Abstract
Background
The interaction between the gastrointestinal tract and the renal excretion of sodium and water is poorly understood. The observation that beneficial effects on blood pressure are seen in bariatric surgery patients before significant weight loss, led us to hypothesise that the Roux-en-Y gastric bypass (gastric bypass) would alter renal salt and water handling.
Material and Methods
21 male wistar rats (Body weight (BW) 348±19g) underwent either gastric bypass (n=14) or sham operation (n=7). Animals were kept on a low sodium diet (DO2051701, Research Diets, Inc., New Brunswick, USA) and deionized water ad libitum. Before and after surgery rats received oral hypertonic sodium solution (1.5 mmol/kg BW) and were placed individually in metabolic cages to measure urine production and water intake for 8 h. Urine sodium concentration was measured by Integrated Chip Technology (ICT) using the Architect ci16200 (Abbott, Illinois, USA).
Results
Rats that underwent gastric bypass had a significantly lower body weight than sham-operated controls over the entire follow-up period. (387.5 ± 18.3g vs. 501.3 ± 8.0g, p=0.0004). An oral sodium load following gastric bypass operation lead to increase in water intake (0.065±0.012 ml/g BW vs. 0.033±0.006 ml/g BW, p= 0.023), urine output (0.034±0.007 ml/g BW vs. 0.015±0.002 ml/g BW, p= 0.027) and natriuresis (65.99±10.7 mol vs. 31.71±8.7 mol, p=0.020). These differences were greater in bypass operated animals that lost weight (“Responders”) compared to those that did not (“Non-Responders”). There was no change in water intake, urine production or sodium excretion after surgery in the sham-operated group.
Conclusions
Urine production, water intake and sodium excretion are increased after gastric bypass in rats following oral salt loading. The magnitude of this effect correlates with the amount of weight loss after surgery. These observations may explain part of the mechanism underlying the effects of bariatric surgery on blood pressure.
Keywords: Gastro-intestinal tract, hypertension, sodium, water
Background
Gastric bypass surgery is currently the most effective treatment for obesity and the beneficial effects on obesity-related comorbidities such as diabetes and hypertension are well documented [1]. The research focus in this field to date has been on the effects of bariatric surgery on gut hormone release and the improvement in glucose homeostasis as a result [2,3]. Less attention has been given to the mechanisms underlying the resolution of hypertension after bariatric surgery. In the Swedish obese subjects (SOS) study, systolic blood pressure decreased by approximately 11 mmHg and diastolic by approximately 7 mmHg in the first 6 months after bariatric surgery [4,5], although this effect was not sustained in the longer term. Several other studies have shown similar observations [1,6-8]. The improvement in blood pressure seen after bypass surgery was initially thought to be a medium-term effect, with the first documented reductions seen at eight weeks. However, Ahmed et al. observed significant reductions in systolic (9 mmHg) and diastolic (7 mmHg) blood pressures as early as one week after gastric bypass surgery [9]. Furthermore, the beneficial effect was maintained for at least one year, and the postoperative usage of antihypertensive medication was reduced by a third [9].
Hypertension is associated with central adiposity and insulin resistance [10,11], but the pathophysiological mechanism remains unclear. There are several plausible hypotheses including insulin resistance [12,13], aldosterone and aldosterone releasing factors (ARF) [14,15], as well as hyperleptinemia [16,17]. The reduction of visceral fat mass and subsequent decrease of sympathetic activation and sodium retention is not immediate and does not explain the early reductions in blood pressure after RYGB described by Ahmed at al [9]. Thus, we hypothesized that salt and water handling of the kidney might be altered after bypass surgery and this modification potentially contributes to the early resolution of hypertension after gastric bypass.
Our aim was to evaluate water intake, urine production and urinary sodium excretion in rats before and shortly after gastric bypass surgery.
Methods
Animals
21 male wistar rats (BW 348±19g) were randomized to have either a gastric bypass (n=14) or sham operation (n=7). The work was performed under UK Home Office licence (PL 70-5569). All animals were kept in identical environmental conditions (temperature 24°C, humidity 60%, light cycle 7.00 – 19.00) with normal chow (RM1 diet, Special Diet Services Ltd, UK) and tap water ad libitum unless otherwise stated. Body weight was measured daily.
Metabolic cage experiments
Metabolic cage experiments were performed one week preoperatively and on postoperative day 30 and 60. Prior to each experiment animals were on low sodium diet and deionized water ad libitum for one week to establish a constant urinary sodium excretion for all animals [18]. The low sodium diet was identical to normal chow except for its salt content (D02051701, Research Diets Inc., New Brunswick, NJ, USA; sodium content 102.6 ppm). For measurements before surgery and on postoperative day 30, salt (1.5 mmol Na/kg BW) was given intragastrically via an oral gavage over 10s in the form of an hypertonic NaCl solution (616 mM) at the beginning of the light phase (7.00 am). Animals were then placed individually into a metabolic cage designed for urine collection and measurement of water intake for eight hours. For baseline measurements on postoperative day 60, animals were placed into the metabolic cage without an oral sodium load. In all experiments urine was collected in pre-weighed plastic tubes. Water was given in pre-weighed plastic bottles which were also re-weighed at the end of the experiment. The cages were cleaned and rinsed with deionized water after each experiment.
Surgery
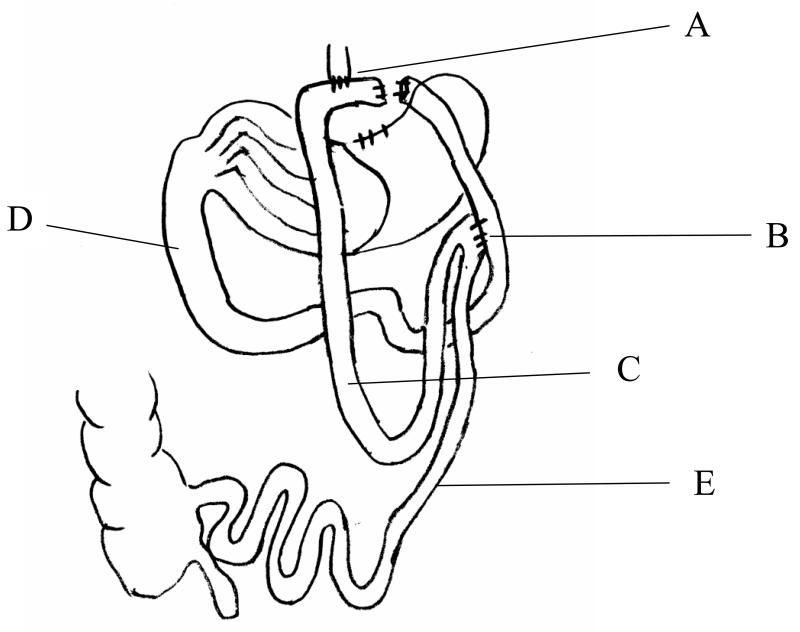
Surgery was performed according to an established and standardized protocol under isoflurane anaesthesia [2]. All operations were carried out by one investigator (M.B.). The rats were fasted overnight, but had access to tap water ad libitum. Briefly, the stomach was transected close to the gastro-oesophageal junction, which was subsequently anastomosed to a loop of jejunum 7 cm distal to the ligament of Treitz in an end-to side fashion. A 7 mm side-to-side small bowel anastomosis was performed between the biliopancreatic and the alimentary limbs to create a common channel of 25 cm, and the omega loop of small bowel was then divided. Figure 1 shows a diagrammatic representation of the RYGB rodent model. The sham operation consisted of a laparotomy, a 7mm gastrotomy on the anterior wall of the stomach with subsequent closure, and a 7 mm jejunotomy with subsequent closure. Preoperatively, gentamicin 8 mg/kg and carprofen 0.01 mL were administered intraperitoneally (ip) as prophylaxis for postoperative pain and infection.
Figure 1.
Diagrammatic representation of the RYGB rodent model. A= Gastro-Jejunostomy, B=Jejuno-Jejunostomy, C=Alimentary limb, D=Biliopancreative Limb (7 cm), E= Common channel (25 cm).
Measurement of urinary sodium
Urine sodium concentration was measured by Integrated Chip Technology (ICT) using the Architect ci16200 (Abbott, Illinois, USA). It obtains millivolt readings, and then converts them to assay-specific analyte conversion units. The measurement of ICT reference solution and ICT samples are used to calculate the assay results.
Statistical analysis
Data is presented as mean ± SEM unless otherwise indicated. Data were compared with the use of 2-tailed, paired Student t tests (Graphpad Prism, USA). P<0.05 was considered significant.
Results
Weight loss
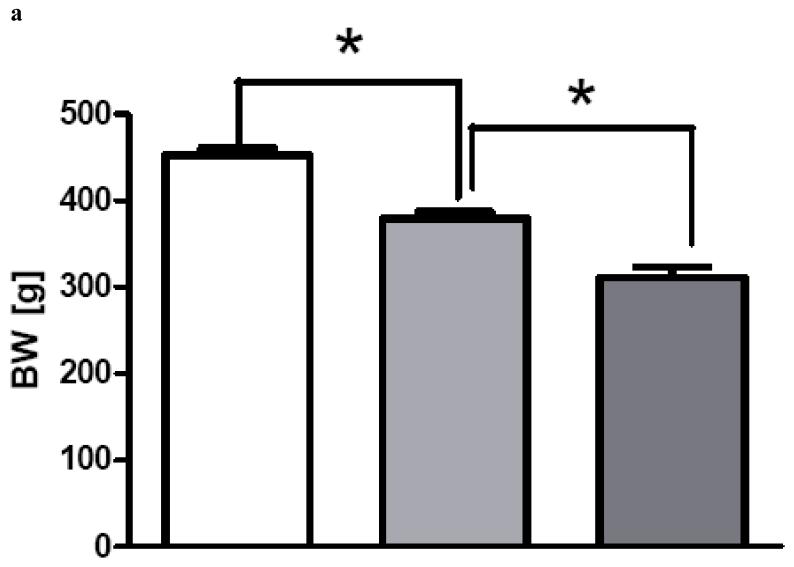
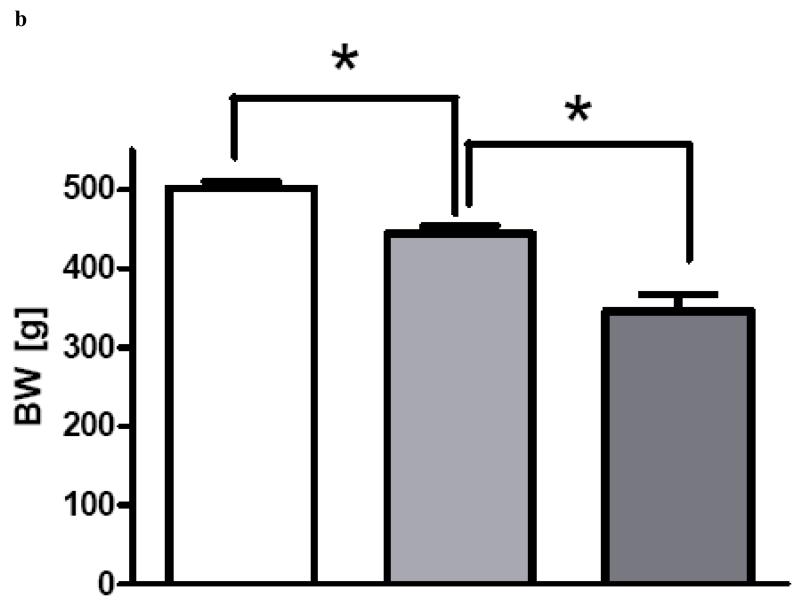
Body weight (BW) was significantly lower in the group of rats that underwent gastric bypass compared with the sham-operated group (Day 60: 388 ± 18g vs. 501 ± 8g, p=0.0004). Figure 2 shows the percentage of initial BW for all bypass (n=14) and sham operated rats (n=7). However, it was clear that not all rats lost weight after the bypass procedure. Those animals that lost weight compared to baseline were defined as ‘responders’ (n=8) while all others were defined as ‘non-responders’ (n=6). The mean BW of these two groups were significantly different from day 8 for the rest of the study (Day 8: 316.6±5.3g vs. 336.5±4.5g, p=0.02). Representative data for postoperative day 30 (311 ± 12g vs. 379 ± 8g, p=0.0011) and day 60 (346 ± 21g vs. 443 ± 10g, p=0.0029) are shown in Figure 3. However, after 60 days the “non-responders” still weighed significantly less than the sham-operated animals (443.3 ± 10.3g vs. 501.3 ± 8g, p=0.0009).
Figure 2.
Percentage weight change for the RYGB (-●-) (n=14) and sham-operated rats (-■-) (n=7). Data are shown as mean values ± SEM. p<0.05 was considered significant (*).
Figure 3.
a and b: Body weight (BW) [g] after sham operation (white columns, n=7), ‘Non-Responders’ (light grey, n=8) and ‘Responders’ (dark grey, n=6) after bypass operations on postoperative day 30 (a) and 60 (b). Data are shown as mean values ± SEM. p<0.05 was considered significant (*).
Urine production
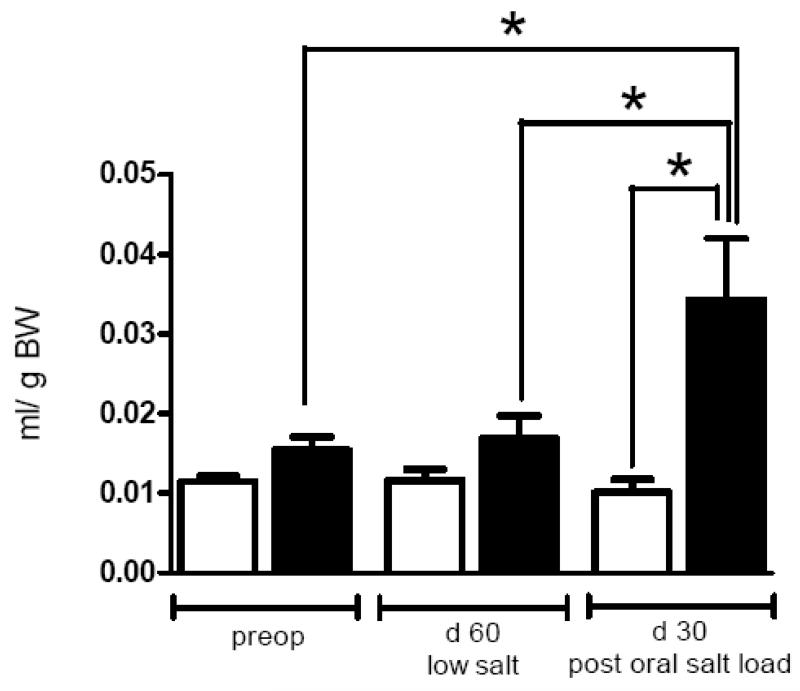
In gastric bypass rats, oral salt loading on postoperative day 30 led to a greater increase in urine output when compared to urine production following oral salt loading prior to surgery (0.034 ± 0.007 ml/g BW vs. 0.015 ± 0.002 ml/g BW, p= 0.027). There was no post-operative change in urine production seen in the sham-operated group (0.010 ± 0.002 ml/g BW vs. 0.011 ± 0.001 ml/g BW, p= 0.44). Furthermore, bypass rats produced significantly more urine than sham-operated rats after oral salt loading (0.034 ± 0.007 ml/g BW vs. 0.010 ± 0.002, p=0.038). There was no difference between urine production on day 60 without oral salt loading and preoperative data (Bypass: 0.017 ± 0.003 ml/g BW vs. 0.015 ± 0.002 ml/g BW, p= 0.66 and Sham: 0.012 ± 0.001 ml/g BW vs. 0.011 ± 0.001 ml/g BW, p= 0.93). Figure 4 summarizes data for urine output. There appeared to be a relationship between BW loss and urine production as “Responders” produced significantly more urine than “Non-Responders” (0.051 ± 0.009 ml/g BW vs. 0.014 ± 0.003, p= 0.006).
Figure 4.
Urine production of bypass operated (black columns, n=14) and Sham operated rats (white columns, n=7) after oral salt loading (1.5 mmol Na/kg BW of a 616 mM NaCl solution) preoperatively and on postoperative day 30. On day 60 urine production was measured without oral salt loading. Data are shown as mean values ± SEM. p<0.05 was considered significant (*).
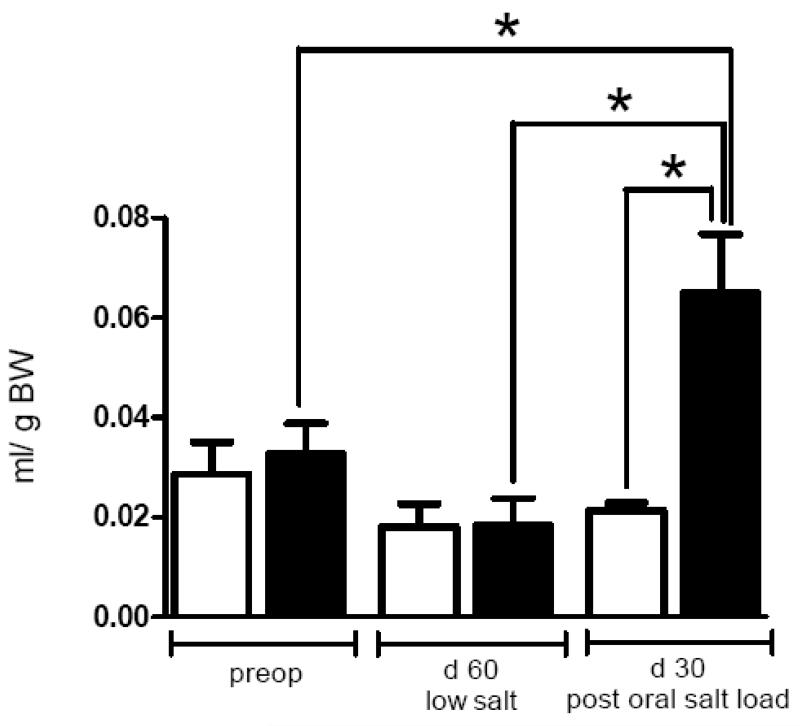
Water intake
Data for water consumption are summarized in Figure 5. Gastric bypass rats consumed significantly more water after the oral salt challenge (1.5 mmol Na/kg BW of a 616 mM NaCl solution) when compared with their water consumption after oral salt loading before surgery (0.065 ± 0.012 ml/g BW vs. 0.033 ± 0.006 ml/g BW, p= 0.023). No changes were observed for water intake in the sham operated animals (0.029 ± 0.006 ml/g BW vs 0.021 ± 0.002 ml/g BW, p=0.30). Moreover, bypass rats drank significantly more water than sham-operated rats (0.065 ± 0.012 ml/g BW vs. 0.02137 ± 0.002 ml/g BW, p= 0.019) after oral salt loading. There was no difference between water intake on day 60 without oral salt challenge and preoperative data (Bypass: 0.018 ± 0.005 ml/g BW vs. 0.033 ± 0.006 ml/g BW, p=0.10 and Sham: 0.018 ± 0.005 ml/g BW vs. 0.029 ± 0.006 ml/g BW, p=0.21). Water intake after bypass surgery showed a positive correlation to the level of BW loss as “Responders” displayed significantly greater water intake than “Non-Responders” (0.090 ± 0.015 ml/g BW vs. 0.031 ± 0.006 ml/g BW, p=0.007).
Figure 5.
Water intake of bypass operated (black columns, n=14) and Sham operated rats (white columns, n=7) after oral salt loading (1.5 mmol Na/kg BW of a 616 mM NaCl solution) preoperatively and on postoperative day 30. On day 60 water intake was measured without oral salt loading. Data are shown as mean values ± SEM. p<0.05 was considered significant (*).
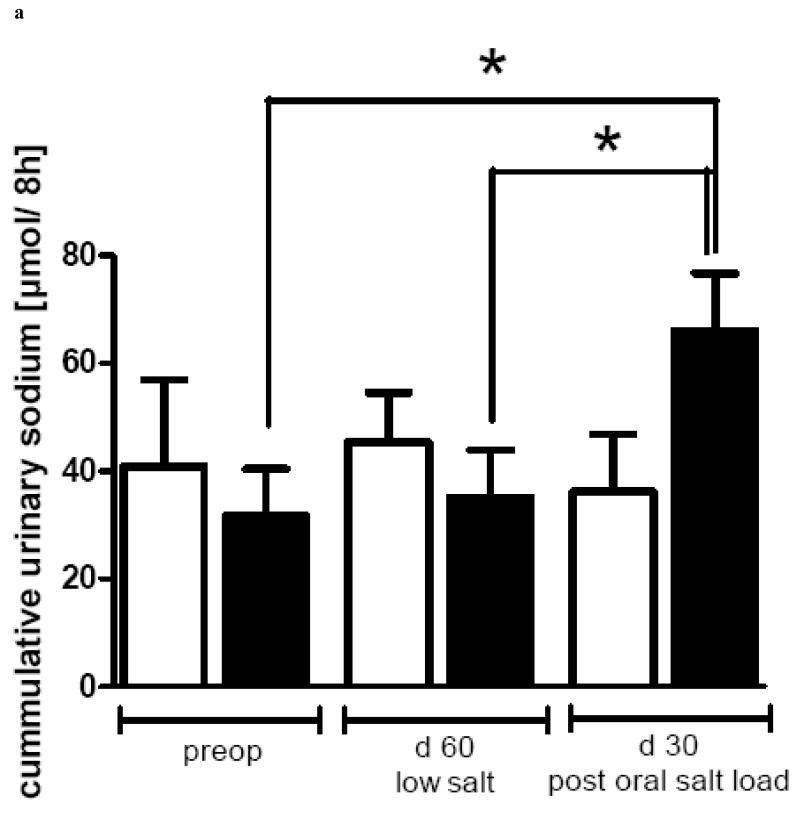
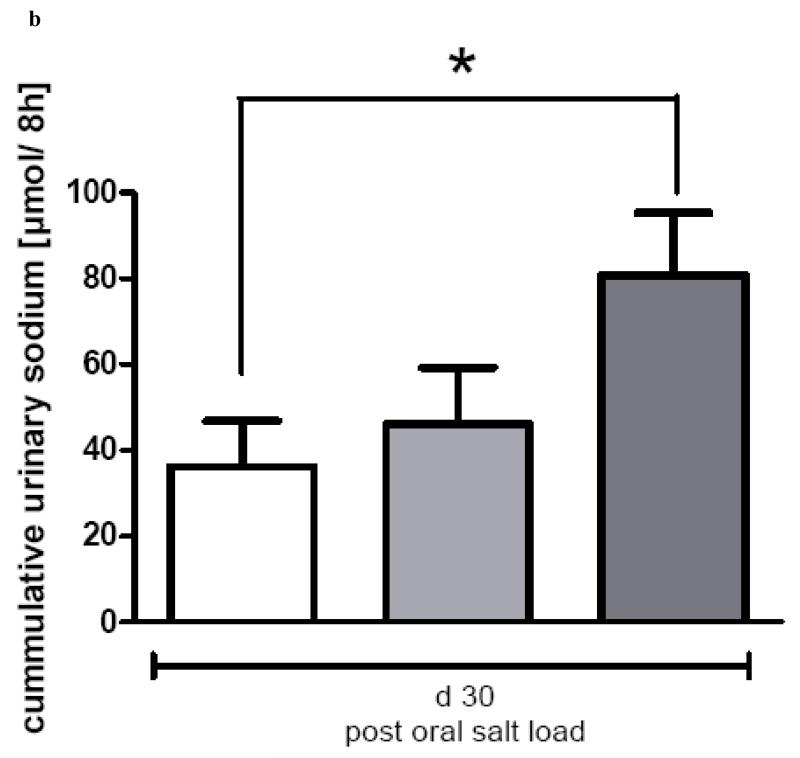
Sodium excretion
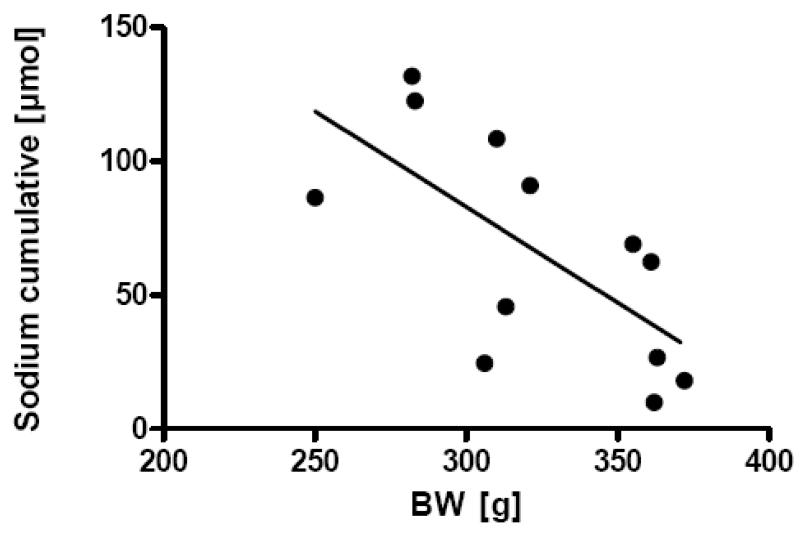
Data for sodium excretion are summarized in Figure 6 and 7. Oral salt loading on postoperative day 30 (1.5 mmol Na/kg BW of a 616 mM NaCl solution) led to an increase in cumulative sodium excretion after 8 hours in the gastric bypass rats compared to preoperatively (65.9 ± 10.7 μmol vs. 31.7 ± 8.7 μmol, p= 0.02). No changes in natriuresis after 8 hours were observed for the sham operated animals (36.2 ± 10.7 μmol vs. 40.9 ± 16.0 μmol, p= 0.81). There was a trend towards increased sodium excretion in bypass rats compared to their sham-operated counterparts, but this did not attain statistical significance (65.9 ± 10.7 μmol vs. 36.2 ± 10.7 μmol, p= 0.09). However, differences became significant when bypass rats were divided into “Responders” and “Non-Responders” (80.9 ± 14.4 μmol vs. 36.2 ± 10.7 μmol, p=0.03) and cumulative sodium excretion after bypass surgery showed a correlation to postoperative BW (r2= 0.45 and p= 0.017, Figure 7). There was no significant difference between preoperative sodium excretion and sodium excretion on day 60 (Bypass: 35.1 ± 8.8 μmol vs. 31.7 ± 8.7 μmol, p= 0.79 and Sham: 45.4 ± 9.2 μmol vs. 40.9 ± 16.0 μmol, p= 0.81).
Figure 6.
a: Cumulative Sodium excretion of bypass operated (black columns, n=14) and Sham operated rats (white columns, n=7) after oral salt loading (1.5 mmol Na/kg BW of a 616 mM NaCl solution) preoperatively and on postoperative day 30. On day 60 sodium excretion was measured without oral salt loading. Data are shown as mean values ± SEM. p<0.05 was considered significant (*).
b: Cumulative Sodium excretion of “Responders” (dark grey columns, n=8) and “Non-Responders” (light grey columns, n=6) and Sham operated rats (white columns, n=7) after oral salt loading (1.5 mmol Na/kg BW of a 616 mM NaCl solution) on postoperative day 30. Data are shown as mean values ± SEM. p<0.05 was considered significant (*).
Figure 7.
Linear regression analysis of cumulative sodium excretion against percentage weight change in RYGB rats after oral salt loading (1.5 mmol Na/kg BW of a 616 mM NaCl solution) on postoperative day 30 (r2=0.57, p=0.0067).
Discussion
Numerous central and peripheral abnormalities account for the development and maintenance of high arterial pressure in obesity [19]. To date, visceral obesity is considered the most important risk factor for hypertension and cardiovascular disease [20]. Visceral obesity is linked to hyperinsulinemia, hyperleptinemia and increased levels of aldosterone and aldosterone releasing factors (ARF) all of which lead to activation of the sympathetic nervous system and the renin-angiotensin-aldosterone system [21-23].
Gastric bypass surgery is currently the most effective treatment for obesity and the beneficial effects on obesity-related comorbidities such as hypertension are well documented [24,25]. However, the reduction of visceral fat mass and subsequent decrease of sympathetic activation and sodium retention is not immediate and does not explain the very early reduction in blood pressure after RYGB described by Ahmed at al [9]. Thus, we reasoned that other mechanisms might be involved in the early resolution of hypertension after gastric bypass and that alteration of renal salt and water handling might be one of them.
We have demonstrated a significant increase in urine production, water intake and sodium excretion after RYGB compared with pre-operative measurements. Sham operated animals show no changes in water intake, urine production or sodium excretion after surgery. Furthermore, we showed that those animals who lost weight after RYGB surgery (“Responders”) demonstrated a greater urine production, water intake and natiuresis compared with animals that did not lose weight after RYGB (“Non-Responders”), suggesting a correlation between weight loss and renal sodium handling.
In many groups of patients, hypertension is related to sodium imbalance [26]. Only a few studies have focused on the possible role of the gastrointestinal tract in the control of sodium homeostasis and arterial hypertension. The idea that dietary intake and composition affects renal function is perhaps self-evident, but an exact definition of this relationship is still lacking. Several physiological mechanisms are involved in controlling sodium balance such as the hormones aldosterone, angiotensin [27] and atrial natriuretic peptide [28]. In addition, there is evidence to suggest the involvement of the gastrointestinal tract. Analogous to the “incretin effect”, characterized by an exaggerated plasma insulin response to an oral glucose load compared with the same amount of glucose administered intravenously, oral ingestion of sodium chloride has a greater natriuretic effect than when given intravenously in subjects on a low sodium diet [29]. This effect has been shown to be independent of changes in aldosterone and atrial natriuretic peptide [29]. In the case of insulin release, the important incretin gut hormone has been shown to be GLP-1, which has since been developed into a successful treatment for type 2 diabetes [30]. The mechanism for the analogous effect on sodium excretion, and therefore potentially, blood pressure control, has yet to be identified.
Animal studies provide some evidence that the gastrointestinal tract may exert direct influence on renal function. Morgan et al observed that salt-sensitive Harlan Sprague Dawley (HSD) rats (SS/Jr) with transplanted kidneys from salt-resistant HSD rats (SR/Jr) developed a significant NaCl-induced hypertension, suggesting that extrarenal factors contribute to the salt-induced hypertension in salt sensitive rats [31]. These observations were not accounted for by any changes in established hormones known to control renal sodium excretion including aldosterone, renin and angiotensin [27] or atrial natriuretic peptide [28]. Hence, the presence of an intestinal natriuretic factor influencing electrolyte transport in renal tubular cells and causing diuresis was suggested [31]. Studies on other animal models for essential hypertension, such as Spontaneously Hypertensive Rats (SHR) and their genetically matched normotensive controls, Wistar-Kyoto (WKY) rats, have shown that WKY rats have greater difficulties than SHR in conserving sodium both via the gastrointestinal tract and via the kidneys [32,33].
A well-recognized problem associated with gastric bypass is weight re-gain after the operation in some patients [34,35]. This clinical observation is reflected by the fact that not all rats exhibited sustained weight loss following the bypass procedure. Our model closely reproduces the RYGB operation in humans. Thus, we defined animals that lost weight as ‘responders’ and all others as ‘non-responders’ [36]. As all operations were carried out by a single surgeon according to a standardized protocol [2], technical variations or inconsistencies are unlikely to account for these postoperative variations in body weight. We previously postulated that poor weight loss or poor weight loss maintenance post RYGB might be correlated with lower levels of PYY and GLP-1 compared with patients with good sustained weight loss [35]. In this study, reduced urine output, less water intake and decreased sodium excretion were also correlated with lower levels of weight loss after RYGB. This correlation points to the altered salt and water homeostasis seen after bypass surgery being modulated by changes in circulating gut hormone concentrations. Potential mediators include Peptide YY (PYY) and glucagon-like peptide (GLP)-1. It is reasonable to speculate that GLP-1 and PYY may provide the missing link between the gastrointestinal tract and the kidney, as effects on renal salt and water handling have been demonstrated for both [37-39].
In conclusion, gastric bypass provides us with a valuable model to facilitate our understanding of the role that the gastrointestinal tract plays in sodium homeostasis and the development of salt-dependent hypertension. RYGB results in a greater urine output, water intake and sodium excretion in salt-restricted rats following an oral salt challenge This observation may provide insight into the mechanism of the early improvement in arterial hypertension seen after gastric bypass.
Acknowledgments
The financial support by the Deutsche Forschungsgemeinschaft is gratefully acknowledged (M.B.).
References
- 1.Buchwald H, Avidor Y, Braunwald E, et al. Bariatric surgery: a systematic review and meta-analysis. JAMA. 2004;292:1724–37. doi: 10.1001/jama.292.14.1724. [DOI] [PubMed] [Google Scholar]
- 2.le Roux CW, Aylwin SJ, Batterham RL, et al. Gut hormone profiles following bariatric surgery favor an anorectic state, facilitate weight loss, and improve metabolic parameters. Ann Surg. 2006;243:108–14. doi: 10.1097/01.sla.0000183349.16877.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rubino F, Gagner M, Gentileschi P, et al. The early effect of the Roux-en-Y gastric bypass on hormones involved in body weight regulation and glucose metabolism. Ann Surg. 2004;240:236–42. doi: 10.1097/01.sla.0000133117.12646.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sjostrom L, Lindroos AK, Peltonen M, et al. Lifestyle, diabetes, and cardiovascular risk factors 10 years after bariatric surgery. N Engl J Med. 2004;351:2683–93. doi: 10.1056/NEJMoa035622. [DOI] [PubMed] [Google Scholar]
- 5.Sjostrom L, Narbro K, Sjostrom CD, et al. Effects of bariatric surgery on mortality in Swedish obese subjects. N Engl J Med. 2007;357:741–52. doi: 10.1056/NEJMoa066254. [DOI] [PubMed] [Google Scholar]
- 6.Carson JL, Ruddy ME, Duff AE, et al. The effect of gastric bypass surgery on hypertension in morbidly obese patients. Arch Intern Med. 1994;154:193–200. [PubMed] [Google Scholar]
- 7.Fernstrom JD, Courcoulas AP, Houck PR, et al. Long-term changes in blood pressure in extremely obese patients who have undergone bariatric surgery. Arch Surg. 2006;141:276–83. doi: 10.1001/archsurg.141.3.276. [DOI] [PubMed] [Google Scholar]
- 8.Sugerman HJ, Wolfe LG, Sica DA, et al. Diabetes and hypertension in severe obesity and effects of gastric bypass-induced weight loss. Ann Surg. 2003;237:751–6. doi: 10.1097/01.SLA.0000071560.76194.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ahmed AR, Rickards G, Coniglio D, et al. Laparoscopic Roux-en-Y Gastric Bypass and Its Early Effect on Blood Pressure. Obes Surg. 2008 doi: 10.1007/s11695-008-9671-z. [DOI] [PubMed] [Google Scholar]
- 10.Kannel WB, Brand N, Skinner JJ, Jr., et al. The relation of adiposity to blood pressure and development of hypertension. The Framingham study. Ann Intern Med. 1967;67:48–59. doi: 10.7326/0003-4819-67-1-48. [DOI] [PubMed] [Google Scholar]
- 11.Stamler R, Stamler J, Riedlinger WF, et al. Weight and blood pressure. Findings in hypertension screening of 1 million Americans. JAMA. 1978;240:1607–10. doi: 10.1001/jama.240.15.1607. [DOI] [PubMed] [Google Scholar]
- 12.DeFronzo RA, Cooke CR, Andres R, et al. The effect of insulin on renal handling of sodium, potassium, calcium, and phosphate in man. J Clin Invest. 1975;55:845–55. doi: 10.1172/JCI107996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Natali A, Quinones GA, Santoro D, et al. Relationship between insulin release, antinatriuresis and hypokalaemia after glucose ingestion in normal and hypertensive man. Clin Sci (Lond) 1993;85:327–35. doi: 10.1042/cs0850327. [DOI] [PubMed] [Google Scholar]
- 14.Connell JM, Davies E. The new biology of aldosterone. J Endocrinol. 2005;186:1–20. doi: 10.1677/joe.1.06017. [DOI] [PubMed] [Google Scholar]
- 15.Laragh J. Laragh’s lessons in pathophysiology and clinical pearls for treating hypertension. Am J Hypertens. 2001;14:397–404. doi: 10.1016/s0895-7061(01)02049-0. [DOI] [PubMed] [Google Scholar]
- 16.Galletti F, D’Elia L, Barba G, et al. High-circulating leptin levels are associated with greater risk of hypertension in men independently of body mass and insulin resistance: results of an eight-year follow-up study. J Clin Endocrinol Metab. 2008;93:3922–6. doi: 10.1210/jc.2008-1280. [DOI] [PubMed] [Google Scholar]
- 17.Rahmouni K, Correia ML, Haynes WG, et al. Obesity-associated hypertension: new insights into mechanisms. Hypertension. 2005;45:9–14. doi: 10.1161/01.HYP.0000151325.83008.b4. [DOI] [PubMed] [Google Scholar]
- 18.Mu JY, Hansson GC, Lundgren O. The intestinal tract and the pathophysiology of arterial hypertension: an experimental study on Dahl rats. Acta Physiol Scand. 1995;155:137–46. doi: 10.1111/j.1748-1716.1995.tb09958.x. [DOI] [PubMed] [Google Scholar]
- 19.Hall JE. The kidney, hypertension, and obesity. Hypertension. 2003;41:625–33. doi: 10.1161/01.HYP.0000052314.95497.78. [DOI] [PubMed] [Google Scholar]
- 20.Sironi AM, Gastaldelli A, Mari A, et al. Visceral fat in hypertension: influence on insulin resistance and beta-cell function. Hypertension. 2004;44:127–33. doi: 10.1161/01.HYP.0000137982.10191.0a. [DOI] [PubMed] [Google Scholar]
- 21.Rahmouni K, Morgan DA, Morgan GM, et al. Hypothalamic PI3K and MAPK differentially mediate regional sympathetic activation to insulin. J Clin Invest. 2004;114:652–8. doi: 10.1172/JCI21737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rahmouni K, Haynes WG, Morgan DA, et al. Role of melanocortin-4 receptors in mediating renal sympathoactivation to leptin and insulin. J Neurosci. 2003;23:5998–6004. doi: 10.1523/JNEUROSCI.23-14-05998.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.El-Atat FA, Stas SN, McFarlane SI, et al. The relationship between hyperinsulinemia, hypertension and progressive renal disease. J Am Soc Nephrol. 2004;15:2816–27. doi: 10.1097/01.ASN.0000133698.80390.37. [DOI] [PubMed] [Google Scholar]
- 24.Adams TD, Gress RE, Smith SC, et al. Long-term mortality after gastric bypass surgery. N Engl J Med. 2007;357:753–61. doi: 10.1056/NEJMoa066603. [DOI] [PubMed] [Google Scholar]
- 25.Buchwald H. Bariatric surgery for morbid obesity: health implications for patients, health professionals, and third-party payers. J Am Coll Surg. 2005;200:593–604. doi: 10.1016/j.jamcollsurg.2004.10.039. [DOI] [PubMed] [Google Scholar]
- 26.Postnov YV. An approach to the explanation of cell membrane alteration in primary hypertension. Hypertension. 1990;15:332–7. doi: 10.1161/01.hyp.15.3.332. [DOI] [PubMed] [Google Scholar]
- 27.Bouhnik J, Richoux JP, Huang H, et al. Hypertension in Dahl salt-sensitive rats: biochemical and immunohistochemical studies. Clin Sci (Lond) 1992;83:13–22. doi: 10.1042/cs0830013. [DOI] [PubMed] [Google Scholar]
- 28.Sterzel RB, Luft FC, Gao Y, et al. Atrial natriuretic factor in sodium-sensitive and sodium-resistant Dahl rats. J Hypertens. 1987;5:17–24. doi: 10.1097/00004872-198702000-00003. [DOI] [PubMed] [Google Scholar]
- 29.Lennane RJ, Carey RM, Goodwin TJ, et al. A comparison of natriuresis after oral and intravenous sodium loading in sodium-depleted man: evidence for a gastrointestinal or portal monitor of sodium intake. Clin Sci Mol Med. 1975;49:437–40. doi: 10.1042/cs0490437. [DOI] [PubMed] [Google Scholar]
- 30.Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology. 2007;132:2131–57. doi: 10.1053/j.gastro.2007.03.054. [DOI] [PubMed] [Google Scholar]
- 31.Morgan DA, DiBona GF, Mark AL. Effects of interstrain renal transplantation on NaCl-induced hypertension in Dahl rats. Hypertension. 1990;15:436–42. doi: 10.1161/01.hyp.15.4.436. [DOI] [PubMed] [Google Scholar]
- 32.Lundin S, Hallback-Nordlander M, Gothberg G, et al. Hemodynamic consequences of early cardiovascular structural adaptation in primary hypertension as studied in rats. Contrib Nephrol. 1982;30:124–31. doi: 10.1159/000406430. [DOI] [PubMed] [Google Scholar]
- 33.Sellden H, Sjovall H, Ricksten SE. Sympathetic nerve activity and central haemodynamics during mechanical ventilation with positive end-expiratory pressure in rats. Acta Physiol Scand. 1986;127:51–60. doi: 10.1111/j.1748-1716.1986.tb07875.x. [DOI] [PubMed] [Google Scholar]
- 34.Christou NV, Look D, MacLean LD. Weight gain after short- and long-limb gastric bypass in patients followed for longer than 10 years. Ann Surg. 2006;244:734–40. doi: 10.1097/01.sla.0000217592.04061.d5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.le Roux CW, Welbourn R, Werling M, et al. Gut hormones as mediators of appetite and weight loss after Roux-en-Y gastric bypass. Ann Surg. 2007;246:780–5. doi: 10.1097/SLA.0b013e3180caa3e3. [DOI] [PubMed] [Google Scholar]
- 36.Guijarro A, Suzuki S, Chen C, et al. Characterization of weight loss and weight regain mechanisms after Roux-en-Y gastric bypass in rats. Am J Physiol Regul Integr Comp Physiol. 2007;293:R1474–R1489. doi: 10.1152/ajpregu.00171.2007. [DOI] [PubMed] [Google Scholar]
- 37.Gutzwiller JP, Tschopp S, Bock A, et al. Glucagon-like peptide 1 induces natriuresis in healthy subjects and in insulin-resistant obese men. J Clin Endocrinol Metab. 2004;89:3055–61. doi: 10.1210/jc.2003-031403. [DOI] [PubMed] [Google Scholar]
- 38.Gutzwiller JP, Hruz P, Huber AR, et al. Glucagon-like peptide-1 is involved in sodium and water homeostasis in humans. Digestion. 2006;73:142–50. doi: 10.1159/000094334. [DOI] [PubMed] [Google Scholar]
- 39.Michell AR, Debnam ES, Unwin RJ. Regulation of renal function by the gastrointestinal tract: potential role of gut-derived peptides and hormones. Annu Rev Physiol. 2008;70:379–403. doi: 10.1146/annurev.physiol.69.040705.141330. [DOI] [PubMed] [Google Scholar]