Abstract
This study evaluated the effects of NaCl on heat resistance, antibiotic susceptibility, and Caco-2 cell invasion of Salmonella. Salmonella typhimurium NCCP10812 and Salmonella enteritidis NCCP12243 were exposed to 0, 2, and 4% NaCl and to sequential increase of NaCl concentrations from 0 to 4% NaCl for 24 h at 35°C. The strains were then investigated for heat resistance (60°C), antibiotic susceptibility to eight antibiotics, and Caco-2 cell invasion efficiency. S. typhimurium NCCP10812 showed increased thermal resistance (P < 0.05) after exposure to single NaCl concentrations. A sequential increase of NaCl concentration decreased (P < 0.05) the antibiotic sensitivities of S. typhimurium NCCP10812 to chloramphenicol, gentamicin, and oxytetracycline. NaCl exposure also increased (P < 0.05) Caco-2 cell invasion efficiency of S. enteritidis NCCP12243. These results indicate that NaCl in food may cause increased thermal resistance, cell invasion efficiency, and antibiotic resistance of Salmonella.
1. Introduction
Salmonella spp. are pathogenic bacteria that are responsible for foodborne diseases, with enteritidis, typhimurium, and Heidelbergare being the serotypes most frequently involved in Salmonella foodborne outbreaks [1]. The pathogen possesses the ability to enter and penetrate the human epithelial cell, causing fever, gastroenteritis, and bacteremia [2, 3]. Since antibiotic resistant Salmonella has been isolated from various foods, the bacteria have become a worldwide food safety issue [4, 5].
Salmonella spp. require high availability of water (a w) ranging between 0.94 and 0.99; however, since the bacteria can still survive below that a w range, it is a widespread pathogen that has been also isolated from high-salted fish and meat products [3, 6]. A study by Oscar [7] showed that previous exposure of Salmonella to NaCl affected the kinetic parameters of the bacteria, such as lag phase duration and maximum specific growth rate. For Staphylococcus aureus, exposure to NaCl increased its thermal resistance in ready-to-heat sauces [8]. Other studies also showed that the exposure of bacteria to sublethal stresses such as acid, heat, and NaCl increased their cross-protection responses, especially following continuous exposure to the stresses [9, 10]. A study by Kieboom et al. [11] revealed that S. enteritidis cells exposed to a w = 0.94 had higher resistance to sodium hypochlorite than control cells.
When Listeria monocytogenes experienced food-related stress conditions such as NaCl, sodium diacetate, and sodium lactate, its virulence characteristics including invasiveness were increased [12]. Prior exposure of L. monocytogenes to mild heat stress and tryptone also augmented the bacterial invasion into human epithelial cells [13, 14].
Therefore, the objective of this study was to elucidate the effects of NaCl on heat resistance, antibiotic susceptibility, and Caco-2 cell invasion efficiency of Salmonella.
2. Materials and Methods
2.1. Preparation of Salmonella Inoculum
The isolated colonies of S. typhimurium NCCP10812 and S. enteritidis NCCP12243 on XLD agar (Difco, Becton Dickinson and Company, Sparks, MD, USA) were cultured in 10 mL tryptic soy broth (TSB; Difco ) at 35°C for 24 h. A fraction (0.1 mL) of the initial culture was then transferred into 10 mL fresh TSB for subculture at 35°C for 24 h, following centrifugation at 1,912 ×g, and 4°C for 15 min. The resulting pellets were washed with phosphate buffered saline (PBS, pH 7.4; 0.2 g of KH2PO4, 1.5 g of Na2HPO4, 8.0 g of NaCl, and 0.2 g of KCl in 1 L of distilled water) twice and diluted in PBS to 4–6 Log CFU/mL of inoculum.
2.2. Exposure of Salmonella Cells to NaCl
To expose S. typhimurium NCCP10812 and S. enteritidis NCCP12243 to NaCl, 0.1 mL portions of the inocula were inoculated into 0, 2 and 4% NaCl (w/v) supplemented with 10 mL TSB and incubated at 35°C for 24 h to obtain stationary phase cells. After exposure to NaCl, 0.1 mL of the cultures were plated on tryptic soy agar (TSA; Difco) containing 0, 2, and 4% NaCl and incubated at 35°C for 24 h to obtain only NaCl-habituated Salmonella cells.
Salmonella cells of inocula were also exposed to a sequential increase of NaCl concentration up to 4% according to the procedure shown in Figure 1. After the incubation of the plates at 35°C for 24 h, 4 mL PBS was added directly on the plates, and the colonies were collected by scraping with a glass rod. The collected bacterial cells were centrifuged (1,912 ×g, and 4°C, for 15 min), and the pellets were washed with PBS twice. These bacterial cell suspensions were then adjusted to OD600 = 0.1 with PBS for the heat resistance and antibiotic susceptibility assays, or to OD600 = 0.03–0.04 for the Caco-2 cell invasion assay.
Figure 1.
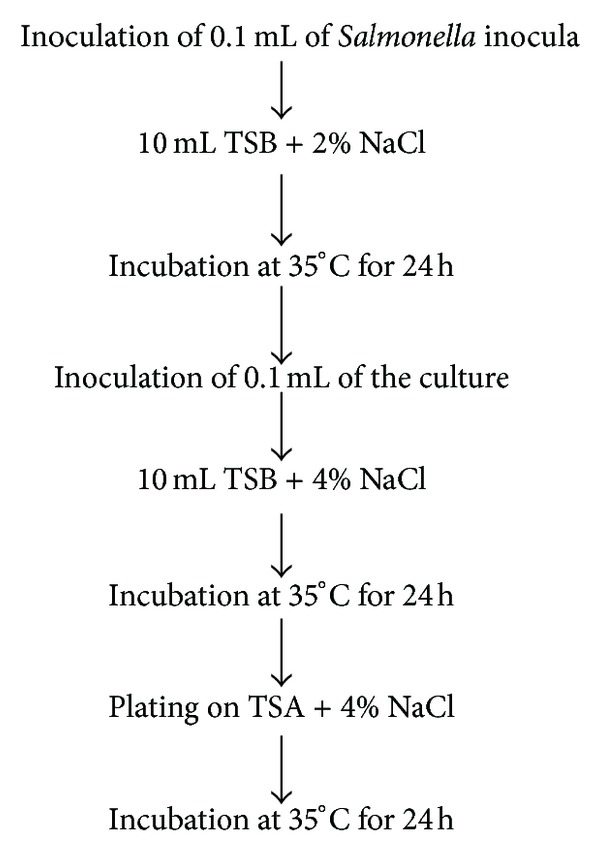
Graphical representation of the preparation of Salmonella cells exposed to sequential increase of NaCl concentration.
2.3. Heat Resistance
One milliliter of each bacterial cell suspension was inoculated into 9 mL TSB at 60°C in a water bath. Samples (1 mL) were withdrawn every 20 minutes for 1 h and diluted with 0.1% buffered peptone water (BPW, Difco). The diluents (0.1 mL) were spread-plated on TSA, and the plates were incubated at 35°C for 24 h.
2.4. Antibiotic Susceptibility
Antibiotic susceptibility was examined by a disc diffusion assay on Mueller-Hinton agar (MHA, Difco) according to the standard procedure outlined in the National Committee for Clinical Laboratory Standards guidelines [15]. A sterile swab was dampened with the bacterial cell suspensions and then spread on the surface of MHA. The MHA plates were then left at room temperature for 10–15 min, and antibiotic discs (Oxoid, Thermo Fisher Scientific, Basingstoke, Hampshire, UK) were placed on their surface using a multidisc dispenser (Oxoid). The tested antibiotics were amoxicillin (10 μg), chloramphenicol (30 μg), ciprofloxacin (5 μg), gentamicin (10 μg), neomycin (30 μg), oxytetracycline (30 μg), streptomycin (10 μg), and tigecycline (15 μg). After the incubation of the plates at 35°C for 24 h, a portion of clear zone in the order of millimeters around each disc was measured.
2.5. Caco-2 Cell Invasion Assay
The bacterial cell suspensions of S. typhimurium NCCP10812 and S. enteritidis NCCP12243 were then diluted to 5 × 105–5 × 106 CFU/mL with PBS, and 0.5 mL of the diluents was inoculated into 4.5 mL Eagle's minimum essential medium (MEM medium, Gibco, Penrose, Auckland, New Zealand) supplemented with 20% fetal bovine serum (FBS, Gibco) to be used as inoculum. The inoculum (1 mL) was then inoculated in a monolayer cell of Caco-2 cells (5 × 104 cells/mL) and incubated in 5% CO2 at 37°C for 4 h. The upper layer of MEM medium was discarded, and the cells were further incubated in fresh MEM medium supplemented with 20% FBS and 50 μg/mL gentamicin at 37°C for 2 h. After the incubation, the upper layer of the media was discarded, and the Caco-2 cells were washed with PBS twice. A solution (1 mL) of 0.5% Triton X-100 was then added into each well of the plate, and the plate was left on ice for 20 min. The resulting suspension was plated on TSA to enumerate invaded Salmonella populations. Invasion efficiency of Salmonella to Caco-2 cells was calculated by the equation suggested by Garner et al. [12] as follows:
| (1) |
2.6. Statistical Analysis
The experiment was repeated twice with three samples per repeat (n = 6). Bacterial populations were converted to Log CFU/mL before statistical analysis. The data values were analyzed with the general linear model procedures of SAS version 9.2 (SAS Institute Inc., USA). The least squares (LS) means among fixed effects were compared using the “pdiff” option to analyze all the pairwise comparisons at alpha = 0.05 [16].
3. Results and Discussion
3.1. Heat Resistance
S. typhimurium NCCP10812 exposed to single NaCl concentrations (0, 2, and 4%) in TSB exhibited increased heat resistance (P < 0.05) as the NaCl concentration increased after 60 min of heat challenge. Similarly, Yuan et al. [17] also showed that the adaptation of S. typhimurium to 5% sodium lactate and 3% sodium acetate enhanced its ability to survive under heat stress. This protective effect of NaCl could be due to a decrease in water activity following NaCl addition. In fact, NaCl causes a poor penetration of heat through the heating medium [18]. However, the heat resistance of S. enteritidis NCCP12243 was not increased upon increasing concentrations of NaCl (Table 1). Moreover, when the Salmonella strains experienced a sequential increase of NaCl concentration, their heat resistances were generally not different from the control (0% NaCl) (Table 1). This result indicates that the effect of NaCl on increased heat resistance of Salmonella depends on the specific strain.
Table 1.
Reductions (mean ± SD; log(N 0/N t)) of S. typhimurium NCCP10812 and S. enteritidis NCCP12243, which were exposed to single concentrations (0, 2, and 4%) of NaCl and sequentially increased NaCl concentrations up to 4%, during heat challenge at 60°C for 60 min.
| Salmonella strains | Heating time (minutes) | Single concentration of NaCl (%) | Sequential increase of NaCl (%) | |||
|---|---|---|---|---|---|---|
| 0 | 2 | 4 | 0→0 | 0→4 | ||
| S. typhimurium NCCP10812 | 0 | 0.0 ± 0.0Aa | 0.0 ± 0.0Aa | 0.0 ± 0.0Aa | 0.0 ± 0.0Zz | 0.0 ± 0.0Zz |
| 20 | −3.7 ± 0.3Ba | −3.8 ± 0.2Ba | −3.9 ± 0.3Ba | −3.5 ± 0.2Yz | −4.4 ± 0.1XWy | |
| 40 | −4.6 ± 0.2Ca | −4.4 ± 0.1Ca | −4.4 ± 0.4Ca | −4.3 ± 0.2Xz | −4.4 ± 0.7XWz | |
| 60 | −5.3 ± 0.4Db | −4.3 ± 0.1Ca | −4.4 ± 0.5Ca | −4.8 ± 0.2Wz | −4.5 ± 0.6XWz | |
|
| ||||||
| S. enteritidis NCCP12243 | 0 | 0.0 ± 0.0Aa | 0.0 ± 0.0Aa | 0.0 ± 0.0Aa | 0.0 ± 0.0Zz | 0.0 ± 0.0Zz |
| 20 | −3.6 ± 0.1Ba | −3.7 ± 0.4Ba | −4.0 ± 0.0Ba | −3.5 ± 0.0Yz | −3.9 ± 0.4Yz | |
| 40 | −4.5 ± 0.3CDb | −4.6 ± 0.3Da | −4.7 ± 0.1Ca | −4.0 ± 0.4Xz | −4.3 ± 0.2Xz | |
| 60 | −5.0 ± 0.6Ca | −4.9 ± 0.5CDa | −4.6 ± 0.2Ca | −5.1 ± 0.2Wz | −4.8 ± 0.3Wz | |
A–EMeans within the same column with different superscript letters are different (P < 0.05). a-bMeans within the same row with different superscript letters are different (P < 0.05). W–ZMeans within the same column with different superscript letters are different (P < 0.05). y–zMeans within the same row with different superscript letters are different (P < 0.05).
3.2. Antibiotic Susceptibility
To evaluate the effect of NaCl on antibiotic susceptibility, S. typhimurium NCCP10812 and S. enteritidis NCCP12243 were exposed to single concentrations of NaCl or sequentially increased NaCl concentration up to 4%. Exposure of the Salmonella strains to single concentrations of NaCl did not change their antibiotic susceptibility (Table 2). However, the antibiotic susceptibility of S. typhimurium NCCP10812 significantly increased (P < 0.05) against chloramphenicol, gentamicin, and oxytetracycline after exposure to sequentially increased NaCl concentration (Table 2). Similarly, S. enteritidis NCCP12243 also displayed (P < 0.05) increased resistance to amoxycillin and ciprofloxacin, while its antibiotic susceptibility against chloramphenicol, gentamicin, oxytetracycline, and streptomycin decreased (P < 0.05) after sequential exposure to 4% of NaCl (Table 2). This finding suggests that consistent exposure of Salmonella strains to NaCl may alter their antibiotic susceptibility, but the influence of NaCl on the susceptibility of Salmonella against antibiotics is dependent on both the strains and antibiotics. Hengge-Aronis [19] found that RpoS, a general stress response regulator, was upregulated when bacteria were exposed to osmotic stress. A study by Huang et al. [20] showed that RpoS has an effect on antibiotic resistances of Escherichia coli against ampicillin, chloramphenicol, and rifampicin. Therefore, it can be hypothesized that NaCl may regulate RpoS expression, thereby influencing antibiotic senstivity of Salmonella NCCP10812 and NCCP12243.
Table 2.
Diameters (mean ± SD; mm) of clear zones formed by S. typhimurium NCCP10812 and S. enteritidis NCCP12243 to single concentrations (0, 2, and 4%) of NaCl and sequentially increased NaCl concentrations up to 4%.
| Salmonella strains | Antibiotic | Single concentration of NaCl (%) | Sequential increase of NaCl (%) | |||
|---|---|---|---|---|---|---|
| 0 | 2 | 4 | 0→0 | 0→4 | ||
| S. typhimurium NCCP10812 | Amoxycillin | 28.0 ± 0.8 | 27.5 ± 0.6 | 27.3 ± 0.5 | 29.0 ± 0.0A | 29.5 ± 0.6A |
| Chloramphenicol | 31.8 ± 1.0 | 33.3 ± 1.5 | 34.0 ± 1.2 | 33.3 ± 1.0A | 12.0 ± 2.4B | |
| Ciprofloxacin | 47.0 ± 1.2 | 49.5 ± 3.4 | 48.0 ± 2.3 | 48.0 ± 1.6A | 46.5 ± 1.9A | |
| Gentamicin | 26.3 ± 1.0 | 26.8 ± 1.0 | 26.8 ± 1.0 | 26.5 ± 0.6A | 14.3 ± 2.2B | |
| Neomycin | 22.0 ± 0.8 | 22.0 ± 0.8 | 22.3 ± 0.5 | 24.0 ± 2.2A | 22.3 ± 2.1A | |
| Oxytetracycline | 26.8 ± 4.9 | 27.0 ± 4.7 | 27.0 ± 3.6 | 30.5 ± 1.7A | 9.5 ± 1.9B | |
| Streptomycin | 19.0 ± 0.0 | 19.3 ± 0.5 | 19.3 ± 1.0 | 20.3 ± 1.5A | 19.5 ± 1.3A | |
| Tigecycline | 25.5 ± 1.0 | 25.8 ± 1.0 | 26.3 ± 0.5 | 25.8 ± 0.5A | 26.8 ± 1.5A | |
|
| ||||||
| S. enteritidis NCCP12243 | Amoxycillin | 27.8 ± 0.5 | 28.0 ± 0.0 | 26.8 ± 2.2 | 28.8 ± 1.3A | 21.3 ± 8.4B |
| Chloramphenicol | 10.8 ± 2.2 | 9.8 ± 0.5 | 11.0 ± 1.6 | 10.3 ± 2.1B | 30.8 ± 2.8A | |
| Ciprofloxacin | 44.5 ± 6.4 | 44.0 ± 4.6 | 39.3 ± 2.2 | 43.5 ± 4.1A | 38.0 ± 6.9B | |
| Gentamicin | 13.3 ± 0.5 | 13.5 ± 1.3 | 12.5 ± 0.6 | 13.8 ± 1.7B | 23.8 ± 5.6A | |
| Neomycin | 20.5 ± 0.6 | 21.0 ± 0.0 | 21.0 ± 0.8 | 21.0 ± 0.8A | 24.3 ± 2.4A | |
| Oxytetracycline | 8.0 ± 0.0 | 8.0 ± 0.0 | 8.0 ± 0.0 | 8.0 ± 0.0B | 29.8 ± 1.3A | |
| Streptomycin | 16.8 ± 0.5 | 15.0 ± 2.0 | 15.0 ± 0.8 | 16.0 ± 0.8B | 25.5 ± 4.8A | |
| Tigecycline | 25.0 ± 1.2 | 25.3 ± 1.0 | 26.8 ± 1.0 | 26.3 ± 1.0A | 26.8 ± 1.5A | |
A-BMeans within the same row with different superscript letters are different (P < 0.05).
3.3. Caco-2 Cell Invasion Assay
The cell invasion efficiency of S. typhimurium NCCP10812 was not affected by NaCl, while S. enteritidis NCCP12243 showed increased (P < 0.05) invasion efficiency into Caco-2 cells from 21% to 39.4% (Figure 2(a)). When S. enteritidis NCCP12243 experienced a sequential increase of NaCl concentration up to 4%, the invasion efficiency dramatically increased (P < 0.05) from 29.5% to 83.3% (Figure 2(b)). This result indicates that the prior exposure to NaCl increases Caco-2 cell invasion efficiency of Salmonella, especially when the pathogen is previously exposed to a sequential increase of NaCl concentration. Similarly, Salmonella was also more invasive on Henle 407 cells when grown in high osmolarity (0.3 M NaCl) than in low osmolarity (0.06 M NaCl) [21]. Moreover, a study by Sirsat et al. [22] showed that stress such as heat increased Salmonella adhesion on Caco-2 cells from 6.3 to 11.7%.
Figure 2.
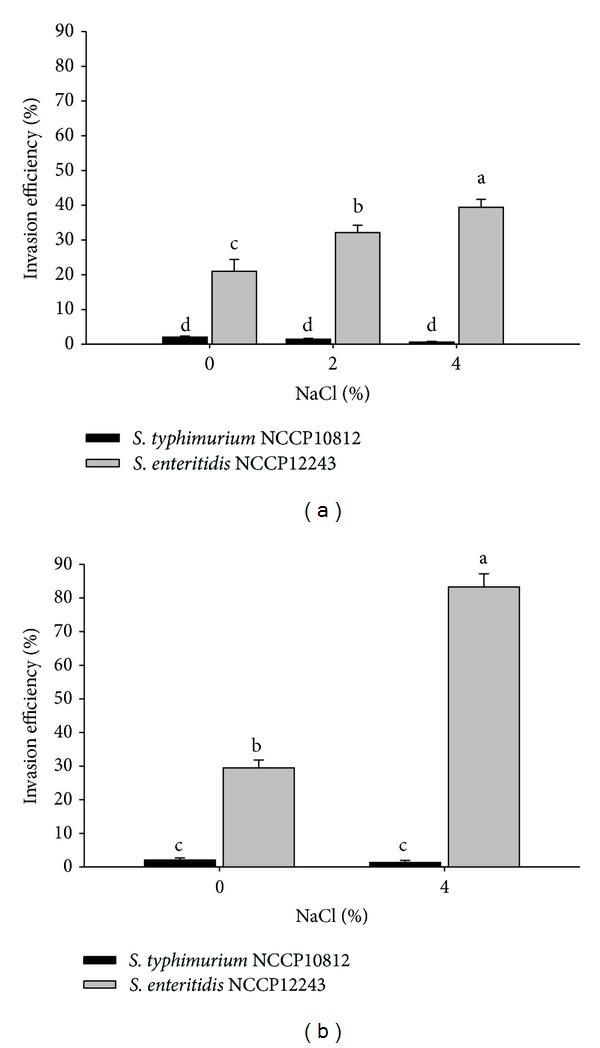
Invasion efficiency of S. typhimurium NCCP10812 and S. enteritidis NCCP12243 exposed to single concentrations (0, 2, and 4%) of NaCl (a) and sequentially increased NaCl concentrations up to 4% (b). a–dMeans with different superscript letters are different (P < 0.05).
In conclusion, the effect of NaCl on the heat resistance of Salmonella is strain dependent, and the sequential increase of NaCl concentration may influence the antibiotic susceptibility of the bacteria. Additionally, NaCl may increase the invasion efficiency of Salmonella strains used in this study into Caco-2 cells.
Acknowledgment
This work was carried out with the support of “Cooperative Research Program for Agriculture Science and Technology Development (Project no. PJ009237)” Rural Development Administration, Republic of Korea.
References
- 1.Boyen F, Haesebrouck F, Maes D, Van Immerseel F, Ducatelle R, Pasmans F. Non-typhoidal Salmonella infections in pigs: a closer look at epidemiology, pathogenesis and control. Veterinary Microbiology. 2008;130(1-2):1–19. doi: 10.1016/j.vetmic.2007.12.017. [DOI] [PubMed] [Google Scholar]
- 2.Shi J, Casanova JE. Invasion of host cells by Salmonella typhimurium requires focal adhesion kinase and p130Cas. Molecular Biology of the Cell. 2006;17(11):4698–4708. doi: 10.1091/mbc.E06-06-0492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pui CF, Wong WC, Chai LC, et al. Salmonella: a foodborne pathogen. International Food Research Journal. 2011;18(2) [Google Scholar]
- 4.Glynn MK, Bopp C, Dewitt W, Dabney P, Mokhtar M, Angulo FJ. Emergence of multidrug-resistant Salmonella enterica serotype typhimurium DT104 infections in the United States. New England Journal of Medicine. 1998;338(19):1333–1338. doi: 10.1056/NEJM199805073381901. [DOI] [PubMed] [Google Scholar]
- 5.Low JC, Angus M, Hopkins G, Munro D, Rankin SC. Antimicrobial resistance of Salmonella enterica typhimurium DT104 isolates and investigation of strains with transferable apramycin resistance. Epidemiology and Infection. 1997;118(2):97–103. doi: 10.1017/s0950268896007339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cabedo L, Picart I Barrot L, Teixidó I Canelles A. Prevalence of Listeria monocytogenes and Salmonella in ready-to-eat food in Catalonia, Spain. Journal of Food Protection. 2008;71(4):855–859. doi: 10.4315/0362-028x-71.4.855. [DOI] [PubMed] [Google Scholar]
- 7.Oscar TP. Response surface models for effects of temperature and previous growth sodium chloride on growth kinetics of Salmonella typhimurium on cooked chicken breast. Journal of Food Protection. 1999;62(12):1470–1474. doi: 10.4315/0362-028x-62.12.1470. [DOI] [PubMed] [Google Scholar]
- 8.Park A, Lee J, Jeong S-J, et al. Thermal inactivation of sodium-habituated Staphylococcus aureus in ready-to-heat sauces. Korean Journal For Food Science of Animal Resources. 2012;32(6):713–717. [Google Scholar]
- 9.Koutsoumanis KP, Kendall PA, Sofos JN. Effect of food processing-related stresses on acid tolerance of Listeria monocytogenes . Applied and Environmental Microbiology. 2003;69(12):7514–7516. doi: 10.1128/AEM.69.12.7514-7516.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lee J, Yoon H, Lee S, et al. Effect of NaCl on thermal resistance, antibiotic resistance, and human epithelial cell invasion of Listeria monocytogenes . Korean Journal For Food Science of Animal Resources. 2012;32(5):545–552. [Google Scholar]
- 11.Kieboom J, Kusumaningrum HD, Tempelaars MH, Hazeleger WC, Abee T, Beumer RR. Survival, elongation, and elevated tolerance of Salmonella enterica serovar enteritidis at reduced water activity. Journal of Food Protection. 2006;69(11):2681–2686. doi: 10.4315/0362-028x-69.11.2681. [DOI] [PubMed] [Google Scholar]
- 12.Garner MR, James KE, Callahan MC, Wiedmann M, Boor KJ. Exposure to salt and organic acids increases the ability of Listeria monocytogenes to invade Caco-2 cells but decreases its ability to survive gastric stress. Applied and Environmental Microbiology. 2006;72(8):5384–5395. doi: 10.1128/AEM.00764-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bradley D, McNeil B, Laffey JG, Rowan NJ. Studies on the pathogenesis and survival of different culture forms of Listeria monocytogenes to pulsed UV-light irradiation after exposure to mild-food processing stresses. Food Microbiology. 2012;30(2):330–339. doi: 10.1016/j.fm.2011.12.024. [DOI] [PubMed] [Google Scholar]
- 14.Kuda T, Nakamura S, An C, Takahashi H, Kimura B. Effect of soy and milk protein-related compounds on Listeria monocytogenes infection in human enterocyte Caco-2 cells and A/J mice. Food Chemistry. 2012;134(4):1719–1723. doi: 10.1016/j.foodchem.2012.03.031. [DOI] [PubMed] [Google Scholar]
- 15.NCCLS (National Committee for Clinical Laboratory Standards) Development of in vitro susceptibility testing criteria and quality control parameters for veterinary antimicrobial agents; Approved guideline. NCCLS document M37-A. NCCLS, Wayne, PA, USA, 1999.
- 16.SAS. SAS version 9.2 User’s guide. 2nd edition, 2012, http://support.sas.com/documentation/cdl/en/statug/63347/HTML/default/viewer.htm#/documentation/cdl/en/statug/63347/HTML/default/titlepage.htm.
- 17.Yuan W, Ágoston R, Lee D, Lee S-C, Yuk H-G. Influence of lactate and acetate salt adaptation on Salmonella typhimurium acid and heat resistance. Food Microbiology. 2012;30(2):448–452. doi: 10.1016/j.fm.2011.12.023. [DOI] [PubMed] [Google Scholar]
- 18.Juneja VK. Predictive model for the combined effect of temperature, sodium lactate, and sodium diacetate on the heat resistance of Listeria monocytogenes in beef. Journal of Food Protection. 2003;66(5):804–811. doi: 10.4315/0362-028x-66.5.804. [DOI] [PubMed] [Google Scholar]
- 19.Hengge-Aronis R. Signal transduction and regulatory mechanisms involved in control of the σs (RpoS) subunit of RNA polymerase. Microbiology and Molecular Biology Reviews. 2002;66(3):373–395. doi: 10.1128/MMBR.66.3.373-395.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Huang Y, Kan B, Lu Y, et al. The effect of osmotic shock on RpoS expression and antibiotic resistance in Escherichia coli . Journal of Experimental Microbiology and Immunology. 2009;13(1):13–17. [Google Scholar]
- 21.Tartera C, Metcalf ES. Osmolarity and growth phase overlap in regulation of Salmonella typhi adherence to and invasion of human intestinal cells. Infection and Immunity. 1993;61(7):3084–3089. doi: 10.1128/iai.61.7.3084-3089.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sirsat SA, Burkholder KM, Muthaiyan A, Dowd SE, Bhunia AK, Ricke SC. Effect of sublethal heat stress on Salmonella typhimurium virulence. Journal of Applied Microbiology. 2011;110(3):813–822. doi: 10.1111/j.1365-2672.2011.04941.x. [DOI] [PubMed] [Google Scholar]


