Abstract
Myeloid-derived suppressor cells (MDSC) accumulate in most cancer patients and experimental animals with cancer. They accumulate in response to pro-inflammatory mediators and they use a variety of mechanisms to block both innate and adaptive antitumor immunity. Because of their critical role in obstructing immune responses, MDSC are a strategic obstacle to immunotherapies that require activation of the host’s cell-mediated and innate immune responses. Following a brief description of the factors that induce MDSC accumulation, this article reviews two newly discovered mechanisms that MDSC use to suppress the activation of CD4+ and CD8+ T cells. The first mechanism is MDSC sequestration of cysteine, an amino acid that T cells are unable to synthesize de novo and that they require for activation. The second mechanism is MDSC-mediated down-regulation of l-selectin. T cells must have an l-selectinhigh phenotype to home to lymph nodes and inflammatory sites where they encounter antigen and are activated. By down-regulating l-selectin on T cells, MDSC perturb T cell trafficking patterns and thereby inhibit T cell activation. Given the complexity of conditions that regulate MDSC accumulation and the variety of suppressive mechanisms used by MDSC, it is essential to understand which conditions and mechanisms are dominant so MDSC accumulation and/or activity can be targeted in individual patients to minimize MDSC-induced immune suppression.
Keywords: Tumor-induced immune suppression, Amino acid metabolism, T cell trafficking, Suppressive myeloid cells
Tumor-induced immune suppression
Most patients with advanced cancer are immune suppressed. As a result, immunotherapies based on activation of a patient’s immune system are unlikely to be effective unless the immune suppression is attenuated. A variety of different cells of hemopoietic origin contribute to tumor-induced immune suppression, including T regulatory cells [1], tumor-associated macrophages [2], type 2 NKT cells [3], mast cells [4], and myeloid-derived suppressor cells (MDSC). One or more of these suppressive cell populations are present in cancer patients; however, MDSC are present in most patients and therefore are a fundamental and virtually universal obstacle to generating antitumor immunity. Because of their widespread presence in most cancer patients, many avenues are being explored to minimize or eliminate immune suppression caused by MDSC. Some of these strategies are aimed at preventing the accumulation of MDSC, while others are focused on inhibiting the mechanisms by which MDSC mediate their effects. Both of these therapeutic approaches are challenging because MDSC are a heterogeneous population of cells that are induced by multiple factors. Likewise, MDSC mediate suppression via multiple mechanisms so that obstructing MDSC activity will require interfering with a diverse spectrum of effector molecules and pathways. Studies conducted during the past decade have identified several conditions that induce the accumulation of MDSC and have documented varied mechanisms used by MDSC to block antitumor immunity. As MDSC continue to be intensively studied, new suppressive mechanisms are being identified. This article will provide a concise background on MDSC biology, briefly review MDSC suppressive mechanisms that have been previously described (and reviewed), and then focus on recently identified suppressive mechanisms employed by MDSC.
MDSC suppress adaptive and innate immunity
Before MDSC were defined as a specific suppressor cell population, studies of tolerance in mice documented a population of so-called “natural suppressor” cells in the spleen [5]. Similar cells were subsequently found in the tumors and lymph nodes of patients with head and neck cancer [6] and in the bone marrow of mice with lung tumors [7]. These cells were characterized as suppressor cells based on their ability to block the activation of T cells and to promote tumor growth. Extensive subsequent studies demonstrated that these suppressor cells were of myeloid origin and the term “MDSC” was coined to refer to this diverse family of cells [8].
The role of MDSC as critical regulatory cells that down-regulate antitumor immunity has been well established by both in vitro and in vivo studies. In vitro, MDSC are potent inhibitors of both antigen-specific and non-specific T cell activation as shown by co-culture experiments of MDSC with peptide-activated transgenic T cells (mouse MDSC) or anti-CD3-activated T cells (mouse and human MDSC), respectively [9–11]. CD4+ and CD8+ T cells are equally suppressed and the suppression requires cell to cell contact; however, suppression can be either MHC restricted [12] or unrestricted [13]. In vivo treatment of tumor-bearing mice with antibodies to Gr1 [14], the chemotherapeutic drug gemcitabine [15, 16], or retinoic acid [17] reduces MDSC levels and leads to improved T cell activation and delayed tumor progression. Retinoic acid has a similar effect in cancer patients [18]. In contrast, adoptive transfer of tumor-induced MDSC into tumor-free mice promotes tumor growth and inhibits T cell activation [19].
In addition to inhibiting T cell activation, MDSC also impact antitumor immunity by perturbing innate immunity through their interactions with macrophages, natural killer (NK) cells, and NKT cells. M1 or classically activated macrophages promote tumor regression; however, in the presence of MDSC, macrophages are converted to an M2 or alternatively activated phenotype which enhance tumor progression [13]. This re-polarization is the result of cross-talk between MDSC and macrophages which increases MDSC production of IL-10 and decreases macrophage production of IL-12 [15]. MDSC have shown mixed effects on NK cells. Some NK subpopulations suppress NK cytotoxicity by blocking NK production of IFN ([16, 20]; however, other subpopulations activated NK cells which in turn eliminated MDSC [21]; whether NKT cells promote or deter the accumulation of MDSC depends on the type of NKT cell. Type II NKT cells produce IL-13 which drives the accumulation of MDSC and M2 macrophages, thereby facilitating tumor progression [13, 22]. In contrast, type I or iNKT cells reduce MDSC accumulation [23].
MDSC are a heterogeneous population of immature myeloid cells
In addition to cancer, MDSC are also induced by bacterial [24, 25], viral [23], and parasitic infection [26], chemotherapy [27], traumatic stress [28], and autoimmunity [29, 30]. In all situations, MDSC arise from myeloid progenitor cells that do not terminally differentiate into mature macrophages, dendritic cells, or granulocytes. They are a heterogeneous population of cells that are characterized by their functional ability to suppress T cell activation and by their expression of particular cell surface markers. Two major classes of MDSC have been described: granulocytic and monocytic MDSC. Granulocytic MDSC are polymorphonuclear and contain high levels of arginase, while monocytic MDSC are mononuclear and contain both arginase and iNOS [31, 32]. In mouse, both populations universally express the granulocyte marker Gr1 and the macrophage/dendritic cell marker CD11b, while monocytic MDSC tend to have high levels of ICAM-1 (CD54), F4/80, and Ly6C, and granulocytic MDSC express high levels of Ly6G. Both subpopulations in the mouse can also express CD115 (c-fms), CD16/32 (FcR), IL-4Rα (CD124), and low levels of CD80. Both granulocytic and monocytic human MDSC express CD33, CD11b, and IL-4Rα, and low levels of CD15, while monocytic MDSC are characterized by their additional expression of CD14 and less CD15 (reviewed in [33, 34]).
The variation in cell surface markers between MDSC isolated from individuals with different tumors indicates there is also heterogeneity within the monocytic and granulocytic subpopulations. The existence of distinct and varied subpopulations is not surprising, since MDSC are induced by multiple environmental conditions and factors. In the case of tumor-induced MDSC, the factors driving MDSC accumulation originate from tumor cells. Therefore, a given tumor producing a particular set of inducer molecules will produce MDSC of a particular phenotype, while a tumor producing a different set of inducer molecules may induce MDSC with a somewhat different phenotype.
MDSC are induced by inflammation
Compelling epidemiological and biological data support the concept that chronic inflammation increases cancer risk by causing genetic alterations, promoting neoangiogenesis, altering responses to hormonal signals, and inducing cell proliferation accompanied by altered cell trafficking [35]. Recent studies have also suggested that chronic inflammation promotes tumor progression by down-regulating adaptive tumor immunity. These findings have led us to hypothesize that chronic inflammation induces MDSC which in turn down-regulate antitumor immunity, thereby allowing the outgrowth and sustained presence of MDSC. Studies by ourselves and others have confirmed this hypothesis.
Numerous tumor-secreted factors are inducers of MDSC. Most of these factors are directly or indirectly pro-inflammatory. For example, the pro-inflammatory cytokines IL-6 and IL-1β, which are present in the microenvironment of many tumors, dramatically increase the rate of accumulation and T cell suppressive activity of MDSC [36, 37]. IL-1β-driven inflammation also increases MDSC suppression of innate immunity by facilitating cross-talk between MDSC and macrophages [38]. The bioactive lipid prostaglandin E2, which is commonly found in inflammatory environments, induces the differentiation of MDSC from c-kit+ hemopoietic stem cells and drives the T cell suppressive activity of mature MDSC [39, 40]. Similarly, the pro-inflammatory S100A8/A9 proteins [19, 41] and the complement component C5a [42] induce MDSC accumulation. Conversely, reducing inflammation within the tumor microenvironment, either by treatment with PGE2 receptor antagonists [40], or by deletion of the receptors for IL-1 [43] or PGE2 [44] on host cells, minimizes MDSC accumulation. GM-CSF, a cytokine that drives myeloid, and specifically dendritic cell differentiation, and VEGF, which drives angiogenesis, are also inflammation-associated molecules and also induce the accumulation of MDSC [44]. Most of these factors directly activate MDSC, while some activate MDSC indirectly via host cells in the tumor microenvironment. Because different tumors produce different quantities and ratios of these pro-inflammatory molecules, tumor microenvironments differ from individual to individual, and therefore give rise to MDSC populations with varied phenotypes.
In addition to responding to inflammatory signals, MDSC also contribute to the inflammatory tumor microenvironment by secreting pro-inflammatory mediators, such as IL-6 and S100A8/A9 [41] producing an autocrine feedback loop that induces and sustains MDSC in the tumor microenvironment.
MDSC use multiple mechanisms to suppress antitumor immunity
Myeloid-derived suppressor cells use a variety of mechanisms to suppress tumor immunity. Monocytic and granulocytic MDSC both use a mechanism in which arginase catabolizes l-arginine. This mechanism has been comprehensively reviewed [45, 46] and, therefore, will only be briefly described here. Like other cells, MDSC and T cells require l-arginine for protein synthesis. MDSC produce high levels of intracellular arginase requiring them to import excess arginine through their CAT-2B transporter. As a result, they deplete their environment of l-arginine and limit l-arginine availability to T cells. T cells deprived of l-arginine are deficient for CD3ζ chain [47, 48] and are arrested in the Go–G1 phase of the cell cycle [49]. Granulocytic MDSC also suppress through their high levels of reactive oxygen species which are caused by increased activation of STAT3 and NADPH. Reactive oxygen species including peroxynitrite and hydrogen peroxide induce apoptosis of T cells, and also nitrate T cell receptors which are then unable to bind peptide/MHC complexes. Hence, T cell activation is blocked [12]. In contrast, monocytic MDSC have activated STAT1 which elevates iNOS and in turn generates NO. MDSC also perturb T cell activation by inducing Foxp3+-regulatory T cells (Tregs). Different studies disagree on the requirement for MDSC production of TGFβ in the induction of Tregs [50, 51], suggesting that different MDSC subpopulations may activate Tregs through disparate mechanisms.
MDSC sequester cystine and prevent T cells from obtaining cysteine
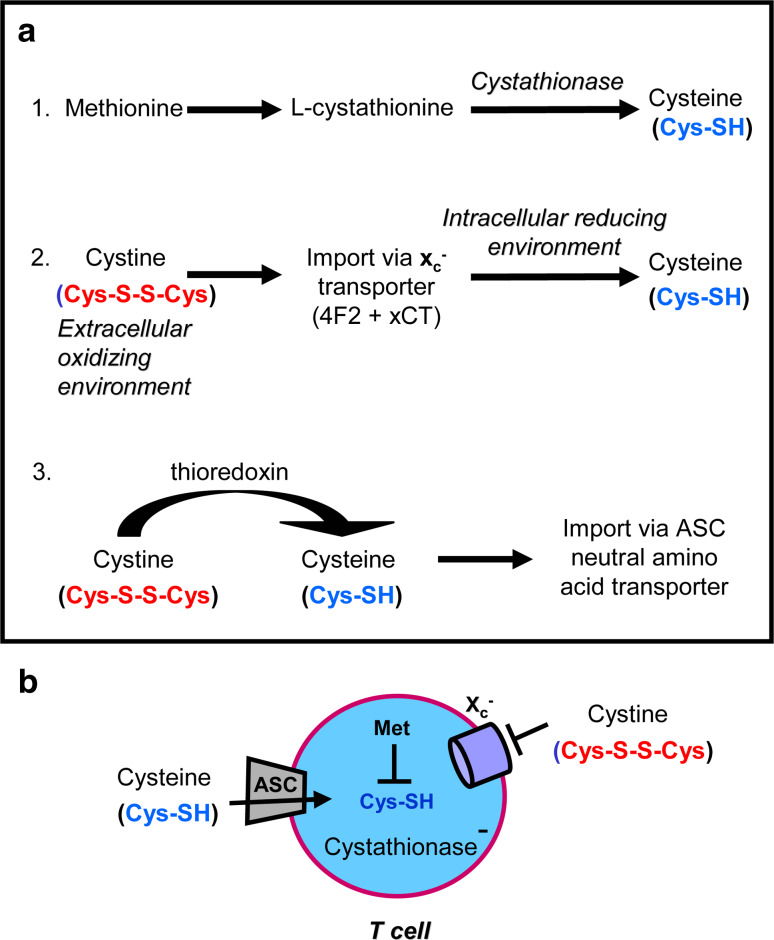
We have recently described another mechanism involving the amino acid cysteine by which MDSC inhibit T cell activation. Cysteine is required by all cells for protein synthesis. Typically, cells synthesize cysteine from their intracellular pool of methionine using the enzyme cystathionase [52, 53]. Alternatively, cells import cystine, the oxidized form of cysteine from the oxidizing extracellular environment, through their plasma membrane x−c cystine/glutamate antiporter [54]. The imported cystine is reduced to cysteine in the intracellular reducing environment [55]. T lymphocytes cannot generate cysteine through either of these mechanisms because they do not express cystathionase and they lack the xCT chain of the x−c cystine transporter [56]. As a result, cysteine is an essential amino acid for T cells and T cells are completely dependent on exogenous sources of cysteine which they import via their ASC neutral amino acid transporter (Fig. 1). Although resting T cells must take up extracellular cysteine to survive, T cells have their greatest requirement for cysteine when they become antigen activated, proliferate, and differentiate. Conveniently, cysteine is provided by antigen-presenting cells (APC) such as dendritic cells (DC) and macrophages during antigen processing and presentation. These APC import cystine through their x−c transporter, reduce it to cysteine and then export the cysteine through their ASC neutral amino acid transporter [57]. In addition, DC and macrophages secrete thioredoxin which reduces extracellular cystine to cysteine, which is then available for the uptake by T cells through their ASC transporter [58, 59] (Fig. 1). Because extracellular spaces are oxidizing environments, cysteine released by APC would normally be oxidized to cystine and therefore not useful to T cells. However, during antigen presentation T cells and APC are in very close proximity so cysteine exported by APC can be directly imported by T cells (Fig. 2). Therefore, the process of antigen presentation not only delivers antigen specific and co-stimulation signals to activate T cells, but also provides the cysteine necessary for T cell activation and subsequent proliferation and differentiation.
Fig. 1.
Mammalian cells, but not T cells, generate cysteine from methionine and/or cystine. a Cells that contain the enzyme cystathionase convert intracellular methionine to cysteine. Cells that contain the x−c plasma membrane transporter, which is a heterodimer of the 4F2 and xCT chains, import oxidized cystine from the extracellular environment and reduce it to cysteine in their intracellular reducing environment. Cells can also obtain cysteine by importing it through their ASC neutral amino acid plasma membrane transporter. Thioredoxin reduces extracellular cystine to cysteine; however, extracellular cysteine is rapidly oxidized back to cystine if it is not quickly taken up by cells. b T cells do not synthesize cystathionase and lack the xCT chain of the x−c transporter so they cannot convert methionine to cysteine and they cannot import cystine. Therefore, cysteine is an essential amino acid for T cells and they must import it through their ASC transporter
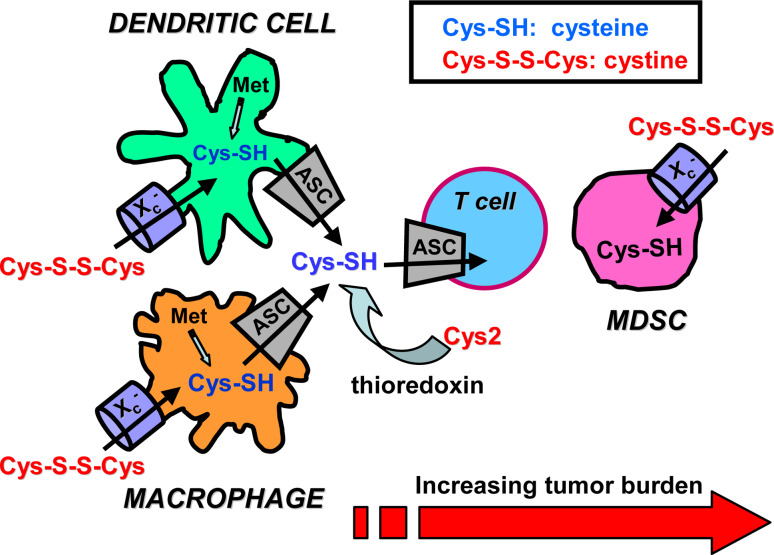
Fig. 2.
MDSC prevent T cell activation by sequestering cystine and limiting the availability of cysteine. As described in Fig. 1, cysteine is an essential amino acid for T cells because T cells lack cystathionase, and have a defective cystine transporter. As a result, T cells must obtain their cysteine from extracellular sources. DC and macrophages normally contain surplus cysteine because they import cystine and reduce it to cysteine and they intracellularly synthesize cysteine from cystine through the action of cystathionase. The surplus cysteine is exported via their ASC transporter. DC and macrophages also generate cysteine through their production of thioredoxin, which reduces extracellular cystine to cysteine. During antigen presentation, APC and T cells are in close proximity, so the cysteine released/produced by APC is readily available for uptake by T cells. In contrast to APC, MDSC do not contain cystathionase or the ASC transporter so their cysteine must be generated from imported cystine. As a result, MDSC deplete their environment of cystine and do not export cysteine. Therefore, when MDSC are present during antigen presentation, the local environment becomes cystine and cysteine-deficient and T cells cannot proliferate and become activated
Because cysteine is an essential amino acid for T cell activation, we hypothesized that MDSC may perturb T cell activation by inhibiting cysteine uptake. To determine if this hypothesis was correct, we increased extracellular cysteine in cultures containing MDSC, transgenic peptide-specific CD4+ or CD8+ T cells, and cognate peptide. Cysteine was increased by addition of the reducing agent β-mercaptoethanol (reduces cystine to cysteine) or N-acetyl cysteine (NAC), a form of cysteine that is stable in the extracellular oxidizing environment. Inclusion of either NAC or β-mercaptoethanol partially reversed MDSC-induced suppression, consistent with the hypothesis that MDSC suppress by perturbing cysteine availability [60].
To decipher the mechanism by which MDSC perturb cysteine uptake, we examined expression of xCT and 4F2 (CD98), the two polypeptide chains that make up the heterodimeric x−c cystine transporter, and ASC, the neutral amino acid transporter for cysteine in MDSC, macrophages, and T cells. As expected, T cells, macrophages and DC contained the ASC transporter, and macrophages and DC contained both chains of the x−c transporter, while T cells lacked the xCT chain. In contrast, MDSC expressed xCT and 4F2 in their plasma membranes; however, they did not contain the ASC transporter, suggesting that MDSC could import cystine, but not export cysteine. This conclusion was confirmed by studies measuring cystine uptake and cysteine export via the x−c and ASC transporters, respectively. MDSC import cysteine with approximately the same kinetics as macrophages and DC; however, in contrast to DC and macrophages, MDSC do not export cysteine. Intracellular pools of cysteine in MDSC are generated exclusively from imported cystine since MDSC, like T cells, do not contain cystathionase and therefore cannot convert methionine to cysteine. Despite their inability to synthesize cysteine from methionine, MDSC contain significantly larger intracellular pools of cysteine as compared to macrophages and DC, consistent with the concept that MDSC sequester cysteine [60] (Fig. 2).
These findings led us to propose that MDSC sequester cystine and thereby prevent macrophages and DC from importing cystine. In the absence of imported cystine, macrophages and DC must generate the cysteine they require from methionine. As a result, macrophages and DC export significantly less cysteine, so T cells do not obtain the cysteine they need for activation and proliferation. This scenario was confirmed by measuring cysteine released from macrophages co-cultured with increasing numbers of MDSC. At ratios of greater than 1:1 macrophages to MDSC, macrophage release of cysteine was reduced by >75%, further demonstrating that MDSC limit the pool of cysteine available for T cells. In addition to their failure to release cysteine, MDSC also limit the pool of extracellular cysteine by perturbing thioredoxin production of cysteine [60]. Because thioredoxin generates cysteine from cystine, if the extracellular pool of cystine is small, thioredoxin will not have sufficient substrate to produce cysteine.
The preceding experiments demonstrate that MDSC dramatically limit cysteine availability in culture. To determine if MDSC also limit cystine and cysteine availability in vivo, we measured the amount of cystine in the serum of mice with mammary tumors. Consistent with the in vitro findings, tumor-bearing mice had statistically significantly less serum cystine relative to tumor-free individuals [60].
In addition to being essential for protein synthesis, cysteine is a substrate for the generation of glutathione (GSH) [61], a major intracellular redox molecule that protects cells from oxidative stress. Oxidative stress is frequent within solid tumors so that even if cysteine-starved T cells are activated they are likely to die when they traffic to tumor sites due to insufficient GSH.
If MDSC-mediated cystine sequestration is biologically relevant in vivo, then provision of additional cysteine to tumor-bearing individuals may delay tumor progression. Epidemiological studies have shown that reduced risk of breast cancer in women correlates with high levels of serum cysteine [62] and NAC has been shown to delay tumor progression in mice [63]. Tumor reduction in these studies was attributed to a decrease in genetic instability resulting from decreased oxidative stress. However, NAC may also limit tumor progression by serving as a source of cysteine and therefore facilitating the activation of tumor-reactive T cells. Our unpublished in vivo studies in which antigen-specific T cell activation was assessed in mice fed NAC water support this conclusion and demonstrate that provision of NAC to tumor-bearing mice partially reverses MDSC-mediated T cell suppression (P. Sinha and S. Ostrand-Rosenberg, unpublished). Therefore, although increasing levels of MDSC impair the ability of T cells to obtain the cysteine required for their activation, provision of NAC, an already FDA-approved compound, may be a useful adjunctive strategy to overcome MDSC suppression in individuals being treated with active immunotherapy.
MDSC impair T cell homing to lymph nodes
We have also recently demonstrated that MDSC impair T cell activation by preventing T cells from homing to sites where they would otherwise become activated. Adaptive T cell-mediated antitumor immunity requires the activation of tumor-specific T cells. Naive T cells typically encounter antigen and become activated in secondary lymph nodes draining tumor sites or within solid tumors themselves [64, 65]. Antigen-naive T cells are directed to these locations because they express high levels of l-selectin (CD62L), a selectin family member that facilitates the extravasation of leukocytes from the blood and lymphatics to lymph nodes and inflammatory locales, such as tumor microenvironments [66, 67].
Despite their naive status, CD4+ and CD8+ T cells in the peripheral blood of tumor-bearing mice and cancer patients frequently have an l-selectin low (l-selectinlow) phenotype, suggesting that they may not be able to home to secondary lymphoid tissue and become activated. l-selectin levels inversely correlate with MDSC levels, since CD4+ and CD8+ T cells from mice with the largest quantities of MDSC have the least l-selectin. In addition, surgical removal of primary tumor, which reduces MDSC levels, resulted in increased expression of l-selectin on circulating T cells [68]. These results were consistent with the hypothesis that MDSC drive l-selectin levels; however, they did not eliminate the possibility that tumor-derived factors, rather than MDSC, regulate l-selectin levels. To distinguish these alternatives, tumor-bearing mice were treated with gemcitabine, a chemotherapeutic drug that reduces the accumulation of MDSC, but does not affect tumor growth [15]. T cells from >8-month-old mice were also studied because MDSC levels in tumor-free mice increase with age. In both of these scenarios, T cell expression of l-selectin and quantities of MDSC in blood were inversely correlated. Furthermore, CD4+ or CD8+ T cells co-cultured with MDSC had an l-selectinlow phenotype demonstrating that MDSC directly down-regulate T cell expression of l-selectin [68].
During standard T cell activation conditions, l-selectin is down-regulated so that once activated, T cells exit peripheral lymph nodes and migrate to their target sites. The down-regulation is mediated by proteolytic cleavage and shedding of the l-selectin ectodomain. The primary “sheddase” is ADAM 17 (a disintegrin and metaloproteinase domain 17) or TACE (TNFα-converting enzyme). ADAM 17 is commonly present in the cytoplasm of many cells, including T cells, and becomes active when it is translocated to the plasma membrane, where it has access to membrane-bound l-selectin [69]. MDSC cleave l-selectin because they constitutively express ADAM17 at their cell surface. Interestingly, Gr1+CD11b+ cells from tumor-free mice also contain plasma membrane ADAM17, suggesting that ADAM17 activity is an inherent property of Gr1+CD11b+ cells and not induced by tumor.
Collectively, these findings suggest that naive T cells in tumor-bearing individuals cannot efficiently traffic to lymph nodes or tumor sites. Therefore, potentially tumor-reactive CD4+ and CD8+ T cells may be present in the periphery, however, because they cannot properly traffic they will not have access to tumor antigens and will not be activated.
Conclusions
Activation of the host’s immune system against endogenous cancer cells was first proposed by Paul Ehrlich over 100 years ago, and continues to be a goal for cancer immunologists. Many innovative and creative cancer immunotherapy strategies have shown promising results in experimental systems. However, their translation to the clinic and for the treatment of established cancer has been disappointing. It is becoming increasingly apparent that active immunotherapy is attenuated by immune suppressive mechanisms present in cancer patients. As one of the major contributors to tumor-induced immune suppression, MDSC are a major obstacle to active immunotherapy. MDSC heterogeneity combined with the varied conditions that activate these cells and the multiple suppressive mechanisms employed by MDSC make it unlikely that a single approach will control MDSC. However, understanding the conditions that induce MDSC and the mechanisms used by MDSC to suppress antitumor immunity is essential for minimizing MDSC-mediated suppression and developing effective immunotherapies. Major questions that remain to be addressed are (1) Are all of the known inducers of MDSC equally responsible for MDSC accumulation or are certain inducers dominant? (2) What is the relative contribution of the various suppressive mechanisms to the overall suppressive activity of MDSC? (3) Are certain suppressive mechanisms dominant and does dominance vary depending on the type of tumor? (4) Is there convergence in the signaling pathways and/or mediators used by the various suppressive mechanisms so one therapy could simultaneously target multiple mechanisms? Tumors are dynamic, complex and heterogeneous mixtures of malignant and host components that vary with tumor progression, so the answers to these questions may be tumor specific, stage specific, and perhaps governed by the genetic make-up of the individual, making it challenging to decipher the relative roles of the various suppressive mechanisms. Regardless of the complexity, in vivo models with deletion of specific genes are essential to parse out the relative contribution of each mechanism. Obtaining this information is essential so we can preferentially target in individual patients the dominant factors that drive MDSC accumulation and suppressive activity and delete or inactivate this detrimental cell population.
Acknowledgments
These studies were supported by National Institutes of Health Grants RO1CA115880 and RO1CA84232. Animal studies described herein were approved by the UMBC Institutional Animal Care and Use Committee.
Conflict of interest statement
The author has no conflict of interest with the organization sponsoring this research.
Footnotes
This paper is a Focussed Research Review based on a presentation given at the Ninth International Conference on Progress in Vaccination against Cancer (PIVAC 9), held in Sofia, Bulgaria, 8–10 October 2009.
References
- 1.Curiel TJ. Regulatory T cells and treatment of cancer. Curr Opin Immunol. 2008;20:241–246. doi: 10.1016/j.coi.2008.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Solinas G, Germano G, Mantovani A, Allavena P. Tumor-associated macrophages (TAM) as major players of the cancer-related inflammation. J Leukoc Biol. 2009;86:1065–1073. doi: 10.1189/jlb.0609385. [DOI] [PubMed] [Google Scholar]
- 3.Berzofsky JA, Terabe M. NKT cells in tumor immunity: opposing subsets define a new immunoregulatory axis. J Immunol. 2008;180:3627–3635. doi: 10.4049/jimmunol.180.6.3627. [DOI] [PubMed] [Google Scholar]
- 4.Groot Kormelink T, Abudukelimu A, Redegeld FA. Mast cells as target in cancer therapy. Curr Pharm Des. 2009;15:1868–1878. doi: 10.2174/138161209788453284. [DOI] [PubMed] [Google Scholar]
- 5.Strober S. Natural suppressor (NS) cells, neonatal tolerance, and total lymphoid irradiation: exploring obscure relationships. Annu Rev Immunol. 1984;2:219–237. doi: 10.1146/annurev.iy.02.040184.001251. [DOI] [PubMed] [Google Scholar]
- 6.Pak AS, Wright MA, Matthews JP, Collins SL, Petruzzelli GJ, Young MR. Mechanisms of immune suppression in patients with head and neck cancer: presence of CD34(+) cells which suppress immune functions within cancers that secrete granulocyte-macrophage colony-stimulating factor. Clin Cancer Res. 1995;1:95–103. [PubMed] [Google Scholar]
- 7.Young MR, Wright MA. Myelopoiesis-associated immune suppressor cells in mice bearing metastatic Lewis lung carcinoma tumors: gamma interferon plus tumor necrosis factor alpha synergistically reduces immune suppressor and tumor growth-promoting activities of bone marrow cells and diminishes tumor recurrence and metastasis. Cancer Res. 1992;52:6335–6340. [PubMed] [Google Scholar]
- 8.Gabrilovich DI, Bronte V, Chen SH, Colombo MP, Ochoa A, Ostrand-Rosenberg S, Schreiber H. The terminology issue for myeloid-derived suppressor cells. Cancer Res. 2007;67:425. doi: 10.1158/0008-5472.CAN-06-3037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bronte V, Apolloni E, Cabrelle A, Ronca R, Serafini P, Zamboni P, Restifo NP, Zanovello P. Identification of a CD11b(+)/Gr-1(+)/CD31(+) myeloid progenitor capable of activating or suppressing CD8(+) T cells. Blood. 2000;96:3838–3846. [PMC free article] [PubMed] [Google Scholar]
- 10.Gabrilovich DI, Velders MP, Sotomayor EM, Kast WM. Mechanism of immune dysfunction in cancer mediated by immature Gr-1+ myeloid cells. J Immunol. 2001;166:5398–5406. doi: 10.4049/jimmunol.166.9.5398. [DOI] [PubMed] [Google Scholar]
- 11.Sinha P, Clements VK, Ostrand-Rosenberg S. Reduction of myeloid-derived suppressor cells and induction of M1 macrophages facilitate the rejection of established metastatic disease. J Immunol. 2005;174:636–645. doi: 10.4049/jimmunol.174.2.636. [DOI] [PubMed] [Google Scholar]
- 12.Nagaraj S, Gupta K, Pisarev V, Kinarsky L, Sherman S, Kang L, Herber DL, Schneck J, Gabrilovich DI. Altered recognition of antigen is a mechanism of CD8+ T cell tolerance in cancer. Nat Med. 2007;13:828–835. doi: 10.1038/nm1609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sinha P, Clements VK, Ostrand-Rosenberg S. Interleukin-13-regulated M2 macrophages in combination with myeloid suppressor cells block immune surveillance against metastasis. Cancer Res. 2005;65:11743–11751. doi: 10.1158/0008-5472.CAN-05-0045. [DOI] [PubMed] [Google Scholar]
- 14.Terabe M, Matsui S, Park JM, Mamura M, Noben-Trauth N, Donaldson DD, Chen W, Wahl SM, Ledbetter S, Pratt B, Letterio JJ, Paul WE, Berzofsky JA. Transforming growth factor-beta production and myeloid cells are an effector mechanism through which CD1d-restricted T cells block cytotoxic T lymphocyte-mediated tumor immunosurveillance: abrogation prevents tumor recurrence. J Exp Med. 2003;198:1741–1752. doi: 10.1084/jem.20022227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sinha P, Clements VK, Bunt SK, Albelda SM, Ostrand-Rosenberg S. Cross-talk between myeloid-derived suppressor cells and macrophages subverts tumor immunity toward a type 2 response. J Immunol. 2007;179:977–983. doi: 10.4049/jimmunol.179.2.977. [DOI] [PubMed] [Google Scholar]
- 16.Suzuki E, Kapoor V, Jassar AS, Kaiser LR, Albelda SM. Gemcitabine selectively eliminates splenic Gr-1+/CD11b+ myeloid suppressor cells in tumor-bearing animals and enhances antitumor immune activity. Clin Cancer Res. 2005;11:6713–6721. doi: 10.1158/1078-0432.CCR-05-0883. [DOI] [PubMed] [Google Scholar]
- 17.Kusmartsev S, Cheng F, Yu B, Nefedova Y, Sotomayor E, Lush R, Gabrilovich D. All-trans-retinoic acid eliminates immature myeloid cells from tumor-bearing mice and improves the effect of vaccination. Cancer Res. 2003;63:4441–4449. [PubMed] [Google Scholar]
- 18.Mirza N, Fishman M, Fricke I, Dunn M, Neuger AM, Frost TJ, Lush RM, Antonia S, Gabrilovich DI. All-trans-retinoic acid improves differentiation of myeloid cells and immune response in cancer patients. Cancer Res. 2006;66:9299–9307. doi: 10.1158/0008-5472.CAN-06-1690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cheng P, Corzo CA, Luetteke N, Yu B, Nagaraj S, Bui MM, Ortiz M, Nacken W, Sorg C, Vogl T, Roth J, Gabrilovich DI. Inhibition of dendritic cell differentiation and accumulation of myeloid-derived suppressor cells in cancer is regulated by S100A9 protein. J Exp Med. 2008;205:2235–2249. doi: 10.1084/jem.20080132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu C, Yu S, Kappes J, Wang J, Grizzle WE, Zinn KR, Zhang HG. Expansion of spleen myeloid suppressor cells represses NK cell cytotoxicity in tumor-bearing host. Blood. 2007;109:4336–4342. doi: 10.1182/blood-2006-09-046201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nausch N, Galani IE, Schlecker E, Cerwenka A. Mononuclear myeloid-derived “suppressor” cells express RAE-1 and activate natural killer cells. Blood. 2008;112:4080–4089. doi: 10.1182/blood-2008-03-143776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Terabe M, Swann J, Ambrosino E, Sinha P, Takaku S, Hayakawa Y, Godfrey DI, Ostrand-Rosenberg S, Smyth MJ, Berzofsky JA. A nonclassical non-Valpha14Jalpha18 CD1d-restricted (type II) NKT cell is sufficient for down-regulation of tumor immunosurveillance. J Exp Med. 2005;202:1627–1633. doi: 10.1084/jem.20051381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.De Santo C, Salio M, Masri SH, Lee LY, Dong T, Speak AO, Porubsky S, Booth S, Veerapen N, Besra GS, Grone HJ, Platt FM, Zambon M, Cerundolo V. Invariant NKT cells reduce the immunosuppressive activity of influenza A virus-induced myeloid-derived suppressor cells in mice and humans. J Clin Invest. 2008;118:4036–4048. doi: 10.1172/JCI36264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Delano MJ, Scumpia PO, Weinstein JS, Coco D, Nagaraj S, Kelly-Scumpia KM, O’Malley KA, Wynn JL, Antonenko S, Al-Quran SZ, Swan R, Chung CS, Atkinson MA, Ramphal R, Gabrilovich DI, Reeves WH, Ayala A, Phillips J, Laface D, Heyworth PG, Clare-Salzler M, Moldawer LL. MyD88-dependent expansion of an immature GR-1(+)CD11b(+) population induces T cell suppression and Th2 polarization in sepsis. J Exp Med. 2007;204:1463–1474. doi: 10.1084/jem.20062602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Haile LA, von Wasielewski R, Gamrekelashvili J, Kruger C, Bachmann O, Westendorf AM, Buer J, Liblau R, Manns MP, Korangy F, Greten TF. Myeloid-derived suppressor cells in inflammatory bowel disease: a new immunoregulatory pathway. Gastroenterology. 2008;135:871–881. doi: 10.1053/j.gastro.2008.06.032. [DOI] [PubMed] [Google Scholar]
- 26.Brys L, Beschin A, Raes G, Ghassabeh GH, Noel W, Brandt J, Brombacher F, De Baetselier P. Reactive oxygen species and 12/15-lipoxygenase contribute to the antiproliferative capacity of alternatively activated myeloid cells elicited during helminth infection. J Immunol. 2005;174:6095–6104. doi: 10.4049/jimmunol.174.10.6095. [DOI] [PubMed] [Google Scholar]
- 27.Angulo I, de las Heras FG, Garcia-Bustos JF, Gargallo D, Munoz-Fernandez MA, Fresno M. Nitric oxide-producing CD11b(+)Ly-6G(Gr-1)(+)CD31(ER-MP12)(+) cells in the spleen of cyclophosphamide-treated mice: implications for T-cell responses in immunosuppressed mice. Blood. 2000;95:212–220. [PubMed] [Google Scholar]
- 28.Makarenkova VP, Bansal V, Matta BM, Perez LA, Ochoa JB. CD11b+/Gr-1+ myeloid suppressor cells cause T cell dysfunction after traumatic stress. J Immunol. 2006;176:2085–2094. doi: 10.4049/jimmunol.176.4.2085. [DOI] [PubMed] [Google Scholar]
- 29.Kerr EC, Raveney BJ, Copland DA, Dick AD, Nicholson LB. Analysis of retinal cellular infiltrate in experimental autoimmune uveoretinitis reveals multiple regulatory cell populations. J Autoimmun. 2008;31:354–361. doi: 10.1016/j.jaut.2008.08.006. [DOI] [PubMed] [Google Scholar]
- 30.Zhu B, Bando Y, Xiao S, Yang K, Anderson AC, Kuchroo VK, Khoury SJ. CD11b+ Ly-6C(hi) suppressive monocytes in experimental autoimmune encephalomyelitis. J Immunol. 2007;179:5228–5237. doi: 10.4049/jimmunol.179.8.5228. [DOI] [PubMed] [Google Scholar]
- 31.Movahedi K, Guilliams M, Van den Bossche J, Van den Bergh R, Gysemans C, Beschin A, De Baetselier P, Van Ginderachter JA. Identification of discrete tumor-induced myeloid-derived suppressor cell subpopulations with distinct T cell-suppressive activity. Blood. 2008;111:4233–4244. doi: 10.1182/blood-2007-07-099226. [DOI] [PubMed] [Google Scholar]
- 32.Youn JI, Nagaraj S, Collazo M, Gabrilovich DI. Subsets of myeloid-derived suppressor cells in tumor-bearing mice. J Immunol. 2008;181:5791–5802. doi: 10.4049/jimmunol.181.8.5791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gabrilovich DI, Nagaraj S (2009) Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol [DOI] [PMC free article] [PubMed]
- 34.Serafini P, Borrello I, Bronte V. Myeloid suppressor cells in cancer: recruitment, phenotype, properties, and mechanisms of immune suppression. Semin Cancer Biol. 2006;16:53–65. doi: 10.1016/j.semcancer.2005.07.005. [DOI] [PubMed] [Google Scholar]
- 35.Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. 2008;454:436–444. doi: 10.1038/nature07205. [DOI] [PubMed] [Google Scholar]
- 36.Bunt SK, Sinha P, Clements VK, Leips J, Ostrand-Rosenberg S. Inflammation induces myeloid-derived suppressor cells that facilitate tumor progression. J Immunol. 2006;176:284–290. doi: 10.4049/jimmunol.176.1.284. [DOI] [PubMed] [Google Scholar]
- 37.Song X, Krelin Y, Dvorkin T, Bjorkdahl O, Segal S, Dinarello CA, Voronov E, Apte RN. CD11b+/Gr-1+ immature myeloid cells mediate suppression of T cells in mice bearing tumors of IL-1beta-secreting cells. J Immunol. 2005;175:8200–8208. doi: 10.4049/jimmunol.175.12.8200. [DOI] [PubMed] [Google Scholar]
- 38.Bunt SK, Clements VK, Hanson EM, Sinha P, Ostrand-Rosenberg S. Inflammation enhances myeloid-derived suppressor cell cross-talk by signaling through Toll-like receptor 4. J Leukoc Biol. 2009;85:996–1004. doi: 10.1189/jlb.0708446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rodriguez PC, Hernandez CP, Quiceno D, Dubinett SM, Zabaleta J, Ochoa JB, Gilbert J, Ochoa AC. Arginase I in myeloid suppressor cells is induced by COX-2 in lung carcinoma. J Exp Med. 2005;202:931–939. doi: 10.1084/jem.20050715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sinha P, Clements VK, Fulton AM, Ostrand-Rosenberg S. Prostaglandin E2 promotes tumor progression by inducing myeloid-derived suppressor cells. Cancer Res. 2007;67:4507–4513. doi: 10.1158/0008-5472.CAN-06-4174. [DOI] [PubMed] [Google Scholar]
- 41.Sinha P, Okoro C, Foell D, Freeze HH, Ostrand-Rosenberg S, Srikrishna G. Proinflammatory S100 proteins regulate the accumulation of myeloid-derived suppressor cells. J Immunol. 2008;181:4666–4675. doi: 10.4049/jimmunol.181.7.4666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Markiewski MM, DeAngelis RA, Benencia F, Ricklin-Lichtsteiner SK, Koutoulaki A, Gerard C, Coukos G, Lambris JD. Modulation of the antitumor immune response by complement. Nat Immunol. 2008;9:1225–1235. doi: 10.1038/ni.1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Bunt SK, Yang L, Sinha P, Clements VK, Leips J, Ostrand-Rosenberg S. Reduced inflammation in the tumor microenvironment delays the accumulation of myeloid-derived suppressor cells and limits tumor progression. Cancer Res. 2007;67:10019–10026. doi: 10.1158/0008-5472.CAN-07-2354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gabrilovich D. Mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat Rev Immunol. 2004;4:941–952. doi: 10.1038/nri1498. [DOI] [PubMed] [Google Scholar]
- 45.Bronte V, Zanovello P. Regulation of immune responses by l-arginine metabolism. Nat Rev Immunol. 2005;5:641–654. doi: 10.1038/nri1668. [DOI] [PubMed] [Google Scholar]
- 46.Rodriguez PC, Ochoa AC. Arginine regulation by myeloid derived suppressor cells and tolerance in cancer: mechanisms and therapeutic perspectives. Immunol Rev. 2008;222:180–191. doi: 10.1111/j.1600-065X.2008.00608.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ezernitchi AV, Vaknin I, Cohen-Daniel L, Levy O, Manaster E, Halabi A, Pikarsky E, Shapira L, Baniyash M. TCR zeta down-regulation under chronic inflammation is mediated by myeloid suppressor cells differentially distributed between various lymphatic organs. J Immunol. 2006;177:4763–4772. doi: 10.4049/jimmunol.177.7.4763. [DOI] [PubMed] [Google Scholar]
- 48.Rodriguez PC, Zea AH, Culotta KS, Zabaleta J, Ochoa JB, Ochoa AC. Regulation of T cell receptor CD3zeta chain expression by l-arginine. J Biol Chem. 2002;277:21123–21129. doi: 10.1074/jbc.M110675200. [DOI] [PubMed] [Google Scholar]
- 49.Rodriguez PC, Quiceno DG, Ochoa AC. L-arginine availability regulates T-lymphocyte cell-cycle progression. Blood. 2007;109:1568–1573. doi: 10.1182/blood-2006-06-031856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Huang B, Pan PY, Li Q, Sato AI, Levy DE, Bromberg J, Divino CM, Chen SH. Gr-1+ CD115+ immature myeloid suppressor cells mediate the development of tumor-induced T regulatory cells and T-cell anergy in tumor-bearing host. Cancer Res. 2006;66:1123–1131. doi: 10.1158/0008-5472.CAN-05-1299. [DOI] [PubMed] [Google Scholar]
- 51.Serafini P, Mgebroff S, Noonan K, Borrello I. Myeloid-derived suppressor cells promote cross-tolerance in B-cell lymphoma by expanding regulatory T cells. Cancer Res. 2008;68:5439–5449. doi: 10.1158/0008-5472.CAN-07-6621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gout PW, Buckley AR, Simms CR, Bruchovsky N. Sulfasalazine, a potent suppressor of lymphoma growth by inhibition of the x(c)- cystine transporter: a new action for an old drug. Leukemia. 2001;15:1633–1640. doi: 10.1038/sj.leu.2402238. [DOI] [PubMed] [Google Scholar]
- 53.Ishii I, Akahoshi N, Yu XN, Kobayashi Y, Namekata K, Komaki G, Kimura H. Murine cystathionine gamma-lyase: complete cDNA and genomic sequences, promoter activity, tissue distribution and developmental expression. Biochem J. 2004;381:113–123. doi: 10.1042/BJ20040243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mansoor MA, Svardal AM, Ueland PM. Determination of the in vivo redox status of cysteine, cysteinylglycine, homocysteine, and glutathione in human plasma. Anal Biochem. 1992;200:218–229. doi: 10.1016/0003-2697(92)90456-H. [DOI] [PubMed] [Google Scholar]
- 55.Arner ES, Holmgren A. Physiological functions of thioredoxin and thioredoxin reductase. Eur J Biochem. 2000;267:6102–6109. doi: 10.1046/j.1432-1327.2000.01701.x. [DOI] [PubMed] [Google Scholar]
- 56.Bannai S. Transport of cystine and cysteine in mammalian cells. Biochim Biophys Acta. 1984;779:289–306. doi: 10.1016/0304-4157(84)90014-5. [DOI] [PubMed] [Google Scholar]
- 57.Sato H, Watanabe H, Ishii T, Bannai S. Neutral amino acid transport in mouse peritoneal macrophages. J Biol Chem. 1987;262:13015–13019. [PubMed] [Google Scholar]
- 58.Angelini G, Gardella S, Ardy M, Ciriolo MR, Filomeni G, Di Trapani G, Clarke F, Sitia R, Rubartelli A. Antigen-presenting dendritic cells provide the reducing extracellular microenvironment required for T lymphocyte activation. Proc Natl Acad Sci USA. 2002;99:1491–1496. doi: 10.1073/pnas.022630299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Castellani P, Angelini G, Delfino L, Matucci A, Rubartelli A. The thiol redox state of lymphoid organs is modified by immunization: role of different immune cell populations. Eur J Immunol. 2008;38:2419–2425. doi: 10.1002/eji.200838439. [DOI] [PubMed] [Google Scholar]
- 60.Srivastava MK, Sinha P, Clements VK, Rodriguez P, Ostrand-Rosenberg S. Myeloid-derived suppressor cells inhibit T-cell activation by depleting cystine and cysteine. Cancer Res. 2010;70:68–77. doi: 10.1158/0008-5472.CAN-09-2587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sakakura Y, Sato H, Shiiya A, Tamba M, Sagara J, Matsuda M, Okamura N, Makino N, Bannai S. Expression and function of cystine/glutamate transporter in neutrophils. J Leukoc Biol. 2007;81:974–982. doi: 10.1189/jlb.0606385. [DOI] [PubMed] [Google Scholar]
- 62.Zhang SM, Willett WC, Selhub J, Manson JE, Colditz GA, Hankinson SE. A prospective study of plasma total cysteine and risk of breast cancer. Cancer Epidemiol Biomarkers Prev. 2003;12:1188–1193. [PubMed] [Google Scholar]
- 63.Gao P, Zhang H, Dinavahi R, Li F, Xiang Y, Raman V, Bhujwalla ZM, Felsher DW, Cheng L, Pevsner J, Lee LA, Semenza GL, Dang CV. HIF-dependent antitumorigenic effect of antioxidants in vivo. Cancer Cell. 2007;12:230–238. doi: 10.1016/j.ccr.2007.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Khan AI, Landis RC, Malhotra R. l-Selectin ligands in lymphoid tissues and models of inflammation. Inflammation. 2003;27:265–280. doi: 10.1023/A:1026056525755. [DOI] [PubMed] [Google Scholar]
- 65.Rosen SD. Ligands for l-selectin: homing, inflammation, and beyond. Annu Rev Immunol. 2004;22:129–156. doi: 10.1146/annurev.immunol.21.090501.080131. [DOI] [PubMed] [Google Scholar]
- 66.Bradley LM, Watson SR, Swain SL. Entry of naive CD4 T cells into peripheral lymph nodes requires l-selectin. J Exp Med. 1994;180:2401–2406. doi: 10.1084/jem.180.6.2401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tedder TF, Steeber DA, Pizcueta P. l-Selectin-deficient mice have impaired leukocyte recruitment into inflammatory sites. J Exp Med. 1995;181:2259–2264. doi: 10.1084/jem.181.6.2259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Hanson EM, Clements VK, Sinha P, Ilkovitch D, Ostrand-Rosenberg S. Myeloid-derived suppressor cells down-regulate l-selectin expression on CD4+ and CD8+ T cells. J Immunol. 2009;183:937–944. doi: 10.4049/jimmunol.0804253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Li Y, Brazzell J, Herrera A, Walcheck B. ADAM17 deficiency by mature neutrophils has differential effects on l-selectin shedding. Blood. 2006;108:2275–2279. doi: 10.1182/blood-2006-02-005827. [DOI] [PMC free article] [PubMed] [Google Scholar]