Abstract
The most evident challenge to treatment of Helicobacter pylori, a bacterium responsible for gastritis, peptic ulcers and gastric cancer, is the increasing rate of resistance to all currently used therapeutic antibiotics. Thus, the development of novel therapies is urgently required. N-geranyl-N'-(2-adamantyl) ethane-1, 2-diamine (SQ109) is an ethylene diamine-based antitubercular drug that is currently in clinical trials for the treatment of tuberculosis (TB). Previous pharmacokinetic studies of SQ109 revealed that persistently high concentrations of SQ109 remain in the stomach 4 hours post oral administration in rats. This finding, combined with the need for new anti- Helicobacter therapies, prompted us to define the in vitro efficacy of SQ109 against H. pylori. Liquid broth micro-dilution was used for susceptibility studies to determine the antimicrobial activity of SQ109 against a total of 6 laboratory strains and 20 clinical isolates of H. pylori; the clinical isolates included a multi-drug resistant strain. All strains tested were susceptible to SQ109 with MIC and MBC ranges of 6-10 µM and 50-60 µM, respectively. SQ109 killing kinetics were concentration- and time-dependent. SQ109 killed H. pylori in 8-10 h at 140 µM (2MBCs) or 4-6 h at 200 µM (~3MBCs). Importantly, though the kinetics of killing were altered, SQ109 retained potent bactericidal activity against H. pylori at low pH. Additionally, SQ109 demonstrated robust thermal stability and was effective at killing slow growing or static bacteria. In fact, pretreatment of cultures with a bacteriostatic concentration of chloramphenicol (Cm) synergized the effects of typically bacteriostatic concentrations of SQ109 to the level of five-logs of bacterial killing. A molar-to-molar comparison of the efficacy of SQ109 as compared to metronidazole (MTZ), amoxicillin (AMX), rifampicin (RIF) and clarithromycin (CLR), revealed that SQ109 was superior to MTZ, AMX and RIF but not to CLR. Finally, the frequency of resistance to SQ109 was low and electron microscopy studies revealed that SQ109 interacted with bacterial inner membrane and cytoplasmic content(s). Collectively, our in vitro data demonstrate that SQ109 is an effective monotherapy against susceptible and multi-drug resistant strains of H. pylori and may be useful alone or in combination with other antibiotics for development as a new class of anti- Helicobacter drugs.
Introduction
H. pylori is a spiral-shaped Gram-negative bacterium that is well adapted to infect the gastric mucosa; with approximately 50% of the world’s population colonized, H. pylori represents one of the most prevalent bacterial infections. Rates of colonization vary by geographic location and economic status and can be as high as 90% [1]. It is believed that H. pylori is typically acquired early in life by most individuals [2]. Post colonization, the bacterium can persist for months, years or decades without inducing obvious clinical symptoms [3]. However, in approximately 20% of infected individuals, H. pylori can induce clinical sequelae that can range from peptic ulcers to gastric cancer [4,5].
The ability to eradicate H. pylori infection plays a critical role in the treatment and prevention of associated gastroduodenal diseases [6,7]. Current treatment strategies typically involve 1-2 week twice-daily administration of a proton pump inhibitor (PPI) and two antibiotics, which is commonly referred to as triple therapy [8]. Though many different antibiotics have been tested for efficacy against H. pylori, combinations of AMX, MTZ or CLR are the antibiotics most often utilized in triple therapy. Beyond these, tetracycline (TET) and levofloxacin are gaining significant use in quadruple and “rescue” therapy regimens, respectively. Levofloxacin is especially useful following treatment failure with CLR-based treatments [9,10]. As implied by the need for quadruple and rescue therapies, H. pylori has developed resistance to nearly all conventional antibiotics currently used for treatment of the infection [11,12]. Indeed, the chronic nature of H. pylori colonization and the difficulty in eradication are responsible for the evolution of H. pylori treatment strategies from mono to dual to triple, and now quadruple, sequential and rescue therapies [13,14]. Adding to the complication of antibiotic resistance, prolonged periods of treatment, combined with higher doses of antibiotics and the use of multiple drugs has increased contraindications and patient non-compliance. This unfortunate cycle likely results in further selection for antibiotic resistance in H. pylori and allows the spread of resistant strains. Given all of these complexities, there is clearly an urgent need to develop new drugs that are effective against resistant strains and that have the ability to traverse into the gastric epithelial cells to eradicate any intracellular H. pylori cells that cannot be reached by other antibiotics.
Despite significant increases in emergence and spread of multi-drug resistant strains of H. pylori over the past 20-30 years, the development of new antibiotics has decreased alarmingly in the same period of time [15–17]. Furthermore, many researchers have switched from traditional drug search to genomic applications, which have unforeseeably taken longer to produce candidate antibiotics than originally expected [18,19]. Indeed, in the past half century only three new classes of antibiotics have entered the clinics: lipopeptides [20], oxazolidinones [21] and streptogramins [22,23]. All three of these antibiotics specifically target Gram-positive bacterial infections, which further limits the availability of effective antibiotics against Gram-negative bacteria such as H. pylori.
A potential antibiotic in clinical development is N-geranyl-N'-(2-adamantyl) ethane-1, 2-diamine (SQ109), which represents a member of a new class of small molecule ethylenediamine compounds that has anti-tubercular activity [24,25]. SQ109 has strong in vitro bactericidal activity against Mycobacterium tuberculosis and traverses host cell membranes to effectively kill M. tuberculosis in macrophage phagolysosomes [24]. Moreover, SQ109 has synergy with two front-line TB drugs (isoniazid and RIF) in both in vitro studies and in pre-clinical animal trials [26,27] and is safe and well-tolerated in humans [28,29]. SQ109 is currently in Phase 2 clinical trials for treatment of adult pulmonary TB.
Pharmacokinetic studies of SQ109 showed promising bioavailability with accumulation of high concentrations of the drug in the stomach [25,30], the natural niche of H. pylori. This finding, along with the fact that SQ109 has proven safe in humans led us to ask whether SQ109 showed antibacterial activity against H. pylori. Herein, we describe the in vitro characterization of SQ109 anti-H. pylori activity. Our data suggest that SQ109 has an interesting potential as a new therapeutic and may be suitable for development as a new antibiotic for the treatment of H. pylori infections.
Materials and Methods
SQ109, reagents and bacterial Strains
SQ109, which was originally developed and identified by combinatorial chemistry and high-throughput screening of over 63,000 library analogs, was synthesized on solid support using a novel acylation-reduction sequence as previously described [24,25], and was provided as a dry powder by Sequella, Inc. (Rockville, MD). SQ109 was dissolved in water to form a 10mg/ml stock solution, which was then stored as 250µl single use aliquots at -20oC. AMX, CLR and MTZ were purchased from Sigma (St. Louis, MO), and vancomycin was obtained from USB Corporation (Cleveland, OH). Each drug was reconstituted according to the manufacturer’s instructions and used at the indicated concentrations.
A total of 26 H. pylori strains and isolates (Table 1 and Table 2) used in the study were obtained as follows: 6 strains (G27, 7.13, HPAG, SS1, J99 and 26695) are common laboratory strains contained in our strain collection, while 20 isolates were low passage clinical isolates collected from patients at several locations. Of these 20 clinical isolates, 6 strains (K3, K93, K154, K260, K264, K266) were obtained from patients presenting for treatment at the Department of Internal Medicine at the College of Medicine of The Catholic University of Korea in Seoul, South Korea, 12 strains (B99, B105, B107, B108, B128, B129, B138, B148, B235, B240, B289, J104) were obtained from patients at Vanderbilt University Medical Center, Nashville TN, and 2 strains (USU102, USU103) were obtained from patients at the Veterans’ Bureau, VA Medical Center, Bethesda, MD. Of these strains, USU103 was obtained from a patient that failed four rounds of antibiotic treatment, including one round of quadruple therapy; antibiotic sensitivity testing showed the strain to be highly resistant to AMX and CLR (Figure S1). Thus, the strain represents a multi-drug resistant isolate.
Table 1. Antimicrobial Activity of SQ109 against H. pylori Laboratory Strains.
| Laboratory Strains | Reference |
SQ109 Concentration (µM)
|
|
|---|---|---|---|
| MICa | MBCb | ||
| G27 | [69] | 8-10 | 65-70 |
| 7.13 | [70] | 8-10 | 75-80 |
| HPAG | [71] | 8-10 | 70-75 |
| SS1 | [72] | 10-15 | 65-70 |
| J99 | [73] | 10-15 | 65-70 |
| 26695 | [74] | 15-20 | 80-100 |
a Minimum inhibitory concentration (100.1% survival); bMinimum bactericidal concentration (99.9% killing). The data represent results from at least three independent experiments.
Determination of SQ109 MIC and MBC against H. pylori
All strains were grown at 37oC under micro-aerobic conditions of 5% O2, 10% CO2, and 85% N2 gas mixture, which was achieved using an Anoxomat instrument (MART 1598, Spiral Biotech, Norwood, MA). The 26 clinical and laboratory strains were used to determine the MIC and MBC of SQ109 by liquid broth micro-dilution as previously described [31]. Briefly, stock cultures stored at -80°C, were used to inoculate horse blood agar (HBA) plates. The strains were cultured for 24 h on HBA plates and then expanded and recultured for an additional 24 h before being used to inoculate Brucella broth (Neogen-Acumedia) liquid starter cultures supplemented with 10 µg/mL vancomycin (Sigma) and 10% FBS (Invitrogen Gibco-BRL, Carlsbad, CA) (complete Brucella broth medium, CBBM). The starter cultures were incubated for 16-18 h and then used to inoculate experimental cultures at an optical density at 600nm wavelength (OD600) of 0.05. These samples were used to perform the MIC and MBC experiments in 1 ml culture aliquots in sterile 15 mm diameter glass tubes. To identify the initial concentration ranges for MIC and MBC determination, we incubated the bacterial cells with two-fold dilutions of SQ109 that ranged from 10–200 µM. The samples were incubated with shaking at 110 rpm for 24 h. Bacterial cells incubated with PBS were included as a control to SQ109-treatment. The MIC and MBC were determined by enumeration of surviving colony forming units (CFU) on HBA plates and were expressed as percent CFU survival using the formula: % CFU Survival= (CFUt24/CFUt0)100; where CFUt24 is the number of CFU at the end of 24 h of culture and /CFUt0 is the starting CFU at the beginning of the assay. After the initial assays, susceptibility assays were next repeated 3 times with smaller concentration intervals; steps of 2-5 µM and 5-10 µM were used for the determination of specific SQ109 MIC and MBC, respectively. The lowest SQ109 concentration at which there was no evidence of growth (100.1% CFU survival) was recorded as the MIC; MBC was defined as the lowest concentration of the drug that killed 99.9% of the starting CFU (0.1% CFU survival).
Since USU103 was reportedly a multi-drug resistant isolate (A. Dubois personal communication), we sought to determine its resistance to selected conventional antibiotics often used against H. pylori (AMX, MTZ, CLR, and TET). Bactericidal assays were conducted in 1 ml liquid cultures as described above and strain 26695 was included for comparison. The percent survival of H. pylori clinical isolate, USU103 was computed against a range of concentrations of AMX (0.5-1000 µg/ml), MTZ (0.5-1000 µg/ml), CLR (0.5-100 µg/ml), and TET (0.5-100 µg/ml) by plating and CFU enumeration following culture in the presence of the indicated drug for 24 h (Figure S1).
Determination of killing kinetics of SQ109
The time required for SQ109 to kill H. pylori cells was determined using time-kill curves by exposing the various strains to a range of SQ109 concentrations (10-200 µM) for up to 24 h, followed by monitoring of CFUs. Briefly, 1 ml liquid bacterial cultures were diluted to an OD600 of 0.05 in CBBM and exposed to SQ109. After 0, 1, 2, 4, 6, 8, 10, 20, 22 and 24 h of culture in the presence of SQ109, 10-fold serial dilutions of the samples were plated on HBA plates. The plates were incubated at 37°C for 5-6 days at which point CFU were enumerated and expressed as CFU/ml. H. pylori cells exposed to similar volumes of PBS (0 µM SQ109) were included as a control.
Determination of the effects of SQ109 on slow-growing H. pylori
A static culture of H. pylori was obtained by using a pre-determined bacteriostatic concentration of Cm; treatment with 30 µM Cm inhibits H. pylori growth but does not kill the bacterium. In order to determine if SQ109 had the ability to kill slowly growing H. pylori, time kill curves were evaluated over a 24-hour period in CBBM liquid cultures as described above but with the following modifications. Briefly, a 5 ml H. pylori starter liquid culture was incubated with 30 µM Cm for 16 h at which point, the OD600 was adjusted to 0.05-0.07 and the sample was divided into four 1 ml samples. The bacterial samples were incubated with shaking with 0 µM, 10 µM, 70 µM or 140 µM SQ109 for 24 h. At 0, 2, 4, 6, 8, 10, 14, 20, 22, and 24 h, an aliquot of each sample was plated and CFU were enumerated. A sample of H. pylori incubated with PBS or Cm-free ethanol for 16 h and then adjusted to an OD600 that matched that of the bacteria cultured in the presence of Cm was included as a control. The time-kill curves of bacterial samples cultured in the presence of combinations of 30 µM Cm with or without SQ109 were compared to those of bacterial samples exposed to PBS or SQ109 alone.
Determination of SQ109 stability under different thermal and pH conditions
SQ109 stability at low pH and following incubation at different temperatures was investigated using time-kill assays as outlined above. To characterize the impact of temperature on stability, 140 µM of SQ109 was incubated at 22oC, 37oC, 60oC or 95oC for 1 h prior to performing the time-kill assay. The samples were then cooled on ice and the killing time was determined as described above.
To examine the stability of SQ109 at low pH, the pH of CBBM was adjusted to pH 4.5 using HCl. The media was subsequently filter-sterilized to remove precipitates and was used for time-kill curve experiments as described above, with the following exceptions: H. pylori cells were suspended in 25 ml of pH 4.5 or pH 6.8 CBBM medium at an OD600 0.05 and subsequently exposed to a final concentration of 140 µM SQ109 or PBS (0 µM). The liquid bacterial cultures were incubated with shaking for up to 24 h. The killing time for both experiments (pH and temperature), were evaluated by enumeration of CFU on HBA plates at 0, 2, 4, 6, 8, 10, 14, 20, 22, and 24 h. Additionally, for the pH stability assays, the pH was monitored across the 24 h culture period to ensure stability. Of note, to control for any effects of nutrients lost by precipitation during the adjustment of pH of the media, the experiment was repeated using pH 6.8 CBBM media whose pH was initially HCl-adjusted to pH 4.5, filtered and then readjusted back to pH 6.8 using NaOH solution. Similar results were obtained.
Comparative efficacy of SQ109, AMX, MTZ and CLR against H. pylori
The antibacterial activity of SQ109 against H. pylori was compared to that of AMX, MTZ and CLR using time-kill assays as described above. For these assays, the H. pylori G27 strain was exposed to 70 µM or 140 µM SQ109, AMX, MTZ and CLR in 1 ml CBBM. Samples were withdrawn at 0, 0.5, 2, 4, 6, 8, 10, 14, 20, 22, and 24 h and the number of viable CFU determined as described above.
Determination of frequency of resistance to SQ109
Frozen H. pylori strains G27, 26695 and B105 were revived on HBA plates as described above (MIC and MBC experiment). The cells were inoculated into 25 ml CBBM liquid cultures, which were then grown for 16-18 h; at this point the cultures showed an OD600 of 0.5-1.0. One milliliter aliquots of each culture were concentrated by centrifugation, resuspended in 200 µl of CBBM and then plated on selective HBA plates that contained 100 µM, 200 µM, or 400 µM SQ109. The total number of bacteria present in the cultures was obtained by serially diluting the starting culture and plating on SQ109-free HBA plates. CFUs appearing on the plates following incubation for 5-6 days at 37oC were enumerated. This entire process was repeated a total of 4 times and the frequency of SQ109-resistant mutants was determined by dividing the total number of CFU obtained on SQ109-selective medium by the total number of CFU obtained on the SQ109-free medium (wild-type) across all of the plating experiments.
To monitor the stability of resistance, and to determine whether resistant colonies might in fact represent persistent strains and not resistant strains, 3-5 individual SQ109-resistant mutant colonies were randomly picked, streaked and expanded on SQ109-free HBA plates to create stock cultures. These variants were then expanded from the freezer and serially passaged as lawns on SQ109-free plates a total of 5 times; each plate was incubated for 24 hours, the resulting lawn was swabbed and the cells used to lawn a new plate. The MIC and MBC of these isolates to SQ109 was then determined and compared to the parental/wild-type strain.
SQ109 membrane permeation assay using 1-N-phenylnaphthylamine (NPN)-uptake
SQ109 was shown to exert its effect on M. tuberculosis by inhibiting the assembly of mycolic acids into the cell wall core [32]. Therefore, to begin to understand mechanism(s) of action of SQ109 against H. pylori, we performed membrane permeation assays as previously described [33] using NPN-uptake as a means to evaluate whether the drug targets and disrupts the bacterial membrane.
Transmission electron microscopy (TEM) studies
TEM analysis was performed as previously described [33]. Briefly, approximately 1.5 x 107 G27 bacterial cells were treated with 140 µM SQ109. Samples were taken at 2 or 8 h, washed twice in PBS and then used for TEM. Bacterial cells treated with 100 µM AMX, 20 µM C12K-2β12 (a peptide previously shown to form pores in H. pylori membranes [33]) or PBS were included as controls. Treated cells were resuspended and fixed in PBS containing 2% formaldehyde/2% glutaraldehyde/0.5% tannic acid (v/v/w) for 1 h at room temperature. Following fixation, the bacterial cells were thoroughly rinsed, enrobed in agarose, and trimmed into 1 mm3 blocks. The enrobed blocks were post-fixed with 1% osmium tetroxide in phosphate buffer, dehydrated by passage through a graduated ethanol series and embedded in Spurr’s resin (Electron Microscopy Sciences, PA) following the manufacturer’s recommendation. The blocks were trimmed and sectioned using a Reichert Jung ultra-microtome UC6. Sections measuring 70-80 nm were collected onto copper grids. Finally, the grids were successively stained with 1% uranyl acetate and Sato’s triple lead stains and subsequently examined in a FEI Tecnai T12 transmission electron microscope at an accelerating voltage of 80 kV. TEM images were examined at low (6,500x) and high (42,000x) magnifications for bacterial morphology and ultra-structure analysis, respectively. The images were acquired with an AMT XR60B digital camera (Advanced Microscopy Techniques) using AMTV600 software.
Results
SQ109 efficacy: Determination of MICs, MBCs and killing kinetics
SQ109 bactericidal activity against M. tuberculosis and the accumulation of the drug in high concentrations in the stomach after oral administration in mice and rats [25] prompted us to determine the activity of this anti-tubercular drug candidate against H. pylori. To determine SQ109 efficacy against the bacterium, we first evaluated the MIC and MBC against a panel of six commonly used laboratory strains. All of the laboratory strains tested were susceptible to SQ109, with MIC ranges of 8-20 µM and MBC ranges of 65-100 µM (Table 1). Among the laboratory strains, H. pylori strain G27 was one of the most susceptible to SQ109 antibacterial activity (MIC=8-10 µM; MBC=65-70 µM), thus G27 was used in subsequent experiments to elucidate SQ109 antimicrobial activity. In contrast, strain 26695 was the least sensitive (MIC=15-20 µM; MBC=80-100 µM). Given that these laboratory strains have been cultured for long periods of time and that H. pylori displays a high rate of genetic variability in culture [34,35], we next analyzed a panel of 20 low passage clinical isolates for susceptibility to SQ109. These included isolates obtained from a diverse number of possible H. pylori-associated diseases (Table 2). Once again, all of the strains were susceptible to SQ109, with MIC ranges of 5-30 µM and MBC ranges of 50-80 µM (Table 2). Of note, USU103, which was obtained from a gastric ulcer patient who had failed rounds of triple and quadruple therapies, was predicted to be a multi-drug resistant strain. Indeed, MIC and MBC testing of AMX, MTZ, CLR and TET against the isolate demonstrated that USU103 was resistant to AMX, CLR and MTZ (Figure S1), whereas 26695 was sensitive to all of the tested antibiotics. En masse, 22 of the 26 examined strains (84.6%) had MICs from 5–15 µM while 4 of 26 (15.4%) had slightly higher MIC ranges of 15-30 µM. Similarly, 20 of the 26 examined strains (76.9%) had MBC ranges of 50-70 µM and 6 of 26 (23.1%) had slightly higher MBCs of 70-100 µM. Taken together, our data suggest that SQ109 has potent antibacterial activity against all tested strains of H. pylori and is effective against at least one multi-drug resistant strain.
Table 2. SQ109 efficacy against H. pylori clinical isolates.
|
H. pylori Clinical History
|
Clinical Isolates Identification |
SQ109 Concentration (µM)
|
||
|---|---|---|---|---|
| Endoscopic and Histologic Diagnosis | Histologic Features | MICa | MBCb | |
| Gastritis | Glandular atrophy score 1 or 2 | K154 | 6-8 | 50-60 |
| K266 | 8-10 | 50-60 | ||
| B99 | 5-10 | 50-60 | ||
| B108 | 8-10 | 60-65 | ||
| B235 | 8-10 | 60-70 | ||
| B289 | 15-20 | 50-60 | ||
| Duodenitis | Glandular atrophy score 1 or 3 | B107 | 15-20 | 60-65 |
| B240 | 8-10 | 60-65 | ||
| B148 | 6-8 | 60-65 | ||
| Gastric Ulcers | Glandular atrophy score 1 or 3 Intestinal metaplasia | B105 | 25-30 | 70-80 |
| B128 | 6-8 | 70-80 | ||
| Multi-drug resistant strain, MDRSc | USU103 | 10-15 | 50-60 | |
| Duodenal Ulcers | Glandular atrophy score 1 or 3 Intestinal metaplasia | K93 | 6-8 | 50-60 |
| K264 | 5-10 | 50-60 | ||
| B129 | 6-8 | 65-70 | ||
| J104 | 10-15 | 60-70 | ||
| Gastric Cancer | Adenocarcinoma | K3 | 5-10 | 50-60 |
| K260 | 5-10 | 65-70 | ||
| B138 | 6-10 | 70-75 | ||
| USU102 | 20-25 | 50-60 | ||
a Minimum inhibitory concentration (100.1% survival); bMinimum bactericidal concentration (99.9% killing); cMDRS = multi-drug resistant strain (quadruple and sequential therapies administered and see Figure S1). The data represent results from at least three independent experiments.
Time kill-curve analysis demonstrates SQ109 time- and dose-dependent killing kinetics
The rate at which an antibiotic exerts its antibacterial activity is particularly important when considering possible treatments for H. pylori infection. This is due to the relatively rapid transit of gastric contents, which has the potential to reduce the exposure time of the bacteria to a particular concentration of an orally-ingested antibiotic. Therefore, to determine the rate of SQ109-dependent killing, we performed time-kill assays against strain G27. As shown in Figure 1, no colonies were recovered after 6 h of treatment with 200 µM SQ109 (approximately three-fold the MBC). Analysis of the slopes of the kill curves revealed that the time required to kill 90% of the bacterial culture could be shortened by increasing the concentration of SQ109; 90% of the G27 strain was killed in approximately 7 h after incubation with 100 µM SQ109 while the same amount of killing could be achieved in 3-4 h upon incubation with 200 µM SQ109. Similar killing kinetics were observed against strain 7.13 (data not shown). Furthermore, similar killing kinetics were also obtained when the SQ109 experiments were conducted in vancomycin-free media; thus, vancomycin in the culture media had no effect on the potency of SQ109 (data not shown). These data indicate dose-dependent and time-dependent killing of H. pylori by SQ109.
Figure 1. Dose-dependent killing of H. pylori strain G27 by SQ109.

Approximately 8 x 107 cells were incubated with increasing concentrations of SQ109. The cultures were monitored for 24 h and sampled at the indicated times to determine surviving CFU by plating. The horizontal dashed line indicates the limit of detection (500 bacteria). The data are representative results from five independent experiments.
SQ109 efficacy against slow-growing bacteria
In vitro, antimicrobial agents are often tested against exponentially growing bacteria in order to achieve maximum bactericidal effects; however, in in vivo environments, pathogens often experience stress and nutrient limitations that slow or prevent bacterial growth [36,37]. Thus, we sought to determine the efficacy of SQ109 against a static or slow-growing culture of H. pylori strain G27. Growth arrest of the culture was achieved by exposure of the bacteria to a pre-determined concentration of Cm (30 µM). As shown in Figure 2A, the addition of 30 µM Cm inhibited H. pylori growth, but had no effect on survival. Conversely, addition of 140 µM or 200 µM SQ109 resulted in complete killing of the culture within 8 h or 6 h, respectively. Simultaneous addition of Cm and SQ109 to the culture similarly resulted in complete killing of H. pylori, though the kinetics of killing were slightly faster; 6 and 4 h were required to achieve complete clearance of bacteria with 140 µM or 200 µM SQ109, respectively (Figure 2A). These results suggest that SQ109 is active against H. pylori regardless of the growth state of the cells. Despite these results, we next considered the fact that the simultaneous growth arrest, through the addition of Cm, might not adequately recapitulate in vivo growth arrest. Colonizing H. pylori within the stomach would likely be in a state of continuous slow or no growth well before the addition of any therapeutic agent. Thus, we next asked whether SQ109 would be effective against bacterial cells that had been in a static state for an extended period of time. To achieve this state, we treated cultures initially with Cm for 16 h before washing and exposing to SQ109, Cm or SQ109 + Cm. Moreover, in order to demonstrate possible synergy in antibacterial activity of SQ109 in combination with Cm, pre-determined bacteriostatic concentrations of the two drugs were used (10 µM SQ109 [Figure 1] and 30 µM Cm [Figure 2A]). As expected, treatment with Cm for 16 h did not significantly damage the H. pylori cells since removal of the drug and exposure to PBS resulted in growth (Figure 2B). Similarly, incubation of cells with similar volumes of ethanol to control for ethanol used in solubilization of Cm, resulted in a growth profile similar to cells incubated with PBS (data not shown), indicating that the small volume of solvent did not impact H. pylori growth or survival. Exposure of the cells to increasing concentrations of SQ109 resulted in dose-dependent killing of H. pylori with killing kinetics similar to those previously observed in Figure 1 (Figure 2A). Continued exposure of the cells to Cm alone resulted in bacteriostasis and no dramatic changes in number of bacterial cells. However, the antibacterial activity of SQ109 was remarkably enhanced against H. pylori when the cells were pretreated with Cm and Cm remained in the culture (30 µM Cm + 10 µM SQ109; [Figure 2B]). While 10 µM SQ109 alone was bacteriostatic, the bacterial samples treated with 10 µM SQ109 + 30 µM Cm resulted in complete killing within 10 h (Figure 2B), indicative of drug synergy. A similar synergy was seen with 70 µM SQ109 + 30 µM Cm, however, the synergy was not as evident at 140 µM SQ109 + 30 µM Cm. This is likely due to rapid killing already observed at the higher concentration of SQ109. Taken together, our results indicate that SQ109 is active against slow-growing bacteria, which further suggest relevance of the antibacterial activity in vivo where the bacteria often show slow growth. These results further suggest that synergy may be achieved in combination therapy by initial pretreatment of the bacterial infection with a bacteriostatic agent, such as Cm.
Figure 2. Antibacterial activity of SQ109 against slow-growing H. pylori and synergy with chloramphenicol, Cm.
A predetermined bacteriostatic concentration of Cm (30 µM), was used to induce slow growth. Synergistic effects of SQ109 and Cm was then examined using two approaches: A) H. pylori strain G27 liquid cell cultures were treated simultaneously with 30 µM Cm and SQ109 (0 µM, 140 µM, and 200 µM) (open symbols), and the cells were monitored for 24 h, and B) H. pylori strain G27 cells were initially incubated with 30 µM Cm for 16 h in liquid culture medium before being split into multiple samples for further incubation with varying concentrations of SQ109 (0 µM, 10 µM, 70 µM or 140 µM) (open symbols) plus Cm. In both approaches, bacterial cells incubated with only PBS, SQ109 (0 µM, 10 µM, 70 µM, 140 µM or 200 µM) or Cm alone, were used as controls (closed symbols). The data are representative results from three independent experiments. The horizontal dashed line on each graph indicates the limit of detection (500 bacteria).
Effect of temperature and pH on antibacterial activity of SQ109
It is well accepted that antimicrobial activity of some drugs is affected by environmental conditions such as temperature and pH. The issue of temperature comes into play when one considers transport and storage of particular therapeutics, while pH stability is particularly important for H. pylori since the bacterium resides in the acidic environment of the stomach. Therefore, we tested the antimicrobial activity of SQ109 after exposure to various temperatures and acidic pH. Pre-exposure of SQ109 for 1 h to temperatures up to 95oC did not prevent the ability of SQ109 to kill H. pylori, though there was a temperature-dependent reduction in the time required to kill all of the bacterial cells (Figure 3A). SQ109 was also active at an acidic pH (pH 4.5), but again showed delayed killing kinetics as compared to activity at a more neutral pH (pH 6.8) (Figure 3B). These data suggest that although the time to kill H. pylori is extended by high temperatures and low pH, the drug remains bactericidal against the microbe.
Figure 3. Effect of temperature and low pH on the stability and bactericidal activity of SQ109.
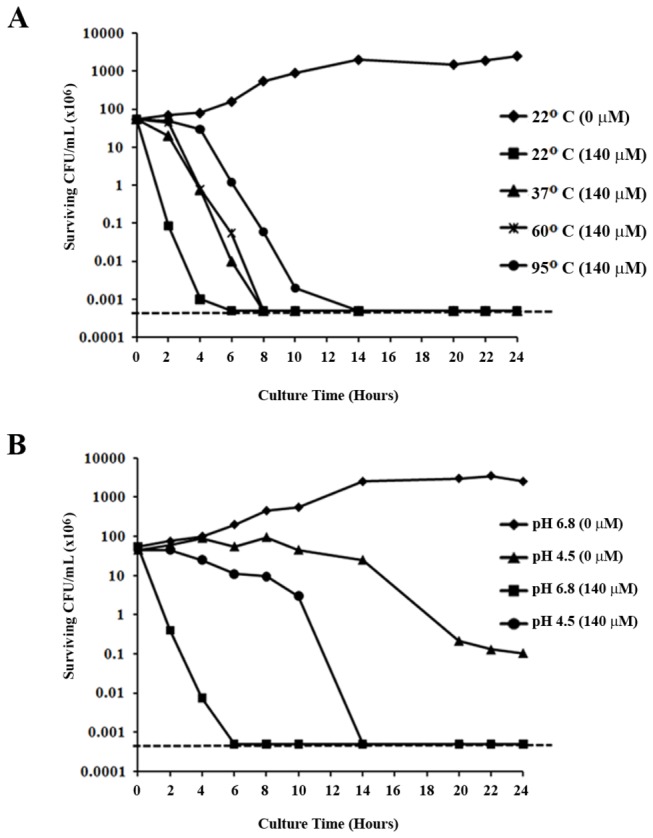
A) The effect of temperature on bactericidal activity of SQ109 was examined by pre-incubation of the drug at various temperatures (22oC, 37oC, 60oC and 95oC) for 1 h prior to use in the time-kill assay. B) In order to determine the effect of pH on SQ109, the time-kill assays were performed in pH-adjusted culture medium. The antibacterial activity of 140µM SQ109 against H. pylori cultured in pH 4.5 medium was compared to bactericidal activity at pH 6.8. The plotted data are representative results from three independent experiments. The horizontal dashed line on each graph indicates the limit of detection (500 bacteria).
Comparison of SQ109 antibacterial activity with conventionally used anti-H. pylori antibiotics
Any newly developed anti-H. pylori drug should show better or at least comparable efficacy to antibiotics currently used in treatment. We directly compared the antibacterial activity of SQ109 to equimolar concentrations of AMX, MTZ and CLR. Treatment with 70 µM of each of the drugs resulted in various rates of killing of the bacterial cells (Figure 4A). At this concentration, no viable H. pylori cells were recovered following 20 h of treatment with SQ109 (limit of detection = 500 bacteria). This was superior to the results obtained with AMX and MTZ, which both showed only 2-3 logs of killing in total, but inferior to CLR, which completely eliminated the bacterial culture after 4 h of treatment (Figure 4A). Increasing the concentration of the drugs to 140 µM increased the slope of the killing curve for each of the drugs, demonstrating time- and concentration-dependent effects of each drug. When the bacteria were exposed to 140 µM SQ109, no detectable CFU were observed after 8 h of exposure (Figure 4B). Once again, this antibacterial activity was superior to the results obtained with AMZ and MTZ, but the time for bacterial clearance was longer than for CLR (2 h). Taken en masse, these results indicate that SQ109 is superior or comparable to drugs currently used in anti-H. pylori therapy.
Figure 4. Molar-to-molar comparison of the antibacterial activity of SQ109, amoxicillin, metronidazole, and clarithromycin.

Time-kill assays for two drug concentrations, 70 µM (A) and 140 µM (B), were used to compare the bactericidal activity of SQ109 to those of conventional antibiotics currently used for the treatment of H. pylori infection. The data are representative results from three independent experiments. The horizontal dashed line on each graph indicates the limit of detection (500 bacteria).
H. pylori displays a low frequency of spontaneous resistance to SQ109 and further analysis suggests that surviving bacteria are largely “persisters”
Given that one of the greatest challenges posed by H. pylori is the development of resistance to nearly all standard drugs, we sought to examine the frequency of spontaneous resistance to SQ109. We examined this frequency for three H. pylori strains, G27 (most sensitive), 26695 (least sensitive), and B105 (least sensitive clinical isolate). Based on the determined MBC of 26695 (Table 1), we chose to test the frequency of resistance by plating on HBA plates containing 100 µM, 200 µM, or 400 µM SQ109. As shown in Table 3, at 100 µM SQ109, the frequency of resistance was 2.02 x 10-11, 2.08 x 10-10 and 1.9 x 10-9 for G27, 26695 and B105, respectively. At 200 µM SQ109, the rates were 2.13 x 10-12, 2.63 x 10-11 and < 9.6 x 10-13, respectively. No colonies were ever obtained on 400 µM SQ109 plates, even after testing more than 1012 cells.
Table 3. H. pylori frequency of resistant mutants to SQ109.
| H. pylori Strain | SQ109 Plate Concentration (µM) | Total CFU# | Frequency of Resistance to SQ109 (Resistant CFU/Total CFU) |
|---|---|---|---|
| G27 | 0 | 9.40E+11 | ND |
| 100 | 19 | 2.02E-11 | |
| 200 | 2 | 2.13E-12 | |
| 400 | 0 | < 1.06E-12 | |
| 26695 | 0 | 1.94E+12 | ND |
| 100 | 403 | 2.08E-10 | |
| 200 | 51 | 2.63E-11 | |
| 400 | 0 | < 5.15E-13 | |
| B105* | 0 | 1.03E+12 | ND |
| 100 | 1976 | 1.90E-09 | |
| 200 | 0 | < 9.63E-13 | |
| 400 | 0 | < 9.63E-13 |
# CFU = Colony Forming Units. * Low passage clinical isolate from a gastric ulcer patient. The data represent results from at least four independent experiments performed in triplicate plates.
The occurrence of persister cells as opposed to true resistant bacteria has been associated with enhanced antibiotic failure for many microbes. Therefore, to determine if the resistant colonies we obtained were in fact stably resistant, we utilized mutant stability tests to further analyze several of the colonies after growth in the absence of SQ109. Data for six of these colonies can be found in Figure S2. For colonies obtained from G27, which displays a MIC of 10-15 µM for SQ109, we observed a slight increase (approximately 2-fold) in the MIC for the resistant colonies. Conversely, no change in the apparent MBC was observed for these strains. Similarly, isolates obtained from B105 showed little to no difference in SQ109 MIC or MBC as compared to the parental wild-type strain. Moreover, when cultures of the resistant strains were spotted on HBA plates containing 100 µM or 200 µM SQ109, the number of colonies obtained, and thus frequencies of resistance, was identical to the wild-type strain (data not shown). These data suggest that the observed resistance was primarily transient in nature and likely originated from physiological adaptation rather than true resistance as a result of spontaneous mutation. Overall, these data demonstrate a very low frequency of resistance to SQ109 for all the strains tested, including a low-passage clinical isolate from a gastric ulcer patient.
SQ109 bactericidal activity is not due to disruption of H. pylori membranes
Antimicrobial agents work through various mechanisms that may target either the bacterial cell wall and/or cytoplasmic components. Antibiotics that target bacterial membranes may directly interact with the lipid bilayer and/or structural proteins found within the membranes. Previous studies using M. tuberculosis demonstrated that SQ109 exerts its antibacterial activity by targeting the cell wall [32]. Thus, in order to initiate characterization of the mechanism of action of SQ109 against H. pylori, we analyzed the ability of the drug to permeabilize the bacterial membrane. For these studies, we utilized a neutral fluorescent dye, NPN, whose fluorescence intensity increases in hydrophobic environments such as those resulting from damaged phospholipid bilayers. NPN is known to be non-fluorescent when excluded by intact cell membranes but fluoresces in the presence of damaged membranes [38,39].
Incubation of the G27 strain with 140 µM SQ109 resulted in no change in NPN fluorescence and showed fluorescence levels that appeared similar to PBS-treated (negative control) bacteria (Figure 5 line graphs). In contrast, samples treated with C12K-2β12 (positive control) showed an exponential increase in fluorescence; C12K-2β12 is known to cause membrane damage via the formation of pores [33]. SQ109 does not inhibit fluorescence of NPN since incubation of H. pylori with a combination of SQ109 and C12K-2β12 resulted in a similar increase in fluorescence intensity as that seen with C12K-2β12-treated bacteria (data not shown). Despite the lack of evidence of membrane disruption, SQ109 antibacterial activity remained potent (Figure 5 histograms). Overall, these data suggest that bactericidal activity of SQ109 may occur via interaction with a cytoplasmic target and not via disruption of the phospholipid cell membrane bilayer.
Figure 5. 1-N-phenylnaphthylamine membrane-permeabilization assay demonstrates that SQ109 bactericidal activity is not via membrane lysis.

Membrane-permeabilization and bactericidal assays were performed in parallel using H. pylori G27 cultured for 24 hours in the presence of 140 µM SQ109. Membrane permeation and bactericidal activity were monitored by fluorescence increase and by CFU enumeration using the micro-dilution plating method, respectively. Bacterial cells grown in the presence of 40 µM C12K-2β12 or PBS (no drugs) were used as controls. Approximately 5 x 106 bacterial cells were sampled at 1 h, 2 h, 4 h, 6 h and 10 h to determine 1-N-phenylnaphthylamine (NPN) uptake and bactericidal activity. Following excitation (λ = 350 nm) and emission (λ = 420 nm), the NPN fluorescence intensity was recorded in arbitrary units (AU) as a correlate of membrane permeation. Bactericidal activity was expressed as percent survival of starting CFU. The data shown are representative results of three independent experiments. The left axis presents NPN fluorescence depicted by lines while percent survival is shown on the right axis and is indicated by bars. The horizontal dashed line indicates the limit of detection (500 bacteria).
Distinct morphological and ultra-structural modifications of SQ109-treated H. pylori cells as visualized with TEM
In M. tuberculosis, SQ109 inhibits cell wall biosynthesis by interfering with the assembly of mycolic acids into the cell wall core [32]. Given that the data from the NPN permeation indicated no membrane disruption of cells exposed to SQ109, we next sought to examine the effect of SQ109 on the morphology and ultra-structure of the H. pylori cell wall and cytoplasm using TEM. Analysis of low magnification images (6,5000x) following 8 h of exposure to SQ109 revealed that nearly all of the H. pylori cells (95-99%) showed a deformed coccoid morphology and contained electron-dense and distorted cytoplasmic structures (Figure 6D). In addition, a small number of apparent ghost cells (<1-2%) were visible (Figure 6D arrowheads). In contrast, the PBS-treated samples showed typical spiral-comma rod-like H. pylori (Figure 6A). Consistent with our previously reported data [33], positive control cells treated with AMX appeared as swollen coccoid forms or ghost cells (Figure 6B) while C12K-2β12-treated cultures showed remarkable lysis (Figure 6C).
Figure 6. Visualization of SQ109-induced morphological changes of H. pylori using transmission electron microscopy (TEM).
Approximately 6 x 107 H. pylori cells were sampled following 8 h of exposure to PBS (A), 100 µM amoxicillin (B), 20 µM C 12K-2β12 peptide (C) or 140 µM SQ109 (D). Cells were examined at low magnification (6,500x) on a TEM and images were acquired and processed with AMT XR60B digital camera and AMTV600 software, respectively. (A) demonstrates normal spiral- or comma-shaped rod morphology of H. pylori cells as well as intact cell membranes following treatment with PBS as a negative control drug. Amoxicillin-treated cells (B) show characteristically swollen cells as well as detachment of the inner membrane from the outer membrane (white arrows). C12K-2β12-treated cells (C) show cell lysis, numerous ghost cells (C, white arrowheads) as well as the formation of electron dense structures within the extracellular medium and inside the cells. Like AMX-treated cells, SQ109-treated cells (D) present with inner membrane detachment from the outer membrane (white arrows), but are not significantly enlarged. Some ghost cells are visible (white arrowheads). Scale bars = 1500 nm. The data are representative images from two independent experiments.
Little or no cell membrane disruption of H. pylori by SQ109 was indicated by the NPN uptake assay (Figure 5). However, morphological changes associated with SQ109 treatment (Figure 6D) contradicted the absence of membrane damage. Thus, in order to conduct a detailed analysis of the effects of SQ109 on the H. pylori cell wall and cytoplasmic contents, we next examined the ultra-structural features of cells treated with SQ109 for 2 h or 8 h at higher magnification (42,000x). Cells treated with PBS, AMX or C12K-2β12 peptide, were included as controls. Representative TEM results of these studies are presented in Figure 7 and Figure S3.
Figure 7. TEM analysis of SQ109-induced morphological and ultra-structural changes in H. pylori-cell wall, membranes, and cytoplasm.
Approximately 6 x 107 H. pylori cells were sampled following 2 h (top panels) or 8 h- (bottom panels) of culture in the presence of PBS (A and B) as a negative control or 140 µM SQ109 (C, D, E, F, G, and H). Bacterial cells cultured in the presence of 100 µM AMX or 20 µM C 12K-2β12 were included as positive controls (data shown in Figure S3). PBS-treated negative control cells showed evidence of normal spiral-comma rod shaped morphology in addition to smooth homogenous cytoplasm (sc) and intact cell walls (A, B). In contrast, after 2 h of culture, SQ109 induced the formation of spindle actin-like cytoskeleton structures (a) in some cells (C) that appeared to condense cytoplasmic contents and lead to detachment of the IM from OM (arrows; C, E, F, G, and H) of nearly all cells resulting in complete disappearance of the periplasmic region; 95-99% of these cells also showed a deformed coccoid morphology (E, G, and H). After 2h and 8 h treatment with SQ109, the cells further showed blebs (b), electron-dense structures (e), evidence of complete loss of the IM and part of the cell wall with only the OM remaining intact (D), formation of two aberrant cytoplasmic bodies from a single cell (arrowheads) (G and H), and formation of outer membrane vesicles (omv) and inner membrane vesicles (imv) (C, F, G, and H). All scale bars are of 200 nm except B and E which measure 1500 nm. Representative images from two independent experiments are presented.
As shown, PBS-treated cells showed an intact cell wall with both inner (IM) and outer (OM) membranes appearing largely unperturbed. In addition, these cells tended to display a uniformly smooth bacterial cytoplasm, sc (Figure 7A and 7B, Figure S3-A, B, and C). In contrast, bacterial samples exposed to AMX showed separation of inner membranes from the outer membranes (arrows), blebs (b), outer membrane vesicles (omv) and ghost cells (gc) indicative of cell lysis (Figure S3-D, E and F). Like PBS-treated cells, AMX-treated bacteria typically showed uniformly smooth cytoplasm (sc). The treatment of the bacteria with C12K-2β12 revealed the formation of pores (p) and sloughing (s) in the outer membrane and electron-dense structures (e) in the cytoplasm (Figure S3-G, H and I). The results observed with AMX- or C12K-2β12-treated cells are similar to our previous data [33].
After 2 h of treatment of H. pylori with SQ109, there was evidence of outer membrane blebs (b) and inner membrane separation from the outer membrane to form bifocal actin-like spindle cytoskeleton structures (a) that appeared to condense the cytoplasmic contents to form inner membrane vesicles (imv) (Figure 7C, Figure S3-J). Figure 7D shows complete dissolution and separation of the inner membrane from the outer membrane with evidence of formation of outer membrane vesicles (omv) and subsequent lack of periplasm. In addition, SQ109 appeared to interact with cytoplamic target(s) as indicated by the presence of electron-dense structures (e) in the cytoplasm of nearly all the cells examined after 2 h or 8 h exposure to the peptide (Figure 7C–H, Figure S3-J–L), suggesting that SQ109 may also interact with bacterial cytoplasmic macromolecules, possibly nucleic acids and/or proteins [33]. There was also evidence of SQ109-induced disintegration of the inner membrane (Figure S3-K arrowheads) as well as an aberrant formation of two cytoplasmic bodies from a single cell (arrowheads) (Figure 7G and H, Figure S3-L) and the formation of inner membrane vesicles (imv) (Figure 7C, F, G , and H; Figure S3-J and K).
SQ109-induced changes in H. pylori showed both similarities and differences to AMX-or, C12K-2β12-treatment (Figure 7C–H, Figure S3-J–L). For instance, both SQ109- and AMX-treated cells showed the presence of vesicles and separation of the inner membrane from the outer membrane, but only AMX-treated cells showed a homogeneous cytoplasm. Both SQ109- and C12K-2β12-treated cells showed electron-dense cytoplasmic structures, but only C12K-2β12 induced pore formation. Overall, these results indicate differences in the mechanism of action of these drugs. Taken together, these data suggest that though the specific target is unclear, SQ109 exerts its antibacterial activity through interaction with both the inner membrane and cytoplasmic contents. In addition, the data further suggest that the mechanism of action of SQ109 is likely different from AMX and C12K-2β12.
Discussion
The significant new finding of this study is that SQ109, an anti-tubercular drug candidate that has shown promise in the treatment of human pulmonary tuberculosis, has a strong in vitro efficacy against H. pylori. Our data show that SQ109 displays robust thermal and pH stability, induces low/no spontaneous drug resistance and shows anti- Helicobacter superiority over MTZ and AMX. Furthermore, the data suggest SQ109 may have a novel mode of action against H. pylori.
Since its discovery in the early 1980s, H. pylori has been causally associated with the development of a diverse number of gastric diseases [40]. Current treatment strategies require the extended use of multiple antibiotics and a PPI [41–44], which leads to contraindications in some patients. This fact, plus the cost of using multiple drugs, has resulted in patient compliance issues [45,46]. Additionally, H. pylori exhibits a high frequency of mutation and genetic recombination [34,35], which no doubt contributes to the rapidly emerging antibiotic resistance of H. pylori in many areas of the world. Moreover, while H. pylori is primarily considered an extracellular pathogen, a sub-population of the infecting bacterial cells can survive as intracellular bacteria [47–49]. This finding when combined with ineffectiveness of currently used antibiotics in targeting intracellular H. pylori, suggests that the intracellular bacterial population of cells may be partially responsible for increasing rates of treatment failure [50,51]. Thus, the combination of the existence of “intracellular persister” bacteria and the emergence and spread of antibiotic resistant strains strongly suggest that new drugs and treatment strategies are needed for H. pylori.
SQ109, a new anti-tubercular drug that is currently in phase 2 clinical trials for adult pulmonary TB, is safe and well-tolerated in humans, and has the capacity to achieve intracellular bactericidal concentrations that kill M. tuberculosis [28,29]. Moreover, the apparent resistance rate of M. tuberculosis against SQ109 is 2.55x10-11, suggesting that the occurrence of clinical resistance to this drug may not be an issue [52,53]. These exciting findings combined with the fact that SQ109 accumulates in the stomach at high concentrations following oral administration in animal models [30,54], led us to test the in vitro susceptibility of H. pylori to SQ109.
For our analysis, we utilized concentration-time-kill assays to determine the MIC, MBC and killing kinetics of SQ109 against H. pylori, since this methodology is superior to measuring the 24 h, 3-log decrease in CFU/ml; the latter method does not adequately predict bactericidal activity [55]. Since strain-specific susceptibility to antibiotics has previously been demonstrated [31,56,57], we included a large collection of H. pylori strains to mimic geographic and clinical diversity. All 26 of the H. pylori strains tested were susceptible to SQ109. Notably, in addition to common laboratory strains and low passage clinical isolates, our collection included a multidrug resistant clinical isolate that was unable to be cleared by successive rounds of traditional anti-H. pylori therapy, suggesting that SQ109 may be useful for treatment of H. pylori infection even if the strain in question is resistant to traditional antibiotics (Figure S1). While the relative range of susceptibility to SQ109 was relatively small, 5 of the 26 strains were slightly less susceptible to the drug. These include strains B105, B107, B289, USU102 (Table 2), and 26695 (Table 1), the last of which was also previously shown to be less susceptible to other novel classes of anti-H. pylori therapies that are being developed [31]. While the nature of these minor strain-specific differences in sensitivity is not clear, it is possible that they are either due to differences in the SQ109 target within each of the strains, or drug accessibility to the target.
Current work in M. tuberculosis indicates that SQ109 interferes with the assembly of mycolic acids into the cell wall of the bacterium [58,59]. Specifically, the drug targets MmpL3, which appears to be a transporter of the mycobacterial cell wall component trehalose monomycolate (TMM) [32]. H. pylori does not produce mycolic acid and does not appear to encode a close protein homolog of MmpL3; BLAST analysis of MmpL3 directly against the H. pylori database shows that the closest H. pylori homolog (E=10-4) is the translocase subunit SecD, which is a component of an export system responsible for translocation of some proteins across the cell membranes. Converse analysis of the H. pylori SecD protein directly against the M. tuberculosis database shows that there is a much more closely related protein (E=10-26) in this bacterium that is likely the functional homolog of SecD; MmpL3 is the second most closely related protein (K. Sacksteder personal communication).
The specific SQ109 target and mechanism(s) of action within H. pylori remains unknown. However, our data broadly suggest that SQ109 exerts its anti-bactericidal effect by a two-fold effect: first, by interacting with the bacterial cell wall to result in retraction of the inner membrane from the outer membrane, and secondly, by targeting cytoplasmic content(s) to induce formation of electron-dense structures in the cytoplasm (Figure 7F and 7G). Notably, the lack of NPN-uptake (Figure 5) suggests that the effect of SQ109 on the H. pylori cell wall occurs without disruption of the hydrophobic phospholipid bilayer. This phenomenon may not be unique to SQ109; similar effects in which an antibacterial agent was able to interact with bacterial membrane to gain entry into the cytoplasm without dramatic cell wall disruption have been previously reported [60]. Consistent with the lack of membrane disruption, we observed that SQ109-treated cells were not dramatically enlarged (Figures 6D and 7E); this suggests minimal or no membrane permeation induced by the drug, which is also consistent with lack of lysis. Moreover, at early time points, SQ109 induced the formation of actin-like filaments in the cytoplasm that appeared to condense the cytoplasmic contents and to cause detachment of the IM from the OM. Similar actin-like cytoskeleton structures have been reported in antibiotic-treated Bacillus subtilis [61], Caulobacter crescentus [62], and Chlamydia trachomatis [63]. Indeed, in studies in which antibiotics targeted MreB, which is a rod shape-determining protein linked to penicillin-binding protein-2 in bacilli that is responsible for bacterial actin-like cytoskeleton structures, the cells changed shape [63]. This finding is consistent with our observation that SQ109 induced coccoid morphology and detachment of the IM from the OM; these changes were similarly evident in AMX-treated cells. AMX exerts its effect by inhibition of assembly of cell wall peptidoglycan through the binding of PBP. Therefore, SQ109-induced detachment of IM from OM may be due to inhibition of cell wall biosynthesis, resulting in a lack of peptidoglycan, the matrix that cross-links the two membranes and keeps them together [64,65]. The differences in SQ109-induced changes as compared to those of AMX (such as the presence of electron-dense structures) and those of C12K-2β12 such as the lack of pore formation, likely relate to differences in mechanism of action. Thus, further studies are required to identify the specific target(s) of SQ109 in H. pylori.
Of note, many antibiotics fail in clinical trials due to an inability to reach sufficient concentrations in infected tissue. This phenomenon, which is also referred to as “first-pass effect”, has been attributed to metabolism of orally administered drugs by gastrointestinal and hepatic enzymes; this results in a significant reduction of the amount of un-metabolized drug reaching the systemic circulation [66]. However, in H. pylori infections, the bacterium colonizes the stomach, thus, the “first-pass effect” can be bypassed, and increased bioavailability of a drug can be achieved by oral administration of many drugs. Moreover, SQ109 has the ability to translocate into the host cell and kill intracellular M. tuberculosis, whereas conventional antibiotics used to treat H. pylori infection likely do not reach suitable concentrations inside the host cell to sterilize the environment [67,68]. Thus, SQ109 may also be effective in killing intracellular H. pylori [47–49], which are believed to be partially responsible for treatment failures through reseeding the infection to the gastric mucosa [47,67,68].
Prospectively, assuming a mouse stomach mass of 200 mg and the previously reported stomach tissue analysis of SQ109 concentration at 1 h and 4 h post-oral administration [30], our results indicate that the SQ109 concentration in the stomach would be approximately two- to five-fold higher than the determined drug MBC (50-100 µM; Tables 1 and 2). It is envisaged that the high concentration of SQ109 maintained for 1-4 h would give the drug time to diffuse into the mucus and/or be absorbed systemically to reach the bacterium via the basolateral surface. Thus, given the observation that the concentration of SQ109 that accumulates in the stomach of animals orally dosed with the drug is well above the MBC for H. pylori, it may be possible to use SQ109 as an effective monotherapy for H. pylori infection. This notion is further supported by our findings that the frequency of resistance is very low (Table 3) and that the antimicrobial activity of SQ109 was superior to the activity of MTZ and AMX on a molar-to-molar basis (Figure 4). Importantly, even if the drug is ineffective as a monotherapy, our finding that a normally bacteriostatic concentration of SQ109 shows synergistic bactericidal activity when administered with Cm suggests that the drug may be effective when co-administered with Cm or other clinically relevant bacteriostatic agents. Our data further describe a preliminary mechanism of action and suggest that the SQ109 acts by interaction with both cell wall and cytoplasmic contents. Thus, our data suggest that SQ109 may represent a powerful new class of drugs against H. pylori that may be used to significantly improve treatment success and eradication of this pathogen.
Supporting Information
Bactericidal assay of selected antibiotics, AMX, MTZ, CLR, and TET to the clinical isolate (USU103) was performed to analyze the resistance levels of the bacteria. The percent survival (vertical axis) of H. pylori clinical isolate, USU103, is shown at the indicated antibiotic concentration (horizontal axis). The bacterial CFU/ml was determined by plating following culture for 24 h in the presence of a range of concentrations of AMX (0.5-1000 µg/ml), MTZ (0.5-1000 µg/ml), CLR (0.5-100 µg/ml), and TET (0.5-100 µg/ml). Percent survival was calculated using the formula: % survival = CFUt24/CFUt0 × 100, where CFUt0 represents CFU at the beginning of the experiment and CFUt24 represents CFU at 24 h of exposure to the antibiotic. Resistance was defined as follows: for AMX (< 1 µg/ml), MTZ (< 8 µg/ml), CLR (< 1 µg/ml), and TET (< 4 µg/ml). The dotted horizontal line represents the MIC. Data are representative of three independent experiments.
(TIF)
A total of six mutants, three from G27 (R1S1, R1S2, and R1S3) and another three from B105 (R870S1, R870S2, and R870S3) were cultured from stocks in a total of five rounds of 24 h-passages by inoculation onto SQ109-free plates. A new bactericidal assay expressed as percent survival was performed to determine MIC and MBC for SQ109 as described and defined in Materials and Methods. The mutant and parental (wild-type) MICs and MBCs were compared to evaluate the presence or absence of persisters. The data are representative of two experiments.
(TIF)
Approximately 6 x 107 H. pylori G27 strain cells were sampled following 2 h (upper panels) or 8 h- (middle and bottom panels) culture in the presence of PBS, no antibiotics (A, B, and C) as negative control, 100 µM AMX (D, E, and F) as positive control, 20 µM C12K-2β12 (G, H, and I) as positive control, or 140 µM SQ109 (J, K, and L). AMX-treated cells (A, B) showed detachment of inner membrane from outer membrane (double arrows) in addition to formation of vesicles (v), outer membrane vesicles (omv), (E), and ghost cells (gc), (F). Like PBS-treated control cells, AMX-treated cells also had smooth homogeneous cytoplasm (sc), (D, E, F). C12K-2β12 induced the formation of pores (p), membrane sloughing (s), and formation of electron-dense structures (e) in the cytoplasm (G, H, I). In contrast, at 2 h of culture SQ109 induced the formation of spindle actin-like cytoskeleton structures (a) that appear to condense cytoplasmic contents leading to detachment of IM from OM (double arrows) similar to AMX-treated cells. At 8 h of exposure to SQ109, cells showed blebs (b) that appeared to pinch off to form outer membrane vesicles (omv) (L) in addition to evidence of formation of inner membrane vesicles (imv) (K), disintegration of IM (arrowheads) and aberrant constriction of cell wall to form two cells (L). Scale bars (white) = 200 nm. The data are representative images from two independent experiments.
(TIF)
Acknowledgments
We are grateful to Drs. Andre Dubois and Jeong-Heon Cha for providing H. pylori strains and to Dr. Carol A. Nacy for valuable comments, suggestions and critical review of the manuscript draft. Technical assistance for TEM studies was provided by Dr. Ru-Ching Hsia and Ms. Johanna M. Sotiris at the Electron Microscopy Core Facility, Department of Microbial Pathogenesis, University of Maryland School of Dentistry, Baltimore, Maryland. Contents of this manuscript are the sole responsibility of the authors and do not necessarily represent the official views of the NIH, the DOD, USUHS or the federal government.
Funding Statement
The authors have no support or funding to report.
References
- 1. Brown LM (2000) Helicobacter pylori: epidemiology and routes of transmission. Epidemiol Rev 22: 283-297. doi:10.1093/oxfordjournals.epirev.a018040. PubMed: 11218379. [DOI] [PubMed] [Google Scholar]
- 2. Torres J, Leal-Herrera Y, Perez-Perez G, Gomez A, Camorlinga-Ponce M et al. (1998) A community-based seroepidemiologic study of Helicobacter pylori infection in Mexico. J Infect Dis 178: 1089-1094. doi:10.1086/515663. PubMed: 9806039. [DOI] [PubMed] [Google Scholar]
- 3. Meurer LN, Bower DJ (2002) Management of Helicobacter pylori infection. Am Fam Physician 65: 1327-1336. PubMed: 11996414. [PubMed] [Google Scholar]
- 4. Sobala GM, Crabtree JE, Dixon MF, Schorah CJ, Taylor JD et al. (1991) Acute Helicobacter pylori infection: clinical features, local and systemic immune response, gastric mucosal histology, and gastric juice ascorbic acid concentrations. Gut 32: 1415-1418. doi:10.1136/gut.32.11.1415. PubMed: 1752479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Suerbaum S, Michetti P (2002) Helicobacter pylori infection. N Engl J Med 347: 1175-1186. doi:10.1056/NEJMra020542. PubMed: 12374879. [DOI] [PubMed] [Google Scholar]
- 6. Graham DY, Hepps KS, Ramirez FC, Lew GM, Saeed ZA (1993) Treatment of Helicobacter pylori reduces the rate of rebleeding in peptic ulcer disease. Scand J Gastroenterol 28: 939-942. doi:10.3109/00365529309098288. PubMed: 8284627. [DOI] [PubMed] [Google Scholar]
- 7. Treiber G, Lambert JR (1998) The impact of Helicobacter pylori eradication on peptic ulcer healing. Am J Gastroenterol 93: 1080-1084. doi:10.1111/j.1572-0241.1998.00333.x. PubMed: 9672334. [DOI] [PubMed] [Google Scholar]
- 8. Matsuzaki J, Suzuki H, Nishizawa T, Hirata K, Tsugawa H et al. (2011) Efficacy of sitafloxacin-based rescue therapy for Helicobacter pylori after failures of first- and second-line therapies. Antimicrob Agents Chemother. [DOI] [PMC free article] [PubMed]
- 9. Gisbert JP (2011) Rescue therapy after Helicobacter pylori eradication failure. Gastroenterol Hepatol 34: 89-99. doi:10.1016/S0210-5705(11)70014-0. [DOI] [PubMed] [Google Scholar]
- 10. Rokkas T, Sechopoulos P, Robotis J, Pistiolas D (2006) Triple levofloxacin-based rescue therapy is an accepted empirical third-line treatment. Am J Gastroenterol 101: 1938-1939. doi:10.1111/j.1572-0241.2006.00684_2.x. PubMed: 16928256. [DOI] [PubMed] [Google Scholar]
- 11. Gisbert JP (2008) "Rescue" regimens after Helicobacter pylori treatment failure. World J Gastroenterol 14: 5385-5402. doi:10.3748/wjg.14.5385. PubMed: 18803350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Mégraud F (2004) H. pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut 53: 1374-1384. doi:10.1136/gut.2003.022111. PubMed: 15306603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Belhoussine-Idrissi L, Boedeker EC (2002) Helicobacter pylori infection: treatment. Curr Opin Gastroenterol 18: 26-33. doi:10.1097/00001574-200201000-00005. PubMed: 17031226. [DOI] [PubMed] [Google Scholar]
- 14. Malfertheiner P, Megraud F, O’Morain C, Bazzoli F, El-Omar E et al. (2007) Current concepts in the management of Helicobacter pylori infection: the Maastricht III Consensus Report. Gut 56: 772-781. doi:10.1136/gut.2006.101634. PubMed: 17170018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Projan SJ (2003) Why is big Pharma getting out of antibacterial drug discovery? Curr Opin Microbiol 6: 427-430. doi:10.1016/j.mib.2003.08.003. PubMed: 14572532. [DOI] [PubMed] [Google Scholar]
- 16. Nathan C (2004) Antibiotics at the crossroads. Nature 431: 899-902. doi:10.1038/431899a. PubMed: 15496893. [DOI] [PubMed] [Google Scholar]
- 17. Piddock LJ (2011) The crisis of no new antibiotics-what is the way forward? Lancet Infect Dis. [DOI] [PubMed] [Google Scholar]
- 18. Baltz RH (2006) Molecular engineering approaches to peptide, polyketide and other antibiotics. Nat Biotechnol 24: 1533-1540. doi:10.1038/nbt1265. PubMed: 17160059. [DOI] [PubMed] [Google Scholar]
- 19. Payne DJ, Gwynn MN, Holmes DJ, Pompliano DL (2007) Drugs for bad bugs: confronting the challenges of antibacterial discovery. Nat Rev Drug Discov 6: 29-40. doi:10.1038/nrd2201. PubMed: 17159923. [DOI] [PubMed] [Google Scholar]
- 20. Marchese A, Schito GC (2001) The oxazolidinones as a new family of antimicrobial agent. Clin Microbiol Infect 7 Suppl 4: 66-74. doi:10.1046/j.1469-0691.2001.00060.x. PubMed: 11688536. [DOI] [PubMed] [Google Scholar]
- 21. Diekema DI, Jones RN (2000) Oxazolidinones: a review. Drugs 59: 7-16. doi:10.2165/00003495-200059010-00002. PubMed: 10718097. [DOI] [PubMed] [Google Scholar]
- 22. Mukhtar TA, Koteva KP, Wright GD (2005) Chimeric streptogramin-tyrocidine antibiotics that overcome streptogramin resistance. Chem Biol 12: 229-235. [DOI] [PubMed] [Google Scholar]
- 23. Mukhtar TA, Wright GD (2005) Streptogramins, oxazolidinones, and other inhibitors of bacterial protein synthesis. Chem Rev 105: 529-542. doi:10.1021/cr030110z. PubMed: 15700955. [DOI] [PubMed] [Google Scholar]
- 24. Protopopova M, Hanrahan C, Nikonenko B, Samala R, Chen P et al. (2005) Identification of a new antitubercular drug candidate, SQ109, from a combinatorial library of 1,2-ethylenediamines. J Antimicrob Chemother 56: 968-974. doi:10.1093/jac/dki319. PubMed: 16172107. [DOI] [PubMed] [Google Scholar]
- 25. Jia L, Tomaszewski JE, Hanrahan C, Coward L, Noker P et al. (2005) Pharmacodynamics and pharmacokinetics of SQ109, a new diamine-based antitubercular drug. Br J Pharmacol 144: 80-87. PubMed: 15644871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Chen P, Gearhart J, Protopopova M, Einck L, Nacy CA (2006) Synergistic interactions of SQ109, a new ethylene diamine, with front-line antitubercular drugs in vitro . J Antimicrob Chemother 58: 332-337. doi:10.1093/jac/dkl227. PubMed: 16751637. [DOI] [PubMed] [Google Scholar]
- 27. Nikonenko BV, Protopopova M, Samala R, Einck L, Nacy CA (2007) Drug therapy of experimental tuberculosis (TB): improved outcome by combining SQ109, a new diamine antibiotic, with existing TB drugs. Antimicrob Agents Chemother 51: 1563-1565. doi:10.1128/AAC.01326-06. PubMed: 17242141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Horwith G, Einck L, Protopopova M, Nacy C (2007) Phase 1 safety and pharmacokinetics of SQ109, a new diamine antituberculosis drug. Meeting Abstract, 45th IDSA; Annual: Meeting, 2007 Oct 4-7; San Diego, CA, USA: Rockville, MD: Sequella, Inc. [Google Scholar]
- 29. National, Institute, of, Allergy, and, et al (2010) Dose escalation Study of SQ109 in healthy adult volunteers US National Institutes of Health, ClinicalTrials.gov NCT00866190. Available: http://www.clinicaltrials.gov [Accessed 28 Jun 2010]
- 30. Jia L, Noker PE, Coward L, Gorman GS, Protopopova M et al. (2006) Interspecies pharmacokinetics and in vitro metabolism of SQ109. Br J Pharmacol 147: 476-485. doi:10.1038/sj.bjp.0706650. PubMed: 16432511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Makobongo MO, Kovachi T, Gancz H, Mor A, Merrell DS (2009) In vitro antibacterial activity of acyl-lysyl oligomers against Helicobacter pylori . Antimicrob Agents Chemother 53: 4231-4239. doi:10.1128/AAC.00510-09. PubMed: 19620333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Tahlan K, Wilson R, Kastrinsky DB, Arora K, Nair V et al. (2012) SQ109 Targets MmpL3 a membrane transporter of trehalose tonomycolate involved in mycolic acid donation to the cell wall core of Mycobacterium tuberculosis . Antimicrob Agents Chemother 56 (4):1797-809 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Makobongo MO, Gancz H, Carpenter BM, McDaniel DP, Merrell DS (2012) The oligo-acyl lysyl antimicrobial peptide C12K-2β 12 exhibits a dual mechanism of action and demonstrates strong in vivo efficacy against Helicobacter pylori . Antimicrob Agents Chemother 56: 378-390. doi:10.1128/AAC.00689-11. PubMed: 22064541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Suerbaum S, Smith JM, Bapumia K, Morelli G, Smith NH et al. (1998) Free recombination within Helicobacter pylori . Proc Natl Acad Sci U S A 95: 12619-12624. doi:10.1073/pnas.95.21.12619. PubMed: 9770535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Björkholm B, Sjölund M, Falk PG, Berg OG, Engstrand L et al. (2001) Mutation frequency and biological cost of antibiotic resistance in Helicobacter pylori . Proc Natl Acad Sci U S A 98: 14607-14612. doi:10.1073/pnas.241517298. PubMed: 11717398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Millar MR, Pike J (1992) Bactericidal activity of antimicrobial agents against slowly growing Helicobacter pylori . Antimicrob Agents Chemother 36: 185-187. doi:10.1128/AAC.36.1.185. PubMed: 1590687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Beste DJ, Espasa M, Bonde B, Kierzek AM, Stewart GR et al. (2009) The genetic requirements for fast and slow growth in mycobacteria. PLOS ONE 4: e5349. doi:10.1371/journal.pone.0005349. PubMed: 19479006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Loh B, Grant C, Hancock RE (1984) Use of the fluorescent probe 1-N-phenylnaphthylamine to study the interactions of aminoglycoside antibiotics with the outer membrane of Pseudomonas aeruginosa . Antimicrob Agents Chemother 26: 546-551. doi:10.1128/AAC.26.4.546. PubMed: 6440475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Hancock RE, Wong PG (1984) Compounds which increase the permeability of the Pseudomonas aeruginosa outer membrane. Antimicrob Agents Chemother 26: 48-52. doi:10.1128/AAC.26.1.48. PubMed: 6433788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Ahmed N, Sechi LA (2005) Helicobacter pylori and gastroduodenal pathology: new threats of the old friend. Ann Clin Microbiol Antimicrob 4: 1. doi:10.1186/1476-0711-4-1. PubMed: 15634357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Chuah SK, Tsay FW, Hsu PI, Wu DC (2011) A new look at anti-Helicobacter pylori therapy. World J Gastroenterol 17: 3971-3975. doi:10.3748/wjg.v17.i35.3971. PubMed: 22046084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Fakheri H, Taghvaei T, Hosseini V, Bari Z (2012) A Comparison between sequential therapy and a modified bismuth-based quadruple therapy for Helicobacter pylori eradication in Iran: A randomized clinical trial. Helicobacter 17: 43-48. doi:10.1111/j.1523-5378.2011.00896.x. PubMed: 22221615. [DOI] [PubMed] [Google Scholar]
- 43. Oh HS, Lee DH, Seo JY, Cho YR, Kim N et al. (2011) Ten-day sequential therapy is more effective than proton-pump inhibitor-based therapy in Korea: a prospective randomized study. J Gastroenterol Hepatol. [DOI] [PubMed] [Google Scholar]
- 44. Park HG, Jung MK, Jung JT, Kwon JG, Kim EY et al. (2012) Randomised clinical trial: a comparative study of 10-day sequential therapy with 7-day standard triple therapy for Helicobacter pylori infection in naive patients. Aliment Pharmacol Ther 35: 56-65. doi:10.1111/j.1365-2036.2011.04902.x. PubMed: 22066530. [DOI] [PubMed] [Google Scholar]
- 45. Ables AZ, Simon I, Melton ER (2007) Update on Helicobacter pylori treatment. Am Fam Physician 75: 351-358. PubMed: 17304866. [PubMed] [Google Scholar]
- 46. Malfertheiner P (1993) Compliance, adverse events and antibiotic resistance in Helicobacter pylori treatment. Scand J Gastroenterol Suppl 196: 34-37. PubMed: 8341989. [DOI] [PubMed] [Google Scholar]
- 47. Amieva MR, Salama NR, Tompkins LS, Falkow S (2002) Helicobacter pylori enter and survive within multivesicular vacuoles of epithelial cells. Cell Microbiol 4: 677-690. doi:10.1046/j.1462-5822.2002.00222.x. PubMed: 12366404. [DOI] [PubMed] [Google Scholar]
- 48. Semino-Mora C, Doi SQ, Marty A, Simko V, Carlstedt I et al. (2003) Intracellular and interstitial expression of Helicobacter pylori virulence genes in gastric precancerous intestinal metaplasia and adenocarcinoma. J Infect Dis 187: 1165-1177. doi:10.1086/368133. PubMed: 12695995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Dubois A (2007) Intracellular Helicobacter pylori and gastric carcinogenesis: an "old" frontier worth revisiting. Gastroenterology 132: 1177-1180. doi:10.1053/j.gastro.2007.01.068. PubMed: 17383438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Kearney DJ (2001) Retreatment of Helicobacter pylori infection after initial treatment failure. Am J Gastroenterol 96: 1335-1339. doi:10.1111/j.1572-0241.2001.03788.x. PubMed: 11374665. [DOI] [PubMed] [Google Scholar]
- 51. Gisbert JP, Bermejo F, Castro-Fernández M, Pérez-Aisa A, Fernández-Bermejo M et al. (2008) Second-line rescue therapy with levofloxacin after H. pylori treatment failure: a Spanish multicenter study of 300 patients. Am J Gastroenterol 103: 71-76. doi:10.1111/j.1572-0241.2007.01500.x. PubMed: 17764498. [DOI] [PubMed] [Google Scholar]
- 52. van den Boogaard J, Kibiki GS, Kisanga ER, Boeree MJ, Aarnoutse RE (2009) New drugs against tuberculosis: problems, progress, and evaluation of agents in clinical development. Antimicrob Agents Chemother 53: 849-862. doi:10.1128/AAC.00749-08. PubMed: 19075046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Sánchez F, López Colomés JL, Villarino E, Grosset J (2011) New drugs for tuberculosis treatment. Enferm Infecc Microbiol Clin 29 Suppl 1: 47-56. doi:10.1016/S0213-005X(11)70018-0. PubMed: 21420567. [DOI] [PubMed] [Google Scholar]
- 54. Jia L, Coward L, Gorman GS, Noker PE, Tomaszewski JE (2005) Pharmacoproteomic effects of isoniazid, ethambutol, and N-geranyl-N'-(2-adamantyl)ethane-1,2-diamine (SQ109) on Mycobacterium tuberculosis H37Rv. J Pharmacol Exp Ther 315: 905-911. doi:10.1124/jpet.105.087817. PubMed: 16085758. [DOI] [PubMed] [Google Scholar]
- 55. Stratton CW, Aldridge KE, Gelfand MS (1995) In vitro killing of penicillin-susceptible, -intermediate, and -resistant strains of Streptococcus pneumoniae by cefotaxime, ceftriaxone, and ceftizoxime: a comparison of bactericidal and inhibitory activity with achievable CSF levels. Diagn Microbiol Infect Dis 22: 35-42. doi:10.1016/0732-8893(95)00094-Q. PubMed: 7587048. [DOI] [PubMed] [Google Scholar]
- 56. Han SR, Bhakdi S, Maeurer MJ, Schneider T, Gehring S (1999) Stable and unstable amoxicillin resistance in Helicobacter pylori: should antibiotic resistance testing be performed prior to eradication therapy? J Clin Microbiol 37: 2740-2741. PubMed: 10405437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Kim JM, Kim JS, Jung HC, Kim N, Kim YJ et al. (2004) Distribution of antibiotic MICs for Helicobacter pylori strains over a 16-year period in patients from Seoul, South Korea. Antimicrob Agents Chemother 48: 4843-4847. doi:10.1128/AAC.48.12.4843-4847.2004. PubMed: 15561865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Lee RE, Protopopova M, Crooks E, Slayden RA, Terrot M et al. (2003) Combinatorial lead optimization of [1,2]-diamines based on ethambutol as potential antituberculosis preclinical candidates. J Comb Chem 5: 172-187. doi:10.1021/cc020071p. PubMed: 12625709. [DOI] [PubMed] [Google Scholar]
- 59. Guy ES, Mallampalli A (2008) Managing TB in the 21st century: existing and novel drug therapies. Ther Adv Respir Dis 2: 401-408. doi:10.1177/1753465808099522. PubMed: 19124385. [DOI] [PubMed] [Google Scholar]
- 60. Rotem S, Radzishevsky IS, Bourdetsky D, Navon-Venezia S, Carmeli Y et al. (2008) Analogous oligo-acyl-lysines with distinct antibacterial mechanisms. FASEB J 22: 2652-2661. doi:10.1096/fj.07-105015. PubMed: 18385215. [DOI] [PubMed] [Google Scholar]
- 61. Tiyanont K, Doan T, Lazarus MB, Fang X, Rudner DZ et al. (2006) Imaging peptidoglycan biosynthesis in Bacillus subtilis with fluorescent antibiotics. Proc Natl Acad Sci U S A 103: 11033-11038. doi:10.1073/pnas.0600829103. PubMed: 16832063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Takacs CN, Poggio S, Charbon G, Pucheault M, Vollmer W et al. (2010) MreB drives de novo rod morphogenesis in Caulobacter crescentus via remodeling of the cell wall. J Bacteriol 192: 1671-1684. doi:10.1128/JB.01311-09. PubMed: 20023035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Ouellette SP, Karimova G, Subtil A, Ladant D (2012) Chlamydia co-opts the rod shape-determining proteins MreB and Pbp2 for cell division. Mol Microbiol 85: 164-178. doi:10.1111/j.1365-2958.2012.08100.x. PubMed: 22624979. [DOI] [PubMed] [Google Scholar]
- 64. Hakenbeck R, König A, Kern I, van der Linden M, Keck W et al. (1998) Acquisition of five high-Mr penicillin-binding protein variants during transfer of high-level beta-lactam resistance from Streptococcus mitis to Streptococcus pneumoniae . J Bacteriol 180: 1831-1840. PubMed: 9537382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Okamoto T, Yoshiyama H, Nakazawa T, Park ID, Chang MW et al. (2002) A change in PBP1 is involved in amoxicillin resistance of clinical isolates of Helicobacter pylori . J Antimicrob Chemother 50: 849-856. doi:10.1093/jac/dkf140. PubMed: 12461003. [DOI] [PubMed] [Google Scholar]
- 66. Calafatti SA, Ortiz RA, Deguer M, Martinez M, Pedrazzoli J Jr. (2001) Effect of acid secretion blockade by omeprazole on the relative bioavailability of orally administered furazolidone in healthy volunteers. Br J Clin Pharmacol 52: 205-209. doi:10.1046/j.0306-5251.2001.01435.x. PubMed: 11488780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Chu YT, Wang YH, Wu JJ, Lei HY (2010) Invasion and multiplication of Helicobacter pylori in gastric epithelial cells and implications for antibiotic resistance. Infect Immun 78: 4157-4165. doi:10.1128/IAI.00524-10. PubMed: 20696835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Wang YH, Wu JJ, Lei HY (2009) When Helicobacter pylori invades and replicates in the cells. Autophagy 5: 540-542. doi:10.4161/auto.5.4.8167. PubMed: 19270492. [DOI] [PubMed] [Google Scholar]
- 69. Baltrus DA, Amieva MR, Covacci A, Lowe TM, Merrell DS et al. (2009) The complete genome sequence of Helicobacter pylori strain G27. J Bacteriol 191: 447-448. doi:10.1128/JB.01416-08. PubMed: 18952803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Franco AT, Israel DA, Washington MK, Krishna U, Fox JG et al. (2005) Activation of beta-catenin by carcinogenic Helicobacter pylori . Proc Natl Acad Sci U S A 102: 10646-10651. doi:10.1073/pnas.0504927102. PubMed: 16027366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Oh JD, Kling-Bäckhed H, Giannakis M, Xu J, Fulton RS et al. (2006) The complete genome sequence of a chronic atrophic gastritis Helicobacter pylori strain: evolution during disease progression. Proc Natl Acad Sci U S A 103: 9999-10004. doi:10.1073/pnas.0603784103. PubMed: 16788065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Lee A, O’Rourke J, De Ungria MC, Robertson B, Daskalopoulos G et al. (1997) A standardized mouse model of Helicobacter pylori infection: introducing the Sydney strain. Gastroenterology 112: 1386-1397. doi:10.1016/S0016-5085(97)70155-0. PubMed: 9098027. [DOI] [PubMed] [Google Scholar]
- 73. Alm RA, Ling LS, Moir DT, King BL, Brown ED et al. (1999) Genomic-sequence comparison of two unrelated isolates of the human gastric pathogen Helicobacter pylori . Nature 397: 176-180. doi:10.1038/16495. PubMed: 9923682. [DOI] [PubMed] [Google Scholar]
- 74. Tomb JF, White O, Kerlavage AR, Clayton RA, Sutton GG et al. (1997) The complete genome sequence of the gastric pathogen Helicobacter pylori . Nature 388: 539-547. doi:10.1038/41483. PubMed: 9252185. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Bactericidal assay of selected antibiotics, AMX, MTZ, CLR, and TET to the clinical isolate (USU103) was performed to analyze the resistance levels of the bacteria. The percent survival (vertical axis) of H. pylori clinical isolate, USU103, is shown at the indicated antibiotic concentration (horizontal axis). The bacterial CFU/ml was determined by plating following culture for 24 h in the presence of a range of concentrations of AMX (0.5-1000 µg/ml), MTZ (0.5-1000 µg/ml), CLR (0.5-100 µg/ml), and TET (0.5-100 µg/ml). Percent survival was calculated using the formula: % survival = CFUt24/CFUt0 × 100, where CFUt0 represents CFU at the beginning of the experiment and CFUt24 represents CFU at 24 h of exposure to the antibiotic. Resistance was defined as follows: for AMX (< 1 µg/ml), MTZ (< 8 µg/ml), CLR (< 1 µg/ml), and TET (< 4 µg/ml). The dotted horizontal line represents the MIC. Data are representative of three independent experiments.
(TIF)
A total of six mutants, three from G27 (R1S1, R1S2, and R1S3) and another three from B105 (R870S1, R870S2, and R870S3) were cultured from stocks in a total of five rounds of 24 h-passages by inoculation onto SQ109-free plates. A new bactericidal assay expressed as percent survival was performed to determine MIC and MBC for SQ109 as described and defined in Materials and Methods. The mutant and parental (wild-type) MICs and MBCs were compared to evaluate the presence or absence of persisters. The data are representative of two experiments.
(TIF)
Approximately 6 x 107 H. pylori G27 strain cells were sampled following 2 h (upper panels) or 8 h- (middle and bottom panels) culture in the presence of PBS, no antibiotics (A, B, and C) as negative control, 100 µM AMX (D, E, and F) as positive control, 20 µM C12K-2β12 (G, H, and I) as positive control, or 140 µM SQ109 (J, K, and L). AMX-treated cells (A, B) showed detachment of inner membrane from outer membrane (double arrows) in addition to formation of vesicles (v), outer membrane vesicles (omv), (E), and ghost cells (gc), (F). Like PBS-treated control cells, AMX-treated cells also had smooth homogeneous cytoplasm (sc), (D, E, F). C12K-2β12 induced the formation of pores (p), membrane sloughing (s), and formation of electron-dense structures (e) in the cytoplasm (G, H, I). In contrast, at 2 h of culture SQ109 induced the formation of spindle actin-like cytoskeleton structures (a) that appear to condense cytoplasmic contents leading to detachment of IM from OM (double arrows) similar to AMX-treated cells. At 8 h of exposure to SQ109, cells showed blebs (b) that appeared to pinch off to form outer membrane vesicles (omv) (L) in addition to evidence of formation of inner membrane vesicles (imv) (K), disintegration of IM (arrowheads) and aberrant constriction of cell wall to form two cells (L). Scale bars (white) = 200 nm. The data are representative images from two independent experiments.
(TIF)





