Abstract
Pancreas plays an important role in maintaining the glucose homeostasis. The deterioration of β-cells in the pancreas is a crucial factor in the progression of diabetes mellitus; therefore, the restoration of β-cell mass and its function is of vital importance for effective therapeutic strategies. The precise mechanism for increase in functional β-cell mass is still unknown. This review focuses on the importance of certain genes which are involved in the rejuvenation of pancreas. These genes are divided according to their functions into three categories: participate either in proliferation (mitotic division of differentiated β-cells), neogenesis/transdifferentiation (development from precursor cells) or inhibition of β-cell apoptosis (programmed cell death). The rate of β-cell rejuvenation is the balance among the rates of β-cell proliferation, neogenesis and apoptosis. Understanding these genes and their pathways may lead to the discovery of new drugs, target based gene delivery and development of safer antidiabetic drugs.
Keywords: Apoptosis, pancreatic genes, proliferation, rejuvenation, transdifferentiation
Introduction
Diabetes is a major cause of health concern in the world and is growing in epidemic proportions. It is assumed that in the next ten years it will become number one disease of the world1. Type-1 diabetes mellitus (T1DM) is an autoimmune disease while type 2 is mostly a lifestyle disease. Majority of people suffer mainly due to type-2 diabetes and is responsible for the current diabetes explosion. The detection early markers for the disease and its prevention is an active area of research to develop target based novel drugs.
Dysfunctional pancreas in diabetes
Insulin, a key polypeptide hormone secreted by the pancreas, targets several tissues for the utilization of glucose and thus maintains the glucose homeostasis. Type 2 diabetes mellitus (T2DM) develops from a combination of genetic and acquired factors (such as changes in metabolic homeostasis) that impair β-cell function on one side, and tissue insulin sensitivity on the other2,3. Normally, β-cell mass can adapt to changes in metabolic homeostasis. Recurrence of these changes in metabolism creates a stress on pancreas often predating the on-set of T2DM by many years. This pancreatic stress causes β-cell mass expansion, through enhanced proliferation and neogenesis. The progression from this stress condition to a state of diabetes is inevitably associated with a decrease in the β-cell mass2,3,4. This β-cell loss arises due to an increase in β-cell apoptosis, which clearly outweighs replication and neogenesis.
The war against diabetes through the development of new drugs is an ongoing continuous process5. With the technological advancement, efforts are being made to rejuvenate the pancreatic cells or create artificial pancreas. Pancreatic rejuvenation can happen either due to proliferation of existing β-cells or differentiation of progenitor cells to β-cells6 (figure), or due to decrease in β-cell apoptosis.
Fig.
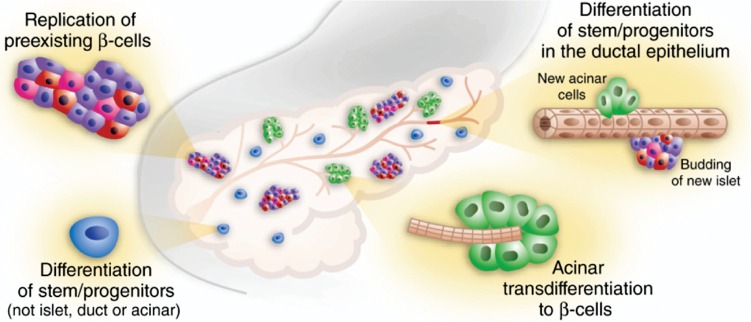
Pancreas as the source of new β-cells. New β-cells can arise from (1) Replication of pre-existing β-cells (2) Differentiaition of stem/progenitors or Transdifferentiation of acinar cells (3) Prevention of apoptosis (not shown in Figure) also contributes for regeneration of pancreas in which gene/gene products play in conjunction either with (1) or (2). All these mechanism may contribute synergistically for the regeneration of pancreas. See Tables 1, 2 and 3 for gene/gene products involved in the above processes for pancreatic rejuvenation. Reproduced with permission from Nature Publishing Group, London, UK6.
β-Cell proliferation
Islets regeneration refers to an increase in β-cell mass by proliferation and replication of existing islet cells. Several mouse studies7,8,9 here shown that β-cells do not proliferate, however, lineage tracing studies10,11,12,13,14 have confirmed that human β-cells proliferate and give rise to a population of progenitor/stem cell. Various genes and transcription factors are involved in this process viz. Reg (Regenerating islets derived proteins), Sox9, Hnf-6, NeuroD1, Neurogenin-3 and Netrin-1 (Table I). Besides these genes, certain peptides or their analogues such as glucagon like peptide-1/exendin-4 are also involved in islet regeneration. These observations are confirmed by using dipeptidyl peptidase (DPP) IV inhibitor sitagliptin in mice15.
Table I.
Genes involved in beta cell proliferation

So far, five REG proteins have been reported in humans that belong to Reg gene family. Some of the members of this family have been implicated in β-cell replication and/or neogenesis as shown in in vivo studies using transgenic and knockout mice16. These also preserve the β-cell mass in autoimmune type 1 diabetes17. This Reg family of genes are expressed in both young and old mice that were subjected to partial pancreatectomy18. In isolated rat islets, Reg1 mRNA levels were significantly increased by glucose, amino acids, foetal serum or specific growth factors such as insulin, growth hormone and platelet-derived growth factors (PDGF)19. PDGF receptor signalling controls age-dependent β-cell proliferation in mouse and human pancreatic islet cells20. Disruption of RegI gene resulted in a significantly decreased rate of DNA synthesis and diminished β-cell hyperplasia in response to obesity, confirming the role of endogenous RegI in islets cell growth21. A study conducted by Huszarik et al22 showed upregulation of RegII during diabetogenic process and also after adjuvant therapy in NOD mice. While all Reg family mRNAs can be detected from total pancreas, RegII and RegIIIα genes have been detected in pancreatic islet cells as confirmed by immunofluorescence23 and RegIIIα expression was remarkably increased during pregnancy in rats24. Mice overexpressing RegIII β was resistant to streptozotocin induced diabetes mellitus25. RegIIIγ, another member of Reg family of genes is also found to be involved in regeneration of pancreas. REG III protein was found to be expressed only in regenerating islets and not in normal rat pancreas26 and its gene expression level induced 10-100 folds on day 3 of pancreatectomy27. These data suggest that there is a strong link between Reg gene family and rejuvenation of pancreatic islets.
Transcription factors in β-cell proliferation
Certain transcription factors (Sox9, Hnf-6, Ngn-3 and NeuroD1) are also found to be involved in the proliferation of β-cells. SOX9 is the first specific marker and maintenance factor of multi-potential progenitors during pancreatic organogenesis. SOX9, in the embryonic pancreas stimulates proliferation and prevents apoptosis of pluripotent progenitor cells. It controls pancreatic progenitor cell maintenance by modulating Notch signal transduction. The phenotypic alterations in the Sox9-deficient pancreas shows a striking resemblance to the pancreatic defects associated with mutations in components of the Notch signalling pathway, thus establishing a possible link between Sox9 and the notch signal transduction pathway for stem cell maintenance28. The hepatocyte nuclear factor 6 (Hnf-6), homeodomain-containing transcription factor, is an important regulator of endocrine development. HNF6 is expressed in early pancreatogenesis in all endodermally derived cells, but is not detected in differentiated endocrine cells at late-gestation29. Hnf-6 null mice embryos showed impaired endocrine differentiation and perturbed duct morphogenesis during embryogenesis30. In addition to defects in endocrine development, Hnf-6 null embryos showed defects in duct development31. Loss of Hnf-6 from Ngn-3 expressing cells did not affect β-cell function or glucose homeostasis suggesting that Hnf-6 is dispensable for later events of endocrine differentiation. These data confirm that HNF6 has both early and late functions in the developing pancreas and is essential for maintenance of Ngn-3 expression and proper pancreatic duct morphology32. NeuroD1, a downstream target of Ngn-3, carries on the endocrine differentiation programme initiated by Ngn3 and participates in the maintenance of the differentiated phenotype of the mature islet cells33. During pancreatic endocrine development, Ngn-3 acts early to determine endocrine cell fate, while NeuroD1 directs endocrine cell differentiation34. At early stage of life, mice lacking a functional NeuroD1 (also called as BETA2) gene exhibit a striking reduction in the number of insulin-producing β-cells and failed to develop mature islets with a marked hyperglycaemia. Attempts to rescue the diabetic phenotype by administration of insulin were unsuccessful, suggesting that the mutant animals were unable to respond to insulin, have become insulin resistant, or perhaps contained additional defects35. Thus BETA-2 is required for the expansion of the pancreatic β-cells population, as well as other islet cell types which are involved in the development of endocrine cells into islets of Langerhans34.
Netrins are laminin-like diffusible chemotactic proteins involved in pancreatic morphogenesis and play a role in the regulation of duct-cell and foetal islet cell migration. In adult rat pancreas, Netrin-1 mRNA was practically undetectable. After duct ligation, its expression was very low in the head part of the pancreas whereas it was strongly upregulated in the tail part at 3rd, 5th and 7th day of post-ligation with the maximum expression on day 536. Netrin-1 mRNA was found to be expressed by islet cells and exocrine cells with ductal characteristics. These observations suggest that Netrin-1 plays a role in pancreatic morphogenesis, both prenatally and in the regenerating adult rat pancreas.
Transdifferentiation of pancreas
Islet neogenesis specifically refers to an increase in β-cell mass via transdifferentiation of adult pancreatic stem cells, putatively found in the ductal epithelium or acinar tissue. Trans-differentiation involves in the conversion of alpha or delta cells of the pancreas into insulin producing β-cells. Various genes/proteins contribute to this process. These include INGAP, Gastrin, MafA, Pdx-1, Foxa2, Nkx2.2, Nkx6.1, Pax4, etc. (Table II).
Table II.
Genes involved in pancreatic neogenesis

INGAP (islets neogenesis associated protein) is a member of the C-lectin protein family that serves as the initiator of a cascade of events that culminates in islet neogenesis and can reverse diabetes in streptozotocin-induced diabetic C57BL/6J mice37. These studies were further confirmed in beagle dogs38. There was also a significant increase in insulin gene expression in the INGAP treated animals. INGAP is also found in human pancreas during pathological states involving islet neogenesis which further suggests that INGAP is of primary importance in the process of islet neogenesis39. Gastrin, a classical gut hormone secreted by G cells in the stomach lining is found to stimulate pancreatic β-cell neogenesis. Intravenous infusion of gastrin into the ligated duct cells, resulted in a doubling of the β-cell mass in rats40, due to high expression of gastrin/cholecystokinin (CCK) B receptors in duct ligated cells41. These observations were confirmed using EGF plus gastrin combinatorial therapy, which showed an increase in insulin positive cells in human islet42. In another study using GLP-1 and gastrin in NOD mice, there was a significant reduction in blood glucose due to increase in pancreatic β-cell mass and insulin content43. MafA a member of Maf subfamily of leucine zippers is capable of strongly activating the insulin promoter. Maf factors play important roles in cellular differentiation of exocrine cells to β-cells44. MafA is restricted to β-cells and known to be important in the embryonic development of pancreas45. It has been observed that MafA expression is decreased during the diabetic condition46.
Transcription factors in pancreatic neogenesis
There are several transcription factors involved both in neogenesis and replication. However, it is not clear whether these work alone or in combination with other transcription factors in a coordinated manner. The list of the transcription factors and their role in transdifferentiation is summarized in Table II. Pancreatic duodenal homeobox-1 (Pdx-1), a homeobox transcription factor, besides being involved as a regulator of pancreatic development (the differentiation and gene expression in the β-cell)47, Pdx1 also turns out to be a major player in the maintenance of an adequate pool of healthy β-cells in adults48,49. It maintains the homeostasis between β-cell neogenesis and apoptosis. In mice with a 50 per cent reduction in Pdx1, the 0isolated islets showed more susceptibility to apoptosis at basal glucose concentrations along with impaired ability to maintain β-cell mass with age. Its expression is shown to be down-regulated during hyperglycaemic condition50. The survival functions of Pdx1 may be mediated by insulin/IGF signaling acting through the forkhead transcription factor Foxo1 (forkhead/winged helix transcription factor). Foxa2 (formerly known as Hnf-3) is a key regulator of foregut development that plays an essential role in the cell type-specific transcription of the Pdx-1 gene in the pancreas51. On deletion of Foxa2 in mice, there is a significant down- regulation of Pdx-1 mRNA52. This shows that Foxa2 is an essential upstream factor that regulates Pdx-1 mRNA levels in β-cells.
Nkx2.2 is another essential pancreatic transcription factor that affects the expression of ghrelin during pancreatic development53. Nkx2.2-null mice lose all β-cells and the majority of the α-cells; and the islet is predominantly populated by gherlin expressing cells. The discovery that ghrelin cells often replace the other endocrine populations in the pancreas54 suggests a lineage relationship between the ghrelin producing epsilon cells and other hormone-producing populations55. In another study, disruption of the Nkx2.2 gene has shown to lead to the accumulation of incomplete differentiated β-cells that express some β-cell markers, but not insulin56. This illustrates the role of Nkx2.2 in pancreatic endocrine cell differentiation. The phenotypic effects of Nkx2.2 mutant mice may in part result from the loss of other homeodomain transcription factor Nkx6.1. In pancreas, Nkx6.1 also follows an expression pattern similar to that of Nkx2.2; but restricted to the β-cells alone57. Deletion of the Nkx6.1 0gene in mice caused a marked reduction of β-cells. In the same study, with the double knockout animal model (Nkx2.2/Nkx6.1) showed the same phenotype as of Nkx2.2 single mutant58. This shows that Nkx6.1 functions downstream of Nkx2.2 in pancreatic development. The paired homeobox transcription factor Pax4 appears to be a strong candidate for specifying β-cell lineages. Mice deficient in Pax4 fail to develop beta and delta cells within the pancreas suggesting that Pax4 expression commits selected endocrine precursors towards the beta and delta cell lineage59. The Pax4 genes are expressed during the embryonic pancreatic development, but later on these are restricted to β-cells alone. Inactivation of Pax4 in mice results in the improper maturation of alpha- and β-cells60. This indicates the role of PAX4 0in the maintenance of progenitors and in maturation of β-cells.
Inhibition of β-cell apoptosis
Under normal circumstances, apoptosis is highly regulated to maintain normal physiological function of the cells. In diabetes, during excess stress, the pancreatic cells not only undergo apoptosis but also become necrotic and are unable to secrete insulin. A study conducted by Butler et al61 indicated that increased apoptosis rather than decreased neogenesis/proliferation might be the main mechanism leading to reduced β-cell mass in T2DM. Thus, decrease in the rate of apoptosis itself, may increase the β-cell mass via proliferation. Several genes/proteins are involved in pancreatic apoptosis and their functions are summarized in Table III.
Table III.
Genes involved in inhibition of beta cell apoptosis

Perforin (pore forming protein) is a cytolytic protein, initiating apoptosis by inducing minimal cell membrane damage while effectively releasing granzymes from the endosomal compartment into the cytosol62. Of the granzyme family, granzyme A and B are the most common in human and mouse63. Granzyme A induces single-strand DNA breaks, while granzyme B cleaves specific substrates including caspases and the pro-apoptotic molecule Bid64. Activated Bid is targeted to the mitochondria where it sequesters anti-apoptotic members of the Bcl-2 family, allowing the oligomerization of Bax and/or Bak which mediates loss of mitochondrial outer membrane potential, release of cytochrome C and causes irreversible apoptosis65. During hyperglycaemic condition its expression is elevated in β-cells, thus increasing the rate of apoptosis51. Deficiency in Bid prevents β-cells from undergoing mitochondrial apoptotic pathway66,67. Caspase-3 has been extensively studied in various tissues due to its role as the principal executioner of apoptosis68. As such, Caspase-3 is an attractive target to inhibit apoptosis in diseased conditions including diabetes69. Caspase-3 null (Casp3-/-) mice were found to be protected from developing diabetes in a multiple-low-dose streptozotocin autoimmune diabetic model70. This illustrates the importance of Caspase-3 in β-cell death and its activity is found to be increased during the diabetic conditions. An attractive regulator of β-cell replication and survival after birth is Survivin. This protein blocks the functions of Caspases in the mitochondria-dependent cell death pathway, protecting cells from apoptosis71. Deletion of Survivin within the mice endocrine pancreas results in diabetes manifested by hyperglycaemia and polyuria. Exogenous expression of Survivin in streptozotocin -induced diabetic model protects the β-cells from apoptosis72. Thus, it may play a role in the replication and/or survival of matured β-cells73. Surprisingly it is modestly upregulated in diabetic patients74 which may help to assuage diabetes.
Beside survivin, there are other proteins present in pancreas which prevent apoptosis. BCL-xL, an anti-apoptotic protein coded by the “survival gene”, is involved in the inhibition of apoptosis. Marked overexpression of Bcl-xL, resulted in a severe defect in insulin secretion and hyperglycaemia in transgenic mice75. Under conditions of stress, β-cells require BCL-xL to maintain their survival in vivo48. Myc is a potent inducer of both β-cell proliferation and apoptosis in vitro76. Myc sensitizes cells to a variety of apoptotic triggers rather than directly inducing apoptosis by itself77. Sustained Myc activation leads to initial hyperplasia and increased apoptosis later on, suggesting that apoptosis ultimately predominates over proliferation78. In chronic hyperglycemia, an increase in the expression of Myc was observed in pancreas79. A thioredoxin-interacting protein (TXNIP), a regulatory protein, is involved in the inhibition of thioredoxin and thereby modulates the cellular redox state and promotes oxidative stress80. Its overexpression in β-cells is found to induce apoptosis81, and the process involves the activation of intrinsic mitochondrial pathway, while the Endoplasmic reticulum (ER)-mediated cell death remains unaffected. It was demonstrated that their mRNA expression levels are elevated during the diabetic conditions82. In a recent study, huntingtin-interacting protein 14 (HIP-14) is found to posses anti-apoptotic property of β-cells. This also plays an important role in glucose-stimulated insulin secretion83. Bace2 (Beta site amyloid precursor protein cleaving enzyme 2), a proteolytic enzyme acts as a negative regulator of β-cell mass via inhibiting Tmem27 expression. Overexpression of Tmem27 or ablation of Bace2 leads to an increased β-cell mass by inhibiting apoptosis84.
Future prespective
Several lifestyle diseases such as T2DM, obesity are increasing significantly. It has been predicted that in near future diabetes will overtake all the current infectious and non-infectious diseases. In spite of rapid progress in our understanding regarding the pathophysiology of diabetes, challenges remain high due to increased complexity of the disease contributed by genetic and environmental factors. In future, there need to be more emphasis on the prognosis and better treatment for diabetes associated diseases, and on the discovery of reliable biomarker(s) for its early detection. Development of new technologies i.e. rejuvenation of the pancreas by small molecules or gene targeted drug delivery, pancreatic transplantation, stem cell therapy and creating artificial pancreas, etc. will be the mainstay of the diabetes research. Thus, it becomes essential to have better understanding of the various pathways at the molecular level that are involved in pancreatic islet cell rejuvenation.
References
- 1.Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract. 2010;87:4–14. doi: 10.1016/j.diabres.2009.10.007. [DOI] [PubMed] [Google Scholar]
- 2.Kahn SE. The relative contributions of insulin resistance and β-cell dysfunction to the pathophysiology of Type 2 diabetes. Diabetologia. 2003;46:3–19. doi: 10.1007/s00125-002-1009-0. [DOI] [PubMed] [Google Scholar]
- 3.Farrannini E, Mari A. β-cell function and its relation to insulin action in humans: a critical appraisal. Diabetologia. 2004;47:943–56. doi: 10.1007/s00125-004-1381-z. [DOI] [PubMed] [Google Scholar]
- 4.Buchanan TA. Pancreatic β-cell loss and preservation in Type 2 diabetes. Clini Ther. 2003;25:32–46. doi: 10.1016/s0149-2918(03)80241-2. [DOI] [PubMed] [Google Scholar]
- 5.Balasubramanyam M, Mohan V. Orally active insulin mimics: where do we stand now? J Biosci. 2001;26:383–90. doi: 10.1007/BF02703748. [DOI] [PubMed] [Google Scholar]
- 6.Bonner-Weir S, Weir GC. New source of pancreatic β-cells. Nat Biotechnol. 2005;23:857–61. doi: 10.1038/nbt1115. [DOI] [PubMed] [Google Scholar]
- 7.Chase LG, Ulloa-Montoya F, Kidder BL, Verfaillie CM. Islet-derived fibroblast-like cells are not derived via epithelial-mesenchymal transition from Pdx-1 or Insulin-Positive cells. Diabetes. 2007;56:3–7. doi: 10.2337/db06-1165. [DOI] [PubMed] [Google Scholar]
- 8.Atouf F, Park CH, Pechhold K, Malancha Ta, Choi Y, Lumelsky NL. No evidence for mouse pancreatic β-cell epithelial-mesenchymal transition in vitro. Diabetes. 2007;56:699–702. doi: 10.2337/db06-1446. [DOI] [PubMed] [Google Scholar]
- 9.Morton RA, Geras-Raaka E, Wilson LM, Raaka BM, Gershengorn MC. Endocrine precursor cells from mouse islets are not generated by epithelial-to-mesenchymal transition of mature β-cells. Mol Cell Endocrinol. 2007;270:87–93. doi: 10.1016/j.mce.2007.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Joglekar MC, Hardikar AA. Epithelial-to-mesenchymal transition in pancreatic islet β-cells. Cell Cycle. 2010;9:4077–9. doi: 10.4161/cc.9.20.13590. [DOI] [PubMed] [Google Scholar]
- 11.Joglekar MV, Joglekar VM, Joglekar VM, Hardikarc AA. Human fetal pancreatic insulin-producing cells proliferate I. J Endocrinol. 2009;201:27–36. doi: 10.1677/JOE-08-0497. [DOI] [PubMed] [Google Scholar]
- 12.Joglekar MV, Patil D, Joglekar VM, Rao GV, Reddy DN, Mitnala S, et al. The miR-30 family microRNAs confer epithelial phenotype to human pancreatic cells. Islets. 2009;1:137–47. doi: 10.4161/isl.1.2.9578. [DOI] [PubMed] [Google Scholar]
- 13.Russ HA, Ravassard P, Kerr-Conte J, Pattou F, Efrat S. Epithelial-mesenchymal transition in cells expanded in vitro from lineage-traced adult human pancreatic β-cells. Plos One. 2009;4:1–8. doi: 10.1371/journal.pone.0006417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bar Y, Russ HA, Knoller S, Ouziel-Yahalom L, Efrat S. HES-1 is involved in adaptation of adult human β-cells to proliferation in vitro. Diabetes. 2008;57:2413–20. doi: 10.2337/db07-1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mu J, Woods J, Zhou YP, Roy RS, Li Z, Zycband E, et al. Chronic inhibition of dipeptidyl peptidase-4 with a sitagliptin analog preserves pancreatic β-cell mass and function in a rodent model of type 2 diabetes. Diabetes. 2006;55:1695–74. doi: 10.2337/db05-1602. [DOI] [PubMed] [Google Scholar]
- 16.Liu J, Cui W, Li B, Lu Y. Possible roles of Reg family of proteins in pancreatic islet cell growth. Endoc Metab Immune Disord-Drug Targets. 2008;8:1–10. doi: 10.2174/187153008783928361. [DOI] [PubMed] [Google Scholar]
- 17.Singh B, Nikoopour E, Huszarik K, Elliott JF, Jevnikar AM. Immunomodulation and regeneration of islet β-cells by cytokines in autoimmune type 1 diabetes. J Interferon Cytokine Res. 2011;31:711–9. doi: 10.1089/jir.2011.0025. [DOI] [PubMed] [Google Scholar]
- 18.Rankin MM, Kushner JA. Aging induces a distinct gene expression program in mouse islets. Islets. 2010;2:345–52. doi: 10.4161/isl.2.6.13376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Francis PJ, Southgate JL, Wilkin TJ, Bone AJ. Expression of an islet regenerating (reg) gene in isolated rat islets: effects of nutrient and non-nutrient growth factors. Diabetologia. 1992;35:238–42. doi: 10.1007/BF00400923. [DOI] [PubMed] [Google Scholar]
- 20.Chen H, Gu X, Liu Y, Wang J, Wirt SE, Bottino R, et al. PDGF signalling controls age-dependent proliferation in pancreatic β-cells. Nature. 2011;478:349–57. doi: 10.1038/nature10502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Unno M, Nata K, Noguchi N, Narushima Y, Akiyama T, Ikeda T, et al. Production and characterization of Reg knockout mice:reduced proliferation of pancreatic β-cells in Reg knockout mice. Diabetes. 2002;51:S478–83. doi: 10.2337/diabetes.51.2007.s478. [DOI] [PubMed] [Google Scholar]
- 22.Huszarik K, Wright B, Keller C, Nikoopour E, Krougly O, Lee-Chan E, et al. Adjuvant immunotherapy increases β-cell regenerative factor Reg2 in the pancreas of diabetic mice. J Immunol. 2010;185:5120–9. doi: 10.4049/jimmunol.1001596. [DOI] [PubMed] [Google Scholar]
- 23.Gurr W, Shaw M, Li Y, Sherwin R. Reg II is a β-cell protein and autoantigen in diabetes of NOD mice. Diabetes. 2007;56:34–40. doi: 10.2337/db06-0669. [DOI] [PubMed] [Google Scholar]
- 24.Xue Y, Liu C, Xu Y, Yuan Q, Xu K, Mao X, et al. Study on pancreatic islet adaptation and gene expression during pregnancy in rats. Endocrine. 2010;37:83–97. doi: 10.1007/s12020-009-9273-0. [DOI] [PubMed] [Google Scholar]
- 25.Xiong X, Wang X, Li B, Chowdhury S, Lu Y, Srikant CB, et al. Pancreatic islet-specific overexpression of Reg3β protein induced the expression of pro-islet genes and protected the mice against streptozotocin induced diabetes mellitus. Am J Physiol Endocrinol Metab. 2011;300:E669–80. doi: 10.1152/ajpendo.00600.2010. [DOI] [PubMed] [Google Scholar]
- 26.Terazono K, Yamamoto H, Takasawa S, Shiga K, Yonemura Y, Tochino Y, et al. A novel gene activated in regenerating islets. J Biol Chem. 1988;263:2111–4. [PubMed] [Google Scholar]
- 27.Joglekar MV, Parekh VS, Mehta S, Bhonde RR, Hardikar AA. Micro RNA profiling of developing and regenerating pancreas reveal post-transcriptional regulation of neurogenin3. Devel Biol. 2007;311:603–12. doi: 10.1016/j.ydbio.2007.09.008. [DOI] [PubMed] [Google Scholar]
- 28.Seymour PA, Freude KK, Tran MN, Mayes EE, Jensen J, Kist R, et al. SOX9 is required for maintenance of the pancreatic progenitor cell pool. Proc Natl Acad Sci USA. 2006;104:1865–70. doi: 10.1073/pnas.0609217104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Landry C, Clotman F, Hioki T, Oda H, Picard JJ, Lemaigre FP, et al. HNF-6 is expressed in endoderm derivatives and nervous system of the mouse embryo and participates to the cross-regulatory network of liver-enriched transcription factors. Devel Biol. 1997;192:247–57. doi: 10.1006/dbio.1997.8757. [DOI] [PubMed] [Google Scholar]
- 30.Jacquemin P, Durviaux SM, Jensen J, Godfraind C, Gradwohl G, Guillemot F, et al. Transcription factor hepatocyte nuclear factor 6 regulates pancreatic endocrine cell differentiation and controls expression of the proendocrine gene ngn3. Mol Cell Biol. 2000;20:4445–54. doi: 10.1128/mcb.20.12.4445-4454.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Pierreux CE, Poll AV, Kemp CR, Clotman F, Maestro MA, Cordi S, et al. The transcription factor hepatocyte nuclear factor-6 controls the development of pancreatic ducts in the mouse. Gastroentrology. 2006;130:532–41. doi: 10.1053/j.gastro.2005.12.005. [DOI] [PubMed] [Google Scholar]
- 32.Zhang H, Ables ET, Pope CF, Washington MK, Hipkens S, Means AL, et al. Multiple, temporal-specific roles for HNF6 in pancreatic endocrine and ductal differentiation. Mech Dev. 2009;126:958–73. doi: 10.1016/j.mod.2009.09.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jensen J. Gene regulatory factors in pancreatic development. Devel Dyna. 2004;229:176–200. doi: 10.1002/dvdy.10460. [DOI] [PubMed] [Google Scholar]
- 34.Gasa R, Mrejen C, Lynn FC, Cox P, Sanchez L, Yang KY, et al. Induction of pancreatic islet cell differentiation by the neurogenin-neuroD cascade. Differentiation. 2008;76:381–91. doi: 10.1111/j.1432-0436.2007.00228.x. [DOI] [PubMed] [Google Scholar]
- 35.Naya FJ, Huang H, Qiu Y, Mutoh H, Demayo FJ, Leiter AB, et al. Diabetes, defective pancreatic morphogenesis, and abnormal enteroendocrine differentiation in BETA2/neurod-deficient mice. Genes Devel. 1997;11:2323–34. doi: 10.1101/gad.11.18.2323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.De Breuck S, Lardon J, Rooman I, Bouwens L. Netrin-1 expression in fetal and regenerating rat pancreas and its effect on the migration of human pancreatic duct and porcine islet precursor cells. Diabetologia. 2003;46:926–33. doi: 10.1007/s00125-003-1125-5. [DOI] [PubMed] [Google Scholar]
- 37.Rosenberg L, Lipsett M, Yoon JW, Prentki M, Wang R, Jun HS, et al. A pentadecapeptide fragment of islet neogenesis-associated protein increases β-cell mass and reverses diabetes in C57BL/6J mice. Ann Surg. 2004;240:875–84. doi: 10.1097/01.sla.0000143270.99191.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pittenger GL, Taylor-Fishwick DA, Johns RH, Burcus N, Kosuri S, Vinik AI. Intramuscular injection of islet neogenesis-associated protein peptide stimulates pancreatic islet neogenesis in healthy dogs. Pancrea. 2007;34:103–11. doi: 10.1097/01.mpa.0000240609.56806.43. [DOI] [PubMed] [Google Scholar]
- 39.Pittenger GL, Taylor-Fishwick D, Vinik AI. The role of islet neogeneis-associated protein (INGAP) in pancreatic islet neogenesis. Curr Protein Pept Sci. 2009;10:37–45. doi: 10.2174/138920309787315211. [DOI] [PubMed] [Google Scholar]
- 40.Rooman I, Lardon J, Bouwens L. Gastrin stimulates β-cell neogenesis and increases islet mass from transdifferentiated but not from normal exocrine pancreas tissue. Diabetes. 2002;51:686–90. doi: 10.2337/diabetes.51.3.686. [DOI] [PubMed] [Google Scholar]
- 41.Rooman I, Lardon J, Flamez D, Schuit F, Bouwens L. Mitogenic effect of gastrin and expression of gastrin receptors in duct-like cells of rat pancreas. Gastroenterology. 2001;121:940–9. doi: 10.1053/gast.2001.27998. [DOI] [PubMed] [Google Scholar]
- 42.Suarez-Pinzon WL, Lakey JR, Brand SJ, Rabinovitch A. Combination therapy with epidermal growth factor and gastrin induces neogenesis of human islet β-cells from pancreatic duct cells and an increase in functional β-cells mass. J Clin Endocrinol Metab. 2005;90:3401–9. doi: 10.1210/jc.2004-0761. [DOI] [PubMed] [Google Scholar]
- 43.Suarez-Pinzon WL, Power RF, Yan Y, Wasserfall C, Atkinson M, Rabinovitch A. Combination therapy with glucagon-like peptide-1 and gastrin restores normoglycemia in diabetic NOD mice. Diabetes. 2008;57:3281–8. doi: 10.2337/db08-0688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhou Q, Brown J, Kanarek A, Rajagopal J, Melton DA. In vivo reprogramming of adult pancreatic exocrine cells to β-cells. Nature. 2008;455:627–33. doi: 10.1038/nature07314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Olbrot M, Rud J, Moss LG, Sharma A. Identification of β-cell-specific insulin gene transcription factor RIPE3b1 as mammalian MafA. Proc Natl Acad Sci USA. 2002;99:6737–42. doi: 10.1073/pnas.102168499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Matsuoka T, Kaneto H, Miyatsuka T, Yamamoto T, Yamamoto K, Kato K, et al. Regulation of MafA expression in pancreatic β-cells in db/db mice with diabetes. Diabetes. 2010;59:1709–20. doi: 10.2337/db08-0693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Taniguchi H, Yamato E, Tashiro F, Ikegami H, Ogihara T, Miyazaki J. β-cell neogenesis induced by adenovirus-mediated gene delivery of transcription factor pdx-1 into mouse pancreas. Gene Therapy. 2003;10:15–23. doi: 10.1038/sj.gt.3301846. [DOI] [PubMed] [Google Scholar]
- 48.Johnson JD, Ahmed NT, Luciani DS, Han Z, Tran H, Fujita J, et al. Increased islet apoptosis in Pdx1+/- mice. J Clin Invest. 2003;111:1147–60. doi: 10.1172/JCI16537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bernardo AS, Hay CW, Docherty K. Pancreatic transcription factors and their role in the birth, life and survival of the pancreatic β-cell. Mol Cell Endocrinol. 2008;294:1–9. doi: 10.1016/j.mce.2008.07.006. [DOI] [PubMed] [Google Scholar]
- 50.Robertson RP, Harmon JS. Diabetes, glucose toxicity, and oxidative stress: A case of double jeopardy for the pancreatic islet β-cell. Free Radic Biol Med. 2006;41:177–84. doi: 10.1016/j.freeradbiomed.2005.04.030. [DOI] [PubMed] [Google Scholar]
- 51.Sharma S, Jhala US, Johnson T, Ferreri K, Leonard J, Montminy M. Hormonal regulation of an islet-specific enhancer in the pancreatic homeobox gene STF-1. Mol Cell Biol. 1997;17:2598–604. doi: 10.1128/mcb.17.5.2598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lee CS, Sund NJ, Vatamaniuk MZ, Matschinsky FM, Stoffers DA, Kaestner KH. Foxa2 controls Pdx1 gene expression in pancreatic β-cells in vivo. Diabetes. 2002;51:2546–51. doi: 10.2337/diabetes.51.8.2546. [DOI] [PubMed] [Google Scholar]
- 53.Hill JT, Chao CS, Anderson KR, Kaufman F, Johnson CW, Sussel L. Nkx2.2 activates the ghrelin promoter in pancreatic islet cells. Mol Endocrinol. 2010;24:381–90. doi: 10.1210/me.2009-0360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Prado CL, Pugh-Bernard AE, Elghazi L, Sosa-Pineda B, Sussel L. Ghrelin cells replace insulin-producing β-cell in two mouse models of pancreas development. Proc Natl Acad Sci USA. 2004;101:2924–9. doi: 10.1073/pnas.0308604100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hill JT, Mastracci TL, Vinton C, Doyle ML, Anderson KR, Loomis ZL, et al. Ghrelin is dispensable for embryonic pancreatic islet development and differentiation. Regul Pept. 2009;157:51–6. doi: 10.1016/j.regpep.2009.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Qiua M, Shimamurab K, Sussel L, Chen S, Rubenstein J. Control of anteroposterior and dorsoventral domains of Nkx-6.1 gene expression relative to other Nkx genes during vertebrate CNS development. Mech Dev. 1998;72:77–88. doi: 10.1016/s0925-4773(98)00018-5. [DOI] [PubMed] [Google Scholar]
- 57.Oster A, Jensen J, Serup P, Galante P, Madsen OD, Larsson LI. Rat endocrine pancreatic development in relation to two homeobox gene products (Pdx-1 and Nkx 6) J Histochem Cytochem. 1998;46:707–15. doi: 10.1177/002215549804600602. [DOI] [PubMed] [Google Scholar]
- 58.Sander M, Sussel L, Conners J, Scheel D, Kalamaras J, Cruz FD, et al. Homeobox gene Nkx6.1 lies downstream of Nkx2.2 in the major pathway of β-cell formation in the pancreas. Development. 2000;127:5533–40. doi: 10.1242/dev.127.24.5533. [DOI] [PubMed] [Google Scholar]
- 59.Ritz-Laser B, Estreicher A, Gauthier BR, Mamin A, Edlund H, Philippe J. The pancreatic β-cell specific transcription factor Pax-4 inhibits glucagon gene expression through Pax-6. Diabetologia. 2002;45:97–107. doi: 10.1007/s125-002-8249-9. [DOI] [PubMed] [Google Scholar]
- 60.Sosa-Pineda B, Chowdry K, Torres M, Oliver G, Gruss P. The Pax-4 gene is essential for differentiation of insulin-producing β-cells in mammalian pancreas. Nature. 1997;386:399–402. doi: 10.1038/386399a0. [DOI] [PubMed] [Google Scholar]
- 61.Butler AE, Janson J, Bonner-Weir S, Ritzel R, Rizza RA, Butler PC. β-cell deficit and increased β-cell apoptosis in humans with type 2 diabetes. Diabetes. 2003;52:102–10. doi: 10.2337/diabetes.52.1.102. [DOI] [PubMed] [Google Scholar]
- 62.Browne KA, Blink E, Sutton VR, Froelich CJ, Jans DA, Trapani JA. Cytosolic delivery of Granzyme B by Bacterial Toxins: Evidence that endosomal disruption, in addition to transmembrane pore formation, is an important function of perforin. Mol Cell Biol. 1999;19:8604–15. doi: 10.1128/mcb.19.12.8604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Chowdhury D, Lieberman J. Death by thousand cuts: Granzyme pathways of programmed cell death. Annu Rev Immunol. 2008;26:389–420. doi: 10.1146/annurev.immunol.26.021607.090404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sutton VR, Davis JE, Cancilla M, Johnstone RW, Ruefli AA, Karin Sedelies, et al. Browne and Joseph A. Trapani. Initiation of apoptosis by granzyme B requires direct cleavage of Bid, but not direct Granzyme B-mediated caspase activation. J Exp Med. 2000;192:1403–13. doi: 10.1084/jem.192.10.1403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Strasser A. The role of BH3-only proteins in the immune system. Nat Rev Immunol. 2005;5:189–200. doi: 10.1038/nri1568. [DOI] [PubMed] [Google Scholar]
- 66.McKenzie MD, Carrington EM, Kaufmann T, Strasser A, Huang DCS, Kay TWH, et al. Proapoptotic BH3-only protein Bid is essential for death receptor-induced apoptosis of pancreatic β-cells. Diabetes. 2008;57:1284–92. doi: 10.2337/db07-1692. [DOI] [PubMed] [Google Scholar]
- 67.Estella E, McKenzie MD, Catterall T, Sutton VR, Bird PI, Trapani JA, et al. Granzyme B-mediated death of pancreatic β-cells requires the proapoptotic BH3-only molecule Bid. Diabetes. 2006;55:2212–9. doi: 10.2337/db06-0129. [DOI] [PubMed] [Google Scholar]
- 68.Fernandes-Alnemri T, Litwack G, Alnemri ES. CPP32, a novel human apoptotic protein with homology to Caenorhabditis elegans cell death protein Ced-3 and mammalian interleukin-l β-converting enzyme. J Biol Chem. 1994;269:30761–4. [PubMed] [Google Scholar]
- 69.Choi D, Woo M. Executioners of apoptosis in pancreatic β-cells: not just for cell death. Am J Physiol Endocrinol Metab. 2010;298:E735–41. doi: 10.1152/ajpendo.00696.2009. [DOI] [PubMed] [Google Scholar]
- 70.Liadis N, Murakami K, Eweida M, Elford AR, Sheu L, Gaisano HY, et al. Caspase-3-dependent β-cell apoptosis in the initiation of autoimmune diabetes mellitus. Mol Cell Biol. 2005;25:3620–9. doi: 10.1128/MCB.25.9.3620-3629.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Schimmer AD. Inhibitor of apoptosis proteins: translating basic knowledge into clinical practice. Cancer Res. 2004;64:7183–90. doi: 10.1158/0008-5472.CAN-04-1918. [DOI] [PubMed] [Google Scholar]
- 72.Dohi T, Salz W, Costa M, Ariyan C, Basadonna GP, Altieri DC. Inhibition of apoptosis by survivin improves transplantation of pancreatic islets for treatment of diabetes in mice. EMBO Rep. 2006;7:438–43. doi: 10.1038/sj.embor.7400640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Jiang Y, Nishimura W, Devor-Henneman D, Kusewitt D, Wang H, Holloway MP, et al. Postnatal expansion of the pancreatic β-cell mass is dependent on Survivin. Diabetes. 2008;57:2718–27. doi: 10.2337/db08-0170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Marselli L, Thorne J, Dahiya S, Sgroi DC, Sharma A, Bonner-Weir S, et al. Gene expression profiles of β-cell enriched tissue obtained by laser capture microdissection from subjects with type 2 diabetes. PLos One. 2010;5:e11499. doi: 10.1371/journal.pone.0011499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yun-Ping Z, Pena JC, Roe MW, Mittal A, Levisetti M, Baldwin AC, et al. Overexpression of Bcl-xl in β-cells prevents cell death but impairs mitochondrial signal for insulin secretion. Am J Physiol Endocrinol Metab. 2000;278:E340–51. doi: 10.1152/ajpendo.2000.278.2.E340. [DOI] [PubMed] [Google Scholar]
- 76.Pelengaris S, Rudolph B, Littlewood T. Action of Myc in vivo - proliferation and apoptosis. Curr Opin Genet Dev. 2000;10:100–5. doi: 10.1016/s0959-437x(99)00046-5. [DOI] [PubMed] [Google Scholar]
- 77.Prendergast GC. Mechanisms of apoptosis by c-Myc. Oncogene. 1999;18:2967–87. doi: 10.1038/sj.onc.1202727. [DOI] [PubMed] [Google Scholar]
- 78.Laybutt DR, Weir GC, Kaneto H, Lebet J, Palmiter RD, Sharma A, et al. Overexpression of c-Myc in β-cells of transgenic mice causes proliferation and apoptosis, downregulation of insulin gene expression, and diabetes. Diabetes. 2002;51:1793–804. doi: 10.2337/diabetes.51.6.1793. [DOI] [PubMed] [Google Scholar]
- 79.Jonas JC, Laybutt DR, Steil GM, Trivedi N, Pertusa JG, Van de Casteele M, et al. High glucose stimulates early response gene c-Myc expression in rat pancreatic β-cells. J Biol Chem. 2001;276:35375–81. doi: 10.1074/jbc.M105020200. [DOI] [PubMed] [Google Scholar]
- 80.Patwari P, Higgins LJ, Chutkow WA, Yoshioka J, Lee RT. The interaction of thioredoxin with Txnip. Evidence for formation of a mixed disulfide by disulfide exchange. J Biol Chem. 2006;281:21884–91. doi: 10.1074/jbc.M600427200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Minn AH, Hafele C, Shalev A. Thioredoxin-interacting protein is stimulated by glucose through a carbohydrate response element and induces β-cell apoptosis. Endocrinology. 2005;145:2397–405. doi: 10.1210/en.2004-1378. [DOI] [PubMed] [Google Scholar]
- 82.Chen J, Saxena G, Mungrue IN, Lusis AJ, Shalev A. Thioredoxin-interacting protein: a critical link between glucose toxicity and β-cell apoptosis. Diabetes. 2008;57:938–44. doi: 10.2337/db07-0715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Berchtold LA, Størling ZM, Ortisc F, Lage K, Bang-Berthelsen C, Bergholdt R, et al. Huntingtin-interacting protein 14 is a type 1 diabetes candidate protein regulating insulin secretion and β-cell apoptosis. Proc Natl Acad Sci USA. 2011;108:E681–8. doi: 10.1073/pnas.1104384108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Esterházy D, Stützer I, Wang H, Rechsteiner MP, Beauchamp J, Döbeli H, et al. Bace2 is a β-cell enriched protease that regulates pancreatic β-cell function and mass. Cell Metabolism. 2011;14:365–77. doi: 10.1016/j.cmet.2011.06.018. [DOI] [PubMed] [Google Scholar]


