Abstract
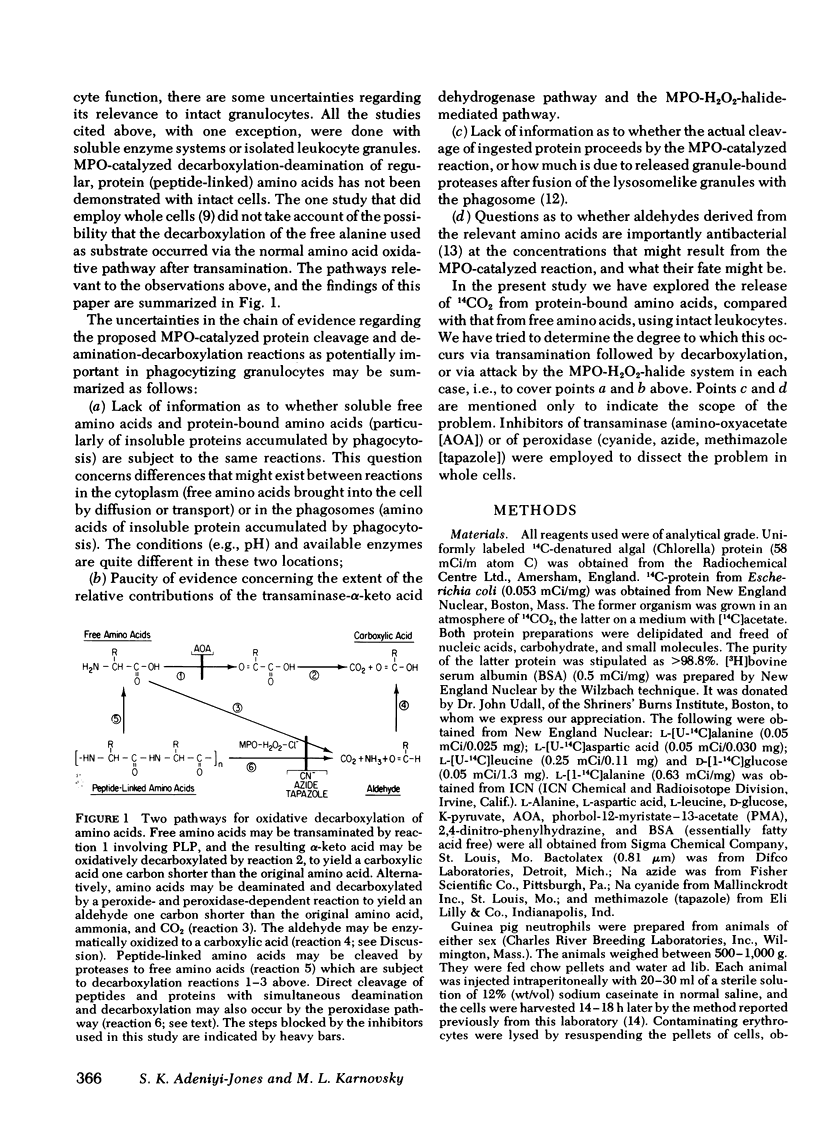
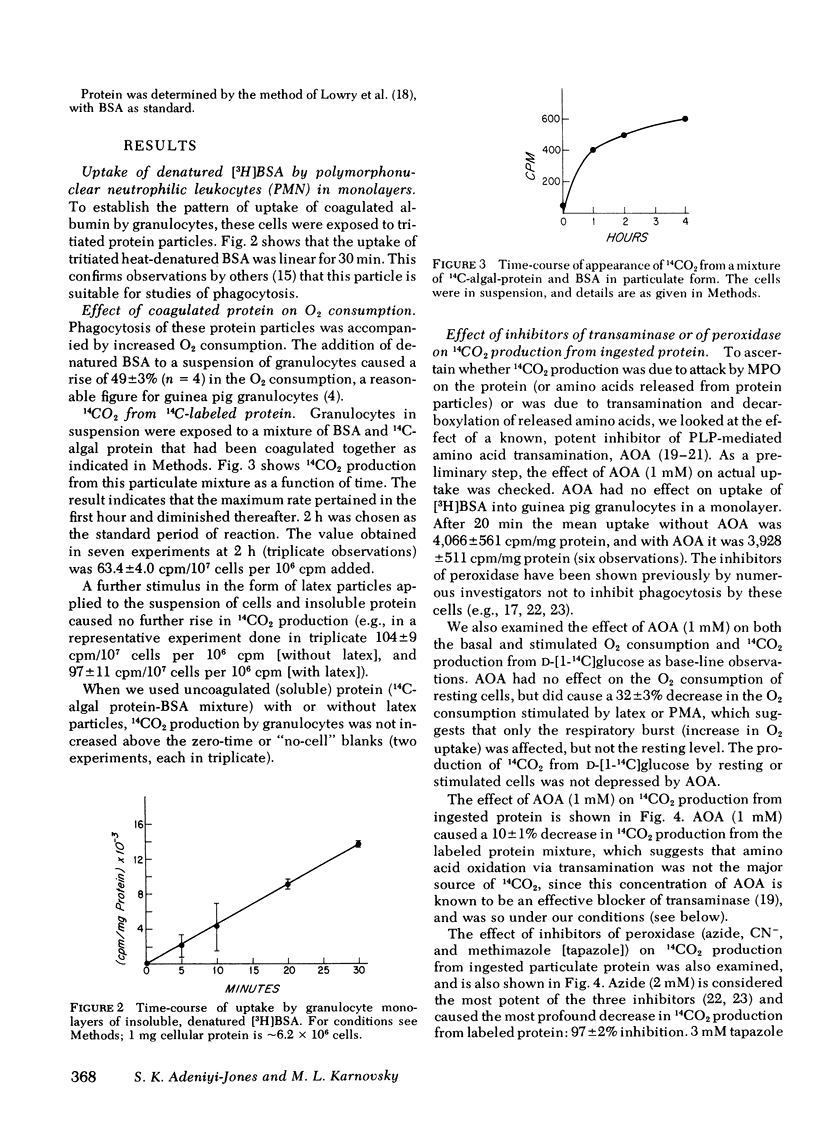
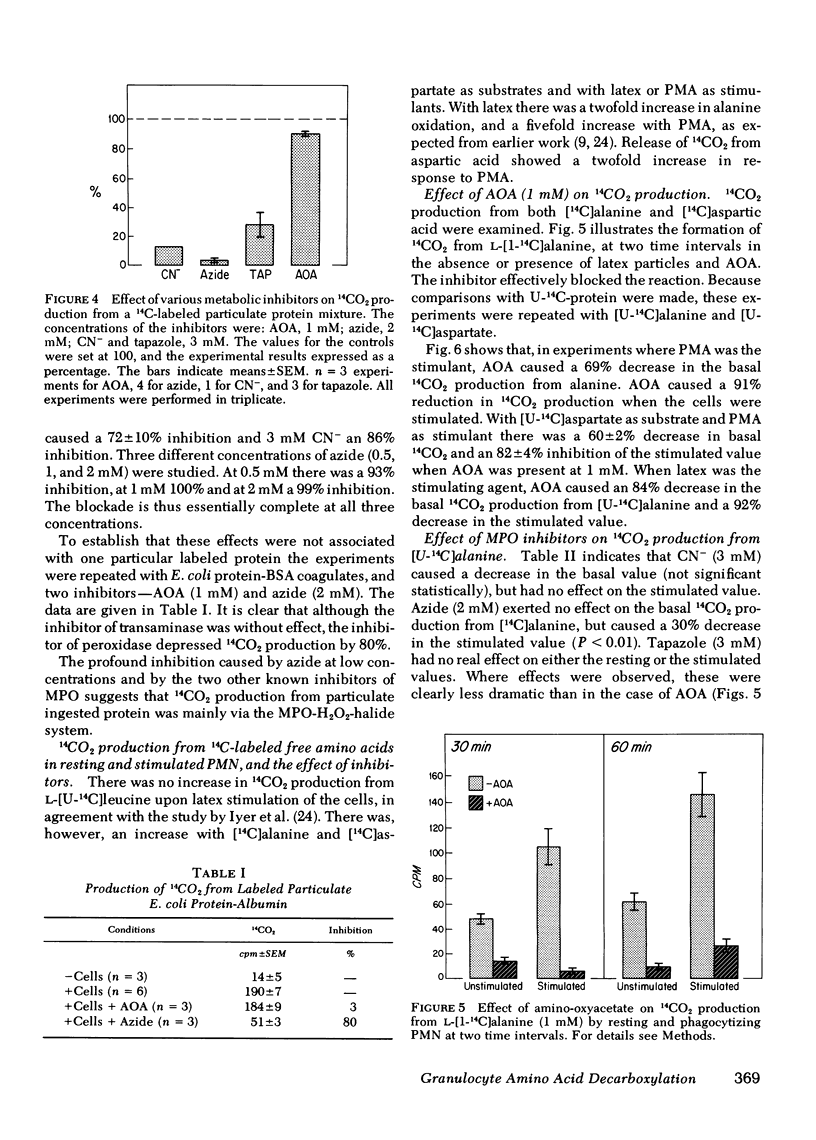
The oxidative decarboxylation of amino acids by a system consisting of myeloperoxidase-hydrogen peroxide-chloride has been demonstrated previously by others and the process has been considered to be part of the microbicidal armamentarium of some phagocytic leukocytes. We were able to translate these earlier observations, made on model systems, to intact guinea pig granulocytes. We could demonstrate differences in the cellular handling of peptide-linked amino acids as particles, compared with free amino acids. Specific inhibitors were used to explore two routes of oxidative decarboxylation: (a) the myeloperoxidase-catalyzed direct decarboxylation-deamination reaction, and (b) oxidation of alpha-keto acids after transamination of amino acids. These inhibitors were cyanide, azide, and tapazole for the former pathway, and amino-oxyacetate for the latter. Amino-oxyacetate profoundly inhibited the decarboxylation of free 14C-amino acids (alanine and aspartate) in both resting and stimulated cells, but had only a minimal effect on 14CO2 production from ingested insoluble 14C-protein. On the other hand, the peroxidase inhibitors cyanide, azide, and tapazole dramatically inhibited the production of 14CO2 from ingested particulate 14C-protein, but had only small effects on the decarboxylation of free amino acid. Soluble, uniformly labeled 14C-protein was not significantly converted to 14CO2 even in the presence of phagocytizable polystyrene beads. These observation suggest that the amino acids taken up by phagocytosis (e.g., as denatured protein particles) are oxidatively decarboxylated and deaminated in the phagosomes by the myeloperoxidase-hydrogen peroxide-chloride system; soluble free amino acids that enter the cytoplasm by diffusion or transport are oxidatively decarboxylated after transamination by the normal cellular amino acid oxidative pathway.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BENACERRAF B., BIOZZI G., HALPERN B. N., STIFFEL C., MOUTON D. Phagocytosis of heat-denatured human serum albumin labelled with 131I and its use as a means of investigating liver blood flow. Br J Exp Pathol. 1957 Feb;38(1):35–48. [PMC free article] [PubMed] [Google Scholar]
- Babior B. M. Oxygen-dependent microbial killing by phagocytes (second of two parts). N Engl J Med. 1978 Mar 30;298(13):721–725. doi: 10.1056/NEJM197803302981305. [DOI] [PubMed] [Google Scholar]
- Badwey J. A., Curnutte J. T., Robinson J. M., Lazdins J. K., Briggs R. T., Karnovsky M. J., Karnovsky M. L. Comparative aspects of oxidative metabolism of neutrophils from human blood and guinea pig peritonea: magnitude of the respiratory burst, dependence upon stimulating agents, and localization of the oxidases. J Cell Physiol. 1980 Dec;105(3):541–545. doi: 10.1002/jcp.1041050319. [DOI] [PubMed] [Google Scholar]
- Badwey J. A., Karnovsky M. L. Active oxygen species and the functions of phagocytic leukocytes. Annu Rev Biochem. 1980;49:695–726. doi: 10.1146/annurev.bi.49.070180.003403. [DOI] [PubMed] [Google Scholar]
- Badwey J. A., Robinson J. M., Karnovsky M. J., Karnovsky M. L. Superoxide production by an unusual aldehyde oxidase in guinea pig granulocytes. Characterization and cytochemical localization. J Biol Chem. 1981 Apr 10;256(7):3479–3486. [PubMed] [Google Scholar]
- Baehner R. L., Karnovsky M. J., Karnovsky M. L. Degranulation of leukocytes in chronic granulomatous disease. J Clin Invest. 1969 Jan;48(1):187–192. doi: 10.1172/JCI105967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger R. R., Karnovsky M. L. Biochemical basis of phagocytosis. V. Effect of phagocytosis on cellular uptake of extracellular fluid, and on the intracellular pool of L-alpha-glycerophosphate. Fed Proc. 1966 May-Jun;25(3):840–845. [PubMed] [Google Scholar]
- Briggs R. T., Drath D. B., Karnovsky M. L., Karnovsky M. J. Localization of NADH oxidase on the surface of human polymorphonuclear leukocytes by a new cytochemical method. J Cell Biol. 1975 Dec;67(3):566–586. doi: 10.1083/jcb.67.3.566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briggs R. T., Karnovsky M. L., Karnovsky M. J. Cytochemical demonstration of hydrogen peroxide in polymorphonuclear leukocyte phagosomes. J Cell Biol. 1975 Jan;64(1):254–260. doi: 10.1083/jcb.64.1.254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- COHN Z. A., HIRSCH J. G. The influence of phagocytosis on the intracellular distribution of granule-associated components of polymorphonuclear leucocytes. J Exp Med. 1960 Dec 1;112:1015–1022. doi: 10.1084/jem.112.6.1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DePierre J. W., Karnovsky M. L. Ecto-enzymes of the guinea pig polymorphonuclear leukocyte. I. Evidence for an ecto-adenosine monophosphatase, adenosine triphosphatase, and -p-nitrophenyl phosphates. J Biol Chem. 1974 Nov 25;249(22):7111–7120. [PubMed] [Google Scholar]
- Edelson P. J., Cohn Z. A. Peroxidase-mediated mammalian cell cytotoxicity. J Exp Med. 1973 Jul 1;138(1):318–323. doi: 10.1084/jem.138.1.318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HOPPER S., SEGAL H. L. Kinetic studies of rat liver glutamicalanine transaminase. J Biol Chem. 1962 Oct;237:3189–3195. [PubMed] [Google Scholar]
- Jacobs A. A., Paul B. B., Strauss R. R., Sbarra A. J. The role of the phagocyte in host-parasite interactions. 23. Relation of bactericidal activity to peroxidase-associated decarboxylation and deamination. Biochem Biophys Res Commun. 1970 Apr 24;39(2):284–289. doi: 10.1016/0006-291x(70)90791-6. [DOI] [PubMed] [Google Scholar]
- Jensen M. S., Bainton D. F. Temporal changes in pH within the phagocytic vacuole of the polymorphonuclear neutrophilic leukocyte. J Cell Biol. 1973 Feb;56(2):379–388. doi: 10.1083/jcb.56.2.379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeunet F. S., Good R. A. Reticuloendothelial function in the isolated perfused liver. II. Phagocytosis of heat-aggregated bovine serum albumin. Demonstration of two components in the blockade of the reticuloendothelial system. J Reticuloendothel Soc. 1969 Feb;6(1):94–107. [PubMed] [Google Scholar]
- Klebanoff S. J. Antimicrobial mechanisms in neutrophilic polymorphonuclear leukocytes. Semin Hematol. 1975 Apr;12(2):117–142. [PubMed] [Google Scholar]
- Klebanoff S. J., Hamon C. B. Role of myeloperoxidase-mediated antimicrobial systems in intact leukocytes. J Reticuloendothel Soc. 1972 Aug;12(2):170–196. [PubMed] [Google Scholar]
- Klebanoff S. J. Myeloperoxidase-halide-hydrogen peroxide antibacterial system. J Bacteriol. 1968 Jun;95(6):2131–2138. doi: 10.1128/jb.95.6.2131-2138.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Michell R. H., Pancake S. J., Noseworthy J., Karnovsky M. L. Measurement of rates of phagocytosis: the use of cellular monolayers. J Cell Biol. 1969 Jan;40(1):216–224. doi: 10.1083/jcb.40.1.216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul B. B., Jacobs A. A., Strauss R. R., Sbarra A. J. Role of the Phagocyte in Host-Parasite Interactions XXIV. Aldehyde Generation by the Myeloperoxidase-H(2)O(2)-Chloride Antimicrobial System: a Possible In Vivo Mechanism of Action. Infect Immun. 1970 Oct;2(4):414–418. doi: 10.1128/iai.2.4.414-418.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paul B., Sbarra A. J. The role of the phagocyte in host-parasite interactions. 13. The direct quantitative estimation of H2O2 in phagocytizing cells. Biochim Biophys Acta. 1968 Feb 1;156(1):168–178. doi: 10.1016/0304-4165(68)90116-5. [DOI] [PubMed] [Google Scholar]
- Rognstad R., Katz J. Gluconeogenesis in the kidney cortex. Effects of D-malate and amino-oxyacetate. Biochem J. 1970 Feb;116(3):483–491. doi: 10.1042/bj1160483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selvaraj R. J., Paul B. B., Strauss R. R., Jacobs A. A., Sbarra A. J. Oxidative peptide cleavage and decarboxylation by the MPO-H2O2-Cl- antimicrobial system. Infect Immun. 1974 Feb;9(2):255–260. doi: 10.1128/iai.9.2.255-260.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith S. B., Briggs S., Triebwasser K. C., Freedland R. A. Re-evaluation of amino-oxyacetate as an inhibitor. Biochem J. 1977 Feb 15;162(2):453–455. doi: 10.1042/bj1620453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss R. R., Paul B. B., Jacobs A. A., Sbarra A. J. Role of the Phagocyte in Host-Parasite Interactions XXVII. Myeloperoxidase-H(2)O(2)-Cl-Mediated Aldehyde Formation and Its Relationship to Antimicrobial Activity. Infect Immun. 1971 Apr;3(4):595–602. doi: 10.1128/iai.3.4.595-602.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss R. R., Paul B. B., Jacobs A. A., Sbarra A. J. Role of the phagocyte in host-parasite interactions. XXII. H2O2-dependent decarbosylation and deamination by myeloperoxidase and its relationship to antimicrobial activity. J Reticuloendothel Soc. 1970 Jun;7(6):754–761. [PubMed] [Google Scholar]
- Tsan M. F., Chen J. W. Oxidation of methionine by human polymorphonuclear leukocytes. J Clin Invest. 1980 May;65(5):1041–1050. doi: 10.1172/JCI109756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zgliczyński J. M., Stelmaszyńska T., Domański J., Ostrowski W. Chloramines as intermediates of oxidation reaction of amino acids by myeloperoxidase. Biochim Biophys Acta. 1971 Jun 16;235(3):419–424. doi: 10.1016/0005-2744(71)90281-6. [DOI] [PubMed] [Google Scholar]
- Zgliczyński J. M., Stelmaszyńska T., Ostrowiski W., Naskalski J., Sznajd J. Myeloperoxidase of human leukaemic leucocytes. Oxidation of amino acids in the presence of hydrogen peroxide. Eur J Biochem. 1968 May;4(4):540–547. doi: 10.1111/j.1432-1033.1968.tb00246.x. [DOI] [PubMed] [Google Scholar]



