Abstract
A method for the selective labeling and imaging of catecholamines in live and fixed secretory cells is reported. The method integrates a tailored approach using a novel fluorescence-based turn-on molecular sensor (NeuroSensor 521) that can exploit the high concentration of neurotransmitters and acidic environment within secretory vesicles for the selective recognition of norepinephrine and dopamine. The utility of the method was demonstrated by selectively labeling and imaging norepinephrine in secretory vesicles such that discrimination between norepinephrine- and epinephrine-enriched populations of chromaffin cells was observed. This method was validated in fixed cells by co-staining with an anti-PNMT antibody.
Keywords: Fluorescent sensor, catecholamine, norepinephrine, cell imaging, chromaffin
The catecholamines dopamine, norepinephrine, and epinephrine are the principal neurotransmitters in the sympathetic nervous system.1 In particular, norepinephrine regulates many critical functions that include attention, memory, learning, emotion, and autonomic and cardiovascular function. In the periphery, norepinephrine increases heart rate, cardiac contractility, vascular tone, renin-angiotensin system activity, and renal sodium reabsorption.2 Norepinephrine is secreted by chromaffin cells, which package catecholamines at high concentrations (0.5–1.0 M) and at low pH (5.0–5.5) in neurosecretory vesicles.3 Chromaffin cells possess approximately 30 000 large dense-core vesicles (LDCV) with norepinephrine and epinephrine.4 Chromaffin cells that store and release mainly epinephrine can be separated from those that utilize mainly norepinephrine through density-gradient centrifugation, though a third subpopulation which secrete both epinephrine and norepinephrine has been identified via cyclic voltametry.5 Over the years, chromaffin cells have become a standard platform for the study of processes related to exocytosis. Thus, chromaffin cells appeared to be an ideal platform for the study of novel sensors for neurotransmitters.
Currently, catecholamines can be studied via electrochemical and chromatographic techniques that provide characterization and quantification, although these techniques can only provide crude spatial information.6 Recently, fluorescent false neurotransmitters (FFNs) have been developed which are selectively loaded into vesicles that express neuronal vesicular monoamine transporter (VMAT) and represent an optical approach for labeling vesicles containing catecholamines and imaging catecholamine release at the single-vesicle level.7 However, FFNs are loaded into all secretory vesicles expressing the VMAT protein without discrimination to cell type and thus, the approach cannot distinguish distinct cell populations that secrete a particular neurotransmitter.
Fluorescent sensors remain a compelling technology for approaching the general problem of selective neurotransmitter detection. In recent years, a number of catecholamine sensors have been reported including RNA aptamers, fluorescent ribonucleopeptide (RNP) complexes, and boronic acid based synthetic receptors.8 However, none of these methods represent a practical approach for in vivo and in vitro cellular analysis and imaging. Indeed, some time ago, we developed a coumarin aldehyde fluorescent sensor for the selective recognition and sensing of amines; however, in the case of dopamine and norepinephrine, the sensor operated in a turn-off mode.9 Herein, we present a fluorescent turn-on sensor based on the coumarin aldehyde scaffold that allows for the selective recognition and sensing of norepinephrine in live and fixed cells.
Results and Discussion
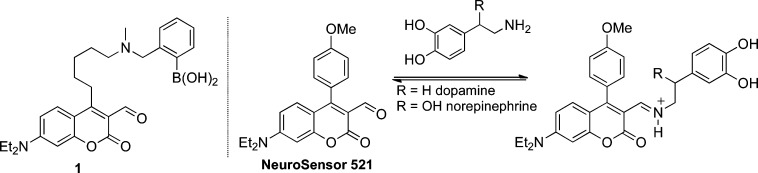
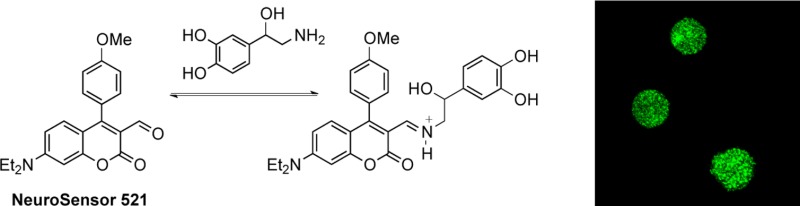
Our original catecholamine sensor design (1, Figure 1) included an aldehyde that would associate with the analyte amine group via iminium ion formation, and a boronic acid that would associate with the catechol group.9 Unfortunately, the catechol group strongly quenched the sensor. In this study, NeuroSensor 521 (NS521) was designed with only the aldehyde group to associate with the analyte amines. Instead of a boronic acid group, a p-methoxyphenyl moiety was incorporated to modulate the fluorescence properties of the coumarin such that it would not be quenched by the catechol group. It was anticipated that the lack of a boronic acid recognition unit would lower the affinity of NeuroSensor 521 for catecholamines relative to sensor 1, but given the extremely high concentration of catecholamine in the secretory vesicles, a lower binding constant was not a concern.
Figure 1.
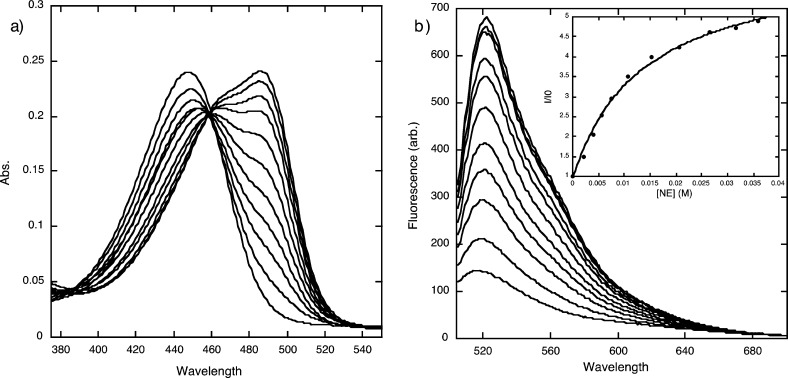
NeuroSensor 521 was prepared and screened with various relevant amines via absorption and fluorescence spectroscopy. As observed with other sensors in this series,10 NeuroSensor 521 binds to all primary amines via iminium ion formation,11 which produces a red shift in absorption from 448 to 488 nm (Figure 2). In fluorescence mode, exciting the sensor at 488 nm and adding norepinephrine produced a marked 5.3-fold increase in fluorescence. Table 1 summarizes binding and spectroscopic data for the interaction of NeuroSensor 521 and a number of relevant amines. As observed with other sensors in this series,10 all primary amines bind with low binding affinity and high fluorescence enhancements. Interestingly, catacholamines such as norepinephrine and dopamine have 10-fold higher binding constants than other alkyl amines such as glycine. As with other sensors in the class, NeuroSensor 521 does not interact with secondary amines such as epinephrine.
Figure 2.
(a) UV/vis titration and (b) fluorescence titration of NS521 (10 μM in 25 mM HEPES, 50 mM Na2S2O3, pH = 5.0, 37 °C) with norepinephrine (λex = 488 nm). Inset in (b) is the fit to a single-site binding isotherm.
Table 1. Association Constants and Spectroscopic Parameters for the Binding of NS521 to Various Analytes.
| amine guest | Ka (M-1)a | Isat/I0b (λex = 488 nm) | quantum yield (Φ) |
|---|---|---|---|
| epinephrine | 0 | nd | nd |
| norepinephrine | 78 | 5.4 | 0.0033 |
| dopamine | 112 | 3.0 | nd |
| glutamate | 10 | 7.8 | 0.0095 |
| lysine | 11 | 15.1 | nd |
| glycine | 8 | 11.1 | nd |
| none | 0.0053 |
Ka measured by fluorescence spectroscopy, λex = 488 nm, λem = 521 nm. Error in Ka values are ±10% based on triplicate titrations.
Isat = fluorescence intensity at saturation taken from the theoretical fit to the binding isotherm. nd = not determined.
The data in Table 1 indicates that the maximum fluorescence response of the catecholamines is lower than that of generic amines such as glutamate. The quantum yield for NeuroSensor 521 was determined both alone and bound to glutamate and norepinephrine. The 3-fold difference in the latter two quantum yields is due to the quenching nature of the catechol group, which can undergo photoelectron transfer (PET) to the coumarin fluorophore. Taken together, these data suggest that NeuroSensor 521 will bind more strongly to catecholamines in a cell, but with lower overall fluorescent enhancements. In a neuroendocrine cell, the concentration of catecholamines (0.5 – 1.0 M) in secretory vesicles is at least an order of magnitude greater than the concentration of other biogenic primary amines. Thus, the moderate selectivity of NeuroSensor 521 for catecholamines over other biogenic amines coupled with the relatively high concentrations of catecholamines in secretory vesicles was expected to overcome the lower fluorescence response from the catecholamine. In addition, the unique spectral properties of NeuroSensor 521 allow for monitoring the unbound and bound states using 440 and 488 nm excitation, respectively.
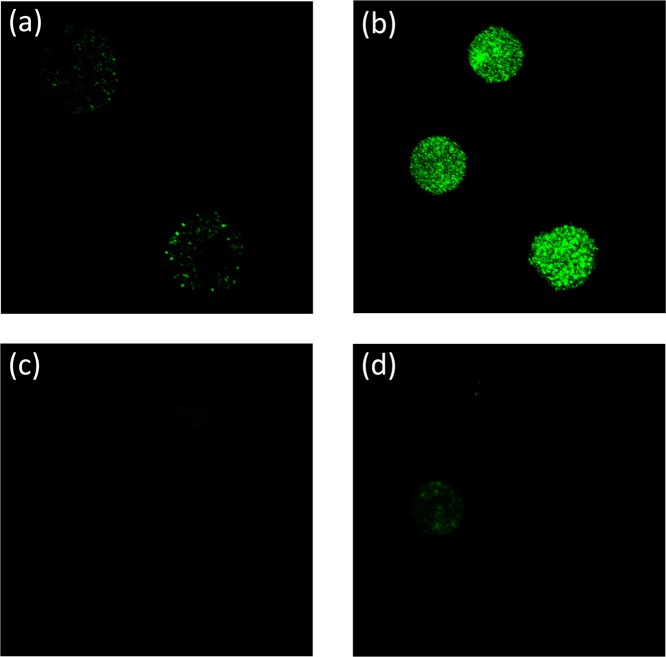
From the titration data, it appeared that NeuroSensor 521 could be used to selectively detect dopamine and norepinephrine over epinephrine. Thus, we chose to demonstrate the utility of our method through the selective labeling and direct visualization of norepinephrine in secretory vesicles to distinguish norepinephrine- from epinephrine-enriched populations of chromaffin cells. Chromaffin cells were separated into norepinephrine-enriched and epinephrine-enriched fractions by centrifugation on a Percoll gradient.12 Both populations were independently incubated with a 0.1 μM solution of NeuroSensor 521 at 37 °C for 30 min and then washed to remove excess sensor and plated.13 The cells were examined by confocal fluorescence microscopy using 488 nm excitation (Figure 3a,b). The norepinephrine-enriched cell population showed strong, punctate fluorescence compared to the epinephrine-enriched cell population, which only showed marginal fluorescence. The punctate fluorescence pattern is consistent with secretory vesicles in chromaffin cells. These results indicate that the sensor is able to enter the vesicle and bind to norepinephrine selectively over epinephrine, as anticipated. The low fluorescence response observed within the epinephrine-containing chromaffin cells was attributed to the binding of NeuroSensor 521 to the low concentration of norepinephrine present in these vesicles. Next, the cells were excited at 440 nm to selectively excite any potential unbound sensor, although some excitation of bound sensor is expected at this wavelength (Figure 3c,d). Weak fluorescence was observed in both cell populations, though again more in the norepinephrine-enriched population. These data suggest that little sensor remains in the epinephrine-enriched cell population. This result supports the notion that NeuroSensor 521 accumulates in vesicles, which is not surprising for a neutral compound that forms a charged complex upon interaction with the target analyte (Figure 1). The charged complex presumably cannot cross the vesicle membrane and thus becomes trapped.14 Thus, the higher fluorescence in the norepinephrine-containing cells is a result of the high concentration of primary catecholamine in the vesicles, which causes accumulation of the sensor in these vesicles. Lacking significant concentrations of primary catecholamines, the epinephrine-containing cells do not accumulate the sensor and show lower fluorescence. The overall low background fluorescence is remarkable given the rather promiscuous binding of NeuroSensor 521 to primary amines. Here the rather low affinity of the sensor for binding amines actually confers an advantage for selective labeling of high concentrations of primary amine analytes in secretory vesicles.
Figure 3.
Epinephrine-containing cells incubated with NS521 (0.1 μM): (a) λex = 488 nm and (c) λex = 440 nm. Norepinephrine-containing cells incubated NS521 (0.1 μM): (b) λex = 488 nm and (d) λex = 440 nm.
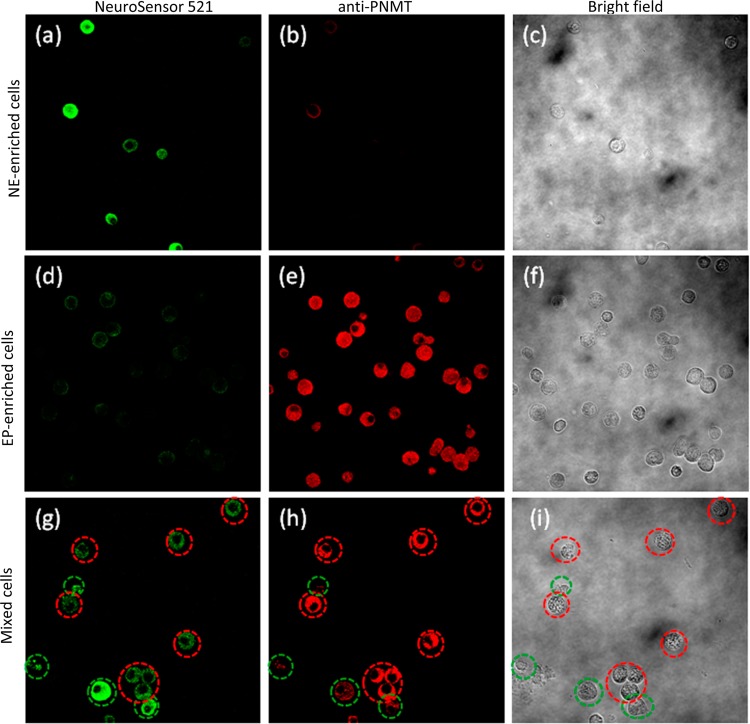
To further validate the selective labeling of norepinephrine-containing vesicles, we labeled fixed cells using an antibody against phenylethanolamine N-methyltransferase (PNMT), an enzyme that converts norepinephrine to epinephrine. Antibody labeling was visualized with a Cy3-conjugated secondary antibody.15 It has been shown that epinephrine-enriched cell populations give the highest level of PNMT staining, while norepinephrine-enriched populations gave lower staining (ca. 20% of that with epinephrine-enriched cells). For this experiment, three populations of chromaffin cells (norepinephrine-enriched, epinephrine-enriched, and mixed) were stained with NeuroSensor 521 and fixed in 4% paraformaldehyde. The fixed cells were stained with the anti-PNMT and secondary antibodies (Figure 4). The top row of Figure 4 shows that the NE-enriched cell population stains brightly with NS521 and weakly with the fluorescent antibody while the opposite is true for the EP-enriched cell population (second row). Indeed the mixed population of cells (third row, Figure 4) contained cells with staining patterns consistent with both NE-enriched cells (circled in green) and EP-enriched cells (circled in red).
Figure 4.
All cells were stained with NS521 (1 μM), fixed (4% formaldehyde), and incubated with an anti-PNMT antibody followed by a Cy3-anti-rabbit antibody. Norepinephrine-enriched cells (a–c) visualized at (a) 525 nm, (b) 585 nm, and (c) bright field. Epinephrine-enriched cells (d–f) visualized at (d) 525 nm (e), 585 nm, and (f) bright field. Mixed cells (g–i) visualized at (g) 525 nm, (h) 585 nm, and (i) bright field. Cells circled in green indicate NE-enriched cells, and cells circled in red indicate EP-enriched cells.
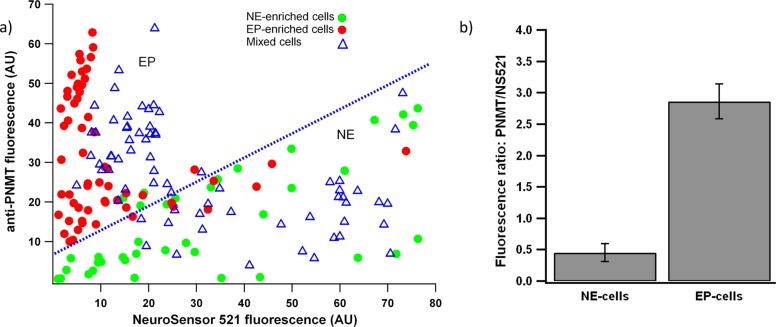
The fluorescence emission in the images shown in Figure 4 were quantified using ImageJ16 (Figure 5). As shown in Figure 5a, EP-enriched cells show an increased anti-PNMT staining compared to NS521. Conversely, the NE-enriched cells showed an increased staining by NS521 compared to the anti-PNMT. As expected, the mixed cell population contained cells which fell into both categories. The average fluorescence intensity ratio of 585 nm vs 525 for epinephrine-enriched cells was 6 times of that of norepinephrine-enriched cells (Figure 5b).
Figure 5.
Quantification of cell fluorescence from the cell populations from Figure 4. (a) Fluorescence intensity at 585 nm plotted on the Y-axis and fluorescence intensity at 525 nm plotted on the X-axis. Each point represents an individual cell. EP-enriched cells fall above the blue line, and NE-enriched cells fall below the blue line. (b) The average ratio of fluorescence intensity at 585 nm vs 525 for norepinephrine-enriched and epinephrine-enriched cells.
Conclusion
NeuroSensor 521 was developed as a turn-on sensor for primary amines. It binds catecholamines such as norepinephrine more tightly than other biogenic amines such as glutamate, and it has no apparent affinity for secondary amines such as epinephrine. This selectivity was demonstrated in chromaffin cells where the norepinephrine-containing cells were stained preferentially over epinephrine-containing cells under identical conditions. Furthermore, the fluorescence is not affected by fixation. NeuroSensor 521 represents a convenient method to selectively stain norepinephrine and dopamine in neurosecretory vesicles.
Methods
Fluorescence Titrations
Fluorescence spectra were recorded on a Shimadzu RF-5301 PC spectrofluorometer at 37 °C. A 1 mg/mL stock solution of NeuroSensor 521 in DMSO was prepared. A stock solution of NeuroSensor 521 in buffer (1 × 10–5 M, 25 mM HEPES, 50 mM Na2S2O3, pH = 5.0) was prepared. Norepinephrine, dopamine, epinephrine, and glutamate stock solutions were prepared by separately dissolving the analytes at the concentration to be used in the titration with the buffered stock solution of NeuroSensor 521 (thus avoiding dilution of NeuroSensor 521 during the experiment). NeuroSensor 521 was titrated with aliquots of analyte solution. The sensor was excited at 488 nm with slit widths of 5 nm.
Chromaffin Cell Preparation
Chromaffin cells were isolated from bovine adrenal glands as previously described.17 Following centrifugation at 18 °C at 13 000 rpm for 45 min in the Percoll gradient, chromaffin cells separate into fractions enriched with either epinephrine-enriched cells (denser band) and norepinephrine-enriched cells (lighter band).12b Over 90% of the catecholamine content found in the cells in the denser fraction is epinephrine, whereas approximately 67% of the catecholamine content in the lighter fraction is norepinephrine.18 The two cell fractions were collected from the Percoll gradient with careful pipetting and separately cultured. We used an alternative culture method to make it easier to detach the cells from the flasks and to reduce cell clumping. Chromaffin cells were cultured in Hibernate A media with calcium (BrainBits LLC, Springfield, IL) in a refrigerator (4 °C) and used 1–6 days after preparation.18
Standard Cell Bath Solution
The standard cell bath solution for live-cell imaging consisted of 150 mM NaCl, 5 mM KCl, 2 mM CaCl2, 1.2 mM MgCl2, 10 mM HEPES, and 11 mM glucose titrated to pH 7.2 with 1 M NaOH.
Live Cell Experiments
Approximately 5 mL of culture media containing suspended norepinephrine cells or epinephrine cells were centrifuged at 1000 rpm for 5 min. The pellet was resuspended in 1 mL Dulbecco’s modified Eagle’s medium (DMEM). Next, the cells were incubated on a 35 mm Petri dish with either 0.1 μM (for live cell experiments) or 1 μM sensor (for fixed cell experiments) in a humidified incubator at 37 °C with 5% CO2 for 30 min. Norepinephrine cells or epinephrine cells were transferred into 15 mL conical tubes, spun, and washed twice with prewarmed PBS (DPBS, Gibco, Invitrogen, Grand Island, NY). Next, the cells were resuspended in prewarmed chromaffin cell regular medium (DMEM supplemented with 10% (v/v) fetal bovine serum and 1% penicillin/streptomycin). The cells were plated onto 0.0025% poly(l-lysine) coated coverslips within 35 mm Petri dishes and incubated at 37 °C with 5% CO2 for 10 min to promote cell adhesion to the coverslips. The coverslip with norepinephrine cells or epinephrine cells was mounted onto the stage of Olympus Optical FluoView FV1000 confocal laser scanning biological microscope. A standard cell bath solution was added, and the images were acquired using a laser with a wavelength of either 488 or 440 nm.
Fixed Cell Experiments
NE-enriched cells, EP-enriched cells and mixed cells were stained with NeuroSensor 521 (1 μM) and plated on 0.0025% poly(l-lysine) coated coverslips. After three washes with prewarmed phosphate-buffered saline (pH = 7.4, PBS), the cells were fixed in 4% paraformaldehyde (Sigma, St. Louis, MO, USA) in PBS for 60 min at room temperature (RT), followed by 6 min in methanol chilled to −20 °C. Cells were incubated at 4 °C with anti-PNMT antibody (1:1000 dilution, Millipore, Temecula, CA) in PBS with 3% BSA and 0.1% Triton X-100.15 After three washes in PBS, bound antibody was detected using Cy3-conjugated AffiniPure donkey anti-rabbit antibody (1:150 dilution, Jackson ImmunoResearch Laboratories, Inc., Nest Grove, PA). Cells were finally washed three times with PBS. NE-enriched cells, EP-enriched cells, and mixed cells on the coverslips were mounted onto the stage of an Olympus Optical Fluoview FV1000 confocal laser scanning biological microscope, prolong gold antifade mounting medium (Invitrogen, Eugene, Oregon) was added, and then the images were acquired. Excitation and emission wavelength of 488 nm and Qdot525 were used for NeuroSensor 521, while 559 nm and Qdot585 were used for PNMT.
Supporting Information Available
Experimental procedures for the preparation of NeuroSensor 521. This material is available free of charge via the Internet at http://pubs.acs.org.
Author Contributions
K.S.H. and T.E.G. designed, prepared, and performed initial spectroscopic characterization of NeuroSensor 521. X.L. and K.D.G. performed the cell staining and imaging.
This work was supported by the NSF (CHE-1112194) and the NIH (R43 MH096650).
The authors declare no competing financial interest.
Funding Statement
National Institutes of Health, United States
Supplementary Material
References
- a Chen Y.; Zhang K.; Wen G.; Rao F.; Sanchez A. P.; Wang L.; Rodriguez-Flores J. L.; Mahata M.; Mahata S. K.; Waalen J.; Ziegler M. G.; Hamilton B. A.; O’Connor D. T. (2011) Human dopamine β-hydroxylase promoter variant alters transcription in chromaffin cells, enzyme secretion, and blood pressure. Am. J. Hypertens. 24, 24–32. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Omiatek D. M.; Dong Y.; Heien M. L.; Ewing A. G. (2010) Only a fraction of quantal content is released during exocytosis as revealed by electrochemical cytometry of secretory vesicles. ACS Chem. Neurosci. 1, 234–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- a Stevens D. R.; Schirra C.; Becherer U.; Rettig J. (2011) Vesicle pools: lessons from adrenal chromaffin cells. Front. Synaptic Neurosci. 3, 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Kalimuthu P.; John S. A. (2011) Selective determination of norepinephrine in the presence of ascorbic and uric acids an ultrathin polymer film modified electrode. Electrochim. Acta 56, 2428–2432. [Google Scholar]
- a Camacho M.; Machado J. D.; Alvarez J.; Borges R. (2008) Intravesicular calcium release mediates the motion and exocytosis of secretory organelles. J. Biol. Chem. 283, 22383–22389. [DOI] [PubMed] [Google Scholar]; b Zhang K.; Chen Y.; Wen G.; Mahata M.; Rao F.; Fung M. M.; Vaingankar S.; Biswas N.; Gayen J. R.; Friese R. S.; Mahata S. K.; Hamilton B. A.; O’Connor D. T. (2011) Catecholamine storage vesicles: role of core protein genetic polymorphisms in hypertension. Curr. Hypertens. Rep. 13, 36–45. [DOI] [PMC free article] [PubMed] [Google Scholar]; c Jankowski J. A.; Schroeder T. J.; Ciolkowski E. L.; Wightman R. M. (1993) Temporal characteristics of quantal secretion of catecholamines from adrenal medullary cells. J. Biol. Chem. 268, 14694–14700. [PubMed] [Google Scholar]; d Borges R.; Pereda D.; Beltrán B.; Prunell M.; Rodríguez M.; Machado J. D. (2010) Intravesicular factors controlling exocytosis in chromaffin cells. Cell Mol. Neurobiol. 30, 1359–1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plattner H.; Artalejo A. R.; Neher E. (1997) Ultrastructural organization of bovine chromaffin cell cortex – analysis by cryofixation and morphometry of aspects pertinent to exocytosis. J. Cell Biol. 139(7), 1709–1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- a Ciolkowski E. L.; Cooper B. R.; Jankowski J. A.; Jorgenson J. W.; Wightman R. M. (1992) Direct observation of epinephrine and norepinephrine concentration from individual adrenal medullary chromaffin cells. J. Am. Chem. Soc. 114, 2815–2821. [Google Scholar]; b Gilabert J. A.; Castejón R.; Vargas J. A.; Duránttez A.; Artalejo A. R. (1999) Characterization of adrenal medullary chromaffin cells by flow cytometry. Cytometry 37, 32–40. [PubMed] [Google Scholar]; c Afework M.; Burnstock G. (2005) Changes in P2Y2 receptor localization on adrenaline- and noradrenaline-containing chromaffin cells in the rat adrenal gland during development and aging. Int. J. Dev. Neurosci. 23, 567–573. [DOI] [PubMed] [Google Scholar]
- a Carrera V.; Sabater E.; Vilanova E.; Sogorb M. A. (2007) A simple and rapid HPLC-MS method for the simultaneous determination of epinephrine, norepinephrine, dopamine, and 5-hydroxytryptamine: application to the secretion of bovine chromaffin cell cultures. J. Chromatogr., B 874, 88–94. [DOI] [PubMed] [Google Scholar]; b Robinson D. L.; Hermans A.; Seipel A. T.; Wightman R. M. (2008) Monitoring rapid chemical communication in the brain. Chem. Rev. 108, 2554–2584. [DOI] [PMC free article] [PubMed] [Google Scholar]; c Albillos A.; Dernick G.; Horstmann H.; Almers W.; Alvarez de Toledo G.; Lindau M. (1997) The exocytotic event in chromaffin cells revealed by patch amperometry. Nature 389, 509–512. [DOI] [PubMed] [Google Scholar]; d Stuart J. N.; Hummon A. B.; Sweedler J. V. (2004) The chemistry of thought: neurotransmitters in the brain. Anal. Chem. 76, 120A–128A. [PubMed] [Google Scholar]; e Kim D.; Koseoglu S.; Manning B. M.; Meyer A. F.; Haynes C. L. (2011) Electroanalytical eavesdropping on single cell communication. Anal. Chem. 83, 7242–7249. [DOI] [PMC free article] [PubMed] [Google Scholar]; f Petrovic J.; Walsh P. L.; Thornley K. T.; Miller C. E.; Wightman R. M. (2010) Real-time monitoring of chemical transmission in slices of the murine adrenal gland. Endocrinology 151, 1773–1783. [DOI] [PMC free article] [PubMed] [Google Scholar]; g Mosharov E. V.; Sulzer D. (2005) Analysis of exocytotic events recorded by amperometry. Nat. Methods 2, 651–658. [DOI] [PubMed] [Google Scholar]
- a Gubernator N. G.; Zhang H.; Staal R. G. W.; Mosharov E. V.; Pereira D. B.; Yue M.; Balsanek V.; Vadola P. A.; Mukherjee B.; Edwards R. H.; Sulzer D.; Sames D. (2009) Fluorescent false neurotransmitters visualize dopamine release from individual presynaptic terminals. Science 324, 1441–1444. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Lee M.; Gubernator N. G.; Sulzer D.; Sames D. (2010) Development of pH-responsive fluorescent false neurotransmitters. J. Am. Chem. Soc. 132, 8828–8830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- a Maue M.; Schrader T. (2005) A color sensor for catecholamines. Angew. Chem., Int. Ed. 44, 2265–2270. [DOI] [PubMed] [Google Scholar]; b Liew F. F.; Hasegawa T.; Fukuda M.; Nakata E.; Morii T. (2011) Construction of dopamine sensors by using fluorescent ribonucleopeptide complexes. Bioorg. Med. Chem. 19, 4473–4481. [DOI] [PubMed] [Google Scholar]; d Kolusheva S.; Molt O.; Herm M.; Schrader T.; Jelinek R. (2005) Selective detection of catecholamines by synthetic receptors embedded in chromatic polydiacetylene vesicles. J. Am. Chem. Soc. 127, 10000–10001. [DOI] [PubMed] [Google Scholar]
- Secor K. E.; Glass T. E. (2004) Selective amine recognition: development of a chemosensor for dopamine and norepinephrine. Org. Lett. 6, 3727–3730. [DOI] [PubMed] [Google Scholar]
- Feuster E. K.; Glass T. E. (2003) Detection of amines and unprotected amino acids in aqueous conditions by formation of highly fluorescent iminium ions. J. Am. Chem. Soc. 125, 16174–16175. [DOI] [PubMed] [Google Scholar]
- A pH titration of NeuroSensor 521 with saturating amounts of norepinephrine gave a pKa of 6.8, which is consistent with an imine of this type. See the Supporting Information.
- a Yang Y.; Craig T. J.; Chen X.; Ciufo L. F.; Takahashi M.; Morgan A.; Gillis K. D. (2007) Phosphomimetic mutation of Ser-187 of SNAP-25 increases both syntaxin binding and highly Ca2+-sensitive exocytosis. J. Gen. Physiol. 129, 233–244. [DOI] [PMC free article] [PubMed] [Google Scholar]; b Moro M. A.; Lépez M. G.; Gandía L.; Michelena P.; García G.. (1990) Separation and culture of living adrenaline- and noradrenaline-containing cells from bovine adrenal medullae. Anal. Biochem. 185, 243–248. [DOI] [PubMed] [Google Scholar]
- NeuroSensor 521 reacts with the poly(lysine) coating on standard coverslips, and thus, staining prior to plating is required.
- Moreno A.; SantoDomingo J.; Fonteriz R. I.; Lobatón C. D.; Montero M.; Alvarez J. (2010) A confocal study on the visualization of chromaffin cell secretory vesicles with fluorescent targeted probes and acidic dyes. J. Struct. Biol. 172, 261–269. [DOI] [PubMed] [Google Scholar]
- Cahill A. L.; Eertmoed A. L.; Mangoura D.; Perlman R. L. (1996) Differential regulation of phenylethanolamine N-methyltransferase expression in two distinct subpopulations of bovine chromaffin cells. J. Neurochem. 67, 1217–1224. [DOI] [PubMed] [Google Scholar]
- Rasband W. S. (1997–2012) ImageJ, U.S. National Institutes of Health, Bethesda, Maryland, http://imagej.nih.gov/ij/. [Google Scholar]
- Yang Y.; Craig T. J.; Chen X.; Ciufo L. F.; Takahashi M.; Morgan A.; Gillis K. D. (2007) Phosphomimetic mutation of Ser-187 of SNAP-25 increases both syntaxin binding and highly Ca2+-sensitive exocytosis. J. Gen. Physiol. 129, 233–244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X.; Barizuddin S.; Shin W.; Mathai C. J.; Gangopadhyay S.; Gillis K. D. (2011) Microwell device for targeting single cells to electrochemical microelectrodes for high-throughput amperometric detection of quantal exocytosis. Anal. Chem. 83, 2445–2451. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.