested for β2 transferrin to confirm CSF leak and 1-mm thickness high-resolution computed tomography (CT) of paranasal sinuses, using BrainLab (Brainlab AG, Feldkirchen, Germany) protocol was performed to allow intraoperative navigation. Magnetic resonance imaging (MRI) was performed when the site of the leak was not seen on CT or where there was a suspicion of a meningocele or encephalocele. All spontaneous leak patients underwent neurological review for assessment and management of any underlying primary pathology.
Operative Technique
An endoscopic approach was used in the management of these patients. Intrathecal fluorescein was used in 67% of patients leading to intraoperative identification of skull base defects in 97% of cases. This included all revision cases alongside those with a suggestion of multiple leak sites. Skin prick testing with fluorescein was performed for all patients prior to surgery to rule out an allergic reaction.7
Various degrees of endoscopic sinus dissection were performed to create adequate exposure depending on the site of the leak following nasal decongestion using Moffat's solution.8 Lateral lamella or ethmoid sinus leaks were accessed with an ethmoidectomy and olfactory groove with a partial middle turbinectomy, whereas access to the sphenoid sinus was typically achieved without disturbing the ethmoid sinus and middle turbinate. In special circumstances such as approaches to the frontal recess, a modified Lothrop was performed.9 Reduction of herniated mucosa or bipolar diathermy of an encephalocele was performed if present. The mucosal perimeter of the leak site was denuded by up to 5 mm if possible.
For small defects (less than 4 mm), a two-layered technique, commonly with fascia lata and mucosa, was utilized.
With regard to defects of at least 4 mm, prior to 2004, a two-layered approach was used. From 2004, an anatomic three-layered repair was employed. The term anatomic refers to mimicking the original anatomy by replacing layer for layer.10 In ethmoid and olfactory defects, fascia lata underlay followed by septal cartilage and free turbinate mucosa was applied. When it was not possible to insert fascia underlay graft, as is often encountered in the olfactory area, it was placed in an overlay fashion. Fascia lata, free turbinate mucosa, and fat were grafted in sphenoid defects to reduce risk of mucocele formation. In some cases of recurrent spontaneous leaks, fascia lata, bone pate, and turbinate mucosa followed by fat for further reinforcement was used. Duragen (Integra LifeSciences, Plainsboro, New Jersey, USA) usage was reserved for when fascia lata was unavailable. In frontal leaks, fat graft was not utilized due to the risk of obstructing the frontal recess with subsequent frontal sinusitis or mucoceles; otherwise a similar technique to ethmoid and olfactory defects was utilized with the caveat that most of these leaks were very small, necessitating two-layered closure with fascia lata and turbinate mucosa. Fibrin sealant Tisseel (Baxter Healthcare Corporation, Deerfield, Illinois, USA) was also applied in all cases in between the first and second layers and superficial to the third layer to provide an impermeable seal.11
A variety of graft materials were used ranging from middle turbinate mucosa, septal cartilage, Duragen, fat, and bone pate.10 Grafts were chosen based on etiology and the site of the leak.
Postoperative Care
Measures were taken to prevent intracranial pressure elevation, including strict bed rest with elevation of the head to 30 degrees for 48 to 72 hours, prevention and treatment of constipation, avoidance of straining and nose blowing, and the use of lumbar drains in selected patients.
Lumbar drains were utilized in all revision cases, defects larger than 15 mm, those with raised intracranial pressure, and spontaneous etiology cases unless the fistula was very small (<3 mm).
In earlier cases, Whitehead's varnish–impregnated gauze nasal packing was inserted and removed in 1 week under a general anesthetic. Since 2009, Nasopore (Polyganics, Groningen, The Netherlands), a biodegradable nasal pack, was used, obviating the need for a second anesthetic for pack removal. Antibiotic (co-amoxiclav) prophylaxis was prescribed for 1 week.
Results
Demographics
Fifty-four patients were identified, of which 18 were male and 36 female. Mean age was 47.7 years. There was a statistically significant difference (p < 0.05) between the mean age of patients with a spontaneous leak (50.4 years) and those with a traumatic etiology (42.8 years).
There was a significant difference between the gender ratios in the spontaneous group, with 75% being female and 25% male as compared with the traumatic group, where there was an equal proportion (50%) of each gender (p < 0.001).
Presentation
The mean duration of CSF rhinorrhea in all patients was 11.8 months with a range of 1 to 36 months (median 8 months). In the spontaneous leak group, the mean duration (14.5 months) was significantly longer than in the traumatic cases (6.4 months) (p < 0.05).
In the spontaneous group, 8 patients (22%) had episodes of meningitis preoperatively, with 2 having 3 incidents, 3 affected twice, and 3 affected once. In the 18 traumatic cases, 4 patients (22%) had meningitis, with 1 enduring 3 bouts and 3 others having a single attack.
Etiology
There were 18 cases of traumatic CSF fistula, including 11 iatrogenic cases, referred to our unit. Spontaneous or nontraumatic leaks occurred in 36 patients (Fig. 1). Ten cases had previous failed attempts at repair in other units, two with traumatic and eight with spontaneous etiologies.
Fig. 1.
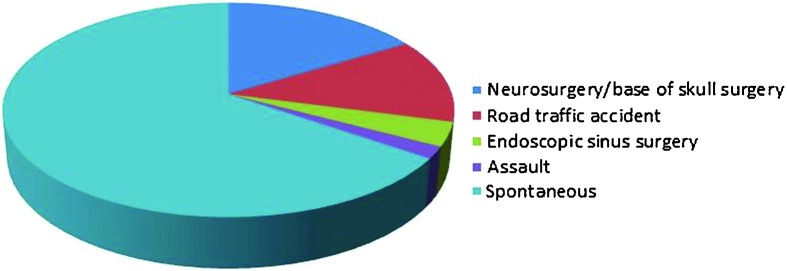
Etiology of cerebrospinal rhinorrhoea.
Size and Site of Leak
The estimated size of the defect ranged from less than 1 mm to 30 mm. Mean size was 10.6 mm (95% confidence interval [CI]: 5.6 to 15.6) and 16.4 mm (95% CI: 8.8 to 23.9) in spontaneous and traumatic leaks, respectively.
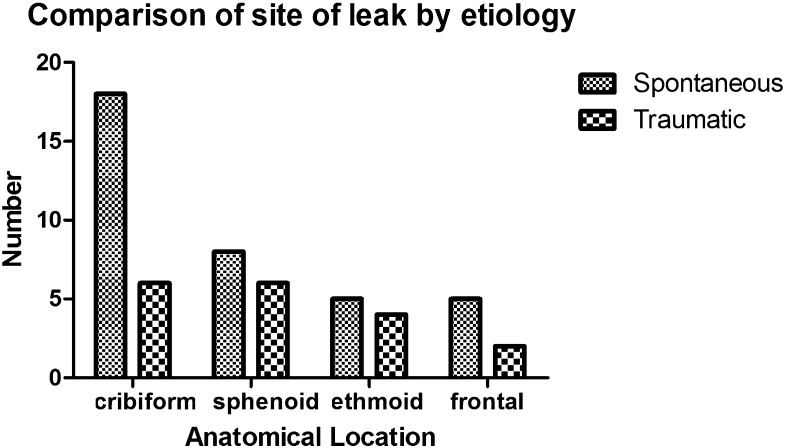
The anatomical defects were in the cribriform plate in 44.4%, sphenoid bone 25.9%, ethmoid bone 16.7%, and frontal recess 13.0% (Fig. 2). Fifty percent of spontaneous leaks (18) were from the cribriform plate, 22% sphenoid, 14% ethmoid, and 14% frontal sinus. In the traumatic CSF leak group, 33.3% were from the cribriform plate, 33.3% sphenoid, 22.2% ethmoid, 11.1% frontal. There was no statistically significant difference in the site of the leak between spontaneous and traumatic leak etiologies nor between iatrogenic and traumatic leak groups (p values > 0.10) (Fig. 3).
Fig. 2.
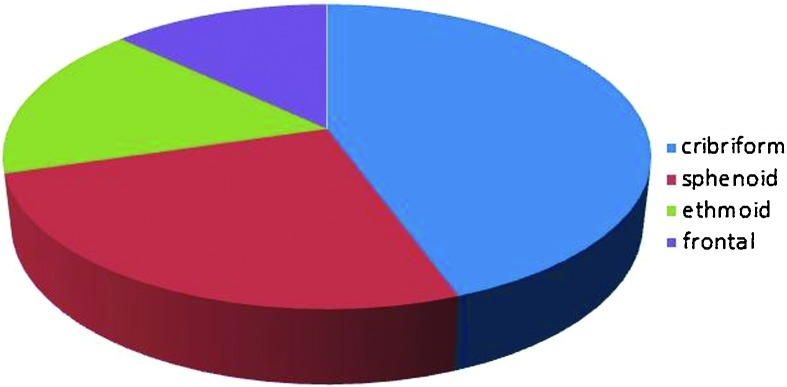
Anatomical site of cerebrospinal fluid leak for all cases.
Fig. 3.
Comparison of anatomical site of leak by etiology.
Body Mass Index (BMI)
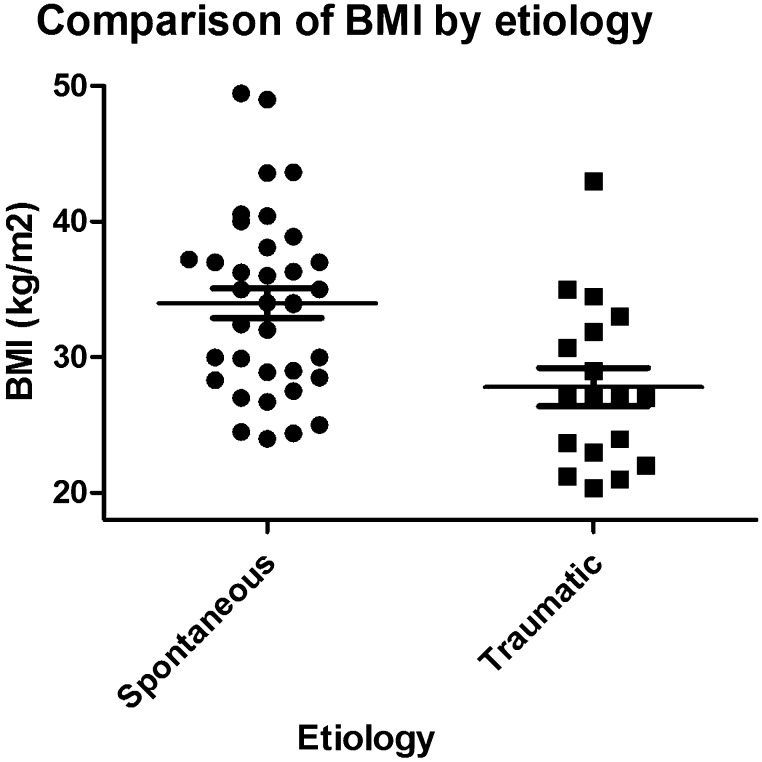
Mean BMI in all patients was 31.9 kg/m2. In those with spontaneous CSF rhinorrhea, mean BMI (34.0 kg/m2) was significantly higher than patients with traumatic etiologies (27.8 kg/m2) (p < 0.05) (Fig. 4).
Fig. 4.
Comparison of body mass index by etiology.
Perioperative Care
Nasal secretion samples sent for β2 transferrin yielded positive results in 87.5%. All patients had confirmed CSF rhinorrhea either in our unit or from the referring center.
A lumbar drain was used in 30 patients (54%), 22 spontaneous leak patients (61.1%) and 8 traumatic ones (44%) (p < 0.05).
Success Rate, Revision Cases, and Duration of Follow-up
Overall success rate was 93% (50/54), inclusive of 10 cases where the initial failed surgeries were performed elsewhere. After a second operation, success rate was 100% (53/53), with one failure case refusing further surgical repair.
Eight patients in the spontaneous group had previous failed attempts in other units. Seven of these had successful repair in our series at first surgery and one, with a BMI of 43.6 kg/m2 and two previous failed attempts, recurred after our repair. She declined further revision surgery and was referred to the bariatric team, who performed gastric bypass surgery with subsequent complete cessation of CSF rhinorrhea to date (18 months). The three other failures in our series were from the spontaneous group with skull base defects of ≥ 4 mm who underwent successful second repairs. These failures were all prior to 2004 when a two-layered repair was used. One patient in the spontaneous group had a successful repair of a leak in the frontoethmoidal recess and presented 2 years later with a novel leak from the posterolateral wall of the same fontal sinus, which was successfully closed.
Two out of 18 traumatic cases had previous failed procedures in other units; both patients suffered extensive injuries following a high-impact road traffic accident. The first patient had two craniotomies to repair the leak by neurosurgeons and the second patient had endoscopic procedures by other otolaryngologists prior to referrals to our department.
Mean follow-up was 21 months with 95% CI (16.1 to 26.6 months) and median of 17 months. There was no statistically significant difference in follow-up duration between the spontaneous and the traumatic patient groups (p = 0.38).
Discussion
This study describes the endoscopic surgical management of patients presenting with CSF rhinorrhea over an 8-year period and represents the largest series over such a time period. The overall success rate of 93% at first surgery and 100% at second surgery compares well with the literature.12,13,14,15 All operations were performed by the senior author (HAS).
We report a much higher proportion of spontaneous (67%) as opposed to traumatic (33%) leaks in contrast to the literature.7,12,13,14,15,16,17 This may be explained by our unit being a tertiary referral center and having close links with a neurosurgical unit, possibly resulting in a skewed patient population. Additionally, several traumatic leaks were successfully managed with conservative measures under the care of the neurosurgeons and did not enter our database.13,16,18
The mean age of patients with traumatic leaks was less than those with spontaneous rhinorrhea, as road traffic accidents and assaults tend to involve the younger demographic. This is comparable to the literature.7,13,14 The duration prior to presentation for the traumatic leaks tended to be shorter than the spontaneous ones, probably due to higher clinical suspicion with the clear history of trauma. Interestingly, the risk of meningitis was similar in both groups of patients and in keeping with current understanding.5
Our series confirms that spontaneous leak patients are more commonly middle-aged females.4,19,20 These patients also had significantly higher BMIs, associating it with benign or idiopathic intracranial hypertension (IIH).19,20 All spontaneous rhinorrhea patients therefore underwent neurological review preoperatively to potentially diagnose and treat IIH. A combination of clinical symptoms, signs, and measurement of opening pressure on lumbar puncture were used.21
The anatomical site of the defect corresponded well with a recent large case series for spontaneous leaks, with the commonest site being the cribriform plate, presumably due to the anatomy of the plate allowing for transmission of olfactory nerve fibers and hence, more easily, permitting a CSF leak.19 Experimental data indicate that the cribriform plate constitutes a substantial pathway of CSF drainage into the lymphatic outflow system.6 A recent systematic review indicates, however, that lateral recess of sphenoid and ethmoid roof defects are more frequent.15
Idiopathic intracranial hypertension is not well understood, and management of these patients lacks a clear evidence base.22 Whatever the cause, the higher pulsatile pressures lead to erosion of the skull base with a predilection for the thinnest areas, namely the cribriform plate and lateral sphenoid recess.19,23
The overall pattern demonstrated for sites of traumatic leaks correlated with a recent series.13 However, some publications cite sphenoid and ethmoid defects as more common.12,15,24 We had two cases of iatrogenic CSF leaks secondary to endoscopic sinus surgery referred to our unit, whereas most cases were secondary to extensive base of skull or neurosurgery in contrast to the literature.2 As a result, a larger proportion of sphenoid defects were noted in iatrogenic injuries, principally after trans-sphenoidal pituitary surgery.
Preoperative work-up in this series fits well with described management algorithms.25 Nasal secretions sent for β2 transferrin yielded positive results in over 87%. Lund also highlighted that adequate samples were attained in a small proportion of patients but, when received, produced positive results in most cases, reflecting its high sensitivity and specificity.7,25
In our experience, CT scans with 1-mm slices employing BrainLab protocol was sufficient, as published management algorithms have indicated.7,25 MRI has a complementary role particularly if there is clinical suspicion of meningoencephalocele (which are more common in the IIH and spontaneous leak demographic) or tumor.25 Although CT cisternography is advocated by some departments, we feel that its use does not add further information.7,16
Our intraoperative use of intrathecal fluorescein was higher than some centres.7,27 It was used in spontaneous leak cases, in those with potential multiple site leaks such as extensive skull base fractures, and in one case where the CT imaging did not clarify an anatomical defect.27 The greater use of intrathecal fluorescein can be attributed to our higher proportion of spontaneous cases, which are often less identifiable on radiological imaging. This series also highlights the efficacy and safety profile of fluorescein, with no adverse reactions noted.28 There were no adverse reactions to intrathecal fluorescein in our series. The routine skin prick testing with fluorescein prior to surgery had ruled out severe allergic reactions. We also used a low concentration of intrathecal fluorescein, and such low concentrations have been shown to have good safety and efficacy.15
We incorporated the use of a variety of grafts, and success rates of differing grafts have been shown to be similar.29 In large skull base defects, the use of vascularized tissue grafts has been correlated with lower failure rates.30 Grafts were chosen depending on etiology alongside the size and site of the leak. Of note, our failed cases all had skull base defects of ≥ 4 mm and all occurred prior to 2004 while using a two-layer repair. The successful three-layer repair of these defects was the reason behind adaptation of this technique. This mimicked normal anatomy by replacing dura by fascia lata, bone by cartilage or bone pate, and mucosa by mucosa. There has been no further failure over the proceeding 7-year period. We therefore recommend employing this technique for defects of ≥ 4 mm.
Postoperatively, there is no clear consensus on the use of lumbar drains, although high success rates have been shown without their use.15,31 In our series, the use of lumbar drains was decided in consultation with the neurosurgeons. We used lumbar drains for all revision cases, spontaneous etiology cases unless the defect was very small (1 to 2 mm), large defects (> 15 mm), and those with raised intracranial pressure.32 This is reflected in significantly more spontaneous leak patients having lumbar drains compared with traumatic etiology cases. CSF pressure was only measured where indicated by neurologists. A recent study in fact suggests that single preoperative CSF pressure measurements in patients with active leaks are not sufficiently reliable to lead clinical decisions.33 However, it was noted intraoperatively that some spontaneous leaks were under high pressure, and 10 to 20 mL of CSF was removed to facilitate a watertight repair, alongside the use of a lumbar drain.
Similarly, there is no accepted protocol for use of antibiotic prophylaxis, and it seems that the risk of meningitis is not reduced.15,34,35 However, some recommend at least 48 hours of antibiotics, whereas others treat those with a record of meningitis or who are undergoing repeat closure attempts.16,36,37 A systematic review of the endoscopic repair of CSF leaks showed that almost half of the total 53 studies used antibiotics routinely.15 In our practice, we used a 1-week course of co-amoxiclav to decrease the risk of possible local infection due to nasal packing.38
One of our four operative failures was a 41-year-old female with a BMI of 43.6 kg/m2 and two previous failed repairs prior to our first surgery. Comorbidities included raised intracranial pressure, hypertension, and type two diabetes. The defect of 10 mm diameter was in the frontoethmoidal recess. She declined further surgery and, having failed to respond to conservative medical management with weight loss and acetazolamide, was referred to have laparoscopic gastric bypass surgery with a subsequent complete resolution of her CSF leak to date.4,19,20 There is evidence that bariatric surgery improves benign intracranial hypertension symptomatology, reduces intracranial pressure, and abates CSF rhinorrhoea.39,40
The revision cases share similarities, namely spontaneous leaks are more difficult to manage and have lower success rates, with these patients typically being obese, middle-aged women with defects in pneumatized sphenoid lateral recesses.4,19,20,23 Our other three failures belonged to this group and had successful revisions, with two being sphenoid and the other a posterior ethmoid roof defect.
A 40-year-old female with confirmed IIH and a BMI of 38.9 kg/m2 was referred to our center after two failed surgeries for a sphenoidal defect of 12 to 15mm. She underwent a successful CSF leak repair using our standard three-layered technique in conjunction with ventriculoperitoneal shunting, which has been demonstrated to be an effective and increasingly common treatment for IIH.19,20,41 In our unit, the use of ventriculoperitoneal shunts was reached in consultation with neurosurgery with criteria including IIH, high BMI (above 35 kg/m2), failed surgery, and failed trials of acetazolamide.
The positive impact of gastric banding and possibly ventriculoperitoneal shunting highlight the importance of a multidisciplinary approach in the management of the spontaneous CSF rhinorrhea. The senior author manages these patients in consultation with neurological colleagues, given the primary disorder is one of CSF circulation, manifesting as a dehiscence of the skull base and, if untreated, may continue to present with further leaks. Treatment of underlying raised intracranial pressure is required but may be coupled with surgery to good effect.4,20 Further research is required to elucidate if, when, and which adjuvant treatments to employ.22 These include conservative (diet and weight loss), medical (loop diuretics and carbonic anhydrase inhibitors), radiological (intracranial venous shunting), and surgical (ventriculoperitoneal shunting or bariatric surgery) options.4,19,20,39,40,41,42,43,44
Complications included three postoperative episodes of transient pyrexia 24 hours postoperatively with negative blood cultures and no meningitis. In follow-up, two patients complained of hyposmia, two patients developed persistent atypical facial pain managed by neurologists, and two patients presented with intranasal synechiae, one of whom required surgical division.
Conclusion
Endoscopic CSF fistula closure is a safe and effective operation that has become the standard of care for managing CSF rhinorrhea. All sites of leak can be accessed endoscopically, including the frontal recess with a modified Lothrop approach. In our experience, improved success followed modifying techniques and adopting a multidisciplinary management approach.
We recommend the use of an anatomic three-layered closure for defects ≥ 4 mm, particularly in revision cases and in patients with spontaneous etiology.
Patients with spontaneous CSF rhinorrhea should be managed in conjunction with the neurology and neurosurgery teams to manage the underlying pathology and may also require ventriculoperitoneal shunting or gastric bypass surgery.
Acknowledgments
Nil to declare.
Conflicts of Interest None Financial Disclosure Information None Funding Nil
Notes
6th International Congress of the World Federation of Skull Base Societies & 10th European Skull Base Society Congress, Brighton, UK. Wednesday 16 May 2012.
6th Australian Rhinology Society Conference, Coolum, QLD, Australia. Sunday 7 October 2012.
References
- 1.Locatelli D Rampa F Acchiardi I Bignami M De Bernardi F Castelnuovo P Endoscopic endonasal approaches for repair of cerebrospinal fluid leaks: nine-year experience Neurosurgery 200658402ONS-246–ONS-256., discussion ONS-256–ONS-257 [DOI] [PubMed] [Google Scholar]
- 2.Carrau R L, Snyderman C H, Kassam A B. The management of cerebrospinal fluid leaks in patients at risk for high-pressure hydrocephalus. Laryngoscope. 2005;115:205–212. doi: 10.1097/01.mlg.0000154719.62668.70. [DOI] [PubMed] [Google Scholar]
- 3.Har-El G. What is “spontaneous” cerebrospinal fluid rhinorrhea? Classification of cerebrospinal fluid leaks. Ann Otol Rhinol Laryngol. 1999;108:323–326. doi: 10.1177/000348949910800401. [DOI] [PubMed] [Google Scholar]
- 4.Wang E W, Vandergrift W A III, Schlosser R J. Spontaneous CSF leaks. Otolaryngol Clin North Am. 2011;44:845–856, vii. doi: 10.1016/j.otc.2011.06.018. [DOI] [PubMed] [Google Scholar]
- 5.Daudia A, Biswas D, Jones N S. Risk of meningitis with cerebrospinal fluid rhinorrhea. Ann Otol Rhinol Laryngol. 2007;116:902–905. doi: 10.1177/000348940711601206. [DOI] [PubMed] [Google Scholar]
- 6.Sakka L, Coll G, Chazal J. Anatomy and physiology of cerebrospinal fluid. Eur ann otorhinolarnygol head neck dis. 2011;128:309–316. doi: 10.1016/j.anorl.2011.03.002. [DOI] [PubMed] [Google Scholar]
- 7.Lund V J. Endoscopic management of cerebrospinal fluid leaks. Am J Rhinol. 2002;16:17–23. [PubMed] [Google Scholar]
- 8.Benjamin E, Wong D K, Choa D. ‘Moffett's’ solution: a review of the evidence and scientific basis for the topical preparation of the nose. Clin Otolaryngol Allied Sci. 2004;29:582–587. doi: 10.1111/j.1365-2273.2004.00894.x. [DOI] [PubMed] [Google Scholar]
- 9.Anverali J K, Hassaan A A, Saleh H A. Endoscopic modified Lothrop procedure for repair of lateral frontal sinus cerebrospinal fluid leak. J Laryngol Otol. 2009;123:145–147. doi: 10.1017/S0022215108002326. [DOI] [PubMed] [Google Scholar]
- 10.Chatrath P, Saleh H A. Endoscopic repair of cerebrospinal fluid rhinorrhea using bone pate. Laryngoscope. 2006;116:1050–1053. doi: 10.1097/01.MLG.0000217644.74806.8F. [DOI] [PubMed] [Google Scholar]
- 11.Kassam A Horowitz M Carrau R et al. Use of Tisseel fibrin sealant in neurosurgical procedures: incidence of cerebrospinal fluid leaks and cost-benefit analysis in a retrospective study Neurosurgery 2003521102–1105., discussion 1105 [PubMed] [Google Scholar]
- 12.Banks C A, Palmer J N, Chiu A G, O'Malley B W Jr, Woodworth B A, Kennedy D W. Endoscopic closure of CSF rhinorrhea: 193 cases over 21 years. Otolaryngol Head Neck Surg. 2009;140:826–833. doi: 10.1016/j.otohns.2008.12.060. [DOI] [PubMed] [Google Scholar]
- 13.Lindstrom D R, Toohill R J, Loehrl T A, Smith T L. Management of cerebrospinal fluid rhinorrhea: the Medical College of Wisconsin experience. Laryngoscope. 2004;114:969–974. doi: 10.1097/00005537-200406000-00003. [DOI] [PubMed] [Google Scholar]
- 14.Mirza S, Thaper A, McClelland L, Jones N S. Sinonasal cerebrospinal fluid leaks: management of 97 patients over 10 years. Laryngoscope. 2005;115:1774–1777. doi: 10.1097/01.mlg.0000175679.68452.75. [DOI] [PubMed] [Google Scholar]
- 15.Psaltis A J, Schlosser R J, Banks C A, Yawn J, Soler Z M. A systematic review of the endoscopic repair of cerebrospinal fluid leaks. Otolaryngol Head Neck Surg. 2012;147:196–203. doi: 10.1177/0194599812451090. [DOI] [PubMed] [Google Scholar]
- 16.Gilat H, Rappaport Z, Yaniv E. Endoscopic transnasal cerebrospinal fluid leak repair: a 10 year experience. Isr Med Assoc J. 2011;13:597–600. [PubMed] [Google Scholar]
- 17.Presutti L, Mattioli F, Villari D, Marchioni D, Alicandri-Ciufelli M. Transnasal endoscopic treatment of cerebrospinal fluid leak: 17 years' experience. Acta Otorhinolaryngol Ital. 2009;29:191–196. [PMC free article] [PubMed] [Google Scholar]
- 18.Liao W, Lin S H, Huang H T. [Traumatic nasal cerebrospinal fluid fistula: report of 86 cases] Di 1 jun yi da xue xue bao = Academic journal of the first medical college of PLA. 2003;23:629–630. [PubMed] [Google Scholar]
- 19.Seth R, Rajasekaran K III, Luong A, Benninger M S, Batra P S. Spontaneous CSF leaks: factors predictive of additional interventions. Laryngoscope. 2010;120:2141–2146. doi: 10.1002/lary.21151. [DOI] [PubMed] [Google Scholar]
- 20.Woodworth B A, Prince A. Chiu AG et al. Spontaneous CSF leaks: a paradigm for definitive repair and management of intracranial hypertension. Otolaryngol Head Neck Surg. 2008;138:715–720. doi: 10.1016/j.otohns.2008.02.010. [DOI] [PubMed] [Google Scholar]
- 21.Wall M. Idiopathic intracranial hypertension (pseudotumor cerebri) Curr Neurol Neurosci Rep. 2008;8:87–93. [PubMed] [Google Scholar]
- 22.Biousse V, Bruce B B, Newman N J. Update on the pathophysiology and management of idiopathic intracranial hypertension. J Neurol Neurosurg Psychiatry. 2012;83:488–494. doi: 10.1136/jnnp-2011-302029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Alexander N S, Chaaban M R, Riley K O, Woodworth B A. Treatment strategies for lateral sphenoid sinus recess cerebrospinal fluid leaks. Arch Otolaryngol Head Neck Surg. 2012;138:471–478. doi: 10.1001/archoto.2012.614. [DOI] [PubMed] [Google Scholar]
- 24.Cassano M, Felippu A. Endoscopic treatment of cerebrospinal fluid leaks with the use of lower turbinate grafts: a retrospective review of 125 cases. Rhinology. 2009;47:362–368. doi: 10.4193/Rhin08.175. [DOI] [PubMed] [Google Scholar]
- 25.Lund V J, Savy L, Lloyd G, Howard D. Optimum imaging and diagnosis of cerebrospinal fluid rhinorrhoea. J Laryngol Otol. 2000;114:988–992. doi: 10.1258/0022215001904572. [DOI] [PubMed] [Google Scholar]
- 26.Bachmann-Harildstad G. Diagnostic values of beta-2 transferrin and beta-trace protein as markers for cerebrospinal fluid fistula. Rhinology. 2008;46:82–85. [PubMed] [Google Scholar]
- 27.Seth R, Rajasekaran K, Benninger M S, Batra P S. The utility of intrathecal fluorescein in cerebrospinal fluid leak repair. Otolaryngol Head Neck Surg. 2010;143:626–632. doi: 10.1016/j.otohns.2010.07.011. [DOI] [PubMed] [Google Scholar]
- 28.Placantonakis D G Tabaee A Anand V K Hiltzik D Schwartz T H Safety of low-dose intrathecal fluorescein in endoscopic cranial base surgery Neurosurgery 2007613, Suppl161–165., discussion 165–166 [DOI] [PubMed] [Google Scholar]
- 29.Prickett K K, Wise S K, Delgaudio J M. Choice of graft material and postoperative healing in endoscopic repair of cerebrospinal fluid leak. Arch Otolaryngol Head Neck Surg. 2011;137:457–461. doi: 10.1001/archoto.2011.12. [DOI] [PubMed] [Google Scholar]
- 30.Harvey R J, Parmar P, Sacks R, Zanation A M. Endoscopic skull base reconstruction of large dural defects: a systematic review of published evidence. Laryngoscope. 2012;122:452–459. doi: 10.1002/lary.22475. [DOI] [PubMed] [Google Scholar]
- 31.Casiano R R, Jassir D. Endoscopic cerebrospinal fluid rhinorrhea repair: is a lumbar drain necessary? Otolaryngol Head Neck Surg. 1999;121:745–750. doi: 10.1053/hn.1999.v121.a98754. [DOI] [PubMed] [Google Scholar]
- 32.Caballero N Bhalla V Stankiewicz J A Welch K C Effect of lumbar drain placement on recurrence of cerebrospinal rhinorrhea after endoscopic repair International Forum of Allergy & Rhinology 2012 [DOI] [PubMed]
- 33.Ramakrishnan V R, Suh J D, Chiu A G, Palmer J N. Reliability of preoperative assessment of cerebrospinal fluid pressure in the management of spontaneous cerebrospinal fluid leaks and encephaloceles. Int Forum Allergy Rhinol. 2011;1:201–205. doi: 10.1002/alr.20010. [DOI] [PubMed] [Google Scholar]
- 34.Eljamel M S. Antibiotic prophylaxis in unrepaired CSF fistulae. Br J Neurosurg. 1993;7:501–505. doi: 10.3109/02688699308995072. [DOI] [PubMed] [Google Scholar]
- 35.Kassam A, Carrau R L, Snyderman C H, Gardner P, Mintz A. Evolution of reconstructive techniques following endoscopic expanded endonasal approaches. Neurosurg Focus. 2005;19:E8. [PubMed] [Google Scholar]
- 36.Carrau R L, Snyderman C, Janecka I P, Sekhar L, Sen C, D'Amico F. Antibiotic prophylaxis in cranial base surgery. Head Neck. 1991;13:311–317. doi: 10.1002/hed.2880130407. [DOI] [PubMed] [Google Scholar]
- 37.Choi D, Spann R. Traumatic cerebrospinal fluid leakage: risk factors and the use of prophylactic antibiotics. Br J Neurosurg. 1996;10:571–575. doi: 10.1080/02688699646880. [DOI] [PubMed] [Google Scholar]
- 38.Shikani A H Use of antibiotics for expansion of the Merocel packing following endoscopic sinus surgery Ear Nose Throat J 199675524–526., 528 [PubMed] [Google Scholar]
- 39.Fridley J, Foroozan R, Sherman V, Brandt M L, Yoshor D. Bariatric surgery for the treatment of idiopathic intracranial hypertension. J Neurosurg. 2011;114:34–39. doi: 10.3171/2009.12.JNS09953. [DOI] [PubMed] [Google Scholar]
- 40.Stangherlin P, Ledeghen S, Scordidis V, Rubay R. Benign intracranial hypertension with recurrent spontaneous cerebrospinal fluid rhinorrhoea treated by laparoscopic gastric banding. Acta Chir Belg. 2008;108:616–618. doi: 10.1080/00015458.2008.11680302. [DOI] [PubMed] [Google Scholar]
- 41.Curry W T Jr Butler W E Barker F G II Rapidly rising incidence of cerebrospinal fluid shunting procedures for idiopathic intracranial hypertension in the United States, 1988-2002 Neurosurgery 20055797–108., discussion 97–108 [DOI] [PubMed] [Google Scholar]
- 42.Ball A K, Howman A, Wheatley K. et al. A randomised controlled trial of treatment for idiopathic intracranial hypertension. J Neurol. 2011;258:874–881. doi: 10.1007/s00415-010-5861-4. [DOI] [PubMed] [Google Scholar]
- 43.Kahle K T, Walcott B P, Staley K J. Resolution of headache and papilledema in idiopathic intracranial hypertension associated with inhibition of Na+−K+−2Cl- cotransport. J Child Neurol. 2011;26:205–208. doi: 10.1177/0883073810391264. [DOI] [PubMed] [Google Scholar]
- 44.Kumpe D A, Bennett J L, Seinfeld J, Pelak V S, Chawla A, Tierney M. Dural sinus stent placement for idiopathic intracranial hypertension. J Neurosurg. 2012;116:538–548. doi: 10.3171/2011.10.JNS101410. [DOI] [PubMed] [Google Scholar]