Abstract
Efficiency in surgical procedures saves time and money and can decrease medical complications. Several sources of inefficiency exist in the operating room, including preoperative and intraoperative. The instruments used during TKA are frequently redundant. Customized instruments and implants can improve efficiency by reducing steps. Additional benefits may include improved alignment and kinematics. This chapter addresses the various sources of inefficiency, provides suggestions to overcome them, and introduces the concept of customized guides and implants as a method to improve efficiency.
Keywords: Knee replacement, Efficiency, Customized instruments, Patient specific implants
Introduction
The primary goal of increased efficiency during TKA is reduced surgical time. Many benefits have been shown from reduced OR time, including deceased risk of thromboembolic disease [1], decreased risk of infection [2], decreased blood loss [3], lower revision rates [4], decreased cost [5••], increased number of surgical cases [5••], and more free time for surgeons [5••]. The latter 2 can also be significantly affected by decreased turnover time between cases. A secondary goal of improved efficiency is reduced cost. This article will discuss areas of inefficiency during the operative event, and how customized implants and patient specific instruments can be implemented to enhance efficiency of TKA.
Sources of inefficiency during TKA
Perioperative
Prior to the start of any operation, instrument preparation is required. For TKA, current implant systems from most manufacturers require 5–7 trays of instruments such as resection guides and trial implants. In any given case, however, only a small percentage of instruments are actually used. Nevertheless, every single instrument has to be cleaned and sterilized prior to the surgery and then re-cleaned and re-sterilized after the surgery. It has been estimated that the cost to re-process a tray is between 75–120 US dollars [6]. So for 7 trays, that translates to 525–840 US dollars per case. Of course, this is a hidden cost which very few hospitals have the ability to measure accurately. Regardless, the instruments do not clean and sterilize themselves, and the cost associated with this step is therefore very real. Similarly, sterilized trays often become contaminated after processing and prior to use. This necessitates repeat processing, adding another hidden cost to the operative episode.
Intraoperative
During the surgery, there are several sources of inefficiency. As mentioned above, many instruments that will never be used are opened and placed on the back tables. Scrub staffs have to arrange the instruments that they anticipate will be needed so that they are handy (Fig. 1). To find these tools, the scrub staff must sort through the unnecessary instruments. Frequently the implant trays are not organized in the manner in which the instruments will be used, thereby increasing the time and effort to organize the instruments. When an unexpected tool is needed, the scrub nurse must first locate the proper tray, and then isolate the proper instrument. This increases their work and leads to unnecessary delays. To avoid these delays, the scrub staff will often hedge their bets and add occasionally used instruments to the sets, further compounding the problem of extraneous instrument setup and reprocessing. In one hospital in which the author works, 363 instruments are opened for every TKA case, when fewer than 50 are routinely used. To compound the ludicrousness of this approach, one retractor in the set has not been actually used in 24 years; yet it has been made available for every surgery, “just in case.”
Fig. 1.
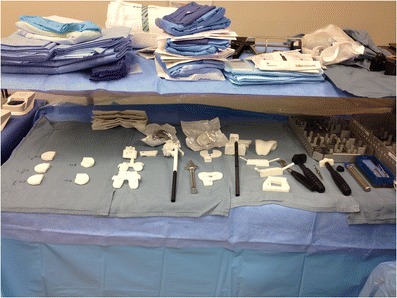
Photograph of the back table showing all the customized instruments needed for customized TKA
Another source of inefficiency is surgeon and scrub staff unfamiliarity with a particular implant system. This occurs when surgeons use many different implant systems, or frequently change systems. Similarly, if scrub staffs are expected to be “jacks of all trades,” covering orthopedic, general surgery, obstetric, and neurosurgical procedures, they will never have the opportunity to be proficient in any one area. It is interesting that hospital administrators accept that highly educated and trained physicians practice in 1 surgical specialty, yet they expect scrub techs and nurses, with less education and training, to be experts in multiple specialties.
Lastly, some blame for inefficiency must be placed at the feet of the surgeon. If a surgeon shows up late for the start of a case, not only is there an unnecessary delay, but it also sets the tone for the rest of the staff that a lackadaisical attitude is acceptable. Frequently, surgeons will not have a set approach to the procedure, changing technique as often as from case to case. This confuses the staff and makes it difficult for them to predict which instruments will be needed and when. Another common mistake is for the surgeon not having a plan B, or if needed a plan C, in mind. Thus, when something unexpected happens, the surgery must be halted while the surgeon first determines the new course, and then second while staff scrambles to find new instrument sets to follow that course. We have all been in the situation where the needed instruments are not sterile or are completely unavailable in the hospital. Although detailed approaches to improve surgeon planning are beyond the scope of this paper, it is nevertheless worthwhile to underscore the importance of preoperative planning and preparation, even for seemingly routine cases.
Methods to improve efficiency
Streamline instrument sets
A simple approach to reduce the number of unutilized instruments in your OR is to streamline your sets. First, decide on 1 or 2 implant systems that you will use for the majority of your cases. Then actually write down in detail the steps of how you prefer to do a TKA. Eliminate redundant or repetitive steps. An example is depicted in Table 1. From there, determine which non-implant specific instruments you use (eg, retractors, power instruments, forceps, etc.) Create a dedicated TKA set of instruments for your TKA cases. This may require your hospital to purchase some new instruments, but quite likely those instruments are just lying around not being used. Regardless, the time saved in turnover and re-processing will easily make up for the effort of being streamlined. Then, go through your implant trays and rearrange them in the way you actually do the operation. If that is not possible because many different surgeons use the same sets, at least you have a written list of what you will need and when to which the scrub staff can easily refer. One approach I used was to laminate this list so that it could be sterilized and placed on the back table for easy reference.
Table 1.
Steps utilized for performance of total knee arthroplasty
| SURGEON | First Assistant | Second Assistant |
|---|---|---|
| Exsanguinate | Hold leg | |
| Leg in leg holder | ||
| Flex to 30 degrees | ||
| Incision | One rake distal | Two rakes proximal |
| Elevate medial skin flap | ||
| Elevate lateral skin flap | ||
| Bovie to incise capsule and VMO | Suction fluid | |
| Kocher to capsule flap | Suction smoke | Hold Kocher |
| Excise synovium | Remove Kocher | |
| Extend knee | Hold one rake | Opposite rake |
| Forceps and bovie to elevate medial tibial soft tissue | Suction smoke | |
| 1/2-inch curved osteotome and bovie | ||
| Bovie to prepat fat pad | ||
| Place Army Navy | Hold Army-Navy | |
| Excise fat pad | ||
| Tilt patella to 90 degrees | ||
| Place 2 towel clips | Hold towel clips | Rake to medial skin |
| Forceps and bovie to clean up fat | ||
| Caliper to measure patella | ||
| Saw to cut patella | Remove bone | |
| Remeasure with caliper | ||
| Adjust with saw | ||
| Measure size | ||
| Patellar drill guide and drill | ||
| Patellar trial and measure | ||
| Readjust if needed | ||
| Fork to lateral femur | Suction | Hold rake to medial muscle |
| Forceps and bovie to excise fat pad | Rotate fork laterally | |
| Flex knee to 60 degrees | Rake to medial skin | |
| 1/2 in. curved osteotome/mallet | Rongeur bone | |
| Kocher/knife excise ACL/PCL | ||
| IM femoral drill | ||
| IM guide and mallet | Place one headed pin | Rotate fork |
| Mallet | ||
| Distal cutter | Two pins | |
| Mallet | Screwdriver | |
| Flex Knee | ||
| Slap hammer to remove sword | ||
| Smiley around MFC | Hold smiley | |
| Saw to cut MFC | Kocher | |
| Move smiley laterally | Hold smiley | |
| Saw to cut LFC | Kocher | Remove smiley |
| Slap hammer | Remove distal cutter | Remove fork |
| Blunt Hohmann to post tib | Rake medially | Hold Hohman |
| Fork to lat tibia | Hold fork | |
| Kocher/bovie to remove menisci | Suction smoke | |
| Adjust tibial cutter height | ||
| Place first pin | ||
| Adjust tibial cutter alignment | ||
| Angel wing to check | Screwdriver | |
| Place second pin | ||
| Sharp Hohmann medially | Hold sharp Hohmann | |
| Saw | ||
| Remove pins | Remove tibial cutter | |
| Finish cut with saw | ||
| Kocher and bovie | ||
| Adjust retractors | ||
| Mark rotation | ||
| Tibial plate and rod | ||
| Adjust cut if needed | ||
| Small pin on Kocher | Mallet | |
| Second pin on Kocher | Mallet | |
| Drill bushing | ||
| Drill | ||
| Keel punch and mallet | Remove small pins | |
| 1/8 in. drill bit | ||
| 10 mm block | Loosen IMP | remove all retractors |
| Alignment rods | ||
| Check extension gap and balance | ||
| Fresh cuts if needed | ||
| Remove block | Rake to medial skin | |
| Place fork laterally | Hold fork | |
| Flex knee to >60 degrees | Rotate fork distally | |
| Place femoral sizer | ||
| 1/8 in. drill | Large headed pins | |
| Mallet | ||
| Extend knee | Rotate fork proximal | |
| Place anterior stylus | Place rotation guide | |
| Two threaded pins | ||
| Check rotation/flexion gap | ||
| Adjust if needed | ||
| Remove stylus/rotation guide | Slaphammer | Rotate fork distally |
| Flex knee | ||
| Place 4 in 1 block | Large headed pins/mallet | |
| Trilogy screws | ||
| Double check rotation | Rotate fork proximal | |
| Saw to anterior cut | ||
| Saw to anterior chamfer | Place jack and smiley medial | |
| Saw to post MFC | ||
| Saw to post med chamfer | Move smiley laterally | remove fork |
| Saw to post LFC | ||
| Saw to post lat chamfer | Remove smiley & jack | |
| Recip saw | ||
| Power screwdriver | Slaphammer | Remove gold block |
| 3/4 in. osteotome/mallet | Kocher | Rakes anterior |
| Small rongeur | ||
| Lamina spreader | Rake | Hold lamina spreader |
| Kocher & bovie | ||
| 1/2 in. curved osteotome | ||
| Angled curette | ||
| Kocher | ||
| Flip lamina spreader | Flip rake | Hold Lamina spreader |
| Kocher & bovie | ||
| 1/2 in. curved osteotome | ||
| Angled curette | ||
| Kocher | ||
| Injection | Remove rake | Hold lamina spreader |
| Spreader | ||
| Tensor at 90 degrees | ||
| Torque wrench | ||
| 12 mm block | ||
| Remove Tensor | Loosen IMP | |
| Place 12 mm block | ||
| Check flexion stability | ||
| Tensor at full extension | ||
| Torque wrench | ||
| 10 mm block | ||
| Remove tensor | ||
| Place 10 mm block | ||
| Check stability | ||
| Adjust bone/ligaments if needed | ||
| Fork laterally | Rake medially | Hold rake and fork |
| Flex knee | Rotate fork distally | |
| Femoral finisher & mallet | 2-Headed pins | |
| Drive in pins | Place jack | |
| Reciprocating saw | Slaphammer | |
| Kocher | ||
| Femoral trial | ||
| Mallet and impactor | Remove retractors | |
| Blunt Hohmann posterior | Hold Hohmann | |
| Tibial trial and insert | Loosen IMP | Remove Hohmann |
| Reduce knee into extension | ||
| Check stability/ROM | ||
| Place in IMP and flex to 90 degrees | ||
| Sharp Hohmann to remove insert | Rake and fork | |
| Femoral slaphammer | Leg in extension | |
| Prepare Mayo for cementing | Pulse evac | Suction |
| Flex knee | ||
| Posterior blunt Hohmann | Hold Hohmann | |
| Fork laterally | Hold fork | |
| Sharp hohmann medially | Hold sharp Hohmann | |
| Remove debris | ||
| Cement to tibia | ||
| Place Tibial component | ||
| Mallet and impactor | ||
| Remove excess cement | Remove excess cement | Remove retractors |
| Fork laterally | Rake medially | Hold fork |
| Army Navy anterior | Hold Army-Navy | |
| Cement blob | ||
| Femoral component | ||
| Mallet and impactor | ||
| Remove excess cement | Remove excess cement | remove retractors |
| Sharp hohmann laterally | Hold sharp Hohmann | |
| Blunt hohmann posterior | Hold blunt Hohmann | |
| Place Tibial insert | ||
| Reduce knee | Loosen IMP | Remove retractors |
| Leg in extension on bump | Hold tibia | |
| Clean patella | ||
| Cement patellar button | Patella clamp | |
| Clean excess cement | ||
| Patellar chamfer | Pulse evac | |
| Hot saline | ||
| Check for debris | ||
| Drain | Rakes proximally | |
| No. 2 quill suture | Suture scissors |
Some companies have started to streamline instrument sets for the surgeon. For example, Stryker has a Precision set that requires staff to only open limited trials once all the measurements are done and cuts have been made. This approach eliminates the need to open and reprocess 2–3 trays of trial components.
Custom cutting blocks
Another approach is to use CT or MRI based measurements to premanufacture cutting blocks that determine implant size, amount of bone to be resected, angle of the cuts, and rotation of the cuts. These can be customized to surgeon preferences, use anatomical standards for alignment and rotation, and attach to the bone using the shape of the patient’s own bone. Several studies have validated the accuracy and efficiency provided by this approach. For example, Spencer et al. [7] showed that deviation from mechanical axis was on average only 1.4 degrees. Nunley et al. [8•] showed that patient specific guides targeted at the mechanical axis were at least as accurate at traditional instruments.
These guides are intended for single use, and so they can be disposed of after case completion, saving significant time and effort in turnover.
However, there often is an additional cost for manufacture of the guides, and so it is important to determine whether the cost savings from the ease of reprocessing is not negated by the added cost [5••] of the custom cutting guides. Also, there is the added cost of the imaging scan, and the 3 week or so time lag from scan to manufacture of the guide.
One word of caution: customized guides cannot solely correct alignment if ligament releases are needed. Similarly, they cannot balance flexion and extension gaps in all cases. Because this approach relies on anatomical landmarks to set rotation, the knee can be imbalanced, necessitating further ligament balancing or repeat bone cutting. Also, if the imaging scan is inaccurate, the guides may not fit properly or the recommended components may be incorrectly sized. So, this approach does not absolve the surgeon of the responsibility of being a surgeon. Lastly, trial implants are still required for final assessment of the reconstruction prior to implantation. Nevertheless, for most surgeons in the majority of cases, this approach has the potential to speed up surgery.
Custom implants and tools
A recently introduced innovation, introduced by a new company called ConforMIS, Inc, (Burlington, MA, USA) has been the use of customized cutting guides and implants. In this approach, a preoperative CT scan is used to create a three dimensional model of the distal femur and proximal tibia, while also calculating the corrected mechanical axis. From these scans, customized cutting guides are prepared, as are patient-specific implants. The guides are similar to those discussed in the section above, although they do require gap balancing prior to making the final bone cuts. The implants are manufactured to recreate the individual patient anatomy, providing an anatomical reconstruction based upon the bone shape and orientation, with the added benefit of fine tuning the ligament tension and balance. The sagittal J curves of the femur are reproduced in the femoral implant and in the articular surfaces. The shape and size of the femoral and tibial components are matched to the predicted bone cuts. Trial implants, which replicate the actual implant are also provided, including multiple poly articular insert thicknesses. Upon conclusion of the case, all guides and trials can be disposed. Again, this can save significant time and effort in turnover. Currently, the cost of the disposable guides and trials are factored into the cost of the implant. In my hospital, both off the shelf and customized implants are competitively priced. Also, there is the added cost of the imaging scan, which may range from $400–$800, but can be an added revenue source for the hospital.
Early data are not yet available, although studies are underway assessing the biomechanics of custom implants, clinical outcomes and CT alignment. The primary pitfalls of using customized implants, besides added cost, are the time to manufacture the implants and the risk of wasted implants. Time to manufacture is typically about 6 weeks or so from the time the imaging scan is completed. This time frame can dissuade patients, who are in pain, and otherwise might be able to receive their surgery within a few weeks, depending upon surgeon availability. Similarly, surgeons may fear losing patients who are compelled to wait 6 weeks or more. Another theoretical concern is that at the time of surgery, if the reconstruction cannot be successfully completed, the custom implants will be necessarily wasted in favor of off the shelf implants.
Conclusions
Optimization of the surgical episode of total knee replacement has been historically inefficient. Recent efforts have focused upon waste reduction before, during, and after the surgery. Besides the obvious benefit of cost and resource conservation, one added benefit may be improved accuracy and possibly outcomes. Further research will be necessary to confirm these possibilities.
Acknowledgments
Disclosure
RK Sinha: board membership with ConforMIS, Inc, consultant to Zimmer and Robodoc, has grants from ConforMIS, Inc, Zimmer, and Superstat, receives royalties from ConforMIS and Zimmer, develops educational presentations for Angiotech, and has stock options with ConforMIS, Inc.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
- 1.Jaffer AK, Barsoum WK, Krebs V, Hurbanek JG, Morra N, Brotman DJ. Duration of anesthesia and venous thromboembolism after hip and knee arthroplasty. Mayo Clin Proc. 2005;80:732–738. doi: 10.1016/S0025-6196(11)61526-7. [DOI] [PubMed] [Google Scholar]
- 2.Willis-Owen CA, Konyves A, Martin DK. Factors affecting the incidence of infection in hip and knee replacement: an analysis of 5277 cases. J Bone Joint Surg Br. 2010;92:1128–1133. doi: 10.1302/0301-620X.92B8.24333. [DOI] [PubMed] [Google Scholar]
- 3.Prasad N, Padmanabhan V, Mullaji A. Blood loss in total knee arthroplasty: an analysis of risk factors. Int Orthop. 2007;31:39–44. doi: 10.1007/s00264-006-0096-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ong KL, Lau E, Manley M, Kurtz SM. Effect of procedure duration on total hip arthroplasty and total knee arthroplasty survivorship in the United States Medicare population. J Arthroplasty. 2008;23(6 Suppl 1):127–132. doi: 10.1016/j.arth.2008.04.022. [DOI] [PubMed] [Google Scholar]
- 5.Watters TS, Mather RC, III, Browne JA, Berend KR, Lombardi AV, Jr, Bolognesi MP. Analysis of procedure-related costs and proposed benefits of using patient-specific approach in total knee arthroplasty. J Surg Orthop Adv. 2011;20(6):112–116. [PubMed] [Google Scholar]
- 6.JFK Memorial Hospital Working Group internal data.
- 7.Spencer BA, Mont MA, McGrath MS, Boyd B, Mitrick MF. Initial experience with custom-fit total knee replacement: intra-operative events and long-leg coronal alignment. Int Orthop. 2009;33:1571–1575. doi: 10.1007/s00264-008-0693-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nunley RM, Ellison BS, Zhu J, Ruh EL, Howell SM, Barrack RL. Do patient-specific guides improve coronal alignment in total knee arthroplasty? Clin Orthop Relat Res. 2012;470:895–902. doi: 10.1007/s11999-011-2222-2. [DOI] [PMC free article] [PubMed] [Google Scholar]


