Abstract
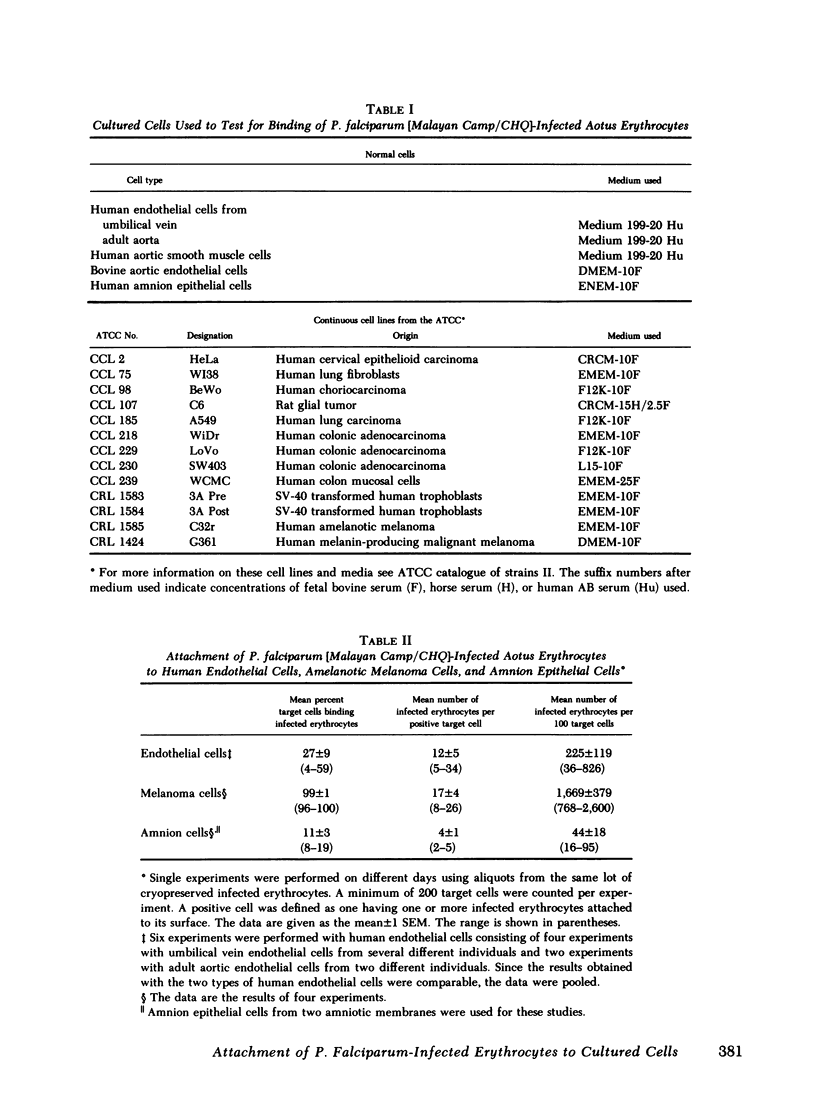
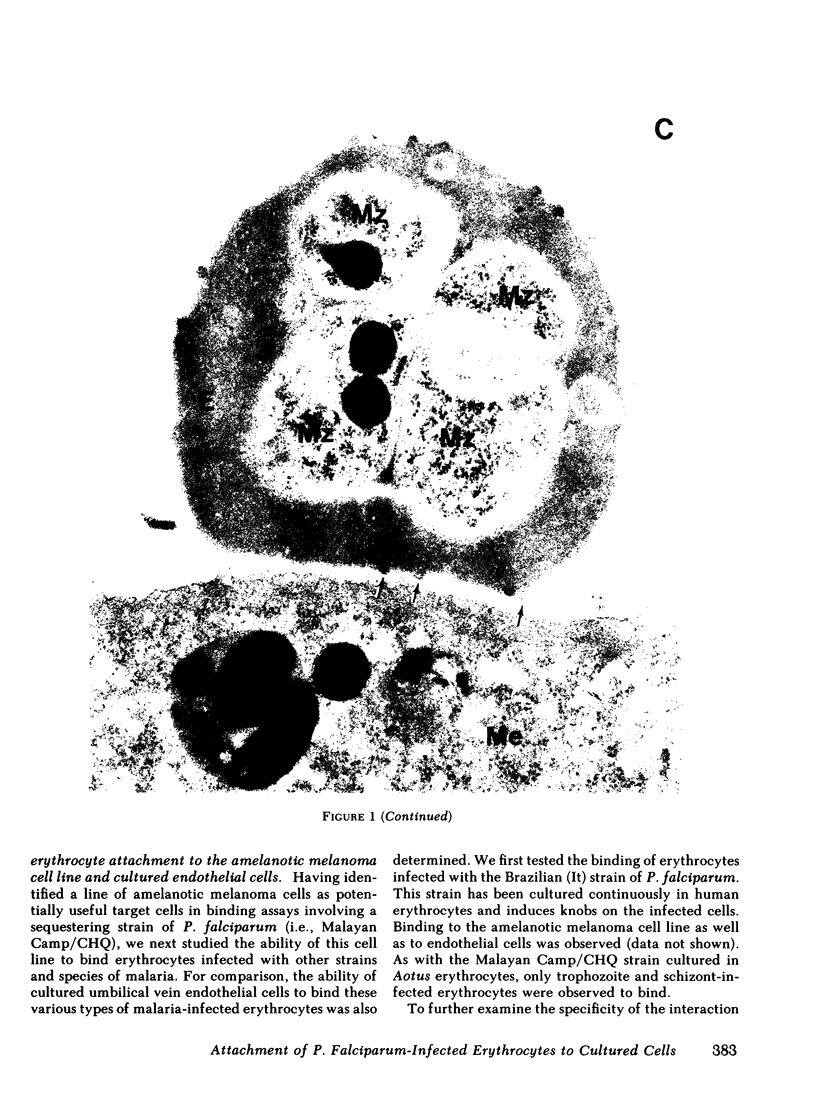
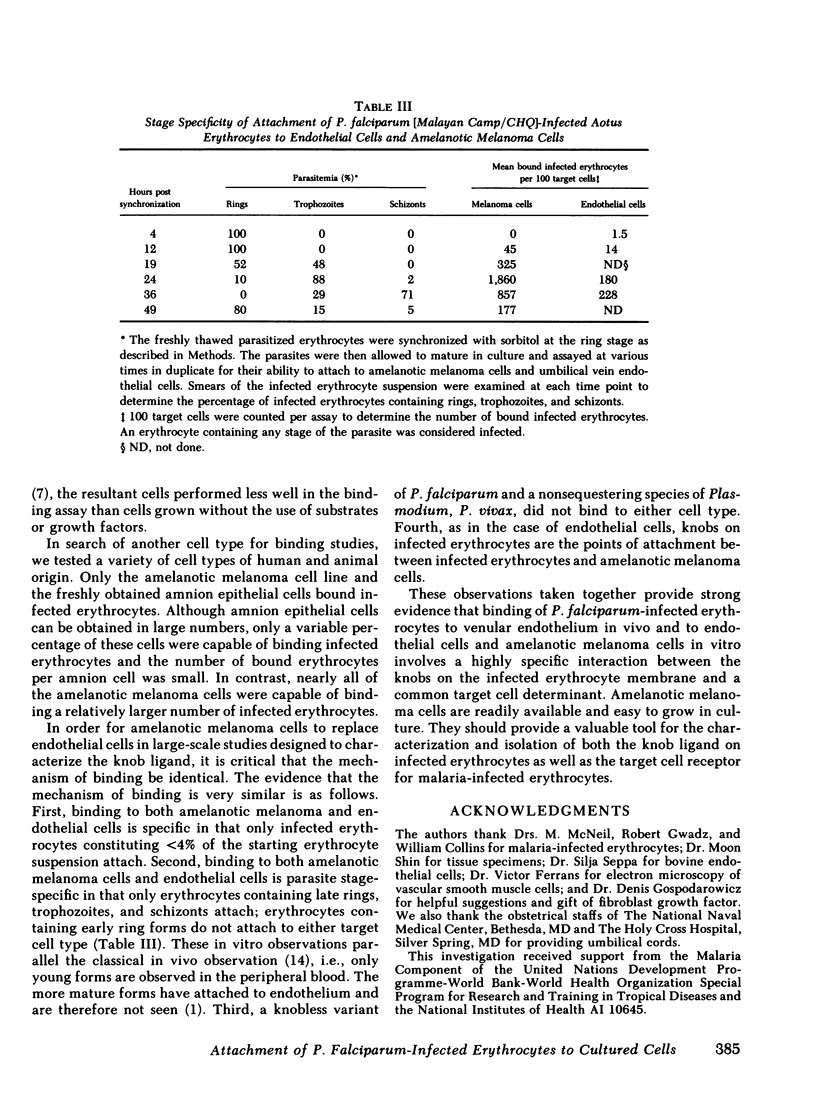
Erythrocytes infected with Plasmodium falciparum trophozoites and schizonts are not seen in the peripheral circulation because they attach to venular endothelium via knoblike structures on the infected erythrocyte membrane. We have recently shown that erythrocytes containing P. falciparum trophozoites and schizonts likewise attach to cultured human venous endothelial cells via knobs. In search of a more practical target cell for large scale binding studies designed to characterize and isolate the knob ligand, we tested various normal cells and continuous cell lines for their ability to bind P. falciparum-infected erythrocytes. Of the 18 cell types tested, binding of infected erythrocytes was observed to a human amelanotic melanoma cell line and amnion epithelial cells as well as to human aortic and umbilical vein endothelial cells. 96-100% of amelanotic melanoma cells bound 17±4 (±1 SEM) infected erythrocytes per positive cell, whereas fewer endothelial cells (4-59%) and amnion epithelial cells (8-19%) were capable of binding 12±5 and 4±1 infected erythrocytes per positive cell, respectively. Further studies designed to compare the mechanism of binding to the amelanotic melanoma cell line and endothelial cells showed the following results. First, that adhesion of infected erythrocytes to these two cell types was parasite stage-specific in that only erythrocytes containing late ring forms, trophozoites, and schizonts bound. Erythrocytes containing early ring forms, which do not attach to venular endothelium in vivo, did not bind to either cell type. Second, erythrocytes infected with trophozoites and schizonts of P. vivax or a knobless strain of P. falciparum, both of which continue to circulate in vivo, did not bind to either target cell type. Third, transmission electron microscopy showed that infected erythrocytes attached to the amelanotic melanoma cells via knobs. We conclude that cultured human endothelial cells and an amelanotic melanoma cell line share common determinants on their surface and that the mechanism of binding to these two different cell types is similar. The amelanotic melanoma cell line offers a useful substitute for endothelial cells in binding studies requiring large numbers of target cells.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aikawa M., Miller L. H., Rabbege J. Caveola--vesicle complexes in the plasmalemma of erythrocytes infected by Plasmodium vivax and P cynomolgi. Unique structures related to Schüffner's dots. Am J Pathol. 1975 May;79(2):285–300. [PMC free article] [PubMed] [Google Scholar]
- Diggs C., Joseph K., Flemmings B., Snodgrass R., Hines F. Protein synthesis in vitro by cryopreserved Plasmodium falciparum. Am J Trop Med Hyg. 1975 Sep;24(5):760–763. doi: 10.4269/ajtmh.1975.24.760. [DOI] [PubMed] [Google Scholar]
- Glaser B. M., D'Amore P. A., Seppa H., Seppa S., Schiffmann E. Adult tissues contain chemoattractants for vascular endothelial cells. Nature. 1980 Dec 4;288(5790):483–484. doi: 10.1038/288483a0. [DOI] [PubMed] [Google Scholar]
- Gospodarowicz D., Ill C. Extracellular matrix and control of proliferation of vascular endothelial cells. J Clin Invest. 1980 Jun;65(6):1351–1364. doi: 10.1172/JCI109799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaffe E. A., Hoyer L. W., Nachman R. L. Synthesis of antihemophilic factor antigen by cultured human endothelial cells. J Clin Invest. 1973 Nov;52(11):2757–2764. doi: 10.1172/JCI107471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaffe E. A., Nachman R. L., Becker C. G., Minick C. R. Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J Clin Invest. 1973 Nov;52(11):2745–2756. doi: 10.1172/JCI107470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambros C., Vanderberg J. P. Synchronization of Plasmodium falciparum erythrocytic stages in culture. J Parasitol. 1979 Jun;65(3):418–420. [PubMed] [Google Scholar]
- Luse S. A., Miller L. H. Plasmodium falciparum malaria. Ultrastructure of parasitized erythrocytes in cardiac vessels. Am J Trop Med Hyg. 1971 Sep;20(5):655–660. [PubMed] [Google Scholar]
- Miller L. H. Distribution of mature trophozoites and schizonts of Plasmodium falciparum in the organs of Aotus trivirgatus, the night monkey. Am J Trop Med Hyg. 1969 Nov;18(6):860–865. doi: 10.4269/ajtmh.1969.18.860. [DOI] [PubMed] [Google Scholar]
- Ricard M. A., Hay R. J. Proliferation and agglutinability of primary and transformed human epithelial cells in culture. J Cell Sci. 1976 Aug;21(3):553–561. doi: 10.1242/jcs.21.3.553. [DOI] [PubMed] [Google Scholar]
- Trager W., Jensen J. B. Human malaria parasites in continuous culture. Science. 1976 Aug 20;193(4254):673–675. doi: 10.1126/science.781840. [DOI] [PubMed] [Google Scholar]
- Udeinya I. J., Schmidt J. A., Aikawa M., Miller L. H., Green I. Falciparum malaria-infected erythrocytes specifically bind to cultured human endothelial cells. Science. 1981 Jul 31;213(4507):555–557. doi: 10.1126/science.7017935. [DOI] [PubMed] [Google Scholar]