Abstract
Many studies in humans have shown that adverse experience in early life is associated with accelerated reproductive timing, and there is comparative evidence for similar effects in other animals. There are two different classes of adaptive explanation for associations between early-life adversity and accelerated reproduction, both based on the idea of predictive adaptive responses (PARs). According to external PAR hypotheses, early-life adversity provides a ‘weather forecast’ of the environmental conditions into which the individual will mature, and it is adaptive for the individual to develop an appropriate phenotype for this anticipated environment. In internal PAR hypotheses, early-life adversity has a lasting negative impact on the individual's somatic state, such that her health is likely to fail more rapidly as she gets older, and there is an advantage to adjusting her reproductive schedule accordingly. We use a model of fluctuating environments to derive evolveability conditions for acceleration of reproductive timing in response to early-life adversity in a long-lived organism. For acceleration to evolve via the external PAR process, early-life cues must have a high degree of validity and the level of annual autocorrelation in the individual's environment must be almost perfect. For acceleration to evolve via the internal PAR process requires that early-life experience must determine a significant fraction of the variance in survival prospects in adulthood. The two processes are not mutually exclusive, and mechanisms for calibrating reproductive timing on the basis of early experience could evolve through a combination of the predictive value of early-life adversity for the later environment and its negative impact on somatic state.
Keywords: developmental plasticity, predictive adaptive response, early-life stress, life history, humans
1. Introduction
Many studies have shown that humans adopt different reproductive schedules according to the early environment that they experience. For example, girls born with low birth weight go on to mature and have their first child relatively early [1–4], as do girls who have experienced psychosocial stress and family disruption in childhood [5–10]. The evidence linking early-life environment to reproductive timing in humans is necessarily correlational (though see [11] for a natural experiment). However, there are genuinely experimental non-human systems that have provided evidence of similar effects [12–15].
Given the breadth of evidence for early-life adversity accelerating reproduction, it is widely agreed that such conditional acceleration must have an adaptive basis. Proposals for what the adaptive advantage of accelerating reproduction in response to early adversity might be can be divided into two general classes. Proposals of both classes can be seen as belonging to the general category of predictive adaptive response (PAR; [16,17]) hypotheses, but they differ in what exactly is being predicted. In external PAR accounts [5,18,19], the early-life environment is hypothesized to provide the developing individual with a ‘weather forecast’ of the kind of adult environment into which she will mature [20]. It is thus advantageous for her to respond by developing a reproductive schedule appropriate for that anticipated environment. For example, according to one version of the hypothesis, the prevailing local rate of extrinsic mortality through the reproductive period determines the optimal reproductive strategy, and early-life stress and low parental investment might be cues that this rate is high [21].
The second class of explanation has been articulated recently by Wells [22], who argues that adverse early conditions cause the development of a soma that is less likely to survive at any given age, whatever the subsequent state of the external environment. The reduced longevity may arise as a consequence of reduced energy available to build somatic tissue during development, reduced self-repair or an increase in the rate of damage processes such as oxidative stress [22–26]. There is abundant empirical evidence for negative effects of early conditions on adult survival, both in humans and other animals [27–31]. On this view, it is adaptive to mature early following early adversity to increase the chances of completing at least some reproduction in the lifetime (see [32,33], for related ideas concerning stressful ontogeny accelerating age-related decline in health). This idea can also be seen as a PAR hypothesis, but what individuals are ‘predicting’ is not the state of the external environment during their adulthood, but rather the future state of their own body.
There has been more previous theory relevant to the external PAR account than to the internal PAR. External PARs are a type of environmental morph determination, and environmental morph determination can only evolve if the cue used to determine phenotype is sufficiently reliable (in the sense of giving accurate information [34]) [35–38]. For the current case, this condition can be decomposed into two sub-requirements: the cue has to be a statistically valid indicator of the state of the environment at the time the cue is received, and the environmental conditions at the time the cue is received have to be likely to persist until the selected life-history tactics impact fitness. In the cases where the empirical evidence for external PARs is very strong, these conditions appear to be met. For example, Storm & Lima [39] exposed gravid field crickets to a predatory spider and showed that the offspring of these mothers exhibited enhanced anti-predator responses to predatory spiders and were more likely to survive in an environment containing such a spider than those who did not receive the maternal cues. In this case, the cue validity is high (the cues came from the gravid mother's direct exposure to the spider), and the environmental persistence is likely to be high, as the crickets face the predation threat only a few days after receiving the prenatal cue and in the same location. Similarly, there are compelling cases of external PARs in plants where seed dispersal is limited, and thus offspring are more or less guaranteed to develop in the same light patch that the parent did [40].
However, whether calibration of reproductive timing by early-life experience in humans could be adaptive in the way envisaged by the external PAR model is debated and not at present clear [41–43]. Several authors [44–46] have argued that external PAR hypotheses are implausible, since the degree of environmental persistence over the length of lifetimes during human evolution is likely to have been insufficient for early experience to be useful as a guide to the later external environment. Baig et al. [47] provide a simple model of the evolution of external PARs, showing that increasing longevity and decreasing environmental persistence disfavour calibration of adult life-history traits by early experience. However, since Baig et al.'s [47] model is based on a simplified environment with only two states (famine and non-famine), it does not yield a criterion for what constitutes ‘sufficient’ environmental persistence in a form that would be easily testable empirically. This is important, since using even a relatively inaccurate weather forecast might provide some adaptive advantage on average [48–50]. We are not yet in a position to specify just how accurate it would have to be for this to be true, or test whether human environments fulfil the criteria for such accuracy to obtain.
In this paper, we explore theoretically the conditions under which it could be adaptive to calibrate reproductive timing according to early-life experience. Our model is the first to include the possibility of both external- and internal-type PAR effects within the same framework, in order to understand how these two potential adaptive processes interact, and which effect is likely to be more important. The internal and external PAR mechanisms are not mutually exclusive. For example, early-life experience might be useful for setting adult life history because of a combination of moderate persistence in the external environment, plus a moderate impact of childhood adversity on adult somatic state. Our model is based on a continuously varying environment and specifically parametrized for the case of human life history, so as to facilitate the empirical investigation of whether reproductive acceleration in response to early-life adversity is adaptive in particular human environments.
In what follows, we set aside questions of the constitutive costs of plasticity (the costs of developing and maintaining the physiological mechanisms involved), and also of why the adult phenotype has to be set early in life rather than remaining uncommitted until immediately before maturation. Instead, we ask, if a cue to the state of the world is available at the beginning of life, under what conditions would natural selection favour using it to set adult phenotype, rather than ignoring it and developing in a non-plastic, genetically determined manner? We first examine the case where the optimal adult strategy is set entirely by external factors such as climate and predation, and the adaptive relevance of early-life experience is the information it provides about these external factors (allowing for an external PAR). We then consider a case where the optimal adult strategy is set by a combination of external environmental factors and somatic state, with somatic state being affected by the level of adversity experienced in early life (allowing for internal as well as external PARs).
2. Methods and results
(a). Basic modelling framework
Table 1 summarizes the key parameters of the model. We model organisms living in an environment that fluctuates from year to year on some continuous variable M. We are envisaging M as the rate of extrinsic mortality, the parameter which determines the optimal reproductive schedule an adult should follow, although M could be equally well interpreted as any other fitness-relevant environmental parameter. Over evolutionary time, M follows a normal distribution with mean  and a standard deviation of 1. In any particular year, M takes the value mt. In the basic model, mt is set entirely by factors external to the individual. We use an autoregressive procedure (see the electronic supplementary material, §1) to generate environments with varying degrees of temporal autocorrelation, henceforth r. The parameter r gives the correlation between successive years, so that when r is close to zero, next year is no more like this year than it is like any randomly chosen year, whereas when r is close to 1, this year's m is a very good guide to next year's. Figure 1 gives an illustration of the environments generated by this procedure for three different values of r. With r > 0.7, distinct runs of good years or bad years begin to emerge. In the electronic supplementary material, §1, we show that the mean length of such runs is approximately equal to 1 + 1/(1 − 0.95r). Thus, with r = 0.95, a year that is worse than
and a standard deviation of 1. In any particular year, M takes the value mt. In the basic model, mt is set entirely by factors external to the individual. We use an autoregressive procedure (see the electronic supplementary material, §1) to generate environments with varying degrees of temporal autocorrelation, henceforth r. The parameter r gives the correlation between successive years, so that when r is close to zero, next year is no more like this year than it is like any randomly chosen year, whereas when r is close to 1, this year's m is a very good guide to next year's. Figure 1 gives an illustration of the environments generated by this procedure for three different values of r. With r > 0.7, distinct runs of good years or bad years begin to emerge. In the electronic supplementary material, §1, we show that the mean length of such runs is approximately equal to 1 + 1/(1 − 0.95r). Thus, with r = 0.95, a year that is worse than  is followed by an average of around 10 more which are also worse than
is followed by an average of around 10 more which are also worse than  .
.
Table 1.
Summary description of the key parameters of the model.
| parameter | description |
|---|---|
| mt | the level of extrinsic mortality in the environment in year t |
| qt | the level of the cue of extrinsic mortality that the individual receives in year t |
| r | the degree of year-to-year autocorrelation of the level of extrinsic mortality in the environment |
| v | the validity of qt as a predictor of mt |
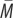 |
the mean of M over evolutionary time |
| d | the extent to which an adult's mortality prospects are influenced by her exposure to environmental harshness in early life |
Figure 1.

Illustrative 200-year periods of values of M for different levels of annual autocorrelation, (a) r = 0, (b) r = 0.7 and (c) r = 0.95. In (b) and particularly (c), there emerge runs of good or bad conditions lasting many years.
Individuals have access to a cue each year (qt) to what mt is. The validity of qt is governed by a parameter v (0 ≤ v ≤ 1), such that v = 0 means that qt carries no information about mt, while v = 1 means that qt perfectly predicts mt. We assume the parameter that determines the individual's optimal adult phenotype is the mean value of M over the years during which she may start reproducing. Based on data from traditional societies [51], we take these to be 16–25, and henceforth, we refer to the mean of m16–m25 as the adult environment. We consider two scenarios. In 1-year sampling, the individual uses q1, the cue received the year she is born, to estimate the adult environment and set her phenotype accordingly. In 5-year sampling, she uses the mean of q1–q5 instead. We investigated 5-year sampling since several approaches to the impact of early life on development have stressed that individuals do not use a single brief cue, but integrate their experience over a number of years of early childhood [5,17].
(b). Adaptive value of external predictive adaptive response
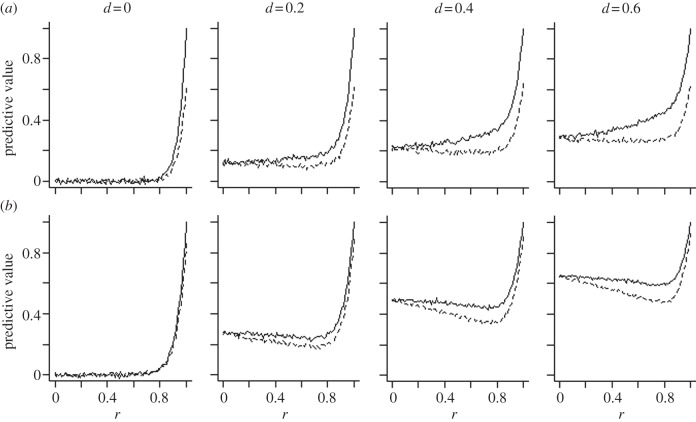
We first consider what the value of the early-life cues is for predicting the adult external environment, for varying values of the temporal autocorrelation r and the cue validity v. We express this predictive value in terms of the regression coefficient of adult environment on early-life cues across a set of 10 000 simulated lifetimes. As figure 2 shows, the predictive value of early-life cues for adult environment declines dramatically as the environmental autocorrelation r decreases from 1 and is essentially zero for r < 0.8. This remains true whether 1-year sampling or 5-year sampling is used. Indeed, the main impact of 5-year sampling is to compensate for low cue validity: note that in figure 2b as compared with figure 2a, there is little difference in predictive value between a cue of validity 1 and a cue of validity 0.6. This is because sampling multiple times improves validity of the aggregate sample, a principle well known in psychometrics and measurement theory. In the electronic supplementary material, §2, we show why the predictive value falls off so steeply with decreasing r by showing that the predictive value of any year for a time point k years in the future is given by 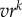 . As long as r < 1, this very rapidly declines to near zero with increasing k.
. As long as r < 1, this very rapidly declines to near zero with increasing k.
Figure 2.

Predictive value of early-life experience for adult environment as a function of the annual autocorrelation r of the environment. The three lines show different levels of the cue validity v, v = 1 (solid line), v = 0.8 (dashed line), v = 0.6 (dotted line). (a) Represents 1-year sampling, where cues from the first year of life only are used, and (b) 5-year sampling, where the mean of the first 5 years is used. Data represent 10 000 simulated lifetimes for each 0.01 increment of r and each value of v.
Even a fairly inaccurate estimate of the future environment could be worth acting on if the benefits of being prepared for possible negative circumstances were sufficiently large. Thus, it is impossible to infer from figure 2 alone, where the ‘cut-off’ in terms of r and v is for an external PAR to be potentially adaptive. However, natural selection could only favour the evolution of an external PAR if using the cues received leads on average to a better match to the adult environment than would be obtained by ignoring the cues and following a genetically fixed developmental strategy instead. In this case, if there were no plasticity, then absent other constraints or complicating factors, natural selection would optimize organisms for the situation where m in the adult environment is always 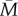 . Thus, we can ask, under what combinations of r and v does the use of early-life cues lead to a phenotype better matched to the adult environment than the fixed strategy of always developing the optimal phenotype for
. Thus, we can ask, under what combinations of r and v does the use of early-life cues lead to a phenotype better matched to the adult environment than the fixed strategy of always developing the optimal phenotype for 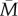 ?
?
To do this, we simulated 2000 lifetimes for each combination of r and v (r and v varying from 0 to 1 in increments of 0.01). For both 1-year sampling and 5-year sampling, we computed the mean of the predictive error, which is the absolute discrepancy between the early-life cues received and the adult environment subsequently experienced, and the mean of the fixed error, which is the absolute discrepancy between 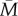 and the adult environment experienced. If the predictive error is on average smaller than the fixed error, then a strategy of using early-life cues to set adult phenotype could be advantageous compared with a genetically fixed strategy. If on the other hand the predictive error is on average larger than the fixed error, then using early-life cues to set adult phenotype could never be advantageous, since using those cues leads on average to a greater mismatch of phenotype to adult environment than is obtained by ignoring the cues and assuming that the adult environment will simply be the mean of adult environments experienced by the lineage over evolutionary time.
and the adult environment experienced. If the predictive error is on average smaller than the fixed error, then a strategy of using early-life cues to set adult phenotype could be advantageous compared with a genetically fixed strategy. If on the other hand the predictive error is on average larger than the fixed error, then using early-life cues to set adult phenotype could never be advantageous, since using those cues leads on average to a greater mismatch of phenotype to adult environment than is obtained by ignoring the cues and assuming that the adult environment will simply be the mean of adult environments experienced by the lineage over evolutionary time.
In figure 3, the dark region shows the combination of parameters for which the predictive error is smaller than the fixed error. For 1-year sampling, this is basically restricted to r ≥ 0.96 and v > 0.5. Five-year sampling expands the region where the predictive error is smaller than the fixed error somewhat, particularly in the direction of low cue validity v. However, responding to early-life cues still only leads to a better match to the adult environment where r ≥ 0.95. For all other combinations of parameters, early-life cues lead to worse estimates of adult environment than simply assuming 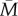 . This is because early-life cues, being based on just a few years’ information, often lead to relatively non-central estimates of what adult environmental conditions will be, whereas actual adult experience is often closer to
. This is because early-life cues, being based on just a few years’ information, often lead to relatively non-central estimates of what adult environmental conditions will be, whereas actual adult experience is often closer to 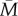 than these estimates (see the electronic supplementary material, §3). Thus, selection would favour simply assuming
than these estimates (see the electronic supplementary material, §3). Thus, selection would favour simply assuming 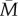 over taking early-life cues as a guide to the adult environment in all but the dark-shaded region of figure 3. This result is not substantially different for a strategy that assumes the adult environment will be halfway between the early-life environment and
over taking early-life cues as a guide to the adult environment in all but the dark-shaded region of figure 3. This result is not substantially different for a strategy that assumes the adult environment will be halfway between the early-life environment and 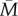 , a scenario that combines the lineage's evolutionary experience and the individual's childhood experience with equal weight (see the electronic supplementary material, §4).
, a scenario that combines the lineage's evolutionary experience and the individual's childhood experience with equal weight (see the electronic supplementary material, §4).
Figure 3.
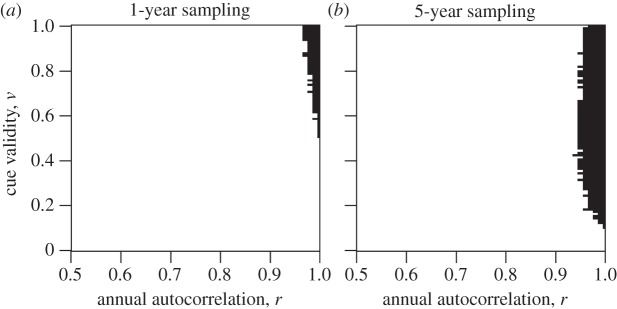
Regions of parameter space (shaded dark) in which an individual ends up on average better matched to her adult environment by using early-life cues to set adult phenotype, rather than following a genetically fixed strategy where she develops matched to the mean of conditions experienced by the lineage over evolutionary time. Data represent 2000 simulated lifetimes for each parameter combination.
(c). Adaptive value of predictive adaptive response based on both external conditions and somatic state
In this section, we expand the model to include the possibility that conditions experienced in early life have a direct impact on the values of M experienced as an adult. This could arise if stress during the developmental period leads to the individual building a soma which is smaller or of poorer quality, such that it will be more likely to fail over the adult period whatever the state of the external environment at that time. To capture this dependency, for each individual, we make M in adulthood depend additively on both external environmental conditions and somatic state. Somatic state is in turn influenced by q1 − 5, the conditions experienced during the early-life period (see the electronic supplementary material, §4 for implementation). The strength of this influence is captured by a new parameter d, with higher d representing a stronger influence of early-life adversity on somatic state. The model analysed in §2b is the special case of this model where d = 0.
We simulated 10 000 lifetimes and computed the predictive power of early-life experience for adult outcome for four different values of d (figure 4a represents 1-year sampling and row (figure 4b) 5-year sampling). As the figure shows, a non-zero d greatly alleviates the decline in predictive power as r decreases from 1. With d ≥ 0.5, early-life experience has a high predictive value for adult outcome even when r is close to zero. This is particularly true for 5-year sampling. This result is unsurprising since in 5-year sampling, the period used by the individual to estimate its future prospects is identical to the period where somatic state gets determined under the assumptions of our model.
Figure 4.
Predictive power of early-life experience for adult environment outcome as a function of r, the degree of annual autocorrelation, for different values of d, the impact of early-life experience on adult internal state. (a) Represents 1-year sampling and (b) 5-year sampling. Solid lines represent v = 1 and dashed lines v = 0.6. Data represent 10 000 simulated lifetimes for each parameter combination.
We again considered whether the plastic error is on average smaller than the fixed error across the range of r and v, with increasing values of d. The results are shown in figure 5. As d increases, the region of the parameter space in which calibrating adult life history to early-life experience produces a better match to adult prospects rapidly becomes larger. More exactly, the minimum required environmental autocorrelation r and cue validity v are progressively relaxed by increasing d, more rapidly for 5-year sampling than for 1-year sampling. For 5-year sampling, once d > 0.5, then it is always adaptive to calibrate to early experience, regardless of r and v.
Figure 5.
Regions of parameter space (shaded dark) in which an individual ends up on average better matched to her adult environment by using early-life cues to set adult phenotype, rather than following a genetically fixed strategy where she develops matched to the mean of conditions experienced by the lineage over evolutionary time, for five values of d, the effect of early experience on somatic state. Data represent 2000 simulated lifetimes for each parameter combination.
3. Discussion
Our model allowed us to explore the conditions that must be met in order for a PAR of human reproductive timing to early-life experience to be selected for, given that multiple years elapse between the early-life period and the expression of the phenotype. We will now discuss implications for the evolution of PARs of both the external and internal types.
(a). External predictive adaptive responses: the weather forecast
Classic statements of the external PAR hypothesis for the impact of early-life conditions on adult phenotype imply that early-life experience provides a forecast of what the external environment will be in adulthood. Our model leads to two main conclusions about the evolution of such a plastic response for the case of human reproductive timing. First, the cues would need to have high levels of validity for a PAR to evolve. That is, they would need to accurately reflect the parameters in the environment relevant to optimal adult reproductive timing, such as the rate of extrinsic mortality [21]. Thus, empirical and theoretical investigation is needed into how well and why proposed early-life growth and psychosocial variables actually relate to this parameter. A strength of our model is that since it is specifically parametrized for the case of human reproductive timing, its key variables could be feasibly estimated empirically from human datasets. Our validity parameter v is just the correlation coefficient between the early-life cue (e.g. the psychosocial stress variable) and the fitness-relevant external environment parameter (e.g. local mortality rates). This should be measurable for particular populations [41,52,53]. Our results regarding cue validity are consistent with theoretical literature on environmental morph determination [54,55] and on the evolution of learning [56,57]. Our model shows that sampling for multiple years partially compensates for low cue validity, and the same would be true for sampling multiple cues contemporaneously. Thus, the model would predict that plastic systems would evolve to make use of the maximum number of different sampling points or cues, especially if cue reliabilities are low.
Our second result is that the level of annual autocorrelation in the environment has to be extremely high for an external PAR for human reproductive timing to be advantageous. Theoreticians have already shown in a general case that environmental persistence is required in order for any kind of predictive plasticity to be adaptive [36,55,58]. Our results are also consistent with previous arguments that temporal environmental fluctuations reduce the adaptive value of external PARs over human lifespans [44,46,47]. Our model shows that, because of the geometric decay of inter-temporal correlation across the years, in order for there to be a substantial persistence of childhood environment to adulthood more than a decade later, the year-to-year autocorrelation of the environment has to be greater than 0.95. This is the level of autocorrelation required for the expected length of a run of successive good or bad years to be of the same order as the period between human birth and maturity. Thus, an external PAR for human reproductive strategy would only have the potential to be adaptive in environments characterized by annual autocorrelation of this magnitude. This has the advantage of being eminently testable: the annual autocorrelation coefficient corresponding to our parameter r could feasibly be directly estimated from any kind of environmental time-series data. If the annual autocorrelation coefficient is not of the order of 0.95 or above, then individuals will do better on average by assuming that the environment will regress to the mean than they do by taking their early-life experience as representative of the world into which they will mature (see [59], for conceptually related results on the optimal duration of memories).
The main contribution of our model to the debate on external PARs in humans is that specific testable criteria for adaptive advantage can be derived from it. The issues surrounding cue validity and environmental persistence in relation to human PARs have been discussed for some time, with arguments on both sides [41–44,48–50,60], but the question of whether the evolveability conditions for an external PAR are met should now be empirically addressable, at least to some extent. Climatic time series (e.g. rainfall) do not generally show autocorrelations of nearly the requisite strength [47]. However, there have been longer scale climatic fluctuations in the course of human evolution [61], and these could be relevant as long as the shorter term variability could be smoothed out by parental buffering, as discussed by Kuzawa [62]. There are also sources of self-consistency in the experienced environment that are social in origin. For example, if human history was often characterized by within-society differences in social status which determined access to resources, and these were rather consistent across the generations as they are in many extant societies [63], then these could provide a plausible basis for effective persistence of individual exposure to high mortality across periods of many years or decades [47].
However, even if the cue validity is high and the environmental autocorrelation very high, an external PAR will not necessarily evolve, for a number of reasons. First, plasticity may be costly, and so the net fitness benefits have not just to exceed zero, but rather, must exceed the constitutive costs of the mechanisms involved. Our model does not incorporate constitutive costs of plasticity since we have no non-arbitrary way of estimating these, but they are nonetheless likely to be important, and their effect will be to make the conditions on the evolution of external PARs more stringent than documented here. Second, because of the geometric decay of predictive power with the passing years (see the electronic supplementary material, figure S2), selection would always favour deferring phenotypic specialization until as close to adulthood as possible, since this would on average lead to a dramatically better fit to the adult environment. Thus, for an external PAR based on very early cues to be plausible, researchers would need to show that the trait was not one whose determination could be deferred until later in ontogeny, such as just before maturation. Third, if the annual autocorrelation becomes extremely high in the experience of a whole lineage, then the selective advantage of plasticity is lost, since the environment can be adapted to genetically [64], thereby avoiding the constitutive costs of plasticity. Thus, external PARs in long-lived organisms are likely to be restricted to cases where within-lifetime variation in the environment is very limited, but there is nonetheless sufficient between-lifetime variation for phenotypic plasticity to be adaptive [65].
(b). Internal predictive adaptive responses: differential weathering
We also considered the case of an internal PAR, where early-life conditions have a direct causal impact on the quality, and thus survival chances, of the soma the developing individual can construct. Our results show that this kind of PAR is likely to be adaptive over a much wider range of environmental conditions than a purely external PAR. Indeed, if the component of variation in adult mortality that is due to developmentally derived somatic state is sizeable compared with the component that is due to fluctuations in the adult external environment, then adjustment of life-history strategy based on early developmental history is always adaptive. Importantly, this is true even if there is no environmental autocorrelation at all, and/or if the early-life cues do not reflect prevailing environmental conditions (e.g. where a child develops in an idiosyncratically unsupportive home in a broader ecology that is quite benign). We would therefore suggest that if PAR-like impacts of early-life on adult life history turn out to be pervasive in long-lived organisms, this is plausibly because the direct impacts of early-life conditions on somatic state are substantial in such organisms. There is substantial empirical evidence for detrimental impacts of conditions experienced during early development on adult somatic state in humans and animals that is compatible with this view [27–31,66].
However, our analysis shows that the external PAR mechanism and internal PAR processes are not mutually exclusive. We explicitly considered a continuum of circumstances defined by our use of variable parameter d. At one extreme (d = 0) are cases, where early life provides only information about prevailing external circumstances, and so for a PAR to be adaptive requires very high cue validity and autocorrelation. At the other extreme (d large) is a purely internal situation, where cue validity and autocorrelation of the environment are not required for calibration of adult phenotype by early-life experience to be adaptive. In the middle are a host of intermediate possibilities, where early-life conditions have both some predictive validity for the external environment, and some implications for somatic state in adulthood. Deciding where on the d continuum the documented human cases fall will be challenging, but may be possible. For example, if the adult environment is harsh, then if d is close to zero, individuals who had harsh early lives should attain greater fitness than those who did not (because they were able to predict the coming conditions), whereas if d is substantial, then they will presumably have lower fitness than individuals from benign early environments, because they have been damaged by their early adversity. This would seem to be the key test in determining where on the external–internal continuum a particular case falls [66]. For example, it has been shown clearly that the maternal effects documented in the herb Campanulastrum americanum represent a PAR of the external, not the internal, type [40]. Such tests have not yet been carried out for human life history.
The results for sizable values of d also confirm that using the whole of early life as the calibrational input (5-year sampling) is generally more advantageous than taking just a single year as representative (1-year sampling). Thus, whether the source of the PAR is external environment or internal state, we ought to expect organisms to evolve to make use of as long a sampling window and as diverse a set of cues as possible. This might explain why, in epidemiological studies of the relationship between early-life events and adult life-history milestones, single early-life variables tend to have rather small effects, but effect sizes are larger when multiple early-life variables are combined into overall indices [6]. We have here assumed that d is an immutable fact of the organism's developmental biology. In reality, d, the degree to which early-life condition affects adult state, may itself be under selection, and could be affected by life-history trade-offs between reproductive effort and energy devoted to self-repair [67].
(c). General implications
We conceptualized our model in terms of the documented associations between early-life conditions and reproductive schedules in humans, but the modelling framework is general. It could thus be applied, with appropriate changes to the parameter values, to other developmentally sensitive phenotypic outcomes in humans, such as the stress response [68,69], and also to PARs in other species. There are cases of developmental plasticity in non-human organisms that appear to correspond to external PARs as defined here [70,71], and other cases that correspond to internal PARs [72,73]. Our model shows that the area of parameter space in which external PARs are adaptive is largest when the plastic phenotype has its fitness benefit immediately after receipt of the cue, and rapidly diminishes as the time gap between the early-life cue and the benefit of the phenotype becomes longer. This suggests that we should find more examples of external PARs in long-lived organisms where the benefit of the plastic phenotype is experienced in early life than when it accrues years later in adulthood, and that very delayed effects may more plausibly represent processes of the internal PAR type. Our model deals with PARs restricted to one generation, but the main conclusions would apply even more strongly to trans-generational effects [74]. For a trans-generational ‘weather forecast’, the conditions in terms of environmental persistence would be even more restrictive than those for an external PAR within a single generation, high enough to enable the persistence of environmental conditions across several generations, but low enough for genetic adaptation not to be possible. Where trans-generational inheritance of phenotype is found, multi-generational persistence of poor somatic state should be considered as one possible adaptive explanation.
References
- 1.Adair LS. 2001. Size at birth predicts age at menarche. Pediatrics 107, e59 (doi:10.1542/peds.107.4.e59) [DOI] [PubMed] [Google Scholar]
- 2.Sloboda DM, Hart R, Doherty DA, Pennell CE, Hickey M. 2007. Rapid communication—age at menarche: influences of prenatal and postnatal growth. J. Clin. Endocrinol. Metab. 92, 46–50 (doi:10.1210/jc.2006-1378) [DOI] [PubMed] [Google Scholar]
- 3.Nettle D, Coall DA, Dickins TE. 2010. Birthweight and paternal involvement predict early reproduction in British women: evidence from the National Child Development Study. Am. J. Hum. Biol. 22, 172–179 (doi:10.1002/ajhb.20970) [DOI] [PubMed] [Google Scholar]
- 4.Cooper C, Kuh D, Egger P, Wadsworth M, Barker D. 1996. Childhood growth and age at menarche. Br. J. Obstet. Gynaecol. 103, 814–817 (doi:10.1111/j.1471-0528.1996.tb09879.x) [DOI] [PubMed] [Google Scholar]
- 5.Belsky J, Steinberg L, Draper P. 1991. Childhood experience, interpersonal development, and reproductive strategy: an evolutionary theory of socialization. Child Dev. 62, 647–670 (doi:10.2307/1131166) [DOI] [PubMed] [Google Scholar]
- 6.Nettle D, Coall DA, Dickins TE. 2011. Early-life conditions and age at first pregnancy in British women. Proc. R. Soc. B 278, 1721–1727 (doi:10.1098/rspb.2010.1726) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Belsky J, Steinberg LD, Houts RM, Friedman SL, DeHart G, Cauffman E, Roisman GI, Halpern-Felsher BL, Susman E. 2007. Family rearing antecedents of pubertal timing. Child Dev. 78, 1302–1321 (doi:10.1111/j.1467-8624.2007.01067.x) [DOI] [PubMed] [Google Scholar]
- 8.Ellis BJ, Essex MJ. 2007. Family environments, adrenarche, and sexual maturation: a longitudinal test of a life history model. Child Dev. 78, 1799–1817 (doi:10.1111/j.1467-8624.2007.01092.x) [DOI] [PubMed] [Google Scholar]
- 9.Tither JM, Ellis BJ. 2008. Impact of fathers on daughters’ age at menarche: a genetically and environmentally controlled sibling study. Dev. Psychol. 44, 1409–1420 (doi:10.1037/a0013065) [DOI] [PubMed] [Google Scholar]
- 10.Ellis BJ, Bates JE, Dodge KA, Fergusson DM, Horwood LJ, Pettit GS, Woodward L. 2003. Does father absence place daughters at special risk for early sexual activity and teenage pregnancy? Child Dev. 74, 801–821 (doi:10.1111/1467-8624.00569) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pesonen A-K, Räikkönen K, Heinonen K, Kajantie E, Forsén T, Eriksson JG. 2008. Reproductive traits following a parent–child separation trauma during childhood: a natural experiment during World War II. Am. J. Hum. Biol. 20, 345–351 (doi:10.1002/ajhb.20735) [DOI] [PubMed] [Google Scholar]
- 12.Maestripieri D. 2005. Effects of early experience on female behavioural and reproductive development in rhesus macaques. Proc. R. Soc. B 272, 1243–1248 (doi:10.1098/rspb.2005.3059) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sloboda DM, Howie GJ, Pleasants A, Gluckman PD, Vickers MH. 2009. Pre- and postnatal nutritional histories influence reproductive maturation and ovarian function in the rat. PLoS ONE 4, 8 (doi:10.1371/journal.pone.0006744) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cameron NM, Fish EW, Meaney MJ. 2008. Maternal influences on the sexual behavior and reproductive success of the female rat. Horm. Behav. 54, 178–184 (doi:10.1016/j.yhbeh.2008.02.013) [DOI] [PubMed] [Google Scholar]
- 15.Cameron NM, Shahrokh D, Del Corpo A, Dhir SK, Szyf M, Champagne FA, Meaney MJ. 2008. Epigenetic programming of phenotypic variations in reproductive strategies in the rat through maternal care. J. Neuroendocrinol. 20, 795–801 (doi:10.1111/j.1365-2826.2008.01725.x) [DOI] [PubMed] [Google Scholar]
- 16.Gluckman PD, Hanson MA, Spencer HG. 2005. Predictive adaptive responses and human evolution. Trends Ecol. Evol. 20, 527–533 (doi:10.1016/j.tree.2005.08.001) [DOI] [PubMed] [Google Scholar]
- 17.Gluckman PD, Hanson MA, Spencer HG, Bateson P. 2005. Environmental influences during development and their later consequences for health and disease: implications for the interpretation of empirical studies. Proc. R. Soc. B 272, 671–677 (doi:10.1098/rspb.2004.3001) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gluckman PD, Hanson MA. 2006. Evolution, development and timing of puberty. Trends Endocrinol. Metab. 17, 7–12 (doi:10.1016/j.tem.2005.11.006) [DOI] [PubMed] [Google Scholar]
- 19.Ellis BJ, Figueredo AJ, Schlomer GL. 2009. Fundamental dimensions of environmental risk: the impact of harsh versus unpredictable environments on the evolution and development of life history strategies. Hum. Nat. 20, 204–268 (doi:10.1007/s12110-009-9063-7) [DOI] [PubMed] [Google Scholar]
- 20.Bateson P, et al. 2004. Developmental plasticity and human health. Nature 430, 419–421 (doi:10.1038/nature02725) [DOI] [PubMed] [Google Scholar]
- 21.Chisholm JS. 1993. Death, hope, and sex: life-history theory and the development of reproductive strategies. Curr. Anthropol. 34, 1–24 (doi:10.1086/204131) [Google Scholar]
- 22.Wells JCK. 2012. Obesity as malnutrition: the role of capitalism in the obesity global epidemic. Am. J. Hum. Biol. 24, 261–276 (doi:10.1002/ajhb.22253) [DOI] [PubMed] [Google Scholar]
- 23.Monaghan P. In press. Organismal stress, telomeres and life histories. J. Evol. Biol. [DOI] [PubMed] [Google Scholar]
- 24.Selman C, Blount JD, Nussey DH, Speakman JR. 2012. Oxidative damage, ageing, and life-history evolution: where now? Trends Ecol. Evol. 27, 570–577 (doi:10.1016/j.tree.2012.06.006) [DOI] [PubMed] [Google Scholar]
- 25.Joergensen A, Broedbaek K, Weimann A, Semba RD, Ferrucci L, Joergensen MB, Poulsen HE. 2011. Association between urinary excretion of cortisol and markers of oxidatively damaged DNA and RNA in humans. PLoS ONE 6, e20795 (doi:10.1371/journal.pone.0020795) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Epel ES, Lin J, Wilhelm FH, Wolkowitz OM, Cawthon R, Adler NE, Dolbier C, Mendes WB, Blackburn EH. 2006. Cell aging in relation to stress arousal and cardiovascular disease risk factors. Psychoneuroendocrinology 31, 277–287 (doi:10.1016/j.psyneuen.2005.08.011) [DOI] [PubMed] [Google Scholar]
- 27.Lummaa V, Clutton-Brock T. 2002. Early development, survival and reproduction in humans. Trends Ecol. Evol. 17, 141–147 (doi:10.1016/S0169-5347(01)02414-4) [Google Scholar]
- 28.Lindstrom J. 1999. Early development and fitness in birds and mammals. Trends Ecol. Evol. 14, 343–348 (doi:10.1016/s0169-534701639-0) [DOI] [PubMed] [Google Scholar]
- 29.Reid JM, Bignal EM, Bignal S, McCracken DI, Monaghan P. 2006. Spatial variation in demography and population growth rate: the importance of natal location. J. Anim. Ecol. 75, 1201–1211 (doi:10.1111/j.1365-2656.2006.01143.x) [DOI] [PubMed] [Google Scholar]
- 30.Taborsky B. 2006. The influence of juvenile and adult environments on life-history trajectories. Proc. R. Soc. B 273, 741–750 (doi:10.1098/rspb.2005.3347) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rickard IJ, Holopainen A, Helema S, Helle S, Russell A, Lummaa V. 2010. Food availability at birth limited reproductive success in historical humans. Ecology 91, 3515–3525 [DOI] [PubMed] [Google Scholar]
- 32.Geronimus AT. 1996. What teen mothers know. Hum. Nat. 7, 323–352 (doi:10.1007/BF02732898) [DOI] [PubMed] [Google Scholar]
- 33.Geronimus AT, Hicken M, Keene D, Bound J. 2006. ‘Weathering’ and age patterns of allostatic load scores among blacks and whites in the United States. Am. J. Public Health 96, 826–833 (doi:10.2105/ajph.2004.060749) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Koops MA. 2004. Reliability and the value of information. Anim. Behav. 67, 103–111 (doi:10.1016/j.anbehav.2003.02.008) [Google Scholar]
- 35.Moran NA. 1992. The evolutionary maintenance of alternative phenotypes. Am. Nat. 139, 971–989 (doi:10.1086/285369) [Google Scholar]
- 36.Fischer B, Taborsky B, Kokko H. 2011. How to balance the offspring quality–quantity tradeoff when environmental cues are unreliable. Oikos 120, 258–270 (doi:10.1111/j.1600-0706.2010.18642.x) [Google Scholar]
- 37.Tufto J. 2000. The evolution of plasticity and nonplastic spatial and temporal adaptations in the presence of imperfect environmental cues. Am. Nat. 156, 121–130 (doi:10.1086/303381) [DOI] [PubMed] [Google Scholar]
- 38.Frankenhuis WE, Panchanathan K. 2011. Balancing sampling and specialization: an adaptationist model of incremental development. Proc. R. Soc. B 278, 3558–3565 (doi:10.1098/rspb.2011.0055) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Storm JJ, Lima SL. 2010. Mothers forewarn offspring about predators: a transgenerational maternal effect on behavior. Am. Nat. 175, 382–390 (doi:10.1086/650443) [DOI] [PubMed] [Google Scholar]
- 40.Galloway LF, Etterson JR. 2007. Transgenerational plasticity is adaptive in the wild. Science 318, 1134–1136 (doi:10.1126/science.1148766) [DOI] [PubMed] [Google Scholar]
- 41.Honekopp J. 2009. Pre-adjustment of adult attachment style to extrinsic risk levels via early attachment style is neither specific, nor reliable, nor effective, and is thus not an adaptation. Behav. Brain Sci. 32, 31 (doi:10.1017/s0140525X09000120) [Google Scholar]
- 42.Ellis BJ. 2004. Timing of pubertal maturation in girls. Psychol. Bull. 130, 920–958 (doi:10.1037/0033-2909.130.6.920) [DOI] [PubMed] [Google Scholar]
- 43.Del Giudice M. 2009. Sex, attachment, and the development of reproductive strategies. Behav. Brain Sci. 32, 1–21 (doi:10.1017/s0140525x09000016) [DOI] [PubMed] [Google Scholar]
- 44.Wells JCK. 2007. Flaws in the theory of predictive adaptive responses. Trends Endocrinol. Metab. 18, 331–337 (doi:10.1016/j.tem.2007.07.006) [DOI] [PubMed] [Google Scholar]
- 45.Jones JH. 2005. Fetal programming: adaptive life-history tactics or making the best of a bad start? Am. J. Hum. Biol. 17, 22–33 (doi:10.1002/ajhb.20099) [DOI] [PubMed] [Google Scholar]
- 46.Rickard IJ, Lummaa V. 2007. The predictive adaptive response and metabolic syndrome: challenges for the hypothesis. Trends Endocrinol. Metab. 18, 94–99 (doi:10.1016/j.tem.2007.02.004) [DOI] [PubMed] [Google Scholar]
- 47.Baig U, Belsare P, Watve M, Jog M. 2011. Can thrifty gene(s) or predictive fetal programming for thriftiness lead to obesity? Journal of Obesity 2011, 861049 (doi:10.1155/2011/861049) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Spencer HG, Hanson MA, Gluckman PD. 2006. Response to Wells: phenotypic responses to early environmental cues can be adaptive in adults. Trends Ecol. Evol. 21, 425–426 (doi:10.1016/j.tree.2006.05.006) [Google Scholar]
- 49.Gluckman PD, Hanson MA, Beedle AS, Spencer HG. 2008. Predictive adaptive responses in perspective. Trends Endocrinol. Metab. 19, 109–110 (doi:10.1016/j.tem.2008.02.002) [DOI] [PubMed] [Google Scholar]
- 50.Bateson P. 2008. Preparing offspring for future conditions is adaptive. Trends Endocrinol. Metab. 19, 111 (doi:10.1016/j.tem.2008.02.001) [DOI] [PubMed] [Google Scholar]
- 51.Walker R, et al. 2006. Growth rates and life histories in twenty-two small-scale societies. Am. J. Hum. Biol. 18, 295–311 (doi:10.1002/ajhb.20510) [DOI] [PubMed] [Google Scholar]
- 52.Quinlan RJ. 2007. Human parental effort and environmental risk. Proc. R. Soc. B 274, 121–125 (doi:10.1098/rspb.2006.3690) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Placek CD, Quinlan RJ. 2012. Adolescent fertility and risky environments: a population-level perspective across the lifespan. Proc. R. Soc. B 279, 4003–4008 (doi:10.1098/rspb.2012.1022) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Leimar O, Hammerstein P, Van Dooren TJM. 2006. A new perspective on developmental plasticity and the principles of adaptive morph determination. Am. Nat. 167, 367–376 (doi:10.1086/499566) [DOI] [PubMed] [Google Scholar]
- 55.Reed TE, Waples RS, Schindler DE, Hard JJ, Kinnison MT. 2010. Phenotypic plasticity and population viability: the importance of environmental predictability. Proc. R. Soc. B 277, 3391–3400 (doi:10.1098/rspb.2010.0771) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Dunlap AS, Stephens DW. 2009. Components of change in the evolution of learning and unlearned preference. Proc. R. Soc. B 276, 3201–3208 (doi:10.1098/rspb.2009.0602) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Stephens DW. 1991. Change, regularity, and value in the evolution of animal learning. Behav. Ecol. 2, 77–89 (doi:10.1093/beheco/2.1.77) [Google Scholar]
- 58.Padilla DK, Adolph SC. 1996. Plastic inducible morphologies are not always adaptive: the importance of time delays in a stochastic environment. Evol. Ecol. 10, 105–117 (doi:10.1007/bf01239351) [Google Scholar]
- 59.Dunlap AS, McLinn CM, MacCormick HA, Scott ME, Kerr B. 2009. Why some memories do not last a lifetime: dynamic long-term retrieval in changing environments. Behav. Ecol. 20, 1096–1105 (doi:10.1093/beheco/arp102) [Google Scholar]
- 60.Wells JCK. 2012. A critical appraisal of the predictive adaptive response hypothesis. Int. J. Epidemiol. 41, 229–235 (doi:10.1093/ije/dyr239) [DOI] [PubMed] [Google Scholar]
- 61.Potts R. 1998. Variability selection in hominid evolution. Evol. Anthropol. 7, 81–96 (doi:10.1002/(sici)1520-6505(1998)7:3<81::aid-evan3>3.0.co;2-a) [Google Scholar]
- 62.Kuzawa CW. 2005. Fetal origins of developmental plasticity: are fetal cues reliable predictors of future nutritional environments? Am. J. Hum. Biol. 17, 5–21 (doi:10.1002/ajhb.20091) [DOI] [PubMed] [Google Scholar]
- 63.Borgerhoff Mulder M, et al. 2009. The intergenerational transmission of wealth and the dynamics of inequality in pre-modern societies. Science 326, 682–688 (doi:10.1126/science.1178336) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Scheiner SM. 1993. Genetics and evolution of phenotypic plasticity. Annu. Rev. Ecol. Syst. 24, 35–68 (doi:10.1146/annurev.ecolsys.24.1.35) [Google Scholar]
- 65.Lachmann M, Jablonka E. 1996. The inheritance of phenotypes: an adaptation to fluctuating environments. J. Theor. Biol. 181, 1–9 (doi:10.1006/jtbi.1996.0109) [DOI] [PubMed] [Google Scholar]
- 66.Monaghan P. 2008. Early growth conditions, phenotypic development and environmental change. Phil. Trans. R. Soc. B 363, 1635–1645 (doi:10.1098/rstb.2007.0011) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Cichon M. 1997. Evolution of longevity through optimal resource allocation. Proc. R. Soc. Lond. B 264, 1383–1388 (doi:10.1098/rspb.1997.0192) [Google Scholar]
- 68.Heiming RS, Jansen F, Lewejohann L, Kaiser S, Schmitt A, Lesch KP, Sachser N. 2009. Living in a dangerous world: the shaping of behavioral profile by early environment and 5-HTT genotype. Front. Behav. Neurosci. 3, 26 (doi:10.3389/neuro.08.026.2009) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Del Giudice M, Ellis BJ, Shirtcliff EA. 2011. The adaptive calibration model of stress responsivity. Neurosci. Biobehavi. Rev. 35, 1562–1592 (doi:10.1016/j.neubiorev.2010.11.007) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.van den Heuvel J, Saastamoinen M, Brakefield PM, Kirkwood TBL, Zwaan BJ, Shanley DP. 2013. The predictive adaptive response: modeling the life-history evolution of the butterfly Bicyclus anynana in seasonal environments. Am. Nat. 181, E28–E42 (doi:10.5061/dryad.kg45v) [DOI] [PubMed] [Google Scholar]
- 71.Dantzer B, Newman AEM, Boonstra R, Palme R, Boutin S, Humphries MM, McAdam AG. 2013. Density triggers maternal hormones that increase adaptive offspring growth in a wild mammal. Science 340, 1215–1217 (doi:10.1126/science.1235765) [DOI] [PubMed] [Google Scholar]
- 72.Smallegange IM. 2011. Complex environmental effects on the expression of alternative reproductive phenotypes in the bulb mite. Evol. Ecol. 25, 857–873 (doi:10.1007/s10682-010-9446-6) [Google Scholar]
- 73.Aubin-Horth N, Dodson JJ. 2004. Influence of individual body size and variable thresholds on the incidence of a sneaker male reproductive tactic in Atlantic salmon. Evolution 58, 136–144 (doi:10.1554/03-256) [DOI] [PubMed] [Google Scholar]
- 74.Youngson NA, Whitelaw E. 2008. Transgenerational epigenetic effects. Annu. Rev. Genomics Hum. Genet. 9, 233–257 (doi:10.1146/annurev.genom.9.081307.164445) [DOI] [PubMed] [Google Scholar]