Summary
The mechanisms by which GM-CSF mediates bacterial clearance and inflammation during mycobacterial infection are poorly understood. The objective of this work was to determine how GM-CSF alters pulmonary mycobacterial infection in vivo. Differences in GM-CSF levels in the lungs of normal mice (GM+/+), transgenic GM-CSF-deficient (GM-CSF−/−), and transgenic mice with high GM-CSF expression only in lung epithelial cells (SP-C-GM-CSF+/+/GM−/−) did not affect pulmonary infection rates caused by either the attenuated Mycobacterium bovis BCG or the virulent Mycobacterium tuberculosis H37Rv. However, in contrast to findings with BCG, all GM-CSF−/− and SP-C-GM-CSF+/+/GM−/− mice succumbed prematurely to virulent H37Rv. Granuloma formation was impaired in both GM-CSF−/− and SP-C-GM-CSF+/+/GM−/− mice regardless of mycobacterial virulence. However, H37Rv-infected GM-CSF−/− mice suffered broncho-alveolar destruction, edema, and necrosis while only short-lived granulomas were observed in SP-C-GM-CSF+/+/GM−/− mice. Bone marrow-derived macrophages, but not dendritic cells of SP-C-GM-CSF+/+/GM−/− mice, were hypo-responsive to mycobacterial infection. Surfactant protein levels were differentially influenced by BCG and H37Rv. We conclude that GM-CSF has an essential protective role first in preserving alveolar structure and second in regulating macrophages and dendritic cells to facilitate containment of virulent mycobacteria in pulmonary granulomas. However, precise regulation of lung GM-CSF is vital to effective control of M. tuberculosis.
Keywords: GM-CSF, Tuberculosis, Macrophages, Granuloma, Alveolar epithelial cells
Introduction
Tuberculosis persists as the leading cause of morbidity and mortality from an infectious disease worldwide. Mycobacterium tuberculosis, the causative agent of tuberculosis, has already infected nearly a third of the world’s population causing approximately 2 million deaths and 8 million new cases of active tuberculosis disease yearly. The high morbidity and mortality from tuberculosis reflects a 10% life-time risk to develop tuberculosis in a vast pool of infected individuals whose immune system restrains but does not eradicate M. tuberculosis infection.1 M. tuberculosis bacteria infect and replicate inside macrophages. Macrophages alone cannot control intracellular mycobacterial growth. Infected macrophages secrete TNFα and chemokines coordinating the recruitment of activated T lymphocytes in granulomas formed at the site of infection. There, activated T cells interact with macrophages and secrete IFNγ that promotes macrophages to kill intracellular mycobacteria. Some mycobacteria, however, undergo a poorly understood adaptation process surviving inside host cells in a latent state without causing clinical disease. Latent M. tuberculosis may re-activate later in life causing secondary tuberculosis that requires antibiotic therapy. The lung is the most susceptible organ to tuberculosis infection representing 85% of clinical tuberculosis cases.1 The pulmonary tropism of tuberculosis disease and the power-lessness of competent cell-mediated immunity in preventing secondary tuberculosis remain unexplained.2,3
Granulocyte–macrophage colony stimulating factor (GM-CSF)5 was originally discovered by its ability to stimulate the differentiation of myeloid progenitors into macrophages and neutrophils.4 Since then its has been established that GM-CSF acts on myeloid cells at all stages of differentiation through a heterodimeric receptor consisting of a and β subunits that mediate binding and intracellular signaling, respectively.4 Mouse lungs were found to have high levels of GM-CSF bio-activity and served as the original source for GM-CSF purification.5 Alveolar macrophages and surrounding epithelial cells, including alveolar Type II epithelial cells, are principal sources of GM-CSF in the lung.6 Stimulation of intercellular interactions among alveolar macrophages and Type II epithelial cells by exposure to pathogens or particulates can elicit local GM-CSF expression and recruitment of bone marrow cells to the lung.7 GM-CSF can also shape the development of dendritic cells. Dendritic cells have a primary responsibility in transport and presentation of antigens to naïve T lymphocytes. GM-CSF acts primarily on macrophages augmenting stimulus-dependent effector functions of macrophages such as induction of antigen-presenting molecules, expression of phagocytic and pattern recognition receptors, and elaboration of inflammatory cytokines, for example, TNFα, which facilitate recruitment of monocytes and T cells to sites of inflammation or infection.4,8
While many cell types secrete GM-CSF after stimulation, constitutive expression of GM-CSF in the lung is critical for steady-state lung function. GM-CSF regulates both pulmonary surfactant homeostasis,9 and the differentiation and proliferation of functionally competent alveolar macro-phages.10,11 Absence or blockade of GM-CSF decreases pulmonary surfactant catabolism,12–17 reduces alveolar macrophage functions,10,18,19 and increases susceptibility to infections.20–24 Complementary studies that assessed the ability of lung-specific expression of GM-CSF to restore pulmonary homeostasis in mice led to the unexpected discovery that GM-CSF influences the proliferation of alveolar Type II epithelial cells, which were shown to express the GM-CSF receptor a subunit.25 The loss of GM-CSF signaling manifests as pulmonary alveolar proteinosis (PAP) in both mice and humans.26 Humans with PAP have apparently intact adaptive immunity even though PAP is associated with increased susceptibility to infectious diseases including infections with mycobacterial organisms.26 Previous studies indicated that GM-CSF augments the mycobactericidal activity of macrophages ex vivo.27,28,29 Consistently, recent studies revealed that GM-CSF-deficient mice are unable to mount effective resistance against M. tuberculosis infection.24 On the other hand, over-expression of GM-CSF in the lungs of GM-CSF-deficient mice24 was also not suitable for effective resistance against tuberculosis30 suggesting a pathogenic role of high GM-CSF levels. Consistent with this notion, the intravenous administration of non-degradable pegylated GM-CSF resulted in high dendritic cell numbers that, paradoxically, increased the susceptibility of the mice to Listeria monocytogenes and M. tuberculosis despite elevated adaptive responses.30
While previous studies demonstrated that GM-CSF levels in the lung influence the clearance rate of a number of pulmonary infections,20–23 the present study reports that GM-CSF levels do not influence the clearance of the attenuated Mycobacterium bovis BCG or the establishment of the virulent M. tuberculosis H37Rv. Our results in GM-CSF−/− mice demonstrate that GM-CSF has a critical early role in preserving the integrity of alveolar epithelial cells in the presence of virulent M. tuberculosis infection. Importantly, we demonstrate that cell-specific expression of GM-CSF in alveolar Type II epithelial cells preserves epithelial integrity in the early stage of M. tuberculosis infection. However, our results also reveal that precise regulation of GM-CSF levels in the lung is necessary to control granulomatous inflammation and long-term containment of M. tuberculosis in the lung. Dys-regulation of GM-CSF may increase the risk for the development of active tuberculosis disease.
Methods
Mice
Specific pathogen-free C57BL/6 mice 4–6 weeks of age were obtained from Jackson Laboratories (Bar Harbor, ME). The generations of GM-CSF-deficient (GM-CSF−/−) 14 and the bitransgenic GM-CSF over-expressing (SP-C-GM-CSF+/+/GM−/−) mice, both bred into the C57BL/6 genetic background, were described previously.12 Both GM-CSF−/− and SP-C-GM-CSF+/+/GM−/− mice were provided by Dr. Jeffrey A. Whitsett (Children’s Hospital Medical Center, Cincinnati, OH). The alveolar Type II epithelial cells of the bi-transgenic SP-C-GM-CSF+/+/GM−/− mice express high levels of GM-CSF only in the lungs of GM-CSF−/− mice. Cell and organ-specific GM-CSF expression was achieved by inserting the GM-CSF gene downstream of the surfactant protein C promoter.12 The level of GM-CSF in the lavage of SP-C-GM-CSF+/+/GM−/− mice was measured by ELISA and confirmed to be in the same range of 200–300 pg/mL as in the original report.12 The physiological concentration of GM-CSF in WT lung lavage is <5pg and below the sensitivity of ELISA assays. All mice were housed and bred under specific pathogen-free conditions. Mice were used in accordance with IACUC approved protocols. Henceforth, the GM-CSF−/− mice will be abbreviated as GMKO and the SP-C-GM-CSF+/+/GM−/− mice will be abbreviated as GMOE.
Bacteria
The M. bovis BCG Tokyo substrain S-1031 (BCG) and M. tuberculosis strain H37Rv (ATCC cat. #27294) (H37Rv) were used in this study. Colony forming units (cfu) grown on 7H11 agar were sub-cultured in 7H9 broth and log phase organisms were harvested 10–12 days later. Bacteria were washed in phosphate buffered saline (PBS) and sonicated at 5W for 15 s to disperse organisms and stored in aliquots at −70 °C in 70% glycerol-PBS. Thawed aliquots was diluted 10-fold in PBS and plated for cfu counts on 7H11 agar.
Infections
Pulmonary infections with BCG were established by intranasal deposition of 1.5×107 BCG under light halothane anesthesia. Sonically dispersed BCG in 40 µL of PBS was distributed slowly in each nostril over 30 s. Aerosol infections with H37Rv were performed in a Middlebrook aerosol exposure chamber (Glascol Inc., Terre Haute, IN). A sonically dispersed suspension of H37Rv at 1 × 106cfu/mL in saline was nebulized for 30 min, which implanted around 100 cfu/ mouse lung. All mouse groups were exposed to the H37Rv aerosol at the same time. Mice were sacrificed on indicated time points and lungs were aseptically removed. Organs were homogenized in PBS with 0.05% Tween-80 and 10-fold dilutions plated on 7H11 agar for enumeration of cfu counts.
Pathology
Lungs were fixed in either 4% paraformaldehyde or 10% formalin overnight and embedded in paraffin. Sections were cut 4.5 µm thick and tissue architecture was visualized following hematoxylin and eosin staining. Images were captured using a Nikon TE 100 microscope equipped with a DXM 1200 digital camera under 2×, 10× or 50 × magnification.
Cytokine measurements
The concentrations of TNFα, GM-CSF or IFNγ were quantitated by standard ELISA assays using commercial kits obtained from either eBiosciences (San Diego, CA) or R&D (Minneapolis, MN). IL-12p70 was quantiated using an ELISA kit from R&D (Minneapolis, MN). The concentration of total IL-12 was measured using an ELISA kit from PEPROTECH (Rocky Hill, NJ). Cytokines were determined in the media of cultured bone marrow-derived macrophages (BMMφ), spleen cells or in lung homogenates obtained from BCG-infected animals. Lung homogenates were centrifuged at 2000 × g, filtered, and stored frozen at −70 °C until use. The protein concentration in lung homogenates was determined spectorophotometrically using the BCA assay (Sigma, St Louis, MO).
Bone marrow-derived macrophages (BMMφ)
BMMφ were obtained by differentiation of bone marrow stem cells in the presence of M-CSF-enriched media from L-cells (LCM) with modifications.32 Briefly, mouse femurs were flushed into a Petri dish with Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% fetal calf serum (FCS) and 1% penicillin-streptomycin solution. Cell aggregates were dispersed using a 1mL pipette tip. Cell suspensions were washed once and transferred into 75 cm2 tissue culture plates in media containing 75% DMEM, 10% FCS, and 25% LCM containing at least 300 pg/mL of M-CSF. The cells were supplemented with 20% LCM on day 3 and an equivalent volume of 20% LCM was exchanged on days 7 and 10 of culture. The LCM-derived BMMφ were used after 7 or 17 days in culture. For experiments, BMMφ were harvested using a trypsin-EDTA solution and cells were re-plated in the same media at a density of 100,000 mm−2 or 80,000 cells per well in 96 well plates. After overnight culture the media were aspirated, and cells placed in DMEM without FCS for 4–8 h prior to infection with BCG. Infections were carried out at a multiplicity of infection (moi) of 5:1 BCG to macrophage. At indicated times the media were collected and kept frozen at −70 °C until use. Alternatively, BMMφ were obtained in the presence of GM-CSF instead of M-CSF-enriched media from L-cells. To induce macrophages, bone marrow stem cells were given 10ng/mL GM-CSF (Cell Sciences, Canton, MA) at the beginning of culture and on day 3. Bone marrow-derived dendritic cells (BMDC) were differentiated in the presence of both 10 ng/mL GM-CSF and 5 ng/mL IL-4 (Cell Sciences) for 7 days. Then, non-adherent CD11c-positive cells were magnetically isolated using beads conjugated with anti-CD11c antibodies (Miltenyi Biotech, Auburn, CA). Media were supplemented with GM-CSF or GM-CS+IL-4 on day 3 of culture. BMMφ and BMDC were studied on day 7 of culture as described above.
Splenocytes
Spleens were removed aseptically and single cell suspensions were obtained by gentle teasing of spleens between sterile glass slides and filtration through a 100-µm cell strainer (Falcon). The red blood cells were lysed in a solution composed of 0.15M NH4Cl, 1 mM KHCO3, and 0.1 mM EDTA, pH 7.4. The suspensions were washed in DMEM supplemented with 10% FCS. Splenocytes were plated in 96 well plates at a concentration of 1 × 106 cells/mL and stimulated with live BCG at an moi of 5:1 BCG per splenocyte. Cytokines were measured in supernatants after 48 h of stimulations.
Northern blot analysis
Total RNA isolation from lung and spleen and Northern blot hybridization procedures were carried out exactly as described previously.33 The 787 bp MARCO DNA probe was generated from a pcDNA plasmid containing the 1.9kb cDNA of mouse MARCO.34 The mouse MARCO cDNA was excised from the plasmid using BamHI and NotI. The excised cDNA was gel-purified as previously described33 and further digested with Xmal, which generated two fragments of 787 and 1100 bp containing the 5′ and 3′ ends of mouse MARCO gene, respectively. The 787 fragment was purified using a Qiagen gel extraction kit (Qiagen, Valencia, CA), radio-actively labeled with 32P, and used on Northern blots as described previously.33
Western blot analysis
Western blot analysis of SP-A and SP-D levels in lung homogenates was carried out according to previously described procedures33 using a rabbit anti-rat SP-A (kind gift of Dr. Francis McCormack, University of Cincinnati, Cincinnati, OH) and rabbit anti-mouse SP-D35 antibodies, respectively.
Statistics
Graphing and statistical analysis was accomplished using Graphpad Prism 4.0 software. Statistical comparisons were assessed by the Student’s t-test, and differences were considered significant at p < 0.05.
Results
Distinct macrophage phenotypes in WT, GMKO, and GMOE mice
Earlier studies reported that GM-CSF influences alveolar macrophage function and the rate of microbial clearance from the lung.20–23 The studies in the present report determined the effect of GM-CSF expression pulmonary mycobacterial infections. Previous studies reported lung-specific arrest in functional activation of alveolar macro-phages marked by depression of innate immune responses including phagocytosis and cytokine secretion in GMKO mice.10,18,36 The phenotypic analysis shown in Figure 1A confirms that alveolar macrophages from GMKO mice have an immature phenotype as judged by expression of the monocytic marker ER-MP20.37 The mRNA analysis shown in Figure 1B demonstrates that the macrophage-specific scavenger receptor MARCO is expressed on WT macro-phages, highly expressed in GMOE, and absent in GMKO alveolar macrophages. MARCO is a type A scavenger receptor that is specifically expressed on highly differentiated alveolar macrophages.34 We determined the expression of MARCO by Northern blot analysis rather than flow cytometry since MARCO protein is mainly stored intracellu-larly in macrophages of naïve mice.38 The absence of MARCO mRNA in GMKO lungs (Figure 1B) is in accordance with the lack of terminal alveolar macrophage differentiation of GMKO alveolar macrophages. However, the high level of MARCO mRNA in GMOE mice is consistent with induction of phagocytic receptors above WT levels by GM-CSF. Thus, the higher expression of innate receptors in GMOE alveolar macrophages may contribute to the enhanced ability of GMOE mice to reduce the burden of infection even more efficiently than WT mice.20–22 Because MARCO is also highly expressed in spleen, we assessed local versus peripheral effects of lung GM-CSF expression (Figure 1B). In contrast to lung, MARCO is expressed in the spleens of GMKO mice indicating that the impact of GM-CSF deficiency on macro-phage differentiation is lung-specific. However, the level of MARCO mRNA in the spleen (Figure 1B) was inversely correlated to the level of GM-CSF in lung suggesting that GM-CSF influences the physiological composition of resident macrophage populations in different organs in addition to having a role in alveolar macrophage differentiation.
Figure 1.
Expression profiles of macrophage differentiation markers in naïve mice. (A) Flow cytometric analysis shows expression of ER-MP20 in GMKO but not in WT or GMOE alveolar macrophages. Data shown are representative of three separate experiments. Open histograms: isotype control IgG2b and shaded gray histograms: anti-ER-MP20. (B) Northern blot hybridization indicates different expression levels of 1.9 kb MARCO mRNA in WTand GMOE lungs. MARCO is not expressed in GMKO lungs. The expression level of MARCO mRNA in spleen is inversely correlated to the level of GM-CSF in the lung. Each lane was loaded with 20µg of total RNA and the results are representative of two independent experiments. Equivalent loading of RNA is indicated by the similar intensity of 28S rRNA on corresponding ethidium bromide-stained gels prior to Northern transfer.
Similar rates of mycobacterial infection in WT, GMKO, and GMOE mice
Based on the above findings and previous results showing that lung GM-CSF significantly alters infection levels in the lungs,20–22 we then tested the hypothesis that high-dose mycobacterial infection would be cleared differently from the lungs of WT, GMKO, and GMOE mice. On the contrary, Figure 2A demonstrates that both the early clearance and persistence of pulmonary BCG was similar in all mice. The number of viable BCG was reduced more rapidly in the first 2 weeks by over 2 log. Subsequently, BCG declined slowly to about 10,000 cfu by 75 days after infection. We then determined the impact of GM-CSF level on establishment of pulmonary infection by virulent mycobacteria. Mice were infected via aerosol with the virulent strain M. tuberculosis H37Rv. Figure 2B demonstrates similar deposition and exponential growth of H37Rv in the lungs of all mice. However, unlike WT and GMOE mice, the proliferation of H37Rv in GMKO mice did not subside after 21 days of infection. On day 29, the H37RV cfu in GMKO lungs was 2 log higher than in WT and GMOE mice. These results indicate that initial infection and proliferation of virulent mycobacteria is not altered by GM-CSF expression. However, GMCSF expression appeared necessary for the long-term control of virulent mycobacteria.
Figure 2.
Infection and survival profiles from pulmonary mycobacterial infections. (A) Similar biphasic reduction of intranasal BCG infection from WT, GMOE, and GMKO lungs. Viable BCG cfu were determined after serial dilution and culture of lung homogenates. Data are means ±S.E.M. Data shown are the combined results from two independent experiments and n = 14 WT mice, 8 GMKO mice, and 8 GMOE mice per time point until day 29. For day 70 n = 3. (B) Similar pulmonary infection and proliferation of aerosolized H37Rv in WT, GMOE, and GMKO mice until day 22. The GMKO lungs had significantly higher burden of H37Rv on day 29. Viable H37Rv cfu were quantitated after serial dilution and culture of lung homogenates. Data are means ±S.E., n = 4 per time point. ****p < 0.001, GMKO versus WT and GMOE on day 29. (C) All WT and GMOE and 80% of GMKO survived the intranasal BCG infection over 150 days of observation. Two GMKO mice were found dead each on days 17 and 19 post-infection without obvious sign of disease. N = 10 for each mouse group. (D) All GMKO and GMOE mice succumbed to H37Rv between 30–35 and 100–150 after infection, respectively. All WT mice survived. WT, n = 21; GMKO, n = 18; and GMOE, n = 22.
Increased susceptibility of GMKO and GMOE mice to virulent mycobacterial infection
The high lung burden of H37Rv in GMKO mice (Figure 2B) indicated increased susceptibility of GMKO mice only to virulent mycobacteria despite similar onset and proliferation of H37Rv as in WT mice. Consequently, mouse survival was assessed as an alternate index of susceptibility to mycobacterial infection. Figure 2C demonstrates that all WT and GMOE mice survived intranasal BCG infection. However, 25% of GMKO mice died at 19–22 days after infection. The cause of death of some GMKO mice could not be evaluated promptly because these mice did not display any signs of morbidity. However, the spike in viable cfu observed in GMKO lungs at 22 days (Figure 2A) indicates that a fraction of GMKO mice lost resistance to the infection. Conversely, Figure 2D demonstrates that all H37Rv-infected GMKO mice and all GMOE suffered 100% mortality between 30–35 and 80–150 days after infection, respectively. These findings revealed that GM-CSF plays a protective role but departure from normal GM-CSF levels compromises long-term resistance to virulent mycobacteria.
GM-CSF regulates granuloma formation
Because granuloma formation is a characteristic host response to mycobacterial infection,39 we determined whether GM-CSF shapes granuloma formation. The comparative histopathology of mouse lungs was evaluated over time after BCG (Figure 3) or H37Rv infection (Figure 4).
Figure 3.
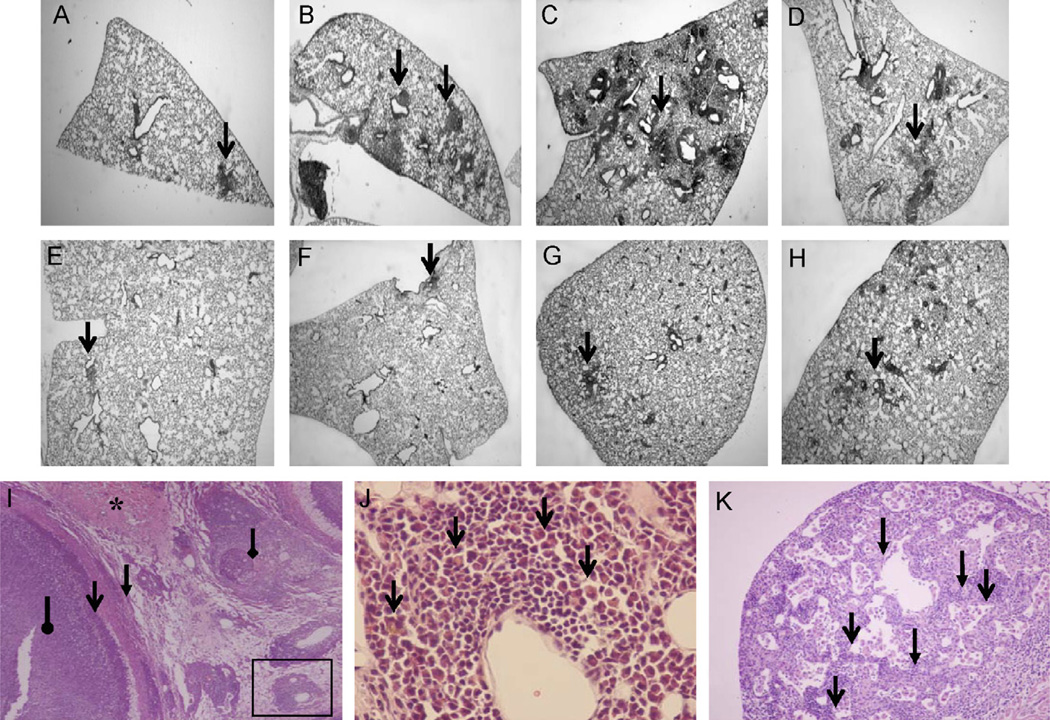
Granuloma formation by intranasal BCG infection. Lung inflammation resulting from intranasal infection of 1.5 × 107 BCG is shown for days 9 (A, E), 14 (B, F), 22 (C, G, I, and J) and 29 (D, H, and K) for WT (A–D), GMKO (E– I and J), and GMOE mice (K). Progressive granulomatous inflammation was observed in WT mice (A–D, arrows). Granulomatous inflammation was attenuated in GMKO mice (E-H, arrows). Two of eight GM−/− mice had encapsulated lesions (I) with a necrotic core (I, oval arrow) surrounded by cellular boundaries (I, open arrow) and fibroblastic granulation tissue (I, closed arrow). Adjacent areas in these lungs contained dense eosinophilic material (I, star), abscesses (I, diamond arrow), and intravascular infiltrates (I, box) rich in plasma cells containing Russell bodies (J, open arrows). The DIP pathology of SP-C-GM+/+ lungs on day 29 is shown on panel (K). Representative images from n = 8 per mouse genotype per time point are shown. Magnification × 2 for images (A–I), × 50 for image (J), and × 10 for image (K).
Figure 4.
Granuloma formation by aerosol infection with H37Rv. Lung inflammation of H37Rv-infected WT (A-E), GMKO (F-J), and GMOE (K-O) mice was evaluated on weekly intervals. Progressive granuloma formation in WT mice (B-E, closed arrows) was typified by the appearance of small interstitial infiltrates on day 7 that became enlarged thereafter, and in close proximity with bronchial and vascular structures. Granuloma formation was completely abrogated in GMKO mice as indicated by the lack of focused inflammation in the parenchyma. Over time, the inflammation in GMKO mice was distinct with scattered alveolar infiltrates (G, closed arrow), peribronchial and perivascular inflammation (H and I, diamond arrows), epithelial necrosis (H, oval arrow), and collapse of broncho-alveolar structures into amorphous necrotic nodules (J, oval arrow). In contrast, GMOE mice developed intense but unstable granulomas in the parenchyma (L-O, closed arrows) and diffuse interstitial inflammation (O, oval arrow). The images shown represent the findings from four mice per group at each time point. Magnification × 10.
Intranasal infection with BCG caused robust bronchogenic and alveolar granulomas only in WT mice (Figure 3A – D). Granulomatous inflammation in WT mice peaked 22 days after infection (Figure 3A – D). In contrast, granuloma formation was markedly attenuated in GMKO mice (Figure 3E – H). Closer examination indicated a diffuse inflammatory disease composed of neutrophils and macrophages (not shown). Interestingly, a fraction of GMKO mice developed well-organized necrotic lesions with a central core of necrosis enveloped by layers of granulation tissue, foamy macrophages, neutrophils, and lymphocytes (Figure 3I). Abscesses were present between necrotic granulomas (Figure 3I). Remarkably, GMKO mice carrying these lesions did not display outward evidence of disease prior to histological assessment, but the discovery of these lesions correlates with the decreased survival of the BCG-infected GMKO mice (Figure 2B). The perivascular inflammation of GMKO lungs was characterized by the presence of undifferentiated plasma cells containing Russell bodies (Figure 3J). Granuloma formation was also absent in GMOE mice (not shown), with a fraction of GMOE mice having developed interstitial lung disease similar to human desquamative interstitial pneumonia with pockets of lymphocytic infiltrates (Figure 3K).
Aerosol infection with H37Rv induced granulomas in WT mice typified by progressive parenchymal, bronchogenic, and perivascular inflammation (Figure 4A – E). In contrast, the inflammatory infiltrates in the lungs of GMKO mice did not consolidate into granulomas (Figure 4F – J). The GMKO lung histology was characterized by sub-mucosal and perivascular mononuclear cell infiltrates, an edematous process beginning as early as 14 days after infection, and formation of amorphous necrotic nodules by day 29 (Figure 4F – J). Under high magnification, the interstitial infiltrates of day 14 infected GMKO lungs (Figure 4H) were composed of lymphocytes, monocytes and plasma cells (not shown). Necrosis, edema, alveolar epithelial destruction, fibrin deposition and plugs of granulation tissue overwhelmed the lung parenchyma of GMKO mice on day 22. On the other hand, local expression of GM-CSF induced intense but transient granulomas and development of interstitial lung disease in GMOE mice (Figure 4K – O). Interstitial infiltrates were readily visible in the parenchyma 7 days after infection (Figure 4K). Many contiguous nodules formed large granulomas on day 15 after infection (Figure 4M). On day 22, the granulomas of GMOE mice were dispersed into many smaller nodules suggesting miliary disease (Figure 4N). The lung pathology on day 29 was characterized by a diffuse interstitial inflammation (Figure 4O). Adjoining lesions resembled desquamative interstitial pneumonia (not shown) as observed in BCG infected lungs (Figure 3K).
Taken together, these results show that granuloma formation in response to both BCG and H37Rv infection was impaired in both GMKO and GMOE mice. Our studies are consistent with previous studies showing the importance of GM-CSF expression in mediating granuloma formation.24,40,41 However, our findings also indicate for the first time that, in addition to granuloma formation, GM-CSF prevents early edema and injury to alveolar cells by virulent mycobacteria. While GM-CSF over-expression was protective early on, the histological analysis indicates that GM-CSF caused abnormal inflammation consistent with the increased susceptibility of the GMOE mice to H37Rv (Figure 2).
Secretion of IL-12 in lung and spleen
Most GMKO and all GMOE were able to control intranasal BCG infection indicating activation of cell-mediated immunity despite the aberrant GM-CSF expression and absence of granulomas in the distal lung. To confirm this observation further, IL-12 levels were measured in lung homogenates of uninfected and BCG-infected mice (Figure 5A). IL-12 is secreted mainly by antigen presenting cells including macrophages, dendritic cells, and B lymphocytes. Expression of IL-12 by dendritic cells is essential for the activation of cell-mediated immunity against both BCG and virulent M. tuberculosis.42 The results in Figure 5A illustrate that the lungs of WT and GMKO mice expressed IL-12 with similar kinetics, indicating that provocation of innate immunity towards a Th1 type immune response is normal even in the absence of GM-CSF. IL-12 was undetectable in uninfected WT and GMKO mice (day 0; Figure 5A). The GMKO lungs secreted more IL-12 on day 22 than WT and GMOE mice. Remarkably, the lungs of uninfected GMOE (day 0) express high levels of IL-12 indicating pre-existing activation of GMOE alveolar macrophages. The level of IL-12 in GMOE mice at 9 days after infection was lower than uninfected mice but was not significantly different on days 22 and 29 compared to uninfected GMOE mice (Figure 5A). Assessment of IL-12 in the spleens of uninfected and BCG immunized mice revealed that splenocytes of previously infected GMKO and GMOE mice secreted significantly more IL-12 (Figure 5B) than WT mice. The basal level of IL-12 (open bars, Figure 5B) in unchallenged splenocytes was similar in all mice and similar to previous observations.43,44 These results suggest that significant changes in GM-CSF levels in the lung alter but do not impair the intrinsic ability of the innate immune system to respond to primary or recalled antigen from pulmonary mycobacterial infection. The IL-12 levels were also similar between H37Rv-infected mice at 22 and 29 days after infection (not shown). The following experiments sought to address the mechanism behind the particular susceptibility of the GM-CSF mice to virulent mycobacteria.
Figure 5.
Secretion of IL-12p70 in lung and spleen. (A) Concentration of IL-12p70 in lung homogenates at indicated times after intranasal BCG infection in WT, GMKO, and GMOE mice. Data are means ±S.E.M. from n = 4–6 mice per time point. (B) Concentration of IL-12p70 in splenocytes from naїve or infected mice 70 days after intranasal BCG infection. Splenocytes were cultured in media (open bars) or in the presence BCG (moi 5:1) (strippled and black bars) for 48h before assessment of IL-12 p40. Spontaneous secretions of IL-12 p40 in media (open bars) are combined data from splenocytes of naïve and infected mice. Data shown are means ±S.E.M. from 4 to 5 mice. *p < 0.05, **p < 0.01, and ***p < 0.001.
Differential effects of mycobacterial infections on alveolar epithelial cells
The tissue injury in H37Rv-infected GMKO lungs (Figure 4) was characterized by edema and necrosis as early as 14 days after infection. Figure 6A shows significant increase in protein levels in GMKO lung homogenates compared to WT and GMOE mice 14 days after infection, consistent with the edematous destructive process observed in the alveoli of H37Rv-infected GMKO mice (Figure 4).
Figure 6.
Effect of BCG and H37Rv on lung protein and GM-CSF levels. (A) Protein concentration in filtered homogenates of H37Rv infected mice was assessed using the BCA assay data are means ±S.D. from 3 to 4 mice. *p < 0.05 and **p < 0.01. The concentration of GM-CSF was measured by ELISA in lung homogenates from BCG (B) and H37Rv-infected (C) GMOE mice. Data shown are means ±S.E.M. from 3 to 4 mice per time point *p < 0.05 and **p < 0.001.
The GMOE lungs failed to either sustain or form granulomas in response to H37Rv or BCG infection (Figure 3 and Figure 4), respectively. Since GMOE mice express GM-CSF only in alveolar Type II cells, we tested the effect of these infections on Type II cell secretory activity. Figure 6B demonstrates that the concentration of GM-CSF declined five-fold between 7 and 22 days remaining at about 50 pg/ mg of protein between 22 and 29 days after infection with H37Rv. The decline of epithelial-derived GM-CSF in GMOE lungs correlates with the dispersal of granulomas and evidence of lung injury in GMOE mice 14 days after infection (Figure 4). In contrast, BCG caused only a small but transient decline in SP-C-driven GM-CSF in GMOE mice (Figure 6C). Together, these results indicate that the GM-CSF is necessary to counteract the cytotoxic effects of virulent H37Rv on alveolar epithelial cells.
The results in Figure 6B and C suggest that alveolar Type II epithelial cells participate in the host response to myco-bacterial infection. To address the effect of mycobacterial infection on alveolar Type II epithelial cells, we evaluated the levels of surfactant proteins A (SP-A) and D (SP-D) of lung homogenates (Figure 7). SP-A and SP-D are secreted mainly by alveolar Type II epithelial cells.45 Western blot analysis of lung homogenates revealed that intranasal BCG infection caused transient reduction of SP-A levels (Figure 7A – C) in all mice and that of SP-D (Figure 7A and B) in WT and GMKO mice. Surfactant protein levels were restored after 14 days of infection. The SP-A levels in GMKO lungs decreased more gradually than WT mice (Figure 7B), likely due to the initial high concentration of surfactant proteins. Given that GMKO lungs are defective in catabolism rather than in surfactant protein synthesis,16,17,46 the observed loss of SP-A and SP-D in Figure 7B is consistent with a direct effect of BCG on secretory functions of alveolar Type II epithelial cells. Together, the temporary alterations in SP-A and SP-D (Figure 7) and the alterations of epithelial-derived GM-CSF levels (Figure 6B and C) indicate that mycobacterial infections influence alveolar Type II cell physiology.
Figure 7.
Expression of SP-A and SP-D after BCG infection. Western blot analysis of lung homogenates from WT (A), GMKO (B), and GMOE (C) following intranasal BCG infection. Each lane was loaded with 20 µg of protein. Proteins were separated on 10% SDS–PAGE gels and blotted nitrocellulose. Blots were probed with anti-SP-A or anti-SP-D antibodies overnight and then with HRP-conjugated goat anti-rabbit antibodies. SP-A and SP-D were visualized by enhanced chemiluminescence for 5–10s on Kodak X-ray film.
Macrophages but not dendritic cells from GMOE mice are hypo-responsive
So far, we have shown that the early death of GMKO mice from H37Rv infection stems from the breach of alveolar epithelium at early stage of infection. Over-expression of GM-CSF protected the mice but was associated with incompetent granuloma formation. The results in Figure 1B suggested that systemic GM-CSF deficiency or over-expression of GM-CSF in a single organ, in this case the lung, altered the physiological composition of macrophages in the spleens of naïve mice as indicated by differences in the levels of MARCO scavenger receptor. Therefore, we designed experiments to address whether there are differences in peripheral functional responses that could contribute to the disorganized granulomatous inflammation in GMOE mice. Figure 8A demonstrates that M-CSF-derived bone marrow macrophages (BMMФ) of WTand GMKO mice secreted similar levels of TNFα in response to BCG infection. In contrast, the GMOE macrophages were hypo-responsive secreting less than 10% of the amount of TNFα produced by WT and GMKO macrophages. In addition, Figure 8B demonstrates that the splenocytes from naїve or previously infected GMOE mice were also hypo-responsive secreting significantly lower levels of TNFα compared to WT and GMKO mice. The spontaneous secretion of TNFα in splenocytes (Figure 8B, open bars) and in media of un-stimulated bone marrow macrophages was negligible. Furthermore, Figure 8C shows that GM-CSF-derived BMMФ from GMOE mice were also hypo-responsive to BCG infection compared to WTand GMKO cells. In contrast, the GM-CSF-derived bone marrow dendritic (BMDC) from GMOE mice were hyper-responsive producing 3–4 times more IL-12p70 than WT and GMKO cells. These findings indicate that the higher level of IL-12p70 in GMOE splenocytes from naïve and BCG infected animals reflects the expression of IL-12p70 in dendritic cells (Figure 8B). The inverse relationship between the functional response of GMOE macrophages and dendritic cells was not stimulus-specific since it was also observed in LPS-treated cells (Figure 8D). However, in this case GMKO dendritic cells were also more responsive to LPS compared to WT mice. Together these findings indicate that over-expression of GM-CSF in the lung disrupts the balance of macrophage and dendritic cells.
Figure 8.
Secretion of TNFα and IL-12p70 in bone marrow and spleen cells. BMMφ from WT, GMKO, and GMOE mice (A, C, and D) were differentiated in either M-CSF (A) or GM-CSF (C and D). Bone marrow-derived dendritic cells (BMDC) cells were differentiated in the presence of GM-CSF and IL-4 and immuno-magnetically separated using anti-CD11c antibodies (C and D). Splenocytes (B) were isolated from either naїve or infected mice 70 days after intranasal immunization with 1 × 105 BCG. Cells were challenged with an moi of 1:1 BCG to cell ratio (A-C), or 100ng/mL LPS (D). The concentrations of TNFα (A and B) or IL-12 p40 (B and D) were measured in supernatants 48h after addition of stimulants. Data shown are means ±S.E.M. from 4 to 5 independent experiments. *p < 0.05, **p < 0.01, and ***p < 0.001.
Discussion
The present study sought to illuminate the mechanisms behind the role of GM-CSF in the pathogenesis of pulmonary tuberculosis. Predicated upon significant differences in macrophage differentiation (Figure 1)10 and rates of pulmonary infection by a number of organisms in WT, GMKO, and GMOE mice,20–22,47 we hypothesized that the rates of mycobacterial infection would also be different. This was not the case as both attenuated BCG and virulent H37Rv mycobacterial strains induced similar infection rates in all mice indicating that susceptibility to tuberculosis is not directly related to the ability of GM-CSF to augment the expression of phagocytic receptors. The major conclusions of the studies undertaken here are two-fold. First, GM-CSF has a fundamental protective role in preserving alveolar integrity and early host responses leading to granuloma formation and containment of virulent mycobacteria. Second, excessive levels of GM-CSF lead to tissue injury and systemic imbalance in macrophages and dendritic cells precluding granuloma formation and long-term containment of virulent mycobacteria.
Our findings in GMKO mice revealed an essential role of GM-CSF in protecting epithelial cells against virulent mycobacteria. In addition, GM-CSF deficiency clearly impaired granuloma formation in response to both BCG and H37Rv-infected mice. These findings are consistent with previous findings showing an important role of GM-CSF in granuloma formation24,40 and the importance of GM-CSF on secretion of macrophage TNFα.10,23,36,48,49 TNFα is a critical mediator of granuloma formation in tuberculosis.24,50,51 However, our findings in GMKO mice indicate that protection of the distal lung epithelial cells must be secured early in the course of the infection even before granuloma formation and immunological containment of virulent mycobacteria can take place locally. The GMKO mice were susceptible only to the virulent H37Rv. The virulent H37Rv strain caused unresolved edema and necrosis in GMKO mice that begun at early stages of infection (Figure 4), indicating that GM-CSF deficiency impaired local cyto-protective responses against virulent mycobacteria. The attenuated BCG strain did not compromise epithelial integrity. However, the BCG infection influenced the secretion of SP-A and SP-D in a transient fashion indicating that alveolar Type II epithelial cells participate in the lung’s response to mycobacterial infection. The ability of both GMKO and GMOE mice to resist BCG infections without granulomas indicates that granulomas are not entirely necessary as long as the broncho-alveolar barrier is not breached and as long as there is sufficient provocation of the local immune system. While intense submucosal infiltration was observed in H37Rv-infected GMKO mice 14 days after infection (Figure 4H), indicating an effort to establish local cell-mediated immunity, these infiltrates remained un-encumbered by the ongoing epithelial destruction in the parenchyma. Virulent mycobacteria have well-known abilities to suppress macrophages,52–54 which may have contributed to local paralysis of macrophage responses and paracrine protection of alveolar epithelial cells in GMKO mice. On the other hand, previous studies showed that alveolar Type II epithelial cells and macrophages collaborate in the translocation of mycobacteria into distal lung parenchyma.55 In addition, alveolar epithelial cell lines in culture are subject to necrosis by virulent but not attenuated mycobacterial organisms.56,57 The attenuation of BCG is related to the loss of the RD1 locus encoding ESAT-6, which is responsible in part for the cytolysis of pneumocytes by virulent mycobacteria.58 The ability of GM-CSF to prevent epithelial cytotoxicity by virulent mycobacteria early in the course of the infection, underlies the significantly reduced rate of mortality of GMOE mice. While it is established that GM-CSF influences the survival and proliferation of macrophages,4 future study is needed to determine how GM-CSF regulates epithelial cell death responses to virulent mycobacterial infection, and, furthermore, to determine why different mycobacteria exhibit different levels of virulence. Our findings support a model where GM-CSF expression is required at the earliest stage of infection to counteract the cytotoxic and immunoevasive activities of virulent mycobacteria on alveolar epithelial cells and macrophages.
Consistently, our findings in GMOE mice indicate that expression of GM-CSF by lung epithelial cells prevented the early edematous and necrotic response to virulent H37Rv increasing the lifespan of the GMOE mice. However, H37Rv also caused swift histological changes characterized by intense but short-lived granulomas in the parenchyma, development of interstitial lung disease, and lung injury in GMOE mice. In a previous study, adenoviral over-expression of GM-CSF was associated with liver injury and inability to form granulomas in response to LPS.59 The liver injury and inflammation was not prevented by neutralization of TNFα.59 The ability of H37Rv, but not BCG, to induce transient granulomas in GMOE mice, indicates that virulent mycobacteria are far more potent at recruitment of inflammatory cells. Both H37Rv and BCG caused DIP-like lesions suggesting over-reactivity of alveolar macrophages. Older reports documented the presence of DIP-like lesions in tuberculosis patients.60,61 However, the DIP-like disease in BCG-infected animals did not render the GMOE mice susceptible to the BCG infection suggesting that DIP was not the cause of death in H37Rv infected mice. The histological analysis indicates the presence of lung injury ensuing 14 days after infection, as evidenced by the disruption of granulomas and the subsequent interstitial inflammation. These changes paralleled the gradual decline in epithelial SP-C-driven GM-CSF levels after 7 days of infection of GMOE mice with H37Rv, suggesting that epithelial cell function was also compromised in GMOE mice but at the later stage of the infection. If epithelial cells are being sacrificed at sites of granuloma formation, these findings also suggest that expression of GM-CSF only by epithelial cells is not sufficient to sustain granulomas. However, our findings that BMMφ and splenocytes from GMOE mice are hypo-responsive to stimulation, secreting low levels of TNFα and IL-12, suggest a functional dichotomy in function and balance between macrophages and dendritic cells that can lead to impairment of granuloma formation and abnormal inflammation in GMOE mice. The hyper-responsive phenotype of GMOE dendritic cells, which secrete high levels of IL-12p70 compared to WT cells, signifies bias of GMOE dendritic cells towards proinflammatory responses. The marked tissue reactivity and demise of GMOE mice to H37Rv but not BCG infections suggests detrimental pathogenic interactions between virulent mycobacteria and IL-12-secreting dendritic cells in vivo.
In conclusion, the present findings support a disease model where GM-CSF, in precisely regulated levels, has a fundamental role in a balanced innate host defense against tuberculosis by its role in preserving the integrity of alveolar epithelial cells and in regulating macrophages and dendritic cells to facilitate containment of virulent mycobacteria in pulmonary granulomas. The present findings support the hypothesis that prolonged dys-regulation of GM-CSF expression imposes a risk for the development of pulmonary tuberculosis in immuno-competent individuals.
Acknowledgements
We thank Devin R. Lindsey (University of Texas Health Sciences Center at Houston, Houston, TX) for technical assistance in carrying out serial dilutions and Dr. Malini Rajagopalan (Department of Microbiology, University of Texas Health Center at Tyler) for providing the M. bovis BCG strain. We thank Dr. Jeffrey A. Whitsett (Cincinnati Children’s Hospital Medical, Cincinnati, OH) and Dr. Francis X. McCormack (University of Cincinnati College of Medicine, Cincinnati, OH) for the critical review of the manuscript. We thank Dr. Jeffrey A. Whitsett and Bruce Trapnell (Cincinnati Children’s Hospital Medical, Cincinnati, OH) for providing SP-A antibodies and the GM-CSF transgenic mice and Dr. Francis X. McCormack for providing the SP-D antibodies.
Funding: This work was supported by NIH grants HL068127 and HL068520.
Abbreviations
- GM-CSF
granulocyte-macrophage colony stimulating factor
- M-CSF
macrophage colony stimulating factor
- LCM
L-cell conditioned medium
- DIP
desquamative interstitial pneumonia
- WT
wild type
- GMKO
GM-CSF knockout
- GMOE
GM-CSF over-expression
- PBS
phosphate buffered saline
- DMEM
Dulbecco’s modified Eagle’s medium
- moi
multiplicity of infection
Footnotes
Competing Interests: None declared
Ethical Approval: Not required
References
- 1.Maher D, Raviglione M. Global epidemiology of tuberculosis. Clin Chest Med. 2005;26:167–182. doi: 10.1016/j.ccm.2005.02.009. v. [DOI] [PubMed] [Google Scholar]
- 2.Flynn JL, Chan J. Immunology of tuberculosis. Annu Rev Immunol. 2001;19:93–129. doi: 10.1146/annurev.immunol.19.1.93. [DOI] [PubMed] [Google Scholar]
- 3.North RJ, Jung YJ. Immunity to tuberculosis. Annu Rev Immunol. 2004;22:599–623. doi: 10.1146/annurev.immunol.22.012703.104635. [DOI] [PubMed] [Google Scholar]
- 4.Fleetwood AJ, Cook AD, Hamilton JA. Functions of granulocy-te-macrophage colony-stimulating factor. Crit Rev Immunol. 2005;25:405–428. doi: 10.1615/critrevimmunol.v25.i5.50. [DOI] [PubMed] [Google Scholar]
- 5.Burgess AW, Metcalf D, Sparrow LG, Simpson RJ, Nice EC. Granulocyte/macrophage colony-stimulating factor from mouse lung conditioned medium Purification of multiple forms and radioiodination. Biochem J. 1986;235:805–814. doi: 10.1042/bj2350805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Christensen PJ, Armstrong LR, Fak JJ, Chen GH, McDonald RA, Toews GB, et al. Regulation of rat pulmonary dendritic cell immunostimulatory activity by alveolar epithelial cell-derived granulocyte macrophage colony-stimulating factor. Am J Respir Cell Mol Biol. 1995;13:426–433. doi: 10.1165/ajrcmb.13.4.7546772. [DOI] [PubMed] [Google Scholar]
- 7.Ishii H, Hayashi S, Hogg JC, Fujii T, Goto Y, Sakamoto N, et al. Alveolar macrophage–epithelial cell interaction following exposure to atmospheric particles induces the release of mediators involved in monocyte mobilization and recruitment. Respir Res. 2005;6:87. doi: 10.1186/1465-9921-6-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cook AD, Braine EL, Hamilton JA. Stimulus-dependent requirement for granulocyte-macrophage colony-stimulating factor in inflammation. J Immunol. 2004;173:4643–4651. doi: 10.4049/jimmunol.173.7.4643. [DOI] [PubMed] [Google Scholar]
- 9.Trapnell BC. Granulocyte macrophage-colony stimulating factor augmentation therapy in sepsis: is there a role? Am J Respir Crit Care Med. 2002;166:129–130. doi: 10.1164/rccm.2205017. [DOI] [PubMed] [Google Scholar]
- 10.Shibata Y, Berclaz PY, Chroneos ZC, Yoshida M, Whitsett JA, Trapnell BC. GM-CSF regulates alveolar macrophage differentiation and innate immunity in the lung through PU.1. Immunity. 2001;15:557–567. doi: 10.1016/s1074-7613(01)00218-7. [DOI] [PubMed] [Google Scholar]
- 11.Lin HS, Lokeshwar BL, Hsu S. Both granulocyte-macrophage CSF and macrophage CSF control the proliferation and survival of the same subset of alveolar macrophages. J Immunol. 1989;142:515–519. [PubMed] [Google Scholar]
- 12.Huffman JA, Hull WM, Dranoff G, Mulligan RC, Whitsett JA. Pulmonary epithelial cell expression of GM-CSF corrects the alveolar proteinosis in GM-CSF-deficient mice. J Clin Invest. 1996;97:649–655. doi: 10.1172/JCI118461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ikegami M, Ueda T, Hull W, Whitsett JA, Mulligan RC, Dranoff G, et al. Surfactant metabolism in transgenic mice after granulo-cyte macrophage-colony stimulating factor ablation. Am J Physiol. 1996;270:L650–L658. doi: 10.1152/ajplung.1996.270.4.L650. [DOI] [PubMed] [Google Scholar]
- 14.Dranoff G, Crawford AD, Sadelain M, Ream B, Rashid A, Bronson RT, et al. Involvement of granulocyte-macrophage colony-stimulating factor in pulmonary homeostasis. Science. 1994;264:713–176. doi: 10.1126/science.8171324. [DOI] [PubMed] [Google Scholar]
- 15.Stanley E, Lieschke GJ, Grail D, Metcalf D, Hodgson G, Gall JA, et al. Granulocyte/macrophage colony-stimulating factor-deficient mice show no major perturbation of hematopoiesis but develop a characteristic pulmonary pathology. Proc Natl Acad Sci USA. 1994;91:5592–5596. doi: 10.1073/pnas.91.12.5592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ikegami M, Jobe AH, Huffman Reed JA, Whitsett JA. Surfactant metabolic consequences of overexpression of GM-CSF in the epithelium of GM-CSF-deficient mice. Am J Physiol. 1997;273:L709–L714. doi: 10.1152/ajplung.1997.273.4.L709. [DOI] [PubMed] [Google Scholar]
- 17.Yoshida M, Ikegami M, Reed JA, Chroneos ZC, Whitsett JA. GM-CSF regulates protein and lipid catabolism by alveolar macro-phages. Am J Physiol Lung Cell Mol Physiol. 2001;280:L379–L386. doi: 10.1152/ajplung.2001.280.3.L379. [DOI] [PubMed] [Google Scholar]
- 18.Paine III R, Morris SB, Jin H, Wilcoxen SE, Phare SM, Moore BB, et al. Impaired functional activity of alveolar macrophages from GM-CSF-deficient mice. Am J Physiol Lung Cell Mol Physiol. 2001;281:L1210–L1218. doi: 10.1152/ajplung.2001.281.5.L1210. [DOI] [PubMed] [Google Scholar]
- 19.Bonfield TL, Raychaudhuri B, Malur A, Abraham S, Trapnell BC, Kavuru MS, et al. PU.1 regulation of human alveolar macro-phage differentiation requires granulocyte–macrophage colony-stimulating factor. Am J Physiol Lung Cell Mol Physiol. 2003;285:L1132–L1136. doi: 10.1152/ajplung.00216.2003. [DOI] [PubMed] [Google Scholar]
- 20.Ballinger MN, Paine III R, Serezani CH, Aronoff DM, Choi ES, Standiford TJ, et al. Role of granulocyte macrophage colony-stimulating factor during gram-negative lung infection with pseudomonas aeruginosa. Am J Respir Cell Mol Biol. 2006;34:766–774. doi: 10.1165/rcmb.2005-0246OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Paine III R, Preston AM, Wilcoxen S, Jin H, Siu BB, Morris SB, et al. Granulocyte-macrophage colony-stimulating factor in the innate immune response to Pneumocystis carinii pneumonia in mice. J Immunol. 2000;164:2602–2609. doi: 10.4049/jimmunol.164.5.2602. [DOI] [PubMed] [Google Scholar]
- 22.LeVine AM, Reed JA, Kurak KE, Cianciolo E, Whitsett JA. GM-CSF-deficient mice are susceptible to pulmonary group B streptococcal infection. J Clin Invest. 1999;103:563–569. doi: 10.1172/JCI5212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Deepe GS, Jr, Gibbons R, Woodward E. Neutralization of endogenous granulocyte-macrophage colony-stimulating factor subverts the protective immune response to Histoplasma capsulatum . J Immunol. 1999;163:4985–4993. [PubMed] [Google Scholar]
- 24.Gonzalez-Juarrero M, Hattle JM, Izzo A, Junqueira-Kipnis AP, Shim TS, Trapnell BC, et al. Disruption of granulocyte macro-phage-colony stimulating factor production in the lungs severely affects the ability of mice to control Mycobacterium tuberculosis infection. J Leukoc Biol. 2005;77:914–922. doi: 10.1189/jlb.1204723. [DOI] [PubMed] [Google Scholar]
- 25.Huffman Reed JA, Rice WR, Zsengeller ZK, Wert SE, Dranoff G, Whitsett JA. GM-CSF enhances lung growth and causes alveolar type II epithelial cell hyperplasia in transgenic mice. Am J Physiol. 1997;273:L715–L725. doi: 10.1152/ajplung.1997.273.4.L715. [DOI] [PubMed] [Google Scholar]
- 26.Seymour JF, Presneill JJ. Pulmonary alveolar proteinosis: progress in the first 44 years. Am J Respir Crit Care Med. 2002;166:215–235. doi: 10.1164/rccm.2109105. [DOI] [PubMed] [Google Scholar]
- 27.Denis M. Tumor necrosis factor, granulocyte macrophage-colony stimulating factor stimulate human macrophages to restrict growth of virulent Mycobacterium avium and to kill avirulent M avium: killing effector mechanism depends on the generation of reactive nitrogen intermediates. J Leukoc Biol. 1991;49:380–387. doi: 10.1002/jlb.49.4.380. [DOI] [PubMed] [Google Scholar]
- 28.Denis M, Ghadirian E. Granulocyte-macrophage colony-stimulating factor restricts growth of tubercle bacilli in human macrophages. Immunol Lett. 1990;24:203–206. doi: 10.1016/0165-2478(90)90049-v. [DOI] [PubMed] [Google Scholar]
- 29.Bermudez LE, Young LS. Recombinant granulocyte-macrophage colony-stimulating factor activates human macrophages to inhibit growth or kill Mycobacterium avium complex. J Leukoc Biol. 1990;48:67–73. doi: 10.1002/jlb.48.1.67. [DOI] [PubMed] [Google Scholar]
- 30.Alaniz RC, Sandall S, Thomas EK, Wilson CB. Increased dendritic cell numbers impair protective immunity to intracellular bacteria despite augmenting antigen-specific CD8+ T lymphocyte responses. J Immunol. 2004;172:3725–3735. doi: 10.4049/jimmunol.172.6.3725. [DOI] [PubMed] [Google Scholar]
- 31.Abe E, Ishimi Y, Jin CH, Hong MH, Sato T, Suda T. Granulocyte-macrophage colony-stimulating factor is a major macrophage fusion factor present in conditioned medium of concanavalin A-stimulated spleen cell cultures. J Immunol. 1991;147:1810–1815. [PubMed] [Google Scholar]
- 32.Chroneos ZC, Abdolrasulnia R, Whitsett JA, Rice WR, Shepherd VL. Purification of a cell-surface receptor for surfactant protein A. J Biol Chem. 1996;271:16375–16383. doi: 10.1074/jbc.271.27.16375. [DOI] [PubMed] [Google Scholar]
- 33.Yang CH, Szeliga J, Jordan J, Faske S, Sever-Chroneos Z, Dorsett B, et al. Identification of the surfactant protein A receptor 210 as the unconventional myosin 18A. J Biol Chem. 2005;280:34447–34457. doi: 10.1074/jbc.M505229200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Elomaa O, Kangas M, Sahlberg C, Tuukkanen J, Sormunen R, Liakka A, et al. Cloning of a novel bacteria-binding receptor structurally related to scavenger receptors and expressed in a subset of macrophages. Cell. 1995;80:603–609. doi: 10.1016/0092-8674(95)90514-6. [DOI] [PubMed] [Google Scholar]
- 35.Zhang L, Hartshorn KL, Crouch EC, Ikegami M, Whitsett JA. Complementation of pulmonary abnormalities in SP-D(−/−) mice with an SP-D/conglutinin fusion protein. J Biol Chem. 2002;277:22453–22459. doi: 10.1074/jbc.M201632200. [DOI] [PubMed] [Google Scholar]
- 36.Berclaz PY, Carey B, Fillipi MD, Wernke-Dollries K, Geraci N, Cush S, et al. GM-CSF regulates a PU.1-dependent transcriptional program determining the pulmonary response to LPS. Am J Respir Cell Mol Biol. 2006 doi: 10.1165/rcmb.2006-0174OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.de Bruijn MF, Slieker WA, van der Loo JC, Voerman JS, van Ewijk W, Leenen PJ. Distinct mouse bone marrow macrophage precursors identified by differential expression of ER-MP12 and ER-MP20 antigens. Eur J Immunol. 1994;24:2279–2284. doi: 10.1002/eji.1830241003. [DOI] [PubMed] [Google Scholar]
- 38.Kvell K, Czompoly T, Pikkarainen T, Balogh P. Species-specific restriction of cell surface expression of mouse MARCO glycoprotein in murine cell lines. Biochem Biophys Res Commun. 2006;341:1193–1202. doi: 10.1016/j.bbrc.2006.01.083. [DOI] [PubMed] [Google Scholar]
- 39.Ulrichs T, Kaufmann SH. New insights into the function of granulomas in human tuberculosis. J Pathol. 2006;208:261–269. doi: 10.1002/path.1906. [DOI] [PubMed] [Google Scholar]
- 40.Wynn AA, Miyakawa K, Miyata E, Dranoff G, Takeya M, Takahashi K. Role of granulocyte/macrophage colony-stimulating factor in zymocel-induced hepatic granuloma formation. Am J Pathol. 2001;158:131–145. doi: 10.1016/S0002-9440(10)63951-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Xing Z, Braciak T, Ohkawara Y, Sallenave JM, Foley R, Sime PJ, et al. Gene transfer for cytokine functional studies in the lung: the multifunctional role of GM-CSF in pulmonary inflammation. J Leukoc Biol. 1996;59:481–488. doi: 10.1002/jlb.59.4.481. [DOI] [PubMed] [Google Scholar]
- 42.Cooper AM, Kipnis A, Turner J, Magram J, Ferrante J, Orme IM. Mice lacking bioactive IL-12 can generate protective, antigen-specific cellular responses to mycobacterial infection only if the IL-12 p40 subunit is present. J Immunol. 2002;168:1322–1327. doi: 10.4049/jimmunol.168.3.1322. [DOI] [PubMed] [Google Scholar]
- 43.Sam H, Stevenson MM. Early IL-12 p70, but not p40, production by splenic macrophages correlates with host resistance to blood-stage Plasmodium chabaudi AS malaria. Clin Exp Immunol. 1999;117:343–349. doi: 10.1046/j.1365-2249.1999.00966.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sam H, Stevenson MM. In vivo IL-12 production and IL-12 receptors beta1 and beta2 mRNA expression in the spleen are differentially up-regulated in resistant B6 and susceptible A/J mice during early blood-stage Plasmodium chabaudi AS malaria. J Immunol. 1999;162:1582–1589. [PubMed] [Google Scholar]
- 45.McCormack FX, Whitsett JA. The pulmonary collectins, SP-A and SP-D, orchestrate innate immunity in the lung. J Clin Invest. 2002;109:707–712. doi: 10.1172/JCI15293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gurel O, Ikegami M, Chroneos ZC, Jobe AH. Macrophage and type II cell catabolism of SP-A and saturated phosphatidylcholine in mouse lungs. Am J Physiol Lung Cell Mol Physiol. 2001;280:L1266–L1272. doi: 10.1152/ajplung.2001.280.6.L1266. [DOI] [PubMed] [Google Scholar]
- 47.Deepe GS, Jr, Gibbons R. Recombinant murine granulocyte-macrophage colony-stimulating factor modulates the course of pulmonary histoplasmosis in immunocompetent and immunodeficient mice. Antimicrob Agents Chemother. 2000;44:3328–3336. doi: 10.1128/aac.44.12.3328-3336.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bozinovski S, Jones JE, Vlahos R, Hamilton JA, Anderson GP. Granulocyte/macrophage-colony-stimulating factor (GM-CSF) regulates lung innate immunity to lipopolysaccharide through Akt/Erk activation of NFkappa B and AP-1 in vivo. J Biol Chem. 2002;277:42808–42814. doi: 10.1074/jbc.M207840200. [DOI] [PubMed] [Google Scholar]
- 49.Bozinovski S, Jones J, Beavitt SJ, Cook AD, Hamilton JA, Anderson GP. Innate immune responses to LPS in mouse lung are suppressed and reversed by neutralization of GM-CSF via repression of TLR-4. Am J Physiol Lung Cell Mol Physiol. 2004;286:L877–L885. doi: 10.1152/ajplung.00275.2003. [DOI] [PubMed] [Google Scholar]
- 50.Flynn JL, Goldstein MM, Chan J, Triebold KJ, Pfeffer K, Lowenstein CJ, et al. Tumor necrosis factor-alpha is required in the protective immune response against Mycobacterium tuberculosis in mice. Immunity. 1995;2:561–572. doi: 10.1016/1074-7613(95)90001-2. [DOI] [PubMed] [Google Scholar]
- 51.Roach DR, Bean AG, Demangel C, France MP, Briscoe H, Britton WJ. TNF regulates chemokine induction essential for cell recruitment, granuloma formation, and clearance of mycobacterial infection. J Immunol. 2002;168:4620–4627. doi: 10.4049/jimmunol.168.9.4620. [DOI] [PubMed] [Google Scholar]
- 52.Rao V, Gao F, Chen B, Jacobs WR, Jr, Glickman MS. Trans-cyclopropanation of mycolic acids on trehalose dimycolate suppresses Mycobacterium tuberculosis-induced inflammation and virulence. J Clin Invest. 2006;116:1660–1667. doi: 10.1172/JCI27335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pennini ME, Pai RK, Schultz DC, Boom WH, Harding CV. Mycobacterium tuberculosis 19-kDa lipoprotein inhibits IFN-gamma-induced chromatin remodeling of MHC2TA by TLR2 and MAPK signaling. J Immunol. 2006;176:4323–4330. doi: 10.4049/jimmunol.176.7.4323. [DOI] [PubMed] [Google Scholar]
- 54.Pathak SK, Basu S, Bhattacharyya A, Pathak S, Kundu M, Basu J. Mycobacterium tuberculosis lipoarabinomannan-mediated IRAK-M induction negatively regulates Toll-like receptor-dependent interleukin-12 p40 production in macrophages. J Biol Chem. 2005;280:42794–42800. doi: 10.1074/jbc.M506471200. [DOI] [PubMed] [Google Scholar]
- 55.Bermudez LE, Sangari FJ, Kolonoski P, Petrofsky M, Goodman J. The efficiency of the translocation of Mycobacterium tuberculosis across a bilayer of epithelial and endothelial cells as a model of the alveolar wall is a consequence of transport within mononuclear phagocytes and invasion of alveolar epithelial cells. Infect Immun. 2002;70:140–146. doi: 10.1128/IAI.70.1.140-146.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Danelishvili L, McGarvey J, Li YJ, Bermudez LE. Mycobacterium tuberculosis infection causes different levels of apoptosis and necrosis in human macrophages and alveolar epithelial cells. Cell Microbiol. 2003;5:649–660. doi: 10.1046/j.1462-5822.2003.00312.x. [DOI] [PubMed] [Google Scholar]
- 57.Dobos KM, Spotts EA, Quinn FD, King CH. Necrosis of lung epithelial cells during infection with Mycobacterium tuberculosis is preceded by cell permeation. Infect Immun. 2000;68:6300–6310. doi: 10.1128/iai.68.11.6300-6310.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hsu T, Hingley-Wilson SM, Chen B, Chen M, Dai AZ, Morin PM, et al. The primary mechanism of attenuation of Bacillus Calmette-Guerin is a loss of secreted lytic function required for invasion of lung interstitial tissue. Proc Natl Acad Sci USA. 2003;100:12420–12425. doi: 10.1073/pnas.1635213100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hirano K, Shimizu Y, Nakayama Y, Minemura M, Yasumura S, Sugiyama T. Overexpression of granulocyte-macrophage colony-stimulating factor in mouse liver enhances the susceptibility of lipopolysaccharide leading to massive apoptosis of hepatocytes. Liver Int. 2005;25:1027–1035. doi: 10.1111/j.1478-3231.2005.01136.x. [DOI] [PubMed] [Google Scholar]
- 60.Wipf R. Desquamative interstitial pneumopathy in a North African schoolchild probably associated with tuberculosis. Schweiz Med Wochenschr. 1970;100:1845–1846. [PubMed] [Google Scholar]
- 61.Thurlbeck W, Abell MR, editors. Clinical-pathologic approach to diffuse infiltrative lung disease. The lung: structure, function, and disease. 1978. p. 78. [Google Scholar]