Abstract
Drugs that interfere with cannabinoid CB1 transmission suppress various food-motivated behaviors, and it has been suggested that such drugs could be useful as appetite suppressants. Biochemical studies indicate that most of these drugs assessed thus far have been CB1 inverse agonists, and although they have been shown to suppress food intake, they also appear to induce nausea and malaise. The present studies were undertaken to characterize the behavioral effects of AM4113, which is a CB1 neutral antagonist, and to examine whether this drug can reduce food-reinforced behaviors and feeding on diets with varying macronutrient compositions. Biochemical data demonstrated that AM4113 binds to CB1 receptors, but does not show inverse agonist properties (ie no effects on cyclic-AMP production). In tests of spontaneous locomotion and analgesia, AM4113 reversed the effects of the CB1 agonist AM411. AM4113 suppressed food-reinforced operant responding with rats responding on fixed ratio (FR) 1 and 5 schedules of reinforcement in a dose-dependent manner, and also suppressed feeding on high-fat, high-carbohydrate, and lab chow diets. However, in the same dose range that suppressed feeding, AM4113 did not induce conditioned gaping, which is a sign of nausea and food-related malaise in rats. These results suggest that AM4113 may decrease appetite by blocking endogenous cannabinoid tone, and that this drug may be less associated with nausea than CB1 inverse agonists.
Keywords: appetite, motivation, operant, feeding, THC, rimonabant
INTRODUCTION
CB1 antagonist/inverse agonists such as SR141716 have been shown repeatedly to suppress feeding and food-motivated behavior. SR141716 attenuated the hyperphagia induced by CB1 agonists (Jamshidi and Taylor, 2001; Kirkham et al, 2002; Williams and Kirkham, 1999), and when administered alone it reduced food intake in a number of different animal models (Arnone et al, 1997; Colombo et al, 1998; Simiand et al, 1998; Williams and Kirkham, 1999). Feeding suppression induced by CB1 antagonists/inverse agonists has been demonstrated in both satiated and food-deprived animals following systemic or central administration, and after either acute or chronic treatment (Chen et al, 2004; Colombo et al, 1998; Shearman et al, 2003; Wiley et al, 2005). Although it is clear that drugs that interfere with CB1 transmission can suppress food intake, the mechanisms by which they accomplish this are less well understood. Biochemical studies indicate that many of these drugs, including SR141716, AM251, and AM1387, act as inverse agonists and exert actions on signal transduction mechanisms when administered in the absence of CB1 receptor stimulation (ie they inhibit GTPγS and increase cAMP production; Landsman et al, 1997; Mato et al, 2002; McLaughlin et al, 2006). In one recent study, CB1-knockout and wild-type mice responded comparably on a progressive ratio schedule reinforced with corn oil, while wild-type mice treated with SR141716 decreased responding, suggesting that SR141716 may exert inverse agonist effects in addition to simply blocking CB1 receptors (Ward and Dykstra, 2005).
There is evidence to suggest that some of the feeding-related effects produced by drugs that act on CB1 receptors may be due to actions such as food avoidance, food aversion, nausea, or malaise. Several studies have shown that CB1 agonists have anti-emetic actions (Gonzalez-Rosales and Walsh, 1997; Simoneau et al, 2001; Darmani and Johnson, 2004). CB1 receptors are present in the brain stem dorsal vagal complex, and CB1 receptors in this area are associated with triggering emetic responses (Van Sickle et al, 2003). Conditioned taste avoidance can be produced by administration of the CB1 inverse agonists SR141716 (De Vry et al, 2004) and AM251 (McLaughlin et al, 2005b). SR141716 potentiated lithium chloride-induced conditioned rejection reactions in rats (Parker et al, 2003), and this drug also produced emesis in the least shrew (Darmani, 2001) and nausea in humans (Despres et al, 2005). Although rats do not vomit, recent studies from our laboratory demonstrated that administration of AM251 led to the production of conditioned gaping in rats (McLaughlin et al, 2005b), which is thought to be a selective marker of nausea in that species (Parker et al, 1998; Parker and Limebeer, 2006). These gaping responses are elicited by treatments that produce vomiting in emetic species (Parker et al, 1998), and treatments that attenuate toxin-induced vomiting in emetic species also attenuate toxin-induced conditioned gaping in rats (Limebeer and Parker, 2000, 2003; Limebeer et al, 2004; for a review, see Parker et al, 2003).
In view of the fact that SR141716 and AM251 can act as inverse agonists, it remains possible that the feeding suppression induced by neutral antagonists (eg Gardner and Mallet, 2006) would not be accompanied by behavioral signs of nausea. For this reason, it is important to evaluate novel CB1 neutral antagonists. The present studies summarize initial research on the biochemical and behavioral effects of AM4113, which is a pyrazole analog structurally related to SR141716 and AM251. Experiment 1a examined the ability of AM4113 to bind to CB1 and CB2 receptors. In order to determine if AM4113 also acts as an inverse agonist at the cellular level, signal transduction effects of this drug were investigated using cAMP assays (Experiment 1b). Experiment 2 characterized the CB1 antagonist activity of AM4113 by assessing the ability of this drug to reverse the behavioral effects of the CB1 agonist AM411 (McLaughlin et al, 2005a) using tests that are associated with cannabinoid receptor activation (eg Martin et al, 1991). Experiments 3–5 assessed the behavioral effects of AM4113, employing the same food-related tasks previously used to characterize the actions of AM251, AM1387, and SR141716 (McLaughlin et al, 2003, 2005b, 2006). Experiment 3 examined the effects of AM4113 on food-reinforced lever pressing using two different fixed-ratio (FR) schedules. Experiment 4 studied the effects of AM4113 on the consumption of diets with different macronutrient compositions (eg high fat, high carbohydrate, and lab chow; McLaughlin et al, 2003, 2005b, 2006). Finally, experiment 5 employed the taste reactivity test developed by Grill and Norgren (1978) to determine if AM4113 can produce conditioned gaping in a manner similar to that previously shown to occur after administration of AM251 (McLaughlin et al, 2005b). It was hypothesized that AM4113 would display neurochemical characteristics of a CB1 neutral antagonist without inverse agonist properties, would be able to block behavioral effects of a CB1 agonist, and would attenuate appetite and food-reinforced behaviors without inducing behaviors associated with nausea and malaise.
MATERIALS AND METHODS
Animals
For the behavioral experiments, adult male Sprague–Dawley rats (Harlan, Indianapolis, IN) were housed in a colony room on a 12-h light–dark cycle (lights on during 0700–1900). All experiments were conducted during the light part of the cycle. For experiments 3–4, rats were food-deprived to 85% of their free-feeding body weight and weighed daily. All animal protocols were approved by the Institution for Animal Care and Use Committee and the methods were in accordance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animals Resources, 1996).
Drugs
AM4113, AM411, and AM251 were synthesized in the laboratory of Alex Makriyannis at the Center for Drug Discovery, Northeastern University. AM4113 is a pyrazole-3-carboxamide analog of SR141716A. For pharmacological studies, AM4113 was suspended in dimethylsulfoxide (DMSO), Tween 80, and 0.9% saline, with a ratio of 1: 1 :8. This combination also served as vehicle control solution. The cannabinoid agonist AM411 also was suspended in DMSO, Tween 80, and 0.9% saline in a 1 :1 :8 ratio.
Experimental Procedures
Experiment 1a—Rat brain CB1 and mouse spleen CB2 binding assay
AM4113, AM251, and SR141716A were tested for binding to the CB1 receptor using a rat brain membrane preparation, and for CB2 receptor binding using HEK293 cell membranes expressing hCB2 membranes, as previously described using [3H]CP-55,940 (Morse et al, 1995; Lan et al, 1999; Makriyannis et al, 2005; McLaughlin et al, 2006). The concentrated stocks (10mM) were diluted into TME buffer (50mM Tris-HCl, 3mM MgCl2, 100mM NaCl, 0.2mM EDTA, pH 7.4) with 0.1% BSA, and transferred to 96-well plates containing [3H]CP-55,940 (specific activity 128 Ci/mmol; NIDA) at a final concentration of 0.76 nM. Nonspecific binding was assessed in the presence of 100 nM CP 55,940. Binding was initiated with the addition of the respective membrane suspension (~50 µg membrane protein) followed by incubation at 30°C with gentle agitation in a shaking water bath for 60 min. Binding was terminated by rapid filtration of the membrane suspension over Unifilter GF/B-96 Well Filter Plates (Packard Instruments) using a Packard Filtermate-196 Cell Harvester. The filter plates were washed four times with ice-cold wash buffer (50mM Tris-base, 5mM MgCl2 with 0.5% BSA) and bound radioactivity was determined using a Packard TopCount Scintillation Counter. Results from the above heterologous competition assays were analyzed using nonlinear regression to determine the actual IC50 of the ligand (Prism by GraphPad Software Inc.) and the Ki values were calculated from the IC50 determinations (Cheng and Prusoff, 1973). All data were in duplicate with IC50 and Ki values determined from at least two independent experiments.
Experiment 1b—CAMP assay
Intracellular cyclic AMP levels were measured with a competitive protein-binding assay using intact HEK293 cells expressing hCB1 or hCB2 as previously described (McLaughlin et al, 2006). These cells were resuspended in 20 mM HEPES buffer, pH 7.3, containing 0.1 mM RO-20-1724 (4-[(3-butoxy-4-methoxyphenyl)-methyl]-2-imidazolidinone) and 1mM IBMX (isobutylmethyl-xanthine) in DME media with 0.1% BSA to a final concentration of 1 × 106 cells/ml. Cells were incubated for 5min at 37°C with forskolin, the HEPES/DME buffer, and various concentrations of compound. The reaction was stopped and the cells were lysed by boiling followed by cooling on ice. Cell membrane pellets were harvested by centrifugation and the cAMP assay kit from Diagnostic Products Corporation (Los Angeles, CA) was used. The results were expressed as percent inhibition of forskolin-stimulated cyclic AMP accumulation, and EC50 curves were generated with the use of GraphPad Prism software. As in previous studies with other compounds (McLaughlin et al, 2006), the concentrations of AM4113 tested ranged from 2 log units (base 10) below to 4 log units above the Ki value obtained from the binding data (ie from 10−11 to 10−5M concentrations of AM4113). For comparison purposes, AM251 and SR141716A were assessed using the same methods.
Experiment 2—Reversal of the effects of AM411 on spontaneous locomotion and analgesia
For assessment of locomotion, rats were placed in small activity chambers (28 × 28 × 28 cm) inside soundproof shells. The floor of each chamber consisted of two wire mesh panels (27 × 13 cm) connected through the center by a metal rod, which served as a fulcrum for the floor panels. Locomotion by the subjects produced a slight deflection of one or more floor panels, which closed one or more of four microswitches mounted on the exterior of the chamber. Microswitch closure sent a signal to an external computer running a custom program written in QBasic, by means of an interface (Med Associates). Each microswitch closure was processed as a single activity count. Animals were tested for an 18-min session. The chambers were novel to the subjects at the time of testing to ensure a high baseline of locomotor counts. Immediately following the locomotion session, subjects were moved to an adjoining room. Rats were then tested for analgesia by measuring latency on a tail-flick apparatus (Ugo Basile, Italy). The body of each animal was wrapped lightly in a cloth towel or shirt to prevent spontaneous movement. The exposed tail of the rat was then placed in contact with a combination of heat source and photosensor, which was turned on using an experimenter-operated foot pedal. Any movement of the tail was detected by the photosensor, which then turned off the heat source and stopped the built-in timer. A cutoff of 10 s was set to prevent tissue damage. Animals in experiment 2 were randomly divided into five dose groups: 4 mg/kg AM4113+ 5 mg/kg AM411 (n = 12), 2 mg/kg AM4113 + 5 mg/kg AM411 (n = 12), vehicle + 5 mg/ kg AM411 (n = 12), 4 mg/kg AM4113 + vehicle (n = 13), or vehicle + vehicle (n= 13). In each group, a dose of AM4113 or vehicle was administered i.p. 60 min before testing followed by a dose of AM411 or vehicle (i.p.) 30 min before testing.
Experiments 3—Operant lever pressing on FR1 and FR5 schedules
Rats were tested in operant chambers (internal dimensions: 20 × 21 × 28 cm) for 30 min/day, 5 days/week for the duration of these experiments. One wall housed a single response lever, 4.5 cm wide, which protruded 3 cm from the wall. After magazine training, all rats were trained for 2 weeks on an FR1 schedule in which each response is reinforced with one 45 mg sucrose pellet (Research Diets, New Brunswick, NJ) delivered into a recessed tray on the same wall that housed the lever. Following the 2 weeks of initial training, some of the rats (n = 6) were maintained on the FR1 schedule for several more weeks, while the others (n= 8) were trained for several weeks on an FR5 schedule, receiving a single pellet for every fifth lever press. Following the initial training period, rats were injected with the drug as described above and then tested once a week on Fridays. Operant-conditioning test sessions were controlled by a QBASIC program, which also gathered the data. Rats in experiment 3 received i.p. injections of 2.0, 4.0, and 8.0 mg/kg doses of AM4113. These rats received i.p. drug injections 30 min before the session and their doses were administered in a randomized order using a repeated measures design.
Experiment 4—Effect of AM4113 on consumption of standard chow, high-fat, or high-carbohydrate diets
Animals were assigned to three different diet conditions (n= 10/group). One group was assigned to a high-fat diet (HF; Diet #D12451, Research Diets, New Brunswick, NJ, 20% protein, 45% fat, 35% carbohydrate). A second group was given a high-carbohydrate diet (HC; Diet #D12450B, Research Diets, New Brunswick, NJ, 20% protein, 10% fat, 70% carbohydrate). The remaining group was fed a standard chow diet (LC, 5P00 Prolab RMH 3000, PMI Nutrition International, St Louis, MO; 26% protein, 14% fat, 60% carbohydrate). Food blocks from each type of diet were nutritionally complete and similar in appearance and weight. Rats were given free access to lab chow in their home cages until the beginning of a 5-day habituation period. On the first day of habituation, rats were assigned to their respective dietary groups and moved into suspended wire mesh test cages containing their assigned food type. After spending 30 min in the test cage, they were returned to their home cages. After this initial habituation period, rats were given free access to lab chow in their home cages every Thursday afternoon through Monday afternoon. Each Tuesday and Wednesday, rats spent 30 min in the test cages with their assigned diets. On Thursdays, the injected animals were placed in the test cages with a pre-weighed amount of assigned food. A piece of cardboard was placed underneath the chamber to catch spillage. Following each session, all remaining food plus any spillage was collected and weighed. The difference between pre- and post-session food weights was considered to be the amount of intake. Rats in experiment 4 received i.p. injections of 2.0, 4.0, and 8.0 mg/kg doses of AM4113. These rats received i.p. drug injections 30 min before the test session, and their doses were administered in a randomized order using a repeated measures design.
Experiment 5—Effects of AM4113 on conditioned avoidance, conditioned gaping, and ingestive responses in the taste reactivity paradigm
One week after arrival in the laboratory, rats (n = 32) were implanted with intraoral cannulae. Twenty-four hours before the surgical procedure, they were administered a prophylactic antibiotic (Derapin, 100 mg/kg, s.c; Ayerst). On the day of surgery, the rats were anesthetized with isofluorane gas and were administered Anafen (7.0 mg/kg, s.c; Merial), a nonsteroidal anti-inflammatory drug (NSAID) with analgesic properties. A 3.0 cm2 patch of fur was shaved at the back of the neck just above the scapula and the area was surgically prepared (Betadine surgical scrub (Purdue Frederick) and alcohol). A thin-walled 15-gauge stainless steel needle was inserted at the back of the neck, directed s.c, around the ear and brought out behind the first molar inside the mouth. A length of IntraMedic polyetheylene tubing with an inner diameter of 0.86 mm and an outer diameter of 1.27 mm was then run through the needle after which the needle was removed. Two circular elastic disks were placed over the tubing and drawn to the exposed skin at the back of the neck for the purpose of stabilizing the cannula. The tubing was held secure in the oral cavity by an o-ring, which was sealed behind the tubing before cannulation surgery. For the purposes of conditioning and testing, the cannula was connected to the infusion pump (Harvard Apparatus, South Natick, MA) for delivery of the solution by slipping the tubing of the cannula inside a second polyethylene tube (inner diameter 1.19 mm, outer diameter 1.70 mm) attached to the infusion pump. Two rats were subsequently removed from the study owing to an ineffective cannula.
Five days after surgery and before conditioning (day 1), rats were individually placed in the Plexiglas taste reactivity chamber (22.5 × 26 × 20 cm) with their cannula attached to the infusion pump for fluid delivery. The rats were habituated to the taste reactivity procedure by infusing them with water for a period of 5 min at a rate of 1.0ml/min after which they were returned to their home cage. On day 2, the rats were individually taken to the chamber for a single conditioning trial. They were intraorally infused with 0.1% saccharin solution for 5 min at a rate of 1.0 ml/min, while their orofacial and somatic responses were videotaped from a mirror at a 45° angle below the chamber. Immediately after the saccharin infusion, the rats were injected with the appropriate dose of AM4113, according to random assignment: 0.0 mg/kg (vehicle; n = 8), 2.0 mg/kg (n = 7), 4.0 mg/kg (n = 7), 8.0 mg/kg (n = 8). On day 5, the animals were given a second adaptation trial with a 5-min intraoral infusion of water.
Taste reactivity testing occurred on day 6, 96 h after conditioning. The rats were taken to the chamber and following a period of 1 min were infused with 0.1% saccharin solution over a period of 5 min (1.0ml/min) while being videotaped. Immediately after the session, the rats were returned to their home cage. On day 7, following 15 h of water deprivation, the rats were given a two-bottle preference test. The rats were presented with a graduated tube containing 0.1% saccharin solution and a graduated tube containing water for a period of 120 min. The amount consumed from each bottle during the 120 min of drinking was converted to a preference score: amount consumed of saccharin solution/amount consumed of saccharin + amount consumed of water. The taste reactivity videotapes were scored using the Observer (Noldus Information Technology, Sterling, VA) event-recording program. The behaviors scored included the frequency of gaping, chin rubs, ingestive reactions, passive drips, and activity. Gaping was defined as rapid, large-amplitude opening of the mandible with retraction of the corners of the mouth. Chin rubbings were defined as chin or mouth in direct contact with the floor or wall of the chamber and forward projections of the body. Ingestive (hedonic) reactions were defined as the frequency of 2 s bouts of tongue protrusions (extensions of the tongue out of the mouth) and mouth movements (movement of the lower mandible without opening the mouth). The scores for tongue protrusions and mouth movements were summed to provide a total ingestive (hedonic) reaction score.
Statistical Analyses
Statistical analysis was performed using SYSTAT 7.0. Analysis of variance (ANOVA) with repeated measures on the dose variable was used to analyze data from experiments 1 and 2. Experiment 3 utilized a diet × drug factorial ANOVA with repeated measures on the dose factor. Experiment 4 data were analyzed using between-subjects ANOVA comparing the five treatment groups. Nonorthogonal planned comparisons (Keppel, 1982) were used to compare each drug treatment with vehicle, and in experiment 4 to compare combination AM411 + AM4113 treatments with AM411 + vehicle. The overall ANOVA error term was used in these calculations, and the number of comparisons was restricted to the number of drug conditions minus one. ED50 and 95% confidence intervals for the drug effect on FR1 and FR5 schedules was estimated using curvilinear regression analysis (GraphPad Prism), employing an exponential decay function.
RESULTS
Experiment 1—Cannabinoid Receptor Binding and cAMP Assays
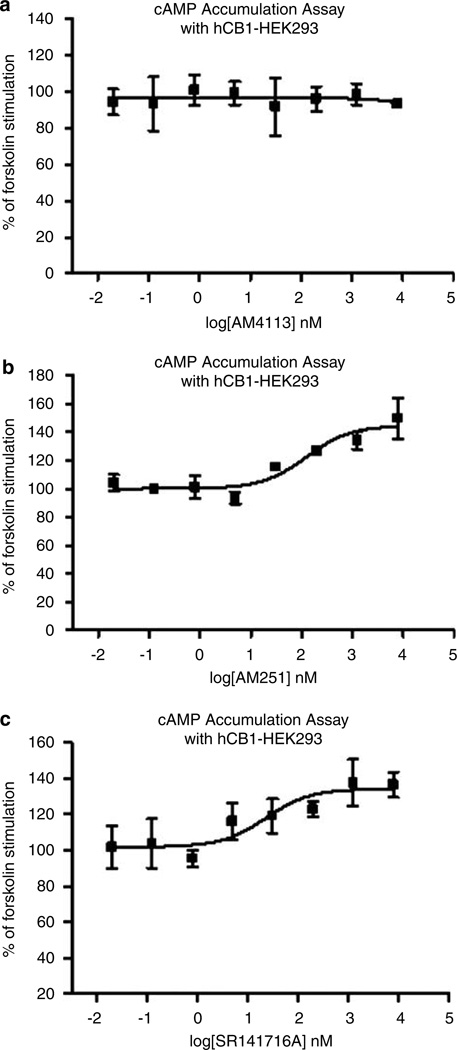
As shown in Table 1, AM4113 was able to bind with high affinity to CB1 receptors, exhibiting 100-fold selectivity for CB1 vs CB2 receptors. AM251 and SR141716A also showed binding selectivity for CB1 receptors relative to CB2 receptors. AM4113 did not change the forskolin-stimulated cAMP accumulation in CB1-transfected HEK cells up to concentrations of 10 µM (Figure 1a), which indicates that this drug is acting as a CB1 neutral antagonist rather than an inverse agonist. In contrast, AM251 and SR141716A both produced substantial increases in cAMP accumulation (ie 30–40% increases; Figure 1b and c).
Table 1.
CB1 and CB2 Binding Data for AM4113, AM251, and SR141716A
| Assay | AM4113 | AM251 | SR141716A |
|---|---|---|---|
| CB1 binding Ki | 0.89±0.44 | 3.43±1.5 | 10.00±0.79 |
| 95% confidence intervals | (0.64, 1.2) | (2.67, 4.45) | (7.15, 14.08) |
| r-Value | 0.936 | 0.953 | 0.944 |
| hCB2 binding Ki | 92±6.9 | 124±10 | 931 |
| 95% confidence intervals | (54, 160) | (94, 1622) | (671, 1291) |
| r-Value | 0.931 | 0.964 | 0.956 |
Values for Ki are in nM±SD of 2–7 assays performed in duplicate (shown with 95% confidence intervals and r2-value).
Figure 1.
Forskolin-stimulated cAMP formation as a function of concentration of cannabinoid drug. (a) AM4113, (b) AM251, (c) SR141716A. These results with the cAMP assay demonstrate that AM4113 is a neutral antagonist for CB1, while AM251 and SR141716A act as inverse agonists. Each graph represents one assay peformed in triplicate. AM215: baseline 95% confidence intervals, 92.7–107%; maximal increase 95% confidence intervals, 133–157%. SR141716A: baseline 95% confidence intervals, 98.8–113%; maximal increase 95% confidence intervals, 122–147%.
Experiment 2—Reversal of the Effects of AM411 on Spontaneous Locomotion and Analgesia
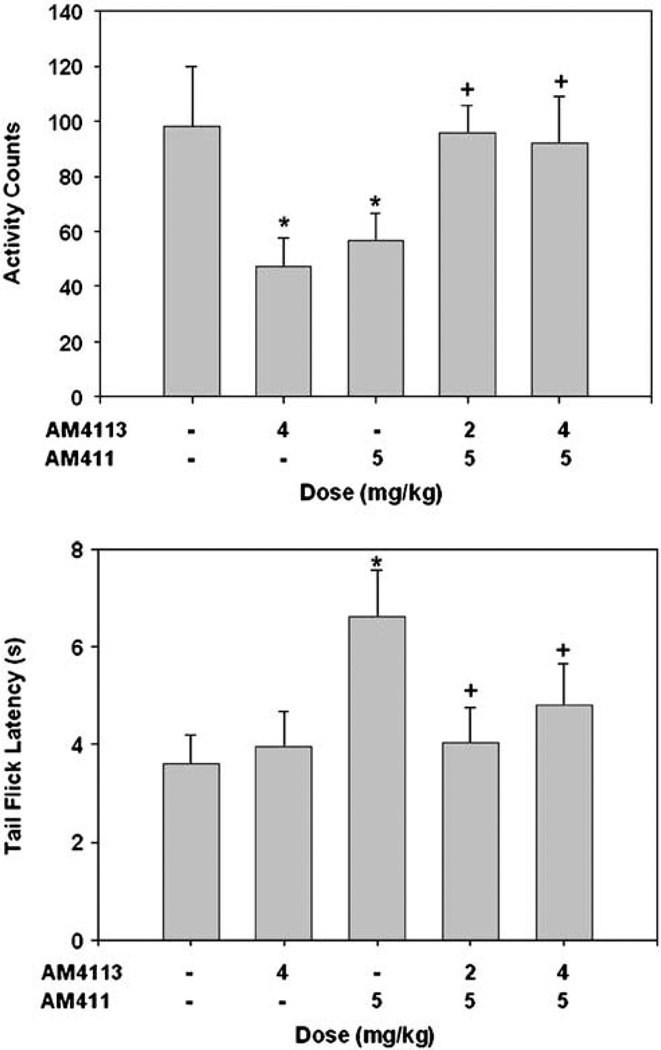
Figure 2 shows results from the tests of spontaneous locomotion and analgesia. ANOVA revealed significant overall treatment group effects for locomotion (F(4, 57) = 2.86, p<0.05) and analgesia (F(4,54) = 2.51, p<0.05). Planned comparisons showed that 5.0 mg/kg of the cannabinoid agonist AM411 significantly suppressed spontaneous locomotion and increased analgesia (p<0.01) relative to the combined vehicle treatment. At both 2.0 and 4.0 mg/kg doses, the antagonist AM4113 attenuated the AM411-induced locomotor suppression (p<0.01 at 2 mg/ kg; p<0.05 at 4 mg/kg) and analgesia (p<0.01 at 2 mg/kg; p<0.05 at 4 mg/kg). In addition, 4.0 mg/kg AM4113 alone significantly suppressed locomotor activity compared to vehicle (p<0.01).
Figure 2.
Mean ( + SEM) effects of cannabinoid agonist AM411 and antagonist AM4113 on tetrad tests of spontaneous locomotion (activity counts; top) and analgesia (tail flick latency; bottom). AM4113 significantly suppressed spontaneous locomotion and attenuated the effects of AM411 on measures of locomotion and analgesia. *Significantly different from vehicle to vehicle at p<0.05; +Significantly different from AM411 plus vehicle at p<0.05.
Experiment 3—Operant Lever Pressing on FR1 and FR5 Schedules
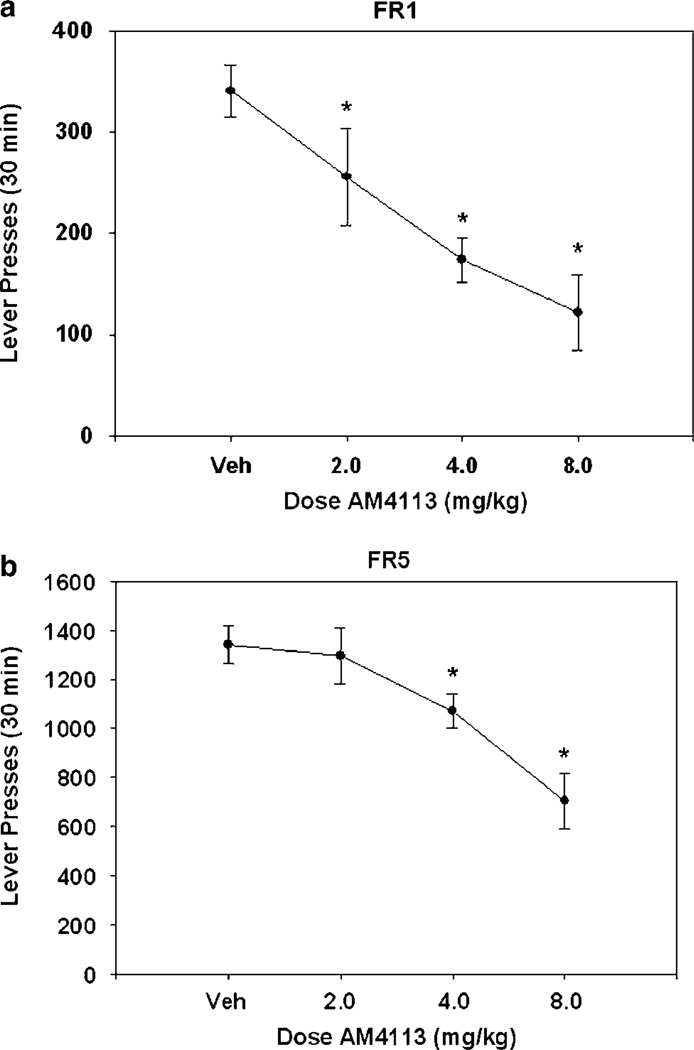
AM4113 decreased lever pressing on an FR1 schedule in a dose-dependent manner (F(3, 15) = 12.0, p<0.001; Figure 3a). Nonorthogonal, planned comparisons showed that every dose of AM4113 significantly decreased lever-pressing response over vehicle (p<0.05). The ED50 for the suppressing effect of AM4113 on FR1 responding was 4.78 mg/kg (r2 = 0.52). AM4113 also produced a dose-dependent decrease in responses on the FR5 schedule (F(3, 21) = 9.4, p<0.001; Figure 3b). Nonorthogonal planned comparisons showed that AM4113 significantly decreased lever-pressing response over vehicle for doses of 4 and 8 mg/kg (p<0.05). The FR5 schedule generated a substantially higher response rate than the FR1 schedule. The ED50 for the effect of AM4113 on FR5 responding was 10.28 mg/kg (r2 = 0.45).
Figure 3.
Mean (±SEM) number of lever presses after injection of vehicle or various doses of AM4113 in experiment 3. (a) Lever pressing on FR1 reinforcement schedule. (b) Lever pressing on FR5 reinforcement schedule. *Significantly different from vehicle (p<0.05) as measured by planned comparisons.
Experiment 4—Effect of AM4113 on Consumption of Standard Chow, High-Fat, or High-Carbohydrate Diets
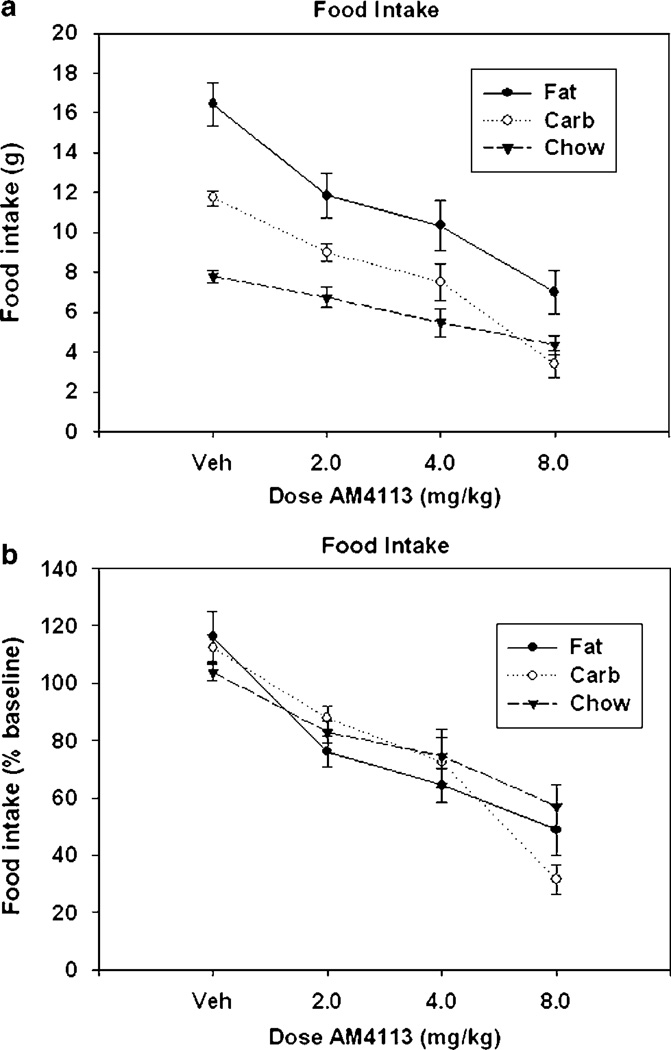
Figure 4a depicts the effects of AM4113 on food consumption. AM4113 significantly suppressed food intake over vehicle across all diet groups (F(3,81) = 37.3, p<0.001). Nonorthogonal, planned comparisons show that every dose of AM4113 significantly decreased consumption over vehicle. There was a significant effect of diet (F(2, 27) = 49.82, p<0.001), and there was a significant interaction between diet group and dose (F(6, 81) = 3.44, p<0.005). Separate analyses showed that intake of each of the three food types was significantly suppressed by AM4113 (p<0.01). Moreover, the interaction effect disappeared when data were transformed to represent a percentage of baseline consumption, defined as the mean consumption of the previous two noninjection sessions (F(6, 81)=1.95, NS; see Figure 4b). There were no significant differences among the three dietary groups in the percent transformed data (F(2, 27) = 0.18, NS), but a robust drug treatment effect remained (F(3, 81) = 54.08, p<0.001).
Figure 4.
Effect of AM4113 on intake of three different diets. (a) Mean (±SEM) raw intake (expressed in grams) of three different diets during 30 min sessions. For all diet groups, every dose was significantly different from vehicle (p<0.05) as measured by planned comparisons. There was also a significant dose–diet interaction. (b) Mean intake expressed as percent of baseline consumption (defined as the mean consumption of the previous two non-injection sessions) of the three different diets during 30-min sessions. There was no significant dose–group interaction; however, a strong dose effect remained.
Experiment 5—Effects of AM4113 on Conditioned Avoidance, Conditioned Gaping, and Ingestive Responses in the Taste Reactivity Paradigm
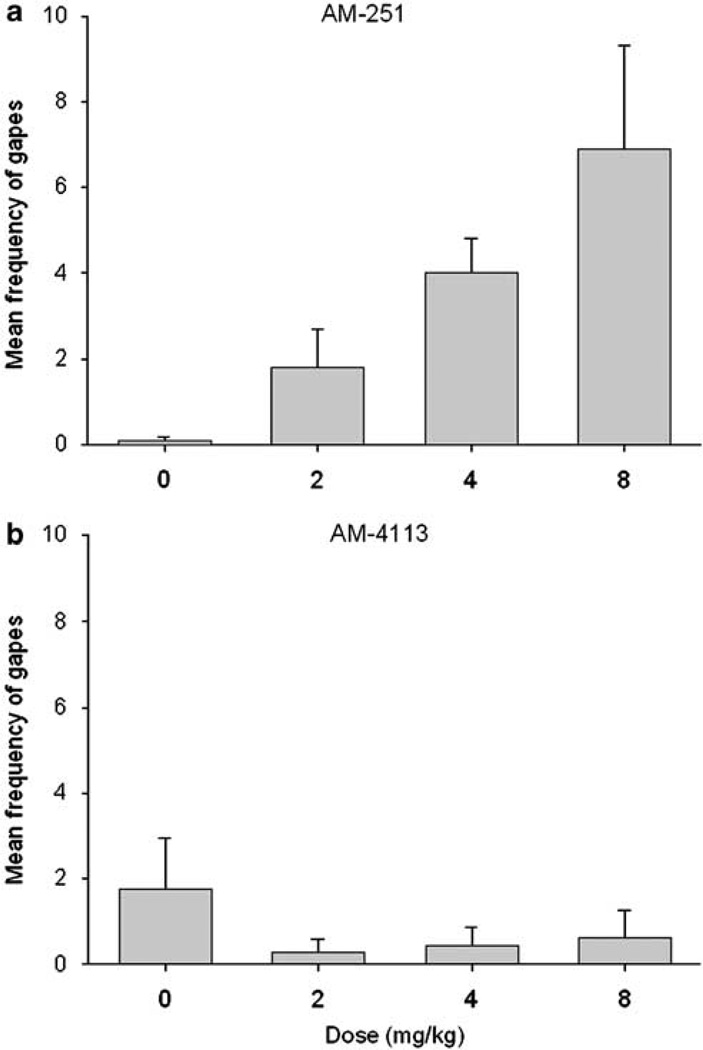
AM4113 produced dose-dependent conditioned taste avoidance and suppression of ingestive (hedonic) taste reactivity scores, but it did not produce conditioned aversion as assessed by gaping and chin rubbing. Mean (±SEM) saccharin preference ratios were as follows: saline, 0.388 (±0.069); 2.0 mg/kg AM4113 0.411 (±0.059); 4.0 mg/kg AM4113 0.298 (±0.074); 8.0 mg/kg AM4113 0.088 (±0.017). A single-factor ANOVA of the saccharin preference ratio scores revealed a significant effect of dose (F(3, 26) = 6.9; p<0.001); subsequent LSD pairwise comparisons revealed that at a dose of 8.0 mg/kg, AM4113 produced conditioned avoidance of saccharin solution that differed significantly from all other groups (p<0.025). AM4113 also produced conditioned suppression of ingestion (hedonic) taste reactions (mean±SEM for each condition were as follows: saline, 9.0 (±3.2); 2.0 mg/kg AM4113, 10.5 (±3.2); 4.0 mg/ kg AM4113, 4.5 (±1.5); 8.0 mg/kg AM4113, 1.3 (±0.5); F(3, 26) = 3.2, p<0.05). Subsequent LSD pairwise comparison tests revealed that a dose of 8.0 mg/kg of AM4113 produced significantly suppressed ingestion reactions relative to vehicle or 2.0 mg/kg of AM4113 (p<0.05), but not 4.0 mg/kg of AM4113. Despite these effects of AM4113, there were no significant effects of AM4113 on chin rubbing or conditioned gaping. Figure 5 depicts the results with the conditioned gaping measure (Figure 5a shows the effects of AM251 from McLaughlin et al (2005b), while Figure 5b displays the present results with AM4113). Although AM251 has been shown to increase conditioned gaping, this effect was not seen with AM4113.
Figure 5.
Effects of AM251 and AM4113 on performance of conditioned gaping in the taste reactivity procedure (ie number of gaping responses (mean (±SEM)). (a) AM251 results from McLaughlin et al (2005b) (F(3, 36) = 4.6; p<0.01). (b) AM4113 results from the present study. *Significantly different from vehicle (p<0.05) as measured by planned comparisons.
DISCUSSION
The biochemical experiments indicated that AM4113 has the characteristics of a neutral antagonist of the cannabinoid CB1 receptor. AM4113 was shown to bind with high affinity to CB1 receptors, and showed 100-fold selectivity for CB1 over CB2 receptors. Previously published results from our laboratory have demonstrated that SR141716, AM1387, and AM251 are 143, 48, and 430 times more selective for CB1 than CB2, respectively (Lan et al, 1999; McLaughlin et al, 2006). Thus, AM4113 showed a level of CB1 selectivity that was within the range of these other CB1 ligands. However, AM4113 also showed characteristics of being a neutral antagonist in the cAMP assay, as there were no changes in forskolin-stimulated cAMP levels up to a concentration of 10 µM AM4113. These findings with AM4113 stand in marked contrast to other studies showing that SR141716, AM251, and AM1387 all increase cAMP production (Mato et al, 2002; McLaughlin et al, 2006). In view of the fact that CB1 receptor stimulation with agonists normally inhibits cAMP formation, the ability of drugs to increase cAMP levels is indicative of their ability to act as inverse agonists. Using the same methods as those employed in the present study, the CB1 inverse agonists AM251 and AM1387 were shown to increase forskolin-stimulated cAMP production by 77–96% in the concentration range that was 2 log units higher than the Ki for those drugs (McLaughlin et al, 2006). Thus, the present binding and cAMP data indicate that AM4113, in contrast to AM251 and AM1387, appears to be acting as a neutral antagonist of the CB1 receptor. AM4113 also was able to produce several behavioral effects. In tests of spontaneous locomotion and analgesia, AM4113 reversed the effects of the CB1 agonist AM411. In addition, AM4113 produced significant suppression of spontaneous locomotion when administered alone. AM4113 suppressed food-reinforced operant responding on FR1 and FR5 schedules of reinforcement in a dose-dependent manner. AM4113 also dose-dependently suppressed feeding on high-fat, high-carbohydrate, and laboratory chow diets. While food-intake suppression was greatest among those animals that are fed high-fat diets and least in rats given laboratory chow, these differences disappeared when data were transformed to account for differences in baseline intake. Taken together, these results suggest that feeding can be suppressed by antagonism of an endogenous cannabinoid tone, and that inverse agonism is not necessary for reductions of food intake to occur. Moreover, AM4113 did not induce conditioned gaping, which suggests that this drug did not suppress feeding because of the induction of nausea or malaise.
Tests of spontaneous locomotion and analgesia were used to assess the ability of AM4113 to attenuate the behavioral effects of the cannabinoid CB1 agonist AM411 (McLaughlin et al, 2005a). Taken together, these results showed that AM4113 blocks some of the behavioral effects that are characteristic of CB1 receptor stimulation. AM4113 also produced a significant suppression of spontaneous locomotion when administered alone. This observation is consistent with effects that were previously reported for O-2050 (Gardner and Mallet, 2006), as well as for the antagonist/ inverse agonists AM251 (McLaughlin et al, 2005a) and SR141716A (Ja¨rbe et al, 2002). The ability of a CB1 antagonist to inhibit motor activity may seem paradoxical, considering that CB1 agonists generally reduce motor activity (McLaughlin et al, 2005b). Possible mechanisms for the CB1 antagonist-induced suppression of locomotion are unclear at this time. Nevertheless, the fact that combined administration of AM4113 and AM411 resulted in levels of activity that did not differ from vehicle shows that the combined drug effects upon locomotion were not additive; this suggests that CB1 antagonists are probably not suppressing locomotion by the same mechanism as CB1 agonists. Moreover, these data indicate that a moderate level of CB1 tone is necessary for normal locomotion, and that either overstimulation or blockade of CB1 receptors can reduce locomotion.
The FR1 and FR5 schedules of reinforcement were chosen for experiment 3 because previous reports have shown that substances acting on CB1 receptors produce reliable, dose-dependent effects on the performance of these schedules (Arizzi et al, 2004; McLaughlin et al, 2003, 2005a, 2006). Moreover, research on the effects of other manipulations (eg nucleus accumbens dopamine depletions, dopamine antagonists) has indicated that the ratio requirement of a schedule can be a critical determinant of the effects of various neurochemical or pharmacological manipulations (Aberman and Salamone, 1999; Ishiwari et al, 2004). In the present study, AM4113 decreased responding on the FR1 schedule with an ED50 of 4.78 mg/kg; however, the potency of AM4113 for suppression of FR5 responding was somewhat less (ie 10.28 mg/kg). This result was somewhat of a surprise, as it was thought that a neutral antagonist blocking endogenous tone would be equipotent across both ratio schedules, similar to the effects of prefeeding (Aberman and Salamone, 1999). Furthermore, this result was quite different from previous studies involving dopaminergic manipulations, which have demonstrated that schedules with higher ratio requirements are much more sensitive to the effects of interference with DA transmission (Aberman and Salamone, 1999; Ishiwari et al, 2004). Interestingly, CB1 agonists also produce decreases in FR5 responding; however, it is thought that the mechanisms causing these response reductions are quite different from those of CB1 antagonists. CB1 antagonists are thought to reduce lever pressing for food by appetite suppression or production of food aversions. On the other hand, CB1 agonists produce catalepsy and ataxia at doses shown to decrease operant responding (Carriero et al, 1998), effects that are not typically observed with CB1 antagonists or inverse agonists. It is possible that the suppression of locomotion induced by AM4113 may be related, at least in part, to the reductions in lever pressing induced by this drug. However, it is unlikely that a decrease in locomotion is related to the suppression of food intake that was observed in experiment 4, because several studies have shown that conditions that decrease locomotion do not necessarily decrease food intake (eg nucleus accumbens DA depletions; Salamone et al, 1993; see review by Salamone and Correa, 2002). More reliable indicators of motor impairments related to food intake include feeding rate and food handling (Salamone et al, 1993); in this regard, it is important to emphasize that a recent study has demonstrated that the CB1 inverse agonist AM251 did not affect these measures in doses that also suppressed feeding (McLaughlin et al, 2005b). In view of the results indicating that food intake was suppressed by AM4113 in the same dose range as food reinforced lever pressing, it is reasonable to suggest that AM4113 is suppressing lever pressing because of actions related to food motivation.
AM4113 suppressed consumption of all three diets, and there was a diet-dose interaction when the raw intake data were analyzed. In terms of raw quantities of food intake, animals consuming the high-fat diet exhibited the greatest suppression of intake, followed by rats on the high-carbohydrate diet. Animals that consumed lab chow showed the lowest suppression of intake with AM4113. Considering that some studies have shown SR141716-induced feeding suppression to be stronger with more palatable diets than less palatable diets (Arnone et al, 1997; Simiand et al, 1998), these results could be interpreted as suggesting a preferential effect on highly palatable foods. However, when intake data were expressed as a percentage of baseline consumption, these differences between diet groups disappeared. It must also be emphasized that there was indeed a significant overall suppression of lab chow intake, consistent with previous studies showing effects of antagonist/inverse agonists on lab chow consumption (Colombo et al, 1998; Gómez et al, 2002; McLaughlin et al, 2003, 2006; Verty et al, 2004). Taken together, these results suggest that AM4113 is not preferentially suppressing feeding of highly palatable diets, but that apparent interactions with diet type or palatability may be owing to differences in baseline consumption and/or scaling. Nevertheless, in considering the potential of this drug as an anti-obesity treatment, the greater feeding suppression in absolute gram quantities observed in those rats consuming calorically dense foods at high baseline rates suggests that CB1 antagonists may reduce caloric intake substantially in patients with the highest baseline levels of food intake, or those who consume more high-calorie foods.
While the feeding data from AM4113 compares closely with those of CB1 antagonist/inverse agonist drugs, it is believed that there may be some differences in the mechanisms by which feeding is decreased. McLaughlin et al (2005b) suggested that the CB1 inverse agonist AM251 may be reducing food intake, at least in part, by inducing nausea and malaise. Using methods that are similar to those used in the present study, McLaughlin et al (2005b) reported that AM251 administration resulted in a significant induction of conditioned gaping, which is thought to be a marker of nausea or malaise in rats (Parker et al, 1998). However, in the present study, AM4113 failed to induce conditioned gaping. Previous clinical work has shown that nausea was one of the most common adverse effects reported in clinical trials with antagonist/inverse agonist SR141716 (Pi-Sunyer et al, 2006; Van Gaal et al, 2005). Considering also the known anti-emetic properties of CB1 agonists, it seems likely that the appetite suppressant effects of SR141716 and AM251 are due at least in part to nausea and malaise induced by their inverse agonist properties. Side effects such as nausea could prove to be problematic for these drugs as anti-obesity treatments, since compliance could become a major issue. On the other hand, AM4113 is a neutral antagonist that may be affecting feeding by blocking endogenous cannabinoid tone instead of stimulating signal transduction effects opposite to those of CB1 agonists. Therefore, it is tempting to suggest that AM4113 has the potential to reduce appetite and food intake without causing nausea, and additional research will be necessary to confirm this hypothesis. Recent data suggest that AM4113, unlike the inverse agonist AM251, does not induce vomiting in ferrets (Chambers et al, 2006).
In summary, AM4113 is a novel neutral cannabinoid CB1 antagonist that can block some of the behavioral effects of the cannabinoid agonist AM411. AM4113 reduced food-reinforced behaviors and suppressed food intake of all diets irrespective of macronutrient composition or palatability at doses comparable to the CB1 antagonist/inverse agonist drugs AM251 and SR141716. Doses of AM4113 that suppressed foodintake and food-reinforced responding, and also decreased ingestive responses and increased food avoidance, nevertheless failed to induce conditioned gaping. These data suggest that neutral CB1 antagonists such as AM4113 may be useful for the suppression of appetite.
ACKNOWLEDGEMENTS
This work was supported by grants to JS and AM from NIH/NIDA. We thank Evan McClure for his technical assistance.
Footnotes
DISCLOSURE/CONFLICT OF INTEREST
The author(s) declare that, except for income received from my primary employer, no financial support or compensation has been received from any individual or corporate entity over the past 3 years for research or professional service and there are no personal financial holdings that could be perceived as constituting a potential conflict of interest.
REFERENCES
- Aberman JE, Salamone JD. Nucleus accumbens dopamine depletions make rats more sensitive to high ratio requirements but do not impair primary food reinforcement. Neuroscience. 1999;92:545–552. doi: 10.1016/s0306-4522(99)00004-4. [DOI] [PubMed] [Google Scholar]
- Arizzi MN, Cervone KM, Aberman JE, Betz A, Liu Q, Lin S, et al. Behavioral effects of inhibition of cannabinoid metabolism: the amidase inhibitor AM374 enhances the suppression of lever pressing produced by exogenously administered anandamide. Life Sci. 2004;74:1001–1011. doi: 10.1016/j.lfs.2003.07.024. [DOI] [PubMed] [Google Scholar]
- Arnone M, Maruani J, Chaperon F, Thiebot MH, Poncelet M, Soubrie P, et al. Selective inhibition of sucrose and ethanol intake by SR141716, an antagonist of central cannabinoid (CB1) receptors. Psychopharmacology (Berl) 1997;132:104–106. doi: 10.1007/s002130050326. [DOI] [PubMed] [Google Scholar]
- Carriero D, Aberman J, Lin SY, Hill A, Makriyannis A, Salamone JD. A detailed characterization of the effects of four cannabinoid agonists on operant lever pressing. Psychopharmacology. 1998;137:147–156. doi: 10.1007/s002130050604. [DOI] [PubMed] [Google Scholar]
- Chambers AP, Vemuri K, Pittman QJ, Makriyannis A, Sharkey KA. Antagonist vs Inverse Agonist Activity at CB 1 Receptors: Effects on Food Intake and Emesis; Atlanta, GA. Program Number 457.15. Meeting Planner. Society for Neuroscience; 2006. (online) [Google Scholar]
- Chen RZ, Huang RR, Shen CP, MacNeil DJ, Fong TM. Synergistic effects of cannabinoid inverse agonist am251 and opioid antagonist nalmefene on food intake in mice. Brain Res. 2004;999:227–230. doi: 10.1016/j.brainres.2003.12.004. [DOI] [PubMed] [Google Scholar]
- Cheng Y, Prusoff WH. Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 percent inhibition (I50) of an enzymatic reaction. Biochem Pharmacol. 1973;22:3099–3108. doi: 10.1016/0006-2952(73)90196-2. [DOI] [PubMed] [Google Scholar]
- Colombo G, Agabio R, Diaz G, Lobina C, Reali R, Gessa GL. Appetite suppression and weight loss after the cannabinoid antagonist SR141716. Life Sci. 1998;63:PL113–PL117. doi: 10.1016/s0024-3205(98)00322-1. [DOI] [PubMed] [Google Scholar]
- Darmani NA. Delta(9)-tetrahydrocannabinol and synthetic cannabinoids prevent emesis produced by the cannabinoid CB(1) receptor antagonist/inverse agonist SR 141716A. Neuropsychopharmacology. 2001;24:198–203. doi: 10.1016/S0893-133X(00)00197-4. [DOI] [PubMed] [Google Scholar]
- Darmani NA, Johnson JC. Central and peripheral mechanisms contribute to the antiemetic actions of delta-9-tetrahydrocannabinol against 5-hydroxytryptophan-induced emesis. Eur J Pharmacol. 2004;488:201–212. doi: 10.1016/j.ejphar.2004.02.018. [DOI] [PubMed] [Google Scholar]
- De Vry J, Schreiber R, Eckel G, Jentzsch KR. Behavioral mechanisms underlying inhibition of food-maintained responding by the cannabinoid receptor antagonist/inverse agonist SR141716A. Eur J Pharmacol. 2004;483:55–63. doi: 10.1016/j.ejphar.2003.10.012. [DOI] [PubMed] [Google Scholar]
- Despres JP, Golay A, Sjostrom L. Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med. 2005;353:2121–2134. doi: 10.1056/NEJMoa044537. [DOI] [PubMed] [Google Scholar]
- Gardner A, Mallet PE. Suppression of feeding, drinking, and locomotion by a putative cannabinoid receptor ‘silent antagonist’. Eur J Pharmacol. 2006;530:103–106. doi: 10.1016/j.ejphar.2005.11.032. [DOI] [PubMed] [Google Scholar]
- Gómez R, Navarro M, Ferrer B, Trigo JM, Bilbao A, Del Arco I, et al. A peripheral mechanism for CB1 cannabinoid receptor-dependent modulation of feeding. J Neurosci. 2002;22:9612–9617. doi: 10.1523/JNEUROSCI.22-21-09612.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Rosales F, Walsh D. Intractable nausea and vomiting due to gastrointestinal mucosal metastases relieved by tetrahydrocannabinol (Dronabinol) J Pain Symptom Manage. 1997;14:311–314. doi: 10.1016/S0885-3924(97)00229-7. [DOI] [PubMed] [Google Scholar]
- Grill HJ, Norgren R. The taste reactivity test. I. Mimetic responses to gustatory stimuli in neurologically normal rats. Brain Res. 1978;143:263–279. doi: 10.1016/0006-8993(78)90568-1. [DOI] [PubMed] [Google Scholar]
- Ishiwari K, Weber SM, Mingote S, Correa M, Salamone JD. Accumbens dopamine and the regulation of effort in food-seeking behavior: modulation of work output by different ratio or force requirements. Behav Brain Res. 2004;151:83–91. doi: 10.1016/j.bbr.2003.08.007. [DOI] [PubMed] [Google Scholar]
- Jamshidi N, Taylor DA. Anandamide administration into the ventromedial hypothalamus stimulates appetite in rats. Br J Pharmacol. 2001;134:1151–1154. doi: 10.1038/sj.bjp.0704379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ja¨rbe TU, Andrzejewski ME, DiPatrizio NV. Interactions between the CB1 receptor agonist Delta 9-THC and the CB1 receptor antagonist SR-141716 in rats: open-field revisited. Pharmacol Biochem Behav. 2002;73:911–919. doi: 10.1016/s0091-3057(02)00938-3. [DOI] [PubMed] [Google Scholar]
- Keppel G. Design and Analysis: A Researcher’s Handbook. Englewood Cliffs: Prentice-Hall; 1982. [Google Scholar]
- Kirkham TC, Williams CM, Fezza F, Di Marzo V. Endocannabinoid levels in rat limbic forebrain and hypothalamus in relation to fasting, feeding and satiation: stimulation of eating by 2-arachidonoyl glycerol. Br J Pharmacol. 2002;136:550–557. doi: 10.1038/sj.bjp.0704767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lan R, Liu Q, Fan P, Lin S, Fernando SR, McCallion D, et al. Structure-activity relationships of pyrazole derivatives as cannabinoid receptor antagonists. J Med Chem. 1999;42:769–776. doi: 10.1021/jm980363y. [DOI] [PubMed] [Google Scholar]
- Landsman RS, Burkey TH, Consroe P, Roeske WR, Yamamura HI. SR141716A is an inverse agonist at the human cannabinoid CB1 receptor. Eur J Pharmacol. 1997;334:R1–R2. doi: 10.1016/s0014-2999(97)01160-6. [DOI] [PubMed] [Google Scholar]
- Limebeer CL, Parker LA. The antiemetic drug ondansetron interferes with lithium-induced conditioned rejection reactions, but not lithium-induced taste avoidance in rats. J Exp Psychol Anim Behav Process. 2000;26:371–384. doi: 10.1037//0097-7403.26.4.371. [DOI] [PubMed] [Google Scholar]
- Limebeer CL, Parker LA. The 5-HT1A agonist 8-OH-DPAT dose-dependently interferes with the establishment and the expression of lithium-induced conditioned rejection reactions in rats. Psychopharmacology. 2003;166:120–126. doi: 10.1007/s00213-002-1309-6. [DOI] [PubMed] [Google Scholar]
- Limebeer CL, Parker LA, Fletcher PJ. 5,7-dihydroxytryptamine lesions of the dorsal and median raphe nuclei interfere with lithium-induced conditioned gaping, but not conditioned taste avoidance, in rats. Behav Neurosci. 2004;118:1391–1399. doi: 10.1037/0735-7044.118.6.1391. [DOI] [PubMed] [Google Scholar]
- Makriyannis A, Guo J, Tian X. Albumin enhances the diffusion of lipophilic drugs into the membrane bilayer. Life Sci. 2005;77:1605–1611. doi: 10.1016/j.lfs.2005.05.008. [DOI] [PubMed] [Google Scholar]
- Martin BR, Compton DR, Thomas BF, Prescott WR, Little PJ, Razdan RK, et al. Behavioral, biochemical, and molecular modeling evaluations of cannabinoid analogs. Pharmacol Biochem Behav. 1991;40:471–478. doi: 10.1016/0091-3057(91)90349-7. [DOI] [PubMed] [Google Scholar]
- Mato S, Pazos A, Valdizan EM. Cannabinoid receptor antagonism and inverse agonism in response to SR141716A on cAMP production in human and rat brain. Eur J Pharmacol. 2002;443:43–46. doi: 10.1016/s0014-2999(02)01575-3. [DOI] [PubMed] [Google Scholar]
- McLaughlin PJ, Lu D, Winston KM, Thakur G, Swezey LA, Makriyannis A, et al. Behavioral effects of the novel cannabinoid full agonist AM411. Pharmacol Biochem Behav. 2005a;81:78–88. doi: 10.1016/j.pbb.2005.02.005. [DOI] [PubMed] [Google Scholar]
- McLaughlin PJ, Qian L, Wood JT, Wisniecki A, Winston KM, Swezey LA, et al. Suppression of food intake and food-reinforced behavior produced by the novel CB1 receptor antagonist/inverse agonist AM1387. Pharmacol Biochem Behav. 2006;83:396–402. doi: 10.1016/j.pbb.2006.02.022. [DOI] [PubMed] [Google Scholar]
- McLaughlin PJ, Winston K, Swezey L, Wisniecki A, Aberman J, Tardif DJ, et al. The cannabinoid CB1 antagonists SR141716A and AM251 suppress food intake and food-reinforced behavior in a variety of tasks in rats. Behav Pharmacol. 2003;14:583–588. doi: 10.1097/00008877-200312000-00002. [DOI] [PubMed] [Google Scholar]
- McLaughlin PJ, Winston KM, Limebeer CL, Parker LA, Makriyannis A, Salamone JD. The cannabinoid CB1 antagonist AM251 produces food avoidance and behaviors associated with nausea but does not impair feeding efficiency in rats. Psychopharmacology (Berl) 2005b;180:286–293. doi: 10.1007/s00213-005-2171-0. [DOI] [PubMed] [Google Scholar]
- Morse KL, Fournier DJ, Li X, Grzybowska J, Makriyannis A. A novel electrophilic high affinity irreversible probe for the cannabinoid receptor. Life Sci. 1995;56:1957–1962. doi: 10.1016/0024-3205(95)00176-7. [DOI] [PubMed] [Google Scholar]
- Parker LA, Limebeer CL. Conditioned gaping in rats: a selective measure of nausea. Auton Neurosci. 2006;129:36–41. doi: 10.1016/j.autneu.2006.07.022. [DOI] [PubMed] [Google Scholar]
- Parker LA, Limebeer CL, Simpson GR. Chlordiazepoxide-induced conditioned place and taste aversion learning in rats. Pharmacol Biochem Behav. 1998;59:33–37. doi: 10.1016/s0091-3057(97)00333-x. [DOI] [PubMed] [Google Scholar]
- Parker LA, Mechoulam R, Schlievert C, Abbott L, Fudge ML, Burton P. Effects of cannabinoids on lithium-induced conditioned rejection reactions in a rat model of nausea. Psychopharmacology (Berl) 2003;166:156–162. doi: 10.1007/s00213-002-1329-2. [DOI] [PubMed] [Google Scholar]
- Pi-Sunyer FX, Aronne LJ, Heshmati HM, Devin J, Rosenstock J RIO-North America Study Group. Effect of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients. RIO-North America: a randomized controlled trial. JAMA. 2006;295:761–775. doi: 10.1001/jama.295.7.761. [DOI] [PubMed] [Google Scholar]
- Salamone JD, Correa M. Motivational views of reinforcement: implications for understanding the behavioral functions of nucleus accumbens dopamine. Behav Brain Res. 2002;137:3–25. doi: 10.1016/s0166-4328(02)00282-6. [DOI] [PubMed] [Google Scholar]
- Salamone JD, Mahan K, Rogers S. Ventrolateral striatal dopamine depletions impair feeding and food handling in rats. Pharmacol Biochem Behav. 1993;44:605–610. doi: 10.1016/0091-3057(93)90174-r. [DOI] [PubMed] [Google Scholar]
- Shearman LP, Rosko KM, Fleischer R, Wang J, Xu S, Tong XS, et al. Antidepressant-like and anorectic effects of the cannabinoid CB1 receptor inverse agonist AM251 in mice. Behav Pharmacol. 2003;14:573–582. doi: 10.1097/00008877-200312000-00001. [DOI] [PubMed] [Google Scholar]
- Simiand J, Keane M, Keane PE, Soubrie P. SR141716, a CB1 cannabinoid receptor antagonist, selectively reduces sweet food intake in marmoset. Behav Pharmacol. 1998;9:179–181. [PubMed] [Google Scholar]
- Simoneau II, Hamza MS, Mata HP, Siegel EM, Vanderah TW, Porreca F, et al. The cannabinoid agonist WIN55,212-2 suppresses opioid-induced emesis in ferrets. Anesthesiology. 2001;94:882–887. doi: 10.1097/00000542-200105000-00029. [DOI] [PubMed] [Google Scholar]
- Van Gaal LF, Rissanen AM, Scheen AJ, Ziegler O, Rossner S RIO Europe Study Group. Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe study. Lancet. 2005;365:1389–1397. doi: 10.1016/S0140-6736(05)66374-X. [DOI] [PubMed] [Google Scholar]
- Van Sickle MD, Oland LD, Mackie K, Davison JS, Sharkey KA. Delta9-tetrahydrocannabinol selectively acts on CB1 receptors in specific regions of dorsal vagal complex to inhibit emesis in ferrets. Am J Physiol Gastrointest Liver Physiol. 2003;285:G566–G576. doi: 10.1152/ajpgi.00113.2003. [DOI] [PubMed] [Google Scholar]
- Verty AN, McGregor IS, Mallet PE. Consumption of high carbohydrate, high fat, and normal chow is equally suppressed by a cannabinoid receptor antagonist in non-deprived rats. Neurosci Lett. 2004;354:217–220. doi: 10.1016/j.neulet.2003.10.035. [DOI] [PubMed] [Google Scholar]
- Ward SJ, Dykstra LA. The role of CB1 receptors in sweet versus fat reinforcement: effect of CB1 receptor deletion, CB1 receptor antagonism (SR141716A) and CB1 receptor agonism (CP-55940) Behav Pharmacol. 2005;16:381–388. doi: 10.1097/00008877-200509000-00010. [DOI] [PubMed] [Google Scholar]
- Wiley JL, Burston JJ, Leggett DC, Alekseeva OO, Razdan RK, Mahadevan A, et al. CB1 cannabinoid receptor-mediated modulation of food intake in mice. Br J Pharmacol. 2005;145:293–300. doi: 10.1038/sj.bjp.0706157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams CM, Kirkham TC. Anandamide induces overeating: mediation by central cannabinoid (CB1) receptors. Psychopharmacology (Berl) 1999;143:315–317. doi: 10.1007/s002130050953. [DOI] [PubMed] [Google Scholar]