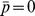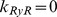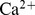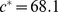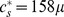Abstract
Intracellular 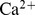 dynamics of airway smooth muscle cells (ASMC) mediate ASMC contraction and proliferation, and thus play a key role in airway hyper-responsiveness (AHR) and remodelling in asthma. We evaluate the importance of store-operated
dynamics of airway smooth muscle cells (ASMC) mediate ASMC contraction and proliferation, and thus play a key role in airway hyper-responsiveness (AHR) and remodelling in asthma. We evaluate the importance of store-operated 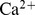 entry (SOCE) in these
entry (SOCE) in these 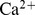 dynamics by constructing a mathematical model of ASMC
dynamics by constructing a mathematical model of ASMC 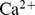 signaling based on experimental data from lung slices. The model confirms that SOCE is elicited upon sufficient
signaling based on experimental data from lung slices. The model confirms that SOCE is elicited upon sufficient 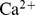 depletion of the sarcoplasmic reticulum (SR), while receptor-operated
depletion of the sarcoplasmic reticulum (SR), while receptor-operated 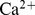 entry (ROCE) is inhibited in such conditions. It also shows that SOCE can sustain agonist-induced
entry (ROCE) is inhibited in such conditions. It also shows that SOCE can sustain agonist-induced 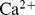 oscillations in the absence of other
oscillations in the absence of other 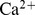 influx. SOCE up-regulation may thus contribute to AHR by increasing the
influx. SOCE up-regulation may thus contribute to AHR by increasing the 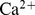 oscillation frequency that in turn regulates ASMC contraction. The model also provides an explanation for the failure of the SERCA pump blocker CPA to clamp the cytosolic
oscillation frequency that in turn regulates ASMC contraction. The model also provides an explanation for the failure of the SERCA pump blocker CPA to clamp the cytosolic 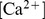 of ASMC in lung slices, by showing that CPA is unable to maintain the SR empty of
of ASMC in lung slices, by showing that CPA is unable to maintain the SR empty of 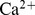 . This prediction is confirmed by experimental data from mouse lung slices, and strongly suggests that CPA only partially inhibits SERCA in ASMC.
. This prediction is confirmed by experimental data from mouse lung slices, and strongly suggests that CPA only partially inhibits SERCA in ASMC.
Introduction
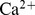 is a ubiquitous cellular messenger, controlling a wide range of biological functions. These include ASMC contraction and proliferation, which are associated with airway hyper-responsiveness (enhanced contractility) and airway remodelling (structural changes) in asthma. The main trigger for cytoplasmic
is a ubiquitous cellular messenger, controlling a wide range of biological functions. These include ASMC contraction and proliferation, which are associated with airway hyper-responsiveness (enhanced contractility) and airway remodelling (structural changes) in asthma. The main trigger for cytoplasmic 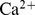 (
(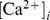 ) increase in ASMC is agonist stimulation at the cell membrane (e.g., by histamine released from mast cells or acethylcholine released from nerves). Binding of agonist to G-protein coupled receptors induces the production of
) increase in ASMC is agonist stimulation at the cell membrane (e.g., by histamine released from mast cells or acethylcholine released from nerves). Binding of agonist to G-protein coupled receptors induces the production of 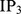 , a second messenger which diffuses into the cytosol and binds to
, a second messenger which diffuses into the cytosol and binds to 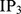 receptor
receptor 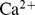 channels (IPR) on the sarcoplasmic reticulum (SR) membrane (Fig. 1). This causes the IPR to open and release
channels (IPR) on the sarcoplasmic reticulum (SR) membrane (Fig. 1). This causes the IPR to open and release 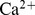 from the SR into the cytosol (the SR being the main
from the SR into the cytosol (the SR being the main 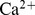 store in ASMC). As
store in ASMC). As 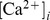 exerts a positive feedback on IPR, this results in so-called
exerts a positive feedback on IPR, this results in so-called 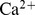 -induced
-induced 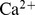 release (CICR). The release is terminated by the inhibition of the IPR at large
release (CICR). The release is terminated by the inhibition of the IPR at large 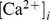 , and
, and 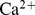 is pumped back into the SR by
is pumped back into the SR by 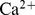 ATP-ases (SERCA). Hence, for sufficient
ATP-ases (SERCA). Hence, for sufficient 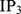 concentration, cycling of
concentration, cycling of 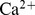 through IPR can occur, and give rise to the repetitive propagation of
through IPR can occur, and give rise to the repetitive propagation of 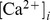 waves through the cytosol. These appear as
waves through the cytosol. These appear as 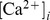 oscillations at the whole-cell level. Importantly, airway contraction increases with the frequency of these
oscillations at the whole-cell level. Importantly, airway contraction increases with the frequency of these 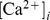 oscillations [1], [2].
oscillations [1], [2]. 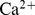 dynamics are also involved in ASMC proliferation [3]–[5], and in the assembly of myosin thick filament and actin thin filament [6]–[8], which form the contractile machinery of ASMC. In addition, several
dynamics are also involved in ASMC proliferation [3]–[5], and in the assembly of myosin thick filament and actin thin filament [6]–[8], which form the contractile machinery of ASMC. In addition, several 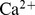 channels and pumps in ASMC are regulated by inflammatory mediators present in asthma (e.g., [4], [9]–[12]).
channels and pumps in ASMC are regulated by inflammatory mediators present in asthma (e.g., [4], [9]–[12]). 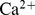 dynamics therefore appear to be involved in multiple interrelated aspects of asthma at the cellular level. In the present work, we use mathematical modelling to investigate the important
dynamics therefore appear to be involved in multiple interrelated aspects of asthma at the cellular level. In the present work, we use mathematical modelling to investigate the important 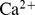 pathways at play in
pathways at play in 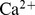 dynamics of ASMC and thus improve our understanding of airway hyper-responsiveness and remodelling in asthma.
dynamics of ASMC and thus improve our understanding of airway hyper-responsiveness and remodelling in asthma.
Figure 1. Schematic of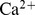 signalling in ASMC.
signalling in ASMC.
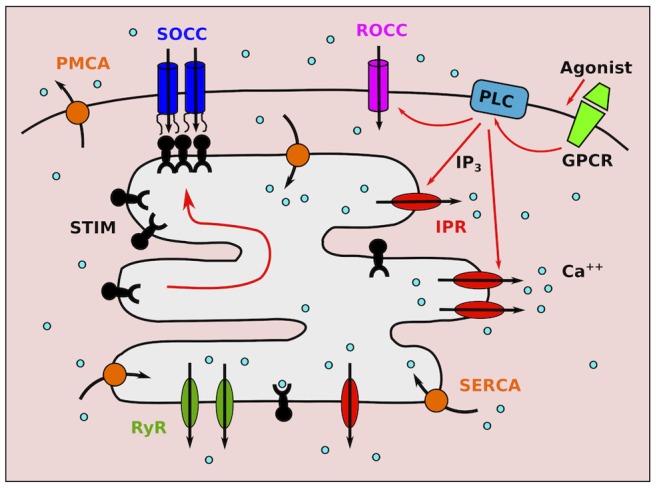
Agonist stimulation of G-protein coupled receptors (GPCR) induces PLC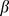 activation, giving rise to
activation, giving rise to 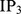 production and
production and 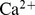 entry through receptor-operated
entry through receptor-operated 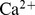 channels (ROCC).
channels (ROCC). 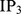 triggers
triggers 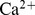 release through IPR. Depletion of the SR from
release through IPR. Depletion of the SR from 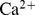 causes STIM protein oligomerisation and migration toward the cell membrane, where they bind and activate store-operated
causes STIM protein oligomerisation and migration toward the cell membrane, where they bind and activate store-operated 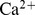 channels (SOCC).
channels (SOCC). 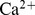 ATP-ases pump
ATP-ases pump 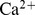 back into the SR (SERCA) and out of the cell (PMCA).
back into the SR (SERCA) and out of the cell (PMCA).
Store-operated 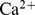 entry (SOCE) is one important
entry (SOCE) is one important 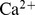 entry mechanism, in which plasma membrane (PM)
entry mechanism, in which plasma membrane (PM) 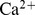 channels open in response to
channels open in response to 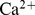 store depletion. These are called store-operated
store depletion. These are called store-operated 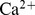 channels (SOCC). Although the concept of SOCE was proposed 25 years ago [13], the mechanism of its activation has been identified only recently [14]. The process is mediated by stromal interaction molecules (STIM), proteins embedded in the SR membrane which are sensitive to SR
channels (SOCC). Although the concept of SOCE was proposed 25 years ago [13], the mechanism of its activation has been identified only recently [14]. The process is mediated by stromal interaction molecules (STIM), proteins embedded in the SR membrane which are sensitive to SR 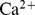 . Upon dissociation of
. Upon dissociation of 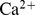 from their SR binding site, they oligomerise and translocate within the SR membrane to the plasma membrane. Here, STIM proteins bind to Orai and/or TRP, the proteins forming the pore of SOCC, and trigger their opening (Fig. 1). Although SOCE has been identified in many cells, it is generally stimulated by artificial emptying of the
from their SR binding site, they oligomerise and translocate within the SR membrane to the plasma membrane. Here, STIM proteins bind to Orai and/or TRP, the proteins forming the pore of SOCC, and trigger their opening (Fig. 1). Although SOCE has been identified in many cells, it is generally stimulated by artificial emptying of the 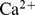 store, as there is unfortunately no specific pharmacological SOCC blocker. Hence, the importance of store depletion, and therefore of SOCE, during physiological conditions such as
store, as there is unfortunately no specific pharmacological SOCC blocker. Hence, the importance of store depletion, and therefore of SOCE, during physiological conditions such as 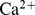 oscillations, remains largely unknown. This may explain why SOCE has been included only in a few mathematical models of
oscillations, remains largely unknown. This may explain why SOCE has been included only in a few mathematical models of 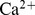 dynamics [15]–[18]. In particular, no prior modelling work on
dynamics [15]–[18]. In particular, no prior modelling work on 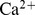 dynamics in ASMC [19]–[23] has taken SOCE into account, even though there is evidence that SOCE is up-regulated by inflammatory mediators found in asthma (TNF-
dynamics in ASMC [19]–[23] has taken SOCE into account, even though there is evidence that SOCE is up-regulated by inflammatory mediators found in asthma (TNF- and IL-13) [9], [11], [24], and is associated with ASMC proliferation [3], [5].
and IL-13) [9], [11], [24], and is associated with ASMC proliferation [3], [5].
In this paper, we develop a mathematical model to evaluate the importance of SOCE in 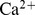 dynamics of ASMC. While there is much evidence that SOCE occurs upon SR depletion in cultured ASMC in vitro (e.g., [3], [25]–[27]), these cultured cells often lose their contractile phenotype, and rarely display agonist-induced
dynamics of ASMC. While there is much evidence that SOCE occurs upon SR depletion in cultured ASMC in vitro (e.g., [3], [25]–[27]), these cultured cells often lose their contractile phenotype, and rarely display agonist-induced 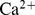 oscillations. Hence, ASMC in lung slices, which retain most of their physiological and morphological characteristics, are a more reliable preparation to study ASMC
oscillations. Hence, ASMC in lung slices, which retain most of their physiological and morphological characteristics, are a more reliable preparation to study ASMC 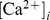 dynamics. Moreover, the available data from lung slices reflect
dynamics. Moreover, the available data from lung slices reflect 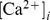 dynamics in individual ASMC, while the majority of works with cultured cells provide only global imaging of
dynamics in individual ASMC, while the majority of works with cultured cells provide only global imaging of 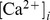 over wells containing thousands of ASMC. Therefore, we base our model on data from lung slices. SOCE has not been studied directly in lung slices, but a treatment with ryanodine-caffeine (Rya-Caf) has previously been used to clamp the cytosolic
over wells containing thousands of ASMC. Therefore, we base our model on data from lung slices. SOCE has not been studied directly in lung slices, but a treatment with ryanodine-caffeine (Rya-Caf) has previously been used to clamp the cytosolic 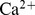 of ASMC [2], [28], [29], which relies on emptying the SR from
of ASMC [2], [28], [29], which relies on emptying the SR from 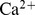 . The results of these experiments therefore provide invaluable information about SOCE. Because agonist stimulation was systematically performed before Rya-Caf treatment to ensure that the lung slice is viable, i.e., that ASMC exhibit normal
. The results of these experiments therefore provide invaluable information about SOCE. Because agonist stimulation was systematically performed before Rya-Caf treatment to ensure that the lung slice is viable, i.e., that ASMC exhibit normal 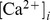 oscillations and contraction, we can construct a mathematical model of
oscillations and contraction, we can construct a mathematical model of 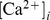 dynamics informed by these data that accounts for both physiological and non-physiological conditions. The model is then used to i) evaluate the effect of SOCE up- and down-regulation on agonist-induced
dynamics informed by these data that accounts for both physiological and non-physiological conditions. The model is then used to i) evaluate the effect of SOCE up- and down-regulation on agonist-induced 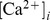 oscillations, and (ii) explain the inability of the SERCA pump blocker CPA to clamp the
oscillations, and (ii) explain the inability of the SERCA pump blocker CPA to clamp the 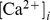 , in contrast with Rya-Caf treatment.
, in contrast with Rya-Caf treatment.
Methods
Ethics Statement
The experimental study followed the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health. The protocol was approved by the Institutional Animal Care Committee of the University of Massachusetts Medical School (Docket Number: A-836–12). Animals were euthanized with sodium pentobarbital before tissue collection.
Experimental data
Data consist of fluorescence recordings of 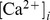 dynamics in ASMC within intact lung slices. All the materials and methods have been previously described (e.g., [2], [28]). Essentially,
dynamics in ASMC within intact lung slices. All the materials and methods have been previously described (e.g., [2], [28]). Essentially, 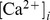 imaging was performed from regions of about 4
imaging was performed from regions of about 4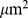 within ASMC (Fig. 2), using two-photon laser scanning microscopy. The fluorescent indicator employed was Oregon Green BAPTA-1-AM, which has a high affinity for
within ASMC (Fig. 2), using two-photon laser scanning microscopy. The fluorescent indicator employed was Oregon Green BAPTA-1-AM, which has a high affinity for 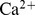 (
(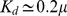 M). We use published data [2] to develop the mathematical model, and new experimental results to test the model predictions (see Results). The latter data can be made freely available upon request for academic, non-commercial use.
M). We use published data [2] to develop the mathematical model, and new experimental results to test the model predictions (see Results). The latter data can be made freely available upon request for academic, non-commercial use.
Figure 2. Fluorescence image of part of a mouse airway wall obtained by two-photon laser scanning microscopy.
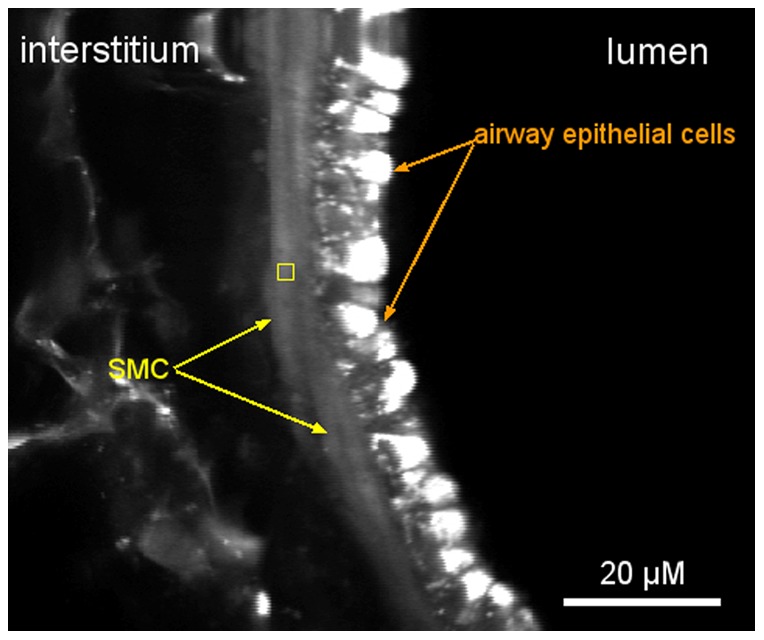
The yellow square shows a typical region, within an ASMC, from which  dynamics is imaged.
dynamics is imaged.
Mathematical model
Intracellular  dynamics are modelled at the whole-cell level, via the following system of ordinary differential equations (e.g., [30]):
dynamics are modelled at the whole-cell level, via the following system of ordinary differential equations (e.g., [30]):
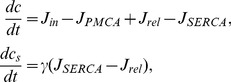 |
(1) |
where 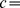
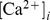 is the free cytosolic
is the free cytosolic 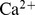 concentration, and
concentration, and 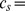
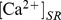 is the free SR
is the free SR 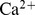 concentration.
concentration.
The term 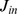 represents the total influx of
represents the total influx of 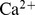 into the cytosol through PM channels;
into the cytosol through PM channels; 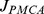 , the
, the 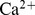 efflux through the PM
efflux through the PM 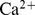 ATP-ase pumps (PMCA);
ATP-ase pumps (PMCA); 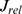 , the
, the 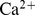 flux of
flux of 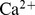 from the SR into the cytosol, and
from the SR into the cytosol, and 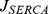 , the flux of
, the flux of 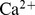 from the cytosol into the SR through the SR/ER
from the cytosol into the SR through the SR/ER 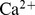 ATP-ases (SERCA). The factor
ATP-ases (SERCA). The factor  represents the ratio of cytoplasmic volume to SR volume, and implicitly incorporates the relative effect of fast, linear (e.g., low affinity)
represents the ratio of cytoplasmic volume to SR volume, and implicitly incorporates the relative effect of fast, linear (e.g., low affinity) 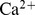 buffers in the SR compared to the effect of similar buffers in the cytosol. Indeed, the effect of fast, linear buffers amounts to a global rescaling of the
buffers in the SR compared to the effect of similar buffers in the cytosol. Indeed, the effect of fast, linear buffers amounts to a global rescaling of the 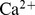 fluxes in the corresponding compartment (e.g., [30]). The other buffers are assumed to have a negligible effect on
fluxes in the corresponding compartment (e.g., [30]). The other buffers are assumed to have a negligible effect on 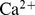 dynamics at the whole-cell level (see also Discussion).
dynamics at the whole-cell level (see also Discussion).
We assume that
| (2) |
where 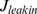 is a constant
is a constant 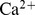 leak through unspecified channels,
leak through unspecified channels, 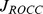 is the
is the 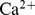 influx through receptor-operated
influx through receptor-operated 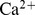 channels (ROCC) and
channels (ROCC) and 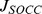 the influx through SOCC. We neglect the
the influx through SOCC. We neglect the 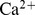 influx through voltage-operated
influx through voltage-operated 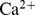 channels (VOCC) because membrane depolarisation plays little role during agonist-induced
channels (VOCC) because membrane depolarisation plays little role during agonist-induced 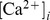 signalling and contraction in ASMC (in contrast to other types of muscle cells, including vascular smooth muscle cells, where action potentials are crucial to contraction) [1], [31]. The
signalling and contraction in ASMC (in contrast to other types of muscle cells, including vascular smooth muscle cells, where action potentials are crucial to contraction) [1], [31]. The 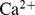 influxes are modelled by:
influxes are modelled by:
| (3a) |
| (3b) |
| (3c) |
where 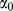 and
and 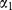 are constants,
are constants, 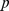 is the agonist concentration,
is the agonist concentration, 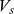 is the maximum SOCC flux, and
is the maximum SOCC flux, and 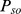 represents the fraction of STIM proteins bound to Orai/TRP proteins, i.e. the fraction of activated SOCC. This fraction adapts slowly to changes in
represents the fraction of STIM proteins bound to Orai/TRP proteins, i.e. the fraction of activated SOCC. This fraction adapts slowly to changes in 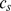 , because the diffusion of STIM within the SR membrane is a slow process [32]. We model this phenomenologically by
, because the diffusion of STIM within the SR membrane is a slow process [32]. We model this phenomenologically by
| (4a) |
| (4b) |
The steady-state function 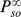 can be interpreted as the fraction of STIM proteins dissociated from SR
can be interpreted as the fraction of STIM proteins dissociated from SR 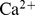 (as a consequence of store depletion), and thus able to oligomerise and move toward the PM to bind with Orai and/or TRP (see also Discussion).
(as a consequence of store depletion), and thus able to oligomerise and move toward the PM to bind with Orai and/or TRP (see also Discussion). 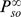 is therefore a decreasing function of
is therefore a decreasing function of 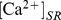 , which we model by the reverse Hill function Eq. (0b), assuming affinity
, which we model by the reverse Hill function Eq. (0b), assuming affinity 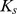 for
for 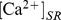 and Hill coefficient
and Hill coefficient 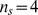 [33].
[33].
The total 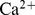 flux from the SR into the cytosol is given by
flux from the SR into the cytosol is given by
| (8) |
where 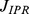 is the
is the 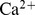 flux through
flux through 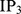 receptors (IPR),
receptors (IPR), 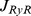 the
the 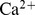 flux through ryanodine receptors (RyR), and
flux through ryanodine receptors (RyR), and 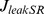 an unspecified
an unspecified 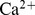 leak out of the SR. We use the formulation (e.g., [30]):
leak out of the SR. We use the formulation (e.g., [30]):
| (9) |
where 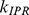 (resp.
(resp. 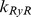 ) is the maximum rate of
) is the maximum rate of 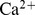 flow through IPR (resp. RyR). Following [23], the IPR opening probability
flow through IPR (resp. RyR). Following [23], the IPR opening probability 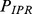 is modelled using the Li-Rinzel/Tang et al. reduction of the De Young-Keizer (DYK) model [34]–[36]:
is modelled using the Li-Rinzel/Tang et al. reduction of the De Young-Keizer (DYK) model [34]–[36]:
| (10) |
where 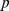 is the
is the 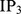 concentration, and
concentration, and 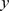 is the fraction of inhibited IPR. The latter obeys
is the fraction of inhibited IPR. The latter obeys
| (11) |
with
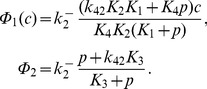 |
(12) |
The parameters 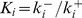 (
(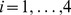 ) are equilibrium constants for
) are equilibrium constants for 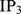 and
and 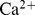 binding/unbinding to the IPR; we use the original values from the DYK model [34]. The value of
binding/unbinding to the IPR; we use the original values from the DYK model [34]. The value of 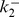 is scaled so that the range of
is scaled so that the range of 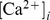 oscillation frequencies matches the experimental range, with the ratio
oscillation frequencies matches the experimental range, with the ratio 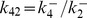 kept constant to the value in ref. [30] (see also Table 1). In the experiments modelled in this work, either RyR play a negligible role, or they are locked open by Rya-Caf treatment (see Results). Hence, we neglect their dynamics and set the fraction of open RyR,
kept constant to the value in ref. [30] (see also Table 1). In the experiments modelled in this work, either RyR play a negligible role, or they are locked open by Rya-Caf treatment (see Results). Hence, we neglect their dynamics and set the fraction of open RyR, 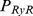 , either to 0 or 1 depending on the experiment considered.
, either to 0 or 1 depending on the experiment considered.
Table 1. Parameter values used in the model.
| Parameter | symbol | value | units | reference |
| PMCA maximum flux | Vp | 7.5 | μM/s | this work |
| PMCA affinity | Kp | 1.5 | μM | 0.1–1 [59] |
| SOCE maximum flux | Vs | 1.57 | μM/s | this work |
| STIM SR Ca2+ affinity | Ks | 50 | μM |
 c*
s/2 c*
s/2 |
| SOCE Hill exponent | ns | 4 | [33] | |
| SOCE timescale | τs | 30 | s | [32] |
| Constant leak influx | α0 | 0 | μM/s |
 Vs
Vs
|
| Cyt/SR vol. × buffer effects | γ | 5.405 | [34], [60] | |
| ROCE rate | α1 | 0.00105 | s−1 | this work |
| SERCA maximum flux | Ve | 5 | μM/s | this work |
| SERCA affinity | Ke | 0.1 | μM | 0.1–1 [61] |
| CPA effect timescale | τe | 30 | s | ∼ min |
| IPR rate | kIPR | 0.667 | s−1 | this work |
| Agonist concentration |
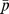
|
1 | μM | this work |
| Agonist effect timescale | τp | 30 | s | ∼ min |
| SR leak rate | JSR | 0.01 | s−1 |
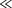 kIPR
kIPR
|
| RyR leak rate (Rya-Caf effect) | KRYR | 0.19 | s−2 | this work |
| Rya-Caf effect timescale | τSR | 10 | s | ∼ min |
IPR affinity for 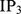
|
K1 | 0.138 | μM | [34] |
| IPR affinity for Ca2+ (inhib. site) | K2 | 1.05 | μM | [34] |
| IPR affinity for IP3 | K3 | 0.943 | μM | [34] |
| IPR affinity for Ca2+ (inhib. site) | K4 | 0.144 | μM | [34] |
| IPR affinity for Ca2+ (activ. site) | K5 | 0.082 | μM | [34] |
| IPR Ca2+ dissoc. rate (inhib. site) | k−2 | 0.167 | s−1 | this work |
| IPR Ca2+ dissoc. rate (inhib. site) | k−4 | 0.138 k−2 | s−1 | [34] |
The 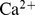 ATP-ases are modelled using the usual expressions (e.g., [30]):
ATP-ases are modelled using the usual expressions (e.g., [30]):
| (13) |
We do not model 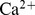 pumping into mitochondria explicitly, but acknowledge that a portion of the extrusion process attributed to PMCA might actually be performed by mitochondria uniporters, as these might be activated at average
pumping into mitochondria explicitly, but acknowledge that a portion of the extrusion process attributed to PMCA might actually be performed by mitochondria uniporters, as these might be activated at average 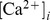 as low as
as low as 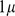 M [20].
M [20].
Gathering all expressions, the model is described by:
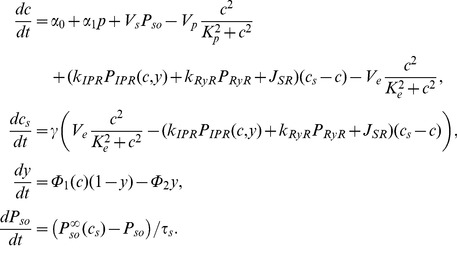 |
(14) |
In addition to Eq. (11), we use the following expressions to account for the time needed by drugs to reach full effect:
| (15) |
| (16) |
| (17) |
These equations describe respectively agonist stimulation, Rya-Caf treatment, and SERCA block by CPA (see Results).
Unless otherwise mentioned, parameter values were freely adapted (within physiological ranges when they are known) to account for the experimental results. The values retained are listed in Table 1. The fitting was performed “by hand” (i.e., no algorithmic method was used) within the Mathematica “Manipulate” environment (a useful framework for fitting an ODE model to several experimental results as it enables visualisation of the effect of a parameter change on several ODE integrations almost instantaneously). The code can be made freely available upon request for academic, non-commercial use.
All simulations were run from the same initial condition as in the experiment, which is usually the physiological equilibrium. Bifurcation diagrams were computed using the numerical continuation software AUTO [37], [38].
Results
Accounting for  dynamics of AMSC in lung slices
dynamics of AMSC in lung slices
Fig. 3A-C shows representative  dynamics of an ASMC in a human lung slice in response to a three-step experimental protocol [2]. This protocol was originally designed to clamp the
dynamics of an ASMC in a human lung slice in response to a three-step experimental protocol [2]. This protocol was originally designed to clamp the 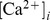 of ASMC, in order to study independently the effects of agonist and
of ASMC, in order to study independently the effects of agonist and 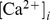 on airway contraction [28]. The slice is first stimulated with agonist (histamine), to verify its viability (Fig. 3A). This induces
on airway contraction [28]. The slice is first stimulated with agonist (histamine), to verify its viability (Fig. 3A). This induces 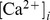 oscillations. Agonist is then washed from the slice, and a Rya-Caf treatment is applied (Fig. 3B). This creates a permanent
oscillations. Agonist is then washed from the slice, and a Rya-Caf treatment is applied (Fig. 3B). This creates a permanent 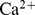 leak through RyR, because caffeine opens RyR and ryanodine locks them open irreversibly. If this
leak through RyR, because caffeine opens RyR and ryanodine locks them open irreversibly. If this 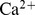 leak is large enough, it keeps the SR empty and prevents any further change in
leak is large enough, it keeps the SR empty and prevents any further change in 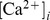 , unless extracellular
, unless extracellular 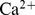 is modified. The effectiveness of the treatment is confirmed by the second application of agonist (Fig. 3C): no further
is modified. The effectiveness of the treatment is confirmed by the second application of agonist (Fig. 3C): no further 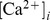 increase is triggered, showing that
increase is triggered, showing that 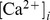 is clamped. It is important to emphasise that these results are not specific to histamine stimulation of human lung slices: similar results have been obtained in mouse and rat lung slices with methylcholine (Fig. 6 in ref. [29], Figs. 5B and 6C-D in ref. [28]).
is clamped. It is important to emphasise that these results are not specific to histamine stimulation of human lung slices: similar results have been obtained in mouse and rat lung slices with methylcholine (Fig. 6 in ref. [29], Figs. 5B and 6C-D in ref. [28]).
Figure 3.
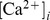 dynamics in ASMC: experiment and model.
dynamics in ASMC: experiment and model.
(A)–(C): Fluorescence imaging of 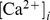 dynamics in an ASMC within a human lung slice, during the following 3-step experiment: (A) Agonist stimulation, (B) Rya-Caf treatment, and (C) second agonist stimulation. Following the irreversible Rya-Caf treatment in (B), agonist stimulation (C) is no longer able to elicit
dynamics in an ASMC within a human lung slice, during the following 3-step experiment: (A) Agonist stimulation, (B) Rya-Caf treatment, and (C) second agonist stimulation. Following the irreversible Rya-Caf treatment in (B), agonist stimulation (C) is no longer able to elicit 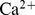 oscillations, nor does it perturb the new elevated
oscillations, nor does it perturb the new elevated 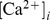 equilibrium. Reprinted from
[2]
under a CC BY license, with permission of the American Thoracic Society, original copyright 2010. Cite: Ressmeyer et al. /2010/Am J Respir Cell Mol Biol/43/179–191. Official journal of the American Thoracic Society. This modified figure is based on the original figure available from
www.atsjournals.org. (D)–(F): Simulations of the experiments in (A)–(C) using Eqs. 114–12 and the parameter values in Table 1. The evolution of
equilibrium. Reprinted from
[2]
under a CC BY license, with permission of the American Thoracic Society, original copyright 2010. Cite: Ressmeyer et al. /2010/Am J Respir Cell Mol Biol/43/179–191. Official journal of the American Thoracic Society. This modified figure is based on the original figure available from
www.atsjournals.org. (D)–(F): Simulations of the experiments in (A)–(C) using Eqs. 114–12 and the parameter values in Table 1. The evolution of 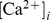 ,
, 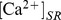 , and
, and 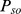 (fraction of open SOCC) are shown (cf. legend in (E)).
(fraction of open SOCC) are shown (cf. legend in (E)).
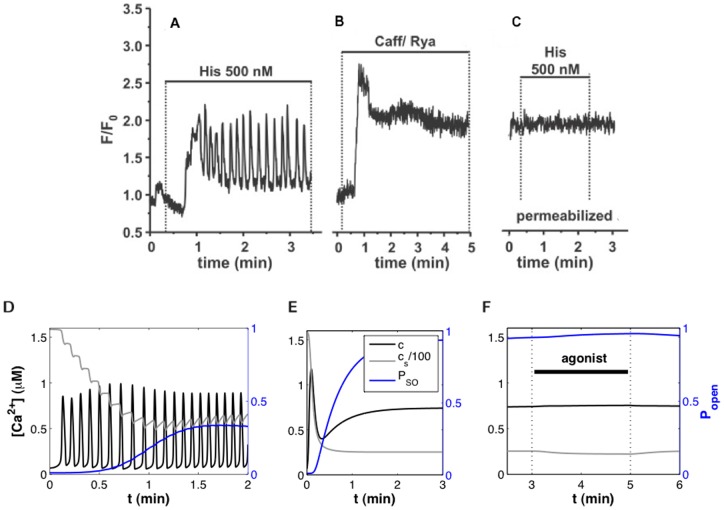
Figure 6. Effect of CPA on 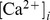 dynamics.
dynamics.
(A) Fluorescence imaging of 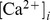 in ASMC of a mouse lung slice treated with agonist and CPA. Agonist removal leads to
in ASMC of a mouse lung slice treated with agonist and CPA. Agonist removal leads to 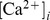 decrease. (B–D) Model simulations of the experiments shown in (A), assuming that (B) CPA quickly blocks the SERCA, (C) CPA slowly blocks the SERCA, (D) CPA partially blocks the SERCA but reaches maximum strength rather quickly. Black solid and dashed curves (left y-axis) represent respectively
decrease. (B–D) Model simulations of the experiments shown in (A), assuming that (B) CPA quickly blocks the SERCA, (C) CPA slowly blocks the SERCA, (D) CPA partially blocks the SERCA but reaches maximum strength rather quickly. Black solid and dashed curves (left y-axis) represent respectively 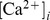 and
and 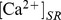 ; blue and red curves (right y-axis) show respectively the fraction of open SOCC and the fraction of operating SERCA (that is,
; blue and red curves (right y-axis) show respectively the fraction of open SOCC and the fraction of operating SERCA (that is, 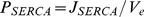 , where
, where 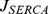 is given by Eq. (0c)).
is given by Eq. (0c)).
Figure 5. Influence of SOCE on agonist-induced 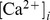 oscillations.
oscillations.
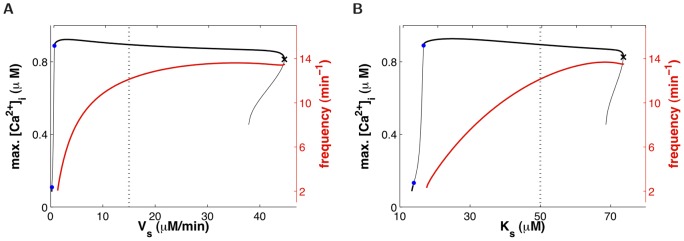
Amplitude (black) and frequency (red) of  oscillations as a function of (A) SOCE maximum rate,
oscillations as a function of (A) SOCE maximum rate, 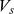 , and (B) STIM affinity for SR
, and (B) STIM affinity for SR 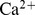 ,
, 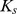 . Dotted lines indicate the “normal” parameter values (Table 1, Figs. 3D–F). As in Fig. 4, only the frequency of the large-amplitude stable
. Dotted lines indicate the “normal” parameter values (Table 1, Figs. 3D–F). As in Fig. 4, only the frequency of the large-amplitude stable 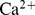 oscillations is shown.
oscillations is shown.
The mathematical model enables the deduction of valuable information from the experimental results. First, from Eq. (14), the new, elevated, 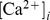 equilbrium reached after Rya-Caf treatment satisfies:
equilbrium reached after Rya-Caf treatment satisfies:
| (13a) |
| (13b) |
where 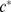 and
and 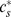 are respectively the equilibrium
are respectively the equilibrium 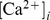 and
and 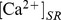 . An important consequence of (18) is that, in the absence of SOCE,
. An important consequence of (18) is that, in the absence of SOCE, 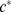 depends only on the
depends only on the 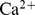 fluxes through the PM. This may seem surprising, as any increase in
fluxes through the PM. This may seem surprising, as any increase in 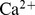 flux out of the SR (
flux out of the SR (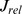 in Eq. (1)) is expected to increase
in Eq. (1)) is expected to increase 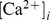 . However, the equilibrium equation (18) tells us that such an increase would only be transient (because the PMCA pumping rate is an increasing function of
. However, the equilibrium equation (18) tells us that such an increase would only be transient (because the PMCA pumping rate is an increasing function of 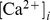 ), unless there is a concomitant permanent increase in
), unless there is a concomitant permanent increase in 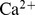 influx through the PM. Hence, the persistence of an elevated
influx through the PM. Hence, the persistence of an elevated 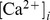 means that a permanent SOCE has been elicited (as SOCE is the only
means that a permanent SOCE has been elicited (as SOCE is the only 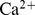 influx capable of increase upon Rya-Caf treatment). Moreover, the model indicates that ROCE is negligible after Rya-Caf treatment. Indeed, if it was not, the addition of agonist would increase
influx capable of increase upon Rya-Caf treatment). Moreover, the model indicates that ROCE is negligible after Rya-Caf treatment. Indeed, if it was not, the addition of agonist would increase 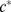 via the increase in
via the increase in 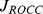 . Hence, we assume that the ROCE rate
. Hence, we assume that the ROCE rate 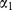 is small (see Table 1 and Discussion).
is small (see Table 1 and Discussion).
Results of “hand-fitting” the model to the experimental results are shown in Figs. 3D–F and Fig. 4, with the corresponding parameter values listed in Table 1. The model reproduces (i) the agonist-induced 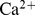 oscillations, (ii) the similar magnitudes of the new equilibrium
oscillations, (ii) the similar magnitudes of the new equilibrium 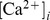 in Fig. 3B and the amplitude of the oscillations in Fig. 3A, and (iii) the negligible effect of agonist stimulation after Rya-Caf treatment. Agonist-induced
in Fig. 3B and the amplitude of the oscillations in Fig. 3A, and (iii) the negligible effect of agonist stimulation after Rya-Caf treatment. Agonist-induced 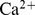 oscillations were simulated with
oscillations were simulated with 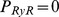 because RyR appear to play a negligible role during agonist-induced
because RyR appear to play a negligible role during agonist-induced 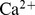 oscillations [2], [39]. On the other hand, the response to Rya-Caf was simulated with
oscillations [2], [39]. On the other hand, the response to Rya-Caf was simulated with 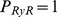 since the treatment locks open the RyR. We did not attempt to reproduce the magnitude of the initial spike response to Rya-Caf treatment relative to that of the subsequent
since the treatment locks open the RyR. We did not attempt to reproduce the magnitude of the initial spike response to Rya-Caf treatment relative to that of the subsequent 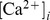 plateau (Fig. 3B) because the fluorescent dye used in the experiments saturates rapidly with
plateau (Fig. 3B) because the fluorescent dye used in the experiments saturates rapidly with 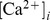 . Parameter values were also adjusted to yield physiological
. Parameter values were also adjusted to yield physiological 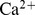 equilibrium concentrations (
equilibrium concentrations (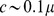 M [40] and
M [40] and 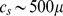 M [41]), realistic
M [41]), realistic 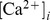 oscillation amplitude (
oscillation amplitude (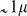 M), and to reproduce the range of
M), and to reproduce the range of 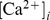 oscillation frequencies observed in human lung slices as a function of agonist (0.5–11/min [2]). More detail on the parameter estimation procedure is given in Supporting Information S1.
oscillation frequencies observed in human lung slices as a function of agonist (0.5–11/min [2]). More detail on the parameter estimation procedure is given in Supporting Information S1.
Figure 4.
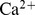 dynamics as a function of agonist concentration.
dynamics as a function of agonist concentration.
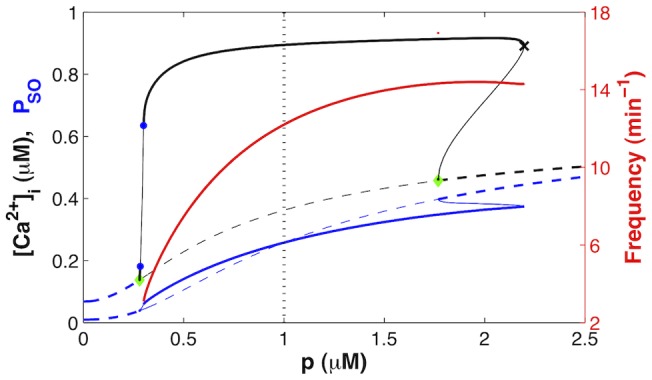
Dashed curves represent steady-states (constant  levels); solid curves, periodic solutions (
levels); solid curves, periodic solutions (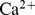 oscillations). The maximum
oscillations). The maximum 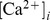 (black) and the maximum fraction of open SOCC (blue) during one solution period are plotted as ordinates. The red curve (right y-axis) shows the frequency of the
(black) and the maximum fraction of open SOCC (blue) during one solution period are plotted as ordinates. The red curve (right y-axis) shows the frequency of the 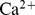 oscillations on the main stable segment (from the upper blue dot to the black cross), which fits the experimental range in human [2]. The stable solutions are represented as thick lines and unstable solutions as thin lines. The green diamonds represent Hopf bifurcations, the black cross, a saddle-node bifurcation, and the blue dots, period-doubling points. Period-doubled branches are not shown because they extend only over a tiny range of
oscillations on the main stable segment (from the upper blue dot to the black cross), which fits the experimental range in human [2]. The stable solutions are represented as thick lines and unstable solutions as thin lines. The green diamonds represent Hopf bifurcations, the black cross, a saddle-node bifurcation, and the blue dots, period-doubling points. Period-doubled branches are not shown because they extend only over a tiny range of 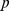 values; moreover it is likely that the deterministic description of
values; moreover it is likely that the deterministic description of 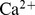 oscillations fails at these low agonist concentrations (see Discussion). The vertical dotted line indicates the value of
oscillations fails at these low agonist concentrations (see Discussion). The vertical dotted line indicates the value of 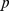 used in Fig. 3 (Table 1).
used in Fig. 3 (Table 1).
Fig. 4 shows the bifurcation diagram of the model as a function of agonist concentration. Periodic solutions (i.e., 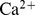 oscillations) arise through a Hopf bifurcation, and disappear through a saddle-node bifurcation of limit cycles. A second Hopf bifurcation is present on the steady-state branch, and is associated with a region of bistability between the steady-state and the periodic solution at the right of the bifurcation diagram. It is not known whether such bistability occurs in reality. It should also be noted that the steady-state
oscillations) arise through a Hopf bifurcation, and disappear through a saddle-node bifurcation of limit cycles. A second Hopf bifurcation is present on the steady-state branch, and is associated with a region of bistability between the steady-state and the periodic solution at the right of the bifurcation diagram. It is not known whether such bistability occurs in reality. It should also be noted that the steady-state 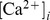 increases with agonist concentration, as is expected (e.g., [30]). This increase is provided by SOCE in our model. Indeed, the
increases with agonist concentration, as is expected (e.g., [30]). This increase is provided by SOCE in our model. Indeed, the 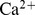 flux through IPR increases with agonist, so that store depletion increases as well.
flux through IPR increases with agonist, so that store depletion increases as well.
Effect of SOCE regulation on agonist-induced 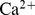 oscillations
oscillations
SOCE is the main 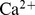 influx in the model, as ROCE is negligible (see above) and the
influx in the model, as ROCE is negligible (see above) and the 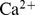 leak influx is (by definition) small. Fig. 3D shows that while SOCE is almost zero at physiological equilibrium (initial condition), it substantially increases during agonist-induced
leak influx is (by definition) small. Fig. 3D shows that while SOCE is almost zero at physiological equilibrium (initial condition), it substantially increases during agonist-induced 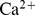 oscillations (final condition; see also Fig. 4), due to significant SR
oscillations (final condition; see also Fig. 4), due to significant SR 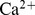 depletion. Therefore, changes in SOCE can be expected to have a substantial effect on
depletion. Therefore, changes in SOCE can be expected to have a substantial effect on 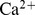 oscillations. This is quantified in Fig. 5, where the amplitude and frequency of
oscillations. This is quantified in Fig. 5, where the amplitude and frequency of 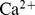 oscillations are plotted as a function of (a) the maximum SOCE rate,
oscillations are plotted as a function of (a) the maximum SOCE rate, 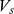 , and (b) STIM affinity for
, and (b) STIM affinity for 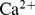 ,
, 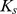 (the
(the 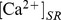 at which half SOCC are open). It is found that the
at which half SOCC are open). It is found that the 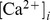 oscillation frequency varies as much with
oscillation frequency varies as much with 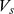 and
and 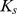 at fixed agonist concentration (Fig. 5) as it varies with agonist concentration at fixed SOCE parameters (Fig. 4). Moreover, a too big departure from the “normal” values (dotted lines, Table 1) leads to the extinction of the
at fixed agonist concentration (Fig. 5) as it varies with agonist concentration at fixed SOCE parameters (Fig. 4). Moreover, a too big departure from the “normal” values (dotted lines, Table 1) leads to the extinction of the 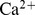 oscillations (via a Hopf bifurcation to the left, and a saddle-node to the right, of the bifurcation diagrams in Figs. 5A–B). These results are not very surprising to the extent that
oscillations (via a Hopf bifurcation to the left, and a saddle-node to the right, of the bifurcation diagrams in Figs. 5A–B). These results are not very surprising to the extent that 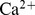 oscillations are expected to depend crucially on
oscillations are expected to depend crucially on 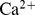 influx (e.g., [42]). However, they suggest that SOCE could play a role in AHR since (i)
influx (e.g., [42]). However, they suggest that SOCE could play a role in AHR since (i) 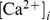 oscillations mediate ASMC contraction, and (ii) SOCE up-regulation (which increases
oscillations mediate ASMC contraction, and (ii) SOCE up-regulation (which increases 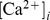 oscillation frequency) can be triggered by inflammatory mediators commonly found in asthma [9], [11], [24].
oscillation frequency) can be triggered by inflammatory mediators commonly found in asthma [9], [11], [24].
Partial inhibition of SERCA by CPA
We now apply the model to experimental data from mouse lung slices showing an attempt to clamp 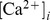 with the SERCA blocker CPA, instead of Rya-Caf treatment (Fig. 6). After inducing
with the SERCA blocker CPA, instead of Rya-Caf treatment (Fig. 6). After inducing 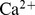 oscillations with agonist, CPA is applied in the presence of agonist (for faster emptying of the SR than CPA alone) and causes a gradual damping of the
oscillations with agonist, CPA is applied in the presence of agonist (for faster emptying of the SR than CPA alone) and causes a gradual damping of the 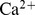 oscillations, together with a rise of the
oscillations, together with a rise of the 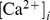 baseline, until the oscillations become undistinguishable from fluctuations around an elevated steady
baseline, until the oscillations become undistinguishable from fluctuations around an elevated steady 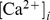 mean. Because CPA is believed to inhibit SERCA, the assumption, at this stage of the experiment, is that the SR is empty and SOCE fully active. However, when agonist is removed (CPA remains),
mean. Because CPA is believed to inhibit SERCA, the assumption, at this stage of the experiment, is that the SR is empty and SOCE fully active. However, when agonist is removed (CPA remains), 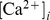 falls. When agonist is reapplied,
falls. When agonist is reapplied, 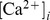 increases. These
increases. These 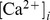 responses to agonist addition and removal are not observed when SOCE is evoked by Rya-Caf treatment. According to our model (Eq.(18)), the decrease in
responses to agonist addition and removal are not observed when SOCE is evoked by Rya-Caf treatment. According to our model (Eq.(18)), the decrease in 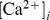 upon agonist removal indicates that SOCE does not remain activated, i.e. that the SR refills with
upon agonist removal indicates that SOCE does not remain activated, i.e. that the SR refills with 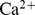 . This suggests that the SERCA are not completely blocked by CPA, as illustrated by the simulations in Fig. 6B–D. If CPA was to fully block the SERCA (Fig. 6B),
. This suggests that the SERCA are not completely blocked by CPA, as illustrated by the simulations in Fig. 6B–D. If CPA was to fully block the SERCA (Fig. 6B), 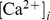 would not decrease upon agonist removal. If
would not decrease upon agonist removal. If 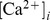 falls, it must be because either CPA requires a longer time than that used in the experiment to fully block the SERCA (Fig. 6C), or CPA achieves only partial block of the SERCA (Fig. 6D).
falls, it must be because either CPA requires a longer time than that used in the experiment to fully block the SERCA (Fig. 6C), or CPA achieves only partial block of the SERCA (Fig. 6D).
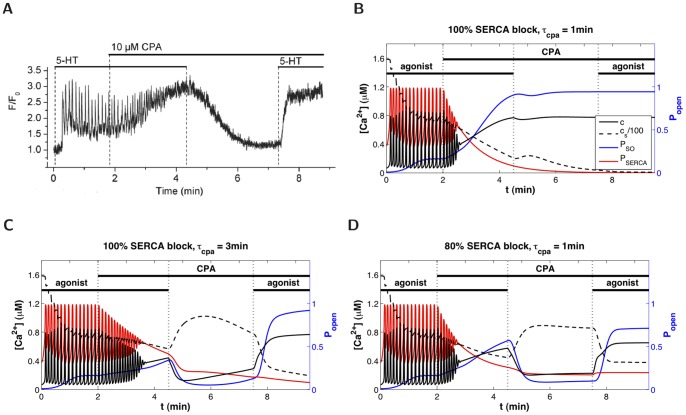
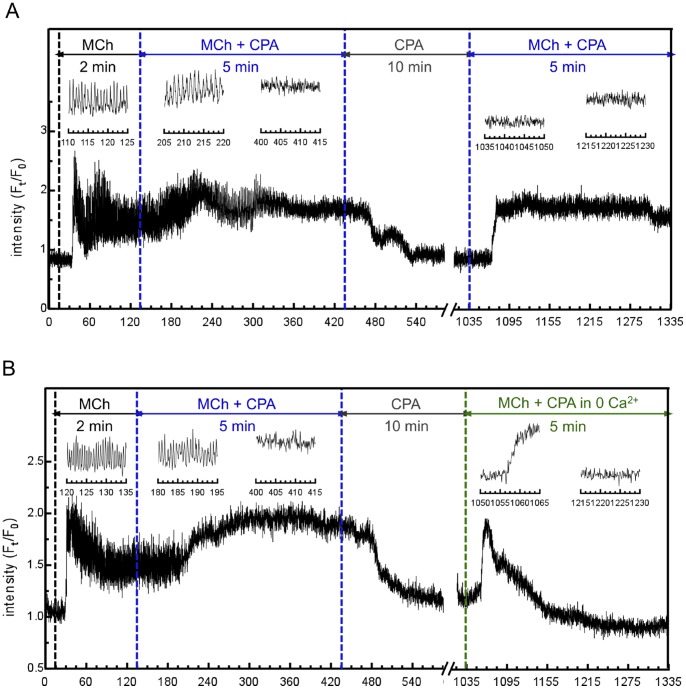
Experiments of longer duration were performed to test the model predictions. Fig. 7A shows that if CPA is applied in the presence of agonist for 5 minutes, followed by CPA only for a further 10 minutes, 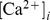 still returns to the original equilibrium level when agonist is removed, and remains low until agonist is reintroduced. This suggests that the explanation in Fig. 6C can be rejected, otherwise the longer exposure to CPA should yield a result similar to Fig. 6B. The inability of CPA to fully empty the SR of
still returns to the original equilibrium level when agonist is removed, and remains low until agonist is reintroduced. This suggests that the explanation in Fig. 6C can be rejected, otherwise the longer exposure to CPA should yield a result similar to Fig. 6B. The inability of CPA to fully empty the SR of 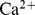 is confirmed by Fig. 7B, where extracellular calcium is removed before agonist is applied a second time, to prevent any potential ROCE. The
is confirmed by Fig. 7B, where extracellular calcium is removed before agonist is applied a second time, to prevent any potential ROCE. The 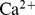 response induced can thus be unambiguously attributed to
response induced can thus be unambiguously attributed to 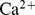 release from the SR.
release from the SR.
Figure 7. Experimental evidence that CPA does not fully empty the SR of ASMC.
Tests of the model predictions shown in Fig. 6B–D, performed with mouse lung slices. (A) Significantly longer exposure to agonist+CPA and to CPA than in Fig. 6A still fails to maintain SOCE. (B) Same experiment as in (A) except that extracellular 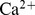 is removed before agonist is applied a second time, confirming the residual presence of
is removed before agonist is applied a second time, confirming the residual presence of 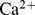 in the SR and hence the partial efficacy of CPA to inhibit SERCA (scenario of Fig. 6D). (Insets show magnifications of selected time windows).
in the SR and hence the partial efficacy of CPA to inhibit SERCA (scenario of Fig. 6D). (Insets show magnifications of selected time windows).
Hence, our combined modelling and experimental study indicates that CPA blocks only partially the SERCA of ASMC in lung slices (scenario simulated in Fig. 6D). This is a potentially important result given the wide use of CPA in cell biology to study SOCE. We note that Figs. 6A and 7 could also be explained by a model assuming that ROCE, instead of SOCE, is the main 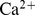 influx (e.g., [23]). However, such a model would fail to explain the outcome of Rya-Caf treatment in human and mouse lung slices (both the persistent elevated
influx (e.g., [23]). However, such a model would fail to explain the outcome of Rya-Caf treatment in human and mouse lung slices (both the persistent elevated 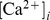 in the absence of agonist, and the absence of effect of agonist on this elevated
in the absence of agonist, and the absence of effect of agonist on this elevated 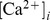 ). In contrast, our model, constructed to account for both agonist-induced oscillations and Rya-Caf treatment, explains the CPA results without requiring any modification. Its prediction holds provided CPA is not a 100% efficient SERCA blocker, and this hypothesis is supported by the experimental data in Fig. 7.
). In contrast, our model, constructed to account for both agonist-induced oscillations and Rya-Caf treatment, explains the CPA results without requiring any modification. Its prediction holds provided CPA is not a 100% efficient SERCA blocker, and this hypothesis is supported by the experimental data in Fig. 7.
Discussion
Modelling SOCE
Our mathematical model accounts for the two main properties of SOCE: 1) SOCE is an increasing function of 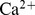 store depletion, and 2) it activates slowly upon store depletion. While the mechanisms of SOCE activation are rather well understood [14], [32], the mechanisms of SOCE termination remain less clear [43], [44]. Hence, we do not explicitly distinguish between SOCE activation and inactivation in the model, and use a single parameter
store depletion, and 2) it activates slowly upon store depletion. While the mechanisms of SOCE activation are rather well understood [14], [32], the mechanisms of SOCE termination remain less clear [43], [44]. Hence, we do not explicitly distinguish between SOCE activation and inactivation in the model, and use a single parameter 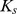 for STIM affinity for SR
for STIM affinity for SR 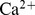 and a single time constant
and a single time constant 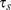 for the slow adaptation to changes in
for the slow adaptation to changes in 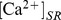 . This is also justified by the fact that most experimental data available on SOCE come from a category of SOCC called CRACC (
. This is also justified by the fact that most experimental data available on SOCE come from a category of SOCC called CRACC (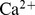 -release-activated
-release-activated 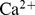 channels), which are highly selective to
channels), which are highly selective to 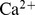 , while there is evidence that SOCE in ASMC (and in other cells) occurs at least in part through non-selective
, while there is evidence that SOCE in ASMC (and in other cells) occurs at least in part through non-selective 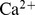 channels (NSCC). It could be that the latter operate somewhat differently from CRACC in response to store depletion or refilling.
channels (NSCC). It could be that the latter operate somewhat differently from CRACC in response to store depletion or refilling.
Our description of SOCE slow activation upon store depletion is continuous, which is easy to handle computationally, and compatible with experimental knowledge. Indeed, it is reasonable to assume that a small fraction of STIM proteins reside in close proximity to the PM, and may thus bind Orai quickly upon store depletion. Hence, a weak SOCE is likely to occur almost instantaneously upon store depletion, rendering unnecessary to introduce a finite activation delay in the model via a delay-differential equation.
We are aware of only few prior works on 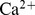 dynamics that include a mathematical description of SOCE, all of which are ODE models [15]–[18]. The first two were published before the molecular basis for SOCE was established. The latter two works include more realistic descriptions of SOCE, but none of them accounts for the slow translocation of oligomerised STIM to the PM, while it is recognised as the rate-limiting event for SOCE activation [32]. Ong et al. however assume a slow diffusion of
dynamics that include a mathematical description of SOCE, all of which are ODE models [15]–[18]. The first two were published before the molecular basis for SOCE was established. The latter two works include more realistic descriptions of SOCE, but none of them accounts for the slow translocation of oligomerised STIM to the PM, while it is recognised as the rate-limiting event for SOCE activation [32]. Ong et al. however assume a slow diffusion of 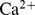 between internal SR and superficial SR (modelled as distinct compartments exchanging
between internal SR and superficial SR (modelled as distinct compartments exchanging 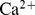 ), with SOCE being triggered by peripheral SR depletion [17]. Liu et al. explicitly model both SR
), with SOCE being triggered by peripheral SR depletion [17]. Liu et al. explicitly model both SR 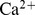 dissociation from STIM and binding of STIM to Orai. Both models are used to study transient
dissociation from STIM and binding of STIM to Orai. Both models are used to study transient 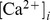 responses only;
responses only; 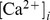 oscillations are not considered. Prior models of
oscillations are not considered. Prior models of 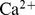 dynamics specific to ASMC did not include SOCE, while we have shown that this is necessary to account for several experimental results obtained with lung slices. The work of Haberichter et al. [19] focused on the influence of the different IPR isoforms on
dynamics specific to ASMC did not include SOCE, while we have shown that this is necessary to account for several experimental results obtained with lung slices. The work of Haberichter et al. [19] focused on the influence of the different IPR isoforms on 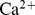 signalling in ASMC. Brumen et al. studied the influence of the total
signalling in ASMC. Brumen et al. studied the influence of the total 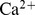 content on the nature (damped or sustained) and frequency of agonist-induced
content on the nature (damped or sustained) and frequency of agonist-induced 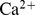 oscillations [21]. Roux et al. did not model
oscillations [21]. Roux et al. did not model 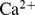 oscillations, but transient
oscillations, but transient 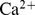 responses to caffeine [20]. Finally, the model by Wang et al.
[23] addressed the different contributions of IPR and RyR to agonist-induced and KCl-induced
responses to caffeine [20]. Finally, the model by Wang et al.
[23] addressed the different contributions of IPR and RyR to agonist-induced and KCl-induced 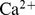 oscillations in ASMC.
oscillations in ASMC.
From the mathematical point of view, the fact that SOCE is an explicit function of store 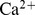 renders the models of
renders the models of 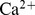 dynamics including this influx qualitatively different from those which do not, as SOCE couples the homogenous steady-state
dynamics including this influx qualitatively different from those which do not, as SOCE couples the homogenous steady-state 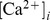 to
to 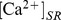 (Eq. (18)). This property is essential for the predictions of our model (in particular, the persistence of an elevated
(Eq. (18)). This property is essential for the predictions of our model (in particular, the persistence of an elevated 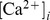 upon sustained store depletion in the absence of agonist). On the other hand, whether SOCE is an instantaneous or delayed function of
upon sustained store depletion in the absence of agonist). On the other hand, whether SOCE is an instantaneous or delayed function of 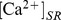 appears to have little effect on our results.
appears to have little effect on our results.
SOCE vs. ROCE
While Fig. 3C (as well as Fig. 6 in ref. [29], Figs. 5B and 6C-D in ref. [28]) shows that no ROCE is elicited by agonist following Rya-Caf treatment, it does not imply that ROCE cannot play a substantial role during other, more physiological, conditions, such as agonist-induced 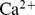 oscillations. It could be that ROCE is inhibited at the large
oscillations. It could be that ROCE is inhibited at the large 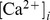 levels induced by SOCE activation following Rya-Caf treatment. Instead of assuming the existence of an inactivation process at large
levels induced by SOCE activation following Rya-Caf treatment. Instead of assuming the existence of an inactivation process at large 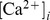 , we assumed, for simplicity, that ROCE is negligible in the model. This approach enabled us to show that
, we assumed, for simplicity, that ROCE is negligible in the model. This approach enabled us to show that 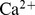 influx through SOCC is sufficient to sustain agonist-induced
influx through SOCC is sufficient to sustain agonist-induced 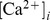 oscillations, and to explain the experimental results obtained with CPA, although the latter could be interpreted as evidence for ROCE at first sight. The fact that there appears to be no selective blocker for SOCE and ROCE makes it difficult to evaluate experimentally the respective contributions of the two
oscillations, and to explain the experimental results obtained with CPA, although the latter could be interpreted as evidence for ROCE at first sight. The fact that there appears to be no selective blocker for SOCE and ROCE makes it difficult to evaluate experimentally the respective contributions of the two 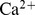 influxes during physiological conditions. These magnitudes are probably also cell-type dependent. Such issues explain the persistence of the controversy regarding SOCE and ROCE [45]–[48]. An informative experiment would be to stimulate ASMC using flash photolysis of caged
influxes during physiological conditions. These magnitudes are probably also cell-type dependent. Such issues explain the persistence of the controversy regarding SOCE and ROCE [45]–[48]. An informative experiment would be to stimulate ASMC using flash photolysis of caged 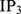 instead of agonist stimulation. Indeed, as
instead of agonist stimulation. Indeed, as 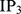 does not induce ROCE, SOCE should be the essential
does not induce ROCE, SOCE should be the essential 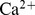 influx left. By comparing the responses to
influx left. By comparing the responses to 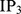 stimulation in the presence and in the absence of extracellular calcium, one could then deduce the importance of SOCE in physiological conditions.
stimulation in the presence and in the absence of extracellular calcium, one could then deduce the importance of SOCE in physiological conditions.
Efficacy of CPA
CPA is widely used as a SERCA blocker, having the advantage over Thapsigargin (Tg) of being reversible, and probably less toxic. Both have been used extensively to study SOCE in different cell types (e.g., [25]–[27], [43], [49]). Although our work indicates that CPA does not fully block the SERCA in intact tissue such as lung slices, it does not imply that CPA should not be used experimentally to induce SOCE. Indeed, CPA might still cause substantial SOCE activation in the presence of agonist. However, our results indicate that CPA is not a good mean to fully empty 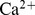 stores, and care should be taken in interpreting the experimental results of its application. We suggest that a combined Rya-Caf treatment is a more reliable way to induce a permanent large SR depletion (Fig. 3B, C). There is evidence that Tg is an efficient SERCA blocker in cell lines such as Hela cells [43], but we have not addressed the effect of Tg on ASMC in lung slices in this study.
stores, and care should be taken in interpreting the experimental results of its application. We suggest that a combined Rya-Caf treatment is a more reliable way to induce a permanent large SR depletion (Fig. 3B, C). There is evidence that Tg is an efficient SERCA blocker in cell lines such as Hela cells [43], but we have not addressed the effect of Tg on ASMC in lung slices in this study.
Modelling IPR
In this work, we followed the approach of Wang et al.
[23], in that we have used one of the simplest models of IPR 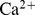 release, namely the Li-Rinzel/Tang et al. reduction of the DYK ODE model [34]–[36]. This category of IPR model produces agonist-induced
release, namely the Li-Rinzel/Tang et al. reduction of the DYK ODE model [34]–[36]. This category of IPR model produces agonist-induced 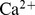 oscillations characterised by significant SR
oscillations characterised by significant SR 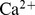 depletion (Fig. 3D and [23]), hence the possibility of SOCE being activated during such
depletion (Fig. 3D and [23]), hence the possibility of SOCE being activated during such 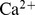 oscillations. This property might be model-dependent, however there is evidence that the SR is actually depleted to some extent during agonist-induced
oscillations. This property might be model-dependent, however there is evidence that the SR is actually depleted to some extent during agonist-induced 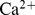 oscillations in ASMC. Indeed, the absence of effect of ryanodine during agonist-induced oscillations can be explained by the average level of
oscillations in ASMC. Indeed, the absence of effect of ryanodine during agonist-induced oscillations can be explained by the average level of 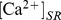 being too low for RyR activation [1], [23]. However, the respective
being too low for RyR activation [1], [23]. However, the respective 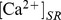 “thresholds” for SOCE and RyR activation are experimentally unknown. In this work, the SOCE activation threshold was deduced from fitting the model simultaneously to Fig. 3A and Figs. 3B–C.
“thresholds” for SOCE and RyR activation are experimentally unknown. In this work, the SOCE activation threshold was deduced from fitting the model simultaneously to Fig. 3A and Figs. 3B–C.
Finally, we note that our whole-cell 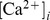 model would likely not benefit from using a recent Markov model of an IPR (e.g., [50]–[52]), because these models are based on steady-state data only (i.e., single-channel opening and closing times in stationary
model would likely not benefit from using a recent Markov model of an IPR (e.g., [50]–[52]), because these models are based on steady-state data only (i.e., single-channel opening and closing times in stationary 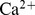 and
and 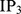 ) and typically miss the long inactivation timescale which was included “ad hoc” in the first IPR models to reproduce the observed behavior at the cell level (i.e.,
) and typically miss the long inactivation timescale which was included “ad hoc” in the first IPR models to reproduce the observed behavior at the cell level (i.e., 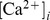 oscillations upon agonist stimulation).
oscillations upon agonist stimulation).
Limitations of the whole-cell model
As we are essentially interested in 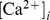 responses of ASMC at the cell level, we have described
responses of ASMC at the cell level, we have described 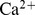 dynamics via a deterministic ODE model. The scope of this model is, however, somewhat limited for the following reasons.
dynamics via a deterministic ODE model. The scope of this model is, however, somewhat limited for the following reasons.
First, there is evidence that IPR are not homogeneously distributed on the SR membrane of cells, but are found as dense clusters. This channel clustering is especially patent upon stimulation by low agonist concentrations, for which local, stochastic 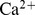 releases may not propagate to neighboring clusters, resulting in spatially isolated, unsynchronised
releases may not propagate to neighboring clusters, resulting in spatially isolated, unsynchronised 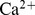 releases, called “puffs”. At higher agonist concentrations, the frequency of these puffs increases, allowing
releases, called “puffs”. At higher agonist concentrations, the frequency of these puffs increases, allowing 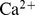 releases from close sites to accumulate and propagate further away. This triggers, via CICR, the firing of more distant clusters, and results in
releases from close sites to accumulate and propagate further away. This triggers, via CICR, the firing of more distant clusters, and results in 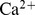 waves propagating repeatedly throughout the cytosol. These waves usually appear as
waves propagating repeatedly throughout the cytosol. These waves usually appear as 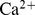 oscillations at the whole-cell level. While
oscillations at the whole-cell level. While 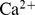 waves are indeed associated with
waves are indeed associated with 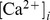 oscillations in ASMC [1], it has, so far, been impossible to detect
oscillations in ASMC [1], it has, so far, been impossible to detect 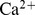 puffs. This could arise from a less clustered distribution of IPR in ASMC, compared to the larger cells (ooycytes and Hela cells) where puffs have been characterised. On the other hand,
puffs. This could arise from a less clustered distribution of IPR in ASMC, compared to the larger cells (ooycytes and Hela cells) where puffs have been characterised. On the other hand, 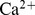 “sparks”, the equivalent of
“sparks”, the equivalent of 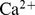 puffs but mediated by RyR, have been detected in ASMC [1], which supports a clustered distribution of RyR. In this study, we did not attempt to consider these spatial/stochastic aspects of the
puffs but mediated by RyR, have been detected in ASMC [1], which supports a clustered distribution of RyR. In this study, we did not attempt to consider these spatial/stochastic aspects of the 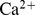 signals. Our model is thus less reliable at low agonist concentrations.
signals. Our model is thus less reliable at low agonist concentrations.
Second, cytoplasmic microdomains often exist between cell organelles (e.g., between peripheral SR and the plasma membrane, between the SR and mitochondria), out of which 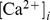 cannot diffuse easily. These have consequences for SOCE dynamics. Indeed, it has been reported that upon store depletion, SERCA can colocalise with STIM proteins, in proximity to the PM [49], [53]. As a consequence, if SOCE is slow enough, the SR can refill with
cannot diffuse easily. These have consequences for SOCE dynamics. Indeed, it has been reported that upon store depletion, SERCA can colocalise with STIM proteins, in proximity to the PM [49], [53]. As a consequence, if SOCE is slow enough, the SR can refill with 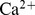 without a concomitant increase in bulk
without a concomitant increase in bulk 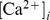 [49]. Upon large SOCE, this is no longer the case; however, mitochondria prevent the local
[49]. Upon large SOCE, this is no longer the case; however, mitochondria prevent the local 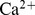 increase to become too large by pumping
increase to become too large by pumping 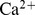 from the subplasmalemmal space and releasing it deeper in the cytoplasm, where it can be absorbed by other SERCA [49]. These spatial effects cannot be accounted for by our current non-compartmentalised model.
from the subplasmalemmal space and releasing it deeper in the cytoplasm, where it can be absorbed by other SERCA [49]. These spatial effects cannot be accounted for by our current non-compartmentalised model.
Finally, 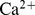 dynamics are modified by
dynamics are modified by 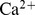 buffers in the cytosol and SR, which bind 99% of the free
buffers in the cytosol and SR, which bind 99% of the free 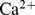 . While the effect of fast, linear buffers can be taken into account by a global rescaling of
. While the effect of fast, linear buffers can be taken into account by a global rescaling of 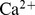 fluxes (see Methods), this is not the case for high affinity buffers, in particular fluorescent dye indicators. Including such buffers in an ODE model of
fluxes (see Methods), this is not the case for high affinity buffers, in particular fluorescent dye indicators. Including such buffers in an ODE model of 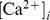 dynamics leads to suppression of
dynamics leads to suppression of 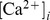 oscillations, because the buffer affinity is close to the amplitude of whole-cell
oscillations, because the buffer affinity is close to the amplitude of whole-cell 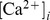 oscillations. In reality,
oscillations. In reality, 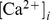 reaches much higher levels locally upon IPR opening, so that the buffers become saturated and cannot prevent
reaches much higher levels locally upon IPR opening, so that the buffers become saturated and cannot prevent 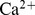 oscillations. Again, this would have to be accounted for by a spatial model of
oscillations. Again, this would have to be accounted for by a spatial model of 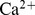 dynamics.
dynamics.
Future work
Although RyR dynamics play a role only during the initial phases of agonist-induced 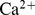 oscillations and Rya-Caf treatment, the interaction between RyR and IPR may become important in other situations, such as drug-induced RyR sensitisation. We plan to extend our model to these dynamics.
oscillations and Rya-Caf treatment, the interaction between RyR and IPR may become important in other situations, such as drug-induced RyR sensitisation. We plan to extend our model to these dynamics.
Since our work is part of a broader effort to improve the understanding of airway hyper-responsiveness and remodelling via mathematical modelling [54]–[57], we also intent to model the interaction of ASMC  signalling with other aspects of lung dynamics. Although mathematical models of ASM contraction have previously been developed [54], [55], [58], modelling of other signalling pathways, such as inflammation and proliferation, is, to our knowledge, still in its infancy.
signalling with other aspects of lung dynamics. Although mathematical models of ASM contraction have previously been developed [54], [55], [58], modelling of other signalling pathways, such as inflammation and proliferation, is, to our knowledge, still in its infancy.
Additionally, experimental studies of ASMC inflammation and proliferation in conjunction with  imaging in lung slices would be desirable. While such studies have been carried out with cultured ASMC [3]–[5], [9]–[12], they do not provide individual
imaging in lung slices would be desirable. While such studies have been carried out with cultured ASMC [3]–[5], [9]–[12], they do not provide individual  dynamics; moreover, cultured ASMC often exhibit a different phenotype from ASMC in intact tissues.
dynamics; moreover, cultured ASMC often exhibit a different phenotype from ASMC in intact tissues.
Conclusions
The inclusion of SOCE in our mathematical model of  dynamics in ASMC enables a better understanding of the experimental physiology of lung slices. It shows that the different abilities of CPA and Rya-Caf treatment to clamp the
dynamics in ASMC enables a better understanding of the experimental physiology of lung slices. It shows that the different abilities of CPA and Rya-Caf treatment to clamp the 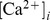 of ASMC can be explained by their different ability to invoke SOCE. The model predicts that CPA, in contrast with Rya-Caf treatment, is unable to empty the SR because of its inefficiency to fully inhibit the SERCA. Furthermore, by accounting for both agonist-induced
of ASMC can be explained by their different ability to invoke SOCE. The model predicts that CPA, in contrast with Rya-Caf treatment, is unable to empty the SR because of its inefficiency to fully inhibit the SERCA. Furthermore, by accounting for both agonist-induced 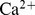 oscillations and SOCE activation by SR
oscillations and SOCE activation by SR 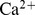 depletion, the model shows that SOCE can be a major determinant of the frequency of agonist-induced
depletion, the model shows that SOCE can be a major determinant of the frequency of agonist-induced 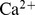 oscillations. Because this frequency of the
oscillations. Because this frequency of the 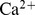 oscillations regulates airway contraction, the model suggests a role for increased SOCE in AHR, a correlation consistent with SOCE up-regulation under inflammatory conditions typical of asthma. These predictions underscore the synergistic role for mathematical modeling in medical research.
oscillations regulates airway contraction, the model suggests a role for increased SOCE in AHR, a correlation consistent with SOCE up-regulation under inflammatory conditions typical of asthma. These predictions underscore the synergistic role for mathematical modeling in medical research.
Supporting Information
Details of the parameter estimation procedure.
(PDF)
Acknowledgments
We thank Ruediger Thul, Charlotte Billington and Ian Hall for fruitful discussions.
Funding Statement
BSB acknowledges funding from the Medical Research Council (http://www.mrc.ac.uk), New Investigator Grant: G0901174. MJS is supported by a grant from National Institutes of Health (http://www.nih.gov): HL103405. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Perez JF, Sanderson MJ (2005) The frequency of calcium oscillations induced by 5-HT, ACH, and KCl determine the contraction of smooth muscle cells of intrapulmonary bronchioles. J Gen Physiol125: 535–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ressmeyer AR, Bai Y, Delmotte PF, Uy KF, Thistlethwaite P, et al. (2010) Human airway contraction and formoterol-induced relaxation is determined by Ca2+ oscillations and Ca2+ sensitivity. Am J Respir Cell Mol Biol 43: 179–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Sweeney M, McDaniel SS, Platoshyn O, Zhang S, Yu Y, et al. (2002) Role of capacitative Ca2+ entry in bronchial contraction and remodeling. J Appl Physiol 92: 1594–602. [DOI] [PubMed] [Google Scholar]
- 4. Mahn K, Hirst SJ, Ying S, Holt MR, Lavender P, et al. (2009) Diminished sarco/endoplasmic reticulum Ca2+ ATPase (SERCA) expression contributes to airway remodelling in bronchial asthma. PNAS 106: 10775–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zou JJ, Gao YD, Geng S, Yang J (2011) Role of STIM1/Orai1-mediated store-operated Ca2+ entry in airway smooth muscle cell proliferation. J Appl Physiol 110: 1256–63. [DOI] [PubMed] [Google Scholar]
- 6. Gerthoffer WT (1991) Regulation of the contractile element of airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 5: L15–L28. [DOI] [PubMed] [Google Scholar]
- 7. Janmey PA (1994) Phosphoinositides and calcium as regulators of cellular actin assembly and disassembly. Annu Rev Physiol 56: 169–91. [DOI] [PubMed] [Google Scholar]
- 8. Herrera AM, Kuo KH, Seow CY (2002) Influence of calcium on myosin thick filament formation in intact airway smooth muscle. Am J Physiol Cell Physiol 282: C310–6. [DOI] [PubMed] [Google Scholar]
- 9. White TA, Xue A, Chini EN, Thompson M, Sieck GC, et al. (2006) Role of transient receptor potential C3 in TNF-alpha-enhanced calcium influx in human airway myocytes. Am J Respir Cell Mol Biol 35: 243–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Moynihan B, Tolloczko B, Michoud MC, Tamaoka M, Ferraro P, et al. (2008) MAP kinases mediate interleukin-13 effects on calcium signaling in human airway smooth muscle cells. Am J Physiol Lung Cell Mol Physiol 295: L171–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sieck GC, White TA, Thompson MA, Pabelick CM, Wylam ME, et al. (2008) Regulation of store-operated Ca2+ entry by CD38 in human airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 294: L378–85. [DOI] [PubMed] [Google Scholar]
- 12. Sathish V, Thompson MA, Bailey JP, Pabelick CM, Prakash YS, et al. (2009) Effect of proinflammatory cytokines on regulation of sarcoplasmic reticulum Ca2+ reuptake in human airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 297: L26–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Putney JW (1986) A model for receptor-regulated calcium entry. Cell Calcium 7: 1–12. [DOI] [PubMed] [Google Scholar]
- 14. Roos J, DiGregorio PJ, Yeromin AV, Ohlsen K, Lioudyno M, et al. (2005) STIM1, an essential and conserved component of store-operated Ca2+ channel function. J Cell Biol 169: 435–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Wiesner TF, Berk BC, Nerem RM (1996) A mathematical model of cytosolic calcium dynamics in human umbilical vein endothelial cells. Am J Physiol Cell Physiol 270: C1556–C1569. [DOI] [PubMed] [Google Scholar]
- 16. Kowalewski JM, Uhlén P, Kitano H, Brismar H (2006) Modeling the impact of store-operated Ca2+ entry on intracellular Ca2+ oscillations. Math Biosci 204: 232–49. [DOI] [PubMed] [Google Scholar]
- 17. Ong HL, Liu X, Tsaneva-Atanasova K, Singh BB, Bandyopadhyay BC, et al. (2007) Relocalization of STIM1 for activation of store-operated Ca2+ entry is determined by the depletion of subplasma membrane endoplasmic reticulum Ca2+ store. J Biol Chem 282: 12176–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Liu W, Tang F, Chen J (2010) Designing Dynamical Output Feedback Controllers for Store-operated Ca2+ Entry. Math Biosci 228: 110–118. [DOI] [PubMed] [Google Scholar]
- 19. Haberichter T, Roux E, Marhl M, Mazat JP (2002) The influence of different InsP3 receptor isoforms on Ca2+ signaling in tracheal smooth muscle cells. Bioelectrochemistry 57: 129–38. [DOI] [PubMed] [Google Scholar]
- 20. Roux E, Marhl M (2004) Role of sarcoplasmic reticulum and mitochondria in Ca2+ removal in airway myocytes. Biophys J 86: 2583–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Brumen M, Fajmut A, Dobovišek A, Roux E (2005) Mathematical Modelling of Ca2+ Oscillations in Airway Smooth Muscle Cells. J Biol Phys 31: 515–524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Roux E, Noble PJ, Noble D, Marhl M (2006) Modelling of calcium handling in airway myocytes. Prog Biophys Mol Biol 90: 64–87. [DOI] [PubMed] [Google Scholar]
- 23. Wang IY, Bai Y, Sanderson MJ, Sneyd J (2010) A mathematical analysis of agonist- and KCl- induced Ca2+ oscillations in mouse airway smooth muscle cells. Biophys J 98: 1170–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Gao YD, Zou JJ, Zheng JW, Shang M, Chen X, et al. (2010) Promoting effects of IL-13 on Ca2+ release and store-operated Ca2+ entry in airway smooth muscle cells. Pulm Pharmacol Ther 23: 182–9. [DOI] [PubMed] [Google Scholar]
- 25. Ay B, Prakash YS, Pabelick CM, Sieck GC (2004) Store-operated Ca2+ entry in porcine airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 286: L909–17. [DOI] [PubMed] [Google Scholar]
- 26. Peel SE, Liu B, Hall IP (2006) A key role for STIM1 in store-operated calcium channel activation in airway smooth muscle. Respir Res 7: 119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Peel SE, Liu B, Hall IP (2008) ORAI and store-operated calcium inux in human airway smooth muscle cells. Am J Respir Cell Mol Biol 38: 744–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Bai Y, Sanderson MJ (2006) Modulation of the Ca2+ sensitivity of airway smooth muscle cells in murine lung slices. Am J Physiol Lung Cell Mol Physiol 291: L208–21. [DOI] [PubMed] [Google Scholar]
- 29. Bai Y, Sanderson MJ (2009) The contribution of Ca2+ signaling and Ca2+ sensitivity to the regulation of airway smooth muscle contraction is different in rats and mice. Am J Physiol Lung Cell Mol Physiol 296: L947–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Keener J, Sneyd J (2008) Mathematical physiology, second edition. Springer.
- 31. Janssen LJ (2002) Ionic mechanisms and Ca2+ regulation in airway smooth muscle contraction: do the data contradict dogma? Am J Physiol Lung Cell Mol Physiol 282: L1161–78. [DOI] [PubMed] [Google Scholar]
- 32. Liou J, Fivaz M, Inoue T, Meyer T (2007) Live-cell imaging reveals sequential oligomerization and local plasma membrane targeting of stromal interaction molecule 1 after Ca2+ store depletion. PNAS 104: 9301–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Luik R, Wang B, Prakriya M, Wu M, Lewis R (2008) Oligomerization of STIM1 couples ER calcium depletion to CRAC channel activation. Nature 454: 538–542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. De Young GW, Keizer J (1992) A single-pool inositol 1, 4, 5-trisphosphate-receptor-based model for agonist-stimulated oscillations in Ca2+ concentration. PNAS 89: 9895–9899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Li Y, Rinzel J (1994) Equations for InsP3 receptor-mediated [Ca2+]i oscillations derived from a detailed kinetic model: a Hodgkin-Huxley like formalism. J Theor Biol 166: 461–473. [DOI] [PubMed] [Google Scholar]
- 36. Tang Y, Stephenson JL, Othmer HG (1996) Simplification and analysis of models of calcium dynamics based on IP3-sensitive calcium channel kinetics. Biophys J 70: 246–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Doedel EJ, Keller HB, Kernevez JP (1991) Numerical analysis and control of bifurcation problems: (I) Bifurcation in finite dimensions. Int J Bifurc Chaos 1: 493–520. [Google Scholar]
- 38. Doedel EJ, Keller HB, Kernevez JP (1991) Numerical analysis and control of bifurcation problems: (II) Bifurcation in infinite dimensions. Int J Bifurc Chaos 1: 745–772. [Google Scholar]
- 39. Bai Y, Edelmann M, Sanderson MJ (2009) The contribution of inositol 1,4,5-trisphosphate and ryanodine receptors to agonist-induced Ca2+ signaling of airway smooth muscle cells. Am J Physiol Lung Cell Mol Physiol 297: L347–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Berridge MJ (2009) Cell Signalling Pathways. In: Cell Signalling Biology, Portland Press, volume 3: 2.1–2.118 Available: www.cellsignallingbiology.org. [Google Scholar]
- 41. Demaurex N, Frieden M (2003) Measurements of the free luminal ER Ca2+ concentration with targeted cameleon fluorescent proteins. Cell Calcium 34: 109–119. [DOI] [PubMed] [Google Scholar]
- 42. Sneyd J, Tsaneva-Atanasova K, Yule DI, Thompson JL, Shuttleworth TJ (2004) Control of calcium oscillations by membrane fluxes. PNAS 101: 1392–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Shen WW, Frieden M, Demaurex N (2011) Local cytosolic Ca2+ elevations are required for stromal interaction molecule 1 (STIM1) de-oligomerization and termination of store-operated Ca2+ entry. J Biol Chem 286: 36448–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Malli R, Naghdi S, Romanin C, Graier WF (2008) Cytosolic Ca2+ prevents the subplasmalemmal clustering of STIM1: an intrinsic mechanism to avoid Ca2+ overload. J Cell Sci 121: 3133–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Shuttleworth TJ (1999) What drives calcium entry during [Ca2+]i oscillations? – challenging the capacitative model. Cell Calcium 25: 237–46. [DOI] [PubMed] [Google Scholar]
- 46. Bird GSJ, Putney JW (2005) Capacitative calcium entry supports calcium oscillations in human embryonic kidney cells. J Physiol 562: 697–706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Trebak M (2011) PLC: Johnny-come-lately to ORAI and the ups and downs of calcium signalling. J Physiol 589: 5337–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Shuttleworth T (2012) STIM and Orai proteins and the non-capacitative ARC channels. Front Biosci 17: 847–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Jousset H, Frieden M, Demaurex N (2007) STIM1 knockdown reveals that store-operated Ca2+ channels located close to sarco/endoplasmic Ca2+ ATPases (SERCA) pumps silently refill the endoplasmic reticulum. J Biol Chem 282: 11456–64. [DOI] [PubMed] [Google Scholar]
- 50. Mak DOD, McBride SMJ, Foskett JK (2003) Spontaneous channel activity of the inositol 1,4,5- trisphosphate (InsP3) receptor (InsP3R). Application of allosteric modeling to calcium and InsP3 regulation of InsP3R single-channel gating. J Gen Physiol 122: 583–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Gin E, Wagner LE, Yule DI, Sneyd J (2009) Inositol trisphosphate receptor and ion channel models based on single-channel data. Chaos 19: 037104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Siekmann I, Wagner LE, Yule D, Crampin EJ, Sneyd J (2012) A Kinetic Model for type I and II IP3R accounting for mode changes. Biophys J 103: 658–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Manjarrés IM, Rodríguez-García A, Alonso MT, García-Sancho J (2010) The sarco/endoplasmic reticulum Ca2+ ATPase (SERCA) is the third element in capacitative calcium entry. Cell Calcium 47: 412–8. [DOI] [PubMed] [Google Scholar]
- 54. Wang I, Politi AZ, Tania N, Bai Y, Sanderson MJ, et al. (2008) A mathematical model of airway and pulmonary arteriole smooth muscle. Biophys J 94: 2053–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Brook BS, Peel SE, Hall IP, Politi AZ, Sneyd J, et al. (2010) A biomechanical model of agonist-initiated contraction in the asthmatic airway. Respir Physiol Neurobiol 170: 44–58. [DOI] [PubMed] [Google Scholar]
- 56. Politi AZ, Donovan GM, Tawhai MH, Sanderson MJ, Lauzon AM, et al. (2010) A multiscale, spatially distributed model of asthmatic airway hyper-responsiveness. J Theor Biol 266: 614–624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Lauzon AM, Bates JHT, Donovan G, Tawhai M, Sneyd J, et al. (2012) A multi-scale approach to airway hyperresponsiveness: from molecule to organ. Front Physiol 3: 191 (1–25).. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Brook BS, Jensen OE (2013) The role of contractile unit reorganization in force generation in airway smooth muscle. Math Med Biol. In press. [DOI] [PMC free article] [PubMed]
- 59. O'Donnell ME, Owen NE (1994) Regulation of ion pumps and carriers in vascular smooth muscle. Physiol Rev 74: 683–721. [DOI] [PubMed] [Google Scholar]
- 60.Alberts B, Johnson A, Lewis J, Raff M, Roberts K, et al. (2008) Molecular Biology of the cell. New York: Garland, fifth edition. Available: http://www.worldcat.org/isbn/0815332181.
- 61. Lytton J, Westlin M, Burk SE, Shull GE, MacLennan DH (1992) Functional comparisons between isoforms of the sarcoplasmic or endoplasmic reticulum family of calcium pumps. J Biol Chem 267: 14483–9. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Details of the parameter estimation procedure.
(PDF)