Abstract
Immunological memory has long considered to be harbored in B cells that express high affinity class-switched IgG. IgM-positive memory B cells can also be generated following immunization, although their physiological role has been unclear. Here we show that bacterial infection elicited a relatively large population of IgM memory B cells that were uniquely identified by their surface expression of CD11c, CD73, and PD-L2. The cells lacked expression of cell surface markers typically expressed by GC B cells, were CD138-negative, and did not secrete antibody ex vivo. The population was also largely quiescent, and accumulated somatic mutations. The IgM memory B cells were located in the region of the splenic marginal zone, and were not detected in blood or other secondary lymphoid organs. Generation of the memory cells was CD4 T cell-dependent, and required IL-21R signaling. In vivo depletion of the IgM memory B cells abrogated the IgG recall responses to specific antigen challenge, demonstrating that the cell population was required for humoral memory, and underwent class switch recombination following antigen encounter. Our findings demonstrate that T cell-dependent IgM memory B cells can be elicited at high frequency, and can play an important role in maintaining long-term immunity during bacterial infection.
Introduction
Immunological memory is a fundamental concept that is key to generating and maintaining immunity to pathogens, and for mediating the protection afforded by vaccines (1, 2). Humoral memory resides in part in antigen-specific memory B cells, which are classically defined as class-switched, somatically-mutated, long-lived cells that are highly responsive to specific antigen challenge (2–7). Class-switched Ig (swIg) memory B cells are generated in germinal centers (GCs), specialized anatomic structures in secondary lymphoid organs where T cell-dependent affinity maturation and class switch recombination of the BCR occurs. Following exposure to cognate antigen, memory B cells proliferate and differentiate into antibody secreting cells (ACSs). The resulting increase in antigen-specific serum Ig aids in the clearance of pathogens from the host (1, 8).
Despite the focus on swIg memory B cells, several studies have shown memory B cell populations to be more diverse than originally envisioned (9). Early studies indicated that IgM-positive memory B cells could be found both humans and mice (10–15). More recent studies have validated the existence of IgM memory B cells, and have demonstrated distinct functions for both IgM and IgG memory B cell subsets. For example, Dogan et al. used an elegant model for the unbiased labeling of antigen-experienced/memory B cells, wherein activation-induced cytidine deaminase (AID)-expressing cells were permanently marked following Cre recombinase regulation of a reporter gene (16). In that study, mice immunized with a particulate antigen, SRBCs, generated both IgG and IgM memory B cells. Following secondary encounter with antigen, the IgM memory B cells initiated a GC reaction, and generated swIg cells, as well as additional IgM memory B cells. In contrast, the IgG memory B cells differentiated directly into ASCs. In other studies, Tomayko and colleagues, using a transgenic mouse model of (4-hydroxy-3-nitrophenyl)-acetyl chicken γ-globulin (NP-CGG) immunization, demonstrated the presence of several swIg and IgM memory B cell subsets that expressed different levels of the maturation markers CD80, PD-L2, and CD73 (17). The varied expression of cell surface markers, and the distinct ontogeny of each subset suggested functional differences between IgM and swIg memory B cells. Pape et al. (18) utilized an antigen-based technique to purify rare antigen-specific memory B cells in a model of PE immunization, and demonstrated that both IgG and IgM memory B cells were generated following immunization. Kinetic analyses of the memory B cell populations revealed the IgM memory subset to be longer lived than the swIg memory subset. As was also reported by Dogan et al., the swIg memory B cells gave rise to ASCs upon antigenic challenge. In contrast to swIg memory B cells, IgM memory B cells were unresponsive to antigen challenge in immune hosts. However, upon transfer into naïve hosts, the IgM memory B cells initiated GC formation, and underwent class-switch recombination when challenged with specific antigen (18). Together, these studies have challenged traditional views of humoral memory by revealing that long-term humoral memory can be retained in IgM memory B cells. Nevertheless, questions regarding the origin, generation, and function of IgM memory B cells remain. Moreover, it has not been demonstrated whether IgM memory B cells are elicited naturally during infections. In the present study, we have identified a population of IgM memory B cells that were elicited in a murine model of human ehrlichiosis. We demonstrate that these cells can be uniquely identified by their expression of CD11c, CD73, and other cell surface markers, and that they require CD4 T cell-mediated helper functions for their generation. Furthermore, we show that these IgM memory B cells are responsible for secondary IgG responses following antigen challenge, and propose that they are correlates of IgM memory B cells identified in humans.
Materials and Methods
Animals
Mice were obtained from the Jackson Laboratory (Bar Harbor, ME), or were bred in the Animal Care Facility at the Wadsworth Center, under microisolater conditions. C57BL/6 mice, and the following transgenic and gene-targeted strains were used: B cell-deficient (B6.129S2-Ighmtm1Cgn/J), CD11c-DTR (B6.FVB-TgItgax-DTR/EGFP57Lan/J), IL-21R-deficient (19), and MHCII-deficient (B6.129S2-H2dlAb1-Ea/J).
Bacterial Infections
Mice were infected with Ehrlichia muris, by i.p. injection, between 6 and 12 weeks of age, with 50,000 bacteria obtained from infected mouse splenocytes, as previously described (20, 21). Mice were infected with the Wisconsin isolate (strain HM543745; ref. 22) as a 10% spleen preparation.
Flow cytometry
Splenocytes were obtained by mechanical disruption in HBSS. The tissues were disaggregated using a 70-μm pore size nylon strainer, and erythrocytes were removed from the suspension by hypotonic lysis. For cell surface staining, single-cell suspensions (4×106 cells) were incubated with Fc blocking solution (1μg/ml anti-CD16/CD32; clone 2.4G2 in 10% normal goat serum/HBSS/0.1% sodium azide), prior to staining with the mAbs. The cells were incubated on ice for 20 minutes, washed twice, and analyzed. Unstained cells were used to establish the flow cytometer voltage settings, and single-color positive controls were used to adjust compensation. Data from stained samples were acquired on a FACSCalibur flow cytometer with Cell Quest software (BD Biosciences, San Jose, CA), and were analyzed with FlowJo software (Tree Star, Inc. Ashland, OR). The antibodies used for flow cytometry included the following: BAFF-R (clone 7H22), BrdU (PRB-1) CD11b (M1/70), CD22 (2D6), CD21/35 (eBio8D9), CD23 (B3B4), CD38 (90), CD40 (1C10), CD49d (R1–2), CD95 (15A7), ICOS-L (HK5.3), IL-21R (4A9), TACI (8F10-3), and IgD (11–26; all from eBiociences, San Diego, CA); and B220 (RA3-6B2), CD19 (1D3), CD11c (HL3), CD62L (MEL-14), CD73 (TY/23), CD80 (16-10A1), CD86 (GL-1), CD138 (281–2), CXCR4 (2B11/CXCR4), GL7, IAb (AF6-120.1), and IgM (R6-60.2; all from BD Biosciences).
ELISA
Antibody titers were determined by ELISA, using purified recombinant E. muris OMP-19 (23). Goat anti-mouse antibodies conjugated to alkaline phosphatase (Southern Biotec, Birmingham, AL) were used to determine the serum titers of IgM, IgG, and the sub-classes of IgG.
Flow cytometric cell sorting
CD11c-positive B cells were identified using PerCP-conjugated CD19 and allophycocyanin (APC)-conjugated CD11c. The CD11c-positive and -negative B cell population were purified using a FACSAria cell sorter equipped with FACSDiva software (BD Biosciences, Santa Cruz, CA); the purity of the sorted populations was greater than 90% in all experiments.
Antibiotic Treatment
Doxycycline treatment was performed as previously described (Racine et al., 2011). E. muris infected mice were administered doxycycline for a two week period (3 times per week; 400 μg/mouse; Sigma-Aldrich, St. Louis, MO). During the treatment period, doxycycline was also provided in drinking water at a concentration of 200μg/ml.
BrdU administration and analysis
Mice were administered BrdU (0.8 mg) by i.p. injection, and were maintained on BrdU in their drinking water (0.8 mg/mL, plus 10% dextrose) for 4 days. BrdU incorporation was assessed by intracellular flow cytometric analysis.
Somatic Mutation Analyses
CD11c-positive and -negative B cells were purified by flow cytometric cell sorting from splenocytes, obtained from 3 mice after day 30 post-infection. Genomic DNA was isolated using DNAzol (Molecular Research Center, Cincinnati, OH). Recombined variable regions of the immunoglobulin heavy chain from CD11c-positive and -negative samples were amplified by nested PCR (Pfu polymerase; Agilent Technologies, Santa Clara, CA) in four separate PCR reactions, using genomic DNA obtained 5,000 cells. Amplified products were pooled, sub-cloned using a TOPO PCR cloning kit (Life Technologies, Grand Island, NY), and sequenced. Ig gene sequence mutation analyses were performed only on the Vh segments. IgBlast (http://www.ncbi.nlm.nih.gov/igblast/) was used to identify germ line V genes with the highest homology for mutation comparison; the Vh region within 4bp of the D segment junction was not included. The following oligonucleotide primers were utilized: 1st round, 5′-ACACAGGACCTCACCATG-3′ and 5′TCACAAGAGTCCGATAGACC-3′ for 12 cycles (98 °C for 10 sec, 60 °C for 30 sec, 72 °C 1.5 min); 2nd round, 5′-GGGTGACAATGACATCCA-3′ and 5′-GAGGAGACTGTGAGAGTGGTGCC-3′ for 18 cycles (98 °C for 10 sec, 62 °C for 30 sec, 72 °C for 30 sec; refs 24, 25).
Immunofluorescence Microscopy
Spleens harvested from E. muris-infected mice were embedded in Optimal Cutting Temperature Compound (OCT; Sakura, Torrence, CA). The tissues were frozen in slurry of dry ice and isopentane, and 7μm cryosections were generated. The cryosections were fixed in 100% ice cold ethanol, blocked in Fc blocking solution, and stained with the following antibodies: PE-CF594-conjugated CD19 (ID3, BD Biosciences, Santa Cruz, CA), Alexa 488-conjugated CD11c (N418, BioLegend, San Diego, CA), Alexa 647-conjugated CD169 (MOMA-1, AbD Serotec, Raleigh, NC), and Alexa 647-conjugated peanut agglutinin (PNA; from Arachis hypogaea, Life Technologies, Santa Clara, CA). Stained sections were mounted with ProLong Gold with DAPI (Life Technologies). Images were captured with a 20x objective on the Nikon TE2000 equipped with a CoolSNAP HQ CCD camera (Roper Scientific, Germany), and ImagePro software (Media Cybernetics, Rockville, MD), and Adobe Photoshop was used to process the images for display.
Antigen Challenge
Mice were administered 100μg purified recombinant OMP-19 (23, 26), with or without alum (Imject; Thermo Fisher Scientific, Waltham, MA).
Generation of bone marrow chimeras
C57BL/6 mice were lethally irradiated (1000 RADs, administered in 2 doses, 4 hours apart), and were reconstituted with 2 × 106 bone marrow cells obtained from B cell-deficient and CD11c-DTR mice. The mice were allowed to recover for at least 6 weeks prior to infection.
Toxin Administration
Bone marrow chimeric mice were administered diphtheria toxin from Corynebacterium diphtheriae (Sigma-Aldrich), at a concentration of 9ng/g of body weight, on days -1 and 6 post-challenge.
Statistical analysis
Statistical analyses were performed with a Student’s t-test, or Mann-Whitney test, using Prism GraphPad Software (LaJolla, CA); a P value of < 0.05 was considered to be significant.
Results
CD19hi CD11c-positive B cells are elicited during E. muris infection
The ehrlichiae are a group of tick-borne obligate intracellular rickettsiae that cause a wide range of diseases in humans and animals. Ehrlichia muris causes a non-fatal acute infection in immunocompetent mice, characterized by splenomegaly and hematological abnormalities (20). Thereafter, the bacterium establishes a low-level chronic infection in immunocompetent mice, and infected mice are resistant to secondary infection. Mice infected with E. muris are also immune to lethal infection by a virulent heterologous ehrlichia (27, 28). Our previous studies demonstrated that acute E. muris infection elicits a large population of extrafollicular T cell-independent B220lo splenic plasmablasts that can be identified in part by their unique expression of CD11c (29). The plasmablasts secrete IgM, and decline in frequency and number by about day 18 post-infection. During the course of this study, we also identified a distinct CD19hi B220+ CD11c-positive cell population in the spleen that emerged within approximately 30 days post-E. muris infection (Figure 1a). The CD11c-positive B cells were detected only in the spleen, and persisted for at least 397 days. The population ranged in frequency from 1.9 to 5.1 % of splenic mononuclear cells (average 3.3%) and represented as many as 8.5 × 106 cells per spleen on day 63 post-infection (Figure 1b). We also addressed whether a recently isolated human ehrlichial isolate (Ehrlichia sp. Wisconsin HM543745; 22) generated a similar B cell population. A phenotypically identical CD11c-positive B cell population was detected in mice infected with the related human pathogen (Figure 1c).
Figure 1. Identification of a long-term splenic B cell population elicited by bacterial infection.
(A) Representative flow cytometry analysis of CD11c expression on CD19-positive B cells in the spleens of E. muris-infected, and uninfected mice. The data in the plots were generated after gating on viable cells, as determined by forward and side scatter parameters. (B) Kinetic analysis of CD11c-positive B cells in E. muris infected mice. The frequency (top panel) and number (bottom panel) of CD11c-positive B cells among total splenocytes are shown. Each datum represents an individual mouse, and horizontal lines represent the mean. The asterisks indicate statistical significance, where * p< 0.01 and ** p < 0.001, relative to the values obtained on day 0. (C) Representative flow cytometry analysis of CD11c-expressing B cells from E. muris infected mice, and from mice infected with a recently identified human isolate (Ehrlichia sp. Wisconsin HM543745), analyzed 49 days post-infection. (D) Representative flow cytometry analyses of CD11c-positive and –negative B cells obtained from E. muris infected mice. (E) Frequency and number of splenic CD11c-positive B cells in untreated, or antibiotic-treated E. muris-infected mice. The mice were treated with doxycycline for two weeks, starting on day 30 post-infection. The splenocytes were analyzed approximately one month following antibiotic treatment. The dot plots are representative of two experiments of similar design.
Flow cytometric analyses revealed that the CD11c-positive B cells were larger and more granular than their CD11c-negativecounterparts (Figure 1d). Although nearly all of the cells expressed IgM, approximately equal portions of the CD11c-positive B cell population were positive and negative for IgD expression. The cells also exhibited high surface expression of IgM and CD23, and low expression of CD21, consistent with a follicular B cell origin. The CD11c-positive B cells were not a canonical GC population, as they exhibited low surface expression of GL7, and high expression of CD38.
We next evaluated the role of chronic infection in the maintenance of the CD11c-positive B cell population. Because the ehrlichiae are susceptible to doxycycline, infected mice were treated weekly with the antibiotic for a two-week period, starting on day 30 post-infection. This protocol has been shown to effectively reduce E. muris infection, and reduce or eliminate clinical signs of disease (30). On day 77 post-infection, the CD11c-positive B cells were found to persist in antibiotic-treated mice. However, the cells were reduced in frequency and number by approximately 2-fold, suggesting that low-level chronic infection may help to maintain the CD11c-positive B cells (Figure 1e). Together, these data identify a long-term pathogen-specific, splenic B cell population elicited by bacterial infection.
CD11c-positive B cells exhibit characteristics of memory B cells
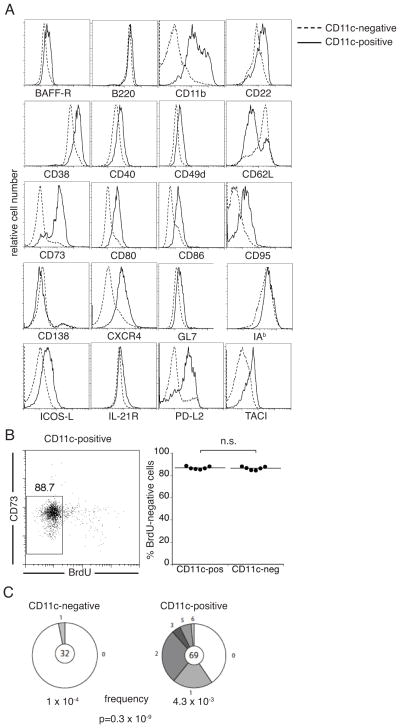
The longevity of the population suggested that the CD11c-expressing B cells were an IgM memory B cell population. To resolve this question, we first addressed whether the CD11c-positive B cells expressed cell surface markers previously shown to be associated with memory B cells. Compared with CD11c-negative B cells, CD11c-positive B cells exhibited high expression of CD38, CD73, CD80, CD95, and PD-L2, all cell surface markers previously identified on memory B cells (17, 31–33; Figure 2a). The expression of these markers, as measured by mean fluorescence intensity (MFI), was significantly higher on the CD11c-positive B cells than their CD11c-negative counterparts (Table I). Most of the CD11c-positive B cells also lacked CD138 expression, consistent with the observation that they did not secrete antibodies ex vivo. Other B cell surface antigens expressed by the CD11c-positive B cells included the B-cell activating factor-R (BAFF-R), B220, CD22, CD40, CD49d, CD62L, CXCR4, IAb, ICOS-L, IL-21R and TACI (transmembrane activator and calcium modulator ligand interactor). Surface marker expression on the CD11c-positive B cell population was largely homogenous, with the exception of CD62L, which was expressed in a bimodal distribution.
Figure 2. The CD11c-positive B cells exhibit characteristics of memory cells.
(A) Representative flow cytometry analysis of CD11c-positive and -negative CD19+ B cells during E. muris infection. The histograms are representative of 5–9 mice that were analyzed at several time-points on or after day 30 post-infection. Basal cell surface staining and voltage settings were established using unstained cells; the data were omitted from the histograms for clarity. Cells obtained from uninfected mice exhibited similar cell surface marker expression as the CD11c-negative B cells obtained from infected mice (data not shown). (B) BrdU incorporation studies of splenic CD11c-positive and -negative B cell from E. muris infected mice. The mice were administered BrdU over a 4 day period, beginning on day 63 post-infection. The splenocytes were analyzed for BrdU incorporation at day 67 post-infection; the cells were also analyzed for CD73 expression, which improved population discrimination. The data are representative of 2 experiments. (C) Mutation analysis of the V region heavy chain J558 family genes from flow cytometrically-purified CD11c-negative, and –positive B cells obtained from infected mice. The segment sizes in the pie charts are proportional to the number of sequences carrying the number of mutations indicated in the periphery of the chart. The number of independent sequences analyzed is indicated in the center of the chart. The mutation frequencies, per base pair, are indicated below the pie charts. The data were compiled from two experiments. Statistical significance was determined using a two-tailed student’s T test, assuming unequal variance, by comparing the CD11c-negative and -positive B cells; the p value is indicated. A single mutation was identified in 9,396 bp from 32 clones obtained from the CD11c-negative B cells, and 87 mutations were identified in 20,247 bp from 69 analyzed clones from the CD11c-positive B cells.
Table I.
Cell Surface Marker Expression
| Surface Marker | B Cell Subset
|
Fold Change | |
|---|---|---|---|
| CD11c-positive | CD11c-negative | ||
| BAFF-R | 21.5 +/− 1.8 | 12.2 +/− 1.0 | 1.8*** |
| CD11b | 246.3 +/− 57.9 | 7.9 +/− 1.0 | 31.0 ** |
| CD19 | 325.6 +/− 15.4 | 213.3 +/− 13.1 | 1.5*** |
| CD22 | 102.7 +/− 48.0 | 96.6 +/− 29.7 | 1.0 |
| CD38 | 2338.2 +/− 627.3 | 1147.7 +/− 74.2 | 1.35* |
| CD40 | 42.8 +/− 4.3 | 32.4 +/− 3.9 | 1.3** |
| CD49d | 33.6 +/− 4.2 | 17.4 +/− 2.5 | 1.9** |
| CD73 | 55.8 +/− 3.9 | 11.0 +/− 1.1 | 5.0*** |
| CD80 | 112.8 +/− 6.4 | 36.1 +/− 4.1 | 2.9*** |
| CD86 | 36.4 +/− 3.1 | 12.0 +/− 1.3 | 2.7*** |
| CD95 | 29.8 +/− 1.0 | 8.29 +/− 1.0 | 3.6*** |
| CXCR4 | 53.6 +/− 11.1 | 12.7 +/− 4.9 | 3.3*** |
| ICOS-L | 18.8 +/− 3.9 | 10.8 +/− 2.0 | 1.7*** |
| PD-L2 | 65.4 +/− 14.2 | 16.3 +/− 5.8 | 4.0*** |
| TACI | 25.3 +/− 3.9 | 8.2 +/− 1.8 | 3.7*** |
The data indicated the mean fluorescent intensity, as determined by flow cytometry plus or minus standard deviation; the data were obtained from the studies described in Figure 2.
The asterisks indicate statistical significance, where
p<0.05,
p<0.005,
p<0.0005.
Multiple studies have demonstrated that memory lymphocytes undergo limited cell division (32, 34, 35). Therefore, we next investigated whether the CD11c-positive B cell population was proliferating in vivo. Infected mice were administered BrdU, and were maintained on BrdU in drinking water over a 4-day period. The CD11c-positiveB cell population was largely quiescent, as the majority of the cells in the population did not incorporate BrdU over the 4-day period (mean frequency of BrdU-negative cells = 86% +/− 1.4; Figure 2b). Thus, the cell surface phenotype and cell proliferation studies provided support for the hypothesis that the CD11c-positive B cells were a population of relatively quiescent IgM memory B cells.
A third criterion we used to establish that the CD11c-positive B cells were memory B cells was somatic mutation, as this is a hallmark of antigen-experienced memory B cells (11). CD11c-positive B cells were purified, and recombined V(D)J regions from the relatively abundant variable heavy chain (Vh) J558 family were analyzed. The J558 family represents the largest Vh gene family, comprising nearly 40% of all such exons; this family was chosen because it provided a representation of the hypermutation dynamics of the CD11c-positive B cell population. The majority of segments that were characterized in the CD11c-positive B cells (41 of 69 clones) contained at least one mutation, and the genes exhibited an overall mutation frequency of 4.3 x10−3 mutations/bp. In contrast, CD11c-negative B cell μ chain genes contained only one mutation in 32 clones, an overall frequency of 1.1×10−4 mutations/bp, a frequency indistinguishable from the PCR incorporation error rate. The mutation frequencies identified in the CD11c-positive B cells are consistent with other reports of mutation frequencies in IgM memory B cells (16, 18, 32).
T cells are required for the generation of CD11c-positive B cells
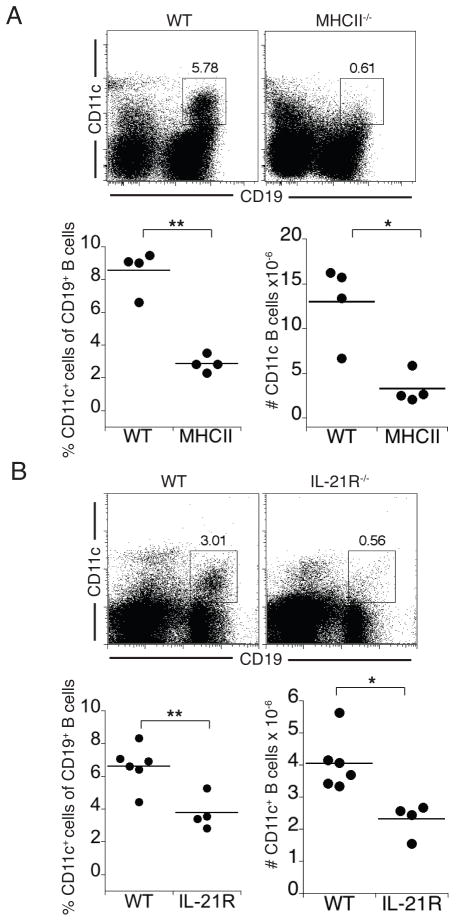
GCs are major sites of T-dependent antibody responses in secondary lymphoid organs, and it is in these specialized structures that most antibody affinity maturation occurs. Memory B cells are considered to be the product of the GC reaction, and as such, are T cell-dependent (36). IgM memory B cells may provide an exception, as at least a subset of these cells have been reported to undergo GC-independent development (37–39), and because affinity maturation can, under some conditions, occur independent of GCs (40–42). CD4 T cells are important for the development of GC-derived memory B cells, as is the cytokine IL-21 (43–46). Therefore, we next addressed the requirement for these factors in the generation of the CD11c-positive B cell population. The frequency and number of CD11c-positive B cells as a proportion of total B cells was much lower in MHCII-deficient mice, which lack CD4 T cells (Figure 3a). CD11c-positive B cells were also significantly reduced in mice deficient for the IL-21R (Figure 3b). These data indicate that CD4 T cell-dependent signals, possibly delivered in GCs, are required and/or important for the generation of the CD11c-positive B cells
Figure 3. CD4+ T cells and IL-21 signaling were required for the generation of the CD11c-positive B cell population.
(A) Flow cytometry analysis of splenic CD11c-positive B cells from wild-type and MHCII-deficient mice analyzed on day 30 post-infection is shown. The frequencies of CD11c-positive B cells in the flow cytometry dot plots represent their percentage among total spleen cells. The data in the panels below indicate the frequency of CD11c-positive cells among total splenic B cells. The average B cell frequency was 67% and 37% in the WT and MHCII-deficient strains, and the average B cell number was 1.6 × 108 and 1.5 × 108 total B cells in the WT and MHCII-deficient strains, respectively. (B) Flow cytometric analysis of CD11c-positive B cells from wild-type and IL-21R-deficient mice on day 70 post-infection. The frequencies of CD11c-positive B cells shown in the dot plots represent their percentage among total splenic B cells. In the experiment shown, the average B cell frequency was 35% and 18% in the WT and IL-21R-deficient strains, and the average B cell number was 5.9 × 107 and 5.8 × 107 B cells, respectively. The data in A and B are representative of two experiments; * p<0.05, **p< 0.005.
CD11c-positive B cells reside in the splenic marginal zone and at the edge of B cell follicles
Lymphocyte positioning within secondary lymphoid organs is critical for their function. The splenic marginal zone (MZ) is a specialized area at the border of the periarteriolar lymphoid sheath (PALS) and B cell follicles, and is populated by macrophages, dendritic cells (DCs), and MZ B cells. Due to the nature of blood flow in the spleen, cells residing in this region have unique access to blood-borne antigens and pathogens (47, 48). Memory B cells have also been shown to localize to the MZ in both rodents and humans (49–52). In situ analyses of spleens from mice on day 63 post-infection identified CD19-positive B cell follicles, with CD11c-positive cell staining in the red pulp (Figure 4a). CD169-positive metallophilic macrophages delineated the MZ, although in infected mice these were found in fewer numbers, and were by appearance less organized than in uninfected mice. We detected distinct co-localization of CD19- and CD11c-co-expressing cells in the MZ, adjacent to CD169-positive macrophages (Figure 4b). In addition, dual CD19- and CD11c-expressing cells were found at the edges of B cell follicles, but were peanut agglutinin (PNA)-negative, and were not detected in GCs (Figure 4c, d). These data indicate that the CD11c-positive B cells are located in the splenic MZ, and at the edge of the B cell follicles, findings that are consistent with the reported localization of memory B cells in rodents and humans.
Figure 4. CD19-positive B cells were detected in the splenic marginal zone and at the edge of B cell follicles.
Cryosections from uninfected (A, C), and infected mice (B, D; analyzed on day 63 post-infection) were stained with Alexa 488-conjugated CD11c (green), and CF594-conjugated CD19 (red), and Alexa 647-conjugated CD169 (blue; panels A and B) or PNA-Alexa 647 (blue; panels C and D). The images show representative images of B cells identified in several randomly selected cryosections. The scale bars represent 40 μm, and yellow arrows indicate several dual CD11c- CD19 -positive B cells.
IgG Recall Responses in Antigen-Challenged Mice
Robust, high-affinity responses to secondary antigen challenge are considered a hallmark of humoral memory. A large proportion of antigen-specific B cells elicited during ehrlichial infections recognize an outer membrane protein (OMP-19), one member of a family of OMPs encoded by the ehrlichiae (53). Both OMP-19-specific IgG and IgM are highly effective at eliciting protective immunity against fatal ehrlichial infection (23, 26, 29). Since secondary E. muris infection is rapidly neutralized by pre-existing antibodies, we evaluated humoral memory responses following intraperitoneal administration of recombinant OMP-19. IgG production was analyzed because we hypothesized that IgM memory B cells would undergo differentiation and class switching in GCs following antigen re-encounter, as was previously reported (16). Mice challenged with recombinant OMP-19, delivered in alum, generated reciprocal IgG titers as high as 17,000–12 days later (Figure 5a); IgG titers in non-challenged mice, and following primary OMP-19 immunization, were by comparison relatively low (Figure 5b). Analysis of IgG subclass utilization showed IgG2c to be the dominant antibody isotype elicited after antigen challenge (reciprocal IgG2c titers were detected as high as 31,200). Mice that were challenged with OMP-19 without alum also produced robust IgG2c responses, indicating that adjuvant was not necessary for IgG production. IgG1, which is not usually detected following ehrlichial infection (54), was detected after antigen challenge, suggesting that the IgG production after antigen challenge was a consequence of class-switching.
Figure 5. Antigen challenge of E. muris-infected mice.
Uninfected and infected mice were administered alum, or were challenged with 100 μg recombinant OMP-19 in alum; challenge of the infected mice was performed on day 30 post-infection. (A) Reciprocal OMP-19-specific serum IgG titers obtained on days 0, 3, 6, and 12 post-antigen challenge, determined by ELISA, are shown. (B) OMP-19-specific IgG subclass titers measured in the serum of infected mice that remained unchallenged, or had been challenged with OMP-19; OMP-19 was delivered in either alum or PBS, as indicated. (C) Serum OMP-19 specific antibody titers were determined on days 0, 6, and 12 post-challenge in MHCII-deficient, IL-21-deficient, and wild-type mice. The data in each of the panels are representative of two experiments. * p<0.05, ** p< 0.005
As the generation of the CD11c-positive B cell population was wholly or in part CD4 T cell- and IL-21R-dependent, we also tested whether these cells and/or factors were required for efficient recall responses following antigen challenge. MHC-II-deficient mice did not generate an IgG recall response, and IL-21R-deficient mice exhibited a 7-fold decrease in OMP19-specific IgG on day 12 post-challenge, relative to challenged to wild-type mice (Figure 5c). These data demonstrate that mice infected with E. muris undergo a robust CD4 T cell- and IL-21R-dependent class-switched memory response to antigen challenge.
CD11c-positive B cells are responsible for amnestic responses to OMP-19 challenge
Since E. muris infected mice generated a strong secondary response to OMP-19 challenge, we next addressed whether the CD11c-positive B cells were directly responsible, by specifically depleting the population in vivo. For these studies, we targeted CD11c-expressing B cells using CD11c-DTR transgenic mice, a mouse strain which encodes a simian diphtheria toxin receptor (DTR)-GFP fusion protein that is regulated by the CD11c promoter (55). Administration of diphtheria toxin (DTX) has been shown to efficiently deplete CD11c-expressing cells in vivo. To avoid possible artifacts associated with the depletion of CD11c-positive DCs, irradiation-induced bone marrow chimeric mice were generated using a 1:1 ratio of donor bone marrow obtained from B cell-deficient, and CD11c-DTR transgenic mice. In this experimental model, all donor-derived CD11c-expressing B cells were anticipated to be DTX-sensitive, but CD11c-negative B cells, and donor-derived DCs, were expected to be DTX-resistant. The CD11c-positive B cells were indeed susceptible to the toxin, as DTX treatment eliminated these cells from infected CD11c-DTR transgenic mice within 24 hours of toxin administration (Figure 6a). To address whether the CD11c-positive B cells were required for the humoral recall response to antigen challenge, infected chimeric mice were administered DTX on days -1, and 6 post-OMP-19 challenge. Serum antibody titers were determined by ELISA, 6 and 12 days post-challenge. Infected chimeric mice that were administered DTX, exhibited a 5-fold reduction in OMP-19 titers relative to non-toxin treated infected mice (Figure 6b). Uninfected chimeric mice that were challenged with OMP-19 exhibited modest IgG titers, and no reduction of IgG titers were observed following toxin administration. These data strongly suggest that the CD11c-positive IgM B cells were responsible for the amnestic IgG response.
Figure 6. In vivo depletion of CD11c-positive B cells abrogated the IgG responses following antigen challenge.
(A) CD11c-DTR transgenic mice were infected, and administered diphtheria toxin (DTX; 9ng/g of body weight) on day 30 post-infection. Twenty-four hours later, splenocytes were analyzed by flow cytometry. The panels shown are representative of 3 mice per group. (B) Chimeric mice were generated using a 1:1 ratio of B cell-deficient and CD11c-DTR bone marrow. The chimeric mice were infected, administered DTX on day 30 post-infection, and challenged with OMP-19, as described in Figure 5. (C) Chimeric mice were generated as in B, using a 9:1 ratio of B cell deficient to CD11c-DTR bone marrow. Serum OMP-19 titers were determined on day 12 post-challenge. The data in each of the studies shown are representative of at least two experiments; * p<0.05
DCs are known for their ability to initiate primary immune responses, and for their important role as professional APCs (56). In addition to these important functions, DCs also have the ability to modulate B cell responses (57, 58). To rule out any possibility that the impaired humoral recall responses observed following DTX treatment were due to partial DC depletion, bone marrow chimeric mice were also generated using a 9:1 ratio of B cell-deficient and CD11c-DTR transgenic bone marrow, respectively. Because in these chimeric mice the only source of B cells was from the transgenic bone marrow, all CD11c-expressing B cells were susceptible to DTX administration, and the majority of DCs were DTX-resistant. Similar to the data obtained using the equicellular chimeric mice, the 9:1 chimeric mice treated with DTX exhibited a 10-fold reduction in OMP-19-specific IgG titers, relative to untreated mice (Figure 6c). Therefore, it is unlikely that the ablation of the secondary IgG responses elicited following antigen challenge was due to the DTX-mediated depletion of CD11c-positive DCs. These data demonstrate that the CD11c-positive B cell population is required for IgG recall responses to antigenic challenge in E. muris infected mice, supporting the conclusion that the CD11c-positive B cells are bona fide IgM memory B cells. The data are also consistent with the notion that IgM memory cells undergo class switching in response to antigen challenge.
Discussion
Our studies have demonstrated that splenic IgM memory B cells are generated in the mouse in response to bacterial infection, and that these cells are responsible for IgG production following secondary challenge. Although many researchers have sought means to monitor and enumerate memory B cells in vivo, definitive assessment of such a population of cells has been difficult to achieve in infected mice. Thus, our findings are significant as they have identified IgM memory B cells in a natural model of infection. Although IgM memory B cells were described in the 1980s, and have been identified in humans, we have show they can be elicited by bacterial infection, and that they can accumulate in large numbers in the spleen. Thus, although it has been generally considered that memory B cells are normally maintained at low frequencies, our data indicate that this is not necessarily the case. Although they were found in unexpectedly large numbers, we have demonstrated on the basis of many independent criteria, that the CD11c-positive B cells we have identified in the spleen during E. muris infection are a bona fide population of IgM memory B cells. Our conclusion is based on the following definitive criteria: 1) cell surface marker expression on the CD11c-positive B cells was nearly identical to that described for memory B cells identified in other non-infectious experimental models; 2) the cell population persisted indefinitely, and was largely quiescent; 3) the cells were CD138-negative, and did not produce IgM or IgG spontaneously; 4) they did not express GC markers, and resided in the splenic MZ, not in GCs; 5) a significant proportion of the cells contained mutated Vh genes, and 6) deletion of CD11c-positive B cells in vivo ablated recall responses to specific antigen. Although it is possible that CD11c-positive B cells in tissue other than the spleen are responsible, we conclude that splenic B cells are involved, as this tissue harbors a large population of cells of this phenotype. It is also possible that the CD11c-positive B cell population performs different or additional functions. Such functions may include their acting as non-antibody secreting, chronically-activated suppressor B cells, or APCs, although, such functions are not supported by any of our data. It is also possible that only a small subset of the CD11c-positive B cells are “true” memory B cells responsible for IgG responses following antigen challenge. This explanation is unlikely correct, however, as the population was homogenous with respect to the expression of most cell surface markers used for their characterization. Thus, although memory B cells are usually considered to be present in low frequencies, our findings indicate that bacterial infection can drive the production and maintenance of large numbers of such cells.
The observation that the CD11c-positive B cells are maintained under conditions of low-level persistent infection does not exclude them as memory B cells. In fact, memory B cells are generated in several chronic infections by pathogens such as HIV, malaria, CMV, and HCV (59–65). Although the IgM memory B cells in our studies were maintained following antibiotic treatment, we were unable to exclude the possibility that antigen persisted under these conditions, and was responsible for memory B cell maintenance. Indeed, it is well established that antigen can persist for months if not years on follicular dendritic cells (66). Although there is conflicting evidence in the literature, it is possible that low antigen exposure or inflammation is required for memory cell maintenance (34, 67–70). Indeed, studies have suggested that subclinical infections may be responsible for the maintenance of memory to bacterial pathogens (64).
Why, then, does ehrlichial infection generate such a large population of IgM memory B cells? Like memory T cells, memory B cells generated under different conditions, are likely heterogeneous, depending on the inflammatory conditions associated with infection or immunization (9, 71). Indeed, we have described extensive spleen disorganization and/or destruction during acute ehrlichia infection that may facilitate the development of large numbers of IgM memory cells (54). We propose that ehrlichial infection drives the expansion of a particularly homogenous subset of CD11c-positive IgM memory B cells that may represent a subset of memory B cells elicited during other infections, or following distinct vaccination regimens. The particularly high frequency of the CD11c-positive B cell population does not undermine the conclusion that they are memory B cells. Rather, our findings suggest that there are significant quantitative and qualitative differences among memory B cells elicited by different pathogens. It is possible that yet other memory B cells were generated during ehrlichial infection that we were unable to identify, including swIg memory B cells. However, the observation that DTX treatment ablated the IgG recall responses, suggested that all of the IgG produced upon antigen challenge was derived from the CD11c-expressing IgM memory B cells. Indeed, we have demonstrated that IgG production and GC formation are inhibited during acute E. muris infection (54), which may explain the apparent paucity of swIg cells in our model. Nevertheless, we cannot formally eliminate the possibility that small numbers of CD11c-positive, IgM-negative, swIg memory B cells were also targeted in the DTX depletion studies. This explanation appears unlikely, and does not undermine the conclusion that the IgM memory B cells are major, if not exclusive contributors to the IgG recall responses.
The IgM memory B cells described in our model were initially identified on the basis of their unique expression of CD11c. Although CD11c expression is atypical in B cells, several studies have identified CD11c expression on B cells in both mice and humans (29, 60, 72–75). CD11c (αx) is an alpha integrin of the leukointegrin family, and is expressed as a dimer with the common β chain CD18 (β2), forming complement receptor 4 (CR4). CD11c can bind a variety of ligands, including cell adhesion molecules, fibrinogen, and iC3b (76). The function of CD11c on IgM memory B cells is unknown, but as an adhesion molecule, CD11c may be partly responsible for proper positioning of the IgM memory B cells within or adjacent to the splenic MZ. In fact, other integrins have been shown to be responsible for proper positioning of B cells within the MZ, and for trafficking through the MZ into the white pulp (77, 78). However, several other surface antigens, in addition to CD11c and IgM, uniquely distinguish these cells from other B cell populations. We noted several co-stimulatory and adhesion molecules that were upregulated on the CD11c-positive B cells, as compared to CD11c-negative B cells. These markers included CD73, CXCR4, PD-L2, TACI, and CD11b. CD73 is an ecto-enzyme with several functions including lymphocyte adhesion, and catalysis of extracellular AMP to adenosine (79). CD73 is often expressed on mature lymphocytes, and as such has been considered a B cell maturation marker (80). Several studies have also shown CD73 expression on memory B cell populations, particularly those with somatic mutations (17, 32, 38). CXCR4 is a chemokine receptor responsible for cell migration towards its ligand CXCL12. This interaction is important for B cell homeostasis, plasma cell homing to the bone marrow, and GC organization (81, 82). The upregulation of CXCR4 on the surface of the CD11c-positive IgM memory B cells may regulate eventual homing of these cells to the bone marrow. PD-L2 (programmed death-ligand 2) is a B7 homolog, known for inhibiting T cell proliferation through its interaction with PD-1 (programmed death molecule-1). Although PD-L2 expression is typically associated with macrophages and DCs, expression has also been shown on B1 cells, and to be highly up-regulated on memory B cell populations (17, 33, 83). TACI is a transmembrane receptor in the tumor necrosis receptor superfamily that binds both BAFF (B cell activating factor of the TNF family) and APRIL (a proliferation-inducing ligand). Signals transmitted by TACI modulate many B cell functions, and survival in the periphery. Available evidence has indicated that BAFF and APRIL signals are not necessary for memory B cell survival, despite high expression of TACI on memory B cells (84), although a possible function for TACI on IgM memory B cells has not yet been evaluated. CD11b expression is characteristic of B-1 cells, and may suggest a possible cellular origin for the IgM memory B cells we have identified. The unique cell surface phenotype of the IgM memory cells we have identified suggest that it may be possible to unambiguously identify such cells during other infections, and in humans.
Although we have demonstrated that the generation of the IgM memory B cells was CD4 T cell-dependent, the IgM memory B cells are similar in cell surface phenotype to the population of splenic CD11clo T cell-independent plasmablasts we have previously described (29). The CD11clo plasmablasts are detected at their highest frequency and number on day 9 post-E. muris infection, and are no longer detected by day 18 post-infection. As the IgM memory B cells described in this study were first detected at about day 30 post-infection, after the plasmablast response declines, it is possible that the IgM memory cells may be derived from the plasmablasts themselves, or from early subset of precursor cells that are then “rescued” upon receipt of CD4 T cell help delivered by T follicular helper cells in GCs.
A requirement for T cell help and GC development in the generation of IgM memory B cells remains controversial. It has been proposed that IgM memory B cells are generated in a GC-independent fashion, and function to provide long-term T-independent immunity (38). This conclusion is based on the observations that not all memory B cells carry somatic mutations, and that IgM memory B cells can be generated in both mice and humans that cannot form GCs (32, 46, 85, 86). Accordingly, Taylor et al., have described a population of murine CD73-negative IgM memory B cells and their precursors that develop independently of GCs (38). In contrast, studies by Dogan et al., described an analogous IgM memory B cell population that was somatically mutated, CD73-positive, and likely GC derived. A GC-derived population of IgM+ IgD+ CD27+ memory B cells has also been described in humans (87). Our data has established an essential role for CD4 T cells, and an important role for IL-21 signaling, in the generation of IgM memory B cells during E. muris infection. In addition, the presence of somatic mutations in a large portion of the IgM memory B cells we have identified is consistent with a role for GC differentiation. Nevertheless, we have not formally resolved whether these memory B cells undergo memory cell development in GCs. An explanation for the apparent differences in the requirement for GC development reported in different studies is that different IgM memory B cell populations with distinct origins predominate in vivo, depending on the particular infection or immunization model being studied.
Several studies have described IgM memory B cells in humans (reviewed in 85). The relationship between the IgM memory B cells identified in humans, and those we and other have identified in mice is not yet known, although the CD11c-positive IgM memory B cells described here bear similarities to memory B cells described in humans. Indeed, expression of CD11c has been reported on tissue-specific Fc receptor-like protein 4 (FCLR4)-positive memory B cells in human tonsils, as well as in the blood of HIV-infected patients (60, 73). CD11c expression has also been identified on a population of atypical memory B cells in malaria patients (64), in various human B cell malignancies, and autoimmune patients (74, 88–90). Thus, it will be important to determine the relationship between the various human IgM memory B cell populations, and those observed in murine models, so that the generation and function of the cells may be studied systematically.
Why should humoral memory be harbored in IgM memory B cells, given that swig memory B cells may respond more quickly, and with higher affinity to secondary challenge? Although IgM is produced early following infection, we have shown that E. muris infection also elicits a long-term protective IgM response (30). IgM memory B cells may be required to maintain long-term IgM production in ehrlichial and other chronic infections, where low concentrations of foreign antigens persist. Alternatively, IgM memory B cells, but not swIg memory B cells, can initiate GC reactions and undergo affinity maturation and class-switch recombination following re-encounter with antigen (16, 18). This process may function to allow greater flexibility when the host encounters variant pathogens. The observation that the CD11c-positive B cells are required for the class-switched antibody response to OMP-19 challenge, also suggests the IgM memory cells undergo GC re-entry. However, we have not demonstrated whether the IgM memory cells re-enter GCs, as we have been unable to engraft the CD11c-positive B cells into recipient mice. Although the limited somatic mutation that occurs in IgM memory B cells may generate higher affinity B cell receptors, it is also possible that antigen affinity is less important that other attributes of IgM memory B cells. These attributes may include their increased number, specialized anatomic localization (i.e. the splenic MZ), and perhaps a better capacity for antigen presentation and cytokine production. Understanding such differences in the properties and functions of IgM memory B cells in mice and humans will facilitate strategies to generate they type of humoral memory most appropriate for particular infections. Our findings underscore the notion that immunological memory is mediated by rich and diverse memory B cell populations that together mediate host defense against the wide range of pathogens encountered in nature.
Acknowledgments
This work was supported by U.S. Public Health Service grant R01AI064678 to G.M.W.
The authors would like to acknowledge R. Song, of the Wadsworth Center Flow Cytometry Core, and R. Cole of the Wadsworth Center Advanced Light Microscopy Core for their technical expertise. The authors also thank K. Chave of the Wadsworth Center Protein Expression Core for production of the recombinant OMP-19 (grant to Dr. I. Lipkin). We also thank P. Maskin, A. Yermakova, M. Tighe, and T. Cookenham for excellent technical assistance, and Dr. L. Warren (National Institutes for Health) for providing the IL-21R-deficient mice. We also thank Drs. W. Lee (Wadsworth Center) and E. Leadbetter (Trudeau Institute) for critical reading of the manuscript.
References
- 1.McHeyzer-Williams LJ, McHeyzer-Williams MG. Antigen-specific memory B cell development. Annu Rev Immunol. 2005;23:487–513. doi: 10.1146/annurev.immunol.23.021704.115732. [DOI] [PubMed] [Google Scholar]
- 2.McHeyzer-Williams M, Okitsu S, Wang N, McHeyzer-Williams L. Molecular programming of B cell memory. Nat Rev Immunol. 2011;12:24–34. doi: 10.1038/nri3128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kaisho T, Schwenk F, Rajewsky K. The roles of gamma 1 heavy chain membrane expression and cytoplasmic tail in IgG1 responses. Science. 1997;276:412–415. doi: 10.1126/science.276.5311.412. [DOI] [PubMed] [Google Scholar]
- 4.Martin SW, Goodnow CC. Burst-enhancing role of the IgG membrane tail as a molecular determinant of memory. Nat Immunol. 2002;3:182–188. doi: 10.1038/ni752. [DOI] [PubMed] [Google Scholar]
- 5.Tangye SG, Avery DT, Deenick EK, Hodgkin PD. Intrinsic differences in the proliferation of naive and memory human B cells as a mechanism for enhanced secondary immune responses. J Immunol. 2003;170:686–694. doi: 10.4049/jimmunol.170.2.686. [DOI] [PubMed] [Google Scholar]
- 6.Wakabayashi C, Adachi T, Wienands J, Tsubata T. A distinct signaling pathway used by the IgG-containing B cell antigen receptor. Science. 2002;298:2392–2395. doi: 10.1126/science.1076963. [DOI] [PubMed] [Google Scholar]
- 7.Weiss U, Rajewsky K. The repertoire of somatic antibody mutants accumulating in the memory compartment after primary immunization is restricted through affinity maturation and mirrors that expressed in the secondary response. J Exp Med. 1990;172:1681–1689. doi: 10.1084/jem.172.6.1681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yoshida T, Mei H, Dorner T, Hiepe F, Radbruch A, Fillatreau S, Hoyer BF. Memory B and memory plasma cells. Immunol Rev. 2010;237:117–139. doi: 10.1111/j.1600-065X.2010.00938.x. [DOI] [PubMed] [Google Scholar]
- 9.Good-Jacobson KL, Tarlinton DM. Multiple routes to B-cell memory. Int Immunol. 2012;24:403–408. doi: 10.1093/intimm/dxs050. [DOI] [PubMed] [Google Scholar]
- 10.Klein U, Kuppers R, Rajewsky K. Evidence for a large compartment of IgM-expressing memory B cells in humans. Blood. 1997;89:1288–1298. [PubMed] [Google Scholar]
- 11.Klein U, Rajewsky K, Kuppers R. Human immunoglobulin (Ig)M+IgD+ peripheral blood B cells expressing the CD27 cell surface antigen carry somatically mutated variable region genes: CD27 as a general marker for somatically mutated (memory) B cells. J Exp Med. 1998;188:1679–1689. doi: 10.1084/jem.188.9.1679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.White H, Gray D. Analysis of immunoglobulin (Ig) isotype diversity and IgM/D memory in the response to phenyl-oxazolone. J Exp Med. 2000;191:2209–2220. doi: 10.1084/jem.191.12.2209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yefenof E, V, Sanders M, Snow EC, Noelle RJ, Oliver KG, Uhr JW, Vitetta ES. Preparation and analysis of antigen-specific memory B cells. J Immunol. 1985;135:3777–3784. [PubMed] [Google Scholar]
- 14.Zan-Bar I, Vitetta ES, Assisi F, Strober S. The relationship between surface immunoglobulin isotype and immune function of murine B lymphocytes. III. Expression of a single predominant isotype on primed and unprimed B cells. J Exp Med. 1978;147:1374–1394. doi: 10.1084/jem.147.5.1374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zan-Bar I, Strober S, Vitetta ES. The relationship between surface immunoglobulin isotype and immune function of murine B lymphocytes. IV. Role of IgD-bearing cells in the propagation of immunologic memory. J Immunol. 1979;123:925–930. [PubMed] [Google Scholar]
- 16.Dogan I, Bertocci B, Vilmont V, Delbos F, Megret J, Storck S, Reynaud CA, Weill JC. Multiple layers of B cell memory with different effector functions. Nat Immunol. 2009;10:1292–1299. doi: 10.1038/ni.1814. [DOI] [PubMed] [Google Scholar]
- 17.Tomayko MM, Steinel NC, Anderson SM, Shlomchik MJ. Cutting edge: Hierarchy of maturity of murine memory B cell subsets. J Immunol. 2010;185:7146–7150. doi: 10.4049/jimmunol.1002163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pape KA, Taylor JJ, Maul RW, Gearhart PJ, Jenkins MK. Different B cell populations mediate early and late memory during an endogenous immune response. Science. 2011;331:1203–1207. doi: 10.1126/science.1201730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ozaki K, Spolski R, Feng CG, Qi CF, Cheng J, Sher A, Morse HC, 3rd, Liu C, Schwartzberg PL, Leonard WJ. A critical role for IL-21 in regulating immunoglobulin production. Science. 2002;298:1630–1634. doi: 10.1126/science.1077002. [DOI] [PubMed] [Google Scholar]
- 20.MacNamara KC, Racine R, Chatterjee M, Borjesson D, Winslow GM. Diminished hematopoietic activity associated with alterations in innate and adaptive immunity in a mouse model of human monocytic ehrlichiosis. Infect Immun. 2009;77:4061–4069. doi: 10.1128/IAI.01550-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stevenson HL, Jordan JM, Peerwani Z, Wang HQ, Walker DH, Ismail N. An intradermal environment promotes a protective type-1 response against lethal systemic monocytotropic ehrlichial infection. Infect Immun. 2006;74:4856–4864. doi: 10.1128/IAI.00246-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pritt BS, Sloan LM, Johnson DK, Munderloh UG, Paskewitz SM, McElroy KM, McFadden JD, Binnicker MJ, Neitzel DF, Liu G, Nicholson WL, Nelson CM, Franson JJ, Martin SA, Cunningham SA, Steward CR, Bogumill K, Bjorgaard ME, Davis JP, McQuiston JH, Warshauer DM, Wilhelm MP, Patel R, Trivedi VA, Eremeeva ME. Emergence of a new pathogenic ehrlichia species, Wisconsin and Minnesota, 2009. N Engl J Med. 2011;365:422–429. doi: 10.1056/NEJMoa1010493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nandi B, Hogle K, Vitko N, Winslow GM. CD4 T-cell epitopes associated with protective immunity induced following vaccination of mice with an ehrlichial variable outer membrane protein. Infect Immun. 2007;75:5453–5459. doi: 10.1128/IAI.00713-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Reinhardt RL, Liang HE, Locksley RM. Cytokine-secreting follicular T cells shape the antibody repertoire. Nature immunology. 2009;10:385–393. doi: 10.1038/ni.1715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.McBride KM, Gazumyan A, Woo EM, Schwickert TA, Chait BT, Nussenzweig MC. Regulation of class switch recombination and somatic mutation by AID phosphorylation. J Exp Med. 2008;205:2585–2594. doi: 10.1084/jem.20081319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li JS, Yager E, Reilly M, Freeman C, Reddy GR, Chu FK, Winslow G. Outer membrane protein specific monoclonal antibodies protect SCID Mice from fatal infection by the obligate intracellular bacterial pathogen Ehrlichia chaffeensis. J Immunol. 2001;166:1855–1862. doi: 10.4049/jimmunol.166.3.1855. [DOI] [PubMed] [Google Scholar]
- 27.Ismail N, Soong L, McBride JW, Valbuena G, Olano JP, Feng HM, Walker DH. Overproduction of TNF-alpha by CD8+ type 1 cells and down-regulation of IFN-gamma production by CD4+ Th1 cells contribute to toxic shock-like syndrome in an animal model of fatal monocytotropic ehrlichiosis. J Immunol. 2004;172:1786–1800. doi: 10.4049/jimmunol.172.3.1786. [DOI] [PubMed] [Google Scholar]
- 28.Bitsaktsis C, Nandi B, Racine R, MacNamara KC, Winslow G. T-Cell-independent humoral immunity is sufficient for protection against fatal intracellular ehrlichia infection. Infect Immun. 2007;75:4933–4941. doi: 10.1128/IAI.00705-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Racine R, Chatterjee M, Winslow GM. CD11c expression identifies a population of extrafollicular antigen-specific splenic plasmablasts responsible for CD4 T-independent antibody responses during intracellular bacterial infection. J Immunol. 2008;181:1375–1385. doi: 10.4049/jimmunol.181.2.1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Racine R, McLaughlin M, Jones DD, Wittmer ST, MacNamara KC, Woodland DL, Winslow GM. IgM production by bone marrow plasmablasts contributes to long-term protection against intracellular bacterial infection. J Immunol. 2011;186:1011–1021. doi: 10.4049/jimmunol.1002836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kaji T, Ishige A, Hikida M, Taka J, Hijikata A, Kubo M, Nagashima T, Takahashi Y, Kurosaki T, Okada M, Ohara O, Rajewsky K, Takemori T. Distinct cellular pathways select germline-encoded and somatically mutated antibodies into immunological memory. J Exp Med. 2012;209:2079–2097. doi: 10.1084/jem.20120127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Anderson SM, Tomayko MM, Ahuja A, Haberman AM, Shlomchik MJ. New markers for murine memory B cells that define mutated and unmutated subsets. J Exp Med. 2007;204:2103–2114. doi: 10.1084/jem.20062571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Good-Jacobson KL, Szumilas CG, Chen L, Sharpe AH, Tomayko MM, Shlomchik MJ. PD-1 regulates germinal center B cell survival and the formation and affinity of long-lived plasma cells. Nat Immunol. 2010;11:535–542. doi: 10.1038/ni.1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ahmed R, Gray D. Immunological memory and protective immunity: understanding their relation. Science. 1996;272:54–60. doi: 10.1126/science.272.5258.54. [DOI] [PubMed] [Google Scholar]
- 35.Schittek B, Rajewsky K. Maintenance of B-cell memory by long-lived cells generated from proliferating precursors. Nature. 1990;346:749–751. doi: 10.1038/346749a0. [DOI] [PubMed] [Google Scholar]
- 36.Allen CD, Okada T, Cyster JG. Germinal-center organization and cellular dynamics. Immunity. 2007;27:190–202. doi: 10.1016/j.immuni.2007.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kruetzmann S, Rosado MM, Weber H, Germing U, Tournilhac O, Peter HH, Berner R, Peters A, Boehm T, Plebani A, Quinti I, Carsetti R. Human immunoglobulin M memory B cells controlling Streptococcus pneumoniae infections are generated in the spleen. J Exp Med. 2003;197:939–945. doi: 10.1084/jem.20022020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Taylor JJ, Pape KA, Jenkins MK. A germinal center-independent pathway generates unswitched memory B cells early in the primary response. J Exp Med. 2012;209:597–606. doi: 10.1084/jem.20111696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Weller S, Braun MC, Tan BK, Rosenwald A, Cordier C, Conley ME, Plebani A, Kumararatne DS, Bonnet D, Tournilhac O, Tchernia G, Steiniger B, Staudt LM, Casanova JL, Reynaud CA, Weill JC. Human blood IgM “memory” B cells are circulating splenic marginal zone B cells harboring a prediversified immunoglobulin repertoire. Blood. 2004;104:3647–3654. doi: 10.1182/blood-2004-01-0346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kim JH, Kim J, Jang YS, Chung GH. Germinal center-independent affinity maturation in tumor necrosis factor receptor 1-deficient mice. J Biochem Mol Biol. 2006;39:586–594. doi: 10.5483/bmbrep.2006.39.5.586. [DOI] [PubMed] [Google Scholar]
- 41.Gardby E, Wrammert J, Schon K, Ekman L, Leanderson T, Lycke N. Strong differential regulation of serum and mucosal IgA responses as revealed in CD28-deficient mice using cholera toxin adjuvant. J Immunol. 2003;170:55–63. doi: 10.4049/jimmunol.170.1.55. [DOI] [PubMed] [Google Scholar]
- 42.Matsumoto M, Lo SF, Carruthers CJ, Min J, Mariathasan S, Huang G, Plas DR, Martin SM, Geha RS, Nahm MH, Chaplin DD. Affinity maturation without germinal centres in lymphotoxin-alpha-deficient mice. Nature. 1996;382:462–466. doi: 10.1038/382462a0. [DOI] [PubMed] [Google Scholar]
- 43.Good-Jacobson KL, Shlomchik MJ. Plasticity and heterogeneity in the generation of memory B cells and long-lived plasma cells: the influence of germinal center interactions and dynamics. J Immunol. 2010;185:3117–3125. doi: 10.4049/jimmunol.1001155. [DOI] [PubMed] [Google Scholar]
- 44.Linterman MA, Beaton L, Yu D, Ramiscal RR, Srivastava M, Hogan JJ, Verma NK, Smyth MJ, Rigby RJ, Vinuesa CG. IL-21 acts directly on B cells to regulate Bcl-6 expression and germinal center responses. J Exp Med. 2010;207:353–363. doi: 10.1084/jem.20091738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ozaki K, Spolski R, Ettinger R, Kim HP, Wang G, Qi CF, Hwu P, Shaffer DJ, Akilesh S, Roopenian DC, Morse HC, 3rd, Lipsky PE, Leonard WJ. Regulation of B cell differentiation and plasma cell generation by IL-21, a novel inducer of Blimp-1 and Bcl-6. J Immunol. 2004;173:5361–5371. doi: 10.4049/jimmunol.173.9.5361. [DOI] [PubMed] [Google Scholar]
- 46.Zotos D, Coquet JM, Zhang Y, Light A, D’Costa K, Kallies A, Corcoran LM, Godfrey DI, Toellner KM, Smyth MJ, Nutt SL, Tarlinton DM. IL-21 regulates germinal center B cell differentiation and proliferation through a B cell-intrinsic mechanism. J Exp Med. 2010;207:365–378. doi: 10.1084/jem.20091777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Martin F, Kearney JF. B-cell subsets and the mature preimmune repertoire. Marginal zone and B1 B cells as part of a “natural immune memory”. Immunol Rev. 2000;175:70–79. [PubMed] [Google Scholar]
- 48.Kraal G. Cells in the marginal zone of the spleen. Int Rev Cytol. 1992;132:31–74. doi: 10.1016/s0074-7696(08)62453-5. [DOI] [PubMed] [Google Scholar]
- 49.Dunn-Walters DK, Isaacson PG, Spencer J. Analysis of mutations in immunoglobulin heavy chain variable region genes of microdissected marginal zone (MGZ) B cells suggests that the MGZ of human spleen is a reservoir of memory B cells. J Exp Med. 1995;182:559–566. doi: 10.1084/jem.182.2.559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Liu YJ, Oldfield S, MacLennan IC. Memory B cells in T cell-dependent antibody responses colonize the splenic marginal zones. Eur J Immunol. 1988;18:355–362. doi: 10.1002/eji.1830180306. [DOI] [PubMed] [Google Scholar]
- 51.Tangye SG, Liu YJ, Aversa G, Phillips JH, de Vries JE. Identification of functional human splenic memory B cells by expression of CD148 and CD27. J Exp Med. 1998;188:1691–1703. doi: 10.1084/jem.188.9.1691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Oldfield S, Liu YJ, Beaman M, MacLennan CM. Memory B cells generated in T cell-dependent antibody responses colonise the splenic marginal zone. Adv Exp Med Biol. 1988;237:93–98. doi: 10.1007/978-1-4684-5535-9_13. [DOI] [PubMed] [Google Scholar]
- 53.Ohashi N, Rikihisa Y, Unver A. Analysis of transcriptionally active gene clusters of major outer membrane protein multigene family in Ehrlichia canis and E. chaffeensis. Infect Immun. 2001;69:2083–2091. doi: 10.1128/IAI.69.4.2083-2091.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Racine R, Jones DD, Chatterjee M, McLaughlin M, Macnamara KC, Winslow GM. Impaired germinal center responses and suppression of local IgG production during intracellular bacterial infection. J Immunol. 2010;184:5085–5093. doi: 10.4049/jimmunol.0902710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jung S, Unutmaz D, Wong P, Sano G, De los Santos K, Sparwasser T, Wu S, Vuthoori S, Ko K, Zavala F, Pamer EG, Littman DR, Lang RA. In vivo depletion of CD11c(+) dendritic cells abrogates priming of CD8(+) T cells by exogenous cell-associated antigens. Immunity. 2002;17:211–220. doi: 10.1016/s1074-7613(02)00365-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Banchereau J, Briere F, Caux C, Davoust J, Lebecque S, Liu YJ, Pulendran B, Palucka K. Immunobiology of dendritic cells. Annu Rev Immunol. 2000;18:767–811. doi: 10.1146/annurev.immunol.18.1.767. [DOI] [PubMed] [Google Scholar]
- 57.Cyster JG. B cell follicles and antigen encounters of the third kind. Nat Immunol. 2010;11:989–996. doi: 10.1038/ni.1946. [DOI] [PubMed] [Google Scholar]
- 58.Ueno H, Palucka AK, Banchereau J. The expanding family of dendritic cell subsets. Nat Biotechnol. 2010;28:813–815. doi: 10.1038/nbt0810-813. [DOI] [PubMed] [Google Scholar]
- 59.Aberle JH, Puchhammer-Stockl E. Age-dependent increase of memory B cell response to cytomegalovirus in healthy adults. Exp Gerontol. 2012;47:654–657. doi: 10.1016/j.exger.2012.04.008. [DOI] [PubMed] [Google Scholar]
- 60.Moir S, Ho J, Malaspina A, Wang W, DiPoto AC, O’Shea MA, Roby G, Kottilil S, Arthos J, Proschan MA, Chun TW, Fauci AS. Evidence for HIV-associated B cell exhaustion in a dysfunctional memory B cell compartment in HIV-infected viremic individuals. J Exp Med. 2008;205:1797–1805. doi: 10.1084/jem.20072683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Mouquet H, Klein F, Scheid JF, Warncke M, Pietzsch J, Oliveira TY, Velinzon K, Seaman MS, Nussenzweig MC. Memory B cell antibodies to HIV-1 gp140 cloned from individuals infected with clade A and B viruses. PLoS One. 2011;6:e24078. doi: 10.1371/journal.pone.0024078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ndungu FM, Cadman ET, Coulcher J, Nduati E, Couper E, Macdonald DW, Ng D, Langhorne J. Functional memory B cells and long-lived plasma cells are generated after a single Plasmodium chabaudi infection in mice. PLoS Pathog. 2009;5:e1000690. doi: 10.1371/journal.ppat.1000690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Scheid JF, Mouquet H, Feldhahn N, Seaman MS, Velinzon K, Pietzsch J, Ott RG, Anthony RM, Zebroski H, Hurley A, Phogat A, Chakrabarti B, Li Y, Connors M, Pereyra F, Walker BD, Wardemann H, Ho D, Wyatt RT, Mascola JR, Ravetch JV, Nussenzweig MC. Broad diversity of neutralizing antibodies isolated from memory B cells in HIV-infected individuals. Nature. 2009;458:636–640. doi: 10.1038/nature07930. [DOI] [PubMed] [Google Scholar]
- 64.Weiss GE, Crompton PD, Li S, Walsh LA, Moir S, Traore B, Kayentao K, Ongoiba A, Doumbo OK, Pierce SK. Atypical memory B cells are greatly expanded in individuals living in a malaria-endemic area. J Immunol. 2009;183:2176–2182. doi: 10.4049/jimmunol.0901297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Roughan JE, Reardon KM, Cogburn KE, Quendler H, Pockros PJ, Law M. Chronic hepatitis C virus infection breaks tolerance and drives polyclonal expansion of autoreactive B cells. Clinical and vaccine immunology: CVI. 2012;19:1027–1037. doi: 10.1128/CVI.00194-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tew JG, Phipps RP, Mandel TE. The maintenance and regulation of the humoral immune response: persisting antigen and the role of follicular antigen-binding dendritic cells as accessory cells. Immunological reviews. 1980;53:175–201. doi: 10.1111/j.1600-065x.1980.tb01044.x. [DOI] [PubMed] [Google Scholar]
- 67.Bernasconi NL, Traggiai E, Lanzavecchia A. Maintenance of serological memory by polyclonal activation of human memory B cells. Science. 2002;298:2199–2202. doi: 10.1126/science.1076071. [DOI] [PubMed] [Google Scholar]
- 68.Maruyama M, Lam KP, Rajewsky K. Memory B-cell persistence is independent of persisting immunizing antigen. Nature. 2000;407:636–642. doi: 10.1038/35036600. [DOI] [PubMed] [Google Scholar]
- 69.Bachmann MF, Odermatt B, Hengartner H, Zinkernagel RM. Induction of long-lived germinal centers associated with persisting antigen after viral infection. J Exp Med. 1996;183:2259–2269. doi: 10.1084/jem.183.5.2259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Gray D. A role for antigen in the maintenance of immunological memory. Nature reviews Immunology. 2002;2:60–65. doi: 10.1038/nri706. [DOI] [PubMed] [Google Scholar]
- 71.Jameson SC, Masopust D. Diversity in T cell memory: an embarrassment of riches. Immunity. 2009;31:859–871. doi: 10.1016/j.immuni.2009.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Postigo A, Corbi A, Sanchez-Madrid F, Landazuri M. Regulated expression and function of CD11c/CD18 integrin on Human B Lymphocytes. Relation between attachment to fibringogen and triggering of proliferation through CD11c/CD18. J Exp Med. 1991;174:1313–1322. doi: 10.1084/jem.174.6.1313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ehrhardt GR, Hijikata A, Kitamura H, Ohara O, Wang JY, Cooper MD. Discriminating gene expression profiles of memory B cell subpopulations. J Exp Med. 2008;205:1807–1817. doi: 10.1084/jem.20072682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Rubtsov AV, Rubtsova K, Fischer A, Meehan RT, Gillis JZ, Kappler JW, Marrack P. TLR7-driven accumulation of a novel CD11c+ B-cell population is important for the development of autoimmunity. Blood. 2011;118:1305–1311. doi: 10.1182/blood-2011-01-331462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Rubtsov AV, Rubtsova K, Kappler JW, Marrack P. TLR7 drives accumulation of ABCs and autoantibody production in autoimmune-prone mice. Immunol Res. 2013;55:210–216. doi: 10.1007/s12026-012-8365-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sadhu C, Ting HJ, Lipsky B, Hensley K, Garcia-Martinez LF, Simon SI, Staunton DE. CD11c/CD18: novel ligands and a role in delayed-type hypersensitivity. J Leukoc Biol. 2007;81:1395–1403. doi: 10.1189/jlb.1106680. [DOI] [PubMed] [Google Scholar]
- 77.Lu TT, Cyster JG. Integrin-mediated long-term B cell retention in the splenic marginal zone. Science. 2002;297:409–412. doi: 10.1126/science.1071632. [DOI] [PubMed] [Google Scholar]
- 78.Lo CG, Lu TT, Cyster JG. Integrin-dependence of lymphocyte entry into the splenic white pulp. J Exp Med. 2003;197:353–361. doi: 10.1084/jem.20021569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Colgan SP, Eltzschig HK, Eckle T, Thompson LF. Physiological roles for ecto-5′-nucleotidase (CD73) Purinergic Signal. 2006;2:351–360. doi: 10.1007/s11302-005-5302-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Thompson LF, Ruedi JM, O’Connor RD, Bastian JF. An explanation for the heterogeneity in B lymphocyte ecto-5′-nucleotidase activity in patients with hypogammaglobulinemia. Adv Exp Med Biol. 1986;195(Pt B):399–405. doi: 10.1007/978-1-4684-1248-2_63. [DOI] [PubMed] [Google Scholar]
- 81.Allen CD, Ansel KM, Low C, Lesley R, Tamamura H, Fujii N, Cyster JG. Germinal center dark and light zone organization is mediated by CXCR4 and CXCR5. Nature immunology. 2004;5:943–952. doi: 10.1038/ni1100. [DOI] [PubMed] [Google Scholar]
- 82.Hargreaves DC, Hyman PL, Lu TT, Ngo VN, Bidgol A, Suzuki G, Zou YR, Littman DR, Cyster JG. A coordinated change in chemokine responsiveness guides plasma cell movements. J Exp Med. 2001;194:45–56. doi: 10.1084/jem.194.1.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhong X, Tumang JR, Gao W, Bai C, Rothstein TL. PD-L2 expression extends beyond dendritic cells/macrophages to B1 cells enriched for V(H)11/V(H)12 and phosphatidylcholine binding. Eur J Immunol. 2007;37:2405–2410. doi: 10.1002/eji.200737461. [DOI] [PubMed] [Google Scholar]
- 84.Benson MJ, Dillon SR, Castigli E, Geha RS, Xu S, Lam KP, Noelle RJ. Cutting edge: the dependence of plasma cells and independence of memory B cells on BAFF and APRIL. J Immunol. 2008;180:3655–3659. doi: 10.4049/jimmunol.180.6.3655. [DOI] [PubMed] [Google Scholar]
- 85.Tangye SG, Good KL. Human IgM+CD27+ B cells: memory B cells or “memory” B cells? J Immunol. 2007;179:13–19. doi: 10.4049/jimmunol.179.1.13. [DOI] [PubMed] [Google Scholar]
- 86.Toyama H, Okada S, Hatano M, Takahashi Y, Takeda N, Ichii H, Takemori T, Kuroda Y, Tokuhisa T. Memory B cells without somatic hypermutation are generated from Bcl6-deficient B cells. Immunity. 2002;17:329–339. doi: 10.1016/s1074-7613(02)00387-4. [DOI] [PubMed] [Google Scholar]
- 87.Seifert M, Kuppers R. Molecular footprints of a germinal center derivation of human IgM+(IgD+)CD27+ B cells and the dynamics of memory B cell generation. J Exp Med. 2009;206:2659–2669. doi: 10.1084/jem.20091087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kost CB, Holden JT, Mann KP. Marginal zone B-cell lymphoma: a retrospective immunophenotypic analysis. Cytometry B Clin Cytom. 2008;74:282–286. doi: 10.1002/cyto.b.20426. [DOI] [PubMed] [Google Scholar]
- 89.Molica S, Dattilo A, Mannella A, Levato D. CD11c expression in B-cell chronic lymphocytic leukemia. A comparison of results obtained with different monoclonal antibodies. Haematologica. 1994;79:452–455. [PubMed] [Google Scholar]
- 90.Schwarting R, Stein H, Wang CY. The monoclonal antibodies alpha S-HCL 1 (alpha Leu-14) and alpha S-HCL 3 (alpha Leu-M5) allow the diagnosis of hairy cell leukemia. Blood. 1985;65:974–983. [PubMed] [Google Scholar]