Abstract
The investigation and development of the cancer stem cell (CSC) model has received much focus during these years. CSC is characterized as a small fraction of cancer cells that have an indefinite ability for self-renewal and pluripotency and are responsible for initiating and sustaining of the bulk of cancer. So, whether current treatment strategies, most of which target the rapid division of cancer cells, could interfere with the slow-cycling CSCs is broadly questioned. Meanwhile, however, the new understanding of tumorigenesis has led to the development of new drug screening strategies. Both stem cells and mesenchymal stem cells have been vigorously used in pre-clinical studies of their anti-tumor potential, mainly due to their inherent tumoritropic migratory properties and their ability to carry anti-tumor transgenes. Here, based on the tumorigenic and tumoritropic characteristics of CSCs, we proposed two hypotheses exploring possible usage of CSCs as novel anti-tumor agents and potential sources for tissue regeneration. Further experimental validation of these hypotheses may unravel some new research topics.
Keywords: Cancer stem cells, Cancer therapy, Hypotheses, Mesenchymal stem cell
Evil enters like a needle and spreads like an oak tree. - by Proverb, Ethiopian
Introduction
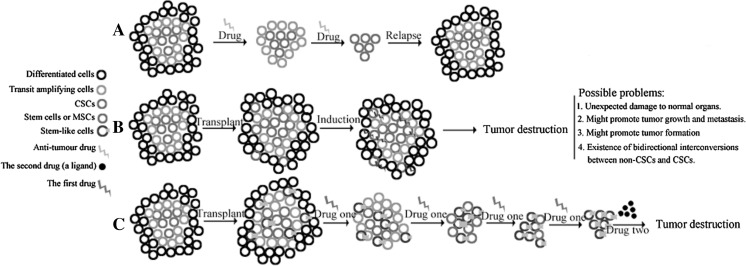
Hierarchical organization of human acute myeloid leukemia (AML) was proposed first by John Dick and colleagues in 1994 and since then similar models were identified for many solid tumors (Lapidot et al. 1994; Visvader and Lindeman 2008). Central to the heterogeneity of human malignancies is the existence of cancer stem cells (CSCs), a rare fraction of slow-cycling cancer cells that have the ability of self-renewal, pluripotency and chemo-resistance. In the presumptive CSC model, only the CSCs are tumorigenic and can produce all of the cells necessary to repopulate a tumor, whereas the bulk of the tumor is comprised of cells that are differentiated and have lost tumorigenic potential. The conceptual similarity between normal tissue-specific stem cells and CSCs implicates that a tumor is an abnormal organ composed of multiple cell types and maintained by pluripotent stem cells (Shackleton 2010; Reya et al. 2001). The existence of CSCs has substantial therapeutic implications, since it well explains why conventional chemo- and radio-therapy, most of which target the rapid division of cancer cells, cannot eradicate tumors and are frequently accompanied by lethal recurrence as shown in Fig. 1a (Jordan et al. 2006). So, there is a recent surge in developing ways to eliminate the CSCs. It was reported recently that dysfunctional proliferation and differentiation of CSCs required the dysregulation of evolutionally conserved mechanisms that were required for the self-renewal and pluripotency of embryonic stem cells (Takebe et al. 2011). This fact has significant therapeutic implications, since a targeted therapy against CSCs may also be hostile to normal tissue stem cells and interfere with homeostatic maintenance of organs and tissues (Bonnett and Dick 1997). Evil enters like a needle and spreads like an oak tree. This proverb well depicts the tumorigenic process as implicated by the CSC model.
Fig. 1.
Schematic representation of possible ways to cure cancer. a The CSC model pointed out that tumors are heterogeneous and are regarded as perturbed versions of the normal tissue, containing a small fraction of drug-resistant CSCs that are responsible for high frequency of relapse after chemotherapy. b Stem cells and in particular MSCs have been experimentally used and clinically explored for efficient delivery of therapeutic transgenes. However, conflicting reports showed that in some cases these cells could promote the metastasis and growth of a tumor. The therapeutic uncertainty of this treatment strategy stalls its clinical application. Our hypothesis is illustrated in c. In this treatment strategy, the stem-like cells transplanted resemble the ‘cancer cells’ of the abnormal tissue–the tumor. In the presence of the drug one, which acts as an environmental pressure favoring the self-renewal of the transplanted cells over that of CSCs, transplanted cells gradually take over the CSC niche. Then, the drug two, which could induce the expression of the incorporated suicide genes in the stem-like cells, kills the progenies of those seeded cells. Since in our model CSCs are replaced by other stem-like cells rather than killed by environmentally hostile agents to kill cancer cells, this strategy may circumvent the shortcomings of treatment methods as illustrated in a and b
In the CSC model, the bulk of a tumor could be regarded as an abnormal organ that expanded constantly. Then, could we also destroy the abnormal ‘organ’ with a spreading ‘evil needle’ as CSCs do to normal organs? Recently, the strategy of stem cell based drug delivery vehicles was proven more efficacious and with lower cytotoxicity, compared with traditional systematic delivery of drug or gene therapy. Theoretically, stem cells migrate towards and engraft into the tumor sites that provide the micro-environment amenable for them to survive. However, controversies and concerns remain regarding the applicability of this treatment method. In this mini-review, we discussed the basic scenario of the CSC model and its implication for future stem cell based cancer treatment. Based on the initial understanding of CSCs’ character, we hypothesize/propose two novel yet promising strategies that might shed some light on future anti-tumor research.
The evolving concept of the cancer stem cell model
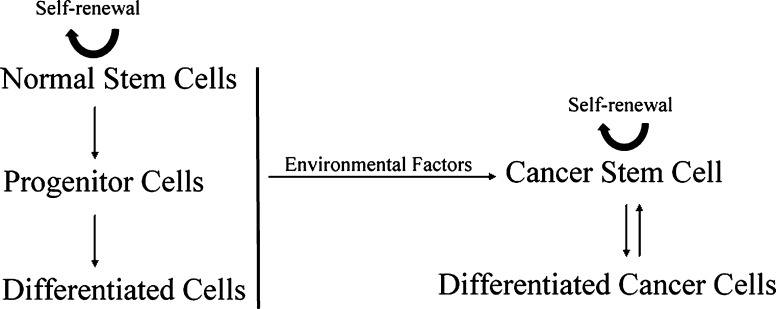
Despite the clonal origin of many cancers, primary tumors are heterogeneous comprised by multiple distinct cell types that arise from a sub-population of pluripotent cancer cells named CSCs or tumor-initiating cells. However, given some intrinsic unresolved issues of the CSC hypothesis, the validity of this model remains a topic of considerable controversy (Rosen and Jordan 2009). For one thing, the precise identification or definition of CSCs is not clear. According to a consensus definition, CSCs can only be defined experimentally by their ability to recapitulate the generation of a continuously growing tumor (Clarke et al. 2006). The concomitant problem of this definition is that the empirically tumorigenic potential of serial transplantation in animal models is also affected by factors such as the tissue site of xenotransplantation and the immune status of the tumor host mice (Cho and Clarke 2008). So, it is possible that a subpopulation of cancer cells is tumorigenic but cannot form xenografts due to inappropriate micro-environment, rendering the frequency of CSCs in tumor tissue highly variable depending on the specific protocols used and procedures undertaken. Of note, the proportion of CSCs in the tumor mass varied remarkably among patients and CSC-rich was believed to be associated with aggressive tumor progression and poor prognosis (Ailles and Weissman 2007; Quintana et al. 2008). A more operational definition of CSC and a more standardized way to identify them are pivotal for future tumor diagnosis and treatment. For this, CSC-specific surface biomarkers that allow antibody-directed cell sorting constitute a promising candidate. However, at present, CSC biomarkers for most cancer cell lines remain largely elusive and no universal biomarker for CSC has yet been identified (Visvader and Lindeman 2008). It was reported that AML cells with CD34+CD38− surface antigens were highly tumorigenic when transplanted into nonobese diabetic severe combined immunodeficiency disease (NOD/SCID) mice. In a breast cancer model, however, cells with CD44+CD24-lineage negative phenotype were believed as CSCs (Bonnett and Dick 1997; Al-Hajj et al. 2003). More recently, CD44+ and CD133+ were identified on the surface of gastric and lung CSCs, respectively (Eramo et al. 2008; Takaishi et al. 2009). It is difficult to find a universal CSC marker for tumors derived from different tissues or even from the same tissue, which implicates that different lineages of CSCs may have host-specific tumorigenic mechanisms. The variability in CSC properties introduces problems when developing new anti-tumor drugs (Hwang-Verslues et al. 2009). Another major issue of the CSC model is the origin of these stem-like cells. So, it was proposed that CSCs might have originated from normal stem cells, early stem cell progenitors or normal differentiated cells in a unidirectional manner (Bjerkvig et al. 2005). However, an unexpected new origin of CSCs was identified recently by Chaffer et al. (2011). They found that transformed human CD44lo cancerous mammary epithelial cells could spontaneously dedifferentiate into CD44hi-CSCs in vivo, indicating the existence of bidirectional inter-conversions between CSC and non-CSC cancer cells. This seminal finding implicates that tumors may regenerate CSCs under the treatment that exclusively target CSCs, compromising the therapeutic significance of CSC specific agents that have been screened (Gupta et al. 2009). The CSC model for tumor progression is illustrated in Fig. 2. Mainly due to the issues outlined above, criticisms concerning the proper conceptualization and nomination of CSCs exist (Maenhaut et al. 2010). However, no one would doubt that the concept of CSCs is a giant leap towards a mechanistic understanding of human malignancies and may lead to a new era of anti-tumor therapy.
Fig. 2.
Possible origins of cancer stem cells. The cancer stem cell might be derived from normal stem cells, early stem cell progenitors or differentiated cells (Bjerkvig et al. 2005). Most recent studies showed differentiated cancer cells could dedifferentiate spontaneously into cancer stem cells (Chaffer et al. 2011)
Homeostatic tissue maintenance and tissue regeneration after injury depend on the delicate balance between the two cardinal characteristics of stem cells that are to differentiate, or to produce specialized cells that comprise the tissue, and to self-renew, or to make more stem cells. The existence of a physiological micro-environment or a niche that could maintain stem cell stemness and sequester them away from differentiation and apoptotic stimuli while protecting the host from excessive stem-cell proliferation was envisaged. Although the existence of stem cell niches has been identified in multiple mammalian tissues, we have little affirmative information about how signaling pathways/bio-factors mediate the interactions between stem cells and the niche (Voog and Jones 2010). However, given that the CSC is intimately associated with its micro-environment, targeting this niche may become a desirable therapeutic target. Anti-angiogenic drugs like thalidomide, bevacizumab and the protease inhibitor bortezomib were proposed to exert their anti-tumor effect by modulating the micro-environment (Burness and Sipkins 2010).
We hypothesize/propose here two potential therapeutic and/or experimental methodologies that might hold some therapeutic significance.
Proposal 1. Cure cancer by replacing CSCs with implanted stem-like cells?
At present, tissue derived stem cells have been largely used in pre-clinical studies of their anti-tumor potential, since these stem cells or mesenchymal stem cells (MSC) can act as vehicles for targeted delivery and releasing of anti-tumor factors, such as anti-angiogenic, cytotoxic and immunostimulatory factors (Shah 2012). However, the major hurdle that thwarts the clinical application of this strategy is the inherent uncertainty after the injection of these stem cells or MSCs. For example, the hostile anti-tumor compounds might also unpredictably harm normal organs, and interleukin-6 (IL-6) and chemokine (C–C motif) ligand 5 (CCL5) endogenously expressed by MSCs were reported to promote the proliferation and metastasis of breast cancer cells (Karnoub et al. 2007; Albarenque et al. 2011). Also, MCSs were reported to promote formation of colorectal tumors (Tsai et al. 2011). Moreover, given the fact that non-CSCs can spontaneously dedifferentiate into CSCs, it is even more difficult to eradicate CSCs by killing them (Gupta et al. 2009). The bidirectional inter-conversions between CSCs and non-CSCs may enable tumors to maintain a relatively constant number of CSCs under treatment (Fig. 2).
Potentially, stem cells derived from the patients’ own tissue would not provoke the immune system. However, most recent pre-clinical studies showed contradictory results. Zhao et al. (2011) found that induced pluripotent stem (iPS) cells could trigger severe immune reactions after injection into the recipient from whom the iPSCs were derived. This finding seriously limits cell types that could be engineered as vehicles for transgenes and we categorized possible limitations of this strategy in Fig. 1b.
Considering all these limitations that seriously limit the applicability of the strategy of using stem cells as drug delivery vehicles, we proposed an alternative strategy as illustrated in Fig. 1c. In this treatment strategy, the replacement of CSCs by the seeded stem-like cells is induced by the presence of environmental stress i.e. ‘drug one’. Technically, it is accomplishable to favor the growth of one cell line while killing other cell lines. For example, we can incorporate the tetR gene into the genome of the seeding cells and at the presence of tetracycline, which is used clinically in treating infections, the seeded stem-like cells may overgrow the CSCs (Speer et al. 1992; Matthess et al. 2005). Subsequently, after successfully replacement, the seeded stem-like cells could be eradicated by adding ‘drug two’ which would activate the suicide genes incorporated before seeding. One possible system fits this scenario was developed by Dong et al. (2004). They found that the solid tumor mass formed by the breast cancer cells, which were genetically modified to incorporate the Grp78 promoter driven HSV-tk suicide gene, could be completely eradicated with the treatment of drug ganciclovir (GCV), which is in clinical use for treating cytomegalovirus (CMV) infections. To make sure that all the progenies of the seed cells could be killed upon induction, multiple suicide genes, flanked by strong controllable promoters, should be recombined into the genome of these stem-like cells before transplantation. In all these procedures, it is not necessary for the seeded cells to secrete hostile anti-tumor substances and consequently, it may reduce therapeutic uncertainty. This strategy may be advantageous given that it incorporates the benefits of both traditional chemo-therapy and the most recent stem cell based drug delivery system.
We hypothesize that this strategy has higher tumor specificity and lower cytotoxicity. Empirically, the seeded cells could be regarded as the ‘needle’ that enters the abnormal ‘organ’ and kill the ‘organ’ by replacing and depriving its initiating cells. The key to the success of this proposal/hypothesis is whether those injected stem-like cells could actually seed in the tumor mass and then replace CSCs in the presence of drug one, which we believe offers survival advantage for injected stem-like cells over the original CSCs (Fig. 1). Recent studies by Kim et al. (2009) showed that mainly mediated by tumor-derived attractants IL-6, IL-8 and proteins matrix metalloproteinase-1 (MMP-1) and fascin-1, circulating tumor cells (CTCs) can re-seed their tumors of origin. The process of tumor-self seeding was believed a process that may select more aggressive cancer cells for the tumor colonization and breeding of more metastatic cancer cell progenies (Kim et al. 2009; Hahnfeldt 2010; Dondossola et al. 2012). Given the tumor formation ability of CTCs and the fact that CSCs may constitute the majority of CTCs, it is reasonable to believe that the re-infiltrated CTCs may have some stem-like properties (Toloudi et al. 2011). However, the amount of stem-like cells is limited by the availability of the niche that provides essential signals maintaining the self-renewal and pluripotency ability of these stem-like cells. Then, some sort of competition may exist between the more aggressive re-seeding CTCs and the original residing CSCs for the limited niche, as what happens between CTCs and the hematopoietic stem cells (HSCs) when CTCs try to establish footholds in bone marrow (Shiozawa et al. 2011). This kind of competition for the limited stem cell niche constitutes the major foundation of our hypothesis and we reasonably hold that in the presence of drug one that favors the out-growth of the engineered stem-like cells, injected stem-like cells may finally replace the original CSCs in the niche, as depicted in Fig. 1.
Proposal 2. Manipulate the CSCs to our advantage: creating functional organs from CSCs?
In the last decade, conceptual progresses in cancer biology highlighted another emerging hallmark of human malignancies-their ability to evade immune destruction (Hanahan and Weinberg 2011). In this case, the so-called donor-related malignancy, in which immunosuppressed organ transplant recipients could potentially develop donor derived cancers, indicating that in immunocompetent donors the cancer cells were held in a dormant state by a fully functional immune system, whereas in the immunosuppressed recipients these cancer cells could be reactivated and progress (Strauss and Thomas 2010). Theoretically, cancer cells paralyze anti-tumor immune surveillance by secreting immunosuppressive factors like TGF-β and chemokine ligand (CCL21), by recruiting inflammatory cells like regulatory T cells (Tregs) and myeloid-derived suppressor cells or by evading the evolutionally deprived immune system of the host (Hanahan and Weinberg 2011; Ostrand-Rosenberg and Sinha 2009; Mougiakakos et al. 2010; Shields et al. 2010; Yang et al. 2010).
Although not well understood, recent studies suggested that CSCs possessed the functional and phenotypic characteristics that helped cancer cells evade host antitumor immunity. For example, a novel type of CSCs, ABCB5+ malignant melanoma initiating cells (MMIC) were reported refractory to current immunotherapeutic treatment strategies (Schatton and Frank 2009). And interestingly, CSCs derived from glioblastoma multiform patients could suppress immune responses by impeding T-cell proliferation and activation, inducing T-cell apoptosis and inhibiting the phagocytosis ability of MΦs/microglia (Wei et al. 2010a, b; Wu et al. 2010).
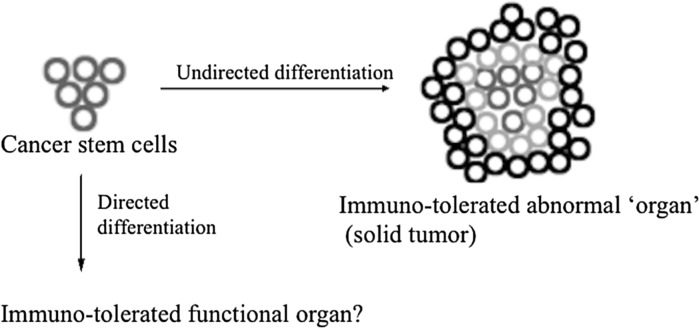
Two hallmarks of CSCs are revealed here: CSCs are pluripotent stem-like cells with self-renewal ability and they are immune suppressive. The ideal combination of these two characteristics promises significant therapeutic implications beyond cancer therapy and we propose/hypothesize here that CSCs could be potentially engineered to create functional tissues and organs for transplantation (Fig. 3). Ideally, these organs are immune-tolerated by universal recipients. Here we tried to define the word ‘universal’ as ‘a group of individuals of the same species’. This definition constitutes the major highlight of this proposal.
Fig. 3.
Schematic representation illustrating proposal 2. Given that cancer stem cells have characteristics of normal stem cells, we hypothesize that we could possibly direct the differentiation of CSCs to normal organs. Since cancer cells could efficiently evade the immuno-surveillance of the host, we propose that the functional organ might also be well tolerated by the recipients, even though those CSCs are derived from other individuals
Technically, directed differentiation of normal embryonic and iPS cells into specific organ cell types is relatively well studied at least for certain tissues. For instance, iPS cells had been engineered to differentiate into liver hepatocytes and pancreatic endocrine (Cai et al. 2007; D’Amour et al. 2006; Song et al. 2009; Zhang et al. 2009). Moreover, a recent study reported the successful directed differentiation of human iPS cells into functional intestinal tissue in vitro by manipulating a temporal series of growth factors (Spence et al. 2011). CSCs could also be differentiated into functional cells, since in glioblastoma multiforme and medulloblastoma, stem-like neurosphere-forming glioma stem cells were manipulated to give rise to neuronal and astroglial-like cells (Hemmati et al. 2003; Singh et al. 2003). In this context, the so-call differentiation therapy is now widely studied. Retinoic acid (RA, Vitamin A), for example, is one of the most thoroughly studied drugs that are capable of inducing CSC differentiation (Sell 2004; Tang and Gudas 2011; Gudas and Wagner 2011). However, mainly due to retinoid resistance and some serious side effects, the clinical outcome of RA treatment is not satisfactory for many solid tumors (Sell 2004). On another side, given their pluripotent and immunosuppressive properties, it might be possible to produce CSCs into functional organs for clinical transplantation. So, instead of killing them, we foresee here a promising application of these novel stem-like cells to the advantage of our healthcare.
Conclusion and prospects
Drugs that target CSCs implicate enormous therapeutic potential for cancer therapy. However, this enthusiasm needs to be tempered. The stark reality that drugable signaling pathways, such as Wnt, Hedgehog, and Notch, that are aberrantly modulated in CSCs are also critical for normal adult stem cell maintenance calls for re-evaluation of current drug-screening methods. However, it is becoming increasingly apparent that the concept of CSCs in tumor progression highlights the significance of stem cell based drug delivery system. In spite of that, the nomination or even the validity of the CSC hypothesis remains critically questioned, mainly due to the problems discussed in this article, the concept is a great leap forward on how to detect the process of tumorigenesis, how to develop efficient therapies and most importantly, if the tumor colonies are maintained by the rare CSCs, and how to prevent their initiation in the first place. The rarity of CSCs in the cancer cell populations, their instability when cultured in vitro and their similarity with normal tissue stem cells thwart the identification and clinical application of CSC specific agents. Meanwhile, the clinical applicability of the stem cell based drug delivery system was seriously compromised by its inherent therapeutic uncertainty.
Instead of killing CSCs by environmental hostile agents, we proposed a novel stem cell based strategy in proposal 1 in which CSCs were replaced by implanted stem-like cells. By doing so, we could circumvent undesirable disadvantages that come along with stem cell based anti-tumor therapy. Moreover, can we manipulate the CSCs to our advantage? Based on pluripotent and immunosuppressive characteristics of CSCs, we hypothesized a novel application of CSCs in proposal 2. A rational question concerning the relationship between the proposal 1 and 2 is that when we know how to direct the differentiation of CSCs into functional tissue cells, why don’t we use a similar strategy to induce CSCs differentiation in vivo and then cure the cancer? Presumably, the directed differentiation of CSCs as highlighted in the proposal 2 is a multiple-step process, happens in a spatial and temporal manner, and consequently, and at least in the actual state of knowledge and development, the differentiation strategy may be inapplicable for cancer treatment in vivo, although, hopefully, this may lead to the development of other more potent drugs that are suitable for the differentiation therapy.
According to our knowledge, the proposals we provided here are not yet experimentally explored. Here, with the help of the world renowned journal, we provide these strategies for possible exploration.
Acknowledgments
This work was supported partially by “National Natural Science Foundation of China” (No.31272100 and No.30901774), the “National High Technology Research and Development Program of China (863 Program)” (No.2012AA020809), the Innovative Research Program for Graduates of Shanxi Province (20113018) and the Program for the Top Young Academic Leaders of Higher Learning Institutions of Shanxi.
Conflict of interest
The authors state that they have no conflict of interest.
References
- Ailles LE, Weissman IL. Cancer stem cells in solid tumors. Curr Opin Biotechnol. 2007;18:460–466. doi: 10.1016/j.copbio.2007.10.007. [DOI] [PubMed] [Google Scholar]
- Albarenque SM, Zwacka RM, Mohr A. Both human and mouse mesenchymal stem cells promote breast cancer metastasis. Stem Cell Res. 2011;7:163–171. doi: 10.1016/j.scr.2011.05.002. [DOI] [PubMed] [Google Scholar]
- Al-Hajj M, Wicha MS, Benito-Hernandez A, Morrison SJ, Clarke MF. Prospective identification of tumourigenic breast cancer cells. Proc Natl Acad Sci USA. 2003;100:3983–3988. doi: 10.1073/pnas.0530291100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bjerkvig R, Tysnes BB, Aboody KS, Najbauer J, Terzis AJ. Opinion: the origin of the cancer stem cell: current controversies and new insights. Nat Rev Cancer. 2005;5:899–904. doi: 10.1038/nrc1740. [DOI] [PubMed] [Google Scholar]
- Bonnett D, Dick JE. Human acute myeloid leukaemia is organized as a hierarchy that originates from a primate haematopoietic cell. Nat Med. 1997;3:730–737. doi: 10.1038/nm0797-730. [DOI] [PubMed] [Google Scholar]
- Burness ML, Sipkins DA. The stem cell niche in health and malignancy. Semin Cancer Biol. 2010;20:107–115. doi: 10.1016/j.semcancer.2010.05.006. [DOI] [PubMed] [Google Scholar]
- Cai J, Zhao Y, Liu Y, Ye F, Song Z, Qin H, Meng S, Chen Y, Zhou R, Song X, Guo Y, Ding M, Deng H. Directed differentiation of human embryonic stem cells into functional hepatic cells. Hepatology. 2007;45:1229–1239. doi: 10.1002/hep.21582. [DOI] [PubMed] [Google Scholar]
- Chaffer CL, Brueckmann I, Scheel C, Kaestli AJ, Wiggins PA, Rodrigues LO, Brooks M, Reinhardt F, Su Y, Polyak K, Arendt LM, Kuperwasser C, Bierie B, Weinberg RA. Normal and neoplastic nonstem cells can spontaneously convert to a stem-like state. Proc Natl Acad Sci USA. 2011;108:7950–7955. doi: 10.1073/pnas.1102454108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cho RW, Clarke MF. Recent advances in cancer stem cells. Curr Opin Genet Dev. 2008;18:48–53. doi: 10.1016/j.gde.2008.01.017. [DOI] [PubMed] [Google Scholar]
- Clarke MF, Dick JE, Dirks PB, Eaves CJ, Jamieson CH, Jones DL, Visvader J, Weissman IL, Wahl GM. Cancer stem cells–perspectives on current status and future directions: AACR workshop on cancer stem cells. Cancer Res. 2006;66:9339–9344. doi: 10.1158/0008-5472.CAN-06-3126. [DOI] [PubMed] [Google Scholar]
- D’Amour KA, Bang AG, Eliazer S, Kelly OG, Agulnick AD, Smart NG, Moorman MA, Kroon E, Carpenter M, Baetge EE. Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat Biotechnol. 2006;24:1392–1401. doi: 10.1038/nbt1259. [DOI] [PubMed] [Google Scholar]
- Dondossola E, Crippa L, Colombo B, Ferrero E, Corti A. Chromogranin A regulates tumor self-seeding and dissemination. Cancer Res. 2012;72:449–459. doi: 10.1158/0008-5472.CAN-11-2944. [DOI] [PubMed] [Google Scholar]
- Dong D, Dubeau L, Bading J, Nguyen K, Luna M, Yu H, Gazit-Bornstein G, Gordon EM, Gomer C, Hall FL, Gambhir SS, Lee AS. Spontaneous and controllable activation of suicide gene expression driven by the stress-inducible grp78 promoter resulting in eradication of sizable human tumors. Hum Gene Ther. 2004;15:553–561. doi: 10.1089/104303404323142006. [DOI] [PubMed] [Google Scholar]
- Eramo A, Lotti F, Sette G, Pilozzi E, Biffoni M, Di Virgilio A, Conticello C, Ruco L, Peschle C, De Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ. 2008;15:504–514. doi: 10.1038/sj.cdd.4402283. [DOI] [PubMed] [Google Scholar]
- Gudas LJ, Wagner JA. Retinoids regulate stem cell differentiation. J Cell Physiol. 2011;226:322–330. doi: 10.1002/jcp.22417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta PB, Onder TT, Jiang G, Tao K, Kuperwasser C, Weinberg RA, Lander ES. Identification of selective inhibitors of cancer stem cells by high-throughput screening. Cell. 2009;138:645–659. doi: 10.1016/j.cell.2009.06.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hahnfeldt P. Significance of tumor self-seeding as an augmentation to the classic metastasis paradigm. Future Oncol. 2010;6:681–685. doi: 10.2217/fon.10.43. [DOI] [PubMed] [Google Scholar]
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- Hemmati HD, Nakano I, Lazareff JA, Masterman-Smith M, Geschwind DH, Bronner-Fraser M, Kornblum HI. Cancerous stem cells can arise from pediatric brain tumors. Proc Natl Acad Sci USA. 2003;100:15178–15183. doi: 10.1073/pnas.2036535100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang-Verslues WW, Kuo WH, Chang PH, Pan CC, Wang HH, Tsai ST, Jeng YM, Shew JY, Kung JT, Chen CH, Lee EY, Chang KJ, Lee WH. Multiple lineages of human breast cancer stem/progenitor cells identified by profiling with stem cell markers. PLoS ONE. 2009;4:e8377. doi: 10.1371/journal.pone.0008377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan CT, Guzman ML, Noble M. Cancer Stem Cells. N Engl J Med. 2006;355:1253–1261. doi: 10.1056/NEJMra061808. [DOI] [PubMed] [Google Scholar]
- Karnoub AE, Dash AB, Vo AP, Sullivan A, Brooks MW, Bell GW, Richardson AL, Polyak K, Tubo R, Weinberg RA. Mesenchymal stem cells within tumour stroma promote breast cancer metastasis. Nature. 2007;449:557–563. doi: 10.1038/nature06188. [DOI] [PubMed] [Google Scholar]
- Kim MY, Oskarsson T, Acharyya S, Nguyen DX, Zhang XH, Norton L, Massagué J. Tumor self-seeding by circulating cancer cells. Cell. 2009;139:1315–1326. doi: 10.1016/j.cell.2009.11.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lapidot T, Sirard C, Vormoor J, Murdoch B, Hoang T, Caceres-Cortes J, Minden M, Paterson B, Caligiuri MA, Dick JE. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature. 1994;367:645–648. doi: 10.1038/367645a0. [DOI] [PubMed] [Google Scholar]
- Maenhaut C, Dumont JE, Roger PP, van Staveren WC. Cancer stem cells: a reality, a myth, a fuzzy concept or a misnomer? An analysis. Carcinogenesis. 2010;31:149–158. doi: 10.1093/carcin/bgp259. [DOI] [PubMed] [Google Scholar]
- Matthess Y, Kappel S, Spänkuch B, Zimmer B, Kaufmann M, Strebhardt K. Conditional inhibition of cancer cell proliferation by tetracycline-responsive, H1 promoter-driven silencing of PLK1. Oncogene. 2005;24:2973–2980. doi: 10.1038/sj.onc.1208472. [DOI] [PubMed] [Google Scholar]
- Mougiakakos D, Choudhury A, Lladser A, Kiessling R, Johansson CC. Regulatory T cells in cancer. Adv Cancer Res. 2010;107:57–117. doi: 10.1016/S0065-230X(10)07003-X. [DOI] [PubMed] [Google Scholar]
- Ostrand-Rosenberg S, Sinha P. Myeloid-derived suppressor cells: linking inflammation and cancer. J Immunol. 2009;182:4499–4506. doi: 10.4049/jimmunol.0802740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quintana E, Shackleton M, Sabel MS, Fullen DR, Johnson TM, Morrison SJ. Efficient tumour formation by single human melanoma cells. Nature. 2008;456:593–598. doi: 10.1038/nature07567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. 2001;414:105–111. doi: 10.1038/35102167. [DOI] [PubMed] [Google Scholar]
- Rosen JM, Jordan CT. The increasing complexity of the cancer stem cell paradigm. Science. 2009;324:1670–1673. doi: 10.1126/science.1171837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schatton T, Frank MH. Antitumor immunity and cancer stem cells. Ann N Y Acad Sci. 2009;1176:154–169. doi: 10.1111/j.1749-6632.2009.04568.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sell S. Stem cell origin of cancer and differentiation therapy. Crit Rev Oncol Hematol. 2004;51:1–28. doi: 10.1016/j.critrevonc.2004.04.007. [DOI] [PubMed] [Google Scholar]
- Shackleton M. Normal stem cells and cancer stem cells: similar and different. Semin Cancer Biol. 2010;20:85–92. doi: 10.1016/j.semcancer.2010.04.002. [DOI] [PubMed] [Google Scholar]
- Shah K (2012) Mesenchymal stem cells engineered for cancer therapy. Adv Drug Deliv Rev 64:739–748 [DOI] [PMC free article] [PubMed]
- Shields JD, Kourtis IC, Tomei AA, Roberts JM, Swartz MA. Induction of lymphoidlike stroma and immune escape by tumors that express the chemokine CCL21. Science. 2010;328:749–752. doi: 10.1126/science.1185837. [DOI] [PubMed] [Google Scholar]
- Shiozawa Y, Pedersen EA, Havens AM, Jung Y, Mishra A, Joseph J, Kim JK, Patel LR, Ying C, Ziegler AM, Pienta MJ, Song J, Wang J, Loberg RD, Krebsbach PH, Pienta KJ, Taichman RS. Human prostate cancer metastases target the hematopoietic stem cell niche to establish footholds in mouse bone marrow. J Clin Invest. 2011;121:1298–12312. doi: 10.1172/JCI43414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh SK, Clarke ID, Terasaki M, Bonn VE, Hawkins C, Squire J, Dirks PB. Identification of a cancer stem cell in human brain tumors. Cancer Res. 2003;63:5821–5828. [PubMed] [Google Scholar]
- Song Z, Cai J, Liu Y, Zhao D, Yong J, Duo S, Song X, Guo Y, Zhao Y, Qin H, Yin X, Wu C, Che J, Lu S, Ding M, Deng H. Efficient generation of hepatocyte-like cells from human induced pluripotent stem cells. Cell Res. 2009;19:1233–1242. doi: 10.1038/cr.2009.107. [DOI] [PubMed] [Google Scholar]
- Speer BS, Shoemaker NB, Salyers AA. Bacterial resistance to tetracycline: mechanisms, transfer, and clinical significance. Clin Microbiol Rev. 1992;5:387–399. doi: 10.1128/cmr.5.4.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spence JR, Mayhew CN, Rankin SA, Kuhar MF, Vallance JE, Tolle K, Hoskins EE, Kalinichenko VV, Wells SI, Zorn AM, Shroyer NF, Wells JM. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature. 2011;470:105–109. doi: 10.1038/nature09691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss DC, Thomas JM. Transmission of donor melanoma by organ transplantation. Lancet Oncol. 2010;11:790–796. doi: 10.1016/S1470-2045(10)70024-3. [DOI] [PubMed] [Google Scholar]
- Takaishi S, Okumura T, Tu S, Wang SS, Shibata W, Vigneshwaran R, Gordon SA, Shimada Y, Wang TC. Identification of gastric cancer stem cells using the cell surface marker CD44. Stem Cells. 2009;27:1006–1020. doi: 10.1002/stem.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takebe N, Harris PJ, Warren RQ, Ivy SP. Targeting cancer stem cells by inhibiting Wnt, Notch, and Hedgehog pathways. Nat Rev Clin Oncol. 2011;8:97–106. doi: 10.1038/nrclinonc.2010.196. [DOI] [PubMed] [Google Scholar]
- Tang XH, Gudas LJ. Retinoids, retinoic acid receptors, and cancer. Annu Rev Pathol. 2011;6:345–364. doi: 10.1146/annurev-pathol-011110-130303. [DOI] [PubMed] [Google Scholar]
- Toloudi M, Apostolou P, Chatziioannou M, Papasotiriou I. Correlation between cancer stem cells and circulating tumor cells and their value. Case Rep Oncol. 2011;4:44–54. doi: 10.1159/000324403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai KS, Yang SH, Lei YP, Tsai CC, Chen HW, Hsu CY, Chen LL, Wang HW, Miller SA, Chiou SH, Hung MC, Hung SC. Mesenchymal stem cells promote formation of colorectal tumors in mice. Gastroenterology. 2011;141:1046–1056. doi: 10.1053/j.gastro.2011.05.045. [DOI] [PubMed] [Google Scholar]
- Visvader JE, Lindeman GJ. Cancer stem cells in solid tumours: accumulating evidence and unresolved questions. Nat Rev Cancer. 2008;8:755–768. doi: 10.1038/nrc2499. [DOI] [PubMed] [Google Scholar]
- Voog J, Jones DL. Cell stem cell—stem cells and the niche: a dynamic duo. Cell Stem Cell. 2010;6:103–115. doi: 10.1016/j.stem.2010.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei J, Barr J, Kong LY, Wang Y, Wu A, Sharma AK, Gumin J, Henry V, Colman H, Priebe W, Sawaya R, Lang FF, Heimberger AB. Glioblastoma cancer-initiating cells inhibit T-cell proliferation and effector responses by the signal transducers and activators of transcription 3 pathway. Mol Cancer Ther. 2010;9:67–78. doi: 10.1158/1535-7163.MCT-09-0734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei J, Barr J, Kong LY, Wang Y, Wu A, Sharma AK, Gumin J, Henry Hyperlink V, Colman H, Sawaya R, Lang FF, Heimberger AB. Glioma-associated cancer-initiating cells induce immunosuppression. Clin Cancer Res. 2010;16:461–473. doi: 10.1158/1078-0432.CCR-09-1983. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Wu A, Wei J, Kong LY, Wang Y, Priebe W, Qiao W, Sawaya R, Heimberger AB. Glioma cancer stem cells induce immunosuppressive macrophages/microglia. Neuro Oncol. 2010;12:1113–1125. doi: 10.1093/neuonc/noq082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang L, Pang Y, Moses HL. TGF-beta and immune cells: an important regulatory axis in the tumor microenvironment and progression. TGF-beta and immune cells: an important regulatory axis in the tumor microenvironment and progression. Trends Immunol. 2010;31:220–227. doi: 10.1016/j.it.2010.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang D, Jiang W, Liu M, Sui X, Yin X, Chen S, Shi Y, Deng H. Highly efficient differentiation of human ES cells and iPS cells into mature pancreatic insulin-producing cells. Cell Res. 2009;19:429–438. doi: 10.1038/cr.2009.28. [DOI] [PubMed] [Google Scholar]
- Zhao T, Zhang ZN, Rong Z, Xu Y. Immunogenicity of induced pluripotent stem cells. Nature. 2011;474:212–215. doi: 10.1038/nature10135. [DOI] [PubMed] [Google Scholar]