Abstract
The CXC chemokines, monokine induced by interferon (IFN)-gamma (MIG) (CXCL9), IFN-gamma-induced protein 10 (IP-10) (CXCL10) and IFN-inducible T cell alpha chemoattractant (I-TAC) (CXCL11), are known to attract CXCR3- (CXCR3A and CXCR3B) T lymphocytes. We investigated MIG, IP-10 and I-TAC mRNAs expression by semi-quantitative multiplex reverse transcription–polymerase chain reaction (RT–PCR) in liver biopsies obtained from patients with a first diagnosis of primary biliary cirrhosis [(PBC) = 20] compared to patients with normal liver biopsy [normal controls (NCs) = 20]. Chemokine production was assessed by enzyme-linked immunosorbent assay (ELISA) in serum. Measurements were repeated 6 months after ursodeoxycholic acid (UDCA) treatment in PBC patients. CXCR3A and CXCR3B mRNAs expression was examined in immunomagnetically sorted CD3+ peripheral blood lymphocytes (PBL) pre- and post-treatment by RT–PCR. Flow cytometry was used to evaluate the expression of CXCR3+ PBLs of NCs and PBC patients. A marked mRNA expression of MIG and IP-10 was found in PBC patients. I-TAC mRNA was not detected. In serum of PBC patients there was a significant increase of MIG and IP-10 compared to NCs. Interestingly, there was a significant reduction of these proteins in patients' serum after UDCA treatment. I-TAC was not statistically different between groups. CXCR3A mRNA expression was found in PBLs from PBC patients as well as in NCs. CXCR3B mRNA was expressed in four of 20 (19%) NCs and 20 of 20 PBC patients. Flow cytometry revealed a significantly lower CXCR3 expression in NCs (13·5%) than in PBC (37·2%), which was reduced (28·1%, P < 0·01) after UDCA administration. These data suggest a possible role for CXCR3-binding chemokines and their receptor in the aetiopathogenetic recruitment of lymphocytes in PBC and a new mechanism of action for UDCA.
Keywords: CXC chemokines, CXCR3 receptor, primary biliary cirrhosis, ursodeoxycholic acid
Introduction
Chemokines and their receptors have been involved in the pathogenesis of a variety of liver diseases during initiation as well as maintenance of hepatic disorder [1]. Growing evidence suggests the implication of T lymphocyte migration in the pathogenesis of liver diseases via chemokine production in the liver tissue [2]. The CXC chemokines, monokine induced by interferon (IFN)-gamma (MIG) (CXC9), IFN-gamma-induced protein 10 (IP-10) (CXC10) and IFN-inducible T cell alpha chemoattractant (I-TAC) (CXC11), have been shown to attract T lymphocytes. CXCR3 is the only receptor on T cells for MIG, IP-10 and I-TAC [3–5]. Recently, the CXCR3 gene was shown to generate two distinct mRNAs by alternative splicing of three different exons, CXCR3A and CXCR3B [6,7].
Primary biliary cirrhosis (PBC) is characterized histologically by the pathological destruction of interlobular small bile ducts and chronic destructive cholangitis. Recruitment of lymphocytes in the portal tracts is a key feature. In inflamed portal tracts of PBC patients, CD4+ T cells of T helper type 1 (Th1) expressing IFN-gamma are detected around injured bile ducts. Infiltrating T cells may play an important role in the destruction of the biliary tract in these patients [8]. Ursodeoxycholic acid (UDCA) has been used for many years as the main treatment of PBC by amelioration of portal inflammation. The mechanisms of its action are multiple [9].
In the present study, we examined the production of CXCR3-binding chemokines and their receptor in PBC patients before and after UDCA administration.
Patients and methods
Patients
PBC diagnosis was established at the Gastroenterology Department, University Hospital of Heraklion on the basis of a cholestatic biochemistry, positive anti-mitochondrial antibodies (AMA) and a compatible liver biopsy. Liver biopsies, obtained from patients with abnormal liver function tests and negative virology studies, with normal architecture, were used as normal controls. All patients with a first diagnosis of PBC were classified as stages II or III, according to Ludwig [10]. Patients with stage IV (cirrhosis) were excluded.
Liver biopsy material was obtained from 20 patients (18 females, age 21–62 years) with primary biliary cirrhosis (PBC) and 20 normal controls (NCs) (17 females, age 32–64 years) (Table 1). As normal controls, we considered patients with a very mild increase in aminotransferases with negative virological and immunological tests and a normal liver biopsy. Peripheral blood (serum and mononuclear cell isolation) was also collected at the time of the biopsy and 6 months after UDCA treatment (13–15 mg/kg body weight). Consent was obtained in all cases, and the local Research Ethics Committee granted approval for these studies.
Table 1.
Patients' characteristics
| Normal values | PBC | Controls | |
|---|---|---|---|
| Number of patients | 20 | 20 | |
| Females, n (%) | 18 (90%) | 17 (85%) | |
| Age, median (range) | 49 (21–62) | 55 (32–64) | |
| %AMA+ | 100% | 0% | |
| Stage II (Ludwig) | 13 (65%) | n.a. | |
| Stage III (Ludwig) | 7 (35%) | n.a. | |
| ALP (median, range) | 35–129 U/l | 165 (120–320) | 89 (40–144) |
| TBL (median, range) | <1·3 mg/dl | 1·2 (0·6–2·2) | 0·6 (0·4–1·2) |
| γGT (median, range) | 5–36 U/l | 44 (22–120) | 34 (22–56) |
| SGOT (median, range) | <31 U/l | 55 (30–144) | 44 (19–140) |
| SGPT (median, range) | <31 U/l | 68 (44–220) | 64 (24–93) |
γGT: gamma-glutamyl transferase; ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotranfrerase; n.a.: not applicable; TBL: total bilirubin; PBC: primary biliary cirrhosis; SGOT: serum glutamic oxaloacetic transaminase; SGPT: serum glutamic pyruvic transaminase; AMA: anti-mitochondrial antibodies.
Purification of peripheral blood lymphocyte (PBL) subpopulations
Peripheral blood was diluted immediately 1:1 in Iscove's modified Dulbecco's medium (IMDM; Gibco, Invitrogen Corporation, Paisley, UK), supplemented with 10 IU/ml preservative-free heparin (Sigma, St Louis, MO, USA). Diluted peripheral blood was centrifuged on Lymphoprep (Nycomed Pharma AS, Oslo, Norway) at 400 g for 30 min at room temperature in order to obtain the peripheral blood mononuclear cells (PBMCs). The CD3+ PB cell subpopulation, representing T lymphocytes, were isolated from the PBMC fraction by magnetic-activated cell sorting (MACS) (Mitenyi Biotec GmbH, Bergisch Gladbach, Germany), according to the manufacturer's protocol. In all experiments, the purity of each subpopulation was >95%, as estimated by flow cytometry. Isolated CD3+ cells were placed immediately into TRIzol in order to extract mRNA [11].
Reverse transcription–polymerase chain reaction (RT–PCR)
Total RNA was extracted from PBLs and liver tissue homogenized on ice with a glass homogenizer using 1 ml RNAzol solution. RNA extraction and RT–PCR were performed as described previously [12,13]. Briefly, 100 ng mRNA was denatured in the presence of 5 mM oligo (dT) 12–18 primer. It was then reverse-transcribed in a 10-ml volume with Superscript II (Gibco), 1 × RT buffer, 1 mM deoxyribonucletide triphosphates (dNTPs), 5 mM dithiothreitol (DDT) and 2·5 U/ml RNAsin (Promega Corp., Southampton, UK) at 42°C for 60 min. One-ml aliquots of cDNA were PCR amplified in a 25-ml reaction, containing: 1 × PCR buffer and 2 mM MgCl2 0·2 mM dNTPs, 0·5 mM sense and anti-sense primers for CXCL9, CXCL10, CXCL11, CXCR3 and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and 0·4 U High Fidelity Expand polymerase (Roche Molecular Biochemicals, Lewes, Sussex, UK). An identical parallel PCR reaction was performed containing starting material, which had not been reverse-transcribed. PCR products were resolved by electrophoresis on 2% agarose gels and visualized by ethidium bromide staining. The ratio of PCR product, as measured by densitometry between tested genes and GAPDH at a constant volume of RT product, was used for cross-sample comparison. The oligonucleotide sequence and product size for specific primer pairs used are shown in Table 2.
Table 2.
Primer sequences used for the reverse transcription–polymerase chain reaction studies
| Gene | Sense | Antisense | bp size |
|---|---|---|---|
| CXCL9 | 5′-CTCTTTCCTGGCTACTCCAT-3′ | 5′-TCTCGGTGGCTATCTTGTTA-3′ | 354 |
| CXCL10 | 5′-CTGAAAGCAGTTAGCAAGGA-3′ | 5′-TATTTGAAGCAGGGTCAGAA-3′ | 330 |
| CXCL11 | 5′-TTTCAGATGCCCTTTTCCAG-3′ | 5′-CAAACATGAGTGTGAAGGGC-3′ | 322 |
| CXCR3 full size | 5′-AAGTGCTAAATGACGCCG-3′ | 5′-CAAAGGCCACCACGACCACCACCA-3′ | 770 |
| CXCR3 splice variant | 5′-CCAAGTGCTAAATGACGCCG-3′ | 5′-CTCCCGGAACTTGACCCCTGTG-3′ | 622 |
| GAPDH | 5′-CAACGGATTTGGTCGTATTG-3′ | 5′-GATGACAAGCTTCCCGTTCT-3′ | 184 |
GAPDH: glyceraldehyde 3-phosphate dehydrogenase; bp: base pairs.
Chemokine enzyme-linked immunosorbent assay (ELISA)
Extracellular chemokine activity of serum was measured by sandwich ELISA, according to the manufacturers' instructions (Duoset® ELISA Development System; R&D Systems, Abingdon, UK). Briefly, mouse anti-human CXCL9, CXCL10 and CXCL11 antibodies (2 μg/ml) were used to coat goat anti-mouse IgG microplates overnight at room temperature (EvenCoat™; R&D Systems). Biotinylated goat anti-human CXCL9, CXCL10 and CXCL11 antibodies were used for detection. ELISA was developed by incubation with streptavidin–horseradish peroxidase followed by tetramethylbenzidine and H2O2 as the substrate.
Flow cytometry
Two-colour flow cytometry was used for the analysis of PBL subpopulations, as described previously [14]. Briefly, 106 isolated PBLs were incubated with staining reagents for 30 min on ice. In particular, anti-CXCR3 phycoerythrin-cyanin 5 (PE-Cy5)-conjugated (clone 1C6; BD Pharmingen™, San Jose, CA, USA) was combined with anti-CD3 PE [monoclonal antibodies (mAbs); Beckman Coulter, Marseille, France]-conjugated mouse anti-human monoclonal antibodies representing the T cell marker. PE-Cy5- and PE-conjugated mouse immunoglobulin (Ig)G of the appropriate isotype served as negative controls. Analysis on 30 000 events was performed in an Epics Elite model flow cytometer (Coulter, Miami, FL, USA). The results of CXCR3 staining were expressed as the percentage of cells expressing CXCR3 within the PBL CD3+ cell population.
Statistical analysis
Statistical analysis was made using the spss statistical package (version 13.0; SPSS, Inc., Chicago, IL, USA). Results are expressed as mean ± standard error of the mean. The Kolmogorov–Smirnov test was used to check the Gaussian data distribution. Statistical comparisons were performed using the one-way analysis of variance (anova) test with Tukey's post-hoc comparisons. If Burtlett's test indicated a significant difference between standard deviations, the non-parametric Kruskal–Wallis test was used instead. A P-value of 0·05 or less was considered statistically significant.
Results
Chemokine expression and production
Marked MIG and IP-10 mRNA expression was confirmed in the liver biopsies of the PBC patients (100%). In contrast, there was almost no expression of MIG and IP-10 mRNA in NCs (Fig. 1). I-TAC mRNA expression was not detected in 35 cycles of amplification in patients with PBC or NCs (data not shown).
Figure 1.
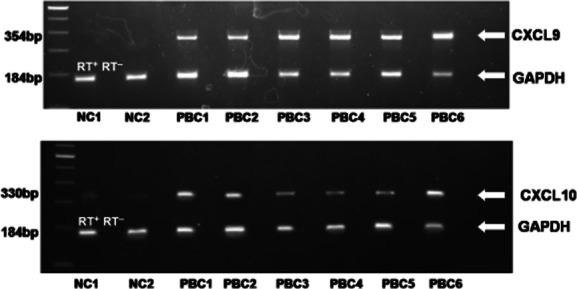
CXCR3 binding chemokines are expressed highly in liver biopsies obtained from primary biliary cirrhosis (PBC) patients. Total RNA was extracted from liver biopsies of PBC patients (n = 20) and normal controls (NCs, n = 20), and multiplex reverse transcription–polymerase chain reaction (RT–PCR) for chemokines and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was performed. (a) Representative PCR blots of mRNA expression from tissue obtained from patients with PBC and NCs, probed for CXCL9/monokine induced by interferon (IFN)-gamma (MIG), CXCL10/IFN-gamma-induced protein-10 (IP-10) and GAPDH. In order to control for genomic contamination each sample has a reverse transcriptase negative control (RT–). The gel shows fluorescence of ethidium bromide-stained PCR products resolved by electrophoresis.
CXCR3-binding chemokines were assessed by ELISA in peripheral blood from PBC patients and NCs. There was a significant increase of MIG (68·49 ± 1·97 pg/ml) and IP-10 (694·85 ± 27·4 pg/ml) levels in serum in PBC patients compared to NCs (30·61 ± 2·52 pg/ml and 34·20 ± 0·81 pg/ml, P < 0·001, respectively), revealing a similar pattern with liver tissue mRNA expression for these chemokines (Fig. 2). Interestingly, there was a significant reduction of both MIG and IP-10 in the serum of PBC patients after UDCA administration (38·64 ± 1·56 pg/ml for MIG, P < 0·001 and 289·20 ± 11·98 pg/ml for IP-10, P < 0·001) compared to untreated patients (Fig. 2). I-TAC serum levels showed no differences between groups (NC 29·8 ± 4·6 pg/ml, PBC before treatment 62·2 ± 22·6 pg/ml and PBC after UDCA 55·6 ± 11·5 pg/ml).
Figure 2.
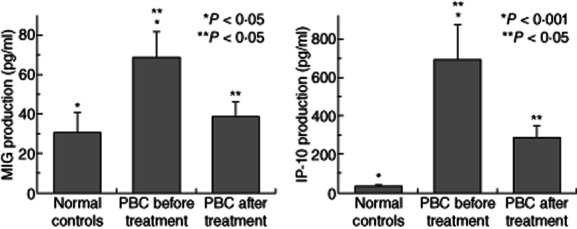
Serum levels of CXCL9/monokine induced by interferon (IFN)-gamma (MIG) CXCL10/IFN-gamma-induced protein-10 (IP-10) are increased preferentially in primary biliary cirrhosis (PBC) patients, while this increase is regulated by ursodeoxycholic acid (UDCA) administration. CXCR3-binding chemokines were measured by enzyme-linked immunosorbent assay in serum of PBC patients (n = 20) and normal controls (NCs, n = 20). Each column represents mean ± standard error of the mean (*P < 0·05).
Chemokine receptor expression
The CXCR3 receptor expression was examined, using RT–PCR and flow cytometry, in CD3+ PBL from PBC patients and NCs. In addition, CXCR3B was examined by RT–PCR in the same groups. mRNA expression of CXCR3A was found in CD3+ PBL in all PBC patients and in 17 of 20 NCs. In contrast, mRNA expression of CXCR3B was detected in four of 20 NCs tested and in all PBC patients (20 of 20) (Fig. 3). Flow cytometry revealed that CXCR3 expression on CD3+ cells was significantly (P < 0·001) lower in NCs (13·54 ± 1·25%) compared to the patients with PBC (37·27 ± 1·41%) (Fig. 4). Treatment with UDCA reduced CXCR3 expression significantly (P < 0·01) in patients with PBC (27·30 ± 1·66%) compared to untreated patients (Fig. 4).
Figure 3.
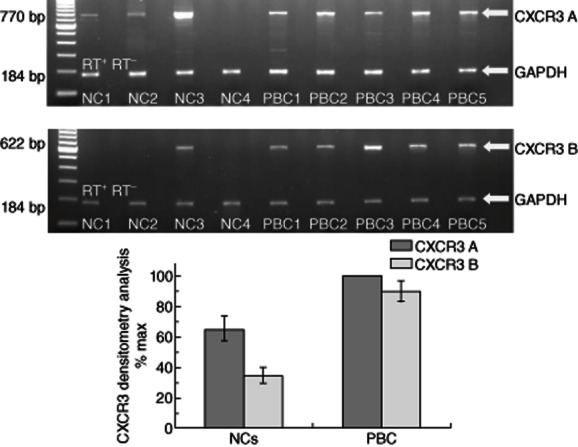
CXCR3A and B are expressed highly in peripheral blood lymphocytes (PBLs) of primary biliary cirrhosis (PBC) patients. Total RNA was extracted from PBLs; the CD3+ PB cell subpopulation representing T lymphocytes were isolated from the peripheral blood mononuclear cell (PBMC) fraction by magnetic-activated cell sorting (MACS) obtained from PBC patients [pre- and post-ursodeoxycholic acid (UDCA) treatment] and normal controls (NC) and multiplex reverse transcription–polymerase chain reaction (RT–PCR) for CXCR3A and B and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was performed. (a) Representative PCR blots of mRNA expression from CD3+ PBL obtained from PBC patients and NCs, probed for CXCR3A and B and GAPDH as internal control. In order to control for genomic contamination, each sample has a reverse transcriptase negative control (RT–). The gel shows fluorescence of ethidium bromide-stained PCR products resolved by electrophoresis. (b) Mean densitometry readings for PCR product of receptors are expressed as a percentage of receptors mRNA expression observed in PBC patients' samples. Each column represents mean ± standard error of the mean of the different groups.
Figure 4.
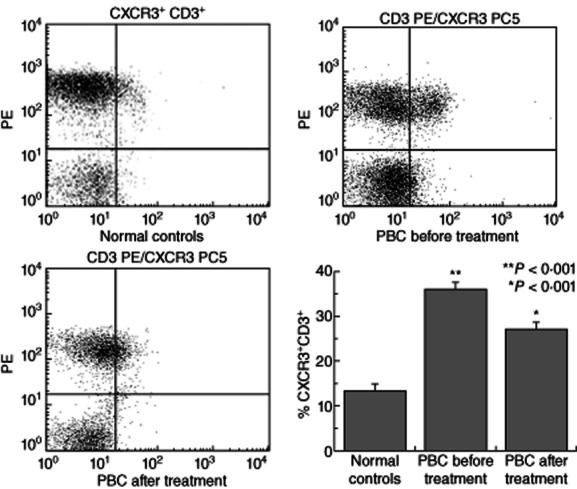
Peripheral T cells from primary biliary cirrhosis (PBC) patients are enriched preferentially in CXCR3+ cells. Peripheral T lymphocytes from PBC (n = 20) and normal controls (NC, n = 20) were isolated from peripheral blood mononuclear cells (PBMCs) via magnetic separation based on CD3 expression. The presence of CXCR3 was assessed by flow cytometry. (a–c) Representative fluorescence activated cell sorter images from normal controls and patients with PBC pre- and post-ursodeoxycholic acid (UDCA) treatment. (d) Mean ± standard error of the mean of CD3+ cells of different groups expressing the CXCR3 receptor.
Discussion
In the present study we revealed increased expression of CXCR3 binding chemokines in liver tissue combined with increased serum levels of these chemokines. CXCR3 receptor was also increased in CD3+ PBL obtained from patients with PBC. Moreover, the spliced variant of the CXCR3 was invariably present in CD3+ PBL obtained from PBC patients compared to PBL from normal controls. UDCA administration could alter the CXCR3 chemokine expression and production.
Chemokine production in liver tissue might play an important role in the migration of CXCR3+CD3+ cells into the inflamed liver of patients with PBC. Our data suggest a role for specific chemokine–chemokine receptor interactions in the pathogenesis of PBC. IP-10, MIG and I-TAC are responsible for T lymphocyte – mainly cytotoxic CD8+ and regulatory T cell (Treg) – migration in tissues. It has been demonstrated recently in patients with hepatitis C virus (HCV) infection that CXCR3high T cells were increased in the liver, correlating with the degree of inflammation [15].
A limitation of our study is the lack of assessment of CXCR3+ cells in the liver of patients and normal controls. However, Chuang et al. [16] have already demonstrated their presence in the portal tract of PBC patients. Moreover, a very recent paper has demonstrated that Tregs not only express CXCR3, but also Th17 and Tc17+ lymphocytes [17]. These findings suggest that a future detailed quantitative study is necessary to clarify the situation.
Increased serum levels of IP-10 and MIG have been reported in PBC patients, with this increase being analogous to disease stage. These chemokines are expressed in the portal areas in PBC patients, but not in NCs. CXCR3 receptor has also been reported to be increased in PBLs of PBC patients [16]. Our findings are in agreement with this study. Interestingly, increased levels of these chemokines, but not of CXCR3+ lymphocytes, were also found in first-degree relatives of PBC patients. The same group has also shown that circulating CD4+CD25+ Tregs are reduced in PBC patients and their first-degree relatives. In addition, the ratio of hepatic Tregs over hepatic CD8+ cells was also lower in PBC patients than in patients with chronic hepatitis C and autoimmune hepatitis [18]. Moreover, recent experimental evidence suggests that deficiency of Tregs results in the development of anti-mitochondrial antibodies and autoimmune cholangitis [19]. Recently, it has been suggested that CXCL10 could mediate the hepatic recruitment of Tregs expressing CXCR3 after natural killer (NK) T cell activation in mice liver [20]. More importantly, recent data suggest that CXCL10 is a pro-fibrotic factor, which participates in cross-talk between hepatocytes, hepatic stellate cells (HSC) and immune cells. NK cells seem to play an important role in controlling HSC activity and fibrosis. CXCL10 blockade may constitute a possible therapeutic intervention for hepatic fibrosis [21]. Therefore, our findings may indicate an anti-fibrotic effect of UDCA, and may also indicate an impaired migration of CXCR3B lymphocytes in the liver of PBC patients. Reduced recruitment could explain the increased CXCR3+ PBLs found in the peripheral blood of PBC patients in our study. Another possible explanation of this impaired migration could be that CXCR3+ cells might be Tregs [22,23]. This needs to be clarified in future studies.
The origin of IP-10, MIG and I-TAC is of particular interest. In atherosclerosis, IP-10 is expressed by endothelial cells, smooth muscle cells and macrophages, whereas MIG and I-TAC are expressed only in endothelial cells and macrophages. Interestingly, nitric oxide (NO) treatment decreased IP-10 induced by IFN-γ [24–26]. This finding is in agreement with already published data from our group [27].
IP-10, I-TAC and MIG are detected on liver sinusoidal endothelium [28]. Conversely, MIG and IP-10 are secreted not only by endothelial cells but also by cholangiocytes and activated stellate cells [29,30]. If so, in PBC, IP-10 and MIG might be secreted by endothelial cells and direct T cells into the portal tracts. We have hypothesized previously that the primary event in PBC is an overproduction of endothelins by liver endothelial cells, leading to ischaemic necrosis of intrahepatic bile ductules [31]. The possibility that sinusoidal endothelial cells might be the source of the increased chemokines is consistent with recently published work by Schrage et al. [32].
In this study, we have also reported a reduction of CXCR3 binding chemokines and CXCR3+ PBLs after UDCA treatment in PBC patients. Although UDCA was administered for a relatively short period of time before reassessment, there was a significant reduction. It is plausible, therefore, that the beneficial effect of UDCA in PBC is also due to chemokine reduction in liver tissue.
In conclusion, a possible model explaining our findings in PBC is that IP-10 and MIG production in liver tissue could create a gradient towards the liver for CXCR3 expressing lymphocytes to migrate into the inflamed bile ductules [33,34]. UDCA, in addition to the previously reported mechanisms [9], may also protect from lymphocyte damage on the bile ductules. This model outlines a working hypothesis for future studies.
Disclosure
There are no conflicts of interest for any of the authors listed. There was no financial or any other support.
References
- 1.Bone-Larson CL, Simpson KJ, Colletti LM, et al. The role of chemokines in the immunopathology of the liver. Immunol Rev. 2000;177:8–20. doi: 10.1034/j.1600-065x.2000.17703.x. [DOI] [PubMed] [Google Scholar]
- 2.Lalor PF, Shields P, Grant A, Adams DH. Recruitment of lymphocytes to the human liver. Immunol Cell Biol. 2002;80:52–64. doi: 10.1046/j.1440-1711.2002.01062.x. [DOI] [PubMed] [Google Scholar]
- 3.Cole KE, Strick CA, Paradis TJ, et al. Interferon-inducible T cell alpha chemoattractant (I-TAC): a novel non-ELR CXC chemokine with potent activity on activated T cells through selective high affinity binding to CXCR3. J Exp Med. 1998;187:2009–2021. doi: 10.1084/jem.187.12.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hyun JG, Lee G, Brown JB, et al. Anti-interferon-inducible chemokine, CXCL10, reduces colitis by impairing T helper-1 induction and recruitment in mice. Inflamm Bowel Dis. 2005;11:799–805. doi: 10.1097/01.mib.0000178263.34099.89. [DOI] [PubMed] [Google Scholar]
- 5.Piali L, Weber C, LaRosa G, et al. The chemokine receptor CXCR3 mediates rapid and shear-resistant adhesion-induction of effector T lymphocytes by the chemokines IP10 and Mig. Eur J Immunol. 1998;28:961–972. doi: 10.1002/(SICI)1521-4141(199803)28:03<961::AID-IMMU961>3.0.CO;2-4. [DOI] [PubMed] [Google Scholar]
- 6.Ehlert JE, Addison CA, Burdick MD, Kunkel SL, Strieter RM. Identification and partial characterization of a variant of human CXCR3 generated by posttranscriptional exon skipping. J Immunol. 2004;173:6234–6240. doi: 10.4049/jimmunol.173.10.6234. [DOI] [PubMed] [Google Scholar]
- 7.Lasagni L, Francalanci M, Annunziato F, et al. An alternatively spliced variant of CXCR3 mediates the inhibition of endothelial cell growth induced by IP-10, Mig, and I-TAC, and acts as functional receptor for platelet factor 4. J Exp Med. 2003;197:1537–1549. doi: 10.1084/jem.20021897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ueno Y, Moritoki Y, Shimosegawa T, Gershwin ME. Primary biliary cirrhosis: what we know and what we want to know about human PBC and spontaneous PBC mouse models. J Gastroenterol. 2007;42:189–195. doi: 10.1007/s00535-007-2019-y. [DOI] [PubMed] [Google Scholar]
- 9.Paumgartner G, Beuers U. Ursodeoxycholic acid in cholestatic liver disease: mechanisms of action and therapeutic use revisited. Hepatology. 2002;36:525–531. doi: 10.1053/jhep.2002.36088. [DOI] [PubMed] [Google Scholar]
- 10.Ludwig J, Dickson ER, McDonald GS. Staging of chronic nonsuppurative destructive cholangitis (syndrome of primary biliary cirrhosis) Virchows Arch A Pathol Anat Histol. 1978;379:103–112. doi: 10.1007/BF00432479. [DOI] [PubMed] [Google Scholar]
- 11.Koumaki V, Papadaki HA, Stefanaki K, et al. Increased cell apoptosis in bone marrow trephine biopsies and immunomagnetically isolated myeloid progenitor cells in patients with chronic idiopathic neutropenia. Ann Hematol. 2003;82:641–645. doi: 10.1007/s00277-003-0709-y. [DOI] [PubMed] [Google Scholar]
- 12.Kolios G, Manousou P, Bourikas L, et al. Ciprofloxacin inhibits cytokine-induced nitric oxide production in human colonic epithelium. Eur J Clin Invest. 2006;36:720–729. doi: 10.1111/j.1365-2362.2006.01710.x. [DOI] [PubMed] [Google Scholar]
- 13.Jordan NJ, Kolios G, Abbot SE, et al. Expression of functional CXCR4 chemokine receptors on human colonic epithelial cells. J Clin Invest. 1999;104:1061–1069. doi: 10.1172/JCI6685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Papadaki HA, Stamatopoulos K, Damianaki A, et al. Activated T-lymphocytes with myelosuppressive properties in patients with chronic idiopathic neutropenia. Br J Haematol. 2005;128:863–876. doi: 10.1111/j.1365-2141.2005.05380.x. [DOI] [PubMed] [Google Scholar]
- 15.Larrubia JR, Calvino M, Benito S, et al. The role of CCR5/CXCR3 expressing CD8+ cells in liver damage and viral control during persistent hepatitis C virus infection. J Hepatol. 2007;47:632–641. doi: 10.1016/j.jhep.2007.04.009. [DOI] [PubMed] [Google Scholar]
- 16.Chuang YH, Lian ZX, Cheng CM, et al. Increased levels of chemokine receptor CXCR3 and chemokines IP-10 and MIG in patients with primary biliary cirrhosis and their first degree relatives. J Autoimmun. 2005;25:126–132. doi: 10.1016/j.jaut.2005.08.009. [DOI] [PubMed] [Google Scholar]
- 17.Oo YH, Banz V, Kavanagh D, et al. CXCR3-dependent recruitment and CCR6-mediated positioning of Th-17 cells in the inflamed liver. J Hepatol. 2012;57:1044–1051. doi: 10.1016/j.jhep.2012.07.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lan RY, Cheng C, Lian ZX, et al. Liver-targeted and peripheral blood alterations of regulatory T cells in primary biliary cirrhosis. Hepatology. 2006;43:729–737. doi: 10.1002/hep.21123. [DOI] [PubMed] [Google Scholar]
- 19.Zhang W, Sharma R, Ju ST, et al. Deficiency in regulatory T cells results in development of antimitochondrial antibodies and autoimmune cholangitis. Hepatology. 2009;49:545–552. doi: 10.1002/hep.22651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Santodomingo-Garzon T, Han J, Le T, Yang Y, Swain MG. Natural killer T cells regulate the homing of chemokine CXC receptor 3-positive regulatory T cells to the liver in mice. Hepatology. 2009;49:1267–1276. doi: 10.1002/hep.22761. [DOI] [PubMed] [Google Scholar]
- 21.Hintermann E, Bayer M, Pfeilschifter JM, Luster AD, Christen U. CXCL10 promotes liver fibrosis by prevention of NK cell mediated hepatic stellate cell inactivation. J Autoimmun. 2010;35:424–435. doi: 10.1016/j.jaut.2010.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Erhardt A, Wegscheid C, Claass B, et al. CXCR3 deficiency exacerbates liver disease and abrogates tolerance in a mouse model of immune-mediated hepatitis. J Immunol. 2011;186:5284–5293. doi: 10.4049/jimmunol.1003750. [DOI] [PubMed] [Google Scholar]
- 23.Oo YH, Weston CJ, Lalor PF, et al. Distinct roles for CCR4 and CXCR3 in the recruitment and positioning of regulatory T cells in the inflamed human liver. J Immunol. 2010;184:2886–2898. doi: 10.4049/jimmunol.0901216. [DOI] [PubMed] [Google Scholar]
- 24.Mach F, Sauty A, Iarossi AS, et al. Differential expression of three T lymphocyte-activating CXC chemokines by human atheroma-associated cells. J Clin Invest. 1999;104:1041–1050. doi: 10.1172/JCI6993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Loetscher M, Gerber B, Loetscher P, et al. Chemokine receptor specific for IP10 and mig: structure, function, and expression in activated T-lymphocytes. J Exp Med. 1996;184:963–969. doi: 10.1084/jem.184.3.963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Farber JM. Mig and IP-10: CXC chemokines that target lymphocytes. J Leukoc Biol. 1997;61:246–257. [PubMed] [Google Scholar]
- 27.Dimoulios P, Kolios G, Notas G, et al. Ursodeoxycholic acid reduces increased circulating endothelin 2 in primary biliary cirrhosis. Aliment Pharmacol Ther. 2005;21:227–234. doi: 10.1111/j.1365-2036.2005.02307.x. [DOI] [PubMed] [Google Scholar]
- 28.Shields PL, Morland CM, Salmon M, Qin S, Hubscher SG, Adams DH. Chemokine and chemokine receptor interactions provide a mechanism for selective T cell recruitment to specific liver compartments within hepatitis C-infected liver. J Immunol. 1999;163:6236–6243. [PubMed] [Google Scholar]
- 29.Shetty S, Lalor PF, Adams DH. Lymphocyte recruitment to the liver: molecular insights into the pathogenesis of liver injury and hepatitis. Toxicology. 2008;254:136–146. doi: 10.1016/j.tox.2008.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Holt AP, Haughton EL, Lalor PF, Filer A, Buckley CD, Adams DH. Liver myofibroblasts regulate infiltration and positioning of lymphocytes in human liver. Gastroenterology. 2009;136:705–714. doi: 10.1053/j.gastro.2008.10.020. [DOI] [PubMed] [Google Scholar]
- 31.Kouroumalis E, Notas G. Pathogenesis of primary biliary cirrhosis: a unifying model. World J Gastroenterol. 2006;12:2320–2327. doi: 10.3748/wjg.v12.i15.2320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Schrage A, Wechsung K, Neumann K, et al. Enhanced T cell transmigration across the murine liver sinusoidal endothelium is mediated by transcytosis and surface presentation of chemokines. Hepatology. 2008;48:1262–1272. doi: 10.1002/hep.22443. [DOI] [PubMed] [Google Scholar]
- 33.Morland CM, Fear J, McNab G, Joplin R, Adams DH. Promotion of leukocyte transendothelial cell migration by chemokines derived from human biliary epithelial cells in vitro. Proc Assoc Am Physicians. 1997;109:372–382. [PubMed] [Google Scholar]
- 34.Murai M, Yoneyama H, Harada A, et al. Active participation of CCR5(+)CD8(+) T lymphocytes in the pathogenesis of liver injury in graft-versus-host disease. J Clin Invest. 1999;104:49–57. doi: 10.1172/JCI6642. [DOI] [PMC free article] [PubMed] [Google Scholar]


