Summary
All materials undergo an aging process which is characterized essentially by changes of the rigidity (stiffness), of the ability to absorb the stresses (toughness) and then ultimately in the mechanical resistance (strength). Both cortical and trabecular bone undergo a continuous process of structural remodeling with the main aim to preserve their biomechanical properties. An imbalance in this process, which promotes bone resorption, results in a quantitative loss of bone tissue and in a qualitative alteration of the skeletal microarchitecture, as you can see in osteoporosis, rheumatoid arthritis or bone metastases.
Cortical component has a prominent role on strength therefore loss of cortical bone that is prevalent in elderly may explain the higher frequency of fractures of bones composed mainly of cortical bone such as the proximal femur.
Remodeling inhibition with denosumab improved structural strength without altering material properties, that can be primarily explained by the combined effects of increased trabecular and cortical bone mass, and reductions in trabecular eroded surfaces and particularly cortical porosity.
Denosumab for its mechanism of action and pharmacokinetics results in a significant, early and continued increase in BMD with enhanced bone strength improving both cortical and trabecular bone.
Keywords: denosumab, cortical bone, bone quality
Background
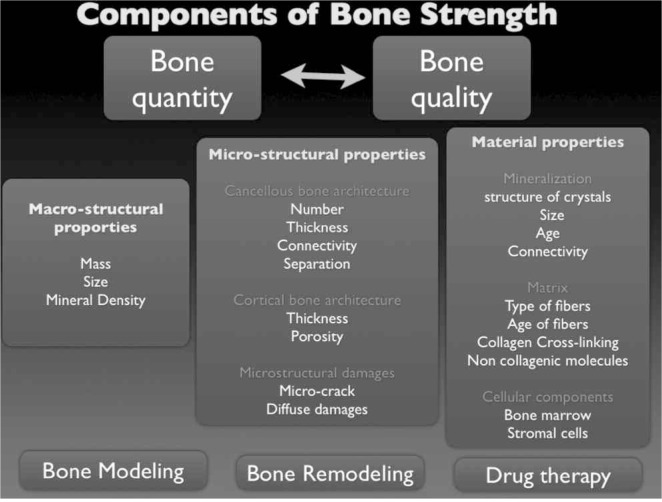
From a historical perspective, clinical researchers were focused primarily on the pathogenesis of bone fragility interpreted as an effect of the increase in bone remodeling of the trabecular tissue, especially at the spine. It’s well known, however, that the most of fragility fractures occurs at non-vertebral sites where bone is composed mainly by compact (or cortical) tissue. This tissue accounts for 80% of the total bone mass of an adult skeleton (1). Both cortical and trabecular bone undergo a continuous process of structural remodeling with the main aim to preserve their biomechanical properties. It’s well known, in fact, that all materials undergo an aging process that is characterized essentially by changes in rigidity (stiffness), ability to absorb the stresses (toughness) and in the mechanical resistance (strength). Bone strength relies on many factors as shown in Figure 1.
Figure 1.
Components of bone strength.
Bone remodeling is carried out by multicellular units that work in sequence in certain micro-clusters of the skeleton. In particular, osteoclasts provide to bone resorption and osteoblasts reform more or less the same amount immediately after. An imbalance in this process, which promotes bone resorption, results in a quantitative loss of bone tissue and in a qualitative alteration of the skeletal microarchitecture, as you can see in clinical conditions such as osteoporosis, rheumatoid arthritis and bone metastases (2).
Soon after menopause, the remodeling process proceeds rapidly in the trabecular bone leading to trabecular resorption and disruption resulting in progressive lower remodeling surfaces. On the contrary the remodeling is progressively increasing with age in the cortical bone as a consequence of increasing resorption at endosteal and intracortical surfaces, with progressive increase of cortical porosity and “spongiosization” of subendosteal bone.
Bjornerem et al. wrote that “intracortical remodeling may be self perpetuating by creating intracortical porosity and so more bone surface for remodeling to occur upon, while remodeling upon the trabecular bone surface is self limiting because it removes trabeculae with their surface”(3). As the trabecular bone is lost, the remaining cortical bone becomes even more important for bone strength.
Zebaze et al. in a cross-sectional study, using a high-resolution peripheral CT to quantify cortical and trabecular bone loss from the distal radius of adult women, and measuring porosity by scanning electron microscopy, demonstrated that rapid bone loss after menopause is mainly trabecular, whereas the majority of bone loss in the 65–79 year age group is cortical, and after the age of 80, almost 90% of bone loss is cortical (4).
These data could partly explain the increasing incidence of hip fractures with age. According to the analyses performed on the National Hospitalizations Database, the overall number of hip and other “minor” fragility fractures occurring each year in Italy has been estimated of almost 410,000 events. The annual incidence of the overall most common fragility fractures (hip, wrist, vertebral, humeral, and ankle fractures) per 100 inhabitants has been estimated up to 1.53 in men aged older than 65 years and up to 3.94 in women of the same age group. The incidence per 100 inhabitants reached 2.35 and 4.67 in men and women aged older than 75 years, respectively (with women aged older than 75 years the age group in which the highest number of fragility fractures was detected); and if the current trends continue, this number is expected to triple in the next 50 years (5).
The medical and social cost of hip fractures in Italy is about 1 billion euros, most of which for surgery and rehabilitation (6). A comparative analysis based on national hospitalization records shows that the burden of hip fractures in Italy is comparable to that of AMI and strokes (7). Half of people surviving a hip fracture can no longer live independently and about 20% die within the first year after hip fracture.
Pharmacological approach to osteoporosis
According to the guidelines promoted by the Italian Society of Orthopedics and Traumatology (SIOT), a comprehensive secondary prevention should include both osteoporosis assessment and treatment (pharmacological and non pharmacological approaches) (8).
Nowadays, there are drug therapies able to prevent or reverse the excessive bone resorption by inhibiting osteoclasts. Nitrogenous Bisphosphonates are still the gold standard of anti-resorptive drugs. They bind to hydroxyapatite crystals with high affinity and the complex hydroxyapatite/bisphosphonate is phagocytosed by the osteoclast during the phase of resorption and the nitrogenous bisphosphonate, non-digestible by lysosomal enzymes, inhibits the metabolic pathway of mevalonate, the same that leads to the synthesis of endogenous cholesterol, with locking of the production of some small proteins and thus apoptosis of the same osteoclast. In this way there is a marked reduction of bone resorption, which will determine as effect of the coupling, a subsequent reduction of new bone apposition (9,10). For their pharmacokinetic characteristics, bisphosphonates are distributed electively in gaps of bone resorption present mainly in trabecular bone, and in smaller amount in compact bone where exert a quantitatively lower action. For this reason the bisphosphonates have a smaller activity on the risk of fracture at level of compact bone, especially at the level of the radio. A new approach, which is also targeted to the inhibition of osteoclastic bone resorption, is the inhibition of RANKL.
Several pathways, involving cytokines, hormones and growth factors, affect bone loss; among them an important role is played by RANK/RANKL/OPG interaction (11). Discovery and characterization of a new pathway involving the receptor activator of nuclear factor-kB (RANK) ligand (RANKL), its receptor RANK, and its soluble decoy receptor osteoprotegerin (OPG) has been a crucial step towards the understanding of bone biology, disclosing an innovative way to develop a targeted treatment for bone diseases characterized by unbalanced osteoclastic activity (12–16). RANKL, by binding to osteoclast surface receptor RANK is a key factor for maturation, proliferation and fusion of pre-osteoclasts, furthermore it is crucial to osteoclast activation and survival. OPG is a soluble glycoprotein receptor that is a competitive inhibitor of RANKL responsible for fine tuning of RANKL activation of osteoclasts (17). The ratio of RANKL-OPG is strictly balanced, allowing physiological bone remodeling, essential for bone health and a functional skeleton (18–21).
Starting from peri-menopause and continuing in the immediate postmenopausal period, levels of estrogens fall, resulting, throughout a complex and diverse cascade of factors, in a significant raise of RANKL secretion by osteoblast and, as a consequence, in an increase of osteoclasts. This enhanced bone resorption activity is not anymore balanced by the osteoblast bone formation activity, causing a loss in bone mass and a progressive destruction of bone microarchitecture.
Induced androgen and estrogen deficiency, systemic glucocorticoid exposure (22), T cell activation as in rheumatoid arthritis and skeletal malignancies (23, 24) also enhance the release of RANKL, ending up in promoting osteoclastogenesis and inducing unbalanced bone loss.
Denosumab is a fully human monoclonal antibody that binds with high affinity and high specificity to RANKL, thereby preventing RANKL from binding to RANK (25). RANKL inhibition causes an important inhibition of osteoclast activity, resulting in restoring a better balance between bone resorption and formation, ending up in an increase in bone mass (26, 27).
Denosumab is available as a subcutaneous prefilled injection to be administered subcutaneously every 6 months. Half-life of approximately 25 days, allows extended-interval dosing (27).
Large reductions in bone resorption markers, specifically serum type 1 C-telopeptide (CTX), are measurable within 3 days, reaching maximum reduction of 89% at one month (28, 29).
Interestingly, after discontinuation of denosumab, markers of bone resorption return to baseline in one year (29–32). Denosumab, coherently with its IgG2 nature, has a nonlinear, dose-dependent pharmacokinetics. Studies did not show any pharmacokinetic difference between postmenopausal women in relation to age, ethnicity, or body weight (30). Furthermore, it shows the typical distribution of a monoclonal antibody, it allows the activity at both the trabecular bone and the compact bone.
The role of denosumab in the treatment of osteoporosis
The FREEDOM trial started in 2004, aimed at evaluating the anti-fracture efficacy of denosumab in women with post-menopausal osteoporosis (31). Dosing schedule, as established after the phase 2 trial, was 60 mg administered subcutaneously every 6 months.
A total number of 7,868 women, ages varying between 60 and 90 years, were enrolled in the study, 3,933 in the denosumab group and 3,935 in the placebo.
BMD measured in terms of T-score at lumbar spine or at total hip was between −2.5 and −4.0 SD. Eligibility criteria excluded women having at baseline any severe or more than two moderate prevalent vertebral fractures. Further exclusion criteria were having prior three-year treatment with oral bisphosphonates, treatment with i.v. bisphosphonates, fluorides, glucocorticoids, PTH, strontium, calcitonin, or other agents, potentially affecting BMD during the 6 weeks prior to the start of the trial. At the end of the third year of study the relative risk reduction versus placebo of new vertebral fractures was 68% (P<0.001); denosumab also showed a relative risk reduction of hip fracture of 40% (P=0.04) and of non vertebral fractures of 20% (P=0.01).
There was no increase in the risk of cancer, infection, cardiovascular disease, delayed fracture healing, or hypocalcemia, and there were no cases of osteonecrosis of the jaw and no adverse reactions to the injection of denosumab. A statistically significant increase of eczema and of cellulitis (including erisipela) was measured.
In order to explore further long term efficacy and safety of denosumab in women with post-menopausal osteoporosis, a 10 year extension of the FREEDOM study was planned. Results of the second year of extension (accounting for 5 years of continuative denosumab treatment) were recently published. Remarkably BMD continued to rise throughout all five years, achieving a 13.7% increase versus baseline at lumbar spine and a 7.0% increase versus baseline at total hip, showing no sign of loss of efficacy in 5 years time. Furthermore the incidence of new vertebral fractures and non-vertebral fractures remained low and there was no increase in the incidence of adverse events up to the end of the fifth year of treatment with denosumab (32).
Effect of denosumab on BMD and bone turnover markers has been evaluated in several further phase III studies. In 2008 a 24 month study exploring efficacy and safety of treatment with denosumab of postmenopausal women with low bone mass (lumbar spine T-scores between −1.0 and −2.5) was published. Results of this study, named DEFEND (Denosumab Fortifies Bone Density), proved denosumab to significantly increase BMD at the lumbar spine of 6.5% versus baseline and of 7.1% versus placebo (33). Significant BMD increases were also described at total hip, one-third radius, and total body (p<0.0001 each vs placebo). Bone turnover markers (CTX and P1NP) decreased significantly compared to placebo. Overall incidence of adverse events that was similar to placebo.
As previously explained, the different mode of action of denosumab, involving inhibition of osteoclast formation, activation and survival, had a peculiar reflection on his action on BMD and bone turnover markers (BTM).
Differences among denosumab and other antiresorptive drugs
To better examine this difference, in particular with bisphosphonates, denosumab was compared with alendronate, as the current gold standard for PMO treatment, in two large randomized, multicentric, double-blind, double-dummy studies: the DECIDE and the STAND studies (34–36).
Both studies compared denosumab 60mg/Q6M + placebo (tablets QW) to branded alendronate QW + placebo (sc injections Q6M) for a total of 12 months. For both studies, the primary endpoint was change from baseline in total hip BMD; secondary endpoints were changes of BMD from baseline at other skeletal sites (including lumbar spine, femoral neck, and one-third radius) and in bone turnover markers (CTX1 and P1NP).
Main difference between the two studies was that to participate to the DECIDE study, subjects (n 1,189) had to be naïve to any PMO treatment, whereas to be eligible for the STAND study, subjects (n 504) had to have received alendronate treatment equivalent to 70mg/week for at least 6 months before the beginning of the study.
Both studies showed a higher effect of denosumab, in terms of BMD increase, in all measured skeletal sites. Denosumab superior efficacy was proved to be independent of the trabecular/cortical bone ratio of the skeletal sites examined and of the presence and duration (up to ten years) of previous alendronate treatment.
The effect of the two treatments on CTX and P1NP appeared also to be very different. Whereas alendronate showed a continuous flat inhibition of both BTMs, denosumab demonstrated a faster and deeper reduction of both BTM, followed to a slow recovery across the 6 months of interval between one denosumab injection and the subsequent.
Actions of denosumab on cortical and trabecular bone
Results from this head to head studies and the difference in the mode of action of denosumab compared to bisphosphonates, suggested a difference in their effect on the diverse compartments of bones (cortical and trabecular). This was further explored in a 3-year sub-study of the FREEDOM trial. Patients enrolled in this analysis were assessed by whole-body spiral computed tomography at baseline and months 12, 24, and 36. Denosumab treated patients showed a significant increase in QCT measured vBMD and DXA measured aBMD from baseline and compared with placebo at the total hip, spine, and femoral neck at every time point (37). At the end of the study, treatment with denosumab produced an increase from baseline in overall vBMD of 12.6% at the trabecular spine, 4.4% at the total hip region, and 2.9% at the femoral neck. When compared to placebo group, denosumab treated patients overall vBMD showed a raise of 21.8% at the trabecular spine, 7.8% at the total hip region, and 5.9% at the femoral neck. Intriguingly, data from the total hip region demonstrated an increase in both vBMD and BMC in the denosumab group, whereas no change in volume was measured. This evidenced an increase in both bone mass and density, likely due to the large effect of denosumab treatment on cortical bone.
Kostenuik et al. examined the effects of denosumab, on bone histomorphometry in adult ovariectomized cynomolgus monkeys, and demonstrated that remodeling inhibition with denosumab improved structural strength without altering material properties, that can be primarily explained by the combined effects of increased trabecular and cortical bone mass, and reductions in trabecular eroded surfaces and particularly cortical porosity (38).
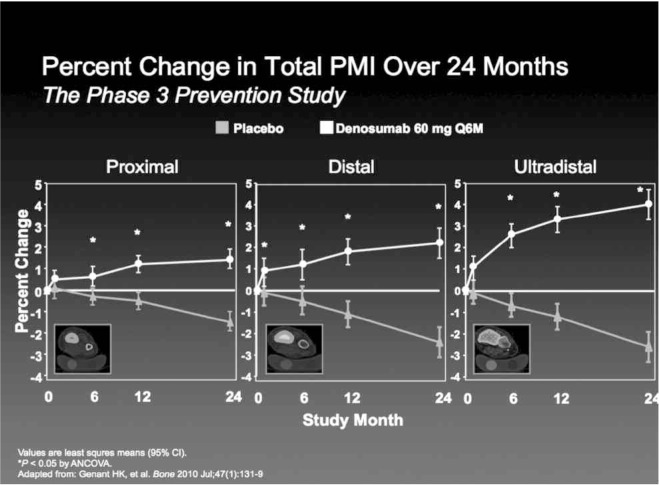
Genant et al. analyzing QCT scans of the radius of patients enrolled in DEFEND trial, obtained data about bone strength parameters such as the density-weighted polar moment of inertia (PMI), which relates to the ability of a bone to resist torsion. This study confirmed that denosumab significantly increased BMD, BMC with positive changes in measures of bone geometry, leading to an increase in derived indices of bone strength across the scanned length of the radius, a bone characterized by a very different composition in cortical or cancellous tissue along its length (39) (Figure 2).
Figure 2.
Denosumab improves density and strength parameters as measured by QCT of the radius in postmenopausal women with low bone mineral density (Adapted from: Genant HK, et al. Bone 2010 Jul; 47(1):131–9).
Conclusion
Bone strength is determined by many factors, ranging from the macroscopic geometry to ultramicroscopic arrangement of the structural components, on which act complex biological phenomena such as modeling, remodeling and aging of the bone tissue.
Cortical component has a prominent role on strength therefore loss of cortical bone that is prevalent in elderly may explain the higher frequency of fractures, of bones composed mainly of cortical bone such as the proximal femur.
Denosumab for its mechanism of action and pharmacokinetics results in a significant, early and continued increase in BMD with enhanced bone strength improving both cortical and trabecular bone.
References
- 1.Riggs BL, Melton LJ, Robb R, et al. A population-based assessment of rates of bone loss at multiple skeletal sites: evidence for substantial trabecular bone loss in young adult women and men. J Bone Miner Res. 2008;23:205–214. doi: 10.1359/JBMR.071020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Raisz LG. Pathogenesis of osteoporosis: concepts, conflicts, and prospects. J Clin Invest. 2005;115:3318–3325. doi: 10.1172/JCI27071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bjornerem A, Ghasem-Zadeh A, Bui M, Wang X, Rantzau C, Nguyen T, Hopper J, Zebaze R, Seeman E. Remodeling markers are associated with larger intracortical surface area but smaller trabecular surface area: a twin study. Bone. 2011;49:1125–1130. doi: 10.1016/j.bone.2011.08.009. [DOI] [PubMed] [Google Scholar]
- 4.Zebaze RM, Ghasem-Zedah A, Bohte A, Iuliano-Burns S, Mirams M, Price R, J Mackei E, Seman E. Intracortical remodeling and porosity in the distal radius and post-mortem femurs of women: a cross-sectional study. The Lancet. 2010;375:1729–1736. doi: 10.1016/S0140-6736(10)60320-0. [DOI] [PubMed] [Google Scholar]
- 5.Tarantino U, Capone A, Planta M, D'Arienzo M, Letizia Mauro G, Impagliazzo A, Formica A, Pallotta F, Patella V, Spinarelli A, Pazzaglia U, Zarattini G, Roselli M, Montanari G, Sessa G, Privitera M, Verdoia C, Corradini C, Feola M, Padolino A, Saturnino L, Scialdoni A, Rao C, Iolascon G, Brandi ML, Piscitelli P. The incidence of hip, forearm, humeral, ankle, and vertebral fragility fractures in Italy: results from a 3-year multicenter study. Arthritis Res Ther. 2010;12(6):R226. doi: 10.1186/ar3213. Epub 2010 Dec 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Piscitelli P, Brandi ML, Tarantino U, Baggiani A, Distante A, Muratore M, Grattagliano V, Migliore A, Granata M, Guglielmi G, Gimigliano R, Iolascon G. Incidence and socio-economic burden of hip fractures in Italy: extension study 2003–2005. Reumatismo. 2010:62. doi: 10.4081/reumatismo.2010.113. [DOI] [PubMed] [Google Scholar]
- 7.Piscitelli P, Iolascon G, Argentiero A, Chitano G, Neglia C, Marcucci G, Pulimeno M, Benvenuto M, Mundi S, Marzo V, Donato D, Baggiani A, Migliore A, Granata M, Gimigliano F, Di Blasio R, Gimigliano A, Renzulli L, Brandi ML, Distante A, Gimigliano R. Incidence and costs of hip fractures vs stroke and acute myocardial infarction in Italy: comparative analysis based on national hospitalization records. Clinical Interventions in Aging. 2012;7:1–9. doi: 10.2147/CIA.S36828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kanis JA, Burlet N, Cooper C, Delmas PD, Reginster JY, Borgstrm F, Rizzoli R. European Guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporosis Int. 2008 doi: 10.1007/s00198-008-0560-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Weinstein RS, Roberson PK, Manolagas SC. Giant osteoclast formation and long-term oral bisphosphonate therapy. N Engl J Med. 2009;360:53–62. doi: 10.1056/NEJMoa0802633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cramer JA, Amonkar MM, Hebborn A, Altman R. Compliance and persistence with bisphosphonate dosing regimens among women with post-menopausal osteoporosis. Curr Med Res Opin. 2005;21:1453–60. doi: 10.1185/030079905X61875. [DOI] [PubMed] [Google Scholar]
- 11.Hofbauer LC, Schoppet M. Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. JAMA. 2004;292:490–495. doi: 10.1001/jama.292.4.490. [DOI] [PubMed] [Google Scholar]
- 12.McClung M. Role of RANKL inhibition in osteoporosis. Arthritis Res Ther. 2007;9:S3. doi: 10.1186/ar2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Eghbali-Fatourechi G, Khosla S, Sanyal A, Boyle WJ, Lacey DL, Riggs BL. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women. Journal of Clinical Investigation. 2003;111:1221–1230. doi: 10.1172/JCI17215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Burgess TL, Qian Y, Kaufman S, et al. The ligand for osteoprotegerin (OPGL) directly activates mature osteoclasts. J Cell Biol. 1999;145:527–538. doi: 10.1083/jcb.145.3.527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lacey DL, Tan HL, Lu J, et al. Osteoprotegerin ligand modulates murine osteoclast survival in vitro and in vivo. Am J Pathol. 2000;157:435–448. doi: 10.1016/S0002-9440(10)64556-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bucay N, Sarosi I, Dunstan CR, Morony S, Tarpley J, Capparelli C, Scully S, Tan HL, Xu W, Lacey DL, Boyle WJ, Simonet WS. Osteoprotegerin-deficient mice develop early onset osteoporosis and arterial calcification. Genes and Development. 1998;12:1260–1268. doi: 10.1101/gad.12.9.1260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kong YY, Yoshida H, Sarosi I, Tan HL, Timms E, Capparelli C, Morony S, Oliveira-dos-Santos AJ, Van G, Itie A, Khoo W, Wakeham A, Dunstan CR, Lacey DL, Mak TW, Boyle WJ, Penninger JM. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature. 1999;397:315–323. doi: 10.1038/16852. [DOI] [PubMed] [Google Scholar]
- 18.Stolina M, Dwyer D, Ominsky MS, Corbin T, Van G, Bolon B, Sarosi I, McCabe J, Zack DJ, Kostenuik P. Continuous RANKL inhibition in osteoprotegerin transgenic mice and rats suppresses bone resorption without impairing lymphorganogenesis or functional immune responses. Journal of Immunology. 2007;179:7497–7505. doi: 10.4049/jimmunol.179.11.7497. [DOI] [PubMed] [Google Scholar]
- 19.Min H, Morony S, Sarosi I, Dunstan CR, Capparelli C, Scully S, Van G, Kaufman S, Kostenuik PJ, Lacey DL, et al. Osteoprotegerin reverses osteoporosis by inhibiting endosteal osteoclasts and prevents vascular calcification by blocking a process resembling osteoclastogenesis. J Exp Med. 2000;192:463–474. doi: 10.1084/jem.192.4.463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Capparelli C, Morony S, Warmington K, Adamu S, Lacey D, Dunstan CR, Stouch B, Martin S, Kostenuik PJ. Sustained antiresorptive effects after a single treatment with human recombinant osteoprotegerin (OPG): a pharmacodynamic and pharmacokinetic analysis in rats. J Bone Miner Res. 2003;18:852–858. doi: 10.1359/jbmr.2003.18.5.852. [DOI] [PubMed] [Google Scholar]
- 21.Bateman TA, Dunstan CR, Ferguson VL, Lacey DL, Ayers RA, Simske SJ. Osteoprotegerin mitigates tail suspension-induced osteopenia. Bone. 2000;26:443–449. doi: 10.1016/S8756-3282(00)00256-8. [DOI] [PubMed] [Google Scholar]
- 22.Tirpitz C, Epp S, Klaus J, Mason R, Hawa G, Brinskelle-Schmal N, Hofbauer LC, Adler G, Kratzer W, Reinshagen M. Effect of systemic glucocorticoid therapy on bone metabolism and the osteoprotegerin system in patients with active Crohn’s disease. European Journal of Gastroenterology & Hepatology. 2003;15:1165–1170. doi: 10.1097/00042737-200311000-00003. [DOI] [PubMed] [Google Scholar]
- 23.Standal T, Seidel C, Hjertner Ø, Plesner T, Sanderson RD, Waage A, Borset M, Sundan A. Osteoprotegerin is bound, internalized, and degraded by multiple myeloma cells. Blood. 2002;100:3002–3007. doi: 10.1182/blood-2002-04-1190. [DOI] [PubMed] [Google Scholar]
- 24.Michigami T, Ihara-Watanabe M, Yamazaki M, Ozono K. Receptor activator of uclear factor-kB ligand (RANKL) is a key molecule of osteoclast formation for bone metastasis in a newly developed model of human neuroblastoma. Cancer Research. 2001;61:1637–1644. [PubMed] [Google Scholar]
- 25.McClung MR, Lewiecki EM, Cohen SB, Bolognese MA, Woodson GC, Moffett AH, Peacock M, Miller PD, Ledermann SN, Chestnut CH, LAin D, Kivitz AJ, Halloway DL, Zhang C, Peterson MC, Bekker PJ. Denosumab in post-menopausal women with low bone mineral density. N Engl J Med. 2006;23:821–831. doi: 10.1056/NEJMoa044459. [DOI] [PubMed] [Google Scholar]
- 26.McClung MR, Lewiecki EM, Cohen SB, Bolognese MA, Woodson GC, Moffett AH, Peacock M, Miller PD, Ledermann SN, Chestnut CH, LAin D, Kivitz AJ, Halloway DL, Zhang C, Peterson MC, Bekker PJ. Denosumab in post-menopausal women with low bone mineral density. N Engl J Med. 2006;23:821–831. doi: 10.1056/NEJMoa044459. [DOI] [PubMed] [Google Scholar]
- 27.Bekker PJ, Holloway DL, Rasmussen AS, Murphy R, Martin SW, Leese PT, et al. A single-dose placebo-controlled study of AMG 162, a fully human monoclonal antibody to RANKL, in postmenopausal women. J Bone Miner Res. 2004;19:1059–66. doi: 10.1359/JBMR.040305. [DOI] [PubMed] [Google Scholar]
- 28.Bone HG, Bolognese MA, Yuen CK, et al. Effects of denosumab treatment and discontinuation on bone mineral density and bone turnover markers in postmenopausal women with low bone mass. J Clin Endocrinol Metab. 2011;96:972–80. doi: 10.1210/jc.2010-1502. [DOI] [PubMed] [Google Scholar]
- 29.Miller PD, Bolognese MA, Lewiecki EM, et al. Effect of denosumab on bone density and turnover in postmenopausal women with low bone mass after long-term continued, discontinued, and restarting of therapy: a randomized blinded phase 2 clinical trial. Bone. 2008;43:222–9. doi: 10.1016/j.bone.2008.04.007. [DOI] [PubMed] [Google Scholar]
- 30.McClung MR, Lewiecki EM, Geller ML, Bolognese MA, Peacock M, WeinsteinRL B, Ding B, Rockabrand E, Wagman RB, Miller PD. Effect of denosumab on bone mineral density and biochemical markers of bone turnover: 8-year results of a phase 2 clinical trial. Osteoporos Int JUL. 2012 doi: 10.1007/s00198-012-2052-4.. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rizzoli R, Boonen S, Bone HG, Minisola S, Wang A, Benhamou CL, et al. The effect of denosumab on vertebral fracture risk by type and subgroup: results from the FREEDOM trial [abstract] Osteoporos Int. 2010;21:S357. [Google Scholar]
- 32.Papapoulos S, Chapurlat R, Libanati C, Brandi ML, Brown JP, Czerwinski E, Krieg M, Man Z, Mellstrom D, Radominski SC, Reginster J, Resch H, Roman Ivorra JA, Roux C, Vittinghoff E, Austin M, Daizadeh N, Bradley MN, Andreas Grauer A, Cummings S, Bone HG. Five Years of Denosumab Exposure in Women With Postmenopausal Osteoporosis: Results From the First Two Years of the FREEDOM Extension. JBMR. 2012;27(3):694–701. doi: 10.1002/jbmr.1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bone HG, Bolognese MA, Yuen CK, et al. Effects of denosumab on bone mineral density and bone turnover in postmenopausal women. J Clin Endocrinol Metab. 2008;93:2149–57. doi: 10.1210/jc.2007-2814. [DOI] [PubMed] [Google Scholar]
- 34.Brown JP, Deal C, de Gregorin LH, et al. Effect of densoumab vs alendronate on bone turnover markers and bone mineral density changes at 12 months based on baseline bone turnover level. Program and abstracts from the American Society of Bone and Mineral Research 30th Annual Meeting; September 12–16, 2008; Montréal, Quebéc, Canada. Abstract 128535. [Google Scholar]
- 35.Kendler DL, Roux C, Benhamou CL, et al. Effects of denosumab on bone mineral density and bone turnover in postmenopausal women transitioning from alendronate therapy. J Bone Miner Res. 2010;25:72–81. doi: 10.1359/jbmr.090716. [DOI] [PubMed] [Google Scholar]
- 36.Kendler DL, Benhamou CL, Brown JP, et al. Effects of denosumab vs alendronate on bone mineral density (BMD), bone turnover markers (BTM), and safety in women previously treated with alendronate. Program and abstracts from the American Society of Bone and Mineral Research 30th Annual Meeting; September 12–16, 2008; Montréal, Quebéc, Canada. Poster abstract 138. [Google Scholar]
- 37.McClung MR, Zanchetta J, Høiseth A, Kendler DL, Yuen CK, Brown JP, Stonkus S, Goemaere S, Recknor C, Woodson G, Bolognese MA, Franek E, Wang A, Cesar Libanati C. Denosumab Densitometric Changes Assessed by Quantitative Computed Tomography at the Spine and Hip in Post-menopausal Women With Osteoporosis. J Clin Densitom. 2012 May 8; doi: 10.1016/j.jocd.2012.02.014.. [DOI] [PubMed] [Google Scholar]
- 38.Kostenuik PJ, Smith SY, Jolette J, Schroeder J, Pyrah I, Ominsky MS. Decreased bone remodeling and porosity are associated with improved bone strength in ovariectomized cynomolgus monkeys treated with denosumab, a fully human RANKL antibody. Bone. 2011;49:151–161. doi: 10.1016/j.bone.2011.03.769. [DOI] [PubMed] [Google Scholar]
- 39.Genant HK, Engelke K, Hanley DA, Brown JP, Omizo M, Bone HG, Kivitz AJ, Fuerst T, Wang H, Austin M, Libanati C. Denosumab improves density and strength parameters as measured by QCT of the radius in post-menopausal women with low bone mineral density. Bone. 2010;47:131–139. doi: 10.1016/j.bone.2010.04.594. [DOI] [PubMed] [Google Scholar]