Abstract
The histone H3 variant, CENP-A, is normally assembled upon canonical centromeric sequences, but there is no apparent obligate coupling of sequence and assembly, suggesting that centromere location can be epigenetically determined. To explore the tolerances and constraints on CENP-A deposition we investigated whether certain locations are favoured when additional CENP-ACnp1 is present in fission yeast cells. Our analyses show that additional CENP-ACnp1 accumulates within and close to heterochromatic centromeric outer repeats, and over regions adjacent to rDNA and telomeres. The use of minichromosome derivatives with unique DNA sequences internal to chromosome ends shows that telomeres are sufficient to direct CENP-ACnp1 deposition. However, chromosome ends are not required as CENP-ACnp1 deposition also occurs at telomere repeats inserted at an internal locus and correlates with the presence of H3K9 methylation near these repeats. The Ccq1 protein, which is known to bind telomere repeats and recruit telomerase, was found to be required to induce H3K9 methylation and thus promote the incorporation of CENP-ACnp1 near telomere repeats. These analyses demonstrate that at non-centromeric chromosomal locations the presence of heterochromatin influences the sites at which CENP-A is incorporated into chromatin and, thus, potentially the location of centromeres.
Introduction
Centromeres are the chromosomal locations at which kinetochores, the machinery responsible for accurate chromosome segregation, are assembled. Kinetochores are assembled on unusual chromatin composed of nucleosomes in which canonical histone H3 is replaced by a centromere specific histone H3 variant, generally known as CENP-A. The incorporation of CENP-A is critical for providing the foundation for the assembly of kinetochores and thus specifying the location of active centromeres in most eukaryotes. Kinetochores are normally assembled on specific sequences such as centromeric repeat sequences (α-satellite in human) and these DNA sequences can direct the de novo assembly of functional kinetochores. Such analyses indicate that specific DNA sequences can direct events that promote CENP-A incorporation and kinetochore assembly. Although kinetochores are normally assembled upon these preferred, defined centromeric sequences in any particular organism, there is now an abundance of examples demonstrating that centromeric sequences are sometimes not sufficient and sometimes not necessary for kinetochore assembly. On human dicentric chromosomes, one centromere can be inactivated despite the continued presence of α-satellite sequence at both centromeric loci [1,2]. However, other observations indicate that specific DNA sequences are not required and that the location of CENP-A incorporation and kinetochore assembly is epigenetically regulated. For example, chromosome rearrangements that delete the normal centromere can allow kinetochores to assemble at novel locations, known as neocentromeres. At such neocentromeres, the underlying DNA sequence can be non-repetitive and bears no sequence similarity to the DNA elements associated with normal centromeres. Neocentromeres have been observed and characterised in a variety of organisms including humans, flies, yeasts and plants [3–10]. Moreover, in several species natural centromeres have been identified which are located at non-repetitive unique DNA sequences [11]. These centromeres are thought to represent recently established centromeres and presumably arise through the mechanisms that also lead to neocentromere formation.
Together such observations suggest that centromeres are normally assembled upon canonical centromeric sequences in any particular species, but there is no obligate coupling of sequence and assembly. These observations provide strong evidence for an epigenetic component to the regulation of centromere assembly. Several lines of evidence point to the histone H3 variant, CENP-A being the epigenetic mark that specifies centromere identity. It is found only at active centromeres, including neocentromeres and is absent from inactivated centromeres [1]. Tethering of CENP-ACID in Drosophila promotes incorporation of endogenous CENP-ACID, even after the tethered version is removed, suggesting that CENP-A chromatin is able to direct its own propagation [12]. This is also supported by experiments in human cells in which tethering of the CENP-A chaperone HJURP to LacO arrays led to CENP-A incorporation and kinetochore assembly at a non centromeric location [13].
The incorporation of CENP-A at novel sites could be influenced in various ways. CENP-A may be rapidly turned over to prevent its accumulation at non-centromeric locations [14,15]. Active processes such as transcription can promote H3 but block CENP-A incorporation [16]. However, particular chromatin features may provide an environment that favours the incorporation of CENP-A and its stabilisation in chromatin relative to canonical histone H3. Indeed the centromeric DNA elements on which CENP-A is naturally assembled, have been shown to be transcribed in a variety of organisms [17].
The three centromeres on fission yeast (Schizosaccharomyces pombe) chromosomes are composed of a central domain (cnt plus imr) flanked by repetitive DNA elements known as the outer repeats (otr). CENP-ACnp1 chromatin and the kinetochore are assembled over the central domain (~10 kb) while heterochromatin, in which histone H3 is methylated on lysine 9 (H3K9me) that allows the binding of specific chromodomain proteins, coats the outer repeats. Central domain DNA alone is unable to direct the de novo assembly of CENP-ACnp1 chromatin on this DNA following its introduction into cells. The flanking heterochromatin is required to provide an environment that somehow allows the replacement of histone H3 with CENP-ACnp1 and kinetochore assembly over the nearby central domain DNA [18,19]. CENP-ACnp1 might be attracted to non-centromeric sites by sequences possessing certain features, but also by chromatin context. The importance of chromatin context is demonstrated by the fact that heterochromatin is required for the establishment of CENP-ACnp1 chromatin on adjacent central core sequences, but once established this heterochromatin is not required for the maintenance of CENP-ACnp1 [18,19].
Apart from centromeres a domain of heterochromatin is formed over the 15 kb region encompassing the silent mating type loci and an extended region internal to telomeres. Heterochromatin is not essential in fission yeast and its formation at all locations is dependent on a single gene encoding the H3 K9 methyltransferase, Clr4. Non-coding transcripts that emanate from repetitive elements at centromeres, telomeres and the mating type locus, are processed to small interfering RNAs which direct H3K9 methylation by Clr4. This modification creates a binding site for the chromodomain protein Swi6. Histones H3 and H4 in the nucleosomes of outer repeat chromatin are underacetylated on lysines in their N-terminal tails and this hypoacetylated state results form the recruitment of the histone deacetylases: Clr3, Clr6 and Sir2 [20].
The engineered deletion of a fission yeast centromere (cen1) has been shown to allow neocentromeres to form at other chromosomal locations. At these neocentromeres CENP-ACnp1 is associated with DNA sequences that lack homology to the central domain of centromeres, where CENP-ACnp1 is normally incorporated. All neocentromeres formed were within 100 kb of either the left or right telomeres. Heterochromatin is associated with fission yeast telomeres and in the absence of heterochromatin the frequency of neocentromere formation was reduced [6].
Fission yeast telomeres are composed of ~300 bp of terminal GT rich repeats that are added by telomerase, internal to these are telomere associated sequences (TAS) on chromosome 1 and 2 [21]. These TAS repeat elements share a short region of homology with centromeric outer repeats [22,23]. The terminal TTAC(A) GG(G1-4) repeats are bound by specific proteins that form a specialised structure that regulates telomerase recruitment, protects chromosomal ends from degradation and anchors them at the nuclear periphery [24]. Telomere repeat length is regulated by telomere associated proteins that are recruited by Taz1, which directly binds double stranded (ds) repeats [25], and Pot1 which binds the single stranded (ss) overhang at the extreme end [26]. Taz1 recruits Rap1 and Rif1 to double stranded telomeric DNA [27,28], whereas Pot1 recruits Tpz1 to the single stranded G tail [29]. In complex with two other factors, Ccq1 (which interacts with Pot1-Tpz1 alone) and Poz1 (which bridges the ss and ds regions of telomeres through direct interactions with Pot1-Tpz1 as well as Rap1), these proteins regulate telomere length and protect the ends of chromosomes [29]. Ccq1 also associates with the SHREC/Clr3 histone deacetylase complex and recruits it to telomeres where it participates in silencing [30].
Taz1 has been shown to inhibit telomerase-mediated telomere elongation, by ensuring cell cycle dependent telomerase recruitment to telomeres [31,32]. Rap1 and Rif1, which associate with Taz1, have also been found to negatively regulate telomere length, both in a Taz1 dependent and independent manner [27,28,33]. Conversely, Pot1 and its associated factors Tpz1 and Ccq1 have been shown to promote telomere elongation and are required for stable telomere maintenance [26,29,34]. In particular, Ccq1 has also been shown to directly interact with telomerase and recruit it to telomeres [35].
To explore the tolerances and constraints on CENP-ACnp1 deposition we investigated whether certain locations are favoured by CENP-ACnp1 when present in excess. We find that when CENP-ACnp1 is moderately overexpressed in fission yeast it accumulates in telomeric regions, the same regions that can form neocentromeres. Here we use engineered telomeres on minichromosomes, integrated arrays of telomeric repeats and cells lacking telomere and heterochromatin proteins to investigate what features promote CENP-ACnp1 incorporation in the telomeric regions of chromosomes. Our analyses show that a short array of telomere repeats, independent of a chromosome end, is sufficient to promote CENP-ACnp1 incorporation and that Ccq1 plays a specific role in promoting the formation of nearby heterochromatin and CENP-ACnp1 incorporation.
Results
Additional CENP-ACnp1 accumulates at ectopic sites
CENP-ACnp1 is normally restricted to the central domain of fission yeast centromeres. However, our previous analyses show that CENP-ACnp1 is able to assemble on non-centromeric DNA inserted within the central domain [36]. Moreover, moderate overexpression of CENP-ACnp1 causes increased repression of a marker gene inserted in the central core and increased levels of CENP-ACnp1 on that marker gene. It has also been shown that the deletion of an endogenous centromere (cen1) results in CENP-ACnp1 accumulating over an extended euchromatic region adjacent to telomeres, forming neocentromeres [6]. These observations suggest that although CENP-ACnp1 is normally restricted to the central domain at centromeres, it can assemble upon non-centromeric sequences.
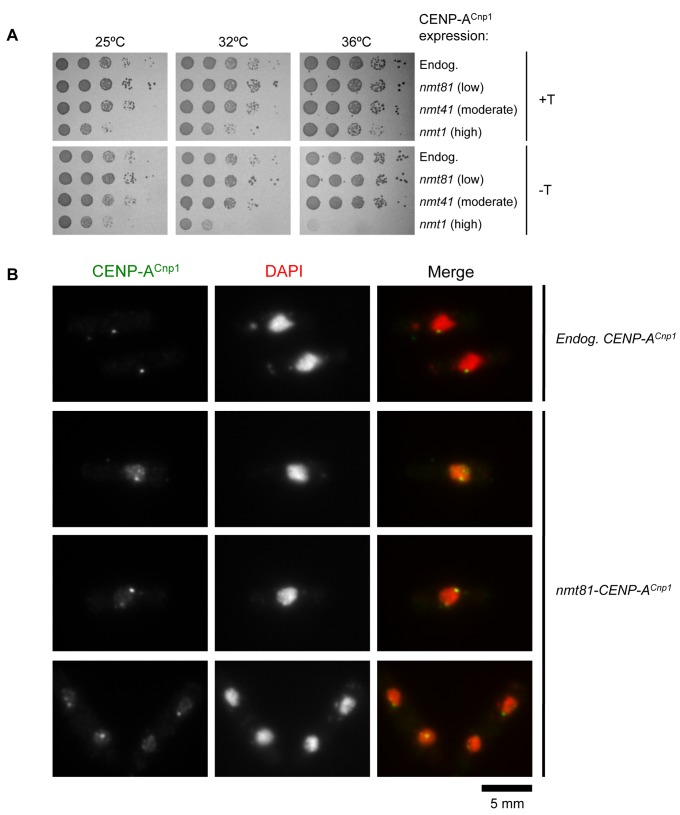
In order to further investigate the constraints or promiscuity of CENP-ACnp1 deposition we expressed additional CENP-ACnp1 at a variety of levels and conditions. The CENP-ACnp1 open reading frame was expressed from different strengths of the thiamine repressible nmt promoter (nmt81 – weak, nmt41 – medium, nmt1 – strong) [37], integrated at the same chromosomal location (this work and [16,36], Figures S1A and S1B). The effect on cell growth and viability was assessed at different temperatures in serial dilution assays (Figure 1A). Whilst expression of moderate or medium additional amounts of CENP-ACnp1 was tolerated, strong overexpression had a highly deleterious effect on cell growth, especially at 36oC.
Figure 1. Additional CENP-ACnp1 accumulates at discrete foci in S. pombe cells.
CENP-ACnp1 was expressed from different strengths of the thiamine repressible nmt promoter (nmt81 – weak, nmt41 – medium, nmt1 – strong); integrated at ars1 in chromosome 1. A strain that has an empty nmt promoter plasmid integrated was used as a control (Endog: CENP-ACnp1 endogenous levels). (A) Serial dilution assays to monitor the effect of additional CENP-ACnp1 on cell growth. Cells were spotted on minimal media with (+T) or without (-T) thiamine at three different temperatures, 25°C, 32°C and 36°C. (B) Immunolocalization of CENP-ACnp1 in cells expressing endogenous levels of CENP-ACnp1 or extra CENP-ACnp1 expressed from the nmt81 promoter. Cells were grown in the absence of thiamine for 24 h at 36oC and stained with anti-CENP-ACnp1 antibody (green) and DAPI (DNA: red). Scale bar, 5 µm.
To determine whether the observed effect on growth was accompanied by deposition of CENP-ACnp1 at non-centromeric loci, CENP-ACnp1 localisation was examined in cells expressing additional CENP-ACnp1 from the three types of promoters. During interphase, the three fission yeast centromeres are clustered adjacent to the spindle pole body (SPB; centrosome equivalent) and appear as a single dot (Figure 1B). When overexpressed at low levels, although the signal intensity was increased, a single CENP-ACnp1 dot was visible at 25oC. In contrast, at 36oC additional CENP-ACnp1 foci were observed that were distinct from the main centromeric cluster (Figure 1B). The most common pattern observed was a few (2–6) bright foci of staining in addition to the centromeric spot. In addition, a general increase of staining of all chromatin, but not the entire nuclear volume, was also observed under moderate or high expression conditions (Figure S1C), suggesting that CENP-ACnp1 was incorporated to some extent more broadly throughout chromatin. These observations indicate that when CENP-ACnp1 is overexpressed it can be deposited at chromatin contexts distinct from centromeres and this is dependent on the level of expression.
Additional CENP-ACnp1 accumulates on chromatin at pericentromeres, and adjacent to telomeres and rDNA
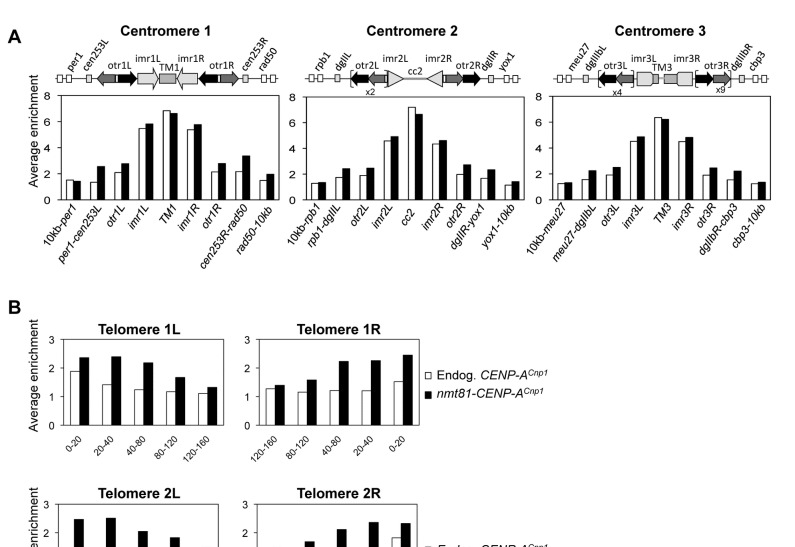
To accurately determine the location of CENP-ACnp1 in cells that display strong extra foci with low background chromatin signal, anti-CENP-ACnp1 genome-wide chromatin immunoprecipitation (ChIP-chip) analysis was performed on cells with low CENP-ACnp1 overexpression (nmt81-CENP-A Cnp1) and cells with wild-type CENP-ACnp1 levels (36oC; see Materials and Methods and Figure 1B). As expected, CENP-ACnp1 was observed at the central domains of all three fission yeast centromeres (Figure 2A) in both cell types.
Figure 2. Additional CENP-ACnp1 is deposited at sub-telomeric and pericentromeric regions.
ChIP-chip analysis of CENP-ACnp1 levels at cells with moderate expression of additional CENP-ACnp1 (nmt81-CENP-ACnp1) or wild-type protein levels (Endog. CENP-ACnp1). (A) Average enrichment of CENP-ACnp1 over different chromosomal regions (cnt, imr, otr) of centromeres I, II and III (X-axis, linear scale). The number of repetitive elements at different loci is indicated (x2, x4, x9). The average enrichment over 10 kb regions immediately adjacent to the centromeric otr repeats is also shown (10 kb region encompassing the most proximal annotated gene on each side of each centromere; per1 and rad50 for cen1, rpb1 and yox1 for cen2 and meu27 and cbp3 for cen3). (B) Average enrichment from ChIP-chip analyses of CENP-ACnp1 over 120 kb regions from the left or right telomere of each chromosome. Signal intensity was averaged within 20 kb or 40 kb windows as indicated. Left panels, left telomeres; right panels, right telomeres of each chromosome.
CENP-ACnp1 is normally restricted to the central domain of centromeres and does not occupy the heterochromatic centromere outer repeats [36,38]. Examination of the ChIP-chip data indicates that additional CENP-ACnp1 accumulated on the outer repeats and pericentromere regions (Figure 2A and Figure S2A), and this was verified by qPCR and duplex PCR using cells expressing low (nmt81) or moderate (nmt41) levels of CENP-ACnp1 (Figure 3A Figure S2B). Interestingly, in cells expressing additional CENP-ACnp1, the kinetochore protein CENP-CCnp3 was also detected on the centromeric outer repeats (Figure 3A). Normally the outer repeats are packaged in chromatin marked by H3K9 methylation which is dependent on the Clr4 histone methyltransferase [20,39] (Figure 3B). Additional CENP-ACnp1 is still detected over the centromere outer repeats in a clr4Δ mutant (Figure 3B). These analyses suggest that additional CENP-ACnp1 can be attracted to centromeric regions by the presence of CENP-ACnp1 and associated kinetochore proteins within the central domain.
Figure 3. CENP-ACnp1 can accumulate on centromeric repeat regions independently of H3K9 methylation.
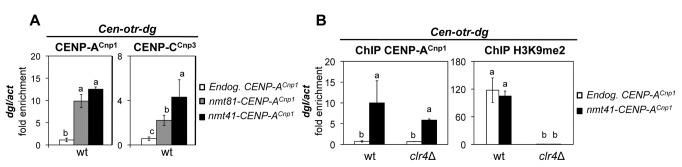
(A) ChIP-qPCR-analysis of CENP-ACnp1 and CENP-CCnp3 levels on centromeric outer repeat sequences (otr dgI) in cells expressing endogenous (Endog. CENP-ACnp1) or additional (nmt81-CENP-ACnp1 or nmt41-CENP-ACnp1) levels of CENP-ACnp1. (B) ChIP-qPCR analysis of CENP-ACnp1 and H3K9me2 levels on centromeric otr dgI repeats in wild-type and clr4∆ cells expressing endogenous (Endog. CENP-ACnp1) or additional levels (nmt41-CENP-ACnp1) of CENP-ACnp1. Enrichment on dgI was normalized to the signal obtained for the euchromatic gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
Strikingly, in cells expressing additional CENP-ACnp1 the levels of CENP-ACnp1 are elevated over an extended region adjacent to the left and right telomeres of chromosomes 1 and 2 (Figure 2B and Figure S2A). These subtelomeric domains of CENP-ACnp1 enrichment encompassed ~100 kb in a series of discrete peaks that were mainly located at intergenic regions. Four S. pombe telomeres (chromosomes 1 and 2) have telomere-associated sequences (TAS) internal to the TTAC(A) GG(G1-4) repeats [21]. Arrays of rDNA directly abut the telomeres of chromosome 3 and increased CENP-ACnp1 levels were observed on the unique DNA immediately internal to the rDNA (Figure 2B and Figure S2A). This increased incorporation of CENP-ACnp1 at subtelomeric regions and adjacent to rDNA was verified by duplex PCR (Figure S2C) and qPCR (Figure S2D).
These analyses show that when expressed in excess, the normally exclusively centromeric histone CENP-ACnp1 is deposited at additional loci including centromeric outer repeats / pericentromeric regions, adjacent to rDNA and over an extended region internal to telomeres.
Telomere repeats alone are sufficient to promote CENP-ACnp1 incorporation nearby
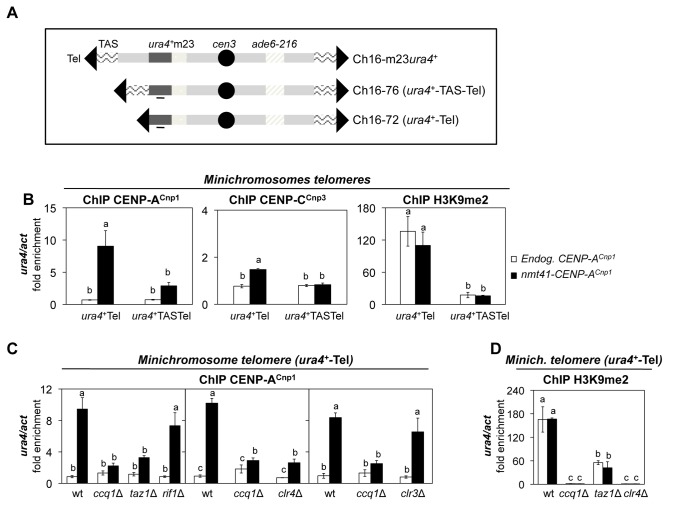
The natural telomeres on fission yeast chromosomes 1 and 2 are composed of a terminal ~300 bp of terminal telomeric repeats (TTAC(A) GG(G1-4)) internal to which are copies of telomere associated sequence (TAS) repeat elements [21]. Part of the TAS regions share homology with centromeric otr repeats [22,23]. To determine if TAS or terminal repeats are required to attract CENP-ACnp1 to domains of telomeric chromatin, we utilised versions of a well characterised minichromosome Ch16 in which new telomeres were created adjacent to a ura4 + marker gene by telomere-mediated chromosome breakage at the m23 locus [40]. On Chs 16–76, 1.2 kb of TAS DNA lies between the terminal telomeric repeat array and the ura4 + gene (ura4 + -TAS-Tel), whereas on Chs 16–72 the terminal telomere repeat array lies directly adjacent to the ura4 + gene (ura4 + -Tel) (Figure 4A). These minichromosomes are non-essential, allowing monitoring of events that might be lethal if they occurred on endogenous chromosomes. The placement of the ura4 + gene next to telomeric sequences allows chromatin-mediated silencing to be monitored and provides unique primer sites for qPCR-ChIP analyses on the Ch16 minichromosome which is otherwise identical in sequence to a ~530 kb section of chromosome 3 encompassing cen3.
Figure 4. Terminal telomere repeats alone can attract CENP-ACnp1 in cells expressing additional CENP-ACnp1.
(A) Schematic representation of Ch16-m23 minichromosome derivatives with the ura4 + gene inserted 50 kb from the minichromosome end (Ch16-m23ura4 +) or adjacent to telomere-associated sequences at a telomere (ura4 +-TAS-Tel, Chs 16–76) or directly abutting terminal telomeric repeats (ura4 +-Tel, Chs 16–72). Black bar indicates the region at ura4 + analyzed by ChIP-qPCR. (B) ChIP-qPCR of CENP-ACnp1, CENP-CCnp3 and H3K9me2 levels on ura4 + in cells containing the ura4 +-Tel or ura4 +-TAS-Tel Ch16 minichromosomes and expressing endogenous (Endog. CENP-ACnp1) or additional CENP-ACnp1 levels (nmt41-CENP-ACnp1) (C) ChIP-qPCR of CENP-ACnp1 levels on ura4 + in wild-type (wt) or mutant cells (ccq1∆, taz1∆, clr4∆ and clr3∆) that contain the ura4 +-Tel or ura4 +-TAS-Tel Ch16 minichromosomes and express endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1 (D) ChIP-qPCR of CENP-ACnp1 and H3K9me2 levels at ura4 + in wild-type (wt) or mutant cells (ccq1∆, taz1∆ and clr4∆) that contain the ura4 +-Tel and express endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. For all ChIP analyses, enrichment on ura4 + was normalized the signal obtained for the gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
When additional CENP-ACnp1 was expressed in cells bearing these minichromosomes, increased levels of CENP-ACnp1 were detected on the ura4 + gene. Significantly more CENP-ACnp1 was detected on ura4 + when it resided immediately adjacent to the terminal telomere repeat (ura4 + -Tel) relative to when TAS sequences were included (ura4 + -TAS-Tel) (Figure 4B and Figure S3A). Moreover, the CENP-CCnp3 kinetochore protein was also enriched at ura4 + -Tel in cells expressing additional CENP-ACnp1 (Figure 4B and Figure S3A). The fact that CENP-ACnp1 is detected on ura4 + -Tel indicates that TAS elements are not required to attract CENP-ACnp1 to telomeric regions. The diminished deposition of additional CENP-ACnp1 on the TAS-adjacent ura4 + could be due to an inhibitory effect of TAS or simply due to the difference in distance of ura4 + from the telomeric repeats in Chs 16–72 versus Chs 16–76. Nonetheless, CENP-ACnp1 and CENP-CCnp3 are clearly attracted to telomeric adjacent regions independently of TAS elements.
Heterochromatin driven by H3K9 methylation coats the outer repeats at centromeres, a region containing the silent mating type genes, and the regions adjacent to telomeres. To determine whether the presence of heterochromatin influences CENP-ACnp1 deposition at telomeres, anti-H3K9me2 ChIP was performed. Our analyses show that H3K9me2 is enriched on ura4 + -Tel, but only weakly enriched on ura4 + -TAS-Tel when compared to the enrichment found at outer repeats (Figure 4B and Figure S3B). Thus, CENP-ACnp1 is incorporated in regions close to a telomere where high levels of H3K9me2 are also present.
The presence of an active ura4 + gene (Ch16-m23-ura4 +) allows growth in the absence of uracil (–ura) and no growth on media containing counter-selective FOA (Figure S4). When placed next to telomeres the ura4 + gene is transcriptionally silenced by telomeric heterochromatin as indicated by enhanced growth of Chs 16–72 (ura4 + -Tel) and Chs 16–76 (ura4 + -TAS-Tel) on FOA plates and/or reduced growth on –uracil plates (Figure S4). The degree of silencing is correlated with H3K9 methylation: ura4 + -TAS-Tel exhibits low levels of H3K9me2 and weak silencing whereas ura4 + -Tel displays high levels of H3K9me2 and stronger silencing (Figure 4B and Figure S4). The lack of heterochromatin may affect the ability of ura4 + -TAS-Tel to incorporate CENP-ACnp1. Indeed expression of additional CENP-ACnp1 enhanced silencing of telomere-adjacent ura4 + -Tel (reduced growth on –ura plates: Figure S4) which is consistent with the increase in CENP-ACnp1 levels detected (Figure 4B), but did not enhance silencing of TAS-adjacent ura4 + -TAS-Tel.
The incorporation of CENP-ACnp1 near telomeres requires Ccq1 and Clr4
Telomeres are bound by several proteins that control telomere length homeostasis, by regulating the recruitment of telomerase [24]. Loss of Taz1, Rap1, or Rif1 results in elongated telomeres [25,27,28] whereas loss of Ccq1, which is involved in telomerase recruitment, results in shortened telomeres [29,34,35]. Ccq1 also associates with the SHREC complex that contains the Clr3 HDAC, and cooperates with Taz1 in recruiting SHREC to telomeres [30].
To investigate the requirements for the incorporation of CENP-ACnp1 near telomeres additional CENP-ACnp1 was expressed in mutants that affect telomere structure and function. The level of CENP-ACnp1 associated with ura4 + -Tel on the minichromosome was markedly reduced in cells lacking Clr4 or Ccq1 relative to wild-type cells (Figure 4C). Cells lacking Rif1 or Clr3 incorporated CENP-ACnp1 on ura4 + -Tel at levels similar to wild-type (Figure 4C), whereas taz1Δ cells displayed intermediate levels of CENP-ACnp1 incorporation. In all cell types analysed (wt, ccq1∆, rif1∆, taz1∆, clr4∆, clr3∆) the observed incorporation of CENP-ACnp1 on the centromeric outer repeats and the central core was unaffected (Figures S5A and S5B), thus indicating that Clr4 and Ccq1, and to a lesser extent Taz1, play a specific role in promoting CENP-ACnp1 deposition near telomeres.
ChIP was also performed to determine the level of H3K9me2 at ura4 + -Tel in these different backgrounds. As expected, all H3K9me2 was lost in clr4∆ cells (Figure 4D). In taz1∆ cells there was some reduction in the level of H3K9me2 at ura4 + -Tel, which might be a consequence of having highly elongated telomeres. Surprisingly, in ccq1∆ cells, H3K9me2 was completely lost from ura4 + -Tel but retained on centromere repeats (Figures 4D and S5C). This observation suggests that the incorporation of CENP-ACnp1 at telomeres is linked to the loss of telomeric H3K9 methylation.
A chromosome end is not required to attract additional CENP-ACnp1
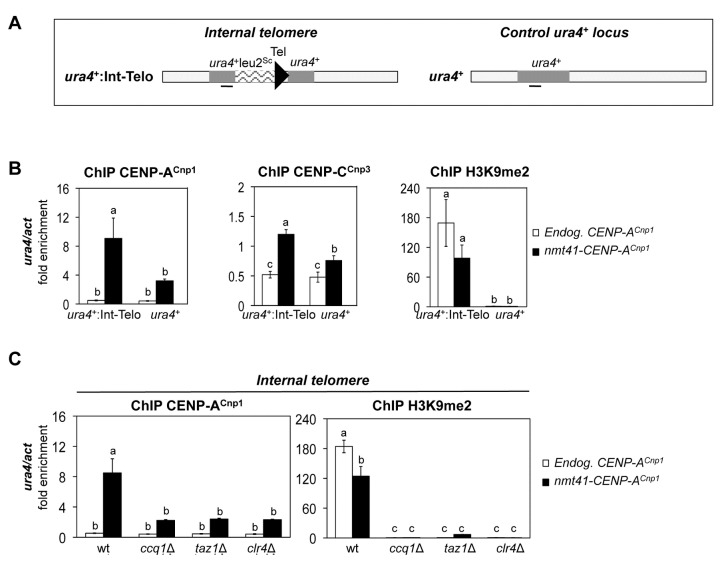
Chromosome ends have a distinct structure that recruits a plethora of factors that could influence the incorporation of CENP-ACnp1 adjacent to telomere repeats. To determine whether the deposition of CENP-ACnp1 near telomeric repeats requires proximity to a chromosome end we examined the association of CENP-ACnp1 adjacent to a 300 bp array of telomeric repeats inserted adjacent to the ura4 + gene within a chromosome arm (Figure 5A [41]). In cells expressing additional CENP-ACnp1, elevated levels of CENP-ACnp1 and CENP-CCnp3 were readily detected adjacent to these interstitial telomere repeats but not the control ura4 + locus alone (Figure 5B), whereas the levels of CENP-ACnp1 on the centromeric outer repeats and the central core was similar in both strains (Figure S6). Moreover, the incorporation of CENP-ACnp1 again correlated with the presence of H3K9me2 in that H3K9me2 was readily detected at ura4 +: Int-Telo (Figure 5B). We conclude that proximity to a chromosome end is not required to promote CENP-ACnp1 deposition.
Figure 5. Telomeric repeat arrays promote CENP-ACnp1 deposition when inserted at an internal chromosomal location.
(A) Schematic representation of the genomic region of S. pombe chromosome III where an array of telomeric repeats was inserted into the ura4 + gene (Internal telomere: ura4 +: Int-Telo) [41]. Black bar indicates the region at ura4 + analyzed by ChIP-qPCR. (B) ChIP-qPCR of CENP-ACnp1, CENP-CCnp3 and H3K9me2 levels on ura4 + in control cells (ura4 +) or cells with telomeric repeats integrated at ura4 + (ura4 +: Int-Telo) and that express endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. (C) ChIP analysis of CENP-ACnp1 and H3K9me2 levels on ura4 +: Int-Telo (qPCR1) in wild-type (wt) or mutant cells (ccq1∆, taz1∆, and clr4∆) expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. For all ChIP analyses, enrichment on ura4 + was normalized to the signal obtained for the gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
We next examined the level of H3K9me2 and the incorporation of CENP-ACnp1 near internal telomere repeats (ura4 +: Int-Telo) in mutant cells expressing additional CENP-ACnp1. In clr4∆, taz1∆ and ccq1∆ cells the level of CENP-ACnp1 at ura4 +: Int-Telo was dramatically reduced compared to the wild type control and the reduced deposition of CENP-ACnp1 correlates with the loss of H3K9me2 at ura4 +: Int-Telo observed in these mutants (Figure 5C). CENP-ACnp1 and H3K9me2 levels at centromeres are not affected in mutants with defective telomere proteins (Figure S7). These analyses suggest that the telomere binding proteins promote the formation of H3K9me-dependent heterochromatin near telomeric repeats and, as at centromeres, this heterochromatin permits the incorporation of surplus CENP-ACnp1.
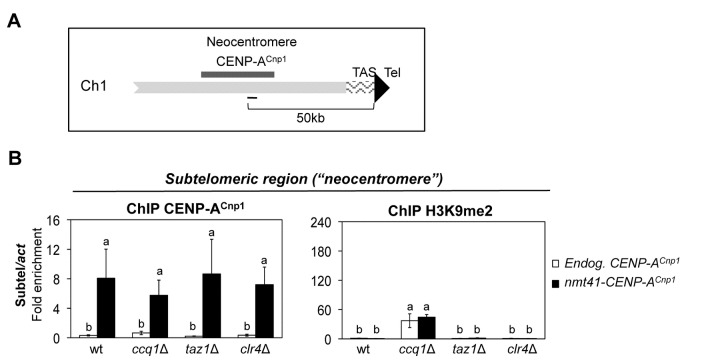
Additional CENP-ACnp1 is attracted independently of heterochromatin to regions capable of neocentromere formation
Fission yeast neocentromeres have been shown to form over a ~ 40 kb domain that is positioned 40-60 kb from chromosome ends [6]. These neocentromeres overlap with the 100 kb region near telomeres where we observe incorporation of CENP-ACnp1 when expressed at elevated levels (Figure 6A and Figure S2A). In contrast to the situation at synthetic telomeres at the ends of minichromosomes or internal telomere repeat arrays, CENP-ACnp1 remains detectable in these neocentromere regions in all mutants analysed, including clr4∆ cells where H3K9 methylation and heterochromatin is completely lost (Figure 6B [23]). Thus, the incorporation of CENP-ACnp1 over these potential neocentromere regions is not dependent on the presence of heterochromatin. It is likely that endogenous sub-telomeric regions possess features that attract CENP-ACnp1 by a route that is independent of heterochromatin and telomere-binding proteins, although these features are also able to attract CENP-ACnp1.
Figure 6. CENP-ACnp1 is deposited on subtelomeric regions of natural chromosomes independently of heterochromatin.
(A) Schematic representation of the right subtelomeric region of S. pombe chromosome I that has been shown to form neocentromeres (thick black line) [6]. Thin black line marks the region analyzed by ChIP-qPCR. (B) ChIP-qPCR of CENP-ACnp1 and H3K9me2 levels associated with the subtelomeric region of chromosome I in wild-type (wt) or mutant cells (ccq1∆, taz1∆, and clr4∆) expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. Enrichment at the sub-telomere region (Subtel) was normalized to the signal obtained for the gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.05).
Discussion
In this study we set out to investigate what factors influence the sites where CENP-A can be incorporated when its levels are elevated. Normally CENP-A is deposited exclusively at centromeres where it forms the basis for assembly of the kinetochore. However, there is strong evidence that CENP-A incorporation can be epigenetically determined and that the normally preferred centromeric sequences are neither necessary nor sufficient for CENP-A deposition in certain circumstances. By expressing additional CENP-ACnp1 in fission yeast we aimed to gain insight into which genomic regions, apart from centromeres, are preferred. This could provide clues to the enigma of neocentromere formation. What constitutes pre-centromeric sites i.e. what features are associated with the potential locations of neocentromeres? Is sequence important or chromatin context or both?
Unlike in S. cerevisiae and Drosophila , CENP-ACnp1 can be overexpressed in fission yeast and thus its levels do not appear to be subject to tight regulation by ubiquitin-mediated degradation [14,15,42]. When the levels of CENP-ACnp1 are raised by a modest amount we find that CENP-ACnp1 accumulates at subtelomeric and pericentromeric regions. Thus, as has been demonstrated in some other species, there is flexibility with respect to the chromosomal locations at which fission yeast CENP-ACnp1 assembles. The CENP-CCnp3 kinetochore protein also becomes localised at some sites where additional CENP-ACnp1 is incorporated, suggesting that these sites have the potential to become functional ectopic kinetochores. The toxicity associated with high levels of CENP-ACnp1 overexpression (Figure 1A) might result from formation of ectopic kinetochores and/or global effects on transcriptional regulation [16]. In Drosophila , overexpression of CENP-ACID also results in recruitment of CENP-C and formation of ectopic kinetochores near telomeres and pericentromeric regions [8,9]. As in S. pombe, heterochromatin influences recruitment of CENP-ACID in Drosophila . In a recent study in Saccharomyces cerevisiae, overexpression of CENP-ACse4 led to its incorporation at additional regions of the genome, termed Centromere-Like-Regions (CLRs). Although budding yeast lacks methyl-H3K9 heterochromatin, many CLRs were found in the regions surrounding centromeres [43]. CENP-A proteins also show a preference for transcribed regions in S. cerevisiae and S. pombe [16,17,43,44]
To investigate the preferential incorporation of additional CENP-ACnp1 at subtelomeric regions we utilised minichromosomes with synthetic telomeres. In cells with elevated CENP-ACnp1, CENP-ACnp1 is incorporated on ura4 + placed adjacent to a terminal array of telomeric repeats (ura4 + -Tel) that lack an intervening TAS element. Thus, TAS sequences are not required to attract CENP-ACnp1 to telomeric regions. As these Ch16-based minichromosomes also lack the 100 kb sub-telomeric region where neocentromeres form, we conclude that these regions are not necessary to attract CENP-ACnp1 to telomeres (but see below). Our finding that telomere repeats inserted at an internal chromosomal location can also attract CENP-ACnp1 indicates that proximity to a chromosome end is not a requirement; thus the specialised structure that caps chromosome ends and involves many telomere associated proteins is not needed. At natural telomeres, heterochromatin formation is dependent on Clr4 methyltransferase which is recruited directly by terminal telomere repeats, and by the action of RNAi on regions of the subtelomeric repeats that are homologous to centromeric outer repeats [23]. At the synthetic ura4 + -Tel telomere and ura4 +: Int-Telo internal telomere repeats, the heterochromatin-specific mark H3K9me2 is dependent on both Clr4 and Ccq1. However, the other components of the SHREC complex do not seem to be involved as lack of the HDAC subunit Clr3 does not affect CENP-ACnp1 deposition (Figure 4C). Thus, unexpectedly, we find that Ccq1 is required to induce H3K9me2 at arrays of TTAC(A) GG(G1-4) telomere repeats regardless of their location or the presence of TAS elements. These analyses suggest that Ccq1 has a role in recruiting Clr4 via telomeric repeats and that it is the resulting heterochromatin that promotes the incorporation of CENP-ACnp1.
Consistent with a role for heterochromatin in promoting CENP-ACnp1 deposition we also detected CENP-ACnp1 accumulation on the heterochromatic outer repeats at centromeres in cells expressing additional CENP-ACnp1. However, unlike at telomeres, when heterochromatin is lost (clr4∆), CENP-ACnp1 is still incorporated on these outer repeats. It is likely that at centromeres the presence of CENP-ACnp1 chromatin, and associated loading factors promotes the incorporation of this additional CENP-ACnp1. Barrier function has been ascribed to tRNA genes that lie between heterochromatin and CENP-ACnp1 chromatin domains at centromeres [38]. The presence of CENP-ACnp1 on the outer repeats in cells expressing elevated levels suggests that these barriers can be overcome so that CENP-ACnp1 can spread beyond the tRNA genes into the flanking heterochromatin region (Figure 2A Figures S2A and S2B).
It is clear that heterochromatin and telomere-associated proteins are required to recruit additional CENP-ACnp1 at synthetic telomeres. However, the situation is more complex at natural telomeric regions. Our ChIP analyses indicate that additional CENP-ACnp1 is incorporated in a 100 kb sub-telomeric domain in wild-type cells. These regions overlap with the regions that have been shown to become neocentromeres [6]. Although neocentromere formation is reduced in heterochromatin-deficient mutants, it is not abolished. Our analyses indicate that heterochromatin is not required for incorporation of CENP-ACnp1 within this sub-telomeric domain (Figure 6). Thus, this region of the genome appears to have an inherent ability to attract CENP-ACnp1. Particular combinations of features, such as proximity to a chromosome end, telomeric repeat sequences, telomere-specific proteins, heterochromatin, subtelomeric regions with homology to the centromeric outer repeats, telomere associated sequences (TAS), association with the nuclear periphery, or even sequence-specific properties of these 100 kb sub-telomeric regions may be responsible for attracting CENP-ACnp1 to regions near chromosome ends.
In S. pombe, heterochromatin is required for establishment, but not maintenance, of CENP-ACnp1 chromatin on adjacent central domain sequences [18]. Here, we have investigated the involvement of heterochromatin in attracting CENP-ACnp1 to other regions of the genome and find that it is sufficient but not necessary for ectopic localisation of CENP-ACnp1. In Drosophila , overproduced CENP-ACID is also attracted to heterochromatic loci [9]. How does heterochromatin promote deposition of CENP-ACnp1 on adjacent/overlapping sequences? One possibility is that the chromatin environment that heterochromatin engenders – such as histone hypoacetylation or transcriptional features - influences CENP-ACnp1 deposition. Alternatively, CENP-ACnp1 incorporation might be a consequence of the compartmentalisation of heterochromatin. Centromeres are clustered at the spindle pole body (SPB), whilst telomeres are located at the nuclear periphery. This tethering constrains the nuclear volume that they explore, increasing the likelihood that they will come into contact with centromeres. It is likely that a domain around the centromeres / SPB contains high concentrations of CENP-ACnp1 and assembly factors. Thus, telomeres and other heterochromatic loci may be favoured sites for CENP-ACnp1 assembly simply because they frequently come into contact with centromeres. Both centromeres and subtelomeres interact with the same inner membrane protein, Man1, at the nuclear periphery [45]. These interactions could favour CENP-A deposition at subtelomeres.
With the exception of organisms with holocentric chromosomes, it is essential that only one kinetochore is assembled per chromosome; dicentric chromosomes are inherently unstable. On the other hand, the existence of neocentromeres indicates that CENP-A is able to assemble on non-centromeric sequences. Indeed, the formation of neocentromeres is thought to be important in genome evolution and speciation [46]. Thus CENP-A must have the ability to be normally specific in its deposition requirements, but occasionally promiscuous. The factors that influence the constraints and permissiveness of CENP-A deposition are clearly complex, but our study indicates that heterochromatin is able to promote CENP-A deposition.
Materials and Methods
Cell growth and manipulation
Standard genetic and molecular techniques were followed. Fission yeast methods were as described [47]. For the strains used in the experiments, see Supporting information (Table S1). The CENP-ACnp1 open reading frame was cloned into three plasmids containing different strengths of the thiamine repressible nmt promoter, pREP81X (pREP81-cnp1+), pREP41X (pREP41-cnp1+ [16], or pREP3X (pREP3-cnp1+). The constructs were linearized using MluI and integrated at ars1 locus. The Ch16 minichromosomes derivatives used in this work were described in [40]. The strain carrying an array of telomeric repeats inserted adjacent to the ura4 + gene within chromosome arm (ura4 +: Int-telo) is described elsewhere [41]
Growth of cells overexpressing CENP-ACnp1 for ChIP analyses
Cells expressing additional CENP-ACnp1 from integrated pREP81-cnp1 + (nmt81-CENP-A Cnp1), pREP41-cnp1 + (nmt41-CENP-A Cnp1) or cells with integrated empty vector were initially grown in rich medium which contains thiamine to repress the expression of additional CENP-ACnp1. Cells were grown in PMG liquid medium (with thiamine) at 25°C and shifted to 36°C in PMG lacking thiamine for 24 h to allow CENP-ACnp1 expression before ChIP analysis. Strains carrying Ch16 minichromosome derivatives were grown in media lacking adenine to select for the minichromosome.
ChIP
ChIP was performed as described [36] using anti-CENP-ACnp1 antibody (10 µl of anti-CENP-ACnp1 antibody per 300 µl chromatin extract), anti-CENP-CCnp3 antibody (10 µl of anti-CENP-CCnp3 antibody per 300 µl chromatin extract) and anti-H3K9me2 antibody (1 µl of mAb 5.1.1 (a kind gift of Takeshi Urano antibody per 300 µl chromatin extract) and subsequently analyzed by quantitative PCR (qPCR) or by multiplex PCR. Table S2 lists primers used in qPCR and Table S3 lists primers used in multiplex PCR. Bars from ChIP-qPCR data represent the means +/- standard deviation (SD) from three independent biological replicates. Mean values marked with the same letter (a, b or c) indicate results not significantly different from each other, as established by one Way ANOVA and Holm-Sidak test for multiple comparisons (P, 0.01) using SigmaPlot 11 software (Systat Software Inc.)
ChIP–chip
DNA was immunoprecipitated as described earlier using 10 µl of anti-CENP-ACnp1 antibody per 100 µl chromatin extract [36]. For analysis on GeneChip S. pombe Tiling 1.0FR Arrays (Affymetrix) 5 mM dUTP was added to the second round of DNA amplification. Fragmentation, labelling and hybridization were performed by the Affymetrix core facility at Karolinska Institiutet (BEA) using standard protocols (http://www.affymetrix.com). Raw data from Affymetrix (.CEL format) were normalized with Affymetrix Tiling Analysis Software (TAS) v 1.1 and analyzed and visualized using Integrated Genome Browser (IGB [48],. Normalized signal of each probe (25nt) were obtained from Tiling Arrays Software (TAS; Affymetrix) according to [49]. For Figure 2, probe values were averaged to give one value for one genomic region.
The microarray data from this publication have been submitted to the GEO database [http://www.ncbi.nlm.nih.gov/geo/] and assigned the accession number GSE46427.
Cytology
Immunolocalization was performed as described [50]. Cells were grown in PMG liquid medium (with thiamine) at 25°C and shifted to 36°C for 24 h to allow CENP-ACnp1 expression before fixation. Cells were fixed for 7 min in 3.7% formaldehyde. The following antibodies were used: sheep CENP-ACnp1 antiserum (1:2000) and Alexa-488-coupled donkey anti-sheep secondary antibody (1:1000) (Invitrogen – Life Technologies). Microscopy was performed as described [50] using a Zeiss Imaging 2 microscope (Zeiss, http://www.zeiss.com). Image acquisition was controlled using Metamorph software (Universal Imaging Corporation, http://www.moleculardevices.com). For comparison of cells expressing CENP-ACnp1 from different promoters, identical exposures are used for imaging of all cells stained for CENP-ACnp1 in any given set. Images are not autoscaled, instead they are scaled relative to the brightest images in the set in order to reflect relative signal intensity obtained from the different strengths of promoter. Thus, images are scaled relative to the nmt81-CENP-ACnp1 cells in Figure 1, and relative to nmt1-CENP-ACnp1 cells in Figure S1.
Supporting Information
(A) CENP-ACnp1 can be detected when overexpressed from the nmt1 promoter. Adapted from Castillo et al., 2007 (Figure S3). Western analyses of extracts from wild-type cells overexpressing CENP-ACnp1 from nmt81 (81xC), nmt41 (41xC) or nmt1 (3xC) promoter to give low, medium and high expression levels compared with cells containing endogenous levels (3x). (B) Western analysis of GFP-CENP-ACnp1 levels in wt and spt16-18 cells. From Choi et al., 2012 (Figure 1D). Western analysis of GFP-CENP-ACnp1 levels in wt and spt16-18 cells expressing GFP-CENP-ACnp1 under endogenous, nmt81 or nmt41 promoter (upper panel). The intensities of GFP-CENP-ACnp1 and TAT-1 (alpha-tubulin) signals were measured using LICOR Odyssey Infrared Imaging System software (Li-COR Bioscience) and the relative intensities of GFP-CENP-ACnp1/TAT-1 were quantified (bottom panel). GFP-CENP-ACnp1 was expressed for 24 h at 25°C before harvest. (C) Comparison of CENP-ACnp1 localisation in cells expressing CENP-ACnp1 from different strengths of nmt promoter. Immunolocalization of CENP-ACnp1 in cells expressing endogenous levels of CENP-ACnp1 or extra CENP-ACnp1 expressed from the nmt81, nmt41 or nmt1 promoters. Cells were grown in the absence of thiamine for 24 h at 36oC and stained with anti-CENP-ACnp1 antibody (green) and DAPI (DNA: red). Images are displayed to indicate the relative signal intensity in cells expressing CENP-ACnp1 from the four different promoters. Images have not been autoscaled. The images for endogenous (Endog.) and nmt81 promoters therefore appear very faint compared to nmt1. Scale bar, 5 µm.
(PDF)
(A) Genome browser view showing ChIP-chip occupancy profiles for wild-type (Endg.) levels in blue or additional CENP-ACnp1 levels in orange (nmt81-CENP-ACnp1) at centromeres, pericentromeric outer repeats (x2, x4, x9 indicate the number of repetitive elements) or subtelomeric / rDNA-adjacent regions. Data on the Y-axis are presented in linear scale and X-axis shows genome positions (distance from the chromosome end at telomeres and rDNA loci is indicated in kb). ChIP-PCR analysis of CENP-ACnp1 association with pericentromeric outer repeats (B) or subtelomeric / rDNA-adjacent regions (C) expressing wild-type (Endog.) or additional (nmt81) levels of CENP-ACnp1. Enrichment of pericentromeric outer repeats (A, otr primers), telomeric (B, primers reside 21.7 kb, 53 kb and 72 kb from the left telomere of chromosome I and 3.7 kb and 47.9 kb from the right end of chromosome II) and rDNA adjacent regions (primers reside 34.5 kb from the rDNA repeats on left arm of chromosome III) was assessed compared to actin (act) in immunoprecipitated (IP) relative to total (T) extract. (D) ChIP-qPCR analysis of CENP-ACnp1 association with a subtelomeric region from the right telomere of chromosome I (53 kb from the end) expressing wild-type (Endog.) or additional (nmt81-CENP-ACnp1) levels of CENP-ACnp1. Enrichment on the sub-telomeric region was normalized to the signal obtained for the euchromatic gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a or b) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
(A) ChIP-qPCR of CENP-ACnp1 and CENP-CCnp3 levels at TM1 in cells containing the ura4 +-Tel or ura4 +-TAS-Tel Ch16 minichromosomes and expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. Enrichment is reported as the percentage of immunoprecipitated chromatin (% IP). Error bars indicate S.D. from 3 biological replicates. (B) ChIP-qPCR of H3K9me2 levels on centromeric otr dgI repeats in the same cells as in A. Enrichment on dgI was normalized to the signal obtained for the gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with the same letter (a) indicate results not significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
Serial dilution growth assay of wild-type cells expressing wild-type levels (Endog.) or moderate levels of additional CENP-ACnp1 (nmt41-CENP-ACnp1) grown at 36°C for three days. Cells containing the m23:ura4 + (ura4 + inserted 50 kb from the left telomere of Ch16), ura4 +-Tel or ura4 +-TAS-Tel Ch16 minichromosomes were plated on the indicated media: no leucine, adenine or no uracil, or with counter-selective 5-FOA added.
(PDF)
ChIP-qPCR of CENP-ACnp1 levels on centromeric otr dgI repeats (A) and at TM1 in the central region of centromere 1 (B) in wild-type (wt) and mutant cells (ccq1∆, taz1∆, rif1∆, clr4∆ and clr3∆) carrying the Chs 16–72 (ura4+-Tel) minichromosome and expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) CENP-ACnp1 levels. (C) ChIP-qPCR of H3K9me2 levels on centromeric otr dgI repeats in wild-type (wt) and mutant cells (ccq1∆, taz1∆, and clr4∆) with the same configuration. Enrichment on dgI was normalized the signal obtained for the gene encoding actin (act1 +). Enrichment at TM1 is reported as the percentage of immunoprecipitated chromatin (% IP). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01 in (A) and (B) or P<0.05 in (C).
(PDF)
ChIP-qPCR of CENP-ACnp1 levels at TM1 in the central region of centromere 1 (A) and centromeric otr dgI repeats (B) in cells containing an array of telomeric repeats integrated at the ura4 + locus (ura4 +: Int-Telo) or control cells (ura4 +) and expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. Enrichment on dgI was normalized the signal obtained for the gene encoding actin (act1 +). Enrichment at TM1 is reported as the percentage of immunoprecipitated chromatin (% IP). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a or b) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
(A) ChIP-qPCR of CENP-ACnp1 levels at TM1 in the central region of centromere 1 and centromeric otr dgI repeats in wild-type (wt) and mutant cells (ccq1∆, taz1∆, and clr4∆) which contain the internal telomere (ura4 +: Int-Telo) and that express endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. (B) ChIP-qPCR of H3K9me2 levels in the same cell backgrounds. Enrichment at TM1 is reported as the percentage of immunoprecipitated chromatin (% IP). Enrichment on dgI was normalized the background signal obtained for the gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
(PDF)
(PDF)
(PDF)
Acknowledgments
We thank members of the Allshire Lab for advice and discussion and we are very grateful to Lakxmi Subramanian for comments on the manuscript and helpful discussions. We thank Julie P. Cooper for providing the ura4 +: Int-Telo strain prior to publication along with taz1∆, ccq1∆, and rif1∆. We also thank Takeshi Urano for providing the H3K9m2 antibody (mAb5.1.1).
Funding Statement
The Wellcome Trust supported the work of RCA [095021], [065061]. Work in the Wellcome Trust Centre for Cell Biology is supported by the Wellcome Trust core funding [092076]. RCA is a Wellcome Trust Principal Fellow. Work in the KE laboratory is supported by grants from the Swedish Cancer Society and the Swedish Research Council. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Earnshaw WC, Migeon BR (1985) Three related centromere proteins are absent from the inactive centromere of a stable isodicentric chromosome. Chromosoma 92: 290-296. doi:10.1007/BF00329812. PubMed: 2994966. [DOI] [PubMed] [Google Scholar]
- 2. Sullivan BA, Schwartz S (1995) Identification of centromeric antigens in dicentric Robertsonian translocations: CENP-C and CENP-E are necessary components of functional centromeres. Hum Mol Genet 4: 2189-2197. doi:10.1093/hmg/4.12.2189. PubMed: 8634687. [DOI] [PubMed] [Google Scholar]
- 3. Topp CN, Okagaki RJ, Melo JR, Kynast RG, Phillips RL et al. (2009) Identification of a maize neocentromere in an oat-maize addition line. Cytogenet Genome Res 124: 228-238. doi:10.1159/000218128. PubMed: 19556776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Nasuda S, Hudakova S, Schubert I, Houben A, Endo TR (2005) Stable barley chromosomes without centromeric repeats. Proc Natl Acad Sci U S A 102: 9842-9847. PubMed: 15998740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Maggert KA, Karpen GH (2001) The activation of a neocentromere in Drosophila requires proximity to an endogenous centromere. Genetics 158: 1615-1628. PubMed: 11514450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Ishii K, Ogiyama Y, Chikashige Y, Soejima S, Masuda F et al. (2008) Heterochromatin integrity affects chromosome reorganization after centromere dysfunction. Science 321: 1088-1091. doi:10.1126/science.1158699. PubMed: 18719285. [DOI] [PubMed] [Google Scholar]
- 7. Shang WH, Hori T, Martins NM, Toyoda A, Misu S et al. (2013) Chromosome Engineering Allows the Efficient Isolation of Vertebrate Neocentromeres. Dev Cell 24: 1-14. doi:10.1016/j.devcel.2012.11.020. PubMed: 23328396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Heun P, Erhardt S, Blower MD, Weiss S, Skora AD et al. (2006) Mislocalization of the Drosophila centromere-specific histone CID promotes formation of functional ectopic kinetochores. Dev Cell 10: 303-315. doi:10.1016/j.devcel.2006.01.014. PubMed: 16516834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Olszak AM, van Essen D, Pereira AJ, Diehl S, Manke T et al. (2011) Heterochromatin boundaries are hotspots for de novo kinetochore formation. Nat Cell Biol 13: 799-808. doi:10.1038/ncb2272. PubMed: 21685892. [DOI] [PubMed] [Google Scholar]
- 10. Ketel C, Wang HS, McClellan M, Bouchonville K, Selmecki A et al. (2009) Neocentromeres form efficiently at multiple possible loci in Candida albicans. PLOS Genet 5: e1000400 PubMed: 19266018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Shang WH, Hori T, Toyoda A, Kato J, Popendorf K et al. (2010) Chickens possess centromeres with both extended tandem repeats and short non-tandem-repetitive sequences. Genome Res 20: 1219-1228. PubMed: 20534883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Mendiburo MJ, Padeken J, Fülöp S, Schepers A, Heun P (2011) Drosophila CENH3 is sufficient for centromere formation. Science 334: 686-690. PubMed: 22053052. [DOI] [PubMed] [Google Scholar]
- 13. Barnhart MC, Kuich PH, Stellfox ME, Ward JA, Bassett EA et al. (2011) HJURP is a CENP-A chromatin assembly factor sufficient to form a functional de novo kinetochore. J Cell Biol 194: 229-243. doi:10.1083/jcb.201012017. PubMed: 21768289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hewawasam G, Shivaraju M, Mattingly M, Venkatesh S, Martin-Brown S et al. (2010) Psh1 is an E3 ubiquitin ligase that targets the centromeric histone variant Cse4. Mol Cell 40: 444-454. PubMed: 21070970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Ranjitkar P, Press MO, Yi X, Baker R, MacCoss MJ et al. (2010) An E3 ubiquitin ligase prevents ectopic localization of the centromeric histone H3 variant via the centromere targeting domain. Mol Cell 40: 455-464. PubMed: 21070971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Choi ES, Strålfors A, Catania S, Castillo AG, Svensson JP et al. (2012) Factors that promote H3 chromatin integrity during transcription prevent promiscuous deposition of CENP-A(Cnp1) in fission yeast. PLOS Genet 8: e1002985 PubMed: 23028377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Choi ES, Strålfors A, Castillo AG, Durand-Dubief M, Ekwall K et al. (2011) Identification of noncoding transcripts from within CENP-A chromatin at fission yeast centromeres. J Biol Chem 286: 23600-23607. doi:10.1074/jbc.M111.228510. PubMed: 21531710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Folco HD, Pidoux AL, Urano T, Allshire RC (2008) Heterochromatin and RNAi are required to establish CENP-A chromatin at centromeres. Science 319: 94-97. doi:10.1126/science.1150944. PubMed: 18174443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kagansky A, Folco HD, Almeida R, Pidoux AL, Boukaba A et al. (2009) Synthetic heterochromatin bypasses RNAi and centromeric repeats to establish functional centromeres. Science 324: 1716-1719. doi:10.1126/science.1172026. PubMed: 19556509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Grewal SI, Jia S (2007) Heterochromatin revisited. Nat Rev Genet 8: 35-46. doi:10.1038/nrg2008. PubMed: 17173056. [DOI] [PubMed] [Google Scholar]
- 21. Sugawara NF (1988) DNA sequences at the telomeres of the fission yeast S. pombe. PhD Thesis, Harvard University, Cambridge, Massachusetts. [Google Scholar]
- 22. Mandell JG, Goodrich KJ, Bähler J, Cech TR (2005) Expression of a RecQ helicase homolog affects progression through crisis in fission yeast lacking telomerase. J Biol Chem 280: 5249-5257. PubMed: 15591066. [DOI] [PubMed] [Google Scholar]
- 23. Kanoh J, Sadaie M, Urano T, Ishikawa F (2005) Telomere binding protein Taz1 establishes Swi6 heterochromatin independently of RNAi at telomeres. Curr Biol 15: 1808-1819. PubMed: 16243027. [DOI] [PubMed] [Google Scholar]
- 24. Jain D, Cooper JP (2010) Telomeric strategies: means to an end. Annu Rev Genet 44: 243-269. doi:10.1146/annurev-genet-102108-134841. PubMed: 21047259. [DOI] [PubMed] [Google Scholar]
- 25. Cooper JP, Nimmo ER, Allshire RC, Cech TR (1997) Regulation of telomere length and function by a Myb-domain protein in fission yeast. Nature 385: 744-747. doi:10.1038/385744a0. PubMed: 9034194. [DOI] [PubMed] [Google Scholar]
- 26. Baumann P, Cech TR (2001) Pot1, the putative telomere end-binding protein in fission yeast and humans. Science 292: 1171-1175. PubMed: 11349150. [DOI] [PubMed] [Google Scholar]
- 27. Chikashige Y, Hiraoka Y (2001) Telomere binding of the Rap1 protein is required for meiosis in fission yeast. Curr Biol 11: 1618-1623. PubMed: 11676924. [DOI] [PubMed] [Google Scholar]
- 28. Kanoh J, Ishikawa F (2001) spRap1 and spRif1, recruited to telomeres by Taz1, are essential for telomere function in fission yeast. Curr Biol 11: 1624-1630. PubMed: 11676925. [DOI] [PubMed] [Google Scholar]
- 29. Miyoshi T, Kanoh J, Saito M, Ishikawa F (2008) Fission yeast Pot1-Tpp1 protects telomeres and regulates telomere length. Science 320: 1341-1344. doi:10.1126/science.1154819. PubMed: 18535244. [DOI] [PubMed] [Google Scholar]
- 30. Sugiyama T, Cam HP, Sugiyama R, Noma K, Zofall M et al. (2007) SHREC, an effector complex for heterochromatic transcriptional silencing. Cell 128: 491-504. doi:10.1016/j.cell.2006.12.035. PubMed: 17289569. [DOI] [PubMed] [Google Scholar]
- 31. Nakamura TM, Cooper JP, Cech TR (1998) Two modes of survival of fission yeast without telomerase. Science 282: 493-496. doi:10.1126/science.282.5388.493. PubMed: 9774280. [DOI] [PubMed] [Google Scholar]
- 32. Dehé PM, Rog O, Ferreira MG, Greenwood J, Cooper JP (2012) Taz1 enforces cell-cycle regulation of telomere synthesis. Mol Cell 46: 797-808. doi:10.1016/j.molcel.2012.04.022. PubMed: 22633956. [DOI] [PubMed] [Google Scholar]
- 33. Miller KM, Ferreira MG, Cooper JP (2005) Taz1, Rap1 and Rif1 act both interdependently and independently to maintain telomeres. EMBO J 24: 3128-3135. doi:10.1038/sj.emboj.7600779. PubMed: 16096639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Flory MR, Carson AR, Muller EG, Aebersold R (2004) An SMC-domain protein in fission yeast links telomeres to the meiotic centrosome. Mol Cell 16: 619-630. doi:10.1016/j.molcel.2004.10.027. PubMed: 15546621. [DOI] [PubMed] [Google Scholar]
- 35. Tomita K, Cooper JP (2008) Fission yeast Ccq1 is telomerase recruiter and local checkpoint controller. Genes Dev 22: 3461-3474. doi:10.1101/gad.498608. PubMed: 19141478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Castillo AG, Mellone BG, Partridge JF, Richardson W, Hamilton GL et al. (2007) Plasticity of fission yeast CENP-A chromatin driven by relative levels of histone H3 and H4. PLOS Genet 3: e121 PubMed: 17677001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Basi G, Schmid E, Maundrell K (1993) TATA box mutations in the Schizosaccharomyces pombe nmt1 promoter affect transcription efficiency but not the transcription start point or thiamine repressibility. Gene 123: 131-136. doi:10.1016/0378-1119(93)90552-E. PubMed: 8422997. [DOI] [PubMed] [Google Scholar]
- 38. Scott KC, Merrett SL, Willard HF (2006) A heterochromatin barrier partitions the fission yeast centromere into discrete chromatin domains. Curr Biol 16: 119-129. doi:10.1016/j.cub.2006.02.013. PubMed: 16431364. [DOI] [PubMed] [Google Scholar]
- 39. Verdel A, Moazed D (2005) RNAi-directed assembly of heterochromatin in fission yeast. FEBS Lett 579: 5872-5878. doi:10.1016/j.febslet.2005.08.083. PubMed: 16223485. [DOI] [PubMed] [Google Scholar]
- 40. Nimmo ER, Cranston G, Allshire RC (1994) Telomere-associated chromosome breakage in fission yeast results in variegated expression of adjacent genes. EMBO J 13: 3801-3811. PubMed: 8070408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Miller KM, Rog O, Cooper JP (2006) Semi-conservative DNA replication through telomeres requires Taz1. Nature 440: 824-828. doi:10.1038/nature04638. PubMed: 16598261. [DOI] [PubMed] [Google Scholar]
- 42. Moreno-Moreno O, Torras-Llort M, Azorín F (2006) Proteolysis restricts localization of CID, the centromere-specific histone H3 variant of Drosophila, to centromeres. Nucleic Acids Res 34: 6247-6255. doi:10.1093/nar/gkl902. PubMed: 17090596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Lefrançois P, Auerbach RK, Yellman CM, Roeder GS, Snyder M (2013) Centromere-like regions in the budding yeast genome. PLOS Genet 9: e1003209 PubMed: 23349633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Lefrancois P, Euskirchen GM, Auerbach RK, Rozowsky J, Gibson T, et al. (2009) Efficient yeast ChIP-Seq using multiplex short-read DNA sequencing. BMC Genomics 10: 37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Steglich B, Filion GJ, van Steensel B, Ekwall K (2012) The inner nuclear membrane proteins Man1 and Ima1 link to two different types of chromatin at the nuclear periphery in S. pombe. Nucleus 3: 77-87. PubMed: 22156748. [DOI] [PubMed] [Google Scholar]
- 46. Marshall OJ, Chueh AC, Wong LH, Choo KH (2008) Neocentromeres: new insights into centromere structure, disease development, and karyotype evolution. Am J Hum Genet 82: 261-282. doi:10.1016/j.ajhg.2007.11.009. PubMed: 18252209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Moreno S, Klar A, Nurse P (1991) Molecular genetic analysis of fission yeast Schizosaccharomyces pombe. Methods Enzymol 194: 795-823. doi:10.1016/0076-6879(91)94059-L. PubMed: 2005825. [DOI] [PubMed] [Google Scholar]
- 48. Nicol JW, Helt GA, Blanchard SG Jr., Raja A, Loraine AE (2009) The Integrated Genome Browser: free software for distribution and exploration of genome-scale datasets. Bioinformatics 25: 2730-2731. doi:10.1093/bioinformatics/btp472. PubMed: 19654113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Durand-Dubief M, Persson J, Norman U, Hartsuiker E, Ekwall K (2010) Topoisomerase I regulates open chromatin and controls gene expression in vivo. EMBO J 29: 2126-2134. PubMed: 20526281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Pidoux AL, Uzawa S, Perry PE, Cande WZ, Allshire RC (2000) Live analysis of lagging chromosomes during anaphase and their effect on spindle elongation rate in fission yeast. J Cell Sci 113 23: 4177-4191. PubMed: 11069763. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(A) CENP-ACnp1 can be detected when overexpressed from the nmt1 promoter. Adapted from Castillo et al., 2007 (Figure S3). Western analyses of extracts from wild-type cells overexpressing CENP-ACnp1 from nmt81 (81xC), nmt41 (41xC) or nmt1 (3xC) promoter to give low, medium and high expression levels compared with cells containing endogenous levels (3x). (B) Western analysis of GFP-CENP-ACnp1 levels in wt and spt16-18 cells. From Choi et al., 2012 (Figure 1D). Western analysis of GFP-CENP-ACnp1 levels in wt and spt16-18 cells expressing GFP-CENP-ACnp1 under endogenous, nmt81 or nmt41 promoter (upper panel). The intensities of GFP-CENP-ACnp1 and TAT-1 (alpha-tubulin) signals were measured using LICOR Odyssey Infrared Imaging System software (Li-COR Bioscience) and the relative intensities of GFP-CENP-ACnp1/TAT-1 were quantified (bottom panel). GFP-CENP-ACnp1 was expressed for 24 h at 25°C before harvest. (C) Comparison of CENP-ACnp1 localisation in cells expressing CENP-ACnp1 from different strengths of nmt promoter. Immunolocalization of CENP-ACnp1 in cells expressing endogenous levels of CENP-ACnp1 or extra CENP-ACnp1 expressed from the nmt81, nmt41 or nmt1 promoters. Cells were grown in the absence of thiamine for 24 h at 36oC and stained with anti-CENP-ACnp1 antibody (green) and DAPI (DNA: red). Images are displayed to indicate the relative signal intensity in cells expressing CENP-ACnp1 from the four different promoters. Images have not been autoscaled. The images for endogenous (Endog.) and nmt81 promoters therefore appear very faint compared to nmt1. Scale bar, 5 µm.
(PDF)
(A) Genome browser view showing ChIP-chip occupancy profiles for wild-type (Endg.) levels in blue or additional CENP-ACnp1 levels in orange (nmt81-CENP-ACnp1) at centromeres, pericentromeric outer repeats (x2, x4, x9 indicate the number of repetitive elements) or subtelomeric / rDNA-adjacent regions. Data on the Y-axis are presented in linear scale and X-axis shows genome positions (distance from the chromosome end at telomeres and rDNA loci is indicated in kb). ChIP-PCR analysis of CENP-ACnp1 association with pericentromeric outer repeats (B) or subtelomeric / rDNA-adjacent regions (C) expressing wild-type (Endog.) or additional (nmt81) levels of CENP-ACnp1. Enrichment of pericentromeric outer repeats (A, otr primers), telomeric (B, primers reside 21.7 kb, 53 kb and 72 kb from the left telomere of chromosome I and 3.7 kb and 47.9 kb from the right end of chromosome II) and rDNA adjacent regions (primers reside 34.5 kb from the rDNA repeats on left arm of chromosome III) was assessed compared to actin (act) in immunoprecipitated (IP) relative to total (T) extract. (D) ChIP-qPCR analysis of CENP-ACnp1 association with a subtelomeric region from the right telomere of chromosome I (53 kb from the end) expressing wild-type (Endog.) or additional (nmt81-CENP-ACnp1) levels of CENP-ACnp1. Enrichment on the sub-telomeric region was normalized to the signal obtained for the euchromatic gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a or b) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
(A) ChIP-qPCR of CENP-ACnp1 and CENP-CCnp3 levels at TM1 in cells containing the ura4 +-Tel or ura4 +-TAS-Tel Ch16 minichromosomes and expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. Enrichment is reported as the percentage of immunoprecipitated chromatin (% IP). Error bars indicate S.D. from 3 biological replicates. (B) ChIP-qPCR of H3K9me2 levels on centromeric otr dgI repeats in the same cells as in A. Enrichment on dgI was normalized to the signal obtained for the gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with the same letter (a) indicate results not significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
Serial dilution growth assay of wild-type cells expressing wild-type levels (Endog.) or moderate levels of additional CENP-ACnp1 (nmt41-CENP-ACnp1) grown at 36°C for three days. Cells containing the m23:ura4 + (ura4 + inserted 50 kb from the left telomere of Ch16), ura4 +-Tel or ura4 +-TAS-Tel Ch16 minichromosomes were plated on the indicated media: no leucine, adenine or no uracil, or with counter-selective 5-FOA added.
(PDF)
ChIP-qPCR of CENP-ACnp1 levels on centromeric otr dgI repeats (A) and at TM1 in the central region of centromere 1 (B) in wild-type (wt) and mutant cells (ccq1∆, taz1∆, rif1∆, clr4∆ and clr3∆) carrying the Chs 16–72 (ura4+-Tel) minichromosome and expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) CENP-ACnp1 levels. (C) ChIP-qPCR of H3K9me2 levels on centromeric otr dgI repeats in wild-type (wt) and mutant cells (ccq1∆, taz1∆, and clr4∆) with the same configuration. Enrichment on dgI was normalized the signal obtained for the gene encoding actin (act1 +). Enrichment at TM1 is reported as the percentage of immunoprecipitated chromatin (% IP). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01 in (A) and (B) or P<0.05 in (C).
(PDF)
ChIP-qPCR of CENP-ACnp1 levels at TM1 in the central region of centromere 1 (A) and centromeric otr dgI repeats (B) in cells containing an array of telomeric repeats integrated at the ura4 + locus (ura4 +: Int-Telo) or control cells (ura4 +) and expressing endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. Enrichment on dgI was normalized the signal obtained for the gene encoding actin (act1 +). Enrichment at TM1 is reported as the percentage of immunoprecipitated chromatin (% IP). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a or b) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
(A) ChIP-qPCR of CENP-ACnp1 levels at TM1 in the central region of centromere 1 and centromeric otr dgI repeats in wild-type (wt) and mutant cells (ccq1∆, taz1∆, and clr4∆) which contain the internal telomere (ura4 +: Int-Telo) and that express endogenous (Endog. CENP-ACnp1) or additional (nmt41-CENP-ACnp1) levels of CENP-ACnp1. (B) ChIP-qPCR of H3K9me2 levels in the same cell backgrounds. Enrichment at TM1 is reported as the percentage of immunoprecipitated chromatin (% IP). Enrichment on dgI was normalized the background signal obtained for the gene encoding actin (act1 +). Error bars indicate S.D. from 3 biological replicates. Mean values marked with different letter (a, b or c) indicate results significantly different from each other, as established by One Way ANOVA and Holm-Sidak test for multiple comparison (P<0.01).
(PDF)
(PDF)
(PDF)
(PDF)