Abstract
Oral cancer has a tendency to be detected at late stage which is detrimental to the patients because of its high mortality and morbidity rates. Early detection of oral cancer is therefore important to reduce the burden of this devastating disease. In this review article, the most common oral precancerous lesions are discussed and the importance of early diagnosis is emphasized. In addition, the most common non-invasive oral cancer devices that can aid the general practitioners in early diagnosis are also discussed.
Keywords: early diagnosis, non-invasive detection tools, oral precancerous lesions
Introduction
The incidence of oral cancer worldwide is around 500 000 new cases every year, accounting for approximately 3% of all malignancies, thus creating a significant worldwide health problem.1 The American Cancer Society estimated 40 250 new cases of these cancers for 2012 in the United States alone. Tobacco use and alcohol consumption are regarded as the main risk factors for oral squamous cell carcinoma (OSCC), while human papilloma virus (HPV) infection is emerging as the leading risk factor in cancers of the oropharynx. The most common form of oral cancer is squamous cell carcinoma (SCC), which accounts for 96% of all cancers of the oral cavity.2
The incidence of oral cancer has risen in the past decade and is usually recognized when symptomatic and at a late stage. The overall 5-year survival rates for oral cancer have remained low at approximately 50% for the past decades and have remained among the worst of all cancer death rates, considerably lower than that for colorectal, cervix and breast origin. This is in part due to the lack of training of health professionals for early detection and diagnosis. Despite significant advances in cancer treatment, early detection of oral cancer and its curable precursors remains the best way to ensure patient survival and improved quality of life.3,4
Occurrence of oral cancer is most frequently after the age of 40 years, with a peak at 60 years of age. It also affects males twice as often as females. The most common risk factors in this group are tobacco and alcohol use. Recently, several studies suggest that head and neck cancer particularly tongue cancer is increasing in young adults both nationally and internationally.5 Factors that contribute to this rise are still unknown, suspected etiologic agents include smokeless tobacco, various forms of drug abuse, environmental factors and HPV (ref. 6).
The purpose of this review article is to summarize the most common oral precancerous conditions that may progress to oral cancer. In addition, a review of the non-invasive detection techniques that are currently being marketed to aid general dentists and other health-care providers for early diagnosis of potential cancerous lesions is presented.
Common oral precancerous lesions
It is well established that oral SCC occurs as a result of several molecular and biochemical cellular alterations and changes in the underlying fibrovascular stroma including neovascularization.7 In conjunction with cellular alterations, clinical changes in the affected epithelial tissues are observed as well, known as precancerous lesions. The clinical significance of oral precancerous lesions lies in its association with malignant transformation into OSCC (refs. 8–9). Oral precancerous lesions can only be diagnosed histologically, although present as a spectrum of epithelial changes, rather than distinct categories. The World Health Organization in 2005 graded precancerous changes into mild, moderate, severe and carcinoma in situ.10 The risk of malignant transformation has been reported to be between 6.6% and 36.4%, although a recent meta-analysis indicated a rate of 12.1%.11,12
The most common precancerous lesions present clinically as white, red or a mix of white and red mucosal changes. These clinical conditions are known as leukoplakia or erythroplakia. There are other pathological conditions that are considered precancerous including oral lichen planus and oral submucous fibrosis. In addition, less common lesions include discoid lupus erythematosus and some rare hereditary conditions such as dyskeraotisis congenita and epidermolysis bullosa.3,4,6 The malignant potential of the above mentioned oral lesions cannot be accurately predicted solely on the basis of their clinical characteristics, histological evaluation is essential for all suspicious lesions. Unfortunately, histological findings only indicate that a given lesion may have malignant potential (dysplasia), and cannot be used for the prediction of malignant changes. Thus, the presence of dysplasia only indicates that an oral lesion may have an increased risk of malignant transformation. Molecular biomarkers capable of identifying the subset of lesions likely to progress to cancer are being widely investigated including genetic and epigenetic alterations observed in oral mucosal precancerous lesions.13,14,15,16,17,18,19
Oral leukoplakia: a target for early oral cancer detection
The earliest detectable morphologic changes of oral cancer are the appearance of the ‘precancerous' lesions, of which the most common ones are leukoplakia and erythroplakia. Oral leukoplakia, a white lesion in the mucosa of the oral cavity, represents the most common precursor lesion of OSCC and its prevalence varies between 0.1% and 0.5%.3 Unfortunately, the reported proportion of oral leukoplakia that develops into oral cancer varies depending on several factors including, the study population, the definition of leukoplakia used and the length of observation time, but an annual transformation rate of 1%–2% per year is a reasonable assumption.13,20 Some of the known risk factors for progression include, female gender, size of lesion and the presence and grade of dysplasia.3,4 Although criteria have been defined by the World Health Organization, it is difficult to make an objective categorization of dysplasia owing to a high inter-observer and intra-observer variation in assessment.10
Microscopically, leukoplakias exhibit either hyperplasia of keratinocytes, which is characterized by hyperorthokeratosis, hyperparakeratosis, and acanthosis or several degrees of cellular atypia, which classifies the lesion as either mild, moderate or severe dysplasia, depending on the number of atypia present and extent of the involved epithelium. The criteria for grading of oral epithelial dysplasia is universally well established and had been taught in most dental school's curriculum. If the epithelial cellular atypia invades the connective tissue then the lesion is defined as an SCC (refs. 3–4, 10). For the oral cavity, up to 20%–25% of biopsies obtained from clinically defined leukoplakias may exhibit dysplasia; lesions located in the floor of the mouth were reported up to 40% prevalence of dysplastic changes.9,11 Silverman and colleagues monitored 257 patients with oral leukoplakias, of which 22 had a diagnosis of epithelial dysplasia; eight of the 22 (36.4%) with dysplasia developed carcinoma. In their assessment, oral leukoplakia had an association with the lifetime development of oral cancer in affected individuals from 5% to 37% of the time. The time from initial diagnosis of dysplasia to carcinoma ranged from 6 months to 39 years.21,22 Surgical excision is the standard treatment of local lesions with epithelial dysplasia in the oral cavity; however, recurrence rates have been reported as high as 50%. Whether recurrence relates to continued exposure to risk factor such as smoking, or to an underlying mechanism that initiates the original lesion remains unclear.13 Proliferative verrucous leukoplakia (PVL), considered to be either a clinical subtype of non-homogeneous oral leukoplakia or to be a distinct clinical entity, is not strongly associated with smoking. It is characterized by multiple verrucous leukoplakic lesions and involves various oral sites. These lesions may progress either to verrucous carcinoma or to SCC. In most cases, PVL is recognized only late in its course since in its initial stages it is identical to an isolated leukoplakia. Unfortunately, PVL lesions have a high tendency for recurrence after excision.23
The problems with histological grading and treatment of the various forms of leukoplakia have driven investigators to assess the risk for progression and to identify targets for treatment. The presence and number of cancer-associated genetic changes have proven to be effective in discriminating leukoplakias with a low risk from those with a high risk of malignant transformation. In addition, to avoid painful biopsies, saliva and exfoliated cells are now used as a source for biomarker-based risk assessment.14,15,16
Erythroplakia
Although erythroplakia is an infrequent oral condition, its risk of malignant progression is the highest among all oral precancerous lesions. Erythroplakia is defined as ‘any lesion of the oral mucosa that presents as bright red velvety plaques which cannot be characterized clinically or pathologically as any other recognizable condition'. It can appear anywhere in the oral cavity, but it predominately occurs in the floor of the mouth, the soft palate, the ventral tongue and the tonsillar fauces. These lesions are usually asymptomatic but sometimes patients complain of a burning sensation and/or sore. Studies have shown that malignant transformation ranges from 20% to 68%.24
Heavy alcohol consumption and tobacco use are known to be important etiological factors of erythroplakia. The epithelium is often atrophic and shows lack of keratin. Sometimes hyperplasia is seen as well. The red color of erythroplakia is due to the epithelial thinness that allows the underlying microvasculature to show through. Erythroplakia needs to be treated because of its high risk of malignant transformation. Surgery, either by cold knife or by laser excision, is the recommended treatment modality. Unfortunately, there are very few data from the literature about the recurrence rate after excision of erythroplakias.20
HPV and oral precancerous lesions
HPVs are DNA viruses that can cause infection of either cutaneous or mucosal epithelium depending on their genotype.25 The ones that infect the mucosal epithelium has been categorized, depending on their oncogenic potential, as either high-risk subtypes (HPV-16, 18, 31, 33 and 35), or low-risk subtypes (HPV-6, 11, 13 and 32). Low-risk HPVs have been implicated in the pathogenesis of the benign oral proliferative epithelial lesions, squamous cell papilloma, common wart (verrucous vulgaris), condyloma acuminatum and focal epithelial hyperplasia (Heck disease), while high-risk types, in particular HPVs 16 and 18, have been associated with precancerous and cancerous oral and oropharyngeal epithelial lesions.26
The role of HPV in cervical cancer is well established and has led to the widespread use of papillomavirus vaccines for young women. In the contrary, the role of HPV in oral and oropharyngeal cancers is still being investigated; although the reported prevalence varies considerably, several studies have confirmed the presence of HPVs in oral samples such as biopsies or brush samples of mucosa.27 In addition, HPV positive normal oral mucosa have also been found in biopsies from healthy mouths, but their prevalence is typically reported to be higher in biopsies from oral lesions such as leukoplakia or cancers. High-risk HPV genotypes, in particular HPV-16, are the most prevalent in oral leukoplakias, including PVL (ref. 25). The role of HPV in the pathogenesis of oral leukoplakia and in its progression to carcinoma is unclear; there is a low viral load in HPV-cytopositive precancerous and cancerous oral lesions, and viral integration is seldom found. But the presence of HPV positive cells in orophayrhngeal carcinoma has been well documented.28,29
The course of malignant transformation in cells infected with oncogenic HPV types is incompletely understood, but some components are known.30 Virally encoded E6 binds simultaneously to the ubiquitin/protein ligase E6AP and p53, resulting in ubiquitination of p53 and its subsequent proteolytic degradation. Although the incidence of tobacco- and alcohol-induced cancers is declining, there are solid indications that the incidence as well as the prevalence of HPV-associated head and neck squamous cell carcinomas is increasing.31
Future studies are needed to investigate the outcomes of HPV-related oropharyngeal dysplastic lesions and evaluate potential screening methods to detect oropharyngeal dysplasia prior to cancer progression. Successful screening measures used for cervical cancer screening such as a Papanicolaou test, HPV polymerase chain reaction testing, or both may be difficult to achieve, but there is meaningful hope that prevention efforts will reduce the burden of HPV-related oropharyngeal cancer.32
The available HPV vaccines have demonstrated significant ability to prevent infection and dysplasia at anogenital sites; unfortunately, data regarding oral HPV infection are lacking. Studies have to be initiated to determine whether the prevalence of oral carcinomas declines along with the expected decline in cervical cancer in women who receive the HPV vaccines at a young age. However, it will take decades before any potential benefit of HPV vaccination in reducing the rates of HPV-related oropharyngeal carcinomas is confirmed.27
Oral submucous fibrosis
Oral submucous fibrosis is a chronic, debilitating disease characterized by inflammation and progressive fibrosis of the submucosal tissues (lamina propria and deeper connective tissues), resulting in marked rigidity and an eventual inability to open the mouth. The buccal mucosa is the most commonly involved site, but any part of the oral cavity can be involved, even the pharynx. The condition is well recognized for its malignant potential and is particularly associated with areca nut chewing, the main component of betel quid.33 The mixture of this quid or chew is a combination of the areca nut (fruit of the Areca catechu palm tree, erroneously termed betel nut) and betel leaf (from the Piper betel, a pepper shrub), tobacco, slaked lime (calcium hydroxide) and catechu (extract of the Acacia catechu tree).34 The pathogenesis of oral submucous fibrosis is not well established and believed to be multifactorial. The cascade begins with a juxtaepithelial inflammatory reaction in the oral mucosa, interspersed with healing and fibrosis, with trismus being the end result.35
Oral lichen planus
Oral lichen planus (OLP) is a chronic inflammatory oral mucosal disease of unknown etiology. OLP typically presents as white striations, white papules or white plaques. Other more symptomatic clinical presentations include erythema, erosions, or blisters, which are generally very painful. OLP affect predominantly the buccal mucosa, tongue and gingiva, although other sites are occasionally involved. OLP affects 1%–2% of the general adult population and is the most common non-infectious oral mucosal disease. The potential malignant transformation of OLP has been a lasting controversial matter. Several retrospective studies have observed a higher incidence of oral cancer in patients with a specific subtype of OLP, mainly erosive OLP ranging from 2% to 8% which represents a greater risk of oral cancer than in the general population. In this regard, OLP should be considered a potentially malignant condition.36
Even though the World Health Organization classified OLP as a precancerous lesion,10 the premalignant or malignant potential of OLP continues to be the subject of an ongoing and controversial debate. Critics contend that the association between OLP and OSCC is due to the misdiagnosis of dysplastic epithelial lesion as OLP, especially as it is often difficult to differentiate one from the other. A recommendation was therefore made to differentiate carefully between OLP lesions with dysplastic lesions.37,38
Lesions that are clinically indistinguishable from OLP but have a distinct etiology are known as oral lichenoid lesions (OLLs), these represent a common end point in response to extrinsic agents (drugs, allergens), altered self-antigens or superantigens.39 These lesions share common clinical and histopathological features with OLP and should be considered oral precancerous conditions when they do not resolve after the removal of the causative factor. Since OLLs are considered to be precancerous, a recall system for patients with OLL might be useful to facilitate the early diagnosis of oral cancer with the aim of reducing morbidity and mortality from oral cancer arising in OLL patients.39
The molecular mechanisms underlying OLP progression to OSCC remains largely unknown. Further investigation of the genetic/protein alterations in OLP and associated OSCC is warranted, which will lead to a more complete understanding of the malignant potential of OLP and help diagnose and treat OLP-associated OSCC patients at early stage.40
Importance of early detection
There is general consensus that the clinical stage at the time of diagnosis is the most important predictor of recurrence and mortality in oral cancer patients. The time to diagnosis is influenced by multiple clinical and sociodemographic variables, including patient reluctance to consult a health-care professional due to lack of access to health care, especially in patients with low socioeconomic status, as well as professional delay in diagnosing and treating the disease.40 Studies have shown that dentists and other health-care providers are in desperate need of systemic educational updates in oral cancer prevention and early detection, as they are remiss in the provision of oral examinations and in the detection of early oral cancers. Clinicians can improve patients' survival rates if a cancerous lesion is detected at an early stage, or if a precursor lesion (dysplasia) is discovered and treated prior to malignant progression.41 A major challenge for early diagnosis of the at-risk tissue is our limited ability to differentiate oral precancerous lesions at high risk of progressing into invasive SCC from those at low risk.42 Thus, the prevention of oral cancer and its associated morbidity and mortality, hinges upon the early detection of oral precancerous lesions, allowing for histological evaluation and subsequent treatment depending on the stage of diagnosis. Early detection and screening for oral cancer has the potential to decrease the morbidity and mortality of disease, but methods for screening have not been proven successful. Although a typical routine oral cancer examination requires a 90-s visual and tactile examination, too few practitioners and dentists in particular are conducting these exams.43
Non-invasive tools for early detection
Recent advancements in oral cancer research have led to the development of potentially useful diagnostic tools at the clinical and molecular level for the early detection of oral cancer. The gold standard for oral cancer diagnosis remains tissue biopsy with histological assessment, but this technique needs a trained health-care provider, and is considered invasive, painful, expensive and time consuming.43 Recent clinical diagnostic tools for early detection of oral cancer include tolonium chloride or toluidine blue dye, Oral CDx brush biopsy kits, salivary diagnostics and lastly optical imaging systems. Depending on the type of light and the imaging approaches used, optical imaging of the oral tissues can detect minimal changes within the tissues, such as alterations in tissue architecture and composition; expression of specific biomarkers, vascularity/angiogenesis and perfusion; microanatomy and tissue boundary integrity (e.g., potential invasiveness of lesions).44,45
The most common ones that have been marketed to dentists include: ViziLite (Zila, Batesville, AR, USA), VELscope (LED Dental Inc., Vancouver, Canada), DIFOTI (Electro-Optical Sciences, Inc., Irvington, NY, USA) and Identafi 3000 (DentalEZ, Bay Minette, AL, USA). All these methods have their own advantages and disadvantages but unfortunately these non-invasive tools have failed in their practical implication in the community setup, as patients are still being diagnosed in advanced stages of oral cancer.46,47
Toluidine blue
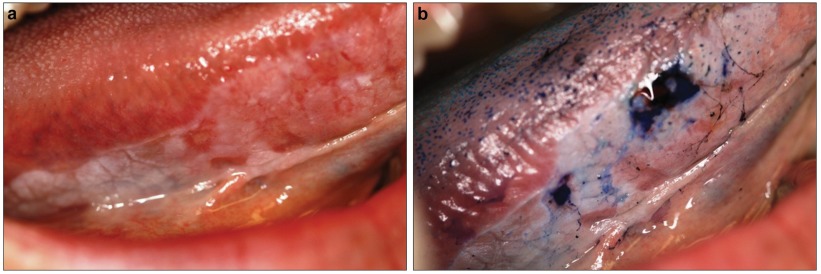
Toluidine blue (TB) is a member of the thiazine group of metachromatic dyes, which binds to DNA and is partially soluble both in water and in alcohol. Theoretically, dysplastic and malignant cells have higher nucleic acid content than normal, and thus, staining of suspicious lesions with this dye can aid recognition of mucosal changes. TB has been used as a vital stain to highlight potentially malignant oral lesions since the early 1980s. A positive staining of TB may appear as a dark royal blue (Figure 1). TB test appears to be highly sensitive (97.8%–93.5%) but less specific (92.9%–73.3%), mainly because of high false-positive results.48,49 Recently, molecular studies on TB stained lesions reported a link between carcinoma and loss of heterozygosity at 3p and 17p, while dysplasia resulted in loss of heterozygosity at 9p. The presence of loss of heterozygosity has also been reported in high frequency of TB-stained lesions without or with low grade dysplasia.50
Figure 1.
TB stain on a suspicious lesion at the right lateral border of the tongue. (a) An erythroplakia lesion on right lateral border of the tongue of a 52-year-old female. (b) Same lesion after application of the TB stain. TB was retained in some areas and not others. A biopsy was taken from the dark stained blue area which showed a well differentiated squamous cell carcinoma lesion. TB, toluidine blue.
Oral CDx
Oral CDx brush biopsy uses the concept of exfoliative cytology to provide a cytological evaluation of a cellular dysplastic changes. The oral CDx provides a complete transepithelial sample as the brush extends deep in the epithelial layers. The oral cytological epithelial samples are fixed onto a glass slide, stained with a modified Papanicolaou test and analyzed microscopically via a computer-based imaging system. However, although exfoliative cytology and brush biopsy techniques are helpful in establishing a more definitive diagnosis of already visible lesions, they are of no value in detecting mucosal changes that are not readily visible to the naked eye.51,52 Advances in the development of automated cytomorphometric methods combined with genetic and proteomic profiling may provide the required tools to refine screening strategies in the future.53,54 A scalpel biopsy is still suggested if there is clinical suspicion of a lesion regardless of the Oral CDx result.
Chemiluminescence: ViziLite
This imaging device has been approved for use in the United States by the Food and Drug Administration since November 2001. It involves the use of a hand-held, single-use, disposable chemiluminescent light stick that emits light at 430, 540 and 580 nm wavelengths. The use of the light stick is intended to improve the visual distinction between normal mucosa and oral white lesions. Normal epithelium will absorb light and appear dark whereas hyperkeratinized or dysplastic lesions appear white. The difference in color could be related to altered epithelial thickness, or to the higher density of nuclear content and mitochondrial matrix that preferentially reflect light in the pathological tissues.55,56 Lately, a combination of both TB and ViziLite systems (ViziLite Plus with TBlue System; Zila, Batesville, AR, USA), received Food and Drug Administration clearance as an adjunct to visual examination of the oral cavity. A recent study of high risk patients showed that the majority of lesions with a histological diagnosis of dysplasia or carcinoma in situ were detected and mapped using ViziLite with TB (ref. 57). Another new chemiluminescence device (MicroLux DL; Zila, Batesville, AR, USA) has been introduced as an adjunct tool for oral lesion identification but few studies have been published to assess its effectiveness in detecting precancerous lesions.58
VELscope System
The use of tissue autofluorescence in the screening and diagnosis of precancerous lesions in the lung, uterine cervix and skin has been well documented. This approach is already in clinical use in the lung, and its mechanism of action and interaction of tissue autofluorescence has been well described in the cervix.59,60
Using the tissue autofluorescence concept for diagnosis of dysplastic lesions in the oral cavity hinges on the changes in the structure and metabolism of the epithelium and the subepithelial stroma when interacting with light. Specifically, loss of autofluorescence in dysplastic and cancerous tissue is believed to reflect a complex mixture of alterations to intrinsic tissue fluorophore distribution, due to tissue remodeling such as the breakdown of the collagen matrix and elastin composition as well as alterations to metabolism such as the decrease in flavin adenine dinucleotide concentration, and increase the reduction form of nicotinamide adenine dinucleotide associated with progression of the disease.61,62,63 Further, these structural changes in tissue morphology are associated with alterations not only in the epithelium but also in the lamina propria (e.g., thickening of the epithelium, hyperchromatin and increased cellular/nuclear pleomorphism, or increased microvascularity). The latter changes lead to increased absorption and/or scattering of light, which in turn reduces and modifies the detectable autofluorescence signal.64
In the past decade, several forms of autofluorescence technology have been developed for inspection of the oral mucosa. In partnership with the British Columbia Cancer Agency, LED Medical Diagnostics Inc markets the hand-held VELscope System. It is a simple hand-held fluorescence visualization tool for the direct visualization of tissue fluorescence, and it is quick and easy to use. The site of interest is viewed through the instrument eye piece. Normal oral mucosa appears pale green due to the tissue autofluorescence resulting from stimulation with intense blue light excitation at 400–460 nm wavelength. In contrast, dysplastic and malignant lesions will appear darker than the surrounding healthy tissues as they have decreased autofluorescence.65,66,67
Two recent studies emphasized the controversial use of this system for early diagnosis. One study, demonstrated that VELscope examination did not provide a definitive diagnosis regarding the presence of epithelial dysplasia, and that loss of autofluorescence is not useful in diagnosing epithelial dysplasia without relevant clinical interpretation.68 While the other study showed that the VELscope was useful in confirming the presence of oral leukoplakia and erythroplakia and other oral mucosal disorders, but the device was unable to discriminate high-risk from low-risk lesions.69
Identafi 3000
The Identafi 3000 technology combines anatomical imaging with fluorescence, fiber optics and confocal microscopy to map and delineate precisely the lesion in the area being screened. The advantage of this device over the Velscope is its small size and easy accessibility to all tissues in the oral cavity.70 Besides detection of autoflurescence similar to the Velscope system (Figure 2b), this device also examines tissue reflectance which is based on the premise of detecting changes in angiogenesis with green-amber light (540- to 575-nm wavelength) illumination (Figure 2c). The amber light is thought to enhance the reflective properties of the oral mucosa, allowing a distinction between normal and abnormal tissue vasculature. Increased angiogenesis is a known process during oral carcinogenesis and oral cancer progression.71,72 It is important to develop imaging technology for evaluating the status of tumor angiogenesis.
Figure 2.
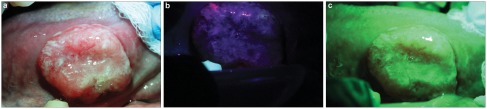
The use of Identafi optical system on a suspicious lesion showing loss of fluorescence and increase vascularity. (a) Picture of a non-healing ulcer on the right lateral border of the tongue of a 62-year-old male taken with the white reflectance (regular light) Identafi 3000 DentalEZ optical device. (b) Application of the Identafi DentalEZ device with violet fluorescent light shows dark area (loss of autoflurescence) in suspicious areas. (c) Application of the Identafi DentalEZ with the green amber reflectance light show increase vascularity in the suspicious areas. A biopsy taken from this area showed a moderately differentiated squamous cell carcinoma.
A recent study using the Identafi 3000 for screening of 124 subjects, demonstrated a sensitivity of 82% and a specificity of 87% in differentiating between neoplastic and non-neoplastic oral conditions. Results appeared to vary between sampling depths, and keratinized vs. non-keratinized tissues.73 Another study using quantitative fluorescence imaging in 56 patients with oral lesions and 11 normal volunteers, showed that healthy tissue could be discriminated from dysplasia and invasive cancer with 95.9% sensitivity and 96.2% specificity in the training set, and with 100% sensitivity and 91.4% specificity in the validation set.74 Further clinical studies are needed in diverse populations to evaluate fully the clinical usefulness of this promising technology.75
Saliva as a diagnostic tool
Saliva from patients has been used in a novel way to provide molecular biomarkers for oral cancer detection. Saliva is a mirror of the body, reflecting virtually the entire spectrum of normal and disease states and its use as a diagnostic fluid meets the demands for an inexpensive, non-invasive and accessible diagnostic tool. Discovery of analytes in saliva of normal and diseased subjects suggests a very promising function of saliva as a local and systematic diagnostic tool.76,77 The ability to analyze saliva to monitor health and disease is a highly desirable goal for oral health promotion and research. So far, saliva has been used to detect caries risk, periodontitis, oral cancer, breast cancer, salivary gland diseases and systemic disorders such as human immunodefficiency virus and hepatitis C virus.78 However, due to lack of knowledge of disease markers and an overall low concentration of these markers in saliva when compared to serum, the diagnostic value of saliva has not been fully realized. However, nowadays, highly sensitive and high-throughput assays such as DNA microarray, mass spectrometry and nanoscale sensors can measure protein and RNA markers at low concentrations in saliva, thus expanding the utility of saliva as a diagnostic tool.79,80
Conclusion and future directions
Dentists' knowledge and education in detecting oral cancer at its precancerous phase is the key to prevent its progression to later stages. In order to improve early detection, it is imperative to increase the health-care providers' depth of knowledge about oral cancer, their risk factors and the most common oral precancerous conditions. Future research can also be directed towards establishing appropriate clinical practice standards for early detection exams. Currently, the new innovative visual-based techniques show promising results, but lack strong evidence to support their effectiveness in early detection. Their utilization in clinic practice is still anecdotal. Limitations that hinder their wide use include lack of methodologically sound clinical trials, their correlation with histological alterations and the impact these techniques have on a patient's survival and risk of disease recurrence.
Acknowledgments
Publication of this manuscript is supported by Open Fund of State Key Laboratory of Oral Diseases, Sichuan University.
References
- Johnson NW, Warnakulasuriya S, Gupta PC, et al. Global oral health inequalities in incidence and outcomes for oral cancer: causes and solutions. Adv Dent Res. 2011;23 2:237–246. doi: 10.1177/0022034511402082. [DOI] [PubMed] [Google Scholar]
- Siegel R, Naishadham D, Jemal A. Cancer statistics. CA Cancer J Clin. 2012;62 1:10–29. doi: 10.3322/caac.20138. [DOI] [PubMed] [Google Scholar]
- Napier SS, Speight PM. Natural history of potentially malignant oral lesions and conditions: an overview of the literature. J Oral Pathol Med. 2008;7 1:1–10. doi: 10.1111/j.1600-0714.2007.00579.x. [DOI] [PubMed] [Google Scholar]
- van der Waal I. Potentially malignant disorders of the oral and oropharyngeal mucosa; terminology, classification and present concepts of management. Oral Oncol. 2009;45 4/5:317–323. doi: 10.1016/j.oraloncology.2008.05.016. [DOI] [PubMed] [Google Scholar]
- Llewellyn CD, Johnson NW, Warnakulasuriya KA. Risk factors for squamous cell carcinoma of the oral cavity in young people—a comprehensive literature review. Oral Oncol. 2001;37 5:401–418. doi: 10.1016/s1368-8375(00)00135-4. [DOI] [PubMed] [Google Scholar]
- Lingen MW, Pinto A, Mendes RA, et al. Genetics/epigenetics of oral premalignancy: current status and future research. Oral Diseases. 2011;17 Suppl 1:7–22. doi: 10.1111/j.1601-0825.2011.01789.x. [DOI] [PubMed] [Google Scholar]
- Albo D, Granick MS, Jhala N, et al. The relationship of angiogenesis to biological activity in human squamous cell carcinomas of the head and neck. Ann Plast Surg. 1994;32 6:588–594. doi: 10.1097/00000637-199406000-00005. [DOI] [PubMed] [Google Scholar]
- Guillaud M, Zhang L, Poh C, et al. Potential use of quantitative tissue phenotype to predict malignant risk for oral premalignant lesions. Cancer Res. 2008;68 9:3099–3107. doi: 10.1158/0008-5472.CAN-07-2113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ho PS, Chen PL, Warnakulasuriya S, et al. Malignant transformation of oral potentially malignant disorders in males: a retrospective cohort study. BMC Cancer. 2009;9:260–267. doi: 10.1186/1471-2407-9-260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnes L, Eveson JW, Reichart P, et al. World health organization. Classification of tumours. Pathology and genetics of head and neck tumours. Lyon; IARC Press; 2005. pp. 283–328. [Google Scholar]
- Arduino PG, Surace A, Carbone M, et al. Outcome of oral dysplasia: a retrospective hospital-based study of 207 patients with a long follow-up. J Oral Pathol Med. 2009;38 6:540–544. doi: 10.1111/j.1600-0714.2009.00782.x. [DOI] [PubMed] [Google Scholar]
- Mehanna HM, Rattay T, Smith J, et al. Treatment and follow-up of oral dysplasia—a systematic review and meta-analysis. Head Neck. 2009;31 12:1600–1609. doi: 10.1002/hed.21131. [DOI] [PubMed] [Google Scholar]
- Leemans CR, Boudewijn JM, Braakhuis JM, et al. The molecular biology of head and neck cancer. Nat Rev Cancer. 2011;11 1:9–22. doi: 10.1038/nrc2982. [DOI] [PubMed] [Google Scholar]
- Bremmer JF, Braakhuis BJ, Brink J, et al. A noninvasive genetic screening test to detect oral preneoplastic lesions. Lab Invest. 2005;85 12:1481–1488. doi: 10.1038/labinvest.3700342. [DOI] [PubMed] [Google Scholar]
- Bremmer JF, Graveland AP, Braakhuis BJ, et al. Screening for oral precancer with noninvasive genetic cytology. Cancer Prev Res. 2009;2 1:128–133. doi: 10.1158/1940-6207.CAPR-08-0128. [DOI] [PubMed] [Google Scholar]
- Shpitzer T, Hamzany Y, Bahar G, et al. Salivary analysis of oral cancer biomarkers. Br J Cancer. 2009;101 7:1194–1198. doi: 10.1038/sj.bjc.6605290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Duan Y. Saliva: a potential media for disease diagnostics and monitoring. Oral Oncol. 2012;48 7:569–577. doi: 10.1016/j.oraloncology.2012.01.021. [DOI] [PubMed] [Google Scholar]
- Mehrotra R, Gupta DK. Exciting new advances in oral cancer diagnosis: avenues to early detection. Head Neck Oncol. 2011;3:33–40. doi: 10.1186/1758-3284-3-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lange CJ. Exploring biomarkers in head and neck cancer. Cancer. 2012;118 16:3882–3892. doi: 10.1002/cncr.26718. [DOI] [PubMed] [Google Scholar]
- Carnelio S, Rodrigues SJ, Shenoy R, et al. A brief review of common oral premalignant lesions with emphasis on their management and cancer prevention. Indian J Surg. 2011;73 4:256–261. doi: 10.1007/s12262-011-0286-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverman S, Jr, Gorsky M, Lozada F. Oral leukoplakia and malignant transformation. A follow-up study of 257 patients. Cancer. 1984;53 3:563–568. doi: 10.1002/1097-0142(19840201)53:3<563::aid-cncr2820530332>3.0.co;2-f. [DOI] [PubMed] [Google Scholar]
- Silverman S., Jr Demographics and occurrence of oral and pharyngeal cancers: the outcomes, the trends, the challenge. J Am Dent Assoc. 2001;132 Suppl 1:7S–11S. doi: 10.14219/jada.archive.2001.0382. [DOI] [PubMed] [Google Scholar]
- Cabay JR, Morton AH, Jr, Epstein JB. Proliferative verrucous leukoplakia and its progression to oral carcinoma: a review of the literature. J Oral Pathol Med. 2007;36 5:255–261. doi: 10.1111/j.1600-0714.2007.00506.x. [DOI] [PubMed] [Google Scholar]
- Villa A, Villa C, Abati S. Oral cancer and oral erythroplakia: an update and implication for clinicians. Aust Dent J. 2011;56 3:253–256. doi: 10.1111/j.1834-7819.2011.01337.x. [DOI] [PubMed] [Google Scholar]
- Feller L, Lemmer J. Oral leukoplakia as it relates to HPV infection: a review. Int J Dent. 2012;2012:540561. doi: 10.1155/2012/540561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grinde B, Olsen I. The role of viruses in oral disease. J Oral Microbiol. 2010;2:2127–2135. doi: 10.3402/jom.v2i0.2127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D'Souza G, Dempsey A. The role of HPV in head and neck cancer and review of the HPV vaccine. Prev Med. 2011;53 Suppl 1:S5–S11. doi: 10.1016/j.ypmed.2011.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ostwald C, Rutsatz K, Schweder JW, et al. Human papillomavirus 6/11, 16 and 18 in oral carcinomas and benign oral lesions. Med Microbiol Immunol. 2003;192 3:145–148. doi: 10.1007/s00430-002-0161-y. [DOI] [PubMed] [Google Scholar]
- Hennessey PT, Westra HW, Califano JA. Human papillomavirus and head and neck squamous cell carcinoma: recent evidence and clinical implications. J Dent Res. 2009;88 4:300–306. doi: 10.1177/0022034509333371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wise-Draper TM, Wells SI. Papillomavirus E6 and E7 proteins and their cellular targets. Front Biosci. 2008;13:1003–1017. doi: 10.2741/2739. [DOI] [PubMed] [Google Scholar]
- Nasman A, Attner P, Hammarstedt L, et al. Incidence of human papillomavirus (HPV) positive tonsillar carcinoma in Stockholm, Sweden: an epidemic of viral-induced carcinoma. Int J Cancer. 2009;125 2:362–366. doi: 10.1002/ijc.24339. [DOI] [PubMed] [Google Scholar]
- Schlecht HP. Oral human papillomavirus infection hazard of intimacy. J Am Med Assoc. 2012;307 7:724–725. doi: 10.1001/jama.2012.117. [DOI] [PubMed] [Google Scholar]
- Ho PS, Yang YH, Shieh TY, et al. Consumption of areca quid, cigarettes, and alcohol related to the comorbidity of oral submucous fibrosis and oral cancer. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104 5:647–652. doi: 10.1016/j.tripleo.2006.12.032. [DOI] [PubMed] [Google Scholar]
- Shahid RA. Coming to America: betel nut and oral submucous fibrosis. J Am Dent Assoc. 2010;141 4:423–428. doi: 10.14219/jada.archive.2010.0194. [DOI] [PubMed] [Google Scholar]
- Bhattacharyya I, Chehal HK. White lesions. Otolaryngol Clin N Am. 2011;44 1:109–131. doi: 10.1016/j.otc.2010.09.009. [DOI] [PubMed] [Google Scholar]
- Sugerman PB, Savage NW, Walsh LJ, et al. The pathogenesis of oral lichen planus. Crit Rev Oral Biol Med. 2002;13 4:350–365. doi: 10.1177/154411130201300405. [DOI] [PubMed] [Google Scholar]
- Sugerman PB, Savage NW, Zhou X, et al. Oral lichen planus. Clin Dermatol. 2000;18 5:533–539. doi: 10.1016/s0738-081x(00)00142-5. [DOI] [PubMed] [Google Scholar]
- Le Cleach L, Chosidow O. Clinical practice. Lichen planus. N Engl J Med. 2012;366 8:723–732. doi: 10.1056/NEJMcp1103641. [DOI] [PubMed] [Google Scholar]
- van der Waal I. Oral lichen planus and oral lichenoid lesions; a critical appraisal with emphasis on the diagnostic aspects. Med Oral Patol Oral Cirt Bucal. 2009;14 7:E310–E314. [PubMed] [Google Scholar]
- Ho MW, Risk JM, Woolger JA, et al. The clinical determinants of malignant transformation in oral epithelial dysplasia. Oral Oncol. 2012;48 10:969–976. doi: 10.1016/j.oraloncology.2012.04.002. [DOI] [PubMed] [Google Scholar]
- Messadi DV, Wilder-Smith P, Wolinsky L. Improving oral cancer survival: the role of dental providers. J Calif Dent Assoc. 2009;37 11:789–798. [PMC free article] [PubMed] [Google Scholar]
- Steele TO, Meyers A. Early detection of premalignant lesions and oral cancer. Otolaryngol Clin N Am. 2011;44 7:221–229. doi: 10.1016/j.otc.2010.10.002. [DOI] [PubMed] [Google Scholar]
- Nair D, Pruthy R, Pawar U, et al. Oral cancer: premalignant conditions and screening—an update. J Can Res Therap. 2012;8 Suppl 1:s57–s66. doi: 10.4103/0973-1482.92217. [DOI] [PubMed] [Google Scholar]
- Fedele S. Diagnostic aids in the screening of oral cancer. Head Neck Oncol. 2009;1:5–10. doi: 10.1186/1758-3284-1-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epstein JB, Silverman S, Jr, Epstein JD, et al. Analysis of oral lesion biopsies identified and evaluated by visual examination, chemiluminescence and toluidine blue. Oral Oncol. 2008;44 6:538–544. doi: 10.1016/j.oraloncology.2007.08.011. [DOI] [PubMed] [Google Scholar]
- Lingen MW, Kalmar JR, Karrison T, et al. Critical evaluation of diagnostic aids for the detection of oral cancer. Oral Oncol. 2008;44 1:10–22. doi: 10.1016/j.oraloncology.2007.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosin MP, Poh CF, Guillard M, et al. Visualization and other emerging technologies as change makers for oral cancer prevention. Ann NY Acad Sci. 2007;1098:167–183. doi: 10.1196/annals.1384.039. [DOI] [PubMed] [Google Scholar]
- Patton LL, Epstein JB, Kerr AR. Adjunctive techniques for oral cancer examination and lesion diagnosis: a systematic review of the literature. J Am Dent Assoc. 2008;139 7:896–905. doi: 10.14219/jada.archive.2008.0276. [DOI] [PubMed] [Google Scholar]
- Su WW, Yen AM, Chiu SY, et al. A community-based RCT for oral cancer screening with toluidine blue. J Dent Res. 2010;89 9:933–937. doi: 10.1177/0022034510373763. [DOI] [PubMed] [Google Scholar]
- Awan KH, Yang YH, Morgan PR, et al. Utility of toluidine blue as a diagnostic adjunct in the detection of potentially malignant disorders of the oral cavity—a clinical and histological assessment. Oral Dis. 2012;18 8:728–733. doi: 10.1111/j.1601-0825.2012.01935.x. [DOI] [PubMed] [Google Scholar]
- Sciubba JJ. Improving detection of precancerous and cancerous oral lesions. Computer-assisted analysis of the oral brush biopsy. U.S. Collaborative OralCDx Study Group. J Am Dent Assoc. 1999;130 10:1445–1457. doi: 10.14219/jada.archive.1999.0055. [DOI] [PubMed] [Google Scholar]
- Rick GM. Oral brush biopsy: the problem of false positives. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;96 3:252–258. doi: 10.1016/s1079-2104(03)00362-7. [DOI] [PubMed] [Google Scholar]
- Svirsky JA, Burns JC, Carpenter WM, et al. Comparison of computer-assisted brush biopsy results with follow up scalpel biopsy and histology. Gen Dent. 2002;50 6:500–503. [PubMed] [Google Scholar]
- Scheifele C, Schimdt-Westhausen A, Detrich T, et al. The sensitivity and specificity of the OralCDx technique: evaluation of 103 cases. Oral Oncol. 2004;40 8:824–828. doi: 10.1016/j.oraloncology.2004.02.004. [DOI] [PubMed] [Google Scholar]
- Huber MA, Bsoul SA, Terezhalmy GT. Acetic acid wash and chemiluminescent illumination as an adjunct to conventional oral soft tissue examination for the detection of dysplasia: a pilot study. Quintessence Int. 2004;35 5:378–384. [PubMed] [Google Scholar]
- Kerr AR, Sirois DA, Epstein JB. Clinical evaluation of chemiluminescent lighting: an adjunct for oral mucosal examinations. J Clin Dent. 2006;17 3:59–63. [PubMed] [Google Scholar]
- Epstein JB, Gorsky M, Lonky S, et al. The efficacy of oral lumenoscopy™ (ViziLite®) in visualizing oral mucosal lesions. Spec Care Dent. 2006;26 4:171–174. doi: 10.1111/j.1754-4505.2006.tb01720.x. [DOI] [PubMed] [Google Scholar]
- McIntosh L, McCullough MJ, Farah CS. The assessment of diffused light illumination and acetic acid rinse (Microlux/DL) in the visualisation of oral mucosal lesions. Oral Oncol. 2009;45 12:227–231. doi: 10.1016/j.oraloncology.2009.08.001. [DOI] [PubMed] [Google Scholar]
- Lam S, MacAulay C, Palcic B. Detection and localization of early lung cancer by imaging techniques. Chest. 1993;103 1 Suppl:12S–14S. doi: 10.1378/chest.103.1_supplement.12s. [DOI] [PubMed] [Google Scholar]
- Park SY, Follen M, Milbourne A, et al. Automated image analysis of digital colposcopy for the detection of cervical neoplasia. J Biomed Opt. 2008;13 1:014029. doi: 10.1117/1.2830654. [DOI] [PubMed] [Google Scholar]
- Kennedy JC, Pottier RH. Endogenous protoporphyrin IX, a clinically useful photosensitizer for photodynamic therapy. J Photochem Photobiol B. 1992;14 4:275–292. doi: 10.1016/1011-1344(92)85108-7. [DOI] [PubMed] [Google Scholar]
- Ebihara A, Liaw LH, Krasieva TB. Detection and diagnosis of oral cancer by light-induced fluorescence. Lasers Surg Med. 2003;32 1:17–24. doi: 10.1002/lsm.10137. [DOI] [PubMed] [Google Scholar]
- Schantz SP, Kolli V, Savage HE, et al. In vivo native cellular fluorescence and histological characteristics of head and neck cancer. Clin Cancer Res. 1998;4 5:1177–1182. [PubMed] [Google Scholar]
- Sweeny L, Dean NR, Magnuson JS, et al. Assessment of tissue autofluorescence and reflectance for oral cavity cancer screening. Otolaryngol Head Neck Surg. 2012;145 6:956–960. doi: 10.1177/0194599811416773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lane PM, Gilhuly T, Whitehead P, et al. Simple device for the direct visualization of oral-cavity tissue fluorescence. J Biomed Opt. 2006;11 2:024006. doi: 10.1117/1.2193157. [DOI] [PubMed] [Google Scholar]
- Poh CF, Ng SP, Williams PM, et al. Direct fluorescence visualization of clinically occult high-risk oral premalignant disease using a simple hand-held device. Head Neck. 2007;29 1:71–76. doi: 10.1002/hed.20468. [DOI] [PubMed] [Google Scholar]
- Balevi B. Evidence-based decision making: should the general dentist adopt the use of the VELscope for routine screening for oral cancer. J Can Dent Assoc. 2007;73 7:603–606. [PubMed] [Google Scholar]
- Camiles F, Mcintosh L, Georgiou A, et al. Efficacy of tissue autofluorescence imaging (Velscope) in the visualization of oral mucosal lesions. Head Neck. 2012;34 6:856–862. doi: 10.1002/hed.21834. [DOI] [PubMed] [Google Scholar]
- Awan KH, Morgan PR, Warnakulasuriya S. Evaluation of an autofluorescence based imaging system (VELscopeTM) in the detection of oral potentially malignant disorders and benign keratosis. Oral Oncol. 2011;47 4:274–277. doi: 10.1016/j.oraloncology.2011.02.001. [DOI] [PubMed] [Google Scholar]
- Roblyer D, Kurachi C, Stepanek V, et al. Objective detection and delineation of oral neoplasia using autofluorescence imaging. Cancer Prev Res. 2009;2 5:423–431. doi: 10.1158/1940-6207.CAPR-08-0229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Folkman J, Watson K, Ingber D, et al. Induction of angiogenesis during the transition from hyperplasia to neoplasia. Nature. 1989;339 6219:58–61. doi: 10.1038/339058a0. [DOI] [PubMed] [Google Scholar]
- Hasina R, Lingen MW. Angiogenesis in oral cancer. J Dent Educ. 2001;65 11:1282–1290. [PubMed] [Google Scholar]
- Schwarz RA, Gao W, Redden Weber C, et al. Noninvasive evaluation of oral lesions using depth-sensitive optical spectroscopy: simple device for the direct visualization of oral-cavity tissue fluorescence. Cancer. 2009;115 8:1669–1679. doi: 10.1002/cncr.24177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGee SA, Mirkovic J, Mardirossian V, et al. Model-based spectroscopic analysis of the oral cavity: impact of anatomy. J Biomed Opt. 2008;13 6:064034. doi: 10.1117/1.2992139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch FP, Kaemmerer PW, Biesterfeld S, et al. Effectiveness of autofluorescence to identify suspicious oral lesions-a prospective, blinded clinical trial. Clin Oral Investig. 2011;15 6:975–982. doi: 10.1007/s00784-010-0455-1. [DOI] [PubMed] [Google Scholar]
- Forde MD, Koka S, Eckert SE, et al. Systemic assessments utilizing saliva: part 1 general considerations and current assessments. Int J Prosthodont. 2006;19 1:43–52. [PubMed] [Google Scholar]
- Pesce MA, Spitalnik SL. Saliva and the clinical pathology laboratory. Ann NY Acad Sci. 2007;1098:192–199. doi: 10.1196/annals.1384.032. [DOI] [PubMed] [Google Scholar]
- Li Y, St John MA, Zhou X, et al. Salivary transcriptome diagnostics for oral cancer detection. Clin Cancer Res. 2004;10 24:8442–8450. doi: 10.1158/1078-0432.CCR-04-1167. [DOI] [PubMed] [Google Scholar]
- Wong DT. Towards a simple, saliva-based test for the detection of oral cancer, oral fluid (saliva), which is the mirror of the body, is a perfect medium to be explored for health and disease surveillance. Expert Rev Mol Diagn. 2006;6 3:267–272. doi: 10.1586/14737159.6.3.267. [DOI] [PubMed] [Google Scholar]
- Wong DT. Salivary diagnostics powered by nanotechnologies, proteomics and genomics. J Am Dent Assoc. 2006;137 3:313–321. doi: 10.14219/jada.archive.2006.0180. [DOI] [PubMed] [Google Scholar]