Abstract
The concreteness effect occurs in both normal and language-disordered populations. Research suggests that abstract and concrete concepts elicit differing neural activation patterns in healthy young adults, but this is undocumented in persons with aphasia. Three persons with aphasia and three age-matched controls were scanned using fMRI while processing abstract and concrete words. Consistent with current theories of abstract and concrete word processing, abstract words elicited activation in verbal areas whereas concrete words additionally activated multi-modal association areas. Persons with aphasia show greater differences in neural activation than age-matched controls between abstract and concrete words, possibly due to an exaggerated concreteness effect.
Introduction
The exploration of language processing in persons with aphasia using fMRI is highly informative for clinical aphasia research in particular and cognitive neuroscience in general. However, since acquired aphasia results from damage to neural tissue and is by definition a deficit in language processing, using fMRI to test theories of language processing in this population becomes highly reliant on task performance. Incorrect responses in the scanner have been shown to elicit different activation patterns than correct responses (e.g., Fridriksson, Baker, & Moser, 2009; Postman-Caucheteux et al., 2010), which can be useful for comparing post-treatment to pre-treatment activations, but are confounding when localizing specific language processes in persons with aphasia. This is especially important when the goal is to tease apart very fine distinctions that are easily found in neurologically healthy cohorts, such as the difference in activations for processing canonical versus non-canonical sentences (Wartenburger et al., 2004) and for processing sentences with object-relative clauses versus those with subject-relative clauses while varying plausibility (Caplan, Stanczak, & Waters, 2008). Such distinctions are revealed behaviorally as differences in reaction time in healthy participants, but as differences in both reaction time and accuracy in persons with aphasia. These psycholinguistic distinctions do not necessarily surface as differences in activations for participants with aphasia (e.g., Thompson, den Ouden, Bonakdarpour, Garibaldi, & Parrish, 2010), but this may be due to poor performance. In other words, the inability to perform a task may overshadow differences in activation. Therefore, in neuroimaging studies of language processing in persons with aphasia, it is important to carefully construct the experimental paradigm such that the participants a) exhibit a behavioral difference in the contrasts of interest and b) are able to perform the task consistently with reasonable accuracy (Price, Crinion, & Friston, 2006).
One contrast that is informative in exploring semantic processing in persons with aphasia is the comparison between abstract and concrete word processing. The term ‘concreteness effect’ is used to describe the difference in performance on lexical semantic processing tasks between abstract (e.g., justice) and concrete (e.g., table) words. Neurologically healthy individuals exhibit faster reaction times and more accurate responses for concrete versus abstract words during lexical decision, word recognition, recall, and sentence comprehension tasks (see Paivio, 1991 for a review). This effect is not only present, but more pronounced in persons with aphasia (Barry & Gerhand, 2003; Berndt, Haendiges, Burton, & Mitchum, 2002; Kiran, Sandberg, & Abbott, 2009; Newton & Barry, 1997). Some studies report a reverse effect, where abstract words are associated with faster reaction times and greater accuracy than concrete words in persons with semantic dementia (SD) or encephalitis (Bonner et al., 2009; Cipolotti & Warrington, 1995; Sirigu, Duhamel, & Poncet, 1991). However, note that Jefferies, Patterson, Jones, and Lambon Ralph (2009) found that the reverse concreteness effect appears to be the exception in SD rather than the rule. Regardless, there appears to be a double dissociation which provides evidence of differing neural substrates for the processing of abstract versus concrete words.
A handful of neuroimaging studies have examined the neural correlates for abstract and concrete word processing in healthy adults, but no clear consensus emerged until meta-analyses of these studies were performed (Binder, Desai, Graves, & Conant, 2009; Wang, Conder, Blitzer, & Shinkareva, 2010). Binder and colleagues (2009) examined 120 functional neuroimaging studies in order to identify specific brain regions that contribute to the semantic aspects of word retrieval. A subset of 17 studies in their analysis focused on the processing of “verbal” (abstract) and “perceptual” (concrete) concepts. Using the activation likelihood estimation (ALE) technique, they found stronger activation for concrete concepts in bilateral angular gyrus, which may play a role in supramodal integration; left mid-fusiform gyrus, which may play a role in retrieving knowledge of visual attributes; left dorsomedial prefrontal cortex, which may direct retrieval of semantic information; and left posterior cingulate cortex, which may act as an interface between episodic memory and semantic retrieval. Stronger activation for abstract concepts was found in left inferior frontal gyrus, which may mediate task efficiency in addition to phonological processing and working memory; and left anterior superior temporal sulcus, whose proximity to left superior temporal gyrus suggests a verbal rather than perceptual role in semantic processing.
Wang and colleagues (2010) examined 19 functional neuroimaging studies of abstract and concrete concept processing, only ten of which overlapped with the Binder et al. (2009) study. Using the multilevel kernel density analysis (MKDA) technique, they found similar results as Binder et al. (2009); namely, that abstract words activated left hemisphere language areas including inferior frontal, superior temporal, and middle temporal gyri, whereas concrete words activated areas related to mental image generation including left precuneus, posterior cingulate, fusiform, and parahippocampal gyri. The results of these meta-analyses are in line with the dual-coding theory, which posits that abstract words are processed in a verbal network, whereas concrete words are processed in a network that bridges language and multi-modal processing (see Paivio, 1991 for a review).
The study of abstract and concrete word processing in persons with aphasia to date has not utilized fMRI, but has relied on comparing behavior with aphasia characteristics (in some cases including lesion characteristics) on a case-by-case basis (Barry & Gerhand, 2003; Berndt et al., 2002; Kiran et al., 2009; Newton & Barry, 1997; Tyler, Moss, & Jennings, 1995). Though informative, these types of studies cannot capture the subtle neuroanatomical differences in abstract and concrete word processing in aphasia; they can only suggest which areas, when damaged, cause deficits. One important reason to study abstract and concrete word processing not only psycholinguisically but also neurolinguistically in persons with aphasia is because this distinction has clinical relevance. Natural conversation requires the frequent use of abstract words and concepts. In aphasia, abstract word use is lessened relative to concrete word use – as mentioned previously – contributing to less natural, albeit functional conversation. Furthermore, a recent study by Kiran et al. (2009) showed that training abstract words results in improvement of those words as well as generalization to untrained target concrete words in the same context-category; however, training concrete words results in improvement of only those trained words. The authors suggest that the differential processing of abstract and concrete words contributes to the differences in generalization, but the neurophysiological support for this explanation is incomplete.
Therefore, the present study examined the processing of abstract versus concrete nouns in persons with aphasia (hereafter PWA) and age-matched neurologically healthy older adults (hereafter NHOA) in order to shed more light on the underlying neural mechanisms for the concreteness effect. As mentioned previously, the concreteness effect is not only present, but more pronounced in persons with aphasia. If indeed abstract nouns are processed in a verbal-only network located in the left hemisphere and concrete nouns are processed in a network that is both verbal and multi-modal sensory, then one would expect damage to the left hemisphere language areas to affect the processing of abstract words more than concrete words. Although this is most often what is seen behaviorally in persons with aphasia, the neural mechanisms for this behavioral phenomenon have not yet been explored, let alone confirmed.
The advantage to using abstract and concrete words to explore semantic processing in persons with aphasia is twofold. First, abstract and concrete words both elicit semantic processing in general, but, according to the dual-coding theory, concrete words additionally elicit perceptual processing. This additional operation should require activation in perceptual areas and in fact, this is what is seen in studies of healthy young adults. As suggested by Caplan (2009), in a model where two processes occur in parallel, the comparison between the two can inform the localization of function when one requires a unique operation. Therefore, abstract and concrete words provide a tight comparison with which to explore the subtleties of semantic processing. Second, persons with aphasia exhibit an exaggerated concreteness effect, suggesting that lesion characteristics may influence the neural correlates of abstract and concrete word processing.
In order to shed more light on the underlying neural mechanisms for the concreteness effect, we examined the neural activation patterns for abstract and concrete word processing of three PWA with varying degrees of lesion during two tasks with varying depth of semantic processing – word judgment and synonym judgment – and compared their data with three age-matched controls. We hypothesize that (a) similar to previous work, abstract and concrete words will produce differing patterns of activation such that abstract words are processed in a verbal network and concrete words additionally recruit multi-modal association areas and (b) PWA will show somewhat different activation patterns than their NHOA counterparts due to lesion characteristics. For example, PWA with very large lesions encompassing much of left hemisphere language areas may reorganize language function to right hemisphere language homologues (Sebastian & Kiran, 2011; Sebastian, Kiran, & Sandberg, 2012; Turkeltaub, Messing, Norise, & Hamilton, 2011).
Methods
Participants
Three (1 female) persons with aphasia subsequent to left middle cerebral artery cerebrovascular accident and three (2 female) neurologically healthy older adults participated in the experiment. PWA ranged in age from 55-59 years (mean = 57.19) and were in the chronic stage of recovery (23-76 months post-onset) (see Table 3 for details). NHOA ranged in age from 56-67 years (mean = 59.7). All participants were right-handed, monolingual English speakers. Handedness was confirmed with the Edinburgh Inventory (Oldfield, 1971). A medical history and demographic questionnaire was used to rule out concomitant neurological disease, head trauma, psychiatric disorders, or developmental speech, language, or learning disabilities. Aided or unaided visual and hearing acuity was determined to be within normal limits. All participants obtained at least a high school education and gave informed consent according to the Human Subjects Protocol for Boston University.
Table 3.
Lesion Information for Participants with Aphasia.
| PWA1 | PWA2 | PWA3 | |
|---|---|---|---|
|
| |||
| Lesion Volume | 85 cc | 133 cc | 228 cc |
| Percent Spared Tissue by Region of Interest | |||
| left inferior frontal gyrus p. opercularis | 7.63% | 23.79% | 1.15% |
| left inferior frontal gyrus p. orbitalis | 85.50% | 87.43% | 70.08% |
| left inferior frontal gyrus p. triangularis | 52.09% | 70.47% | 2.06% |
| left middle frontal gyrus | 86.08% | 73.31% | 21.38% |
| left superior frontal gyrus | 100.00% | 99.77% | 85.99% |
| left middle temporal gyrus | 99.97% | 13.71% | 82.45% |
| left angular and supramarginal gyri | 67.94% | 69.49% | 11.15% |
PWA were given a battery of standardized language tests, including the Western Aphasia Battery (WAB; Kertesz, 1982) to establish the type and severity of aphasia, the Boston Naming Test (BNT; Goodglass, Kaplan, & Weintraub, 1983) to determine confrontation naming ability, the Psycholinguistic Assessment of Language Processing in Aphasia (PALPA; Kay, Lesser, & Coltheart, 1992) to determine specific deficits of access to the semantic system, the Pyramids and Palm Trees (PAPT; Howard & Patterson, 1992) to determine overall soundness of the semantic system, and the Cognitive Linguistic Quick Test (CLQT; Helm-Estabrooks, 2001) to determine the relative contribution of cognitive deficits such as attention and memory to language dysfunction (see Table 1 for details).
Table 1.
Diagnostic and Demographic Information for Participants with Aphasia.
| PWA1 | PWA2 | PWA3 | |
|---|---|---|---|
|
| |||
| Age | 56 | 55 | 59 |
| Sex | Female | Male | Male |
| Education | B.A. | M.B.A. | M.B.A. |
| Months Post Stroke | 38 | 76 | 23 |
| Lesion Region | Left MCAa | Left MCA | Left MCA |
| Western Aphasia Battery | |||
| Aphasia Quotient | 96.70 | 77.70 | 78.60 |
| Aphasia Type | Anomic | Conduction | Transcortical Motor |
| Boston Naming Test | 91.67% | 86.67% | 66.67% |
| Psycholinguistic Assessment of Language Processing in Aphasia | |||
| Auditory Lexical Decision: High Imageability | 100.00% | 100.00% | 97.50% |
| Auditory Lexical Decision: Low Imageability | 100.00% | 97.50% | 97.50% |
| Visual Lexical Decision: High Imageablility | 100.00% | 100.00% | 100.00% |
| Visual Lexical Decision: Low Imageablility | 96.67% | 100.00% | 93.33% |
| Auditory Synonym Judgment: High Imageability | 100.00% | 90.00% | 93.33% |
| Auditory Synonym Judgment: Low Imageability | 96.67% | 90.00% | 76.67% |
| Written Synonym Judgment: High Imageability | 100.00% | 96.67% | 86.67% |
| Written Synonym Judgment: Low Imageability | 100.00% | 83.33% | 76.67% |
| Semantic Association: High Imageability | 80.00% | DNTb | 73.33% |
| Semantic Association: Low Imageability | 80.00% | DNT | 86.67% |
| Pyramids and Palm Trees | |||
| Pictures | 96.15% | 98.08% | 94.23% |
| Written Words | 98.08% | 96.15% | 94.23% |
| Cognitive Linguistic Quick Test | |||
| Composite Severity | WNLc | WNL | WNL |
Note:
MCA = middle cerebral artery,
DNT = did not test,
WNL = within normal limits. Note that most cases of differences in performance between high and low imagability reflect better performance for lexical items with high imageability, which are more concrete in nature.
NHOA completed the Mini Mental State Exam (MMSE; Folstein, Folstein, & McHugh, 1975) and the Boston Naming Test Short Form (BNT; Goodglass et al., 1983) to rule out cognitive and semantic deficits. Average scores for these tests were 97% for the MMSE and 100% for the BNT.
Tasks and Stimuli
Two tasks were used to elicit specific semantic processing for abstract and concrete words: word judgment (WJ) and synonym judgment (SJ). In both tasks, participants made a semantic judgment and responded with a button press using the left hand. The left hand was used to equate the task between populations, since patients with aphasia are often hemiplegic on the right side. The two tasks were designed to elicit semantic processing at two different levels. The WJ task is a more metalinguistic task that requires only cursory semantic processing, whereas the SJ task requires access to meaning and is therefore a deeper semantic task. All participants were allowed to practice both tasks until they felt comfortable performing each task.
In the WJ task, participants decided whether a word was abstract or concrete. The control condition for this task was deciding whether letter strings were composed of vowels or consonants. The words abstract / concrete for noun stimuli and vowel / consonant for letter stimuli appeared at the bottom of each screen in order to minimize possible confounding effects of participants not remembering which button to push.
The 50 abstract and 50 concrete words used for the WJ task, as well as their ratings for concreteness, imageability, and frequency were obtained from the Medical Research Council psycholinguistic database (www.psy.uwa.edu.au/mrcdatabase/uwa_mrc.htm; Frances & Kucera, 1983; Gilhooly & Logie, 1980; Toglia & Battig, 1978). Abstract and concrete words were chosen so that they were as far apart on the concreteness and imageability spectrums as possible to ensure distinct behavioral and neurophysiological processing between word types. Therefore, abstract words had significantly lower concreteness (F (1, 98) = 2161.20, p < .01) and imageability (F (1, 98) = 494.70, p < .01) than concrete words. Abstract and concrete words were matched on frequency (F (1, 84) = 2.27, p = .13). The length of each letter string was matched to the letter length of each concrete and abstract word.
In the SJ task, participants decided whether or not paired abstract and paired concrete words were similar in meaning. The control condition was to decide whether or not paired nonwords were the same.
The 50 abstract and 50 concrete words used in the SJ task were taken from a previous treatment study in our lab (Kiran et al., 2009) and from a subtest of the PALPA (Kay et al., 1992). Psycholinguistic information for these words was also obtained from the MRC database (www.psy.uwa.edu.au/mrcdatabase/uwa_mrc.htm; Frances & Kucera, 1983; Gilhooly & Logie, 1980; Toglia & Battig, 1978). Again, abstract words had significantly lower concreteness (F (1, 65) = 846.43, p < .01) and imageability (F (1, 68) = 456.34, p < .01) than concrete words, but abstract and concrete words were matched on frequency (F (1, 54) = .27, p = .61).
The 50 nonwords used in the SJ task were obtained from the ARC nonword database (Rastle, Harrington, & Coltheart, 2002) and were matched in letter length with the abstract and concrete words. Additionally, nonwords with no phonological or orthographic neighbors were chosen so that unintentional processing of real words did not occur due to similarities between nonwords and real words.
Experimental Design
For both tasks, an event-related design was implemented using E-Prime software (Psychology Software Tools, Inc.). Each event stimulus lasted 4 seconds and presentation was randomized within tasks. During each inter-stimulus interval (ISI), which randomly lasted 2, 3, or 4 seconds, a small cross centered on the screen was presented to maintain the subject’s visual attention. The WJ task consisted of three runs, lasting a total of 17.45 minutes. The SJ task consisted of four runs, lasting a total of 23.27 minutes.
Data Collection
Magnetic resonance images were acquired at the Boston University Center for Biomedical Imaging from a 3T Phillips MRI scanner. High-resolution T1 images were acquired with the following parameters:140 sagittal slices, 1 mm3 voxels, 240 × 240 matrix, FOV = 240 mm, flip angle = 8, fold-over direction = AP, TR = 8.2 ms, TE = 3.8 ms. Blood-oxygen-level-dependent (BOLD) sensitive functional images were collected using the following parameters: 31 axial slices (3 mm thick, 0.3 interslice gap), 3mm3 voxels, FOV = 240, flip angle = 90, fold-over direction = AP, TR = 2000 ms, TE = 35 ms. The visual stimuli were presented on a screen behind the scanner, which projected to a mirror fitted to the head coil. Padding was used to minimize head motion and corrective optical lenses were used when necessary to correct visual acuity. After bore entry, the magnet was shimmed to achieve maximum homogeneity.
Data Analysis
SPM8 software (Wellcome Trust Centre for Neuroimaging) was used to analyze the fMRI data. Preprocessing was performed to correct for slice time differences, remove movement and baseline function artifacts, align the structural and functional images to each other, and normalize both structural and functional images to the MNI (Montreal Neurological Institute) template. Baseline functions were filtered out with a high-pass filter of 128 s, motion artifacts were corrected using the ArtRepair toolbox for SPM8 (Mazaika, Hoeft, Glover, & Reiss, 2009). In addition to these basic steps, the preprocessing of PWA data required that lesion masks be drawn using MRIcron software (http://www.cabiatl.com/mricro/) and used during normalization to minimize deformities during warping (Brett, Leff, Rorden, & Ashburner, 2001). MRIcron was also then used to calculate the lesion volume for each patient. Additionally, patient data were not smoothed because (a) this data is not intended to be analyzed in a group analysis – which is the primary reason for smoothing data -- and (b) smoothing may mask small but important activations in patient data (see Meinzer et al., 2012, for a discussion regarding smoothing in patient populations).
After preprocessing, statistical analyses were performed at the individual level with a general linear model (GLM) convolved with a canonical hemodynamic response function (HRF) with a temporal derivative. Only correct responses were analyzed. The contrasts of interest were: abstract words > control items, concrete words > control items, abstract words > concrete words, and concrete words > abstract words.
Results
Behavioral Results
Analyses were performed on both accuracy and reaction time (RT) data (see Table 2). Nonparametric Kruskal-Wallis and Mann-Whitney U-tests were used due to small sample sizes. For the Kruskal-Wallis tests, there were three levels of the factor condition: abstract, concrete, and control. Overall, PWA and NHOA did not differ significantly in reaction time on either task (p = .51 for both), nor in accuracy on the WJ task (p = .82), but during SJ, NHOA were more accurate than PWA (U = 3.86, p = .05). This indicates that the SJ task was more difficult for PWA than for NHOA. For specific effects of condition, PWA data were analyzed separately from NHOA data.
Table 2.
Means and Standard Deviations of Accuracy and Reaction Times.
| Synonym Judgment | Word Judgment | |||
|---|---|---|---|---|
|
| ||||
| NHOAa (N = 3) | PWAb (N = 3) | NHOA (N = 3) | PWA (N = 3) | |
|
| ||||
| Abstract | ||||
| Accuracy | 89.93% (5.84%) | 80.52% (5.50%) | 98.65% (2.33%) | 80.67% (18.15%) |
| Reaction Time | 1954.38 (426.94) | 2020.27 (147.50) | 1517.23 (214.33) | 1727.07 (328.85) |
| Concrete | ||||
| Accuracy | 98.67% (2.31%) | 95.06% (4.61%) | 93.33% (3.05%) | 98.67% (1.15%) |
| Reaction Time | 1741.65 (447.39) | 1762.45 (33.23) | 1541.68 (468.49) | 1377.92 (55.11) |
| Control | ||||
| Accuracy | 99.33% (1.15%) | 98.89% (1.02%) | 95.33% (1.16%) | 100.00% (0.00%) |
| Reaction Time | 1757.41 (626.19) | 1330.72 (34.90) | 1821.95 (410.52) | 1324.60 (355.49) |
Note:
NHOA = Neurologically healthy older adults,
PWA = Persons with aphasia. Reaction time is given in milliseconds. Standard deviations shown in parentheses.
Participants with aphasia
Accuracy
The Kruskal-Wallis test revealed a significant effect of condition on accuracy for both tasks (SJ: H (2) = 32.20, p < .001; WJ: H (2) = 46.59, p < .001). Post-hoc Mann-Whitney U-tests revealed that PWA were less accurate for abstract than concrete words (SJ: U = 8.66, p < .01; WJ: U = 23.42, p< .001). For the SJ task, PWA were less accurate for concrete words than nonwords (U = 4.76, p < .05), but in the WJ task, there was no difference in accuracy between concrete words and letter strings (U = 2.02, p = .16). These results confirm the existence of the concreteness effect in PWA.
Reaction time
The Kruskal-Wallis test showed a significant effect of condition on RT for both tasks (SJ: H (2) = 101.76, p < .001; WJ: H (2) = 38.76, p < .001). Post-hoc Mann-Whitney U-tests revealed that PWA were faster for concrete than abstract words (SJ: U = 11.64, p = .001; WJ: U = 22.66, p < .001). For the SJ task, PWA were faster for nonwords than concrete words (U = 55.28, p < .001), but in the WJ task, there was no difference in reaction time between concrete words and letter strings (U = 1.96, p = .16). These results again confirm the concreteness effect in PWA.
Neurologically healthy older adults
Accuracy
The Kruskal-Wallis test revealed a significant effect of condition on accuracy for SJ (H (2) = 17.62, p < .001), but not for WJ (H (2) = 4.00, p = .14). Post-hoc Mann-Whitney U-tests for the SJ task revealed that NHOA were less accurate for abstract words than concrete words (U = 6.11, p = .01), but there was no difference in accuracy between concrete words and nonwords (U = .51, p = .48). These results confirm the existence of the concreteness effect in NHOA for SJ but not WJ.
Reaction time
The Kruskal-Wallis test showed a significant effect of condition on RT for both tasks (SJ: H (2) = 9.23, p = .01; WJ: H (2) = 20.50, p < .001). Post-hoc Mann-Whitney U-tests revealed differing patterns for each task. For SJ, NHOA exhibited longer RTs for abstract words than concrete words (U = 4.79, p < .05), but there was no significant difference in RT between concrete words and nonwords (U = .33, p = .57). For WJ, NHOA exhibited longer RTs for letter strings than for both abstract words (U = 19.28, p< .001) and concrete words (U = 11.46, p = .001), but there was no significant difference in RT between abstract and concrete words (U = .08, p = .78). These results again confirm the concreteness effect in NHOA, depending on the task.
Neuroimaging results
Data from each participant were analyzed at the individual level, corrected with an FDR of p < .05. FDR was used rather than FWE because the data were not smoothed and the FWE procedure requires the data to be smoothed.
Participants with aphasia
Importantly, all PWA showed patterns of activation that showed certain consistencies (see Figures 1 and 2). First, all PWA showed activation in some portion of left IFG, regardless of task or word type. Second, all PWA exhibited bilateral activation, again, regardless of task or word type. Third, all PWA showed greater activation for abstract than concrete words. Each PWA’s specific neuroimaging results are described separately below.
Figure 1.
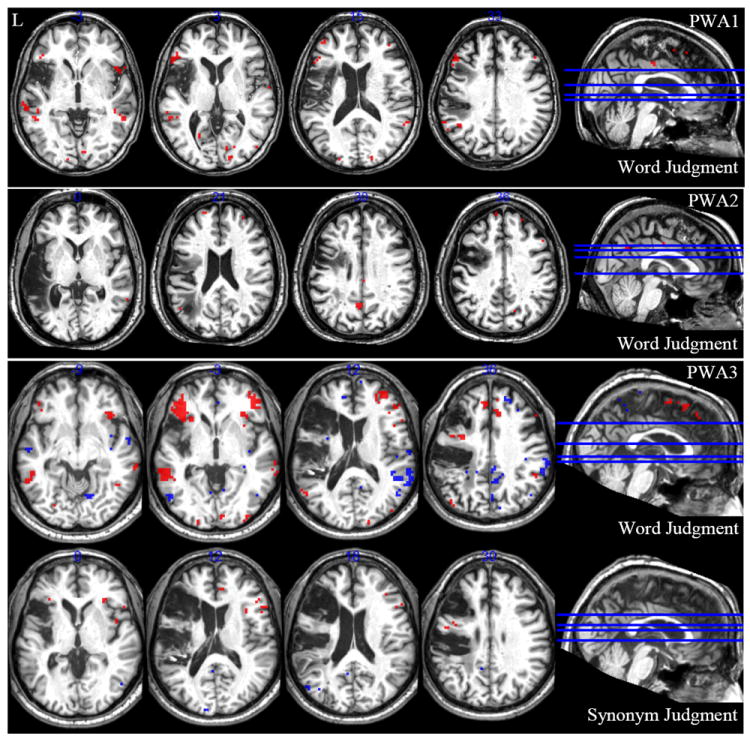
Activations for persons with aphasia (PWA). Activation shown at FDR of p < .05 for tight contrasts abstract > concrete (red) and concrete > abstract (blue).
Figure 2.
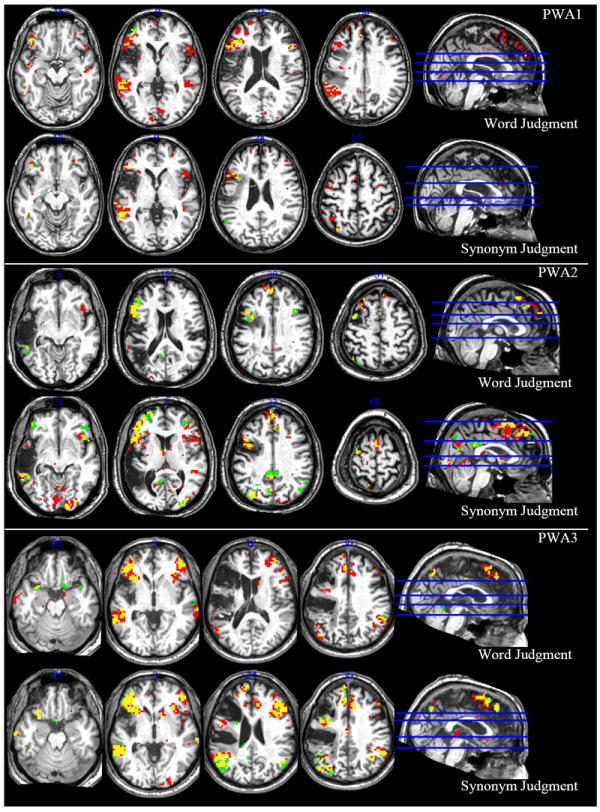
Activations for persons with aphasia (PWA). Activation shown at FDR of p < .05 for contrasts abstract > control (red) and concrete > control (green). Yellow = overlap between contrasts.
PWA1
The first PWA was a 56 year old female with a lesion encompassing about 85 cc of the left hemisphere including roughly 48% of left IFGtri and 32% AG/SMG (see Table 3). During the word judgment task, for the abstract > concrete contrast, this participant activated left IFGorb, left IFGtri, and left MTG, as well as many additional areas, including right hemisphere homologues (see Table 4 and Figure 1). The concrete > abstract contrast did not result in any significant activation. The broader contrasts of abstract > control and concrete > control show much overlap between abstract and concrete word processing (see Table 4 and Figure 2) which aligns with the fact that the tight contrast of concrete > abstract was not significant.
Table 4.
MNI Coordinates and T Values of Areas of Activation for PWA1.
| Region | Abstract vs. Concrete | Abstract vs. Control | Concrete vs. Abstract | Concrete vs. Control | ||||
|---|---|---|---|---|---|---|---|---|
|
| ||||||||
| T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | |
| Anterior Left | Word Judgment | |||||||
|
| ||||||||
| Middle Cingulate Cortex | 3.71 | -3 -21 42 | 3.38 | -3 -36 36 | 3.81 | -3 -36 39 | ||
| Superior Medial Gyrus | 4.66 | -3 39 45 | 6.19 | -12 63 18 | 5.17 | -6 45 42 | ||
| Superior Frontal Gyrus | 5.24 | -24 45 45 | 6.29 | -24 21 54 | 4.87 | -18 27 42 | ||
| Middle Frontal Gyrus | 5.29 | -48 30 33 | 9.95 | -42 6 57 | 6.18 | -42 6 57 | ||
| Inferior Frontal Gyrus p. Orbitalis | 5.21 | -45 30 -3 | 3.38 | -45 21 -9 | 5.77 | -45 33 -15 | ||
| Inferior Frontal Gyrus p. Triangularis | 4.87 | -51 27 3 | 11.08 | -45 30 15 | 7.94 | -42 21 21 | ||
| Middle Orbital Gyrus | 3.97 | -39 51 -9 | 4.01 | -45 48 -6 | ||||
| Insula Lobe | 3.42 | -36 -15 21 | 4.89 | -30 24 -3 | 3.59 | -27 24 0 | ||
| Putamen | 3.38 | -30 6 0 | ||||||
| Pallidum | 3 | -9 6 -6 | ||||||
| Supplementary Motor Area | 3.96 | 0 21 54 | 5.9 | 0 21 54 | ||||
| Precentral Gyrus | 3.61 | -36 -15 57 | 4.57 | -39 -9 60 | ||||
|
Anterior Right
|
||||||||
| Middle Cingulate Cortex | 3.35 | 3 -3 39 | ||||||
| Superior Medial Gyrus | 4.37 | 6 63 18 | 4.7 | 9 60 24 | ||||
| Superior Frontal Gyrus | 3.78 | 30 9 66 | 3.56 | 15 33 54 | ||||
| Middle Frontal Gyrus | 4.7 | 33 57 21 | 5.84 | 45 42 27 | ||||
| Inferior Frontal Gyrus p. Orbitalis | 3.69 | 48 39 -12 | 3.04 | 39 39 -18 | ||||
| Inferior Frontal Gyrus p. Triangularis | 4.06 | 45 39 24 | 4.56 | 45 24 12 | 3.98 | 45 24 15 | ||
| Inferior Frontal Gyrus p. Opercularis | 3.87 | 57 18 0 | 4.82 | 54 18 0 | ||||
| Insula Lobe | 4.72 | 45 18 -3 | 5.15 | 45 18 -6 | ||||
| Rolandic Operculum | 3.49 | 54 9 3 | ||||||
| Caudate Nucleus | 3.55 | 9 12 3 | ||||||
| Supplementary Motor Area | 3.26 | 6 -15 66 | ||||||
| Precentral Gyrus | 4.71 | 24 -24 66 | 4.17 | 39 -27 63 | 3.79 | 36 -27 63 | ||
|
Posterior Left
|
||||||||
| Postcentral Gyrus | 3.15 | -33 -27 45 | ||||||
| Temporal Pole | 4.81 | -39 24 -21 | 3.42 | -42 18 -15 | ||||
| Superior Temporal Gyrus | 3.41 | -45 -15 -6 | ||||||
| Middle Temporal Gyrus | 5.36 | -66 -27 -3 | 10.07 | -60 -45 0 | 9.15 | -54 -57 21 | ||
| Inferior Temporal Gyrus | 3.38 | -42 -63 -9 | 3.66 | -54 -57 -12 | 3.49 | -54 -39 -15 | ||
| Fusiform Gyrus | 3.35 | -42 -48 -24 | ||||||
| Superior Parietal Lobule | 4.11 | -33 -48 69 | 5.24 | -33 -51 69 | ||||
| Inferior Parietal Lobule | 5.46 | -51 -54 48 | 6.79 | -48 -57 57 | 4.06 | -54 -30 48 | ||
| SupraMarginal Gyrus | 4.88 | -60 -51 30 | 7.76 | -60 -48 33 | 4.31 | -57 -48 30 | ||
| Angular Gyrus | 5.4 | -48 -54 30 | 8.57 | -57 -57 24 | 4.61 | -54 -63 30 | ||
| Posterior Cingulate Cortex | 3.07 | -3 -51 24 | ||||||
| Superior Occipital Gyrus | 3.4 | -15 -93 24 | 3.57 | -15 -93 24 | ||||
| Middle Occipital Gyrus | 3.47 | -21 -93 15 | ||||||
| Inferior Occipital Gyrus | 4.02 | -48 -66 -12 | ||||||
| Calcarine Gyrus | 4.46 | -12 -93 -3 | 5.69 | -9 -93 3 | ||||
| Precuneus | 3.9 | -12 -66 36 | 3.88 | -9 -54 27 | ||||
| Cuneus | 3.64 | -9 -93 24 | ||||||
| Lingual Gyrus | 3.95 | -21 -66 3 | 4.86 | 3 -81 0 | ||||
| Cerebellum | 3.8 | -12 -78 -15 | 3.92 | -9 -84 -24 | ||||
|
Posterior Right
|
||||||||
| Postcentral Gyrus | 3.05 | 24 -30 63 | ||||||
| Superior Temporal Gyrus | 4.5 | 63 -48 18 | 4.32 | 66 -48 21 | ||||
| Heschls Gyrus | 4.93 | 51 -9 6 | 3.4 | 54 -6 6 | ||||
| Middle Temporal Gyrus | 4.79 | 54 -42 -3 | 6.35 | 51 -39 0 | ||||
| Hippocampus | 3.64 | 39 -24 -15 | ||||||
| SupraMarginal Gyrus | 3.18 | 69 -39 24 | ||||||
| Superior Occipital Gyrus | 3.81 | 24 -90 21 | 3.33 | 27 -93 15 | ||||
| Middle Occipital Gyrus | 3.94 | 48 -78 3 | ||||||
| Inferior Occipital Gyrus | 4.04 | 42 -81 -12 | ||||||
| Calcarine Gyrus | 5.22 | 15 -93 6 | 4.88 | 15 -93 6 | 4.81 | 6 -87 12 | ||
| Cuneus | 3.87 | 18 -90 15 | 3.1 | 15 -87 21 | ||||
| Lingual Gyrus | 4.27 | 9 -90 -9 | ||||||
| Anterior Left | Synonym Judgment | |||||||
|
| ||||||||
| Anterior Cingulate Cortex | 3.44 | 0 30 27 | ||||||
| Middle Cingulate Cortex | ||||||||
| Superior Medial Gyrus | 4.69 | -12 63 21 | ||||||
| Superior Frontal Gyrus | 3.45 | -12 18 51 | 3.88 | -12 33 54 | ||||
| Middle Frontal Gyrus | 6.27 | -42 6 57 | 4.08 | -24 42 18 | ||||
| Inferior Frontal Gyrus p. Orbitalis | 4.96 | -24 27 -15 | 5.54 | -42 36 -12 | ||||
| Inferior Frontal Gyrus p. Triangularis | 7.53 | -45 39 6 | 6.94 | -45 39 6 | ||||
| Inferior Frontal Gyrus p. Opercularis | 3.56 | -54 15 12 | ||||||
| Middle Orbital Gyrus | 4.15 | -36 54 -3 | ||||||
| Insula Lobe | 5 | -30 21 -3 | 3.92 | -30 21 -3 | ||||
| Putamen | 3.51 | -15 6 6 | ||||||
| Supplementary Motor Area | 4.36 | -3 15 48 | ||||||
| Precentral Gyrus | 5.23 | -39 6 45 | 4.33 | -57 0 36 | ||||
|
Anterior Right
|
||||||||
| Middle Cingulate Cortex | 3.41 | 12 12 45 | ||||||
| Superior Medial Gyrus | 3.84 | 9 60 27 | ||||||
| Superior Frontal Gyrus | 5.35 | 33 54 12 | ||||||
| Middle Frontal Gyrus | 4.99 | 45 42 24 | ||||||
| Inferior Frontal Gyrus p. Triangularis | 4.8 | 57 24 15 | 4.11 | 57 24 15 | ||||
| Insula Lobe | 6.86 | 45 18 -3 | 4.19 | 45 18 -3 | ||||
| Rolandic Operculum | 4.24 | 45 -3 12 | ||||||
| Putamen | 3.81 | 30 -9 12 | ||||||
| Caudate Nucleus | 3.59 | 12 6 6 | ||||||
| Precentral Gyrus | 3.72 | 33 0 51 | 3.6 | 33 -15 66 | ||||
|
Posterior Left
|
||||||||
| Postcentral Gyrus | 6.17 | -48 -39 57 | ||||||
| Temporal Pole | 3.88 | -48 21 -12 | ||||||
| Superior Temporal Gyrus | 3.53 | -48 -18 0 | ||||||
| Middle Temporal Gyrus | 7.65 | -60 -48 3 | 7.16 | -57 -45 0 | ||||
| Inferior Temporal Gyrus | 4.53 | -48 -39 -21 | 4.39 | -48 -39 -18 | ||||
| Fusiform Gyrus | 4.43 | -33 -9 -27 | 4.53 | -42 -51 -12 | ||||
| Inferior Parietal Lobule | 5.3 | -30 -66 42 | 4.41 | -30 -66 45 | ||||
| SupraMarginal Gyrus | 4.37 | -48 -45 30 | ||||||
| Angular Gyrus | 4.52 | -57 -57 24 | ||||||
| Posterior Cingulate Cortex | 3.59 | -3 -33 33 | ||||||
| Superior Occipital Gyrus | 3.94 | -24 -69 33 | ||||||
| Middle Occipital Gyrus | 3.51 | -27 -75 39 | 4.05 | -24 -72 30 | ||||
| Inferior Occipital Gyrus | 3.96 | -48 -60 -18 | ||||||
| ParaHippocampal Gyrus | 3.28 | -24 -27 -18 | ||||||
|
Posterior Right
|
||||||||
| Pallidum | 3.62 | 21 -3 6 | ||||||
| Inferior Parietal Lobule | 4.1 | 57 -36 51 | ||||||
Note: All activation significant at FDR p ≤ .05.
During the synonym judgment task, PWA1 did not show significant activation for either the abstract > concrete contrast or the concrete > abstract contrast. Again, the broader contrasts of abstract > control and concrete > control show much overlap between abstract and concrete word processing, but some subtle differences emerge, such as activation in left AG for the concrete > control contrast and left STG for the abstract > control contrast (see Table 4 and Figure 2).
PWA2
The second PWA was a 55 year old male with a lesion encompassing about 133 cc of the left hemisphere including roughly 30% of IFGtri, 27% of MFG, 86% of MTG, and 31% of AG/SMG (see Table 3). During the word judgment task, this participant activated left MTG for the abstract > concrete contrast, as well as bilateral middle cingulate cortex (MCC) and precuneus, right MFG and MTG, and left SFG (see Table 5 and Figure 1). The concrete > abstract contrast was not significant. Like PWA1, the broader contrasts (abstract > control and concrete > control) reveal much overlap between abstract and concrete word processing (see Table 5 and Figure 2 for details).
Table 5.
MNI Coordinates and T Values of Areas of Activation for PWA2.
| Region | Abstract vs. Concrete | Abstract vs. Control | Concrete vs. Abstract | Concrete vs. Control | ||||
|---|---|---|---|---|---|---|---|---|
|
| ||||||||
| T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | |
| Anterior Left | Word Judgment | |||||||
|
| ||||||||
| Anterior Cingulate Cortex | 3.34 | -9 39 21 | ||||||
| Middle Cingulate Cortex | 4.29 | 0 -12 39 | 4.06 | -3 -45 36 | ||||
| Superior Medial Gyrus | 6.19 | -3 51 30 | 5.57 | -3 54 30 | ||||
| Superior Frontal Gyrus | 4.86 | -9 57 36 | 6.12 | -12 57 27 | 4.81 | -12 57 27 | ||
| Middle Frontal Gyrus | 4.96 | -27 36 48 | 5.16 | -27 24 45 | ||||
| Inferior Frontal Gyrus p. Orbitalis | 4.36 | -42 27 -18 | 4.57 | -45 45 -6 | ||||
| Inferior Frontal Gyrus p. Triangularis | 9.21 | -57 24 15 | 8.89 | -57 21 15 | ||||
| Inferior Frontal Gyrus p. Opercularis | 3.4 | -39 3 24 | ||||||
| Rolandic Operculum | 5.75 | -60 3 6 | ||||||
| Supplementary Motor Area | 5.25 | 0 21 57 | 5.06 | 0 21 57 | ||||
| Precentral Gyrus | 6.81 | -42 9 48 | 6.39 | -33 9 30 | ||||
|
Anterior Right
|
||||||||
| Anterior Cingulate Cortex | 4.52 | 6 48 18 | ||||||
| Middle Cingulate Cortex | 4 | 3 -27 30 | ||||||
| Superior Medial Gyrus | 4.21 | 9 42 51 | 4.36 | 3 33 45 | ||||
| Superior Frontal Gyrus | 3.79 | 15 6 75 | ||||||
| Middle Frontal Gyrus | 4.23 | 24 51 33 | ||||||
| Inferior Frontal Gyrus p. Orbitalis | 3.74 | 48 27 -15 | ||||||
| Inferior Frontal Gyrus p. Triangularis | 6.57 | 48 42 0 | 3.52 | 45 42 0 | ||||
| Inferior Frontal Gyrus p. Opercularis | 4.36 | 51 18 12 | 4.59 | 39 18 30 | ||||
| Insula Lobe | 5.92 | 36 27 -6 | 3.99 | 39 24 -6 | ||||
|
Posterior Left
|
||||||||
| Postcentral Gyrus | 4.31 | -54 -6 45 | 3.55 | -54 -3 42 | ||||
| Temporal Pole | 4.68 | -54 6 -12 | ||||||
| Superior Temporal Gyrus | 4.28 | -63 -42 15 | ||||||
| Middle Temporal Gyrus | 4.09 | -51 -63 21 | 4.21 | -54 -42 -9 | 5.04 | -54 -42 -9 | ||
| Inferior Temporal Gyrus | 3.42 | -48 -48 -9 | 4.01 | -51 -51 -9 | ||||
| Superior Parietal Lobule | 3.82 | -33 -66 57 | 4.14 | -33 -66 57 | ||||
| Inferior Parietal Lobule | 3.65 | -36 -78 42 | 4.46 | -33 -72 51 | ||||
| Angular Gyrus | 3.53 | -45 -63 51 | 3.61 | -45 -63 51 | ||||
| Posterior Cingulate Cortex | 3.76 | 0 -48 30 | 5.6 | -3 -45 33 | ||||
| Superior Occipital Gyrus | 3.29 | -18 -93 24 | ||||||
| Middle Occipital Gyrus | 4.4 | -39 -78 36 | 6.6 | -39 -78 36 | ||||
| Calcarine Gyrus | 3.36 | -3 -69 18 | 5.06 | -3 -69 18 | ||||
| Precuneus | 5.27 | -3 -57 30 | 4.53 | -9 -60 15 | ||||
|
Posterior Right
|
||||||||
| Middle Temporal Gyrus | 4.07 | 63 -51 0 | ||||||
| Fusiform Gyrus | 3.32 | 45 -36 -21 | ||||||
| Posterior Cingulate Cortex | 3.84 | 9 -39 24 | ||||||
| Superior Occipital Gyrus | 4.11 | 27 -90 24 | 3.37 | 27 -90 24 | ||||
| Middle Occipital Gyrus | 4.48 | 30 -90 15 | ||||||
| Calcarine Gyrus | 3.63 | 12 -99 3 | ||||||
| Precuneus | 4.3 | 9 -57 39 | ||||||
| Anterior Left | Synonym Judgment | |||||||
|
| ||||||||
| Anterior Cingulate Cortex | 3.75 | -6 27 30 | ||||||
| Middle Cingulate Cortex | 3.11 | 0 -6 36 | 3.28 | -12 -6 48 | ||||
| Superior Medial Gyrus | 2.94 | 0 69 9 | 6.6 | -3 48 42 | ||||
| Superior Frontal Gyrus | 4.05 | -24 21 54 | 4.25 | -12 72 6 | ||||
| Middle Frontal Gyrus | 6.07 | -27 9 57 | 6.1 | -24 21 48 | ||||
| Inferior Frontal Gyrus p. Orbitalis | 6.06 | -42 27 -18 | 4.32 | -39 30 -15 | ||||
| Inferior Frontal Gyrus p. Triangularis | 10.75 | -57 18 12 | 10.3 | -57 24 15 | ||||
| Inferior Frontal Gyrus p. Opercularis | 3.13 | -39 6 21 | ||||||
| Middle Orbital Gyrus | 3.33 | -27 33 -18 | ||||||
| Insula Lobe | 4.43 | -36 6 6 | ||||||
| Rolandic Operculum | 3.18 | -48 3 9 | 3.84 | -60 0 9 | ||||
| Putamen | 3.02 | -30 -9 6 | ||||||
| Supplementary Motor Area | 3.18 | -3 -3 75 | 5.26 | 0 9 66 | ||||
| Precentral Gyrus | 9.12 | -42 9 48 | 7.9 | -42 9 48 | ||||
|
Anterior Right
|
||||||||
| Anterior Cingulate Cortex | 3.92 | 3 33 24 | ||||||
| Middle Cingulate Cortex | 2.79 | 3 -33 30 | 4.15 | 9 -39 33 | ||||
| Superior Medial Gyrus | 3.76 | 6 60 3 | 3.96 | 6 57 30 | ||||
| Superior Frontal Gyrus | 3.07 | 18 66 21 | ||||||
| Middle Frontal Gyrus | 4.09 | 39 39 6 | 4.62 | 36 45 3 | ||||
| Inferior Frontal Gyrus p. Orbitalis | 6.31 | 45 21 -12 | 5.25 | 42 36 -12 | ||||
| Inferior Frontal Gyrus p. Triangularis | 6.38 | 48 42 0 | 5.48 | 45 45 0 | ||||
| Inferior Frontal Gyrus p. Opercularis | 3 | 60 18 36 | ||||||
| Insula Lobe | 7.78 | 36 27 -6 | 4.32 | 36 24 -3 | ||||
| Putamen | 3.19 | 30 -9 6 | ||||||
| Caudate Nucleus | 3.05 | 9 9 9 | 3.45 | 15 -6 18 | ||||
| Supplementary Motor Area | 3.89 | 6 -15 78 | ||||||
| Precentral Gyrus | 4.09 | 54 6 45 | ||||||
| Paracentral Lobule | 3.42 | 9 -30 63 | ||||||
|
Posterior Left
|
||||||||
| Postcentral Gyrus | 3.25 | -21 -36 75 | 3.96 | -54 -6 45 | ||||
| Temporal Pole | 3.62 | -51 9 -15 | 3 | -54 9 -6 | ||||
| Superior Temporal Gyrus | 5.1 | -66 -39 15 | 4.48 | -60 0 0 | ||||
| Middle Temporal Gyrus | 7.5 | -63 -42 -9 | 7.29 | -60 -42 -12 | ||||
| Inferior Temporal Gyrus | 4.38 | -51 -57 -18 | 4.45 | -51 -48 -9 | ||||
| Superior Parietal Lobule | 2.77 | -21 -57 66 | 4.88 | -21 -81 48 | ||||
| Inferior Parietal Lobule | 7.59 | -42 -60 51 | 5.74 | -36 -72 48 | ||||
| SupraMarginal Gyrus | 3.79 | -63 -24 27 | ||||||
| Angular Gyrus | 2.8 | -45 -72 30 | 6.84 | -39 -63 48 | ||||
| Posterior Cingulate Cortex | 6.13 | -3 -45 33 | 8.74 | -3 -45 33 | ||||
| Superior Occipital Gyrus | 3.8 | -21 -69 33 | ||||||
| Middle Occipital Gyrus | 7.66 | -39 -78 36 | 8.32 | -33 -81 36 | ||||
| Inferior Occipital Gyrus | 3.39 | -24 -87 -9 | ||||||
| Calcarine Gyrus | 4.76 | -6 -63 15 | 5.96 | -3 -66 18 | ||||
| Precuneus | 4.57 | -3 -54 12 | 5.41 | -6 -69 36 | ||||
| Cuneus | 4.43 | 0 -72 30 | 3.42 | 3 -84 24 | ||||
| ParaHippocampal Gyrus | 2.8 | -21 -21 -27 | ||||||
| Lingual Gyrus | 6.36 | -6 -84 -9 | 3.06 | -12 -84 -6 | ||||
| Cerebellum | 5.61 | -3 -63 -6 | 3.11 | -3 -63 -6 | ||||
|
Posterior Right
|
||||||||
| Postcentral Gyrus | 3.8 | 15 -51 72 | ||||||
| Superior Temporal Gyrus | 6.38 | 51 -36 6 | ||||||
| Middle Temporal Gyrus | 3.24 | 57 -66 0 | 3.41 | 63 -27 -3 | ||||
| Inferior Temporal Gyrus | 2.85 | 51 -72 -6 | ||||||
| Fusiform Gyrus | 5.35 | 27 -63 -15 | 3.16 | 30 -72 -15 | ||||
| Thalamus | 3.52 | 9 -21 12 | ||||||
| Superior Parietal Lobule | 4.62 | 24 -75 51 | ||||||
| Inferior Parietal Lobule | 3.51 | 42 -33 48 | 3.11 | 45 -54 48 | ||||
| SupraMarginal Gyrus | 3.36 | 51 -30 42 | ||||||
| Angular Gyrus | 3.25 | 45 -63 30 | 4.99 | 45 -66 30 | ||||
| Superior Occipital Gyrus | 4.47 | 33 -72 42 | 4.43 | 33 -75 42 | ||||
| Middle Occipital Gyrus | 5.61 | 42 -81 3 | 4.51 | 27 -93 3 | ||||
| Inferior Occipital Gyrus | 3.94 | 48 -75 -12 | 4.13 | 30 -87 -6 | ||||
| Calcarine Gyrus | 4.79 | 12 -63 12 | 3.41 | 27 -69 3 | ||||
| Precuneus | 2.96 | 9 -66 66 | 3.19 | 12 -66 36 | ||||
| Cuneus | 5.3 | 21 -87 39 | 3.51 | 9 -93 15 | ||||
| Lingual Gyrus | 6.01 | 15 -96 -9 | 4.52 | 15 -99 -9 | ||||
|
Posterior Central
|
||||||||
| Cerebellar Vermis | 3.87 | 3 -48 0 | 3.7 | -3 -51 6 | ||||
Note: All activation significant at FDR p ≤ .05.
During the synonym judgment task, PWA2 did not show significant activation for either the abstract > concrete contrast or the concrete > abstract contrast. The broader contrasts of abstract > control and concrete > control again showed similar patterns of activation (see Table 5 and Figure 2 for details), but notable differences included bilateral anterior cingulate cortex (ACC) and left insula for the abstract > control contrast and bilateral IFGop for the concrete > control contrast. Additional areas of activation for these contrasts are listed in Table 5.
PWA3
The third participant was a 59 year old male with a lesion encompassing about 228 cc of the left hemisphere including roughly 30% of IFGorb, 98% of IFGtri, 79% of MFG, and 89% of AG/SMG (see Table 3). During the word judgment task, he showed significant activation in left IFGorb and MTG for the abstract > concrete contrast and for the concrete > abstract contrast, left dorsomedial prefrontal cortex (DMPFC), AG, precuneus, and parahippocampal gyrus were significantly active. Additional areas that were active for these contrasts are listed in Table 6 and shown in Figure 1. The broader contrasts of abstract > control and concrete > control showed some similar areas of activation, but again, notable differences surfaced, including left STG for the abstract > control contrast and left fusiform gyrus for the concrete > control contrast. Additional areas of activation for these contrasts are listed in Table 6 and shown in Figure 2.
Table 6.
MNI Coordinates and T Values of Areas of Activation for PWA3.
| Region | Abstract vs. Concrete | Abstract vs. Control | Concrete vs. Abstract | Concrete vs. Control | ||||
|---|---|---|---|---|---|---|---|---|
|
| ||||||||
| T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | |
| Anterior Left | Word Judgment | |||||||
|
| ||||||||
| Anterior Cingulate Cortex | 4.87 | -3 45 18 | 3.56 | -12 48 3 | 3.24 | -3 45 18 | ||
| Middle Cingulate Cortex | 3.61 | -3 -12 39 | 4.27 | -6 -36 45 | ||||
| Superior Medial Gyrus | 6.48 | -6 30 57 | 11.08 | -6 33 45 | 3.72 | -12 48 12 | 5.51 | -3 27 57 |
| Superior Frontal Gyrus | 4.01 | -15 33 51 | 6.96 | -15 33 51 | 4.05 | -24 45 42 | 4.26 | -15 33 54 |
| Middle Frontal Gyrus | 4.22 | -42 12 45 | 7.17 | -27 21 51 | 4.49 | -30 36 48 | 4.15 | -27 21 54 |
| Inferior Frontal Gyrus p. Orbitalis | 6.25 | -51 33 -3 | 11.82 | -39 39 -3 | 7.31 | -39 42 -3 | ||
| Inferior Frontal Gyrus p. Triangularis | 5.17 | -51 33 0 | ||||||
| Insula Lobe | 5.05 | -27 21 -3 | 10.58 | -30 21 -3 | 6.98 | -30 21 -3 | ||
| Supplementary Motor Area | 2.89 | -6 -12 75 | 4.67 | -6 -9 75 | ||||
| Precentral Gyrus | 6.02 | -33 3 36 | 6.27 | -33 3 36 | ||||
|
Anterior Right
|
||||||||
| Anterior Cingulate Cortex | 5.47 | 12 30 24 | 6.34 | 12 30 24 | 3.68 | 9 42 0 | ||
| Middle Cingulate Cortex | 5.96 | 9 18 45 | 3.64 | 6 -12 33 | 5.46 | 6 -51 33 | ||
| Superior Medial Gyrus | 4.67 | 6 45 45 | 4.18 | 12 54 30 | 4.17 | 9 66 12 | 3.47 | 12 54 30 |
| Superior Frontal Gyrus | 3.45 | 21 66 3 | 4.07 | 21 66 3 | 4.71 | 21 45 36 | 4.07 | 24 63 6 |
| Middle Frontal Gyrus | 6.3 | 36 45 21 | 8.29 | 51 18 48 | 5.25 | 33 33 39 | 5.79 | 45 51 0 |
| Inferior Frontal Gyrus p. Orbitalis | 5.75 | 33 30 -6 | 10.84 | 48 45 -3 | 4.69 | 42 42 -6 | ||
| Inferior Frontal Gyrus p. Triangularis | 6.75 | 48 18 18 | 9.83 | 48 18 18 | 4.67 | 51 42 0 | ||
| Insula Lobe | 4.16 | 33 18 3 | 2.94 | 33 24 -18 | 3.51 | 39 -15 -3 | ||
| Rolandic Operculum | 3.5 | 48 -27 18 | ||||||
| Putamen | 3.29 | 15 9 -6 | ||||||
| Caudate Nucleus | 3.9 | 9 15 6 | 6.14 | 9 6 6 | 4.05 | 9 12 0 | ||
| Supplementary Motor Area | 4.05 | 6 18 66 | 6.29 | 3 21 57 | ||||
| Precentral Gyrus | 3.81 | 27 -21 75 | ||||||
| Paracentral Lobule | 3.41 | 6 -27 75 | ||||||
|
Posterior Left
|
||||||||
| Superior Temporal Gyrus | 3 | -63 -33 12 | ||||||
| Middle Temporal Gyrus | 8.63 | -60 -39 -3 | 13.05 | -63 -39 -3 | 4.6 | -51 -69 18 | 5.99 | -60 -48 0 |
| Inferior Temporal Gyrus | 5.29 | -57 -51 -9 | ||||||
| Fusiform Gyrus | 3.65 | -27 -69 -12 | ||||||
| Superior Parietal Lobule | 3.85 | -18 -48 72 | ||||||
| Inferior Parietal Lobule | 4.11 | -36 -78 45 | ||||||
| Angular Gyrus | 3.52 | -45 -60 42 | 4.31 | -57 -57 30 | ||||
| Posterior Cingulate Cortex | 3.9 | -6 -51 21 | 4.41 | -9 -48 27 | ||||
| Superior Occipital Gyrus | 3.02 | -3 -84 45 | 3.49 | -18 -78 27 | ||||
| Middle Occipital Gyrus | 3.92 | -45 -75 36 | 3.16 | -24 -99 9 | 5.13 | -36 -75 24 | 5.17 | -42 -78 30 |
| Inferior Occipital Gyrus | 3.63 | -27 -78 -9 | 4.7 | -54 -69 -3 | ||||
| Calcarine Gyrus | 4.08 | -3 -90 15 | ||||||
| Precuneus | 3.78 | -3 -54 45 | 6.85 | 0 -69 45 | 4.1 | 3 -69 36 | 5.19 | 0 -63 51 |
| ParaHippocampal Gyrus | 4.21 | -21 -24 -21 | 4.49 | -18 -27 -18 | ||||
| Lingual Gyrus | 3.45 | -24 -48 -6 | 3.34 | -15 -51 0 | ||||
| Cerebellum | 4.7 | -12 -36 -18 | 3.38 | -3 -48 -15 | 3.92 | -3 -48 -15 | ||
|
Posterior Right
|
||||||||
| Postcentral Gyrus | 4.26 | 54 -15 30 | ||||||
| Superior Temporal Gyrus | 3.16 | 45 -12 -9 | 4.79 | 60 0 -9 | 4.26 | 66 -15 -6 | ||
| Middle Temporal Gyrus | 5.84 | 66 -45 0 | 8.49 | 66 -45 0 | 5.6 | 66 -51 9 | 4.77 | 54 -66 21 |
| Inferior Temporal Gyrus | 3.69 | 45 -54 -9 | 3.92 | 60 -21 -18 | 3.4 | 48 -66 -3 | 3.92 | 60 -45 -12 |
| Hippocampus | 3.83 | 18 -24 -12 | 3.23 | 33 -33 -3 | ||||
| Thalamus | 3.2 | 3 -21 3 | ||||||
| Superior Parietal Lobule | 3.45 | 15 -57 57 | ||||||
| Inferior Parietal Lobule | 5.09 | 54 -48 48 | 8.39 | 54 -51 48 | 3.97 | 48 -33 48 | 5.86 | 57 -51 42 |
| SupraMarginal Gyrus | 4.22 | 54 -42 39 | 7.26 | 60 -45 36 | 6.46 | 66 -21 21 | 5.71 | 63 -45 39 |
| Angular Gyrus | 3.8 | 54 -60 24 | 6.86 | 39 -57 42 | 4.18 | 48 -63 33 | ||
| Superior Occipital Gyrus | 3.56 | 21 -66 45 | ||||||
| Middle Occipital Gyrus | 4.35 | 24 -96 6 | ||||||
| Inferior Occipital Gyrus | 4.52 | 36 -93 -3 | ||||||
| Calcarine Gyrus | 4.62 | 6 -90 0 | 3.41 | 6 -87 0 | 4.07 | 30 -63 6 | ||
| Precuneus | 5.49 | 15 -66 39 | 7.74 | 15 -66 39 | 5.73 | 12 -54 42 | ||
| Cuneus | 3.41 | 18 -99 9 | 3.24 | 12 -102 9 | 3.72 | 3 -78 39 | ||
| Lingual Gyrus | 4.91 | 15 -66 -9 | 4.38 | 18 -42 -12 | ||||
| Posterior Central | ||||||||
| Cerebellar Vermis | 4.15 | 0 -42 -18 | 3.66 | 0 -48 -6 | ||||
| Anterior Left | Synonym Judgment | |||||||
|
| ||||||||
| Anterior Cingulate Cortex | 3.76 | -3 12 27 | 4.83 | -3 45 18 | ||||
| Middle Cingulate Cortex | 4.54 | -3 -9 36 | 3.68 | 0 -12 36 | ||||
| Superior Medial Gyrus | 4.16 | -6 33 54 | 12.98 | -3 30 57 | 10.82 | -3 30 57 | ||
| Superior Frontal Gyrus | 6.34 | -12 57 27 | 3.85 | -12 51 30 | ||||
| Middle Frontal Gyrus | 4.38 | -27 45 15 | 3.79 | -33 57 6 | ||||
| Inferior Frontal Gyrus p. Orbitalis | 4.21 | -39 24 -3 | 12.84 | -42 42 -6 | 4.28 | -36 33 -9 | 9.63 | -39 39 -3 |
| Inferior Frontal Gyrus p. Triangularis | 3.81 | -33 30 0 | 13.28 | -45 33 0 | 9.94 | -45 33 0 | ||
| Insula Lobe | 4.84 | -27 18 -6 | 10.69 | -27 21 -3 | 3.33 | -36 0 -6 | ||
| Putamen | 2.93 | -30 -12 3 | ||||||
| Pallidum | 4.87 | -9 3 0 | ||||||
| Supplementary Motor Area | 5.07 | -3 12 60 | 8.02 | -3 15 63 | ||||
| Precentral Gyrus | 4.65 | -51 -3 30 | 9.46 | -33 3 36 | 5.66 | -45 6 36 | ||
|
Anterior Right
|
||||||||
| Anterior Cingulate Cortex | 4.22 | 6 42 12 | 4.68 | 12 30 24 | ||||
| Middle Cingulate Cortex | 3.34 | 6 -21 33 | ||||||
| Superior Medial Gyrus | 4.86 | 15 54 30 | 5.3 | 6 48 45 | ||||
| Superior Frontal Gyrus | 5.21 | 24 15 42 | 3.63 | 24 66 6 | ||||
| Middle Frontal Gyrus | 4.57 | 30 6 54 | 10.42 | 33 6 54 | 7.16 | 33 9 57 | ||
| Inferior Frontal Gyrus p. Orbitalis | 4.33 | 42 30 -9 | 4.74 | 27 33 -18 | 5.79 | 45 48 -3 | ||
| Inferior Frontal Gyrus p. Triangularis | 4.74 | 51 24 21 | 10.19 | 48 18 18 | 7.45 | 48 18 18 | ||
| Inferior Frontal Gyrus p. Opercularis | 5.59 | 57 21 12 | 3.23 | 63 21 27 | 3.18 | 39 15 33 | ||
| Insula Lobe | 4.27 | 45 3 0 | 3.88 | 48 12 -9 | 3.43 | 36 21 -3 | ||
| Rolandic Operculum | 4.15 | 57 -3 9 | ||||||
| Putamen | 3.17 | 21 18 -6 | ||||||
| Caudate Nucleus | 5.1 | 12 15 12 | 5.82 | 9 6 6 | ||||
| Supplementary Motor Area | 3.95 | 6 15 57 | 2.72 | 9 -15 75 | ||||
| Precentral Gyrus | 4.25 | 57 3 48 | 7.16 | 57 3 48 | 4.04 | 60 6 33 | 3.96 | 60 9 30 |
|
Posterior Left
|
||||||||
| Postcentral Gyrus | 4.29 | -60 3 18 | 3.18 | -54 -18 21 | ||||
| Superior Temporal Gyrus | 4.27 | -66 -21 9 | 3.92 | -51 -33 21 | ||||
| Middle Temporal Gyrus | 4.96 | -54 -63 9 | 12.48 | -60 -48 -3 | 11.58 | -63 -48 -3 | ||
| Inferior Temporal Gyrus | 4.09 | -51 -54 -15 | 3.2 | -45 -36 -21 | ||||
| Fusiform Gyrus | 2.93 | -36 -66 -12 | 2.98 | -36 -24 -21 | ||||
| Inferior Parietal Lobule | 8.42 | -33 -78 45 | 9.72 | -36 -78 42 | ||||
| Inferior Parietal Cortex | 5.43 | -54 -72 18 | ||||||
| Angular Gyrus | 10.89 | -51 -66 24 | ||||||
| Posterior Cingulate Cortex | 6.04 | -3 -39 30 | 4.11 | -9 -51 30 | 7.91 | -3 -39 30 | ||
| Superior Occipital Gyrus | 4.79 | -9 -99 12 | ||||||
| Middle Occipital Gyrus | 3.26 | -27 -69 27 | 5.07 | -39 -75 21 | ||||
| Inferior Occipital Gyrus | 2.7 | -45 -75 -9 | ||||||
| Calcarine Gyrus | 4.17 | -9 -63 15 | 3.87 | -12 -60 15 | ||||
| Precuneus | 7.05 | -3 -69 42 | 4.3 | -6 -57 15 | 7.46 | -3 -72 42 | ||
| Lingual Gyrus | 2.86 | -12 -39 0 | ||||||
|
Posterior Right
|
||||||||
| Postcentral Gyrus | 2.73 | 27 -27 60 | ||||||
| Superior Temporal Gyrus | 4.46 | 54 -36 6 | ||||||
| Heschls Gyrus | 4.51 | 54 -12 9 | 3.55 | 51 -15 6 | ||||
| Middle Temporal Gyrus | 6.01 | 72 -30 -3 | 4.82 | 51 -69 0 | 5.29 | 45 -57 15 | ||
| Inferior Temporal Gyrus | 3.49 | 48 -54 -12 | 4.43 | 66 -42 -9 | ||||
| Fusiform Gyrus | 4.66 | 39 -60 -12 | 3.55 | 36 -42 -15 | ||||
| Thalamus | 3.04 | 9 -6 6 | ||||||
| Superior Parietal Lobule | 3.06 | 30 -78 51 | ||||||
| Inferior Parietal Lobule | 9.25 | 54 -48 45 | 6.12 | 54 -51 42 | ||||
| SupraMarginal Gyrus | 3.86 | 57 -45 42 | 2.93 | 57 -27 39 | 5.53 | 57 -45 33 | ||
| Angular Gyrus | 10.77 | 39 -57 39 | 7.67 | 39 -57 39 | ||||
| Middle Occipital Gyrus | 3.01 | 33 -93 9 | ||||||
| Inferior Occipital Gyrus | 4.46 | 27 -96 -3 | 3.22 | 42 -84 -3 | ||||
| Calcarine Gyrus | 3.59 | 18 -102 0 | 3.04 | 3 -60 18 | ||||
| Precuneus | 4.87 | 3 -54 48 | 4.13 | 3 -54 45 | ||||
| Cuneus | 3.26 | 21 -99 9 | 4.43 | 12 -75 21 | ||||
| Lingual Gyrus | 3.48 | 27 -87 -6 | 3.02 | 12 -45 0 | ||||
Note: All activation significant at FDR p ≤ .05.
During the synonym judgment task, PWA3 showed activation in left IFGorb, IFGtri, and MTG for the abstract > concrete contrast and in left inferior parietal cortex, PCC, and precuneus for the concrete > abstract contrast, in line with the existing literature. Additional areas for these contrasts are listed in Table 6 and shown in Figure 1. Again, the broader contrasts of abstract > control and concrete > control showed some activation similarities, but again some differences stand out, such as significant activation in left AG for the concrete > control contrast (see Table 6 and Figure 2 for details).
Neurologically healthy older adults
As in PWA, NHOA showed certain consistencies in their patterns of activation (see Figure 3). First, all NHOA showed activation in left IFG. Unlike in PWA, this activation appeared to be mediated by task and word type. Second, NHOA showed fewer regions of activation overall than PWA, constrained to mainly left hemisphere language areas, but notable exceptions exist, including bilateral activations which appear to be mediated by task and word type. Third, all NHOA showed greater activation for abstract than concrete words, which effect also appeared to be mediated by task. Each NHOA’s specific neuroimaging results are described separately below.
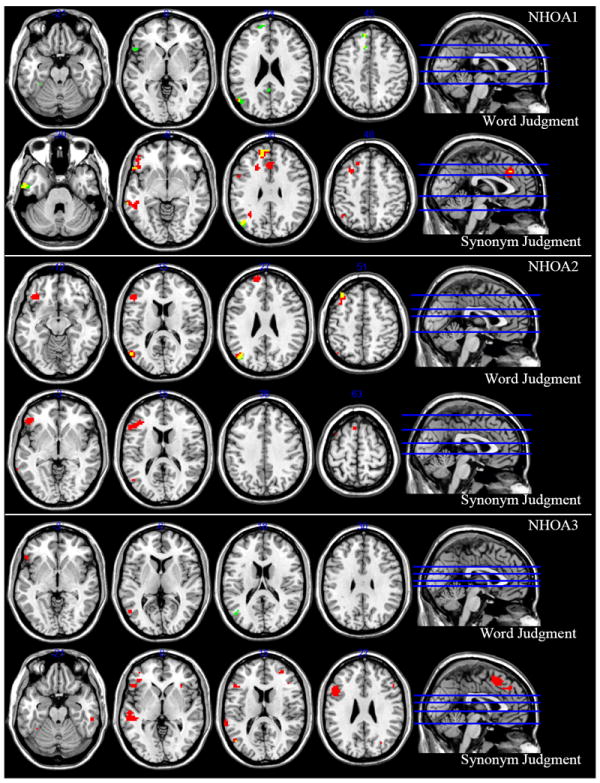
Figure 3.
Activations for neurologically healthy older adults (NHOA). Activation shown at FDR p < .05 for contrasts abstract > control (red) and concrete > control (green). Yellow = overlap between contrasts.
NHOA1
he first participant was a 67 year old female. During the word judgment task, this participant did not show activation for the tight contrasts of abstract > concrete and concrete > abstract. The broader contrasts of abstract > control and concrete > control showed much overlap, but differences included activation in left superior medial gyrus for the abstract > control contrast and activation in left fusiform gyrus, PCC, and precuneus for the concrete > control contrast (see Table 7 and Figure 3 for details).
Table 7.
MNI Coordinates of Activation and T Values for NHOA1.
| Region | Abstract vs. Concrete | Abstract vs. Control | Concrete vs. Abstract | Concrete vs. Control | ||||
|---|---|---|---|---|---|---|---|---|
|
| ||||||||
| T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | |
| Word Judgment | ||||||||
|
| ||||||||
|
Anterior Left
|
||||||||
| Superior Medial Gyrus | 5.23 | -9 45 45 | ||||||
| Superior Frontal Gyrus | 5.78 | -12 45 36 | 7.02 | -12 60 24 | ||||
| Inferior Frontal Gyrus p. Triangularis | 4.85 | -51 21 0 | 5.93 | -54 21 0 | ||||
|
Posterior Left
|
||||||||
| Middle Temporal Gyrus | 5.06 | -51 -60 15 | 6.92 | -51 -66 21 | ||||
| Inferior Temporal Gyrus | 4.21* | -51 -12 -27 | 5.1 | -54 -3 -36 | ||||
| Fusiform Gyrus | 4.92 | -33 -39 -21 | ||||||
| Angular Gyrus | 5.55 | -51 -69 24 | 6.21 | -45 -72 27 | ||||
| Posterior Cingulate Cortex | 5.34 | -3 -51 24 | ||||||
| Precuneus | 4.93 | -6 -57 15 | ||||||
|
| ||||||||
| Synonym Judgment | ||||||||
|
| ||||||||
|
Anterior Left
|
||||||||
| Superior Medial Gyrus | 5.97 | 3 33 33 | 8.47 | 0 30 33 | 5.05 | 0 27 36 | ||
| Superior Frontal Gyrus | 5.35 | -21 27 39 | 9.42 | -21 27 39 | 6.75 | -15 48 30 | ||
| Middle Frontal Gyrus | 4.91 | -27 18 45 | 6.95 | -30 18 48 | 5.48 | -21 30 39 | ||
| Inferior Frontal Gyrus p. Orbitalis | 4.85 | -48 42 -9 | 9.4 | -45 42 -12 | 6.32 | -54 24 -3 | ||
| Inferior Frontal Gyrus p. Triangularis | 5.65 | -54 33 0 | ||||||
| Inferior Frontal Gyrus p. Opercularis | 4.9 | -45 6 27 | ||||||
| Insula Lobe | 4.39 | -33 21 -9 | ||||||
| SMA | 4.71 | 0 12 45 | ||||||
|
Anterior Right
|
||||||||
| Middle Cingulate Cortex | 4.47 | 3 21 39 | ||||||
| Inferior Frontal Gyrus p. Orbitalis | 4.3* | 30 24 -12 | 4.79 | 33 18 -21 | ||||
|
Posterior Left
|
||||||||
| Superior Temporal Gyrus | 6.67 | -51 -42 18 | ||||||
| Middle Temporal Gyrus | 4.69 | -51 -45 -6 | 6.63 | -54 -42 -6 | 4.73 | -66 -36 -6 | ||
| Inferior Temporal Gyrus | 7.61 | -60 -9 -30 | ||||||
| Angular Gyrus | 4.82 | -42 -57 45 | 9.41 | -45 -69 36 | 9.57 | -45 -69 33 | ||
|
Posterior Right
|
||||||||
| Cerebellum | 6.44 | 15 -78 -39 | 4.59 | 12 -78 -39 | ||||
Note: Activation significant at FDR p < .05. Asterisks indicate activations that were significant at p < .10.
During the synonym judgment task, the abstract > concrete contrast showed activation in left IFG (p. orbitalis and opercularis) and MTG, as well as additional areas listed in Table 7, but the concrete > abstract contrast did not reveal any significant areas of activation. The broader contrasts of abstract > control and concrete > control showed overlap, but notable differences included activation in right IFGorb and left STG for the abstract > control contrast and activation in left ITG for the concrete > control contrast (see Table 7 for details).
NHOA2
The second participant was a 56 year old male. During the word judgment task, this participant did not show activation for the tight contrasts of abstract > concrete and concrete > abstract. The broader contrasts of abstract > control and concrete > control showed similar activation patterns, but the abstract > control contrast additionally activated left superior medial gyrus, SFG, AG, and PCC (see Table 8 and Figure 3 for details).
Table 8.
MNI Coordinates of Activation and T Values for NHOA2.
| Region | Abstract vs. Concrete | Abstract vs. Control | Concrete vs. Abstract | Concrete vs. Control | ||||
|---|---|---|---|---|---|---|---|---|
|
| ||||||||
| T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | |
|
| ||||||||
| Word Judgment | ||||||||
|
Anterior Left
|
||||||||
| Superior Medial Gyrus | 7.27 | -12 60 30 | ||||||
| Superior Frontal Gyrus | 8.49 | -15 54 42 | ||||||
| Middle Frontal Gyrus | 7.22 | -33 33 51 | 5.09 | -27 27 57 | ||||
| Inferior Frontal Gyrus p. Orbitalis | 7.18 | -36 30 -12 | 4.55 | -36 33 -9 | ||||
| Inferior Frontal Gyrus p. Triangularis | 6.5 | -48 27 9 | 4.59 | -45 30 9 | ||||
|
Posterior Left
|
||||||||
| Middle Temporal Gyrus | 7.4 | -51 -66 18 | 5.76 | -51 -66 15 | ||||
| Angular Gyrus | 5.98 | -42 -72 45 | ||||||
| Posterior Cingulate Cortex | 4.59 | -3 -45 30 | ||||||
| Middle Occipital Gyrus | 7.1 | -42 -72 27 | 7.19 | -39 -72 27 | ||||
|
| ||||||||
| Synonym Judgment | ||||||||
|
| ||||||||
|
Anterior Left
|
||||||||
| Middle Frontal Gyrus | 4.35 | -36 12 63 | ||||||
| Inferior Frontal Gyrus p. Orbitalis | 5.88 | -48 33 -3 | ||||||
| Inferior Frontal Gyrus p. Triangularis | 5.26 | -36 33 15 | ||||||
| SMA | 5.33 | -6 24 66 | ||||||
|
Posterior Left
|
||||||||
| Middle Temporal Gyrus | 4.78 | -51 -66 15 | ||||||
Note: All activation significant at FDR p ≤ .05.
Like the word judgment task, the synonym judgment task showed no significant activation for the tight contrasts of abstract > concrete and concrete > abstract. Additionally, there was no significant activation for the broad contrast of concrete > control. However, the abstract > control contrast showed activation in left IFG (p. orbitalis and triangularis) and MTG (see Table 8 and Figure 3 for details).
NHOA3
The third participant was a 56 year old female. During the word judgment task, this participant showed activation in left superior parietal lobule for the tight contrast of abstract > concrete, but did not show activation for the tight contrast of concrete > abstract. The broader contrast of abstract > control approached significance in left IFG and MTG, and the concrete > control contrast showed activation in left MTG (see Table 9 and Figure 3 for details).
Table 9.
MNI Coordinates of Activation and T Values for NHOA3.
| Region | Abstract vs. Concrete | Abstract vs. Control | Concrete vs. Abstract | Concrete vs. Control | ||||
|---|---|---|---|---|---|---|---|---|
|
| ||||||||
| T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | T value | MNI x,y,z coordinates | |
| Word Judgment | ||||||||
|
| ||||||||
|
Anterior Left
|
||||||||
| Inferior Frontal Gyrus p. Orbitalis | 4.78* | -54 27 -3 | ||||||
|
Posterior Left
|
||||||||
| Middle Temporal Gyrus | 4.59* | -54 -66 6 | 5.07 | -45 -69 18 | ||||
| Superior Parietal Lobule | 5.43 | -24 -54 48 | ||||||
|
| ||||||||
| Synonym Judgment | ||||||||
|
| ||||||||
|
Anterior Left
|
||||||||
| Superior Medial Gyrus | 6.14 | -3 39 39 | ||||||
| Inferior Frontal Gyrus p. Triangularis | 5.09 | -51 27 0 | 8.13 | -51 18 27 | ||||
| Insula Lobe | 4.62 | -27 24 -6 | ||||||
| SMA | 5.39 | -3 18 48 | 8.39 | 0 21 51 | ||||
| Precentral Gyrus | 5.04 | -39 3 57 | ||||||
|
Anterior Right
|
||||||||
| Middle Frontal Gyrus | 5.54 | 33 48 18 | ||||||
| Inferior Frontal Gyrus p. Triangularis | 4.34* | 48 30 18 | 5.68 | 48 30 18 | ||||
| Inferior Frontal Gyrus p. Opercularis | 6.63 | 54 18 36 | ||||||
| Insula Lobe | 5 | 33 24 0 | ||||||
|
Posterior Left
|
||||||||
| Superior Temporal Gyrus | 4.63 | -66 -39 15 | 7 | -66 -39 15 | ||||
| Middle Temporal Gyrus | 5.2 | -51 -30 0 | 8.09 | -54 -36 -6 | 4.63 | -45 -69 12 | ||
| Fusiform Gyrus | 4.34* | -39 -57 -12 | 5.56 | -39 -57 -12 | ||||
| Cerebellum | 4.91 | -18 -90 -21 | ||||||
|
Posterior Right
|
||||||||
| Inferior Temporal Gyrus | 4.78 | 57 -30 -21 | ||||||
| Middle Occipital Gyrus | 4.45 | 30 -72 27 | ||||||
| Cerebellum | 6.17 | 9 -87 -30 | ||||||
Note: Activation significant at FDR p < .05. Asterisks indicate activations that were significant at p < .10.
During the synonym judgment task, the abstract > concrete contrast showed activation in areas including left IFGtri, STG, and MTG, but the concrete > abstract contrast did not reveal any significant areas of activation (see Table 9). The broader contrast of abstract > control showed much more activation than the concrete > control contrast, with the only overlap occurring in left MTG (see Table 9 and Figure 3 for details).
Discussion
We examined the neural activation patterns for abstract and concrete word processing of three PWA with varying degrees of lesion during two tasks with varying depth of semantic processing and compared their data with three age-matched controls in order to shed more light on the concreteness effect seen in both neurologically intact adults and PWA. As mentioned in the introduction, the abstract-concrete distinction provides a unique opportunity for studying semantic processing in PWA because abstract and concrete words have been shown to elicit differing patterns of activation in neurologically intact adults (e.g., Binder et al., 2009; Wang et al., 2010) and can be differentially impaired due to brain damage.
Both NHOA and PWA showed differing patterns of activation for abstract and concrete word processing, confirming our first hypothesis. The direct contrast of abstract > concrete was significant in two of the three NHOA and in all three PWA, and the direct contrast of concrete > abstract was significant in one PWA, but none of the NHOA. It is interesting to note that the NHOA did not show as robust differences between abstract and concrete word processing as PWA. This lack of robustness and agreement among NHOA is not altogether surprising, since there were only three participants. The studies leading up to the Binder et al. (2009) and Wang et al. (2010) meta-analyses were not in agreement until all their data were pooled together. That being said, it is somewhat surprising that the PWA show such robust neural activation differences. It is possible that the exaggerated concreteness effect seen in patients behaviorally translates to more robust differences in neural activation between abstract and concrete word processing.
When the direct contrasts of abstract > concrete and concrete > abstract were significant, areas that were shown to be active were in line with previous work and with the dual-coding theory, as can be seen in Table 10 and Figure 1. Interestingly, additional areas of activation for these contrasts were also noted for both NHOA and PWA, but to a much greater extent for the PWA (see Tables 4-9). These additional areas that were not also observed in the Binder et al. (2009) and Wang et al. (2010) meta-analyses may have been recruited due to increased effort.
Table 10.
Comparison of Activations between Persons with Aphasia, Neurologically Healthy Older Adults, and Existing Literature
| Area | Binder et al., 2009 | Wang et al., 2010 | Persons with Aphasia | Neurologically Healthy Older Adults | |||||
|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||
| Various Tasks | Word Judgment | Synonym Judgment | Word Judgment | Synonym Judgment | |||||
|
Abstract > Concrete
| |||||||||
| LIFGtri | X | X | X | X | |||||
| LIFGorb | X | X | X | X | X | X | |||
| LSTG | X (STS) | X | X | ||||||
| LMTG | X | X | X | X | X | X | X | ||
|
| |||||||||
|
Concrete > Abstract
| |||||||||
| L DMPFC | X | X | |||||||
| L fusiform | X | X | |||||||
| LAG | X | X (IPC) | |||||||
| RAG | X | X | |||||||
| LPCC | X | X | X | X | |||||
| L precuneus | X | X | X | ||||||
| L para-hippocampal | X | ||||||||
Note:Only areas matching with previous meta-analyses shown. For current study, each X represents activation for one participant. See text for abbreviations of anatomical areas.
Furthermore, there appears to be an effect of task. For PWA, there are more areas active for both abstract > concrete and concrete > abstract contrasts in the WJ task than in the SJ task. For NHOA, this trend is reversed. The WJ task may require deeper semantic processing from the PWA than the NHOA, resulting in greater differences between abstract and concrete word processing for PWA in this task. The SJ task, which is meant to require deeper semantic processing than the WJ task, may be the depth of semantic processing necessary to show the maximum differences in abstract and concrete word processing for NHOA, but for PWA, the increased difficulty for this task may overshadow inherent differences between abstract and concrete word processing. These speculations are tentative and would need to be confirmed with more subjects.
It is also informative to qualitatively compare the activation patterns of abstract and concrete words when each has had the control condition removed. PWA activated areas consistent with their age-matched counterparts as well as areas consistent with previous work. Specifically, regardless of task, all three PWA activated left IFG, STG, and MTG for abstract word processing (see Figure 2 and Tables 4-6), which is consistent with both the existing literature and all three NHOA (see Figure 3 and Tables 7-9). For concrete word processing, again, regardless of task, all three PWA activated left AG, PCC, and portions of DMPFC (see Figure 2 and Tables 4-6), in line with the existing literature and at least one NHOA (see Figure 3 and Table 7). Thus, the qualitative comparison of abstract > control and concrete > control contrasts for both the WJ task and the SJ task confirms the results of the abstract > concrete and concrete > abstract contrasts and suggests that with more data, these tentative results are likely to become more robust.
Importantly, NHOA and PWA show surprisingly similar patterns of activation for abstract and concrete words in this experiment. Most notably, both NHOA and PWA activated left IFG (p. triangularis and orbitalis) and MTG for both abstract and concrete words. In fact, all three PWA activated all three of these areas, which is quite remarkable considering the fact that PWA2 is missing roughly 85% of left MTG and PWA3 is missing roughly 95% of IFGtri. This result reinforces the importance of left IFG and MTG for semantic processing in general (Lau, Phillips, & Poeppel, 2008; Vigneau et al., 2006).
The differences that surface appear to be influenced by cognitive demand, task, and lesion characteristics; therefore, our second hypothesis that differences between NHOA and PWA would be due to lesion characteristics is supported and extended to task and cognitive demand. PWA in general, show increased activity in comparison to NHOA. This is in the face of similar task performance; in fact, for the WJ task, there was no statistically significant difference in behavioral performance between the two groups. In order to maintain this level of performance, PWA almost certainly require more cognitive effort due to their language deficit caused by damage to the left perisylvian region. This increased effort may be reflected in increased global activity.
Additionally, all three PWA showed perilesional activation. PWA1 showed activation in left IFGtri and AG, even though both areas were lesioned. PWA2’s lesion was much larger than PWA1’s, but he too showed activation in lesioned areas including left IFG, MFG, MTG, and AG areas for abstract and/or concrete words, depending on the task. When PWA2 did not show activation in left MTG and AG for abstract words as NHOA did, the amount of damage in these areas could be to blame. Although PWA3’s lesion was the largest, he also activated lesioned areas, including left IFGtri, MFG, and AG/SMG for abstract and/or concrete words, depending on the task. When these areas were active in NHOA, but not PWA3, the right hemisphere homologue was often active (see Results for details).
These individual patterns of activation can be interpreted in light of the amount of spared tissue in each ROI and with respect to the existing literature examining the neural correlates of language recovery after stroke. For all three PWA, the activation in spared tissue surrounding the lesion in areas such as left IFG and MTG, suggest restoration of function (e.g., Fridriksson, Richardson, Fillmore, & Cai, 2012; Meinzer et al., 2008; Saur et al., 2006; Turkeltaub et al., 2011). Additionally, right hemisphere homologues were recruited for all three PWA, which may be indicative of a reorganization of language function or may represent supplementary – possibly non-essential – processing. For PWA3, who had the largest lesion and the most right hemisphere activation, RH activation may reflect compensation, but for the other PWAs, whose lesions were not as extensive and who had less right hemisphere activation than PWA3, this may simply reflect increased effort. Because of the small sample size, it is impossible to conduct meaningful statistics to determine the relationship between lesion characteristics and activation patterns and therefore substantial conclusions regarding the relative contributions of perilesional and contralesional activations cannot be made at this time.
Conclusion
The results of this study are in line with previous neuroimaging studies of abstract and concrete word processing showing that abstract words are processed in a mainly verbal network whereas concrete words are processed in a mainly perceptual network (e.g., Binder et al., 2009 and Wang et al., 2010). We found this to be true in PWA and their neurologically healthy age-matched counterparts. PWA and NHOA exhibit remarkably similar activation patterns, although notable differences do exist. These differences are most likely due to lesion characteristics and help to inform the process of language recovery after stroke. Notably, PWA activated both spared tissue and right hemisphere homologues. Although this work is tentative due to the small sample size, these activations appear to be reorganization of function rather than maladaptive compensation, due to the relatively high performance of these participants.
Some limitations of this study are the small sample sizes and the similarity in aphasic profiles of the patients. Future work will incorporate more participants with greater variation in aphasia severity, with correlations between behavior and activation being conducted. Additionally, we plan to explore changes in abstract and concrete word processing as a function of a theoretically-based treatment focused on abstract word training. As mentioned in the introduction, PWA show an exaggerated concreteness effect which has been successfully manipulated in treatment to boost its therapeutic effects (Kiran, Sandberg, & Abbott, 2009). Knowing where abstract and concrete words are normally processed and how that processing may shift due to aging and left-hemisphere stroke will aid in discovering why the concreteness effect is exaggerated in PWA and why training abstract words seems to be more beneficial than training concrete words. This study is a step forward in a continued effort to understand the mechanisms underlying word retrieval deficits in aphasia in order to adopt more effective treatment methods.
References
- Barry C, Gerhand S. Both concreteness and age-of-acquisition affect reading accuracy but only concreteness affects comprehension in a deep dyslexic patient. Brain Lang. 2003;84(1):84–104. doi: 10.1016/s0093-934x(02)00522-9. [DOI] [PubMed] [Google Scholar]
- Berndt RS, Haendiges AN, Burton MW, Mitchum CC. Grammatical class and imageability in aphasic word production: their effects are independent. Journal of Neurolinguistics. 2002;15:353–371. [Google Scholar]
- Binder JR, Desai Rutvik H, Graves William W, Conant Lisa L. Where Is the Semantic System? A Critical Review and Meta-Analysis of 120 Functional Neuroimaging Studies. Cereb Cortex. 2009 doi: 10.1093/cercor/bhp055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonner MF, Vesely L, Price C, Anderson C, Richmond L, Farag C, Grossman M, et al. Reversal of the concreteness effect in semantic dementia. Cogn Neuropsychol. 2009;26(6):568–579. doi: 10.1080/02643290903512305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brett M, Leff AP, Rorden C, Ashburner J. Spatial normalization of brain images with focal lesions using cost function masking. Neuroimage. 2001;14:486–500. doi: 10.1006/nimg.2001.0845. [DOI] [PubMed] [Google Scholar]
- Caplan D, Stanczak L, Waters G. Syntactic and thematic constraint effects on blood oxygenation level dependent signal correlates of comprehension of relative clauses. J Cogn Neurosci. 2008;20(4):643–656. doi: 10.1162/jocn.2008.20044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cipolotti L, Warrington EK. Semantic memory and reading abilities: a case report. J Int Neuropsychol Soc. 1995;1(1):104–110. doi: 10.1017/s1355617700000163. [DOI] [PubMed] [Google Scholar]
- Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- Fridriksson J, Baker JM, Moser D. Cortical mapping of naming errors in aphasia. Human Brain Mapping. 2009;30(8):2487–2498. doi: 10.1002/hbm.20683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fridriksson J, Richardson JD, Fillmore P, Cai B. Left hemisphere plasticity and aphasia recovery. NeuroImage. 2012;60(2):854–863. doi: 10.1016/j.neuroimage.2011.12.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodglass H, Kaplan E, Weintraub S. Boston Naming Test. Philadelphia: Lea & Febiger; 1983. [Google Scholar]
- Helm-Estabrooks N. Cognitive Linguistic Quick Test: The Psychological Corporation 2001 [Google Scholar]
- Howard D, Patterson K. The Pyramids and Palm Trees Test. Bury St. Edmunds: Thames Valley Test Company; 1992. [Google Scholar]
- Jefferies Elizabeth, Patterson Karalyn, Jones Roy W, Lambon Ralph Matthew A. Comprehension of concrete and abstract words in semantic dementia. Neuropsychology. 2009;23(4):492–499. doi: 10.1037/a001545210.1037/a0015452.supp. Supplemental. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kay J, Lesser RP, Coltheart M. The Psycholinguistic Assessment of Language Processing in Aphasia (PALPA) Hove, UK: Erlbaum; 1992. [Google Scholar]
- Kertesz A. The Western Aphasia Battery. Philadelphia: Grune & Stratton; 1982. [Google Scholar]
- Kiran S, Sandberg C, Abbott K. Treatment for lexical retrieval using abstract and concrete words in persons with aphasia: Effect of complexity. Aphasiology. 2009;23:835–853. doi: 10.1080/02687030802588866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lau Ellen F, Phillips Colin, Poeppel David. A cortical network for semantics: (de)constructing the N400. Nature Reviews Neuroscience. 2008;9(12):920–933. doi: 10.1038/nrn2532. [DOI] [PubMed] [Google Scholar]
- Mazaika Paul, Hoeft Fumiko, Glover Gary H, Reiss Allan L. Methods and Software for fMRI Analysis for Clinical Subjects; Paper presented at the Human Brain Mapping.2009. [Google Scholar]
- Meinzer M, Beeson PM, Cappa S, Crinion J, Kiran S, Saur D, Thompson CK, et al. Neuroimaging in aphasia treatment research: Consensus and practical guidelines for data analysis. Neuroimage. 2012 doi: 10.1016/j.neuroimage.2012.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meinzer M, Flaisch T, Breitenstein C, Wienbruch C, Elbert T, Rockstroh B. Functional re-recruitment of dysfunctional brain areas predicts language recovery in chronic aphasia. Neuroimage. 2008;39(4):2038–2046. doi: 10.1016/j.neuroimage.2007.10.008. [DOI] [PubMed] [Google Scholar]
- Newton PK, Barry C. Concreteness Effects in Word Production but Not Word Comprehension in Deep Dyslexia. Cognitive Neuropsychology. 1997;14:481–509. [Google Scholar]
- Paivio A. Dual Coding Theory: Retrospect and current status. Canadian Journal of Psychology. 1991;45(3):255–287. [Google Scholar]
- Postman-Caucheteux WA, Birn RM, Pursley RH, Butman JA, Solomon JM, Picchioni D, Braun AR, et al. Single-trial fMRI Shows Contralesional Activity Linked to Overt Naming Errors in Chronic Aphasic Patients. Journal of Cognitive Neuroscience. 2010;22(6):1299–1318. doi: 10.1162/jocn.2009.21261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price CJ, Crinion J, Friston KJ. Design and analysis of fMRI studies with neurologically impaired patients. J Magn Reson Imaging. 2006;23:816–826. doi: 10.1002/jmri.20580. [DOI] [PubMed] [Google Scholar]
- Rastle K, Harrington J, Coltheart M. 358,534 nonwords: the ARC Nonword Database. Quarterly Journal of Experimental Psychology Section a-Human Experimental Psychology. 2002;55(4):1339–1362. doi: 10.1080/02724980244000099. [DOI] [PubMed] [Google Scholar]
- Saur D, Lange R, Baumgaertner A, Schraknepper V, Willmes K, Rijntjes M, Weiller C. Dynamics of language reorganization after stroke. Brain. 2006;129(Pt 6):1371–1384. doi: 10.1093/brain/awl090. [DOI] [PubMed] [Google Scholar]
- Sebastian Rajani, Kiran Swathi. Task-modulated neural activation patterns in chronic stroke patients with aphasia. Aphasiology. 2011;25(8):927–951. doi: 10.1080/02687038.2011.557436. [DOI] [Google Scholar]
- Sebastian Rajani, Kiran Swathi, Sandberg Chaleece. Semantic processing in Spanish–English bilinguals with aphasia. Journal of Neurolinguistics. 2012;25(4):240–262. doi: 10.1016/j.jneuroling.2012.01.003. [DOI] [Google Scholar]
- Sirigu A, Duhamel JR, Poncet M. The role of sensorimotor experience in object recognition. A case of multimodal agnosia. Brain. 1991;114(Pt 6):2555–2573. doi: 10.1093/brain/114.6.2555. [DOI] [PubMed] [Google Scholar]
- Thompson CK, den Ouden D, Bonakdarpour B, Garibaldi K, Parrish TB. Neural plasticity and treatment-induced recovery of sentence processing in agrammatism. Neuropsychologia. 2010;48(11):3211–3227. doi: 10.1016/j.neuropsychologia.2010.06.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turkeltaub PE, Messing S, Norise C, Hamilton RH. Are networks for residual language function and recovery consistent across aphasic patients? Neurology. 2011;76(20):1726–1734. doi: 10.1212/WNL.0b013e31821a44c1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tyler LK, Moss HE, Jennings F. Abstract word deficits in aphasia: Evidence from semantic priming. Neuropsychology. 1995;9(3):354–363. doi: 10.1037/0894-4105.9.3.354. [DOI] [Google Scholar]
- Vigneau M, Beaucousin V, Hervé PY, Duffau H, Crivello F, Houdé O, Tzourio-Mazoyer N, et al. Meta-analyzing left hemisphere language areas: phonology, semantics, and sentence processing. Neuroimage. 2006;30(4):1414–1432. doi: 10.1016/j.neuroimage.2005.11.002. [DOI] [PubMed] [Google Scholar]
- Wang Jing, Conder Julie A, Blitzer David N, Shinkareva Svetlana V. Neural representation of abstract and concrete concepts: A meta-analysis of neuroimaging studies. Human Brain Mapping. 2010;31(10):1459–1468. doi: 10.1002/hbm.20950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wartenburger Isabell, Heekeren Hauke R, Burchert Frank, Heinemann Steffi, De Bleser Ria, Villringer Arno. Neural correlates of syntactic transformations. Human Brain Mapping. 2004;22(1):72–81. doi: 10.1002/hbm.20021. [DOI] [PMC free article] [PubMed] [Google Scholar]


