Abstract
Background
Cancers in the proximal colon, distal colon, and rectum are frequently studied together; however, there are biological differences in cancers across these sites, particularly in the prevalence of microsatellite instability.
Objective
We assessed differences in survival by colon or rectal cancer site, considering the contribution of microsatellite instability to such differences.
Design
This is a population-based prospective cohort study for cancer survival.
Settings
This study was conducted within the Colon Cancer Family Registry, an international consortium. Participants were identified from population-based cancer registries in the United States, Canada, and Australia.
Patients
Information on tumor site, microsatellite instability, and survival after diagnosis was available for 3284 men and women diagnosed with incident invasive colon or rectal cancer between 1997–2002, with ages at diagnosis ranging from 18–74.
Main Outcome Measures
Cox regression was used to calculate hazard ratios for the association between all-cause mortality and tumor location, overall and by microsatellite instability status.
Results
Distal colon (hazard ratio=0.59, 95% confidence interval: 0.49–0.71) and rectal cancers (hazard ratio=0.68, 95% confidence interval: 0.57–0.81) were associated with lower mortality than proximal colon cancer overall. Compared specifically to cases with proximal colon cancer exhibiting no/low microsatellite instability, cases with distal colon and rectal cancers experienced lower mortality, regardless of microsatellite instability status; cases with proximal colon cancer exhibiting high microsatellite instability had the lowest mortality.
Limitations
Study limitations include the absence of stage at diagnosis and cause of death information for all but a subset of study participants. Some case groups defined jointly by tumor site and microsatellite instability status are subject to small numbers.
Conclusion
Proximal colon cancer survival differs from survival for distal colon and rectal cancer in a manner apparently dependent on microsatellite instability status. These findings support the premise that proximal colon, distal colon, and rectal cancers are clinicopathologically distinct.
Keywords: Colorectal cancer, Colon cancer, Rectal cancer, Survival, Microsatellite instability
INTRODUCTION
Cancers arising in sites from the proximal to distal portions of the colon are frequently studied in combination with each other (i.e., colon cancer), and in combination with cancers arising in the rectum (i.e., colorectal cancer). Increasing evidence, however, indicates that risk factors1–5 and molecular profiles6–9 of cancers may differ across these sites. For example, family history is more strongly associated with risk of proximal colon cancer than rectal cancer,1 and alcohol consumption is more strongly associated with risk of rectal cancer than colon cancer. 4 Cancers in the proximal colon are more likely than cancers in the distal colon and rectum to be diagnosed in women,10,11 to exhibit microsatellite instability (MSI),7–9,12 and to be diagnosed at a later age.7,10–12
Recent studies suggest that differences in biological characteristics and risk factors across cancer site within the colon and rectum may translate to differences in survival. In particular, proximal colon cancer has been associated with poorer survival than distal colon cancer,10,11,13 but there appears to be little difference in survival for cancers arising in the distal colon versus rectum.10 This finding is contrary to observations that proximal colon cancers are more likely to exhibit high MSI (MSI-H) and that MSI-H tumors are, overall, associated with a more favorable prognosis than tumors exhibiting low or no MSI (MSS/MSI-L).14 Two recent studies have suggested that finer distinctions in tumor localization (i.e., subsite within the proximal colon, distal colon, or rectum) are informative of survival,10,13 although these studies have not evaluated the contribution of MSI status to survival differences.
Using data from the Colon Cancer Family Registry, we evaluated differences in all-cause mortality after colon or rectal cancer diagnosis according to tumor site, both overall and by MSI status.
MATERIALS AND METHODS
Study Population
The study population included men and women diagnosed with incident invasive primary colon or rectal cancer who were enrolled in the Colon Cancer Family Registry (C-CFR). The C-CFR is an international resource representing a collaboration between six study centers in Canada, the United States, and Australia. Recruitment protocols and eligibility criteria have been described in detail elsewhere.15 The present analysis was restricted to cases identified from population-based cancer registries and enrolled through four sites (Fred Hutchinson Cancer Research Center, Seattle, Washington, USA; Mayo Clinic, Rochester, Minnesota, USA; Cancer Care Ontario, Toronto, Ontario, Canada; University of Melbourne, Melbourne, Victoria, Australia). Eligible cases were diagnosed between January 1997 and June 2002, with ages at diagnosis ranging from 18 to 74 years (N=4073). A small number of cases were diagnosed with synchronous tumors at different sites (N=36 cases with 2 tumors and N=3 cases with 3 tumors); those cases were excluded from the present analysis. Cases completed risk factor surveys within five years of diagnosis including information regarding: family history, demographic and anthropometric factors, medical history, smoking history, and use of nonsteroidal anti-inflammatory drugs (NSAIDs) (https://cfrisc.georgetown.edu/isc/dd.questionnaires.do). Most cases were interviewed within two years of diagnosis (>85%).
Vital Status
Vital status and date of death were determined via passive follow-up by routine linkage to cancer registries and national death indices. This information was also obtained through active follow-up with cases and/or relatives at, on average, five-year intervals after study recruitment.
Tumor Subsite and Microsatellite Instability Assessment
Tumor subsite was determined based on review of pathology records. Proximal colon cancer was defined as cancer arising in the cecum, ascending colon, hepatic flexure, transverse colon, or splenic flexure (ICD-O-3 codes C180, C182, C183, C184, and C185);16 cancers in the descending (C186) or sigmoid colon (C187) were classified as distal colon cancers, and cancers in the rectosigmoid junction (C199) or rectum (C209) were grouped together as rectal cancers. Cases with unknown tumor site were excluded from the present analysis (N=78).
MSI status was evaluated for cases with available tumor tissue (N=3284). MSI was determined via genetic analysis for most cases (N=2969), based on a 10-marker panel (BAT25, BAT26, BAT40, MYCL, D5S346, D17S250, ACTC, D18S55, D10S197, and BAT34C4) using DNA extracted from formalin-fixed paraffin-embedded normal and tumor tissue as previously described.17,18 Tumors were classified as MSI-H if instability was observed for ≥30% of markers (N=423), and as MSS/MSI-L if instability was observed for <30% of markers (N=2546). For other cases (N=315), MSI was approximated based on immunohistochemistry testing of four markers: hMLH1, hMSH2, hMSH6, and hPMS2.18,19 Cases who exhibited positive staining for all markers were considered MSS/MSI-L (N=278); cases who were negative on at least one marker were considered MSI-H (N=37). Thus, the study population included 2824 MSS/MSI-L and 460 MSI-H cases. Cases with unknown MSI status were excluded (N=630).
Statistical Analysis
We used Cox proportional hazards regression to evaluate the association between tumor location and all-cause mortality after cancer diagnosis, where the time axis was defined as days since diagnosis and death was the outcome of interest. In separate analyses, we evaluated associations with grouped tumor sites (i.e., proximal colon, distal colon, rectum) and with individual tumor subsites (e.g., cecum, descending colon, rectosigmoid junction). For the analysis of grouped tumor sites, we used the most common group (i.e., proximal colon) as the referent category; for analyses of individual tumor subsites, we used the most common subsite within the proximal colon (i.e., cecum) as the referent category.
We also conducted analyses jointly stratifying cases by tumor location and MSI status (MSS/MSI-L, MSI-H). We assessed heterogeneity by MSI status in associations with tumor site using likelihood ratio tests to compare models distinguishing tumors at each site by MSI status to models combining tumors by site regardless of MSI status.
Proportional hazards assumptions were verified by testing for a non-zero slope of the scaled Schoenfeld residuals on ranked failure times.20 Due to a violation of proportional hazards when evaluating associations across the full duration of study follow-up, follow-up was truncated at five years postdiagnosis; thus, all cases still alive at five years postdiagnosis were censored at that time.
Regression models were adjusted for age at diagnosis (10-year strata), year of diagnosis (1997–1998, 1999, 2000–2002), sex, study site, education level (high school graduate or less, some college or vocational school, college graduate), and body mass index (<25.0, 25.0–29.9, ≥30.0 kg/m2); analyses not stratified by MSI were adjusted for MSI. We also evaluated confounding by NSAID use (yes, no), family history of colorectal cancer in first-degree relatives (yes, no), smoking (never, former, current smoker), history of endoscopic screening at least two years prior to diagnosis (yes, no), and race (non-Hispanic white, other); however, these variables did not influence estimates sufficiently to be included in the final model (<10% change). Because information on stage at diagnosis was not available for a large proportion of cases (46%, N=1516), we did not adjust for stage in our primary analyses. We did, however, conduct sensitivity analyses assessing the impact of stage adjustment in cases with known stage at diagnosis, and conducted sensitivity analyses adjusting for T-stage and nodal status (i.e., the T and N components of TNM stage) which were known for a larger proportion of cases (84%, N=2757). Because hereditary nonpolyposis colorectal cancer (HNPCC) may be associated with a better prognosis than sporadic disease,21 and because HNPCC is more likely to be MSI-H and proximally-located than sporadic disease, we also conducted sensitivity analyses excluding cases with germline mutations in one of four DNA mismatch repair genes (MLH1, MSH2, MSH6, and PMS2), which could reflect HNPCC. All analyses were conducted using STATA SE version 11.0 (College Station, Texas).
RESULTS
After a median follow-up of 6.7 years (range: 0.7–14.5 years), approximately 35% of enrolled cases were no longer alive (N=1144); of those who died, 66% died in the first 5 years after diagnosis (N=754) (Table 1). Compared to cases who were alive at the end of follow-up or at five years postdiagnosis (whichever came first), cases who died within five years of diagnosis were more likely to be male (58% versus 51%), more likely to have ever been a smoker (64% versus 59%), more likely to have poorly differentiated tumors (26% versus 16%), and less likely to have MSI-H tumors (8% versus 16%) (p-values all <0.05).
TABLE 1.
Patient population characteristics by vital status at the end of follow-up or 5 years after diagnosis (whichever came first)
| Alive n (%) |
Deceased n (%) |
pa | |
|---|---|---|---|
| Age at diagnosis, y, mean (SD) | 57.5 (10.5) | 57.7 (11.1) | 0.69 |
| Sex | <0.001 | ||
| Male | 1278 (51) | 437 (58) | |
| Female | 1252 (49) | 317 (42) | |
| Study site | |||
| Ontario, Canada | 781 (31) | 218 (28) | 0.01 |
| Melbourne, Victoria, Australia | 411 (16) | 148 (20) | |
| Minnesota, USA | 292 (12) | 62 (8) | |
| Puget Sound, Washington, USA | 1046 (41) | 332 (44) | |
| Family history of colon or rectal cancer in first-degree relatives | |||
| No | 1935 (76) | 594 (79) | 0.19 |
| Yes | 595 (24) | 160 (21) | |
| Race/ethnicity | |||
| Non-Hispanic white | 2288 (91) | 671 (90) | 0.35 |
| Other | 233 (9) | 78 (10) | |
| Education | |||
| ≤High school graduate | 1030 (41) | 332 (45) | 0.14 |
| Some college/vocational school | 782 (31) | 231 (31) | |
| College graduate | 695 (28) | 182 (24) | |
| Unknown | 23 | 9 | |
| BMI, kg/m2 | |||
| <25.0 | 892 (36) | 250 (34) | 0.51 |
| 25.0–29.9 | 997 (40) | 301 (40) | |
| ≥30.0 | 612 (24) | 194 (26) | |
| Unknown | 29 | 9 | |
| Smoking status | |||
| Never smoker | 1023 (41) | 269 (36) | 0.02 |
| Ever smoker | 1496 (59) | 481 (64) | |
| Unknown | 11 | 4 | |
| Tumor grade | |||
| Well differentiated | 214 (9) | 44 (6) | <0.001 |
| Moderately differentiated | 1709 (75) | 476 (68) | |
| Poorly differentiated | 360 (16) | 182 (26) | |
| Unknown | 247 | 52 | |
| MSI status | |||
| MSS/MSI-L | 2133 (84) | 691 (92) | <0.001 |
| MSI-H | 397 (16) | 63 (8) |
MSI = microsatellite instability; MSS = microsatellite stable; MSI-L = low microsatellite instability; MSI-H = high microsatellite instability.
p values for comparison by vital status are based on a t test of group means (age at diagnosis) or χ2(all other variables).
Cases with proximal colon cancer accounted for 38% of the study population; distal colon and rectal cancers accounted for 28% and 34% of cases, respectively (Table 2). The distribution of cancer subsite was similar for women and men, with the exception that women had a greater proportion of cancers located in the cecum (18% versus 13%) and a lower proportion of cancers located in the rectum (20% versus 27%). The distribution of tumor site differed by MSI status: 82% of MSI-H tumors were located in the proximal colon, compared to 31% of MSS/MSI-L tumors. Tumors in individuals with germline mismatch repair mutations (N=70) were also more likely to be located in the proximal colon (71% vs. 37% in other cases, not shown).
Table 2.
Distribution of individual tumor subsites by sex, and microsatellite instability statusa
| Overall N (column %) |
Men N (column %) |
Women N (column %) |
MSS / MSI-L N (column %) |
MSI-H N (column %) |
|
|---|---|---|---|---|---|
| Proximal colon: | |||||
| Overall | 1240 (38) | 568 (33) | 672 (43) | 863 (31) | 377 (82) |
| Cecum | 502 (15) | 217 (13) | 285 (18) | 359 (13) | 143 (31) |
| Ascending colon | 377 (11) | 170 (10) | 207 (13) | 241 (9) | 136 (30) |
| Hepatic flexure | 85 (3) | 45 (3) | 40 (3) | 53 (2) | 32 (7) |
| Transverse colon | 202 (6) | 92 (6) | 110 (7) | 156 (6) | 46 (10) |
| Splenic flexure | 74 (2) | 44 (3) | 30 (2) | 54 (2) | 20 (4) |
| Distal colon: | |||||
| Overall | 933 (28) | 486 (28) | 447 (28) | 887 (31) | 47 (10) |
| Descending colon | 146 (5) | 76 (5) | 70 (4) | 129 (5) | 17 (4) |
| Sigmoid colon | 787 (24) | 410 (24) | 377 (24) | 758 (27) | 30 (6) |
| Rectum: | |||||
| Overall | 1111 (34) | 661 (39) | 450 (28) | 1074 (38) | 37 (8) |
| Rectosigmoid junction | 317 (10) | 182 (10) | 135 (9) | 304 (11) | 13 (3) |
| Rectum | 794 (24) | 479 (27) | 315 (20) | 770 (27) | 24 (5) |
MSS = microsatellite stable; MSI-L = low microsatellite instability; MSI-H = high microsatellite instability
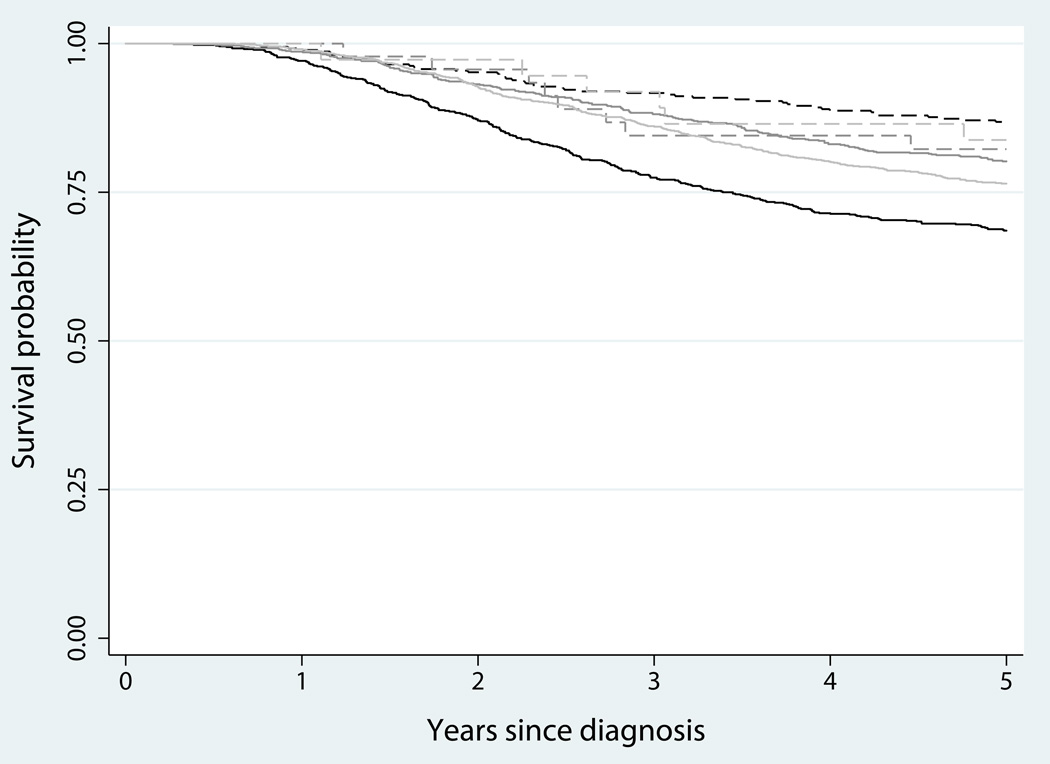
Overall, cases with distal colon cancer and rectal cancer experienced significantly lower mortality than cases with proximal colon cancer (Table 3). However, there was evidence of interaction in this association with tumor site by MSI status (pinteraction=0.04 and 0.15 for distal colon and rectal cancer, respectively). Compared to cases with MSS/MSI-L proximal colon cancer, cases with MSI-H cancer had lower mortality regardless of grouped tumor site (Figure 1); cases with MSS/MSI-L distal colon or MSS/MSI-L rectal cancer also had significantly lower mortality than cases with MSS/MSI-L proximal colon cancer. Compared to cases with MSI-H proximal colon cancer, all MSS/MSI-L case groups experienced significantly higher mortality, regardless of grouped tumor site. Very few MSI-H cases had distal colon or rectal cancer, but patterns of association for these case groups were the same as for MSS/MSI-L distal colon and rectal cancer case groups. MSI status was not associated with all-cause mortality for cases with distal colon or rectal cancer (HR=0.90, 95% CI: 0.44–1.86; HR=0.72, 95% CI: 0.32–1.63, for MSI-H versus MSS/MSI-L in distal colon and rectal cancer cases, respectively, not shown). Additional adjustment for stage, or for components of TNM stage, had little effect on point estimates; all statistically significant findings remained significant with stage adjustment (results not shown). Exclusion of cases with germline mismatch repair mutations also had no impact on findings.
Table 3.
All-cause mortality by grouped tumor site (proximal colon, distal colon, rectum) and microsatellite instability statusa
| Cases N (column %) |
Deaths b N (column %) |
Estimated 5- Year Overall Survival (95% CI) |
HR Relative to Proximal Colon Cancer HR (95% CI)cd |
HR Relative to MSS/MSI-L Proximal Colon Cancer HR (95% CI)c |
HR Relative to MSI-H Proximal Colon Cancer HR (95% CI)c |
p-heterogeneity by MSI status |
|
|---|---|---|---|---|---|---|---|
| Proximal Colon: | |||||||
| Overall | 1240 (38) | 318 (42) | 74% (72–76%) | 1.0 (ref) | <0.001 | ||
| MSS/MSI-L | 863 (27) | 269 (36) | 69% (65–72%) | 1.0 (ref) | 2.63 (1.93–3.57) | ||
| MSI-H | 377 (12) | 49 (7) | 87% (83–90%) | 0.38 (0.28–0.52) | 1.0 (ref) | ||
| Distal Colon: | |||||||
| Overall | 933 (28) | 181 (24) | 80% (78–83%) | 0.59 (0.49–0.71) | 0.67 | ||
| MSS/MSI-L | 887 (27) | 173 (23) | 80% (77–83%) | 0.56 (0.46–0.68) | 1.48 (1.07–2.03) | ||
| MSI-H | 46 (1) | 8 (1) | 82% (68–91%) | 0.48 (0.24–0.98) | 1.27 (0.60–2.68) | ||
| Rectum: | |||||||
| Overall | 1111 (34) | 255 (34) | 77% (74–79%) | 0.68 (0.57–0.81) | 0.42 | ||
| MSS/MSI-L | 1074 (32) | 249 (33) | 77% (74–79%) | 0.66 (0.55–0.78) | 1.73 (1.26–2.36) | ||
| MSI-H | 37 (1) | 6 (1) | 83% (67–92%) | 0.47 (0.21–1.07) | 1.25 (0.53–2.94) |
MSS = microsatellite stable; MSI-L = low microsatellite instability; MSI-H = high microsatellite instability
Includes only deaths occurring within the first five years after diagnosis (N=754 of 1144 total deaths over study period, i.e., 66%)
Adjusted for age at diagnosis, year of diagnosis, sex, study site, education, and body mass index
Also adjusted for MSI status
Figure 1.
Kaplan-Meier curves for overall survival after colorectal cancer diagnosis according to MSI status (solid line = MSS/MSI-L, dashed line = MSI-H) and tumor site (black = proximal colon, medium gray = distal colon, light gray = rectum)
Based on observed differences in associations with tumor site by MSI status, analyses of all-cause mortality by individual tumor subsites were stratified by MSI. Among MSS/MSI-L cases, those with cancer located within the descending colon, sigmoid colon, rectosigmoid junction, and rectum experienced significantly lower mortality than cases with cecal cancer (Table 4). There was no difference in associations across individual subsites within the distal colon (pheterogeneity=0.73) or rectum (pheterogeneity=0.81). Among MSS/MSI-L proximal colon cancer cases, those with cancer located in the hepatic flexure experienced significantly lower mortality than individuals with cancer in the cecum (HR=0.50, 95% CI: 0.26–0.95), but mortality was otherwise similar across proximal colon subsites (pheterogeneity=0.14). Results were unchanged when adjusting for stage and when excluding cases with germline mismatch repair mutations (not shown). Small numbers limited analyses by tumor subsite for MSI-H cases; however, cases with MSI-H sigmoid colon cancer were found to have significantly higher mortality relative to cases with MSI-H cecal cancer (HR=2.64, 95% CI: 1.00–6.96). When we analyzed the 8 subsites from cecum to rectum as a continuous variable, there was a significant association between increasing distance from the cecum and lower mortality (HR=0.93, 95% CI: 0.90–0.95), with evidence of interaction by MSI status (p=0.009) (not shown).
Table 4.
All-cause mortality by tumor subsite and microsatellite instability statusa
| Cases N (column %) |
Deathsb N (column %) |
MSS/MSI-L Cases HR (95% CI)c |
MSI-H Cases HR (95% CI)c |
|
|---|---|---|---|---|
| Proximal colon: | ||||
| Cecum | 502 (15) | 137 (18) | 1.0 (ref) | 1.0 (ref) |
| Ascending colon | 377 (11) | 96 (13) | 0.90 (0.67–1.21) | 1.31 (0.64–2.66) |
| Hepatic flexure | 85 (3) | 16 (2) | 0.50 (0.26–0.95) | 1.75 (0.65–4.72) |
| Transverse colon | 202 (6) | 51 (7) | 0.77 (0.55–1.08) | 1.47 (0.58–3.74) |
| Splenic flexure | 74 (2) | 18 (2) | 0.78 (0.45–1.35) | --d |
| Distal colon: | ||||
| Descending colon | 146 (4) | 26 (3) | 0.46 (0.30–0.72) | --d |
| Sigmoid colon | 787 (24) | 155 (21) | 0.50 (0.39–0.64) | 2.64 (1.00–6.96) |
| Rectum: | ||||
| Rectosigmoid junction | 317 (10) | 72 (10) | 0.59 (0.44–0.80) | --d |
| Rectum | 794 (24) | 183 (24) | 0.57 (0.45–0.72) | --d |
MSS = microsatellite stable; MSI-L = low microsatellite instability; MSI-H = high microsatellite instability
Includes only deaths occurring within the first five years after diagnosis (N=754 of 1144 total deaths over study period, i.e., 66%)
Adjusted for age at diagnosis, year of diagnosis, sex, study site, education, and body mass index
Estimate based on <5 deaths
DISCUSSION
In this cohort of individuals with invasive colon or rectal cancer, we found that those with tumors in the distal colon and rectum experienced lower all-cause mortality in the five years after diagnosis relative to individuals with proximal colon cancer. However, comparisons of mortality by tumor site differed by MSI status: MSS/MSI-L proximal colon cancer was associated with the poorest prognosis, whereas MSI-H proximal colon cancer was associated with the best prognosis. We found little difference in these patterns when more finely evaluating associations across subsites within the proximal colon, distal colon, and rectum.
Certain limitations should be considered in interpreting these findings. In particular, inclusion in the study population was contingent on surviving long enough after cancer diagnosis to be enrolled in the C-CFR. Selection bias is possible if otherwise eligible cases who died before they could be enrolled differed from included cases with respect to tumor location or covariates. Additionally, we were unable to evaluate treatment variables as confounders or effect modifiers since we did not have this information; however, it is unlikely that treatment or access to treatment differed substantially by tumor location. Similarly, we did not adjust for stage at diagnosis because this information was not available for a large fraction of cases; we were also concerned that stage classification and the assessment of stage components could differ across tumor subsites, in which case, stage adjustment could create difficulties for interpretation. We did, however, conduct sensitivity analyses adjusting for components of stage and adjusting for stage in the subset of cases with known stage and found that any confounding by stage was not responsible for observed survival differences. Although we had a large study population, some case groups became small after stratification by MSI status and tumor site. Specifically, as has been reported by previous studies,7,9,12 we had few cases with MSI-H distal colon cancer or MSI-H rectal cancer; thus, interpretation of comparisons with these small case groups should be made cautiously. Last, although the vast majority of cases (75%) were followed beyond five years postdiagnosis, we truncated follow-up at five years in this analysis to avoid violations of proportional hazards assumptions. As a result, cases who died more than five years postdiagnosis were censored before they experienced the study outcome. However, by truncating follow-up it also became much more likely that cases who died during the period of analysis died as a result of their disease and not due to some other cause. Although we did not have cause of death information for most cases, within the subset of cases for whom we did have cause of death data (41%), approximately 80% of deaths occurring within the first 5 years postdiagnosis were attributable to colon or rectal cancer, compared to only 41% of deaths occurring more than 5 years postdiagnosis (not shown).
Consistent with our results, most prior studies have suggested that proximal colon cancers are associated with greater mortality than distal colon7,10,11,13 and rectal cancers.10 Recently, a study from the Swedish Family-Cancer Database demonstrated significantly greater cause-specific mortality in patients diagnosed with proximal colon cancer relative to patients with rectal cancer (HR=1.16, 95% CI: 1.07–1.27), but found no difference in survival between patients with distal colon versus rectal cancer (HR=1.04, 95% CI: 0.95–1.15).10 In another analysis, Wray et al. found lower all-cause mortality in patients diagnosed with cancer located in the sigmoid colon than in those with cancer located in the cecum, ascending colon, or hepatic flexure (HR=0.90, 95% CI: 0.87–0.92).13
Previous observations that proximal colon cancer is associated with poorer survival than distal colon or rectal cancer7,10,11,13 appear inconsistent with observations that proximal colon cancers are more likely to be MSI-H,7,9,12 as MSI-H cancers generally have a more favorable prognosis than MSS/MSI-L cancers.14 We found that the greater mortality associated with proximal colon tumor location was restricted to MSS/MSI-L proximal colon cancer, and that the more favorable survival associated with MSI-H status was limited to proximal colon cancer. Very few prior studies have considered MSI in evaluating survival differences by tumor site or, conversely, considered tumor site in evaluating associations by MSI.12,22,23 One study by Jernvall et al. reported that the survival advantage associated with MSI-H status was limited to patients with proximal colon cancer.22 Another analysis of patients enrolled in clinical trials demonstrated greater mortality in distal colon cancer patients relative to patients with proximal colon cancer, and found this association to be diminished after excluding MSI-H cases.12 These studies support our finding that the interaction between MSI and tumor location is of critical importance in evaluating survival differences by MSI status or tumor location. However, given the rarity of the MSI-H phenotype in distal colon and rectal cancers in these prior studies12,21,23 and in ours, there remains a need to better understand the epidemiologic and clinical profile of MSI-H distal colon and rectal cancers.
It is possible that differences in all-cause mortality by tumor site reflect underlying differences in tumor aggressiveness or amenability to screening. Consistent with such a hypothesis, previous studies have found that sigmoidoscopy24,25 and colonoscopy26,27 are associated with lower incidence and mortality for distal but not proximal colon cancer. Proximal colon cancers are more likely to be diagnosed as interval cancers,28,29 which could imply a more rapid pattern of tumor progression, and are also more likely to be BRAF-mutated and to exhibit a CpG island methylator phenotype (CIMP);9 these characteristics, when observed in combination with MSS/MSI-L status, have been associated with poorer prognosis.30,31 We did not have information on BRAF-mutation or CIMP status, but found that cancers at all sites within the proximal colon were more likely to be poorly differentiated than cancers at sites within the distal colon or rectum (results not shown). Differences in the distribution of MSI status for proximal colon cancer versus distal colon and rectal cancer may also be indicative of differences in pathways of tumor development. Previous studies have hypothesized that MSI-H status reflects a pathway(s) of tumor development distinct from MSS/MSI-L tumors, as reflected by differences in the prevalence of certain somatic mutations and epigenetic alterations;32 these differences, in turn, may have an important impact on survival.
In summary, all-cause mortality after diagnosis of colon or rectal cancer differed significantly for patients with tumors located in the proximal colon relative to patients with distal colon or rectal cancer. For individuals with proximal colon cancer, mortality risk differed significantly according to MSI status. MSS/MSI-L proximal colon cancer was associated with a greater mortality than MSI-H proximal colon cancer, and was also associated with greater mortality than MSS/MSI-L or MSI-H distal colon or rectal cancers. Conversely, MSI-H proximal colon cancers were associated with the most favorable survival. These findings are consonant with a more aggressive clinicopathology of MSS/MSI-L tumors located in the proximal colon.
Acknowledgments
Financial Support: This work was supported by the National Cancer Institute, National Institutes of Health (RFA CA-95-011) and through cooperative agreements with members of the Colon Cancer Family Registry and Principal Investigators. Collaborating centers include the Australian Colorectal Cancer Family Registry (5U24CA097735, M.A.J., A.K.W.), the Mayo Clinic Cooperative Family Registry for Colon Cancer Studies (5U24CA074800, N.M.L.), the Ontario Registry for Studies of Familial Colorectal Cancer (5U24CA074783, S.G., R.G.), the Seattle Colon Cancer Family Registry (5U24CA074794, P.A.N.), and the University of Southern California (5U24CA074799, J.A.B.). This publication was also supported by the National Cancer Institute career grant (K05CA152715, P.A.N.) and training grant R25CA94880 (A.I.P.).
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Disclaimers: The content of this manuscript does not necessarily reflect the views or policies of the National Cancer Institute or any of the collaborating centers in the CFRs, nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government or the CFR.
Author Contributions: AIP, NML, MAJ, JAB, AKW, SG, RG, and PAN contributed to the study conception and design. NML, MAJ, JAB, SG, RG, and PAN were involved with the collection and assembly of data. AIP led the data analysis and, along with NML, MAJ, JAB, AKW, SG, RG, and PAN, contributed to the interpretation of analytic findings. AIP also led the manuscript writing process. All co-authors contributed to the manuscript writing and editing process and provided final approval of the manuscript. NML, MAJ, JAB, SG and PAN provided financial and administrative support for the original study.
REFERENCES
- 1.Andrieu N, Launoy G, Guillois R, Ory-Paoletti C, Gignoux M. Estimation of the familial relative risk of cancer by site from a French population based family study on colorectal cancer (CCREF study) Gut. 2004;53:1322–1328. doi: 10.1136/gut.2003.036376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Wei EK, Giovannucci E, Wu K, et al. Comparison of risk factors for colon and rectal cancer. Int J Cancer. 2004;108:433–442. doi: 10.1002/ijc.11540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Limburg PJ, Vierkant RA, Cerhan JR, et al. Cigarette smoking and colorectal cancer: long-term, subsite-specific risks in a cohort study of postmenopausal women. Clin Gastroenterol Hepatol. 2003;1:202–210. doi: 10.1053/cgh.2003.50030. [DOI] [PubMed] [Google Scholar]
- 4.Bongaerts BW, van den Brandt PA, Goldbohm RA, de Goeij AF, Weijenberg MP. Alcohol consumption, type of alcoholic beverage and risk of colorectal cancer at specific subsites. Int J Cancer. 2008;123:2411–2417. doi: 10.1002/ijc.23774. [DOI] [PubMed] [Google Scholar]
- 5.Mahipal A, Anderson KE, Limburg PJ, Folsom AR. Nonsteroidal anti-inflammatory drugs and subsite-specific colorectal cancer incidence in the Iowa women's health study. Cancer Epidemiol Biomarkers Prev. 2006;15:1785–1790. doi: 10.1158/1055-9965.EPI-05-0674. [DOI] [PubMed] [Google Scholar]
- 6.Kim GP, Colangelo LH, Wieand HS, et al. Prognostic and predictive roles of high-degree microsatellite instability in colon cancer: a National Cancer Institute-National Surgical Adjuvant Breast and Bowel Project Collaborative Study. J Clin Oncol. 2007;25:767–772. doi: 10.1200/JCO.2006.05.8172. [DOI] [PubMed] [Google Scholar]
- 7.Minoo P, Zlobec I, Peterson M, Terracciano L, Lugli A. Characterization of rectal, proximal and distal colon cancers based on clinicopathological, molecular and protein profiles. Int J Oncol. 2010;37:707–718. doi: 10.3892/ijo_00000720. [DOI] [PubMed] [Google Scholar]
- 8.Deng G, Kakar S, Tanaka H, et al. Proximal and distal colorectal cancers show distinct gene-specific methylation profiles and clinical and molecular characteristics. Eur J Cancer. 2008;44:1290–1301. doi: 10.1016/j.ejca.2008.03.014. [DOI] [PubMed] [Google Scholar]
- 9.Yamauchi M, Morikawa T, Kuchiba A, et al. Assessment of colorectal cancer molecular features along bowel subsites challenges the conception of distinct dichotomy of proximal versus distal colorectum. Gut. 2012;61:847–854. doi: 10.1136/gutjnl-2011-300865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hemminki K, Santi I, Weires M, Thomsen H, Sundquist J, Bermejo JL. Tumor location and patient characteristics of colon and rectal adenocarcinomas in relation to survival and TNM classes. BMC Cancer. 2010;10:688. doi: 10.1186/1471-2407-10-688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Meguid RA, Slidell MB, Wolfgang CL, Chang DC, Ahuja N. Is there a difference in survival between right-versus left-sided colon cancers? Ann Surg Oncol. 2008;15:2388–2394. doi: 10.1245/s10434-008-0015-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sinicrope FA, Rego RL, Halling KC, et al. Prognostic impact of microsatellite instability and DNA ploidy in human colon carcinoma patients. Gastroenterology. 2006;131:729–737. doi: 10.1053/j.gastro.2006.06.005. [DOI] [PubMed] [Google Scholar]
- 13.Wray CM, Ziogas A, Hinojosa MW, Le H, Stamos MJ, Zell JA. Tumor subsite location within the colon is prognostic for survival after colon cancer diagnosis. Dis Colon Rectum. 2009;52:1359–1366. doi: 10.1007/DCR.0b013e3181a7b7de. [DOI] [PubMed] [Google Scholar]
- 14.Guastadisegni C, Colafranceschi M, Ottini L, Dogliotti E. Microsatellite instability as a marker of prognosis and response to therapy: A meta-analysis of colorectal cancer survival data. Eur J Cancer. 2010;46:2788–2798. doi: 10.1016/j.ejca.2010.05.009. [DOI] [PubMed] [Google Scholar]
- 15.Newcomb PA, Baron J, Cotterchio M, et al. Colon Cancer Family Registry: an international resource for studies of the genetic epidemiology of colon cancer. Cancer Epidemiol Biomarkers Prev. 2007;16:2331–2343. doi: 10.1158/1055-9965.EPI-07-0648. [DOI] [PubMed] [Google Scholar]
- 16.World Health Organization. International Classification of Diseases for Oncology. In: Editor ed., editor. Book International Classification of Diseases for Oncology. 3rd ed. City: WHO; 2000. [Google Scholar]
- 17.Boland CR, Thibodeau SN, Hamilton SR, et al. A National Cancer Institute Workshop on Microsatellite Instability for cancer detection and familial predisposition: development of international criteria for the determination of microsatellite instability in colorectal cancer. Cancer Res. 1998;58:5248–5257. [PubMed] [Google Scholar]
- 18.Cicek MS, Lindor NM, Gallinger S, et al. Quality assessment and correlation of microsatellite instability and immunohistochemical markers among population- and clinic-based colorectal tumors results from the Colon Cancer Family Registry. J Mol Diagn. 2011;13:271–281. doi: 10.1016/j.jmoldx.2010.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lindor NM, Burgart LJ, Leontovich O, et al. Immunohistochemistry versus microsatellite instability testing in phenotyping colorectal tumors. J Clin Oncol. 2002;20:1043–1048. doi: 10.1200/JCO.2002.20.4.1043. [DOI] [PubMed] [Google Scholar]
- 20.Therneau TM, Grambsch PM. Modeling survival data: Extending the Cox model. New York: Springer; 2000. [Google Scholar]
- 21.Stigliano V, Assisi D, Cosimelli M, et al. Survival of hereditary non-polyposis colorectal cancer patients compared with sporadic colorectal cancer patients. J Exp Clin Cancer Res. 2008;27:39. doi: 10.1186/1756-9966-27-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Jernvall P, Makinen MJ, Karttunen TJ, Makela J, Vihko P. Microsatellite instability: impact on cancer progression in proximal and distal colorectal cancers. Eur J Cancer. 1999;35:197–201. doi: 10.1016/s0959-8049(98)00306-2. [DOI] [PubMed] [Google Scholar]
- 23.Sanchez JA, Krumroy L, Plummer S, et al. Genetic and epigenetic classifications define clinical phenotypes and determine patient outcomes in colorectal cancer. Br J Surg. 2009;96:1196–1204. doi: 10.1002/bjs.6683. [DOI] [PubMed] [Google Scholar]
- 24.Newcomb PA, Storer BE, Morimoto LM, Templeton A, Potter JD. Long-term efficacy of sigmoidoscopy in the reduction of colorectal cancer incidence. J Natl Cancer Inst. 2003;95:622–625. doi: 10.1093/jnci/95.8.622. [DOI] [PubMed] [Google Scholar]
- 25.Atkin WS, Edwards R, Kralj-Hans I, et al. Once-only flexible sigmoidoscopy screening in prevention of colorectal cancer: a multicentre randomised controlled trial. Lancet. 2010;375:1624–1633. doi: 10.1016/S0140-6736(10)60551-X. [DOI] [PubMed] [Google Scholar]
- 26.Baxter NN, Goldwasser MA, Paszat LF, Saskin R, Urbach DR, Rabeneck L. Association of colonoscopy and death from colorectal cancer. Ann Intern Med. 2009;150:1–8. doi: 10.7326/0003-4819-150-1-200901060-00306. [DOI] [PubMed] [Google Scholar]
- 27.Brenner H, Hoffmeister M, Arndt V, Stegmaier C, Altenhofen L, Haug U. Protection from right- and left-sided colorectal neoplasms after colonoscopy: population-based study. J Natl Cancer Inst. 2010;102:89–95. doi: 10.1093/jnci/djp436. [DOI] [PubMed] [Google Scholar]
- 28.Baxter NN, Sutradhar R, Forbes SS, Paszat LF, Saskin R, Rabeneck L. Analysis of administrative data finds endoscopist quality measures associated with postcolonoscopy colorectal cancer. Gastroenterology. 2011;140:65–72. doi: 10.1053/j.gastro.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 29.Shaukat A, Arain M, Thaygarajan B, Bond JH, Sawhney M. Is BRAF mutation associated with interval colorectal cancers? Dig Dis Sci. 2010;55:2352–2356. doi: 10.1007/s10620-010-1182-9. [DOI] [PubMed] [Google Scholar]
- 30.Dahlin AM, Palmqvist R, Henriksson ML, et al. The role of the CpG island methylator phenotype in colorectal cancer prognosis depends on microsatellite instability screening status. Clin Cancer Res. 2010;16:1845–1855. doi: 10.1158/1078-0432.CCR-09-2594. [DOI] [PubMed] [Google Scholar]
- 31.Phipps AI, Makar KW, Baron J, et al. BRAF V600E mutation status and survival after colorectal cancer diagnosis according to patient and tumor characteristics. In preparation. 2012 doi: 10.1158/1055-9965.EPI-12-0674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ogino S, Goel A. Molecular classification and correlates in colorectal cancer. J Mol Diagn. 2008;10:13–27. doi: 10.2353/jmoldx.2008.070082. [DOI] [PMC free article] [PubMed] [Google Scholar]