Abstract
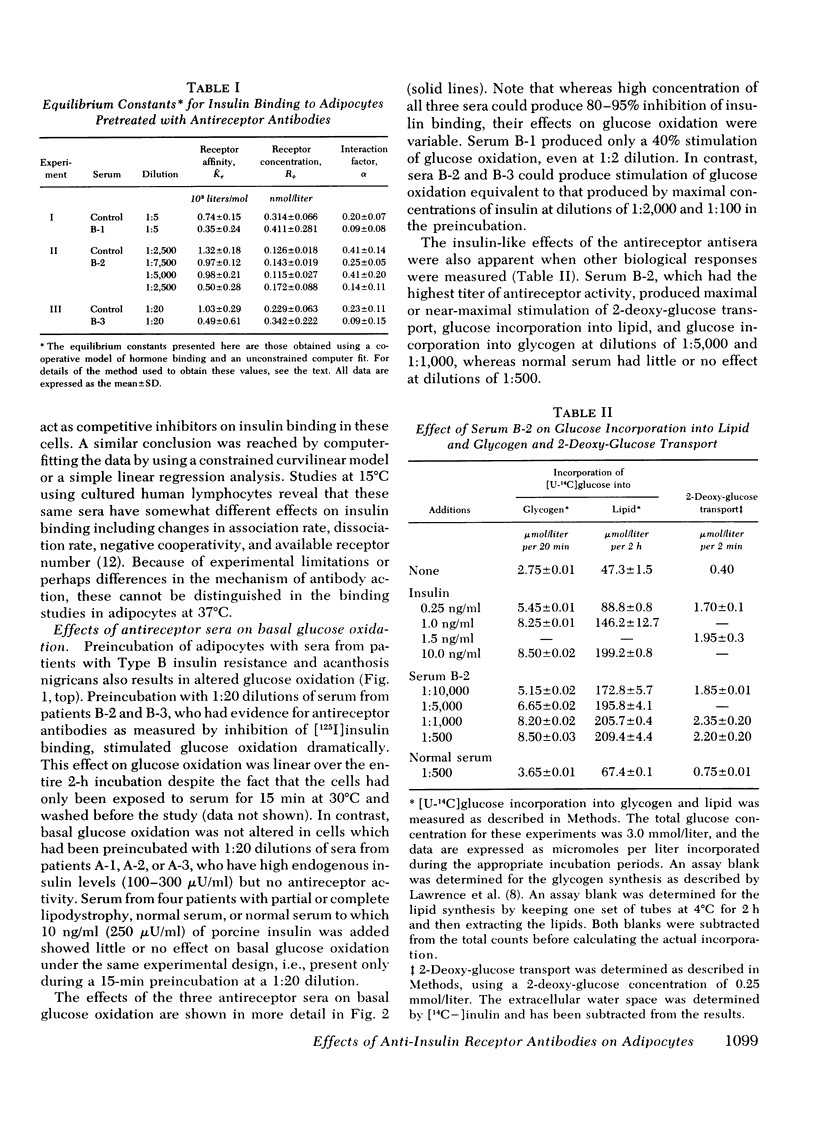
Autoantibodies to the insulin receptor have been detected in the sera of several patients with the Type B syndrome of insulin resistance and acanthosis nigricans. In this study we have used three of these sera (B-1, B-2, and B-3) as probes of the insulin receptor in isolated rat adipocytes. Preincubation of adipocytes with each of the three sera resulted in an inhibition of subsequent [125I]insulin binding. 50% inhibition of binding occurred with serum dilutions of 1:5 to 1:7,500. As in our previous studies with other tissues, Scatchard analysis of the insulin-binding data was curvilinear consistent with negative cooperativity. Computer analysis suggested that in each case the inhibition of binding was due to a decrease in receptor affinity rather than a change in available receptor number.
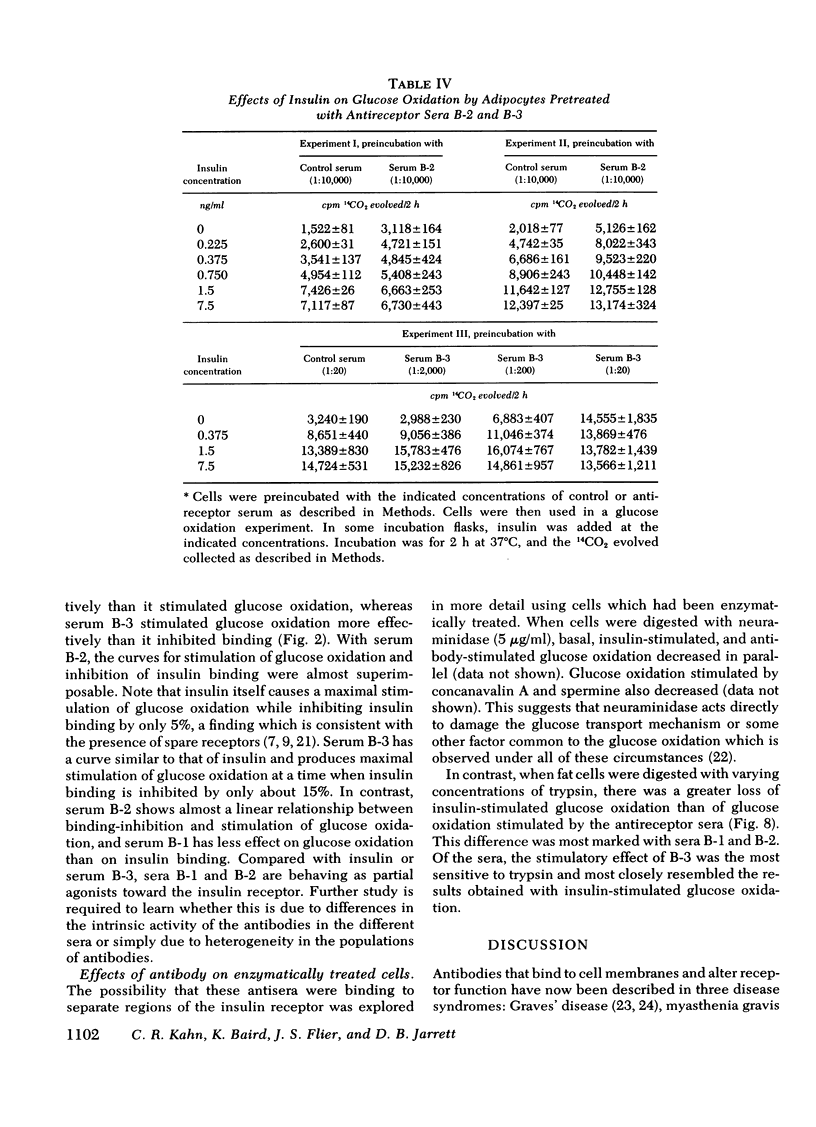
In addition to the effects on insulin binding, adipocytes pretreated with antireceptor sera also showed alterations in biological responses. All three sera produced some stimulation of basal glucose oxidation. With serum B-3, maximal stimulation of glucose oxidation occurred at a serum concentration that inhibited binding by only 10-15%, whereas with serum B-2 the dilution curves for inhibition of binding and stimulation of glucose oxidation were superimposable. Serum B-1 behaved as a partial agonist; that is, it inhibited binding more effectively than it stimulated glucose oxidation. Cells pretreated with this serum in a concentration which inhibited binding by 80% also showed a five-fold shift to the right in the dose response of insulin-stimulated glucose oxidation, whereas spermine-stimulated glucose oxidation was unaffected. Serum B-2, which contained the highest titer of antireceptor antibodies, also stimulated 2-deoxy-glucose transport, as well as glucose incorporation into lipid and glycogen.
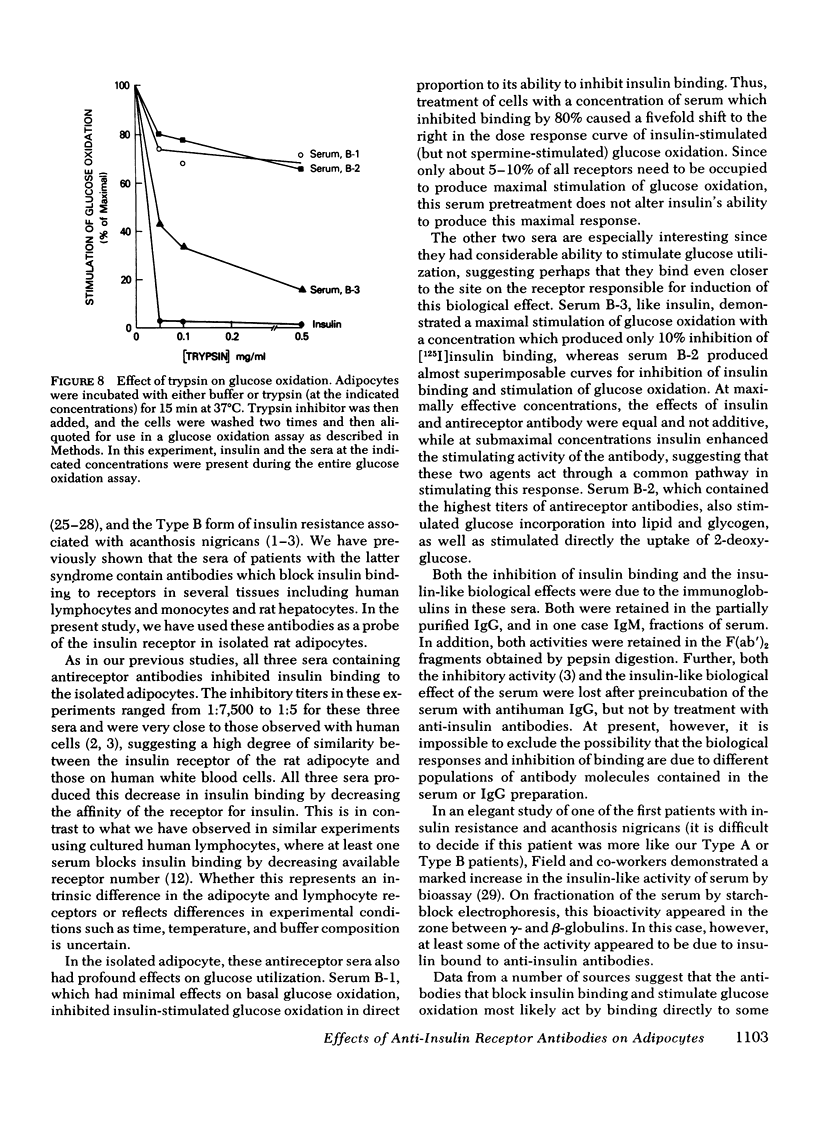
Both the ability of the serum to inhibit binding and stimulate glucose utilization were enriched in purified immunoglobulin fractions and retained in the F(ab′)2 fragment of the IgG. In addition, the bioactivity was blocked by antihuman IgG but not by anti-insulin antibodies. Enzymatic digestion of adipocytes with trypsin resulted in a complete loss of insulin-stimulated bioactivity of serum B-3, but had only minor effects on the glucose oxidation produced by serum B-1 or B-2.
These data suggest that the antibodies present in these three sera bind to different determinants on the insulin receptor. Thus, these antibodies may be useful probes of receptor structure and function.
Full text
PDF









Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aharonov A., Abramsky O., Tarrab-Hazdai R., Fuchs S. Humoral antibodies to acetylcholine receptor in patients with myasthenia gravis. Lancet. 1975 Aug 23;2(7930):340–342. doi: 10.1016/s0140-6736(75)92779-8. [DOI] [PubMed] [Google Scholar]
- Almon R. R., Andrew C. G., Appel S. H. Serum globulin in myasthenia gravis: inhibition of alpha-bungarotoxin binding to acetylcholine receptors. Science. 1974 Oct 4;186(4158):55–57. doi: 10.1126/science.186.4158.55. [DOI] [PubMed] [Google Scholar]
- Bender A. N., Ringel S. P., Engel W. K., Daniels M. P., Vogel Z. Myasthenia gravis: a serum factor blocking acetylcholine receptors of the human neuromuscular junction. Lancet. 1975 Mar 15;1(7907):607–609. doi: 10.1016/s0140-6736(75)91886-3. [DOI] [PubMed] [Google Scholar]
- Crothers D. M., Metzger H. The influence of polyvalency on the binding properties of antibodies. Immunochemistry. 1972 Mar;9(3):341–357. doi: 10.1016/0019-2791(72)90097-3. [DOI] [PubMed] [Google Scholar]
- Cuatrecasas P., Illiano G. Membrane sialic acid and the mechanism of insulin action in adipose tissue cells. Effects of digestion with neuraminidase. J Biol Chem. 1971 Aug 25;246(16):4938–4946. [PubMed] [Google Scholar]
- Cuatrecasas P., Tell G. P. Insulin-like activity of concanavalin A and wheat germ agglutinin--direct interactions with insulin receptors. Proc Natl Acad Sci U S A. 1973 Feb;70(2):485–489. doi: 10.1073/pnas.70.2.485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Czech M. P., Lynn W. S. Stimulation of glucose metabolism by lectins in isolated white fat cells. Biochim Biophys Acta. 1973 Feb 28;297(2):368–377. doi: 10.1016/0304-4165(73)90084-6. [DOI] [PubMed] [Google Scholar]
- De Meyts P., Roth J. Cooperativity in ligand binding: a new graphic analysis. Biochem Biophys Res Commun. 1975 Oct 27;66(4):1118–1126. doi: 10.1016/0006-291x(75)90473-8. [DOI] [PubMed] [Google Scholar]
- DeMeyts P., Bainco A. R., Roth J. Site-site interactions among insulin receptors. Characterization of the negative cooperativity. J Biol Chem. 1976 Apr 10;251(7):1877–1888. [PubMed] [Google Scholar]
- FIELD J. B., JOHNSON P., HERRING B. Insulin-resistant diabetes associated with increased endogenous plasma insulin followed by complete remission. J Clin Invest. 1961 Sep;40:1672–1683. doi: 10.1172/JCI104390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flier J. S., Kahn C. R., Jarrett D. B., Roth J. Autoantibodies to the insulin receptor. Effect on the insulin-receptor interaction in IM-9 lymphocytes. J Clin Invest. 1977 Oct;60(4):784–794. doi: 10.1172/JCI108832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flier J. S., Kahn C. R., Jarrett D. B., Roth J. Characterization of antibodies to the insulin receptor: a cause of insulin-resistant diabetes in man. J Clin Invest. 1976 Dec;58(6):1442–1449. doi: 10.1172/JCI108600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flier J. S., Kahn C. R., Roth J., Bar R. S. Antibodies that impair insulin receptor binding in an unusual diabetic syndrome with severe insulin resistance. Science. 1975 Oct 3;190(4209):63–65. doi: 10.1126/science.170678. [DOI] [PubMed] [Google Scholar]
- Freychet P., Kahn R., Roth J., Neville D. M., Jr Insulin interactions with liver plasma membranes. Independence of binding of the hormone and its degradation. J Biol Chem. 1972 Jun 25;247(12):3953–3961. [PubMed] [Google Scholar]
- Gammeltoft S., Gliemann J. Binding and degradation of 125I-labelled insulin by isolated rat fat cells. Biochim Biophys Acta. 1973 Aug 17;320(1):16–32. doi: 10.1016/0304-4165(73)90161-x. [DOI] [PubMed] [Google Scholar]
- Gavin J. R., 3rd, Roth J., Neville D. M., Jr, de Meyts P., Buell D. N. Insulin-dependent regulation of insulin receptor concentrations: a direct demonstration in cell culture. Proc Natl Acad Sci U S A. 1974 Jan;71(1):84–88. doi: 10.1073/pnas.71.1.84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ginsberg B. H., Kahn C. R., Roth J., De Meyts P. Insulin-induced dissociation of its receptor into subunits: possible molecular concomitant of negative cooperativity. Biochem Biophys Res Commun. 1976 Dec 20;73(4):1068–1074. doi: 10.1016/0006-291x(76)90232-1. [DOI] [PubMed] [Google Scholar]
- Jarrett D. B., Roth J., Kahn C. R., Flier J. S. Direct method for detection and characterization of cell surface receptors for insulin by means of 125I-labeled autoantibodies against the insulin receptor. Proc Natl Acad Sci U S A. 1976 Nov;73(11):4115–4119. doi: 10.1073/pnas.73.11.4115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KRISS J. P., PLESHAKOV V., CHIEN J. R. ISOLATION AND IDENTIFICATION OF THE LONG-ACTING THYROID STIMULATOR AND ITS RELATION TO HYPERTHYROIDISM AND CIRCUMSCRIBED PRETIBIAL MYXEDEMA. J Clin Endocrinol Metab. 1964 Oct;24:1005–1028. doi: 10.1210/jcem-24-10-1005. [DOI] [PubMed] [Google Scholar]
- Kahn C. R., Flier J. S., Bar R. S., Archer J. A., Gorden P., Martin M. M., Roth J. The syndromes of insulin resistance and acanthosis nigricans. Insulin-receptor disorders in man. N Engl J Med. 1976 Apr 1;294(14):739–745. doi: 10.1056/NEJM197604012941401. [DOI] [PubMed] [Google Scholar]
- Kahn C. R., Megyesi K., Bar R. S., Eastman R. C., Flier J. S. Receptors for peptide hormones. New insights into the pathophysiology of disease states in man. Ann Intern Med. 1977 Feb;86(2):205–219. doi: 10.7326/0003-4819-86-2-205. [DOI] [PubMed] [Google Scholar]
- Kono T., Barham F. W. The relationship between the insulin-binding capacity of fat cells and the cellular response to insulin. Studies with intact and trypsin-treated fat cells. J Biol Chem. 1971 Oct 25;246(20):6210–6216. [PubMed] [Google Scholar]
- Lawrence J. C., Jr, Guinovart J. J., Larner J. Activation of rat adipocyte glycogen synthase by insulins. J Biol Chem. 1977 Jan 25;252(2):444–450. [PubMed] [Google Scholar]
- Lindstrom J. M., Seybold M. E., Lennon V. A., Whittingham S., Duane D. D. Antibody to acetylcholine receptor in myasthenia gravis. Prevalence, clinical correlates, and diagnostic value. Neurology. 1976 Nov;26(11):1054–1059. doi: 10.1212/wnl.26.11.1054. [DOI] [PubMed] [Google Scholar]
- Manley S. W., Bourke J. R., Hawker R. W. The thyrotrophin receptor in guinea-pig thyroid homogenate: interaction with the long-acting thyroid stimulator. J Endocrinol. 1974 Jun;61(3):437–445. doi: 10.1677/joe.0.0610437. [DOI] [PubMed] [Google Scholar]
- McKenzie J. M. Does LATS cause hyperthyroidism in Graves' disease? (A review biased toward the affirmative). Metabolism. 1972 Sep;21(9):883–894. doi: 10.1016/0026-0495(72)90012-1. [DOI] [PubMed] [Google Scholar]
- Mehdi S. Q., Nussey S. S. A radio-ligand receptor assay for the long-acting thyroid stimulator. Inhibition by the long-acting thyroid stimulator of the binding of radioiodinated thyroid-stimulating hormone to human thyroid membranes. Biochem J. 1975 Jan;145(1):105–111. doi: 10.1042/bj1450105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittag T., Kornfeld P., Tormay A., Woo C. Detection of anti-acetylcholine receptor factors in serum and thymus from patients with myasthenia gravis. N Engl J Med. 1976 Mar 25;294(13):691–694. doi: 10.1056/NEJM197603252941303. [DOI] [PubMed] [Google Scholar]
- Mukhtar E. D., Smith B. R., Pyle G. A., Hall R., Vice P. Relation of thyroid-stimulating immunoglobulins to thyroid function and effects of surgery, radioiodine, and antithyroid drugs. Lancet. 1975 Mar 29;1(7909):713–715. doi: 10.1016/s0140-6736(75)91629-3. [DOI] [PubMed] [Google Scholar]
- Olefsky J. M. Effect of dexamethasone on insulin binding, glucose transport, and glucose oxidation of isolated rat adipocytes. J Clin Invest. 1975 Dec;56(6):1499–1508. doi: 10.1172/JCI108231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orgiazzi J., Williams D. E., Chopra I. J., Solomon D. H. Human thyroid adenyl cyclase-stimulating activity in immunoglobulin G of patients with Graves' disease. J Clin Endocrinol Metab. 1976 Feb;42(2):341–354. doi: 10.1210/jcem-42-2-341. [DOI] [PubMed] [Google Scholar]
- Patrick J., Lindstrom J. Autoimmune response to acetylcholine receptor. Science. 1973 May 25;180(4088):871–872. doi: 10.1126/science.180.4088.871. [DOI] [PubMed] [Google Scholar]
- RODBELL M. METABOLISM OF ISOLATED FAT CELLS. I. EFFECTS OF HORMONES ON GLUCOSE METABOLISM AND LIPOLYSIS. J Biol Chem. 1964 Feb;239:375–380. [PubMed] [Google Scholar]
- Roth J. Methods for assessing immunologic and biologic properties of iodinated peptide hormones. Methods Enzymol. 1975;37:223–233. doi: 10.1016/s0076-6879(75)37018-3. [DOI] [PubMed] [Google Scholar]
- Shiu R. P., Friesen H. G. Blockade of prolactin action by an antiserum to its receptors. Science. 1976 Apr 16;192(4236):259–261. doi: 10.1126/science.176727. [DOI] [PubMed] [Google Scholar]
- Smith B. R., Hall R. Thyroid-stimulating immunoglobulins in Graves' disease. Lancet. 1974 Aug 24;2(7878):427–431. doi: 10.1016/s0140-6736(74)91815-7. [DOI] [PubMed] [Google Scholar]
- Tarrab-Hazdai R., Aharonov A., Silman I., Fuchs S., Abramsky O. Experimental autoimmune myasthenia induced in monkeys by purified acetylcholine receptor. Nature. 1975 Jul 10;256(5513):128–130. doi: 10.1038/256128a0. [DOI] [PubMed] [Google Scholar]
- Yamashita K., Field J. B. Effects of long-acting thyroid stimulator on thyrotropin stimulation of adenyl cyclase activity in thyroid plasma membranes. J Clin Invest. 1972 Mar;51(3):463–472. doi: 10.1172/JCI106834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Meyts P., Roth J., Neville D. M., Jr, Gavin J. R., 3rd, Lesniak M. A. Insulin interactions with its receptors: experimental evidence for negative cooperativity. Biochem Biophys Res Commun. 1973 Nov 1;55(1):154–161. doi: 10.1016/s0006-291x(73)80072-5. [DOI] [PubMed] [Google Scholar]


