Abstract
Background
MicroRNAs (miRNAs, miRs) are involved in most physiological, developmental, and pathological processes. miR-192 and miR-205 are expressed preferentially in the renal cortex and closely relevant to the renal cell biology. In the present study, we aim to measure the serum levels of miR-192 and miR-205 and their correlation with clinicopathological data in patients with primary focal segmental glomerulosclerosis (FSGS) and minimal change disease (MCD).
Methods
Fifty-six patients (35 male, 21 female) with idiopathic nephrotic syndrome (FSGS 30, MCD 26) and 20 healthy controls were enrolled in the study. We quantified the serum levels of miR-192 and miR-205 in patients with FSGS and MCD by RT-qPCR.
Results
Patients with FSGS had higher serum levels of miR-192 and miR-205 than those with MCD (324.49 ± 42.74 fmol/l versus 90.19 ± 27.14 fmol/l, p < 0.01, 2.25 ± 0.69 fmol/l versus 0.60 ± 0.51 fmol/l, p < 0.01, respectively). The level of miR-192 was positively correlated with the proteinuria in patients with FSGS and MCD (r = 0.62, p < 0.001, r = 0.84, p < 0.001, respectively). Similarly, the level of miR-205 was positively correlated with the proteinuria in patients with FSGS (r = 0.54, p = 0.002). In addition, the serum level of miR-192 was significantly correlated with the degree of interstitial fibrosis in patients with FSGS (r = 0.342, p < 0.05).
Conclusions
miR-192 and miR-205 have the potential as markers to differentiate FSGS from MCD.
Keywords: Focal segmental glomerulosclerosis, Minimal change disease, Nephrotic syndrome, miRNAs
Introduction
Primary focal segmental glomerulosclerosis (FSGS) and minimal change disease (MCD), lumped together as podocytopathy, are both characterized by heavy proteinuria and the nephrotic syndrome (NS) as well as extensive flattening of podocyte foot process in ultrastructural morphology [1]. However, FSGS often shows lesions as glomerulosclerosis, tubular atrophy, and interstitial fibrosis, followed by therapeutic dilemma, steroid resistance, which shows a tendency to process to end-stage renal disease (ESRD) [2, 3]. Currently, clinicians predict the severity and prognosis of the idiopathic nephrotic syndrome in children through their response to steroids, rather than through kidney biopsy, which is the precise approach to distinguish MCD from FSGS. The consequence of an imprecise diagnosis in these children is exposure to the high risk of morbidity due to immunosuppression [4]. To our knowledge, there has not been a reliable biomarker for FSGS and MCD.
MicroRNAs (miRNAs, miRs) appear to play roles in most physiological, developmental, and pathological processes. Many of the miRNAs can be found in extracellular fluid such as plasma, serum [5, 6], and urine [7] in addition to tissues and cells. miRNAs are relatively stable, which makes them ideal as biomarkers for disease. Although plenty of miRNAs are widely expressed in many tissues, some miRNAs are found to be highly organ-specific [8]. miR-192 and miR-205 are expressed preferentially in the renal cortex and closely relevant to the renal cell biology [9, 10]. miR-192 and miR-205 are more abundant in the kidney than other organs(rat) [10]. Wang et al. [11] showed miR-192 and miR-205 were found elevated in renal tissues of patients with hypertensive glomerulosclerosis. In the present study, we examine the serum levels of miR-192 and miR-205 and their correlation with clinicopathological data.
Patients and methods
Patient cohort and blood collection
Fifty-six children and adolescents (31 male, 25 female) diagnosed with active idiopathic nephrotic syndrome based on the International Study of Kidney Disease in Children [12] were enrolled in the study. Thirty patients with FSGS and 26 patients with MCD were confirmed by kidney biopsy between March 2011 and April 2012 at the Department of Pediatrics, Jinling Hospital, Nanjing, China. We selected another 20 healthy, age- and sex-matched individuals (12 male, 8 female) as controls. Demographic and clinical data (urinalysis, biochemical results, response to steroid, duration of disease, and current treatment) were obtained at the time of enrollment. Patients with congenital nephrotic syndrome, secondary nephrotic syndrome such as systemic lupus erythematosus (SLE), and idiopathic nephrotic syndrome in remission status were excluded. Steroid-dependent nephrotic syndrome (SDNS) was defined when a patient had two consecutive relapses during corticosteroid therapy, or within 14 days of ceasing therapy. Steroid-dependent nephrotic syndrome (SRNS) was defined as no response to the initial 8 weeks of corticosteroid treatment. Blood samples obtained from all participants were processed immediately. Serum samples were separated by a 15-min centrifugation at 3,000 rpm and then stored at −70 °C until analysis.
Total RNA extraction and quantification
Total RNA was extracted from each serum sample (100 μl) using TRIzol Reagent (Invitrogen, Carlsbad, CA, USA) in accordance with the manufacturer’s protocols.
We used a TaqMan PCR kit (Applied Biosystems, Foster City, CA, USA) to carry out assays following the manufacturer’s instructions. In brief, 2 μl of total RNA was reverse transcribed to cDNA using a specific stem-loop RT primer (Applied Biosystems) under the condition of 16 °C for 30 min, 42 °C for 30 min, and 85 °C for 5 min. RT–QPCR was performed at 95 °C for 5 min, followed by 40 cycles at 95 °C for 15 s and 60 °C for 1 min. The results were analyzed on the ABI 7900 Real-Time PCR System (Applied Biosystems). All reactions, including non-template controls, were performed in triplicate. The calibration curves for concentrations of the synthetic mature miR-192 and miR-205 calibrators were linear from 102 to 105 fmol/l.
Assessment of interstitial fibrosis
Assessment of mesangial proliferation and interstitial fibrosis was determined on 4-μm paraffin-embedded sections stained by periodic acid-Schiff stain (PAS). Ten microscopic fields were viewed at magnification of 200× and scored from 0 to 100 % for each patient. An experienced pathologist blinded to the laboratory results evaluated the degree of mesangial proliferation and interstitial fibrosis.
Statistics
Statistical analysis was performed with the software SPSS 17.0. The results were presented in mean ± SD. Comparisons were made using the ANOVA analysis followed by SNK test for three parametric groups. Student’s t test was used for two parametric groups. Chi-square test was used to examine the differences in proportions between groups. Pearson correlation coefficient (r) was used to test the relationship between miRNAs and clinical parameters. All probabilities were two-tailed. Statistical significance was determined by p < 0.05.
Results
The demographic and baseline data of participants are summarized in Table 1. There was no significant difference in age, sex, serum creatinine, urinary protein/creatinine ratio, hepatic function, glomerular filtration rate (GFR), or duration of disease. The proportion of patients receiving distinct therapies were similar (p = 0.53), as well as that of the patients’ response to the steroid at the time of enrollment (p = 0.83). The glomerular filtration rate was estimated using the Schwartz formula [13].
Table 1.
The demographic and baseline data of participants
| Group | FSGS | MCD | HC |
|---|---|---|---|
| No. | 30 | 26 | 20 |
| Gender (male/female) | 16/14 | 15/11 | 12/8 |
| Age | 6.67 ± 2.90 | 6.84± 3.56 | 7.50 ± 3.12 |
| uPCR (mg/mg) | 5.02 ± 1.18 | 4.48 ± 1.04 | |
| Serum creatinine(μmol/l) | 41.13 ± 15.61 | 38.90 ± 24.63 | |
| ALT | 22.10 ± 4.66 | 24.00 ± 5.12 | |
| AST | 27.10 ± 5.47 | 28.54 ± 5.79 | |
| GFR (ml/min/1.73 m2) | 139.51± 49.11 | 152.71 ± 54.26 | |
| Duration of disease (months) | 16.34 ± 6.87 | 16.02 ± 5.32 | |
| Treatment (steroid + non-CNI/steroid + CNI) | 7/23 | 8/18 | |
| Response to corticosteroid (SRNS/SDNS) | 17/13 | 14/12 |
Student’s t test was used for two parametric groups. The Chi-square test was used to examine the differences in proportions between groups
FSGS focal segmental glomerulosclerosis, MCD minimal change disease, HC healthy controls, GFR glomerular filtration rate, uPCR urinary protein/creatinine ratio, CNI calcineurin inhibitors, ALT alanine aminotransferase, AST aspartate aminotransferase, SRNS steroid-resistant nephrotic syndrome, SDNS steroid-dependent nephrotic syndrome
Serum levels of miR-192 and miR-205
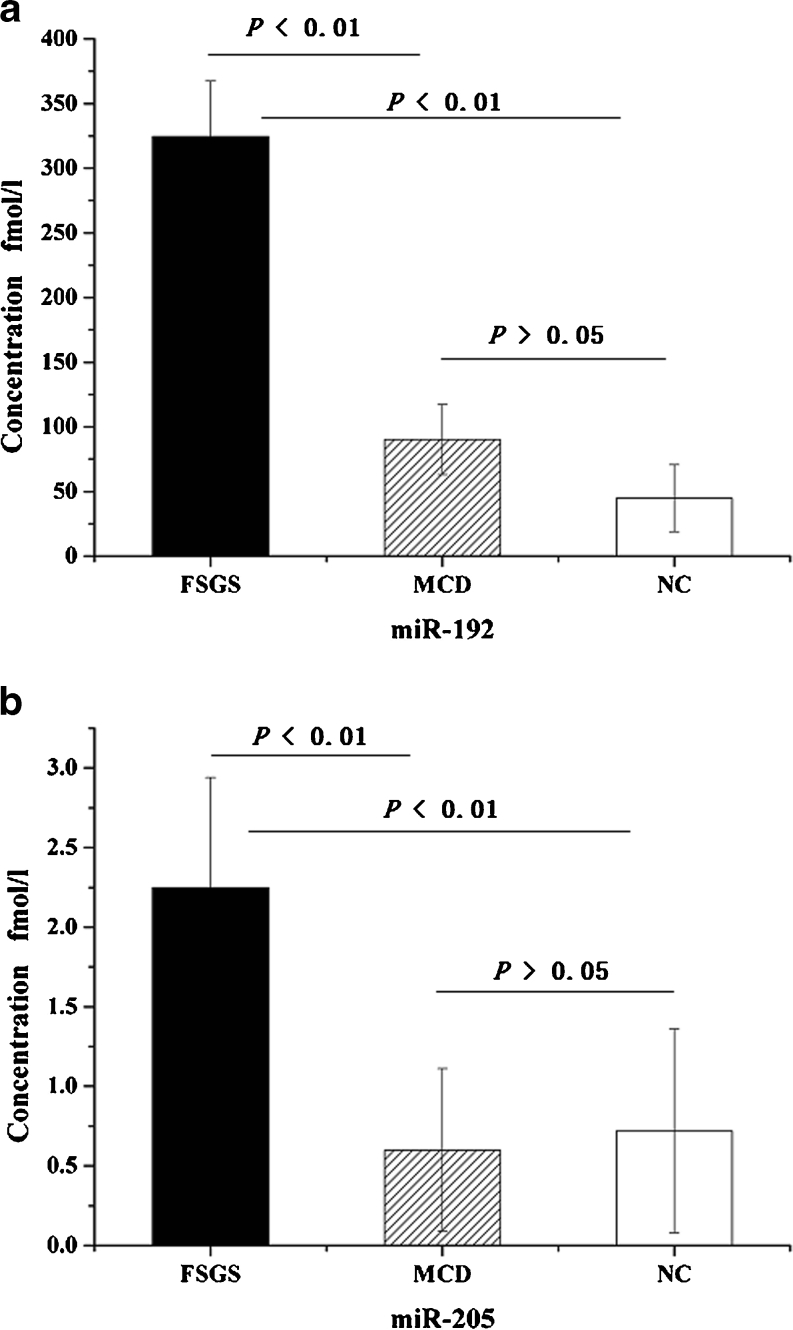
As compared to patients with MCD and healthy controls, patients with FSGS had higher serum concentration of miR-192 (324.49 ± 42.74 fmol/l versus 90.19 ± 27.14 fmol/l, p < 0.01, 324.49 ± 42.74 fmol/l versus 44.81 ± 26.03 fmol/l, p < 0.01, respectively) (Fig. 1a). Similarly, patients with FSGS had higher serum concentration of miR-205 than those with MCD and healthy controls (2.25 ± 0.69 fmol/l versus 0.60 ± 0.51 fmol/l, p < 0.01, 2.25 ± 0.69 fmol/l versus 0.72 ± 0.64 fmol/l, p < 0.01, respectively), but there was no difference between MCD and controls (0.60 ± 0.51 fmol/l versus 0.72 ± 0.64 fmol/l, p > 0.05) (Fig. 1b).
Fig. 1.
Comparison of serum concentrations of (a) miR-192 and (b) miR-205 among patients with FSGS, MCD, and HC. Data were compared by ANOVA analysis followed by SNK test. FSGS focal segmental glomerulosclerosis, MCD minimal change disease, HC healthy controls
In the FSGS group, patients received calcineurin inhibitors or not had similar serum miRNAs levels (miR-192: 323.54 ± 39.75 versus 327.60 ± 54.94, p = 0.83; 2.21 ± 0.65 versus 2.39 ± 0.85, p = 0.56, respectively). The serum levels of miRNAs did not show any significant difference between patients with distinct therapies within MCD group (miR-192: 95.03 ± 26.71 versus 79.29 ± 26.49, p = 0.18; 0.53 ± 0.43 versus 0.77 ± 0.64, p = 0.35; respectively).
Relation with clinical parameters
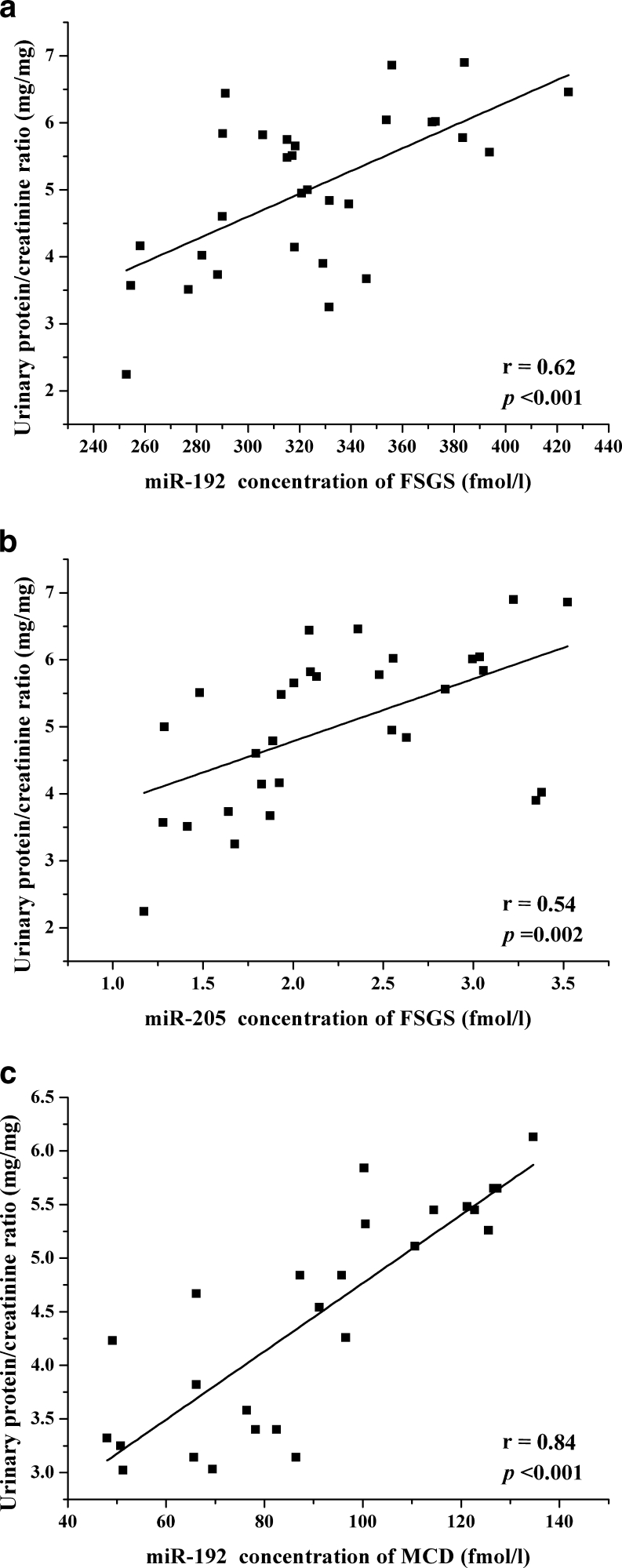
Serum concentration levels of miR-192 and miR-205 had a positive correlation with proteinuria in patients with FSGS (miR-192: r = 0.62, p < 0.001; miR-205: r = 0.54, p = 0.002) (Fig. 2a, b). The serum level of miR-192 was also correlated with proteinuria in MCD (r = 0.84, p < 0.001) (Fig. 2c). There was no significant relation between particular miRNA level and other clinical parameters.
Fig. 2.
Correlation between urinary protein and serum concentrations of miR-192 (a), miR-205 in patients with FSGS (b), and miR-192 in patients with MCD (c). Data were compared by Pearson correlation coefficient (r). FSGS focal segmental glomerulosclerosis, MCD minimal change disease
Relation with interstitial fibrosis
The degree of mesangial proliferation of FSGS was higher that of MCD (35.2 ± 12.3 % versus 16.2 ± 5.6 %). There was no correlation was found between the serum levels of the miRNAs and the degree of mesangial proliferation in FSGS and MCD. The percentage of interstitial fibrosis was 19.6 ± 13.7 %. The degree of interstitial fibrosis was correlated with serum level of miR-192 (r = 0.342, p < 0.05), but no significant correlation with that of miR-205.
Discussion
FSGS and MCD have similar presentations but absolutely different outcomes. We use the responsiveness to steroids to extrapolate the severity of renal histology, however the response was nothing but a time-dependent and elementary way to eschew the confirmation of the exact pathology [14]. In addition, the functional mode of steroids eliminating the proteinuria has not been understood. In the present work, we analyzed the serum miRNAs according to the definite histology including MCD and FSGS rather than responsiveness to steroids.
Historically early FSGS often cannot be distinguished from MCD, when it does not present with glomerulosclerosis or interstitial fibrosis [15]. FSGS and MCD often have heavy proteinuria, while renal fibrosis occurs in FSGS predominantly, which indicates that other mechanisms accelerating the process of fibrosis apart from proteinuria [16]. Many miRNAs have been found to play a pivotal role in the physiology and pathogenesis of kidney disease [17, 18]. Tissue specificity and relative stability of miRNAs make them promising candidates as biomarkers to reflect various disease states. miR-192 and miR-205 have been reported in multiple kidney diseases. In the current work, we found that higher levels of serum miR-192 and miR-205 in FSGS compared to that in MCD and healthy controls. miR-192 and miR-205 were positively correlated with proteinuria in patients with FSGS. Results indicated that miR-192 level positively correlated with the degree of renal fibrosis. Our study was consistent with previous studies. Kato et al. [19] showed that elevated miR-192 level in mouse mesangial cell increased the expression of collagen contributing to matrix accumulation and glomerulosclerosis. Kato et al. [20, 21] also identified that increased miR-192 level could lead to glomerulosclerosis in mesangial cell through TGF-β1 auto-introduction and Akt activation. Additionally, Putta et al. [22] found that suppression of miR-192 in diabetic mice attenuated proteinuria and ameliorated renal fibrosis. Multiple evidence has emerged that miR-192 and miR-205 participated in the process of epithelial mesenchymal transition (EMT) [23–26], which was proved to contribute to the renal fibrosis [27–30].
This is an innovative study on analyzing the serum levels of miR-192 and miR-205 in FSGS and MCD. There are some inadequacies. First, the single-centre study limited the sample size. Therefore we do not have enough statistical evidence for a powerful conclusion. Second, the treatment of each patient could be heterogenous, and the drugs they were receiving might affect the results, especially the calcineurin inhibitors. In order to eliminate bias, we compared the serum miRNAs levels between patients with or without calcineurin inhibitors in each group. In each group, the serum levels of miR-192 were similar between patients with distinct treatment, as well as that of miR-205. Additionally, we tested the miRNAs in serum without exploring the cellular sources of them. Kato et al. showed that miR-192 were largely expressed in mesangial cells [20]. Further studies are warranted to explore the underlying mechanism to validate the hypothesis that circulating miRNAs play a part in cell-to-cell communication [6].
In summary, our study, for the first time, showed that serum miR-192 and miR-205 have higher expression levels, which were correlated with the urine protein and the degree of tubulointerstitial fibrosis in patients with FSGS. The results suggest that miR-192 and miR-205 have the potential as markers to differentiate FSGS from MCD. The in-depth mechanism by which the miR-192 and miR-205 accelerate the progressive process of FSGS needs to be explored.
Acknowledgments
This work was supported by a grant from the National Natural Science Foundation of China (nos. 81270800). We thank Gao Chunlin, Shi Zhuo, Sun Tao, and Peng Hongjun, pediatricians, for the excellent contribution and the patients for their generous involvement in this study. We appreciate Wang Xiaomeng for her excellent suggestions for our manuscript.
References
- 1.Nezhad ST, Momeni B, Basiratnia M. Glomerular malondialdehyde levels in patients with focal and segmental glomerulosclerosis and minimal change disease. Saudi J Kidney Dis Transpl. 2010;21:886–891. [PubMed] [Google Scholar]
- 2.Gulati S, Sengupta D, Sharma RK, Sharma A, Gupta RK, Singh U, Gupta A. Steroid resistant nephrotic syndrome: role of histopathology. Indian Pediatr. 2006;43:55–60. [PubMed] [Google Scholar]
- 3.Savin VJ, Sharma R, Sharma M, McCarthy ET, Swan SK, Ellis E, Lovell H, Warady B, Gunwar S, Chonko AM, Artero M, Vincenti F. Circulating factor associated with increased glomerular permeability to albumin in recurrent focal segmental glomerulosclerosis. N Engl J Med. 1996;334:878–883. doi: 10.1056/NEJM199604043341402. [DOI] [PubMed] [Google Scholar]
- 4.Czech KA, Bennett M, Devarajan P. Distinct metalloproteinase excretion patterns in focal segmental glomerulosclerosis. Pediatr Nephrol. 2011;26:2179–2184. doi: 10.1007/s00467-011-1897-7. [DOI] [PubMed] [Google Scholar]
- 5.Cortez MA, Calin GA. MicroRNA identification in plasma and serum: a new tool to diagnose and monitor diseases. Expert Opin Biol Ther. 2009;9:703–711. doi: 10.1517/14712590902932889. [DOI] [PubMed] [Google Scholar]
- 6.Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, Peterson A, Noteboom J, O’Briant KC, Allen A, Lin DW, Urban N, Drescher CW, Knudsen BS, Stirewalt DL, Gentleman R, Vessella RL, Nelson PS, Martin DB, Tewari M. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA. 2008;105:10513–10518. doi: 10.1073/pnas.0804549105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hanke M, Hoefig K, Merz H, Feller AC, Kausch I, Jocham D, Warnecke JM, Sczakiel G. A robust methodology to study urine microRNA as tumor marker: microRNA-126 and microRNA-182 are related to urinary bladder cancer. Urol Oncol. 2010;28:655–661. doi: 10.1016/j.urolonc.2009.01.027. [DOI] [PubMed] [Google Scholar]
- 8.Liang Y, Ridzon D, Wong L, Chen C. Characterization of microRNA expression profiles in normal human tissues. BMC Genomics. 2007;8:166. doi: 10.1186/1471-2164-8-166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sun Y, Koo S, White N, Peralta E, Esau C, Dean NM, Perera RJ. Development of a micro-array to detect human and mouse microRNAs and characterization of expression in human organs. Nucleic Acids Res. 2004;32:e188. doi: 10.1093/nar/gnh186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Tian Z, Greene AS, Pietrusz JL, Matus IR, Liang M. MicroRNA-target pairs in the rat kidney identified by microRNA microarray, proteomic, and bioinformatic analysis. Genome Res. 2008;18:404–411. doi: 10.1101/gr.6587008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang G, Kwan BC, Lai FM, Choi PC, Chow KM, Li PK, Szeto CC. Intrarenal expression of miRNAs in patients with hypertensive nephrosclerosis. Am J Hypertens. 2010;23:78–84. doi: 10.1038/ajh.2009.208. [DOI] [PubMed] [Google Scholar]
- 12.Eddy AA, Symons JM. Nephrotic syndrome in childhood. Lancet. 2003;362:629–639. doi: 10.1016/S0140-6736(03)14184-0. [DOI] [PubMed] [Google Scholar]
- 13.Schwartz GJ, Haycock GB, Edelmann CM, Jr, Spitzer A. A simple estimate of glomerular filtration rate in children derived from body length and plasma creatinine. Pediatrics. 1976;58:259–263. [PubMed] [Google Scholar]
- 14.Meyrier AY. Treatment of focal segmental glomerulosclerosis with immunophilin modulation: when did we stop thinking about pathogenesis? Kidney Int. 2009;76:487–491. doi: 10.1038/ki.2009.204. [DOI] [PubMed] [Google Scholar]
- 15.Cameron JS, Davlson AM, GrUnfcld JP (1992) Oxford textbook of clinical nephrology. Abstracts of the Renal Association Sheffield UK:1142
- 16.Wen Q, Huang Z, Zhou SF, Li XY, Luo N, Yu XQ. Urinary proteins from patients with nephrotic syndrome alters the signalling proteins regulating epithelial-mesenchymal transition. Nephrology (Carlton) 2010;15:63–74. doi: 10.1111/j.1440-1797.2009.01152.x. [DOI] [PubMed] [Google Scholar]
- 17.Kato M, Arce L, Natarajan R. MicroRNAs and their role in progressive kidney diseases. Clin J Am Soc Nephrol. 2009;4:1255–1266. doi: 10.2215/CJN.00520109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Saal S, Harvey SJ. MicroRNAs and the kidney: coming of age. Curr Opin Nephrol Hypertens. 2009;18:317–323. doi: 10.1097/MNH.0b013e32832c9da2. [DOI] [PubMed] [Google Scholar]
- 19.Kato M, Zhang J, Wang M, Lanting L, Yuan H, Rossi JJ, Natarajan R. MicroRNA-192 in diabetic kidney glomeruli and its function in TGF-beta-induced collagen expression via inhibition of E-box repressors. Proc Natl Acad Sci USA. 2007;104:3432–3437. doi: 10.1073/pnas.0611192104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kato M, Arce L, Wang M, Putta S, Lanting L, Natarajan R. A microRNA circuit mediates transforming growth factor-beta1 autoregulation in renal glomerular mesangial cells. Kidney Int. 2011;80:358–368. doi: 10.1038/ki.2011.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kato M, Putta S, Wang M, Yuan H, Lanting L, Nair I, Gunn A, Nakagawa Y, Shimano H, Todorov I, Rossi JJ, Natarajan R. TGF-beta activates Akt kinase through a microRNA-dependent amplifying circuit targeting PTEN. Nat Cell Biol. 2009;11:881–889. doi: 10.1038/ncb1897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Putta S, Lanting L, Sun G, Lawson G, Kato M, Natarajan R. Inhibiting microRNA-192 ameliorates renal fibrosis in diabetic nephropathy. J Am Soc Nephrol. 2012;23:458–469. doi: 10.1681/ASN.2011050485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gregory PA, Bert AG, Paterson EL, Barry SC, Tsykin A, Farshid G, Vadas MA, Khew-Goodall Y, Goodall GJ. The miR-200 family and miR-205 regulate epithelial to mesenchymal transition by targeting ZEB1 and SIP1. Nat Cell Biol. 2008;10:593–601. doi: 10.1038/ncb1722. [DOI] [PubMed] [Google Scholar]
- 24.Park SM, Gaur AB, Lengyel E, Peter ME. The miR-200 family determines the epithelial phenotype of cancer cells by targeting the E-cadherin repressors ZEB1 and ZEB2. Genes Dev. 2008;22:894–907. doi: 10.1101/gad.1640608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Korpal M, Lee ES, Hu G, Kang Y. The miR-200 family inhibits epithelial-mesenchymal transition and cancer cell migration by direct targeting of E-cadherin transcriptional repressors ZEB1 and ZEB2. J Biol Chem. 2008;283:14910–14914. doi: 10.1074/jbc.C800074200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Burk U, Schubert J, Wellner U, Schmalhofer O, Vincan E, Spaderna S, Brabletz T. A reciprocal repression between ZEB1 and members of the miR-200 family promotes EMT and invasion in cancer cells. EMBO Rep. 2008;9:582–589. doi: 10.1038/embor.2008.74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rastaldi MP, Ferrario F, Giardino L, Dell’Antonio G, Grillo C, Grillo P, Strutz F, Muller GA, Colasanti G, D’Amico G. Epithelial-mesenchymal transition of tubular epithelial cells in human renal biopsies. Kidney Int. 2002;62:137–146. doi: 10.1046/j.1523-1755.2002.00430.x. [DOI] [PubMed] [Google Scholar]
- 28.Frazier KS, Paredes A, Dube P, Styer E. Connective tissue growth factor expression in the rat remnant kidney model and association with tubular epithelial cells undergoing transdifferentiation. Vet Pathol. 2000;37:328–335. doi: 10.1354/vp.37-4-328. [DOI] [PubMed] [Google Scholar]
- 29.Fan JM, Ng YY, Hill PA, Nikolic-Paterson DJ, Mu W, Atkins RC, Lan HY. Transforming growth factor-beta regulates tubular epithelial-myofibroblast transdifferentiation in vitro. Kidney Int. 1999;56:1455–1467. doi: 10.1046/j.1523-1755.1999.00656.x. [DOI] [PubMed] [Google Scholar]
- 30.Nangaku M. Mechanisms of tubulointerstitial injury in the kidney: final common pathways to end-stage renal failure. Intern Med. 2004;43:9–17. doi: 10.2169/internalmedicine.43.9. [DOI] [PubMed] [Google Scholar]