Abstract
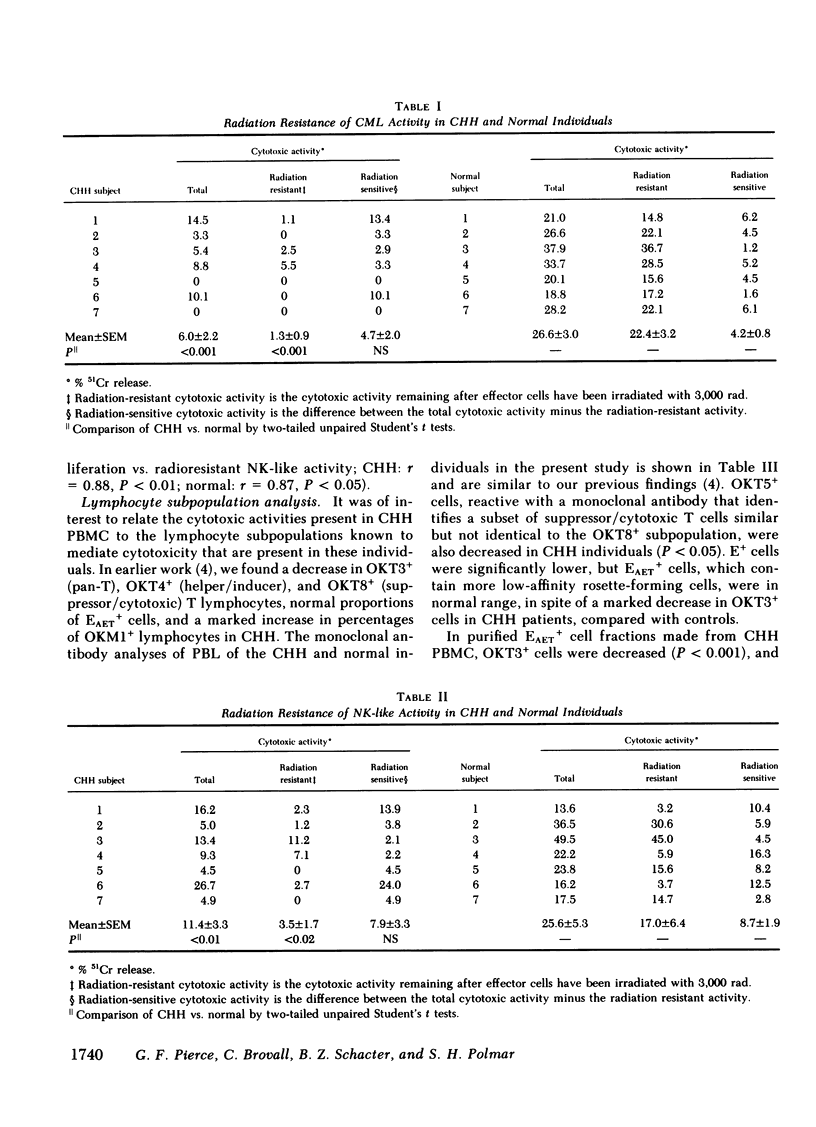
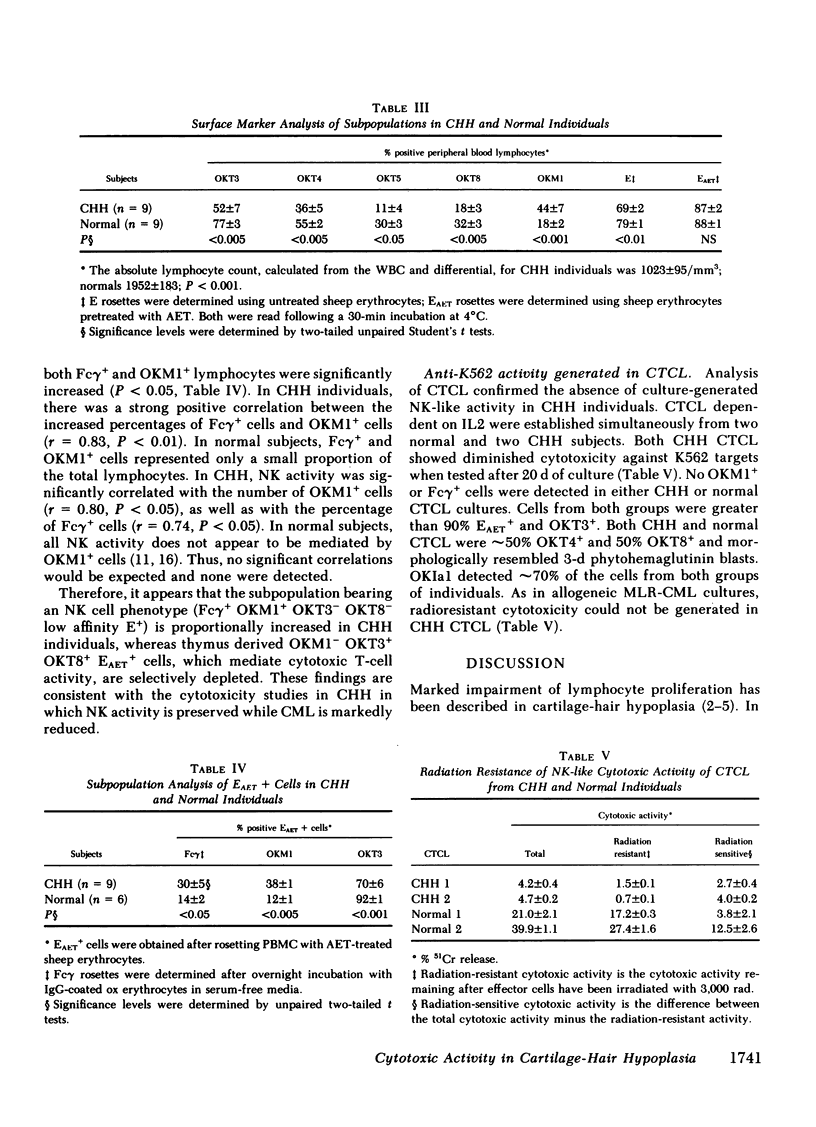
Recent studies of cartilage-hair hypoplasia (CHH), a form of short-limbed dwarfism, have shown that all affected individuals have a cellular proliferation defect that results in a cellular immunodeficiency. However, only a minority of CHH individuals suffer from severe, life-threatening infections. For this reason, relevant immune defense mechanisms that may be responsible for maintaining intact host defenses in the majority of CHH individuals were studied. Spontaneous and allogeneic culture-induced (mixed lymphocyte response-MLR) specific and nonspecific (NK-like) cytotoxic mechanisms were analyzed and correlated with lymphocyte subpopulations present in CHH and normal individuals. Spontaneous natural-killer (NK) activity was present at or above normal levels, but culture-induced specific cytotoxicity and NK-like cytotoxicity as well as NK-like activity by T cell lines were significantly reduced in CHH individuals. The generation of radiation-resistant cytotoxicity, which normally occurs during allogeneic MLR, was markedly diminished in CHH, and was correlated with the decreased proliferation observed in CHH cultures. Preservation of spontaneous NK activity and loss of all forms of culture-induced cytotoxicity was associated with an increase in the proportion of lymphocytes bearing a thymic independent NK phenotype (OKM1+ OKT3- Fc gamma + low-affinity E+), and a significant decrease in thymic derived OKT3+ cytolytic T cell sub-populations in CHH individuals. Therefore, an intact cellular cytotoxic effector mechanism has been identified in CHH (i.e., NK activity). Natural cytotoxicity may be of importance in maintaining host resistance to viral infections despite diminished thymic-derived effector mechanisms in cartilage-hair hypoplasia.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abo T., Balch C. M. A differentiation antigen of human NK and K cells identified by a monoclonal antibody (HNK-1). J Immunol. 1981 Sep;127(3):1024–1029. [PubMed] [Google Scholar]
- Biddison W. E., Sharrow S. O., Shearer G. M. T cell subpopulations required for the human cytotoxic T lymphocyte response to influenza virus: evidence for T cell help. J Immunol. 1981 Aug;127(2):487–491. [PubMed] [Google Scholar]
- Bolhuis R. L., Schellekens H. Induction of natural killer cell activity and allocytotoxicity in human peripheral blood lymphocytes after mixed lymphocyte culture. Scand J Immunol. 1981;13(5):401–412. doi: 10.1111/j.1365-3083.1981.tb00151.x. [DOI] [PubMed] [Google Scholar]
- Brovall C., Schacter B. Radiation sensitivity of human natural killer cell activity: control by X-linked genes. J Immunol. 1981 Jun;126(6):2236–2239. [PubMed] [Google Scholar]
- Fast L. D., Hansen J. A., Newman W. Evidence for T cell nature and heterogeneity within natural killer (NK) and antibody-dependent cellular cytotoxicity (ADCC) effectors: a comparison with cytolytic T lymphocytes (CTL). J Immunol. 1981 Aug;127(2):448–452. [PubMed] [Google Scholar]
- Ferrarini M., Cadoni A., Franzi A. T., Ghigliotti C., Leprini A., Zicca A., Grossi C. E. Ultrastructure and cytochemistry of human peripheral blood lymphocytes. Similarities between the cells of the third population and TG lymphocytes. Eur J Immunol. 1980 Jul;10(7):562–570. doi: 10.1002/eji.1830100714. [DOI] [PubMed] [Google Scholar]
- Herberman R. B., Ortaldo J. R. Natural killer cells: their roles in defenses against disease. Science. 1981 Oct 2;214(4516):24–30. doi: 10.1126/science.7025208. [DOI] [PubMed] [Google Scholar]
- Kay H. D., Horwitz D. A. Evidence by reactivity with hybridoma antibodies for a probable myeloid origin of peripheral blood cells active in natural cytotoxicity and antibody-dependent cell-mediated cytotoxicity. J Clin Invest. 1980 Oct;66(4):847–851. doi: 10.1172/JCI109923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kung P. C., Goldstein G. Functional and developmental compartments of human T lymphocytes. Vox Sang. 1980 Sep;39(3):121–127. doi: 10.1111/j.1423-0410.1980.tb01846.x. [DOI] [PubMed] [Google Scholar]
- López-Botet M., Silva A., Rodríguez J., de Landazuri M. O. Generation of T cell blasts with NK-like activity in human MLC: cellular precursors, IL 2 responsiveness, and phenotype expression. J Immunol. 1982 Sep;129(3):1109–1115. [PubMed] [Google Scholar]
- Moretta L., Mingari M. C., Sekaly P. R., Moretta A., Chapuis B., Cerottini J. C. Surface markers of cloned human T cells with various cytolytic activities. J Exp Med. 1981 Aug 1;154(2):569–574. doi: 10.1084/jem.154.2.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ortaldo J. R., Bonnard G. D., Kind P. D., Herberman R. B. Cytotoxicity by cultured human lymphocytes: characteristics of effector cells and specificity of cytotoxicity. J Immunol. 1979 Apr;122(4):1489–1494. [PubMed] [Google Scholar]
- Ortaldo J. R., Sharrow S. O., Timonen T., Herberman R. B. Determination of surface antigens on highly purified human NK cells by flow cytometry with monoclonal antibodies. J Immunol. 1981 Dec;127(6):2401–2409. [PubMed] [Google Scholar]
- Pawelec G. P., Hadam M. R., Ziegler A., Lohmeyer J., Rehbein A., Kumbier I., Wernet P. Long-term culture, cloning, and surface markers of mixed leukocyte culture-derived human T lymphocytes with natural killer-like cytotoxicity. J Immunol. 1982 Apr;128(4):1892–1896. [PubMed] [Google Scholar]
- Perrin L. H., Tishon A., Oldstone M. B. Immunologic injury in measles virus infection. III. Presence and characterization of human cytotoxic lymphocytes. J Immunol. 1977 Jan;118(1):282–290. [PubMed] [Google Scholar]
- Perrin L. H., Zinkernagel R. M., Oldstone M. B. Immune response in humans after vaccination with vaccinia virus: generation of a virus-specific cytotoxic activity by human peripheral lymphocytes. J Exp Med. 1977 Oct 1;146(4):949–969. doi: 10.1084/jem.146.4.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierce G. F., Polmar S. H. Lymphocyte dysfunction in cartilage hair hypoplasia. II. Evidence for a cell cycle specific defect in T cell growth. Clin Exp Immunol. 1982 Dec;50(3):621–628. [PMC free article] [PubMed] [Google Scholar]
- Pierce G. F., Polmar S. H. Lymphocyte dysfunction in cartilage-hair hypoplasia: evidence for an intrinsic defect in cellular proliferation. J Immunol. 1982 Aug;129(2):570–575. [PubMed] [Google Scholar]
- Quinnan G. V., Jr, Kirmani N., Rook A. H., Manischewitz J. F., Jackson L., Moreschi G., Santos G. W., Saral R., Burns W. H. Cytotoxic t cells in cytomegalovirus infection: HLA-restricted T-lymphocyte and non-T-lymphocyte cytotoxic responses correlate with recovery from cytomegalovirus infection in bone-marrow-transplant recipients. N Engl J Med. 1982 Jul 1;307(1):7–13. doi: 10.1056/NEJM198207013070102. [DOI] [PubMed] [Google Scholar]
- Reinherz E. L., Schlossman S. F. The differentiation and function of human T lymphocytes. Cell. 1980 Apr;19(4):821–827. doi: 10.1016/0092-8674(80)90072-0. [DOI] [PubMed] [Google Scholar]
- Roder J. C., Haliotis T., Klein M., Korec S., Jett J. R., Ortaldo J., Heberman R. B., Katz P., Fauci A. S. A new immunodeficiency disorder in humans involving NK cells. Nature. 1980 Apr 10;284(5756):553–555. doi: 10.1038/284553a0. [DOI] [PubMed] [Google Scholar]
- Seeley J. K., Masucci G., Poros A., Klein E., Golub S. H. Studies on cytotoxicity generated in human mixed lymphocyte cultures. II. Anti-K562 effectors are distinct from allospecific CTL and can be generated from NK-depleted T cells. J Immunol. 1979 Sep;123(3):1303–1311. [PubMed] [Google Scholar]
- Sullivan J. L., Byron K. S., Brewster F. E., Purtilo D. T. Deficient natural killer cell activity in x-linked lymphoproliferative syndrome. Science. 1980 Oct 31;210(4469):543–545. doi: 10.1126/science.6158759. [DOI] [PubMed] [Google Scholar]
- Timonen T., Ortaldo J. R., Herberman R. B. Characteristics of human large granular lymphocytes and relationship to natural killer and K cells. J Exp Med. 1981 Mar 1;153(3):569–582. doi: 10.1084/jem.153.3.569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trojak J. E., Polmar S. H., Winkelstein J. A., Hsu S., Francomano C., Pierce G. F., Scillian J. J., Gale A. N., McKusick V. A. Immunologic studies of cartilage-hair hypoplasia in the Amish. Johns Hopkins Med J. 1981 Apr;148(4):157–164. [PubMed] [Google Scholar]
- Virolainen M., Savilahti E., Kaitila I., Perheentupa J. Cellular and humoral immmunity in cartilage-hair hypoplasia. Pediatr Res. 1978 Oct;12(10):961–966. doi: 10.1203/00006450-197810000-00002. [DOI] [PubMed] [Google Scholar]
- Wagner H., Hardt C., Stockinger H., Pfizenmaier K., Bartlett R., Röllinghoff M. Impact of thymus on the generation of immunocompetence and diversity of antigen-specific MHC-restricted cytotoxic T-lymphocyte precursors. Immunol Rev. 1981;58:95–129. doi: 10.1111/j.1600-065x.1981.tb00351.x. [DOI] [PubMed] [Google Scholar]
- Zarling J. M., Kung P. C. Monoclonal antibodies which distinguish between human NK cells and cytotoxic T lymphocytes. Nature. 1980 Nov 27;288(5789):394–396. doi: 10.1038/288394a0. [DOI] [PubMed] [Google Scholar]
- van de Griend R. J., ten Berge I., Tanke H. J., Roos D., Schellekens P. T., Melief C. J., Zeijlemaker W. P., Astaldi A. Characterization of two subsets of human T gamma cells. J Immunol. 1982 May;128(5):1979–1985. [PubMed] [Google Scholar]



