Abstract
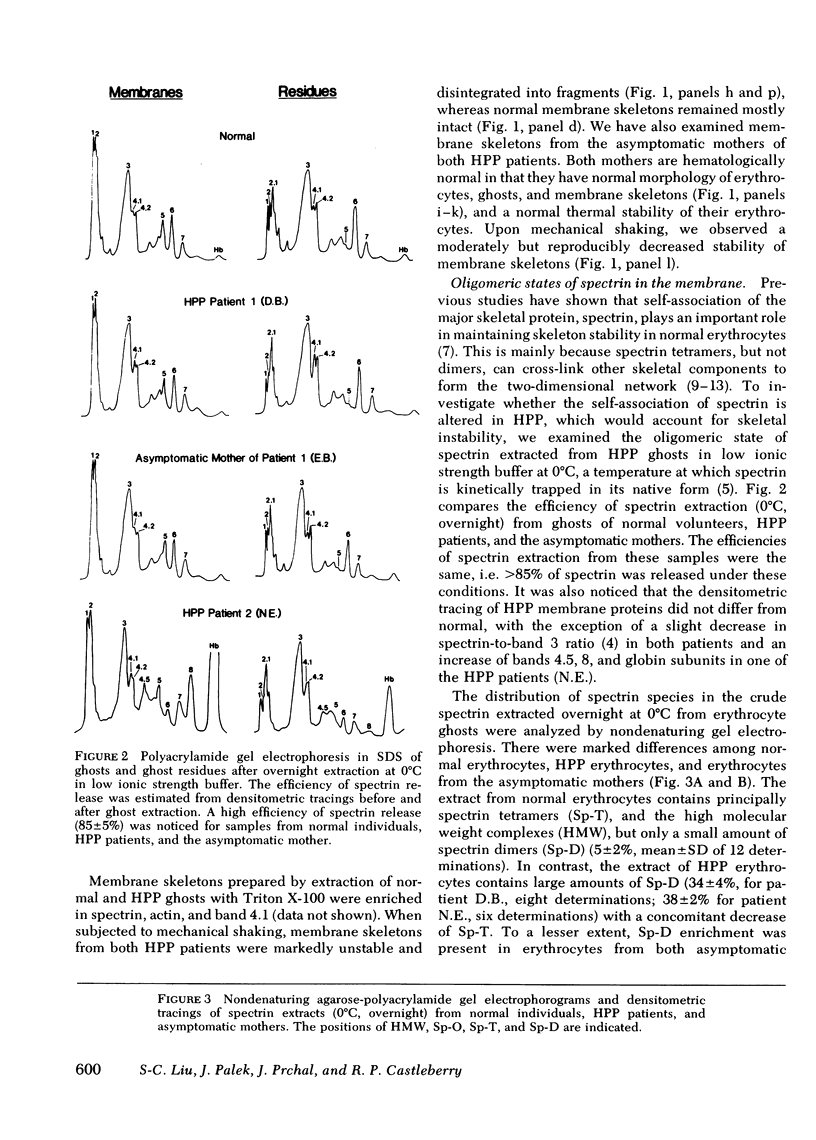
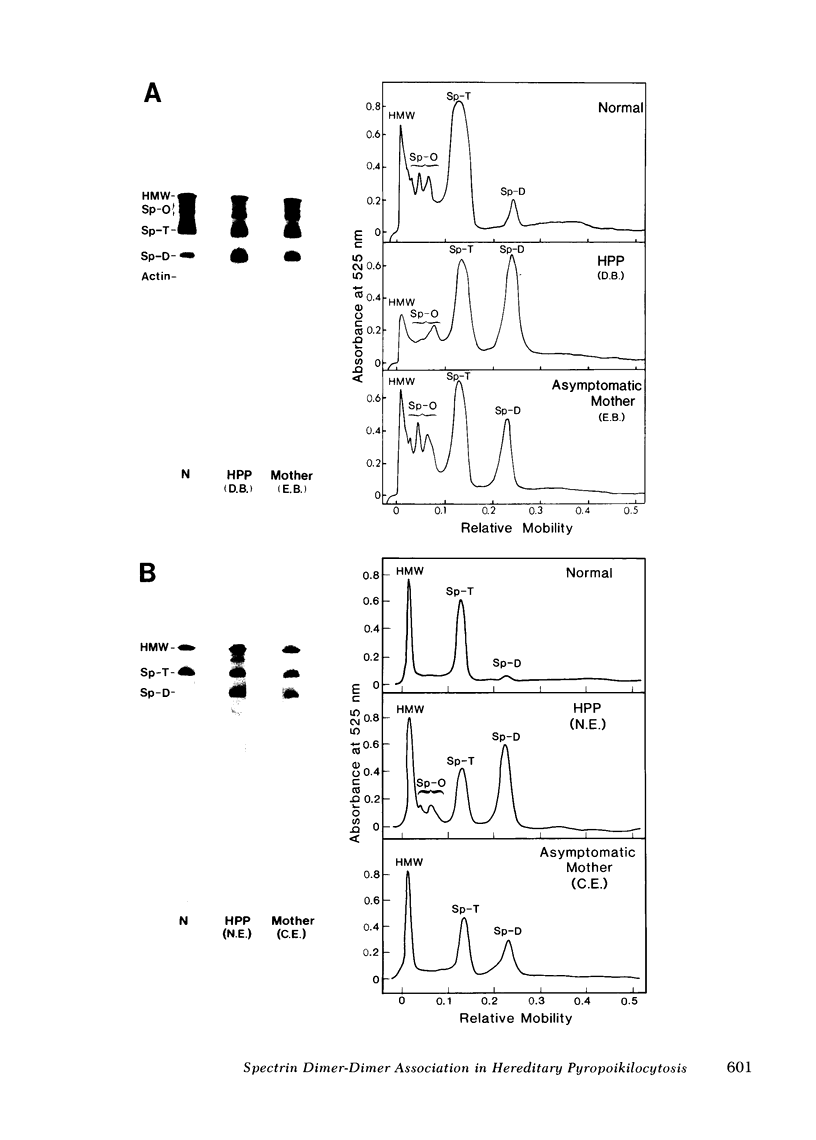
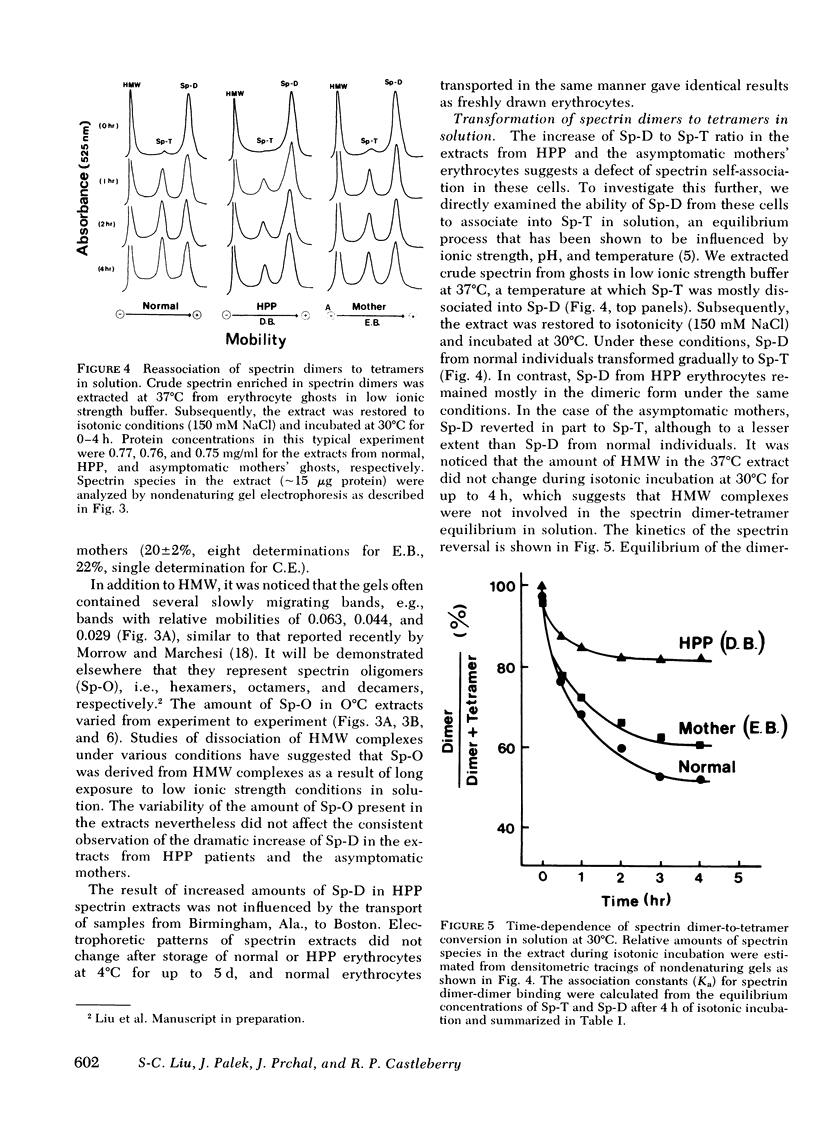
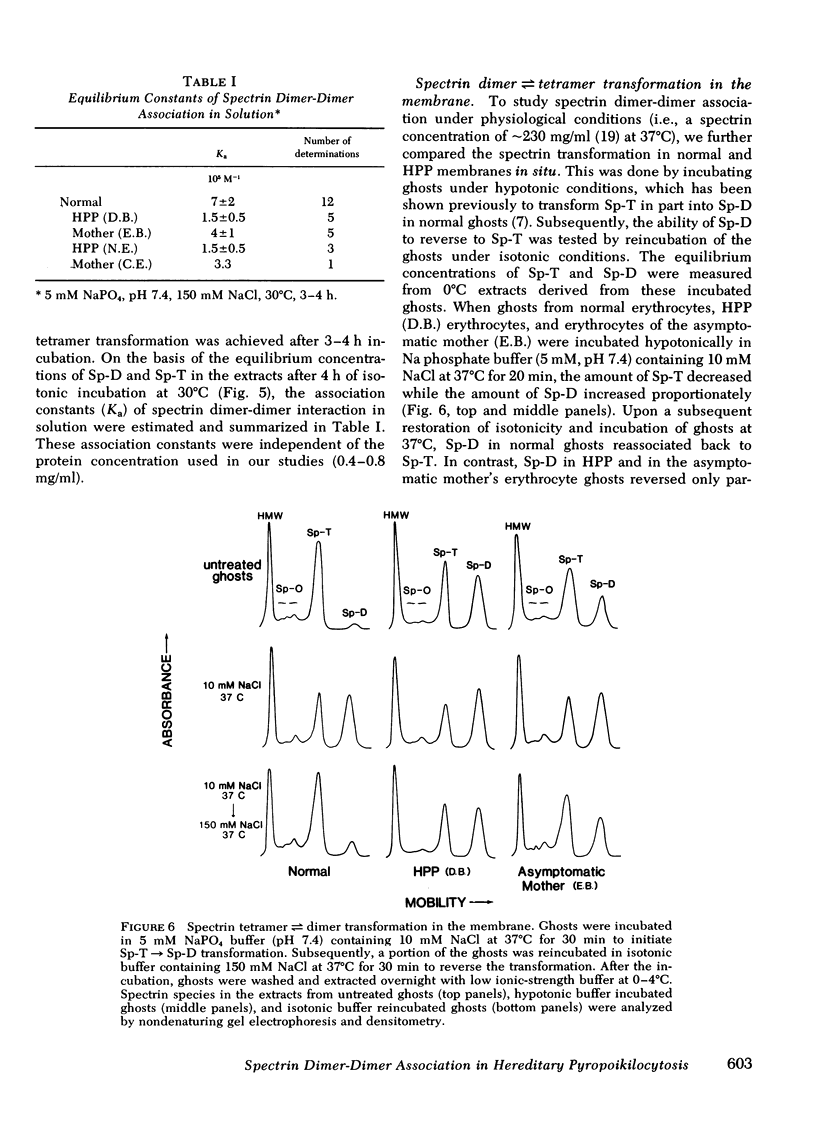
Hereditary pyropoikilocytosis (HPP) is a hemolytic anemia characterized by microspherocytosis, poikilocytosis, and an unusual thermal sensitivity of erythrocytes. We have investigated the contribution of abnormal membrane skeletal assembly to these abnormal HPP erythrocyte properties. Skeletons prepared from fresh HPP ghosts with Triton X-100 were considerably more fragile than skeletons from control erythrocytes. Spectrin, the major skeleton component, extracted at 0 degrees C from normal erythrocytes, was present primarily as tetramers and high molecular weight complexes. In contrast, spectrin extracted from HPP erythrocytes under identical conditions contained a significant amount of dimers with a concomitant decrease of tetramers. Furthermore, spectrin dimers from HPP erythrocytes differed from normal spectrin dimers in their failure to reassociate into tetramers both in solution and in the membrane. Presumptive HPP carriers (asymptomatic mothers of the two patients) exhibited a mild but reproducible increase of spectrin dimers in 0 degrees C extracts and a defective reassociation of spectrin dimers of tetramers both in solution and in the membrane. We conclude that in HPP, self-association of spectrin dimers into tetramers is defective, which accounts for the instability of membrane skeletons.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brenner S. L., Korn E. D. Spectrin-actin interaction. Phosphorylated and dephosphorylated spectrin tetramer cross-link F-actin. J Biol Chem. 1979 Sep 10;254(17):8620–8627. [PubMed] [Google Scholar]
- Chang K., Williamson J. R., Zarkowsky H. S. Effect of heat on the circular dichroism of spectrin in hereditary pyropoikilocytosis. J Clin Invest. 1979 Jul;64(1):326–328. doi: 10.1172/JCI109456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen C. M., Foley S. F. Spectrin-dependent and -independent association of F-actin with the erythrocyte membrane. J Cell Biol. 1980 Aug;86(2):694–698. doi: 10.1083/jcb.86.2.694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DODGE J. T., MITCHELL C., HANAHAN D. J. The preparation and chemical characteristics of hemoglobin-free ghosts of human erythrocytes. Arch Biochem Biophys. 1963 Jan;100:119–130. doi: 10.1016/0003-9861(63)90042-0. [DOI] [PubMed] [Google Scholar]
- Fairbanks G., Steck T. L., Wallach D. F. Electrophoretic analysis of the major polypeptides of the human erythrocyte membrane. Biochemistry. 1971 Jun 22;10(13):2606–2617. doi: 10.1021/bi00789a030. [DOI] [PubMed] [Google Scholar]
- Fowler V., Taylor D. L. Spectrin plus band 4.1 cross-link actin. Regulation by micromolar calcium. J Cell Biol. 1980 May;85(2):361–376. doi: 10.1083/jcb.85.2.361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ji T. H., Kiehm D. J., Middaugh C. R. Presence of spectrin tetramer on the erythrocyte membrane. J Biol Chem. 1980 Apr 10;255(7):2990–2993. [PubMed] [Google Scholar]
- Liu S. C., Fairbanks G., Palek J. Spontaneous, reversible protein cross-linking in the human erythrocyte membrane. Temperature and pH dependence. Biochemistry. 1977 Sep 6;16(18):4066–4074. doi: 10.1021/bi00637a020. [DOI] [PubMed] [Google Scholar]
- Liu S. C., Palek J. Spectrin tetramer-dimer equilibrium and the stability of erythrocyte membrane skeletons. Nature. 1980 Jun 19;285(5766):586–588. doi: 10.1038/285586a0. [DOI] [PubMed] [Google Scholar]
- Lux S. E. Spectrin-actin membrane skeleton of normal and abnormal red blood cells. Semin Hematol. 1979 Jan;16(1):21–51. [PubMed] [Google Scholar]
- Morrow J. S., Marchesi V. T. Self-assembly of spectrin oligomers in vitro: a basis for a dynamic cytoskeleton. J Cell Biol. 1981 Feb;88(2):463–468. doi: 10.1083/jcb.88.2.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palek J., Liu S. C., Liu P. Y., Prchal J., Castleberry R. P. Altered assembly of spectrin in red cell membranes in hereditary pyropoikilocytosis. Blood. 1981 Jan;57(1):130–139. [PubMed] [Google Scholar]
- Sheetz M. P. DNase-I-dependent dissociation of erythrocyte cytoskeletons. J Cell Biol. 1979 Apr;81(1):266–270. doi: 10.1083/jcb.81.1.266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shotton D. M., Burke B. E., Branton D. The molecular structure of human erythrocyte spectrin. Biophysical and electron microscopic studies. J Mol Biol. 1979 Jun 25;131(2):303–329. doi: 10.1016/0022-2836(79)90078-0. [DOI] [PubMed] [Google Scholar]
- Tyler J. M., Hargreaves W. R., Branton D. Purification of two spectrin-binding proteins: biochemical and electron microscopic evidence for site-specific reassociation between spectrin and bands 2.1 and 4.1. Proc Natl Acad Sci U S A. 1979 Oct;76(10):5192–5196. doi: 10.1073/pnas.76.10.5192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ungewickell E., Bennett P. M., Calvert R., Ohanian V., Gratzer W. B. In vitro formation of a complex between cytoskeletal proteins of the human erythrocyte. Nature. 1979 Aug 30;280(5725):811–814. doi: 10.1038/280811a0. [DOI] [PubMed] [Google Scholar]
- Ungewickell E., Gratzer W. Self-association of human spectrin. A thermodynamic and kinetic study. Eur J Biochem. 1978 Aug 1;88(2):379–385. doi: 10.1111/j.1432-1033.1978.tb12459.x. [DOI] [PubMed] [Google Scholar]
- Zarkowsky H. S. Heat-induced erythrocyte fragmentation in neonatal elliptocytosis. Br J Haematol. 1979 Apr;41(4):515–518. doi: 10.1111/j.1365-2141.1979.tb05889.x. [DOI] [PubMed] [Google Scholar]
- Zarkowsky H. S., Mohandas N., Speaker C. B., Shohet S. B. A congenital haemolytic anaemia with thermal sensitivity of the erythrocyte membrane. Br J Haematol. 1975 Apr;29(4):537–543. doi: 10.1111/j.1365-2141.1975.tb02740.x. [DOI] [PubMed] [Google Scholar]





