Abstract
European-like field isolates of porcine reproductive and respiratory syndrome virus (PRRSV) have recently emerged in North America. The full-length genomic sequence of an index isolate characterized in 1999, strain EuroPRRSV, served as the reference strain for further studies of the evolution and epidemiology of European-like isolates (type 1) in the United States. Strain EuroPRRSV shared 90.1 to 100% amino acid identity with the prototype European strain, Lelystad, within the structural and nonstructural open reading frames (ORFs) and 95.3% overall nucleotide identity. The 5′ untranslated region and two nonstructural regions within ORF 1 were closely examined due to significant divergence from strain Lelystad. A 51-bp deletion in a region within ORF 1a, coding for nonstructural protein 2 (NSP2), was observed. Sequence analysis of the structural ORFs 2 to 7 of additional European-like isolates indicated that these isolates share 93% nucleotide identity with one another and 95 to 96% identity with the Lelystad strain but only 70% identity with the North American reference strain VR-2332. Phylogenetic analysis with published PRRSV ORF 3, 5, and 7 nucleotide sequences indicated that these newly emerging isolates form a clade with the Lelystad and United Kingdom PRRSV isolates. Detailed analysis of four of these isolates with a panel of 60 monoclonal antibodies directed against the structural proteins confirmed a recognition pattern that was more consistent with strain Lelystad than with other North American isolates.
Porcine reproductive and respiratory syndrome (PRRS) virus (PRRSV) is the causative agent of PRRS, a disease characterized by reproductive failure in sows, including early farrowing with stillborn piglets and late-term abortions, as well as respiratory distress in young pigs and an influenza-like disease in grow-finish swine (58, 65). This syndrome was first recognized as a mystery swine disease in the United States in 1987 (27) and was subsequently identified in Europe (Lelystad virus [LV]) (74) and in the United States (ATCC VR-2332) (3, 9). The disease is now endemic in many swine-producing countries (M. L. Meredith, PRRS, Pig Disease Information Centre, Cambridge, United Kingdom [http://www.pighealth.com]) and is the most economically important swine disease in the United States (59, 86).
PRRSV is a small enveloped virus with a single-stranded positive-sense RNA genome and is a member of the family Arteriviridae in the order Nidovirales (6, 14). Other members of the arterivirus family include lactate dehydrogenase-elevating virus of mice, equine arteritis virus (EAV), and simian hemorrhagic fever virus (55). Each of these viruses replicates preferentially in macrophages and establishes a persistent infection in its natural host (54). Several members of the nidovirus order have been shown to undergo high-frequency recombination (50, 81, 83). Because of unique recombinatorial properties (81, 83), widespread persistence, intermingling of virus types, and changes in swine management, PRRSV has been a key subject of evolutionary interest (17, 18, 19, 33, 39, 67, 69).
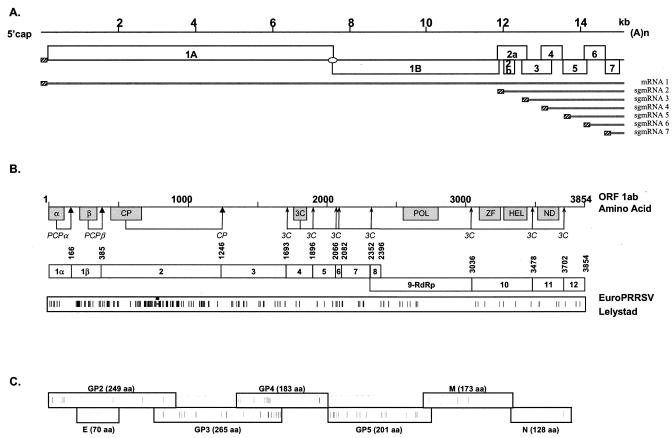
The 5′-capped and 3′-polyadenylated genome of PRRSV is 15.1 to 15.5 kb in length and consists of at least eight open reading frames (ORFs). ORF 1a and ORF 1b are located immediately downstream of the 5′ untranslated region (UTR) and occupy more than two-thirds of the genome. ORF 1a is translated directly from the genomic RNA. ORF 1b is expressed by a ribosomal translational frameshift yielding a large ORF 1ab polyprotein that is proteolytically cleaved into products involved in virus transcription and replication. ORFs 2 to 7 code for PRRSV structural proteins and are located at the 3′ end of the genome, with ORFs 2 to 6 encoding polypeptides characteristic of membrane-associated proteins. These genes are expressed from a 3′-coterminal nested set of functionally monocistronic subgenomic mRNAs (sgmRNAs) (Fig. 1A) (10, 36, 54). The sgmRNAs encode four glycoproteins (sgmRNA2/GP2, sgmRNA3/GP3, sgmRNA4/GP4, and sgmRNA5/GP5), two unglycosylated proteins (sgmRNA2/2b envelope [E] and sgmRNA6 membrane [M]), and a nucleocapsid protein (sgmRNA7/N) (36, 42, 79).
FIG. 1.
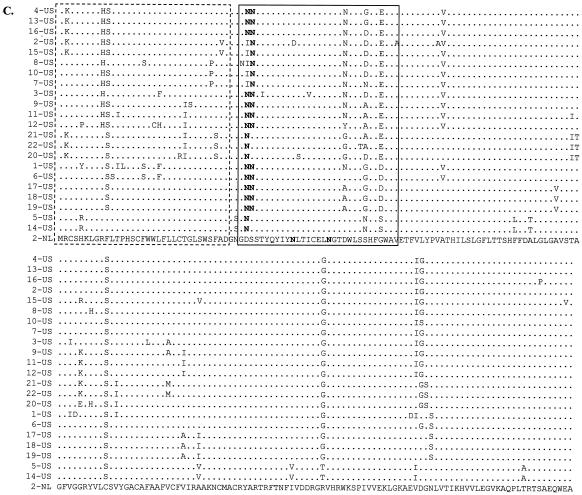
(A) PRRSV genome organization and expression strategy include a leader sequence found in all mRNAs (hatched boxes) and a ribosomal frameshift (oval) used to produce ORF 1ab (see text). (B) ORF 1ab polypeptide predicted functional domains (papain-like cysteine proteases [PCPα and PCPβ], cysteine protease [CP], 3C-like serine protease [3C], polymerase [POL], zinc finger [ZF], helicase [HEL], and a domain found in all nidoviruses [ND]), the putative proteolytic cleavage sites and resultant individual nonstructural proteins (NSPs 1 to 12), and a representation of the amino acid differences between EuroPRRSV and LV within the replicase ORFs. Amino acid numbering is based on the ORF 1ab protein of strain Lelystad. (C) Graphical representation of the amino acid differences between EuroPRRSV and LV within the structural proteins coded for by LV nt 11796 to 14984. The length of each protein is shown in parentheses.
The North American and European isolates of PRRSV share morphological and structural similarities, but despite their nearly simultaneous emergence on their respective continents, they display significant molecular and antigenic variation. The prototype virus strains from the two continents share approximately 60% nucleotide identity at the genome level (39) and have been divided into two main PRRSV genotypes: European (type 1) and North American (type 2). Antigenically, PRRSV has also been divided into two subgroups, with subgroup A representing the European prototype LV isolate and subgroup B representing the U.S. prototype ATCC VR-2332 isolate (41). Antigenic variations among PRRSV isolates are well documented (26, 32, 75; reviewed in reference 34). Since the emergence of PRRSV, intermingling of the two genotypes has occurred in Europe as a result of the use of a live, United States-derived (type 2) PRRSV vaccine (5, 31). There have been several recent reports of European-like PRRSV strains appearing in North America. Dewey et al. (15) reported an LV-like strain of PRRSV in swine imported to Canada from Europe. European-like PRRSV in American swine herds with no apparent European source has been reported by a number of veterinary diagnostic laboratories (Minnesota Veterinary Diagnostic Laboratory [C. Mahlum, K. Rossow, J. Collins, and K. Faaberg, Proc. Conf. Res. Work. Anim. Dis., abstr. 164, 2000], South Dakota Animal Disease Research and Diagnostic Laboratory [S. Ropp, Y. Fang, M. Bien, B. Arndt, S. Preszler, P. Steen, J. Christopher-Hennings, D. Benfield, and E. Nelson, Proc. Conf. Res. Work. Anim. Dis., abstr. 166, 2002], and Missouri-Columbia Veterinary Medical Diagnostic Laboratory [S. K. Schommer, R. Stammer, E. Vaughn, D. Polson, W. Chittick, and S. B. Kleiboeker, GenBank accession no. AF514803]). Some of the unique characteristics of these European-like isolates can have a significant impact on diagnostic approaches to detecting PRRSV in U.S. herds and PRRSV management strategies within U.S. swine herds. This report details the unique genomic features of the European-like type 1 U.S. reference isolate EuroPRRSV and provides fundamental genetic and antigenic characterization of several European-type isolates that have emerged within the United States since 1999.
MATERIALS AND METHODS
Virus isolates.
EuroPRRSV was identified clinically as a mild case of PRRS in Iowa in 1999. The virus isolate was reamplified in swine due to poor growth in vitro and produced clinical symptoms similar to those of the original isolate. Other isolates have since been identified in diverse geographic locations including the Southeastern, Midwestern, and Western United States (Table 1).
TABLE 1.
PRRSV isolates utilized in this study
| Designation | Isolate | Country of isolation | Isolation datea | Accession no. (segment) | Reference |
|---|---|---|---|---|---|
| 1-US | EuroPRRSV | United States | 22/09/99 | AY366525 (complete genome) | This study |
| 2-US | MN-02-02_EU | United States | 10/04/02 | AY422798 (ORF 5) | This study |
| 3-US | MN-01-03_EU | United States | 19/09/01 | AY422799 (ORF 5) | This study |
| 4-US | MN-01-04_EU | United States | 21/08/01 | AY422800 (ORF 5) | This study |
| 5-US | MN-01-05_EU | United States | 21/08/01 | AY422801 (ORF 5) | This study |
| 6-US | MN-00-06_EU | United States | 17/11/00 | AY422802 (ORF 5) | This study |
| 7-US | MN-00-07_EU | United States | 17/11/00 | AY422803 (ORF 5) | This study |
| 8-US | MN-00-08_EU | United States | 03/08/00 | AY422804 (ORF 5) | This study |
| 9-US | MN-00-09_EU | United States | 03/08/00 | AY422805 (ORF 5) | This study |
| 10-US | MN-00-10_EU | United States | 03/08/00 | AY422806 (ORF 5) | This study |
| 11-US | MN-00-11_EU | United States | 03/08/00 | AY422807 (ORF 5) | This study |
| 12-US | MN-00-12_EU | United States | 07/07/00 | AY422808 (ORF 5) | This study |
| 13-US | SD-01-08 | United States | 08/08/01 | AY395080 (ORFs 2-7) | This study |
| 14-US | SD-01-07 | United States | 01/08/01 | AY395079 (ORFs 2-7) | This study |
| 15-US | SD-02-10 | United States | 05/05/02 | AY395081 (ORFs 2-7) | This study |
| 16-US | SD-03-12 | United States | 11/03/03 | AY395074 (ORF 5) | This study |
| 17-US | SD-03-17 | United States | 18/01/03 | AY395075 (ORF 5) | This study |
| 18-US | SD-03-15 | United States | 22/01/03 | AY395076 (ORF 5) | This study |
| 19-US | SD-03-16 | United States | 12/01/03 | AY395077 (ORF 5) | This study |
| 20-US | SD-02-11 | United States | 23/12/02 | AY395078 (ORF 5) | This study |
| 21-US | MN-01-21_EU | United States | 06/07/00 | AY422809 (ORF 5) | This study |
| 1-DK | 111/92 | Denmark | 09/04/92 | AF17167 (ORF 3), AY035944 (ORF 5-7) | 19, 46 |
| 2-DK | 12654 | Denmark | 19/12/95 | AF171672 (ORF 3), AY035902 (ORF 5), AY035947 (ORF 7) | 19, 46 |
| 3-DK | 12770/95 | Denmark | 21/12/95 | AY035903 (ORF 5), AY035948 (ORF 7) | 19 |
| 4-DK | 12985 | Denmark | 04/01/96 | AF171673 (ORF 3), AY035904 (ORF 5), AY035949 (ORF 7) | 19, 46 |
| 5-DK | 14474B | Denmark | 04/03/96 | AF171675 (ORF 3), AY035905 (ORF 5), AY035950 (ORF 7) | 19, 46 |
| 6-DK | 18794 | Denmark | 26/03/93 | AY035906 (ORF 5), AY035951 (ORF 7) | 19 |
| 7-DK | 20567 A | Denmark | 26/03/97 | AF171678 (ORF 3), AY035907 (ORF 5), AY035952 (ORF 7) | 19, 46 |
| 8-DK | 21191 | Denmark | 09/05/97 | AF171679 (ORF 3), AY035908 (ORF 5), AY035953 (ORF 7) | 19, 46 |
| 9-DK | 228 A | Denmark | 16/11/93 | AF171691 (ORF 3), AY035909 (ORF 5), AY035954 (ORF 7) | 19, 46 |
| 10-DK | 24554/97 | Denmark | 19/11/97 | AF171680 (ORF 3), AY035910 (ORF 5), AY035955 (ORF 7) | 19, 46 |
| 11-DK | 25434/98 | Denmark | 15/01/98 | AF171681 (ORF 3), AY035911 (ORF 5), AY035956 (ORF 7) | 19, 46 |
| 12-DK | 28639/98 | Denmark | 16/06/98 | AF171684 (ORF 3), AY035912 (ORF 5), AY035957 (ORF 7) | 19, 46 |
| 13-DK | 32-10/92 | Denmark | 17/03/92 | AF171687 (ORF 3), AY035913 (ORF 5), AY035958 (ORF 7) | 19, 46 |
| 14-DK | 340-1 | Denmark | 09/01/94 | AY035914 (ORF 5), AY035959 (ORF 7) | 19 |
| 15-DK | 361-4 | Denmark | 18/01/94 | AY035915 (ORF 5), AY035960 (ORF 7) | 19 |
| 16-DK | 48/92-1 | Denmark | 20/03/92 | AF171690 (ORF 3), AY035916 (ORF 5), AY035961 (ORF 7) | 19, 46 |
| 17-DK | 5767-6 | Denmark | 10/02/95 | AF171692 (ORF 3), AY035917 (ORF 5), AY035962 (ORF 7) | 19, 46 |
| 18-DK | 26666 | Denmark | 18/03/98 | AF171683 (ORF 3) | 46 |
| 19-DK | 34/92 | Denmark | 17/03/92 | AF171688 (ORF 3) | 46 |
| 20-DK | 38/8 | Denmark | 18/03/92 | AF171689 (ORF 3) | 46 |
| 21-DK | 18009/4 | Denmark | 28/09/92 | AF171677 (ORF 3) | 46 |
| 22-DK | 146/92 | Denmark | 24/09/92 | AF171676 (ORF 3) | 46 |
| 23-DK | 5544 A | Denmark | 01/02/95 | AF171696 (ORF 3) | 46 |
| 24-DK | 6501 | Denmark | 22/03/95 | AF171693 (ORF 3) | 46 |
| 25-DK | 6504 | Denmark | 22/03/95 | AF171694 (ORF 3) | 46 |
| 26-DK | 6617 | Denmark | 29/03/95 | AF171695 (ORF 3) | 46 |
| 27-DK | 13759 B | Denmark | 26/01/96 | AF171674 (ORF 3) | 46 |
| 28-DK | 26371 B | Denmark | 04/03/98 | AF171682 (ORF 3) | 46 |
| 29-DK | 31540 | Denmark | 18/11/98 | AF171685 (ORF 3) | 46 |
| 30-DK | 31690 | Denmark | 25/11/98 | AF171686 (ORF 3) | 46 |
| 31-DK | 32413 | Denmark | 08/01/99 | AF303361 (ORF 3) | 18 |
| 32-DK | 32351 | Denmark | 05/01/99 | AF303360 (ORF 3) | 18 |
| 33-DK | 32929 | Denmark | 01/02/99 | AF303362 (ORF 3) | 18 |
| 34-DK | 33792 | Denmark | 11/03/99 | AF303363 (ORF 3) | 18 |
| 35-DK | 34229 | Denmark | 26/03/99 | AF303365 (ORF 3) | 18 |
| 1-UK | BE1 | United Kingdom | 18/02/93 | L77913 | 17 |
| 3-UK | H3 | United Kingdom | 21/06/91 | L77915 | 17 |
| 8-UK | NO1 | United Kingdom | 25/03/92 | L77923 | 17 |
| 4-UK | HA1 | United Kingdom | 18/06/92 | L77917 | 17 |
| 7-UK | LE1 | United Kingdom | 11/05/92 | L77921 | 17 |
| 6-UK | L2 | United Kingdom | 05/10/92 | L77919 | 17 |
| 1-BE | 92V58 | Belgium | 02/92 | AY035900 (ORF 5), AY035945 (ORF 7) | 19 |
| 2-BE | AV30 | Belgium | 06/92 | AY035901 (ORF 5), AY035946 (ORF 7) | 19 |
| 1-FR | 5A | France | U40697 (ORF 5) | 69 | |
| 2-FR | 6A | France | U40698 (ORF 5) | 69 | |
| 3-FR | 8D | France | U40699 (ORF 5) | 69 | |
| 4-FR | II/91 | France | 11/91 | Z92526 (ORF 7) | 30 |
| 5-FR | IV/92 | France | 09/92 | Z92528 (ORF 7) | 30 |
| 6-FR | IX/95 | France | 01/95 | Z92529 (ORF 7) | 30 |
| 7-FR | SDRPII7A | France | 29/11/91 | AY035918 (ORF 5), AY035963 (ORF 7) | 19 |
| 8-FR | SDRPIV4A | France | 23/09/92 | AY035919 (ORF 5), AY035964 (ORF 7) | 19 |
| 9-FR | SDRPV4A | France | 17/05/93 | AY035920 (ORF 5), AY035965 (ORF 7) | 19 |
| 10-FR | V/93 | France | 05/93 | Z92706 (ORF 7) | 30 |
| 11-FR | VI/93 | France | 05/93 | Z92707 (ORF 7) | 30 |
| 1-DE | 2.25 | Germany | 1993 | AY035921 (ORF 5), Z92538 (ORF 7) | 19, 30 |
| 2-DE | 2.35 | Germany | 1993 | AY035922 (ORF 5), AY035966 (ORF 7) | 19 |
| 3-DE | 2.46 | Germany | 1993 | AY035923 (ORF 5), AY035967 (ORF 7) | 19 |
| 4-DE | 2.72 | Germany | 1993 | AY035924 (ORF 5), Z92537 (ORF 7) | 19, 30 |
| 5-DE | 2.96 | Germany | 1993 | AY035925 (ORF 5), AY035968 (ORF 7) | 19 |
| 1-IT | 1/93 | Italy | 19/01/93 | AY035926 (ORF 5), AY035969 (ORF 7) | 19 |
| 2-IT | 1142/97 | Italy | 17/02/97 | AY035941 (ORF 5-7) | 19 |
| 3-IT | 1751/93 | Italy | 23/02/93 | AY035927 (ORF 5), AY035970 (ORF 7) | 19 |
| 4-IT | 1828 | Italy | AY035928 (ORF 5), AY035971 (ORF 7) | 19 | |
| 5-IT | 1999/93 | Italy | 23/06/93 | AY035929 (ORF 5), AY035972 (ORF 7) | 19 |
| 6-IT | 2029/97 | Italy | 08/04/97 | AF303358 (ORF 3), AY035930 (ORF 5), AY035973 (ORF 7) | 18, 19 |
| 7-IT | 2156 | Italy | U40696 (ORF 5), AY035974 (ORF 7) | 19, 69 | |
| 8-IT | 2481/97 | Italy | 24/04/97 | AF303359 (ORF 3), AY035931 (ORF 5), AY035975 (ORF 7) | 18, 19 |
| 9-IT | 2567/96 | Italy | 07/05/96 | AY035932 (ORF 5), AY035976 (ORF 7) | 19 |
| 10-IT | 3391/93 | Italy | 24/11/93 | AF303364 (ORF 3), AY035942 (ORF 5-7) | 18, 19 |
| 11-IT | 3943/96 | Italy | 21/06/96 | AF303366 (ORF 3), AY035933 (ORF 5), AY035977 (ORF 7) | 18, 19 |
| 12-IT | 7571/96 | Italy | 14/11/96 | AY035943 (ORF 5-7) | 19 |
| 13-IT | 974/98 | Italy | 10/02/98 | AF303367 (ORF 3), AY035934 (ORF 5), AY035978 (ORF 7) | 18, 19 |
| 1-NL | Boxmeer 10 | The Netherlands | L04493 (ORF 2-7) | 10 | |
| 2-NL | Lelystad | The Netherlands | 1991 | M96262 (ORF 1-7) | 36 |
| 3-NL | NL2.2 | The Netherlands | 15/11/91 | Z92533 (ORF 7) | 30 |
| 4-NL | NL3.1 | The Netherlands | 1992 | U40695 (ORF 5) | 69 |
| 5-NL | NL4.1 | The Netherlands | Z92534 (ORF 7) | 30 | |
| 1-SP | L56/2/91 | Spain | 26/10/91 | AY035935 (ORF 5), AY035979 (ORF 7) | 19 |
| 2-SP | 2228 | Spain | 1992 | U40688 (ORF 5) | 69 |
| 3-SP | 3211 | Spain | 1992 | U40689 (ORF 5) | 69 |
| 4-SP | 4606 | Spain | 1991 | U40690 (ORF 5) | 69 |
| 5-SP | 5710 | Spain | 1991 | U40702 (ORF 5) | 69 |
| 6-SP | 5999 | Spain | 1991 | U40691 (ORF 5) | 69 |
| 7-SP | 65/2/91 | Spain | 25/06/91 | AY035936 (ORF 5), AY035980 (ORF 7) | 19 |
| 8-SP | 705 | Spain | 1993 | U40692 (ORF 5) | 69 |
| 9-SP | L51/2/92 | Spain | AY035937 (ORF 5), Z92531 (ORF 7) | 19, 30 | |
| 10-SP | Olot/91 | Spain | X92942 (ORF 2-7) | 19 | |
| 11-SP | P035 | Spain | 1995 | U40694 (ORF 5) | 69 |
| 1-UK | BE1 | United Kingdom | 18/02/93 | L77914 (ORF 7) | 19 |
| 2-UK | H2-D768 | United Kingdom | 30/10/91 | AY035938 (ORF 5), AY035981 (ORF 7) | 19 |
| 3-UK | H3 | United Kingdom | 21/06/91 | L77916 (ORF 7) | 19 |
| 4-UK | HA1 | United Kingdom | 18/06/92 | L77918 (ORF 7) | 19 |
| 5-UK | L1-D767 | United Kingdom | 14/02/92 | AY035939 (ORF 5), AY035982 (ORF 7) | 19 |
| 6-UK | L2 | United Kingdom | 05/10/92 | L77920 (ORF 7) | 19 |
| 7-UK | LE1 | United Kingdom | 11/05/92 | L77922 (ORF 7) | 19 |
| 8-UK | NO1 | United Kingdom | 25/03/92 | L77924 (ORF 7) | 19 |
| 9-UK | NY3-D769 | United Kingdom | 22/10/92 | AY035940 (ORF 5), Z92536 (ORF 7) | 19, 30 |
| 10-UK | NY4 5 | United Kingdom | 22/06/94 | L77926 (ORF 7) | 19 |
| 11-UK | Ox1 | United Kingdom | L77927 (ORF 7) | 19 | |
| 10-UK | NY4 | United Kingdom | 22/06/94 | L77925 | 17 |
Dates are given in the forms day/month/year, month/year, and year.
Virus isolation.
PRRSV field isolates were obtained from diagnostic sera or tissues on primary porcine alveolar macrophages as previously described (84). Cell harvest methods are similar to a procedure described previously, with some modifications (74). Briefly, swine alveolar macrophages were obtained from conventionally raised 3- to 9-week-old pigs. The lungs were excised and washed three or four times with phosphate-buffered saline, pH 7.2. Cells were centrifuged for 10 min at 800 × g at 5°C. Supernatant fluid was decanted, and cells were washed in phosphate-buffered saline and repelleted two times. Cells were resuspended in RPMI 1640 medium supplemented with 10% irradiated fetal bovine serum and appropriate levels of antibiotics. Macrophages were seeded at a rate of 106 cells/ml to 12- or 24-well plates and allowed to adhere for 7 h. Nonadherent cells were decanted, and wells were refilled with 10% fetal bovine serum and RPMI medium. If swine alveolar macrophages could not be used the same day, they were frozen and stored in liquid nitrogen for later use. Inoculation was at 72 h postseeding. Infection was confirmed by a direct fluorescent antibody test on macrophages removed (scraped) from the well, air dried on glass slides, fixed in acetone, and reacted with monoclonal antibody (MAb) conjugate against the N protein (40). When possible, U.S. and European isolates of PRRSV were propagated on MARC-145 cells as previously described (3, 28, 57).
RT-PCR.
Viral RNA was extracted from infected cell culture with the RNAeasy mini kit (Qiagen) according to the manufacturer's instructions. Reverse transcription (RT) was performed by using approximately 1 μg of total infected cell RNA, Superscript II reverse transcriptase (Gibco-BRL), and oligo(dT) or sequence-specific primers. PCR was performed with high-fidelity Elongase enzyme (Gibco-BRL) and sequence-specific primers according to the manufacturer's instructions with the addition of dimethyl sulfoxide to a final concentration of 10% in the reaction mixture. Primers were based on published sequences of the European prototype PRRSV strain LV (GenBank accession no. M96262) (Table 2). The primer pairs used to amplify and subsequently sequence ORFs 2 to 7 of 13-US, 14-US, and 15-US were L11304F/L12315R, E2F11579F/L12315R, E2F11579F/E3R13263R, L11792F/L12315R, L12245F/L12747R, L12523F/L13022R, L12523F/L13573R, L12523F/E3R13263R, E3F12910F/E3R13263R, L13138F/L13640R, 5EUF/5EUR, L13526F/L14030R, E4F13884F/L14691R, E4F13884F/E4R14347R, L14200F/E7R15023R, L14621F/E7R15023R, and L14800F/3′EndR. For isolates 16-US to 20-US, RT was primed with 5EUR, and the products were then PCR amplified with primer pair 5EUF/5EUR. For generation of the ORF 5 sequences of isolates 2-US to 12-US and 21-US, RT was primed with random hexamers, PCR amplified with the primer pair LV55/LV6, and then submitted for sequencing with primers EUFor1, EUFor3, EURev2, and EURev4. To determine the full-length genomic sequence of EuroPRRSV, primers were first selected based on the known nucleotide sequence of strain LV and then used to obtain RT-PCR fragments of 0.5 to 3 kb by employing the procedure described previously (83) (Table 2). Alternate primers were synthesized based on newly obtained EuroPRRSV sequences (Table 2). To determine the 5′ terminus of EuroPRRSV, we employed Euro21R for the RT and tailed the RT product with terminal deoxynucleotide transferase in the presence of guanosine or adenine in separate reactions, followed by PCR amplification of the products with primer pairs Qc/Euro40R or Qt/Euro40R, by using established conditions (82).
TABLE 2.
Oligonucleotide primers utilized in RT-PCR amplification and nucleotide sequencing of type 1 isolates
| Primera | nt positions for strain:
|
Sequence | |
|---|---|---|---|
| EuroPRRSV | LV | ||
| L11304F/b | 11263-11282 | 11314-11333 | 5′-TTTCCAGTTGGAAGGGCTTA |
| E2F11579F/ | 11538-11557 | 11589-11608 | 5′-ACACTGAGGATTGGGCAATC |
| L11792F/ | 11751-11770 | 11802-11821 | 5′-TGGGGTCACTGTGGAGTAAA |
| L12245F/ | 12204-12223 | 12255-12274 | 5′-GATTCTTGCCGCTTTCTCAG |
| /L12315R | 12255-12274 | 12306-12325 | 5′-TGTAGGCTCACATTGCCAAC |
| L12523F/ | 12482-12501 | 12533-12552 | 5′-TCATTCGAGCTGACCATCAA |
| /L13573R | 12640-12659 | 12691-12710 | 5′-GTTTGAGGTTGTCGTACCCG |
| /L12747R | 12687-12706 | 12738-12757 | 5′-CGCGTAGGAAAAGGACAAAA |
| E3F12910F/ | 12869-12888 | 12920-12939 | 5′-GACGGGGGCAATTGGTTTCA |
| /L13022R | 12962-12981 | 13013-13032 | 5′-CGTCGAGAAACAGGGCTTAC |
| L13138F/ | 13097-13116 | 13148-13167 | 5′-GAAAGTCGTCCCAATGTCGT |
| /E3R13263R | 13205-13222 | 13256-13273 | 5′-CGTAGAAAAGGCACGCAG |
| 5EUF/ | 13322-13341 | 13373-13392 | 5′-CCCAACATACCCAGCAGCAT |
| LV55/ | 13366-13385 | 13417-13436 | 5′-TTGCTGCATTTCCTGACACC |
| EUFor1/ | 13398-13414 | 13449-13465 | 5′-GTGGGCTACAACCATTG |
| L13526F/ | 13485-13504 | 13536-13555 | 5′-TCTTGCTTCTGGTGGCTTTT |
| /L13640R | 13580-13599 | 13631-13650 | 5′-TCAGCTCGCATATCGTCAAG |
| EUFor3/ | 13823-13841 | 13874-13892 | 5′-CCGTGCTGCTAAAAATTGC |
| E4F13884F/ | 13843-13862 | 13894-13913 | 5′-TGGCTTGCCGCTACGCCGTA |
| /EURev4 | 13912-13930 | 13963-13981 | 5′-GGAAGTCTCCAATAGTGGT |
| /L14030R | 13970-13989 | 14021-14040 | 5′-GGACGACATGTTTGATGGTG |
| /5EUR | 14065-14082 | 14116-14133 | 5′-TTTTGTGCGGCGGTAGGA |
| L14200F/ | 14159-14178 | 14210-14229 | 5′-GGGCTGTTGCACATCCTAAT |
| /E4R14347R | 14286-14306 | 14337-14357 | 5′-ATGACTCTGTGAGGCTGTAGA |
| /EURev2 | 14352-14369 | 14403-14420 | 5′-GATACATTCTGGCCCCTG |
| /LV6 | 14510-14529 | 14561-14580 | 5′-CGAGCTGTTAAACGAGGAGT |
| L14621F/ | 14631-14650 | 14682-14701 | 5′-TTGCTGGGTGCAATGATAAA |
| /L14691R | 14631-14650 | 14682-14701 | 5′-TTTATCATTGCACCCAGCAA |
| L14800F/ | 14783-14802 | 14834-14853 | 5′-GATCCAGACGGCTTTCAATC |
| /E7R15023R | 14961-14982 | 15012-15033 | 5′-GTGACTCAGAGGCCACACGCCA |
| /LEndR | 15028-15047 | 15079-15098 | 5′-ATTTTCGGTCACATGGTTCC |
| LVLP1/c | 20-40 | 20-40 | 5′-CCCTACATACACGACACTTCT |
| /LVLP4 | 92-11 | 92-111 | 5′-CCTGTTGGGCTAGAACAGGG |
| LVLP3/ | 92-111 | 92-111 | 5′-CCCTGTTCTAGCCCAACAGG |
| /LVLP2 | 204-222 | 204-222 | 5′-GATCTCCACCCTTTAACCA |
| Euro41F/ | 426-443 | 426-443 | 5′-GAATGTACTCCATCCGGG |
| Euro42F/ | 441-457 | 441-457 | 5′-GGGTGCTGTTGGCTCTC |
| /Euro42R | 562-579 | 562-579 | 5′-GAAGCTCACGGAGGTGTC |
| 5′-592/ | 603-622 | 603-622 | 5′-TGGTAYCCKATCACKGGSCC |
| /LAR1 | 677-698 | 677-698 | 5′-CCCTGGTGCCACCCATGTGTTG |
| /Euro41R | 825-841 | 825-841 | 5′-GGCGTCCACATAATGCG |
| Euro43F/ | 906-923 | 906-923 | 5′-CTCATTCGGAGTTTCCCT |
| /Euro41RA | 928-945 | 928-945 | 5′-CCAGGTTGACAGGGTGAT |
| /LAR2 | 1176-1196 | 1176-1196 | 5′-CGCGGCATTCGTGCTGTAGTC |
| 5′-1223/ | 1235-1258 | 1235-1258 | 5′-CCCSSAGTCTTRSATCAGGCACMT |
| /Euro40R | 1272-1290 | 1272-1290 | 5′-CAAATCCTGGGGTGACATC |
| 5′-1748/ | 1758-1776 | 1758-1776 | 5′-TCTCSTGAATGTGTRKTGG |
| /Euro21R | 1770-1786 | 1770-1786 | 5′-GAACAAACGCCGACCAC |
| 5′-2002/ | 2014-2030 | 2014-2030 | 5′-TTCCGCWAAARWGCGGW |
| Euro2158/ | 2168-2187 | 5′-CCCTGCAGATGTTCCGGCCG | |
| /Euro20R | 2257-2274 | 2257-2274 | 5′-CTTCCTCGAACCCTTCTG |
| Euro2644/ | 2603-2518 | 2654-2669 | 5′-GCTCCGTCATGTTGAG |
| /Euro2714 | 2653-2673 | 2704-2724 | 5′-GCGCATAAGACAGATCCAAAG |
| Euro3080 | 3039-3057 | 3090-3108 | 5′-GTGTATGAACGGTGCCTCC |
| /Euro3207 | 3151-3166 | 3202-3217 | 5′-GCTTGGAACTGCGAGG |
| /Euro3228 | 3169-3186 | 3220-3237 | 5′-GGGACGCAAGAATGCGAC |
| Euro22F/ | 3522-3538 | 3573-3589 | 5′-GTTTTCTCCCACCTCCC |
| Euro70F/ | 3771-3787 | 3822-3838 | 5′-TCTTCTTGTGACCACGA |
| /Euro79R | 3813-3832 | 3864-3883 | 5′-TGGCGCTGCTCAAGAGCCAA |
| Euro71F/ | 3821-3837 | 3872-3888 | 5′-TGAGCAGCGCCAACTTT |
| /Euro80R | 3870-3889 | 3921-3940 | 5′-ATGACACATAAGAGGCCCGA |
| Euro23FA/ | 3903-3923 | 3954-3974 | 5′-CTCGGTGGGTCACGTTATCTC |
| Euro23F/ | 3933-3951 | 3984-4002 | 5′-CTCCTACGTTTATGCATGC |
| /Euro73R | 3999-4014 | 4050-4065 | 5′-CCCAACACTTGTGACA |
| /Euro72R | 4280-4299 | 4331-4350 | 5′-TCAGGCCATCAAATGCCTGA/PICK> |
| LV4234F/ | 4329-4345 | 4380-4396 | 5′-GACCAGCCTACACCTGA |
| /LV4235R | 4329-4345 | 4380-4396 | 5′-TCAGGTGTAGGCTGGTC |
| /Euro74R | 4394-4413 | 4445-4464 | 5′-AATCTGGGTTGACTGGAACT |
| Euro75F/ | 4461-4480 | 4512-4531 | 5′-GGTTACTCGACAGCACAACT |
| /Euro83R | 4493-4512 | 4544-4563 | 5′-TCAACTTGGCAAAGTTGCCC |
| Euro76F/ | 4631-4650 | 4682-4701 | 5′-TACATCACCTCAAGTGTGTG |
| 5′-4688/ | 4647-4661 | 4698-4712 | 5′-TGTGGMCGAGGAACC |
| Euro81F/ | 4753-4772 | 4804-4823 | 5′-TCACCTTGCCATTGCTCTCA |
| /Euro88R | 4753-4772 | 4804-4823 | 5′-TGAGAGCAATGGCAAGGTGA |
| /Euro89R | 4753-4772 | 4804-4823 | 5′-TGAGAACAATGGCAAGGTGA |
| 5′-5022/ | 4981-5000 | 5032-5051 | 5′-TTTGGGTGCAMTYWTTCTTG |
| Euro91F/ | 4982-5001 | 5033-5052 | 5′-TTGGGTGCACTCATTCTTGG |
| Euro90F/ | 5024-5043 | 5075-5094 | 5′-CATCCTCTCATTAGGGACAA |
| /Euro92R | 5473-5492 | 5524-5543 | 5′-CTTGACCACAGGAGCAACGC |
| /Euro93R | 5534-5553 | 5585-5604 | 5′-TGCCGGGTTCGACACCAGTT |
| LAF4/ | 5632-5650 | 5683-5701 | 5′-TTATTGGAATCCACACCGG |
| 5′-5682/ | 5641-5661 | 5692-5712 | 5′-TCCACACYGGTTCAAACAAAC |
| 5′-5690/ | 5649-5670 | 5700-5721 | 5′-GGTTCAAACAAACKYGGYTCWG |
| /Euro84R | 5752-5770 | 5803-5821 | 5′-TCCCCAAGAGGAACGCTCG |
| Euro86F/ | 5802-5821 | 5853-5872 | 5′-GTGACATCCATTCCGAGTGA |
| /Euro85R | 5802-5821 | 5853-5872 | 5′-TCACTCGGAATGGATGTCAC |
| 5′-5922/ | 5883-5904 | 5934-5957 | 5′-TTGTGTGTCTTYTTCYTTCTSTGG |
| Euro87F/ | 5940-5959 | 5991-6010 | 5′-GCCGTGGGCTTCTTTCTACT |
| /3′-6050L | 5995-6016 | 6046-6071 | 5′-CAMARAKAGKGCAAAAGAAAACA |
| /3′-6050 | 6000-6017 | 6051-7068 | 5′-CAMARAKAGKGCAAAACA |
| /Euro77R | 6020-6039 | 6071-6090 | 5′-ACCAGGTGGTGACCCATGCA |
| Euro11F/ | 6061-6078 | 6112-6129 | 5′-GACTCCTCACGGCATCTC |
| /Euro78R | 6088-6106 | 6139-6157 | 5′-AACGCCAGAGAGAACTTGT |
| LAF3/ | 6310-6328 | 6361-6379 | 5′-ACATGCTGGTTGGTGAGGG |
| /Euro23R | 6314-6331 | 6365-6382 | 5′-CTCCCATCACCAACCAGC |
| /Euro22R | 6391-6407 | 6442-6458 | 5′-CATGCCACAGGACTGTG |
| LAF2/ | 6493-6515 | 6544-6566 | 5′-GCTTTGTATCTGCTTCAAACATG |
| /Euro22RA | 6565-6582 | 6616-6633 | 5′-GAGAGGCCAACTCTTGGC |
| Euro18FA/ | 6765-6782 | 6816-6833 | 5′-CTTGGCGGCTCCAAATTC |
| 5′-7016/ | 6975-6994 | 7026-7045 | 5′-TGGSAYAAGKCTACYGGTGA |
| /Euro19RA | 7073-7092 | 7124-7143 | 5′-CGGTTTGCACACCTTCATAG |
| Euro19F/ | 7285-7302 | 7336-7353 | 5′-CTGCCGAGGTGGAAAAG |
| /Euro19R | 7575-7594 | 7626-7645 | 5′-CAGAAGAACGTCAACAAGGG |
| /Euro18RA | 7592-7609 | 7643-7660 | 5′-GTCAAGTCCGGGTTTCAG |
| LAF1/ | 7762-7781 | 7813-7832 | 5′-CCTCCAACTCCCTTACAAGC |
| 5′-7865/ | 7824-7844 | 7875-7895 | 5′-TKWTCAATACCRGSTTTGGAG |
| 5′-7905 | 7854-7871 | 7905-7922 | 5′-YGTCAYTGGGGGTTTTGT |
| Euro19FA/ | 7916-7932 | 7967-7983 | 5′-GCCCCTGTGTCTGATGG |
| /Euro18R | 8002-8021 | 8053-8072 | 5′-GTGAATCGAGGTACTCCATG |
| Euro10F/ | 8051-8068 | 8102-8119 | 5′-CATGGCACTTCCAAGGCT |
| 5′-8242/ | 8201-8219 | 8252-8270 | 5′-CCCGCYAAGACCTCKATGG |
| 5′-8342/ | 8301-8322 | 8352-8373 | 5′-AGGAGMAYTGGCAAACYGTGAC |
| /Euro17R | 8494-8509 | 8545-8560 | 5′-GACGGTGCAATGCAGC |
| Euro12F/ | 8517-8534 | 8568-8585 | 5′-GGTGTCTTGAGGCCGACT |
| /Euro16R | 8909-8927 | 8960-8978 | 5′-CCCACCAGTGGTAATTAGG |
| Euro13F/ | 8929-8947 | 8980-8998 | 5′-CGAGCACCTTGACCTAATG |
| Euro13aF/ | 9206-9225 | 9257-9276 | 5′-CAGGATGGTTATAGCTTCCC |
| /Euro15R | 9307-9326 | 9358-9377 | 5′-CATAGTCAGCTTTGGCATCG |
| Euro14F/ | 9402-9420 | 9453-9471 | 5′-GTCATCATGCCGGTTCAAG |
| /Euro14R | 9729-9745 | 9780-9796 | 5′-GGTCTTTCCGGAACCTG |
| Euro15F/ | 9847-9863 | 9898-9914 | 5′-CATCCCAGGAGCCTCAG |
| /Euro13R | 10212-10230 | 10263-10281 | 5′-GTCAGCACCTGACCAAAGG |
| Euro16F/ | 10336-10352 | 10387-10403 | 5′-CAAATCCCGAGCACTTG |
| /Euro12R | 10663-10682 | 10714-10733 | 5′-GAAGTGCAAATGCTGGACTG |
| Euro17F/ | 10757-10775 | 10808-10826 | 5′-GTTAGTATGCGCCCAATTG |
| /Euro11R | 11138-11157 | 11189-11208 | 5′-GGGAGATACACATCGGTGAG |
| Euro18F/ | 11177-11193 | 11228-11244 | 5′-CAACCTGAGACGGCATC |
| LV1/ | 11333-11353 | 11384-11404 | 5′-GCCGTTGTATACATTGATCCG |
| /Euro10R | 11568-11587 | 11619-11638 | 5′-GTCCTTGCCGTCATTCATAC |
| LV11739/ | 11698-11716 | 11749-11767 | 5′-AGGTAAACCCCGGCTGCCG |
| Euro1F/ | 11716-11733 | 11767-11784 | 5′-GCCTGGGCAGGTGCCGTG |
| Euro1F-a/ | 11728-11747 | 11779-11798 | 5′-GCCGTGAATTTGGAGTAATG |
| /LV2 | 12047-12066 | 12098-12117 | 5′-CCATCTCATCAATCAAGTGG |
| Euro2F/ | 12202-12219 | 12253-12270 | 5′-CGGATTCTTGTCGCTTTC |
| /Euro4R | 12619-12637 | 12670-12688 | 5′-CGGGATGGACATTAACAAC |
| /Euro3R | 13036-13055 | 13087-13106 | 5′-CGGAAACAATTGATGTCCTG |
| LV55/ | 13366-13385 | 13417-13436 | 5′-TTGCTGCATTTCCTGACACC |
| /Euro2R | 13422-13440 | 13473-13491 | 5′-CGCCAAGAGAATGGCGAAC |
| /Euro2aR | 13453-13472 | 13504-13523 | 5′-GAAACGCCCCAATTTGTGAG |
| /LV11850 | 13808-13829 | 13859-13880 | 5′-AGCACGGATGACAAAACATACG |
| /3′-6800 | 13971-1391 | 14022-14042 | 5′-CAGGTCTACAAGTTGGGTGGT |
| LV6/ | 14071-14090 | 14122-14141 | 5′-CGCCGCACAAAAGCTCGTGC |
| /LV6 | 14510-14529 | 14561-14580 | 5′-CGAGCTGTTAAACGAGGAGT |
| /Euro1R | 14522-14540 | 14573-14591 | 5′-CGAGGTTAACCACTCCTCG |
| LV7/ | 14534-14553 | 14585-14604 | 5′-AACCTCGTCAAGTATGGCCG |
| /LV56 | 14641-14661 | 14692-14712 | 5′-CAATGATAAAGTCCCAGCGCA |
| Euro4F/ | 14850-14867 | 14901-14918 | 5′-CAGGTTGAGTTCATGCTG |
| /LV7 | 14914-14933 | 14965-14984 | 5′-CCAGTCAGGGTGCAAGTTAA |
| Qc | 5′-CCAGTGAGCAGAGTGACGAGGACTCGAG CTCAAGCCCCCCCCCCCCCCCCCC | ||
| Qt | 5′-CCAGTGAGCAGAGTGACGAGGACTCGAG CTCAAGTTTTTTTTTTTTTTTTTT | ||
A slash (/) after an oligonucleotide name indicates a forward primer and a slash before an oligonucleotide name indicates a reverse primer.
The first group of oligonucleotides (L11304F/ to /LEndR) was used for the determination of ORFs 2 to 7 of 13-US, 14-US, and 15-US and of ORF5 of the remaining isolates.
The second group of oligonucleotides (LVLP1/ to Qt) was used for the determination of the EuroPRRSV full-length sequence.
DNA sequencing.
The PCR amplicons were purified with the Wizard Prep kit (Promega) according to the manufacturer's instructions, and then the nucleotide sequence determination was completed in both directions with the PCR primers used to generate the amplicons. Cycle sequencing was performed with fluorescent Big Dye chain terminators (Applied Biosystems), and sequencing products were electrophoresed on an ABI Prism 310 Genetic Analyzer (Applied Biosystems). In the case of EuroPRRSV, isolates 2-US to 12-US and 21-US, purified RT-PCR-generated DNA fragments, and relevant primers based on type 1 strain LV were submitted to the Advanced Genetic Analysis Center at the University of Minnesota for sequence determination with an ABI 377 automated DNA fragment analyzer. A quality sequence representing at least threefold genome coverage was obtained.
Sequence data analysis.
Sequence data were assembled and analyzed by using the GeneTool sequence analysis program (BioTools, Inc., Edmonton, Alberta, Canada) or Lasergene (DNASTAR, Inc., Madison, Wis.) and Wisconsin Package, version 10.3 (Accelrys, San Diego, Calif.) sequence analysis software suites. Multiple-sequence alignments were generated with CLUSTALX (71). Phylogenetic trees were inferred from these alignments by using TREE-PUZZLE, version 5.0 (68), implementing the HKY+γ substitution model. Transition/transversion ratios were estimated by TREE-PUZZLE, version 5.0, from the data. Quartet-puzzling (QP) trees were constructed with the described settings and 10,000 puzzling steps to obtain support percentage values (QP reliability) for each internal branch. Representations of the QP trees were constructed with TreeView. Phylogeny analysis was completed by using the sequences listed in Table 1. Full-length PRRSV strains U87392, JA142 (accession no. AY424271), and accession numbers AF325691, AY032626, AF331831, AF303356, AF046869, AF184212, and AF094476 were used for nonstructural region comparison.
MAbs.
MAbs were produced by immunizing mice with whole, intact virus particles or selected ORFs subcloned into a pCDNA3.1/Zeo(+) vector, as previously described (41, 80). The protein specificity of the MAbs was determined by radioimmunoprecipitation and/or immunoblotting. Immunoglobulin isotypes were determined by using a commercial immunodiffusion assay. For this study, we selected 60 MAbs representing multiple epitopes of products of ORFs 2 through 7, including 2 to the ORF 2 product, 3 each to the ORF 3, 5, and 6 products, 6 for the ORF 4 product, and 43 to the ORF 7 products. MAbs to ORFs 3, 5, and 6 and some of ORF 7 were generously provided by Kenneth Platt (Iowa State University).
Indirect fluorescent antibody assay.
Isolates were tested in the MAb panel at the second or third passage in cell culture to minimize the opportunity for antigenic changes. An indirect fluorescent antibody assay was used to determine the reactivity of each MAb with each PRRSV isolate (80). Confluent monolayers of MARC-145 cells in 96-well plates were infected with each PRRSV isolate. Fixed cells for each isolate were stained with each of the 60 different MAbs at a 1:100 dilution followed by fluorescein isothiocyanate-conjugated goat anti-mouse immunoglobulin A (IgA), IgM, and IgG. Cells were then observed for the presence of virus-specific staining and rated as reactive (+), nonreactive (−), or weakly reactive (+/−).
RESULTS
Genomic sequence analysis.
Discovered in 1999 as a routine clinical submission for the Minnesota Veterinary Diagnostic Laboratory, EuroPRRSV represents the initial virus of a group of newly emerging European-like (type 1) PRRSV isolates detected in the United States (U.S. Animal Health Association, 2000 Report of the Committee on Transmissible Diseases of Swine [http://www.usaha.org/reports/reports00/r00swine.html]) and the second European strain for which the full-length genome nucleotide sequence has been determined. The completed sequence allowed the first full-length genome comparison among PRRSV type 1 viruses (Table 1). Excluding the poly(A) tail, the genomic sequence of EuroPRRSV was 15,047 nucleotides (nt) in length, 51 nt shorter than the genome of the prototypic European strain LV. Full-length sequence analysis determined that the genome of this U.S. index isolate was 95.3% identical (704-nt differences of 15,047 nt) to strain LV at the nucleotide level, but identity varied for individual ORFs (ORF 1a, 94.2% identical with a 51-bp deletion; ORF 1b, 96.2% identidcal; ORFs 2 to 7, 95.8% identical). This high level of identity was not expected for two type 1 PRRSV field isolates of a virus known to undergo a high rate of recombination and mutation and known to evolve rapidly (18, 19, 25, 35, 81, 83; W. L. Mengeling, D. F. Clouser, A. C. Vorwald, and K. M. Lager, Proc. Conf. Res. Work. Anim. Dis., abstr. 134, 2000). Considering the immense diversity of PRRSV isolates found worldwide, the 10-year interval between identification of isolates LV and EuroPRRSV, and the fact that EuroPRRSV was isolated in North America, we were anticipating much greater dissimilarity between the two strains (18, 19, 21, 25). As a likely circumstance, the Minnesota Veterinary Diagnostic Laboratory sequenced 242 type 2 (NA) isolates in 1999, yet less than 16% displayed 95% or greater similarity to the type 2 prototype VR-2332, which was isolated at approximately the same time as strain LV. However, the lack of overall heterogeneity between strains EuroPRRSV and LV did allow comparison of two similar sequences for the discrimination of variable genomic regions from those regions that had remained highly conserved over this time frame.
An interesting global pattern of nucleotide and amino acid differences was seen when the two strains were analyzed in detail, with the major differences clustering near the 5′ end and in regions coding for three glycoproteins (Table 3). The predicted proteins for much of ORFs 1a and 1b as well as ORFs 2a, 2b, 6, and 7 displayed greater than 97% identity with LV, and evaluation of the amino acid differences between strains EuroPRRSV and LV in these regions led to the conclusion that specific changes in NSP1α, NSP3 to 12, ORFs 2a, 2b, 6, and 7, and the 3′ UTR were not noteworthy. No deletions or insertions were seen in these genomic regions, and the functional significance of many of these proteins is not known (data not shown). However, the 5′ UTR, sequences near the 5′ end of ORF 1a and ORFs 3, 4, and 5 displayed less than 95% identity with LV, with ORF 5 displaying the lowest degree of conservation at 90.2% identity. The 5′ UTR, NSP1β, and NSP2, as well as three variant glycoproteins (ORFs 3 to 5), were thus studied in more detail. A graphical representation of all amino acid differences observed between EuroPRRSV and LV is shown in Fig. 1B (ORF 1ab) and C (ORFs 2 to 7).
TABLE 3.
EuroPRRSV representative nucleotide and amino acid lengths and detailed comparison to strain LV
| Nucleotide or amino acid | Lengthb | No. of changesb | % Identity |
|---|---|---|---|
| Nucleotides | |||
| 5′ UTR | 221 | 15 | 93.2 |
| ORF 1aa | 7,137 | 386 | 94.6 |
| ORF 1b | 4,374 | 165 | 96.2 |
| ORFs 2-7 | 3,203 | 136 | 95.8 |
| 3′ UTR | 124 | 2 | 98.2 |
| Complete | 15,047 | 704 | 95.3 |
| Amino acids | |||
| NSP1α | 166 | 8 | 95.1 |
| NSP1β | 219 | 18 | 91.3 |
| NSP2a | 861 | 70 | 91.6 |
| NSP3 | 447 | 17 | 96.2 |
| NSP4 | 203 | 3 | 98.0 |
| NSP5 | 170 | 6 | 96.5 |
| NSP6 | 16 | 0 | 100 |
| NSP7 | 270 | 7 | 97.4 |
| NSP8 | 38 | 0 | 100 |
| NSP9 | 646 | 10 | 98.5 |
| NSP10 | 442 | 5 | 98.9 |
| NSP11 | 224 | 4 | 98.2 |
| NSP12 | 152 | 3 | 98.0 |
| ORF 2a/GP2 | 249 | 6 | 97.6 |
| ORF 2b/E | 70 | 1 | 98.6 |
| ORF 3/GP3 | 265 | 17 | 93.6 |
| ORF 4/GP4 | 183 | 10 | 94.5 |
| ORF 5/GP5 | 201 | 20 | 90.1 |
| ORF 6/M | 173 | 3 | 96.5 |
| ORF 7/N | 128 | 1 | 99.2 |
Gap in EuroPRRSV when compared to strain LV.
Units for length and number of changes are base pairs for nucleotides and amino acids for amino acids.
5′-Leader sequence variation.
In PRRSV-infected cells, the major subgenomic transcripts (sgmRNAs 2 to 7), coding for largely structural proteins, are produced by joining the 5′ 221-base leader sequence to specific sites downstream by discontinuous transcription (Fig. 1A) (reference 50 and references therein). The 3′ terminus of the leader 5′ UTR ends in a motif (UUAACC) known as the transcription regulatory sequence (TRS), which has been predicted to be situated in a loop of a stem-loop structure (72). In addition, the leader sequence is also proposed to bind to viral and cellular proteins involved in PRRSV transcription, translation, and replication (24).
The 5′ UTR of EuroPRRSV displayed differences in 15 of 221 nt (93.2% similarity) when compared to strain LV (Table 3; Fig. 2). The only other type 1 leader sequence currently available is a field isolate identified in Denmark in 1992, which showed 90.1% identity to strain LV (111/92/DK, GenBank accession no. AF094476) (45). In 1999, Oleksiewicz and coinvestigators recognized common domains in this important region of the virus by using available PRRSV type 1 (LV and 111/92/DK) and type 2 (VR-2332) leader sequences (45). They identified a conserved 5′-leader start motif of 12 nt and three invariant nucleotide stretches just upstream of the leader TRS (Fig. 2). Comparison of the EuroPRRSV leader sequence with a larger panel of isolates (two type 1 viruses [Fig. 2] and type 2 virus [data not shown]) provided evidence that the three invariant motifs were not strictly conserved, as the EuroPRRSV sequence contained nucleotide changes in all three domains. However, the 12-nt 5′-leader start motif and the leader TRS were accurately maintained. Also, as previously noted (45), one of the motifs contained a conserved CACCC site, with another conserved CACCC site occurring approximately 100 nt upstream in the leader sequences (Fig. 2). CACCC stretches have been shown to be important in binding to host cell transcription factors important in growth control, apoptosis, and angiogenesis (4, 44) and are also utilized for double-stranded DNA virus and retroviral transcription (38, 44, 56). Strict maintenance of these CACCC domains was seen in all but a few known PRRSV leader sequences (Fig. 2 and data not shown), but their significance for PRRSV transcription remains to be determined.
FIG. 2.
(A) Alignment of the 5′ UTR of the EuroPRRSV isolate with strain LV and a pathogenic European PRRSV isolate from 1992, 111/92/DK (45). Of the domains previously thought to be absolutely conserved (grey boxes), only the 12-nt start motif and the leader TRS (black box) were shown to remain inerrant (45). As identified by others (45), the CACCC motifs (shown in bold) implicated in regulating growth of cells but not shown to be similarly active in nonretroviral RNA viruses are also maintained (4, 44) (see text).
Amino acid alterations in EuroPRRSV NSP1β and NSP2.
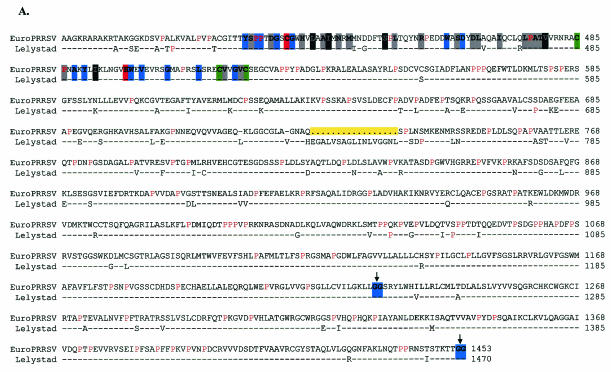
Genomic RNA (mRNA 1) serves as the template for production of the replicase protein, which is catalytically processed by virus-encoded proteases as shown most clearly for EAV (Fig. 1A and B). All previously identified putative cleavage sites were maintained in EuroPRRSV, based on similarity to EAV, LV, and VR-2332 (1, 13; reviewed in reference 85). As briefly discussed previously, the accumulation of differences between EuroPRRSV and LV was most concentrated in NSP1β (amino acids [aa] 167 to 385) and NSP2 (aa 386 to 1247), with a notable 51-bp deletion within NSP2 (Fig. 1B).
NSP1β contains a papain-like cysteine protease (13). Amino acid alignment of this EuroPRRSV region with strain LV (Fig. 3) revealed that sites previously suggested to be maintained in most arteriviruses were not altered, including the putative catalytic cysteine and histidine residues and the invariant residues, but both conserved and nonconserved amino acid mutations are scattered throughout this protease domain. The data suggest that mutations were allowed in noncritical amino acids while amino acids crucial to the function of this protein segment, which is autocatalytically cleaved from the ORF 1 polyprotein during translation (12, 13, 39, 62, 63, 64, 85), were maintained.
FIG. 3.
Nsp1β amino acid alignment between strains EuroPRRSV and LV. Boldface type and colored backgrounds represent the following: red, (putative) catalytic residues; blue, invariant nidovirus residues; black and grey, residues conserved in 100% and 50 to 75%, respectively, of the arteriviral sequences, as shown previously (85). Conservation groups are I, V, L, and M; F and Y; K and R; D and N; E and Q; and S and T. The downward arrow indicates the putative position of the cleavage site.
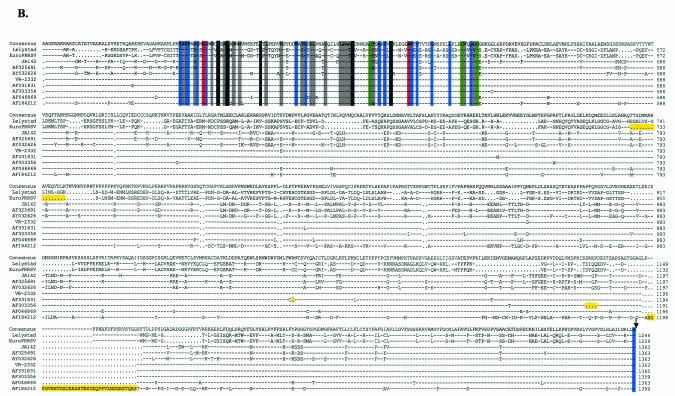
A chymotrypsin-like (3C-like) cysteine protease domain resides at the N-terminal end of NSP2 (16). NSP2 has been shown to be variable in length among sequenced isolates of arteriviruses (1, 36, 39, 61). PRRSV type 1 strain LV NSP2 is 861 aa in length while type 2 strains have an additional 100 aa or more, and lactate dehydrogenase-elevating virus and EAV encode NSP2 proteins which are 733 and 401 aa in length, respectively (1, 20, 36, 82, 85). Snijder et al. (66) found that the NSP2 protein of EAV was involved in proteolysis of replicase precursors and membrane association of the virus replication complex, but no confirming work has been reported for other arteriviruses. Similarity is observed only in the amino- and carboxy-terminal domains among different arteriviruses (14). An alignment of this region in EuroPRRSV with the corresponding region in LV, the only other type 1 sequence available, is shown in Fig. 4A. There are 96 and 101 proline residues in strains EuroPRRSV and LV representing 11.1 and 11.5%, respectively, of the NSP2 amino acids, and each represents over 57% of the proline residues in the ORF 1a protein. Ten and 11 of these proline-rich domains, respectively, are of the pattern PXXP, long recognized as the signature binding motif of Src homology 3 (SH3) domains (7, 8, 51). SH3 domains participate in a number of signal transduction mechanisms and cell-cell communication by binding to such polyproline-rich peptides (7, 8, 51) and have been shown to be active in a number of viruses (2, 29, 43, 70). However, an interaction between SH3 domains and the NSP2 protein of arteriviruses remains to be elucidated. The 17-aa deletion in EuroPRRSV lies within this NSP2 proline-rich region. Two potential protease cleavage sites have been identified for NSP2, one or both of which may be utilized (Fig. 4A) (1, 85).
FIG. 4.
(A) NSP2 (CP2) amino acid alignment between strains EuroPRRSV and LV. Downward arrows indicate the putative positions of a cleavage site at ORF 1 aa 1246 in EuroPRRSV (1) or at aa 1452 in EuroPRRSV (85). Boldface type and colored backgrounds represent the following: red, (putative) catalytic residues; blue, invariant nidovirus residues; black and grey, residues conserved in 100% and <75%, respectively, of the arteriviral sequences. Conservation groups are I, V, L, and M; F and Y; K and R; D and N; E and Q; and S and T. The green backgrounds at several positions of the CP2 alignment are completely conserved between arteriviruses and are thought to bind Zn2+ (22, 64). Proline residues, comprising approximately 11% of the total protein, are shown in red. The 17-aa deletion in EuroPRRSV is shaded yellow. (B) Nsp2 amino acid alignment between type 1 (LV and EuroPRRSV) and type 2 (JA142, AF325691, AY032626, VR-2332, AF331831, AF303356, AF046869, and AF184212) PRRSV strains show conservation of identified putative protease residues but considerable divergence outside the domain. The downward arrow indicates the putative position of the cleavage site at ORF 1 aa 1246 in EuroPRRSV (1). Insertions or deletions present in individual viruses are shaded yellow. Key protease residues are shaded as in Fig. 4A.
Comparison of the NSP2 predicted protein of the two PRRSV subtypes demonstrated that most residues are strictly maintained within a subtype (84 to 99% similarity) but differ greatly between subtypes (<45% similarity) (data not shown). Alignment of the NSP2 proteins of type 1 and type 2 PRRSV strains exhibiting moderate to extensive sequence divergence also revealed that the predicted proteolytic residues remain well conserved, but the two subtypes show little amino acid conservation outside this predicted domain (Fig. 4B) (85). Further, the insertions and deletions occurred in both subtypes but did not cluster in the same region. These data suggest that the NSP2 ORF contains a protease motif necessary for the replication cycle of PRRSV but is highly susceptible to mutation in other regions of the NSP2 protein without deleterious effects on the virus.
Genetic analysis of structural ORFs.
The nucleotide sequence of the structural ORFs (ORFs 2 to 7) for three additional type 1 isolates were generated. Each isolate revealed comparable overall nucleotide identity with strain LV (13-US, 95.8% identity; 14-US, 95.6% identity; 15-US, 95.2% identity; EuroPRRSV [1-US], 95.8% identity). Amino acid sequence comparison of 13-US, 14-US, 15-US, EuroPRRSV, and VR-2332 with LV indicated that the type 1 viruses isolated near the beginning of U.S. emergence shared 89.6 to 99.2% identity with prototype strain LV. In comparison, the North American prototype strain VR-2332 shared only 59.5 to 78.6% identity with strain LV (Table 4). From these initial data, we found that GP3, GP4, and GP5 were the least conserved of the structural proteins and indicated that these ORFs were of interest to study more closely. Accordingly, multiple alignments of these proteins were produced (Fig. 5).
TABLE 4.
Percent amino acid identity of selected proteins of PRRSV isolates to the European strain LV
| Protein | % Amino acid identity to strain LV for isolate:
|
||||
|---|---|---|---|---|---|
| 1-US | 14-US | 13-US | 15-US | VR-2332 | |
| ORF 2a/GP2 | 97.6 | 97.6 | 97.2 | 96.0 | 63.1 |
| ORF 2b/E | 98.6 | 95.7 | 98.6 | 98.6 | 75.7 |
| ORF 3/GP3 | 93.6 | 92.3a | 94.0 | 92.8 | 59.5 |
| ORF 4/GP4 | 94.5 | 90.7 | 90.2 | 89.6 | 68.5 |
| ORF 5/GP5 | 90.1 | 93.5 | 93.5 | 91.5 | 56.5 |
| ORF 6/M | 96.5 | 98.3 | 96.5 | 96.5 | 78.6 |
| ORF 7/N | 99.2 | 96.1 | 99.2 | 99.2 | 65.9 |
18-aa C-terminal truncation.
FIG. 5.
The alignment of structural proteins was completed by using GeneTool sequence analysis software. (A) GP3; (B) GP4; (C) GP5. Amino acids representing hypervariable domains (solid boxes) and putative signal sequences (dotted line boxes) are indicated. Potential N glycosylation sites are shown in boldface type.
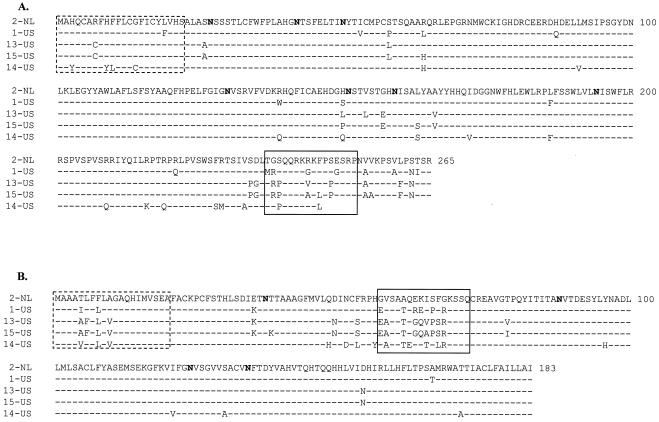
As graphically represented in Fig. 1C, a concentration of amino acid changes was seen between EuroPRRSV and LV in ORFs 3 and 4, which encode the minor structural proteins GP3 and GP4, respectively. The locations of these differences concur with the mutational hotspots identified previously (Fig. 5A and B) (46). The hypervariable region encompasses aa 237 to 252 in ORF 3, where 4 of 17 aa changes between EuroPRRSV (1-US) and LV (2-NL) were observed, and aa 57 to 72 in ORF 4, where 6 of 10 aa differences between these two strains were observed (Fig. 5A and B). Nonconserved amino acid differences between the type 1 isolates of this study were also observed in these hypervariable regions, although differences were seen elsewhere in these proteins as well (Fig. 5A and B). The 14-US ORF 3 is truncated by a premature stop codon 18 aa from the C terminus. Similar truncations have been previously reported among European isolates and are consistent with previous observations that indicated that the carboxy-terminal portion of type 1 GP3 plays a nonessential role in the viral life cycle (18, 46). The predicted glycosylation sites remained unaffected by the differences (Fig. 5A and B).
ORF 5 sequences for 15 additional European-like U.S. isolates were determined. A significant number of differences were observed in ORF 5, which encodes the major envelope glycoprotein GP5 (Fig. 1 and 5C) (37). This protein has also been shown to contain the primary neutralization site of the virus (48, 53, 76), has been reported to be the most heterogeneous PRRSV structural protein, and is disulfide linked at an invariant cysteine (located at LV aa 50) to an invariant cysteine of the M protein (aa 8) (11, 55). The hypervariable region observed among type 2 PRRSV field isolates includes the signal peptide sequence and the distal portion of the ectodomain, and the variation observed in this region tends to affect the number of potential N glycosylation sites (Fig. 5C) (52, 60). Similarly, more than half of the differences observed between strains EuroPRRSV (1-US) and LV (2-NL) within the ORF 5 region are found in this hypervariable region, with 5 of 20 differences observed in the signal sequence and 6 differences found in the ectodomain (Fig. 5C). Figure 5C illustrates that nonconserved amino acid differences among the U.S. type 1 PRRSV isolates are also seen in this hypervariable region. In addition, the U.S. type 1 isolates acquired one or two new potential N glycosylation sites at amino acid positions 37 and 38 while aa 46 to 48 and 53 to 55 were unaffected, with the exception of isolate 2-US, which lost a potential glycosylation site as a result of an N-to-D mutation at aa 46. All U.S. type 1 isolates differed from LV at amino acid positions 37, 60, 63, 111, and 153. At amino acid position 111, all U.S. strains contained a serine (S) instead of the cysteine (C) that is present in the LV ORF 5 sequence. At position 153, all U.S. strains contained a glycine (G) instead of an arginine (R), with the exception of 5-US and 14-US, which were shown somewhat diverged from the other U.S. strains (see below).
Phylogenetic analysis.
For evolutionary studies, the nucleotide sequences corresponding to ORF 3, 5, or 7 were aligned to previously published database sequences (10, 17, 18, 19, 30, 36, 46, 69) by using CLUSTAL X (gap opening penalty, 15; gap extension penalty, 6.66) (71). Following alignment, the phylogeny of the isolates was estimated by using the TREE-PUZZLE program and yielded the phylogenetic trees shown in Fig. 6.
FIG. 6.
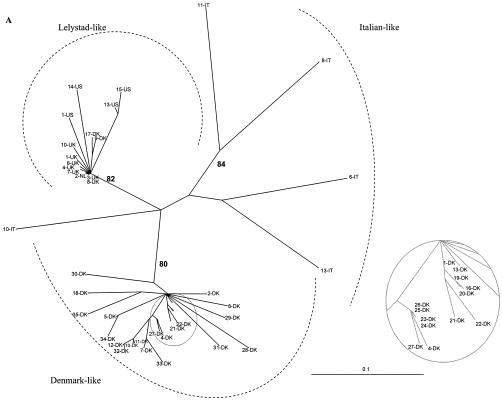
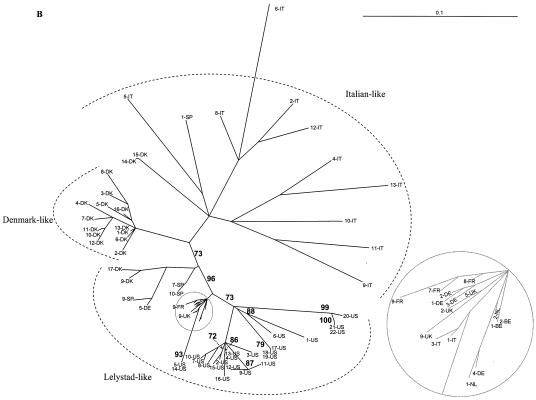
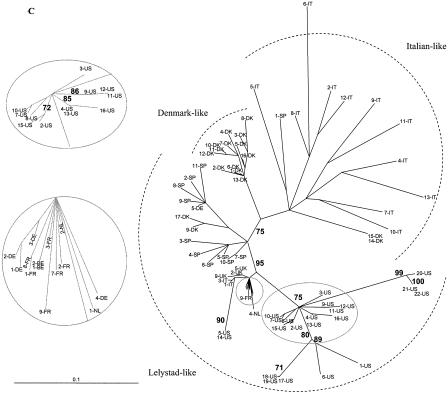
Sequence alignments were generated by using CLUSTAL X. Phylogenies of the aligned isolates were estimated by using the TREE-PUZZLE program (10,000 puzzling steps). The scale bars indicate a maximum-likelihood branch length of 0.1 inferred substitutions per site. (A) Phylogenetic tree based on the ORF 3 nucleotide sequences. Newly generated ORF 3 nucleotide sequences of 13-US, 14-US, 15-US, and EuroPRRSV were aligned with previously published ORF 3 sequences. The 14-US ORF 3 sequence is truncated by a premature stop codon 18 aa from the C terminus. (B and C) Phylogenetic tree based on ORF 5 nucleotide sequences. The newly generated ORF 5 sequences of U.S. type 1 isolates 1 to 21 were aligned with previously published type 1 ORF 5 sequences. (B) Evolutionary analyses were completed with only full-length ORF 5 sequences. (C) Phylogenetic analysis was undertaken with both complete and incomplete ORF 5 sequences. Sequences located in the grey circles were magnified in the companion insets to clearly discern some European-like isolates.
Forsberg et al. (18) and Oleksiewicz et al. (46) established a panel of well-characterized and highly divergent ORF 3 sequences as a reference point for molecular epidemiologic studies. In another study, ORF 5 and 7 genes from 46 European-type PRRSV isolates were also sequenced and combined with existing database sequences to determine ORF 5- and 7-based genealogies (19). In this study, we expanded the data sets presented in those papers to include sequences from several PRRSV type 1 sequences generated from U.S. swine herds (Table 1). The genealogy of ORF 7 was star-like, as observed by Forsberg et al. (19), likely due to little variation or signal in the data because of the highly conserved sequence of ORF 7 (data not shown). When ORFs 3 and 5 were examined, all U.S. type 1 isolates sequenced in our laboratories consistently formed a clade with the European-like isolates from The Netherlands and the United Kingdom (Fig. 6). For the ORF 3-based phylogenies, the QP reliability value for this clade was 82%. For ORF 5-based genealogies (the analysis which contained the most U.S. type 1 isolate sequence information), this value increased to 95 to 96%. This finding suggests there was a limited introduction of selected PRRSV isolates from Europe; it does not support numerous introductions of several diverse type 1 isolates into the United States. Mateu et al. (33) recently reported phylogenetic analysis of ORF 5 of type 1 PRRSV strains in Spain. Phylogenetic analysis with these additional ORF 5 sequences did not provide any new insights into the origin of the U.S. type 1 isolates (data not shown). Within the European-like isolates, the new U.S. type 1 isolates displayed some marked diversity. The relatedness of 13-US and 15-US and the diversity of 1-US and 14-US were consistent for all three ORFs examined, regardless of the inclusion of incomplete ORF 5 sequences into the phylogenetic analysis (Fig. 6).
Antigenic profile of selected isolates.
Three type 1 PRRSV field isolates (13-US, 14-US, and 15-US) were obtained from diagnostic sera or tissues on primary porcine alveolar macrophages and then propagated on MARC-145 cells to obtain suitable concentrations for antigenic profile analysis. These isolates were screened with a panel of 60 MAbs directed against the products of the structural proteins: 2 MAbs prepared to GP2; 3 MAbs specific for each of GP3, GP5, and M; 5 MAbs to GP4; and 43 MAbs specific to N protein (Table 5). The MAb reactivity pattern was highly consistent with that of strain LV and strikingly different from that of our North American reference strain, SD-23983. Several of the MAbs directed against the nucleocapsid protein have been previously characterized, and their reactivities with these new type 1 PRRSV isolates, as well as the original isolate MAb characterization, are shown in Table 6 (41, 78). Specific epitopes of the remaining MAbs have not yet been determined.
TABLE 5.
Reactivities of 13-US, 14-US, and 15-US PRRSV with MAbs directed against specific PRRSV structural proteins
| ORF/protein | MAb | Reactivity with isolatea:
|
||||
|---|---|---|---|---|---|---|
| LV | 13-US | 14-US | 15-US | SD-23983 | ||
| 2/GP2 | C5b | − | − | − | − | +/− |
| C5c | − | − | − | − | +/− | |
| 3/GP3 | P9-1 | − | − | + | − | − |
| P9-2 | − | − | − | − | − | |
| P9-7 | − | − | − | − | + | |
| 4/GP4 | CI82 | + | + | − | + | + |
| CI83 | + | + | − | − | + | |
| NI26 | + | + | − | + | + | |
| NI37 | + | + | − | + | + | |
| NI69 | + | + | − | + | + | |
| 5/GP5 | P6-28 | − | − | − | − | + |
| P6-246 | − | − | − | − | + | |
| P15-1 | − | − | − | − | + | |
| 6/M | P14-1 | − | − | + | − | + |
| P14-2 | − | − | + | − | + | |
| P14-3 | − | − | − | − | +/− | |
| 7/N | A23 | − | − | − | − | + |
| B5 | + | + | + | + | + | |
| C10 | + | + | + | + | + | |
| D12 | − | − | − | − | + | |
| D14 | − | − | − | − | + | |
| E5 | − | − | − | − | + | |
| G1 | − | − | − | − | + | |
| G2 | − | − | − | − | + | |
| G3 | + | + | − | +/− | + | |
| G5 | − | − | + | − | + | |
| G6 | − | − | − | − | + | |
| G7 | + | + | − | − | + | |
| G9 | − | − | − | − | + | |
| H1 | − | − | − | − | + | |
| I1 | +/− | +/− | − | − | + | |
| I2 | − | − | − | − | + | |
| I5 | − | − | − | − | + | |
| J1 | − | − | − | − | + | |
| J2 | +/− | − | − | − | + | |
| J3 | + | + | + | + | + | |
| J4 | + | + | + | + | + | |
| P5-51 | − | − | + | − | + | |
| P5-53 | + | + | − | − | + | |
| P7-11 | − | − | − | − | + | |
| P7-32 | − | − | − | − | + | |
| P7-62 | − | − | − | − | + | |
| P7-66 | − | − | − | − | + | |
| P9-3 | − | − | − | − | + | |
| P9-5 | + | + | + | + | + | |
| P9-6 | + | + | + | + | + | |
| P9-8 | + | + | − | − | + | |
| P9-9 | − | − | − | − | + | |
| P9-10 | + | + | − | + | + | |
| 26 | − | − | + | − | + | |
| CF13 | + | + | − | − | + | |
| EP147 | − | − | − | − | + | |
| JP25 | − | + | − | + | + | |
| MR39 | − | − | − | − | + | |
| MR40 | − | − | − | − | + | |
| NS99 | + | + | + | + | + | |
| SDOW17 | + | + | + | + | + | |
| SR30 | + | + | + | + | + | |
| VO17 | − | − | − | − | + | |
LV is the prototypic European representative, and SD-23983 is a reference North American strain. −, no reactivity; +, definitive strong reactivity; +/−, weak reactivity.
TABLE 6.
Reactivities with MAbs directed against PRRSV nucleocapsid epitopes
| MAb | Reactivity with isolatea:
|
Required domainsb | |||||
|---|---|---|---|---|---|---|---|
| SD-23983 | Lelystad | 13-US | 14-US | 15-US | 20-US | ||
| SDOW17 | + | + | + | + | + | + | I + V |
| VO17 | + | − | − | − | − | − | III + V |
| EP147 | + | − | − | − | − | − | III + V |
| SR30 | + | + | + | + | + | + | IV + V |
| MR40 | + | − | − | − | − | − | III + V |
| JP25 | + | − | + | − | + | + | III + V |
−, no reactivity; +, definitive strong reactivity.
Domains I (aa 30 to 52), II (aa 37 to 52), III (aa 52 to 69), IV (aa 69 to 112), and V (aa 112 to 123) of the ORF 7 protein product were previously identified by Wootton et al. (78).
No antigenic differences were seen in proteins GP2 to GP5 and M between strain LV and isolate 13-US, and only one difference was noted when strain LV was compared to 15-US (anti-GP4 MAb CI83). Several unique differences were observed for isolate 14-US when compared to the antigenic profile of strain LV. MAb P9-1, directed against GP3, reacted strongly with 14-US but did not recognize the other type 1 and type 2 isolates tested. As shown in Fig. 5A, the 14-US GP3 sequence contains 14 unique amino acid differences compared to the other type 1 isolates and is truncated. None of the MAbs directed against GP4 (CI82, CI83, NI26, NI37, and NI69) reacted with isolate 14-US, setting this isolate apart from the type 1 and type 2 viruses screened. As shown in Fig. 5B, the GP4 sequence of isolate 14-US contains 12 unique amino acid differences compared to the other type 1 viruses screened, three of which are located within the hypervariable region, a likely immunogenic target. MAbs P14-1 and 14-2, directed against the M protein, reacted with 14-US and SD-23983 but did not recognize the other type 1 isolates tested. The reason for this is unclear. Alignment of the amino acid sequence encoded by ORF 6 indicates that 14-US contains only one unique amino acid difference compared to the other type 1 viruses (data not shown). This difference in reactivity may be due to the involvement of another viral protein.
Antigenic differences between these new type 1 isolates were also seen with MAbs directed against the nucleocapsid protein. Characterization of isolate 14-US revealed 10 differences in relation to strain LV. Five of these differences were shared by isolate 15-US (G7, I1, P5-53, P9-8, and CF-13). Three MAbs (G5, P5-51, and 26) reacted only with 14-US and the U.S. type 2 isolate SD-23983. Two MAbs (G3 and P9-10) set isolate 14-US (no reactivity) apart from the other type 1 and type 2 viruses included in this survey. As shown in Fig. 7, 13-US and 15-US share 100% identity in the N protein while 14-US has four unique N amino acids. The reason for these differences remains unclear, but they may be due to involvement of another viral protein or to subtle alterations in nucleocapsid shape when associated with viral RNA. Only one anti-N MAb (JP25) was able to discriminate between strain LV and 14-US and isolates 13-US and 15-US (see below). The unique MAb pattern observed for 14-US is consistent with the phylogenetic analysis, which indicated that 14-US clustered tightly with 5-US and was unique from the cluster of U.S. type 1 viruses that included isolates 13-US and 15-US.
FIG. 7.
Alignment of the amino acid sequences of the nucleocapsids of 1-US, 13-US, 14-US, 15-US, 2-NL, VR2332, PA8, and SD23983. The required domains as defined by Wootton et al. (78) are indicated.
A subset of MAbs directed against the nucleocapsid protein have been characterized by Wootton et al. (78) (Fig. 7). They identified five specific domains of a type 2 isolate (PA-8) (77) required for recognition by the antibodies SDOW17, VO17, EP147, SR30, MR40, and JP25: domains I (aa 30 to 52), II (aa 37 to 52), III (aa 52 to 69), IV (aa 69 to 112), and V (aa 112 to 123). Domains I and V were required for SDOW17 recognition. Domains IV and V were required for recognition by SR30. Domains III and V were required for recognition by VO17, EP147, MR40, and JP25. As shown in Table 5, the recognition pattern of these MAbs with 13-US, 14-US, and 15-US was consistent with that of LV, with the exception of JP25. Simultaneous characterization of a subset of panel reactivity reaffirmed that MAb JP25 reacts with North American strain SD-23983 as well as 13-US, 15-US, and 20-US but does not recognize 14-US or strain LV. The reason for this is unclear. An alignment of nucleocapsid amino acid sequence for isolates LV, 13-US, 14-US, 15-US, and 1-US as well as type 2 strains VR-2332, PA-8, and SD-23983 demonstrates that 13-US and 15-US differ in sequence from LV by only the carboxyl amino acid (Fig. 7). VR-2332, PA-8, and SD-23983, recognized by JP25, are truncated at the carboxyl-terminal end in relation to all type 1 isolates examined (Fig. 7). The North American strains are quite diverse from the 13-US and 15-US isolates (68% similar), yet they share a common reactivity with JP25, likely due to the conserved domain III. However, 14-US and LV also share sequence similarity with VR-2332 in domain III, leading one to conclude that conformation of domain V is critical or another undescribed viral interaction is needed.
DISCUSSION
The genetic and antigenic analyses presented in this paper indicate that type 1 PRRSV isolates are present in U.S. swine herds from diverse geographic regions. The full-length genomic sequence of EuroPRRSV, the U.S. index type 1 isolate originating from a herd in the Midwest, was utilized to compare and contrast type 1 isolates identified since 1999. The result of such a study was the first genetic characterization of European-like PRRSV isolates originating from dispersed areas of the United States. Selected U.S. type 1 PRRSV isolates were then mapped antigenically.
The second full-length type 1 PRRSV sequence to be reported worldwide, EuroPRRSV, is remarkably similar to LV, sharing 95.3% overall nucleotide identity, but the 10-year interval between strain identification resulted in considerable divergence in localized domains. Although the origin of EuroPRRSV is unclear, comparison to strain LV suggests certain regions of the PRRSV type 1 genome may tolerate mutation more than others and thus may evolve more rapidly. These regions include the 5′ UTR, NSP1β, NSP2, and three envelope glycoproteins, GP3 to GP5. The 5′ UTR, critical in replication, transcription, and translation (24), was shown to possess only 93.2% nucleotide identity to LV, yet the 3′ UTR sequence was accurately maintained (98.2% identity). The reason for this disparity is unknown, as evidence suggests that these two regions may be in close proximity during viral replication (23, 49). NSP2 has been reported to be the most variable part of the genome (11, 61), and the ORF 1a sequence of EuroPRRSV was consistent with that notion, displaying a 17-aa deletion within the NSP2 protein, the only deletion or insertion observed in relation to the LV genomic sequence. Although NSP2 has been shown to be variable in type 2 viruses, this is the first instance of variation in this region for type 1 viruses and suggests that this PRRSV protein is uniformly unstable, perhaps due to immunologic pressure, as NSP2 was previously shown to contain a cluster of B-cell epitopes (47). The protein is well conserved at residues thought to be important in proteolytic cleavage events but differs in downstream amino acids. The fact that NSP2 is proline-rich (10 to 11% in both types) is particularly intriguing. PXXP, a signature SH3 binding motif, is seen a minimum of 10 times in both PRRSV subtypes, and its existence demands further exploration of protein function. Other aspects of the variable region, surrounding and including the deletion, are the subject of further investigations in our laboratories.
The high occurrence of amino acid mutations in GP3 to GP5 is in accordance with the results of other studies (11, 18, 46, 52, 60). The GP5 amino acids highlighted in Fig. 5C are predicted to lie in the extravirion domain of the protein shown to be responsible for cell attachment. The conserved amino acids have been shown to be critical in binding to the host cell, and the hypervariable regions surrounding these conserved amino acids have been implicated in enabling virus escape from host immunity, due to the presence of predicted N glycosylation (48, 53). It is of interest that U.S. type 1 isolates acquired additional predicted N glycosylation sites compared to strain Lelystad, and 22 sequenced isolates seem to show a tendency for variation at the same amino acids in each protein but not necessarily a mutation from strain Lelystad to an identical amino acid. Further analysis of GP5 variation may lead to increased significance for the mutational hotspots seen. The role of GP3 and GP4 in the virus replication cycle must be determined before importance can be attributed to the hypervariable regions of these proteins.
Phylogenetic analyses of published PRRSV ORF 3, 5, and 7 nucleotide sequences indicate that these newly emerging isolates form a clade with strain LV and the United Kingdom type 1 PRRSV isolates. Previous studies have found very low levels of sequence variability in European-type PRRSV from Great Britain, Belgium, France, Germany, The Netherlands, and Spain (17, 19, 30, 69). Forsberg et al. (19) confirmed the scant isolate variation in these countries but found diversity to be high among type 1 PRRSV when isolates from Italy and Denmark were included in the analysis. ORF 5 phylogenetic analysis also revealed a tight clustering of isolates from The Netherlands, Great Britain, France, and Belgium around LV, but quite divergent strains were shown to be present in Eastern Europe (67). Although high genetic variability is common among North American type 2 isolates, these new U.S. type 1 isolates cluster around the LV isolate despite their wide geographic distribution, suggesting a common source of entry into the United States. The exact time and source of North American introduction remain unknown for several reasons. These European-like isolates were identified only when swine were exhibiting symptoms consistent with PRRS disease, yet no evidence of type 2 sequence could be elucidated. This fact does not preclude the prior or present existence of type 1 isolates that may not have been diagnosed. Furthermore, tracking movement of PRRSV isolates by geography is difficult, as animal husbandry has changed significantly so that large swine producers own individual farms in several contiguous and noncontiguous U.S. states, with producers sharing animals within a production system as well as outside production systems. Lastly, PRRSV can be transmitted through artificial insemination of semen derived from boar studs, often of unknown PRRSV status.
Adaptation of three U.S. type 1 PRRSV isolates (13-US, 14-US, and 15-US) to grow to high titers (105 50% tissue culture infective doses) in MARC-145 cells enabled MAb screening, confirming a European-like serotype for these isolates. We are further exploring a variety of culture variations including pH control, gamma interferon inhibition, and macrophage coculture to facilitate the consistent isolation and propagation of additional type 1 PRRSV isolates. The MAb panel identified well-characterized antibodies that react strongly with these newly emerging type 1 isolates and are thus extremely useful in identifying and differentiating them in diagnostic samples. To illustrate the value of MAb analysis, anti-N MAb SDOW17 recognizes essentially all PRRSV strains tested, regardless of genetic type. MAbs MR39 and MR40 can then differentiate between European and North American isolates, recognizing only type 2 isolates to date. JP25 will recognize some U.S. type 1 isolates and thus differentiate them from LV but will recognize type 2 isolates as well. Other MAbs further distinguish PRRSV isolate antigenicity, such that a profile of individual isolates can be derived for downstream use. As stated previously, the difference in reactivities of conserved nucleocapsids with the large anti-N MAb panel may not be due merely to amino acid differences in the N protein but rather to an as-yet-uncharacterized interaction of the nucleocapsid with another viral protein or viral RNA. The MAb panel may therefore be a valuable tool in elucidating PRRSV protein-protein or protein-RNA interactions.
There are a number of challenges complicating the detection of European-like PRRSV in American swine herds. Currently, no commercially available serologic tests differentiate between European and North American PRRSV strains. The IDEXX PRRSV enzyme-linked immunosorbent assay utilizes antigen-coated plates that include both North American and LV-like antigens. Additionally, many European isolates have been reported to grow poorly in porcine alveolar macrophage cultures and often not at all on MARC-145 cells. Many North American diagnostic labs utilize RT-PCR-based tests with primers specific for identification of only type 2 PRRSV strains or do not differentiate between European and North American PRRSV strains. Submitted virus must then be sequenced directly from the clinical sample or isolated on alveolar macrophages. Both techniques are time-consuming and laborious, potentially leading to sample misdiagnosis. The information provided in this report will aid in overcoming these challenges.
Lastly, RNA recombination between the European and North American genotypes of PRRSV may be at least 10,000 times less likely to occur than RNA recombination between diverse European isolates (73). Indeed, phylogenetic analysis of nucleotide sequences of three separate ORFs (3, 5, and 7) from 13-US, 14-US, 15-US, and EuroPRRSV provided no evidence of recombination with type 2 isolates. If intertype RNA recombinants are generated at a low level under field conditions, it is likely that such recombinants would not be viable due to the low level of similarity between the strains (73) or that one of the original virus isolates would eventually dominate the intertype recombinant, as observed in cell culture (81). However, preexisting immunity to the parental viruses in the pig population may instill a selective advantage to the intertype recombinant, and even an extremely rare nonhomologous recombination event could rapidly spread the virus in the pig population (73). The possibility of new intertype PRRSV recombinants should be considered when evaluating diagnostic procedures. Intertype recombination could result in problems with virus typing and vaccination and may potentially generate a virus with new biological properties. Therefore, it is essential to characterize the type 1 isolates now circulating in United States. The unique characteristics of these European-like isolates may have a significant impact on diagnostic approaches to viral detection and to future swine management strategies.
Acknowledgments
We thank Adam Woodbury, Linda Schuveiller, and Dan Mickelson for excellent technical assistance.
Funding for this work was derived from USDA NRI 0002114, NPB (National Pork Board) 01-102, Department of Veterinary Science South Dakota State PRRS Royalty Fund, USDA-CSRES-National Research Initiative Competitive Grants Program, South Dakota Animal Disease Research and Diagnostic Laboratory, South Dakota Agricultural Experiment Station, and USDA NRI Postdoctoral grant 2002-35204-11643.
REFERENCES
- 1.Allende, R., T. L. Lewis, Z. Lu, D. L. Rock, G. F. Kutish, A. Ali, A. R. Doster, and F. A. Osorio. 1999. North American and European porcine reproductive and respiratory syndrome viruses differ in non-structural protein coding regions. J. Gen. Virol. 80:307-315. [DOI] [PubMed] [Google Scholar]
- 2.Arold, S., P. Franken, M. P. Strub, F. Hoh, S. Benichou, R. Benarous, and C. Dumas. 1997. The crystal structure of HIV-1 Nef protein bound to the Fyn kinase SH3 domain suggests a role for this complex in altered T cell receptor signaling. Structure 5:1361-1372. [DOI] [PubMed] [Google Scholar]
- 3.Benfield, D. A., E. Nelson, J. E. Collins, L. Harris, S. M. Goyal, D. Robison, W. T. Christianson, R. B. Morrison, D. Gorcyca, and D. Chladek. 1992. Characterization of swine infertility and respiratory syndrome (SIRS) virus (isolate ATCC VR-2332). J. Vet. Diagn. Investig. 4:127-133. [DOI] [PubMed] [Google Scholar]
- 4.Black, A. R., J. D. Black, and J. Azizkhan-Clifford. 2001. Sp1 and kruppel-like factor family of transcription factors in cell growth regulation and cancer. J. Cell. Physiol. 188:143-160. [DOI] [PubMed] [Google Scholar]
- 5.Botner, A., B. Strandbygaard, K. J. Sorensen, P. Have, K. G. Madsen, E. S. Madsen, and S. Alexandersen. 1997. Appearance of acute PRRS-like symptoms in sow herds after vaccination with a modified live PRRS vaccine. Vet. Rec. 141:497-499. [DOI] [PubMed] [Google Scholar]
- 6.Cavanaugh, D. 1997. Nidovirales: a new order comprising Coronaviridae and Arteriviridae. Arch. Virol. 142:629-633. [PubMed] [Google Scholar]
- 7.Cicchetti, P., B. J. Mayer, G. Thiel, and D. Baltimore. 1992. Identification of a protein that binds to the Sh3 region of Abi and is similar to Bcr and Gap-Rho. Science 257:803-806. [DOI] [PubMed] [Google Scholar]
- 8.Cohen, G. B., R. B. Ren, and D. Baltimore. 1995. Modular binding domains in signal-transduction proteins. Cell 80:237-248. [DOI] [PubMed] [Google Scholar]
- 9.Collins, J. E., D. A. Benfield, W. T. Christianson, L. Harris, J. C. Hennings, D. P. Shaw, S. M. Goyal, S. McCullough, R. B. Morrison, H. S. Joo, D. E. Gorcyca, and D. W. Chladek. 1992. Isolation of swine infertility and respiratory syndrome virus (isolate ATCC VR-2332) in North America and experimental reproduction of the disease in gnotobiotic pigs. J. Vet. Diagn. Investig. 4:117-126. [DOI] [PubMed] [Google Scholar]
- 10.Conzelmann, K., N. Visser, P. van Woensel, and H. Thiel. 1993. Molecular characterization of porcine reproductive and respiratory syndrome virus, a member of the arterivirus group. Virology 193:329-339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dea, S., C. A. Gagnon, H. Mardassi, B. Pirzadeh, and D. Rogan. 2000. Current knowledge on the structural proteins of porcine reproductive and respiratory syndrome (PRRS) virus: comparison of the North American and European isolates. Arch. Virol. 145:659-688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Den Boon, J. A., E. J. Snijder, E. D. Chirnside, A. F. de Vries, M. C. Horzinek, and W. J. M. Spaan. 1991. Equine arteritis virus is not a togavirus but belongs to the cornaviruslike superfamily. J. Virol. 65:2910-2920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Den Boon, J. A., K. S. Faaberg, J. J. M. Meulenberg, A. L. M. Wassenaar, P. G. W. Plagemann, A. E. Gorbelenya, and E. J. Snijder. 1995. Processing and evolution of the N-terminal region of the arterivirus replicase ORF1a protein: identification of two papainlike cysteine proteases. J. Virol. 69:4500-4505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.de Vries, A. A. F., E. D. Chirnside, P. J. Bredenbeek, L. A. Gravestein, M. C. Horzinek, and W. J. M. Spaan. 1990. The genome organization of the nidovirales: similarities and difference between arteri-, toro- and coronaviruses. Semin. Virol. 8:33-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dewey, C., G. Charbonneau, S. Carman, A. Hamel, G. Nayar, R. Friendship, K. Eernisse, and S. Swenson. 2000. Lelystad-like strain of porcine reproductive and respiratory syndrome virus (PRRSV) identified in Canadian swine. Can. Vet. J. 21:493-494. [PMC free article] [PubMed] [Google Scholar]
- 16.Dougherty, W. G., and B. L. Semler. 1993. Expression of virus-encoded proteinases: functional and structural similarities with cellular enzymes. Microbiol. Rev. 57:781-822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Drew, T. W., J. P. Lowings, and F. Yapp. 1997. Variation in open reading frames 3, 4, and 7 among porcine reproductive and respiratory syndrome virus isolates in the UK. Vet. Microbiol. 55:209-221. [DOI] [PubMed] [Google Scholar]
- 18.Forsberg, R., M. B. Oleksiewicz, A.-M. K. Petersen, J. Hein, A. Botner, and T. Storgaard. 2001. A molecular clock dates the common ancestor of European-type porcine reproductive and respiratory syndrome virus at more than 10 years before the emergence of disease. Virology 289:174-179. [DOI] [PubMed] [Google Scholar]
- 19.Forsberg, R., T. Storgaard, H. S. Nielsen, M. B. Oleksiewicz, P. Cordioli, G. Sala, J. Hein, and A. Botner. 2002. The genetic diversity of European type PRRSV is similar to that of the North American type but is geographically skewed within Europe. Virology 299:38-47. [DOI] [PubMed] [Google Scholar]
- 20.Godeny, E. K., L. Chen, S. N. Kumar, S. L. Methven, E. V. Koonin, and M. A. Brinton. 1993. Complete genomic sequence and phylogenetic analysis of the lactate dehydrogenase-elevating virus (LDV). Virology 194:585-596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Goldberg, T. L., E. C. Hahn, R. M. Weigel, and G. Scherba. 2000. Genetic, geographical and temporal variation of porcine reproductive and respiratory syndrome virus in Illinois. J. Gen. Virol. 81:171-179. [DOI] [PubMed] [Google Scholar]
- 22.Herold, J., A. E. Gorbalenya, V. Thiel, B. Schelle, and S. G. Siddell. 1998. Proteolytic processing at the amino terminus of human coronavirus 229E gene 1-encoded polyproteins: identification of a papain-like proteinase and its substrate. J. Virol. 72:910-918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang, P., and M. M. Lai. 2001. Heterogeneous nuclear ribonucleoprotein a1 binds to the 3′-untranslated region and mediates potential 5′-3′-end cross talks of mouse hepatitis virus RNA. J. Virol. 75:5009-5017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hwang, Y.-K., and M. A. Brinton. 1998. A 68-nucleotide sequence within the 30 noncoding region of simian hemorrhagic fever virus negative-strand RNA binds to four MA 104 cell proteins. J. Virol. 72:4341-4351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kapur, V., M. R. Elam, T. M. Pawlovich, and M. P. Murtaugh. 1996. Genetic variation in porcine reproductive and respiratory syndrome virus isolates in the midwestern United States. J. Gen. Virol. 77:1271-1276. [DOI] [PubMed] [Google Scholar]
- 26.Katz, J. B., A. L. Shafer, K. A. Eernisse, J. G. Landgraf, and E. A. Nelson. 1995. Antigenic differences between European and American isolates of porcine reproductive and respiratory syndrome virus (PRRSV) are encoded by the carboxyterminal portion of viral open reading frame 3. Vet. Microbiol. 44:65-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Keffaber, K. K. 1989. Reproductive failure of unknown etiology. Am. Assoc. Swine Prac. Newsl. 1:1-10. [Google Scholar]
- 28.Kim, H. S., J. Kwang, I. J. Yoon, H. S. Joo, and M. L. Frey. 1993. Enhanced replication of porcine reproductive and respiratory syndrome (PRRS) virus in a homogeneous subpopulation of MA-104 cell line. Arch. Virol. 133:477-483. [DOI] [PubMed] [Google Scholar]
- 29.Lee, C. H., B. Leung, M. A. Lemmon, J. Zheng, D. Cowburn, J. Kuriyan, and K. Saksela. 1995. A single amino acid in the SH3 domain of Hck determines its high affinity and specificity in binding to HIV-1 Nef protein. EMBO J. 14:5006-5015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Le Gall, A. L., E. Albine, R. Magar, and J. P. Gauthier. 1998. Antigenic variability of porcine reproductive and respiratory syndrome (PRRS) virus isolates: influence of virus passage in pig. Virus Res. 28:247-257. [PubMed] [Google Scholar]
- 31.Madsen, K. G., C. M. Hansen, E. S. Madsen, B. Strandbygaard, A. Botner, and K. J. Sorensen. 1998. Detection of porcine reproductive and respiratory syndrome virus of the American type in Danish swine herds. Arch. Virol. 143:1683-1700. [DOI] [PubMed] [Google Scholar]
- 32.Magar, R., R. Larochelle, E. A. Nelson, and C. Charreyre. 1997. Differential reactivity of a monoclonal antibody directed to the membrane protein of porcine reproductive and respiratory syndrome virus. Can. J. Vet. Res. 61:69-71. [PMC free article] [PubMed] [Google Scholar]
- 33.Mateu, E., M. Martin, and D. Vidal. 2003. Genetic diversity and phylogenetic analysis of glycoprotein 5 of European-type porcine reproductive and respiratory virus strains in Spain. J. Gen. Virol. 84:529-534. [DOI] [PubMed] [Google Scholar]
- 34.Meng, X. J. 2000. Heterogeneity of porcine reproductive and respiratory syndrome virus: implications for current vaccine efficacy and future vaccine development. Vet. Microbiol. 74:309-329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mengeling, W. L., K. M. Lager, A. C. Vorwald, and D. F. Clouser. 2003. Comparative safety and efficacy of attenuated single-strain and multi-strain vaccines for porcine reproductive and respiratory syndrome. Vet. Microbiol. 93:25-38. [DOI] [PubMed] [Google Scholar]
- 36.Meulenberg, J. J. M., M. M. Hulst, E. J. de Meijer, R. J. L. M. Moonen, A. den Besten, E. P. de Kluyver, G. Wensvoort, and R. J. M. Moormann. 1993. Lelystad virus, the causative agent of porcine epidemic abortion and respiratory syndrome (PEARS), is related to LDV and EAV. Virology 192:62-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mounir, S., H. Mardassi, and S. Dea. 1995. Identification and characterization of the porcine reproductive and respiratory virus ORFs 7, 5 and 4 products. Adv. Exp. Med. Biol. 380:317-320. [DOI] [PubMed] [Google Scholar]
- 38.Nakagawa, H., T. Inomoto, and A. K. Rustgi. 1997. A CACCC box-like cis-regulatory element of the Epstein-Barr virus ED-L2 promoter interacts with a novel transcriptional factor in tissue-specific squamous epithelia. J. Biol. Chem. 272:16688-16699. [DOI] [PubMed] [Google Scholar]
- 39.Nelsen, C. J., M. P. Murtaugh, and K. S. Faaberg. 1999. Porcine reproductive and respiratory syndrome virus comparison: divergent evolution on two continents J. Virol. 73:270-280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nelson, E., J. C. Hennings, L. Harris, J. E. Collins, D. E. Gorcyca, D. W. Chladek, and D. A. Benfield. 1992. Characterization of monoclonal antibodies to an isolate of porcine epidemic abortion and respiratory syndrome virus. Proc. Int. Pig Vet. Soc. 12:121. [Google Scholar]
- 41.Nelson, E. A., J. Christopher-Hennings, T. Drew, G. Wensvoort, J. Collins, and D. A. Benfield. 1993. Differentiation of U. S. and European isolates of porcine reproductive and respiratory syndrome virus by monoclonal antibodies. J. Clin. Microbiol. 31:3184-3189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nelson, E. A., J. Christopher-Hennings, and D. A. Benfield. 1995. Structural proteins of porcine reproductive and respiratory syndrome virus (PRRSV). Adv. Exp. Med. Biol. 380:321-323. [DOI] [PubMed] [Google Scholar]
- 43.Nelson, J. W., J. Zhu, C. C. Smith, M. Kulka, and L. Aurelian. 1996. ATP and SH3 binding sites in the protein kinase of the large subunit of herpes simplex virus type 2 of ribonucleotide reductase (ICP10). J. Biol. Chem. 271:17021-17027. [DOI] [PubMed] [Google Scholar]
- 44.Okumura, K., G. Sakaguchi, S. Takagi, K. Naito, T. Mimori, and H. Igarashi. 1996. Sp1 family proteins recognize the U5 repressive element of the long terminal repeat of human T cell leukemia virus type I through binding to the CACCC core motif. Proc. Natl. Acad. Sci. USA 271:12944-12950. [DOI] [PubMed] [Google Scholar]
- 45.Oleksiewicz, M. B., A. Botner, J. Nielsen, and T. Storgaard. 1999. Determination of 5′-leader sequences from radically disparate strains of porcine reproductive and respiratory syndrome virus reveals the presence of highly conserved sequence motifs. Arch. Virol. 144:981-987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Oleksiewicz, M. B., A. Botner, P. Toft, T. Grubbe, J. Nielsen, S. Kamstrup, and T. Storgaard. 2000. Emergence of porcine reproductive and respiratory syndrome virus deletion mutants. Correlation with the porcine antibody response to a hypervariable site in the ORF 3 structural glycoprotein. Virology 267:135-140. [DOI] [PubMed] [Google Scholar]
- 47.Oleksiewicz, M. B., A. Botner, P. Toft, P. Normann, and T. Storgaard. 2001. Epitope mapping porcine reproductive and respiratory syndrome virus by phage display: the nsp2 fragment of the replicase polyprotein contains a cluster of B-cell epitopes. J. Virol. 75:3277-3290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ostrowski, M., J. A. Galeota, A. M. Jar, K. B. Platt, F. A. Osorio, and O. J. Lopez. 2002. Identification of neutralizing and nonneutralizing epitopes in the porcine reproductive and respiratory syndrome virus GP5 ectodomain. J. Virol. 76:4241-4250. (Erratum, 76:6863.) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Palmenberg, A. C., and J.-Y. Sgro. 1997. Topological organization of picornaviral genomes: statistical prediction of RNA structural signals. Semin. Virol. 8:231-241. [Google Scholar]
- 50.Pasternak, A. O., E. van den Born, W. J. Spaan, and E. J. Snijder. 2001. Sequence requirements for RNA strand transfer during nidovirus discontinuous subgenomic RNA synthesis. EMBO J. 20:7220-7228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Pawson, T. 1995. Protein modules and signaling networks. Nature 373:573-580. [DOI] [PubMed] [Google Scholar]
- 52.Pirzadeh, B., C. A. Gagnon, and S. Dea. 1998. Genetic and antigenic variation of porcine reproduction and respiratory syndrome virus major envelope GP5 glycoprotein. Can. J. Vet. Res. 62:170-177. [PMC free article] [PubMed] [Google Scholar]
- 53.Plagemann, P. G., R. R. Rowland, and K. S. Faaberg. 2002. The primary neutralization epitope of porcine respiratory and reproductive syndrome virus strain VR-2332 is located in the middle of the GP5 ectodomain. Arch. Virol. 147:2327-2347. [DOI] [PubMed] [Google Scholar]
- 54.Plagemann, P. G. W., and V. Moenning. 1992. Lactate dehydrogenase elevating virus, equine arteritis virus and simian hemorrhagic fever virus: a new group of positive strand RNA virus. Adv. Virus Res. 41:99-192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Plagemann, P. G. W. 1996. Lactate dehydrogenase-elevating virus and related viruses, p. 1105-1120. In B. Fields (ed.), Fields virology, 3rd ed. Lippincott-Raven, Philadelphia, Pa.
- 56.Ramachandran, A., A. Jain, P. Arora, M. D. Bashyam, U. Chatterjee, S. Ghosh, V. K. Parnaik, and S. E. Hasnain. 2001. Novel Sp family-like transcription factors are present in adult insect cells and are involved in transcription from the polyhedrin gene initiator promoter. J. Biol. Chem. 276:23440-23449. [DOI] [PubMed] [Google Scholar]
- 57.Rossow, K. D., E. M. Bautista, S. M. Goyal, T. W. Molitor, M. P. Murtaugh, R. B. Morrison, D. A. Benfield, and J. E. Collins. 1994. Experimental porcine reproductive and respiratory syndrome virus infection in one-, four-, and 10-week old pigs. J. Vet. Diagn. Investig. 6:3-12. [DOI] [PubMed] [Google Scholar]
- 58.Rossow, K. D. 1998. Porcine reproductive and respiratory syndrome. Vet. Pathol. 35:1-20. [DOI] [PubMed] [Google Scholar]
- 59.Rossow, K. D., J. L. Shiver, P. E. Yeske, D. D. Polson, R. R. R. Rowland, S. R. Lawson, M. P. Murtaugh, E. A. Nelson, and J. E. Collins. 1999. Porcine reproductive and respiratory syndrome virus infection in neonatal pigs characterized by marked neurovirulence. Vet. Rec. 144:444-448. [DOI] [PubMed] [Google Scholar]
- 60.Rowland, R. R., M. Steffen, T. Ackerman, and D. A. Benfield. 1999. The evolution of porcine reproductive and respiratory syndrome virus: quasispecies and emergence of a virus subpopulation during infection of pigs with VR-2332. Virology 259:262-266. [DOI] [PubMed] [Google Scholar]
- 61.Shen, S., J. Kwang, W. Liu, and D. X. Lui. 2000. Determination of the complete nucleotide sequence of a vaccine strain of porcine reproductive and respiratory syndrome virus and identification of the nsp2 gene with a unique insertion. Arch. Virol. 145:871-883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Snijder, E. J., A. L. M. Wassenaar, and W. J. M. Spaan. 1992. The 5′ end of the equine arteritis virus replicase gene encodes a papainlike cysteine protease. J. Virol. 66:7040-7048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Snijder, E. J., A. L. M. Wassenaar, and W. J. M. Spaan. 1994. Proteolytic processing of the replicase ORF1a protein of equine arteritis virus. J. Virol. 68:5755-5764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Snijder, E. J., A. L. M. Wassenaar, W. J. M. Spaan, and A. E. Gorbalenya. 1995. The arterivirus NSP2 protease. An unusual cysteine protease with primary structure similarities to both papain-like and chymotrypsin-like proteases. J. Biol. Chem. 270:16671-16676. [DOI] [PubMed] [Google Scholar]
- 65.Snijder, E. J., and J. J. M. Meulenberg. 1998. The molecular biology of arteriviruses. J. Gen. Virol. 79:961-979. [DOI] [PubMed] [Google Scholar]
- 66.Snijder, E. J., H. van Tol, N. Roos, and K. W. Pedersen. 2001. Non-structural proteins 2 and 3 interact to modify host cell membranes during the formation of the arterivirus replication complex. J. Gen. Virol. 82:985-994. [DOI] [PubMed] [Google Scholar]
- 67.Stadejek, T., A. Stankevicius, T. Storgaard, M. B. Oleksiewicz, S. Belak, T. W. Drew, and Z. Pejsak. 2002. Identification of radically different variants of porcine reproductive and respiratory syndrome virus in Eastern Europe: towards a common ancestor for European and American viruses. J. Gen. Virol. 83:1861-1873. [DOI] [PubMed] [Google Scholar]
- 68.Strimmer, K., and A. von Haeseler. 1996. Quartet puzzling: a quartet maximum likelihood method for reconstructing tree topologies. Mol. Biol. Evol. 13:964-969. [Google Scholar]
- 69.Suarez, P., R. Zardoya, M. J. Martin, C. Prieto, J. Dopazo, A. Soana, and J. M. Castro. 1996. Phylogenetic relationship of European strains of porcine reproductive and respiratory syndrome virus (PRRSV) inferred from DNA sequences of putative ORF-5 and ORF-7 genes. Virus Res. 42:159-165. [DOI] [PubMed] [Google Scholar]
- 70.Tan, S. L., H. Nakao, Y. He, S. Vijaysri, P. Neddermann, B. L. Jacobs, B. J. Mayer, and M. G. Katze. 1999. NS5A, a nonstructural protein of hepatitis C virus, binds growth factor receptor-bound protein 2 adaptor protein in a Src homology 3 domain/ligand-dependent manner and perturbs mitogenic signaling. Proc. Natl. Acad. Sci. USA 96:5533-5538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Thompson, J. D., T. J. Gibson, F. Plewniak, F. Jeanmoughin, and D. G. Higgins. 1997. The CLUSTAL X windows interface: flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucleic Acids Res. 25:4876-4882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.van Marle, G., J. C. Dobbe, A. P. Gultyaev, W. Luytjes W. J. Spaan, and E. J. Snijder. 1999. Arterivirus discontinuous mRNA transcription is guided by base pairing between sense and antisense transcription-regulating sequences. Proc. Natl. Acad. Sci. USA 96:12056-12061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.van Vugt, J. J., T. Storgaard, M. B. Oleksiewicz, and A. Botner. 2001. High frequency RNA recombination in porcine reproductive and respiratory syndrome virus occurs preferentially between parental sequences with high similarity J. Gen. Virol. 82:2615-2630. [DOI] [PubMed] [Google Scholar]
- 74.Wensvoort, G., C. Terpstra, J. M. Pol, E. A. Ter Laak, M. Bloemraad, E. P. De Kluyver, C. Kragten, L. Van Buiten, A. den Besten, and F. Wagenaar. 1991. Mystery swine disease in The Netherlands: the isolation of Lelystad virus. Vet. Q. 13:121-130. [DOI] [PubMed] [Google Scholar]
- 75.Wensvoort, G., E. P. de Kluyver, E. A. Luitjze, A. den Besten, L. Harris, J. E. Collins, W. T. Christianson, and D. Chladek. 1992. Antigenic comparison of Lelystad virus and swine infertility and respiratory syndrome (SIRS) virus. J. Vet. Diagn. Investig. 4:134-138. [DOI] [PubMed] [Google Scholar]
- 76.Wissink, E. H., H. A. van Wijk, M. V. Kroese, E. Weiland, J. J. Meulenberg, P. J. Rottier, and P. A. van Rijn. 2003. The major envelope protein, GP5, of a European porcine reproductive and respiratory syndrome virus contains a neutralization epitope in its N-terminal ectodomain. J. Gen. Virol. 84:1535-1543. [DOI] [PubMed] [Google Scholar]
- 77.Wootton, S., D. Yoo, and D. Rogan. 2000. Full-length sequence of a Canadian porcine reproductive and respiratory syndrome virus (PRRSV) isolate. Arch. Virol. 145:2297-2323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wootton, S. K., E. A. Nelson, and D. Yoo. 1998. Antigenic structure of the nucleocapsid protein of porcine reproductive and respiratory syndrome virus. Clin. Diagn. Lab. Immunol. 5:773-779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Wu, W. H., Y. Fang, R. Farwell, M. Steffen-Bien, R. R. Rowland, J. Christopher-Hennings, and E. A. Nelson. 2001. A 10-kDa structural protein of porcine reproductive and respiratory syndrome virus encoded by ORF2b. Virology 287:183-191. [DOI] [PubMed] [Google Scholar]
- 80.Yang, L., K. J. Yoon, Y. Li, J. H. Lee, J. J. Zimmerman, M. L. Frey, K. M. Harmon, and K. B. Platt. 1999. Antigenic and genetic variations of the 15 kD nucleocapsid protein of porcine reproductive and respiratory syndrome virus isolates. Arch. Virol. 144:525-546. [DOI] [PubMed] [Google Scholar]
- 81.Yuan, S., C. J. Nelsen, M. P. Murtaugh, B. J. Schmitt, and K. S. Faaberg. 1999. Recombination between North American strains of porcine reproductive and respiratory syndrome virus. Virus Res. 61:87-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yuan, S., D. Mickelson, M. P. Murtaugh, and K. S. Faaberg. 2001. Complete genome comparison of porcine reproductive and respiratory syndrome virus parental and attenuated strains. Virus Res. 79:189-200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yuan, S., M. P. Murtaugh, and K. S. Faaberg. 2000. Heteroclite RNAs are produced in porcine reproductive syndrome virus infection. Virology 275:158-169. [DOI] [PubMed] [Google Scholar]
- 84.Zeman, D., R. Neiger, M. Yaeger, E. Nelson, D. Benfield, P. Leslie-Steen, J. Thomson, D. Miskimins, R. Daly, and M. Minehart. 1993. Laboratory investigation of PRRS virus infection in three swine herds. J. Vet. Diagn. Investig. 5:522-528. [DOI] [PubMed] [Google Scholar]
- 85.Ziebuhr, J., E. J. Snijder, and A. E. Gorbalenya. 2000. Virus-encoded proteinases and proteolytic processing in the Nidovirales. J. Gen. Virol. 81:853-879. [DOI] [PubMed] [Google Scholar]
- 86.Zimmerman, J., K. J. Yoon, G. Stevenson, and S. A. Dee (ed.). 1998. PRRS compendium: a comprehensive reference on porcine reproductive and respiratory syndrome for pork producers, veterinary practitioners, and researchers, 1st ed., p. 14-17. National Pork Producers Council, Des Moines, Iowa.