Abstract
Stromal factors play a critical role in the development of the mammary gland. Using a three dimensional-coculture model we demonstrate a significant role for stromal fibroblasts in the regulation of normal mammary epithelial morphogenesis and the control of tumor growth. Both soluble factors secreted by fibroblasts and fibroblast-derived modifications of the matrix compliance contribute to the regulation of epithelial cell morphogenesis. Readjustment of matrix tension by fibroblasts can even induce a phenotypic reversion of breast carcinoma cells. These data offer a basis to develop new strategies for the normalization of the tumor stroma as an innovative target in cancer therapy.
Keywords: Mammary fibroblasts, Microenvironment, Breast cancer, Acinar morphogenesis, Matrix compliance
1. Introduction
The normal mammary gland, formed by the branching epithelium and the surrounding microenvironment, is composed of a multitude of cell types that participate in a complex functional network. Cell–matrix and cell–cell interactions play important roles in the development of the normal mammary gland and in tumorigenesis. Our understanding of tumorigenesis has progressed enormously over the past several years. Historically, cancer research has focused mainly on tumor cells themselves whereas recent findings have highlighted the significance of interactions between epithelial cells and the surrounding microenvironment. Bissell et al. demonstrated that stromal factors influence the differentiation and morphogenesis of the normal mammary gland as well as tumor growth and progression [1]. Furthermore, it is known that perturbations in the microenvironment are associated with breast cancer and that the restoration of a normal tissue architecture can revert the malignant phenotype [1,2].
The microenvironment accounts for nearly 80% of the breast volume and is composed of extracellular matrix (ECM) and stromal cells including inflammatory cells, endothelial cells and fibroblasts [3]. Known functions of fibroblasts include the deposition of the ECM and the regulation of differentiation of the neighboring epithelium [4,5]. Tumor-associated fibroblasts differ from normal mammary fibroblasts with respect to growth factor expression and growth behavior [6–8]. Recent findings imply a key role for fibroblasts in tumorigenesis, however, the underlying mechanism remains incompletely understood.
Apart from paracrine fibroblast-derived signals, mechanical tension of the microenvironmental ECM can regulate carcinoma cell growth behavior. Boyd et al. described an association between mammographic density and breast cancer risk, and Paszek et al. found that increasing the ECM rigidity leads to perturbation of tissue architecture and enhances tumor growth [9,10]. Due to their contractile ability stromal fibroblasts are able to remodel the ECM leading to alteration of matrix compliance.
Multifaceted microenvironmental variables, such as paracrine cell-stroma interactions, ECM composition and compliance are difficult to replicate in cell culture systems. Three-dimensional (3D)-cell culture approximates the architecture of the mammary gland in vivo and provides the functional context to study reciprocal signaling between epithelium and its microenvironment that controls differentiation and tumorigenesis.
The aim of our present study was to investigate the role of mammary stromal fibroblasts in regulating differentiation and proliferation of mammary epithelial cells in a 3D context. Using normal and cancer-associated fibroblasts and normal and tumorigenic breast epithelial cells we show that stromal fibroblasts are able to control morphogenesis independent of their original background. Furthermore, we demonstrate that modifications of matrix compliance by stromal factors contribute to fibroblast-induced morphogenesis and control growth behavior of breast carcinoma cells.
2. Materials and methods
2.1. Tissue samples and isolation of primary fibroblasts
Tissue was obtained with approval from the Institutional Review Board of the University Medical Center Schleswig–Holstein, Campus Kiel. Fresh surgicalspecimens (mastectomies) were available from nine patients with primary invasive breastcarcinomas and from three reduction mammoplasty. Patients did not receive any systemictherapy prior to surgery and were enrolled in the study if they provided written informed consent. Primary cancer associated fibroblast (CAF) were isolated from grossly recognizable tumors and primary normal mammary fibroblasts (NF) were isolated from adjacent normal breast tissue or from reduction mammoplasty specimens. Hematoxylin and eosin-stained frozen sections were prepared from each tissue sample to confirm benignity or malignancy. Primary fibroblasts were isolated as previously described [8].
2.2. Culture and characterization of primary fibroblasts
CAF and NF were routinely maintained in DMEM and 10% fetal bovine serum at 37 °C in a humidified atmosphere containing 5% CO2. After one to two passages, epithelial cells had disappeared from confluent fibroblasts cultures and fibroblasts were used for the preparation of 3D-cultures. If sufficient numbers of fibroblasts remained after preparation of 3D-cultures, they were used for further characterization by immunoflourescence. The purity of the fibroblasts cultures was confirmed by immunolabeling for the epithelial cell marker pancytokeratin (rabbit; 1:100; abcam) and the mesenchymal marker vimentin (rabbit; 1:100; LabVision). For further characterization, the fibroblast cultures were labeled with an antibody against a-SMA (mouse; 1:100; LabVision).
2.3. Cell lines
The spontaneously immortalized nonmalignant human breast epithelial cell line HMT-3522 (S1 cells) was kindly provided by O.W. Petersen (Department of Cellular and Molecular Medicine, Panum Institute, University of Copenhagen, Denmark) [11]. S1 cells were grown in H14-medium consisting of DME-F12 medium (HyClone, Thermo Fisher Scientific, Bonn, Germany), containing 250 ng/ml insulin, 10 µg/ml transferrin, 2.6 ng/ml sodium selenite, 1010 M estradiol, 1.4×10−6 M hydrocortison, 5 µg/ml prolactin, 10 ng/ml epidermal growth factor (EGF) (all from SIGMA-Aldrich Chemie GmbH, Steinheim, Germany), and penicillin–streptomycin (Biochrom AG, Berlin, Germany). The spontaneously transformed and tumorigenic human breast epithelial line HMT-3522 T4–2 was routinely grown in H14 medium without epidermal growth factor on collagen-coated plates. Immortalized normal human mammary fibroblasts (HMF) labeled with green fluorescent protein were used in cocultures [12]. HMF were cultured on plastic in DMEM (Invitrogen GmbH, Darmstadt, Germany) with 10% fetal bovine serum (Biochrom AG, Berlin, Germany) supplemented with streptomycin and penicillin.
2.4. Three-dimensional cell culture
3D-cell cultures were routinely prepared by trypsinization of cells followed by embedding in collagen type I (BD Biosciences, Heidelberg, Germany) gels with a final collagen concentration of 1.3 mg/ml. To conduct experiments of decreasing and increasing matrix rigidity, collagen concentrations of 1.0 mg/ml, 1.5 mg/ml, 2.0 mg/ ml and 3.0 mg/ml were used. Monocultures consisted of 0.1 × 106 S1 cells/ml or 0.07 × 106 T4–2 cells/ml. In cocultures, epithelial cells and fibroblasts were used at a ratio of 1:2 (0.2 × 106 or 0.14 × 106 HMF/ml; 0.2 × 106 NF or CAF/ml). 750 µl of the gel-cell-mix were dispensed into each well of a 12-well plate (Sarstedt AG & Co., Nürnberg, Germany). 3D-cultures were maintained in H14-medium at 37 °C in a humidified atmosphere containing 5% CO2 for 9–12 days. Medium was changed every other day.
Experiments with conditioned media were conducted using the same protocol as described above, except the gels were maintained in conditioned media derived from HMF. 3D-cultures with H14-Medium served as control. To generate conditioned media, HMF were plated in H14 medium and grown to 50–80% of confluence. To remove remaining trypsin, medium was changed for a minimum of 2–3 times. Then fibroblasts were cultured for an additional 72 h, medium was removed and centrifuged to remove cell debris.
For floating gel experiments, cells were cultured in collagen type I gels at a final collagen concentration of 1.3 mg/ml as described above. When gels became attached, they were detached from the sides and bottom of the dish and were maintained in H14-medium at 37 °C in a humidified atmosphere containing 5% CO2 for 7–10 days. As a control, one set of gels was left attached to the dish.
2.5. Immunofluorescence analysis and image acquisition
After 9–12 days of culture, gels were fixed and stained as described previously [13]. The following primary antibodies were used: Rabbit polyclonal anti-cytokeratin wide spectrum AK (1:100; abcam, Berlin, Germany), mouse monoclonal anti-cytokeratin (1:100; Medac GmbH, Wedel, Germany), rabbit monoclonal anti-Ki-67 (1:100; Labvision/Thermo Fisher Scientific GmbH, Dreieich, Germany), mouse monoclonal anti-golgin-97 (1:100; Invitrogen GmbH, Darmstadt, Germany), mouse monoclonal anti-p4-integrin (1:50–1:200; BD Biosciences, Heidelberg, Germany), mouse monoclonal anti-p1-integrin (1:200; Labvision/Thermo Fisher Scientific GmbH, Dreieich, Germany), rabbit polyclonal anti-β-catenin (1:25–1:50; Labvision/Thermo Fisher Scientific GmbH, Dreieich, Germany), rabbit polyclonal antivimentin (1:100; Labvision/Thermo Fisher Scientific GmbH, Dreieich, Germany) and rabbit polyclonal anti-ZO-I (1:25; New England Biolabs GmbH, Ipswich Massachusetts, USA). The secondary antibodies Alexa Fluor®488 goat anti-rabbit and Alexa Fluor® 555 goat anti-mouse (Invitrogen GmbH, Darmstadt, Germany) were diluted 1:500 and incubated for 1 h at room temperature. All antibodies were diluted with blocking solution composed of 3% goat serum (DAKO, Hamburg, Germany), 5% PBS (20×) and 92% Milli Qwater. Nuclei were conterstained with DAPI (0.13 µg/ml). Immunofluorescence staining procedure without the use of primary antibodies served as negative controls.
Immunofluorescence images were acquired using an inverted microscope (Axioplan2, Carl Zeiss Jena, Jena, Germany) equipped with a digital camera and Axio Vision Rel.4.6.3 software (Carl Zeiss Jena, Jena, Germany). Confocal analysis was performed using a LSM510 UV Meta confocal microscopy system (Carl Zeiss Jena, Jena, Germany). The images shown are representative of three or more independent experiments.
For analysis of cell morphology, ten visual fields of each sample were analyzed for morphology of cell clusters (inverted microscope; magnification 250x). Polarized acini were defined as structures composed of 4 or more epithelial cells that form a single layer, express apical golgin-97 and surround a small hollow lumen. Disorganized cell clusters are defined as aggregates composed of 4 or more cells with an irregular shape (length > 2 fold width) and randomly distributed golgin-97 protein.
For quantification of cell growth, ten visual fields of each sample were analyzed for total cell area (ratio of all cell areas [pixel2] and total image size [pixel2]) (inverted microscope; magnification: 100×) using customized macros generated in NIH ImageJ 1.40 g (http://rsb.info.nih.gov/ij/). The area size of cell clusters immuno-labeled with pancytokeratin was shown to be significantly correlated with the number of cell nuclei [14]. Thus, total area was used as a surrogate measure of cell number.
2.6. Apoptosis assay
To study apoptosis of normal mammary epithelial cells in 3D-mono- and co-culture gels were fixed after 10 days of culture and epithelial cells were identified by labeling with anti-cytokeratin as described before [13]. Apoptotic cells were visualized using the In Situ Cell Death Detection Kit (POD, Roche Diagnostics, Mannheim, Germany), and nuclei were counterstained with DAPI. For quantification of apoptosis, the apoptosis index of epithelial cells was determined in a visual field. 14 visual fields were evaluated each containing a minimum of 50 epithelial cells (inverted microscope; magnification 630×).
2.7. Statistical analysis
To evaluate differences in morphology and growth of S1 and T4–2 cells in 3D-cocultures and monocultures, a t-test was used. p-Values ≤ 0.05 were considered statistically significant.
2.8. Fluorescence-activated cell sorting
After 9 days of culture, gels were dissolved and cells were released by incubation with 2 mg/ml collagenase VIII (SIGMA-Aldrich) at 37 °C for 20 min. HMF were separated from S1 cells by fluorescence-activated cell sorting based on GFP expression, using the BD FACSAria, equipped with FACSDiva software (BD Biosciences, Heidelberg, Germany). S1 cells and HMF were collected in 1 × PBS. To provide identical conditions, monocultures were subjected to the same procedure. For cell cycle analyses, cells were washed and fixed in 100% ethanol for 30 min at room temperature. Then, cells were incubated with 20 µl RNase A (1 mg/ml) in100 µl PBS/EDTA (5 mM)/propidium iodide (100 µg/ml) at room temperature for 30 min. DNA content was measured by flow cytometry using the BD FACSCalibur (Becton-Dickinson, Heidelberg, Germany).
2.9. 1D-SDS-PAGE and nano-HPLC–ESI-MS/MS
To analyze the composition of conditioned medium, HMF medium was separated by centrifugation through a membrane (Millipore GmbH, Schwalbach/Ts., Germany) in two fractions, fraction A > 3 kDa and fraction B < 3 kDa, respectively. Both fractions were used as cell culture medium in 3D-monocultures. Normal H14 + medium was used as control. In a first step 60 µg (determined by Bradfords assay [15]) of fraction A were separated via 12% 1D-SDS–PAGE and Coomassie stained [16]. The gel lane was sliced in 9 identical gel bands (each 10 × 2.3 mm). After reduction with dithiotreitol and alkylation with iodoacetamide each gel band was digested over night at 37 °C with 50 ng modified, sequencing grade porcine trypsin (Promega, Mannheim, Germany) in 5% (v/v) acetonitrile (ACN)/50 mM ammonium bicarbonate buffer, pH 8. Tryptic peptides were eluted from the gel bands, dried by evaporation in a vacuum concentrator and redissolved in 12 µl 0.1% (v/v) trifluoroacetic acid (TFA), 3% (v/v) ACN in 18.2 MΩ cm water (loading buffer). The digests were separated by reversed-phase liquid chromatography on a UltiMate 3000 nano-HPLC system (Dionex, Germering, Germany) coupled online to an LTQ Orbitrap Velos mass spectrometer (Thermo Fisher Scientific, Bremen, Germany) equipped with a nano electrosprayion source. Briefly, 10 µl redissolved digests were loaded on a PepMap C18 trap column (300 µm i.D × 5 mm; Acclaim PepMap 100 C18,3 µm, 100 Å, Dionex, Germering, Germany) and desalted by washing with loading buffer at a flow rate of 30 µl/min for 6 min. Peptides were separated by a 90 min gradient from 0% to 60% eluent B (eluent A: 0.05% (v/v) formic acid (FA) in 18.2 MΩ cm water; eluent B: 0.1% (v/v) FA, 80% (v/v) ACN in 18.2 MΩ cm water) on a PepMap RSLC analytical column (75 µm i.D.× 15 cm; Acclaim PepMap RSLC C18. 3 µm, 100 Å, Dionex, Germering, Germany) at a flow rate of 250 nl/min. MS scans were acquired in the Orbitrap mass analyzer in the m/z range of 300–1600 at a resolution of 60,000. Up to 15 CID (collision induced dissociation) MS/MS spectra of the most abundant precursors per full-scan with a signal intensity ≥500 and a charge state ≥2 were acquired in the linear ion trap (LTQ Velos). For fragmentation, a normalized collision energy of 35% at an activation Q-value of 25 for 10 ms was used. Fragmented precursors (±10 ppm) were added automatically on an exclusion list for 45 s (size 500 precursor).
Data analysis: Raw MS/MS data of all 9 measurements were merged and searched against a FASTA-database containing all entries for taxonomy human [9606] (www.uniprot.org; build date: 02–17–2011, 97,060 entries) using the proteome discoverer 1.1 software (Thermo Fischer Scientific) with the implemented Sequest-algorithm. Precursor and fragment ion tolerances were set to 10 ppm and 0.8 Da respectively. Two missed cleavages for trypsin were allowed; carbamidomethylation was set as fixed and methionine oxidation as variable modification. For false discovery rate (FDR) calculation Raw MS/MS data were searched against a decoy database, which resulted in Xcores for high confident (p ≤ 0.01) and medium confident peptide identifications (p ≤ 0.05). For a positive identification one protein had to be identified by at least two high confident or three medium confident unique peptides.
3. Results
3.1. Normal mammary fibroblasts regulate growth and differentiation of mammary epithelial cells
Normal mammary epithelial cells undergo a process of acinar morphogenesis regulated by the tissue microenvironment. Components of the microenvironment like the ECM and stromal fibroblasts play a critical role in the regulation of epithelial differentiation [1,2,17]. In three-dimensional cell culture, a laminin-rich reconstituted basement membrane (BM) has been shown to be crucial for establishment and maintenance of apicobasal polarized and growth-arrested acini [18–20].
To investigate the influence of stromal fibroblasts on growth and morphogenesis of mammary epithelial cells, phenotypically normal human non-malignant HMT-3522 cells were cultured as either monoculture or in coculture with normal human mammary fibroblasts in a 3D-collagen I matrix, a defined ECM free of BM components.
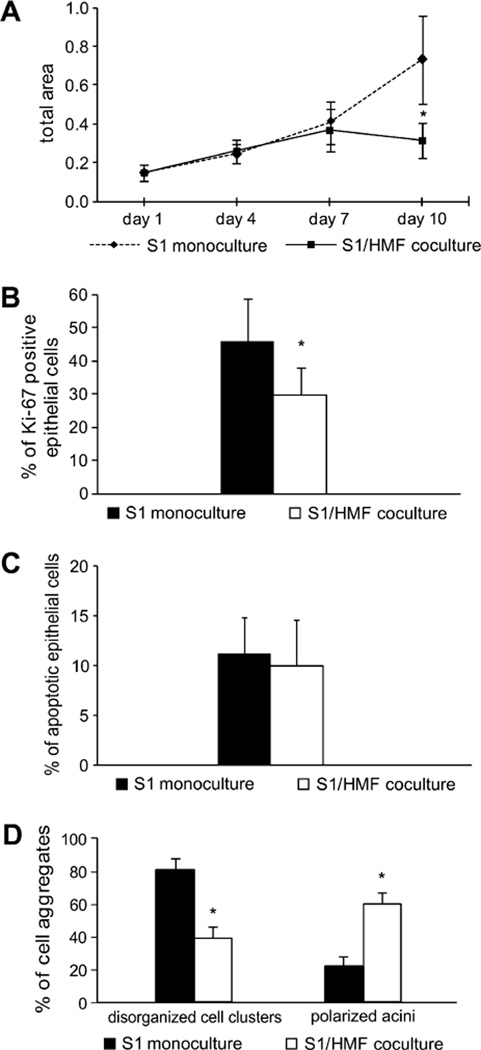
Growth of S1 cells in mono- and cocultures was analyzed at day 1, 4, 7 and 10 after immunolabeling for pancytokeratin. Up to day 7, no significant difference in cell growth was observed between mono- and coculture. While growth of epithelial cells in cocultures plateaued after 7 days of culture, they demonstrated progressive growth in monocultures. At day 10, growth of epithelial cells in monoculture was 2-fold higher than in coculture (p ≤ 0.01) (Fig. 1A). The growth difference was maintained if culture time was extended for up to 20 days (data not shown).
Fig. 1.
Evaluation of growth and morphology of S1 cells in 3D-monocultures and cocultures with HMF. (A) Growth of S1 cells in mono- and coculture in a time course at day 1, 4, 7 and 10. (B) Ki67-proliferation index of S1 cells at day 9 in mono- and coculture was calculated by counting Ki-67 positive cells in 13 visual fields of each gel (inverted microscope; magnification 630×). The Ki67 proliferation index is the ratio of Ki-67 positive nuclei and all nuclei present in a visual field. (C) Apoptotic index (AI) of S1 cells after 10 days of culture in 3D-mono- and coculture. The AI was determined using the TUNEL method. 14 visual fields were evaluated each containing at least 50 epithelial cells (inverted microscope, magnification 630×). (D) Morphology of S1 cells in monoculture and in coculture with HMF was evaluated by analyzing disorganized multicellular cell clusters and polarized acini. The diagrams are representative for more than three independent experiments. Asterisk indicates statistical significance compared to monoculture (p ≤ 0.01).
To ascertain that the presence of mammary fibroblasts leads to an inhibition of epithelial cell proliferation, we labeled the cultures with the proliferation marker Ki-67. At day nine, the fraction of Ki67-positve epithelial cells was significantly higher in monocultures than in cocultures (p ≤ 0.01) (Fig. 1B). To confirm the growth data, 3D-gels were analyzed after 9 days of culture in some experiments and S1 cells were quantified by FACS, revealing a 1.9 to 2.6-fold higher number of epithelial cells in monoculture than in coculture (Additional file 1).
Additionally, we asked whether increased apoptosis would contribute to the reduced growth of epithelial cells observed in 3D-cocultures. Using a TUNEL-assay, no difference in the number of apoptotic cells could be detected under both culture conditions. In monocultures, the apoptotic index was 11.2, whereas apoptosis could be detected in 9.95% of S1 cells in coculture (p = 0.42) (Fig. 1C). Thus, increased apoptosis does not account for decreased growth of S1 cells in 3D-coculture.
Assessment of fibroblast growth in coculture and HMF-monoculture did not reveal a significant increase in cell number after 10–12 days of 3D-culture and HMF did not show positive staining for Ki-67 (data not shown). Furthermore, proliferation of HMF in cocultures was evaluated performing cell-cycle-analysis per FACS. After 9 days of culture about 95% of HMF in cocultures rested in G1/G0-phase suggesting proliferation arrest of fibroblasts in cocultures.
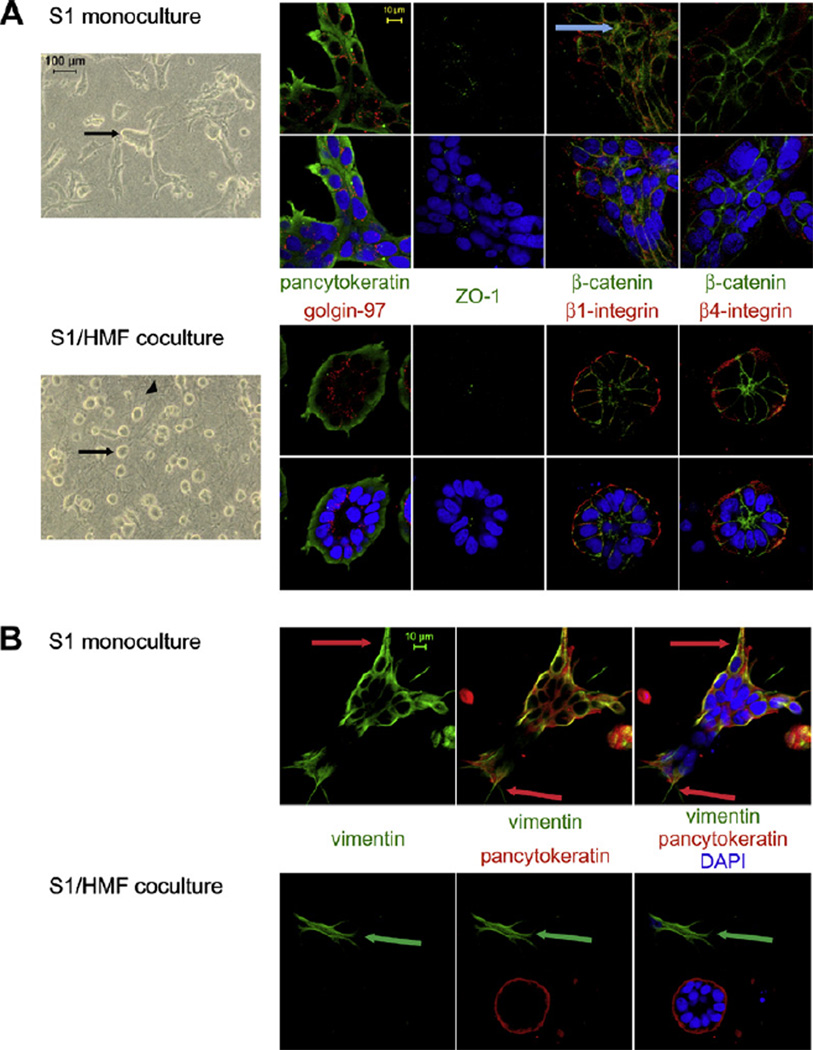
Apart from the different growth behavior, S1 cells growing in coculture with HMF showed distinct morphological differences compared to monoculture. After 9–10 days of 3D-culture the majority of epithelial cells in coculture underwent acinar morphogenesis resulting in formation of well-ordered acini-like spheroids. In monocultures, only 20% of cell colonies showed acini-like morphology compared to more than 60% observed in cocultures (p ≤ 0.01) (Fig. 1D). The spheroids were composed of a single layer of epithelial cells surrounding a small hollow lumen (Fig. 2A). In contrast, S1 cells in monoculture mostly formed large, proliferative, disorganized colonies or were present as single cells (Fig. 2A). Interestingly, some of these aggregates showed cord-like structures, but in contrast to accomplished tubulogenesis [21] these structures were lacking a hollow lumen (Additional file 2).
Fig. 2.
Morphology of S1 cells in 3D-mono- and cocultures. (A) Morphology of S1 cells (arrow) in monoculture (upper panel) compared with S1 cells in coculture (lower panel) with HMF (arrowhead) as shown by phase contrast microscopy (column 1) and immunoflourescence staining (columns 2–5). Cells were labeled for apical (golgin-97 (red) and ZO-1 (green)), basal (β4-integrin (red)), basolateral (β1-integrin (red)) and lateral (β-catenin (green)) polarity markers and epithelial (pancytoceratin (green)) markers. A nuclear counterstain (DAPI (blue)) was employed. Blue arrow: cytoplasmic expression of β-catenin in monoculture. (B) Vimentin (green) expression is upregulated in S1 cells in monoculture (upper panel) compared to coculture (lower panel). Epithelial marker pancytokeratin (red). Red arrow: cytoplasmic process formed by epithelial cells in monoculture. Green arrow: Vimentin positive fibroblasts in coculture. Nuclear counterstain (DAPI (blue)).
Spheroids of S1 cells in cocultures showed apicobasal polarization of the cellular axis. Polarization was indicated by apical expression of a golgi marker (golgin-97) and basally localized β4-integrin. β1-integrin was expressed at the basolateral surface (Fig. 2A). In monocultures, disorganized colonies contained randomly distributed golgin-97, and β1- and β4-integrin were present on the cell membrane circumferentially (Fig. 2A). Integrin expression was decreased in monocultures compared to cocultures.
The morphology of cell clusters in mono- and cocultures was further characterized by immunolabeling for β-catenin and ZO-I. Approximately 95% of epithelial cells in cocultures showed apicolateral expression of β-catenin (Fig. 2A). In monocultures, β-catenin expression was found to be decreased compared to the cocultures. Furthermore, disorganized cell clusters displayed cortical and, less frequently, cytoplasmic localization of β-catenin. ZO-I showed weak expression under both culture conditions. In cocultures, acinus-like spheroids displayed apical expression of ZO-I whereas in monocultures expression was more random (Fig. 2A).
Since growth of S1 cells in monocultures in collagen is characterized by unrestrained proliferation and increased morphological disorganization, the epithelial cells resemble a carcinoma-like growth pattern [22]. Additionally, S1 cells in monoculture have an elongated shape with cytoplasmic extensions reminiscent of mesenchymal cell types (Fig. 2B). Therefore we hypothesized that S1 cells in monocultures may undergo epithelial-mensenchymal transition (EMT) as is often observed in cancer cells [23]. Vimentin is a commonly used molecular marker for EMT [24,25]. Furthermore, when establishing and characterizing the HMT-3522 cell line, Briand et al. showed positive vimentin expression in S1 cells in 2D culture on plastic [26].
To investigate the effect of HMF on vimentin expression in S1 cells, 3D-gels from mono- and cocultures were labeled with antibodies to vimentin. In monocultures 100% of S1 cells showed strong vimentin expression. Surprisingly, S1 cells forming polarized acini in coculture with HMF displayed negative or very weak vimentin staining, whereas epithelial cells growing in disorganized structures showed positive vimentin expression even in cocultures (Fig. 2B). Nuclear expression of β-catenin, an additional marker for EMT, could not be observed under either culture conditions.
These data demonstrate that non-malignant mammary epithelial cells establish acinar morphogenesis in a collagen-I matrix in the presence of normal mammary fibroblasts, whereas in monoculture, in 3D-collagen gels, they form proliferative, apolar structures reminiscent of malignant tumors in vivo.
3.2. HMF stimulate growth of tumorigenic T4–2 cells but do not influence morphogenesis
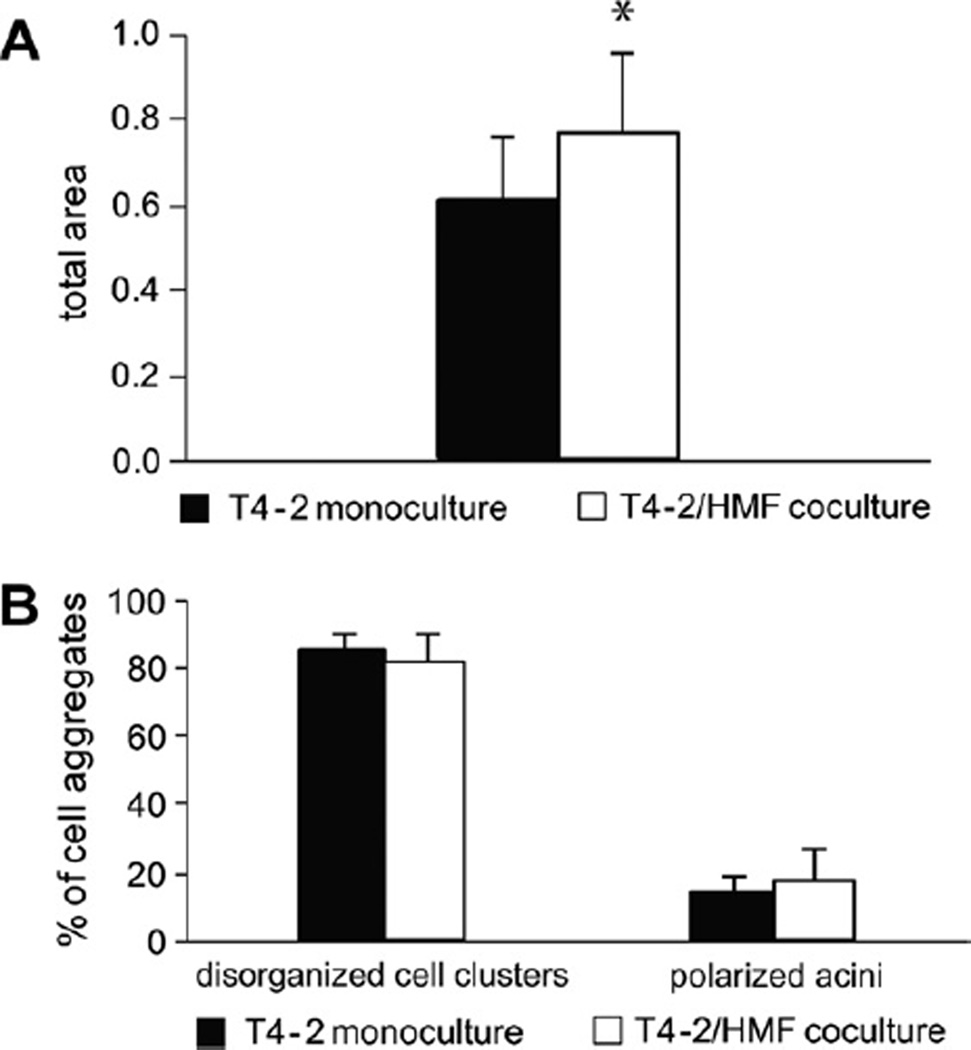
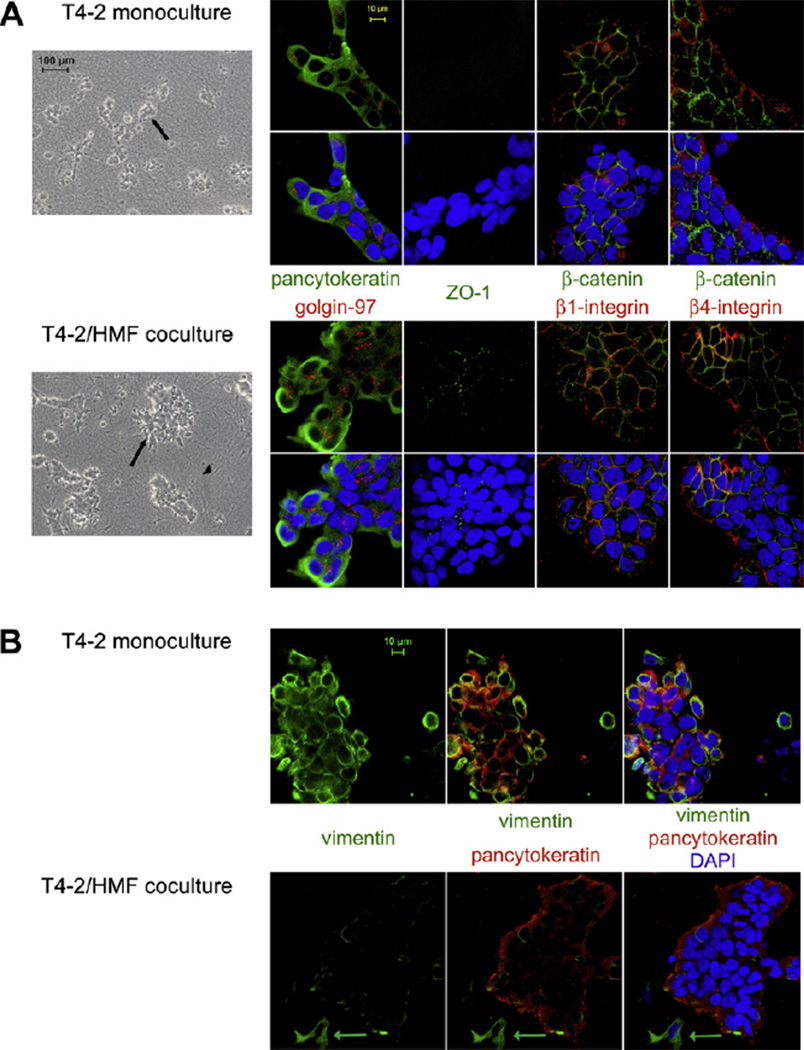
In view of our findings that HMF regulate growth and differentiation of non-malignant breast epithelial cells, we sought to examine the influence of normal mammary fibroblasts on growth behavior of breast carcinoma cells in a 3D-collagen I matrix. Therefore, we took advantage of the HMT-3522 breast tumor progression series. Tumorigenic T4–2 cells, generated from the nontumorigenic parental line HMT-3522 by withdrawal of epidermal growth factor, were used as malignant derivative of S1 cells [26]. Cells were cultured for 4–8 days in mono- and coculture with HMF and growth and morphology were analyzed. In contrast to S1/ HMF cocultures, growth of T4–2 cells in cocultures was about 30% increased compared to monocultures (p ≤ 0.05) (Fig. 3A). No difference in morphology of T4–2 cells grown in mono- or cocultures was observed. Carcinoma cells formed disorganized, continuously growing structures in both mono- and coculture. Less than 20% of T4–2 cells underwent acinar morphogenesis (Fig. 3B). Disorganized cell colonies as observed in mono- and cocultures showed a diffuse distribution of golgin-97 and ZO-I. Expression of both differentiation markers was reduced in monocultures (Fig. 4A). β1-and β4-integrin showed membraneous distribution and β-catenin assumed a cortical location in mono- and cocultures. As observed with S1 cells, expression of β-catenin was increased in cocultures (Fig. 4A). All T4–2 cells in mono- and cocultures were vimentin positive but expression was clearly decreased in cocultures (Fig. 4B). No nuclear expression of β-catenin could be detected in either coor monoculture.
Fig. 3.
HMF stimulate growth of T4-2 cells but do not influence morphogenesis. (A) Growth of T4-2 cells in 3D-monoculture and coculture with HMF after 6 days of culture. Growth in coculture was significantly increased compared to monoculture (*p < 0.05). (B) Assessment of morphology of T4-2 cells in 3D-monoculture and coculture. In both mono- and coculture T4-2 cells showed disorganized growth behavior.
Fig. 4.
Morphology of T4–2 cells in 3D-mono- and cocultures. (A) Morphology of T4–2 cells in monoculture (upper panel) compared with T4–2 cells in coculture (lower panel) with HMF (arrowhead) as shown by phasecontrast microscopy (column 1) and immunoflourescence staining (columns 2–5). Cells were labeled for apical (golgin-97 (red) and ZO-1 (green)), basal (4-integrin (red)), basolateral (1-integrin (red)) and lateral (-catenin (green)) polarity markers and epithelial markers (pancytoceratin (green)). (B) The mesenchymal marker vimentin (green) is downregulated in T4–2 cells in coculture with HMF (lower panel) compared to monoculture (upper panel). Cells were labeled with the epithelial marker pancytoeratin (red). Nuclear counterstain (DAPI (blue)). Green arrow: Vimentin positive fibroblast in coculture.
Thus, in contrast to cocultures with non-malignant breast epithelial cells, HMF stimulate growth but do not regulate morphology of tumorigenic mammary epithelial cells.
3.3. Ability of stromal fibroblasts to control growth and morphogenesis of mammary epithelial cells does not depend on their origin
Fibroblasts are heterogeneous in terms of gene expression and function [27]. To examine whether the fibroblast type determines the response of epithelial cells in coculture, we grew S1 cells in cocultures with primary cancer associated fibroblasts or primary normal human mammary fibroblasts. Cocultures of S1 with HMF served as controls.
Fibroblasts were isolated from breast carcinomas, from adjacent normal breast tissue or normal breast tissue obtained from reduction mammoplasties. Tissue samples from four patients allowed isolation of both CAF and NF (patient ID ANF/CAF, BNF/CAF, CNF/CAF, DNF/CAF), whereas four additional samples served for isolation of either NF (patient ID ENF, FNF, GNF, HNF) or CAF (patient ID ICAF, JCAF, KCAF, LCAF).
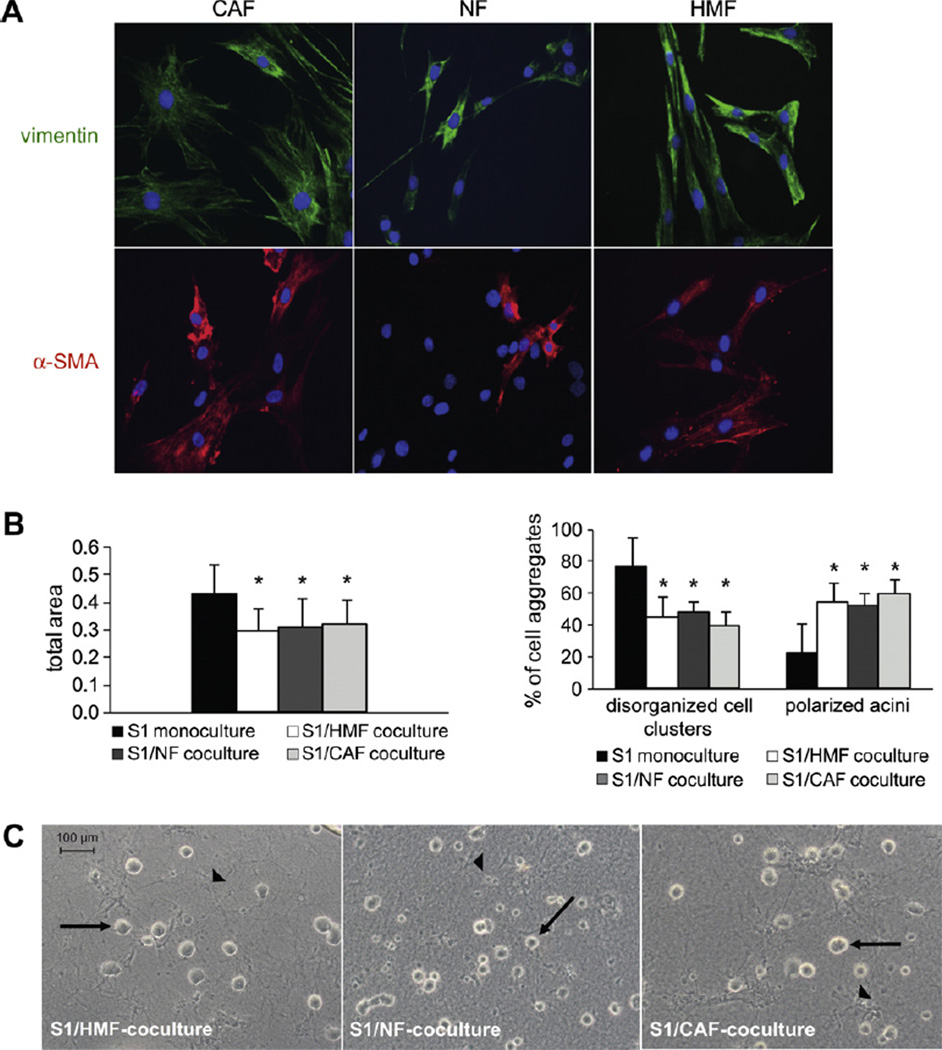
Labeling with antibodies to pancytokeratin and vimentin was performed to assure the purity of fibroblast cultures. Cultures were universally positive for vimentin, whereas no expression of cytokeratin was found, confirming the absence of epithelial contamination (Fig. 5A and not shown). For further characterization of fibroblasts, immunoflourescent labeling for the myofibroblast marker a-smooth muscle actin (α-SMA) was performed (Fig. 5A; Tables 1 and 2). On average, the frequency of α-SMA positive cells was higher in CAF than in NF. Furthermore, the intensity of α-SMA expression was increased in CAF compared to NF. HMF cultured on slides were entirely positive for vimentin and α-SMA. Additionally, CAF and NF showed differences in cell shape (Fig. 5A).
Fig. 5.
Influence of NFand CAFon differentiation of mammary epithelial cells. (A) Characterization of fibroblasts: Fibroblasts isolated from human breast tissue samples and HMF were charactetrized by immunolabeling for mesenchymal marker vimentin (upper panel (green)) and myofibroblast marker α-SMA (lower panel (red)). Note that CAF show heterogenous morphology, whereas NF and HMF are more uniform. Nuclear counterstain (DAPI (blue)). Original magnification: 250x for all images. (B) Assessment of growth (left bar graph) and morphology (right bar graph) of S1 cells in 3D-monoculture and in coculture with either CAF, NF or HMF. Growth in all three cocultures was significantly decreased compared to monoculture (*p < 0.05). In cocultures, number of apicobasal polarized glandular structures was significantly increased compared to monoculture (*p < 0.05). Diagrams are representative for more than 3 independent experiments. (C) Acinar morphogenesis of S1 cells in coculture with NF, CAF and HMF as shown by phase contrast microscopy. Arrow: glandular structures formed by S1 cells. Arrowhead: fibroblasts.
Table 1.
α-SMA expression in primary normal mammary fibroblasts.
| Patient ID | (%) of α-SMA positive cells |
|---|---|
| ANF/CAF | 1 |
| BNF/CAF | 26 |
| CNF/CAF | 5 |
| DNF/CAF | 30 |
| ENF | 10 |
| FNF | 0 |
Bold, patients with corresponding CAF samples.
Table 2.
α-SMA expression in primary cancer associated fibroblasts.
| Patient ID | (%) of α-SMA positive cells |
|---|---|
| ANF/CAF | 33 |
| BNF/CAF | 89 |
| CNF/CAF | 17 |
| DNF/CAF | NA |
| ICAF | 100 |
| JCAF | 11 |
Bold, patients with corresponding NF samples.
NA, no material for α-SMA staining available.
S1 cells in cocultures with NF showed reduced cell growth at day 9 compared to monoculture in six out of eight cases, whereas growth arrest of S1 cells was induced in five out of eight cocultures with CAF (Fig. 5B; Tables 3 and 4). Notably, in samples of two patients (CNF/CAF and DNF/CAF) neither NF nor CAF influenced growth of S1 cells in cocultures (Tables 3 and 4). Thus, growth of normal breast epithelial cells appears to be influenced by interindividual characteristics of fibroblasts rather than by fibroblast type.
Table 3.
Influence of primary normal mammary fibroblasts on growth and differentiation of S1 cells in 3D-collagen I matrix.
| Patient ID | Induction of growth arrest in coculture with NF at day 9 |
(%) of polarized acini in coculture with NF |
(%) of polarized acini in coculture with HMF |
(%) of polarized acini in monoculture |
|---|---|---|---|---|
| ANF/CAF | + | 59a | 50a | 22 |
| BNF/CAF | + | 64a | 67a | 30 |
| CNF/CAF | − | 26 | 54a | 24 |
| DNF/CAF | − | 52a | 55a | 23 |
| ENF | + | 72a | 51a | 20 |
| FNF | + | 65a | 70a | 41 |
| GNF | + | 44a | 44a | 16 |
| HNF | + | 59a | 60a | 20 |
NF induced growth arrest of S1 cells in coculture compared to monoculture.
NF did not influence growth of S1 cells in coculture compared to monoculture. Bold, patients with corresponding CAF samples.
Significant difference compared to monoculture (p < 0.01).
Table 4.
Influence of primary cancer associated fibroblasts on growth and differentiation of S1 cells in 3D-collagen I matrix.
| Patient-ID | Induction of growth arrest in coculture with CAF at day 9 |
(%) of polarized acini in coculture with CAF |
(%) of polarized acini in coculture with HMF |
(%) of polarized acini in monoculture |
|---|---|---|---|---|
| ANF/CAF | + | 65a | 4a | 28 |
| BNF/CAF | + | 60a | 67a | 30 |
| CNF/CAF | − | 28 | 54a | 24 |
| DNF/CAF | − | 60a | 55a | 23 |
| ICAF | – | 54a | 54a | 26 |
| JCAF | + | 70a | 56a | 30 |
| KCAF | + | 81a | 62a | 17 |
| LCAF | + | 53a | 59a | 33 |
CAF induced growth arrest of S1 cells in coculture compared to monoculture.
CAF did not influence growth of S1 cells in coculture compared to monoculture. Bold, patients with corresponding NF samples.
Significant difference compared to monoculture (p < 0.01).
Furthermore, we examined morphogenesis of S1 cells in cocultures with NF and CAF (Fig. 5B and C). In seven out of eight cases, S1 cells underwent acinar morphogenesis in cocultures with NF (Table 3). S1 cells grown in cocultures with CAF exhibited growth patterns similar to those observed in cocultures with HMF or NF (Fig. 5B and C). In one case, CAF did not induce morphogenesis as S1 cells mainly formed disorganized and apolar colonies in mono- and coculture. Remarkably, NF isolated from the same patient (CNF/CAF) likewise did not promote acinar morphogenesis of S1 cells (Tables 3 and 4).
Hence, morphogenesis of normal breast epithelial cells seems to be induced by different mesenchymal cell types regardless of whether they originate from normal or tumorigenic breast tissue. Therefore, we reasoned that integration of common signals from the microenvironment like fibroblast-derived modifications of the mechanical and biochemical properties of the ECM rather than particular cell specific features account for the establishment of growth-arrested polarized acini in cocultures.
3.4. Mammary epithelial cell differentiation is influenced by fibroblast-induced changes in matrix compliance
It is well known that breast density is strongly associated with the risk of breast cancer [9]. Furthermore, high breast density is associated with an increase of ECM components like collagen [28]. Roeder et al. demonstrated that elevating collagen concentration leads to increased collagen matrix stiffness [29]. Paszek et al. showed that increasing the matrix rigidity inhibits differentiation and enhances growth of mammary epithelial cells [10].
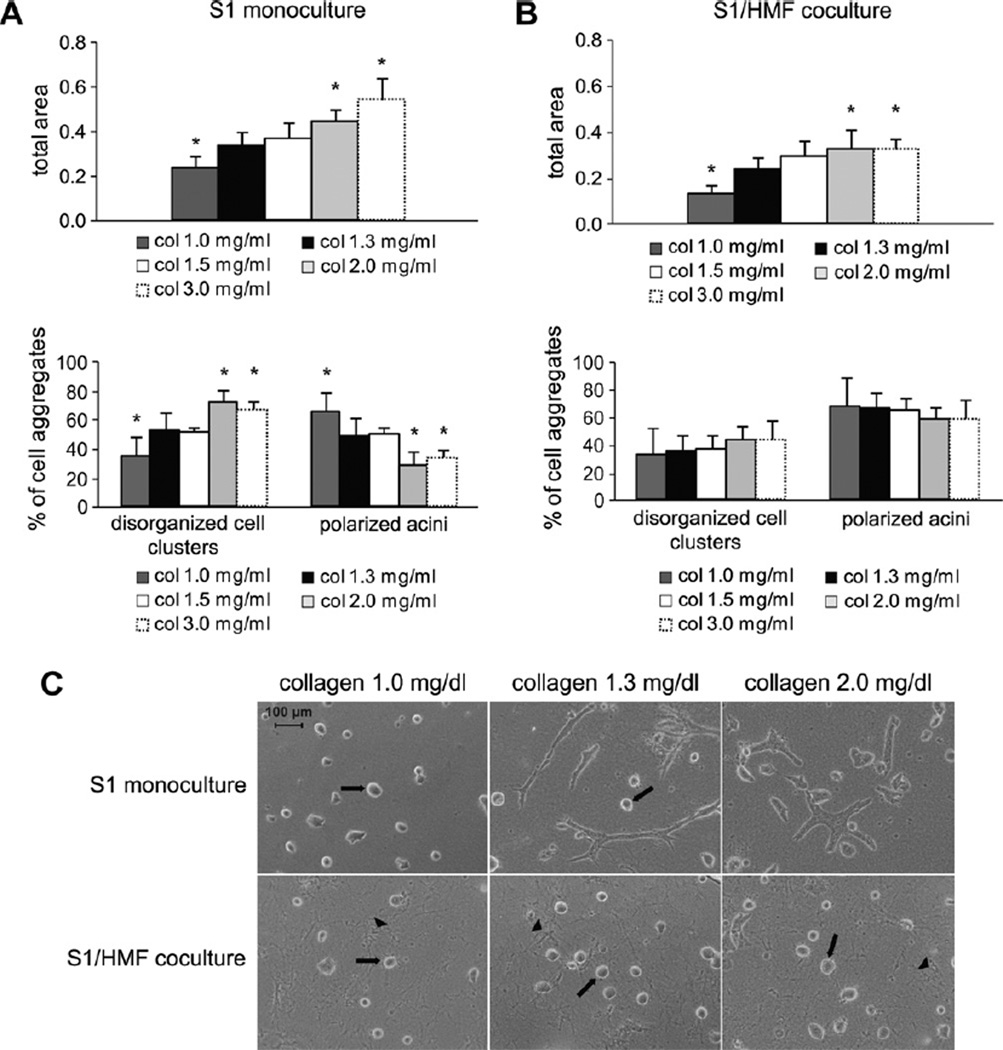
To expand on these results, S1 cells were grown as monocultures for 10 days in 3D-collagen I gels of different concentrations ranging from 1.0 mg/ml to 3.0 mg/ml. Our standard collagen concentration of 1.3 mg/ml served as control. When cultured in collagen gels of low rigidity (1.0 mg/ml), growth of S1 cells decreased about 1.8-fold compared to monocultures with 1.3 mg/ml collagen concentration. Furthermore, 65% of S1 cells in monoculture of low rigidity (1.0 mg/ml) formed polarized acini- like structures (Fig. 6A). Increasing the matrix stiffness led to disorganized and progressive growth of S1 cells. Using collagen concentrations of 2.0 mg/ml only 28% of S1 cells in monoculture differentiated into polarized acinus-like structures and growth was increased about 1.4-fold compared to the standard collagen concentration. Further increase of collagen concentration to 3.0 mg/ml did not further change the growth behavior seen with 2.0 mg/ml. No difference in growth or morphology was observed between gels of collagen concentrations of 1.3 mg/ml and 1.5 mg/ml (Fig. 6A). In agreement with the results from Paszek et al., increasing matrix tension inhibits differentiation and enhances total growth of S1 cells.
Fig. 6.
The matrix stiffness regulates epithelial cell growth and differentiation and is influenced by fibroblasts. (A) Evaluation of growth (upper bar graph) and differentiation (lower bar graph) of S1 cells grown in 3D-monoculture in collagen gels of different collagen concentrations after 10 days of culture. Asterisk indicates statistical significance compared to 3D-cultures in collagen gels of a collagen concentration of 1.3 mg/ml (p < 0.05). (B) Evaluation of growth (upper bar graph) and differentiation (lower bar graph) of S1 cells grown in 3D-coculture with HMF in collagen gels of different collagen concentrations after 10 days of culture. Asterisk indicates statistical significance compared to 3D-cultures in collagen gels of a collagen concentration of 1.3 mg/ml (p < 0.05). (C) Influence of increasing the matrix stiffness on morphology of S1 cells in monoculture (upper panel) and in coculture with HMF (lower panel) after 9 days of culture as shown by phase contrast microscopy. Arrow: glandular structures formed by S1 cells. Arrowhead: fibroblasts.
To investigate whether fibroblasts could modify the ECM to overcome the effects of matrix rigidity on morphogenesis observed in monocultures, we cocultured S1 cells with HMF in collagen gels of the same concentrations used in monoculture experiments. In cocultures of low matrix rigidity (1.0 mg/ml), fibroblasts did not increase the number of polarized acini compared to monocultures of low rigidity. Under both conditions the number of well-ordered polar spheroids was about 65%, whereas cell growth in cocultures of low rigidity decreased about 1.8-fold (Fig. 6B and C).
In gels of high rigidity (2.0 mg/ml) fibroblasts significantly increased the number of polarized acini compared to monocultures of high rigidity from 28% to 57% (p ≤ 0.001). Furthermore, growth of S1 cells in cocultures of high rigidity was increased compared to control. Remarkably, comparison of cocultures of high and low rigidity revealed no difference in morphogenesis of S1 cells (Fig. 6B and C). These findings demonstrate that mammary fibroblasts are able to reverse the perturbations of morphogenesis caused by increased matrix stiffness and can induce acinar morphogenesis of S1 cells even in collagen gels of high rigidity.
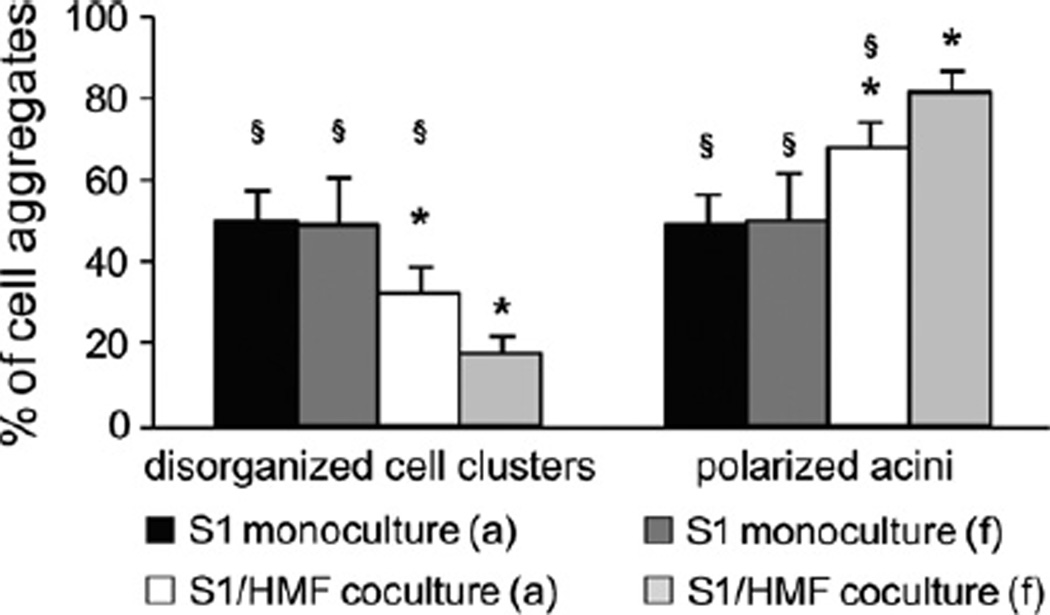
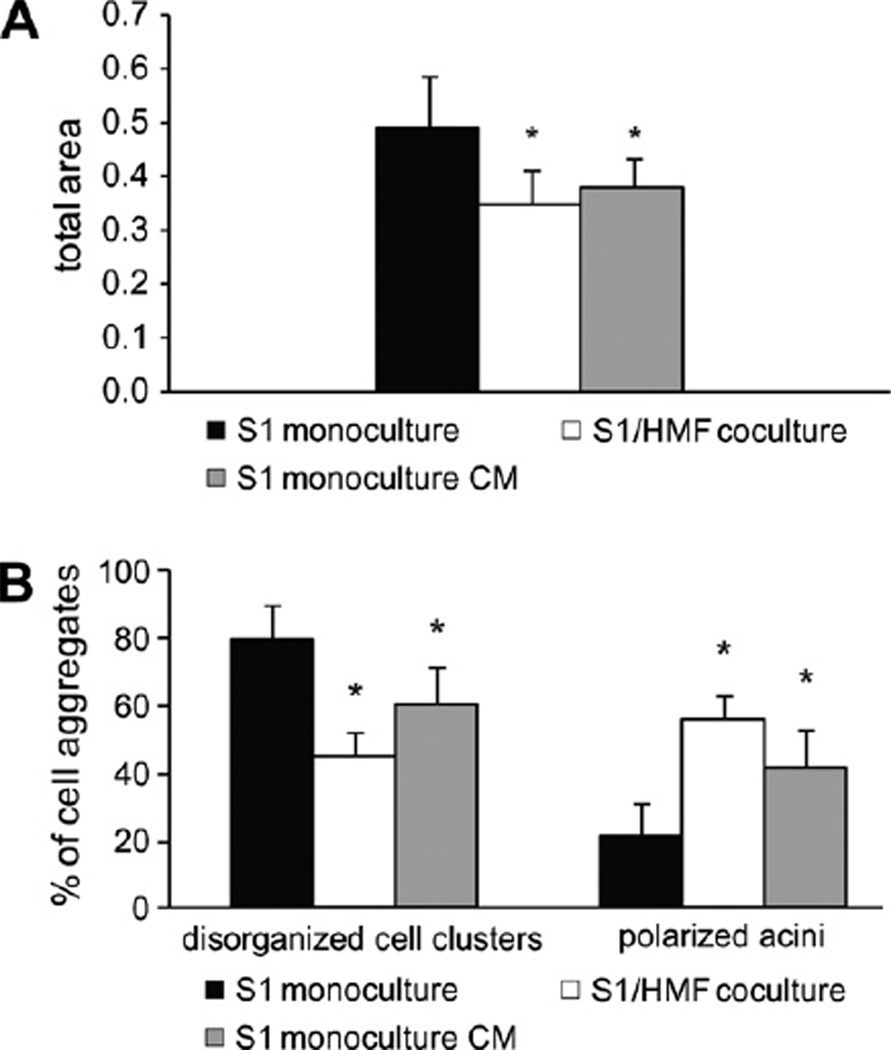
Besides matrix compliance, Wozniak et al. described contractility as an important attribute determining the ability of a cell to remodel matrix and thus to induce differentiation [30]. To investigate the ability of mammary epithelial cells to contract a collagen-I matrix, S1 cells were cultured in floating 3D-collagen I gels. When cultured under floating conditions no visible contraction of gels could be observed and S1 cells did not express contractile proteins as confirmed by immunolabeling for α-SMA (data not shown). As expected, no difference was observed between the number of polarized acini in floating and attached collagen gels (Fig. 7). Thus, S1 cells fail to undergo morphogenesis by contracting the gel.
Fig. 7.
Acinar morphogenesis of S1 cells in floating 3D-collagen I gels S1 cells were cultured as monoculture and coculture with HMF in floating and attached gels for 9 days (abbreviations: a, attached gel; f, floating gel). Asterisk indicates statistical significance compared to attached monoculture (p < 0.001); Section sign indicates statistical significance compared to floating coculture (p < 0.001).
Since fibroblast have been shown to be contractile [31] and HMF, CAF and NF were proven to express α-SMA (Fig. 5A), we hypothesized that contractility may contribute to the induction of differentiation observed in cocultures. Thus, we wondered if remodeling the ECM by enhancing contractile forces would increase fibroblast-induced differentiation of S1 cells in cocultures. Therefore, S1 cells were cocultured with HMF in floating 3D-collagen I gels. Under these conditions, a visible contraction of gels could be detected. Analysis of gels showed that gel contraction caused by fibroblasts significantly increased the number of polarized acini by 15–20% (p ≤ 0.001) (Fig. 7). Quantitative growth analyses of S1 cells grown in floating gels could not be performed because of differences in the degree of gel contraction. Therefore, we conclude that fibroblasts are able to influence matrix tension leading to cell differentiation by contraction of gels.
3.5. Influence of fibroblast-induced changes in matrix compliance on tumorigenic T4–2 cells
Because our data suggest that growth behavior and differentiation of phenotypically normal mammary epithelial cells are influenced by changes in matrix compliance, we sought to evaluate the impact of fibroblast-initiated changes of matrix compliance on malignant breast epithelial cells. Tumorigenic T4–2 cells were cultured in either mono- or coculture with HMF under floating conditions and growth and morphology were analyzed as previously described.
Similar to their benign counterpart (S1 cells), T4–2 cells did not contract collagen-I-gels under floating conditions. Consistent with this observation, T4–2 cells did not express contractile proteins as confirmed by immunolabeling for α-SMA (data not shown).
Floating cocultures showed strong contraction of the 3D-gels after 4–6 days of culture. Thus, growth analyses of T4–2 cells under floating conditions could not be performed by measuring the cytokeratin-positive cell clusters. Instead, we used the Ki67 proliferation index to evaluate differences in epithelial cell proliferation between the different culture conditions.
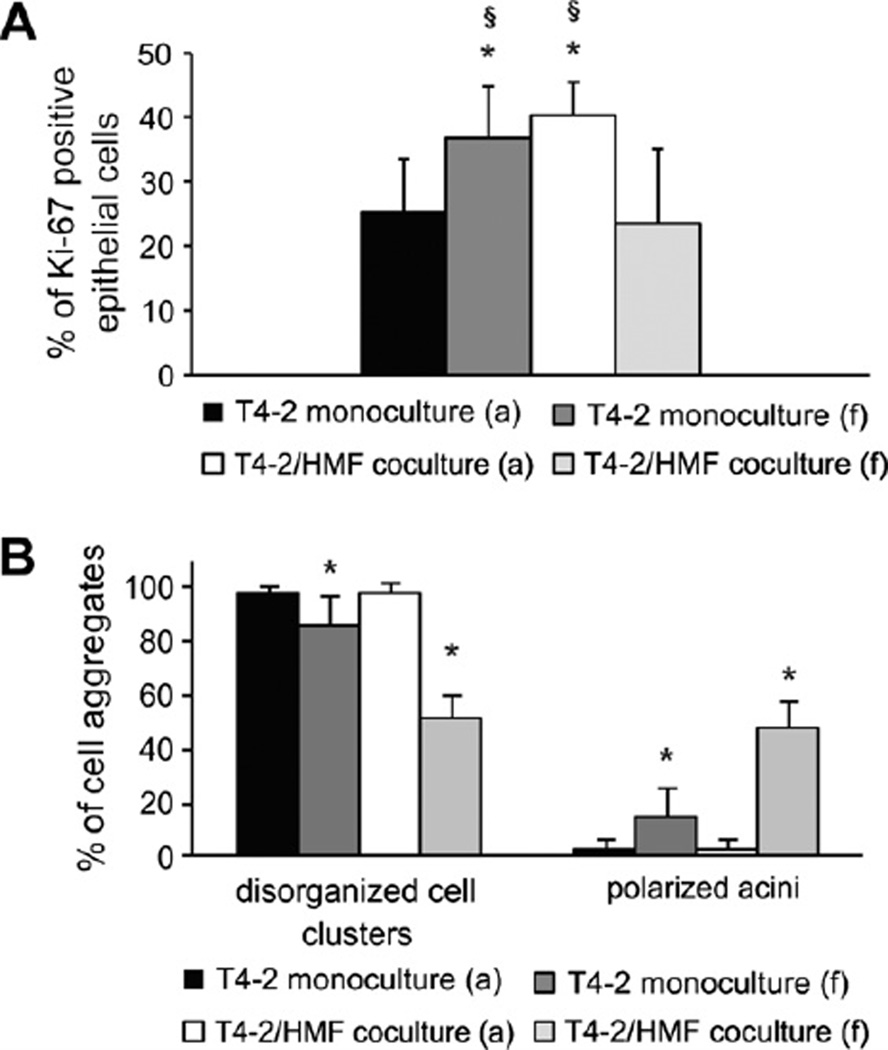
In line with the data presented in Fig. 3, proliferation of T4–2 cells was increased in attached coculture compared to attached monoculture (p < 0.0001). Furthermore, in T4–2 monocultures 3D-culture under floating conditions significantly increased proliferation of T4–2 cells (p = 0.002). Interestingly, proliferation of breast carcinoma cells in floating coculture was clearly decreased compared to attached coculture and floating monoculture (p < 0.01) (Fig. 8A).
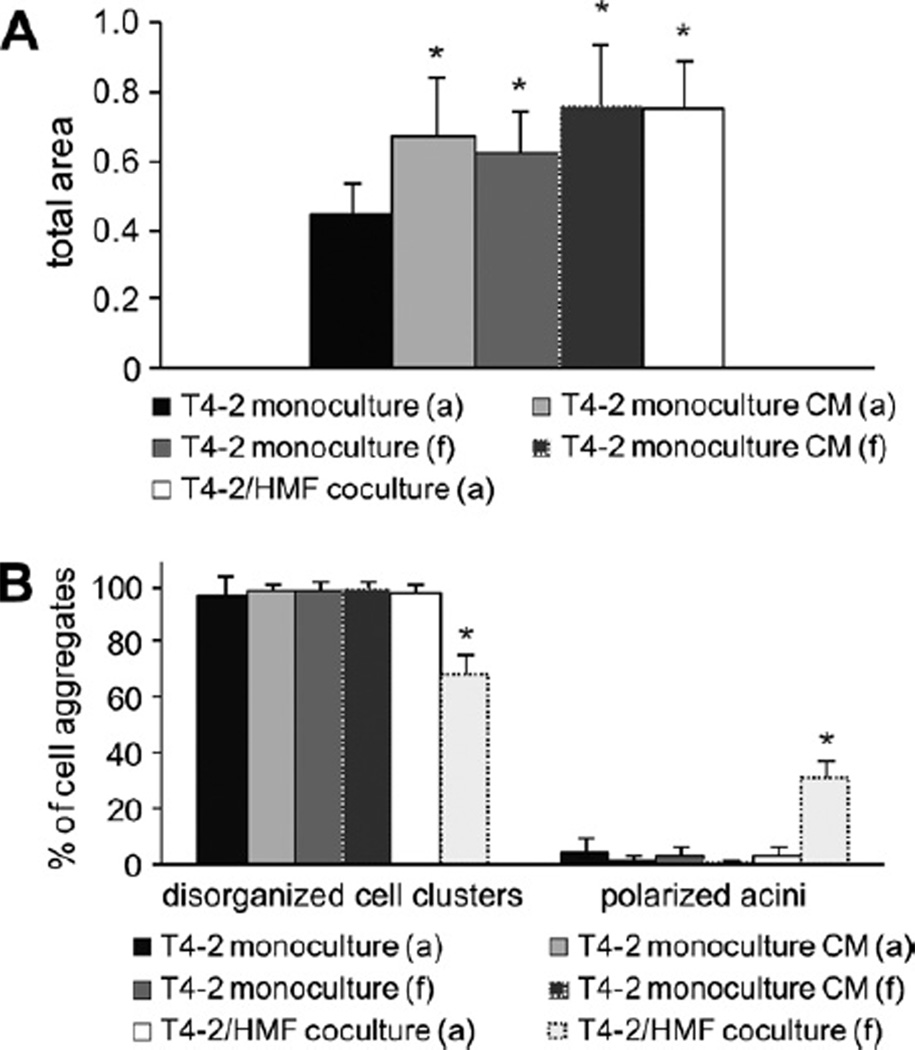
Fig. 8.
Proliferation and acinar morphogenesis of T4-2 cells in floating 3D-collagen I gels. (A) Ki67-proliferation index of T4-2 cells at day 8 in attached and floating mono- and cocultures was calculated by counting Ki-67 positive epithelial cells in at least 12 visual fields of each gel (inverted microscope; magnification 630×). The Ki67 proliferation index is the ratio of Ki-67 positive nuclei and all nuclei present in a visual field. Asterisk indicates statistical significance compared to attached monoculture (p < 0.01); Section sign indicates statistical significance compared to floating coculture (p < 0.01). (B) T4-2 cells were cultured as mono- and cocultures with HMF in floating and attached gels for days 7 days and morphology of cell clusters was evaluated (abbreviations: a, attached gel; f, floating gel). Asterisk indicates statistical significance compared to attached monoculture (p < 0.05).
When cultured in floating gels, the number of polarized acini in monoculture was mildly but significantly increased compared to attached monoculture (Fig. 8B). Remarkably, in cocultures the number of organized cell clusters under floating conditions was significantly increased about 15-fold compared to attached cocultures (p ≤ 0.001) (Fig. 8B).
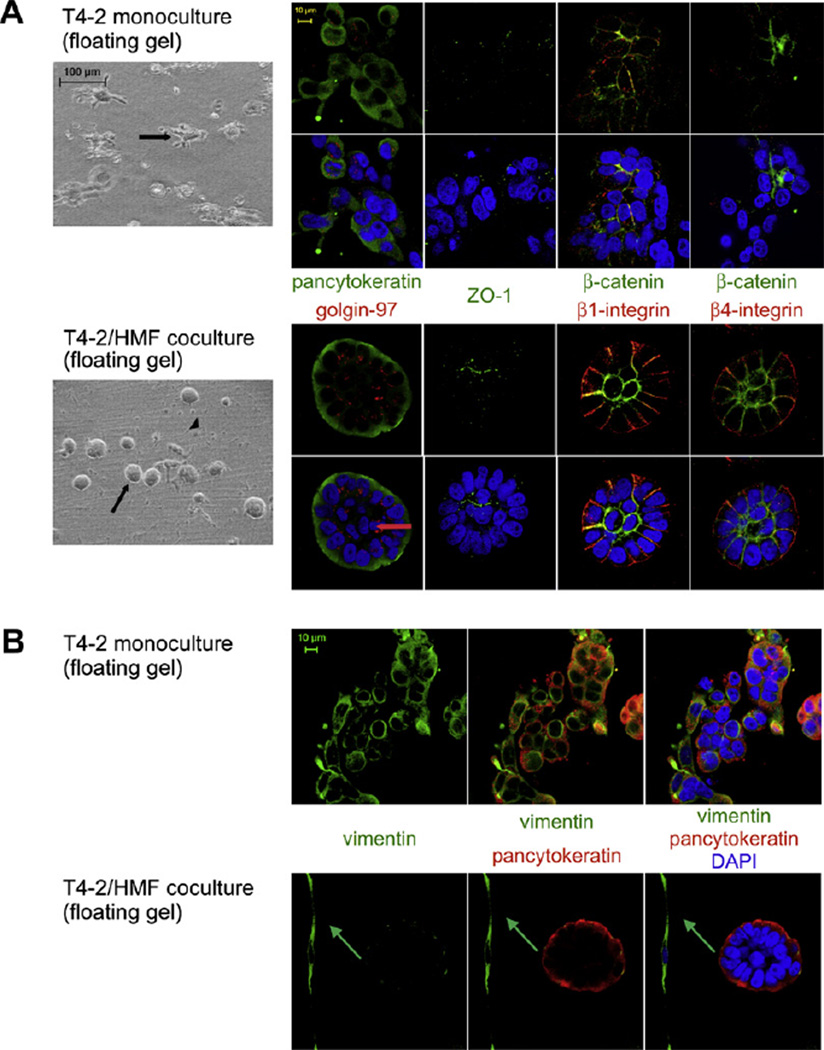
In floating cocultures, T4–2 cells were able to undergo morphogenetic events as observed in S1 cocultures resulting in formation of acini-like spheroids. In these glandular structures, T4–2 cells were organized in a single layer surrounding a luminal space. Most of these lumina contained residual single cells, implying that the process of lumen formation has not been completed during the culture time of 4–8 days (Fig. 9A).
Fig. 9.
Morphology of breast carcinoma cells in floating 3D-collagen I gels. (A) T4–2 cells (arrow) in monoculture (upper panel) compared with T4–2 cells in coculture (lower panel) with HMF (arrowhead) as shown by phasecontrast microscopy (column 1) and immunoflourescence staining (columns 2–5). Cells were stained for apical (golgin-97 (red)) and ZO-1 (green)), basal (4-integrin (red)), basolateral (1-integrin (red)) and lateral (-catenin (green)) polarity markers and epithelial (pancytoceratin (green)) markers. Nuclear counterstain (DAPI (blue)). Red arrow: Lumina of acini containing single cells indicating that the process of lumen formation has not been completed. (B) Vimentin expression is strongly reduced in polarized acini in coculture (lower panel) compared to disorganized cell clusters in monoculture (upper panel). Epithelial marker pancytokeratin (red). Nuclear counterstain (DAPI (blue)). Green arrow: Vimentin positive fibroblast in coculture.
T4–2 cells organized in acinus-like structures acquired apicobasal polarization resulting in apical expression of golgin-97 and ZO-1, basally located β4-integrin and basolaterally expressed β1-integrin, whereas β-catenin assumed an apicolateral location (Fig. 9A). As observed in S1 cocultures, T4–2 cells in floating cocultures showed no or weak vimentin expression in contrast to floating monocultures, in which vimentin was strongly expressed (Fig. 9B). T4–2 cells in floating monocultures mostly displayed randomly distributed expression of differentiation markers comparable to attached monocultures (Fig. 9A).
Whereas T4–2 cells in monoculture display striking perturbations in tissue architecture, they are able to reestablish phenotypically normal mammary tissue structures in floating 3D-cocultures. These findings suggest that gel contraction caused by fibroblasts induces changes in matrix compliance leading to a reversion of the malignant phenotype of breast carcinoma cells.
3.6. Growth behavior and differentiation of mammary epithelial cells are influenced by soluble stromal factors
Our findings reveal that acinar morphogenesis of mammary epithelial cells is regulated by modifications of the ECM caused by direct contact between fibroblasts and ECM. To investigate whether fibroblast-derived soluble factors additionally contribute to the establishment of glandular structures, we cultured S1 cells with conditioned media (CM) from HMF. After 10 days of culture, S1 cells in monoculture with conditioned medium showed similar growth behavior as S1 cells grown in coculture with HMF. Cell growth of S1 cells was 1.3-fold increased in normal monocultures compared to cocultures or monocultures with conditioned medium (Fig. 10A). However, in monoculture with conditioned medium, only 40% of cell colonies showed acinus-like morphology compared to 55% observed in cocultures and 20% observed in normal monocultures (p ≤ 0.01) (Fig. 10B). These data imply that, in addition to direct interactions between fibroblasts, ECM and epithelium, secreted stromal proteins may play a role in the regulation of mammary epithelial cell morphogenesis.
Fig. 10.
Soluble stromal factors influence growth behavior and differentiation of mammary epithelial cells. (A) Growth of S1 cells at day 9 in culture with conditioned media was decreased compared to control monoculture (abbreviations: CM, conditioned medium). Asterisk indicates statistical significance compared to monoculture (p < 0.05). (B) Culturing S1 cells with conditioned medium from HMF induced differentiation of S1 cells comparable to coculture with HMF (abbreviations: CM, conditioned medium). Asterisk indicates statistical significance compared to monoculture (p < 0.01).
Therefore, we asked whether the identification of the protein composition of the conditioned medium produced by HMF would help us to delineate possible signaling pathways involved in stromal–epithelial interactions leading to differentiation.
Analysis of conditioned medium from HMF was performed with 1D-SDS–PAGE, tryptic in gel digest and Nano-LC–ESI-MS/MS. Using a false discovery rate of 1% fragment spectra, 6591 peptide spectrum matches (PSMs) were identified from a database obtained from www.uniprot.org. A false discovery rate of 5% fragment spectra provided 7985 PSM, and a total of 401 proteins could be identified.
These proteins included major components of the basement membrane like laminin, nidogen-1 and −2 and collagen IV, which are known to be essential for mammary epithelial cell differentiation [5,32]. Furthermore, the proteoglycans decorin and lumican and the glycoprotein dystroglycan, that play an important role in proper ECM constitution and tumor suppression [33–36], were found to be secreted by HMF. Additionally, CM comprised regulators of the tensional homeostasis required for morphogenesis of mammary epithelial cells like filamin A, vinculin and actinin [10,30,37] (Additional file 3).
3.7. Soluble stromal factors do not contribute to reversion of tumorigenic T4–2 cells
3D-coculture with HMF induces growth arrest and morphogenesis of normal mammary epithelial cells, whereas HMF exhibit protumoral effects in attached coculture with breast carcinoma cells. Our data reveal that both mechanical cues and stroma-derived soluble factors are involved in regulation of growth and morphogenesis of S1 cells. Since HMF revert the malignant phenotype of T4–2 cells only under floating conditions, we hypothesized that mechanical rather than soluble fibroblast-derived signals are responsible for this effect. To determine whether this was the case, we cultured T4–2 cells in a 3D-collagen I matrix with conditioned media from HMF in either attached or floating gels. Under both conditions, growth of T4–2 cells in monoculture with CM was increased about 1.5–1.7-fold compared to the level observed in attached monoculture (p < 0.01) (Fig. 11A). 3D-monoculture under floating conditions caused a slight but significant increase in growth compared to attached monoculture, which had not been observed in our previous experiments. A strong contraction of the 3D-coculture gel under floating conditions impeded growth assessment of T4–2 cells.
Fig. 11.
Soluble stromal factors do not contribute to reversion of tumorigenic T4–2 cells A. Growth of tumorigenic T4–2 cells in 3D-monoculture and coculture with different matrix compliance and different culture media. Growth of T4–2 cells is stimulated by HMF in coculture or in monoculture with conditioned media (CM) from HMF. Asterisk indicates statistical significance compared to attached monoculture (p < 0.01). (abbreviations: a, attached gel; f, floating gel). (B) Morphology of breast carcinoma cells in attached or floating 3D-collagen gels with and without conditioned media. CM from HMF does not influence morphogenesis of T4–2 cells under either attached or floating conditions as >95% of carcinoma cells demonstrate disorganized growth behavior. Morphogenesis of T4–2 cells is only induced in coculture under floating conditions. Asterisk indicates statistical significance compared to attached monoculture (p < 0.01) (abbreviations: a, attached gel; f, floating gel; CM, conditioned medium).
Morphology of T4–2 cells in either attached or floating monoculture with CM was indistinguishable from monocultures without CM as > 95% of carcinoma cell clusters formed disorganized apolar structures. However, in floating cocultures the number of polarized acini was highly significantly increased as previously described (p < 0.01) (Fig. 11B).
These findings suggest that secreted stromal factors stimulate growth of tumorigenic mammary cells and are not involved in the regulation of acinar morphogenesis. Collectively, our data confirm that fibroblast-induced gel contraction is required for reversion of the malignant phenotype of breast carcinoma cells.
4. Discussion
Breast tissue is a dynamically interacting complex 3D multicellular network. The development of the normal mammary gland is not only cell autonomous but also partly controlled by the surrounding microenvironment [38]. The microenvironment is composed of soluble proteins such as growth factors, the ECM and stromal cells including endothelial cells, adipocytes, infiltrating immune cells and fibroblasts [39]. Fibroblasts are the predominant cell type of the stroma and are considered critical for tumor progression [7].
The findings presented here imply that stromal fibroblasts play an important role in the regulation of morphogenesis and control of growth behavior of normal and tumorigenic breast epithelial cells. Using a 3D-collagen I cell culture model, which mimics aspects of the physiological tissue context in vitro, we found that mammary stromal fibroblasts are crucial for the maintenance of the tissue tensional homeostasis, thus regulating epithelial morphogenesis and reverting the malignant phenotype of breast carcinoma cells.
During normal epithelial organogenesis, mammary epithelial cells organize into acini, spherical structures containing a hollow lumen [40]. Here, we show that phenotypically normal mammary epithelial cells (S1) in 3D-collagen I cocultures with normal mammary fibroblasts (HMF) arrange into growth-arrested, acini like structures with a central lumen. In vivo, a defined apical–basal polarity of normal epithelial cells is essential for tissue function [1]. Consistent with literature reports using 3D-cultures in laminin-rich matrices, we demonstrate polarized orientation of the cellular axis in 3D-collagen I gels in coculture with HMF [41]. In contrast, S1 cells in monoculture are characterized by progressive growth, disruption of tissue organization as shown by random distribution of basal (β4-integrin) and apical (golgin-97, ZO-1) polarity markers and destabilization of adherent junctions indicated by diffuse β-catein expression. Moreover, normal mammary epithelial cells in monoculture display decreased integrin expression, upregulation of the mesenchymal marker vimentin and formation of celular extensions as frequently observed in carcinoma cells [42]. Collectively, S1 cells growing in the absence of fibroblasts recapitulate many hallmarks of breast cancer such as loss of normal tissue architecture, morphological disorganization and loss of proliferation control [43].
Contrasting with our findings, Howlett et al. and Gudjonsson et al. showed that S1 cells and normal primary mammary epithelial cells (MECs) produced well-ordered spheres when cultured as monocultures in collagen-I gels. Despite the organization of cells into ordered structures, both papers describe a lack of differentiation of MECs and S1 cells in collagen gels. Whereas Howlett et al. merely observed the absence of a lumen and an endogenous deposition of BM, which would be consistent with differentiation, Gudjonsson et al. found that MECs exhibit inside-out polarity [18,44]. This apparent discrepancy to our findings could be explained by their use of a higher cell number and a different collagen concentration both of which would produce an altered matrix compliance.
Matrices composed of BM substitutes like Matrigel® have been the gold standard for 3D experiments to study differentiation of mammary epithelial cells. Solubilized BM preparations contain laminin as major component [45]. Normal breast epithelial cell lines and MECs recapitulate the phenotypic characteristics of normal breast tissue in vivo when cultured three-dimensionally with-in reconstituted BM [41,46]. These experiments imply that BM components are required to induce polarization and differentiation of normal mammary epithelial cells. Particularly, laminin or a combination of laminin and nidogen-1 have been shown to be critically required for the induction of differentiation of normal mammary epithelial cells [5,47]. Our results reveal that major components of the BM like laminin, nidogen−1 and −2 and collagen IV can be produced by mammary fibroblasts (Additional file 3). Furthermore, we show that soluble factors secreted by HMF alone are sufficient to induce morphogenesis of S1 cells in collagen-I monoculture. Therefore, we hypothesize that the production of BM components by fibroblasts in 3D-collagen I cocultures may contribute to the formation of growth arrested polarized acini by S1 cells.
Whereas coculture with normal fibroblasts or exposure to soluble factors secreted by HMF induces differentiation of normal mammary epithelial cells, tumorigenic T4–2 cells demonstrate increased and disorganized growth under both conditions (Figs. 3 and 11).
These findings are consistent with the results of Su et al., who showed growth stimulation of the human breast cancer cell line T47D by HMF [13]. In concert with observations from S1-cell experiments, these results indicate that the ability of fibroblasts to inhibit or to stimulate cell growth and morphogenesis is context dependent. Cancer-associated fibroblasts have been reported to exhibit distinct morphological and functional alterations compared to normal fibroblasts [7,8,48]. We conducted gene expression analysis of HMF cocultured with S1 or T4–2 cells and observed fundamental differences in gene expression profiles [8]. Hence, the inverse growth behavior of S1 and T4–2 cells in coculture with HMF is likely caused by altered stromal–epithelial interactions. Furthermore, increased growth of T4–2 cells in coculture with HMF indicates that the growth arrest of S1 cells in coculture with HMF cannot be explained by additional medium consumption by fibroblasts.
T4–2 cells have been shown to form large, loosely disorganized colonies that continue to grow even when cultured in reconstituted BM [41], which supports our findings, that T4–2 cells do not undergo morphogenesis when cultured with BM component-containing media (Fig. 11). Considering those and our findings, we hypothesize that tumorigenic T4–2 cells have lost the ability to respond to stromal factors normally leading to glandular differentiation.
Interestingly, Krause et al. demonstrated that a matrix containing both type I collagen and reconstituted BM, and primary normal breast fibroblasts, induces a nearly complete reversion of the malignant phenotype of MCF7 cells in 3D-culture [49]. These results indicate that in principle, not all breast carcinoma cells are incapable of responding to stromal differentiation factors. Unpublished data by our group support this hypothesis. Coculture with HMF differentiates primary human mammary carcinoma cells into a collective of revertible and a collective of nonrevertible carcinomas.
Using a variety of primary normal human mammary fibroblasts in 3D-cocultures, we determined that the induction of differentiation of S1 cells is a more universal activity of mammary fibroblasts rather than a phenomenon specific to HMF. Interestingly, we found that there was no difference between growth behavior of S1 cells in coculture with either NF or CAF. In support of these findings, Sadlonova et al. showed that both NF and CAF inhibit the growth of the non-tumorigenic breast epithelial cell line MCF10A when cocultured in Matrigel [17].
Recently, we showed that the variability of gene expression is higher in NF than in CAF, suggesting an interindividual heterogeneity of normal stroma [8]. Because NF and CAF isolated from the same person yielded the same results in coculture with S1 cells, this heterogeneity did not seem to contribute to the induction of differentiation of epithelial cells. However, our results imply an interindividual heterogeneity of mammary fibroblasts rather than differences between NF and CAF isolated from the same patient to account for the ability to induce morphogenesis in mammary epithelial cells.
Collectively, these findings let us assume that the mechanism by which this is accomplished, is not controlled by cell specific features. Instead, we reason that fibroblasts generally influence the mechanical and biochemical properties of the ECM, leading to differentiation of mammary epithelial cells.
This assumption is supported by several studies showing that cell behavior is influenced by the stiffness of their microenvironment [10,30,50,51]. Increasing the collagen concentration in the ECM of tissues leads to an increase in the elastic modulus and therefore stiffness [37]. We and others demonstrate that normal mammary epithelial cells are able to sense the stiffness of their microenvironment and respond to it by regulating their cell behavior (Figs. 6 and 7; [10]). In vivo, an increase in stromal density in the breast is correlated with a significantly increased risk of developing breast cancer [9]. These findings correlate with our observations of cancer-like cell growth in monocultures of high density and acini-like structures in monocultures of low density similar to the soft fatty mammary gland in vivo. Since we found differentiation of S1 cells in cocultures with HMF even in high-density collagen gels, we reasoned that mammary fibroblasts are able to counterbalance the perturbations of morphogenesis caused by increased matrix stiffness.
Several published literature reports postulate a relationship between cell differentiation and the ability of a cell to contract the ECM. For example Gehler et al. describe that high-density collagen gels are too stiff for cells to adequately contract and therefore inhibit ductal morphogenesis [37]. As a possible molecular mechanism Wozniak et al. describe the molecular link between ECM stiffness and ROCK-mediated contractility as well as a subsequent down-regulation of Rho and FAK function [30].
As early as 1977, floating collagen gels were used to show that normal breast epithelial cells need a deformable 3D-ECM for differentiation [52]. Wozniak et al. demonstrated that noncancerous MCF10A breast epithelial cells did not form differentiated structures when cultured in attached 3D-collagen gels, whereas in floating 3D-collagen gels they were able to contract the gel and undergo morphogenesis [30]. Our results show that S1 cells themselves are not able to contract collagen gels leading to unstructured growth even in floating monocultures. Coculturing of S1 cells and HMF under free-floating conditions leads to visible gel contraction and increased cell differentiation. Therefore, we propose that fibroblasts are able to influence matrix tension by contraction of gels leading to cell differentiation. These conclusions are consistent with those of Larsen et al., who postulated that the ability of a cell to contract and remodel the matrix is influenced by a balance between the stiffness of the matrix and cell contractility [53].
Accordingly, we sought to investigate whether breast carcinoma cells would be influenced in their growth behavior by fibroblast-induced remodeling of the mechanical characteristics of the ECM. T4–2 cells in floating cocultures with HMF differentiated into polarized acini with reduced proliferative activity, unlike T4–2 cells in floating monocultures or attached mono- and cocultures. Gehler et al. showed that human T47D ductal breast carcinoma cells underwent ductal morphogenesis when cultured in low-density, floating collagen gels [37]. These results are in contrast to our observations in floating monocultures of T4–2 cells. Interestingly, T47D cells, but not T4–2 cells, are able to contract the gels similar to HMF. These findings support our hypothesis that gel contraction caused by fibroblasts induces changes in matrix compliance leading to differentiation of tumorigenic T4–2 cells in floating cocultures. Supporting this reasoning, Paszek et al. demonstrated that the malignant phenotype of T4–2 cells can be reverted by reducing intracellular tension in response to the stiffness of the microenvironment [10]. Interestingly, exposure to soluble stromal factors (CM) stimulates growth of T4–2 cells in both attached and floating monoculture, but does not inhibit morphogenesis in floating coculture [39]. Hence, these findings reinforce the notion that a readjustment of the tensional homeostasis is required for reversion of the malignant phenotype and is furthermore sufficient to counterbalance progression promoted by secreted stromal factors.
In conclusion, we demonstrate that stromal mammary fibroblasts induce morphogenesis of normal mammary epithelial cells in a matrix free of BM components, whereas the absence of fibroblasts results in tumor-like growth. Our results prove that the regulation of morphogenesis of mammary epithelial cells relies on a complex interplay of cell–matrix interactions, cell–cell contacts and matrix compliance. Our findings point to a key role for stromal fibroblasts within this network. As a potential mechanism of action, we postulate the production of BM components and fibroblast-mediated remodeling of matrix compliance. In addition, we show that the readjustment of matrix tension can even induce a phenotypical reversion of breast carcinoma cells. Uncovering the molecular underpinnings of these observations will be the subject of future work. In summary, our results demonstrate a significant role for stromal factors in regulating the differentiation of normal epithelial cells and in the control of tumor growth. Therefore the mammary stroma promises to be an innovative therapeutic target in the treatment of breast cancer.
Supplementary Material
Acknowledgments
We acknowledge Prof. Dr. med. Dieter Kabelitz for enabling confocal analysis and Prof. Dr. rer. nat. Sabine Adam for support with the cell cycle analyses. We also thank Regina Grunewald and Sandra Ussat for excellent technical assistance.
Footnotes
Appendix A. Supplementary material
Supplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.canlet.2012.06.014.
References
- 1.Bissell MJ, Rizki A, Mian IS. Tissue architecture: the ultimate regulator of breast epithelial function. Curr. Opin. Cell Biol. 2003;15:753–762. doi: 10.1016/j.ceb.2003.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chrenek MA, Wong P, Weaver VM. Tumour-stromal interactions. Integrins and cell adhesions as modulators of mammary cell survival and transformation. Breast Cancer Res. 2001;3:224–229. doi: 10.1186/bcr300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ronnov-Jessen L, Petersen OW, Bissell MJ. Cellular changes involved in conversion of normal to malignant breast: importance of the stromal reaction. Physiol. Rev. 1996;76:69–125. doi: 10.1152/physrev.1996.76.1.69. [DOI] [PubMed] [Google Scholar]
- 4.Krause S, Maffini MV, Soto AM, Sonnenschein C. A novel 3D in vitro culture model to study stromal–epithelial interactions in the mammary gland. Tissue Eng. Part C Methods. 2008;14:261–271. doi: 10.1089/ten.tec.2008.0030. [DOI] [PubMed] [Google Scholar]
- 5.Pujuguet P, Simian M, Liaw J, Timpl R, Werb Z, Bissell MJ. Nidogen-1 regulates laminin-1-dependent mammary-specific gene expression. J. Cell Sci. 2000;113(Pt 5):849–858. doi: 10.1242/jcs.113.5.849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Singer C, Rasmussen A, Smith HS, Lippman ME, Lynch HT, Cullen KJ. Malignant breast epithelium selects for insulin-like growth factor II expression in breast stroma: evidence for paracrine function. Cancer Res. 1995;55:2448–2454. [PubMed] [Google Scholar]
- 7.Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat. Rev. Cancer. 2006;6:392–401. doi: 10.1038/nrc1877. [DOI] [PubMed] [Google Scholar]
- 8.Bauer M, Su G, Casper C, He R, Rehrauer W, Friedl A. Heterogeneity of gene expression in stromal fibroblasts of human breast carcinomas and normal breast. Oncogene. 2010;29:1732–1740. doi: 10.1038/onc.2009.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boyd NF, Lockwood GA, Martin LJ, Knight JA, Byng JW, Yaffe MJ, Tritchler DL. Mammographic densities and breast cancer risk. Breast Dis. 1998;10:113–126. doi: 10.3233/bd-1998-103-412. [DOI] [PubMed] [Google Scholar]
- 10.Paszek MJ, Zahir N, Johnson KR, Lakins JN, Rozenberg GI, Gefen A, Reinhart-King CA, Margulies SS, Dembo M, Boettiger D, Hammer DA, Weaver VM. Tensional homeostasis and the malignant phenotype. Cancer Cell. 2005;8:241–254. doi: 10.1016/j.ccr.2005.08.010. [DOI] [PubMed] [Google Scholar]
- 11.Briand P, Petersen OW, Van Deurs B. A new diploid nontumorigenic human breast epithelial cell line isolated and propagated in chemically defined medium. In Vitro Cell Dev. Biol. 1987;23:181–188. doi: 10.1007/BF02623578. [DOI] [PubMed] [Google Scholar]
- 12.Kuperwasser C, Chavarria T, Wu M, Magrane G, Gray JW, Carey L, Richardson A, Weinberg RA. Reconstruction of functionally normal and malignant human breast tissues in mice. Proc. Natl. Acad. Sci. USA. 2004;101:4966–4971. doi: 10.1073/pnas.0401064101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Su G, Blaine SA, Qiao D, Friedl A. Shedding of syndecan-1 by stromal fibroblasts stimulates human breast cancer cell proliferation via FGF2 activation. J. Biol. Chem. 2007;282:14906–14915. doi: 10.1074/jbc.M611739200. [DOI] [PubMed] [Google Scholar]
- 14.Bauer M, Su G, Beebe DJ, Friedl A. 3D microchannel co-culture: method and biological validation. Integr. Biol. (Camb) 2010;2:371–378. doi: 10.1039/c0ib00001a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- 16.Scheler C, Lamer S, Pan Z, Li XP, Salnikow J, Jungblut P. Peptide mass fingerprint sequence coverage from differently stained proteins on two-dimensional electrophoresis patterns by matrix assisted laser desorption/ ionization-mass spectrometry (MALDI-MS) Electrophoresis. 1998;19:918–927. doi: 10.1002/elps.1150190607. [DOI] [PubMed] [Google Scholar]
- 17.Sadlonova A, Novak Z, Johnson MR, Bowe DB, Gault SR, Page GP, Thottassery JV, Welch DR, Frost AR. Breast fibroblasts modulate epithelial cell proliferation in three-dimensional in vitro co-culture. Breast Cancer Res. 2005;7:R46–R59. doi: 10.1186/bcr949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gudjonsson T, Ronnov-Jessen L, Villadsen R, Rank F, Bissell MJ, Petersen OW. Normal and tumor-derived myoepithelial cells differ in their ability to interact with luminal breast epithelial cells for polarity and basement membrane deposition. J. Cell Sci. 2002;115:39–50. doi: 10.1242/jcs.115.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Streuli CH, Bissell MJ. Expression of extracellular matrix components is regulated by substratum. J. Cell Biol. 1990;110:1405–1415. doi: 10.1083/jcb.110.4.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lelièvre SA, Bissell MJ. Three-dimensional cell culture: the importance of context in regulation of function. Encyclopedia Mol. Cell Biol. Mol. Med. 2005;14:383–420. [Google Scholar]
- 21.Mailleux AA, Overholtzer M, Brugge JS. Lumen formation during mammary epithelial morphogenesis: insights from in vitro and in vivo models. Cell Cycle. 2008;7:57–62. doi: 10.4161/cc.7.1.5150. [DOI] [PubMed] [Google Scholar]
- 22.Park CC, Zhang H, Pallavicini M, Gray JW, Baehner F, Park CJ, Bissell MJ. Beta1 integrin inhibitory antibody induces apoptosis of breast cancer cells, inhibits growth, and distinguishes malignant from normal phenotype in three dimensional cultures and in vivo. Cancer Res. 2006;66:1526–1535. doi: 10.1158/0008-5472.CAN-05-3071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Thiery JP. Epithelial-mesenchymal transitions in tumour progression. Nat. Rev. Cancer. 2002;2:442–454. doi: 10.1038/nrc822. [DOI] [PubMed] [Google Scholar]
- 24.Kokkinos MI, Wafai R, Wong MK, Newgreen DF, Thompson EW, Waltham M. Vimentin and epithelial–mesenchymal transition in human breast cancer–observations in vitro and in vivo. Cells Tissues Organ. 2007;185:191–203. doi: 10.1159/000101320. [DOI] [PubMed] [Google Scholar]
- 25.Lee JM, Dedhar S, Kalluri R, Thompson EW. The epithelial–mesenchymal transition: new insights in signaling, development, and disease. J. Cell Biol. 2006;172:973–981. doi: 10.1083/jcb.200601018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Briand P, Nielsen KV, Madsen MW, Petersen OW. Trisomy 7p and malignant transformation of human breast epithelial cells following epidermal growth factor withdrawal. Cancer Res. 1996;56:2039–2044. [PubMed] [Google Scholar]
- 27.Chang HY, Chi JT, Dudoit S, Bondre C, van de Rijn M, Botstein D, Brown PO. Diversity, topographic differentiation, and positional memory in human fibroblasts. Proc. Natl. Acad. Sci. USA. 2002;99:12877–12882. doi: 10.1073/pnas.162488599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Guo YP, Martin LJ, Hanna W, Banerjee D, Miller N, Fishell E, Khokha R, Boyd NF. Growth factors and stromal matrix proteins associated with mammographic densities. Cancer Epidemiol. Biomarkers Prev. 2001;10:243–248. [PubMed] [Google Scholar]
- 29.Roeder BA, Kokini K, Sturgis JE, Robinson JP, Voytik-Harbin SL. Tensile mechanical properties of three-dimensional type I collagen extracellular matrices with varied microstructure. J. Biomech. Eng. 2002;124:214–222. doi: 10.1115/1.1449904. [DOI] [PubMed] [Google Scholar]
- 30.Wozniak MA, Desai R, Solski PA, Der CJ, Keely PJ. ROCK-generated contractility regulates breast epithelial cell differentiation in response to the physical properties of a three-dimensional collagen matrix. J. Cell. Biol. 2003;163:583–595. doi: 10.1083/jcb.200305010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Danowski BA. Fibroblast contractility and actin organization are stimulated by microtubule inhibitors. J. Cell. Sci. 1989;93(Pt 2):255–266. doi: 10.1242/jcs.93.2.255. [DOI] [PubMed] [Google Scholar]
- 32.Plachot C, Chaboub LS, Adissu HA, Wang L, Urazaev A, Sturgis J, Asem EK, Lelievre SA. Factors necessary to produce basoapical polarity in human glandular epithelium formed in conventional and high-throughput three-dimensional culture: example of the breast epithelium. BMC Biol. 2009;7:77. doi: 10.1186/1741-7007-7-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Eshchenko TY, Rykova VI, Chernakov AE, Sidorov SV, Grigorieva EV. Expression of different proteoglycans in human breast tumors. Biochemistry (Mosc) 2007;72:1016–1020. doi: 10.1134/s0006297907090143. [DOI] [PubMed] [Google Scholar]
- 34.Kelemen LE, Couch FJ, Ahmed S, Dunning AM, Pharoah PD, Easton DF, Fredericksen ZS, Vierkant RA, Pankratz VS, Goode EL, Scott CG, Rider DN, Wang X, Cerhan JR, Vachon CM. Genetic variation in stromal proteins decorin and lumican with breast cancer: investigations in two case-control studies. Breast Cancer Res. 2008;10:R98. doi: 10.1186/bcr2201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ishiwata T, Cho K, Kawahara K, Yamamoto T, Fujiwara Y, Uchida E, Tajiri T, Naito Z. Role of lumican in cancer cells and adjacent stromal tissues in human pancreatic cancer. Oncol. Rep. 2007;18:537–543. [PubMed] [Google Scholar]
- 36.Leonoudakis D, Singh M, Mohajer R, Mohajer P, Fata JE, Campbell KP, Muschler JL. Dystroglycan controls signaling of multiple hormones through modulation of STAT5 activity. J. Cell Sci. 2010;123:3683–3692. doi: 10.1242/jcs.070680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gehler S, Baldassarre M, Lad Y, Leight JL, Wozniak MA, Riching KM, Eliceiri KW, Weaver VM, Calderwood DA, Keely PJ. Filamin A-beta1 integrin complex tunes epithelial cell response to matrix tension. Mol. Biol. Cell. 2009;20:3224–3238. doi: 10.1091/mbc.E08-12-1186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Nelson CM, Bissell MJ. Of extracellular matrix, scaffolds, and signaling: tissue architecture regulates development, homeostasis, and cancer. Ann. Rev. Cell Dev. Biol. 2006;22:287–309. doi: 10.1146/annurev.cellbio.22.010305.104315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Muschler J, Streuli CH. Cell–matrix interactions in mammary gland development and breast cancer, Cold Spring Harb. Perspect Biol. 2010;2:a003202. doi: 10.1101/cshperspect.a003202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Debnath J, Mills KR, Collins NL, Reginato MJ, Muthuswamy SK, Brugge JS. The role of apoptosis in creating and maintaining luminal space within normal and oncogene-expressing mammary acini. Cell. 2002;111:29–40. doi: 10.1016/s0092-8674(02)01001-2. [DOI] [PubMed] [Google Scholar]
- 41.Petersen OW, Ronnov-Jessen L, Howlett AR, Bissell MJ. Interaction with basement membrane serves to rapidly distinguish growth and differentiation pattern of normal and malignant human breast epithelial cells. Proc. Natl. Acad. Sci. USA. 1992;89:9064–9068. doi: 10.1073/pnas.89.19.9064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Poincloux R, Lizarraga F, Chavrier P , Matrix invasion by tumour cells: a focus on MT1-MMP trafficking to invadopodia. J. Cell Sci. 2009;122:3015–3024. doi: 10.1242/jcs.034561. [DOI] [PubMed] [Google Scholar]
- 43.Harris J, Lippman M, Morrow M, Osborne C. Diseases of the Breast Philadelphia. PA: Lippincott Williams and Wilkins; 1999. [Google Scholar]
- 44.Howlett AR, Bailey N, Damsky C, Petersen OW, Bissell MJ. Cellular growth and survival are mediated by beta 1 integrins in normal human breast epithelium but not in breast carcinoma. J. Cell Sci. 1995;108(Pt 5):1945–1957. doi: 10.1242/jcs.108.5.1945. [DOI] [PubMed] [Google Scholar]
- 45.Hughes CS, Postovit LM, Lajoie GA. Matrigel: a complex protein mixture required for optimal growth of cell culture. Proteomics. 2010;10:1886–1890. doi: 10.1002/pmic.200900758. [DOI] [PubMed] [Google Scholar]
- 46.Streuli CH, Bailey N, Bissell MJ. Control of mammary epithelial differentiation: basement membrane induces tissue-specific gene expression in the absence of cell–cell interaction and morphological polarity. J. Cell Biol. 1991;115:1383–1395. doi: 10.1083/jcb.115.5.1383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Streuli CH, Schmidhauser C, Bailey N, Yurchenco P, Skubitz AP, Roskelley C, Bissell MJ. Laminin mediates tissue-specific gene expression in mammary epithelia. J. Cell Biol. 1995;129:591–603. doi: 10.1083/jcb.129.3.591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Singer CF, Gschwantler-Kaulich D, Fink-Retter A, Haas C, Hudelist G, Czerwenka K, Kubista E. Differential gene expression profile in breast cancer-derived stromal fibroblasts. Breast Cancer Res. Treat. 2008;110:273–281. doi: 10.1007/s10549-007-9725-2. [DOI] [PubMed] [Google Scholar]
- 49.Krause S, Maffini MV, Soto AM, Sonnenschein C. The microenvironment determines the breast cancer cells’ phenotype: organization of MCF7 cells in 3D cultures. BMC Cancer. 2010;10:263. doi: 10.1186/1471-2407-10-263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Provenzano PP, Eliceiri KW, Campbell JM, Inman DR, White JG, Keely PJ. Collagen reorganization at the tumor–stromal interface facilitates local invasion. BMC Med. 2006;4:38. doi: 10.1186/1741-7015-4-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Alcaraz J, Xu R, Mori H, Nelson CM, Mroue R, Spencer VA, Brownfield D, Radisky DC, Bustamante C, Bissell MJ. Laminin and biomimetic extracellular elasticity enhance functional differentiation in mammary epithelia. EMBO J. 2008;27:2829–2838. doi: 10.1038/emboj.2008.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Emerman JT, Pitelka DR. Maintenance and induction of morphological differentiation in dissociated mammary epithelium on floating collagen membranes. In Vitro. 1977;13:316–328. doi: 10.1007/BF02616178. [DOI] [PubMed] [Google Scholar]
- 53.Larsen M, Artym VV, Green JA, Yamada KM. The matrix reorganized: extracellular matrix remodeling and integrin signaling. Curr. Opin. Cell Biol. 2006;18:463–471. doi: 10.1016/j.ceb.2006.08.009. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.