Abstract
Neutral endopeptidase (NEP), which degrades substance P (SP), may regulate neutrophil activation during Mg-deficiency (MgD). Male Sprague-Dawley rats (180g) were fed MgD (~50 mg Mg/kg) or Mg-sufficient (MgS, 608 mg Mg/kg) diets for 7 days ± NEP inhibitor phosphoramidon (PR, 5 mg/kg/day, s. c.). MgD alone induced a 9-fold (vs. MgS, p <0.01) elevation in plasma SP; MgD+PR enhanced it further to 18-fold (p <0.001). Neutrophils from MgD+PR rats displayed a 3.9-fold higher (p <0.01) basal ·O2- generation, but those from MgD or PR alone were not activated. Plasma PGE2-metabolite levels rose 2.67- (p <0.01) and 1.56- (p <0.05) fold, respectively, in MgD+PR and MgD groups; the corresponding red blood cell glutathione levels were decreased 21 % (p <0.025) and 7 % (NS). MgD+PR significantly reduced neutrophil NEP activity by 48 % (p <0.02); PR or MgD alone only reduced this activity 26 % and 15 %, respectively. We conclude that NEP inhibition potentiates SP-mediated neutrophil ·O2- production and may promote other inflammatory activities during MgD.
Keywords: NEP inhibition, Substance P, Mg-deficiency, Neutrophil activation, Oxidative stress
Introduction
We have established with a rat model that severe Mg-deficiency (MgD) results in a significant elevation in circulating substance P (SP) levels within the first week after initiating the MgD diet [1–3]. Other data strongly suggested the early increase in circulating SP results from neuronal release due to N-methyl-D-aspartate (NMDA)-receptor activation [4]. Subsequent to the elevation of SP, we also observed that isolated neutrophils from the 2nd and 3rd week of diet, but not the 1st week, displayed elevated basal activity to generate superoxide anions (O2-) during MgD [5]. Further study indicated that the neutrophil basal activity was substantially (>80 %) attenuated by in vivo treatment of rats with the neurokinin-1 (NK-1) receptor blocker, L-703,606, consistent with the notion that neutrophil activation was directly mediated by SP/NK-1 receptor signaling [6].
Neutral endopeptidase or neprilysin (NEP, EC 3.4.24.11) is a cell-surface metallopeptidase [7] which hydrolyzes a variety of naturally occurring peptides including neuropeptides [8] and humoral mediators [7, 9]. NEP is widely distributed in many tissues and is abundant especially in the kidney, brain, and lung [10]. Among neuropeptides, SP is effectively targeted and inactivated by NEP [8]. NEP is also prominently expressed on normal mature neutrophils [11]; it is presumed that neutrophil NEP may play a key role in controlling its own responses to neuropeptide-mediated activation. In this communication, we examined whether inhibition of NEP in vivo affected the status of neutrophils in generating free radicals and the influence on the associated inflammatory indices during the early phase (week 1) of dietary MgD.
Materials and methods
Animals, Diets and Drug treatment
All animal experiments were guided by the principles for the care and use of laboratory animals as recommended by the US Department of Health and Human Services and approved by the The George Washington University Animal Care and Use Committee. Male Sprague-Dawley rats (180 gm, Hilltop Labs, Scottdale, PA) were fed either a Mg-deficient (MgD) diet containing ~50 mg magnesium/kg diet (Harlan Teklad, Madison, WI) or the same diet supplemented to 608 mg magnesium/kg with magnesium oxide (MgS group, [12]). Animals were placed on the diets for 7 days. The NEP inhibitor, phosphoramidon (Sigma-Aldrich Chemicals, St. Louis, MO) was formulated into continuous-release pellets by Innovative Research of America (Sarasota, Fl). The pellets were then administered by subcutaneous implantation to release the drug at a dose of 5 mg/kg/day for the entire dietary period.
Blood sample collection/plasma preparation
Tail bleed (aseptic) samples (0.37 ml) were collected periodically (days 0, 3, 5 or 6) during each diet/treatment regimen using heparinized glass capillary tubes, and spun (3,500 rpm, 10 min, RT) in aprotinin (protease inhibitor, 0.016 U/ml) + heparin (358 U/ml)-containing (12.6 μl) centrifuge tubes. On sacrifice day (day 7), heparinized blood was drawn by cardiac puncture; the plasma fraction was isolated by differential centrifugation as described above and all plasma samples were stored at −80°C until use. Plasma from the tail bleed was used mainly to measure SP whereas blood from the cardiac puncture was a source for neutrophils and red blood cells (RBC).
Neutrophil isolation and superoxide measurement
Neutrophils were obtained from the whole blood using a modified ficoll hypaque reagent which is essentially a step-gradient (upper layer: specific gravity = 1.077 and lower layer: 1.113 g/ml) centrifugation as previously described [5, 6]. Neutrophils were recovered from the lower band, washed and recentrifuged in phosphate buffered saline (PBS), pH 7.4. The contaminating erythrocytes were lysed with hypotonic solution (10 % PBS) for 30 sec; the resulting neutrophil fraction was examined morphologically and found > 90 % purity. The basal and stimulated superoxide anion production was assayed in the neutrophil suspensions (0.5–0.75 × 106 cells/ml of Krebs-Ringer phosphate buffer containing 5 mM glucose, 1 mM CaCl2, 1 mM MgCl2, pH 7.6) in the absence or presence of 100 ng/ml phorbol myristate acetate (PMA) plus 75 μM cytochrome c ± 50 μg superoxide dismutase (SOD). After 20 min of incubation at 30 °C, all samples were immediately immersed in an ice-bath and centrifuged at 600 g for 5 min at 4 °C; superoxide anion production was estimated as SOD-inhibitable reduction of cytochrome c in the supernatant using the extinction coefficient: E550 = 2.1×104M−1cm−1
Substance P measurement
Plasma SP levels were determined using a commercial colorimetric 96 well ELISA kit from R&D Systems (Minneapolis, MN). The assay is a competitive binding technique in which SP in a sample (50 μl of 1:4 diluted plasma read in triplicate) competes with a fixed amount of horseradish - labeled SP for sites on a murine monoclonal antibody. Color development is inversely proportional to SP concentration and absorbance is read with a microplate reader (VersaMax, Molecular Devices, Sunnyvale, CA) at 450 nm using wavelength correction at 540 nm. After normalizing to baseline, time-course data were area integrated over the specified times to obtain total plasma SP levels for that period.
Neprilysin activity assay
NEP enzymatic activity was determined according to the fluorimetric procedure of Miners et al [13] with slight modification. Briefly, using the microtiter plate, 50 μl of fluorogenic peptide substrate [Mca-Arg-Pro-Gly-Phe-Ser-Ala-Phe-Lys-(Dnp)-OH from R&D Systems (Minneapolis, MN)], was added to 100 μl neutrophil suspension in N-[2-hydroxyethyl]piperazine-N’-[2-ethanesulfonic acid] (HEPES) buffer (pH 7.4) and to the same number of cells preincubated for 30 min with 50 μl of 30 μM Thiorphan (Sigma-Aldrich Chemicals, St. Louis, MO). All the assays were performed in duplicate and samples with substrate alone in HEPES buffer were used to monitor nonspecific fluorescence. After 30 min incubation at 37 °C, the relative fluorescence was read on the fluorimeter (Victor 3, Perkin Elmer) with an excitation wavelength at 320 nm and emission 405 nm.
RBC glutathione and Plasma PGE2 metabolite
RBC were centrifuged at 200 g × 10 min; total [reduced glutathione (GSH) +1/2 GSSG] and oxidized (GSSG) glutathione levels were determined by the ‘enzymatic recycling method’ as described previously (12, 14). In data not shown, the level of GSSG represented <5 % of the total glutathione for all samples with or without PR treatment. Plasma PGE2 levels were determined by its 13, 14-dihydro-15-keto metabolite (PGEM), 13, 14-dihydro-15-keto-Prostaglandin A2, which is a stable breakdown product and can reflect a more reliable estimate of PGE2 production in vivo. PGEM levels in the plasma samples were assessed by using a PGEM EIA Kit from Cayman Chemical (Ann Arbor, MI). In brief, derivatization of the single stable PGEM from plasma aliquots (25–50 μl) was performed overnight and the levels were quantified by a competitive enzyme immunoassay according to the manufacturer’s instructions.
Statistics
Results were expressed as means ± standard errors unless otherwise stated. Statistical comparisons were performed by Student’s t test when only two groups were compared; a value of p <0.05 is considered significant. Selected data were analyzed by one-way ANOVA followed by a Tukey’s test.
Results
NEP inhibition and circulating SP levels
We investigated the impact of the NEP inhibitor, phosphoramidon (PR) on circulating SP levels in MgD rats. Previously, we found significant increases in circulating SP levels within the first week on the MgD diet [1–3]. In the current study, we observed that during wk 1, MgD alone induced a substantial elevation in plasma SP (569 ± 34 pg/ml/wk) beyond the baseline; this elevation is 9-fold (p <0.01) higher than that of the MgS controls (62.9 ± 10 pg/ml/wk). PR-treatment of MgD rats during week 1 led to a further 18.3-fold (p <0.001) increase in SP (1156 ± 110 pg/ml/wk) compared to MgS controls. This observation is consistent with systemic inhibition of SP degradation in the PR treated animals.
Neutrophil activation
Without in vitro stimulation, neutrophils isolated from MgS rats displayed only a low level of basal superoxide producing activity (0.133 ± 022 nmol/min/106 cells). Results in Fig. 2 indicate that despite the significant rise in circulating SP observed in the MgD alone animals, only a minor and non-significant (NS) activation in the basal activity of superoxide production (0.204 ± 032 nmol/min) was evident. However, the activity of the neutrophils was markedly and significantly elevated 3.9-fold by PR treatment in the MgD rats (0.513 ± 0.062 nmol/min, p < 0.01 vs. MgS, p <0.025 vs. MgD alone). PR treatment alone in the MgS rats produced no significant effect (0.192 ± 030 nmol/min). In the presence of PMA, neutrophils from all groups were stimulated to a comparable extent (1.1 to 1.3 nmol/min/106 cells), suggesting that the full capacity of the neutrophils to generate free radicals in response to in vitro challenge remained unchanged by the PR treatment. In data not shown, we found that a higher dose of PR (10 mg/kg/day) did not significantly enhance basal superoxide anion production from the MgD neutrophils beyond that achieved by the dose of 5 mg/kg/day.
Fig. 2.

Influence of NEP inhibition by phosphoramidon on basal generation of superoxide anions in rat neutrophils. The rats were fed MgS or MgD diets for 7 days with or without receiving PR as s. c. implantation for the entire period. The neutrophils were isolated from whole blood by step gradient centrifugation; basal superoxide anion production was determined as SOD-inhibitable cytochrome c reduction. Results are means ± SEM of 4–6 animal samples; ** p <0.01 vs. MgS and, # p <0.025 vs. MgD alone.
Neutrophil NEP activity
The effect of PR treatment on the relative neutrophil NEP activity was examined. As represented by Fig. 3, PR alone moderately (26 %, though NS) decreased the NEP activity of neutrophils in the MgS group, and MgD alone only had a modest lowering effect (15 %, NS). However, PR+MgD resulted in a substantial and significant reduction (48 %, p <0.05) in NEP activity compared to the MgS controls.
Fig. 3.

The relative NEP enzymatic activities in neutrophils isolated from MgS or MgD rats (7 days) with or without PR treatment. NEP activity on intact neutrophils were estimated as thiorphan-inhibitable relative fluorescence (excitation = 320 nm, emission = 405 nm). Values are means ± SEM from 4–6 samples; *p < 0.05 vs. MgS control and MgD alone.
PGE2 Metabolite and RBC GSH levels
We previously observed that COX-derived vasoactive mediators, PGE2 and PGI2 were up-regulated during MgD [2, 6]. In this study, we assessed the effect of PR treatment on the plasma levels of the PGE2 metabolite. As indicated by Fig. 4A. MgD alone for 7 days resulted in a moderate, but significant increase (1.56-fold, p <0.05) in PGEM content. The PR and MgD combination elicited a further elevation in this vasoactive mediator to 2.67-fold (p <0.01) above the MgS control level. PR alone in MgS rats did not produce any effect.
Fig. 4.
Effects of NEP inhibition by PR on (A) circulating PGE2-metabolite (PGEM) level and (B) RBC glutathione content. PGEM content in plasma samples were determined by an EIA kit from Cayman Chemical and RBC glutathione was determined by an enzymatic “recycling” method. Values are means ± SEM from 4-6 samples; * p <0.05, **p <0.01 vs. MgS controls; + p <0.05 vs MgD alone or MgS+PR.
Dietary MgD resulted in oxidative depletion of RBC GSH during the 2nd and 3rd weeks of the diet [12, 15]; but the loss of GSH was insignificant during the 1st week. In the present study, we observed that MgD alone for 7 days only resulted in a minor insignificant reduction (7 %) of the RBC GSH (Fig. 4B). However, PR treatment augmented the loss of GSH to 21 %, which was significantly lower than the MgS (p <0.01) or MgD (p <0.05) groups. PR alone did not elicit any change in the GSH level of MgS animals (Fig. 4B).
Discussion
We [5, 6] and others [16] have demonstrated that neutrophil activation represents an ‘important cellular response which may contribute significantly to the systemic oxidative stress associated with MgD. However, the immediate signaling events leading to neutrophil activation are less clear [17]). Severe dietary MgD leads to a dramatic decrease in plasma Mg within one week [18]. Since Mg may act as a natural Ca antagonist [19], one may speculate that decreased extracellular Mg leads to a Ca influx in white cells to activate superoxide production by the NADPH oxidase. However, it seems unlikely that low extracellular Mg alone would directly provide the activation signal, since extracellular Mg decreased greater than 50 % 3 days after initiating the MgD diet [18], yet neutrophil activation did not occur until the 2nd week and continued into the 3rd week [5]. This would suggest that additional mediator(s) are required to promote neutrophil activation. Low extracellular Mg alone can trigger NMDA receptor activation leading to neuronal release of neuropeptides [20, 21]. Indeed, we previously showed that MgD resulted in early elevations of circulating SP and calcitonin gene-related peptide (CGRP) levels, in rodent models [1, 2]. We subsequently presented evidence that SP played a key role in modulating the inflammatory events leading to cardiac lesion development during the progression of MgD [1, 2, 22]. Moreover, we found that activation of neutrophils from MgD rats was blocked by treatment with the NK-1 receptor blocker, L-703,606 [6], strongly suggesting that the activation process was triggered directly by SP binding to cellular NK-1 receptors. In the present study, we found that MgD alone resulted in significant increases in plasma SP levels (Fig. 1). In another study [23], we observed that the early increase in circulating SP levels could be attenuated by in vivo treatment with the NMDA receptor/channel blocker, MK-801. The latter observation further supports the notion that the early increase in plasma SP levels resulted from neuronal release due to NMDA receptor complex activation during the 1st week of MgD.
Fig. 1.
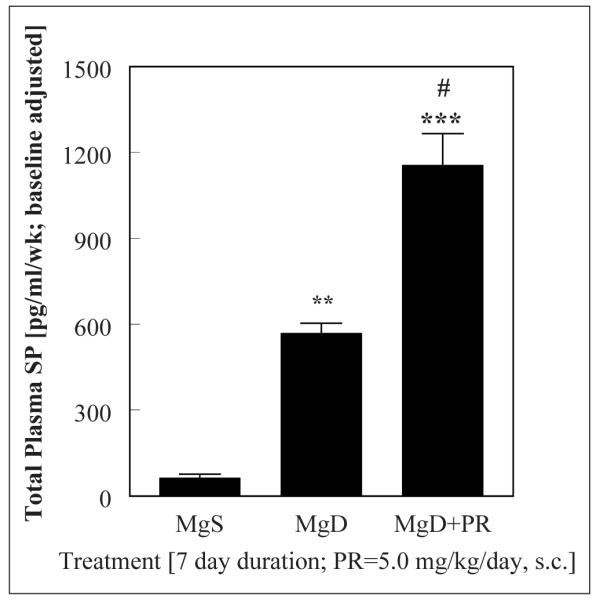
Effect of the neutral endopeptidase inhibitor, phosphoramidon (PR: 5 mg/kg/day, s. c.) on total plasma SP levels (colorimetric ELISA) during week 1 of MgD treatment. After normalizing to baseline (0.447 ng/ml), time-course data were integrated over the specified time. Values are means ± SEM from 4–5 rats/group; **p <0.01, ***p <0.001 vs MgS; # p <0.025 vs MgD alone.
NEP is the major proteolytic enzyme responsible for the degradation of SP [7–10]. Therefore, we investigated the impact of the NEP inhibitor, phosphoramidon, on circulating SP levels in vivo, in conjunction with changes in neutrophil activation and oxidative stress parameters. Rats were treated with PR, which should produce a systemic inhibitory effect on NEP activity in all tissues. In agreement, we found that PR treatment led to a significantly higher circulating SP level in MgD animals. While neutrophils isolated from the MgD rats after 7 days experienced minimal changes in superoxide generation, the cells from PR+MgD rats displayed up to 4-fold higher basal generation of superoxide anions (Fig. 2). In association, while neutrophils from MgD alone rats still retained 85 % of their NEP activity, neutrophils from the PR treated MgD group lost close to 50 % of the NEP activity compared to the MgS controls. NEP on the neutrophil surface may play an important role in controlling the functional level of SP accessible to its NK-1 receptor, and this may modulate SP stimulatory activity. In support of this notion, a related study with isolated human neutrophils [24], showed that NEP inhibition by either thiorphan, or PR, potentiated both neutrophil chemotaxis and adhesion molecule expression towards N-formyl-Met-Leu-Phe and SP. In another in vitro study [25], NEP inhibition by PR reduced the amount of Met-enkephalin required to trigger neutrophil inflammatory responses by several orders of magnitude [25]. These studies suggest that the NEP of neutrophils may function to control the cellular responsiveness to multiple inflammatory peptides.
The pathophysiological importance of NEP related to SP had been documented in several in vivo models of inflammation [26–28]. In a mouse model of acute intestinal inflammation induced by Clostridium difficile toxin A (TxA), Kirkwood et al. [26] demonstrated that inflammation was associated with SP release and activation of NK-1 receptors; genetic deletion of NEP (by using NEP−/− mice) greatly reduced the threshold dose of TxA and exacerbated the inflammatory response by more than two-fold [26]. In the same study, pretreatment of wild-type mice with the NEP inhibitor, PR, exacerbated the induced enteritis. In a related study, Sturiatle et al. [27] showed that the SP level in the colon of NEP−/− mice was 2.5-fold higher than in wild –type mice. Moreover, the extravasation of Evans’ blue-labeled plasma protein in the colon of NEP−/− mice was 4-fold higher than in wild type mice; this elevated plasma protein leak was attenuated by the NK-1 receptor antagonist, SR1540333 [27]. In a mouse model of nematode-induced intestinal inflammation [28], neutrophil accumulation (assessed by myeloperoxidase [MPO] as the enzymatic marker) was delayed at least 24 hrs in NEP+/+ mice, but the onset was accelerated by 12 hrs in NEP−/− mice, which displayed increased SP staining and a 2.5-fold increase in MPO activity. It was concluded that NEP activity prevented the early onset of nematode-induced intestinal inflammation through degradation of SP [28]. In our study, it is noteworthy that SP-mediated inflammation was induced without any external pathogen. It is within the realm of possibility that the immune response to nematode invasion could be accentuated by Mg-deficiency.
In summary, we have demonstrated that inhibition of NEP activity by PR, enhanced the oxidative activity of neutrophils and accelerated RBC GSH loss and PGE2 elevation during the early phase (week 1) of MgD. PGE2 is most likely a COX-2 derived prostanoid produced from the endothelium. COX-2 up-regulation may also be governed by SP, as we observed that PGE2 elevation during MgD was attenuated by SP-receptor blockade [2, 6]. The current findings and those from our previous reports [1, 6, 22] strongly suggest that the enhanced neutrophil activation and other inflammatory activities were due to increased bioavailability of SP. In a preliminary study [29], we observed that neutrophils isolated from rats after two weeks on the MgD diet displayed a 45 % loss in NEP activity and an elevated basal generation of superoxide anions. These observations suggest that NEP plays an important role in modulating neurogenic inflammation during the early weeks of MgD.
Acknowledgments
This study was supported by PHS grants: NIH-HL-62282 and NIH-HL-65178.
References
- [1].Weglicki WB, Mak IT, Phillips TM. Blockade of cardiac inflammation in Mg2+-deficiency by substance P receptor inhibition. Circ Res. 1994;24:1009–13. doi: 10.1161/01.res.74.5.1009. [DOI] [PubMed] [Google Scholar]
- [2].Weglicki WB, Mak IT, Stafford RE, Dickens BF, Cassidy MM, Phillips TM. Neurogenic peptides and the cardiomyopathy of magnesium deficiency: Effects of substance P-receptor inhibition. Mol Cell Biochem. 1994;130:103–9. doi: 10.1007/BF01457391. [DOI] [PubMed] [Google Scholar]
- [3].Kramer JH, Mak IT, Phillips TM, Weglicki WB. Dietary magnesium intake influence circulating pro-inflammatory neuropeptide levels and loss of myocardial tolerance to postischemic stress. Exp Biol Med. 2003;228:665–73. doi: 10.1177/153537020322800604. [DOI] [PubMed] [Google Scholar]
- [4].Tejero-Taldo MI, Chmielinska JJ, Gonzalez G, Mak IT, Weglicki WB. N-Methyl-D-aspartate receptor blockade inhibits cardiac inflammation in the Mg-deficient rat. J Pharmacol Exp Ther. 2004;311:8–13. doi: 10.1124/jpet.104.070003. [DOI] [PubMed] [Google Scholar]
- [5].Mak IT, Dickens BF, Komarov AM, Phillips TM, Weglicki WB. Activation of the neutrophil and loss of plasma glutathione during Mg-deficiency – modulation effect by nitric oxide synthase inhibition. Mol Cell Biochem. 1997;176:35–39. [PubMed] [Google Scholar]
- [6].Mak IT, Kramer JK, Weglicki WB. Suppression of neutrophil and endothelial activation by substance P receptor blockade in the Mg-deficient rat. Magnes Res. 2003;16:91–7. [PubMed] [Google Scholar]
- [7].Erdös EG, Skidgel RA. Neutral endopeptidase 24.11(enkephalinase) and related regulators of peptide hormones. FASEB J. 1989;3:145–51. [PubMed] [Google Scholar]
- [8].Stimler-Gerard NP. Neutral endopeptidase-like enzyme controls the contractile activity of substance P in guinea pig lung. J Clin Invest. 1987;79:1819–25. doi: 10.1172/JCI113023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Yong T, Gao XP, Koizumi S, Conlon JM, Rennard SI, Mayhan WG, et al. Role of peptidase in bradykinin-induced increase in vascular permeability in vivo. Circ Res. 1992;70:952–9. doi: 10.1161/01.res.70.5.952. [DOI] [PubMed] [Google Scholar]
- [10].Kubota E, Dean RG, Hubner RA, Casley DJ, Johnston CI, Burrell LM. Differential tissue and enzyme inhibitory effects of the vasopeptidase inhibitor omapatrilat in the rat. Clin Sci. 2003;105:339–45. doi: 10.1042/CS20030097. [DOI] [PubMed] [Google Scholar]
- [11].Iwamoto I, Kimura A, Ochiai K, Tomioka H, Yoshida S. Distribution of neutral endopeptidase activity in human blood leukocytes. J Leukocyte Biol. 1991;49:116–25. doi: 10.1002/jlb.49.2.116. [DOI] [PubMed] [Google Scholar]
- [12].Mak IT, Komarov AM, Wagner TL, Stafford RE, Dickens BF, Weglicki WB. Enhanced NO production during Mg-deficiency and its role in mediating red cell glutathione loss. Am J Physiol. 1996;271:C385–C390. doi: 10.1152/ajpcell.1996.271.1.C385. [DOI] [PubMed] [Google Scholar]
- [13].Miners JS, Van Helmond Z, Chalmers K, Wilcock G, Love S, Kehoe PG. Decreased expression and activity of neprilysin in Alzheimer disease are associated with cerebral amyloid angiopathy. J Neuropathol Exp Neurol. 2006;65:1012–21. doi: 10.1097/01.jnen.0000240463.87886.9a. [DOI] [PubMed] [Google Scholar]
- [14].Mak IT, Weglicki WB. Antioxidant activity of calcium channel blocking drugs. Methods Enzymol. 1994;234(Part D):620–30. doi: 10.1016/0076-6879(94)34133-8. [DOI] [PubMed] [Google Scholar]
- [15].Mak IT, Stafford RE, Weglicki WB. Loss of red cell glutathione during Mg-deficiency: prevention by vitamin E, D-propranolol and chloroquine. Am J Physiol. 1994;267:C1366–C1370. doi: 10.1152/ajpcell.1994.267.5.C1366. [DOI] [PubMed] [Google Scholar]
- [16].Bussiere FI, Zimowska W, Gueux E, Rayssiguier Y, Mazur A. Stress protein expression cDNA array study supports activation of neutrophils during acute magnesium deficiency in rats. Magnes Res. 2002;15:37–42. [PubMed] [Google Scholar]
- [17].Mazur A, Maier JA, Rock E, Gueux E, Nowacki W, Rayssiguier Y. Magnesium and the inflammatory response: potential physiopathogical implications. Arch Biochem Biophys. 2007;458:48–56. doi: 10.1016/j.abb.2006.03.031. [DOI] [PubMed] [Google Scholar]
- [18].Kurantsin-Mills J, Cassidy MM, Stafford RE, Weglicki WB. Marked Alterations in circulating inflammatory cells during cardiomyopathy development in a magnesium- deficient rat model. Br J Nutr. 1997;78:845–55. doi: 10.1079/bjn19970200. [DOI] [PubMed] [Google Scholar]
- [19].Altura BM, Altura BT. New perspective on the role of Mg in the pathophysiology of the cardiovascular system. Magnesium. 1985;4:226–44. [PubMed] [Google Scholar]
- [20].Mayer ML, Westbrook GL, Guthrie PB. Votalge-dependent block by Mg2+ of NMDA responses in spinal cord neurons. Nature. 1984;309:261–3. doi: 10.1038/309261a0. [DOI] [PubMed] [Google Scholar]
- [21].Liu H, Mantyh PW, Basbaum AI. NMDA-receptor regulation of substance P release from primary afferent nociceptors. Nature. 1997;386:721–4. doi: 10.1038/386721a0. [DOI] [PubMed] [Google Scholar]
- [22].Weglicki WB, Mak IT, Kramer JH, Dickens BF, Cassidy MM, Stafford RE, et al. Role of free radicals and substance P in magnesium deficiency. Cardiovasc Res. 1996;31:677–82. [PubMed] [Google Scholar]
- [23].Kramer JH, Mak IT, Dickens BF, Weglicki WB. Modulation of circulating substance P in Mg-deficient (MgD) rats by NMDA receptor and neutral endopeptidase inhibitors. FASEB J. 2007;21:A1066. (Abs.) [Google Scholar]
- [24].Shipp MA, Stefano GB, Switzer SN, Griffin JD, Reinherz EL. CD10 (CALLA)/neutral endopeptidase 24.11 modulates inflammatory peptide-induced changes in neutrophil morphology, migration and adhesion proteins and is itself regulated by neutrophil activation. Blood. 1991;78:1834–41. [PubMed] [Google Scholar]
- [25].Shipp MA, Stefano GB, D’Adamio L, Switzer SN, Howard FD, Sinisterra J, et al. Downregulation of enkephalin-mediated inflammatory responses by CD10/neutral endopeptidase 24.11. Nature. 1990;347:394–400. doi: 10.1038/347394a0. [DOI] [PubMed] [Google Scholar]
- [26].Kirkwood KS, Bunnett NW, Maa J, Castagliolo I, Liu B, Gerard N, et al. Deletion of neutral endopeptidase exacerbates intestinal inflammation induced by Clostridium difficile toxin A. Am J Physiol Gastrointest Liver Physiol. 2001;281:G544–51. doi: 10.1152/ajpgi.2001.281.2.G544. [DOI] [PubMed] [Google Scholar]
- [27].Sturiale S, Barbar G, Qiu B, Figini M, Geppetti P, Gerard N, et al. Neutral endopeptidase (EC 3.4.24.11) terminates colitis by degrading substance P. Proc Nat Acad Sci. 1999;96:11653–8. doi: 10.1073/pnas.96.20.11653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Barbara G, De Giorgio R, Stanghellini V, Corinaldesi R, Cremon C, Gerard N, et al. Neutral endopeptidase downregulates the onset of intestinal inflammation in the nematode infected mouse. Gut. 2003;52:1457–64. doi: 10.1136/gut.52.10.1457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Chmielinska JJ, Mak IT, Kramer JH, Viswalingam K, Weglicki WB. Decreased neutral endopeptidase activity in cardiac neurogenic inflammation due to Mg-deficiency. J Mol Cell Cardiol. 2007;42:S114. (Abs.) [Google Scholar]



