Abstract
Introduction
HLA-G is a major histocompatibility complex (MHC), class Ib molecule that is selectively expressed at the fetal–maternal interface. It is thought to play a role in protecting the fetus from the maternal immune response. Interestingly, the preimplantation embryo development (Ped) gene product Qa-2 is also a mouse MHC class Ib protein that affects cleavage and division of preimplantation mouse embryos and subsequent embryonic survival. Data from many human in vitro fertilization (IVF) clinics suggest that the mouse Ped phenomenon also exists in human because embryos fertilized at the same time have different cleavage rates and consequently different IVF outcomes. As HLA-G is expressed in human early embryos, it is highly regarded as the functional homologue of Qa-2. Whether HLA-G expression is correlated with the cleavage rate of human embryos has great potential clinical value.
Methods
In this study, 45 human early abnormal fertilized embryos (3 PN) from patients undergoing in vitro fertilization were used to test the effects of HLA-G knock-down via infection with adenovirus carrying its specific siRNA on the cleavage rate in a 2-day culture period. One-way ANOVA, Post hoc and Chi-square were used to compare groups. A p-value smaller than 0.05 was considered statistically significant.
Results
Knocking-down HLA-G in human pre-implantation stage embryos resulted in a higher cell arrest rate and a slower cleavage rate.
Conclusion
The results from the present study suggested that HLA-G might play an important role in early human embryo development.
Keywords: Adenovirus vector, Cleavage, HLA-G, In vitro fertilization, Mouse Qa-2 antigen, Preimplantation embryo, RNAi
Introduction
Human leukocyte antigen (HLA)-G is a non-classical major histocompatibility complex class I or Class Ib molecule that is primarily expressed at the fetal-maternal interface. It is thought to play a role in protecting the fetus from the maternal immune response. HLA-G is also expressed in preimplantation human embryos (1–4). Embryos capable of secreting soluble HLA-G are associated with a higher implantation rate, ongoing gestation and live birth rates in in vitro fertilization (IVF) cycles (5–7). In human, HLA-G protein is expressed in 75% of embryos at preblastocyst stage and in all at blastocyst stage. More importantly, HLA-G expressing embryos have an increased development rate (1).
Human HLA-G sequence possesses the highest homologue to that of the mouse Q7 and preimplantation embryo development (Ped) gene encoding Qa-2 protein (8). HLA-G and Qa-2 proteins also have similar functions (2, 8–12). HLA-G/Qa-2 proteins are restricted in the plasma membrane lipid rafts in the fast-cleaving human/ mouse embryos (9, 11). It is also reported that the mouse Ped gene regulates the cleavage and division and subsequent survival of preimplantation mouse embryos (13). Qa-2 is expressed in mouse preimplantation embryos from 2-cell through the blastocyst stage (14, 15). Two phenotypes of the Ped gene, fast and slow, have been defined. Ped fast preimplantation embryos develop at a significantly faster rate, both in vivo and in vitro, compared with Ped slow embryos (13, 16, 17). HLA-G is suggested to be the human functional homologue of the mouse Qa-2 protein (2, 8, 9).
The Ped gene phenotype, fast and slow preimplantation embryo development, also exists in human. Data from many IVF clinics showed that embryos from pools of oocytes fertilized simultaneously were not necessarily synchronized in their development, suggesting that gene(s) similar to Ped exist in human which could influence human preimplantation embryo cleavage (18). In addition, embryos that divide more rapidly are more likely to lead to a pregnancy. One report has suggested the fast rate of human embryonic development is associated with expression of HLA-G mRNA (2). The cleavage rate of embryos at 48 h after fertilization is significantly correlated with sHLA antigens in the culture medium (19). HLA-G can act as a signaling molecule to induce proliferation of resting T cells (11).
We hypothesized that HLA-G could affect early human embryonic development. In this study, we designed recombinant adenoviral vectors encoding siRNA against HLA-G for down-regulating HLA-G expression. We infected human early embryos with these siRNA viral vectors to test the effects of HLA-G on embryo cleavage rate.
Methods
Construction of HLA-G siRNA vectors
The construction of the HLA-G siRNA vectors and adenoviral infection in vitro was described elsewhere (20). Hairpin siRNA template that targets the HLA-G gene was inserted into the shuttle vector 1.0-CMV at the XhoI and SpeI restriction enzyme sites (Ambion, Austin, TX, USA). The 21 nt HLA-G target sequence (GenBank accession no. NM002127,3217-3228,3674-3680) was served as a basis for the design of the two complementary 55-mer oligonucleotides. The 55 bp oligonucleotide of hairpin siRNA template was designed by Ambion's Web-Based siRNA Design Resources to interfere exclusively with HLA-G. For a negative control, the oligo sequence was scrambled and arranged not to affect any known human genes (Table 1). After confirmation of the knock-down effect of HLA-G on JEG-3 cell line, the siRNA was cloned into the adenoviral vector (Ad5). The viruses were harvested, amplified and titered using a kit according to the manufacturer's instructions (Ambion, Austin, TX, USA). A multiplicity of infection (MOI) of 100 was applied for both the mouse and human embryos.
Table 1.
Oligonucleotide sequences used for HLA-G RNAi
| Target sequence | 5’-TCGAGAGAAGAGCTCAGATTGAAATTCAAGAGATTTCAATCTGAGCTCTTCTTTA-3’ |
|
| |
|---|---|
| Srambled control | 5’-TCGAGATAGAAGAGACGAGTTAAATTCAAGAGATTTAACTCGTCTCTTCTATTTA-3’ |
Adenoviral infection of mouse embryos
Mouse embryos at the pronuclear stage were flushed out from the oviducts of the super-ovulated Kunming mice in M2 culture medium (Sigma-Aldrich, M7167, UK). Adenoviral vectors with scrambled HLA-G siRNA sequence were added in concentrations of 104 pfu/ml, 105 pfu/ml, 106 pfu/ml and 107 pfu/ml, respectively to the zona-free embryos in M2 medium. Then it was supplemented by Bovine Serum albumin (BSA) to a final concentration of 4 mg/ml. Polybrene (4 μg/ml) was also added to improve infection efficiency. After 2 h of culture, embryos were rinsed with M2 medium and transferred to a CO2 balanced fresh M16 medium covered with mineral oil. The growth of the embryos was observed in the following two days. In the same way, 4-cell embryos were exposed to the adenoviral vectors for 2 h and then cultured for 24 h. Embryos were fixed with 4% parafor-maldehyde (PFA) in phosphate buffer solution (PBS) and then were put in the X-Gal solution (20 mg/ml Bromo-chloro-indolyl-galactopyranoside dissolved in dimethylformamide) overnight at 37°C to stain the galactose-expressing embryos.
Experimental design and adenoviral infection of human embryos
Collection of human embryos and their research use herein were approved by the Ethics Committee of Tangdu Hospital of the FMMU. Embryos were obtained with informed consent from patients who had undergone IVF in the Fertility Center of Tangdu Hospital from March to July 2007. The embryos with abnormal fertilization (3 PN) that were not appropriate for transfer to uterus were selected for this study. The protocols of ovarian induction and in vitro fertilization have been previously described in detail (1). The day of egg retrieval was termed as day 0; insemination was performed on day 0 and the next days were termed as day 1- day 3. The 4- to 8-cell staged embryos collected on day 3 were deprived from their zona-pellucida and were grouped randomly into 3 groups: the mock control (group 1), the scrambled control (group 2) and the HLAG-siRNA group (group 3). The zona-free embryos were cultured in culture medium with or without adenoviral vectors for 1 hour before being put back in normal culture media. In group 1, the embryos were treated with 4 μg/ml polybrene without any adenoviral vectors. In group 2, the embryos transfected with adenoviral vector containing the scrambled siRNA. In group 3, the embryos transfected with the adenoviral vector containing HLAG-siRNA. After 2 days of culture (day 5), embryos were fixed and the number of arrested embryos and blastomeres per embryo were counted.
The adenoviral vectors were diluted to a concentration of 106 pfu/ml in the medium supplemented with 4 μg/ml polybrene. After incubation for an hour, the zona-free embryos were rinsed and cultured individually in 10 μl of fresh culture medium under mineral oil. On day 5, the embryos were fixed in 4% PFA for 30 min, permeabilized with 0.2% Triton X-100 for 10 min and stained with 5 μg/ml fluorochrome 4,6-diamidino-2-phe-nylindole (DAPI, Roche 236276, UK) in PBS for 20 min. The number of blastomeres was counted under a fluorescence microscope.
The adenoviral vector with HLA-G siRNA target sequence exerted a powerful and specific knockdown effect on HLA-G expressing cells (JEG-3 cells).
The expression of LacZ reporter gene in the blastomeres which were stained blue by X-Gal was regarded as the evidence of being infected by the adenoviral vectors.
Statistical analysis
One-way ANOVA, Post hoc and chi-square were used to compare the number of blastomeres per embryo in groups and the percentage of arrested embryos. The means were shown as Mean ± SD. A p-value of <0.05 was considered statistically significant.
Results
Effects of HLA-G adenoviral infection on the cleavage of human embryos
A total of 45 human embryos were obtained. They were abnormally fertilized embryos with three pronuclei (3 PN) at 4-8 cell stage including 14 grade 1 and 31 grade 2 embryos. The donors’ mean age was 29.64 ± 0.48 (ranging from 24 to 44, Table 2). There was no statistical differences regarding the average cell count, grade or the donors’ age among groups 1, 2 and 3 (Table 2). After 2 days of culture following transfection, 53.33% and 92.31% embryos, respectively from group 2 and 3 arrested which were significantly higher than that of group 1 (17.65%) (Table 3). The number of blastomeres after 4 days of transfection was 17.71 ± 6.57, 10.87 ± 4.16 and 7.69 ± 1.55, respectively in groups 1, 2 and 3 (Table 3). There was significant differences between any two groups among the three, with a p-value of 0.004 (between group 1 and 2), 0.000 (between group 1 and 3) and 0.039 (between group 2 and 3).
Table 2.
Cell number, grade and the donor's age of day 3 embryos at the time of transfection
| Group | Number of embryos | Number of blastomeres at the time of transfection (M±SD) | Grade (M±SD) | Donors’ age (M±SD) |
|---|---|---|---|---|
| 1 | 17 | 7.53 ± 1.62 | 1.76 ± 0.44 | 29.18 ± 4.43 |
| 2 | 15 | 7.60 ± 0.99 | 1.60 ± 0.51 | 30.20 ± 2.48 |
| 3 | 13 | 7.38 ± 0.87 | 1.69 ± 0.48 | 29.62 ± 1.89 |
Group 1: Mocked trasnfection; Group 2: Scrambled siRNA transfection. Group 3: HLAG siRNA transfection. None of the difference was statistically significant.
Table 3.
Effect of HLA-G siRNA transfection of human day 3 embryos on embryo arrest and number of blastomers after 2 days in vitro cultre
| Group | Number of embryos | Arrested embryos (M±SD) | No. of blastomeres per embryo after 2 days of culture (M±SD) |
|---|---|---|---|
| 1 | 17 | 17.65% a | 17.71 ± 6.57 a |
| 2 | 15 | 53.33% b | 10.87 ± 4.16 b |
| 3 | 13 | 92.31% c | 7.69 ± 1.55 c |
Group 1: Mocked trasnfection; Group 2: Scrambled siRNA transfection; Group 3: HLAG siRNA transfection
indicate that the difference was statistically significant between groups (P < 0.05)
Efficiency of adenoviral vector transfection on HLA-G expressing cells and mouse embryos
The HLA-G mRNA's relative expression decreased significantly (about 24% less) in HLAG-siRNA group compared with the scrambled control 48 h after infection. The HLA-G protein's relative expression decreased significantly (about 50% less) in the Ad5-siRNA group versus the Ad5-scrambled control.
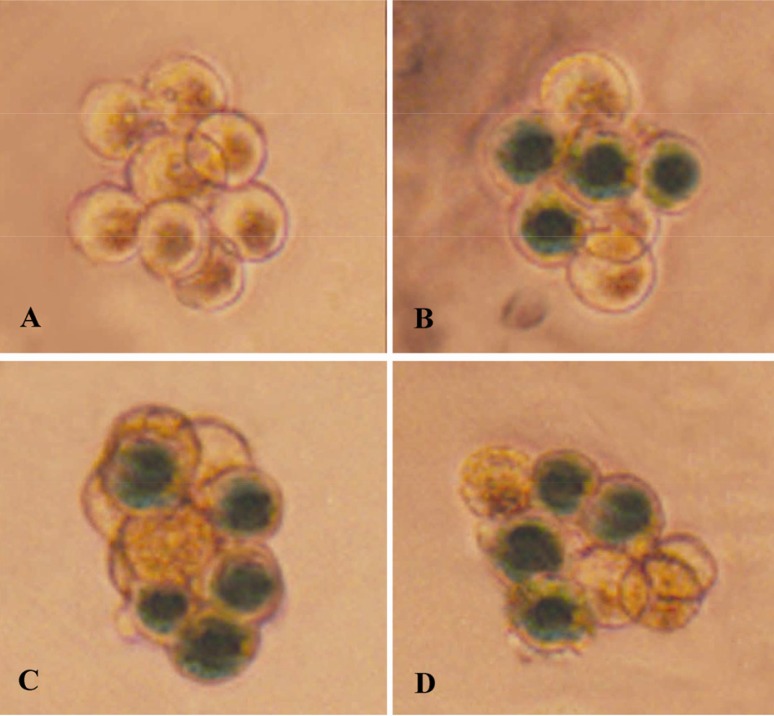
Mouse 4-8 cell embryos can be infected with ≥106 pfu/ml Ad5 vectors with about 50% efficiency (Figure 1). No obvious toxicity was found during embryo culture. However, at 107 pfu/ml, the adenoviral vectors showed toxicity to the embryos despite a higher infection rate (Data not shown). We then chose to use 106 pfu/ml for the human embryo studies.
Figure 1.
Expression of LacZ on the siRNA transfected mouse embryos. A) Embryos without zona pellucida cultured to 8-cell stage without virus infection; B-D) Adenoviral infection group, LacZ expression blastomeres stained blue)
Discussion
RNA interference, known as RNAi, is an efficient gene knock-out technology. One of the biggest challenges in the use of RNAi to study gene function in mammalian cells is the efficiency of successful delivery of small interfering RNAs (siRNAs) or siRNA expression vectors into the cells. The Ad serotype 5 (Ad5) vector used in this study was a replication defective vector encoding bacterial β-galactosidase (lacZ) gene as a marker. The Ad vector has high infection efficiency with a remarkable gene knockdown effect in the JEG-3 cells cultured in vitro. The NK-mediated cytotoxicity of HLA-G knocking-down cells is increased (20). Furthermore, they are easy and safe to manipulate. The purified virus has less toxicity to the cells as compared with lipophilic transfection reagents. Despite the application of the Ad vectors in basic research and gene therapy, little information is available on Ad vector-mediated gene transfer into human embryos. We observed that transfection of the virus in a concentration of 107 pfu/ml has potential toxicity to 2-cell human embryos by delaying their developmental speed and destroying their morphology (data not shown). Non-infectious viral particles can provoke a cell-mediated immune response, exert direct toxic effects on cells and consequently disturb surface interactions between blastomeres which all are harmful for pre-implantation-stage embryos. In order to reduce the toxicity to embryos, 106 pfu/ml titer was applied in this study.
To our knowledge, this is the first report using siRNA-expressing Ad vector to transfect human early embryo in HLA-G function study. The ade-noviral vector targeting HLA-G provides a possibility to investigate and compare embryos with HLA-G expression with their deficient analogue. As embryo cleavage speed is an indicator of successful IVF outcome (5–7), our findings may also shed light on the clinical work. From our data in mock control group, the embryos which had not been exposed to Ad vector showed an unusual low cleavage and high arrest rate during the 2 days of culture (Table 3). The average number of blastomeres per embryo was only 17.71 ± 6.57 and most of the morulas could not form blastocysts (Table 3). It might have been caused by the abnormally fertilized three pronuclei (3 PN) human embryos we used whose cleaving rate were always slowed-down (17). In the study by Jurisicova et al. (4), the blastocyst formation rate from 3 PN human embryos was significantly lower than that from 2 PN (normal) embryos (20% versus 56%). Aneuploidy embryos are more likely to be blocked on day 3 than the euploidy ones. In our study, the HLA-G down-regulated embryos had a significant slower cleavage rate, consistent with the previous findings (2, 20). In contrast, embryos expressing HLA-G were associated with a higher cleaving rate and better morphology (5–7).
In this study, we used 3-PN embryos instead of normally fertilized embryos. It is estimated that only about 50% of the IVF embryos are euploid, and self-correction occurs during the development toward the blastocyst stage (21). With slight discontent, we believed that the effects of HLA-G on 3PN human embryos could represent its effects on normal embryos which were difficult ethically to be used in research. Importantly, we used the 3PN human embryos as the controls to reduce the influences of chromosomes on development.
Mice embryos expressing Qa-2 are characterized by the Ped fast phenotypes, which exhibit faster pre-implantation cleavage rate (both in vitro and in vivo), better survival to term, and higher birth and weaning weights compared with the embryos not expressing Qa-2 and showing Ped slow phenotypes (13, 22). Chemical removal of the Qa-2 protein from the embryonic cell surface results in a slowing down of embryo development (23). Additionally, antisense oligonucleotides to Qa-2 mRNA decrease the cleavage rate of the Ped fast embryos (24). Conversely, transferring Qa-2 gene to the Qa-2-negative zygotes leads to the expression of Qa-2 and an increased cleavage rate (19, 25). Addition of exogenous Qa-2 protein into the membrane of Ped-negative embryos also increases the rate of cleavage (26).
Similar to Qa-2, HLA-G is also present in the lipid rafts in the cell membrane and can act as a signaling molecule to induce proliferation of resting T cells (11). To date, seven HLA-G mRNA transcripts have been identified, which encode four membrane-bound isoforms (G1, G2, G3 and G4) and three soluble isoforms (G5, G6 and G7). Yao et al. (1) found that HLA-G3 and -G4 were the predominant isoforms expressed in human embryos. Transcription of HLA-G mRNA was triggered at 6-8 cell stage and increased as the embryos developed (1). All human blastocysts and 75% of pre-blastocyst expressed HLA-G protein.
Conclusion
We presumed that the expression of HLA-G protein increased human early embryo cleavage and its expression might facilitate blastomere cleavage rate and blastocyst formation in 4- to 8-cell-stage embryos. The adenoviral vector we constructed in this study was highly effective on transfection and thus provided a potent tool for further investigation in HLA-G function.
Acknowledgement
This study was supported by The Chinese Natural Science Foundation (No. 30471812) and the National Basic Research Program of China (grant number: 2007CB948102). A consent form from each embryo donor was obtained before starting the experiment.
To cite this article: Sun LL, Ming Wang A, J Haines Ch, Han Y, Qing Yao Y. Down-regulation of HLA-G Attenuates Cleavage Rate in Human Triploid Embryos. J Reprod Infertil. 2011;12(3):215-220.
References
- 1.Yao YQ, Barlow DH, Sargent IL. Differential expression of alternatively spliced transcripts of HLA-G in human preimplantation embryos and inner cell masses. J Immunol. 2005;175(12):8379–85. doi: 10.4049/jimmunol.175.12.8379. [DOI] [PubMed] [Google Scholar]
- 2.Jurisicova A, Casper RF, MacLusky NJ, Mills GB, Librach CL. HLA-G expression during preimplantation human embryo development. Proc Natl Acad Sci U S A. 1996;93(1):161–5. doi: 10.1073/pnas.93.1.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cao W, Brenner CA, Alikani M, Cohen J, Warner CM. Search for a human homologue of the mouse Ped gene. Mol Hum Reprod. 1999;5(6):541–7. doi: 10.1093/molehr/5.6.541. [DOI] [PubMed] [Google Scholar]
- 4.Jurisicova A, Antenos M, Kapasi K, Meriano J, Casper RF. Variability in the expression of trophectodermal markers beta-human chorionic gonadotrophin, human leukocyte antigen-G and pregnancy specific beta-1 glycoprotein by the human blastocyst. Hum Reprod. 1999;14(7):1852–8. doi: 10.1093/humrep/14.7.1852. [DOI] [PubMed] [Google Scholar]
- 5.Noci I, Fuzzi B, Rizzo R, Melchiorri L, Criscuoli L, Dabizzi S, et al. Embryonic soluble HLA-G as a marker of developmental potential in embryos. Hum Reprod. 2005;20(1):138–46. doi: 10.1093/humrep/deh572. [DOI] [PubMed] [Google Scholar]
- 6.Yie SM, Balakier H, Motamedi G, Librach CL. Secretion of human leukocyte antigen-G by human embryos is associated with a higher in vitro fertilization pregnancy rate. Fertil Steril. 2005;83(1):30–6. doi: 10.1016/j.fertnstert.2004.06.059. [DOI] [PubMed] [Google Scholar]
- 7.Fisch JD, Keskintepe L, Ginsburg M, Adamowicz M, Sher G. Graduated Embryo Score and soluble human leukocyte antigen-G expression improve assisted reproductive technology outcomes and suggest a basis for elective single-embryo transfer. Fertil Steril. 2007;87(4):757–63. doi: 10.1016/j.fertnstert.2006.08.088. [DOI] [PubMed] [Google Scholar]
- 8.Geraghty DE, Koller BH, Orr HT. A human major histocompatibility complex class I gene that encodes a protein with a shortened cytoplasmic segment. Proc Natl Acad Sci U S A. 1987;84(24):9145–9. doi: 10.1073/pnas.84.24.9145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Comiskey M, Goldstein CY, De Fazio SR, Mammolenti M, Newmark JA, Warner CM. Evidence that HLA-G is the functional homolog of mouse Qa-2, the Ped gene product. Hum Immunol. 2003;64(11):999–1004. doi: 10.1016/j.humimm.2003.08.352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Comiskey M, Warner CM, Schust DJ. MHC molecules of the preimplantation embryo and trophoblast. In: Markert UR, editor. Immunology of pregnancy. Switzerland: Karger Publishers; 2005. pp. 72–80. [Google Scholar]
- 11.Comiskey M, Domino KE, Warner CM. HLA-G is found in lipid rafts and can act as a signaling molecule. Hum Immunol. 2007;68(1):1–11. doi: 10.1016/j.humimm.2006.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Clements CS, Kjer-Nielsen L, Kostenko L, Hoare HL, Dunstone MA, Moses E, et al. Crystal structure of HLA-G: a nonclassical MHC class I molecule expressed at the fetal-maternal interface. Proc Natl Acad Sci U S A. 2005;102(9):3360–5. doi: 10.1073/pnas.0409676102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Warner CM, Gollnick SO, Flaherty L, Goldbard SB. Analysis of Qa-2 antigen expression by preimplantation mouse embryos: possible relationship to the preimplantation-embryo-development (Ped) gene product. Biol Reprod. 1987;36(3):611–6. doi: 10.1095/biolreprod36.3.611. [DOI] [PubMed] [Google Scholar]
- 14.McElhinny AS, Warner CM. Detection of major histocompatibility complex class I antigens on the surface of a single murine blastocyst by immuno-PCR. Biotechniques. 1997;23(4):660–2. doi: 10.2144/97234bm26. [DOI] [PubMed] [Google Scholar]
- 15.McElhinny AS, Kadow N, Warner CM. The expression pattern of the Qa-2 antigen in mouse pre-implantation embryos and its correlation with the Ped gene phenotype. Mol Hum Reprod. 1998;4(10):966–71. doi: 10.1093/molehr/4.10.966. [DOI] [PubMed] [Google Scholar]
- 16.Goldbard SB, Verbanac KM, Warner CM. Role of the H-2 complex in preimplantation mouse embryo development. Biol Reprod. 1982;26(4):591–6. doi: 10.1095/biolreprod26.4.591. [DOI] [PubMed] [Google Scholar]
- 17.Magli MC, Gianaroli L, Munné S, Ferraretti AP. Incidence of chromosomal abnormalities from a morphologically normal cohort of embryos in poor-prognosis patients. J Assist Reprod Genet. 1998;15(5):297–301. doi: 10.1023/A:1022596528036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Biezinová J, Svobodová M, Oborná I, Fingerová H, Dostá J, Krsková M. [Embryo quality evaluation according to the speed of the first cleavage after conventional IVF] Ceska Gynekol. 2006;71(2):105–10. Czech. [PubMed] [Google Scholar]
- 19.Xu Y, Jin P, Mellor AL, Warner CM. Identification of the Ped gene at the molecular level: the Q9 MHC class I transgene converts the Ped slow to the Ped fast phenotype. Biol Reprod. 1994;51(4):695–9. doi: 10.1095/biolreprod51.4.695. [DOI] [PubMed] [Google Scholar]
- 20.Sun LL, Han Y, Chen JH, Yao YQ. Down-regulation of HLA-G boosted natural killer cell-mediated cytolysis in JEG-3 cells cultured in vitro. Fertil Steril. 2008;90(6):2398–405. doi: 10.1016/j.fertnstert.2007.10.076. [DOI] [PubMed] [Google Scholar]
- 21.Barbash-Hazan S, Frumkin T, Malcov M, Yaron Y, Cohen T, Azem F, et al. Preimplantation aneuploid embryos undergo self-correction in correlation with their developmental potential. Fertil Steril. 2009;92(3):890–6. doi: 10.1016/j.fertnstert.2008.07.1761. [DOI] [PubMed] [Google Scholar]
- 22.Brownell MS, Warner CM. Ped gene expression by embryos cultured in vitro. Biol Reprod. 1988;39(4):806–11. doi: 10.1095/biolreprod39.4.806. [DOI] [PubMed] [Google Scholar]
- 23.Tian Z, Xu Y, Warner CM. Removal of Qa-2 antigen alters the Ped gene phenotype of preimplantation mouse embryos. Biol Reprod. 1992;47(2):271–6. doi: 10.1095/biolreprod47.2.271. [DOI] [PubMed] [Google Scholar]
- 24.Xu Y, Jin P, Warner CM. Modulation of preimplantation embryonic development by antisense oligonucleotides to major histocompatibility complex genes. Biol Reprod. 1993;48(5):1042–6. doi: 10.1095/biolreprod48.5.1042. [DOI] [PubMed] [Google Scholar]
- 25.Wu L, Feng H, Warner CM. Identification of two major histocompatibility complex class Ib genes, Q7 and Q9, as the Ped gene in the mouse. Biol Reprod. 1999;60(5):1114–9. doi: 10.1095/biolreprod60.5.1114. [DOI] [PubMed] [Google Scholar]
- 26.McElhinny AS, Exley GE, Warner CM. Painting Qa-2 onto Ped slow preimplantation embryos increases the rate of cleavage. Am J Reprod Immunol. 2000;44(1):52–8. doi: 10.1111/j.8755-8920.2000.440108.x. [DOI] [PubMed] [Google Scholar]