Abstract
Resistance of estrogen receptor positive (ER+) breast cancer cells to tamoxifen has been linked in part to activation of (i) certain receptor tyrosine kinases, such as HER2, and (ii) the PI3K→AKT pathway. Mucin 1 (MUC1) is aberrantly overexpressed in about 90% of human breast cancers and the oncogenic MUC1-C subunit associates with ERα. The present studies using HER2 overexpressing BT-474 breast cancer cells, which are constitutively resistant to tamoxifen, demonstrate that silencing MUC1-C is associated with (i) downregulation of p-HER2 levels, and (ii) sensitivity to tamoxifen-induced growth inhibition and loss of clonogenic survival. The results also demonstate that overexpression of MUC1-C in tamoxifen-sensitive MCF-7 breast cancer cells results in upregulation of p-AKT and tamoxifen resistance. We show that MUC1-C forms complexes with ERα on the estrogen-responsive promoter of the Rab31 gene and that MUC1-C blocks tamoxifen-induced decreases in ERα occupancy. MUC1-C also attenuated tamoxifen-induced decreases in (i) recruitment of the coactivator CREB binding protein, (ii) Rab31 promoter activation, and (ii) Rab31 mRNA and protein levels. The importance of MUC1-C is further supported by the demonstration that targeting MUC1-C with the cell-penetrating peptide inhibitor, GO-203, sensitizes tamoxifen-resistant cells to tamoxifen treatment. Moreover, we show that targeting MUC1-C in combination with tamoxifen is highly synergistic in the treatment of tamoxifen-resistant breast cancer cells. These findings indicate that MUC1-C contributes to tamoxifen resistance and provide support for the investigation of MUC1-C inhibitors in the setting of tamoxifen refractory disease.
Keywords: MUC1-C, estrogen receptor α, HER2, AKT, tamoxifen, resistance
Introduction
The estrogen receptor α (ERα) signaling pathway contributes to the development and progression of human breast cancers. Over 70% of all breast cancers express ERα with a somewhat higher frequency in tumors from postmenopausal women (1). Endocrine therapy of patients with ER+ breast cancer has included (i) blocking estrogen binding with selective ER modulators, such as tamoxifen, (ii) decreasing ER expression with fulvestrant, and (iii) inhibiting estrogen synthesis with aromatase inhibitors. These endocrine therapies have had a major impact on the natural history of hormone-dependent breast cancer; however, their effectiveness is often limited by intrinsic or acquired resistance (1, 2). For example, adjuvant therapy of ER+ breast cancers with tamoxifen is associated with recurrent disease in about one-third of patients (2). One mechanism of acquired tamoxifen resistance is the downregulation of ERα expression, although this response has been observed in only 15–20% of breast cancers (3). Tamoxifen resistance has also been linked to cross-talk between ERα and receptor tyrosine kinases (RTKs), specifically epidermal growth factor receptor (EGFR), the epidermal growth factor receptor 2 (HER2/ERBB2) and the insulin-like growth factor receptor (IGF1-R) (1, 2). In this context, amplification and overexpression of HER2 has been associated with endocrine resistance (4–6). However, only about 10% of ER+ breast cancers overexpress HER2, indicating that additional mechanisms confer tamoxifen resistance in the majority of these tumors. Other work has shown that hyperactivation of the phosphatidylinositol 3-kinase (PI3K) pathway confers resistance to endocrine therapy through both direct and indirect ERα interactions (7). Accordingly, PI3K pathway inhibitors are being evaluated for the treatment of patients with tamoxifen-resistant ER+ breast cancer (8). Nonetheless, new therapeutic targets are needed for the treatment of tamoxifen-resistant disease.
The mucin 1 (MUC1) heterodimeric protein is aberrantly overexpressed in about 90% of human breast cancers (9). The two MUC1 subunits are generated by autocleavage of a single polypeptide and, in turn, form a stable non-covalent complex (9, 10). The MUC1 N-terminal subunit (MUC1-N) is the heavily glycosylated mucin component of the heterodimer. MUC1-N is positioned extracellularly in a complex at the cell membrane with the MUC1 C-terminal (MUC1-C) transmembrane subunit (10). MUC1-C functions as an oncoprotein by interacting with RTKs, such as EGFR and HER2, at the breast cancer cell surface and by contributing to their downstream signaling pathways (9). In this regard, the 72 amino acid MUC1-C cytoplasmic domain acts as a substrate for EGFR and other RTKs. The MUC1-C cytoplasmic domain also contains a YHPM motif, that when phosphorylated on tyrosine, functions as a binding site for PI3K SH2 domains and thereby activation of the PI3K→AKT pathway (11, 12). The MUC1-C subunit is, in addition, targeted to the nucleus where it interacts with certain transcription factors (10). Of relevance to breast cancer, MUC1-C associates with ERα and this interaction is stimulated by 17β-estradiol (E2) (13). MUC1-C binds directly to the ERα DNA binding domain and stabilizes ERα by blocking its ubiquitination and degradation. MUC1-C also enhances ERα promoter occupancy, increases recruitment of coactivators and stimulates ERα-mediated transcription (13). Notably, tamoxifen has no effect on MUC1-C/ERα complexes and MUC1-C antagonizes the inhibitory effects of tamoxifen on ERα-mediated transcription (13). In other studies, a MUC1-C-induced 38-gene set was applied to the analysis of a database obtained from ER+ breast cancer patients treated with tamoxifen and (i) demonstrated a strong association with ER-dependent signaling, and (ii) predicted failure to tamoxifen treatment, as measured by disease-free and overall survival (14). These findings have supported a potential link between MUC1-C and tamoxifen resistance; however, there has been no direct evidence to date for such an association.
The present studies using loss and gain of MUC1-C function demonstrate that MUC1-C is sufficient to confer tamoxifen resistance in breast cancer cells. The mechanistic basis for these results is supported by the demonstration that MUC1-C (i) contributes to HER2 and AKT activation, and (ii) blocks tamoxifen-induced decreases in ERα occupancy on an estrogen-responsive promoter. The results also demonstrate that targeting MUC1-C is synergistic with tamoxifen in the treatment of tamoxifen-resistant breast cancer cells.
Materials and Methods
Cell culture
Human HER2 overexpressing BT-474 breast cancer cells (ATCC) were grown in Dulbecco’s Modified Eagle’s Medium (DMEM)/Ham F12 medium (1:1 ratio), 10% heat-inactivated fetal bovine serum (HI-FBS), 100 µg/ml streptomycin, 100 units/ml penicillin and 2 mM L-glutamine. BT-474 cells were infected with lentiviruses expressing a MUC1 shRNA (Sigma) or a scrambled control shRNA (CshRNA; Sigma). Human MCF-7 breast cancer and 293T renal cells (ATCC) were maintained in DMEM, 10% HI-FBS, antibiotics and L-glutamine. MCF-7 cells were transfected to stably express a control pHR-CMV-GFP vector or one expressing MUC1-C. For certain experiments performed in the absence of estrogen stimulation, cells were grown in phenol red-free Iscove’s Modified Eagle’s Medium (IMEM), 10% charcoal-stripped serum (CSS), antibiotics and L-glutamine. Cells were treated with tamoxifen (TAM; Sigma-Aldrich) or 4-hydroxytamoxifen (OHTAM; Sigma-Aldrich) dissolved in DMSO and, as a control, with a corresponding dilution of DMSO. The cells were also treated with the MUC1-C inhibitor GO-203 (Genus Oncology) (12) or the PI3K inhibitor LY294002 (Sigma).
Immunoblot analysis
Cell lysates were analyzed by immunoblotting with anti-MUC1-C (15), anti-ERα (Santa Cruz Biotechnology, Cat. #SC-8005), anti-p-HER2 (Cell Signaling Technology, Cat. #D66B7), anti-HER2 (Cell Signaling Technology, Cat. #2242), anti-p-AKT (Cell Signaling Technology, Cat. #S473), anti-AKT (Santa Cruz Biotechnology, Cat. #C67E7) or anti-β-actin (Sigma, Cat. #A5441) as described (12, 13). Immune complexes were detected with horseradish peroxidase-conjugated secondary antibodies and enhanced chemiluminescence (GE Healthcare).
Colony formation assays
Cells were seeded in 6-well plates for 24 h and then left untreated or treated with inhibitor. After 7–14 d, the cells were washed and stained with 0.5% crystal violet in 25% methanol. Colonies >30 cells were counted in triplicate wells.
Chromatin immunoprecipitation (ChIP) assays
Soluble chromatin was prepared as described (16) and precipitated with anti-ERα (2 µg; Neomarkers, Cat. #TE111.5D11) or a control nonimmune IgG. For re-ChIP assays, complexes from the initial ChIP were eluted and reprecipitated with anti-MUC1-C (Neomarkers, Cat. #HM-1630-PABX) as described (16). For PCR, 2 µl from a 50 µl DNA sample was used with Rab31 promoter primers (16) and 25–35 cycles of amplification. Fold enrichment was calculated as described (17).
Promoter-reporter assays
Control pGL3 or pRab31-Luc constructs (16) were transfected into cells with the Renilla plasmid in the presence of Superfect (Qiagen). Luciferase activity was measured using the Promega Dual Glo kit as described (16).
qRT-PCR
Total RNA was isolated from cells using an RNeasy Mini kit (Qiagen). cDNAs were synthesized from RNA using the first-strand cDNA synthesis kit (Invitrogen) as described (16). The SYBR green qPCR assay kit (Applied Biosystems) was used with 5 µl of 20-fold diluted cDNA. The samples were amplified with the ABI Prism 7300 machine (Applied Biosystems). Rab31 and GAPDH primers used for qRT-PCR are listed in Supplemental Table S1.
Results
Silencing MUC1-C confers sensitivity of BT-474 cells to tamoxifen treatment
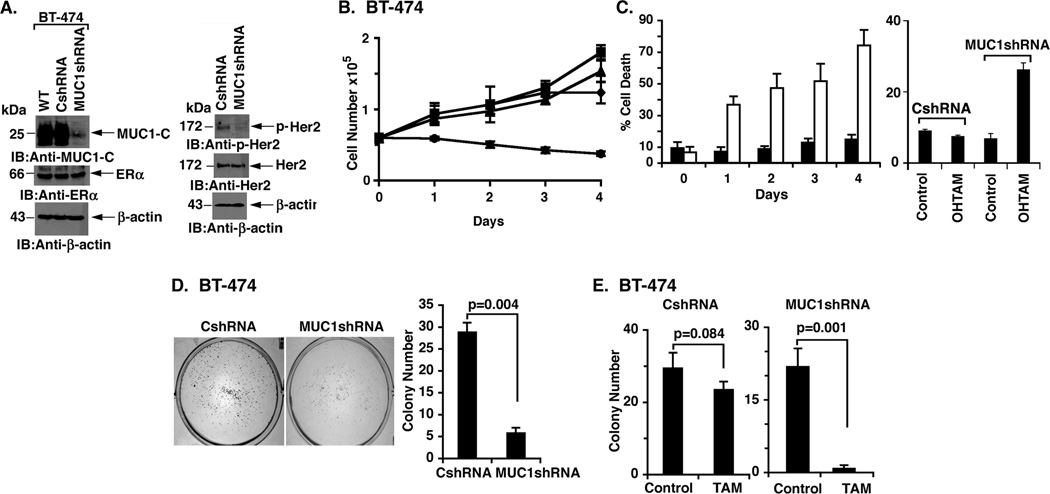
BT-474 breast cancer cells overexpress HER2, are ER positive and are resistant to tamoxifen (18, 19). Immunoblot analysis further demonstrated that BT-474 cells express MUC1-C (Fig. 1A, left). To determine whether MUC1-C plays a role in tamoxifen resistance, we transduced cells with a lentiviral vector expressing a control scrambled shRNA (CshRNA) or one expressing a MUC1 shRNA (Fig. 1A, left). Compared to wild-type (WT) BT-474 cells and those stably expressing the CshRNA, there was downregulation of MUC1-C in the cells expressing the MUC1shRNA (Fig. 1A, left). As a control, the partial silencing of MUC1-C had little if any effect on ERα levels (Fig. 1A, left). MUC1 interacts with HER2 and promotes HER2-mediated signaling (20, 21). In this context, partial silencing of MUC1-C in BT-474 cells was associated with downregulation of p-HER2 and no detectable effect on HER2 levels (Fig. 1A, right). With regard to tamoxifen resistance, growth of BT-474 and BT-474/CshRNA cells was unaffected by the addition of tamoxifen as compared to that obtained with untreated cells (Fig. 1B). By contrast, proliferation of BT-474/MUC1shRNA cells was partially slowed as compared to BT-474/CshRNA cells and was clearly further inhibited by tamoxifen treatment (Fig. 1B). To exclude an off-target effect of the MUC1shRNA, we infected BT-474 cells with a lentivirus expressing another MUC1 shRNA, designated MUC1shRNA(#2). Studies of BT-474/MUC1shRNA(#2) cells also demonstrated (i) downregulation of MUC1 abundance (Supplemental Fig. S1A, left), (ii) decreases in p-HER2 levels (Supplemental Fig. S1A, right), and (iii) tamoxifen-induced growth inhibition (Supplemental Fig. S1B), confirming the effects of silencing MUC1 on reversal of tamoxifen resistance. In concert with these results, BT-474/MUC1shRNA cells exhibited a marked loss of viability in response to tamoxifen as compared to that obtained for BT-474/CshRNA cells (Fig. 1C, left). Moreover, similar results were obtained when the cells were treated with 4-hydroxytamoxifen (OHTAM), the active metabolite of tamoxifen (Fig. 1C, right). Plating efficiency of BT-474/MUC1shRNA cells was also significantly decreased compared to BT-474/CshRNA cells (Figs. 1D, left and right). As expected, tamoxifen had little if any effect on the ability of BT-474/CshRNA to form colonies (Fig. 1E, left). Notably, however, tamoxifen treatment was associated with a marked decrease in BT-474/MUC1shRNA cell colony formation (Fig. 1E, right). These findings indicate that MUC1-C contributes to tamoxifen resistance in BT-474 cells.
Figure 1. Resistance of HER2-overexpressing BT-474 cells to tamoxifen is conferred by MUC1-C expression.
A. Lysates from wild-type (WT) BT-474 cells, BT-474/CshRNA and BT-474/MUC1shRNA cells were immunoblotted with the indicated antibodies (left and right). B. Control BT-474/CshRNA (squares) and BT-474/MUC1shRNA (diamonds) cells were left untreated. BT-474/CshRNA (triangles) and BT-474/MUC1shRNA (circles) were also treated with 5 µM tamoxifen on days 0 and 2. Cell number is expressed as the mean±SD of three replicates. C. BT-474/CshRNA (solid bars) and BT-474/MUC1shRNA (open bars) cells were treated with 5 µM tamoxifen (left) or 10 nM OHTAM (right) on days 0 and 2. The results (mean±SD of three replicates) are expressed as the percentage of cell death as determined by trypan blue staining on the indicated days (left) or on day 4 (right). D. BT-474/CshRNA and BT-474/MUC1shRNA cells were seeded at 1000 cells/well (6-well plate), grown for 14 days and then stained with crystal violet (left). Colony number (>30 cells) is expressed as the mean±SD of three replicates (right). E. BT-474/CshRNA (1000 cells/well; left) and BT-474/MUC1shRNA (2000 cells/well; right) cells were seeded in 6-well plates and left untreated (Control) or treated with 5 µM tamoxifen (TAM) every other day for 14 days. Colony number (>30 cells) is expressed as the mean±SD of three replicates.
Overexpression of the MUC1-C subunit confers resistance of MCF-7 cells to tamoxifen
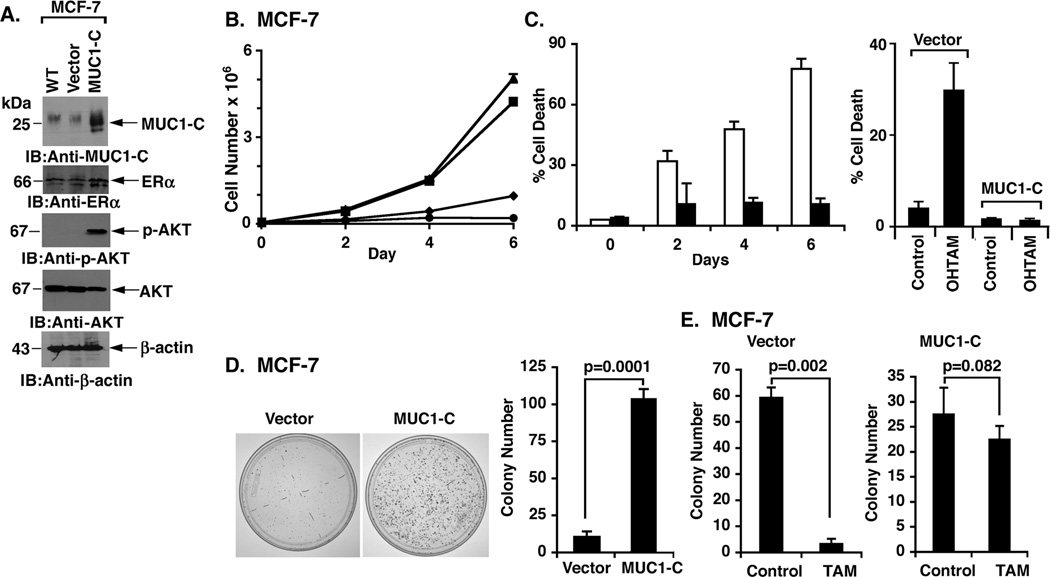
In contrast to BT-474 cells, MCF-7 breast cancer cells are ER+ and sensitive to tamoxifen. To extend the analysis of MUC1-C involvement in tamoxifen resistance, MCF-7 cells were stably transfected with a control vector or one expressing MUC1-C (Fig. 2A). Overexpression of MUC1-C resulted in a modest increase in ERα levels (Fig. 2A). In addition and consistent with the reported effects of MUC1-C on upregulation of the PI3K→AKT pathway (11, 12), overexpression of MUC1-C in MCF-7 cells was associated with a marked induction of p-AKT activation (Fig. 2A). Growth of MCF-7/vector cells was inhibited by tamoxifen (Fig. 2B). Significantly, however, tamoxifen treatment had little effect on proliferation of MCF-7/MUC1-C cells (Fig. 2B). The MCF-7/MUC1-C cells were also less sensitive to tamoxifen- and OHTAM-induced loss of viability as compared to MCF-7/vector cells (Figs. 2C, left and right). Plating efficiency of MCF-7/MUC1-C cells was substantially increased compared to MCF-7/vector cells (Figs. 2D, left and right). Moreover, tamoxifen was effective in decreasing formation of MCF-7/vector cell colonies (Fig. 2E, left), but not clonogenic survival of MCF-7/MUC1-C cells (Fig. 2E, right). To determine whether activation of AKT contributes to resistance of MCF-7/MUC1-C cells to tamoxifen, we incubated these cells with a sub-lethal concentration of the PI3K inhibitor, LY294002 (22). As expected, treatment with LY294002 was associated with downregulation of p-AKT levels (Supplemental Fig. S2A). We also found that LY294002 partially reversed the sensitivity of MCF-7/MUC1-C cells to tamoxifen (Supplemental Fig. S2B). These findings indicate that overexpression of MUC1-C in MCF-7 cells confers tamoxifen resistance and that this effect is conferred in part by AKT activation.
Figure 2. Overexpression of MUC1-C in MCF-7 cells is associated with tamoxifen resistance.
A. Lysates from wild-type (WT) MCF-7 cells, MCF-7/vector and MCF-7/MUC1-C cells were immunoblotted with the indicated antibodies. B. Control MCF-7/vector (diamonds) and MCF-7/MUC1-C (triangles) cells were left untreated. MCF-7/vector (circles) and MCF-7/MUC1-C (squares) cells were also treated with 5 µM tamoxifen on days 0, 2 and 4. Cell number is expressed as the mean±SD of three replicates. C. MCF-7/vector (open bars) and MCF-7/MUC1-C (solid bars) cells were treated with 5 µM tamoxifen on days 0, 2 and 4 (left) or 10 nM OHTAM on days 0 and 2 (right). The results (mean±SD of three replicates) are expressed as the percentage of cell death as determined by trypan blue staining on the indicated days (left) or on day 4 (right). D. MCF-7/vector and MCF-7/MUC1-C cells were seeded at 500 cells/well (6-well plate), grown for 7 days and then stained with crystal violet (left). Colony number (>30 cells) is expressed as the mean±SD of three replicates (right). E. MCF-7/vector (2000 cells/well; left) and MCF-7/MUC1-C (500 cells/well; right) cells were seeded in 6-well plates and left untreated (Control) or treated with 5 µM tamoxifen (TAM) every other day for 7 days. Colony number (>30 cells) is expressed as the mean±SD of three replicates.
MUC1-C confers MCF-7 cell growth in the absence of estrogen
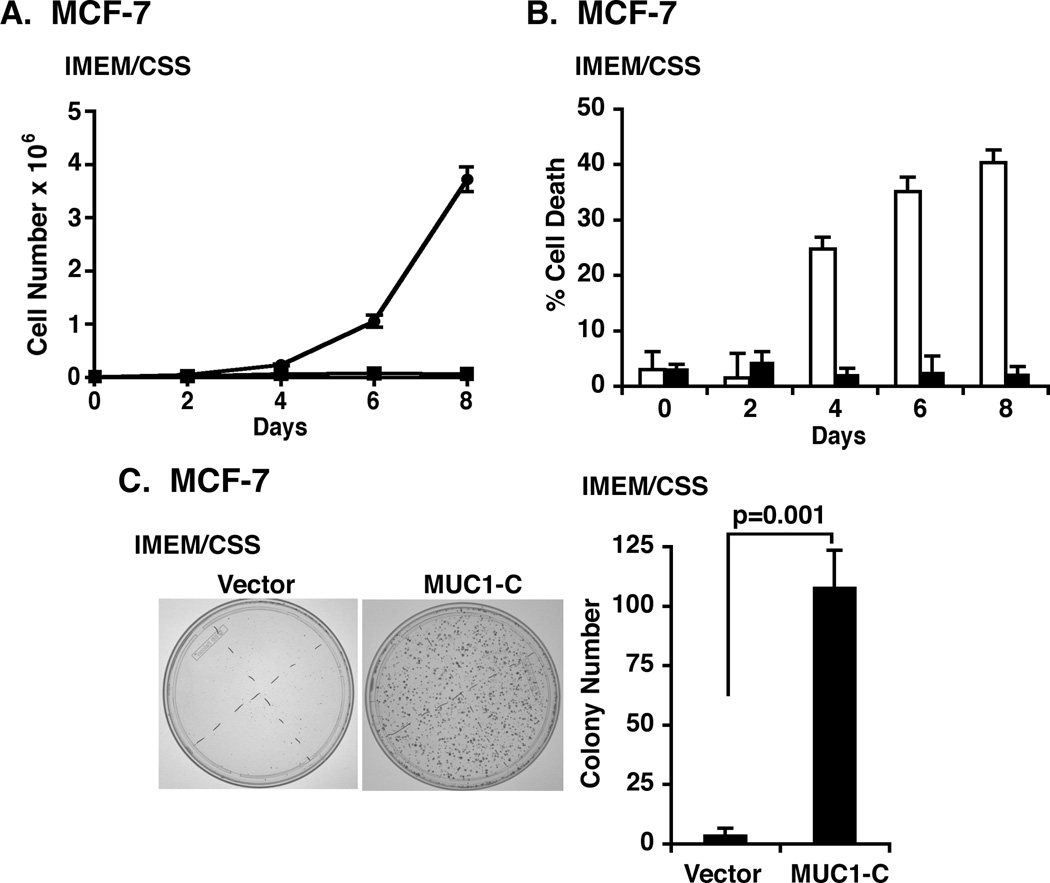
Growth of MCF-7 cells is dependent on estrogen (23). The demonstration that MUC1-C confers resistance to tamoxifen prompted studies to determine whether MCF-7/MUC1-C cells are also estrogen-independent. Culture of MCF-7/vector cells in estrogen-depleted IMEM/CSS medium was associated with inhibition of growth (Fig. 3A). Strikingly, however, proliferation of MCF-7/MUC1-C cells was readily apparent in the setting of estrogen depletion (Fig. 3A). Loss of MCF-7/vector cell viability in the absence of estrogen stimulation was also abrogated by MUC1-C overexpression (Fig. 3B). In addition, the plating efficiency of MCF-7/MUC1-C cells in IMEM/CSS medium was substantially greater than that found for MCF-7/vector cells (Figs. 3C, left and right). These findings demonstrate that overexpression of MUC1-C in MCF-7 cells confers estrogen independence.
Figure 3. MCF-7 cells overexpressing MUC1-C are estrogen-independent.
A. MCF-7/vector (squares) and MCF-7/MUC1-C (circles) were seeded at 1 × 104 cells/ml in IMEM/CSS medium for the indicated number of days. The results are expressed as the cell number × 106/ml (mean±SD of three replicates). B. MCF-7/vector (open bars) and MCF-7/MUC1-C (solid bars) cells were seeded in IMEM/CSS medium for the indicated number of days. The results are expressed as the percentage of cell death (mean±SD of three replicates) as determined by trypan blue staining. C. MCF-7/vector and MCF-7/MUC1-C cells were seeded at 500 cells/well (6-well plate), grown for 7 days in IMEM/CSS medium and then stained with crystal violet (left). Colony number (>30 cells) is expressed as the mean±SD of three replicates (right).
MUC1-C abrogrates effects of tamoxifen on ERα-mediated gene transcription
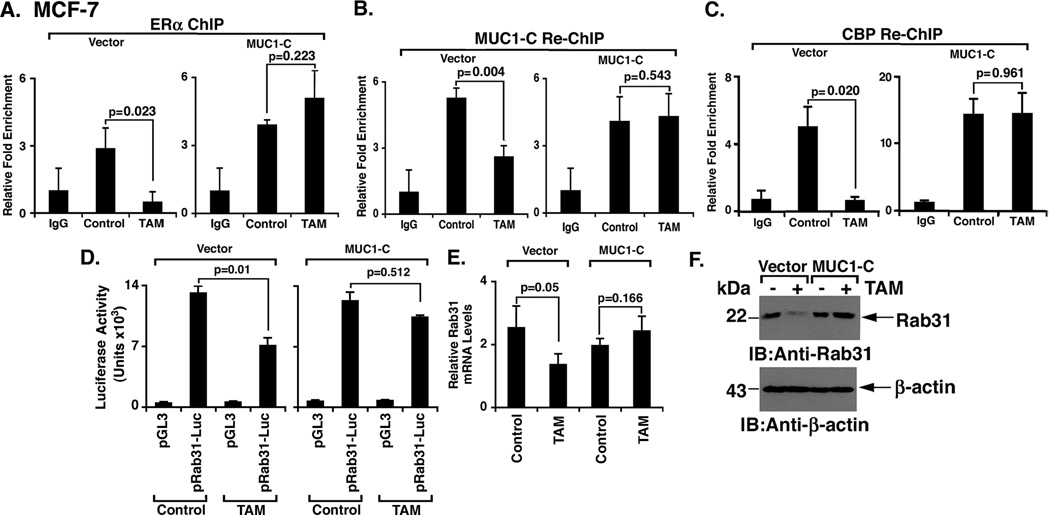
To study the effects of MUC1-C on the response of an estrogen-responsive gene to tamoxifen, we first examined ERα and MUC1-C occupancy of the Rab31 promoter by ChIP analysis (16). In this context, recent work showed that MUC1-C forms a complex with ERα on the Rab31 promoter and activates Rab31 gene transcription in an estrogen-dependent manner (16). Tamoxifen treatment of MCF-7/vector and MCF-7/MUC1-C cells had little if any effect on ERα levels (Supplemental Fig. S3). However, in MCF-7/vector cells, tamoxifen treatment was associated with decreased ERα occupancy of the Rab31 promoter (Fig. 4A, left). By contrast, tamoxifen treatment of MCF-7/MUC1-C cells had no apparent effect on ERα occupancy (Fig. 4A, right). In re-ChIP studies, occupancy of the Rab31 promoter by ERα and MUC1-C was also decreased by tamoxifen in MCF-7/vector (Fig. 4B, left), but not MCF-7/MUC1-C, cells (Fig. 4B, right). The CREB binding protein (CBP) is a histone acetyltransferase that is recruited to ligand-activated, DNA-bound ERα, and enhances ERα-mediated gene transcription (24, 25). Recruitment of CBP to the Rab31 promoter in MCF-7/vector cells was decreased by tamoxifen treatment (Fig. 4C, left); however, tamoxifen had no significant effect on CBP occupancy in MCF-7/MUC1-C cells (Fig. 4C, right). To extend these results, we studied activation of the Rab31 promoter using a Rab31 promoter-luciferase reporter construct. Tamoxifen treatment was associated with a decrease in Rab31 promoter activity in MCF-7/vector, but not in MCF-7/MUC1-C, cells (Fig. 4D). In concert with these results, tamoxifen-induced downregulation of Rab31 mRNA levels as observed in MCF-7/vector cells was attenuated in MCF-7/MUC1-C cells (Fig. 4E). Moreover, Rab31 protein was decreased by tamoxifen treatment of MCF-7/vector, but not MCF-7/MUC1-C, cells (Fig. 4F). These findings demonstrate that MUC1-C blocks the inhibitory effects of tamoxifen on ERα occupancy, CBP recruitment and Rab31 promoter activity.
Figure 4. MUC1-C blocks the effects of tamoxifen on occupancy and activation of the Rab31 promoter.
A–C. MCF-7/vector (left) and MCF-7/MUC1-C (right) cells were left untreated (Control) or treated with 5 µM tamoxifen for 2 days. A. Soluble chromatin was precipitated with anti-ERα or a control IgG. The final DNA samples were analyzed for Rab31 promoter estrogen-responsive element (ERE) or control region (CR) sequences (16). The results (mean+SD of three determinations) are expressed as the relative fold enrichment compared to that obtained with the IgG control. B and C. In re-ChIP experiments, the anti-ERα precipitates were released, reimmunoprecipitated with anti-MUC1-C (B) or anti-CBP (C) and a control IgG, and then analyzed for Rab31 promoter sequences. The results (mean±SD of three determinations) are expressed as the relative fold enrichment compared to that obtained with the IgG control. D. MCF-7/vector and MCF-7/MUC1-C cells were transfected with the pGL3 vector or pRab31-Luc and Renilla plasmid as an internal control. The cells were then left untreated (Control) or treated with 5 µM tamoxifen for 2 days and then assayed for luciferase activity. The results are expressed as the mean±SD of two experiments each performed with three determinations. E. MCF-7/vector and MCF-7/MUC1-C cells were left untreated (Control) or treated with 5 µM tamoxifen for 2 days. Rab31 and GAPDH mRNA levels were determined by qRT-PCR. The results (mean±SD of three determinations) are expressed as relative Rab31 mRNA levels as compared to that obtained for GAPDH. F. MCF-7/vector and MCF-7/MUC1-C cells were left untreated (Control) or treated with 5 µM tamoxifen for 2 days. Lysates from the indicated cells were immunoblotted with anti-Rab31 and anti-β-actin.
Tamoxifen-resistant breast cancer cells are sensitive to MUC1-C inhibition
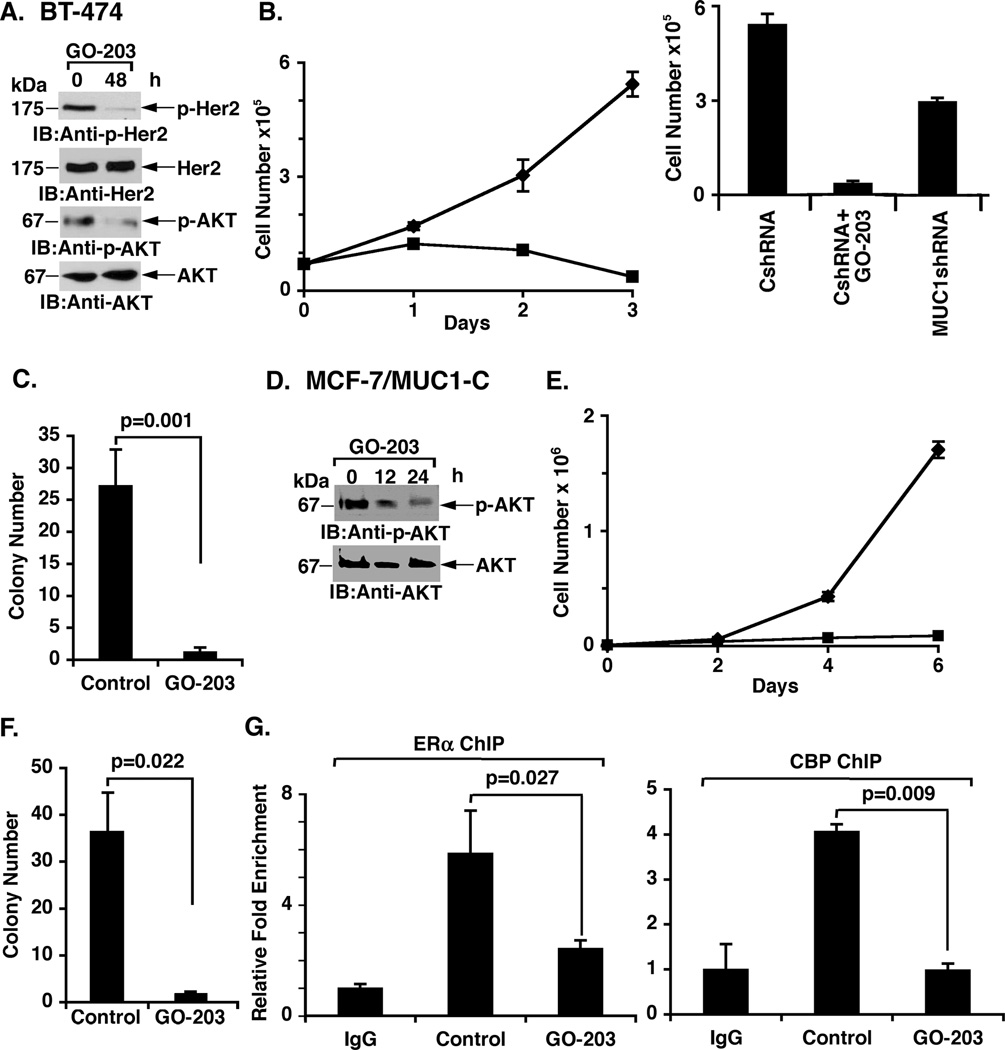
The finding that MUC1-C confers tamoxifen resistance invoked the possibility that MUC1-C inhibitors could be effective in tamoxifen-resistant cells. Accordingly, we treated BT-474 cells with the MUC1-C inhibitor GO-203, a cell-penetrating D-amino acid peptide ([R]9-CQCRRKN)(12). GO-203 treatment of BT-474 cells was associated with marked downregulation of p-HER2 and p-AKT (Fig. 5A). In addition, treatment of BT-474 cells with GO-203 was associated with marked inhibition of growth (Fig. 5B, left). The effects of GO-203 on BT-474 growth were more pronounced than that obtained with partial silencing of MUC1-C in these cells (Fig. 5B, right), consistent with the potential of GO-203 to effectively block MUC1-C homodimerization and thereby function (26). As found with silencing of MUC1 in BT-474 cells, GO-203 treatment also resulted in a substantial loss of clonogenic survival (Fig. 5C). By contrast, GO-203 had little if any effect on survival of MUC1-negative 293T cells (Supplemental Fig. S4). GO-203 treatment of MCF-7/MUC1-C cells also inhibited AKT activation (Fig. 5D), growth (Fig. 5E) and colony formation (Fig. 5F). Moreover, inhibition of MUC1-C with GO-203 decreased recruitment of ERα and CBP to the Rab31 promoter (Fig. 5G). These results indicate that tamoxifen-resistant BT-474 and MCF-7/MUC1-C cells are dependent on MUC1-C function for their growth and survival.
Figure 5. Tamoxifen-resistant cells are sensitive to MUC1-C inhibition.
A. BT-474 cells were left untreated (Control) or treated with 5 µM GO-203 each day for 2 days. Lysates were immunoblotted with the indicated antibodies. B. BT-474/CshRNA cells were left untreated (diamonds) or treated with 5 µM GO-203 (squares) each day for the indicated days (left). The results (mean±SD of three replicates) are expressed as the viable cell number. BT-474/CshRNA and BT-474/MUC1shRNA cells were seeded at 5 × 104 cells/well and left untreated (right). The BT-474/CshRNA cells were also treated with 5 µM GO-203 each day for 3 days. The results (mean±SD of three replicates) are expressed as the viable cell number on day 4. C. BT-474/CshRNA cells were seeded at 1000 cells/well in 6-well plates and left untreated (Control) or treated with 5 µM GO-203 each day for 7 days. Colony number (>30 cells) is expressed as the mean±SD of three replicates. D. MCF-7/MUC1-C cells were left untreated (Control) or treated with 5 µM GO-203 each day for 2 days. Lysates were immunoblotted with the indicated antibodies. E. MCF-7/MUC1-C cells were left untreated (diamonds) or treated with 5 µM GO-203 (squares) each day for the indicated days. The results (mean±SD of three replicates) are expressed as the viable cell number. F. MCF-7/MUC1-C cells were seeded at 200 cells/well in 6-well plates and left untreated (Control) or treated with 5 µM GO-203 each day for 7 days. Colony number (>30 cells) is expressed as the mean±SD of three replicates. G. MCF-7/MUC1-C cells were left untreated (Control) or treated with 5 µM GO-203 each day for 2 days. Soluble chromatin was precipitated with anti-ERα (left), anti-CBP (right) or a control IgG. The precipitates were analyzed for Rab31 promoter sequences. The results (mean±SD of three determinations) are expressed as the relative fold enrichment compared to that obtained with the IgG control.
Synergy between GO-203 and tamoxifen in tamoxifen-resistant breast cancer cells
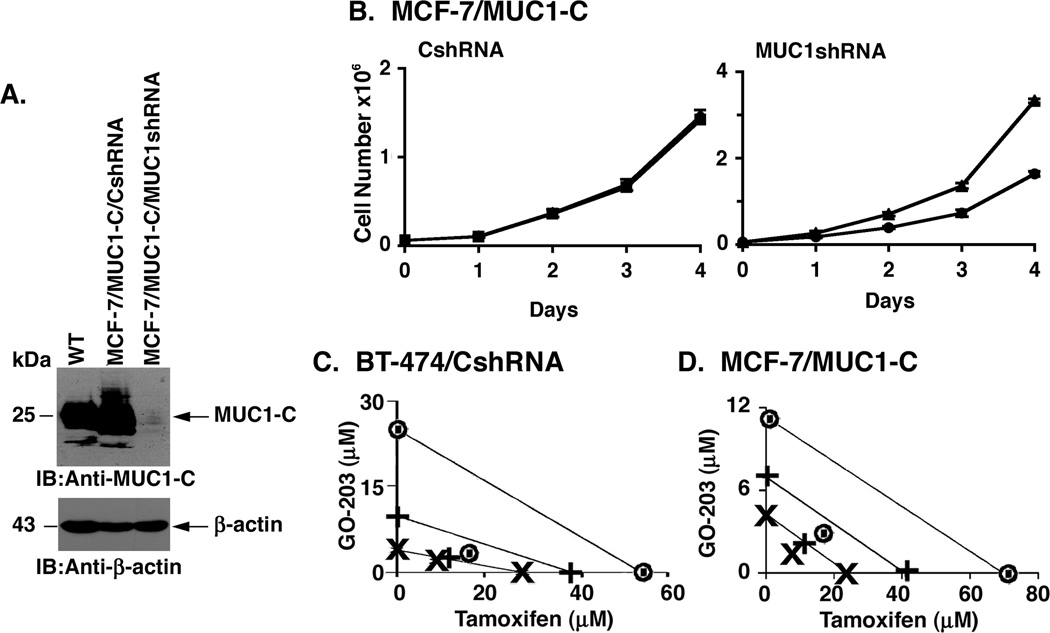
Our above findings demonstrate that silencing MUC1-C in BT-474 cells results in tamoxifen sensitivity. To further substantiate that MUC1-C confers tamoxifen resistance, we silenced MUC1-C in MCF-7/MUC1-C cells (Fig. 6A). Notably, the MCF-7/MUC1-C/MUC1shRNA cells regained sensitivity to tamoxifen treatment (Fig. 6B). These results suggested that targeting MUC1-C in tamoxifen-resistant cells could potentiate tamoxifen treatment. To address this line of reasoning, we used the Chou-Talalay method for evaluating drug combinations (27, 28). For the tamoxifen-resistant BT-474/CshRNA cells, we had to select a concentration of tamoxifen for the combination studies. Accordingly, we used the IC50 (16.6 µM) obtained for the tamoxifen-sensitive BT-474/MUC1shRNA cells, based on the reasoning that targeting MUC1-C in BT-474 cells reverses tamoxifen resistance. Using the half-maximal inhibitory concentration for GO-203 (3.9 µM), GO-203 and tamoxifen were tested alone for their effects on BT-474/CshRNA cell growth at 1/8X, 1/4X, 1/2X, 1X, 2X and 4X the IC50 values. GO-203 and tamoxifen were also tested at equipotent concentrations at the same ratios in combination. Isobologram analysis at the ED50, ED75 and ED90 values demonstrated synergy for the GO-203/tamoxifen combination (Fig. 6C) with combination indices (CIs) of less than 1 (ED50=0.81; ED75=0.56, ED90=0.43). A synergistic interaction between GO-203 and tamoxifen with CIs of <1 (ED50=0.79; ED75=0.57; ED90=0.50) was also observed in the tamoxifen-sensitive BT-474/MUC1shRNA cells (Supplemental Fig. S5A). In assessing the combined effects of GO-203 and tamoxifen against MCF-7/vector cells, we found that the activity of GO-203 and tamoxifen is synergistic at the ED50 (CI=0.69) and ED75 (CI=0.88), and additive at the ED90 (CI=1.12) (Supplemental Fig. S5B). To assess these drug interactions in the tamoxifen-resistant MCF-7/MUC1-C cells, we used the half-maximal inhibitory concentration identified for tamoxifen in the treatment of MCF-7/vector cells, based on the demonstration that targeting MUC1-C in MCF-7/MUC1-C cells reverses tamoxifen resistance. Under these experimental conditions, a synergistic interaction between GO-203 and tamoxifen was observed for MCF-7/MUC1-C cells with CIs of <1 (ED50=0.65; ED75=0.56; ED90=0.49) (Fig. 6D). These results and those obtained with BT-474 cells demonstrate that GO-203 and tamoxifen are synergistic in the treatment of both tamoxifen-sensitive and -resistant cells.
Figure 6. Synergistic interaction between GO-203 and tamoxifen.
A. MCF-7/MUC1-C cells were infected with lentiviruses expressing the control shRNA (CshRNA) or the MUC1shRNA. Lysates from wild-type (WT) MCF-7 cells, MCF-7/MUC1-C/CshRNA and MCF-7/MUC1-C/MUC1shRNA cells were immunoblotted with the indicated antibodies. B. MCF-7/MUC1-C/CshRNA (left) and MCF-7/MUC1-C/MUC1shRNA (right) cells were left untreated (triangles) or treated (circles) with 5 µM tamoxifen days 0 and 2. The results (mean±SD of three replicates) are expressed as viable cell number. C and D. BT-474/CshRNA (C) and MCF-7/MUC1-C (D) cells were treated with fixed IC50 ratios of (i) GO-203 alone on days 0, 1, 2, 3 and 4, (ii) tamoxifen alone on days 0, 2 and 4 and (iii) the GO-203/tamoxifen combination. For tamoxifen-resistant BT-474/CshRNA and MCF-7/MUC1-C cells, tamoxifen was used at the half-maximal inhibitory concentrations obtained for the tamoxifen-sensitive BT-474/MUC1shRNA and MCF-7/vector cells, respectively. The multiple effect-level isobologram analyses are shown for the ED50 (×), ED75 (+) and ED90 (◉) values.
Discussion
The overexpression of HER2 in breast cancers has been linked to tamoxifen resistance (4). The present studies provide evidence that the MUC1-C oncoprotein promotes resistance to tamoxifen in the HER2 overexpressing BT-474 breast cancer cell model. Previous work had shown that MUC1-C forms a complex with HER2 and contributes to heregulin-induced downstream signals (20, 21). In concert with those findings, silencing MUC1-C in BT-474 cells was associated with downregulation of p-HER2 levels. Moreover, silencing MUC1-C and thereby suppressing HER2 activation reversed the resistance of BT-474 cells to tamoxifen, consistent with cross-talk between HER2 signaling and the ER pathway. In further support of a role for MUC1-C in resistance of HER2-overexpressing BT-474 cells to tamoxifen, we found that treatment with the MUC1-C inhibitor, GO-203, suppresses HER2 activation and confers sensitivity to tamoxifen-induced inhibition of growth and colony formation. GO-203 disrupts MUC1-C homodimerization and blocks the interaction of MUC1-C with HER2 at the cell membrane (9). In this way, silencing MUC1-C or blocking its function with an inhibitor results in HER2 downregulation and reversal of tamoxifen resistance. In addition to HER2, activation of EGFR and IGF1-R can confer tamoxifen resistance (2). Moreover, like EGFR and IGF1-R, HER2 activates downstream signals that confer phosphorylation of ER and can result in tamoxifen-mediated activation or ligand-independence (1, 6, 29). Activation of pathways downstream to these RTKs can also contribute to tamoxifen resistance (2). For example, signaling by the PI3K→AKT→mTOR pathway as a consequence of HER2 overexpression or loss of PTEN can regulate responsiveness to tamoxifen (6, 30–33). However, the precise mechanisms that confer tamoxifen resistance have not been fully defined and may involve activation of mitogenic and anti-apoptotic pathways (1, 2).
Our studies further demonstrate that overexpression of MUC1-C in MCF-7 cells induces tamoxifen resistance. MCF-7 cells constitutively express MUC1; however, MUC1-C levels are not sufficient to activate the PI3K→AKT pathway. Indeed, as has been shown in other cell types (11, 12), overexpression of MUC1-C in MCF-7 cells was associated with marked upregulation of AKT activation. In this way, MUC1-C interacts directly with PI3K through binding of PI3K SH2 domains to a consensus pYHPM motif in the MUC1-C cytoplasmic domain and activates the PI3K→AKT pathway (11, 12, 34). The PI3K pathway is hyperactivated in response to the development of resistance to estrogen deprivation (23). In addition, activation of PI3K signaling has been linked to antiestrogen resistance in breast cancer cells (7). In concert with these findings, MCF-7 cells that overexpress MUC1-C were found to be resistant to estrogen deprivation. Moreover, MUC1-C overexpression was sufficient to confer resistance to tamoxifen-induced loss of proliferation and clonogenic survival. Notably, treatment of MCF-7/MUC1-C cells with the MUC1-C inhibitor GO-203 was associated with a block in AKT activation, consistent with MUC1-C function in activating the PI3K→AKT pathway. Treatment with GO-203 or the PI3K→AKT inhibitor LY294002 was also associated with reversal of MUC1-C-induced tamoxifen resistance. These findings and those obtained in the BT-474 model of HER2 overexpression indicate that MUC1-C is sufficient to confer tamoxifen resistance by contributing, at least in part, to signaling pathways, such as AKT, that have been linked to ER activity. Other models of tamoxifen resistance have been described that are induced by chronic tamoxifen exposure (35). As such, further studies will be needed to determine whether MUC1-C plays a role in this setting of induced tamoxifen resistance.
Previous findings have demonstrated that MUC1-C binds directly to the ERα DNA binding domain and associates with ERα on estrogen-responsive promoters (13). MUC1-C was also found to enhance ERα promoter occupancy and increase recruitment of coactivators (13). In the present work, we studied the effects of MUC1-C on ERα occupancy in the response to tamoxifen treatment. MUC1-C forms a complex with ERα on the ERα-responsive Rab31 promoter and activates Rab31 gene transcription in an estrogen-dependent manner (16). Treatment with tamoxifen was associated with a decrease in ERα occupancy on the Rab31 promoter and this response was blocked by overexpression of MUC1-C. Tamoxifen competes with estrogen for binding to ERα and induces changes in ERα conformation that block recruitment of coactivators (36, 37). In this context, tamoxifen treatment was associated with decreases in recruitment of CBP to the Rab31 promoter and this response was also attenuated by a MUC1-C-dependent mechanism. MUC1-C contributes to the availability of ERα/E2 complexes for occupancy of estrogen-responsive elements (13). In addition, through a direct interaction with ERα and increasing ERα/E2 complexes, MUC1-C attenuates the competitive effects of tamoxifen on estrogen binding (13). These findings further indicated that overexpression of MUC1-C promotes the transcription of ER-dependent genes and thereby survival of ER+ breast cancer cells (13). Thus, the available evidence indicates that, in concert with MUC1-C-induced regulation of the HER2 and AKT pathways, binding of MUC1-C to ERα may also contribute to attenuating the effects of tamoxifen.
The MUC1-C cytoplasmic domain contains a CQC motif that is necessary for its homodimerization and nuclear localization (38). Accordingly, cell penetrating peptides and small molecules have been developed to block the CQC dimerization motif (39, 40). In this way, treatment with MUC1-C inhibitors abrogates the formation of MUC1-C homodimers and thereby MUC1-C function in breast cancer cells (26, 39). The present work demonstrates that treatment of tamoxifen-resistant BT-474 cells with the MUC1-C inhibitor, GO-203, is associated with inhibition of growth and loss of clonogenicity, supporting a lack of cross-resistance to targeting MUC1-C. MCF-7 cells with induced resistance to tamoxifen as a result of overexpressing MUC1-C were also sensitive to GO-203 treatment. These findings invoked the possibility that MUC1-C-induced tamoxifen resistance could be reversed by targeting MUC1-C. Tamoxifen-resistant cells were therefore treated with GO-203 in combination with tamoxifen. The demonstration that GO-203 and tamoxifen are highly synergistic against tamoxifen-resistant cells provided further support that MUC1-C is of importance to tamoxifen resistance. By extension, GO-203 and tamoxifen were also found to be synergistic in the treatment of tamoxifen-sensitive cells. These results lend support to the concept that targeting MUC1-C could be effective in the treatment of patients with breast cancers that develop resistance to tamoxifen. A Phase I trial of GO-203 is presently underway for patients with refractory solid tumors to identify a maximum tolerated dose for Phase II studies. Based on the present findings, this agent may be a candidate for evaluation in the treatment of tamoxifen-resistant breast cancers.
Supplementary Material
Acknowledgments
Grant Support
Research reported in this publication was supported by the National Cancer Institute of the National Institutes of Health under award numbers CA97098 and CA166480.
Abbreviations
- MUC1
mucin 1
- MUC1-C
MUC1 C-terminal subunit
- MUC1-CD
MUC1 cytoplasmic domain
- ERα
estrogen receptor α
- RTK
receptor tyrosine kinase
- PI3K
phosphatidylinositol 3-kinase
- CBP
CREB binding protein
Footnotes
Conflict of Interest: D. Kufe is a founder of Genus Oncology and holds equity in the company. The other authors disclosed no potential conflicts of interest.
References
- 1.Osborne CK, Schiff R. Mechanisms of endocrine resistance in breast cancer. Annu Rev Med. 2011;62:233–247. doi: 10.1146/annurev-med-070909-182917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Musgrove EA, Sutherland RL. Biological determinants of endocrine resistance in breast cancer. Nat Rev Cancer. 2009;9(9):631–643. doi: 10.1038/nrc2713. [DOI] [PubMed] [Google Scholar]
- 3.Gutierrez MC, Detre S, Johnston S, et al. Molecular changes in tamoxifen-resistant breast cancer: relationship between estrogen receptor, HER-2, and p38 mitogen-activated protein kinase. J Clin Oncol. 2005;23(11):2469–2476. doi: 10.1200/JCO.2005.01.172. [DOI] [PubMed] [Google Scholar]
- 4.De Laurentiis M, Arpino G, Massarelli E, et al. A meta-analysis on the interaction between HER-2 expression and response to endocrine treatment in advanced breast cancer. Clin Cancer Res. 2005;11(13):4741–4748. doi: 10.1158/1078-0432.CCR-04-2569. [DOI] [PubMed] [Google Scholar]
- 5.Ellis MJ, Tao Y, Young O, et al. Estrogen-independent proliferation is present in estrogen-receptor HER2-positive primary breast cancer after neoadjuvant letrozole. J Clin Oncol. 2006;24(19):3019–3025. doi: 10.1200/JCO.2005.04.3034. [DOI] [PubMed] [Google Scholar]
- 6.Arpino G, Wiechmann L, Osborne CK, Schiff R. Crosstalk between the estrogen receptor and the HER tyrosine kinase receptor family: molecular mechanism and clinical implications for endocrine therapy resistance. Endocr Rev. 2008;29(2):217–233. doi: 10.1210/er.2006-0045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Miller TW, Balko JM, Arteaga CL. Phosphatidylinositol 3-kinase and antiestrogen resistance in breast cancer. J Clin Oncol. 2011;29(33):4452–4461. doi: 10.1200/JCO.2010.34.4879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Miller TW, Balko JM, Fox EM, et al. ERalpha-dependent E2F transcription can mediate resistance to estrogen deprivation in human breast cancer. Cancer Discov. 2011;1(4):338–351. doi: 10.1158/2159-8290.CD-11-0101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kufe D. MUC1-C oncoprotein as a target in breast cancer: activation of signaling pathways and therapeutic approaches. Oncogene. 2012 doi: 10.1038/onc.2012.158. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kufe D. Mucins in cancer: function, prognosis and therapy. Nature Reviews Cancer. 2009;9(12):874–885. doi: 10.1038/nrc2761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Raina D, Kharbanda S, Kufe D. The MUC1 oncoprotein activates the anti-apoptotic PI3K/Akt and Bcl-xL pathways in rat 3Y1 fibroblasts. J Biol Chem. 2004;279:20607–20612. doi: 10.1074/jbc.M310538200. [DOI] [PubMed] [Google Scholar]
- 12.Raina D, Kosugi M, Ahmad R, et al. Dependence on the MUC1-C oncoprotein in non-small cell lung cancer cells. Mol Cancer Therapeutics. 2011;10(5):806–816. doi: 10.1158/1535-7163.MCT-10-1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wei X, Xu H, Kufe D. MUC1 oncoprotein stabilizes and activates estrogen receptor α. Mol Cell. 2006;21(2):295–305. doi: 10.1016/j.molcel.2005.11.030. [DOI] [PubMed] [Google Scholar]
- 14.Pitroda S, Khodarev N, Beckett M, Kufe D, Weichselbaum R. MUC1-induced alterations in a lipid metabolic gene network predict response of human breast cancers to tamoxifen treatment. Proc Natl Acad Sci USA. 2009;106:5837–5841. doi: 10.1073/pnas.0812029106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Panchamoorthy G, Rehan H, Kharbanda A, Ahmad R, Kufe D. A monoclonal antibody against the oncogenic mucin 1 cytoplasmic domain. Hybridoma. 2011;30(6):531–535. doi: 10.1089/hyb.2011.0070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jin C, Rajabi H, Pitroda S, et al. Cooperative interaction between the MUC1 oncoprotein and the Rab31 GTPase in human breast cancer cells. PLoS One. 2012;7(7):e39432. doi: 10.1371/journal.pone.0039432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ahmad R, Rajabi H, Kosugi M, et al. MUC1-C oncoprotein promotes STAT3 activation in an auto-inductive regulatory loop. Science Signaling. 2011;4:ra9. doi: 10.1126/scisignal.2001426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chung Y, Sheu M, Yang S, Lin C, Yen S. Resistance to tamoxifen-induced apoptosis is associated with direct interaction between Her2/neu and cell membrane estrogen receptor in breast cancer. Int J Cancer. 2002;97:306–312. doi: 10.1002/ijc.1614. [DOI] [PubMed] [Google Scholar]
- 19.Ross-Innes CS, Stark R, Teschendorff AE, et al. Differential oestrogen receptor binding is associated with clinical outcome in breast cancer. Nature. 2012;481(7381):389–393. doi: 10.1038/nature10730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li Y, Yu W-H, Ren J, et al. Heregulin targets γ-catenin to the nucleolus by a mechanism dependent on the DF3/MUC1 protein. Mol Cancer Res. 2003;1:765–775. [PubMed] [Google Scholar]
- 21.Ren J, Bharti A, Raina D, Chen W, Ahmad R, Kufe D. MUC1 oncoprotein is targeted to mitochondria by heregulin-induced activation of c-Src and the molecular chaperone HSP90. Oncogene. 2006;25(1):20–31. doi: 10.1038/sj.onc.1209012. [DOI] [PubMed] [Google Scholar]
- 22.Liu P, Cheng H, Roberts TM, Zhao JJ. Targeting the phosphoinositide 3-kinase pathway in cancer. Nat Rev Drug Discov. 2009;8(8):627–644. doi: 10.1038/nrd2926. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Miller TW, Hennessy BT, Gonzalez-Angulo AM, et al. Hyperactivation of phosphatidylinositol-3 kinase promotes escape from hormone dependence in estrogen receptor-positive human breast cancer. J Clin Invest. 2010;120(7):2406–2413. doi: 10.1172/JCI41680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Acevedo ML, Kraus WL. Mediator and p300/CBP-steroid receptor coactivator complexes have distinct roles, but function synergistically, during estrogen receptor alpha-dependent transcription with chromatin templates. Mol Cell Biol. 2003;23(1):335–348. doi: 10.1128/MCB.23.1.335-348.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Jaber BM, Mukopadhyay R, Smith CL. Estrogen receptor-alpha interaction with the CREB binding protein coactivator is regulated by the cellular environment. J Mol Endocrinol. 2004;32(1):307–323. doi: 10.1677/jme.0.0320307. [DOI] [PubMed] [Google Scholar]
- 26.Raina D, Ahmad R, Rajabi H, Panchamoorthy G, Kharbanda S, Kufe D. Targeting cysteine-mediated dimerization of the MUC1-C oncoprotein in human cancer cells. Int J Oncol. 2012;40:1643–1649. doi: 10.3892/ijo.2011.1308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chou TC, Talalay P. Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul. 1984;22:27–55. doi: 10.1016/0065-2571(84)90007-4. [DOI] [PubMed] [Google Scholar]
- 28.Chou TC. Drug combination studies and their synergy quantification using the Chou-Talalay method. Cancer Res. 2010;70(2):440–446. doi: 10.1158/0008-5472.CAN-09-1947. [DOI] [PubMed] [Google Scholar]
- 29.Shou J, Massarweh S, Osborne CK, et al. Mechanisms of tamoxifen resistance: increased estrogen receptor-HER2/neu cross-talk in ER/HER2-positive breast cancer. J Natl Cancer Inst. 2004;96(12):926–935. doi: 10.1093/jnci/djh166. [DOI] [PubMed] [Google Scholar]
- 30.Faridi J, Wang L, Endemann G, Roth RA. Expression of constitutively active Akt-3 in MCF-7 breast cancer cells reverses the estrogen and tamoxifen responsivity of these cells in vivo. Clin Cancer Res. 2003;9(8):2933–2939. [PubMed] [Google Scholar]
- 31.deGraffenried LA, Friedrichs WE, Fulcher L, et al. Eicosapentaenoic acid restores tamoxifen sensitivity in breast cancer cells with high Akt activity. Ann Oncol. 2003;14(7):1051–1056. doi: 10.1093/annonc/mdg291. [DOI] [PubMed] [Google Scholar]
- 32.deGraffenried LA, Friedrichs WE, Russell DH, et al. Inhibition of mTOR activity restores tamoxifen response in breast cancer cells with aberrant Akt Activity. Clin Cancer Res. 2004;10(23):8059–8067. doi: 10.1158/1078-0432.CCR-04-0035. [DOI] [PubMed] [Google Scholar]
- 33.Riggins RB, Schrecengost RS, Guerrero MS, Bouton AH. Pathways to tamoxifen resistance. Cancer Lett. 2007;256(1):1–24. doi: 10.1016/j.canlet.2007.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jin C, Rajabi H, Rodrigo C, Porco J, J A, D K. Targeting the eIF4A RNA helicase blocks translation of the MUC1-C oncoprotein. Oncogene. 2012 Jun 11; doi: 10.1038/onc.2012.236. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ring A, Dowsett M. Mechanisms of tamoxifen resistance. Endocr Relat Cancer. 2004;11(4):643–658. doi: 10.1677/erc.1.00776. [DOI] [PubMed] [Google Scholar]
- 36.Brzozowski AM, Pike AC, Dauter Z, et al. Molecular basis of agonism and antagonism in the oestrogen receptor. Nature. 1997;389(6652):753–758. doi: 10.1038/39645. [DOI] [PubMed] [Google Scholar]
- 37.Shang Y, Hu X, DiRenzo J, Lazar MA, Brown M. Cofactor dynamics and sufficiency in estrogen receptor-regulated transcription. Cell. 2000;103(6):843–852. doi: 10.1016/s0092-8674(00)00188-4. [DOI] [PubMed] [Google Scholar]
- 38.Leng Y, Cao C, Ren J, et al. Nuclear import of the MUC1-C oncoprotein is mediated by nucleoporin Nup62. J Biol Chem. 2007;282(27):19321–19330. doi: 10.1074/jbc.M703222200. [DOI] [PubMed] [Google Scholar]
- 39.Raina D, Ahmad R, Joshi M, et al. Direct targeting of the MUC1 oncoprotein blocks survival and tumorigenicity of human breast carcinoma cells. Cancer Res. 2009;69(12):5133–5141. doi: 10.1158/0008-5472.CAN-09-0854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhou J, Rajabi H, Kufe D. MUC1-C oncoprotein is a target for small molecule inhibitors. Mol Pharm. 2011;79(5):886–893. doi: 10.1124/mol.110.070797. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.