Abstract
Background:
A defined role for reactive oxygen species (ROS) in the tissue destruction that characterizes periodontitis has been described. Protein carbonyl (PC) is the most widely used biomarker for oxidative damage to proteins, and reflects cellular damage induced by multiple forms of ROS. The purpose of this study is to determine the presence of PC in gingival crevicular fluid (GCF) in healthy, gingivitis, and chronic periodontitis (CP) subjects and to find an association, if any.
Materials and Methods:
A total number of 75 subjects (38 males and 37 females) were selected based on their clinical parameters into three groups: Group 1 (25 healthy subjects), Group 2 (25 gingivitis subjects), and Group 3 (25 CP subjects). GCF samples were collected to estimate the levels of PC.
Results:
The PC concentration in GCF was highest in subjects with CP as compared to gingivitis and healthy subjects and a significant association was observed between GCF PC levels and all periodontal parameters.
Conclusion:
There was an increase in PC levels in GCF as the disease process progressed from healthy to gingivitis and CP, suggesting a role for increased oxidative stress in CP.
Keywords: Chronic periodontitis, gingival crevicular fluid, oxidative stress, protein carbonyl
Introduction
Periodontal disease is an inflammatory disorder in which tissue damage occurs through the complex interactions between periodontal pathogens and components of the host defense mechanism.[1,2] Diagnosis of periodontal disease has been primarily based upon clinical and radiographic measures of periodontal tissue destruction. While these parameters provide a measure of past destruction, they are of limited use in diagnosis.[3,4,5] The periodontitis phenotype is characterized by hyperinflammation involving excess oxygen radical release by neutrophilic polymorphonuclear leucocytes[6] and excess release of proteolytic enzymes such as neutrophil elastase.[7] Evidence is emerging to implicate oxidative stress in the pathogenesis of periodontitis.
Oxidative stress is a state of altered physiological equilibrium within a cell or tissue/organ, defined as “a condition arising when there is a serious imbalance between the levels of free radicals in a cell and its antioxidant defences in favour of the former.”[8] It is estimated that 1-3 billion reactive oxygen species (ROS) are generated/cell/day, and given this, the importance of the body's antioxidant defense systems to the maintenance of health becomes clear.[9]
ROS is a collective term which includes oxygen-derived free radicals (ODFR), such as the superoxide radical, hydroxyl radical, and nitric oxide radical species, and non-radical derivatives of oxygen, such as hydrogen peroxide and hypochlorous acid. The presence of one or more unpaired electrons in the outer orbitals of ODFR makes such species, especially the hydroxyl species, extremely reactive in nature.[10]
Protein carbonylation is a type of protein oxidation that can be promoted by ROS. ROS can react directly with the protein or they can react with molecules such as sugars and lipids, generating products (reactive carbonyl species) which then react with the protein. Direct oxidation of proteins by ROS yields highly reactive carbonyl derivatives resulting either from oxidation of the side chains of lysine, arginine, proline, and threonine residues – particularly via metal-catalyzed oxidation[11] – or from the cleavage of peptide bonds by the α-amidation pathway or by oxidation of glutamyl residues. Glutamic semialdehyde, a product of oxidation of arginine and proline, and aminoadipic semialdehyde, a product of oxidation of lysine, are the main carbonyl products of metal-catalyzed oxidation of proteins, this reaction being a major route leading to the generation of protein carbonyls (PCs) in biological samples.[12] PC is the most widely used biomarker for oxidative damage to proteins, and reflects cellular damage induced by multiple forms of ROS.[13,14,15]
Individuals manifesting the most advanced periodontal disease exhibit the high levels of salivary PCs[16,17] and also gingival crevicular fluid (GCF) and serum PC levels are significantly higher in chronic periodontitis (CP) group.[18]
Thus, in view of the aforementioned findings, this clinico-biochemical study was undertaken to estimate the PC levels in GCF in clinically healthy periodontium, gingivitis, and CP subjects and to assess the role of PC as a marker of oxidative stress.
Materials and Methods
The study population consisted of 75 age- and gender-balanced subjects (37 females and 38 males; age range: 25-45 years) attending the outpatient section of the Department of Periodontics, Government Dental College and Research Institute, Bangalore, Karnataka, India. Written informed consent was obtained from those who agreed to participate voluntarily. Ethical clearance was obtained from the institution's Ethical Committee and Review Board. Subjects with aggressive periodontitis, hypertension, a smoking habit, gross oral pathology, heart diseases, rheumatoid arthritis, tumors, or any other systemic disease that can alter the course of periodontal disease, or those who had any course of medication affecting periodontal status or had received periodontal therapy in the preceding 6 months were excluded from the study. Each subject underwent a full mouth periodontal probing and charting, along with periapical radiographs using the long-cone technique. Radiographic bone loss was recorded dichotomously (presence or absence) to differentiate CP patients from other groups. Furthermore, no delineation was attempted within the CP group based on the extent of alveolar bone loss. Based on the gingival index (GI),[19] probing pocket depth (PPD), clinical attachment loss (CAL), and radiographic evidence of bone loss, subjects were categorized into three groups. Group 1 (healthy) consisted of 25 subjects with clinically healthy periodontium, with GI = 0, PPD ≤ 3 mm, and CAL = 0, with no evidence of bone loss on radiograph. Group 2 (gingivitis) consisted of 25 subjects who showed clinical signs of gingival inflammation, GI > 1, PPD ≤ 3 mm, and had no attachment loss or radiographic bone loss. Group 3 (CP) consisted of 25 subjects who had signs of clinical inflammation, GI > 1, CAL > 1, in 30% of sites with radiographic evidence of bone loss and PPD ≥ 5 mm 4 mm in 30% of sites.
Site selection and GCF fluid collection
All clinical examinations, radiographs, group allocations, and sampling site selections were performed by one examiner (ARP), and the samples were collected on the subsequent day by a second examiner (PB). This was to prevent contamination of GCF with blood associated with the probing of inflamed sites. A calibrated examiner performed all the clinical assessments using a University of North Carolina-15 periodontal probe, to ensure adequate intra-examiner reproducibility. In patients with CP, the site showing the greatest CAL and signs of inflammation, along with radiographic confirmation of bone loss, was selected for sampling. After making the subjects sit comfortably in an upright position on the dental chair, the selected test site was air-dried and isolated with cotton rolls. Without touching the marginal gingiva, supragingival plaque was removed to avoid contamination of the paper strips and GCF was collected with Periopaper (Ora Flow Inc., Amityville, NY, USA) using the intra-crevicular method “superficially” developed by Loe and Holm-Pederson.[20] The absorbed GCF volume of each strip was determined by electronic impedance (Periotron 8000; ProFlow Inc., Amityville, NY, USA). The same method was used to obtain GCF samples from the control group.
Two Periopaper strips that absorbed GCF for each subject were pooled and the Periopaper strips were placed in a sterile Eppendorf vial containing 400 μl of phosphate buffer saline and kept at −70°C until analyzed. Periopaper strips, contaminated with blood and saliva, were excluded or discarded. Periodontal treatment, scaling for gingivitis patients and scaling and root planing for periodontitis patients, was performed at the same appointment after GCF collection.
Blood collection
Two milliliters of blood were collected from the antecubital fossa by venipuncture using a 20-gauge needle with 2-ml syringe and immediately transferred to the laboratory. The blood sample was allowed to clot at room temperature and after 1 h, serum was separated from blood by centrifuging at 3000 g for 5 min. The serum was immediately transferred to a plastic vial and stored at −70°C until the time of assay.
PC analysis
The samples were analyzed using PC Assay Kit (Cayman Chemical Company, Ann Arbor, MI, USA). The GCF sample tubes were homogenized for 30 s and centrifuged for 5 min at 1,500 g to elute. Assays were carried out according to the manufacturer's recommendations.
Statistical analysis
The data were analyzed using statistical software (SPSS version 10.5, SPSS, Chicago, USA). Power calculations were performed before the study was initiated. To achieve 90% power and detect mean differences of the clinical parameters between groups, 25 sites per group were required. Analysis of variance (ANOVA) and Bonferroni's test were carried out for a comparison of GCF PC levels between the groups. Using Pearson's correlation coefficient, the relationship between GCF PC levels and the clinical parameters were analyzed. P < 0.05 were considered statistically significant.
Results
The descriptive statistics along with the mean ± SD of age, sex, PPD, and CAL of the sites of GCF sample collection (of all groups) are tabulated in Table 1. All the samples in each group tested positive for PC. Table 2 shows that mean PC concentration in GCF was highest for Group 3 followed by Group 2 and least in Group 1. When Groups 1 and 2, 1 and 3, and 2 and 3 were compared, the differences in the mean GCF PC concentrations were statistically significant [Table 2]. Pearson correlation coefficients between the clinical parameters and PC levels are tabulated in Table 3. The correlation between PC levels and PPD was found to be significantly strong in all the groups. However, the correlation between PC levels and GI was found to be significant only in Group 2 and weak positive (not statistically significant at P > 0.05) correlation in Group 3. The correlation between PC levels and CAL was found to be positive and statistically significant (P > 0.05) in Group 3.
Table 1.
Descriptive statistics of study population (mean±SD)
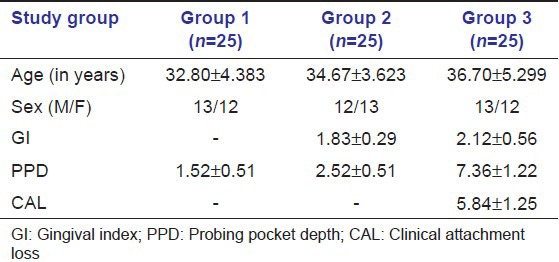
Table 2.
Results of analysis of variance and pair-wise comparison using Bonferroni's test of the mean gingival crevicular fluid protein carbonyl levels between three groups
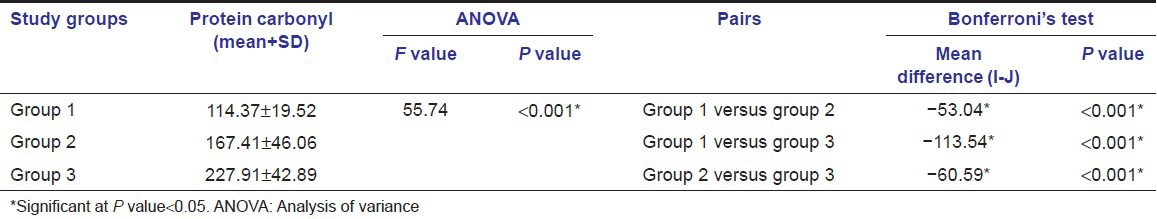
Table 3.
Relationship of gingival crevicular fluid protein carbonyl levels to clinical parameters

Discussion
There now exists convincing evidence that oxidative stress and ROS play an important role in the etiology and/or progression of a number of human diseases.[21] Actually, the medical significance of oxidative stress has become increasingly recognized to the point that it is now considered to be a component of virtually every disease process. More recently, evidence has also emerged for a crucial role of ROS in periodontal tissue destruction.[22] ROS generation secondary to polymorphonuclear leukocyte infiltration, a key host defense mechanism against bacterial invasion,[23,24,25,26,27] has been implicated in “bystander” destruction of periodontal tissue. To investigate the role of oxidative stress and ROS in the pathogenesis and/or progression of human diseases, the use of appropriate biomarkers is necessary. In determining whether to use lipids, DNA, or proteins as a marker of oxidative stress, the nature of the ROS will play a significant role.
Many different types of protein oxidative modification can be induced directly by ROS or indirectly by reactions of secondary by-products of oxidative stress.[28] Protein carbonylation is a type of protein oxidation that can be promoted by ROS and protein carbonylation is the most widely used biomarker for oxidative damage to proteins, and reflects cellular damage induced by multiple forms of ROS.[13,14,15] PC has a major advantage over lipid peroxidation products as markers of oxidative stress: Oxidized proteins are generally more stable. PC forms early and circulate in the blood for longer periods (its elevation in serum is stable for at least 4h),[29] compared with other parameters of oxidative stress, such as glutathione disulfide and malondialdehyde. The formation of PC seems to be a common phenomenon during oxidation, and its quantification can be used to measure the extent of oxidative modification.
Our study comprised of three groups (healthy, gingivitis, and CP); these groups helped us to evaluate the role of PC in periodontal health and disease. The influence of age and gender on the PC concentration was minimized by including an equal number of males and females in each group and selecting the subjects within the specified age group (25-45 years). In this study, significantly higher levels of serum and GCF PC in CP than gingivitis and periodontal healthy subjects were found and it may be a sign of significant increase in local periodontal oxidative damage in CP.
The results of this study indicated that concentration of PC in GCF increased progressively from healthy to periodontitis sites, whereas in periodontitis sites (Group 3), the mean concentration of PC was higher than the concentrations obtained in Groups 1 and 2, suggesting that oxidative stress increases as the periodontal disease advances from health to gingivitis and is much higher in CP. The results of this study are also in accordance with those of Baltacioglu et al., which reported an increase in PC levels in both serum and GCF of patients with CP as compared to healthy controls. Also, the concentration of PC in healthy controls and CP patients in both serum and GCF in their study was of similar range as found in this study.[18]
In this study, GCF was collected using the absorbent filter paper strips. The advantages of the technique are that it is quick and easy to use, can be applied to individual sites and possibly, is the least traumatic when correctly used. The electronic measuring device, the Periotron, allowed accurate determination of the GCF volume and subsequent laboratory investigation of the sample composition. GCF samples were pooled per subject to ensure sufficient assay sensitivity and the patient was used as the unit of analysis. Site-specific differences were therefore not analyzed. The PC levels found in healthy subjects in absence of diseased sites may be because many of our diseased sites were probably stable, and some healthy sites may have been undergoing active attachment loss.
Elevated levels of PC are generally a sign not only of oxidative stress but also of disease-derived protein dysfunction and oxidative damage contributing to complicate the pathophysiology of the disease.[14,30] In addition to the well-established roles of protein carbonylation in oxidative stress, this oxidation process may also play roles in cell signal transduction as described by Suzuki et al.[31,32] This suggests that cellular regulatory mechanisms of protein carbonylation may be complex, which might include means to promote and eliminate PC.
These pathophysiological roles of protein carbonylation in oxidation stress and oxidant signaling suggest that compounds, which regulate carbonyl content, may have clinical value. Identification of carbonylated-proteins may provide new diagnostic biomarkers for oxidative damage which occurs in periodontitis.
Conclusion
Increase in GCF concentration of PC confirms the critical role of PC in periodontal destruction and shows that oxidative stress increases as the periodontal disease advances from health to CP. Within the limitations of this study, it can be postulated that increased concentrations of PC can be detected in GCF in healthy, gingivitis, and CP subjects. However, further longitudinal studies are needed to validate PC as a “marker of oxidative stress” in periodontal disease progression.
Footnotes
Source of Support: Nil
Conflict of Interest: None declared
References
- 1.Goodson JM, Tanner AC, Haffajee AD, Sornberger GC, Socransky SS. Patterns of progression and regression of advanced destructive periodontal disease. J Clin Periodontol. 1982;9:472–81. doi: 10.1111/j.1600-051x.1982.tb02108.x. [DOI] [PubMed] [Google Scholar]
- 2.The Research, Science and Therapy Committee of The American Academy of Periodontology. The pathogenesis of periodontal diseases. J Periodontol. 1999;70:457–70. [Google Scholar]
- 3.Badersten A, Nilvéus R, Egelberg J. Effect of nonsurgical periodontal therapy. VII. Bleeding, suppuration and probing depth in sites with probing attachment loss. J Clin Periodontol. 1985;12:432–40. doi: 10.1111/j.1600-051x.1985.tb01379.x. [DOI] [PubMed] [Google Scholar]
- 4.Greenstein G. The role of bleeding upon probing in the diagnosis of periodontal disease. A literature review. J Periodontol. 1984;55:684–8. doi: 10.1902/jop.1984.55.12.684. [DOI] [PubMed] [Google Scholar]
- 5.Haffajee AD, Socransky SS, Goodson JM. Clinical parameters as predictors of destructive periodontal disease activity. J Clin Periodontol. 1983;10:257–65. doi: 10.1111/j.1600-051x.1983.tb01274.x. [DOI] [PubMed] [Google Scholar]
- 6.Gustafsson A, Asman B. Increased release of free oxygen radicals from peripheral neutrophils in adult periodontitis after Fc delta-receptor stimulation. J Clin Periodontol. 1996;23:38–44. doi: 10.1111/j.1600-051x.1996.tb00502.x. [DOI] [PubMed] [Google Scholar]
- 7.Figueredo CM, Gustafsson A, Asman B, Bergström K. Increased release of elastase from in vitro activated peripheral neutrophils in patients with adult periodontitis. J Clin Periodontol. 1999;26:206–11. doi: 10.1034/j.1600-051x.1999.260402.x. [DOI] [PubMed] [Google Scholar]
- 8.Halliwell B, Gutteridge JM. 2nd ed. Oxford, UK: Oxford University Press; 1989. Free Radicals in Biology and Medicine. [Google Scholar]
- 9.Ames BN, Shigenaga MK, Hagen TM. Oxidants, antioxidants, and the degenerative diseases of aging. Proc Natl Acad Sci U S A. 1993;90:7915–22. doi: 10.1073/pnas.90.17.7915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Halliwell B, Gutteridge JM, Cross CE. Free radicals, antioxidants, and human disease: Where are we now? J Lab Clin Med. 1992;119:598–620. [PubMed] [Google Scholar]
- 11.Stadtman ER. Metal ion-catalyzed oxidation of proteins: Biochemical mechanism and biological consequences. Free Radic Biol Med. 1990;9:315–25. doi: 10.1016/0891-5849(90)90006-5. [DOI] [PubMed] [Google Scholar]
- 12.Stadtman ER, Berlett BS. Fenton chemistry. Amino acid oxidation. J Biol Chem. 1991;266:17201–11. [PubMed] [Google Scholar]
- 13.Dalle-Donne I, Scaloni A, Giustarini D, Cavarra E, Tell G, Lungarella G, et al. Proteins as biomarkers of oxidative/nitrosative stress in diseases: The contribution of redox proteomics. Mass Spectrom Rev. 2005;24:55–99. doi: 10.1002/mas.20006. [DOI] [PubMed] [Google Scholar]
- 14.Dalle-Donne I, Giustarini D, Colombo R, Rossi R, Milzani A. Protein carbonylation in human diseases. Trends Mol Med. 2003;9:169–76. doi: 10.1016/s1471-4914(03)00031-5. [DOI] [PubMed] [Google Scholar]
- 15.Dalle-Donne I, Rossi R, Colombo R, Giustarini D, Milzani A. Biomarkers of oxidative damage in human disease. Clin Chem. 2006;52:601–23. doi: 10.1373/clinchem.2005.061408. [DOI] [PubMed] [Google Scholar]
- 16.Su H, Gornitsky M, Velly AM, Yu H, Benarroch M, Schipper HM. Salivary DNA, lipid, and protein oxidation in nonsmokers with periodontal disease. Free Radic Biol Med. 2009;46:914–21. doi: 10.1016/j.freeradbiomed.2009.01.008. [DOI] [PubMed] [Google Scholar]
- 17.Sculley DV, Langley-Evans SC. Periodontal disease is associated with lower antioxidant capacity in whole saliva and evidence of increased protein oxidation. Clin Sci (Lond) 2003;105:167–72. doi: 10.1042/CS20030031. [DOI] [PubMed] [Google Scholar]
- 18.Baltacioğlu E, Akalin FA, Alver A, Değer O, Karabulut E. Protein carbonyl levels in serum and gingival crevicular fluid in patients with chronic periodontitis. Arch Oral Biol. 2008;53:716–22. doi: 10.1016/j.archoralbio.2008.02.002. [DOI] [PubMed] [Google Scholar]
- 19.Loe H, Silness J. Periodontal disease in pregnancy. I. prevalence and severity. Acta Odontol Scand. 1963;21:533–51. doi: 10.3109/00016356309011240. [DOI] [PubMed] [Google Scholar]
- 20.Loe H, Holm-Pedersen P. Absence and presence of fluid from normal and inflamed gingivae. Periodontics. 1965;149:171–7. [PubMed] [Google Scholar]
- 21.Halliwell B, Gutteridge J. NY: Oxford University Press; 1989. Free Radicals in Biology and Medicine. [Google Scholar]
- 22.Chapple IL, Matthews JB. The role of reactive oxygen and antioxidant species in periodontal tissue destruction. Periodontol 2000. 2007;43:160–232. doi: 10.1111/j.1600-0757.2006.00178.x. [DOI] [PubMed] [Google Scholar]
- 23.Sculley DV, Langley-Evans SC. Salivary antioxidants and periodontal disease status. Proc Nutr Soc. 2002;61:137–43. doi: 10.1079/pns2001141. [DOI] [PubMed] [Google Scholar]
- 24.Chapple IL. Reactive oxygen species and antioxidants in inflammatory diseases. J Clin Periodontol. 1997;24:287–96. doi: 10.1111/j.1600-051x.1997.tb00760.x. [DOI] [PubMed] [Google Scholar]
- 25.Canakçi CF, Tatar A, Canakçi V, Cicek Y, Oztas S, Orbak R. New evidence of premature oxidative DNA damage: Mitochondrial DNA deletion in gingival tissue of patients with periodontitis. J Periodontol. 2006;77:1894–900. doi: 10.1902/jop.2006.060108. [DOI] [PubMed] [Google Scholar]
- 26.Król K. Reactive oxygen species and antioxidant mechanisms in the pathogenesis of periodontitis. Ann Acad Med Stetin. 2004;50:135–48. [PubMed] [Google Scholar]
- 27.Waddington RJ, Moseley R, Embery G. Reactive oxygen species: A potential role in the pathogenesis of periodontal diseases. Oral Dis. 2000;6:138–51. doi: 10.1111/j.1601-0825.2000.tb00325.x. [DOI] [PubMed] [Google Scholar]
- 28.Berlett BS, Stadtman ER. Protein oxidation in aging, disease, and oxidative stress. J Biol Chem. 1997;272:20313–6. doi: 10.1074/jbc.272.33.20313. [DOI] [PubMed] [Google Scholar]
- 29.Pantke U, Volk T, Schmutzler M, Kox WJ, Sitte N, Grune T. Oxidized proteins as a marker of oxidative stress during coronary heart surgery. Free Radic Biol Med. 1999;27:1080–6. doi: 10.1016/s0891-5849(99)00144-6. [DOI] [PubMed] [Google Scholar]
- 30.Trombetta D, Gangemi S, Saija A, Minciullo PL, Cimino F, Cristani M, et al. Increased protein carbonyl groups in the serum of patients affected by thalassemia major. Ann Hematol. 2006;85:520–2. doi: 10.1007/s00277-006-0115-3. [DOI] [PubMed] [Google Scholar]
- 31.Wong CM, Cheema AK, Zhang L, Suzuki YJ. Protein carbonylation as a novel mechanism in redox signaling. Circ Res. 2008;102:310–8. doi: 10.1161/CIRCRESAHA.107.159814. [DOI] [PubMed] [Google Scholar]
- 32.Wong CM, Marcocci L, Liu L, Suzuki YJ. Cell signaling by protein carbonylation and decarbonylation. Antioxid Redox Signal. 2010;12:393–404. doi: 10.1089/ars.2009.2805. [DOI] [PMC free article] [PubMed] [Google Scholar]


