Abstract
Exendin-(9,39) is a competitive antagonist of glucagon-like peptide-1 (GLP-1) at its receptor. However, it is unclear if it has direct and unique effects of its own. We tested the hypothesis that exendin-(9,39) and GLP-1-(9,36)amide have direct effects on hormone secretion and β-cell function as well as glucose metabolism in healthy subjects. Glucose containing [3-3H]glucose was infused to mimic the systemic appearance of glucose after a meal. Saline, GLP-1-(9,36)amide, or exendin-(9,39) at 30 pmol/kg/min (Ex 30) or 300 pmol/kg/min (Ex 300) were infused in random order on separate days. Integrated glucose concentrations were slightly but significantly increased by exendin-(9,39) (365 ± 43 vs. 383 ± 35 vs. 492 ± 49 vs. 337 ± 50 mmol per 6 h, saline, Ex 30, Ex 300, and GLP-1-[9,36]amide, respectively; P = 0.05). Insulin secretion did not differ among groups. However, insulin action was lowered by exendin-(9,39) (25 ± 4 vs. 20 ± 4 vs. 18 ± 3 vs. 21 ± 4 10−4 dL/kg[min per μU/mL]; P = 0.02), resulting in a lower disposition index (DI) during exendin-(9,39) infusion (1,118 ± 118 vs. 816 ± 83 vs. 725 ± 127 vs. 955 ± 166 10−14 dL/kg/min2 per pmol/L; P = 0.003). Endogenous glucose production and glucose disappearance did not differ significantly among groups. We conclude that exendin-(9,39), but not GLP-1-(9,36)amide, decreases insulin action and DI in healthy humans.
The incretin hormone glucagon-like peptide-1 (GLP-1) arises by posttranslational processing of preproglucagon in the enteroendocrine L cells distributed throughout the intestine. GLP-1 secretion occurs within minutes of food ingestion, is a potent insulin secretagogue, and suppresses glucagon (1). However, the active form(s) of GLP-1 are rapidly deactivated by a serine protease dipeptidyl peptidase-4, which cleaves the two NH2-terminal amino acids necessary for activation of the GLP-1 receptor (GLP-1R). This enzyme is widely distributed so that the half-life of active GLP-1 in the circulation is ∼1 min (2). The resulting metabolite GLP-1-(9,36) has been proposed as a potential antagonist of GLP-1R, although at present there is no evidence of an effect of this peptide on insulin secretion (3).
Exendin-(7,39) is a naturally occurring analog of GLP-1-(7,36) and is an agonist of the GLP-1R. This compound binds to GLP-1R with greater affinity than the natural ligand due to a nine–amino acid COOH-terminal sequence absent in native GLP-1 (4). On the other hand, exendin-(9,39), which arises from the removal of the two NH2-terminal amino acids, is a competitive antagonist of GLP-1 at the GLP-1R (5). It has been used to examine the effects of endogenous GLP-1 secretion on glucose homeostasis (6). Although it is presumed that exendin-(9,39) has no direct effects on glucose metabolism, it alters gastric emptying and capacitance through vagal mechanisms, thereby altering glucose tolerance independent of its ability to inhibit GLP-1-(7,36) effects on insulin and glucagon secretion (7,8). A direct effect of GLP-1-(9,36) signaling on glucose metabolism has been reported (9).
The present studies were undertaken to determine whether exendin-(9,39) and GLP-1-(9,36)amide have direct effects on β-cell function, insulin action, glucagon secretion, and glucose metabolism. We did so by infusing glucose in a manner that mimicked the systemic appearance of glucose after ingestion of carbohydrate. Since glucose was infused intravenously, this created a model that resulted in the stimulation of insulin and suppression of glucagon in the absence of a change in endogenous GLP-1 concentrations. Subjects were studied on four occasions: receiving, in random order, saline, exendin-(9,39) infused at 30 pmol/kg/min (Ex 30) and at 300 pmol/kg/min (Ex 300), and GLP-1-(9,36)amide. Glucose turnover was measured on each occasion using [3-3H]glucose; insulin secretion and action were measured using the minimal model.
RESEARCH DESIGN AND METHODS
Subjects.
After approval by the Mayo institutional review board, we recruited 11 healthy subjects (3 males and 8 females) with no history of prediabetes. Subjects were taking no medications other than oral contraceptives or stable doses of thyroid hormone. Fasting glucose was 4.62 ± 0.13 mmol/L and mean age was 31.0 ± 2.1 years. All subjects were at a stable weight and did not engage in regular exercise. The mean weight and BMI were 82.1 ± 7.1 kg and 27.5 ± 2.0 kg/m2, respectively. Participants were instructed to follow a weight-maintenance diet containing 55% carbohydrate, 30% fat, and 15% protein for at least 3 days prior to the initial study and then throughout the duration of the study. There was no prior abdominal surgery. Body composition was measured using dual-energy X-ray absorptiometry (DEXA scanner; Hologic, Waltham, MA) to determine lean body mass (48.3 ± 3.1 kg). No gastrointestinal symptoms were detected by the bowel disease questionnaire (10). The study was registered at www.clinicaltrials.gov (NCT01218633). The use of exendin-(9,39) and GLP-1-(9,36) was approved as U.S. Food and Drug Administration investigational new drugs (109555 and 109858, respectively).
Experimental design.
Participants were studied on four occasions in random order. On each occasion, subjects were admitted to the Mayo Clinic Clinical Research Unit at 1730 h on the evening prior to the study. Immediately after admission, subjects ate a standard mixed meal (10 kcal/kg; 55% carbohydrate, 30% fat, and 15% protein) and then fasted overnight. The next morning, an 18-gauge cannula was inserted in a retrograde fashion into a dorsal hand vein of the nondominant arm. The hand was placed in a heated box (55°C) to enable sampling of arterialized venous blood. Another cannula was placed in the contralateral arm to enable infusion. At 0600 h (−120), a primed continuous infusion of [3-3H]glucose was initiated (10-μCi bolus followed by 0.1 μCi/min). At 0800 h (0), a variable glucose infusion also labeled with [3-3H]glucose was started so as to produce glucose concentrations similar to those observed after oral ingestion of 50 g of glucose as previously described (11).
All infused glucose contained [3-3H]glucose in amounts equal to the estimated baseline plasma glucose specific activity. In addition, the basal infusion of [3-3H]glucose was altered so as to approximate the anticipated pattern of fall of glucose production in an effort to minimize changes in specific activity throughout the experiment.
On the saline control day, at 0800 h (0 min), normal saline was infused at a rate of 0.1 mL/min (after a 0.4-mL bolus) for the 360-min duration of the experiment (saline). On the exendin 30 day, at 0800 h, exendin-(9,39) was administered as a bolus of 120 pmol/kg followed by an infusion at 30 pmol/kg/min (Ex 30). On the exendin 300 day, at 0800 h, exendin-(9,39) was administered as a 1,200 pmol/kg bolus followed by infusion at 300 pmol/kg/min. The GLP-1-(9,36) day differed from the other study days in that at 0800 h, GLP-1-(9,36)amide was infused at 1.2 pmol/kg/min (after a 4.8 pmol/kg bolus) (GLP). The order of the four study days was random.
Analytical techniques.
Arterialized plasma samples were placed in ice, centrifuged at 4°C, separated, and stored at −20°C until assay. Plasma glucose concentrations were measured using a glucose oxidase method (Yellow Springs Instruments, Yellow Springs, OH). Plasma insulin was measured using a chemiluminescence assay (Access Assay; Beckman, Chaska, MN). Plasma glucagon and C-peptide concentrations were measured by radioimmunoassay (Linco Research, St. Louis, MO).
Calculations and statistical analysis.
Specific activity was smoothed using the method of Bradley et al. (12). Glucose appearance and disappearance were calculated using non–steady-state Steele equations (13,14) using the tracer infusion rate for each interval. The volume of distribution of glucose was assumed to equal 200 mL/kg and the pool correction factor to equal 0.65. Endogenous glucose production was determined by subtracting the glucose infusion rate from the tracer-determined rate of glucose appearance. All rates of infusion and turnover were expressed per kilogram of lean body mass.
Net insulin sensitivity (Si) was estimated from insulin and glucose concentrations using the unlabeled minimal model. A global β-cell responsivity index (ϕ) was estimated from glucose and C-peptide concentrations by using the C-peptide minimal model, incorporating age-associated changes in C-peptide kinetics. Disposition indices (DIs) were calculated as the product of ϕ and Si. Hepatic extraction was also calculated (15).
A repeated-measures ANCOVA was used to test whether fasting, peak, nadir, and integrated hormonal concentrations differed among the four study days, incorporating BMI as a covariate. A compound symmetry correlation structure was assumed, and the Dunnett-Hsu multiple comparison method was used to compare each treatment with saline. A similar approach was used to assess effects on Si and DI. A P value ≤0.05 was considered significant. The analyses used SAS software version 9.3 (SAS Institute Inc., Cary, NC).
RESULTS
Plasma glucose, insulin, C-peptide, and glucagon concentrations.
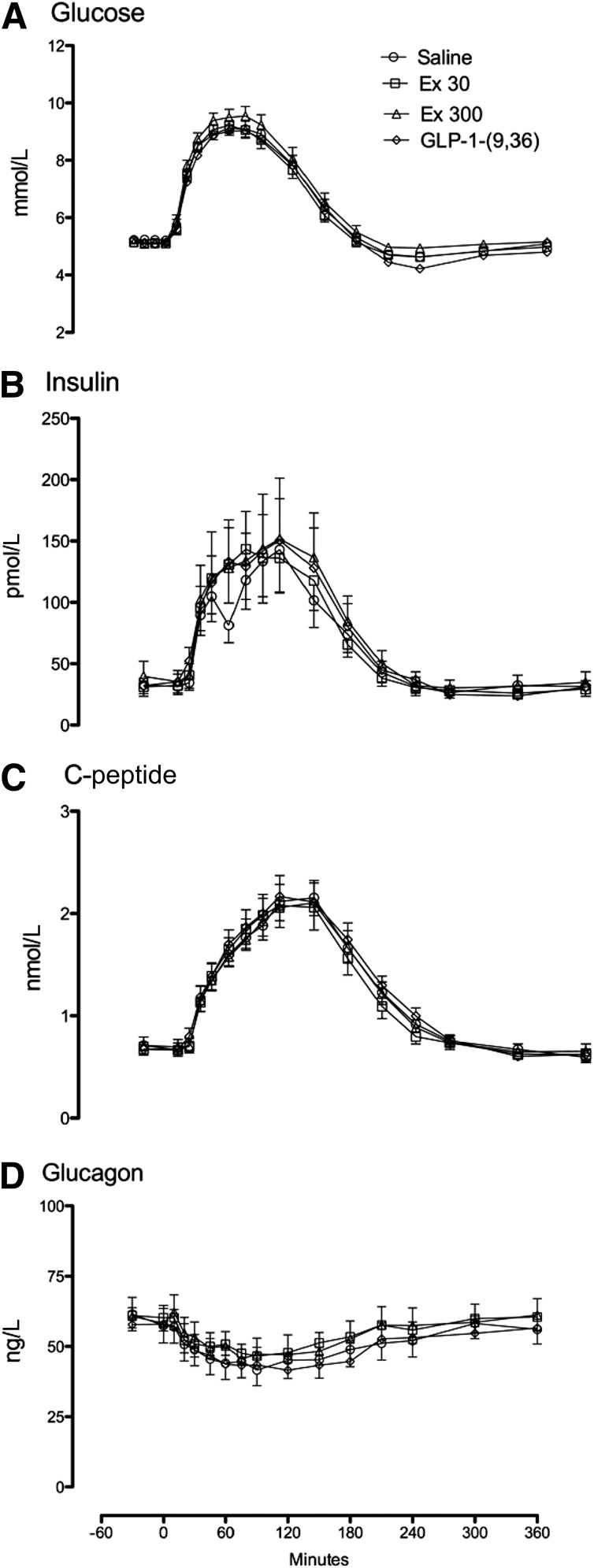
Fasting glucose concentrations did not differ among study days (5.2 ± 0.1 vs. 5.1 ± 0.1 vs. 5.1 ± 0.1 vs. 5.1 ± 0.1 mmol/L for the saline, Ex 30, Ex 300, and GLP-1-[9,36] study days, respectively; P = 0.14). Similarly, peak glucose concentrations did not differ (9.3 ± 0.2 vs. 9.3 ± 0.2 vs. 9.7 ± 0.3 vs. 9.2 ± 0.3 mmol/L; P = 0.28). However, integrated area above basal glucose concentrations (AAB) differed slightly but significantly between study days (P = 0.05) (Fig. 1A), so that glucose concentrations were higher in the presence of exendin infused at 300 pmol/kg/min compared with saline (365 ± 43 vs. 492 ± 49 mmol per 6 h; P = 0.05). Integrated glucose concentrations did not differ on the Ex 30 and GLP-1-(9,36) study days.
FIG. 1.
Glucose (A), insulin (B), C-peptide (C), and glucagon (D) concentrations during the saline, Ex 30, Ex 300, and GLP-1-(9,36)amide infused at a rate of 1.2 pmol/kg/min (GLP-1-[9,36]) study days.
Fasting and integrated AAB insulin, C-peptide, and glucagon concentrations did not differ among study days.
Endogenous glucose production and glucose disappearance.
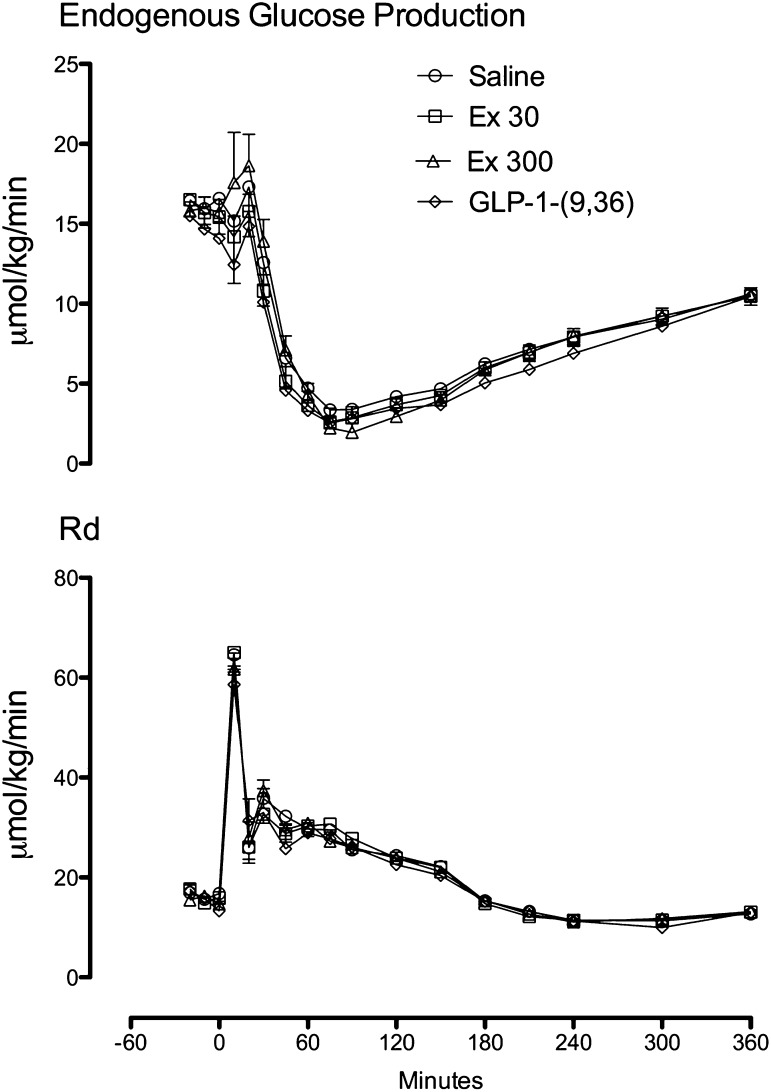
Fasting rates of endogenous glucose production and disappearance did not differ among groups. In addition, suppression of endogenous glucose production (Fig. 2, top panel) and stimulation of glucose disappearance (Fig. 2, bottom panel) did not differ among groups.
FIG. 2.
Rates of endogenous glucose production (top panel) and glucose disappearance (bottom panel) during the saline, Ex 30, Ex 300, and GLP-1-(9,36)amide infused at a rate of 1.2 pmol/kg/min (GLP-1-[9,36]) study days.
Insulin action, β-cell responsivity, DI, and hepatic extraction.
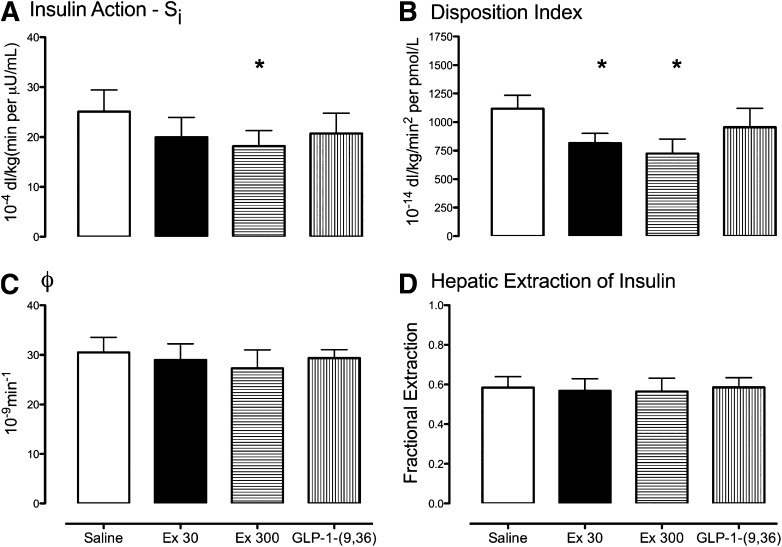
Insulin action (Si) differed among groups (P = 0.023) and was lower with Ex 300 than saline (18 ± 3 vs. 25 ± 4 10−4 dL/kg[min per μU/mL]; P = 0.03) and did not differ on the Ex 30 (20 ± 4 10−4 dL/kg[min per μU/mL]) or GLP-1-(9,36) (21 ± 4 10−4 dL/kg[min per μU/mL]) study days.
In contrast, no differences in β-cell responsivity (ϕ) were observed (31 ± 3 vs. 29 ± 3 vs. 27 ± 4 vs. 30 ± 2 10−9 min−1; P = 0.26) (Fig. 3C). This resulted in a DI that differed between study days (P = 0.003) (Fig. 3B) and was lower on the Ex 30 day compared with saline (816 ± 83 vs. 1,118 ± 118 10−14 dL/kg/min2 per pmol/L; P = 0.02). This was also the case on the Ex 300 day (725 ± 127 10−14 dL/kg/min2 per pmol/L, P = 0.002) compared with saline.
FIG. 3.
Insulin action (Si, A), β-cell responsivity (ϕ, C), DIs (B), and fractional extraction of insulin (D) during the saline, Ex 30, Ex 300, and GLP-1-(9,36)amide infused at a rate of 1.2 pmol/kg/min (GLP-1-[9,36]) study days. *P < 0.05.
Hepatic insulin extraction (Fig. 3D) did not differ among groups (0.58 ± 0.05 vs. 0.57 ± 0.06 vs. 0.56 ± 0.07 vs. 0.58 ± 0.05; P = 0.50).
DISCUSSION
In otherwise healthy subjects, under conditions where there is little endogenous incretin secretion, exendin-(9,39) infusion leads to a decrease in insulin action with an accompanying decrease in DI. This ultimately results in a slight increase in glucose concentrations. Such alterations in insulin secretion and action were not observed with GLP-1-(9,36), and neither compound altered glucagon concentrations. These data suggest that some of the observed effects when exendin-(9,39) is used as a competitive antagonist of GLP-1 at the GLP-1R are attributable to a direct effect of exendin-(9,39), in addition to competitive antagonism of GLP-1, with effects on incretin-mediated insulin secretion, gastric compliance, and gastric emptying (7,8), effects that were not extant under the current experimental conditions.
Although no effect of exendin-(9,39) on absolute β-cell responsivity (ϕ) was observed, when ϕ was expressed as a function of the prevailing level of insulin action, the resulting DI was impaired at both infusion rates, implying a failure of β-cell compensation to the decrease in insulin action. The mechanism by which this occurs is uncertain. One possibility is that inhibition of the actions of fasting concentrations of GLP-1 impedes compensatory insulin secretion. This would not explain the effect on insulin action given the absent effects of GLP-1-(7,36) on this parameter under similar experimental conditions (11).
Exendin has a unique interaction with the GLP-1R (4), but it is uncertain that insulin signaling can be modulated through ligand-GLP-1R interactions (16). Moreover, it seems that GLP-1-(9,36) has actions that are subject to interference by exendin-(9,39) but are not mediated by the GLP-1R (17). Whether this novel, and alternate, signaling pathway can explain our observations also remains unclear. No direct effect of GLP-1-(9,36), or indeed of exendin-(9,39), on whole glucose metabolism was observed, although this does not preclude small effects on specific tissue compartments such as the myocardium.
Peripheral insulin concentrations represent the sum total of insulin secretion into the portal circulation and hepatic extraction as insulin appears in the systemic circulation. This is not a passive process and is affected by insulin secretion (18,19). In rodents, GLP-1 appears to decrease insulin clearance (20,21) but this is not the case in humans (22,23). In the current experiment, neither exendin-(9,39) nor GLP-1-(9,36) altered insulin clearance (15).
Acute infusion of GLP-1-(7,36) in pharmacologic concentrations is associated with increased cortisol concentrations (11). To ensure that our observations were not explained by increased secretion of counterregulatory hormones, we measured growth hormone and cortisol (as well triglycerides and free fatty acid) concentrations during the experiment (see Supplementary Appendix). No significant differences in these concentrations were observed, suggesting that effects on cortisol or growth hormone could not explain our observations. The time course of the effects of these hormones on glucose metabolism would also make this an unlikely explanation (24,25). Of note, neither exendin-(9,39) nor GLP-1-(9,36) lower glucagon, the latter observation implying that circulating GLP-1-(9,36) has little, if any, effect on the suppression of glucagon in the presence of hyperglycemia and hyperinsulinemia.
The current data indicate that exendin-(9,39) causes a slight but significant decrease in both insulin action and insulin secretion that needs to be taken into consideration when this agent is used as a GLP-1-(7,36) antagonist. It remains to be determined whether these effects are also present in individuals who already have impaired insulin action and insulin secretion (as in type 2 diabetes) and also whether they are more pronounced than those observed in the present experiment, with otherwise healthy nondiabetic subjects.
ACKNOWLEDGMENTS
The authors acknowledge the support of the Mayo Clinic General Clinical Research Center. A.V. and C.C. are supported by DK-78646 and DK-82396.
A.V. has received research grants from Merck and Daiichi-Sankyo and has consulted for Sanofi, Novartis, and Bristol-Myers Squibb. The authors thank Merck for providing support for the purchase of exendin-(9,39) and GLP-1-(9,36) in this investigator-initiated study. No other potential conflicts of interest relevant to this article were reported.
M.S. researched data and ran the studies. L.P.F. assisted with data collection and analysis. J.M.M. measured free fatty acid and triglyceride concentrations, contributed to discussion, and reviewed and edited the manuscript. F.P. and C.D.M. performed mathematical modeling of insulin secretion and action. A.R.Z. performed statistical analysis. C.C. reviewed and edited the manuscript. R.A.R. contributed to discussion and reviewed and edited the manuscript. A.V. researched data and wrote the manuscript. A.V. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
Clinical trial reg. no. NCT01218633, clinicaltrials.gov.
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db13-0140/-/DC1.
REFERENCES
- 1.Drucker DJ. Minireview: the glucagon-like peptides. Endocrinology 2001;142:521–527 [DOI] [PubMed] [Google Scholar]
- 2.Deacon CF, Johnsen AH, Holst JJ. Degradation of glucagon-like peptide-1 by human plasma in vitro yields an N-terminally truncated peptide that is a major endogenous metabolite in vivo. J Clin Endocrinol Metab 1995;80:952–957 [DOI] [PubMed] [Google Scholar]
- 3.Vahl TP, Paty BW, Fuller BD, Prigeon RL, D’Alessio DA. Effects of GLP-1-(7-36)NH2, GLP-1-(7-37), and GLP-1- (9-36)NH2 on intravenous glucose tolerance and glucose-induced insulin secretion in healthy humans. J Clin Endocrinol Metab 2003;88:1772–1779 [DOI] [PubMed] [Google Scholar]
- 4.Doyle ME, Theodorakis MJ, Holloway HW, Bernier M, Greig NH, Egan JM. The importance of the nine-amino acid C-terminal sequence of exendin-4 for binding to the GLP-1 receptor and for biological activity. Regul Pept 2003;114:153–158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.D’Alessio DA, Vogel R, Prigeon R, et al. Elimination of the action of glucagon-like peptide 1 causes an impairment of glucose tolerance after nutrient ingestion by healthy baboons. J Clin Invest 1996;97:133–138 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schirra J, Sturm K, Leicht P, Arnold R, Göke B, Katschinski M. Exendin(9-39)amide is an antagonist of glucagon-like peptide-1(7-36)amide in humans. J Clin Invest 1998;101:1421–1430 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schirra J, Nicolaus M, Roggel R, et al. Endogenous glucagon-like peptide 1 controls endocrine pancreatic secretion and antro-pyloro-duodenal motility in humans. Gut 2006;55:243–251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schirra J, Nicolaus M, Woerle HJ, Struckmeier C, Katschinski M, Goke B. GLP-1 regulates gastroduodenal motility involving cholinergic pathways. Neurogastroenterol Motil 2009;21:609–618, e21–22 [DOI] [PubMed]
- 9.Elahi D, Egan JM, Shannon RP, et al. GLP-1 (9-36) amide, cleavage product of GLP-1 (7-36) amide, is a glucoregulatory peptide. Obesity (Silver Spring) 2008;16:1501–1509 [DOI] [PubMed] [Google Scholar]
- 10.Talley NJ, Phillips SF, Melton J, 3rd, Wiltgen C, Zinsmeister AR. A patient questionnaire to identify bowel disease. Ann Intern Med 1989;111:671–674 [DOI] [PubMed] [Google Scholar]
- 11.Vella A, Shah P, Basu R, Basu A, Holst JJ, Rizza RA. Effect of glucagon-like peptide 1(7-36) amide on glucose effectiveness and insulin action in people with type 2 diabetes. Diabetes 2000;49:611–617 [DOI] [PubMed] [Google Scholar]
- 12.Bradley DC, Steil GM, Bergman RN. Quantitation of measurement error with Optimal Segments: basis for adaptive time course smoothing. Am J Physiol 1993;264:E902–E911 [DOI] [PubMed] [Google Scholar]
- 13.Steele R, Bjerknes C, Rathgeb I, Altszuler N. Glucose uptake and production during the oral glucose tolerance test. Diabetes 1968;17:415–421 [DOI] [PubMed] [Google Scholar]
- 14.Steele R, Wall JS, De Bodo RC, Altszuler N. Measurement of size and turnover rate of body glucose pool by the isotope dilution method. Am J Physiol 1956;187:15–24 [DOI] [PubMed] [Google Scholar]
- 15.Campioni M, Toffolo G, Basu R, Rizza RA, Cobelli C. Minimal model assessment of hepatic insulin extraction during an oral test from standard insulin kinetic parameters. Am J Physiol Endocrinol Metab 2009;297:E941–E948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vella A, Rizza RA. Extrapancreatic effects of GIP and GLP-1. Horm Metab Res 2004;36:830–836 [DOI] [PubMed] [Google Scholar]
- 17.Ban K, Kim K-H, Cho C-K, et al. Glucagon-like peptide (GLP)-1(9-36)amide-mediated cytoprotection is blocked by exendin(9-39) yet does not require the known GLP-1 receptor. Endocrinology 2010;151:1520–1531 [DOI] [PubMed] [Google Scholar]
- 18.Sathananthan A, Dalla Man C, Zinsmeister AR, et al. A concerted decline in insulin secretion and action occurs across the spectrum of fasting and postchallenge glucose concentrations. Clin Endocrinol (Oxf) 2012;76:212–219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Meier JJ, Veldhuis JD, Butler PC. Pulsatile insulin secretion dictates systemic insulin delivery by regulating hepatic insulin extraction in humans. Diabetes 2005;54:1649–1656 [DOI] [PubMed] [Google Scholar]
- 20.Ahrén B, Thomaseth K, Pacini G. Reduced insulin clearance contributes to the increased insulin levels after administration of glucagon-like peptide 1 in mice. Diabetologia 2005;48:2140–2146 [DOI] [PubMed] [Google Scholar]
- 21.Pacini G, Thomaseth K, Ahrén B. Dissociated effects of glucose-dependent insulinotropic polypeptide vs. glucagon-like peptide-1 on beta-cell secretion and insulin clearance in mice. Metabolism 2010;59:988–992 [DOI] [PubMed] [Google Scholar]
- 22.Meier JJ, Holst JJ, Schmidt WE, Nauck MA. Reduction of hepatic insulin clearance after oral glucose ingestion is not mediated by glucagon-like peptide 1 or gastric inhibitory polypeptide in humans. Am J Physiol Endocrinol Metab 2007;293:E849–E856 [DOI] [PubMed] [Google Scholar]
- 23.Brandt A, Katschinski M, Arnold R, Polonsky KS, Göke B, Byrne MM. GLP-1-induced alterations in the glucose-stimulated insulin secretory dose-response curve. Am J Physiol Endocrinol Metab 2001;281:E242–E247 [DOI] [PubMed] [Google Scholar]
- 24.Nielsen MF, Dinneen S, Basu A, Basu R, Alzaid A, Rizza RR. Failure of nocturnal changes in growth hormone to alter carbohydrate tolerance the following morning. Diabetologia 1998;41:1064–1072 [DOI] [PubMed] [Google Scholar]
- 25.Dinneen S, Alzaid A, Miles J, Rizza R. Effects of the normal nocturnal rise in cortisol on carbohydrate and fat metabolism in IDDM. Am J Physiol 1995;268:E595–E603 [DOI] [PubMed] [Google Scholar]