Abstract
Background
The pathologic identification of 12 or more lymph nodes (LN) after colectomy for colon cancer became a quality indicator for surgery in 2001. We investigated whether this new standard of care was differentially adopted across racial and socioeconomic lines.
Methods
We identified 111,339 stage I-III colon cancer patients identified as black or white in the Surveillance, Epidemiology, and End Results (SEER) database from 1988-2004 who underwent colectomy. We performed multivariable logistic regression to investigate the influence of race, area socioeconomic status (SES), and other clinical and demographic characteristics on the number of lymph nodes examined.
Results
Between 1988 and 2004, white patients were more likely than black patients to have ≥12 LNs identified, (OR=1.06, 95%CI 1.02-1.10) after adjustment for age, year of diagnosis, sex, marital status, tumor grade, stage, and subsite within the colon. After adjustment for SES, race was no longer significant (adjusted OR 1.00, 95%CI 0.96-1.04). There was, however, a significant positive trend between a patient's SES and having ≥12 LNs examined (p-trend<0.0001), with a 30% increased odds comparing the highest to the lowest quintiles of SES. We found that the association between SES and the dissection of ≥12 LNs was only present in individuals diagnosed after 1999.
Conclusions
The association between high SES and the examination of ≥12 LNs is only apparent from 2000 onward, and coincides with its dissemination and acceptance as a new standard of care. This suggests that the emergence of lymph node dissection as a quality indicator may have been more rapidly disseminated into higher SES groups.
Introduction
Despite advances in the prevention, screening, and treatment of colon cancer in the United States, there remain persistent disparities in colon cancer outcomes based on race and socioeconomic status (SES). Black men and women have higher colon cancer incidence and mortality rates,(1) lower rates of screening,(2) are more likely to be diagnosed at an advanced stage,(3) and have worse stage-for-stage survival when compared to whites.(4) Groups of lower socioeconomic status likewise exhibit lower screening rates(5) and higher mortality related to colorectal cancer than their higher socioeconomic counterparts, when matched stage for stage.(1, 6)
Observational studies in the late 1990's revealed that the proportion of patients with positive LNs increased when more LNs are examined, suggesting that an increased number of LNs examined yields more accurate staging.(7, 8) Subsequent population-based studies(9) as well as randomized clinical trials evaluating chemotherapy,(10) demonstrated a relationship between the number of LNs dissected and increased survival rates. The relationship between the quantity of LNs dissected and survival gained increasing acceptance in the oncologic and surgical communities, and beginning in 2001 a minimum recommended number of LNs (defined as 12) dissected during tumor resection was incorporated into surgical guidelines as a quality measure.(11, 12, 13) We aimed to determine the extent to which race or SES are associated with the extent of LN dissection, and to identify other factors that contribute to such differences.
Materials and Methods
Study Population
We identified 142,425 patients with histologically confirmed, invasive, AJCC stage I-III colon cancer who underwent colectomy between January 1, 1988, and December 31, 2004, in the Surveillance, Epidemiology, and End Results (SEER) program using SEER*Stat version 6.2.3. The SEER program has recorded incident cancers from nine registries since 1973, but has only included AJCC staging information since 1988. In 1992, the program expanded to include four additional sites. In 2000, it was expanded again to its current set of 17 registries. The SEER Public Use Data includes data on patient age, sex, race, year of diagnosis, and geographic location, and provide information regarding stage of disease; tumor grade, tumor extension, and histology; LN status; and overall survival. SEER also contains county level socioeconomic data, including poverty level, educational attainment, and median household income.
Patients were sequentially excluded if they had a diagnosis of familial adenomatous polyposis (n=93), a non-specified number of LNs dissected (n= 4,803), a non-specified number of positive LNs (n=77), or inconsistent stage/positive nodes information (n=95). Finally, we excluded all cases with T1tumors(n=15,311), as well as any patients who were not identified in SEER as being of either “White” or “Black” race (n=10,707), yielding a final sample size of n=111,339.
Study Variables
Clinical and Demographic Characteristics
Variables for tumor extent, number of nodes examined, number of positive nodes, the subsite of the tumor within the colon, tumor grade, and staging information were used. Tumor grade was categorized as ‘high’ if it was grade 3 or 4 (poorly differentiated, undifferentiated), ‘low’ if it was grade 1 or 2 (well differentiated, moderately differentiated), and ‘unknown’ if it was not recorded. The number of nodes examined was categorized as less than 12 vs. ≥12 in accordance with current guidelines.(13) Tumor subsite was grouped into four categories corresponding to the proximal colon (cecum, appendix, and right ascending colon), the transverse colon (hepatic flexure, transverse colon, splenic flexure), the distal colon (descending left colon, sigmoid), and unspecified (overlapping lesion, colon NOS).
Demographic information included year of diagnosis, age at diagnosis, race, sex, and marital status. Age at diagnosis was grouped into four categories: <60 years, 60-69 years, 70-79 years, and ≥80 years of age. Patients were categorized by marital status at the time of diagnosis as married, unmarried (single, divorced, widowed, or separated), or unknown.
SES
The SEER database contains a variety of socioeconomic data from the 1990 and 2000 census. Due to concerns over maintaining patient confidentiality, only the county level census data is publicly available. Although inferior to data collected at the ZIP code or census block group, county level census data are generally accepted as crude proxy measures of the elements of a patient's social environment—such as level of economic deprivation, social inequality, resource availability, opportunity structure, or living conditions.(14,15,16,17,18) Based on an exhaustive review of area-based socioeconomic measures by Krieger et al., we used a composite measure of area SES by incorporating three different variables from the county level 1990 census data: median household income, percentage of persons 25 years of age or older with at least a high school education, and percentage of people below the poverty level. For each of the three county level SES measures, patients were ranked into quintiles such that those the first quintile consisted of patients living in areas in the highest fifth for household income, educational attainment, and the lowest fifth with respect to percentage of people below the poverty level. Each patient was then assigned a composite SES score based on the average of these three rankings. Our decision to use a composite area-based socioeconomic measure was made in the interests retaining the most parsimonious statistical model, and in an effort to make our results comparable to other SEER based health disparities studies.(17)
To test the sensitivity of the SES findings, we generated three alternate measures of area SES for use in the logistic models. In the first, we used the adjusted median household income, educational attainment, and poverty level data fields from the 2000 census. The second method used the race-specific data from the 2000 Census data for educational attainment and poverty in addition to adjusted median household income. In the third method, we assigned each patient his/her average area SES rank based on the closest available census data, in which patients diagnosed prior to 1996 were linked to the 1990 census data and those diagnosed after used the 2000 census.
Statistical Analysis
We used the chi-square test to compare the distributions of demographic and clinical characteristics (age, marital status, stage, tumor extension, number of positive LNs, number of LNs examined, year of diagnosis, tumor grade, tumor subsite) of the patients by race and SES, separately. We conducted univariate analyses of tumor stage, nodes examined, and number of positive LNs to identify differences in these distributions by race and SES, respectively. We used the Kolmogorov-Smirnoff procedure to test for the normality of the distributions of the continuous variables and, for non-normally distributed data, and the nonparametric Wilcoxon test to assess statistically significant differences in the distribution of number of nodes examined between races and SES quintiles. Logistic regression models were used to measure the independent effects of race and/or SES on the odds of having ≥12 LNs examined. Each model was adjusted for selected covariates, including year of diagnosis, age at diagnosis, sex, marital status, T-stage, grade, and subsite. We then tested for multiplicative statistical interactions between race and year of diagnosis, and SES and year of diagnosis, respectively. Finally, we stratified the final logistic models by year of diagnosis to examine temporal trends. All statistical analyses were performed using SAS version 9.1.3.
Results
Table 1 presents the distributions of age at diagnosis, sex, and selected clinical and demographic characteristics, among 11,127 black and 100,212 white patients who received colectomy for T2-4 colon cancer. Black patients were on average 5 years younger at diagnosis, with a median age of 66 for blacks and 71 for whites (p<0.0001). Black patients were less frequently married at the time of diagnosis when compared to white (40.6% vs. 55.7%, p<0.0001), respectively. Black and white patients differed significantly by composite area SES score, with 23.6% of black patients ranked in the wealthiest two quintiles, compared to 42.2% of white patients (p<0.0001). Differences of this magnitude and significance were consistent across the individual elements of SES (income, education, poverty), as well as the three alternate area SES measures. Clinically, black patients had more advanced disease compared to whites with respect to both stage at diagnosis (p<0.0001), and T-stage (p=0.0024). Overall, more black patients than white had nodal involvement (p<0.0001); however, among those with one or more positive nodes, black patients had fewer positive nodes identified when compared to white patients, with 3.2 vs. 3.5 respectively (p<0.0001). The tumors of Black patients were 15.3% high grade compared to 21.1% among whites (p<0.0001). The overall proportions of black and white patients who had ≥12 nodes dissected were similar with 47.2% of black and 46.2% of white patients (p=0.04).
Table 1. Clinical and demographic characteristics of patients diagnosed with stage I-III colon cancer by race, SEER, 1988-2004 (n =111,339).
| Total | % | Black | % | White | % | p value | |
|---|---|---|---|---|---|---|---|
| Sample Size | 111,339 | 100.0 | 11,127 | 10.0 | 100,212 | 90.0 | |
| Mean Age (yr) | 70.4 | 66 | 71 | <0.0001 | |||
| Sex | <0.0001 | ||||||
| Male | 62,252 | 46.9 | 4,949 | 44.5 | 47,303 | 47.2 | |
| Female | 59,087 | 53.1 | 6,178 | 55.5 | 52,909 | 52.8 | |
| AJCC Stage | <0.0001 | ||||||
| I | 15,451 | 13.9 | 1,372 | 12.3 | 14,079 | 14.0 | |
| IIa | 45,300 | 40.7 | 4,235 | 38.1 | 41,065 | 41.0 | |
| IIb | 8,960 | 8.1 | 892 | 8.0 | 8,068 | 8.1 | |
| IIIa | 2,719 | 2.4 | 291 | 2.6 | 2,428 | 2.4 | |
| IIIb | 25,266 | 22.7 | 2,912 | 26.2 | 22,354 | 22.3 | |
| IIIc | 13,643 | 12.2 | 1,425 | 12.8 | 12,218 | 12.2 | |
| T Stage | 0.0024 | ||||||
| 2 | 18,680 | 16.8 | 1,737 | 15.6 | 16,943 | 16.9 | |
| 3 | 75,214 | 67.6 | 7,619 | 68.5 | 67,595 | 67.5 | |
| 4 | 17,445 | 15.7 | 1,771 | 15.9 | 15,674 | 15.6 | |
| N Stage | <0.0001 | ||||||
| 0 | 69,711 | 62.6 | 6,499 | 58.4 | 63,212 | 67.8 | |
| 1 (1-3) | 27,985 | 25.1 | 3,203 | 28.8 | 24,782 | 24.7 | |
| 2 (4+) | 13,643 | 12.3 | 1,425 | 12.8 | 12,218 | 12.2 | |
| Tumor Grade | <0.0001 | ||||||
| Low | 84,443 | 75.8 | 8,990 | 80.8 | 75,453 | 75.3 | |
| High | 22,902 | 20.6 | 1,704 | 15.3 | 21,198 | 21.1 | |
| Unknown | 3,994 | 3.6 | 433 | 3.9 | 3,561 | 3.6 | |
| Colon Site | 0.0009 | ||||||
| Proximal | 29,917 | 26.9 | 3,012 | 27.1 | 26,905 | 26.9 | |
| Transverse | 42,620 | 38.3 | 4,320 | 38.8 | 38,300 | 38.2 | |
| Distal | 37,179 | 33.4 | 3,593 | 32.3 | 33,586 | 33.5 | |
| Unspecified | 1,623 | 1.5 | 202 | 1.8 | 1,421 | 1.4 | |
| Marital Status ‡ | <0.0001 | ||||||
| Married | 60,319 | 54.2 | 4,522 | 40.6 | 55,797 | 55.7 | |
| Unmarried | 51,020 | 45.8 | 6,605 | 59.4 | 44,415 | 44.3 | |
| Nodes Examined | 0.11 | ||||||
| 0 | 1,919 | 1.7 | 192 | 1.7 | 1,727 | 1.7 | |
| 1-11 | 57,905 | 52.0 | 5,682 | 51.1 | 52,223 | 52.1 | |
| 12+ | 51,515 | 46.3 | 5,253 | 47.2 | 46,262 | 46.2 | |
| Summary SES | <0.0001 | ||||||
| 1st | 17,472 | 15.7 | 1086 | 9.8 | 16,386 | 16.4 | |
| 2nd | 27,389 | 24.6 | 1535 | 13.8 | 25,854 | 25.8 | |
| 3rd | 18,709 | 16.8 | 1316 | 11.8 | 17,393 | 17.4 | |
| 4th | 30,930 | 27.8 | 3653 | 32.8 | 27,277 | 27.2 | |
| 5th | 16,839 | 15.1 | 3537 | 31.8 | 13,302 | 13.3 | |
|
| |||||||
| Mean/Median | SD | Mean/Median | SD | Mean/Median | SD | ||
|
| |||||||
| Nodes Examined | 12.7/11.0 | 9.1 | 12.8/11.0 | 8.9 | 12.7/11.0 | 9.1 | |
| >12 | 51,515 | 46.3 | 5,253 | 47.2 | 46,262 | 46.2 | 0.04 |
| Positive Nodes | |||||||
| Node +/− | 1.29/0.0 | 2.7 | 1.34/0.0 | 2.6 | 1.28/0.0 | 2.7 | 0.02 |
| Node +only | 3.45/2.0 | 3.5 | 3.24/2.0 | 3.2 | 3.47/2.0 | 3.5 | <0.0001 |
Unmarried = single, widowed, divorced, separated at time of diagnosis.
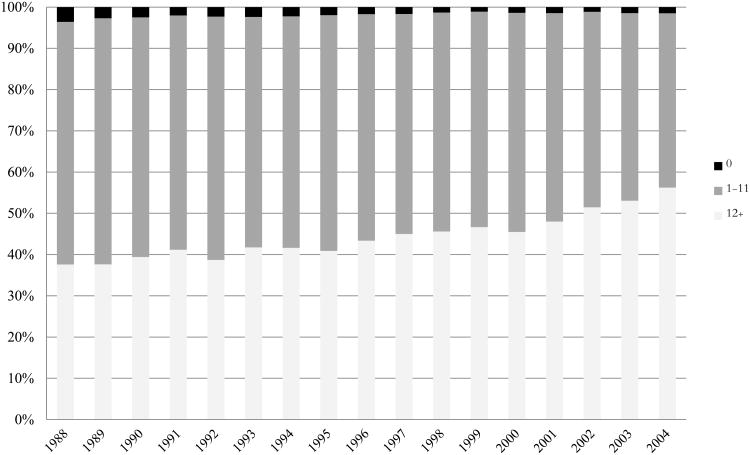
Figure 1 illustrates the temporal increase in the number of LNs examined using a histogram of the overall percentages of colon cancer cases within different categories of nodes examined. Between 1988 and 2004, the proportion of individuals having ≥12 LNs examined after receiving a colectomy for a T2 or greater colon cancer rose from 37.6% to 56.2%.
Figure 1. Distribution of number of LNs examined for stage I-III colon cancer from 1988-2004.
We used multivariate logistic regression models to identify factors associated with having ≥12 LNs examined. Among the findings that were consistent with previous study results,(19) we found that later year of diagnosis (OR=1.05, 95%CI 1.04-1.06), younger age at diagnosis (1.18, 95%CI 1.17-1.19) per 10 years, and high grade vs. low grade (1.17, 95%CI 1.13-1.20) were each independently associated with significantly greater odds of having ≥12 LN s examined. In addition, we found that patients with T3 (1.51, 95%CI 1.46- 1.56) or T4 (1.35, 95%CI 1.29-1.40) tumors, were at an increased odds compared to those with T2 tumors, and that tumors of the transverse (0.90, 95%CI 0.87-0.93) or distal colon (0.48, 95%CI 0.46-0.49) had lower odds when compared with tumors of the proximal colon.
White patients had a small but significantly increased odds of having ≥12 LNs examined compared to black patients (1.06, 95%CI 1.02-1.10), however, after adjustment for SES (in addition to the aforementioned clinical and demographic factors), the effect of race effectively nullified (1.00, 95%CI 0.96-1.04). Moreover, the addition of SES to the model as a set of ordinal categorical variables, revealed a positive trend in the odds for having ≥12 LNs examined with each increase in SES category when compared to the lowest (p for trend <0.001).
We also found a statistically significant positive interaction between SES and year of diagnosis (p<0.001), suggesting that the association between a patient's SES and their odds of having ≥12 LNs examined has increased each year significantly more than would be expected from multiplying their individual effects (p<0.0001), based on a generalized Wald test to simultaneously test all four interaction terms. A positive interaction was also noted between race and year of diagnosis (p<0.001), which was similarly rendered statistically insignificant after adjustment for SES.
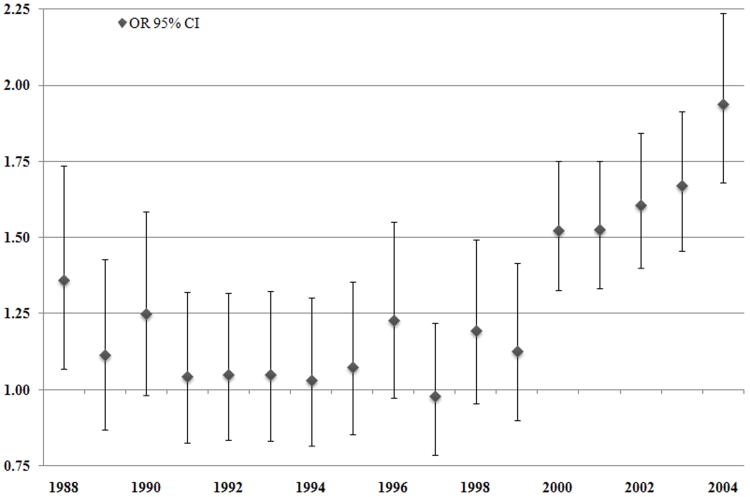
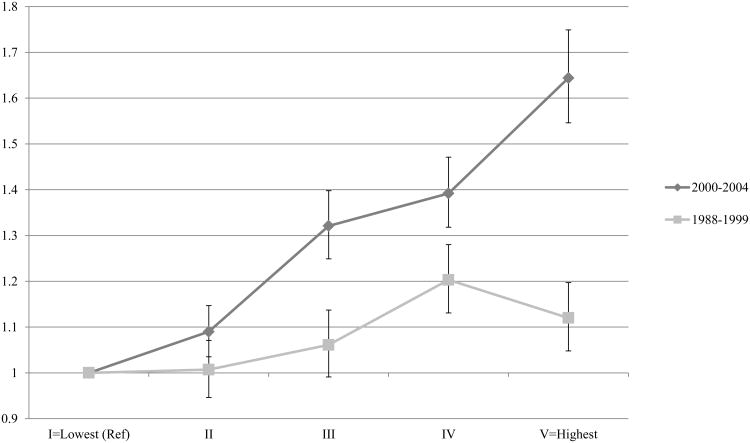
To further explore these interactions, we performed stratified analyses of the models of SES, adjusted for race, as well as clinical and demographic factors. Figure 2 shows the odds ratios of having ≥12 LNs examined comparing the lowest to highest area SES groups, by year of diagnosis. This figure illustrates that prior to 2000, a patient's odds of having ≥12 LNs examined were equivalent, whether they were in the highest or lowest area SES quintile, while from 2000 to 2004 these OR's begin to increase significantly. Figure 3 examines this relationship comparing the low SES reference category to increasing quintiles of SES, grouping year of diagnosis into categories corresponding with the early published findings regarding the clinical significance of the extent of LN dissection. From the year 2000 onward, an individual's odds of having ≥12 LNs examined increase as a linear function of their area SES quintile (p for trend <0.001).
Figure 2.
Odds ratios for examination of ≥ 12 lymph nodes between the highest vs. lowest quintile of area SES, by year of diagonis, after adjustment for age, sex, marital status, tumor grade, T-stage, and subsite within the colon.
Figure 3.
Odds of ≥ 12 lymph nodes examined across quintiles of area SES comparing patients diagonised in 1988-1999 vs. 2000-2004, after adjustment for age, year of diagnosis, sex, marital status, tumor grade, T-stage, and subsite within the colon.
A similar interaction was noted between patient race and year of diagnosis (p<0.001), when unadjusted for SES. Prior to 2000, black and white patients did not differ significantly with regard to the odds of having ≥12 LNs examined. Whereas, each year thereafter, white patient's had a modest but significantly greater odds of having ≥12 LNs examined when compared to black patients. This relationship was, similarly, rendered non-significant after adjustment for SES.
Discussion
We found in this database of 111,339 patients with stage I-III colorectal cancer, that white patients were more likely than black patients to undergo an adequate LN dissection, after adjustment for age, year of diagnosis, sex, tumor grade, T-stage, and subsite within the colon. However, after adjustment for SES, this disparity was nullified. We also found that SES disparities in rates of adequate LN dissection emerged only in the most recent years of the analysis, temporally coinciding with the emerging consensus that more LN dissection is associated with improved outcomes. Our finding that SES was the primary driver of treatment disparity rather than race is consistent with prior analyses of colorectal cancer outcomes which found that observed racial disparities were greatly diminished after adjusting for SES.(17,24)
The extent of LN dissection is an important predictor of survival in colon cancer.(9,10,20) There are multiple explanations for this phenomenon. The number of LNs resected may be a surrogate for the extent of resection, with a higher number of LNs reflecting a more complete resection of tumor and its lymphatic supply. Alternatively, the number of resected LNs may reflect a more complete examination by the pathologist, and may lead to a more accurate overall staging of the tumor. Indeed, a low ratio of metastatic to examined LNs is associated with an improved prognosis, but only if a sufficient quantity of LNs is examined; a low so-called LN ratio is not prognostic in patients with fewer than ten LNs removed.(21)
The consideration of LN dissection as a quality standard has generated controversy, as there is no consensus on a mechanistic explanation for this proposed protective effect. Moreover, since the studies associating adequate LN dissection with improved outcomes are all observational, the possibility remains that LN dissection may be a correlate, but not a cause of increased survival.(22) For example, colectomy specimens with more LNs present may reflect a greater immunologic response to the tumor rather than the extent of dissection, or thoroughness of pathological examination.
Despite the uncertainty raised by some authors regarding the mechanism and true causation, using the number of LNs dissected as a metric has been gradually accepted in clinical practice. In the late 1990's, data emerged that indicated that increasing numbers of LNs examined was associated with an increased probability of identifying metastatic disease.(7,8) Adequate LN dissection was then formally accepted as a quality measure in surgical guidelines beginning in 2001.(11,12,13) Despite its emerging acceptance, this standard is often not achieved in clinical practice. A SEER-based study revealed that in 2001, only 44 percent of patients undergoing surgical resection for colon cancer had ≥12 LNs removed.(19) Our results closely matched these previously reported findings for 2000, and show that by 2004 the rate of adequate LN dissection had reached only 56%. Given the striking gap between the standard of care and everyday clinical practice, we hypothesized that the racial and socioeconomic disparities observed in colon cancer outcomes may be due to differences in the degree of LN dissection, and sought to test whether such treatment differences exist.
We were surprised to discover that disparities in LN dissection are only present in more recent years. Prior to the year 2000, there were no observed differences in the odds of receiving an adequate LN dissection between black and white patients, or between those of the highest and lowest categories of county level socioeconomic status, after adjusting for clinical and demographic characteristics. However, beginning in 2000, we found a significant increase in the difference of LN dissection rates between these groups. Intriguingly, the emergence of this disparity coincides temporally with the dissemination of observational evidence of a beneficial association of adequate LN dissection with survival.
As these racial and socioeconomic differences of adequate LN dissection have only developed in recent years, they are not an adequate explanation of longstanding disparities in outcomes. Rather, this emerging racial and socioeconomic disparity in LN dissection rate more likely reflects a well-described phenomenon that occurs whenever a medical advance (or, in this case, a proposed quality measure) develops; those who initially benefit from the advance tend to be those with the greatest social resources, such as education, and wealth.(23) Thus, the dissemination of this new quality of care standard occurs within a preexisting social structure, being differentially adopted across socioeconomic strata, which is then reflected in the emergence of the described disparities. Of note, the emergence of this disparity does not prove a beneficial effect of adequate LN dissection. Rather, the disparity reflects disparate adoption of a medical innovation based on the resources described above.
Our study has several limitations. As this analysis utilizes a population database, we were unable to include variables that may have an effect on the odds of adequate LN dissection, such as the type of hospital, the experience of the surgeon, and the resources and experience of the examining pathologist. Another limitation is that we lacked information on comorbidities and performance status which may be associated with SES, and influence the extent of the LN dissection. An alternate explanation of our findings that we were not able to test using this dataset involves the increase in the rate of obesity in the last 10 years. Obesity disproportionately affects lower SES individuals, and may reduce the facility with which the pathologist is able to identify lymph nodes within a surgical specimen, thereby creating the appearance that lower SES predicts fewer nodes examined.
The primary limitation of our study is the fact that our ascertainment of socioeconomic status was limited to county level census data which has some distinct and well described limitations.(25,26,27,28) Krieger et al have shown that effects of socio-demographic factors on mortality vary when different size geographic units are used. For instance, when evaluating area based socioeconomic effects, smaller units of measurement, (such as the census tract, or block group) are more sensitive to gradients than larger area metrics (zip code, or county) and offer the strongest and most consistent results.(29) The use of county level SES measurements are at a distinctly increased likelihood of type II error, which would bias any SES effects toward the null. To examine the possibility that our findings were due to chance, we reproduced our findings using three alternate measures of SES. Yet, despite these limitations, our findings appear robust, and were replicated using a variety of single as well as composite area SES measures from both 1990 and 2000. We also tested the sensitivity of our results to changes in the SEER source population, due to the expansions of the number of registries in 1992 and 2000. Despite large differences in the patient sample, our findings were similar with respect to magnitude, statistical significance, temporal and dose response trends.
We conclude that in this analysis of a large cancer database, SES disparities in adequate LN dissection rates began to appear in the year 2000, which may reflect the preferential adoption of an emerging standard of care among those patients with greater social resources. Future efforts are needed to elucidate the mechanistic relationship between LN dissection and prognosis. Future efforts should also focus on improving adherence to quality guidelines across all socioeconomic strata.
Acknowledgments
Grant support: Supported by an R25 Award from the National Cancer Institute (NCI) (CA94061 to R.B.M.), and a T32 Award (ULI RR024156 to R.B.M.) from a grant from the National Center for Research Resources (NCRR) of the National Institutes of Health; a T32 Award from the National Cancer Institute (CA095929 to B.L.); a grant from the Department of Defense (BC043120 to A.I.N.).
References
- 1.Jemal A, Siegel R, Ward E, et al. Cancer statistics, 2008. CA Cancer J Clin. 2008;58:71–96. doi: 10.3322/CA.2007.0010. [DOI] [PubMed] [Google Scholar]
- 2.Ananthakrishnan AN, Schellhase KG, Sparapani RA, Laud PW, Neuner JM. Disparities in colon cancer screening in the Medicare population. Arch Intern Med. 2007;167:258–64. doi: 10.1001/archinte.167.3.258. [DOI] [PubMed] [Google Scholar]
- 3.Chien C, Morimoto LM, Tom J, Li CI. Differences in colorectal carcinoma stage and survival by race and ethnicity. Cancer. 2005;104:629–39. doi: 10.1002/cncr.21204. [DOI] [PubMed] [Google Scholar]
- 4.Dignam JJ, Colangelo L, Tian W, et al. Outcomes among African-Americans and Caucasians in colon cancer adjuvant therapy trials: findings from the National Surgical Adjuvant Breast and Bowel Project. J Natl Cancer Inst. 1999;91:1933–40. doi: 10.1093/jnci/91.22.1933. [DOI] [PubMed] [Google Scholar]
- 5.Beydoun HA, Beydoun MA. Predictors of colorectal cancer screening behaviors among average-risk older adults in the United States. Cancer Causes Control. 2008;19:339–59. doi: 10.1007/s10552-007-9100-y. [DOI] [PubMed] [Google Scholar]
- 6.Albano JD, Ward E, Jemal A, et al. Cancer mortality in the United States by education level and race. J Natl Cancer Inst. 2007;99:1384–94. doi: 10.1093/jnci/djm127. [DOI] [PubMed] [Google Scholar]
- 7.Wong JH, Severino R, Honnebier MB, Tom P, Namiki TS. Number of nodes examined and staging accuracy in colorectal carcinoma. J Clin Oncol. 1999;17:2896–900. doi: 10.1200/JCO.1999.17.9.2896. [DOI] [PubMed] [Google Scholar]
- 8.Goldstein NS, Sanford W, Coffey M, Layfield LJ. Lymph node recovery from colorectal resection specimens removed for adenocarcinoma. Trends over time and a recommendation for a minimum number of lymph nodes to be recovered. Am J Clin Pathol. 1996;106:209–16. doi: 10.1093/ajcp/106.2.209. [DOI] [PubMed] [Google Scholar]
- 9.Chen SL, Bilchik AJ. More extensive nodal dissection improves survival for stages I to III of colon cancer: a population-based study. Ann Surg. 2006;244:602–10. doi: 10.1097/01.sla.0000237655.11717.50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Le Voyer TE, Sigurdson ER, Hanlon AL, et al. Colon cancer survival is associated with increasing number of lymph nodes analyzed: a secondary survey of intergroup trial INT-0089. J Clin Oncol. 2003;21:2912–9. doi: 10.1200/JCO.2003.05.062. [DOI] [PubMed] [Google Scholar]
- 11.Nelson H, Petrelli N, Carlin A, et al. Guidelines 2000 for colon and rectal cancer surgery. J Natl Cancer Inst. 2001;93:583–96. doi: 10.1093/jnci/93.8.583. [DOI] [PubMed] [Google Scholar]
- 12.Otchy D, Hyman NH, Simmang C, et al. Practice parameters for colon cancer. Dis Colon Rectum. 2004;47:1269–84. doi: 10.1007/s10350-004-0598-8. [DOI] [PubMed] [Google Scholar]
- 13.McGory ML, Shekelle PG, Ko CY. Development of quality indicators for patients undergoing colorectal cancer surgery. J Natl Cancer Inst. 2006;98:1623–33. doi: 10.1093/jnci/djj438. [DOI] [PubMed] [Google Scholar]
- 14.Singh GK, Miller BA, Hankey BF, Edwards BK. Area Socioeconomic Variations in US Cancer Incidence, Mortality, Stage, Treatment, and Survival 1975-1999. Bethesda, MD: National Cancer Institute; 2003. [Google Scholar]
- 15.Krieger N, Williams DR, Moss NE. Measuring social class in US public health research: concepts, methodologies, and guidelines. Annu Rev Public Health. 1997;18:341–78. doi: 10.1146/annurev.publhealth.18.1.341. [DOI] [PubMed] [Google Scholar]
- 16.Yost K, Perkins C, Cohen R, Morris C, Wright W. Socioeconomic status and breast cancer incidence in California for different race/ethnic groups. Cancer Causes Control. 2001;12:703–11. doi: 10.1023/a:1011240019516. [DOI] [PubMed] [Google Scholar]
- 17.Du XL, Fang S, Vernon SW, et al. Racial disparities and socioeconomic status in association with survival in a large population-based cohort of elderly patients with colon cancer. Cancer. 2007;110:660–9. doi: 10.1002/cncr.22826. [DOI] [PubMed] [Google Scholar]
- 18.Berkman LF, Macintyre S. The measurement of social class in health studies: old measures and new formulations. IARC Sci Publ. 1997:51–64. [PubMed] [Google Scholar]
- 19.Baxter NN, Virnig DJ, Rothenberger DA, Morris AM, Jessurun J, Virnig BA. Lymph node evaluation in colorectal cancer patients: a population-based study. J Natl Cancer Inst. 2005;97:219–25. doi: 10.1093/jnci/dji020. [DOI] [PubMed] [Google Scholar]
- 20.Chang GJ, Rodriguez-Bigas MA, Skibber JM, Moyer VA. Lymph node evaluation and survival after curative resection of colon cancer: systematic review. J Natl Cancer Inst. 2007;99:433–41. doi: 10.1093/jnci/djk092. [DOI] [PubMed] [Google Scholar]
- 21.Berger AC, Sigurdson ER, LeVoyer T, et al. Colon cancer survival is associated with decreasing ratio of metastatic to examined lymph nodes. J Clin Oncol. 2005;23:8706–12. doi: 10.1200/JCO.2005.02.8852. [DOI] [PubMed] [Google Scholar]
- 22.Markman M. Assigning a cause for a particular outcome in oncology: a serious note of caution. Cancer. 2008;113:668–70. doi: 10.1002/cncr.23631. [DOI] [PubMed] [Google Scholar]
- 23.Link BG, Phelan JC. Understanding sociodemographic differences in health--the role of fundamental social causes. Am J Public Health. 1996;86:471–3. doi: 10.2105/ajph.86.4.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Du XL, Meyer TE, Franzini L. Meta-analysis of racial disparities in survival in association with socioeconomic status among men and women with colon cancer. Cancer. 2007;109:2161–70. doi: 10.1002/cncr.22664. [DOI] [PubMed] [Google Scholar]
- 25.Rehkopf DH, Haughton LT, Chen JT, Waterman PD, Subramanian SV, Krieger N. Monitoring socioeconomic disparities in death: comparing individual-level education and area-based socioeconomic measures. Am J Public Health. 2006;96:2135–8. doi: 10.2105/AJPH.2005.075408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen JT, Coull BA, Waterman PD, Schwartz J, Krieger N. Methodologic implications of social inequalities for analyzing health disparities in large spatiotemporal data sets: an example using breast cancer incidence data (Northern and Southern California, 1988--2002) Stat Med. 2008;27:3957–83. doi: 10.1002/sim.3263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Smigal C, Jemal A, Ward E, et al. Trends in breast cancer by race and ethnicity: update 2006. CA Cancer J Clin. 2006;56:168–83. doi: 10.3322/canjclin.56.3.168. [DOI] [PubMed] [Google Scholar]
- 28.Ward E, Jemal A, Cokkinides V, et al. Cancer disparities by race/ethnicity and socioeconomic status. CA Cancer J Clin. 2004;54:78–93. doi: 10.3322/canjclin.54.2.78. [DOI] [PubMed] [Google Scholar]
- 29.Krieger N, Chen JT, Waterman PD, Rehkopf DH, Subramanian SV. Race/ethnicity, gender, and monitoring socioeconomic gradients in health: a comparison of area-based socioeconomic measures--the public health disparities geocoding project. Am J Public Health. 2003;93:1655–71. doi: 10.2105/ajph.93.10.1655. [DOI] [PMC free article] [PubMed] [Google Scholar]