Abstract
Objectives
To compare patterns of arteriographic lesions of the aorta and primary branches in patients with Takayasu’s arteritis (TAK) and giant cell arteritis (GCA).
Methods
Patients were selected from two North American cohorts of TAK and GCA. The frequency of arteriographic lesions was calculated for 15 large arteries. Cluster analysis was used to derive patterns of arterial disease in TAK versus GCA and in patients categorised by age at disease onset. Using latent class analysis, computer derived classification models based upon patterns of arterial disease were compared with traditional classification.
Results
Arteriographic lesions were identified in 145 patients with TAK and 62 patients with GCA. Cluster analysis demonstrated that arterial involvement was contiguous in the aorta and usually symmetric in paired branch vessels for TAK and GCA. There was significantly more left carotid (p=0.03) and mesenteric (p=0.02) artery disease in TAK and more left and right axillary (p<0.01) artery disease in GCA. Subclavian disease clustered asymmetrically in TAK and in patients ≤55 years at disease onset and clustered symmetrically in GCA and patients >55 years at disease onset. Computer derived classification models distinguished TAK from GCA in two subgroups, defining 26% and 18% of the study sample; however, 56% of patients were classified into a subgroup that did not strongly differentiate between TAK and GCA.
Conclusions
Strong similarities and subtle differences in the distribution of arterial disease were observed between TAK and GCA. These findings suggest that TAK and GCA may exist on a spectrum within the same disease.
BACKGROUND
Takayasu’s arteritis (TAK) and giant cell arteritis (GCA) are the two most common types of large vessel vasculitis. Historically, TAK and GCA have been considered distinct diseases on the basis of differences in age at disease onset, ethnic distribution and clinical features including predilection for certain arterial territories. Recent observations, however, have prompted speculation as to whether TAK and GCA compromise a spectrum within a single disease.1 All patients with TAK have disease involvement of the aorta or its primary branches. In contrast, GCA is traditionally considered a disease of the cranial arteries, yet recent reports estimate that 20%–30% of patients with GCA also have radiographic evidence of arterial disease in the aorta and its primary branches2–4 and one older study found that, at necropsy, all patients with GCA had vasculitic changes in the large arteries.5 Furthermore, arterial histology may be indistinguishable in TAK and GCA.1,5
The 1990 American College of Rheumatology (ACR) classification criteria for vasculitis are used to distinguish between TAK and GCA but do not adequately differentiate between TAK and the subset of GCA with large vessel involvement.6,7 The criteria for TAK focus on large vessel disease of the aorta and primary branches. In contrast, the criteria for GCA were developed at a time when large vessel involvement was not a well-recognised feature of GCA and focus instead on the cranial aspects of the disease. Ultimately, age at the time of disease onset is often what is used to classify patients with large vessel involvement as having either TAK or GCA. Using the age criterion proposed in the 1990 ACR classification criteria, patients with large vessel vasculitis and disease onset at less than 40 years of age are classified as having TAK and those with disease onset at greater than 50 years of age are classified as having GCA; it is unclear how to classify patients with the onset of large vessel vasculitis between the ages of 40 and 50 years as this age group is not specified in the criteria.
In a recent study, cluster analysis was used to demonstrate novel patterns of arteriographic lesions in 82 patients with TAK.8 Lesions were generally symmetrical in paired arteries (eg, right and left carotid arteries) and contiguous in the aorta. Whether similar patterns exist in patients with GCA and large vessel involvement is unknown. Similarity of arteriographic patterns in TAK and GCA would suggest that TAK and GCA exist on a spectrum within the same disease. If arteriographic patterns are different in TAK and GCA, these potential differences could be used in the ongoing development of new classification criteria for vasculitis.9
The objectives of the current study were: (1) to compare the occurrence of arteriographic lesions in the aorta and primary branches in patients with large vessel vasculitis; (2) to determine if patterns of arteriographic disease differ between patients with TAK and GCA and between patients categorised by age at disease onset; and (3) to explore novel ways to classify large vessel vasculitis using computer-generated models of disease classification based upon patterns of arterial involvement.
METHODS
Study sample
Patients with TAK and GCA were selected from two cohorts, the Vasculitis Clinical Research Consortium (VCRC) and the Cleveland Clinic Foundation (CCF). The VCRC is a National Institutes of Health supported, international, multicentre research infrastructure dedicated to conducting clinical research in different forms of vasculitis. Patients with TAK and GCA enrolled in VCRC longitudinal, observational cohorts from 2006 to 2010 were selected for this study. Six VCRC centres provided patients for analysis. The CCF cohort consists of patients with TAK and GCA who were under the care of a single investigator from 1992 to 2006. Since the CCF is a participating centre within the VCRC, data from patients enrolled in both the cohorts were assigned to only one cohort.
All patients with TAK fulfilled the 1990 ACR criteria for classification of TAK. Patients with GCA were included in this study if they had arteriographic lesions of the aorta or primary branch vessels clinically attributed to vasculitis and were either 50 years or older at disease onset or fulfilled the 1990 ACR criteria for classification of GCA.
Data elements
Baseline demographic information including age, sex, race, ethnicity, time of disease onset and disease duration was recorded. Disease onset was defined as the date of initial identification of vascular symptoms that were subsequently determined to be compatible with vasculitis and not attributable to comorbid conditions.
All patients underwent magnetic resonance angiography of the aorta and its primary branches. Clinical radiologists at each participating institution assessed the angiograms for the presence of artery-specific lesions, and study investigators reviewed and recorded angiogram findings using standardised data collection forms. In the VCRC and CCF cohorts, arteriographic lesions were defined as stenosis, occlusion or aneurysm. Presence of abnormal arterial wall thickness was recorded in the CCF cohort, but given the unclear prognostic significance of this finding, wall thickness was not recorded in the VCRC cohort. For patients who underwent multiple angiograms over time, the most recent angiogram was selected for analysis.
Analytic methods
The frequency of artery-specific lesions was calculated for the following 15 arteries: thoracic aorta, abdominal aorta, carotid arteries, vertebral arteries, subclavian arteries, axillary arteries, renal arteries, mesenteric arteries (celiac, superior mesenteric and inferior mesenteric arteries) and iliofemoral arteries. The frequencies of artery-specific lesions were compared between TAK and GCA using Fisher’s exact test.
Cluster analysis was used to identify patterns of arterial disease. Individual arteries were clustered on the presence or absence of arteriographic lesions, and agglomerative, hierarchical clustering was performed.10 The cluster algorithm started with each individual artery as a single cluster. In successive iterations, the two nearest clusters were merged together on the basis of a measure of similarity to form a new, unique cluster. The process was repeated until all of the data were contained in one cluster.
An important step in cluster analysis is to choose a measure of similarity to define the relationship of the data variables, and a common critique of cluster analysis is that different methods can sometimes yield disparate results. Therefore, three different measures of similarity were analysed in parallel to determine if results were consistent across different clustering approaches. The three different measures of similarity were based upon: (1) ϕ coefficient correlation, (2) tetrachoric coefficient correlation and (3) principal component analysis. To allow for comparison with the recently reported cluster analysis in TAK,8 correlation matrices were created using the ϕ coefficient. The ϕ coefficient is a measure of correlation between categorical data with two categories (ie, presence or absence of arteriographic disease).11 The tetrachoric coefficient, like the ϕ coefficient, is a measure of correlation between categorical data but also assumes that the variable underlying each dichotomous measure is normally distributed.12 Distance between clusters in the ϕ and tetrachoric coefficient correlation techniques was calculated using the Euclidian distance, and Ward’s method was used to agglomerate the clusters. In the principal component approach, clusters were determined by calculating the first principal component and maximising the sum across clusters of the variation accounted for by the cluster components. Distance between clusters in the principal component approach was calculated using average linkage. Since only the CCF cohort incorporated wall thickness into the definition of arterial disease, sensitivity analyses were performed to see if cluster patterns differed according to the definition of arterial disease.
Tree dendograms were created to visualise cluster patterns. Individual tree dendograms from patients with TAK and GCA were visually compared. Tree dendograms were also derived in patients categorised by age at the time of disease onset as follows: <40 years, 40–55 years and >55 years.
Latent class analysis was used to explore novel ways of classifying large vessel vasculitis based upon patterns of arterial lesions.13 Latent class analysis relates a set of observed categorical variables to a set of latent variables. A latent class model assumes that the variables of interest (ie, individual arteries) are conditionally independent and related only via an underlying latent class (eg, disease entity) which causes the association. A latent class model with the optimal number of computer derived subgroups (latent classes) was determined using model fit statistics, including the likelihood ratio G2, Akaike information criteria and Bayesian information criteria values. Posterior probabilities of latent class membership were calculated for each patient using Bayes’s theorem. Logistic regression was used to assess whether a patient’s probability of membership to a latent class was associated with the traditional diagnoses of TAK and GCA.
All statistical analyses were done using SAS 9.1 (SAS Institute, Cary, North Carolina, USA).
RESULTS
Subject characteristics
One-hundred and forty-five patients with TAK and 62 patients with GCA were included in the analysis. Baseline demographics, reported by cohort and disease, are displayed in table 1.
Table 1.
Baseline patient characteristics
| Takayasu’s arteritis (n=145) | Giant cell arteritis (n=62) | |||
|---|---|---|---|---|
| Cohort | VCRC (n=70) | CCF (n=75) | VCRC (n=28) | CCF (n=34) |
| Age* (years) (mean, range) | 31 (9–61) | 31 (8–59) | 67 (57–89) | 66 (49–93) |
| Disease duration (years) (mean, SD) | 8.7 (±7.1) | 5.1 (±9.1) | 2.9 (±2.5) | 2.4 (±3.4) |
| Sex (% female) | 66 (94%) | 68 (91%) | 27 (96%) | 27 (77%) |
| Race (% white) | 59 (84%) | 66 (88%) | 26 (92%) | 30 (86%) |
Age at time of angiogram assessment.
CCF, Cleveland Clinic Foundation; VCRC, Vasculitis Clinical Research Consortium.
In both cohorts, patients with GCA underwent angiography at the discretion of the physician if there was a clinical suspicion for large vessel disease. Among patients with GCA, 10/28 (36%) in the VCRC cohort and 11/34 (32%) in the CCF cohort had large vessel disease in absence of features of cranial arteritis. Among patients with GCA who had features of cranial arteritis, biopsy proven temporal artery involvement was demonstrated in 6/18 (33%) in the VCRC cohort and 10/23 (44%) in the CCF cohort.
Frequency of arteriographic lesions
The occurrence of arteriographic lesions in TAK and GCA is compared in table 2. No significant differences in the frequency of arterial disease were observed between TAK and GCA for 11 of the 15 arteries of interest. There was significantly more left carotid (p=0.03) and mesenteric (p=0.02) artery disease in TAK and substantially more left and right axillary (p<0.01) artery disease in GCA. The highest frequency of arterial disease in patients with TAK was observed in the left subclavian artery. Asymmetric involvement of the subclavian arteries was seen more frequently in TAK (63 of 139; 45%) compared with GCA (10 of 57; 18%) (p<0.01). Ninety-seven per cent of all arterial lesions were stenotic or occlusive, 3% of all lesions were aneurysmal and there were no significant differences observed in the type of arterial lesions between TAK and GCA.
Table 2.
Frequency of arteriographic lesions
| Artery | Takayasu’s arteritis | Giant cell arteritis | Difference in frequency (p value) |
|---|---|---|---|
| Left carotid | 52/139 (37%) | 12/57 (21%) | 16% (0.03) |
| Right carotid | 34/136 (25%) | 10/58 (17%) | 8% (0.27) |
| Left vertebral | 17/137 (13%) | 10/58 (17%) | 4% (0.44) |
| Right vertebral | 16/139 (12%) | 6/57 (11%) | 1% (1.00) |
| Left subclavian | 96/140 (69%) | 35/57 (61%) | 8% (0.41) |
| Right subclavian | 56/139 (40%) | 27/57 (39%) | 1% (0.43) |
| Left axillary | 16/137 (11%) | 22/57 (39%) | 28% (<0.01) |
| Right axillary | 12/138 (9%) | 25/57 (44%) | 35% (<0.01) |
| Thoracic aorta | 61/137 (46%) | 34/56 (61%) | 15% (0.06) |
| Abdominal aorta | 45/122 (37%) | 22/53 (42%) | 5% (0.49) |
| Mesenteric | 47/131 (36%) | 10/54 (18%) | 18% (0.02) |
| Left renal | 19/125 (15%) | 4/49 (8%) | 7% (0.32) |
| Right renal | 20/125 (16%) | 8/51 (16%) | 0% (1.00) |
| Left iliofemoral | 23/119 (19%) | 5/46 (17%) | 2% (0.25) |
| Right iliofemoral | 24/119 (20%) | 6/46 (13%) | 7% (0.37) |
When stratified by cohort, there were no significant differences in the frequency of arteriographic lesions in any of the aortic branch vessels. There was significantly more thoracic and abdominal aortic disease (p<0.01) in the CCF cohort than in the VCRC cohort. When wall thickness was not incorporated into the definition of arteriographic lesions in the CCF cohort, there were no longer significant differences in aortic involvement between the CCF and VCRC cohorts.
Cluster analysis
Correlation of each pair of the 15 specific large arteries was assessed using a 2×2 correlation matrix. Results from the ϕ coefficient correlation matrices for TAK and GCA are presented elsewhere (see online supplementary appendix 1). For 10 of the 12 paired arteries studied in TAK and 8 of the 12 paired arteries in GCA, the highest correlation was observed with the contralateral arterial bed. In GCA and TAK, the thoracic aorta most strongly correlated with the abdominal aorta. Sensitivity analyses excluding wall thickness in the definition of arterial disease did not change the degree of correlation in paired branch vessels but the thoracic aorta no longer correlated strongly with the abdominal aorta (data not shown).
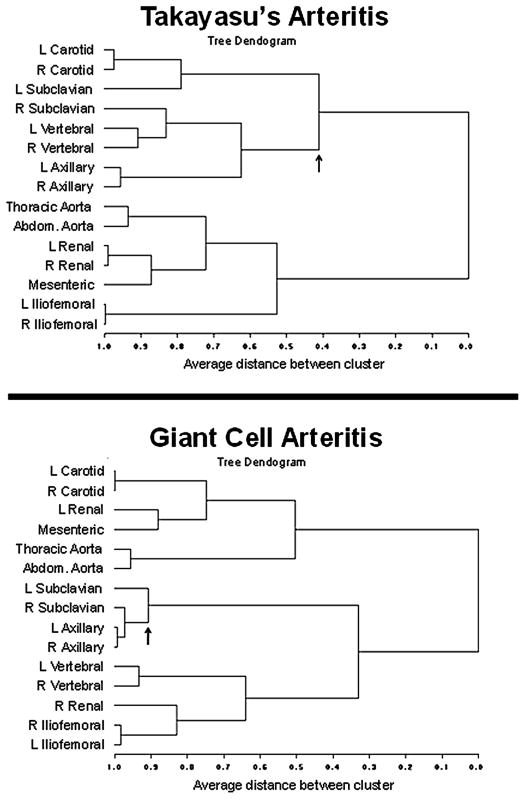
Tree dendograms demonstrated similarities and differences in clustering patterns between TAK and GCA (figure 1). In TAK and GCA, arterial disease tended to be symmetric in paired branch vessels and contiguous in the aorta. Subclavian disease clustered asymmetrically in TAK with the split point between clusters containing the right and left subclavian arteries occurring late into the clustering algorithm. In contrast, subclavian disease tended to be symmetric in GCA with the split point between the subclavian arteries occurring early in the clustering algorithm. Left subclavian disease in TAK clustered with the carotid arteries. In GCA, both subclavian arteries clustered more closely with the axillary arteries than the carotid arteries. Cluster patterns were consistent across different measures of similarity and when stratified by cohort.
Figure 1.
Cluster patterns in Takayasu’s arteritis and giant cell arteritis. Arrows denote the split point between clusters containing the right and left subclavian arteries. Tree dendograms in this figure were derived using ϕ correlation coefficients.
When patients were categorised according to age at disease onset, there were 120 patients <40 years of age at disease onset, and all were clinically diagnosed with TAK. There were 28 patients 40–55 years old at disease onset (TAK 21; GCA 7). There were 55 patients >55 years at disease onset, and all were clinically diagnosed with GCA. Tree dendograms, restricted to arteries above the diaphragm to reduce the number of variables in the model, are presented for each age category (figure 2). Subclavian disease clustered asymmetrically (similar to TAK) in those <40 years and aged 40–55 years at disease onset and clustered symmetrically (similar to GCA) in patients >55 years at disease onset.
Figure 2.
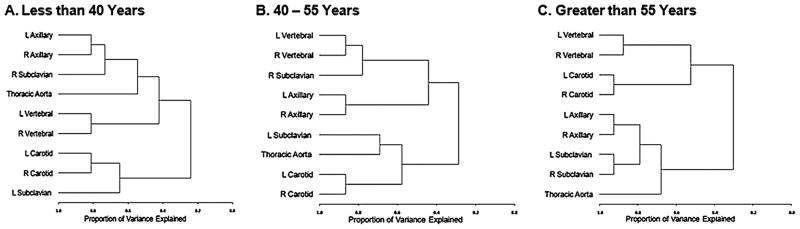
Cluster patterns stratified by age at disease onset. Patterns of arterial disease for patients in the <40 and the 40–55 year age groups at the time of disease onset resembled TAK, and arterial patterns for patients in the >55 year age group resembled giant cell arteritis. Cluster patterns in this figure were derived using principal component analysis.
Latent class analysis
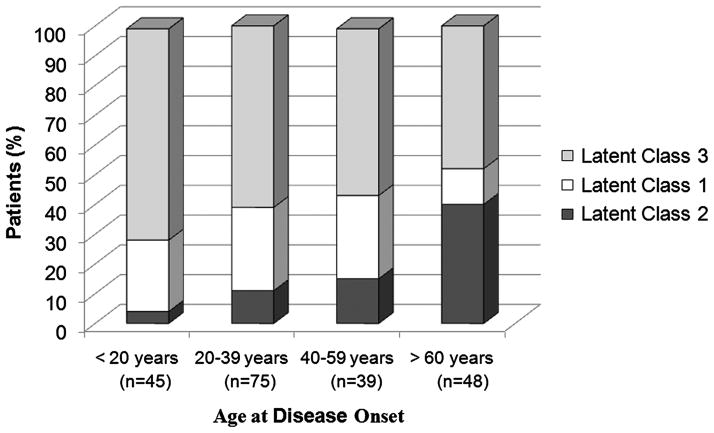
Model fit statistics suggested that a latent class model with three latent classes best fits the dataset. The probabilities that a patient was assigned to a certain latent class based upon class membership probability scores were: latent class 1: 26%; latent class 2: 18%; and latent class 3: 56% (see online supplementary appendix 2). Latent class 1 was defined in part by lack of disease in the axillary arteries, the highest probability of carotid and mesenteric artery involvement, and the highest probability of asymmetric subclavian disease. Posterior probability scores for latent class 1 were significantly associated with a diagnosis of TAK (OR=2.71; 95% CI 1.11 to 6.66). Latent class 2 was defined in part by high probability for bilateral axillary and bilateral subclavian involvement. Posterior probability scores for latent class 2 were significantly associated with a diagnosis of GCA (OR=5.56; 95% CI 2.49 to 12.50). The majority of patients had the highest probability for inclusion into latent class 3. Posterior probability scores for latent class 3 were associated with a diagnosis of GCA (OR=1.76; 95% CI 0.90 to 3.43), but the magnitude of association was small and not statistically significant. To visualise the relationship between latent class membership probability scores and patient age, the percentage of patients assigned to each latent class were stratified by age at disease onset (figure 3).
Figure 3.
Latent class membership stratified by age at disease onset. Membership in latent class 1 was associated with an increased likelihood for Takayasu’s arteritis. Membership in latent class 2 was associated with an increased likelihood for giant cell arteritis and was more frequent in subjects >60 years at disease onset. Membership in latent class 3 was not associated with an increased likelihood for either Takayasu’s arteritis or giant cell arteritis and comprised the majority of patients with large vessel vasculitis across all age strata.
DISCUSSION
When comparing the distribution of arteriographic lesions in patients with large vessel vasculitis, there were strong similarities in the frequency and patterns of arterial involvement between TAK and GCA. There were no significant differences in the frequency of arterial involvement between TAK and GCA for 11 out of 15 arteries of interest. Findings from cluster analysis demonstrated that arterial involvement was generally symmetric in paired vessels and contiguous in the aorta in TAK and GCA, which confirms reported patterns of arterial involvement in TAK8 and extends these patterns to include GCA. Using latent class analysis as an alternative way to classify patients with large vessel vasculitis, modelling based upon patterns of arterial involvement did not strongly differentiate between TAK and GCA in the majority of cases. Overall, these findings provide evidence to support the hypothesis that TAK and GCA exist on a spectrum within the same disease.
There were a few differences in patterns of arterial involvement between TAK and GCA. Carotid and mesenteric arterial disease was seen more frequently in TAK, and axillary disease was more frequent in GCA. Subclavian disease tended to be asymmetric in TAK with a high frequency of left subclavian artery involvement, and symmetric subclavian with concomitant axillary involvement was seen more frequently in GCA.
Age at disease onset is strongly incorporated into the existing classification criteria for TAK and GCA. In this study, analyses of arterial patterns based on age at disease onset rather than the clinical diagnoses circumvent the circularity of age as a primary determinant of disease classification. As evidenced by latent class analysis, patterns of arterial involvement in the majority of patients with large vessel vasculitis were similar across all age strata and were not associated with traditional diagnoses. However, in a subset of patients, differences in patterns of arterial disease that associated with traditional diagnoses were observed across different age strata. Arterial disease patterns in patients who were between 40 and 55 years of age at disease onset, a challenging subgroup of patients to classify using existing classification criteria, resembled TAK rather than GCA.
In patients with TAK, patterns of arterial disease observed in this study were similar to patterns reported in the study by Arnaud et al8 with one notable exception: subclavian disease in TAK clustered asymmetrically in the current study and symmetrically in the prior study. However, in the study by Arnaud et al, there was a suggestion of asymmetric subclavian involvement (left subclavian disease – 68%; right subclavian disease – 50%) and differences in sample size (82 vs 145 patients) may account for the differences in subclavian artery cluster patterns between the two studies. Even though asymmetric subclavian disease is not a universal feature of TAK, a high prevalence of left subclavian disease has been observed in several cohorts.14–18
The current study has several strengths. This study has the largest sample size to date for a comparative study between TAK and GCA. Additionally, sophisticated analytic techniques (cluster analysis, latent class analysis) were used to compare complex patterns of arterial disease in TAK and GCA. To minimise the potential that results were merely artefacts of computer-generated algorithms, cluster analysis findings were confirmed across different measures of similarity, quantified using latent class analysis and assessed within the clinical context of disease classification.
This study also has some potential limitations to consider. Since there are no existing guidelines regarding screening for large vessel involvement in patients with GCA, selection bias could have made arterial disease patterns in TAK and GCA more similar. Patients with GCA underwent angiography only when there was clinical suspicion of large vessel disease. It is possible that arterial disease in this selected subset of patients may not adequately represent the spectrum of arterial involvement in patients with GCA. Angiograms were not scored by two independent reviewers; however, the consistency of the findings across two independent cohorts (VCRC and CCF) provides reassurance that the reliability of angiogram assessment for arterial lesions was high. Additionally, angiography may not reflect the full extent of arterial disease in comparison with histology as the gold standard. The impact of duration of disease and immunosuppressive treatment on the distribution of arterial disease was also not assessed.
These study findings have important implications for future classification criteria in large vessel vasculitis. Although patterns of arterial involvement are only one aspect of clinical comparison between TAK and GCA, the strong similarities observed in TAK and GCA provide evidence to support the theory that these diseases exist on a spectrum within the same disease. Furthermore, the subtle differences in patterns of arterial involvement may represent age-related phenotypic variation of the same disease. Alternatively, arteriography may not meaningfully contribute to the classification of these conditions. Future classification criteria should explore the possibility of redefining subsets of disease within large vessel vasculitis. For example, the presence or absence of cranial involvement may be more clinically relevant than age at disease onset when distinguishing between subgroups of patients with large vessel vasculitis.
In conclusion, patterns of arterial disease were similar between TAK and GCA. For both diseases, arterial involvement was often symmetric in paired arteries and contiguous in the aorta. These findings suggest that rather than being distinct diseases, TAK and GCA may exist on a spectrum within the same disease.
Supplementary Material
Acknowledgments
This work was sponsored by the Vasculitis Clinical Research Consortium which has received support from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (U54AR057319 and U01 AR51874 04), the National Center for Research Resources (U54 RR019497), and the Office of Rare Diseases Research. Dr Grayson also received support from an NIH Training Program in Rheumatic Diseases grant (AR 007598) and through a Rheumatology Scientist Development Award from the Research and Education Foundation of the American College of Rheumatology.
Funding US National Institutes of Health.
Footnotes
Additional appendixes are published online only. To view these files please visit the journal online (http://ard.bmj.com/content/early/recent).
Contributors Study conception and design: PCG, K M-M, GSH, PAM. Acquisition of data: all authors. Analysis and interpretation of data: all authors.
Competing interests None.
Ethics approval Multiple IRB/Ethics Committees in USA and Canada.
Provenance and peer review Not commissioned; externally peer reviewed.
References
- 1.Maksimowicz-McKinnon K, Clark TM, Hoffman GS. Takayasu arteritis and giant cell arteritis: a spectrum within the same disease? Medicine (Baltimore) 2009;88:221–6. doi: 10.1097/MD.0b013e3181af70c1. [DOI] [PubMed] [Google Scholar]
- 2.García-Martínez A, Hernández-Rodríguez J, Arguis P, et al. Development of aortic aneurysm/dilatation during the followup of patients with giant cell arteritis: a cross-sectional screening of fifty-four prospectively followed patients. Arthritis Rheum. 2008;59:422–30. doi: 10.1002/art.23315. [DOI] [PubMed] [Google Scholar]
- 3.Nuenninghoff DM, Hunder GG, Christianson TJ, et al. Incidence and predictors of large-artery complication (aortic aneurysm, aortic dissection, and/or large-artery stenosis) in patients with giant cell arteritis: a population-based study over 50 years. Arthritis Rheum. 2003;48:3522–31. doi: 10.1002/art.11353. [DOI] [PubMed] [Google Scholar]
- 4.Cid MC, Prieto-González S, Arguis P, et al. The spectrum of vascular involvement in giant-cell arteritis: clinical consequences of detrimental vascular remodelling at different sites. APMIS Suppl. 2009;127:10–20. doi: 10.1111/j.1600-0463.2009.02471.x. [DOI] [PubMed] [Google Scholar]
- 5.Ostberg G. Morphological changes in the large arteries in polymyalgia arteritica. Acta Med Scand Suppl. 1972;533:135–59. [PubMed] [Google Scholar]
- 6.Arend WP, Michel BA, Bloch DA, et al. The American College of Rheumatology 1990 criteria for the classification of Takayasu arteritis. Arthritis Rheum. 1990;33:1129–34. doi: 10.1002/art.1780330811. [DOI] [PubMed] [Google Scholar]
- 7.Fries JF, Hunder GG, Bloch DA, et al. The American College of Rheumatology 1990 criteria for the classification of vasculitis. Summary. Arthritis Rheum. 1990;33:1135–6. doi: 10.1002/art.1780330812. [DOI] [PubMed] [Google Scholar]
- 8.Arnaud L, Haroche J, Toledano D, et al. Cluster analysis of arterial involvement in Takayasu arteritis reveals symmetric extension of the lesions in paired arterial beds. Arthritis Rheum. 2011;63:1136–40. doi: 10.1002/art.30240. [DOI] [PubMed] [Google Scholar]
- 9.Watts RA, Suppiah R, Merkel PA, et al. Systemic vasculitis–is it time to reclassify? Rheumatology (Oxford) 2011;50:643–5. doi: 10.1093/rheumatology/keq229. [DOI] [PubMed] [Google Scholar]
- 10.Massart DLK. The interpretation of analytical chemical data by the use of cluster analysis. Wiley (New York) 1983:65. [Google Scholar]
- 11.Bishop YM, Feinberg SE, Holland PW. Discrete multivariate analysis: theory and practice. Cambridge (MA): MIT Press; 1975. p. 557. [Google Scholar]
- 12.Harris B. Tetrchoric correlation coefficient. In: Kotz L, Johnson NL, editors. Encyclopedia of statistical sciences. Vol. 9. 1998. pp. 223–5. [Google Scholar]
- 13.Lanza ST, Collins LM, Lemmon DR, et al. PROC LCA: A SAS procedure for latent class analysis. Struct Equ Modeling. 2007;14:671–94. doi: 10.1080/10705510701575602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Maksimowicz-McKinnon K, Clark TM, Hoffman GS. Limitations of therapy and a guarded prognosis in an American cohort of Takayasu arteritis patients. Arthritis Rheum. 2007;56:1000–9. doi: 10.1002/art.22404. [DOI] [PubMed] [Google Scholar]
- 15.Kerr GS, Hallahan CW, Giordano J, et al. Takayasu arteritis. Ann Intern Med. 1994;120:919–29. doi: 10.7326/0003-4819-120-11-199406010-00004. [DOI] [PubMed] [Google Scholar]
- 16.Ueda H, Morooka S, Ito I, et al. Clinical observation of 52 cases of aortitis syndrome. Jpn Heart J. 1969;10:277–88. doi: 10.1536/ihj.10.277. [DOI] [PubMed] [Google Scholar]
- 17.Lupi-Herrera E, Sánchez-Torres G, Marcushamer J, et al. Takayasu’s arteritis. Clinical study of 107 cases. Am Heart J. 1977;93:94–103. doi: 10.1016/s0002-8703(77)80178-6. [DOI] [PubMed] [Google Scholar]
- 18.Jain S, Kumari S, Ganguly NK, et al. Current status of Takayasu arteritis in India. Int J Cardiol. 1996;54 (Suppl):S111–16. doi: 10.1016/s0167-5273(96)88780-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.