Abstract
Predicting species distributions with changing climate has often relied on climatic variables, but increasingly there is recognition that disturbance regimes should also be included in distribution models. We examined how changes in rainfall and disturbances along climatic gradients determined demographic patterns in a widespread and long-lived tree species, Callitris glaucophylla in SE Australia. We examined recruitment since 1950 in relation to annual (200–600 mm) and seasonal (summer, uniform, winter) rainfall gradients, edaphic factors (topography), and disturbance regimes (vertebrate grazing [tenure and species], fire). A switch from recruitment success to failure occurred at 405 mm mean annual rainfall, coincident with a change in grazing regime. Recruitment was lowest on farms with rabbits below 405 mm rainfall (mean = 0–0.89 cohorts) and highest on less-disturbed tenures with no rabbits above 405 mm rainfall (mean = 3.25 cohorts). Moderate levels of recruitment occurred where farms had no rabbits or less disturbed tenures had rabbits above and below 405 mm rainfall (mean = 1.71–1.77 cohorts). These results show that low annual rainfall and high levels of introduced grazing has led to aging, contracting populations, while higher annual rainfall with low levels of grazing has led to younger, expanding populations. This study demonstrates how demographic patterns vary with rainfall and spatial variations in disturbances, which are linked in complex ways to climatic gradients. Predicting changes in tree distribution with climate change requires knowledge of how rainfall and key disturbances (tenure, vertebrate grazing) will shift along climatic gradients.
Keywords: Callitris, climatic gradients, demography, disturbances, grazing, tree distribution
Introduction
A current challenge in ecology is to determine the processes influencing species distributions, so shifts in distributions under climate change can be predicted. Many models identify empirical relationships between current distributions of species and climatic variables that are used to estimate future distributions of species under climate scenarios (Thomas et al. 2004; Araújo and New 2007). As they do not consider population dynamics, biotic factors, and disturbances, it is uncertain whether species will track predicted shifts in suitable climate space (Pearson and Dawson 2003; Guisan and Thuiller 2005; Keith et al. 2008). In the few studies where these factors have been measured, biotic factors and disturbance regimes were just as important as climate in determining distributions and potential range shifts (Miller and Halpern 1998; García et al. 1999; Gworek et al. 2007; Boulant et al. 2009). This highlights the need to include biotic factors and disturbances in studies identifying drivers of species distributions.
For widespread species, different processes are likely to control demographic patterns in different parts of their distributions (Gworek et al. 2007). Climate can directly affect species distributions through variations in rainfall and temperature patterns (Gworek et al. 2007). For example, establishment of woody species in low rainfall systems is highly dependent on infrequent periods of above average rainfall (Denham and Auld 2004; Castro et al. 2005; Holmgren et al. 2006a; Squeo et al. 2007), resulting in fewer opportunities for establishment than in high rainfall systems. The timing of rainfall may be more important in low than high rainfall systems, as soil moisture stress in summer is a primary cause of plant mortality, especially for small, shallow-rooted seedlings (Denham and Auld 2004; Holmgren et al. 2006b; Squeo et al. 2007; López et al. 2008).
Climate can also affect species distributions indirectly through disturbance regimes (Gworek et al. 2007), which may be influenced by land use, the distribution of which is also determined by climate (Dale 1997; Luck 2007). For example, high levels of livestock grazing in arid systems (Walker 1993; Bureau of Rural Sciences 2009) have contributed to aging populations of woody species in many regions (García et al. 1999; Meyer and Pendleton 2005; Auld and Keith 2009). By comparison, in higher rainfall systems, where a range of land uses are economically viable (Dale 1997), disturbance regimes may be more variable across space. For instance, livestock grazing levels are likely to be higher on farms than on other tenures, such as roadsides (Lunt 1995; Fensham 1998), which may act as refuges, especially for palatable woody species. While fire frequencies have declined in some agricultural systems, leading to an expansion of fire sensitive woody species (Belsky and Blumenthal 1997; Noble 1997; Noss et al. 2006), large patches of woodland may maintain a regime of regular fires (Lunt et al. 2012). Identification of the way in which such processes vary and interact along climatic gradients will provide insight into how consequent demographic effects are likely to be altered by climate change.
Demographic patterns of widespread and long-lived species, associated with climatic factors and disturbance regimes can be identified in large-scale surveys (Peters et al. 2007; Prior et al. 2011). The distribution of trees is especially valuable because their longevity tells us about past influences (Dale et al. 2001). Seedlings of trees often experience demographic “bottle necks” (Grubb 1977; Richardson and Bond 1991), because their recruitment is more sensitive to changes in climate and disturbances than the mortality of adults (Lloyd 1997; Hanson and Weltzin 2000). Changes in the distribution of climatic and disturbance regimes associated with the demographic patterns of a species will influence future species distributions (Thuiller et al. 2008).
Our aim was to examine how rainfall and disturbances along climatic gradients affect demographic patterns of a long-lived and widespread tree species, Callitris glaucophylla Thompson & Johnson (Fig. 1). This species, with a life span of at least 250 years and dominant in arid to temperate woodlands of Australia (Bowman et al. 1995; Prior et al. 2011), is ideal for examining how demographic processes in woody species may vary along climatic and disturbance gradients. C. glaucophylla is a slow-growing obligate-seeding conifer, which is sensitive to disturbances, as it does not resprout (Bowman et al. 1995). A number of studies have identified varying demographic patterns in Callitris species, which have been variously linked to climatic and disturbance variables. In a continental-scale survey of C. columellaris, recruitment was successful in tropical and temperate climates but absent in arid climates and linked to domestic stock grazing (Prior et al. 2011). In smaller scale studies, recruitment of Callitris species was associated with above average rainfall and absent under the influence of grazing by sheep or cattle and high intensity fires (Austin and Williams 1988; Bowman and Latz 1993; Noble 1997; Prior et al. 2010; Russell-Smith et al. 2012). No studies have aged C. glaucophylla individuals to determine relationships between recruitment, rainfall events, and disturbance regimes across climatic gradients. Relationships between these factors are likely to govern the way that the distribution of C. glaucophylla and other woody species will change in the future under climate change.
Figure 1.

A Callitris glaucophylla tree (center), which established in the 1890s surrounded by more recent recruits in north eastern New South Wales, Australia.
In this study, we explored the way that rainfall and disturbances interact at macro-scales across the distribution of C. glaucophylla. We hypothesized: (1) disturbance regimes will vary along rainfall gradients; (2) in the absence of inter- and intra-specific competition, fewer cohorts are more likely where annual rainfall is low and winter dominant, especially where disturbances are frequent; and (3) more cohorts are expected where annual rainfall is higher, especially where disturbances are less frequent. We then use these insights to predict future demographic and distributional changes in C. glaucophylla populations across south eastern Australia in response to climate change.
Methods
Stands of C. glaucophylla were sampled along three transects of declining mean annual rainfall and varying seasonal rainfall dominance in New South Wales, Australia (Colls and Whitaker 2001; Bureau of Meteorology 2010; Fig. 2). Transects extended from the arid zone in the west (200 mm annual rainfall) to the temperate zone in the east (600 mm annual rainfall; Specht and Specht 1999; Bureau of Meteorology 2006). Rainfall varies seasonally from north to south with summer dominant rainfall in the north (60% excess in summer cf. winter, Lacey 1973), uniform rainfall in the middle and winter dominant rainfall in the south (40% excess rainfall in winter cf. summer). Hereafter, transects will be referred to as the summer (29.36°S 149.76°E to 29.03°S 141.44°E), uniform (33.15°S 148.17°E to 32.77°S 141.63°E), and winter rainfall zones (34.80°S 147.20°E to 34.01°S 141.05°E). Each transect was approximately 600 km in length. Fieldwork was undertaken from May to August 2008.
Figure 2.

The location of transects used to survey Callitris glaucophylla recruitment in New South Wales, Australia. Transects follow gradients of declining mean annual rainfall (200–600 mm) from east to west and are located in seasonal rainfall zones from summer in the north, uniform in the middle to winter in the south. Sites where wood samples were taken for radiocarbon dating are marked from S1 to S6. Isohyets are labeled with the mean annual rainfall.
Sites were chosen to sample the maximum number of cohorts at intervals along each transect. In the higher rainfall area to the east, sites were on average 30 km apart, however, in the lower rainfall areas in the west, potential sites were less frequent, and previous vegetation surveys were used to identify sites within 50 km north or south of each transect (Fox 1991; Scott 1992; Porteners 1993; Porteners et al. 1997; Keith 2004; Benson et al. 2006). A total of 56 sites were sampled including 16 in the summer, 23 in the uniform and 17 in the winter rainfall zone.
A prerequisite for site inclusion was the presence of mature C. glaucophylla trees (late-19th century recruitment; Lacey 1972) as a seed source. This cohort established before rabbits reached large numbers from approximately 1879–1897 and there was little C. glaucophylla regeneration until rabbit numbers were significantly reduced in the mid 20th century (Thompson and Eldridge 2005). Other prerequisites included gaps between the canopies of the mature C. glaucophylla trees with at least 10% bare ground to provide space for potential recruitment, flat to undulating topography, no evidence of earthworks, logging, or fires since 1950s and minimal stock grazing. Where possible, fenced roadsides and state forests were sampled, since these were generally less intensely grazed than travelling stock routes and paddocks (Fensham 1998; Lunt 2005). However, there were few less disturbed sites in low rainfall areas in the west, where roadsides were often unfenced from adjacent farms, and state forests were less common.
At each site a number of variables were measured within a 0.1 ha quadrat. The number of cohorts, which had established since 1950s was counted. Cohorts were distinguished by differences in height and canopy shape, presence of lower stems, lichens on bark, and local knowledge of land managers (Read 1995; Whipp 2009). Cohorts were allocated to a relative age category from A to D, with A being the oldest and D the youngest at each site. Where there was uncertainty in differentiating between two cohorts they were grouped into the one category, resulting in a higher probability of underestimating than overestimating cohort numbers. The height and diameter of the two tallest individuals from each cohort were measured.
Bomb-pulse radiocarbon (14C) dating was used to age C. glaucophylla individuals (Hua 2009; Pearson et al. 2011). Radiocarbon dating of representative basal disks of C. glaucophylla individuals from two sites along each transect (distant where possible) was used to confirm recruitment dates assessed by ring counting (Hua 2009; Fig. 2). This involved dating one wood specimen from each cohort at four sites (winter and uniform) and duplicate wood specimens from each cohort at two sites (summer) to examine within site reliability of cohort assessment. Disks from the younger trees were removed near ground level and disks from the oldest trees were removed 20 cm from the ground. Radiocarbon dating on the third ring from the pith of each sample was carried out using the STAR accelerator mass spectrometry facility at Australian Nuclear Science and Technology Organisation (Hua et al. 2001; Fink et al. 2004). As the precision of radiocarbon dating is low for the most recent recruits (≥2003), they were dated by ring counting only.
Recent grazing was assessed by recording the presence of native macropods (Macropodidae) and introduced vertebrates including cattle (Bos taurus L), sheep (Ovis aries L), European rabbits (Oryctolagus cuniculus L), and goats (Capra hircus L). Herbivores were either sighted directly or indirectly by scats, warrens or diggings. Evidence of fire since 1950s was based on charred stems or stags of trees that established after 1950. Land tenure was used as a surrogate for longer term grazing intensity, measured as low to moderate and high: low to moderate for non-farms, including roadsides fenced from adjacent farms, travelling stock routes, state forests, and national parks; high for farms and adjacent unfenced roadsides.
Statistical analyses
We used regression tree analyses to examine the suite of variables associated with the number of cohorts at each site (Table 1). Overfitting was avoided by pruning the tree to minimize the cross-validated error (Venables and Ripley 2002). Categorical variables which occurred in fewer than 30% of sites were excluded from the analyses (e.g., fire). Spearman's rank coefficient was used to examine collinearity between remaining pairs. In the first regression tree analysis all explanatory variables with |r| ≤ 0.5 were included (Tabachnick and Fidell 2007), i.e., annual rainfall (mm), latitude, topographic position (flat, dune), land use (non-farm, farm), and the presence of rabbits, sheep, cattle, goats, and kangaroos. In the second regression tree analysis, annual rainfall was removed, as a number of explanatory variables were significantly correlated with annual rainfall including land use (r = −0.46, P < 0.001), rabbits (r = −0.48, P < 0.001), goats (r = −0.34, P < 0.01), and topographic position (r = −0.34, P < 0.01). No other explanatory variables were significantly correlated. All analyses were undertaken in SPLUS software version 8 for Windows (Insightful Corp 2007).
Table 1.
A description of the variables used in the regression tree analyses and how they were assessed
| Variable | Values | How assessed |
|---|---|---|
| Annual rainfall (mm yr−1) | 200–600 | Bureau of Meteorology (2006) |
| Seasonal rainfall | Summer, uniform, winter | Bureau of Meteorology (2010) |
| Land use type | Fenced roadside (l-m) | Maps/Observations |
| State forest (l-m) | ||
| National park (l-m) | ||
| Travelling stock route (l-m) | ||
| Unfenced roadside (h) | ||
| Farm (h) | ||
| Herbivores (rabbits, sheep cattle, goats, kangaroos) | Present/Absent | Sighting scats, warrens, diggings |
| Fire after 1950 | Present/Absent | Charred post-1950 Callitris |
Land use type indicated long-term grazing intensity, which was rated low-moderate (l-m) or high (h). BOM is the Bureau of Meteorology.
On the basis of the results from the regression tree analysis, we examined trends in the number of cohorts with annual rainfall in each seasonal rainfall zone, by fitting linear, loess, and sigmoidal relationships. Curves of best fit were determined using the residual sum of squares (RSS) and Akaike Information Criterion (AIC), with lower values of either indicating a better fit (Quinn and Keough 2002). A goodness of fit was calculated for each model using R2 = RSS/Total Sum Squares (TSS).
We examined the potential opportunities for C. glaucophylla regeneration since 1950 by deriving a rainfall index. This index was derived at each site by assuming that C. glaucophylla regeneration is dependent on high rainfall in summer, a well documented phenomenon (Forestry Commission of New South Wales 1988). First, we defined the summer rainfall (December–February) value associated with the estimated time of establishment for each radiocarbon dated sample (Fig. 3). We then selected the lowest summer rainfall estimate from the values defined in the first step and assumed that this equated to the minimum amount of summer rain needed for establishment. Finally, we estimated the number of summers from 1950 to 2008 that this minimum value was reached at each site, and from this we constructed an index of potential opportunities for C. glaucophylla regeneration along each seasonal rainfall transect. The higher the index, the greater the potential opportunities for C. glaucophylla regeneration. We examined the effects of mean annual rainfall, seasonal rainfall (summer, uniform, winter) and their interaction on the index of potential opportunities for C. glaucophylla regeneration from 1950, using an Analysis of Covariance (ANCOVA). Data were square root transformed to satisfy homogeneity of variances. Post hoc pairwise comparisons used Bonferroni correction.
Figure 3.

Recruitment periods of Callitris glaucophylla specimens, derived from radiocarbon dating versus mean annual rainfall in each seasonal rainfall zone (summer 1 = duplicate 1, summer 2 = duplicate 2, uniform, winter). Cohorts identified in the field are indicated by A, B, C, or D and represented by vertical lines. Cohort A is likely to be older than the dating indicates, as disks were taken at least 20 cm above the ground. Periods of La Niña are also shown.
We used non-parametric proportion tests to examine the proportional occurrence of disturbances, namely farms, rabbits, sheep, cattle, goats, and kangaroos with mean annual rainfall (<405 mm, ≥405 mm) and seasonal rainfall zone (summer, uniform, winter). Evidence of fires was too infrequent to analyze. Post hoc tests used the Bonferroni correction.
Results
While field recognition of discrete cohorts within sites was reliable, recognition of the same age class between sites was not always reliable (Appendix 1). The oldest cohort (Cohort A) was consistently dated from 1959 to 1964 by radiocarbon, although it is likely that these established in the 1950s (Thompson and Eldridge 2005), as samples were cut 20 cm above the ground, giving an underestimate of their ages (Fig. 3). Cohort B was considered older than C in the field, however, dating showed overlap between sites. Cohort B dated from 1972 to 1996 and cohort C from 1988 to 2002. This agreed with the ring counts, which also indicated that Cohort D, which established from 2003 to 2006 was the youngest. In five of the six cases where replicate wood specimens were dated within a site, radiocarbon results of these specimens delivered the same recruitment year. On the one occasion where this did not occur, there was only 1 year difference between the duplicate specimens.
The first regression tree analysis indicated that cohort number was associated with annual rainfall, seasonal rainfall (i.e., latitude), and land tenure (adjusted R2 = 0.65, Fig. 4A). The root or primary division in the regression tree occurred at 405 mm mean annual rainfall. There were fewer cohorts below 405 mm mean annual rainfall, especially in the winter rainfall zone compared with the summer rainfall zone (0.15 c.f. 1.78). Above 405 mm mean annual rainfall, farms had fewer cohorts than less disturbed tenures, such as roadsides, travelling stock routes, state forests, and national parks (2.13 c.f. 3.28).
Figure 4.
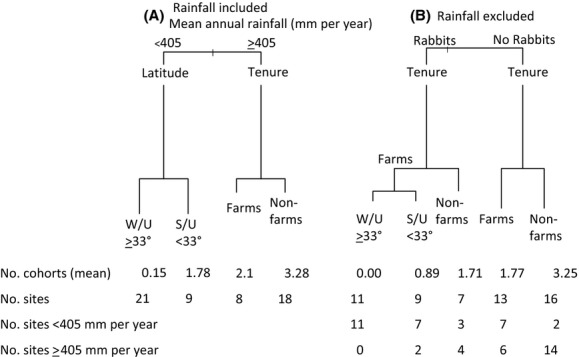
Results from regression tree analyses showing the factors associated with the average number of cohorts since 1950, when mean annual rainfall was (A) included and (B) excluded. In (B) the number of sites less than (<) and greater than or equal to (≥) 405 mm mean annual rainfall is given below each branch. Seasonally dominant rainfall is in summer (S), uniform (U), and winter (W).
The second regression tree analysis, which excluded mean annual rainfall, indicated that cohort number was associated with rabbit occurrence and land tenure (adjusted R2 = 0.65, Fig. 4B). The mean number of cohorts was lowest on farms with rabbits in the winter and uniform rainfall zones (0) and marginally higher in the summer and uniform rainfall zones (0.89). Most of these sites occurred below 405 mm mean annual rainfall. The highest number of cohorts occurred where there were no rabbits on less disturbed tenures, namely roadsides, travelling stock routes, state forests, and national parks (3.25). Most of these sites occurred above 405 mm mean annual rainfall. Moderate numbers of cohorts were on farms without rabbits (1.77) and less disturbed tenures with rabbits (1.71). These sites were distributed equally above and below 405 mm mean annual rainfall.
Curve fitting identified varying trends in the number of cohorts with mean annual rainfall, depending on the seasonal rainfall zone (Fig. 5). Abrupt declines in the number of cohorts at approximately 405 mm annual rainfall in the winter and uniform rainfall zones were best described by a sigmoid and loess curve, respectively, based on low values for RSS and AIC (R2 = 0.94, 0.92; Appendix 2). In the summer rainfall zone, there was little relationship between cohort number and annual rainfall using linear or loess curves (R2 = 0.18, 0.001; Appendix 2).
Figure 5.

Curves of best fit (based on AIC) for the number of cohorts since 1950 versus mean annual rainfall in each seasonal rainfall zone.
The index of potential opportunities for C. glaucophylla regeneration (square root transformed) since 1950, was significantly and positively associated with mean annual rainfall treated as a continuous variable (P < 0.001) and significantly associated with seasonal rainfall (summer, uniform, winter (P < 0.001)), but not their interaction (P = 0.31) using ANCOVA (Fig. 6, Appendix 3). Post hoc pairwise tests indicated that the index was significantly greater in summer than either the uniform or winter rainfall zones, but not significantly different between the winter and uniform rainfall zones.
Figure 6.

Linear trends in an index of potential opportunities for Callitris glaucophylla regeneration in summer from 1950 to 2008 versus mean annual rainfall in each seasonal rainfall zone (summer, uniform, winter).
Proportion tests were used to examine the occurrence of disturbances with mean annual rainfall and seasonal rainfall (Fig. 7, Appendix 4). A significantly greater proportion of sample sites contained farms (P = 0.002), rabbits (P = 0.001), and sheep (P = 0.048), below 405 mm mean annual rainfall than above. The proportion of sites with farms (P = 0.190), sheep (P = 0.810), cattle (P = 0.480), and kangaroos (P = 0.480) was not significantly different between the seasonal rainfall zones. The proportion of sites with rabbits was significantly greater in the winter than the summer rainfall zone (P = 0.030), and for goats was significantly greater in the uniform than the winter rainfall zone (P = 0.008). Although not statistically analyzed, a greater proportion of fires occurred at less than 405 mm mean annual rainfall than above.
Figure 7.

Proportion of sample sites with farms, grazing species, and fire versus mean annual rainfall (< and ≥405 mm) in each seasonal rainfall zone (summer, winter, uniform). Asterisks and pluses indicate significant differences between < 405 and ≥405 mm mean annual rainfall and seasons, respectively. As few sites were burned, fire was not statistically analyzed.
Discussion
These results indicate major differences in C. glaucophylla recruitment patterns across the species’ range in western New South Wales. Since 1950s, C. glaucophylla has recruited frequently in high-rainfall areas (≥405 mm mean annual rainfall), and in areas with summer-dominated rainfall. In contrast, most populations in low rainfall areas (<405 mm) with winter-dominated rainfall have experienced no recruitment since the late 1800s (Thompson and Eldridge 2005). At the scale of the study, disturbance regimes were not independent of climatic patterns. Consequently, recruitment patterns were closely associated with regional patterns of disturbance and land use, especially grazing by livestock, feral, and native mammals. C. glaucophylla recruited more frequently on tenures that were less frequently grazed by livestock and in areas with no evidence of recent grazing by feral rabbits.
Rainfall and recruitment
Many studies have reported infrequent, pulsed recruitment of woody plants in arid and semi-arid regions (Watson et al. 1997b; Holmgren and Scheffer 2001; Wiegand et al. 2004; Holmgren et al. 2006b; Prior et al. 2011). Inadequate soil moisture in most years prevents seedling establishment and causes high mortality of young plants (Watson et al. 1997a; Holmgren et al. 2006a; Gutiérrez et al. 2007). C. glaucophylla, however, is a long-lived tree with a life-span of over 200 years (Bowman et al. 1995). Consequently, an absence of recruitment for a century does not necessarily indicate a decline in long-term population viability, as rare recruitment events may enable populations to persist (e.g., Wiegand et al. 2004). However, tree mortality was apparent at some sites, indicating that populations may have declined in recent decades.
Additionally, historical rainfall records indicate that the winter-rainfall region has experienced a number of high rainfall (La Niña) events over the past century, especially in 1948–1949, 1955–1956, 1973–1974, 1988–1989, and 1998–2000 (Nicholls et al. 1992; Bureau of Meteorology 2005). Rainfall in these years was well above the mean annual rainfall or in the top decile rank (Bureau of Meteorology 2005). These events triggered widespread recruitment of woody plants including C. glaucophylla in SE Australia (Austin and Williams 1988; Harrington 1991; Denny 1992; Read 1995; Noble 1997; Parker and Lunt 2000), but only limited recruitment of woody species in arid and semi-arid regions (Hall et al. 1964; Chesterfield and Parsons 1985), and sometimes only within fenced areas (Hall et al. 1964). Consequently, the absence of C. glaucophylla recruitment during the 1900s from this low rainfall region (especially winter-rainfall region) is unlikely to be due to inadequate rainfall. Indeed, other authors have attributed declines of woody species in this region to unfavorable disturbance regimes, particularly grazing (Auld and Keith 2009; Prior et al. 2011).
In contrast, C. glaucophylla has recruited frequently over the past 60 years in high rainfall areas (>405 mm mean annual rainfall), consistent with trends in a continental-scale survey by Prior et al. (2011). For the first half of the 20th century, C. glaucophylla populations contained plants derived from the late 1800s and earlier, with no younger cohorts (Lacey 1972; Austin and Williams 1988). Repeated recruitment since1950s means that population density has increased (Lacey 1972; Austin and Williams 1988) and mean population age has declined. C. glaucophylla stands self-thin extremely slowly (Lacey 1972; Forestry Commission of New South Wales 1988). Consequently, these fire-sensitive stands are becoming increasingly dense as recruitment continues. A current lack of high intensity fires, prevalent before agriculture and land fragmentation (Noble 1997; Noble and Dargavel 2001) also promotes these changes (Ross et al. 2012). This phenomenon is similar to that found in fire-sensitive Pinus ponderosa populations in the long-term absence of fire in mixed Pinus-Quercus forests in the western U. S. A. (Noss et al. 2006).
Seasonal rainfall had a strong effect on the number of C. glaucophylla cohorts, with establishment being particularly restricted in low rainfall areas (<405 mm) in winter as opposed to summer-dominated rainfall areas. In dry regions, soil moisture stress in summer is a primary cause of plant mortality, especially for small, shallow-rooted seedlings (Denham and Auld 2004; Holmgren et al. 2006b; Squeo et al. 2007; López et al. 2008). Consequently, for a given amount of annual rainfall, summer or uniform rainfall supports greater plant productivity than winter rainfall (Holmgren et al. 2006b; Lopez et al. 2006; Squeo et al. 2007). The coincidence of seed shedding in summer by C. glaucophylla (Lacey 1972) and summer rainfall may also increase opportunities for establishment in the summer-dominated rainfall zone.
Land use, herbivory, and recruitment
Land use typically changes with climate, with greater agricultural productivity in higher rainfall areas, and extensive (“rangeland”) livestock grazing in low rainfall zones (Walker 1993). In this study, fencing patterns which control livestock movements varied across the rainfall gradient. In low rainfall areas, extensive rangeland grazing occurs, and relatively few roadsides or patches of remnant vegetation are fenced to prevent access by livestock. In contrast, in high rainfall areas, fences separate all paddocks from roadsides, and most patches of remnant vegetation on public land area are fenced. Consequently, C. glaucophylla stands on roadsides in high rainfall areas are less likely to be frequently grazed by livestock than stands on roadsides in semi-arid areas. This may explain why the number of C. glaucophylla cohorts was consistently greatest in “non-farm” land tenures (including roadsides and public land reserves) in areas of high rainfall.
Rabbits and sheep cause recruitment failure in C. glaucophylla and other woody species (e.g., Acacia, Casuarina spp.) as indicated by exclosure experiments and studies of population structure in semi-arid and arid Australia (Crisp and Lange 1976; Crisp 1978; Lange and Graham 1983; Chesterfield and Parsons 1985; Auld 1995; Tiver and Andrew 1997; Auld and Denham 2001; Prior et al. 2011). Similarly, our results suggest that the paucity of C. glaucophylla recruitment in semi-arid areas with winter and uniform rainfall is likely to be strongly influenced by grazing patterns. These trends are consistent with studies on woody shrubs in semi-arid regions elsewhere in the world (Bowers 1997; Gutiérrez et al. 2007; Holmgren et al. 2006b).
Our results highlight complex interactions among recruitment, disturbance regimes, and climate (Pearson and Dawson 2003; Peters et al. 2008). At a regional scale, variation in land use can provide refuges from livestock grazing and influence the abundance of feral animals. At a patch scale, grazing by livestock and feral animals directly influences establishment of woody plants. Broad-scale climate variation (e.g., high vs. low, and winter vs. summer rainfall) also affects these processes as shown by the variations in tree recruitment over a 60-year time-frame in this study. Future trends in recruitment will therefore depend on the way changes to management and climate interact (Fig. 7; Pearson and Dawson 2003; Guisan and Thuiller 2005; Keith et al. 2008).
Future trends
Under climate change, temperatures are expected to increase in the study area in the future (Commonwealth Scientific and Industrial Research Organisation & Bureau of Meteorology 2007; IPCC et al. 2007; Hennessy et al. 2008). Although future rainfall patterns are less certain, projections indicate an increase in summer rainfall in the north of the region, strengthening existing patterns of summer dominant rainfall (Commonwealth Scientific and Industrial Research Organisation & Bureau of Meteorology 2007; Hennessy et al. 2008). In contrast, in southern areas, winter rainfall is expected to decline substantially, with only a slight increase in summer rainfall (Commonwealth Scientific and Industrial Research Organisation & Bureau of Meteorology 2007; Hennessy et al. 2008).
While infrequent high rainfall events may continue to drive major recruitment pulses, as has occurred in the past (Austin and Williams 1988), increasing aridity is likely to further restrict opportunities for C. glaucophylla recruitment, especially in dry, winter-rainfall areas. Thus, projected climatic changes are likely to intensify rather than ameliorate recruitment problems currently experienced in dry winter-rainfall areas. In contrast, the “leading edge” of the species distribution in high rainfall areas may expand eastward into adjacent Eucalyptus-dominated woodlands, in which C. glaucophylla is currently absent or rare, particularly if low fire frequency is maintained by land management practices.
A recent projection of climate change impacts on Callitris dominated woodlands in Western Australia based on bioclimatic envelope modeling suggested a major range expansion with no decline in areas occupied (Prober et al. 2012). In contrast, our results suggest that climate change is likely to intensify stresses on patches in dry winter-rainfall areas, potentially leading to range contractions in this sub-region. However, we emphasize that our results reflect historical inter-dependencies among land management practices, and these may not be continued in the future. In particular, recruitment opportunities and population viability may increase if greater control is exerted over grazing by feral animals and livestock in dry, winter-rainfall populations. If current grazing regimes persist or intensify, there appears to be a bleak future for C. glaucophylla populations and other woody species in this region regardless of climate change. This uncertainty may be resolved by experimentally manipulating grazing regimes in this sub-region.
Conclusion
Demographic patterns in the widespread and long-lived tree species, C. glaucophylla, were closely associated with rainfall gradients and disturbance regimes. Disturbance regimes were associated with rainfall through different land use types. As climate changes, rainfall, land use, and disturbance regimes will shift along climatic gradients, influencing species distributions. To predict changes in the distribution of C. glaucophylla and other species, a greater understanding is required of how land management, disturbance regimes, and climate interact. Prediction of the outcome of novel combinations of these factors in the future will be challenging.
Acknowledgments
Australian Research Council (ARC) Linkage Grant between Charles Sturt University (CSU) and NSW Department of Environment and Climate Change (DECC) provided the funding for this research (Project number LP066829). A grant from Australian Institute for Nuclear Science and Energy (AINSE) provided funding for the radiocarbon dating of C. glaucophylla wood samples (AINGRA09075). We thank Stephen Campbell and Alan Smith from State Forests NSW for their assistance and permission to take samples. We also thank John Warren and Kate Hammill for assistance in the field.
Appendix 1
Radiocarbon dating results for Callitris glaucophylla basal disks.
| Percent modern carbon (pMC) | Calibrated ages at 95% confidence level | |||||||
|---|---|---|---|---|---|---|---|---|
| Lab ID | Sample ID | Transect | δ13C (‰) | Mean | 1σ | Age range (AD) | Probability | |
| 1 | OZL812 | s2 1950/1 | Summer | −22.9 | 117.09 | 0.45 | 1959.64–1961.76 | 0.14 |
| 1987.33–1992.56 | 0.86 | |||||||
| 2 | OZL813 | s2 1950/2 | Summer | −23.0 | 119.10 | 0.44 | 1959.68–1962.4 | 0.47 |
| 1985.32–1990.12 | 0.53 | |||||||
| 3 | OZL814 | s2 1970/1 | Summer | −23.8 | 114.16 | 0.40 | 1959.07–1959.64 | 0.16 |
| 1990.18–1994.34 | 0.83 | |||||||
| 4 | OZL815 | s2 1970/2 | Summer | −24.4 | 113.12 | 0.39 | 1958.77–1959.62 | 0.13 |
| 1992.36–1996.29 | 0.85 | |||||||
| 5 | OZL816 | s2 1990/2000/1 | Summer | −24.9 | 111.34 | 0.42 | 1958.72–1959.09 | 0.18 |
| 1995.15–1999.99 | 0.81 | |||||||
| 6 | OZL817 | s2 1990/2000/2 | Summer | −25.2 | 114.66 | 0.43 | 1959.09–1959.65 | 0.12 |
| 1988.46–1993.94 | 0.88 | |||||||
| 7 | OZL818 | s1 1950/1 | Summer | −22.6 | 146.13 | 0.65 | 1963.78–1964.17 | 0.07 |
| 1972.27–1973.62 | 0.93 | |||||||
| 8 | OZL819 | s1 1950/2 | Summer | −23.2 | 138.41 | 0.56 | 1963.51–1963.91 | 0.09 |
| 1974.37–1976.24 | 0.91 | |||||||
| 9 | OZL820 | s1 1970/1 | Summer | −22.6 | 119.71 | 0.47 | 1959.92–1962.43 | 0.52 |
| 1985.26–1989.33 | 0.47 | |||||||
| 10 | OZL821 | s1 1970/2 | Summer | −23.9 | 121.96 | 0.49 | 1961.84–1962.5 | 0.12 |
| 1983.11–1986.43 | 0.88 | |||||||
| 11 | OZL822 | s1 1990/2000/1 | Summer | −24.0 | 113.36 | 0.44 | 1959.04–1959.62 | 0.15 |
| 1990.36–1995.48 | 0.84 | |||||||
| 12 | OZL823 | s1 1990/2000/2 | Summer | −23.3 | 114.47 | 0.42 | 1959.09–1959.64 | 0.13 |
| 1990.17–1993.94 | 0.86 | |||||||
| 13 | OZL824 | s4 1950/2 | Uniform | −24.1 | 134.31 | 0.48 | 1963.42–1963.61 | 0.07 |
| 1976.56–1978.17 | 0.93 | |||||||
| 14 | OZL825 | s4 1970/2 | Uniform | −23.8 | 114.11 | 0.45 | 1959.07–1959.64 | 0.17 |
| 1990.18–1994.36 | 0.82 | |||||||
| 15 | OZL826 | s3 1950/4 | Uniform | −23.2 | 137.76 | 0.43 | 1963.5–1963.89 | 0.08 |
| 1974.42–1976.25 | 0.92 | |||||||
| 16 | OZL827 | s3 1970/1 | Uniform | −22.8 | 134.09 | 0.50 | 1963.4–1963.59 | 0.06 |
| 1976.57–1978.79 | 0.94 | |||||||
| 17 | OZL828 | s3 1990/2000/2 | Uniform | −22.8 | 114.26 | 0.44 | 1959.07–1959.64 | 0.15 |
| 1990.18–1994.33 | 0.84 | |||||||
| 18 | OZL829 | s6 1950/2 | Winter | −23.1 | 126.18 | 0.51 | 1962.56–1963.26 | 0.16 |
| 1980.4–1982.79 | 0.83 | |||||||
| 19 | OZL830 | s6 1970/2 | Winter | −23.9 | 123.59 | 0.42 | 1962.25–1962.72 | 0.05 |
| 1982.09–1984.67 | 0.95 | |||||||
| 20 | OZL831 | s5 1950/1 | Winter | −23.8 | 145.69 | 0.53 | 1963.76–1964.17 | 0.07 |
| 1972.42–1973.64 | 0.93 | |||||||
| 21 | OZL832 | s5 1970/1 | Winter | −22.8 | 145.33 | 0.54 | 1963.76–1964.2 | 0.08 |
| 1972.43–1973.65 | 0.92 | |||||||
| 22 | OZL833 | s5 2000/2 | Winter | −23.1 | 109.24 | 0.41 | 1958.64–1958.72 | 0.03 |
| 1997.45–2002.87 | 0.97 | |||||||
Appendix 2
Results of fitting curves to the number of cohorts with mean annual rainfall in each seasonal rainfall zone according to residual sum of squares (RSS), Akaikie Information Criterion (AIC), and R2.
| Rainfall zone | Curve | RSS | AIC | R2 |
|---|---|---|---|---|
| Summer | Linear | 32.96 | 15.56 | 0.18 |
| Loess | 26.96 | 20.40 | 0.001 | |
| Uniform | Loess | 5.42 | −21.43 | 0.92 |
| Sigmoid | 7.40 | −18.08 | 0.89 | |
| Linear | 29.53 | 9.75 | 0.57 | |
| Winter | Sigmoid | 1.83 | −29.86 | 0.94 |
| Loess | 2.20 | −22.87 | 0.93 | |
| Linear | 9.24 | −6.36 | 0.70 |
Within each rainfall zone, models are ordered from best to worst using AIC.
Appendix 3
Results of ANCOVA used to examine the effects of mean annual rainfall (continuous) and seasonal rainfall (summer s, uniform u, winter w) on an index of potential opportunities for Callitris glaucophylla regeneration in summer from 1950 to 2008 (R2 = 0.93).
| Source | SS | df | MS | F | P |
|---|---|---|---|---|---|
| Corrected model | 79.60 | 5 | 15.92 | 138.21 | <0.001 |
| Intercept | 0.11 | 1 | 0.11 | 0.98 | 0.327 |
| Annual rainfall (mm yr−1) | 45.32 | 1 | 45.88 | 393.44 | <0.001 |
| Seasonal rainfall (s, u, w) | 0.79 | 2 | 0.39 | 3.44 | <0.001 |
| Annual × seasonal rainfall | 0.27 | 2 | 0.14 | 1.18 | 0.315 |
| Error | 5.76 | 50 | 0.12 | ||
| Total | 978.00 | 56 | |||
| Corrected total | 85.37 | 55 |
The index was square root transformed.
Appendix 4
Results of proportion tests used to examine the occurrence of disturbances with mean annual rainfall (<405 mm, >405 mm) and seasonal rainfall (summer s, winter w, uniform u).
| Disturbance | Mean annual rainfall (mm) | Seasonal rainfall zone | ||||||
|---|---|---|---|---|---|---|---|---|
| X2 | df | P | Directionality | X2 | df | P | Directionality | |
| Farms | 13.80 | 1 | 0.002 | Below > above | 3.37 | 2 | 0.190 | |
| Rabbits | 10.20 | 1 | 0.001 | Below > above | 6.86 | 2 | 0.030 | w > s |
| Sheep | 3.91 | 1 | 0.048 | Below > above | 0.41 | 2 | 0.810 | |
| Cattle | 1.31 | 1 | 0.250 | 1.49 | 2 | 0.480 | ||
| Goats | 1.31 | 1 | 0.290 | 9.61 | 2 | 0.008 | u > w | |
| Kangaroos | 0.04 | 1 | 0.840 | 0.11 | 2 | 0.950 | ||
Fire was not statistically analyzed.
Conflict of Interest
None declared.
References
- Araújo MB, New M. Ensemble forecasting of species distributions. Trends Ecol. Evol. 2007;22:42–47. doi: 10.1016/j.tree.2006.09.010. [DOI] [PubMed] [Google Scholar]
- Auld TD. The impact of herbivores on regeneration in four trees from arid Australia. Rangeland J. 1995;17:213–227. [Google Scholar]
- Auld TD, Denham AJ. Flora conservation issues at Kinchega National Park, western NSW. Cunninghamia. 2001;7:27–40. [Google Scholar]
- Auld TD, Keith DA. Dealing with threats: integrating science and management. Ecol. Manage. Restor. 2009;10:s79–s87. [Google Scholar]
- Austin MP, Williams OB. Influence of climate and community composition on the population demography of pasture species in semi-arid Australia. Vegetatio. 1988;77:43–49. [Google Scholar]
- Belsky AJ, Blumenthal DM. Effects of livestock grazing on stand dynamics and soils in upland forests of the interior west. Conserv. Biol. 1997;11:315–327. [Google Scholar]
- Benson JS, Allen CB, Togher C, Lemmon J. New South Wales vegetation classification and assessment: part 1 plant communities of NSW western plains. Cunninghamia. 2006;9:383–450. [Google Scholar]
- Boulant N, Garnier A, Curt T, Lepart J. Disentangling the effects of land use, shrub cover and climate on the invasion speed of native and introduced pines in grasslands. Divers. Distrib. 2009;15:1047–1059. [Google Scholar]
- Bowers JE. Demography patterns of Ferocactus cylindraceus in relation to substrate age and grazing history. Plant Ecol. 1997;133:37–48. [Google Scholar]
- Bowman DMJS, Latz PK. Ecology of Callitris glaucophylla (Cupressaceae) on the MacDonnell Ranges, Central Australia. Aust. J. Bot. 1993;41:217–225. [Google Scholar]
- Bowman DMJS, Harris S. Ecology of the Southern Conifers. In: Enright NJ, Hill Robert S, editors. Conifers of Australia's dry forests and open woodlands. 1st ed. Melbourne: Melbourne University Press; 1995. pp. 252–270. [Google Scholar]
- Bureau of Meteorology. Melbourne: Australian Government; 2005. Australian rainfall deciles [Online] Available at http://reg.bom.gov.au/silo/products/ClimMaps.shtml (accessed 14 October 2011) [Google Scholar]
- Bureau of Meteorology. Melbourne: Bureau of Meteorology; 2006. Special climate statement 9: an exceptionally dry decade in parts of southern and eastern Australia: October 1996-September 2006. [Google Scholar]
- Bureau of Meteorology. Major seasonal rainfall zones of Australia [Online] 2010. Commonwealth of Australia. Available at http://www.bom.gov.au/lam/climate/levelthree/ausclim/zones.htm (accessed October 14, 2011)
- Bureau of Rural Sciences. 2001/02 Land uses of Australia, V.3 [Online] 2009. Available at http://adl.brs.gov.au/mapserv/landuse/index.cfm?fa=app.mapping&tab=mapping: Australian Government. 2010 (accessed October 14, 2011)
- Castro J, Zamora R, Hódar JA, Gómez JM. Alleviation of summer drought boosts establishment success of Pinus sylvestris in a mediterranean mountain: an experimental approach. Plant Ecol. 2005;181:191–202. [Google Scholar]
- Chesterfield CJ, Parsons RF. Regeneration of three tree species in arid south-eastern Australia. Aust. J. Bot. 1985;33:715–732. [Google Scholar]
- Colls K, Whitaker R. The Australian weather book. Sydney: Reed New Holland Pty Ltd; 2001. [Google Scholar]
- Commonwealth Scientific and Industrial Research Organisation & Bureau of Meteorology. Climate change in Australia: technical report 2007. Canberra: 2007. CSIRO, Bureau of Meteorology and Australian Greenhouse Office in partnership with the Australian Climate Change Program. [Google Scholar]
- Crisp MD. Demography and survival under grazing of three Australian semi-arid desert shrubs. Oikos. 1978;30:520–528. [Google Scholar]
- Crisp MD, Lange RT. Age structure, distribution and survival under grazing of the arid-zone shrub Acacia burkittii. Oikos. 1976;27:86–92. [Google Scholar]
- Dale VH. The relationship between land-use change and climate change. Ecol. Appl. 1997;7:753–769. [Google Scholar]
- Dale VH, Joyce LA, McNulty S, Neilson RP, Ayres MP, Flannigan MD, et al. Climate change and forest disturbances (Cover story) Bioscience. 2001;51:723. [Google Scholar]
- Denham AJ, Auld TD. Survival and recruitment of seedlings and suckers of trees and shrubs of the Australian arid zone following habitat management and the outbreak of Rabbit Calicivirus Disease (RCD) Austral Ecol. 2004;29:585–599. [Google Scholar]
- Denny M. Historical and ecological study of the effects of european settlement on inland NSW. Sydney: Nature Conservation Council of New South Wales; 1992. [Google Scholar]
- Fensham RJ. The grassy vegetation of the Darling Downs, south-eastern Queensland, Australia, Floristics and grazing effects. Biol. Conserv. 1998;84:301–310. [Google Scholar]
- Fink D, Hotchkis M, Hua Q, Jacobsen GE, Smith AM, Zoppi U, et al. The ANTARES AMS facility at ANSTO. Nucl. Instrum. Methods Phys. Res., Sect. B. 2004;223–224:109–115. [Google Scholar]
- Forestry Commission of New South Wales. Sydney: Forestry Commission of NSW; 1988. Notes on the Silviculture of Major NSW Forest Types.10. Cypress Pine Types. [Google Scholar]
- Fox MD. The natural vegetation of the Ana Branch-Mildura 1:250000 map sheets. Cunninghamia. 1991;2:443–493. [Google Scholar]
- García D, Zamora R, Hódar JA, Gómez JM. Age structure of Juniperus communis L. in the Iberian peninsula: conservation of remnant populations in Mediterranean mountains. Biol. Conserv. 1999;87:215–220. [Google Scholar]
- Grubb PJ. The maintenance of species-richness in plant communities: the importance of the regeneration niche. Biol. Rev. 1977;52:107–145. [Google Scholar]
- Guisan A, Thuiller W. Predicting species distribution: offering more than simple habitat models. Ecol. Lett. 2005;8:993–1009. doi: 10.1111/j.1461-0248.2005.00792.x. [DOI] [PubMed] [Google Scholar]
- Gutiérrez JR, Holmgren M, Manrique R, Squeo FA. Reduced herbivore pressure under rainy ENSO conditions could facilitate dryland reforestation. J. Arid Environ. 2007;68:322–330. [Google Scholar]
- Gworek JR, Vander Wall SB, Brussard PF. Changes in biotic interactions and climate determine recruitment of Jeffrey pine along an elevation gradient. For. Ecol. Manage. 2007;239:57–68. [Google Scholar]
- Hall E, Specht R, Eardley C. Regeneration of the vegetation on Koonamore vegetation Reserve, 1926–1962. Aust. J. Bot. 1964;12:205–264. [Google Scholar]
- Hanson PJ, Weltzin JF. Drought disturbance from climate change: response of United States forests. Sci. Total Environ. 2000;262:205–220. doi: 10.1016/s0048-9697(00)00523-4. [DOI] [PubMed] [Google Scholar]
- Harrington GN. Effects of soil moisture on shrub seedling survival in a semi-arid grassland. Ecology. 1991;72:1138–1149. [Google Scholar]
- Hennessy K, Fawcett R, Kirono D, Mpelasoka F, Jones D, Bathols J, et al. An assessment of the impact of climate change on the nature and frequency of exceptional climatic circumstances. Canberra: Commonwealth of Australia; 2008. [Google Scholar]
- Holmgren M, Scheffer M. El Niño as a window of opportunity for the restoration of degraded arid ecosystems. Ecosystems. 2001;4:151–159. [Google Scholar]
- Holmgren M, Stapp P, Dickman CR, Gracia CA, Graham S, Gutierrez JR, et al. Extreme climatic events shape arid and semiarid ecosystems. Front. Ecol. Environ. 2006a;4:87–95. [Google Scholar]
- Holmgren M, Lopez BC, Gutierrez JR, Squeo FA. Herbivory and plant growth rate determine the success of El Nino Southern Oscillation-driven tree establishment in semiarid South America. Glob. Change Biol. 2006b;12:2263–2271. [Google Scholar]
- Hua Q. Radiocarbon: a chronological tool for the recent past. Quat. Geochronol. 2009;4:378–390. [Google Scholar]
- Hua Q, Barbetti M. Review of tropospheric bomb 14C data for carbon cycle modeling and age calibration purposes. Radiocarbon. 2004;46:1273–1298. [Google Scholar]
- Hua Q, Jacobsen GE, Zoppi U, Lawson EM, Willaims AA, Smith AM, et al. Progress in radiocarbon target preparation at the ANTARES AMS Centre. Radiocarbon. 2001;43:275–282. [Google Scholar]
- Insightful Corp. S-Plus 8 Guide to Statistics. Seattle, WA: Insightful Corporation; 2007. [Google Scholar]
- IPCC. Climate change 2007: the physical basis. In: Solomon S, Quin D, Manning M, Marquis M, Averyt KB, Tignor MB, Miller HL, Chen Z, editors. Contributions of working group 1 to the fourth assessment report of the intergovernmental panel on climate change. Cambridge, U.K., New York: Cambridge Univ. Press; 2007. pp. 1–196. [Google Scholar]
- Keith DA. Ocean shores to desert dunes: the native vegetation of New South Wales and the ACT. Sydney: Dept Environment and Climate Change (NSW); 2004. [Google Scholar]
- Keith DA, Akcakaya HR, Thuiller W, Midgely GF, Pearson RG, Phillips SJ, et al. Predicting extinction risks under climate change: coupling stochastic population models with dynamic bioclimatic habitat models. Biol. Lett. 2008;4:560–563. doi: 10.1098/rsbl.2008.0049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lacey CJ. Factors influencing occurrence of cypress pine regeneration in New South Wales. Sydney: Forestry Commission of NSW; 1972. [Google Scholar]
- Lacey CJ. Silvicultural characteristics of white cypress pine. Sydney: Forestry Commission of NSW; 1973. [Google Scholar]
- Lange RT, Graham CR. Rabbits and the failure of regeneration in Australian arid zone Acacia. Austral Ecol. 1983;8:377–381. [Google Scholar]
- Lloyd AH. Response of tree-line populations of foxtail pine (Pinus balfouriana) to climate variation over the last 1000 years. Can. J. For. Res. 1997;27:936–942. [Google Scholar]
- Lopez BC, Rodriguez R, Gracia CA, Sabate S. Climatic signals in growth and its relation to ENSO events of two Prosopis species following a latitudinal gradient in South America. Glob. Change Biol. 2006;12:897–906. [Google Scholar]
- López BC, Holmgren M, Sabaté S, Gracia CA. Estimating annual rainfall threshold for establishment of tree species in water-limited ecosystems using tree-ring data. J. Arid Environ. 2008;72:602–611. [Google Scholar]
- Luck GW. The relationships between net primary productivity, human population density and species conservation. J. Biogeogr. 2007;34:201–212. [Google Scholar]
- Lunt ID. European management of remnant grassy forests and woodlands in south-eastern Australia. Vic. Nat. 1995;112:239–249. [Google Scholar]
- Lunt ID. Canberra: Environment ACT; 2005. Effects of stock grazing on biodiversity values in temperate native grasslands and grassy woodlands in SE Australia: a literature review. Technical Report 18. [Google Scholar]
- Lunt ID, Prober SM, Morgan JW. How do fire regimes affect ecosystem structure, function and diversity in grasslands and grassy woodlands of southern Australia? In: Bradstock RA, Gill MA, Williams RJ, editors. Flammable Australia: fire regimes, biodiversity and ecosystems in a changing world. Melbourne: CSIRO Publishing; 2012. pp. 253–270. [Google Scholar]
- Meyer SE, Pendleton BK. Factors affecting seed germination and seedling establishment of a long-lived desert shrub (Coleogyne ramosissima: Rosaceae) Plant Ecol. 2005;178:171–187. [Google Scholar]
- Miller EA, Halpern CB. Effects of environment and grazing disturbance on tree establishment in meadows of the central Cascade Range, Oregon, USA. J. Veg. Sci. 1998;9:265–282. [Google Scholar]
- Nicholls N. Historical El Nino/Southern Oscillation variability in the Australasian region. In: Diaz HF, Markgraf V, editors. El Nino: historical and paleo aspects of the Southern Oscillation. Cambridge: Cambridge Univ. Press; 1992. pp. 149–173. [Google Scholar]
- Noble JC. The delicate and noxious scrub. Canberra: CSIRO; 1997. [Google Scholar]
- Noble JC. Regulating Callitris populations: tale of two pineries. In: Dargavel J, editor. Perfumed pineries: environmental history of Australia's Callitris forests. Canberra: Australian National University; 2001. pp. 173–183. [Google Scholar]
- Noss RF, Franklin JF, Baker WL, Schoennagel T, Moyle PB. Managing fire-prone forests in western United States. Front. Ecol. Environ. 2006;4:481–487. [Google Scholar]
- Parker D, Lunt ID. Stand structure and recruitment patterns in Callitris-Eucalyptus woodlands in Terrick Terrick National Park, Victoria. Vic. Nat. 2000;117:207–213. [Google Scholar]
- Pearson RG, Dawson TP. Predicting the impacts of climate change on the distribution of species: are bioclimate envelope models useful? Glob. Ecol. Biogeogr. 2003;12:361–371. [Google Scholar]
- Pearson S, Hua Q, Allen K, Bowman DMJS. Validating putatively cross-dated Callitris tree-ring chronologies using bomb-pulse radiocarbon analysis. Aust. J. Bot. 2011;59:7–17. [Google Scholar]
- Peters D, Bestelmeyer B, Turner M. Cross–scale interactions and changing pattern–process relationships: consequences for system dynamics. Ecosystems. 2007;10:790–796. [Google Scholar]
- Peters DPC, Groffman PM, Nadelhoffer KJ, Grimm NB, Collins SL, Michener WK, et al. Living in an increasingly connected world: a framework for continental-scale environmental science. Front. Ecol. Environ. 2008;6:229–237. [Google Scholar]
- Porteners MF. The natural vegetation of the Hay Plain: Booligal-Hay and Deniliquin-Bendigo 1:250 000 maps. Cunninghamia. 1993;3:1–122. [Google Scholar]
- Porteners MF, Ashby EM, Benson JS. The natural vegetation of Pooncarie 1:250 000 map. Cunninghamia. 1997;5:139–231. [Google Scholar]
- Prior LD, Lee Z, Brock C, Williamson GJ, Bowman DMJS. What limits the distribution and abundance of the native conifer Callitris glaucophylla (Cupressaceae) in the West MacDonnell Ranges, central Australia? Aust. J. Bot. 2010;58:554–564. [Google Scholar]
- Prior LD, McCaw WL, Grierson PF, Murphy BP, Bowman DMJS. Population structures of the widespread Australian conifer Callitris columellaris are a bio-indicator of continental environmental change. For. Ecol. Manage. 2011;262:252–262. [Google Scholar]
- Prober S, Hilbert D, Ferrier S, Dunlop M, Gobbett D. Combining community-level spatial modelling and expert knowledge to inform climate adaptation in temperate grassy eucalypt woodlands and related grasslands. Biodivers. Conserv. 2012;21:1627–1650. [Google Scholar]
- Quinn GP, Keough MJ. Experimental design and data analysis for biologists. Cambridge: Cambridge Univ. Press; 2002. [Google Scholar]
- Read J. Recruitment characteristics of the white cypress pine (Callitris glaucophylla) in arid South Australia. Rangeland J. 1995;17:228–240. [Google Scholar]
- Reimer PJ, Bailllie MGL, Bard E, Bayliss A, Warren BJ, Bertrand CJH, et al. IntCal04 terrestrial radiocarbon age calibration, 0–26 cal kyr BP. Radiocarbon. 2004;46:1029–1058. [Google Scholar]
- Richardson DM, Bond WJ. Determinants of plant distribution: evidence from pine invasions. Am. Nat. 1991;137:639–668. [Google Scholar]
- Ross KA, Lunt ID, Bradstock RA, Bedward M, Ellis MV. Did historical tree removal promote woody plant encroachment in Australian woodlands? J. Veg. Sci. 2012;23:304–312. [Google Scholar]
- Russell-Smith J, Edwards AC, Price OF. Simplifying the savanna: the trajectory of fire-sensitive vegetation mosaics in northern Australia. J. Biogeogr. 2012;39:1303–1317. [Google Scholar]
- Scott JA. The natural vegetation of the Balranald-Swan Hill area. Cunninghamia. 1992;2:597–652. [Google Scholar]
- Specht RL, Specht A. Australian plant communities: dynamics of structure, growth and biodiversity. Melbourne: Oxford Univ. Press; 1999. [Google Scholar]
- Squeo FA, Holmgren M, Jimenez M, Alban L, Reyes J, Gutierrez JR. Tree establishment along an ENSO experimental gradient in the Atacama desert. J. Veg. Sci. 2007;18:195–202. [Google Scholar]
- Tabachnick BG, Fidell LS. Using multivariate statistics. Boston: Pearson Education Inc; 2007. [Google Scholar]
- Thomas CD, Cameron A, Green RE, Bakkenes M, Beaumont LJ, Collingham YC, et al. Extinction risk from climate change. Nature. 2004;427:145–148. doi: 10.1038/nature02121. [DOI] [PubMed] [Google Scholar]
- Thompson WA, Eldridge DA. White cypress pine (Callitris glaucophylla): a review of its roles in landscape and ecological processes in eastern Australia. Aust. J. Bot. 2005;53:555–570. [Google Scholar]
- Thuiller W, Albert C, Araújo MB, Berry PM, Cabeza M, Guisan A, et al. Predicting global change impacts on plant species’ distributions: future challenges. Perspect. Plant Ecol. Evol. Syst. 2008;9:137–152. [Google Scholar]
- Tiver F, Andrew MH. Relative effects of herbivory by sheep, rabbits, goats and kangaroos on recruitment and regeneration of shrubs and trees in eastern South Australia. J. Appl. Ecol. 1997;34:903–914. [Google Scholar]
- Venables WN, Ripley BD. Modern applied statistics with S. New York, NY: Springer-Verlag; 2002. [Google Scholar]
- Walker BH. Rangeland ecology: understanding and managing change. Ambio. 1993;22:80–87. [Google Scholar]
- Watson IW, Westoby M, Holm AM. Demography of two shrub species from an arid grazed ecosystem in Western Australia 1983–93. J. Ecol. 1997a;85:815–832. [Google Scholar]
- Watson IW, Westoby M, Holm AM. Continuous and episodic components of demographic change in arid zone shrubs: models of two Eremophila species from Western Australia compared with published data on other species. J. Ecol. 1997b;85:833–846. [Google Scholar]
- Whipp RK. Albury, Australia: Charles Sturt University; 2009. Historical vegetation change in relation to forest management in the Pilliga State Forests of northern NSW, Australia. PhD. [Google Scholar]
- Wiegand K, Jeltsch F, Ward D. Minimum recruitment frequency in plants with episodic recruitment. Oecologia. 2004;141:363–372. doi: 10.1007/s00442-003-1439-5. [DOI] [PubMed] [Google Scholar]


