Abstract
Alternaria is a ubiquitous fungal genus that includes saprobic, endophytic and pathogenic species associated with a wide variety of substrates. In recent years, DNA-based studies revealed multiple non-monophyletic genera within the Alternaria complex, and Alternaria species clades that do not always correlate to species-groups based on morphological characteristics. The Alternaria complex currently comprises nine genera and eight Alternaria sections. The aim of this study was to delineate phylogenetic lineages within Alternaria and allied genera based on nucleotide sequence data of parts of the 18S nrDNA, 28S nrDNA, ITS, GAPDH, RPB2 and TEF1-alpha gene regions. Our data reveal a Pleospora/Stemphylium clade sister to Embellisia annulata, and a well-supported Alternaria clade. The Alternaria clade contains 24 internal clades and six monotypic lineages, the assemblage of which we recognise as Alternaria. This puts the genera Allewia, Brachycladium, Chalastospora, Chmelia, Crivellia, Embellisia, Lewia, Nimbya, Sinomyces, Teretispora, Ulocladium, Undifilum and Ybotromyces in synonymy with Alternaria. In this study, we treat the 24 internal clades in the Alternaria complex as sections, which is a continuation of a recent proposal for the taxonomic treatment of lineages in Alternaria. Embellisia annulata is synonymised with Dendryphiella salina, and together with Dendryphiella arenariae, are placed in the new genus Paradendryphiella. The sexual genera Clathrospora and Comoclathris, which were previously associated with Alternaria, cluster within the Pleosporaceae, outside Alternaria s. str., whereas Alternariaster, a genus formerly seen as part of Alternaria, clusters within the Leptosphaeriaceae. Paradendryphiella is newly described, the generic circumscription of Alternaria is emended, and 32 new combinations and 10 new names are proposed. A further 10 names are resurrected, while descriptions are provided for 16 new Alternaria sections.
Taxonomic novelties:
New combinations - Alternaria abundans (E.G. Simmons) Woudenb. & Crous, Alternaria alternariae (Cooke) Woudenb. & Crous, Alternaria atra (Preuss) Woudenb. & Crous, Alternaria bornmuelleri (Magnus) Woudenb. & Crous, Alternaria botrytis (Preuss) Woudenb. & Crous, Alternaria caespitosa (de Hoog & C. Rubio) Woudenb. & Crous, Alternaria cantlous (Yong Wang bis & X.G. Zhang) Woudenb. & Crous, Alternaria caricis (E.G. Simmons) Woudenb. & Crous, Alternaria cinerea (Baucom & Creamer) Woudenb. & Crous, Alternaria didymospora (Munt.-Cvetk.) Woudenb. & Crous, Alternaria fulva (Baucom & Creamer) Woudenb. & Crous, Alternaria hyacinthi (de Hoog & P.J. Mull. bis) Woudenb. & Crous, Alternaria indefessa (E.G. Simmons) Woudenberg & Crous, Alternaria leptinellae (E.G. Simmons & C.F. Hill) Woudenb. & Crous, Alternaria lolii (E.G. Simmons & C.F. Hill) Woudenb. & Crous, Alternaria multiformis (E.G. Simmons) Woudenb. & Crous, Alternaria obclavata (Crous & U. Braun) Woudenb. & Crous, Alternaria obovoidea (E.G. Simmons) Woudenb. & Crous, Alternaria oudemansii (E.G. Simmons) Woudenb. & Crous, Alternaria oxytropis (Q. Wang, Nagao & Kakish.) Woudenb. & Crous, Alternaria penicillata (Corda) Woudenb. & Crous, Alternaria planifunda (E.G. Simmons) Woudenb. & Crous, Alternaria proteae (E.G. Simmons) Woudenb. & Crous, Alternaria scirpinfestans (E.G. Simmons & D.A. Johnson) Woudenb. & Crous, Alternaria scirpivora (E.G. Simmons & D.A. Johnson) Woudenb. & Crous, Alternaria septospora (Preuss) Woudenb. & Crous, Alternaria slovaca (Svob.-Pol., L. Chmel & Bojan.) Woudenb. & Crous, Alternaria subcucurbitae (Yong Wang bis & X.G. Zhang) Woudenb. & Crous, Alternaria tellustris (E.G. Simmons) Woudenb. & Crous, Alternaria tumida (E.G. Simmons) Woudenb. & Crous, Paradendryphiella salina (G.K. Sutherl.) Woudenb. & Crous, Paradendryphiella arenariae (Nicot) Woudenb. & Crous. New names - Alternaria aspera Woudenb. & Crous, Alternaria botryospora Woudenb. & Crous, Alternaria brassicae-pekinensis Woudenb. & Crous, Alternaria breviramosa Woudenb. & Crous, Alternaria chlamydosporigena Woudenb. & Crous, Alternaria concatenata Woudenb. & Crous, Alternaria embellisia Woudenb. & Crous, Alternaria heterospora Woudenb. & Crous, Alternaria papavericola Woudenb. & Crous, Alternaria terricola Woudenb. & Crous. Resurrected names - Alternaria cetera E.G. Simmons, Alternaria chartarum Preuss, Alternaria consortialis (Thüm.) J.W. Groves & S. Hughes, Alternaria cucurbitae Letendre & Roum., Alternaria dennisii M.B. Ellis, Alternaria eureka E.G. Simmons, Alternaria gomphrenae Togashi, Alternaria malorum (Ruehle) U. Braun, Crous & Dugan, Alternaria phragmospora Emden, Alternaria scirpicola (Fuckel) Sivan. New sections, all in Alternaria - sect. Chalastospora Woudenb. & Crous, sect. Cheiranthus Woudenb. & Crous, sect. Crivellia Woudenb. & Crous, sect. Dianthicola Woudenb. & Crous, sect. Embellisia Woudenb. & Crous, sect. Embellisioides Woudenb. & Crous, sect. Eureka Woudenb. & Crous, sect. Infectoriae Woudenb. & Crous, sect. Japonicae Woudenb. & Crous, sect. Nimbya Woudenb. & Crous, sect. Phragmosporae Woudenb. & Crous, sect. Pseudoulocladium Woudenb. & Crous, sect. Teretispora Woudenb. & Crous, sect. Ulocladioides Woudenb. & Crous, sect. Ulocladium Woudenb. & Crous, sect. Undifilum Woudenb. & Crous. New genus - Paradendryphiella Woudenb. & Crous.
Key words: Allewia, Chalastospora, Crivellia, Embellisia, Lewia, Nimbya, Paradendryphiella, Sinomyces, systematics, Teretispora, Ulocladium, Undifilum
INTRODUCTION
Alternaria is a ubiquitous fungal genus that includes saprobic, endophytic and pathogenic species. It is associated with a wide variety of substrates including seeds, plants, agricultural products, animals, soil and the atmosphere. Species of Alternaria are known as serious plant pathogens, causing major losses on a wide range of crops. Several taxa are also important postharvest pathogens, causative agents of phaeohyphomycosis in immuno-compromised patients or airborne allergens. Because of the significant negative health effects of Alternaria on humans and their surroundings, a correct and rapid identification of Alternaria species would be of great value to researchers, medical mycologists and the public alike.
Alternaria was originally described by Nees (1816), based on A. tenuis as the only species. Characteristics of the genus included the production of dark-coloured phaeodictyospores in chains, and a beak of tapering apical cells. Von Keissler (1912) synonymised both A. tenuis and Torula alternata (Fries 1832) with Alternaria alternata, due to ambiguities in Nees’s description of A. tenuis. Two additional genera, Stemphylium (Wallroth 1833) and Ulocladium (Preuss 1851) were subsequently described for phaeodictyosporic hyphomycetes, further complicating the taxonomic resolution in this group of fungi. Several re-descriptions and revised criteria of these genera (Saccardo 1886, Elliot 1917, Wiltshire 1933, 1938, Joly 1964) resulted in a growing number of new species. Results of a lifetime study on Alternaria taxonomy based upon morphological characteristics were summarised in Simmons (2007), in which 275 Alternaria species were recognised. One species was transferred to the genus Prathoda and three new genera, Alternariaster, Chalastospora and Teretispora, were segregated from Alternaria.
Molecular studies revealed multiple non-monophyletic genera within the Alternaria complex and Alternaria species clades, which do not always correlate to species-groups based upon morphological characteristics (Pryor & Gilbertson 2000, Chou & Wu 2002, de Hoog & Horré 2002, Pryor & Bigelow 2003, Hong et al. 2005, Inderbitzin et al. 2006, Pryor et al. 2009, Runa et al. 2009, Wang et al. 2011, Lawrence et al. 2012). The A. alternata, A. brassicicola, A. infectoria, A. porri and A. radicina species-groups were strongly supported by these studies and two new species-groups, A. sonchi (Hong et al. 2005) and A. alternantherae (Lawrence et al. 2012) and three new genera, Crivellia (Inderbitzin et al. 2006), Undifilum (Pryor et al. 2009) and Sinomyces (Wang et al. 2011), were described. The latest molecular revision of Alternaria (Lawrence et al. 2013) introduced two new species groups, A. panax and A. gypsophilae, and elevated eight species-groups to sections within Alternaria. The sexual phylogenetic Alternaria lineage, the A. infectoria species-group, did not get the status of section, in contrast to the eight asexual phylogenetic lineages in Alternaria. The Alternaria complex currently comprises the genera Alternaria, Chalastospora (Simmons 2007), Crivellia, Embellisia, Nimbya, Stemphylium, Ulocladium, Undifilum and the recently described Sinomyces together with eight sections of Alternaria and the A. infectoria species-group.
The aim of the present study was to delineate the phylogenetic lineages within Alternaria and allied genera, and to create a robust taxonomy. Phylogenetic inferences were conducted on sequence data of parts of the 18S nrDNA (SSU), 28S nrDNA (LSU), the internal transcribed spacer regions 1 and 2 and intervening 5.8S nrDNA (ITS), glyceraldehyde-3-phosphate dehydrogenase (GAPDH), RNA polymerase second largest subunit (RPB2) and translation elongation factor 1-alpha (TEF1) gene regions of ex-type and reference strains of Alternaria species and all available allied genera.
MATERIAL AND METHODS
Isolates
Based on the ITS sequences of all ex-type or representative strains from the Alternaria identification manual present at the CBS-KNAW Fungal Biodiversity Centre (CBS), Utrecht, The Netherlands (data not shown), 66 Alternaria strains were included in this study together with 61 ex-type or representative strains of 16 related genera (Table 1). Alternaria is represented by the ex-type or representative strains of the seven species-groups and species that clustered outside known Alternaria clades. Because of the size and complexity of the A. alternata, A. infectoria and A. porri species-groups, we only included known species; the complete species-groups will be treated in future studies.
Table 1.
Isolates used in this study and their GenBank accession numbers. Bold accession numbers were generated in other studies.
|
Old species name |
New species name |
Alternaria Section |
Strain
number1 |
Status2 | Host / Substrate | Country |
Other collection
number1 |
GenBank accession numbers |
|||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| SSU | LSU | RPB2 | ITS | GAPDH | TEF1 | ||||||||
| Alternaria alternantherae | Alternaria alternantherae | Althernantherae | CBS 124392 | Solanum melongena | China | HSAUP2798 | KC584506 | KC584251 | KC584374 | KC584179 | KC584096 | KC584633 | |
| Alternaria alternata | Alternaria alternata | Alternata | CBS 916.96 | T | Arachis hypogaea | India | EGS 34.016 | KC584507 | DQ678082 | KC584375 | AF347031 | AY278808 | KC584634 |
| Alternaria anigozanthi | Alternaria anigozanthi | Eureka | CBS 121920 | T | Anigozanthus sp. | Australia | EGS 44.066 | KC584508 | KC584252 | KC584376 | KC584180 | KC584097 | KC584635 |
| Alternaria arborescens | Alternaria arborescens | Alternata | CBS 102605 | T | Lycopersicon esculentum | USA | EGS 39.128 | KC584509 | KC584253 | KC584377 | AF347033 | AY278810 | KC584636 |
| Alternaria argyranthemi | Alternaria argyranthemi | CBS 116530 | T | Argyranthemum sp. | New Zealand | EGS 44.033 | KC584510 | KC584254 | KC584378 | KC584181 | KC584098 | KC584637 | |
| Alternaria armoraciae | Alternaria armoraciae | Chalastospora | CBS 118702 | T | Armoracia rusticana | New Zealand | EGS 51.064 | KC584511 | KC584255 | KC584379 | KC584182 | KC584099 | KC584638 |
| Alternaria avenicola | Alternaria avenicola | Panax | CBS 121459 | T | Avena sp. | Norway | EGS 50.185 | KC584512 | KC584256 | KC584380 | KC584183 | KC584100 | KC584639 |
| Alternaria axiaeriisporifera | Alternaria axiaeriisporifera | Gypsophilae | CBS 118715 | T | Gypsophila paniculata | New Zealand | EGS 51.066 | KC584513 | KC584257 | KC584381 | KC584184 | KC584101 | KC584640 |
| Alternaria brassicae | Alternaria brassicae | CBS 116528 | R | Brassica oleracea | USA | EGS 38.032 | KC584514 | KC584258 | KC584382 | KC584185 | KC584102 | KC584641 | |
| Alternaria brassicicola | Alternaria brassicicola | Brassicicola | CBS 118699 | R | Brassica oleracea | USA | EGS 42.002; ATCC 96836 | KC584515 | KC584259 | KC584383 | JX499031 | KC584103 | KC584642 |
| Alternaria calycipyricola | Alternaria calycipyricola | Panax | CBS 121545 | T | Pyrus communis | China | EGS 52.071; RGR 96.0209 | KC584516 | KC584260 | KC584384 | KC584186 | KC584104 | KC584643 |
| Alternaria capsici-annui | Alternaria capsici-annui | Ulocladium | CBS 504.74 | Capsicum annuum | - | KC584517 | KC584261 | KC584385 | KC584187 | KC584105 | KC584644 | ||
| Alternaria carotiincultae | Alternaria carotiincultae | Radicina | CBS 109381 | T | Daucus carota | USA | EGS 26.010 | KC584518 | KC584262 | KC584386 | KC584188 | KC584106 | KC584645 |
| Alternaria cheiranthi | Alternaria cheiranthi | Cheiranthus | CBS 109384 | R | Cheiranthus cheiri | Italy | EGS 41.188 | KC584519 | KC584263 | KC584387 | AF229457 | KC584107 | KC584646 |
| Alternaria chlamydospora | Alternaria chlamydospora | Phragmosporae | CBS 491.72 | T | Soil | Egypt | EGS 31.060; ATCC 28045; IMI 156427 | KC584520 | KC584264 | KC584388 | KC584189 | KC584108 | KC584647 |
| Alternaria cinerariae | Alternaria cinerariae | Sonchi | CBS 116495 | R | Ligularia sp. | USA | EGS 49.102 | KC584521 | KC584265 | KC584389 | KC584190 | KC584109 | KC584648 |
| Alternaria conjuncta | Alternaria conjuncta | Infectoriae | CBS 196.86 | T | Pastinaca sativa | Switzerland | EGS 37.139 | KC584522 | KC584266 | KC584390 | FJ266475 | AY562401 | KC584649 |
| Alternaria cumini | Alternaria cumini | Eureka | CBS 121329 | T | Cuminum cyminum | India | EGS 04.158a | KC584523 | KC584267 | KC584391 | KC584191 | KC584110 | KC584650 |
| Alternaria dauci | Alternaria dauci | Porri | CBS 117097 | R | Daucus carota | USA | EGS 46.006 | KC584524 | KC584268 | KC584392 | KC584192 | KC584111 | KC584651 |
| Alternaria daucifolii | Alternaria daucifolii | Alternata | CBS 118812 | T | Daucus carota | USA | EGS 37.050 | KC584525 | KC584269 | KC584393 | KC584193 | KC584112 | KC584652 |
| Alternaria dianthicola | Alternaria dianthicola | Dianthicola | CBS 116491 | R | Dianthus × allwoodii | New Zealand | EGS 51.022 | KC584526 | KC584270 | KC584394 | KC584194 | KC584113 | KC584653 |
| Alternaria elegans | Alternaria elegans | Dianthicola | CBS 109159 | T | Lycopersicon esculentum | Burkina Faso | EGS 45.072; IMI 374542 | KC584527 | KC584271 | KC584395 | KC584195 | KC584114 | KC584654 |
| Alternaria ellipsoidea | Alternaria ellipsoidea | Gypsophilae | CBS 119674 | T | Dianthus barbatus | USA | EGS 49.104 | KC584528 | KC584272 | KC584396 | KC584196 | KC584115 | KC584655 |
| Alternaria eryngii | Alternaria eryngii | Panax | CBS 121339 | R | Eryngium sp. | - | EGS 41.005 | KC584529 | KC584273 | KC584397 | JQ693661 | AY562416 | KC584656 |
| Alternaria ethzedia | Alternaria ethzedia | Infectoriae | CBS 197.86 | T | Brassica napus | Switzerland | EGS 37.143 | KC584530 | KC584274 | KC584398 | AF392987 | AY278795 | KC584657 |
| Alternaria gaisen | Alternaria gaisen | Alternata | CBS 632.93 | R | Pyrus pyrifolia cv. Nijiseiki | Japan | EGS 90.512 | KC584531 | KC584275 | KC584399 | KC584197 | KC584116 | KC584658 |
| Alternaria geniostomatis | Alternaria geniostomatis | Eureka | CBS 118701 | T | Geniostoma sp. | New Zealand | EGS 51.061 | KC584532 | KC584276 | KC584400 | KC584198 | KC584117 | KC584659 |
| Alternaria gypsophilae | Alternaria gypsophilae | Gypsophilae | CBS 107.41 | T | Gypsophila elegans | - | EGS 07.025; IMI 264349 | KC584533 | KC584277 | KC584401 | KC584199 | KC584118 | KC584660 |
| Alternaria helianthiinficiens | Alternaria helianthiinficiens | CBS 117370 | R | Helianthus annuus | UK | EGS 50.174; IMI 388636 | KC584534 | KC584278 | KC584402 | KC584200 | KC584119 | KC584661 | |
| Alternaria helianthiinficiens | Alternaria helianthiinficiens | CBS 208.86 | T | Helianthus annuus | USA | EGS 36.184 | KC584535 | KC584279 | KC584403 | JX101649 | KC584120 | EU130548 | |
| Alternaria infectoria | Alternaria infectoria | Infectoriae | CBS 210.86 | T | Triticum aestivum | UK | EGS 27.193 | KC584536 | KC584280 | KC584404 | DQ323697 | AY278793 | KC584662 |
| Alternaria japonica | Alternaria japonica | Japonicae | CBS 118390 | R | Brassica chinensis | USA | EGS 50.099 | KC584537 | KC584281 | KC584405 | KC584201 | KC584121 | KC584663 |
| Alternaria juxtiseptata | Alternaria juxtiseptata | Gypsophilae | CBS 119673 | T | Gypsophila paniculata | Australia | EGS 44.015; DAR 43414 | KC584538 | KC584282 | KC584406 | KC584202 | KC584122 | KC584664 |
| Alternaria limaciformis | Alternaria limaciformis | Phragmosporae | CBS 481.81 | T | Soil | UK | EGS 07.086; IMI 052976; QM 1790 | KC584539 | KC584283 | KC584407 | KC584203 | KC584123 | KC584665 |
| Alternaria limoniasperae | Alternaria limoniasperae | Alternata | CBS 102595 | T | Citrus jambhiri | USA | EGS 45.100 | KC584540 | KC584284 | KC584408 | FJ266476 | AY562411 | KC584666 |
| Alternaria longipes | Alternaria longipes | Alternata | CBS 540.94 | R | Nicotiana tabacum | USA | EGS 30.033; QM 9589 | KC584541 | KC584285 | KC584409 | AY278835 | AY278811 | KC584667 |
| Alternaria macrospora | Alternaria macrospora | Porri | CBS 117228 | T | Gossypium barbadense | USA | EGS 50.190 | KC584542 | KC584286 | KC584410 | KC584204 | KC584124 | KC584668 |
| Alternaria mimicula | Alternaria mimicula | Brassicicola | CBS 118696 | T | Lycopersicon esculentum | USA | EGS 01.056; QM 26a | KC584543 | KC584287 | KC584411 | FJ266477 | AY562415 | KC584669 |
| Alternaria molesta | Alternaria molesta | Phragmosporae | CBS 548.81 | T | Phocaena phocaena | Denmark | EGS 32.075 | KC584544 | KC584288 | KC584412 | KC584205 | KC584125 | KC584670 |
| Alternaria mouchaccae | Alternaria mouchaccae | Phragmosporae | CBS 119671 | T | Soil | Egypt | EGS 31.061 | KC584545 | KC584289 | KC584413 | KC584206 | AY562399 | KC584671 |
| Alternaria nepalensis | Alternaria nepalensis | Japonicae | CBS 118700 | T | Brassica sp. | Nepal | EGS 45.073; IMI 374543 | KC584546 | KC584290 | KC584414 | KC584207 | KC584126 | KC584672 |
| Alternaria nobilis | Alternaria nobilis | Gypsophilae | CBS 116490 | R | Dianthus caryophyllus | New Zealand | EGS 51.027; NZMAF Lynfield 743 | KC584547 | KC584291 | KC584415 | KC584208 | KC584127 | KC584673 |
| Alternaria oregonensis | Alternaria oregonensis | Infectoriae | CBS 542.94 | T | Triticum aestivum | USA | EGS 29.194 | KC584548 | KC584292 | KC584416 | FJ266478 | FJ266491 | KC584674 |
| Alternaria panax | Alternaria panax | Panax | CBS 482.81 | R | Aralia racemosa | USA | EGS 29.180 | KC584549 | KC584293 | KC584417 | KC584209 | KC584128 | KC584675 |
| Alternaria perpunctulata | Alternaria perpunctulata | Althernantherae | CBS 115267 | T | Alternanthera philoxeroides | USA | KC584550 | KC584294 | KC584418 | KC584210 | KC584129 | KC584676 | |
| Alternaria petroselini | Alternaria petroselini | Radicina | CBS 112.41 | T | Petroselinum sativum | - | EGS 06.196 | KC584551 | KC584295 | KC584419 | KC584211 | KC584130 | KC584677 |
| Alternaria photistica | Alternaria photistica | Panax | CBS 212.86 | T | Digitalis purpurea | UK | EGS 35.172 | KC584552 | KC584296 | KC584420 | KC584212 | KC584131 | KC584678 |
| Alternaria porri | Alternaria porri | Porri | CBS 116698 | R | Allium cepa | USA | EGS 48.147 | KC584553 | KC584297 | KC584421 | DQ323700 | KC584132 | KC584679 |
| Alternaria pseudorostrata | Alternaria pseudorostrata | Porri | CBS 119411 | T | Euphorbia pulcherrima | USA | EGS 42.060 | KC584554 | KC584298 | KC584422 | JN383483 | AY562406 | KC584680 |
| Alternaria radicina | Alternaria radicina | Radicina | CBS 245.67 | T | Daucus carota | USA | EGS 03.145; ATCC 6503; IMI 124939; QM 1301; QM 6503 | KC584555 | KC584299 | KC584423 | KC584213 | KC584133 | KC584681 |
| “Alternaria resedae” | Alternaria sp. | Cheiranthus | CBS 115.44 | Reseda odorata | - | EGS 07.030 | KC584556 | KC584300 | KC584424 | KC584214 | KC584134 | KC584682 | |
| Alternaria saponariae | Alternaria saponariae | Gypsophilae | CBS 116492 | R | Saponaria officinalis | USA | EGS 49.199 | KC584557 | KC584301 | KC584425 | KC584215 | KC584135 | KC584683 |
| Alternaria selini | Alternaria selini | Radicina | CBS 109382 | T | Petroselinum crispum | Saudi Arabia | EGS 25.198; IMI 137332 | KC584558 | KC584302 | KC584426 | AF229455 | AY278800 | KC584684 |
| Alternaria septorioides | Alternaria septorioides | Brassicicola | CBS 106.41 | T | Reseda odorata | Netherlands | EGS 52.089; MUCL 20298 | KC584559 | KC584303 | KC584427 | KC584216 | KC584136 | KC584685 |
| Alternaria simsimi | Alternaria simsimi | Dianthicola | CBS 115265 | T | Sesamum indicum | Argentina | EGS 13.110 | KC584560 | KC584304 | KC584428 | JF780937 | KC584137 | KC584686 |
| Alternaria smyrnii | Alternaria smyrnii | Radicina | CBS 109380 | R | Smyrnium olusatrum | UK | EGS 37.093 | KC584561 | KC584305 | KC584429 | AF229456 | KC584138 | KC584687 |
| Alternaria solani | Alternaria solani | Porri | CBS 116651 | R | Solanum tuberosum | USA | EGS 45.020 | KC584562 | KC584306 | KC584430 | KC584217 | KC584139 | KC584688 |
| Alternaria soliaridae | Alternaria soliaridae | CBS 118387 | T | Soil | USA | EGS 33.024 | KC584563 | KC584307 | KC584431 | KC584218 | KC584140 | KC584689 | |
| Alternaria solidaccana | Alternaria solidaccana | Brassicicola | CBS 118698 | T | Soil | Bangladesh | EGS 36.158; IMI 049788 | KC584564 | KC584308 | KC584432 | KC584219 | KC584141 | KC584690 |
| Alternaria sonchi | Alternaria sonchi | Sonchi | CBS 119675 | R | Sonchus asper | Canada | EGS 43.131; IMI 366167 | KC584565 | KC584309 | KC584433 | KC584220 | KC584142 | KC584691 |
| Alternaria tagetica | Alternaria tagetica | Porri | CBS 479.81 | R | Tagetes erecta | UK | EGS 33.081 | KC584566 | KC584310 | KC584434 | KC584221 | KC584143 | KC584692 |
| Alternaria tenuissima | Alternaria tenuissima | Alternata | CBS 918.96 | R | Dianthus sp. | UK | EGS 34.015 | KC584567 | KC584311 | KC584435 | AF347032 | AY278809 | KC584693 |
| Alternaria thalictrigena | Alternaria thalictrigena | CBS 121712 | T | Thalictrum sp. | Germany | KC584568 | KC584312 | KC584436 | EU040211 | KC584144 | KC584694 | ||
| Alternaria triglochinicola | Alternaria triglochinicola | Eureka | CBS 119676 | T | Triglochin procera | Australia | EGS 41.070 | KC584569 | KC584313 | KC584437 | KC584222 | KC584145 | KC584695 |
| Alternaria vaccariae | Alternaria vaccariae | Gypsophilae | CBS 116533 | R | Vaccaria hispanica | USA | EGS 47.108 | KC584570 | KC584314 | KC584438 | KC584223 | KC584146 | KC584696 |
| Alternaria vaccariicola | Alternaria vaccariicola | Gypsophilae | CBS 118714 | T | Vaccaria hispanica | USA | EGS 46.003; ATCC 26038 | KC584571 | KC584315 | KC584439 | KC584224 | KC584147 | KC584697 |
| Alternariaster helianthi | Alternariaster helianthi | CBS 119672 | R | Helianthus sp. | USA | EGS 36.007 | KC584626 | KC584368 | KC584493 | ||||
| Alternariaster helianthi | Alternariaster helianthi | CBS 327.69 | Helianthus annuus | - | KC584627 | KC584369 | KC584494 | ||||||
| Ascochyta pisi | Ascochyta pisi | CBS 126.54 | Pisum sativum | Netherlands | EU754038 | DQ678070 | DQ677967 | ||||||
| Boeremia exigua | Boeremia exigua | CBS 431.74 | Solanum tuberosum | Netherlands | PD 74/2447 | EU754084 | EU754183 | GU371780 | |||||
| Brachycladium papaveris | Alternaria papavericola | Crivellia | CBS 116606 | T | Papaver somniferum | USA | KC584579 | KC584321 | KC584446 | FJ357310 | FJ357298 | KC584705 | |
| Brachycladium penicillatum | Alternaria penicillata | Crivellia | CBS 116608 | T | Papaver rhoeas | Austria | DAOM 230457 | KC584572 | KC584316 | KC584440 | FJ357311 | FJ357299 | KC584698 |
| Chaetodiplodia sp. | Chaetodiplodia sp. | CBS 453.68 | Halimione portulacoides | Netherlands | DQ678001 | DQ678054 | KC584499 | ||||||
| Chaetosphaeronema hispidulum | Chaetosphaeronema hispidulum | CBS 216.75 | Anthyllis vulneraria | Germany | EU754045 | EU754144 | GU371777 | ||||||
| Chalastospora cetera | Alternaria cetera | Chalastospora | CBS 121340 | T | Elymus scabrus | Australia | EGS 41.072 | KC584573 | KC584317 | KC584441 | JN383482 | AY562398 | KC584699 |
| Chalastospora ellipsoidea | Alternaria breviramosa | Chalastospora | CBS 121331 | T | Triticum sp. | Australia | KC584574 | KC584318 | KC584442 | FJ839608 | KC584148 | KC584700 | |
| Chalastospora obclavata | Alternaria obclavata | Chalastospora | CBS 124120 | T | Air | USA | EGS 12.128 | KC584575 | FJ839651 | KC584443 | KC584225 | KC584149 | KC584701 |
| Chmelia slovaca | Alternaria slovaca | Infectoriae | CBS 567.66 | T | Human | Slovakia | ATCC 24279 | KC584576 | KC584319 | KC584444 | KC584226 | KC584150 | KC584702 |
| Clathrospora elynae | Clathrospora elynae | CBS 161.51 | Carex curvula | Switzerland | KC584628 | KC584370 | KC584495 | ||||||
| Clathrospora elynae | Clathrospora elynae | CBS 196.54 | Carex curvula | Switzerland | KC584629 | KC584371 | KC584496 | ||||||
| Clathrospora heterospora | Alternaria sp. | Alternata | CBS 175.52 | Juncus mertensianus | USA | EGS 35.1619; IMI 068085; QM 1277 | KC584577 | KC584320 | KC584445 | KC584227 | KC584151 | KC584703 | |
| Cochliobolus heterostrophus | Cochliobolus heterostrophus | CBS 134.39 | Zea mays | - | DSM 1149 | AY544727 | AY544645 | DQ247790 | |||||
| Cochliobolus sativus | Cochliobolus sativus | DAOM 226212 | Hordeum vulgare | Canada | DQ677995 | DQ678045 | DQ677939 | ||||||
| Comoclathris magna | Alternaria sp. | Alternata | CBS 174.52 | Anemone occidentalis | USA | EGS 39.1613; IMI 068086; QM 1278 | KC584578 | DQ678068 | DQ677964 | KC584228 | KC584152 | KC584704 | |
| Comoclathris compressa | Comoclathris compressa | CBS 156.53 | Castilleja miniata | USA | EGS No. C-20285-I | KC584630 | KC584372 | KC584497 | |||||
| Comoclathris compressa | Comoclathris compressa | CBS 157.53 | Ligusticum purpureum | USA | EGS No. 1952a-1633 | KC584631 | KC584373 | KC584498 | |||||
| Coniothyrium palmarum | Coniothyrium palmarum | CBS 400.71 | Chamaerops humilis | Italy | EU754054 | EU754153 | DQ677956 | ||||||
| Crivellia papaveracea | Alternaria penicillata | Crivellia | CBS 116607 | T | Papaver rhoeas | Austria | DAOM 230456 | KC584580 | KC584322 | KC584447 | KC584229 | KC584153 | KC584706 |
| Dendryphiella arenariae | Paradendryphiella arenariae | CBS 181.58 | T | Coastal sand | France | DAOM 63738; IMI 067735; MUCL 4129 | KC793336 | KC793338 | DQ470924 | ||||
| Dendryphiella salina | Paradendryphiella salina | CBS 142.60 | Spartina sp. | UK | MUCL 9639 | KC793337 | KC793339 | KC793340 | |||||
| Embellisia abundans | Alternaria abundans | Chalastospora | CBS 534.83 | T | Fragaria sp. | New Zealand | EGS 29.159 | KC584581 | KC584323 | KC584448 | JN383485 | KC584154 | KC584707 |
| Embellisia allii | Alternaria embellisia | Embellisia | CBS 339.71 | R | Allium sativum | USA | ATCC 22412; IMI 155707; MUCL 18571; QM 8609 | KC584582 | KC584324 | KC584449 | KC584230 | KC584155 | KC584708 |
| Embellisia annulata | Cicatricea salina | CBS 302.84 | T | Cancer pagurus | North Sea, Skagerrak | KC584583 | KC584325 | KC584450 | JN383486 | JN383467 | KC584709 | ||
| Embellisia chlamydospora | Alternaria chlamydosporigena | Embellisia | CBS 341.71 | R | Air | USA | EGS 10.073; ATCC 22409; IMI 155709; MUCL 18573; QM 7287 | KC584584 | KC584326 | KC584451 | KC584231 | KC584156 | KC584710 |
| Embellisia conoidea | Alternaria conoidea | Brassicicola | CBS 132.89 | Ricinus communis | Saudi Arabia | KC584585 | KC584327 | KC584452 | AF348226 | FJ348227 | KC584711 | ||
| Embellisia dennisii | Alternaria dennisii | CBS 110533 | Senecio jacobaea | New Zealand | KC584586 | KC584328 | KC584453 | KC584232 | KC584157 | KC584712 | |||
| Embellisia dennisii | Alternaria dennisii | CBS 476.90 | T | Senecio jacobaea | Isle of Man | IMI 151744 | KC584587 | KC584329 | KC584454 | JN383488 | JN383469 | KC584713 | |
| Embellisia didymospora | Alternaria didymospora | Phragmosporae | CBS 766.79 | Seawater | Adriatic Sea | KC584588 | KC584330 | KC584455 | FJ357312 | FJ357300 | KC584714 | ||
| Embellisia eureka | Alternaria eureka | Eureka | CBS 193.86 | T | Medicago rugosa | Australia | IMI 273162 | KC584589 | KC584331 | KC584456 | JN383490 | JN383471 | KC584715 |
| Embellisia hyacinthi | Alternaria hyacinthi | Embellisioides | CBS 416.71 | T | Hyacinthus orientalis | Netherlands | EGS 19.102; IMI 279179 | KC584590 | KC584332 | KC584457 | KC584233 | KC584158 | KC584716 |
| Embellisia indefessa | Alternaria indefessa | Cheiranthus | CBS 536.83 | T | Soil | USA | EGS 30.195 | KC584591 | KC584333 | KC584458 | KC584234 | KC584159 | KC584717 |
| Embellisia leptinellae | Alternaria leptinellae | Eureka | CBS 477.90 | T | Leptinella dioica | New Zealand | EGS 39.101 | KC584592 | KC584334 | KC584459 | KC584235 | KC584160 | KC584718 |
| Embellisia lolii | Alternaria lolii | Embellisioides | CBS 115266 | T | Lolium perenne | New Zealand | KC584593 | KC584335 | KC584460 | JN383492 | JN383473 | KC584719 | |
| Embellisia novaezelandiae | Alternaria botryospora | Embellisioides | CBS 478.90 | T | Leptinella dioica | New Zealand | EGS 39.099 | KC584594 | KC584336 | KC584461 | AY278844 | AY278831 | KC584720 |
| Embellisia phragmospora | Alternaria phragmospora | Phragmosporae | CBS 274.70 | T | Soil | The netherlands | EGS 27.098; ATCC 18914 | KC584595 | KC584337 | KC584462 | JN383493 | JN383474 | KC584721 |
| Embellisia planifunda | Alternaria planifunda | Embellisioides | CBS 537.83 | T | Triticum aestivum | Australia | IMI 115034 | KC584596 | KC584338 | KC584463 | FJ357315 | FJ357303 | KC584722 |
| Embellisia proteae | Alternaria proteae | Embellisioides | CBS 475.90 | T | Protea sp. | Australia | IMI 320290; IMI 341684 | KC584597 | KC584339 | KC584464 | AY278842 | KC584161 | KC584723 |
| Embellisia tellustris | Alternaria tellustris | Embellisia | CBS 538.83 | T | Soil | USA | EGS 33.026 | KC584598 | KC584340 | KC584465 | FJ357316 | AY562419 | KC584724 |
| Embellisia tumida | Alternaria tumida | Embellisioides | CBS 539.83 | T | Triticum aestivum | Australia | KC584599 | KC584341 | KC584466 | FJ266481 | FJ266493 | KC584725 | |
| Heterospora chenopodii | Heterospora chenopodii | CBS 115.96 | Chenopodium album | Netherlands | PD 94/1576 | EU754089 | EU754188 | GU371775 | |||||
| Julella avicenniae | Julella avicenniae | BCC 18422 | Mangrove wood | Thailand | GU371831 | GU371823 | GU371787 | ||||||
| Leptosphaerulina australis | Leptosphaerulina australis | CBS 317.83 | Eugenia aromatica | Indonesia | GU296160 | GU301830 | GU371790 | ||||||
| Loratospora aestuarii | Loratospora aestuarii | JK 5535B | Juncus roemerianus | USA | GU296168 | GU301838 | GU371760 | ||||||
| Neophaeosphaeria filamentosa | Neophaeosphaeria filamentosa | CBS 102202 | Yucca rostrata | Mexico | GQ387516 | GQ387577 | GU371773 | ||||||
| Nimbya caricis | Alternaria caricis | Nimbya | CBS 480.90 | T | Carex hoodii | USA | EGS 13.094 | KC584600 | KC584342 | KC584467 | AY278839 | AY278826 | KC584726 |
| “Nimbya gomphrenae” | Alternaria sp. | Alternata | CBS 108.27 | Gomphrena globosa | - | KC584601 | KC584343 | KC584468 | KC584236 | KC584162 | KC584727 | ||
| Nimbya scirpicola | Alternaria scirpicola | Nimbya | CBS 481.90 | R | Scirpus sp. | UK | EGS 19.042 | KC584602 | KC584344 | KC584469 | KC584237 | KC584163 | KC584728 |
| Ophiosphaerella herpotricha | Ophiosphaerella herpotricha | CBS 620.86 | Bromus erectus | Switzerland | ETH 9373 | DQ678010 | DQ678062 | DQ677958 | |||||
| Paraleptosphaeria dryadis | Paraleptosphaeria dryadis | CBS 643.86 | Dryas octopetala | Switzerland | ETH 9446 | KC584632 | GU301828 | GU371733 | |||||
| Peyronellaea glomerata | Peyronellaea glomerata | CBS 528.66 | Chrysanthemum sp. | Netherlands | PD 63/590 | EU754085 | EU754184 | GU371781 | |||||
| Peyronellaea zeaemaydis | Peyronellaea zeaemaydis | CBS 588.69 | T | Zea mays | USA | EU754093 | EU754192 | GU371782 | |||||
| Phaeosphaeria ammophilae | Phaeosphaeria ammophilae | CBS 114595 | Ammophila arenaria | Sweden | UPSC 3568 | GU296185 | GU304859 | GU371724 | |||||
| Phaeosphaeria avenaria | Phaeosphaeria avenaria | DAOM 226215 | Avena sativa | Canada | OSC 100096 | AY544725 | AY544684 | DQ677941 | |||||
| Phaeosphaeria eustoma | Phaeosphaeria eustoma | CBS 573.86 | Dactylis glomerata | Switzerland | ETH 9239 | DQ678011 | DQ678063 | DQ677959 | |||||
| Phoma complanata | Phoma complanata | CBS 268.92 | Anglica sylvestris | Netherlands | PD 75/3 | EU754081 | EU754180 | GU371778 | |||||
| Phoma herbarum | Phoma herbarum | CBS 276.37 | Wood pulp | Sweden | DQ678014 | DQ678066 | DQ677962 | ||||||
| Plenodomus lingam | Plenodomus lingam | DAOM 229267 | Brassica sp. | France | DQ470993 | DQ470946 | DQ470894 | ||||||
| Pleospora betae | Pleospora betae | CBS 109410 | Beta vulgaris | Netherlands | PD 77/113 | EU754079 | EU754178 | GU371774 | |||||
| Pleospora calvescens | Pleospora calvescens | CBS 246.79 | Atriplex hastata | Germany | PD 77/655 | EU754032 | EU754131 | KC584500 | |||||
| Pleospora chenopodii | Pleospora chenopodii | CBS 206.80 | Chenopodium quinoa | Bolivia | PD 74/1022 | JF740095 | JF740266 | KC584501 | |||||
| Pleospora fallens | Pleospora fallens | CBS 161.78 | Olea europaea | New Zealand | GU238215 | GU238074 | KC584502 | ||||||
| Pleospora halimiones | Pleospora halimiones | CBS 432.77 | Halimione portulacoides | Netherlands | IMI 282137 | JF740096 | JF740267 | KC584503 | |||||
| Pleospora incompta | Pleospora incompta | CBS 467.76 | Olea europaea | Greece | GU23822 | GU238087 | KC584504 | ||||||
| Pleospora tarda | Pleospora tarda | CBS 714.68 | T | Medicago sativa | Canada | EGS 04.118C; IMI 135456; MUCL 11717; QM 1379 | KC584603 | KC584345 | AF107804 | KC584238 | AF443881 | KC584729 | |
| Pleospora typhicola | Pleospora typhicola | CBS 132.69 | Typha angustifolia | Netherlands | JF740105 | JF740325 | KC584505 | ||||||
| Pyrenochaeta nobilis | Pyrenochaeta nobilis | CBS 407.76 | T | Laurus nobilis | Italy | EU754107 | DQ678096 | DQ677991 | |||||
| Pyrenophora phaeocomes | Pyrenophora phaeocomes | DAOM 222769 | Calamagrostis villosa | Switzerland | DQ499595 | DQ499596 | DQ497614 | ||||||
| Saccothecium sepincola | Saccothecium sepincola | CBS 278.32 | Ribes nigrum | USA | GU296195 | GU301870 | GU371745 | ||||||
| Setomelanomma holmii | Setomelanomma holmii | CBS 110217 | Picea pungens | USA | GU296196 | GQ37633 | GU371800 | ||||||
| Sinomyces alternariae | Alternaria alternariae | Ulocladium | CBS 126989 | T | Daucus carota | USA | EGS 46.004 | KC584604 | KC584346 | KC584470 | AF229485 | AY278815 | KC584730 |
| Stemphylium herbarum | Stemphylium herbarum | CBS 191.86 | T | Medicago sativa | India | EGS 36.138; IMI 276975 | GU238232 | GU238160 | KC584471 | KC584239 | AF443884 | KC584731 | |
| Teretispora leucanthemi | Alternaria leucanthemi | Teretispora | CBS 421.65 | T | Chrysanthemum maximum | Netherlands | ATCC 16028; IFO 9085; IMI 111986; QM 7227 | KC584605 | KC584347 | KC584472 | KC584240 | KC584164 | KC584732 |
| Teretispora leucanthemi | Alternaria leucanthemi | CBS 422.65 | R | Chrysanthemum maximum | USA | EGS 17.063; ATCC 16029; IMI 111987; QM 8579 | KC584606 | KC584348 | KC584473 | KC584241 | KC584165 | KC584733 | |
| Ulocladium arborescens | Alternaria aspera | Pseudoulocladium | CBS 115269 | T | Pistacia vera | Japan | IMI 369777 | KC584607 | KC584349 | KC584474 | KC584242 | KC584166 | KC584734 |
| Ulocladium atrum | Alternaria atra | Ulocladioides | CBS 195.67 | T | Soil | USA | ATCC 18040; IMI 124944; QM 8408 | KC584608 | KC584350 | KC584475 | AF229486 | KC584167 | KC584735 |
| Ulocladium botrytis | Alternaria botrytis | Ulocladium | CBS 197.67 | T | Contaminant | USA | ATCC 18042; IMI 124942; MUCL 18556; QM 7878 | KC584609 | KC584351 | KC584476 | KC584243 | KC584168 | KC584736 |
| Ulocladium botrytis | Alternaria sp. | Ulocladioides | CBS 198.67 | R | Soil | USA | ATCC 18043; IMI 124949; MUCL 18557; QM 8619 | KC584610 | KC584352 | KC584477 | AF229487 | KC584169 | KC584737 |
| Ulocladium brassicae | Alternaria brassicaepekinensis | Ulocladioides | CBS 121493 | T | Brassica pekinensis | China | HSAUPwy0037 | KC584611 | KC584353 | KC584478 | KC584244 | KC584170 | KC584738 |
| Ulocladium cantlous | Alternaria cantlous | Ulocladioides | CBS 123007 | T | Cucumis melo | China | HSAUP0209 | KC584612 | KC584354 | KC584479 | KC584245 | KC584171 | KC584739 |
| Ulocladium capsici | Alternaria concatenata | Pseudoulocladium | CBS 120006 | T | - | - | HSAUPIII00035 | KC584613 | KC584355 | KC584480 | KC584246 | AY762950 | KC584740 |
| Ulocladium chartarum | Alternaria chartarum | Pseudoulocladium | CBS 200.67 | T | Populus sp. | Canada | ATCC 18044; DAOM 59616b; IMI 124943; MUCL 18564; QM 8328 | KC584614 | KC584356 | KC584481 | AF229488 | KC584172 | KC584741 |
| Ulocladium consortiale | Alternaria consortialis | Ulocladioides | CBS 104.31 | T | - | - | KC584615 | KC584357 | KC584482 | KC584247 | KC584173 | KC584742 | |
| Ulocladium cucurbitae | Alternaria cucurbitae | Ulocladioides | CBS 483.81 | R | Cucumis sativus | New Zealand | EGS 31.021; LEV 7067 | KC584616 | KC584358 | KC584483 | FJ266483 | AY562418 | KC584743 |
| Ulocladium multiforme | Alternaria multiformis | Ulocladioides | CBS 102060 | T | Soil | Canada | KC584617 | KC584359 | KC584484 | FJ266486 | KC584174 | KC584744 | |
| Ulocladium obovoideum | Alternaria obovoidea | Ulocladioides | CBS 101229 | Cucumis sativus | New Zealand | KC584618 | KC584360 | KC584485 | FJ266487 | FJ266498 | KC584745 | ||
| Ulocladium oudemansii | Alternaria oudemansii | Ulocladium | CBS 114.07 | T | - | - | ATCC 18047; IMI 124940; MUCL 18563; QM 1744 | KC584619 | KC584361 | KC584486 | FJ266488 | KC584175 | KC584746 |
| Ulocladium septosporum | Alternaria septospora | Pseudoulocladium | CBS 109.38 | Wood | Italy | KC584620 | KC584362 | KC584487 | FJ266489 | FJ266500 | KC584747 | ||
| Ulocladium solani | Alternaria heterospora | Ulocladioides | CBS 123376 | T | Lycopersicon esculentum | China | HSAUP 0521 | KC584621 | KC584363 | KC584488 | KC584248 | KC584176 | KC584748 |
| Ulocladium subcucurbitae | Alternaria subcucurbitae | Ulocladioides | CBS 121491 | T | Chenopodium glaucum | China | KC584622 | KC584364 | KC584489 | KC584249 | EU855803 | KC584749 | |
| Ulocladium tuberculatum | Alternaria terricola | Ulocladioides | CBS 202.67 | T | Soil | USA | ATCC 18048; IMI 124947; MUCL 18560; QM 8614 | KC584623 | KC584365 | KC584490 | FJ266490 | KC584177 | KC584750 |
| Undifilum bornmuelleri | Alternaria bornmuelleri | Undifilum | DAOM 231361 | T | Securigera varia | Austria | DAOM 231361 | KC584624 | KC584366 | KC584491 | FJ357317 | FJ357305 | KC584751 |
| Ybotromyces caespitosus | Alternaria caespitosa | Infectoriae | CBS 177.80 | T | Human | Spain | KC584625 | KC584367 | KC584492 | KC584250 | KC584178 | KC584752 | |
ATCC: American Type Culture Collection, Manassas, VA, USA; BCC: BIOTEC Culture Collection, Thailand; CBS: Culture collection of the Centraalbureau voor Schimmelcultures, Fungal Biodiversity Centre, Utrecht, The Netherlands; DAOM: Canadian Collection of Fungal Cultures, Ottawa, Canada; DAR: Plant Pathology Herbarium, Orange Agricultural Institute, Australia; DSM: German Collection of Microorganisms and Cell Cultures, Leibniz Institute, Braunschweig, Germany; EGS: Personal collection of Dr. E.G. Simmons; ETH: Swiss Federal Institute of Technology, Switzerland; HSAUP: Department of Plant Pathology, Shandong Agricultural University, China; IFO: Institute for Fermentation Culture Collection, Osaka, Japan; IMI: Culture collection of CABI EUrope UK Centre, Egham UK; JK: Personal collection of Dr. J. Kohlmeyer; LEV: Plant Health and Diagnostic Station, Levin, New Zealand; MUCL: (Agro)Industrial Fungi and Yeast Collection of the Belgian Co-ordinated Collections of Micro-organisms (BCCM), Louvain-la Neuve, Belgium; NZMAF: New Zealand Ministry of Agriculture and Forestry; OSC: Oregon State University Herbarium, USA; PD: Plant Protection Service, Wageningen, The Netherlands; RGR: Personal collection of Dr. R.G. Roberts; UPSC: Uppsala University Culture Collection, Sweden; QM: Quarter Master Culture Collection, Amherst, MA, USA.
T: ex-type strain; R: representative strain.
Freeze-dried strains were revived in 2 mL malt/peptone (50 % / 50 %) and subsequently transferred to oatmeal agar (OA) (Crous et al. 2009a). Strains of the CBS collection stored in liquid nitrogen were transferred to OA directly from -80 °C. DNA extraction was performed using the UltraClean Microbial DNA Isolation Kit (MoBio laboratories, Carlsbad, CA, USA), according to the manufacturer’s instructions.
Taxonomy
Morphological descriptions were made for isolates grown on synthetic nutrient-poor agar plates (SNA, Nirenberg 1976) with a small piece of autoclaved filter paper placed onto the agar surface. Cultures were incubated at moderate temperatures (∼ 22 °C) under CoolWhite fluorescent light with an 8 h photoperiod for 7 d. The sellotape technique was used for making slide preparations (Crous et al. 2009a) with Shear’s medium as mounting fluid. Photographs of characteristic structures were made with a Nikon Eclipse 80i microscope using differential interference contrast (DIC) illumination. Growth rates were measured after 5 and 7 d. Colony characters were noted after 7 d, colony colours were rated according to Rayner (1970). Nomenclatural data were deposited in MycoBank (Crous et al. 2004).
PCR and sequencing
The SSU region was amplified with the primers NS1 and NS4 (White et al. 1990), the LSU region with LSU1Fd (Crous et al. 2009b) and LR5 (Vilgalys & Hester 1990), the ITS region with V9G (De Hoog & Gerrits van den Ende 1998) and ITS4 (White et al. 1990), the GAPDH region with gpd1 and gpd2 (Berbee et al. 1999), the RPB2 region with RPB2-5F2 (Sung et al. 2007) and fRPB2-7cR (Liu et al. 1999) and the TEF1 gene with the primers EF1-728F and EF1-986R (Carbone & Kohn 1999) or EF2 (O’Donnell et al. 1998). The PCRs were performed in a MyCycler™ Thermal Cycler (Bio-Rad Laboratories B.V., Veenendaal, The Netherlands) in a total volume of 12.5 μL. The SSU and LSU PCR mixtures consisted of 1 μL genomic DNA, 1′ GoTaq® Flexi buffer (Promega, Madison, WI, USA), 2 μM MgCl2, 40 μM of each dNTP, 0.2 μM of each primer and 0.25 Unit GoTaq® Flexi DNA polymerase (Promega). The ITS and GAPDH PCR mixtures differed from the original mix by containing 1 μM MgCl2, the RPB2 and TEF1 PCR mixtures differed from the original mix by containing 2 μL genomic DNA and the RPB2 mixture differed from the original mix by containing 0.5 U instead of 0.25 U GoTaq® Flexi DNA polymerase. Conditions for PCR amplification consisted of an initial denaturation step of 5 min at 94 °C followed by 35 cycles of 30 s at 94 °C, 30 s at 48 °C and 90 s at 72 °C for SSU, LSU, ITS and 40 cycles of 30 s at 94 °C, 30 s at 52 °C / 59 °C and 45 s at 72 °C for TEF1 using respectively EF2 or EF1-986R as reverse primer and a final elongation step of 7 min at 72 °C. The partial RPB2 gene was obtained by using a touchdown PCR protocol of 5 cycles of 45 s at 94 °C, 45 s at 60 °C and 2 min at 72 °C, followed by 5 cycles with a 58 °C annealing temperature and 30 cycles with a 54 °C annealing temperature. The PCR products were sequenced in both directions using the PCR primers and the BigDye Terminator v. 3.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA, USA), according to the manufacturer’s recommendations, and analysed with an ABI Prism 3730XL Sequencer (Applied Biosystems) according to the manufacturer’s instructions. Consensus sequences were computed from forward and reverse sequences using the BioNumerics v. 4.61 software package (Applied Maths, St-Martens-Latem, Belgium). All generated sequences were deposited in GenBank (Table 1).
Phylogenetic analyses
Multiple sequence alignments were generated with MAFFT v. 6.864b (http://mafft.cbrc.jp/alignment/server/index.html), and adjusted by eye. Two different datasets were used to estimate two phylogenies; an Alternaria complex phylogeny and a Pleosporineae family tree. The first tree focusses on the Alternaria complex, the second one was produced to place the genera Comoclathris, Clathrospora and Alternariaster in the context of the Alternaria complex. The relatives of the three genera were determined with standard nucleotide blast searches, with both the SSU and LSU sequences, against the nucleotide database in GenBank. This resulted in a selection of 35 species (Table 1) for which the SSU, LSU and RPB2 sequence data set was present or could be completed. Blast searches with Embellisia annulata gave hits with two marine Dendryphiella species, Dendryphiella arenariae and Dendryphiella salina, which we also included. Phylogenetic analyses of the sequence data consisted of Bayesian and Maximum likelihood analyses of both the individual data partitions as well as the combined aligned dataset. Bayesian analyses were performed with MrBayes v. 3.2.1 (Huelsenbeck & Ronquist 2001, Ronquist & Huelsenbeck 2003). The Markov Chain Monte Carlo (MCMC) analysis used four chains and started from a random tree topology. The sample frequency was set at 100 and the temperature value of the heated chain was 0.1. The temperature value was lowered to 0.05 when the average standard deviation of split frequencies did not fall below 0.01 after 5M generations (RPB2 and Pleosporineae phylogeny). Burn-in was set to 25 % after which the likelihood values were stationary. Maximum likelihood analyses including 500 bootstrap replicates were run using RAxML v. 7.2.6 (Stamatakis & Alachiotis 2010). The online tool Findmodel (http://www.hiv.lanl.gov/content/sequence/findmodel/findmodel.html) was used to determine the best nucleotide substitution model for each partition. For the SSU (Pleosporineae family tree), LSU, ITS, RPB2 and TEF1 partitions a GTR model with a gamma-distributed rate variation was suggested, and for the SSU (Alternaria complex) and GAPDH partitions a TrN model with gamma-distributed rate variation. Sequences of Stemphylium herbarum (CBS 191.86) were used as outgroup in the Alternaria phylogeny and those of Jullella avenicae (BCC 18422) in the Pleosporineae phylogeny. The resulting trees were printed with TreeView v. 1.6.6 (Page 1996) and together with the alignments deposited into TreeBASE (http://www.treebase.org).
RESULTS
Phylogeny
For defining the taxonomy of Alternaria and allied genera, 121 strains were included in the Alternaria complex alignment. The alignment length and unique site patterns of the different genes and gene combinations are stated in Table 2. The original ITS alignment consisted of 577 characters of which the first 78 are excluded as this contained a non-alignable region. In the original TEF1 alignment (375 characters) we coded the major inserts (Table 3), which otherwise would negatively influence the phylogeny, resulting in a TEF1 alignment of 269 characters. All phylogenies, different phylogenetic methods and gene regions or gene combinations used on this dataset (data not shown, trees and alignments lodged in TreeBASE), show a weak support at the deeper nodes of the tree. The only well-supported node (Bayesian posterior probability of 1.0, RAxML Maximum Likelihood support value of 100) in all phylogenies separates Embellisia annulata CBS 302.84 and the Pleospora/Stemphylium clade from the Alternaria complex (Fig. 1). In the Alternaria clade, six monotypic lineages and 24 internal clades occur consistently in the individual and combined phylogenies, although positions vary between the different gene regions or combinations used. The support values for the clades within Alternaria (called sections) are plotted in a heat map (Table 2) per gene and phylogenetic method used. The support values for the different phylogenetic methods vary, with the Bayesian posterior probabilities being higher than the RAxML bootstrap support values (Table 2). The SSU, LSU and ITS phylogenies display a low resolution, which reflects in poor to no support of the sections. Therefore, we chose not to include them in the multi-gene alignments, except in the all-gene alignment. In the GAPDH phylogenies, sect. Cheiranthus, sect. Nimbya and sect. Pseudoulocladium are poorly supported and “A. resedae” clusters separate from sect. Cheiranthus. In the RPB2 phylogenies the support values for sect. Alternata, sect. Embellisioides and sect. Eureka are relatively low; A. cumini clusters in sect. Embellisioides instead of sect. Eureka and U. capsici clusters separate from sect. Pseudoulocladium. The TEF1 phylogenies did not support sect. Nimbya and show relative low support for sect. Cheiranthus, sect. Dianthicola, sect. Embellisioides, sect. Panax, sect. Phragmosporae and sect. Radicina, and A. cumini clusters outside sect. Eureka. In the 2-region phylogenies U. capsici clusters outside sect. Pseudoulocladium based on GAPDH and RPB2, E. indefessa clusters outside sect. Cheiranthus based on GAPDH and TEF1, and sect. Eureka is poorly supported based on RPB2 and TEF1. The combined phylogeny based on the GAPDH, RPB2 and TEF1 sequences (Fig. 1) is displayed, as these are the genes with the best resolution.
Table 2.
Summary of locus and phylogenetic results as well as a heat map of the Bayesian posterior probabilities and RAxML boostrap support values per Alternaria section.
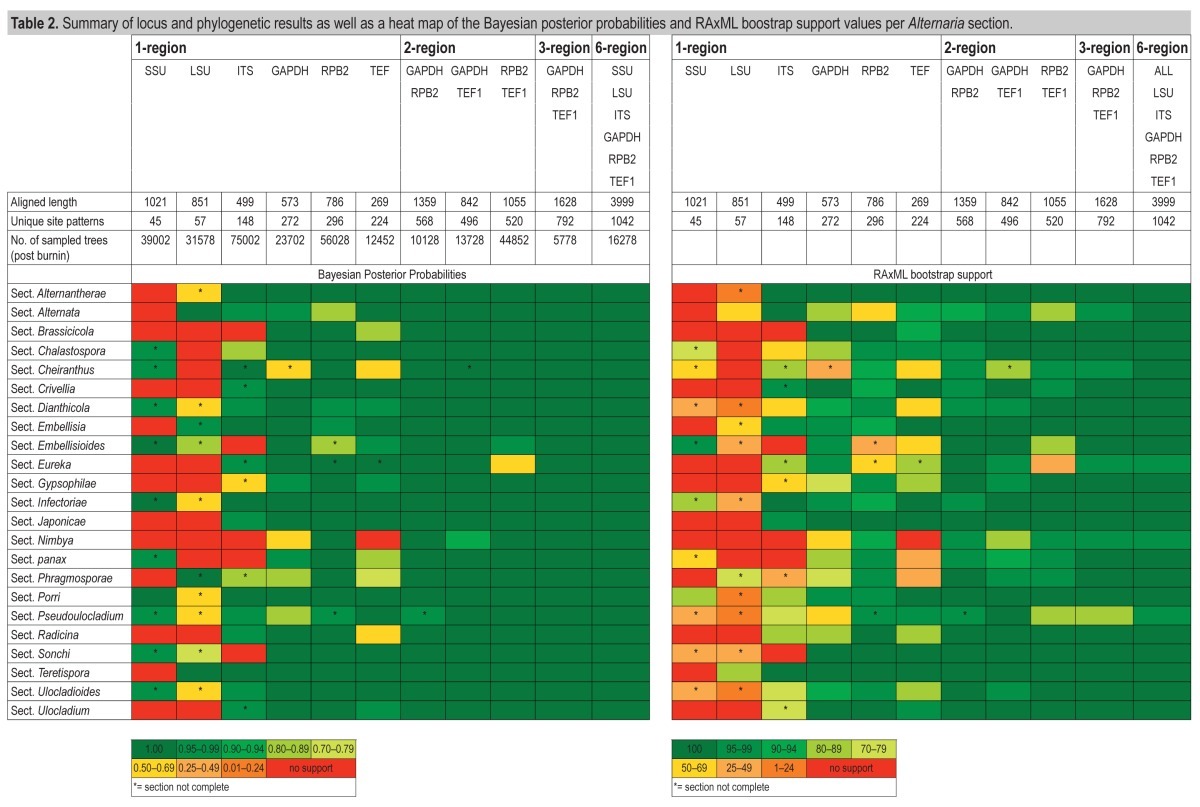
Table 3.
Coded inserts in the TEF1 sequence alignment.
| Species | Nt position | Coded | Nt position | Coded |
|---|---|---|---|---|
| Alternaria elegans | 23 to 39 | TC | ||
| Alternaria simsimi | 23 to 39 | TCC | ||
| Alternaria dauci | 186 to 205 | C | 221 to 269 | TACTT |
| Alternaria macrospora | 186 to 205 | C | 221 to 269 | TCCCC |
| Alternaria porri | 186 to 205 | C | 221 to 269 | ACTTA |
| Alternaria pseudorostrata | 186 to 205 | C | 221 to 269 | TGGTA |
| Alternaria solani | 186 to 205 | C | 221 to 269 | -AAGG |
| Alternaria tegetica | 186 to 205 | C | 221 to 269 | CACAC |
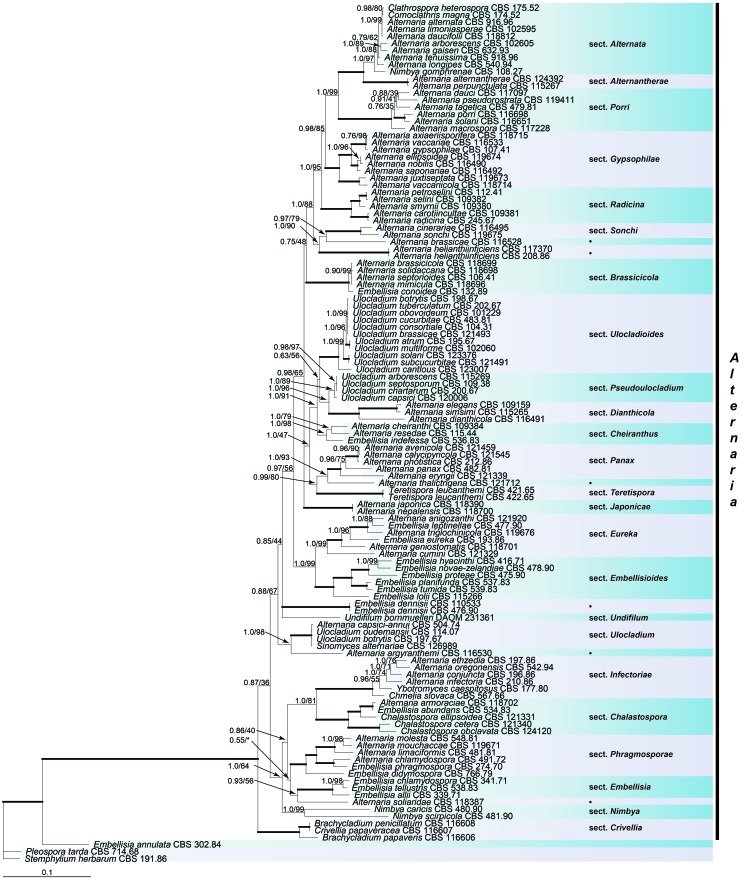
Fig. 1.
Bayesian 50 % majority rule consensus tree based on the GAPDH, RPB2 and TEF1 sequences of 121 strains representing the Alternaria complex. The Bayesian posterior probabilities (PP) and RAxML bootstrap support values (ML) are given at the nodes (PP/ML). Thickened lines indicate a PP of 1.0 and ML of 100. The tree was rooted to Stemphylium herbarum (CBS 191.86). The monotypic lineages are indicated by black dots.
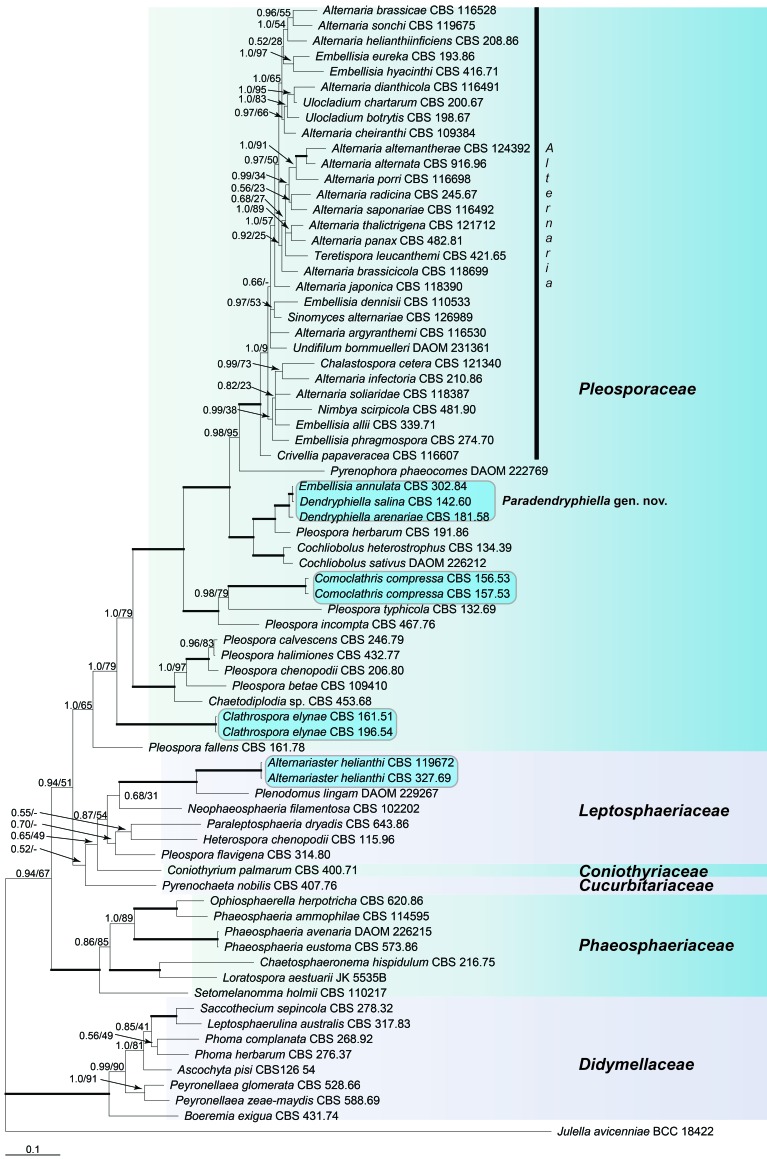
The final Pleosporineae alignment included 74 strains, representing six families, and consisted of 2 506 characters (SSU 935, LSU 796, RPB2 775) of which 700 were unique site patterns (SSU 111, LSU 145, RPB2 444). In the SSU alignment a large insertion at position 446 in the isolates Chaetosphaeronema hispidulum CBS 216.75, Pleospora fallens CBS 161.78, Pleospora flavigena CBS 314.80 and Ophiosphaerella herpotrichia CBS 620.86 was excluded from the phylogenetic analyses. A total of 43 202 trees were sampled after the burn-in. The type species of Clathrospora, C. elynae, forms a well-supported clade, located basal to the Pleosporaceae (Fig. 2), outside the Alternaria complex. The type species of Comoclathris, C. lanata, was not available for study but the two Comoclathris compressa strains cluster in a well-supported clade within the Pleosporaceae outside Alternaria s. str. The genus Alternariaster, with Alternariaster helianthi as type and only species, also clusters outside the Alternaria complex and even outside Pleosporaceae; it belongs to the Leptosphaeriaceae instead (Fig. 2). Embellisia annulata is identical to Dendryphiella salina, and forms a well-supported clade in the Pleosporaceae together with Dendryphiella arenariae. As the type species of Dendryphiella, D. vinosa, clusters outside the Pleosporineae (dela Cruz 2006, Jones et al. 2008), Dendryphiella salina and D. arenariae are placed in a new genus, Paradendryphiella, below.
Fig. 2.
Bayesian 50 % majority rule consensus tree based on the SSU, LSU and RPB2 sequences of 74 strains representing the Pleosporineae. The Bayesian posterior probabilities (PP) and RAxML bootstrap support values (ML) are given at the nodes (PP/ML). Thickened lines indicate a PP of 1.0 and ML of 100. The tree was rooted to Julella avicenniae (BCC 18422).
Taxonomy
Based on DNA sequence data in combination with a review of literature and morphology, the species within the Alternaria clade are all recognised here as Alternaria (Fig 1). This puts the genera Allewia, Brachycladium, Chalastospora, Chmelia, Crivellia, Embellisia, Lewia, Nimbya, Sinomyces, Teretispora, Ulocladium, Undifilum and Ybotromyces in synonymy with Alternaria, resulting in the proposal of 32 new combinations, 10 new names and the resurrection of 10 names. Species of Alternaria were assigned to 24 Alternaria sections, of which 16 are newly described, and six monotypic lineages. The (emended) description of the genus Alternaria, the Alternaria sections and monotypic lineages with new Alternaria names and name combinations are treated below in alphabetical order. Finally the description of the new genus Paradendryphiella is also provided.
Alternaria Nees, Syst. Pilze (Würzburg): 72. 1816 [1816-1817].
= Elosia Pers., Mycol. Eur. (Erlanga) 1: 12. 1822.
= Macrosporium Fr., Syst. Mycol. (Lundae) 3: 373. 1832.
= Rhopalidium Mont., Ann. Sci. Nat., Bot., Sér. 2, 6: 30. 1836.
= Brachycladium Corda, Icon. Fungorum hucusque Cogn. (Prague) 2: 14. 1838.
= Ulocladium Preuss, Linnaea 24: 111. 1851.
= Chmelia Svob.-Pol., Biologia (Bratislava) 21: 82. 1966.
= Embellisia E.G. Simmons, Mycologia 63: 380. 1971.
= Trichoconiella B.L. Jain, Kavaka 3: 39. 1976 [1975].
= Botryomyces de Hoog & C. Rubio, Sabouraudia 20: 19. 1982. (nom. illegit.)
= Lewia M.E. Barr & E.G. Simmons, Mycotaxon 25: 289. 1986.
= Ybotromyces Rulamort, Bull. Soc. Bot. Centre-Ouest, Nouv. Sér. 17: 192. 1986.
= Nimbya E.G. Simmons, Sydowia 41: 316. 1989.
= Allewia E.G. Simmons, Mycotaxon 38: 260. 1990.
= Crivellia Shoemaker & Inderb., Canad. J. Bot. 84: 1308. 2006.
= Chalastospora E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 668. 2007.
= Teretispora E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 674. 2007.
= Undifilum B.M. Pryor, Creamer, Shoemaker, McLain-Romero & Hambl., Botany 87: 190. 2009.
= Sinomyces Yong Wang bis & X.G. Zhang, Fungal Biol. 115: 192. 2011.
Colonies effuse, usually grey, dark blackish brown or black. Mycelium immersed or partly superficial; hyphae colourless, olivaceous-brown or brown. Stroma rarely formed. Setae and hyphopodia absent. Conidiophores macronematous, mononematous, simple or irregularly and loosely branched, pale brown or brown, solitary or in fascicles. Conidiogenous cells integrated, terminal becoming intercalary, polytretic, sympodial, or sometimes monotretic, cicatrized. Conidia catenate or solitary, dry, ovoid, obovoid, cylindrical, narrowly ellipsoid or obclavate, beaked or non-beaked, pale or medium olivaceous-brown to brown, smooth or verrucose, with transverse and with or without oblique or longitudinal septa. Septa can be thick, dark and rigid and an internal cell-like structure can be formed. Species with meristematic growth are known.
Ascomata small, solitary to clustered, erumpent to (nearly) superficial at maturity, globose to ovoid, dark brown, smooth, apically papillate, ostiolate. Papilla short, blunt. Peridium thin. Hamathecium of cellular pseudoparaphyses. Asci few to many per ascoma, (4-6-)8-spored, basal, bitunicate, fissitunicate, cylindrical to cylindro-clavate, straight or somewhat curved, with a short, furcate pedicel. Ascospores muriform, ellipsoid to fusoid, slightly constricted at septa, yellow-brown, without guttules, smooth, 3-7 transverse septa, 1-2 series of longitudinal septa through the two original central segments, end cells without septa, or with 1 longitudinal or oblique septum, or with a Y-shaped pair of septa.
Type species: Alternaria alternata (Fr.) Keissl.
ALTERNARIA SECTIONS
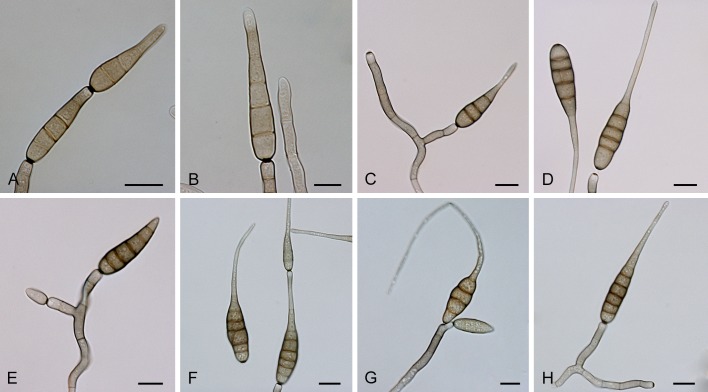
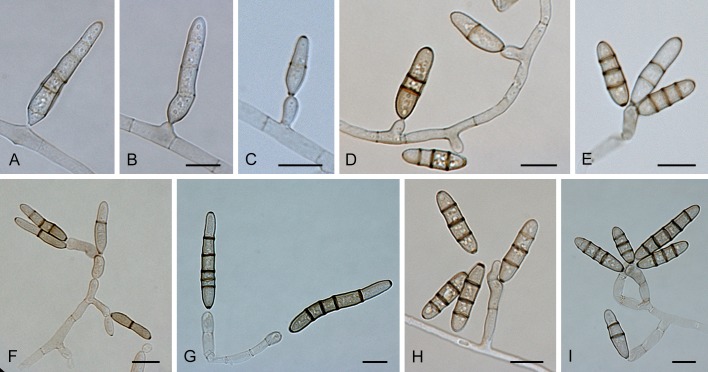
Section Alternantherae D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 540. 2013. Fig. 3.
Fig. 3.
Alternaria sect. Alternantherae: conidia and conidiophores. A-D. A. alternantherae. E-H. A. perpunctulata. Scale bars = 10 μm.
Type species: Alternaria alternantherae Holcomb & Antonop.
Diagnosis: Section Alternantherae contains short to moderately long conidiophores with a conidiogenous tip which can be enlarged. Conidia are narrowly ellipsoid or ovoid, sometimes subcylindrical, solitary or rarely paired, sometimes slightly constricted near some septa, longitudinal or oblique septa occasionally occur, disto- and euseptate, with a long apical narrow beak. The conidial beak is unbranched, septate or aseptate, long filiform, and sometimes swollen at the end. Internal compartmentation occurs, cell lumina tend to be broadly octagonal to rounded.
Notes: Section Alternantherae was recently established by Lawrence et al. (2013) after first being described as species-group A. alternantherae (Lawrence et al. 2012). The described section consists of three former Nimbya species which formed a separate clade amidst the Alternaria species-groups based on sequences of the GAPDH, ITS and Alt a 1 genes (Lawrence et al. 2012). Nimbya celosiae is placed in this section based on the data of Lawrence et al. (2012), while N. gomphrenae is placed in the section based on ITS sequence data from Chou & Wu (2002).
Alternaria alternantherae Holcomb & Antonop., Mycologia 68: 1126. 1976.
≡ Nimbya alternantherae (Holcomb & Antonop.) E.G. Simmons & Alcorn, Mycotaxon 55: 142. 1995.
Alternaria celosiicola Jun. Nishikawa & C. Nakash., J. Phytopathol.: doi: 10.1111/jph.12108 (p. 3). 2013.
Basionym: Nimbya celosiae E.G. Simmons & Holcomb, Mycotaxon 55: 144. 1995.
≡ Alternaria celosiae (E.G. Simmons & Holcomb) D.P. Lawr., M.S. Park & B.M. Pryor, Mycol. Progr. 11: 811. 2012. (nom. illegit., homonym of Alternaria celosiae (Tassi) O. Savul. 1950).
Alternaria gomphrenae Togashi, Bull. Imp. Coll. Agric. 9: 6. 1926.
≡ Nimbya gomphrenae (Togashi) E.G. Simmons, Sydowia 41: 324. 1989.
Alternaria perpunctulata (E.G. Simmons) D.P. Lawr., M.S. Park & B.M. Pryor, Mycol. Progr. 11: 811. 2012.
Basionym: Nimbya perpunctulata E.G. Simmons, Stud. Mycol. 50: 115. 2004.
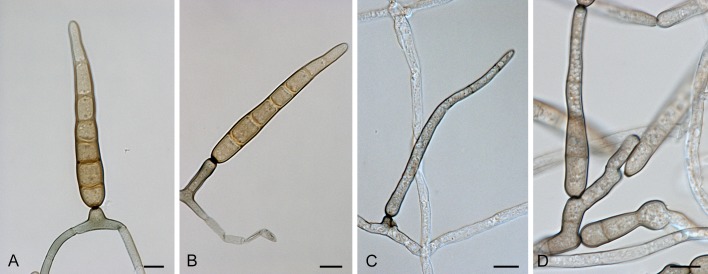
Section Alternata D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 538. 2013. Fig. 4.
Fig. 4.
Alternaria sect. Alternata: conidia and conidiophores. A, N. A. daucifolii. B, L-M. A. arborescens. C, H-J. A. alternata. D, O. A. gaisen. E. A. limoniasperae. F, K. A. tenuissima. G, P. A. longipes. Scale bars = 10 μm.
Type species: Alternaria alternata (Fr.) Keissl.
Diagnosis: Section Alternata contains straight or curved primary conidiophores, short to long, simple or branched, with one or several apical conidiogenous loci. Conidia are obclavate, long ellipsoid, small or moderate in size, septate, slightly constricted near some septa, with few longitudinal septa, in moderately long to long, simple or branched chains. The conidium body can narrow gradually into a tapered beak or secondary conidiophore. Secondary conidiophores can be formed apically or laterally with one or a few conidiogenous loci.
Notes: Next to the species that are displayed in our phylogeny, 14 more are included in sect. Alternata based on the study of Lawrence et al. (2013) and confirmed by our molecular data (not shown). We chose not to include 11 species from the study of Lawrence et al. (2013). The species A. gossypina, A. grisae, A. grossulariae, A. iridis, A. lini, A. maritima and A. nelumbii were not recognised by Simmons (2007) and the strains of A. malvae, A. rhadina, A. resedae and A. tomato used by Lawrence et al. (2013) were not authentic. Section Alternata comprises almost 60 Alternaria species based on ITS sequence data (data not shown). The molecular variation within this section is low.
Alternaria alternata (Fr.) Keissl., Beih. Bot. Centralbl., Abt. 2, 29: 434. 1912.
Basionym: Torula alternata Fr., Syst. Mycol. (Lundae) 3: 500. 1832 (nom. sanct.).
= Alternaria tenuis Nees, Syst. Pilze (Würzburg): 72. 1816 [1816-1817].
Additional synonyms listed in Simmons (2007)
Alternaria angustiovoidea E.G. Simmons, Mycotaxon 25: 198. 1986.
Alternaria arborescens E.G. Simmons, Mycotaxon 70: 356. 1999.
Alternaria burnsii Uppal, Patel & Kamat, Indian J. Agric. Sci. 8: 49. 1938.
Alternaria cerealis E.G. Simmons & C.F. Hill, CBS Biodiversity Ser. (Utrecht) 6: 600. 2007.
Alternaria citriarbusti E.G. Simmons, Mycotaxon 70: 287. 1999.
Alternaria citrimacularis E.G. Simmons, Mycotaxon 70: 277. 1999.
Alternaria colombiana E.G. Simmons, Mycotaxon 70: 298. 1999.
Alternaria daucifollii E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 518. 2007.
Alternaria destruens E.G. Simmons, Mycotaxon 68: 419. 1998.
Alternaria dumosa E.G. Simmons, Mycotaxon 70: 310. 1999.
Alternaria gaisen Nagano ex Hara, Sakumotsu Byorigaku, Edn 4: 263. 1928.
= Alternaria gaisen Nagano, J. Jap. Soc. Hort. Sci. 32: 16-19. 1920. (nom. illegit.)
= Alternaria kikuchiana S. Tanaka, Mem. Coll. Agric. Kyoto Univ., Phytopathol. Ser. 28: 27. 1933.
= Macrosporium nashi Miura, Flora of Manchuria and East Mongolia, Part III Cryptogams, Fungi: 513. 1928.
Alternaria herbiphorbicola E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 608. 2007.
Alternaria limoniasperae E.G. Simmons, Mycotaxon 70: 272. 1999.
Alternaria longipes (Ellis & Everh.) E.W. Mason, Mycol. Pap. 2: 19. 1928.
Basionym: Macrosporium longipes Ellis & Everh., J. Mycol. 7: 134. 1892.
= Alternaria brassicae var. tabaci Preissecker, Fachliche Mitt. Österr. Tabakregie 16: 4. 1916.
Alternaria perangusta E.G. Simmons, Mycotaxon 70: 303. 1999.
Alternaria postmessia E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 598. 2007.
Alternaria tangelonis E.G. Simmons, Mycotaxon 70: 282. 1999.
Alternaria tenuissima (Nees & T. Nees: Fr.) Wiltshire, Trans. Brit. Mycol. Soc. 18: 157. 1933.
Basionym: Macrosporium tenuissimum (Nees & T. Nees) Fr., Syst. Mycol. (Lundae) 3: 374. 1832 (nom. sanct.).
≡ Helminthosporium tenuissimum Kunze ex Nees & T. Nees, Nova Acta Acad. Caes. Leop.-Carol. German. Nat. Cur. 9: 242. 1818.
Additional synonyms listed in Simmons (2007).
Alternaria toxicogenica E.G. Simmons, Mycotaxon 70: 294. 1999.
Alternaria turkisafria E.G. Simmons, Mycotaxon 70: 290. 1999.
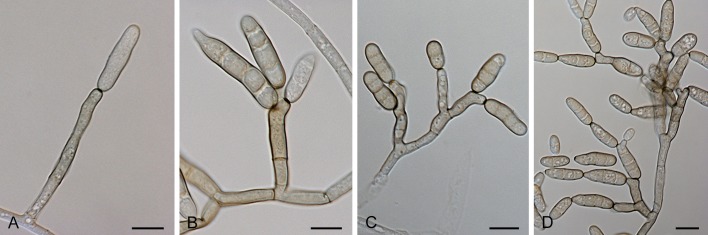
Section Brassicicola D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 541. 2013. Fig. 5.
Fig. 5.
Alternaria sect. Brassicicola: conidia and conidiophores. A, H. A. brassicicola. B, I, L-M. A. mimicola. C, G. A. solidaccana. D, J-K. A. conoidea. E-F. A. septorioides. Scale bars = 10 μm.
Type species: Alternaria brassicicola (Schwein.) Wiltshire
Diagnosis: Section Brassicicola contains short to moderately long, simple or branched primary conidiophores with one or several apical conidiogenous loci. Conidia are ellipsoid, ovoid or somewhat obclavate, small or moderate in size, septate, slightly or strongly constricted at most of their transverse septa, with no to many longitudinal septa, in moderately long to long, simple or branched chains, with dark septa and cell walls. Secondary conidiophores can be formed apically or laterally with one or a few conidiogenous loci. Chlamydospores may occur.
Notes: Our molecular data support the morphological placement of A. septorioides and A. solidaccana in section Brassicicola (Simmons 2007). The other three species were already assigned to this section based on previous molecular studies (Pryor et al. 2009, Runa et al. 2009, Lawrence et al. 2012). Alternaria japonica was previously linked to the A. brassicicola species-group (Pryor & Gilbertson 2000, Pryor & Bigelow 2003, Lawrence et al. 2013), but this association was questioned by Hong et al. (2005). In our analyses, A. japonica clustered in sect. Japonicae.
Alternaria brassicicola (Schwein.) Wiltshire, Mycol. Pap. 20: 8. 1947.
Basionym: Helminthosporium brassicicola Schwein., Trans. Amer. Philos. Soc., Ser. 2, 4: 279. 1832.
Additional synonyms listed in Simmons (2007)
Alternaria conoidea (E.G. Simmons) D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 542. 2013.
Basionym: Embellisia conoidea E.G. Simmons, Mycotaxon 17: 226. 1983.
Alternaria mimicula E.G. Simmons, Mycotaxon 55: 129. 1995.
Alternaria septorioides (Westend.) E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 570. 2007.
Basionym: Sporidesmium septorioides Westend., Bull. Acad. Roy. Sci. Belgique., Cl. Sci., Sér. 2, 21: 236. 1854.
= Alternaria resedae Neerg., Annual Rep. Phytopathol. Lab. J.E. Ohlsens Enkes, Seed Growers, Copenhagen 7: 9. 1942 (nom. nud.).
= Alternaria resedae Neerg., Danish species of Alternaria & Stemphylium: 150. 1945.
Alternaria solidaccana E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 572. 2007.
Section Chalastospora (E.G. Simmons) Woudenb. & Crous, comb. et stat. nov. MycoBank MB803733. Fig. 6.
Fig. 6.
Alternaria sect. Chalastospora: conidia and conidiophores. A. A. cetera. B. A. obclavata. C. A. breviramosa. D, H. A. armoraciae. E-G. A. abundans. Scale bars = 10 μm.
Basionym: Chalastospora E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 668. 2007.
Type species: Alternaria cetera E.G. Simmons
Diagnosis: Section Chalastospora contains short to long, simple or branched primary conidiophores with one or several conidiogenous loci. Conidia are pale to medium brown, narrowly ellipsoid to ellipsoid or ovoid, beakless, with no to multiple transverse eusepta and rarely longitudinal septa, solitary or in chains. Secondary conidiophores can be formed apically or laterally with one or a few conidiogenous loci.
Notes: Previous studies already placed E. abundans in the Chalastospora-clade (Andersen et al. 2009, Lawrence et al. 2012). Our study also placed Alternaria armoraciae in this section, while Crous et al. (2009c) showed that Chalastospora gossypii, formerly Alternaria malorum, belonged to this section based on sequences of the ITS and LSU genes.
Alternaria abundans (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803688.
Basionym: Embellisia abundans E.G. Simmons, Mycotaxon 17: 222. 1983.
Alternaria armoraciae E.G. Simmons & C.F. Hill, CBS Biodiversity Ser. (Utrecht) 6: 660. 2007.
Alternaria breviramosa Woudenb. & Crous, nom. nov. MycoBank MB803690.
Basionym: Chalastospora ellipsoidea Crous & U. Braun, Persoonia 22: 145. 2009, non Alternaria ellipsoidea E.G. Simmons, 2002.
Etymology: Name refers to the short lateral branches.
Alternaria cetera E.G. Simmons, Mycotaxon 57: 393. 1996.
≡ Chalastospora cetera (E.G. Simmons) E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 668. 2007.
Alternaria malorum (Ruehle) U. Braun, Crous & Dugan, Mycol. Progr. 2: 5. 2003.
Basionym: Cladosporium malorum Ruehle, Phytopathology 21: 1146. 1931.
-
= Cladosporium gossypii Jacz., Khlopkovoe Delo, 1929 (5-6): 564. 1929, non Alternaria gossypii (Jacz.) Y. Nisik., K. Kimura & Miyaw., 1940.
≡ Chalastospora gossypii (Jacz.) U. Braun & Crous, Persoonia 22: 144. 2009.
= Cladosporium malorum Heald, Wash. State Agric. Exp. Sta. Bull., Special Ser. 245: 48. 1930. (nom. nud.)
Additional synonyms in Crous et al. (2009c).
Alternaria obclavata (Crous & U. Braun) Woudenb. & Crous, comb. nov. MycoBank MB803689.
Basionym: Chalastospora obclavata Crous & U. Braun, Persoonia 22: 146. 2009.
Section Cheiranthus Woudenb. & Crous, sect. nov. MycoBank MB803734. Fig. 7.
Fig. 7.

Alternaria sect. Cheiranthus: conidia and conidiophores. A-B. A. indefessa. B-C. A. cheiranthi. Scale bars = 10 μm.
Type species: Alternaria cheiranthi (Lib.) P.C. Bolle
Diagnosis: Section Cheiranthus contains short to moderately long, simple or branched primary conidiophores with one or several conidiogenous loci. Conidia are ovoid, broadly ellipsoid with transverse and longitudinal septa, slightly or strongly constricted at the septa, in short to long, simple or branched chains. Secondary conidiophores can be formed apically or laterally with a single conidiogenous locus.
Notes: Next to Alternaria cheiranthi and Embellisia indefessa, sect. Cheiranthus contains a non-sporulating strain formerly known as Alternaria resedae, CBS 115.44. Because Alternaria resedae is synonymised with Alternaria septorioides (Simmons 2007), which clusters in section Brassisicola, CBS 115.44 will be treated as “Alternaria sp.”. Alternaria cheiranthi and E. indefessa have been linked to Ulocladium (Pryor & Gilbertson 2000, Pryor & Bigelow 2003, Hong et al. 2005, Pryor et al. 2009, Runa et al. 2009, Lawrence et al. 2012), but based on morphology could not be placed here. Our extensive dataset showed that they form a sister section to section Ulocladioides.
Alternaria cheiranthi (Lib.) P.C. Bolle, Meded. Phytopathol. Lab. “Willie Commelin Scholten” 7: 43. 1924.
Basionym: Helminthosporium cheiranthi Lib. [as “Helmisporium”], in Desmazières, Plantes Cryptogames du Nord de la France, edn 1: 213. 1827.
≡ Macrosporium cheiranthi (Lib.) Fr., Syst. Mycol. (Lundae) 3: 374. 1832.
Alternaria indefessa (E.G. Simmons) Woudenberg & Crous, comb. nov. MycoBank MB803691.
Basionym: Embellisia indefessa E.G. Simmons, Mycotaxon 17: 228. 1983.
Section Crivellia (Shoemaker & Inderb.) Woudenb. & Crous, comb. et stat. nov. MycoBank MB803735. Fig. 8.
Fig. 8.
Alternaria sect. Crivellia: conidia and conidiophores. A-B. A. papavericola. C-D. A. penicillata. Scale bars = 10 μm.
Basionym: Crivellia Shoemaker & Inderb., Canad. J. Bot. 84: 1308. 2006.
Type species: Alternaria penicillata (Corda) Woudenb. & Crous (= Cucurbitaria papaveracea De Not.).
Diagnosis: Section Crivellia is characterised by straight or curved, simple or branched primary conidiophores, with geniculate, sympodial proliferations. Conidia are cylindrical, straight to curved to inequilateral, with transverse eusepta, rarely constricted at septa, single or in short, simple or branched chains. Secondary conidiophores are formed apically or laterally. Microsclerotia or chlamydospores may occur. Sexual morphs observed.
Notes: Section Crivellia contains the type species of the sexual morph Crivellia, C. papaveracea, with Brachycladium penicillatum asexual morph, and Brachycladium papaveris. The genus was established by Inderbitzin et al. (2006) based on the finding that C. papaveraceae, formerly Pleospora papaveraceae, belonged to the Alternaria-complex instead of Pleospora s. str. based on ITS, GAPDH and TEF1 sequences.
Alternaria papavericola Woudenb. & Crous, nom. nov. MycoBank MB803749.
Basionym: Helminthosporium papaveris Sawada, J. Nat. Hist. Soc. Formosa 31: 1. 1917.
≡ Dendryphion papaveris (Sawada) Sawada, Special Publ. Coll. Agric. Natl. Taiwan Univ. 8: 200. 1959, non Alternaria papaveris (Bres.) M.B. Ellis, 1976.
≡ Brachycladium papaveris (Sawada) Shoemaker & Inderb., Canad. J. Bot. 84: 1310. 2006.
Etymology: Name refers to the host.
Alternaria penicillata (Corda) Woudenb. & Crous, comb. nov. MycoBank MB803692.
Basionym: Brachycladium penicillatum Corda, Icon. Fungorum hucusque Cogn. (Prague) 2: 14. 1838.
≡ Dendryphion penicillatum (Corda) Fr., Summa Veg. Scand., Sect. Post. (Stockholm): 504. 1849.
-
= Cucurbitaria papaveracea De Not., Sferiacei Italici: 62. 1863.
≡ Pleospora papaveracea (De Not.) Sacc., Syll. Fungorum (Abellini) 2: 243. 1883.
≡ Crivellia papaveracea (De Not.) Shoemaker & Inderb., Canad. J. Bot. 84: 1308. 2006.
Note: The asexual name, Brachycladium penicillatum is older than the sexual name, Cucurbitaria papaveracea, and therefore the species epithet penicillatum is chosen above papaveracea.
Section Dianthicola Woudenb. & Crous, sect. nov. MycoBank MB803736. Fig. 9.
Fig. 9.
Alternaria sect. Dianthicola: conidia and conidiophores. A-B. A. dianthicola. C-E. A. simsimi. F-H. A. elegans. Scale bars = 10 μm.
Type species: Alternaria dianthicola Neerg.
Diagnosis: Section Dianthicola contains simple or branched primary conidiophores, with or without apical geniculate proliferations. Conidia are narrowly ovoid or narrowly ellipsoid with transverse and few longitudinal septa, slightly constricted at the septa, with a long (filamentous) beak or apical secondary conidiophore, solitary or in short chains.
Note: Based on the ITS sequence, Alternaria dianthicola clustered near Ulocladium (Chou & Wu 2002). Our extensive dataset places it in a sister section to section Ulocladioides.
Alternaria dianthicola Neerg., Danish species of Alternaria & Stemphylium: 190. 1945.
Alternaria elegans E.G. Simmons & J.C. David, Mycotaxon 75: 89. 2000.
Alternaria simsimi E.G. Simmons, Stud. Mycol. 50: 111. 2004.
Section Embellisia (E.G. Simmons) Woudenb. & Crous, comb. et stat. nov. MycoBank MB803737. Fig. 10.
Fig. 10.
Alternaria sect. Embellisia: conidia and conidiophores. A-D. A. embellisia. E-H. A. tellustris. Scale bars = 10 μm.
Basionym: Embellisia E.G. Simmons, Mycologia 63: 380. 1971.
Type species: Alternaria embellisia Woudenb. & Crous (≡ Helminthosporium allii Campan., Embellisia allii (Campan.) E.G. Simmons).
Diagnosis: Section Embellisia contains simple, septate conidiophores, straight or with geniculate sympodial proliferation. Condia are solitary, ovoid to subcylindrical, straight to inequilateral, transseptate; septa can be thick, dark and rigid in contrast to the external wall. Chlamydospores may occur.
Notes: Section Embellisia contains the first two species described in the genus Embellisia, Embellisia allii (type species) and Embellisia chlamydospora (Simmons 1971) together with Embellisia tellustris. This clade is also resolved in the latest molecular revision of Embellisia based on sequences of the GAPDH, ITS and Alt a 1 genes as Embellisia group I (Lawrence et al. 2012).
Alternaria chlamydosporigena Woudenb. & Crous, nom. nov. MycoBank MB803694.
Basionym: Pseudostemphylium chlamydosporum Hoes, G.W. Bruehl & C.G. Shaw, Mycologia 57: 904. 1965, non Alternaria chlamydospora Mouch., 1973.
≡ Embellisia chlamydospora (Hoes, G.W. Bruehl & C.G. Shaw) E.G. Simmons, Mycologia 63: 384. 1971.
Etymology: Name refers to the formation of chlamydospores during growth.
Alternaria embellisia Woudenb. & Crous, nom. nov. MycoBank MB803693.
Basionym: Helminthosporium allii Campan., Nuovi Ann. Agric. Roma 4: 87. 1924, non Alternaria allii Nolla, 1927.
≡ Embellisia allii (Campan.) E.G. Simmons, Mycologia 63: 382. 1971.
Etymology: Name refers to the genus Embellisia for which it served as type species.
Alternaria tellustris (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803695.
Basionym: Embellisia tellustris E.G. Simmons [as “telluster”], Mycotaxon 17: 234. 1983.
Section Embellisioides Woudenb. & Crous, sect. nov. MycoBank MB803738. Fig. 11.
Fig. 11.
Alternaria sect. Embellisioides: conidia and conidiophores. A-B. A. hyacinthi. C-E. A. lolii. F-H. A. botryospora. I-K. A. planifunda. L-N. A. proteae. O-P. A. tumida. Scale bars = 10 μm.
Type species: Alternaria hyacinthi (de Hoog & P.J. Mull. bis) Woudenb. & Crous
Diagnosis: Section Embellisioides contains simple, septate conidiophores, straight or with multiple, geniculate, sympodial proliferations. Apical or lateral, short secondary conidiophores may occur. Condia are solitary or in short chains, obovoid to ellipsoid, with transverse and longitudinal septa; transverse septa can be thick, dark and rigid in contrast to the external wall. Chlamydospores and a sexual morph may occur.
Note: In Lawrence et al. (2012) the section is named Embellisia group III.
Alternaria botryospora Woudenb. & Crous, nom. nov. MycoBank MB803705.
Basionym: Embellisia novae-zelandiae E.G. Simmons & C.F. Hill, Mycotaxon 38: 252. 1990, non Alternaria novae-zelandiae E.G. Simmons, 2002.
Etymology: Name refers to the clusters of conidia.
Alternaria hyacinthi (de Hoog & P.J. Mull. bis) Woudenb. & Crous, comb. nov. MycoBank MB803703.
Basionym: Embellisia hyacinthi de Hoog & P.J. Mull. bis, Netherlands J. Pl. Pathol. 79: 85. 1973.
Alternaria lolii (E.G. Simmons & C.F. Hill) Woudenb. & Crous, comb. nov. MycoBank MB803704.
Basionym: Embellisia lolii E.G. Simmons & C.F. Hill, Stud. Mycol. 50: 113. 2004.
Alternaria planifunda (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803706.
Basionym: Embellisia planifunda E.G. Simmons, Mycotaxon 17: 233. 1983.
Alternaria proteae (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803707.
Basionym: Embellisia proteae E.G. Simmons, Mycotaxon 38: 258. 1990.
= Allewia proteae E.G. Simmons, Mycotaxon 38: 262. 1990.
Alternaria tumida (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803708.
Basionym: Embellisia tumida E.G. Simmons, Mycotaxon 17: 236. 1983.
Section Eureka Woudenb. & Crous, sect. nov. MycoBank MB803739. Fig. 12.
Fig. 12.
Alternaria sect. Eureka: conidia and conidiophores. A-B. A. anigozanthi. C-D. A. cumini. E-F. A. leptinellae. G-H. A. triglochinicola. I-J. A. geniostomatis. K-L. A. eureka. Scale bars = 10 μm.
Type species: Alternaria eureka E.G. Simmons
Diagnosis: Section Eureka contains simple, septate conidiophores, straight or with geniculate, sympodial proliferations. Apical or lateral, short secondary conidiophores may occur. Condia are solitary or in short chains, narrowly ellipsoid to cylindrical, with transverse and longitudinal septa, slighty constricted at the septa, with a blunt rounded apex. Chlamydospores and a sexual morph may occur.
Notes: Section Eureka contains four Alternaria species and two former Embellisia species. From the Alternaria species only the ITS sequence of A. geniostomatis was previously used in a molecular study (Toth et al. 2011), showing it to cluster separate from the other Alternaria spp. The two Embellisia species were included in the latest molecular-based revision of Embellisia (Lawrence et al. 2012) where they formed Embellisia group IV. A sexual morph is known for the type species of this section.
Alternaria anigozanthi Priest, Australas. Pl. Pathol. 24: 239. 1995.
Alternaria cumini E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 664. 2007.
Alternaria eureka E.G. Simmons, Mycotaxon 25: 306. 1986.
≡ Embellisia eureka (E.G. Simmons) E.G. Simmons, Mycotaxon 38: 260. 1990.
-
= Lewia eureka E.G. Simmons, Mycotaxon 25: 304. 1986.
≡ Allewia eureka (E.G. Simmons) E.G. Simmons, Mycotaxon 38: 264. 1990.
Alternaria geniostomatis E.G. Simmons & C.F. Hill, CBS Biodiversity Ser. (Utrecht) 6: 412. 2007.
Alternaria leptinellae (E.G. Simmons & C.F. Hill) Woudenb. & Crous, comb. nov. MycoBank MB803696.
Basionym: Embellisia leptinellae E.G. Simmons & C.F. Hill, Mycotaxon 38: 254. 1990.
Alternaria triglochinicola Alcorn & S.M. Francis, Mycotaxon 46: 359. 1993.
Section Gypsophilae D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 541. 2013. Fig. 13
Fig. 13.
Alternaria sect. Gypsophilae: conidia and conidiophores. A-B. A. axiariisporifera. C-D. A. ellipsoidea. E-G. A. saponariae. H-I. A. vaccariae. J-K. A. nobilis. L-M. A. juxtiseptata. N-P. A. vaccariicola. Scale bars = 10 μm.
Type species: Alternaria gypsophilae Neerg.
Diagnosis: Section Gypsophilae contains simple, or occasionally branched, primary conidiophores, with one or a few conidiogenous loci. Conidia are ellipsoid to long ovoid, with multiple transverse and longitudinal septa, conspicuously constricted near some transverse septa, solitary or in short chains. Secondary conidiophores are formed apically with one or two conidiogenous loci or laterally with a single conidiogenous locus. Species from this section occur on Caryophyllaceae.
Notes: Section Gypsophilae was recently established by Lawrence et al. (2013) containing the four Alternaria species, A. gypsophilae, A. nobilis, A. vaccariae and A. vaccariicola. Our dataset adds four Alternaria species, A. axiaeriisporifera, A. ellipsoidea, A. saponariae, and A. juxtiseptata to this section. Simmons (2007) noted the similarity of the primary conidia of A. ellipsoidea to A. gypsophilae, A. nobilis, A. saponariae and A. vaccariae. This section contains all Alternaria species that occur on Caryophyllaceae (Simmons 2002), except A. dianthicola which resides in sect. Dianthicola.
Alternaria axiaeriisporifera E.G. Simmons & C.F. Hill, CBS Biodiversity Ser. (Utrecht) 6: 662. 2007.
Alternaria ellipsoidea E.G. Simmons, Mycotaxon 82: 31. 2002.
Alternaria gypsophilae Neerg., Danish species of Alternaria & Stemphylium: 207. 1945.
Alternaria juxtiseptata E.G. Simmons, Mycotaxon 82: 32. 2002.
Alternaria nobilis (Vize) E.G. Simmons, Mycotaxon 82: 7. 2002.
Basionym: Macrosporium nobile Vize, Grevillea 5(35): 119. 1877.
Alternaria saponariae (Peck) Neerg., Annual Rep. Phytopathol. Lab. J.E. Ohlsens Enkes, Seed Growers, Copenhagen 3: 6. 1938 [1937-1938].
Basionym: Macrosporium saponariae Peck, Rep. (Annual) NewYork State Mus. Nat. Hist. 28: 62. 1876 [1875].
Alternaria vaccariae (Săvul. & Sandu) E.G. Simmons & S.T. Koike, Mycotaxon 82: 21. 2002.
Basionym: Macrosporium vaccariae Săvul. & Sandu, Hedwigia 73: 130. 1933.
Alternaria vaccariicola E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 594. 2007.
Section Infectoriae Woudenb. & Crous, sect. nov. MycoBank MB803740. Fig. 14.
Fig. 14.
Alternaria sect. Infectoriae: conidia and conidiophores. A-B. A. ethzedia. C-D. A. infectoria. E-F. A. conjuncta. G-H. A. oregonensis. Scale bars = 10 μm.
Type species: Alternaria infectoria E.G. Simmons
Diagnosis: Section Infectoriae contains short to long, simple or branched primary conidiophores with one or several conidiogenous loci. Conidia are obclavate, long-ellipsoid, small or moderate in size, septate, slightly constricted near some septa, with few longitudinal septa, in moderately long to long, branched chains. Long, geniculate, multi-locus secondary conidiophores can be formed apically or laterally. Sexual morphs are known, and meristematic growth has been reported.
Notes: In addition to the six species that are displayed in our phylogeny, 19 more are included based on the study of Lawrence et al. (2013), confirmed with our molecular data (not shown). From these 25 species, nine species have a known sexual morph in Lewia. Three species from the study of Lawrence et al. (2013) are not included; A. photistica (sect. Panax) and A. dianthicola (sect. Dianthicola) cluster elsewhere in our phylogenies and A. peglionii is marked as a taxon incertae sedis by Simmons (2007). The human pathogenic genera Ybotromyces and Chmelia are also embedded in sect. Infectoriae.
Alternaria alternarina E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 644. 2007.
-
= Pyrenophora alternarina M.D. Whitehead & J. Dicks., Mycologia 44: 748. 1952.
≡ Lewia alternarina (M.D. Whitehead & J.G. Dicks.) E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 644. 2007.
Alternaria arbusti E.G. Simmons, Mycotaxon 48: 103. 1993.
Alternaria caespitosa (de Hoog & C. Rubio) Woudenb. & Crous, comb. nov. MycoBank MB803698.
Basionym: Botryomyces caespitosus de Hoog & C. Rubio, Mycotaxon 14: 19. 1982.
≡ Ybotromyces caespitosus (de Hoog & C. Rubio) Rulamort, Bull. Soc. Bot. Centre-Ouest, Nouv. Sér. 21: 512. 1990.
Alternaria californica E.G. Simmons & S.T. Koike, CBS Biodiversity Ser. (Utrecht) 6: 602. 2007.
Alternaria conjuncta E.G. Simmons, Mycotaxon 25: 294. 1986.
-
= Sphaeria scrophulariae Desm., Ann. Sci. Nat., Bot., Sér. 2, 6: 245. 1836.
≡ Leptosphaeria scrophulariae (Desm.) Sacc., Syll. Fungorum (Abellini) 2: 57. 1883.
≡ Heptameria scrophulariae (Desm.) Cooke, Grevillea 18(no. 86): 31. 1889.
≡ Pleospora scrophulariae (Desm.) Höhn., Sitzungsber. Kaiserl. Akad. Wiss., Math.-Naturwiss. Cl., Abt. 1. 126(4-5): 374. 1917.
≡ Lewia scrophulariae (Desm.) M.E. Barr & E.G. Simmons, Mycotaxon 25: 294. 1986.
Alternaria daucicaulis E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 640. 2007.
= Lewia daucicaulis E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 640. 2007.
Alternaria ethzedia E.G. Simmons, Mycotaxon 25: 300. 1986.
= Lewia ethzedia E.G. Simmons, Mycotaxon 25: 299. 1986.
Alternaria frumenti E.G. Simmons & C.F. Hill, CBS Biodiversity Ser. (Utrecht) 6: 620. 2007.
Alternaria graminicola E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 626. 2007.
Alternaria hordeiaustralica E.G. Simmons & Alcorn, CBS Biodiversity Ser. (Utrecht) 6: 614. 2007.
= Lewia hordeiaustralica E.G. Simmons & Alcorn, CBS Biodiversity Ser. (Utrecht) 6: 614. 2007.
Alternaria hordeicola E.G. Simmons & Kosiak, CBS Biodiversity Ser. (Utrecht) 6: 630. 2007.
= Lewia hordeicola Kwaśna & Kosiak, Mycologia 98: 663. 2006.
Alternaria humuli E.G. Simmons, Mycotaxon 83: 139. 2002.
Alternaria incomplexa E.G. Simmons, Mycotaxon 57: 394. 1996.
Alternaria infectoria E.G. Simmons, Mycotaxon 25: 298. 1986.
-
= Pleospora infectoria Fuckel, Jahrb. Nassauischen Vereins Naturk. 23-24: 132. 1870 [1869-70].
≡ Sphaeria infectoria (Fuckel) Cooke, Handb. Brit. Fungi 2: 897. 1871.
≡ Pleospora phaeocomoides var. infectoria (Fuckel) Wehm., A World Monograph of the Genus Pleospora and its Segregates: 121. 1961.
≡ Lewia infectoria (Fuckel) M.E. Barr & E.G. Simmons, Mycotaxon 25: 296. 1986.
Alternaria intercepta E.G. Simmons, Mycotaxon 83: 134. 2002.
= Lewia intercepta E.G. Simmons & McKemy, Mycotaxon 83: 133. 2002.
Alternaria merytae E.G. Simmons, Mycotaxon 83: 136. 2002.
Alternaria metachromatica E.G. Simmons, Mycotaxon 50: 418. 1994.
Alternaria novae-zelandiae E.G. Simmons, Mycotaxon 83: 142. 2002.
Alternaria oregonensis E.G. Simmons, Mycotaxon 50: 417. 1994.
Alternaria slovaca (Svob.-Pol., L. Chmel & Bojan.) Woudenb. & Crous, comb. nov. MycoBank MB803699.
Basionym: Aureobasidium slovacum Svob.-Pol., L. Chmel & Bojan., Conspect. Verruc. 5: 116. 1966.
≡ Chmelia slovaca (Svob.-Pol., L. Chmel & Bojan.) Svob.-Pol., Biologia (Bratislava) 21: 83. 1966.
Alternaria triticimaculans E.G. Simmons & Perelló, Mycotaxon 50: 413. 1994.
Alternaria triticina Prasada & Prabhu, Indian Phytopathol. 15 (3-4): 292. 1963. [1962]
Alternaria ventricosa R.G. Roberts, Mycotaxon 100: 164. 2007.
Alternaria viburni E.G. Simmons, Mycotaxon 83: 132. 2002.
= Lewia viburni E.G. Simmons & McKemy, Mycotaxon 83: 130. 2002.
Section Japonicae Woudenb. & Crous, sect. nov. MycoBank MB803741. Fig. 15.
Fig. 15.
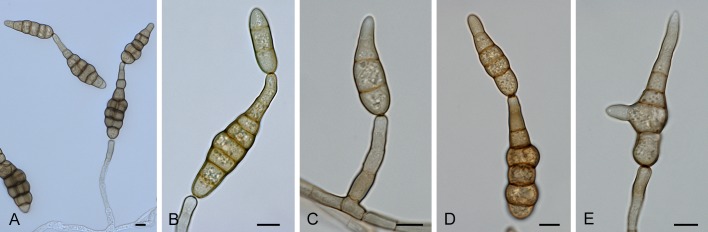
Alternaria sect. Japonicae: conidia and conidiophores. A-B. A. japonica. C-E. A. nepalensis. Scale bars = 10 μm.
Type species: Alternaria japonica Yoshii
Diagnosis: Section Japonicae contains short to long, simple or occasionally branched primary conidiophores with a single conidiogenous locus. Conidia are short, to long-ovoid with transverse and longitudinal septa, conspicuously constricted at most of the transverse septa, in short chains. Apical secondary conidiophores are produced with a single conidiogenous locus. The species within this section occur on Brassicaceae.
Note: Alternaria japonica was previously connected to the A. brassicicola species-group (Pryor & Gilbertson 2000, Pryor & Bigelow 2003, Lawrence et al. 2013), but this association was questioned by Hong et al. (2005).
Alternaria japonica Yoshii, J. Pl. Protect. 28: 17. 1941.
= Alternaria matthiolae Neerg., Danish species of Alternaria and Stemphylium: 184. 1945.
Alternaria nepalensis E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 480. 2007.
Section Nimbya (E.G. Simmons) Woudenb. & Crous, comb. et stat. nov. MycoBank MB803742. Fig. 16.
Fig. 16.
Alternaria sect. Nimbya: conidia and conidiophores. A-B. A. caricis. C-D. A. scirpicola. Scale bars = 10 μm.
Basionym: Nimbya E.G. Simmons, Sydowia 41: 316. 1989.
Type species: Alternaria scirpicola (Fuckel) Sivan.
Diagnosis: Section Nimbya contains simple, short to moderately long conidiophores, which may form one or a few short to long, geniculate, sympodial proliferations. Conidia are narrowly elongate-obclavate, gradually tapering apically, solitary or in short chains, with transverse disto- and eusepta, sometimes slightly constricted near eusepta. Apical condiophores with a single conidiogenous locus can be formed. Internal compartmentation occurs, cell lumina tend to be broadly octagonal to rounded. A sexual morph may occur.
Notes: Section Nimbya contains the type species of Nimbya, N. scirpicola, and N. caricis (Simmons 1989). A more extensive study on Nimbya (Lawrence et al. 2012) found that N. scirpinfestans and N. scirpivora also belonged to this section based on sequences of the GAPDH, ITS and Alt a 1 genes.
Alternaria caricis (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803700.
Basionym: Nimbya caricis E.G. Simmons, Sydowia 41: 328. 1989.
Alternaria scirpicola (Fuckel) Sivan., Bitunicate Ascomycetes and their Anamorphs (Vaduz): 526. 1984.
Basionym: Sporidesmium scirpicola Fuckel, Jahrb. Nassauischen Vereins Naturk. 23-24: 140. 1870 [1869-70].
≡ Clasterosporium scirpicola (Fuckel) Sacc., Syll. Fungorum (Abellini) 4: 393. 1886.
≡ Cercospora scirpicola (Fuckel) Zind.-Bakker, Rev. Mycol. (Paris) 5: 66. 1940.
≡ Alternaria scirpicola (Fuckel) M.T. Lucas & J. Webster, Čas. Slez. Mus., Ser. A, Hist. Nat. 23: 151. 1974 (nom. inval.).
≡ Nimbya scirpicola (Fuckel) E.G. Simmons, Sydowia 41: 316. 1989.
-
= Sphaeria scirpicola DC., in Lamarck & de Candolle, Fl. Franç., Edn 3 (Paris) 2: 300. 1805.
≡ Clathrospora scirpicola (DC.) Höhn., Ann. Mycol. 18(1/3): 77. 1920.
≡ Macrospora scirpicola (DC.) Fuckel, Jahrb. Nassauischen Vereins Naturk. 23-24: 139. 1870 [1869-70].
≡ Pyrenophora scirpicola (DC.) E. Müll., Sydowia 5(3-6): 256. 1951.
Note: Although Sphaeria scirpicola DC. (de Candolle 1805) predates Sporidesmium scirpicola Fuckel (Fuckel 1870), a valid combination in Alternaria already exists, thus we choose to retain Alternaria scirpicola (Fuckel) Sivan., which is also a well established name.
Alternaria scirpinfestans (E.G. Simmons & D.A. Johnson) Woudenb. & Crous, comb. nov. MycoBank MB803701.
Basionym: Nimbya scirpinfestans E.G. Simmons & D.A. Johnson, Mycotaxon 84: 420. 2002.
= Macrospora scirpinfestans E.G. Simmons & D.A. Johnson, Mycotaxon 84: 417. 2002.
Alternaria scirpivora (E.G. Simmons & D.A. Johnson), Woudenb. & Crous, comb. nov. MycoBank MB803702.
Basionym: Nimbya scirpivora E.G. Simmons & D.A. Johnson, Mycotaxon 84: 424. 2002.
= Macrospora scirpivora E.G. Simmons & D.A. Johnson, Mycotaxon 84: 422. 2002.
Section Panax D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 541. 2013. Fig. 17.
Fig. 17.
Alternaria sect. Panax: conidia and conidiophores. A-B. A. avenicola. C-D. A. calycipyricola. E-F. A. panax. G-H. A. photistica. Scale bars = 10 μm.
Type species: Alternaria panax Whetzel
Diagnosis: Section Panax contains simple or branched, short to moderately long primary conidiophores, with one or a few conidiogenous loci. Conidia are obclavate to ovoid, with multiple transverse and longitudinal septa, conspicuously constricted near several transverse septa, solitary or in simple or branched, short chains. Apical secondary conidiophores are formed with one or several conidiogenous loci, multiple lateral secondary conidiophores with a single conidiogenous locus may occur.
Notes: Section Panax was recently described by Lawrence et al. (2013) and consists of A. calycipyricola, A. eryngii and A. panax. Our extended dataset added the species A. avenicola and A. photistica to this section. Three species, A. avenicola, A. calycipyricola, and A. photistica have earlier been placed in the A. infectoria species-group based on their morphological characters (Simmons 2007), and two of them have a known sexual morph; Lewia avenicola (Simmons 2007) and Lewia photistica (Simmons 1986). A phylogenetic study based on Alt a 1 and GAPDH sequences placed A. photistica in the A. infectoria species-group (Hong et al. 2005) but an extensive study on the A. infectoria species-group (Andersen et al. 2009) confirmed our finding, and placed this species outside the A. infectoria species-group. Additional research performed on multiple A. photistica strains support our sequence data (data not shown).
Alternaria avenicola E.G. Simmons, Kosiak & Kwaśna, in Simmons, CBS Biodiversity Ser. (Utrecht) 6: 114. 2007.
= Lewia avenicola Kosiak & Kwaśna, Mycol. Res. 107: 371. 2003.
Alternaria calycipyricola R.G. Roberts, Mycotaxon 100: 162. 2007.
Alternaria eryngii (Pers.) S. Hughes & E.G. Simmons, Canad. J. Bot. 36: 735. 1958.
Basionym: Conoplea eryngii Pers., Mycol. Eur. (Erlanga) 1: 11. 1822.
≡ Exosporium eryngianum (Pers.) Chevall., Flore Générale des Environs de Paris 1: 39. 1826.
≡ Exosporium eryngii (Pers.) Duby, Bot. Gallicum., Edn 2 (Paris) 2: 882. 1830.
≡ Helminthosporium eryngii (Pers.) Fr., Syst. Mycol. (Lundae) 3: 361. 1832.
Alternaria panax Whetzel, Bull. U.S.D.A. 250: 11. 1912.
= Macrosporium araliae Dearn. & House, Circ. New York State Mus. 24: 58. 1940.
= Alternaria araliae H.C. Greene, Trans. Wisconsin Acad. Sci. 42: 80. 1953.
Alternaria photistica E.G. Simmons, Mycotaxon 25: 304. 1986.
= Lewia photistica E.G. Simmons, Mycotaxon 25: 302. 1986.
Section Phragmosporae Woudenb. & Crous, sect. nov. MycoBank MB803743. Fig. 18.
Fig. 18.
Alternaria sect. Phragmosporae: conidia and conidiophores. A-B. A. didymospora. C. A. phragmospora. D-E. A. limaciformis. F-G. A. molesta. H-I. A. mouchaccae. Scale bars = 10 μm.
Type species: Alternaria phragmospora Emden
Diagnosis: Section Phragmosporae contains simple, short to moderately long, primary conidiophores, with one or multiple geniculate, sympodial proliferations. Conidia are (broad) ovoid to long ovoid, ellipsoid, curved, or limaciform, with multiple transverse and few to multiple longitudinal septa, some septa darkened, slightly to conspicuously constricted near several transverse septa, solitary or in simple short chains. Apical secondary conidiophores are formed with one or several conidiogenous loci. All species within the section are known from soil and seawater environments.
Note: Section Phragmosporae contains six species of which two were linked to Embellisia.
Alternaria chlamydospora Mouch. [as “chlamydosporum”], Mycopathol. Mycol. Appl. 50: 217. 1973.
Alternaria didymospora (Munt.-Cvetk.) Woudenb. & Crous, comb. nov. MycoBank MB803709.
Basionym: Embellisia didymospora Munt.-Cvetk., Mycologia 68: 49. 1976.
Alternaria limaciformis E.G. Simmons, Mycotaxon 13: 24. 1981.
Alternaria molesta E.G. Simmons, Mycotaxon 13: 17. 1981.
Alternaria mouchaccae E.G. Simmons, Mycotaxon 13: 18. 1981.
≡ Ulocladium chlamydosporum Mouch., Rev. Mycol. (Paris) 36: 114. 1971, non Alternaria chlamydospora Mouch., 1973.
Alternaria phragmospora Emden, Acta Bot. Neerl. 19: 393. 1970.
≡ Embellisia phragmospora (Emden) E.G. Simmons, Mycotaxon 17: 232. 1983.
Section Porri D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 541. 2013. Fig. 19
Fig. 19.
Alternaria sect. Porri: conidia and conidiophores. A-C. A. daucii. D-F. A. pseudorostrata. G-H. A. solani. Scale bars = 10 μm.
Type species: Alternaria porri (Ellis) Cif.
Diagnosis: Section Porri is characterised by broadly ovoid, obclavate, ellipsoid, subcylindrical or obovoid (medium) large conidia, disto- and euseptate, solitary or in short to moderately long chains, with a simple or branched, long to filamentous beak. Conidia contain multiple transverse and longitudinal septa and are slightly constricted near some transverse septa. Secondary conidiophores can be formed apically or laterally.
Notes: In addition to the six species that are displayed in our phylogeny, 40 more are included based on the study of Lawrence et al. (2013), confirmed with own molecular data (not shown). With almost 80 species section Porri is the largest Alternaria section (data not shown). The section displays a higher level of genetic variation than the second largest section; section Alternata.
Alternaria acalyphicola E.G. Simmons, Mycotaxon 50: 260. 1994.
Alternaria agerati Sawada ex E.G. Simmons, Mycotaxon 65: 63. 1997.
= Alternaria agerati Sawada, Rep. Dept. Agric. Gov. Res. Inst. Formosa 86: 165. 1943. (nom. inval., Art. 36.1)
Alternaria agripestis E.G. Simmons & K. Mort., Mycotaxon 50: 255. 1994.
Alternaria anagallidis A. Raabe, Hedwigia 78: 87. 1939.
Alternaria aragakii E.G. Simmons, Mycotaxon 46: 181. 1993.
Alternaria argyroxiphii E.G. Simmons & Aragaki, Mycotaxon 65: 40. 1997.
Alternaria bataticola Ikata ex W. Yamam., Trans. Mycol. Soc. Japan 2(5): 89. 1960.
= Macrosporium bataticola Ikata, Agric. Hort. (Tokyo) 22: 241. 1947 (nom. inval., Art. 36.1).
Alternaria blumeae E.G. Simmons & Sontirat, Mycotaxon 65: 81. 1997.
Alternaria calendulae Ondřej, Čas. Slez. Mus. v Opave˘, Ser. A, Hist. Nat. 23(2): 150. 1974.
= Alternaria calendulae W. Yamam. 1939 (nom. nud.).
= Macrosporium calendulae Nelen, Bull. Centr. Bot. Gard. (Moscow) 35: 90. 1959 (nom. inval., Art. 36.1).
= Macrosporium calendulae Nelen, Bot. Mater. Otd. Sporov. Rast. Bot. Inst. Akad. Nauk S.S.S.R. 15: 144. 1962.
= Alternaria calendulae Nirenberg, Phytopathol. Z. 88(2): 108. 1977 (nom. illegit., Art. 53.1).
Alternaria capsici E.G. Simmons, Mycotaxon 75: 84. 2000.
Alternaria carthami S. Chowdhury, J. Indian Bot. Soc. 23: 65. 1944.
= Macrosporium anatolicum A. Săvul., Bull. Sect. Sci. Acad. Roumaine 26: 709. 1944.
Alternaria cassiae Jurair & A. Khan, Pakistan J. Sci. Industr. Res. 3(1): 72. 1960.
Alternaria cichorii Nattrass, First List of Cyprus Fungi: 29. 1937.
≡ Alternaria porri f.sp. cichorii (Natrass) T. Schmidt, Pflanzenschutz-berichte 32: 181. 1965.
≡ Macrosporium cichorii (Nattrass) Gordenko, Mikol. Fitopatol. 9(3): 241. 1975.
Alternaria cirsinoxia E.G. Simmons & K. Mort., Mycotaxon 65: 72. 1997.
Alternaria crassa (Sacc.) Rands, Phytopathology 7: 337. 1917.
Basionym: Cercospora crassa Sacc., Michelia 1(no. 1): 88. 1877.
Alternaria cretica E.G. Simmons & Vakal., Mycotaxon 75: 64. 2000.
Alternaria cucumerina (Ellis & Everh.) J.A. Elliott, Amer. J. Bot. 4: 472. 1917.
Basionym: Macrosporium cucumerinum Ellis & Everh., Proc. Acad. Nat. Sci. Philadelphia 47: 440. 1895.
Alternaria cyphomandrae E.G. Simmons, Mycotaxon 75: 86. 2000.
Alternaria danida E.G. Simmons, Mycotaxon 65: 78. 1997.
Alternaria dauci (J.G. Kühn) J.W. Groves & Skolko, Canad. J. Res., Sect. C, Bot. Sci. 22: 222. 1944.
Basionym: Sporidesmium exitiosum var. dauci J.G. Kühn, Hedwigia 1: 91. 1855.
Additional synonyms in Simmons 2007.
Alternaria dichondrae Gambogi, Vannacci & Triolo, Trans. Brit. Mycol. Soc. 65(2): 323. 1975.
Alternaria euphorbiicola E.G. Simmons & Engelhard, Mycotaxon 25: 196. 1986.
≡ Macrosporium euphorbiae Reichert, Bot. Jahrb. Syst. 56: 723. 1921. (nom. illegit., Art 53.1).
Alternaria grandis E.G. Simmons, Mycotaxon 75: 96. 2000.
Alternaria hawaiiensis E.G. Simmons, Mycotaxon 46: 184. 1993.
Alternaria limicola E.G. Simmons & M.E. Palm, Mycotaxon 37: 82. 1990.
Alternaria linicola J.W. Groves & Skolko, Canad. J. Res., Sect. C, Bot. Sci. 22: 223. 1944.
Alternaria macrospora Zimm., Ber. Land-Forstw. Deutsch-Ostafrika 2: 24. 1904.
≡ Macrosporium macrosporum (Zimm.) Nishikado & Oshima, Agric. Res. (Kurashiki) 36: 391. 1944.
-
= Sporidesmium longipedicellatum Reichert, Bot. Jahrb. Syst. 56: 723. 1921.
≡ Alternaria longipedicellata (Reichert) Snowden, Rep. Dept. Agric. Uganda: 31. 1927 [1926].
Alternaria multirostrata E.G. Simmons & C.R. Jacks., Phytopathology 58: 1139. 1968.
Alternaria nitrimali E.G. Simmons & M.E. Palm, Mycotaxon 75: 93. 2000.
Alternaria passiflorae J.H. Simmonds, Proc. Roy. Soc. Queensland. 49: 151. 1938.
Alternaria poonensis Ragunath, Mycopathol. Mycol. Appl. 21: 315. 1963.
Alternaria porri (Ellis) Cif., J. Dept. Agric. Porto Rico 14: 30. 1930 [1929].
Basionym: Macrosporium porri Ellis, Grevillea 8 (no. 45): 12. 1879.
Alternaria protenta E.G. Simmons, Mycotaxon 25: 207. 1986.
Alternaria pseudorostrata E.G. Simmons, Mycotaxon 57: 398. 1996.
Alternaria ricini (Yoshii) Hansf., Proc. Linn. Soc. Lond.: 53. 1943.
Basionym: Macrosporium ricini Yoshii, Bult. Sci. Fak. Terk. Kjusu Imp. Univ. 3(4): 327. 1929.
Alternaria rostellata E.G. Simmons, Mycotaxon 57: 401. 1996.
Alternaria scorzonerae (Aderh.) Loer., Netherlands J. Pl. Pathol. 90(1): 37. 1984.
Basionym: Sporidesmium scorzonerae Aderh.,Arbeiten Kaiserl. Biol. Anst. Land-Forstw. 3: 439. 1903.
Alternaria sesami (E. Kawam.) Mohanty & Behera, Curr. Sci. 27: 493. 1958.
Basionym: Macrosporium sesami E. Kawam., Fungi 1(2): 27. 1931.
Alternaria solani Sorauer, Z. Pflanzenkrankh. Pflanzenschutz 6: 6. 1896.
-
= Macrosporium solani Ellis & G. Martin, Amer. Naturalist 16(12): 1003. 1882
≡ Alternaria solani (Ellis & G. Martin) L.R. Jones & Grout, Vermont Agric. Exp. Sta. Annual Rep. 9: 86. 1896.
Additional synonyms in Simmons (2007).
Alternaria solani-nigri R. Dubey, S.K. Singh & Kamal [as “solani-nigrii”], Microbiol. Res. 154(2): 120. 1999.
Alternaria steviae Ishiba, T. Yokoy. & Tani, Ann. Phytopathol. Soc. Japan 48(1): 46. 1982.
Alternaria subcylindrica E.G. Simmons & R.G. Roberts, Mycotaxon 75: 62. 2000.
Alternaria tagetica S.K. Shome & Mustafee, Curr. Sci. 35: 370. 1966.
Alternaria tomatophila E.G. Simmons, Mycotaxon 75: 53. 2000.
Alternaria tropica E.G. Simmons, Mycotaxon 46: 187. 1993.
Alternaria zinniae H.Pape ex M.B. Ellis, Mycol. Pap. 131: 22. 1972.
= Alternaria zinniae H. Pape, Angew. Bot. 24: 61. 1942. (nom. inval., Art. 36.1)
Section Pseudoulocladium Woudenb. & Crous, sect. nov. MycoBank MB803744. Fig. 20.
Fig. 20.
Alternaria sect. Pseudoulocladium: conidia and conidiophores. A-B. A. aspera. C-D. A. concatenata. E-F. A. chartarum. G-H. A. septospora. Scale bars = 10 μm.
Type species: Alternaria chartarum Preuss
Diagnosis: Section Pseudoulocladium is characterised by simple or branched conidiophores with short, geniculate, sympodial proliferations. Conidia are obovoid, non-beaked with a narrow base, in simple or (mostly) branched chains. Apical secondary conidiophores with multiple conidiogenous loci and lateral secondary conidiophores with a single conidiogenous locus can be formed.
Note: It forms a sister clade to section Ulocladioides.
Alternaria aspera Woudenb. & Crous, nom. nov. MycoBank MB803712.
Basionym: Ulocladium arborescens E.G. Simmons, Stud. Mycol. 50: 117. 2004, non Alternaria arborescens E.G. Simmons, 1999.
Etymology: Name refers to the conspicuously ornamented conidia.
Alternaria chartarum Preuss, Bot. Zeitung 6: 412, 1848.
≡ Sporidesmium polymorphum var. chartarum (Preuss) Cooke, Fungi Brit. Exs., ser. 2: 329. 1875.
≡ Ulocladium chartarum (Preuss) E.G. Simmons, Mycologia 59: 88. 1967.
-
= Alternaria stemphylioides Bliss, Mycologia 36: 538. 1944.
≡ Alternaria chartarum f. stemphylioides (Bliss) P. Joly, Encycl. Mycol. (Paris) 33: 161. 1964.
Alternaria concatenata Woudenb. & Crous, nom. nov. MycoBank MB803713.
Basionym: Ulocladium capsici F. Xue & X.G. Zhang [as “capsicuma”], Sydowia 59: 174. 2007, non Alternaria capsici E.G. Simmons, 2000.
Eymology: Name refers to the concatenated conidia.
Alternaria septospora (Preuss) Woudenb. & Crous, comb. nov. MycoBank MB803714.
Basionym: Helminthosporium septosporum Preuss, Linnaea 24: 117. 1851.
≡ Macrosporium septosporum (Preuss) Rabenh., Bot. Zeitung 9: 454. 1851.
≡ Ulocladium septosporum (Preuss) E.G. Simmons, Mycologia 59: 87. 1967.
Section Radicina D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 541. 2013. Fig. 21.
Fig. 21.
Alternaria sect. Radicina: conidia and conidiophores. A-C. A. carotiincultae. D-E. A. petroselini. F-G. A. radicina. H-I. A. selini. J-L. A. smyrnii. Scale bars = 10 μm.
Type species: Alternaria radicina Meier, Drechsler & E.D. Eddy
Diagnosis: Section Radicina contains straight, simple or branched, short or long, primary conidiophores with multiple, short geniculate, sympodial proliferations with single or a few conidiogenous loci at the apex. Sporulation resembles a cluster or clumps of conidia. Conidia are widely ovoid to narrowly ellipsoid, moderate in size, beakless, with several transverse and longitudinal septa, solitary or in short chains. Solitary, short, apical secondary conidiophores may occur. The species from this section occur on Umbelliferae.
Note: This section was first recognised by Pryor & Gilbertson (2000) based on sequence data of the ITS and mitochondrial SSU.
Alternaria carotiincultae E.G. Simmons, Mycotaxon 55: 103. 1995.
Alternaria petroselini (Neerg.) E.G. Simmons, More dematiaceous hyphomycetes (Kew): 417. 1976.
Basionym: Stemphylium petroselini Neerg., Zentralbl. Bakteriol., 2. Abt., 104: 411. 1942.
≡ Stemphylium radicinum var. petroselini (Neerg.) Neerg., Danish species of Alternaria & Stemphylium: 357. 1945.
≡ Alternaria radicina var. petroselini (Neerg.) Neerg., Encycl. Mycol. 33: 123. 1964.
Alternaria radicina Meier, Drechsler & E.D. Eddy, Phytopathology 12: 157. 1922.
≡ Stemphylium radicinum (Meier, Drechsler & E.D. Eddy) Neerg., Annual Rep. Phytopathol. Lab. J.E. Ohlsens Enkes, Seed Growers, Copenhagen 4: 14. 1939.
≡ Thyrospora radicina (Meier, Drechsler & E.D. Eddy) Neerg., Bot. Tidsskr. 44: 361. 1939.
≡ Pseudostemphylium radicinum (Meier, Drechsler & E.D. Eddy) Subram., Curr. Sci. 30: 423. 1961.
Alternaria selini E.G. Simmons, Mycotaxon 55: 109. 1995.
Alternaria smyrnii (P. Crouan & H. Crouan) E.G. Simmons, Mycotaxon 55: 41. 1995.
Basionym: Helminthosporium smyrnii P. Crouan & H. Crouan, Florule Finistère (Paris): 11. 1867.
≡ Macrosporium smyrnii (P. Crouan & H. Crouan) Sacc., Syll. Fungorum (Abellini) 4: 527. 1886.
Section Sonchi D.P. Lawr., Gannibal, Peever & B.M. Pryor, Mycologia 105: 542. 2013. Fig. 22.
Fig. 22.
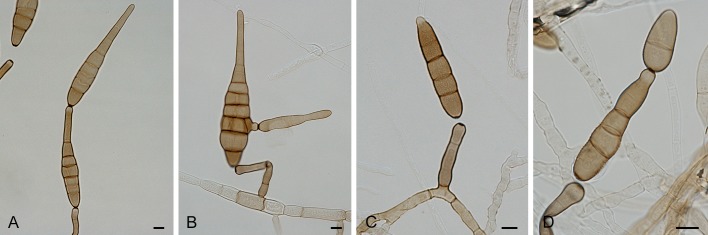
Alternaria sect. Sonchi: conidia and conidiophores. A-B. A. cinerariae. C-D. A. sonchi. Scale bars = 10 μm.
Type species: Alternaria sonchi Davis
Diagnosis: Section Sonchi is characterised by subcylindrical, broadly ovoid, broadly ellipsoid or obclavate, (medium) large conidia, single or in short chains, with multiple transverse and few longitudinal septa, slightly constricted at the septa, with a blunt taper which can form secondary conidiophores.
Notes: The species-group was described by Hong et al. (2005) based on molecular data of the GAPDH and Alt a 1 regions. Lawrence et al. (2013) included A. brassicae as a basal lineage in sect. Sonchi, which is supported as a monotypic lineage in our analyses. The species from section Sonchi occur on multiple hosts within the Compositae.
Alternaria cinerariae Hori & Enjoji, J. Pl. Protect. 18: 432. 1931.
Alternaria sonchi Davis, in Elliott, Bot. Gaz. 62: 416. 1916.
Section Teretispora (E.G. Simmons) Woudenb. & Crous, comb. et stat. nov. MycoBank MB803745. Fig. 23.
Fig. 23.
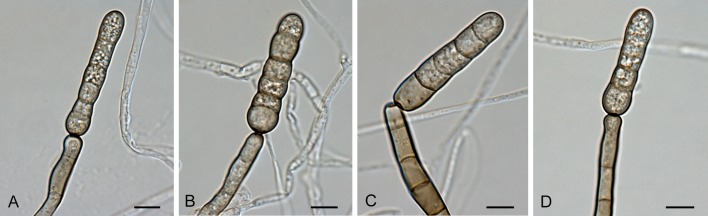
Alternaria sect. Teretispora: conidia and conidiophores. A-D. A. leucanthemi. Scale bars = 10 μm.
Basionym: Teretispora E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 674. 2007.
Type species: Alternaria leucanthemi Nelen
Diagnosis: Section Teretispora is characterised by simple conidiophores, sometimes extending at the apex with one or two, geniculate, sympodial proliferations, bearing single, long cylindrical mature conidia lacking a beak portion, with many transverse and a few longitudinal septa, constricted at most of the transverse septa. Secondary conidiophores with a single conidium are rarely formed at the apex; instead, they may form from the base of the primary conidium.
Notes: The genus Teretispora had Teretispora leucanthemi, formerly Alternaria leucanthemi (= Alternaria chrysanthemi), as type and only species (Simmons 2007). We choose to treat this as a section, which retains the name Teretispora, rather than a monotypic lineage.
Alternaria leucanthemi Nelen, in Nelen & Vasiljeva, Bot. Mater. Otd. Sporov. Rast. Bot. Inst. Akad. Nauk S.S.S.R. 15: 148. 1962.
≡ Teretispora leucanthemi (Nelen) E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 674. 2007.
= Alternaria leucanthemi Nelen, Bull. Centr. Bot. Gard. (Moscow) 35: 83. 1959. (nom. inval., Art. 36.1)
= Alternaria chrysanthemi E.G. Simmons & Crosier, Mycologia 57: 142. 1965.
Section Ulocladioides Woudenb. & Crous, sect. nov. MycoBank MB803746. Fig. 24.
Fig. 24.
Alternaria sect. Ulocladioides: conidia and conidiophores. A-B. A. atra. C-D. A. brassicae-pekinensis. E-F. A. cantlous. G-H. A. multiformis. I-J. A. obovoidea. K-L. A. heterospora. M-N. A. subcucurbitae. O-P. A. terricola. Scale bars = 10 μm.
Type species: Alternaria cucurbitae Letendre & Roum.
Diagnosis: Section Ulocladioides is characterised by conidiophores with short, geniculate, sympodial proliferations. Conidia are obovoid, non-beaked with a narrow base, single or in chains, which may form secondary conidiophores at the apex.
Note: Section Ulocladioides resembles section Ulocladium and contains the majority of the species included in this study from the genus Ulocladium (11/17).
Alternaria atra (Preuss) Woudenb. & Crous, comb. nov. MycoBank MB803717.
Basionym: Ulocladium atrum Preuss, Linnaea 25: 75. 1852.
≡ Stemphylium atrum (Preuss) Sacc., Syll. Fungorum (Abellini) 4: 520. 1886.
Alternaria brassicae-pekinensis Woudenb. & Crous, nom. nov. MycoBank MB803723.
Basionym: Ulocladium brassicae Yong Wang bis & X.G. Zhang, Mycologia 100: 457. 2008, non Alternaria brassicae (Berk.) Sacc., 1880.
Etymology: Name refers to the host from which it was originally isolated.
Alternaria cantlous (Yong Wang bis & X.G. Zhang) Woudenb. & Crous, comb. nov. MycoBank MB803719.
Basionym: Ulocladium cantlous Yong Wang bis & X.G. Zhang, Mycologia 102: 376. 2010.
Alternaria consortialis (Thüm.) J.W. Groves & S. Hughes [as “consortiale”], Canad. J. Bot. 31: 636. 1953.
Basionym: Macrosporium consortiale Thüm., Herb. Mycol. Oecon. 9: no. 450. 1876.
≡ Stemphylium consortiale (Thüm.) J.W. Groves & Skolko, Canad. J. Res., Sect. C, Bot. Sci.: 196. 1944.
≡ Pseudostemphylium consortiale (Thüm.) Subram., Curr. Sci. 30: 423. 1961.
≡ Ulocladium consortiale (Thüm.) E.G. Simmons, Mycologia 59: 84. 1967.
= Stemphylium ilicis Tengwall, Meded. Phytopathol. Lab. “Willie Commelin Scholten” 6: 44. 1924.
Alternaria cucurbitae Letendre & Roum., in Roumeguère, Rev. Mycol. (Toulouse) 8 (no. 30): 93. 1886.
≡ Ulocladium cucurbitae (Letendre & Roum.) E.G. Simmons, Mycotaxon 14: 48. 1982.
Alternaria heterospora Woudenb. & Crous, nom. nov. MycoBank MB803724.
Basionym: Ulocladium solani Yong Wang bis & X.G. Zhang, Mycol. Progr. 8: 209. 2009, non Alternaria solani Sorauer, 1896.
Etymology: Name refers to the various conidial morphologies observed during growth.
Alternaria multiformis (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803720.
Basionym: Ulocladium multiforme E.G. Simmons, Canad. J. Bot. 76: 1537. 1999 [1998].
Alternaria obovoidea (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803721.
Basionym: Ulocladium obovoideum E.G. Simmons, Mycotaxon 37: 104. 1990.
Alternaria subcucurbitae (Yong Wang bis & X.G. Zhang) Woudenb. & Crous, comb. nov. MycoBank MB803722.
Basionym: Ulocladium subcucurbitae Yong Wang bis & X.G. Zhang, Mycologia 100: 456. 2008.
Alternaria terricola Woudenb. & Crous, nom. nov. MycoBank MB803725.
Basionym: Ulocladium tuberculatum E.G. Simmons, Mycologia 59: 83. 1967, non Alternaria tuberculata M. Zhang & T.Y. Zhang, 2006. Etymology: Name refers to soil from which it was originally isolated.
Section Ulocladium (Preuss) Woudenb. & Crous, comb. et stat. nov. MycoBank MB803747. Fig. 25.
Fig. 25.
Alternaria sect. Ulocladium: conidia and conidiophores. A-B. A. capsici-annui. C-D. A. oudemansii. E-F. A. alternariae. G-H. A. botrytis. Scale bars = 10 μm.
Basionym: Ulocladium Preuss, Linnaea 24: 111. 1851.
Type species: Alternaria botrytis (Preuss) Woudenb. & Crous
Diagnosis: Section Ulocladium is characterised by simple conidiophores, or with one or two short, geniculate, sympodial proliferations, with (mostly) single, obovoid, non-beaked conidia with a narrow base.
Notes: Section Ulocladium resembles sect. Ulocladioides. The epitype of Ulocladium, U. botrytis CBS 197.67, and the isotype of U. oudemansii (CBS 114.07) cluster with the Sinomyces representative, as do many other strains stored as U. botrytis in the CBS collection (data not shown). Furthermore, a strain stored as A. capsici-annui (CBS 504.74) in the CBS collection clusters within the Sinomyces clade and displays identical morphological features.
Alternaria alternariae (Cooke) Woudenb. & Crous, comb. nov. MycoBank MB803716.
Basionym: Sporidesmium alternariae Cooke, Handb. Brit. Fungi 1: 1440. 1871.
≡ Stemphylium alternariae (Cooke) Sacc., Syll. Fungorum (Abellini) 4: 523. 1886.
≡ Ulocladium alternariae (Cooke) E.G. Simmons, Mycologia 59: 82. 1967.
≡ Sinomyces alternariae (Cooke) Yong Wang bis & X.G. Zhang, Fungal Biol. 115: 194. 2011.
Alternaria botrytis (Preuss) Woudenb. & Crous, comb. nov. MycoBank MB803718.
Basionym: Ulocladium botrytis Preuss, Linnaea 24: 111. 1851.
≡ Stemphylium botryosum var. ulocladium Sacc. (nom. nov.), Syll. Fungorum (Abellini) 4: 522. 1886.
≡ Stemphylium botryosum var. botrytis (Preuss) Lindau, Rabenhorst’s. Kryptog.-Fl., Edn 2 (Leipzig) 1(9): 219. 1908.
Alternaria capsici-annui Săvul. & Sandu, Hedwigia 75: 228. 1936.
Alternaria oudemansii (E.G. Simmons) Woudenb. & Crous, comb. nov. MycoBank MB803715.
Basionym: Ulocladium oudemansii E.G. Simmons, Mycologia 59: 86. 1967.
Section Undifilum (B.M. Pryor, Creamer, Shoemaker, McLain-Romero & Hambl.) Woudenb. & Crous, comb. et stat. nov. MycoBank MB803748. Fig. 26.
Fig. 26.
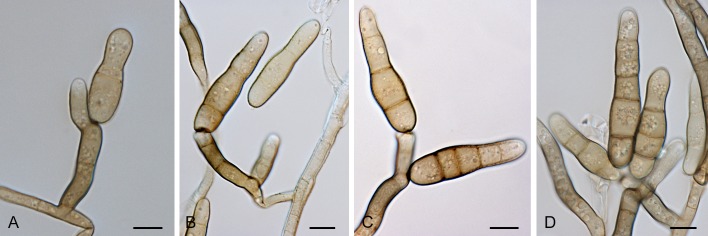
Alternaria sect. Undifilum: conidia and conidiophores. A-D. A. bornmuelleri. Scale bars = 10 μm.
Basionym: Undifilum B.M. Pryor, Creamer, Shoemaker, McLain-Romero & Hambl., Botany 87: 190. 2009.
Type species: Alternaria bornmuelleri (Magnus) Woudenb. & Crous
Diagnosis: Section Undifilum is characterised by ovate to obclavate to long ellipsoid, straight to inequilateral, single, transseptate conidia; septa can be thick, dark and rigid, and form unique germ tubes, which are wavy or undulate until branching. Species of this section occur on Fabaceae and almost all produce the toxic compound swaisonine.
Notes: Section Undifilum shares morphological features with section Embellisia, but is characterised by the formation of a wavy germ tube upon germination (Pryor et al. 2009). Based on previous studies, the swaisonine producing species U. oxytropis (Pryor et al. 2009, Lawrence et al. 2012), U. fulvum and U. cinereum (Baucom et al. 2012) also belong to this section, although the type species, A. bornmuelleri, does not produce swaisonine.
Alternaria bornmuelleri (Magnus) Woudenb. & Crous, comb. nov. MycoBank MB803726.
Basionym: Helminthosporium bornmuelleri Magnus, Hedwigia 38 (Beibl.): 73. 1899.
≡ Undifilum bornmuelleri (Magnus) B.M. Pryor, Creamer, Shoemaker, McLain-Romero & Hambl., Botany 87: 190. 2009.
Alternaria cinerea (Baucom & Creamer) Woudenb. & Crous, comb. nov. MycoBank MB803731.
Basionym: Undifilum cinereum Baucom & Creamer, Botany 90: 872. 2012
Alternaria fulva (Baucom& Creamer) Woudenb. & Crous, comb. nov. MycoBank MB803732.
Basionym: Undifilum fulvum Baucom & Creamer, Botany 90: 871. 2012
Alternaria oxytropis (Q. Wang, Nagao & Kakish.) Woudenb. & Crous, comb. nov. MycoBank MB803727.
Basionym: Embellisia oxytropis Q. Wang, Nagao & Kakish., Mycotaxon 95: 257. 2006.
≡ Undifilum oxytropis (Q. Wang, Nagao & Kakish.) B.M. Pryor, Creamer, Shoemaker, McLain-Romero & Hambl., Botany 87: 191. 2009.
Monotypic lineages
The following six species are not assigned to one of the 24 above described Alternaria sections and are treated as separate, single species, lineages in this study. Future studies, including more and/or new Alternaria species, might eventually give rise to the formation of new sections, when these new species show to be closely related to one of these monotypic lineages.
Alternaria argyranthemi E.G. Simmons & C.F. Hill, Mycotaxon 65: 32. 1997.
Alternaria brassicae (Berk.) Sacc., Michelia 2(no. 6): 129. 1880.
Basionym: Macrosporium brassicae Berk., Engl. Fl., Fungi (Edn 2) (London) 5: 339. 1836.
Additional synonyms listed in Simmons (2007).
Alternaria dennisii M.B. Ellis, Mycol. Pap. 125: 27. 1971.
≡ Embellisia dennisii (M.B. Ellis) E.G. Simmons, Mycotaxon 38: 257. 1990.
Alternaria helianthiinficiens E.G. Simmons, Walcz & R.G. Roberts [as “helianthinficiens”], Mycotaxon 25: 204. 1986.
Alternaria soliaridae E.G. Simmons, CBS Biodiversity Ser. (Utrecht) 6: 374. 2007.
Alternaria thalictrigena K. Schub. & Crous, Fungal Planet No. 12: 2. 2007.
Paradendryphiella Woudenb. & Crous, gen. nov. MycoBank MB803750. Fig. 27.
Fig. 27.
Paradendryphiella gen. nov.: conidia and conidiophores. A-B, D-E, G-I. P. salina. C, F. P. arenariae. Scale bars = 10 μm.
Colonies on SNA effuse, entire, velvety, olivaceous. Reverse olivaceous-grey to iron-grey. Mycelium consisting of branched, septate hypha, (sub)hyaline, smooth. Conidiophores subhyaline, simple or branched, septate or not, straight or flexuous, often nodose with conspicuous, brown pigmentation at the apical region; at times reduced to conidiogenous cells. Conidiogenous cells terminal or lateral, with denticles aggregated at apex, with prominent conidial scars, thickened but not darkened; sometimes proliferating with a new head or a short, inconspicuous sympodial rachis. Conidia produced holoblastically, on narrow denticle, smooth, cylindrical to obclavate, straight or slightly flexuous, 1-7 transverse septa, pale to medium brown, often with dark septa (often constricted), and a darkened zone of pigmentation at the apex, and at the hilum, which is thickened, and somewhat protruding, with a minute marginal frill. Chlamydospores and sexual state not observed.
Type species: Paradendryphiella salina (G.K. Sutherl.) Woudenb. & Crous
Paradendryphiella salina (G.K. Sutherl.) Woudenb. & Crous, comb. nov. MycoBank MB803751.
Basionym: Cercospora salina G.K. Sutherl., New Phytol. 15: 43. 1916.
≡ Dendryphiella salina (G.K. Sutherl.) Pugh & Nicot, Trans. Brit. Mycol. Soc. 47(2): 266. 1964.
≡ Scolecobasidium salinum (G.K. Sutherl.) M.B. Ellis, More dematiaceous hyphomycetes (Kew): 192. 1976.
= Embellisia annulata de Hoog, Seigle-Mur., Steiman & K.-E. Erikss., Antonie van Leeuwenhoek J. Microbiol. Serol. 51: 409. 1985.
Paradendryphiella arenariae (Nicot) Woudenb. & Crous, comb. nov. MycoBank MB803752.
Basionym: Dendryphiella arenariae Nicot, [as “arenaria”] Rev. Mycol. (Paris) 23: 93. 1958.
≡ Scolecobasidium arenarium (Nicot) M.B. Ellis, More dematiaceous hyphomycetes (Kew): 194. 1976.
DISCUSSION
The well-supported node for the Alternaria clade obtained in the present study, and the low bootstrap support at the deeper nodes within the Alternaria complex is also consistently seen in previous phylogenetic studies published on these genera (Pryor & Bigelow 2003, Inderbitzin et al. 2006, Pryor et al. 2009, Runa et al. 2009, Wang et al. 2011, Lawrence et al. 2012). The only phylogenetic study which displays a second fully supported node is based on a five-gene combined dataset of GAPDH, Alt a 1, actin, plasma membrane ATPase and calmodulin (Lawrence et al. 2013). This node, called clade A by the authors, supports eight “asexual” Alternaria species-groups and an Ulocladium (sect. Ulocladioides in our phylogenies) clade. By resolving these eight asexual phylogenetic lineages of Alternaria together with Ulocladium, which is sister to the sexual A. infectoria species-group and other sexual genera, Lawrence et al. (2013) elevated the asexual species-groups to sections within Alternaria. If we take this node as cut-off for the genus Alternaria in our phylogenies, this would leave an Alternaria clade with 14 internal clades (sections) and three monotypic lineages. In order to create a stable phylogenetic taxonomy, seven new genera need to be described of which three would be monotypic; E. dennissii, A. argyranthemi and A. soliaridae. Embellisia species would be assigned to five different genera of which four would be new, leaving only E. allii, E. chlamydospora and E. tellustris in the genus Embellisia. The well-known (medical) A. infectoria species-group would also have to be transferred to a new genus. This node is not supported in our study (0.98 PP /65 ML Fig 1) and also the strict asexual/sexual division is not supported as two sexual morphs are found in section Panax. This approach would therefore give rise to multiple small genera, and would not end up in a logical and workable situation.
Based on our phylogenetic study on parts of the SSU, LSU, ITS, GAPDH, RPB2 and TEF1 gene regions of ex-type and reference strains of Alternaria species and all available allied genera, we resolved a Pleospora/Stemphylium-clade sister to Embellisia annulata, and a well-supported Alternaria clade. The Alternaria clade contains 24 internal clades and six monotypic lineages. In combination with a review of literature and morphology, the species within the Alternaria clade are all recognised here as Alternaria s. str. This puts the genera Allewia, Brachycladium, Chalastospora, Chmelia, Crivellia, Embellisia, Lewia, Nimbya, Sinomyces, Teretispora, Ulocladium, Undifilum and Ybotromyces in synonymy with Alternaria.
The support values for the different sections described in this study are plotted in a heatmap per gene/gene combination and phylogenetic method used (Table 2). This shows that the Bayesian method provides greater support than the Maximum Likelihood bootstrap support values, which is in congruence with previous reports (e.g. Douady et al. 2003). The sections Cheiranthus, Eureka and Nimbya have the lowest support values. For sect. Eureka this is mainly caused by the position of A. cumini, which clusters within sect. Embellisioides based on its RPB2 sequence and as a monotypic lineage based on its TEF1 sequence. Section Cheiranthus and Nimbya are small sections, with relative long branches. Future studies, including more strains and/or species in these sections, are necessary to check the stability of these long branches.
The sexual genus Crivellia with its Brachycladium asexual morph was described by Inderbitzin et al. (2006) with Crivellia papaveraceae (asexual morph Brachycladium penicillatum) as type species and B. papaveris, with an unnamed sexual morph, as second species. The genus Brachycladium, which was synonymised with Dendryphion (Ellis 1971), was resurrected for the non-sexual stage based on polyphyly within Dendryphion and morphological distinction from its type species, D. comosum. The type species of Brachycladium, B. penicillatum, resides in Alternaria sect. Crivellia, which places Brachycladium in synonymy with Alternaria instead of Dendryphion.
The genus Chalastospora was established by Simmons (2007) based on Chalastospora cetera, formerly Alternaria cetera. Two new Chalastospora species, C. ellipsoidea and C. obclavata, and A. malorum as C. gossypii were later added to the genus, based on sequence data of the ITS and LSU regions (Crous et al. 2009c). The genus is characterised by conidia which are almost always narrowly ellipsoid to narrowly ovoid with 1-6 transverse eusepta, generally lacking oblique or longitudinal septa (Crous et al. 2009c). Our study shows that Alternaria armoraciae and Embellisia abundans also belong to this clade. Juvenile conidia of A. armoraciae are ovoid, but vary from being narrow to broadly ovoid and ellipsoid, with 3-5 transverse septa and a single longitudinal septum in up to four of the transverse segments (Simmons 2007). Embellisia abundans was already mentioned as part of the Chalastospora clade (Andersen et al. 2009, Lawrence et al. 2012), and has long ovoid or obclavate conidia with 3-6 transverse septa and rarely any longitudinal septa (Simmons 1983). The description of sect. Chalastospora does therefore not completely follow the original description of the genus Chalastospora.
The genus Embellisia is characterised by the thick, dark, rigid conidial septa and the scarcity of longitudinal septa (Simmons 2007). It was first described by Simmons (1971), with Embellisia allii as type and E. chlamydospora as second species. Multiple Embellisia species followed after the description of the genus, which was later linked to the sexual genus Allewia (Simmons 1990). The latest molecular-based revision was performed based on sequences of the GAPDH, ITS and Alt a 1 genes (Lawrence et al. 2012). They found that Embellisia split into four clades and multiple species, which clustered individually amidst Alternaria, Ulocladium or Stemphylium spp. Our results mostly support these data, but with the inclusion of more ex-type/representative strains of Alternaria some additions were made to the different Embellisia groups mentioned by Lawrence et al. (2012). Group I (sect. Embellisia) and III (sect. Embellisioides) are identical to the treatment of Lawrence et al. (2012) but group II (section Phragmosporae) and IV (section Eureka) are both expanded with four Alternaria species. As not all species from group II and IV display the typical morphological characters of Embellisia, we chose to name these Alternaria sections based on the oldest species residing in the respective sections. Embellisia abundans was already mentioned as being part of the Chalastospora-clade and E. indefessa formed a clade close to Ulocladium, which we now assign to sect. Cheiranthus. Embellisia dennisii also forms a separate lineage in our phylogenies; therefore the old name Alternaria dennissii is resurrected. Furthermore, the clustering of E. conoidea within the A. brassicicola species-group and E. annulata close to Stemphylium, now assigned as Paradendryphiella gen. nov., is confirmed by our phylogenetic data. The morphological character of thick, dark, rigid septa seems to have evolved multiple times and does not appear to be a valid character for taxonomic distinction at generic level.
The sexual morphs Lewia (Simmons 1986) and Allewia (Simmons 1990) were linked to Alternaria and Embellisia respectively, with the only difference between these genera being the morphology of their asexual morphs. Lewia chlamidosporiformans and L. sauropodis are transferred to the genus Leptosphaerulina (Simmons 2007), which leaves 11 Lewia species with a known Alternaria anamorph. Most of them (9/11) reside in sect. Infectoriae, the others are found in sect. Panax. Allewia only contains two species of which one resides in sect. Eureka and one in sect. Embellisioides. With the establishment of the new International Code of Nomenclature for algae, fungi and plants (ICN), the dual nomenclature system for sexual and asexual fungal morphs was abandoned and replaced by a single-name nomenclature (Hawksworth et al. 2011, Norvell 2011). In order to implement the new rules of the ICN, we synonymised Lewia and Allewia with Alternaria.
Although multiple molecular studies included Nimbya isolates in their phylogenies (Chou & Wu 2002, Pryor & Bigelow 2003, Hong et al. 2005, Inderbitzin et al. 2006, Pryor et al. 2009), a more extensive molecular-based study was recently published by Lawrence et al. (2012). Based on sequences of the GAPDH, ITS and Alt a 1 genes, the authors found a Nimbya clade which contained the type species N. scirpicola together with N. scirpinfestans, N. scirpivora and N. caricis. The N. scirpicola isolate which we included in our study, was assigned to this genus by Simmons (1989) based on morphological characters, as is the one used in other molecular studies (Pryor & Bigelow 2003, Hong et al. 2005, Lawrence et al. 2012). The sequences of the ITS, GAPDH and Alt a 1 genes of these isolates are however not identical, but do cluster in the same clade in the two phylogenies (data not shown), together with the isolate of N. caricis. The N. gomphrenae isolate we included in our phylogeny was not representative of the name. Simmons mentioned in 1989 that Togashi (1926) described two different fungi and deposited the small-spored species in the CBS collection, instead of the large-spored N. gomphrenae isolate. Nimbya gomphrenae CBS 108.27, which does not sporulate anymore, will therefore be treated as “Alternaria sp.”, and resides in sect. Alternata. The ITS sequence of N. gomphrenae from Chou & Wu (2002) actually clusters within sect. Alternantherae. This section was described by Lawrence et al. (2012) and consists of three Nimbya species, which they renamed to Alternaria based on the position of the clade amidst the Alternaria species-groups. Based on the data from Chou & Wu (2002), the name Alternaria gomphrenae is resurrected and placed in sect. Alternantherae.
The genus Sinomyces was described in by Wang et al. (2011) to accommodate Ulocladium alternariae and two new species from China, S. obovoideus and S. fusoides (type). The genus was differentiated from Ulocladium based on its simple conidiophores with a single apical pore or 1-2 short, uniperforate, geniculate sympodial proliferations. Unfortunately, our DNA sequence analyses of the ex-type cultures of the two new species from China (CBS 124114 and CBS 123375) were not congruent with the GAPDH (both species) and Alt a 1 (S. obovoideus) sequences deposited in GenBank (data not shown), leading us to doubt the authenticity of these strains. This matter could not be resolved in spite of contacting the original depositors. The ex-type strain of S. alternariae (CBS 126989) was therefore included as representative of the genus Sinomyces. The presence of the epitype of Ulocladium, U. botrytis CBS 197.67, in this section resulted in us rejecting the name Sinomyces, and calling this sect. Ulocladium. In addition, the presence of U. oudemansii in this section, with conidiophores with 1-5 uniperforate geniculations (Simmons 1967), also disagrees with the mentioned differentiation of Sinomyces from Ulocladium.
The type species of Ulocladium, U. botrytis, was typified by two representative strains QM 7878 (CBS 197.67) and QM 8619 (CBS 198.67) (Simmons 1967). Molecular studies performed afterwards showed that these strains are not identical (de Hoog & Horré 2002). Most molecular studies performed used CBS 198.67 as representative of U. botrytis (Pryor & Gilbertson 2000, Pryor & Bigelow 2003, Hong et al. 2005, Xue & Zhang 2007, Pryor et al. 2009, Runa et al. 2009, Wang et al. 2010, Wang et al. 2011, Lawrence et al. 2012), which clusters in section Ulocladioides. However, de Hoog & Horré (2002) epitypified U. botrytis with CBS 197.67, which clusters with Sinomyces strains, as does Ulocladium oudemansii, now named sect. Ulocladium. Extended phylogenetic analyses on all U. botrytis strains present in the CBS culture collection (16 isolates) also highlight this issue as they cluster either within sect. Ulocladium or sect. Ulocladioides (data not shown), both with one of the representative strains described by Simmons (1967). The suggestion to synonymise Ulocladium with Alternaria has been made several times in the past (Pryor & Gilbertson 2000, Chou & Wu 2002). The latest systematic revision of the genus Ulocladium (Runa et al. 2009) based on sequences from the ITS, GAPDH and Alt a 1 genes supported previous findings of poly- and paraphyletic relationships of Ulocladium among Alternaria, Embellisia and Stemphylium spp. (de Hoog & Horré 2002, Pryor & Bigelow 2003, Hong et al. 2005). Ulocladium alternariae and U. oudemansii, now known as sect. Ulocladium, cluster separately. The core Ulocladium clade, containing the two sister clades now called sect. Ulocladioides and sect. Pseudoulocladium, was confirmed by later studies (Wang et al. 2010, Lawrence et al. 2012). Alternaria cheiranthi and Embellisia indefessa have been linked to Ulocladium (Pryor & Gilbertson 2000, Pryor & Bigelow 2003, Hong et al. 2005, Pryor et al. 2009, Runa et al. 2009, Lawrence et al. 2012), but missed the diagnostic feature of Ulocladium. Our study showed that they form a sister section, sect. Cheiranthus, to sect. Ulocladioides. The confusing taxonomy in this genus strengthens our decision to reduce Ulocladium to synonymy with Alternaria. The characteristics of the former genus Ulocladium are added to the new broader Alternaria generic circumscription.
The genus Undifilum was described by Pryor et al. (2009) to accommodate the species U. oxytropis and U. bornmuelleri. It shares the morphological feature of thick, dark and rigid septa with the genus Embellisia, but was characterised by the formation of a wavy germ-tube upon germination (Pryor et al. 2009). A recent study on fungal endophytes in locoweeds in the US described two new Undifilum species (Baucom et al. 2012). Both new species produce the toxic compound swaisonine, which is also produced by U. oxytropis. Swaisonine is the cause of a neurological disease, locism, of grazing animals, resulting in economic losses in livestock (James & Panter 1989). The production of swaisonine seems to be related to this section, although the type-species, U. bornmuelleri, does not produce this toxin.
The genus Ybotromyces contains one species, Y. caespitosus (originally Botryomyces caespitosus), which was isolated from a skin lesion of a human patient (de Hoog & Rubio 1982). De Hoog et al. (1997) discovered a high similarity to Alternaria spp. based on restriction patterns of the ITS and SSU rDNA. A phylogeny study of melanised meristematic fungi based on their SSU and ITS rDNA sequences (Sterflinger et al. 1999) placed Y. caespitosus within the Pleosporales together with Alternaria and Pleospora. De Hoog & Horré (2002) hypothesized that the ex-type strain of Y. caespitosus, CBS 177.80, is likely a synanamorph of a yet undescribed Alternaria species. Our phylogeny supports this hypothesis, and places the genus in sect. Infectoriae.
Chmelia slovaca, described from dermatic lesions of a human (Svobodová 1966), also clusters with sect. Infectoriae as was shown previously (de Hoog & Horré 2002). The genus produces different types of chlamydospores and sporadically blastospores, but no conidia or conidiophores, which makes it difficult to identify based on morphology. De Hoog & Horré (2002) were confident that Chmelia is a sterile member of A. infectoria, which is in agreement with our results.
Genera unrelated to Alternaria
The placement of the sexual genus Pleospora (1863) with Stemphylium (1833) asexual morphs as basal sister clade to the Alternaria complex is well-documented in multiple molecular studies (Chou & Wu 2002, Pryor & Bigelow 2003, Hong et al. 2005, Pryor et al. 2009, Lawrence et al. 2012). Therefore, we only included the type species of both genera in our phylogenies and used them as outgroup in the Alternaria phylogeny. Pleospora herbarum with its Stemphylium herbarum (CBS 191.86) asexual morph is the type species of the genus Pleospora. Stemphylium botryosum with its Pleospora tarda (CBS 714.68) sexual morph is the type species of the genus Stemphylium.
Embellisia annulata proved to be identical to the marine species Dendryphiella salina, and forms a well-supported clade in the Pleosporaceae together with D. arenariae. Several DNA-based studies (dela Cruz 2006, Jones et al. 2008, Zhang et al. 2009) concluded that the marine Dendryphiella species, D. arenariae and D. salina, belonged to the Pleosporaceae as sister clade to the Pleospora/Stemphylium complex. Furthermore, they showed the type species of Dendryphiella, D. vinosa, to be only distantly related, based on sequences of the ITS, SSU, LSU (Jones et al. 2008) and ITS, TEF1, RPB2 (dela Cruz 2006) gene regions. The transfer of the marine Dendryphiella species to Scolecobasidium (Ellis 1976), was also disputed. Scolecobasidium does not belong to the Pleosporales based on ITS, TEF1, and RPB2 sequences (dela Cruz 2006) and the morphology of the two Dendryphiella species does not fit the generic circumscription of Scolecobasidium (dela Cruz 2006, Jones et al. 2008). Ellis (1976) described denticles on the conidiogenous cells when the conidia become detached. However other observers describe a marginal basal frill on the conidia after detachment, leaving a scar on the conidiophore. We propose to place the two species in the new genus Paradendryphiella as C. arenariae and C. salina. The need for a new genus to accommodate the two species was already suggested by Jones et al. (2008).
A recent study on Diademaceae, a family which is characterised by a flat circular operculum and bitunicate asci (Shoemaker & Babcock 1992), excluded the sexual genera Comoclathris and Clathrospora, and (provisionally) placed them in the Pleosporaceae with alternaria-like asexual morphs (Zhang et al. 2011). Molecular data of two strains (Dong et al. 1998, Schoch et al. 2009) placed them within the Pleosporaceae. A confusing factor is that Dong et al. (1998) use the name Comoclathris baccata in their paper for strain CBS 175.52, but submitted their sequences under the name Clathrospora diplospora to GenBank. Shoemaker & Babcock (1992) synonymised Clathrospora diplospora with Comoclathris baccata, which renders Comoclathris as the correct generic name. The confusion around these genera is illustrated by the fact that the CBS collection currently harbours six strains named as Clathrospora species of which four were renamed by Shoemaker & Babcock in 1992 based on morphological studies, and three of these four strains were even transferred to the genus Comoclathris. The type species of Clathrospora, C. elynae is represented by two strains of which one, CBS 196.54, was also studied morphologically by Shoemaker and Babcock (1992). They form a well-supported clade, located basal to the Pleosporaceae (Fig. 2), outside the Alternaria complex. The type species of Comoclathris, Comoclathris lanata, was not available to us, but the two Comoclathris compressa strains cluster together in a well-supported clade within the Pleosporaceae, also outside the Alternaria complex, which we believe to be the correct phylogenetic placement of the genus. Two other strains, named Comoclathris magna (CBS 174.52) and Clathrospora heterospora (CBS 175.52) by Shoemaker and Babcock (1992), cluster amidst sect. Alternata. Culture studies performed by Simmons (1952) showed the presence of alternaria-like conidia in these cultures and no (mature) ascospore formation. Presumably the species observed by Shoemaker and Babcock (1992) on plant material were lost during cultivation and became replaced by A. alternata species-group isolates. Both strains will be treated as “Alternaria sp.”
The genus Alternariaster was first described by Simmons (2007) with Alternariaster helianthi, formerly Alternaria helianthi or Helminthosporium helianthi, as type and only species. It is distinct from Alternaria by the lack of a pigmented conspicuous internal, circumhilar ring in its conidia and conidiophores. Our study showed that this genus is clearly not part of the Alternaria complex and belongs to the Leptosphaeriaceae (Fig. 2) (Alves et al. 2013).
In the recently published book “The genera of Hyphomycetes” (Seifert et al. 2011) three more genera are linked to Alternaria, namely Pantospora, Briansuttonia and Rhexoprolifer. A recent study on Pantospora included ITS and LSU sequence data of the type species Pantospora guazumae, which placed the genus in Mycosphaerellaceae (Minnis et al. 2011). This refutes the link with Alternaria. The genus Rhexoprolifer was described in 1996 by Matsushima with R. variabilis as type and only species, isolated from South Africa. Rhexoprolifer variabilis has rhexolytic conidial liberation and proliferating conidiophores with both phragmosporous and dictyosporous conidia. Briansuttonia was described in 2004 to accommodate Corynespora alternarioides (Castañeda Ruiz et al. 2004). The distoseptate muriform conidia of Briansuttonia do resemble Alternaria and Stemphylium, but the conidiogenous loci and euseptate conidia of Alternaria and holoblastic conidial ontogeny and euseptate muriform conidia of Stemphylium were enough for the authors to regard their taxon as a different genus. Both asexual genera presently lack molecular data, and we were unable to obtain any living specimens of these taxa. It would be valuable to include both genera in a future study to resolve the connection among genera with muriform conidia and Alternaria.
The description of Alternaria s. str. in the present study is supported by i) a well-supported phylogenetic node in multiple analyses, ii) high similarity of clades within Alternaria based on SSU, LSU and ITS data, and iii) variation in the order of the clades between the different gene phylogenies, which is in congruence with low support values at these deeper nodes. We follow the precedence introduced by Lawrence et al. (2013) to assign the taxonomic status of sections of Alternaria for the different clades found, thus allowing us to retain the former generic names but associated with a different taxonomic status. For end-users, this also results in a more stable and understandable taxonomy and nomenclature.
DEDICATION
We would like to dedicate this manuscript to the late Dr E.G. Simmons, who spent over 50 years of his life researching the systematics of the genus Alternaria. Without the time EGS spent on characterising the species included in this study, and his impeccable strain collection, which he placed in CBS for preservation and further study, the present study would not have been possible.
Acknowledgments
Mrs M. Vermaas is thanked for preparing the photoplates, Mrs J. Bloem for assisting with the molecular work, and Prof. dr B.M. Pryor for sending us the Undifilum isolate. This research was supported by the Dutch Ministry of Education, Culture and Science through an endowment of the FES programme “Making the tree of life work”.
REFERENCES
- Alves JL, Woudenberg JHC, Duarte LL, Crous PW, Barreto RW. (2013). Reappraisal of the genus Alternariaster (Dothideomycetes). Persoonia 31: 77–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen B, Sørensen JL, Nielsen KF, Gerrits van den Ende AHG, Hoog GS de. (2009). A polyphasic approach to the taxonomy of the Alternaria infectoria species-group. Fungal Genetics and Biology 46: 642–656 [DOI] [PubMed] [Google Scholar]
- Baucom DL, Romero M, Belfon R, Creamer R. (2012). Two new species of Undifilum, fungal endophytes of Astragalus (locoweeds) in the United States. Botany 90: 866–875 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berbee ML, Pirseyedi M, Hubbard S. (1999). Cochliobolus phylogenetics and the origin of known, highly virulent pathogens, inferred from ITS and glyceraldehyde-3-phosphate dehydrogenase gene sequences. Mycologia 91: 964–977 [Google Scholar]
- Carbone I, Kohn LM. (1999). A method for designing primer sets for speciation studies in filamentous ascomycetes. Mycologia 91: 553–556 [Google Scholar]
- Castañeda Ruiz RF, Heredia GP, Arias RM, Saikawa M, Minter DW, et al. (2004). Two new hyphomycetes from rainforests of México, and Briansuttonia, a new genus to accommodate Corynespora alternarioides. Mycotaxon 89: 297–305 [Google Scholar]
- Chou HH, Wu WS. (2002). Phylogenetic analysis of internal transcribed spacer regions of the genus Alternaria, and the significance of filament-beaked conidia. Mycological Research 106: 164–169 [Google Scholar]
- Crous PW, Gams W, Stalpers JA, Robert V, Stegehuis G. (2004). MycoBank: an online initiative to launch mycology into the 21st century. Studies in Mycology 50: 19–22 [Google Scholar]
- Crous PW, Verkley GJM, Groenewald JZ, Samson RA. (eds) (2009a). Fungal Biodiversity. CBS laboratory Manual Series 1. CBS-KNAW Fungal Biodiversity Centre, Utrecht, The Netherlands: [Google Scholar]
- Crous PW, Schoch CL, Hyde KD, Wood AR, Gueidan C, et al. (2009b). Phylogenetic lineages in the Capnodiales. Studies in Mycology 64: 17–47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crous PW, Braun U, Wingfield MJ, Wood AR, Shin HD, et al. (2009c). Phylogeny and taxonomy of obscure genera of microfungi. Persoonia 22: 139–161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruz TEE dela. (2006). Marine Dendryphiella species from different geographical locations: an integrated, polyphasic approach to its taxonomy and physioecology. Ph.D. dissertation. Fakültat für Lebenswissenschaften der Technischen Universität Carolo-Wilhelmina, Braunschweig, Germany: [Google Scholar]
- Dong J, Chen W, Crane JL. (1998). Phylogenetic studies of the Leptosphaeriaceae, Pleosporaceae and some other Loculoascomycetes based on nuclear ribosomal DNA sequences. Mycological Research 102: 151–156 [Google Scholar]
- Douady CJ, Delsuc F, Boucher Y, Doolittle WF, Douzery EJP. (2003). Comparison of Bayesian and Maximum Likelihood Bootstrap Measures of Phylogenetic Reliability. Molecular Biology and Evolution 20: 248–254 [DOI] [PubMed] [Google Scholar]
- Elliott JA. (1917). Taxonomic characters of the genera Alternaria and Macrosporium. American Journal of Botany 4: 439–476 [Google Scholar]
- Ellis MB. (1971). Dematiaceous hyphomycetes. Commonwealth Mycological Institute, Kew, UK: [Google Scholar]
- Ellis MB. (1976). More dematiaceous hyphomycetes. Commonwealth Mycological Institute, Kew, UK: [Google Scholar]
- Fries EM. (1832). Systema mycologicum. vol. 3 E. Moritz, Greifswald, Germany: [Google Scholar]
- Hawksworth DL, Crous PW, Redhead SA, Reynolds DR, Samson RA, et al. (2011). The Amsterdam Declaration on Fungal Nomenclature. IMA Fungus 2: 105–112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong SG, Cramer RA, Lawrence CB, Pryor BM. (2005). Alt a 1 allergen homologs from Alternaria and related taxa: analysis of phylogenetic content and secondary structure. Fungal Genetics and Biology 42: 119–129 [DOI] [PubMed] [Google Scholar]
- Hoog GS de, Rubio C. (1982). A new dematiaceous fungus from human skin. Sabouradia 20: 15–20 [DOI] [PubMed] [Google Scholar]
- Hoog GS de, Uijthof JMJ, Gerrits van den Ende AHG, Figge MJ, Weenink XO. (1997). Comparative rDNA diversity in medically significant fungi. Microbiology and Culture Collections 13: 39–48 [Google Scholar]
- Hoog GS de, Gerrits van den Ende AHG. (1998). Molecular diagnostics of clinical strains of filamentous Basidiomycetes. Mycoses 41: 183–189 [DOI] [PubMed] [Google Scholar]
- Hoog GS de, Horré R. (2002). Molecular taxonomy of the Alternaria and Ulocladium species from humans and their identification in the routine laboratory. Mycoses 45: 259–276 [DOI] [PubMed] [Google Scholar]
- Huelsenbeck JP, Ronquist F. (2001). MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17: 754–755 [DOI] [PubMed] [Google Scholar]
- Inderbitzin P, Shoemaker RA, O’Neill NR, Turgeon BG, Berbee ML. (2006). Systematics and mating systems of two fungal pathogens of opium poppy: the heterothallic Crivellia papaveracea with a Brachycladium penicillatum asexual state and a homothallic species with a Brachycladium papaveris asexual state. Canadian Journal of Botany 84: 1304–1326 [Google Scholar]
- James LF, Panter KE. (1989). Locoweed poisoning in livestock. In: Swaisonine and related Glycosidase inhibitors (James LF, Elbein AD, Molyneux RJ, Warren CD, eds). Iowa State University Press, Ames, Iowa, USA: 23–38 [Google Scholar]
- Joly P. (1964). Le genre Alternaria. Encyclopédie mycologique XXXIII, P. Lechevalier, Paris, France: [Google Scholar]
- Jones EBG, Klaysuban A, Pang K-L. (2008). Ribosomal DNA phylogeny of marine anamorphic fungi: Cumulospora varia, Dendryphiella species and Orbimyces spectabilis. The Raffles Bulletin of Zoology Supplement 19: 11–18 [Google Scholar]
- Keissler K von. (1912). Zur kenntnis der pilzflora krains. Beihefte zum Botanischen Zentralblatt 29: 395–440 [Google Scholar]
- Lawrence DP, Park MS, Pryor BM. (2012). Nimbya and Embellisia revisited, with nov. comb for Alternaria celosiae and A. perpunctulata. Mycological Progress 11: 799–815 [Google Scholar]
- Lawrence DP, Gannibal PB, Peever TL, Pryor BM. (2013). The sections of Alternaria: Formalizing species-groups concepts. Mycologia 105: 530–546 [DOI] [PubMed] [Google Scholar]
- Liu YJ, Whelen S, Hall BD. (1999). Phylogenetic relationships among ascomycetes: evidence from an RNA polymerse II subunit. Molecular Biology and Evolution 16: 1799–1808 [DOI] [PubMed] [Google Scholar]
- Matsushima T. (1996). Matsushima Mycological Memoirs No. 9 Matsushima Fungus Collection, Kobe: (published by the author). [Google Scholar]
- Minnis AM, Kennedy AH, Grenier DB, Rehner SA, Bischoff JF. (2011). Asperisporium and Pantospora (Mycosphaerellaceae): epitypifications and phylogenetic placement. Persoonia 27: 1–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nees von Esenbeck CG. (1816). Das system der pilze und schwämme. Wurzburg, Germany: [Google Scholar]
- Nirenberg HI. (1976). Untersuchungen über die morphologische und biologische Differenzierung in der Fusarium-Section Liseola. Mitteilungen aus der Biologischen Bundesanstalt für Land- und Forstwirtschaft Berlin-Dahlem 169: 1–117 [Google Scholar]
- Norvell LL. (2011). Fungal nomenclature. 1. Melbourne approves a new code. Mycotaxon 116: 481–490 [Google Scholar]
- O’Donnell K, Kistler HC, Cigelnik E, Ploetz RC. (1998). Multiple evolutionary origins of the fungus causing Panama disease of banana: Concordant evidence from nuclear and mitochondrial gene genealogies. Proceedings of the National Academy of Sciences of the United States of America 95: 2044–2049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Page RDM. (1996). TreeView: an application to display phylogenetic trees on personal computers. Computer Applications in the Biosciences 12: 357–358 [DOI] [PubMed] [Google Scholar]
- Preuss CGT. (1851). Übersicht untersuchter pilze, besonders aus der Umgegend von Hoyerswerda. Linnaea 24: 99–153 [Google Scholar]
- Pryor BM, Gilbertson RL. (2000). Molecular phylogenetic relationships amongst Alternaria species and related fungi based upon analysis of nuclear ITS and mt SSU rDNA sequences. Mycological Research 104: 1312–1321 [Google Scholar]
- Pryor BM, Bigelow DM. (2003). Molecular characterization of Embellisia and Nimbya species and their relationship to Alternaria, Ulocladium and Stemphylium. Mycologia 95: 1141–1154 [DOI] [PubMed] [Google Scholar]
- Pryor BM, Creamer R, Shoemaker RA, McLain-Romero J, Hambleton S. (2009). Undifilum, a new genus for endophytic Embellisia oxytropis and parasitic Helminthosporium bornmuelleri on legumes. Botany 87: 178–194 [Google Scholar]
- Rayner RW. (1970). A Mycological Colour Chart. Commonwealth Mycological Institute, Kew, UK: [Google Scholar]
- Ronquist F, Huelsenbeck JP. (2003). MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19: 1572–1574 [DOI] [PubMed] [Google Scholar]
- Runa F, Park M, Pryor B. (2009). Ulocladium systematics revisited: phylogeny and taxonomic status. Mycological Progress 8: 35–47 [Google Scholar]
- Saccardo PA. (1886). Sylloge Fungorum Omnium hucusque cognitorum, Volume 4 Padua, Italy: [Google Scholar]
- Schoch CL, Crous PW, Groenewald JZ, Boehm EWA, Burgess TI, et al. (2009). A class-wide phylogenetic assessment of Dothideomycetes. Studies in Mycology 64: 1–15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seifert K, Morgan-Jones G, Gams W, Kendrick B. (2011). The genera of Hyphomycetes. CBS Biodiversity Series 9. CBS Fungal Biodiversity Centre, Utrecht, The Netherlands: [Google Scholar]
- Shoemaker RA, Babcock CE. (1992). Applanodictyosporous Pleosporales: Clathrospora, Comoclathris, Graphyllium, Macrospora, and Platysporoides. Canadian Journal of Botany 70: 1617–1658 [Google Scholar]
- Simmons EG. (1952). Culture studies in the genera Pleospora, Clathrospora, and Leptosphaeria. Mycologia 44: 330–365 [Google Scholar]
- Simmons EG. (1967). Typification of Alternaria, Stemphylium, and Ulocladium. Mycologia 59: 67–92 [PubMed] [Google Scholar]
- Simmons EG. (1971). Helminthosporium allii as type of a new genus. Mycologia 63: 380–386 [Google Scholar]
- Simmons EG. (1983). An aggregation of Embellisia species. Mycotaxon 17: 216–241 [Google Scholar]
- Simmons EG. (1986). Alternaria themes and variations (22-26). Pleospora/Stemphylium and Lewia / Alternaria. Mycotaxon 25: 287–308 [Google Scholar]
- Simmons EG. (1989). Macrospora Fuckel (Pleosporales) and related anamorphs. Sydowia 41: 314–329 [Google Scholar]
- Simmons EG. (1990). Embellisia and related teleomorphs. Mycotaxon 38: 251–265 [Google Scholar]
- Simmons EG. (2002). Alternaria themes and variations (287-304). Species on Caryophyllaceae. Mycotaxon 82: 1–40 [Google Scholar]
- Simmons EG. (2007). Alternaria. An identification manual. CBS Biodiversity Series 6. CBS Fungal Biodiversity Centre, Utrecht, The Netherlands: [Google Scholar]
- Stamatakis A, Alachiotis N. (2010). Time and memory efficient likelihood-based tree searches on phylogenomic alignments with missing data. Bioinformatics 26: i132–i139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sterflinger K, Hoog GS de, Haase G. (1999). Phylogeny and ecology of meristematic ascomycetes. Studies in Mycology 43: 5–22 [Google Scholar]
- Sung G-H, Sung J-M, Hywel-Jones NL, Spatafora JW. (2007). A multi-gene phylogeny of Clavicipitaceae (Ascomycota, Fungi): Identification of localized incongruence using a combinational bootstrap approach. Molecular Phylogenetics and Evolution 44: 1204–1223 [DOI] [PubMed] [Google Scholar]
- Svobodová Y. (1966). Chmelia slovaca gen. nov., a dematiaceous fungus, pathogenic for man and animals. Biológia, Bratislava 21: 81–88 [PubMed] [Google Scholar]
- Togashi K. (1926). On a new species of Alternaria causing a leafspot disease of Gomphrena globosa L. Bulletin of Imperial College of Agriculture and Forestry (Morioka) 9: 1–16 [Google Scholar]
- Toth B, Csosz M, Szabo-Hever A, Simmons EG, Samson RA, Varga J. (2011). Alternaria hungarica sp. nov., a minor foliar pathogen of wheat in Hungary. Mycologia 103: 94–100 [DOI] [PubMed] [Google Scholar]
- Vilgalys R, Hester M. (1990). Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. Journal of Bacteriology 172: 4238–4246 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue F, Zhang XG. (2007). Ulocladium capsicuma, a new species identified by morphological and molecular phylogenetic data. Sydowia 59: 161–178 [Google Scholar]
- Wallroth CFW. (1833). Flora Cryptogamica Germaniae Sectio 2. J.L. Schrag, Nürnberg, Germany: [Google Scholar]
- Wang Y, Pei Y-F, O’Neill NR, Zhang X-G. (2010). Ulocladium cantlous sp. nov. isolated from northwestern China: its morphology and molecular phylogenetic position. Mycologia 102: 374–383 [DOI] [PubMed] [Google Scholar]
- Wang Y, Geng Y, Ma J, Wang Q, Zhang X-G. (2011). Sinomyces: a new genus of anamorphic Pleosporaceae. Fungal Biology 115: 188–195 [DOI] [PubMed] [Google Scholar]
- White TJ, Bruns T, Lee S, Taylor J. (1990). Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: PCR protocols: a guide to methods and applications (Innis MA, Gelfand DH, Sninsky JJ, White TJ, eds). Academic Press, San Diego, California, USA: 315–322 [Google Scholar]
- Wiltshire SP. (1933). The foundation species of Alternaria and Macrosporium. Transactions of the British Mycological Society 18: 135–160 [Google Scholar]
- Wiltshire SP. (1938). The original and modern conceptions of Stemphylium. Transactions of the British Mycological Society 21: 211–239 [Google Scholar]
- Zhang Y, Schoch CL, Fournier J, Crous PW, Gruyter J de, et al. (2009). Multi-locus phylogeny of Pleosporales: a taxonomic, ecological and evolutionary re-evaluation. Studies in Mycology 64: 85–102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang YM, Koko TW, Hyde KD. (2011). Towards a monograph of Dothideomycetes: Studies on Diademaceae. Cryptogamie, Mycologie 32: 115–126 [Google Scholar]