Abstract
Somatic cells have been reprogrammed into induced pluripotent stem (iPS) cells that recapitulate the pluripotent nature of embryonic stem (ES) cells. Reduced pluripotency and variable differentiation capacities have hampered progress with this technology for applications in regeneration medicine. We have previously shown that Germ Cell Nuclear Factor (Gcnf) is required for the repression of pluripotency genes during ES cell differentiation and embryonic development. Here we report that iPS cell lines, in which the Gcnf gene was properly re-programmed, allowing expression of Gcnf, repress pluripotency genes during subsequent differentiation. In contrast, iPS clones in which the Gcnf gene was not re-programmed maintained pluripotency gene expression during differentiation and did not differentiate properly either in vivo or in vitro. These mal-reprogrammed cells re-capitulated the phenotype of Gcnf knock out (Gcnf−/−) ES cells. Re-introduction of Gcnf into either the Gcnf negative iPS cells or the Gcnf−/− ES cells, rescued repression of Oct4 during differentiation. Our findings establish a key role for Gcnf as a regulator of iPS cell pluripotency gene expression. It also demonstrates that reactivation of the Gcnf gene may serve as a marker to distinguish completely re-programmed iPS cells from incompletely pluripotent cells, which would make therapeutic use of iPS cells safer and more practical as it would reduce the oncogenic potential of iPS cells.
Keywords: Gcnf, iPS cells, somatic cell reprogramming, epigenetics, stem cells
Introduction
Overexpression of a defined set of transcription factors suffices to reprogram somatic cells into induced pluripotent stem (iPS) cells, which have been derived from various tissues, including embryonic fibroblasts and adult tail-tip fibroblasts, hepatocytes, gastric epithelial cells, pancreatic cells, neural cell and B lymphocytes in the mouse [1–5], and skin fibroblasts, keratinocytes and peripheral blood cells in the human [6–8]. iPS cells have enormous therapeutic potential because they can be derived from not only from normal but also patient-specific cells [9,10]. There are significant similarities between ES cells and iPS cells, including indistinguishable global histone modification and gene expression patterns [11,12]. Despite these similarities, there is emerging evidence of differences between ES cells and iPS cells and even between different iPS colonies. For example, differences in mRNA and microRNA (miRNA) expression [13–15], as well as in DNA methylation patterns [16–18], have been reported between ES cells and iPS cells. These observations indicate that transcription factor mediated reprogramming can result in abnormalities in subsequent iPS cells, which could impede their therapeutic utility [16].
Pluripotency genes, which are required to maintain ES and iPS cell self-renewal, function in part through inhibition of somatic gene expression [19]. Thus, pluripotency genes are repressed and/or silenced in a timely manner during differentiation to ensure the expression of lineage determinants, which in turn allows differentiation into the three germ layers and their derivatives [19,20]. It has been demonstrated that pluripotent gene expression persists in some differentiated iPS cell clones, while in others they are silenced as normal [1]. Germ Cell Nuclear Factor (Gcnf; NR6a1), a transcriptional repressor and an orphan member of the nuclear receptor gene family, is required for the repression and silencing of pluripotency genes such as Oct4 and Nanog in vitro and in vivo [21–23]. Loss of Gcnf function in Gcnf knock out (Gcnf−/−) mice results in embryonic lethality by E10.5 [24]. Importantly, there is loss of normal repression of the Oct4 gene in somatic cells after gastrulation, where it is generally silenced. Likewise Gcnf−/− ES cells maintain Oct4 expression during differentiation upon retinoic acid (RA) treatment, which impedes full differentiation of these cells [24]. Whether Gcnf plays a similar pivotal role in iPS cell differentiation is unknown.
Here we report that the Gcnf gene, which is silenced in fibroblasts, can be re-activated during somatic cell reprogramming using Oct4, Sox2, c-Myc and Klf4, however the Gcnf gene is not reprogrammed in all iPS colonies. Oct4 expression was repressed in those clones in which Gcnf expression was reactivated during subsequent differentiation with RA. In contrast, in the clones within which Gcnf was still silenced Oct4 expression was maintained during differentiation. Maintained Oct4 expression after differentiation affects the pluripotency of iPS cells, leading to poor differentiation in vitro, aberrant expression of differentiation genes, as well as failure to produce chimeras. Re-introduction of Gcnf rescues Oct4 repression during differentiation. Thus, re-programming of the Gcnf gene is an important variable affecting quality that should be considered during iPS formation.
Materials and Methods
Generation of iPS cells
pMXs-base retroviral vectors-Oct4, Sox2, Klf4 and C-myc (Addgene, catalog #s 13366, 13367, 13370 and 13375) [1] were introduced into each 10 cm plate with Plat-E cells respectively using Fugene 6 transfection reagent (Roche, catalogue no.11814443001) according to the manufacturer’s recommendations. One day before infection, MEFs were seeded at 8 ×105 cells per 10 cm gelatin coated plate. After 24 hours of transfection, virus-containing supernatants derived from these Plat-E cultures were filtered through a 0.45 mm cellulose acetate filter and supplemented with 4 mg/ml polybrene. Target cells were incubated in the virus/polybrene-containing supernatants overnight. MEFs were infected twice. After 24 hr of infection, the media were replaced with 10 ml fresh ES cell medium. No selection markers were used in this experiment, thus iPS cells were generated in the absence of antibiotics [25]. Three weeks later, alkaline phosphatase staining (Vector Laboratories, Catalogue no SK-5100) was carried out to evaluate reprogramming efficiency, and colonies were picked based on a morphology of characteristic ES colonies and GFP positive. To establish stable iPS cell lines, single iPS cell colonies were each picked into one well of a 24-well plate. iPS colonies in which the viral ectopic genes (Oct4, Sox2, c-Myc and Klf4) were silenced were used in the experiments described here.
Teratoma formation and histological analysis
The iPS clones that expressed Gcnf at day 1.5 of differentiation were designated Gcnfon, while the iPS clones that did not express Gcnf were designated Gcnfoff. 1×106 wt ES cells, Gcnf−/− ES cells, Gcnfon iPS cells and Gcnfoff iPS cells were injected into the rear leg muscle of 4 to 6 week-old severe combined immuno-deficient (SCID) mice. Four weeks later, tumors were surgically dissected from the mice. Samples were fixed in 10% formalin and embedded in paraffin. Sections were stained with hematoxylin and eosin.
Chimera generation and germ line transmission
To determine the ability of Gcnfoff iPS cells to contribute to adult chimeras, Gcnfon iPS cells or Gcnfoff iPS cells were injected into the blastocysts of C57 Albino white genetic background at passage 10. Chimeric blastocysts were subsequently transferred to day 2.5 pseudopregnant recipient CD-1 females, and the percentage of chimera contribution was estimated by scoring the level of coat color pigmentation. To evaluate the capacity for germ line transmission of iPS cells, chimeric males were bred with C57BL/6 females. Pups were identified by coat pigmentation and genotype after birth.
iPS cell differentiation assays
Differentiation of iPS cells and ES cells was induced by LIF withdrawal and addition of presence of 1 µM RA. The differentiating ES cells or iPS cells were collected over the course of several days, and the expression of pluripotent genes and germ layer markers were analyzed by regular reverse transcription PCR (RT-PCR) or quantitative RT (Q-PCR) with SYBR (Qiagen, catalogue no. 204056). Primers used in this paper are described in table S1. Protein samples were analyzed by western blot. For EB formation the iPS cells and ES cells were dispersed into single cells using trypsin/EDTA and were plated into bacterial grade Petri dishes. Both ES cell and iPS cells aggregated into embryoid bodies (EBs) for four days and iPS cell EBs were plated on to gelatin-coated dishes for differentiation for another four days. The differentiated cells were identified by the cellular morphology.
Polymerase chain reaction (PCR) analysis
Total RNA was prepared using the Trizol reagent (Invitrogen, catalog no.15596-018) and quantified by NanoDropTM 1000 Spectrophotometer v3.7 (Thermo Fisher Scientific). RNA samples were reverse transcribed to cDNA using SuperScript III First-Strand Synthesis System (Invitrogen, Catalogue no. 18080-051). PCR primers are described in supplemental information Table S1.
Western analysis
Whole cell lysates were subjected to western analyses. Antibodies against mouse Nanog (Abcam, ab80892), Sox2 (Millipore, ab5603), Oct4 (Santa Cruz, catalogue no. sc-5279) and Gcnf were used at 1:1000 dilution. β-Actin (Sigma, catalogue no. A1978) was used at a 1:5000 dilution. The secondary antibodies, goat anti-mouse IgG-HRP (Santa Cruz, catalogue no. sc-2055) and goat anti-rabbit IgG-HRP (Santa Cruz, catalogue no. sc-2004) were used at a 1:2000 dilution. HRP activity was detected by a chemiluminescence using Pierce ECL Western Blotting Substrate kit (Thermo scientific, catalog no. 32106). The emitted light was detected by photographic film.
DNA CpG Methylation analysis
Bisulfite treatment was performed [18] using the EZ DNA Methylation Kit (Zymo research, Catalogue no. D5001) according to the manufacturers’ instructions. CpG methylation was analyzed within the Oct4 promoter in Gcnfon (m5 iPSCs), Gcnfoff (m8 iPSCs) and MEF cells. PCR primers are: forward: CTGAAAATCACCACCACC; reverse: CTAATAATGAGCCTTTCC. Amplified PCR products were cloned into pCR2.1-TOPO vector (Invitrogen, catalogue no. K2040-01). Ten randomly selected clones were sequenced with the M13 forward and M13 reverse primers.
Gcnf Rescue
A full-length mouse Gcnf complementary DNA (cDNA) was obtained from reverse transcribed PCR products generated from d1.5 differentiated ES cell mRNA. The resulting cDNA was cloned into the Gateway/Topo TA vector (Invitrogen, catalogue no. K250020), and then was recombined into the destination retroviral plasmid with the mscv promoter by Gateway LR Clonase (Invitrogen, catalogue no. 11791-020). For packaging the virus, 1.5×106 BOSC cells were plated in 6 well of plates (10%FBS DMEM), and were cultured overnight. The mscv-Gcnf and mscv-GFP plasmids along with the packaging plasmid pDuo were introduced into each 6 cm plate with BOSC cells respectively using Fugene 6 transfection reagent (Roche, catalogue no.11814443001) according to the manufacturer’s recommendations. After 24 hours of transduction, virus-containing supernatants derived from these BOSC cell cultures were filtered through a 0.45 mm cellulose acetate filter and supplemented with 4mg/ml polybrene. 3 ×105 target cells were seeded into 24 well plates that were incubated with 0.5 ml of the virus/polybrene-containing supernatants overnight and infected twice. After 24 hr of infection, the media was replaced with 0.5 ml fresh ES cell medium. The transfected cells were selected for two weeks with 1 µg/ml of puromycin after three days of transfection. The selected cells were used for the described analysis.
Results
The degree of Gcnf reprogramming determines the level of Oct4 repression during iPS cell differentiation
Mouse iPS cells were generated by virally introducing four factors: Oct4, Sox2, c-Myc and Klf4 [1] into Oct4-GFP mouse embryonic fibroblasts (MEF) (supplemental information Fig. S1A). The selected iPS colonies were positive for Oct4-GFP fluorescence (supplemental information Fig. S1B) and alkaline phosphatase (ALP) staining (supplemental information Fig. S1C, S1D). We induced iPS cell differentiation by treatment with RA for 6 days. Wild type (wt) and Gcnf−/− ES cells were used as controls. The highest levels of Gcnf expression are induced at 1.5 days of RA differentiation in wt ES cells [21]. Thus, Gcnf expression was analyzed at 1.5 days of RA treatment by reverse transcription-PCR (RT-PCR) and quantitative RT-PCR (Q-RT-PCR) in nine different iPS clones that had been selected based on optimal ES cell morphology (25), GFP positive and silencing of the viral genes (Oct4, Sox2, c-Myc and Klf4) (supplemental information Fig. S1E, S1F, S1G, S1H). Unexpectedly, we observed Gcnf expression in only four of the iPS clones (44.4%) (Fig. 1A). The remaining five iPS clones (55.6%) did not express Gcnf, suggesting that the Gcnf gene had not been properly reprogrammed during the process of iPS cell formation. The iPS clones that expressed Gcnf at day 1.5 of differentiation were designated Gcnfon, while the iPS clones that did not express Gcnf were designated Gcnfoff.
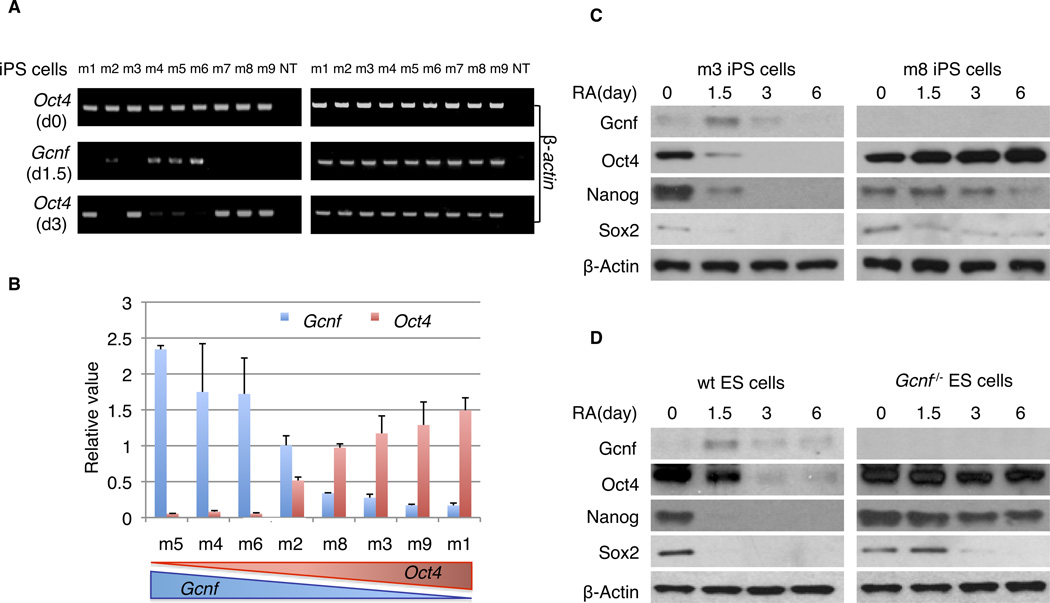
Figure 1.
Repression of Oct4 expression is dependent on induced Gcnf expression during iPS cell differentiation. (A) Using RT-PCR, Oct4 was detected in individual undifferentiated iPS clones and in differentiated iPS cells treated with RA for 3 days; Gcnf expression was detected at day 1.5 of RA induced differentiation. β-Actin served as a loading and integrity control (B). Q-RT-PCR was used to quantify the mRNA expression level of Oct4 at day 3 of RA-induced differentiation and Gcnf at 1.5 days of differentiation and compared with undifferentiated cells respectively. (C) Protein levels of Gcnf, Oct4, Nanog, Sox2 and control β-Actin were detected by western analysis in undifferentiated iPS cells (day 0) and differentiated cells treated with RA at days 1.5, 3, and 6 in Gcnfon iPS cells (m3) and Gcnfoff iPS cells (m8). (D) Protein levels for Gcnf, Oct4, Nanog, Sox2 and control β-actin were detected by western analysis in undifferentiated wt and Gcnf−/− ES cells (day 0) and differentiated wt and Gcnf−/− ES cells treated with RA at days 1.5, 3, and 6.
Oct4 expression was detected in undifferentiated ES cells (d0) and at three days of RA induced differentiation (d3) (Fig. 1A). Oct4 expression was repressed by day 3 of RA treatment in the four Gcnf positive (Gcnfon) iPS clones. In contrast, the five Gcnf negative (Gcnfoff) clones maintained Oct4 expression at day 3 of differentiation. Confirmation of this finding with Q-RT-PCR showed that the levels of Oct4 at day 3 of differentiation inversely correlated with the level of induction of Gcnf at day 1.5 (Fig. 1B). Silencing of pluripotency gene expression is necessary for normal differentiation to proceed so that lineage determinants can be properly expressed.
Two cell lines were selected to further characterize the importance of reactivation of Gcnf expression during iPS cell formation. The m8 iPS clone was observed to be a typical Gcnfoff cell, whereas the m5 iPS clone was a typical Gcnfon cell. Western analysis showed that the Gcnf and Oct4, Nanog and Sox2 expression patterns in Gcnfon and Gcnfoff clones are similar to that of their genetic counterparts, wt and Gcnf−/− ES cells, respectively. Oct4, Nanog and Sox2 expression in wt ES cells and Gcnfon iPS cells is silenced after differentiation; as a result of Gcnf induction in both cell lines (Fig. 1C, 1D). In contrast, Gcnf was undetectable in Gcnf−/− ES cells and Gcnfoff iPS cells, and Oct4, Nanog and Sox2 expression was maintained even at day 6 of differentiation (Fig. 1C, 1D). Thus, Gcnfoff iPS cells cannot efficiently repress Oct4, Nanog and Sox2 expression during differentiation because of the lack of Gcnf. The Gcnfoff iPS cells behaved just like the Gcnf−/− ES cells and thus represent an epigenetic knock out (KO) of Gcnf, equivalent to the genetic KO. The level of Oct4 repression is dependent on the level of Gcnf induction during differentiation.
Aberrant Gcnfoff iPS cell differentiation in vitro
To investigate the effect of the level of Gcnf expression on iPS cell pluripotency and the capacity for differentiation into the three germ layers in vitro, a standard differentiation protocol was adopted using monolayer culture or embryoid body (EB) formation and stepwise differentiation [1,21]. First, these cell lines were induced to differentiate by withdrawal of LIF and the addition of RA. The ability of these two iPS cell lines to differentiate into derivatives of the three different embryonic germ layers was characterized by analyzing marker gene expression. Expression of the endoderm markers, Afp and FoxA2 increased during Gcnfon iPS cell and wt ES cell differentiation [26]. In contrast, Afp and FoxA2 expression was undetectable in differentiated Gcnfoff iPS cells and Gcnf−/− ES cells (Fig. 2A, 2B). Induction of FoxA2 requires FoxD3, which binds to a response element located in the FoxA2 promoter [27]. Oct4 and FoxD3 bind identical regulatory sequences, implying that Oct4 likely competes with FoxD3 for DNA binding on the FoxA2 promoter. The loss of FoxA2 induction in Gcnfoff clones is likely due to maintained Oct4 expression. To further investigate whether the loss induction of endoderm gene expression correlated to Gcnf expression, we used Q-RT-PCR to detect Sox17 expression. The results showed that Sox17 expression increased rapidly during RA induced differentiation in wt ES cells and Gcnfon iPS cells (m4 and m5 iPS cells), in contrast Sox 17 was barely induced in Gcnf−/− ES cells and Gcnfoff iPS cells (m3 and m8 iPS cells) (Fig. 2C). We also found that the level of Sox 17 gene expression positively correlated with the level of Gcnf induction at 1.5 days of differentiation (Fig. 2D).
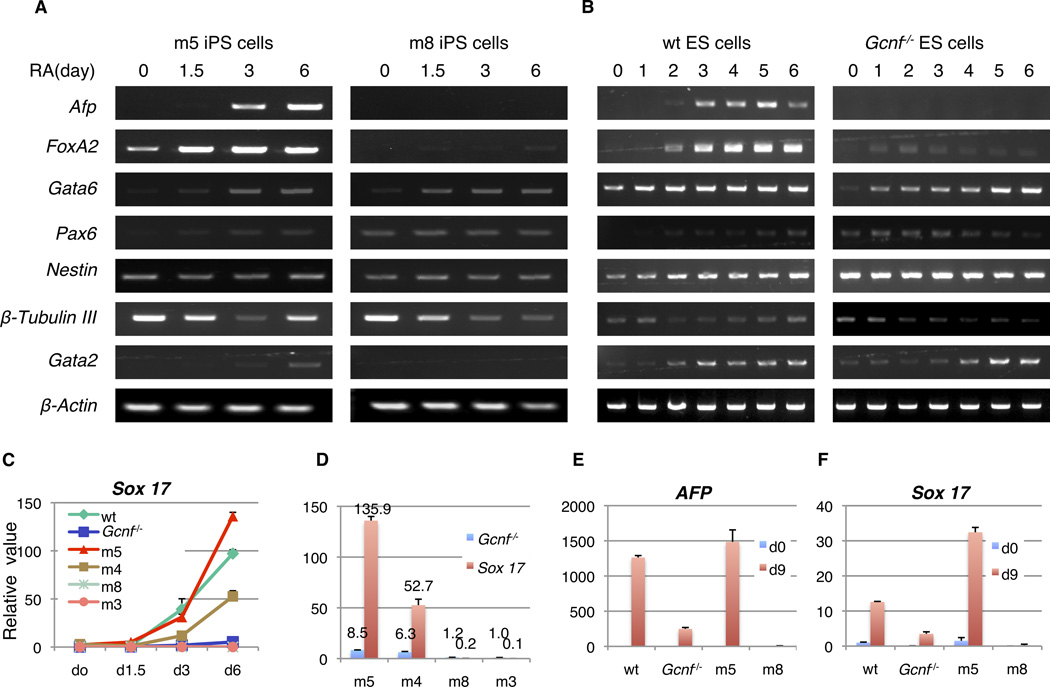
Figure 2.
Loss of Gcnf reprogramming results in defective activation of endoderm genes during iPS cell differentiation with RA treatment in vitro. Endoderm markers (Afp and FoxA2), mesoderm markers (Gata6 and Pax6), ectoderm markers (Nestin and β-Tubulin III) were detected by RT-PCR in (A) Gcnfon (m5) and Gcnfoff (m8) iPS cells, (B) wt and Gcnf−/− cells during RA induced differentiation. (C) The mRNA levels of Sox 17 was detected by Q-RT-PCR in wt ES cells, Gcnf−/− ES cells, Gcnfon iPS cells and Gcnfoff iPS cells. (D) Relationship of Gcnf and Sox 17 mRNA levels, Gcnf was detected at 1.5 days of differentiation, and Sox 17 was detected at 6 days of differentiation. (E) Comparison of Afp mRNA levels in EBs at d0 and d9 of differentiation Afp mRNA was detected by Q-RT-PCR. (F) Comparison of Sox 17 mRNA levels in EBs at d0 and d9 of differentiation Sox 17 mRNA was detected by Q-RT-PCR.
The expression of the mesoderm markers Pax6, gata1, and the ectoderm markers, β-Tubulin III and Nestin were also determined by RT-PCR. Gata6 expression showed no significant differences between the Gcnfoff and Gcnfon iPS and ES cell lines. Pax6 expression gradually increased in both m5 iPS cells (Gcnfon) and wt ES cells, opposite to what was observed in both m8 iPS (Gcnfoff) cells and Gcnf−/− ES cells during differentiation (Fig. 2A, 2B). Nestin expression displayed no significant difference between the different cell lines. Expression of β-Tubulin III was detected in all four undifferentiated cell types analyzed, and decreased to the lowest level at day 3 of differentiation. Subsequently, the expression of β-Tubulin gradually began to increase in m5 iPS cells (Gcnfon) and wt ES cells. In contrast, the m8 iPS cells (Gcnfoff) and Gcnf−/− ES cells exhibited no induction of β-Tubulin III expression at later stages of differentiation (Fig. 2A, 2B), affirming the deficiency reported in neural cell development attributed to the loss of Gcnf [28,29].
EB formation demonstrates the capacity of pluripotent cells to generate many cell types. To analyze the ability of Gcnfoff iPS cells to form EBs, the m5 and m8 iPS cells, as well as wt and Gcnf−/− ES cells, were dispersed as single cells in Petri dishes. After four days, all four cell lines formed EBs and their morphology did not display significant differences between the different cell types (supplemental information Fig. S2A). After plating the EBs onto gelatin coated tissue culture plates the m5 iPS cells (Gcnfon) showed extensive differentiation in contrast to the m8 cells (Gcnfoff), which differentiated into cells exhibiting a fibroblast-like morphology, instead of extensive differentiation (supplemental information Fig. S2B).
In order to verify that endoderm genes are also not activated during EB formation in Gcnf−/− and Gcnfoff iPS cells, the expression of the markers Sox17 and Afp were tested by Q-RT-PCR at day 9 of EB differentiation. The results showed that Sox 17 and Afp were activated in Gcnfon iPS cells (m5) and wt ES cells indicative of the formation of endoderm, however Sox 17 and Afp expression was not induced in Gcnfoff or Gcnf−/− EBs (Fig. 2E, F).
Gcnfoff iPS cell differentiation in vivo
The differentiation capability of Gcnfoff iPS cells was assessed in vivo via teratoma formation. The m5 and m8 iPS cells, as well as control wt and Gcnf−/− ES cells, were injected into Severe Combined Immunodeficiency (SCID) mice. All four cell types gave rise to teratomas. Histological analysis showed that well-differentiated teratomas with components of all three germ layers were found both in m5 iPS and in wt ES cell teratomas (Fig. 3A), whereas extensive differentiation derivatives of the three germ layers was not observed in m8 iPS derived teratomas (Fig. 3B). Gcnf−/− ES cells differentiated slightly better than the m8 (Gcnfoff) iPS cells, but not better than wt ES cells or m5 (Gcnfon) iPS cells (supplemental information Fig. S3). This result indicates that loss of Gcnf affected the capacity of the cells to differentiate into all cell types in vivo.
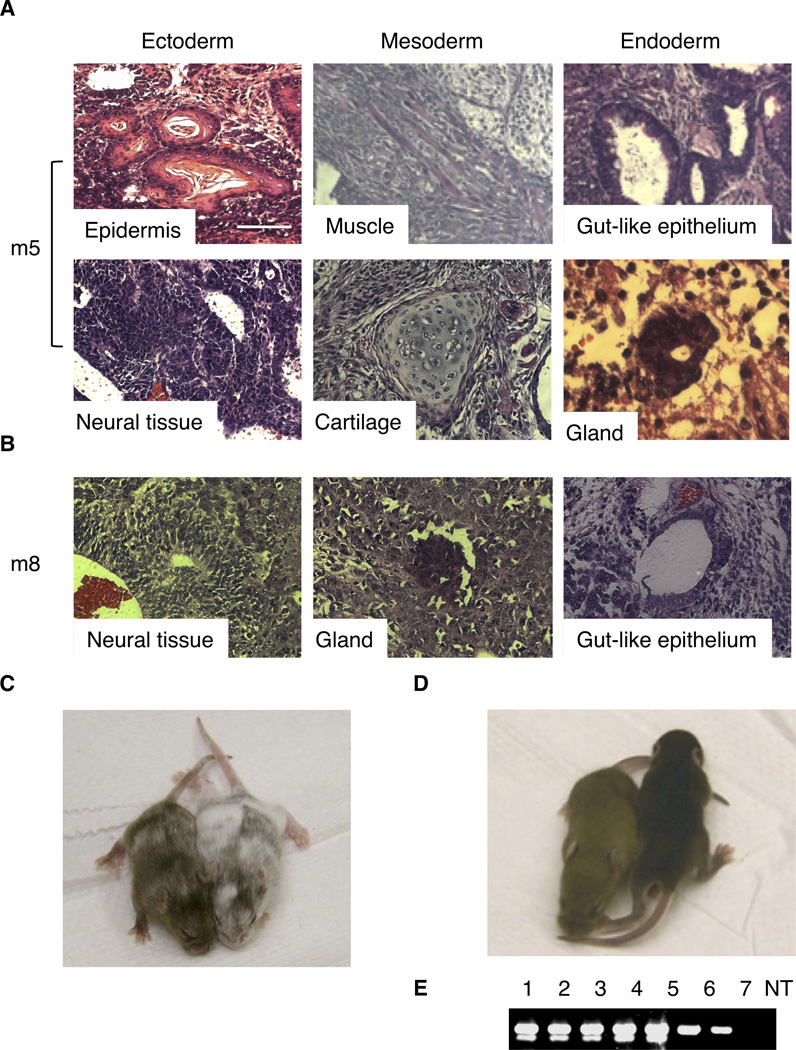
Figure 3.
Assessment of pluripotency of iPS Cells in vivo. (A) Gcnfon iPS cells were injected into the rear legs of SCID mice. Three weeks later teratomas were collected and histologic H & E staining was used to determine the differentiation into cell types distinctive of the three germ layers. (B) Teratomas from Gcnfoff iPS cells were analyzed by H & E staining. (C) Chimera generation from Gcnfon iPS cells by injection into albino C57 mouse blastcysts. No chimeras were generated from Gcnfoff (m8) iPS cell injections. (D) Mice generated from germ line transmission from Gcnfon iPS cell derived chimeric mice. (E) Mice with germ line transmission were confirmed by PCR genotyping for the Oct4 GFP reporter. Bars = 50 µm.
Loss of Chimeric Potential in Gcnfoff iPS cells
Chimerism and germ line transmission, which are the most stringent assays for developmental potential and pluripotency of ES or iPS cells [30,31], was also used to further evaluate the differentiation capacity of the iPS clones in vivo. The experimental schedule is diagrammed in supplemental information Figure S4. The m8 (Gcnfoff) iPS cells were unable to give rise to chimeras after blastocyst injection, whereas m5 (Gcnfon) iPS cells were able to generate live postnatal animals with high coat color chimerism (Fig. 3C). A chimeric male mouse was mated with albino C57/BL6 females in order to test for germ line transmission and true pluripotency. Germ line transmission was achieved from a m5 chimera (Fig. 3D), which was confirmed by PCR genotyping for the Oct4-GFP reporter contained in the original reprogrammed MEFs (Fig. 3E) and coat color, indicating that Gcnfon iPS cells (m5) were competent for germ line transmission (Fig. 3D). These results indicate that Gcnfon iPS cells are pluripotent, and loss of Gcnf expression through improper reprogramming leads to loss of iPS cell pluripotency.
Epigenetic changes in the Gcnf gene promoter during reprogramming
Reprogramming of somatic cells requires removal of epigenetic modifications on chromatin that are laid down during differentiation and developmental processes, allowing reactivation of pluripotency genes that are necessary for maintaining self-renewal and pluripotency of iPS cells. One type of epigenetic modification is DNA methylation of CpG dinucleotides, a hallmark of silenced genes [13,18]. However, somatic cell reprogramming does not completely demethylate CpG dinucleotides of genes that are epigenetically silenced during differentiation because iPS cells display greater levels of methylation than the ES cells [11]. The DNA methylation status at CpG dinucleotides correlates with gene expression. To determine the DNA methylation status of the Gcnf gene, bisulfite sequencing was performed to determine the extent of CpG methylation at a CpG island located between the Gcnf translational start site (TSS) and 66 bp upstream (Fig. 4A). This region of the Gcnf gene is hyper-methylated in the MEFs, which were re-programmed to form the iPS cells (Fig. 4B). As expected, the m5 (Gcnfon) iPS cells were predominantly demethylated in this region in the undifferentiated state indicative of re-programming, and then re-methylated during RA-induced differentiation (day 6) as Gcnf is transiently induced (Fig. 4B). This is consistent with epigenetic remodeling of the Gcnf promoter after retroviral infection. In contrast, m8 (Gcnfoff) cells were predominantly methylated at the Gcnf CpG island in the undifferentiated and differentiated states, which is indicative of a failure to re-program this locus (Fig. 4B). The methylation of the Gcnf promoter region in m8 clones correlates with the failure to induce Gcnf expression during iPS cell differentiation. Thus, during somatic cell reprogramming, epigenetic characteristics of iPS cells exhibit diversity among different iPS cell lines [11]. Interestingly, the m8 iPS cells represent an epigenetic KO of the Gcnf gene that phenocopies the genetic Gcnf KO in many ways.
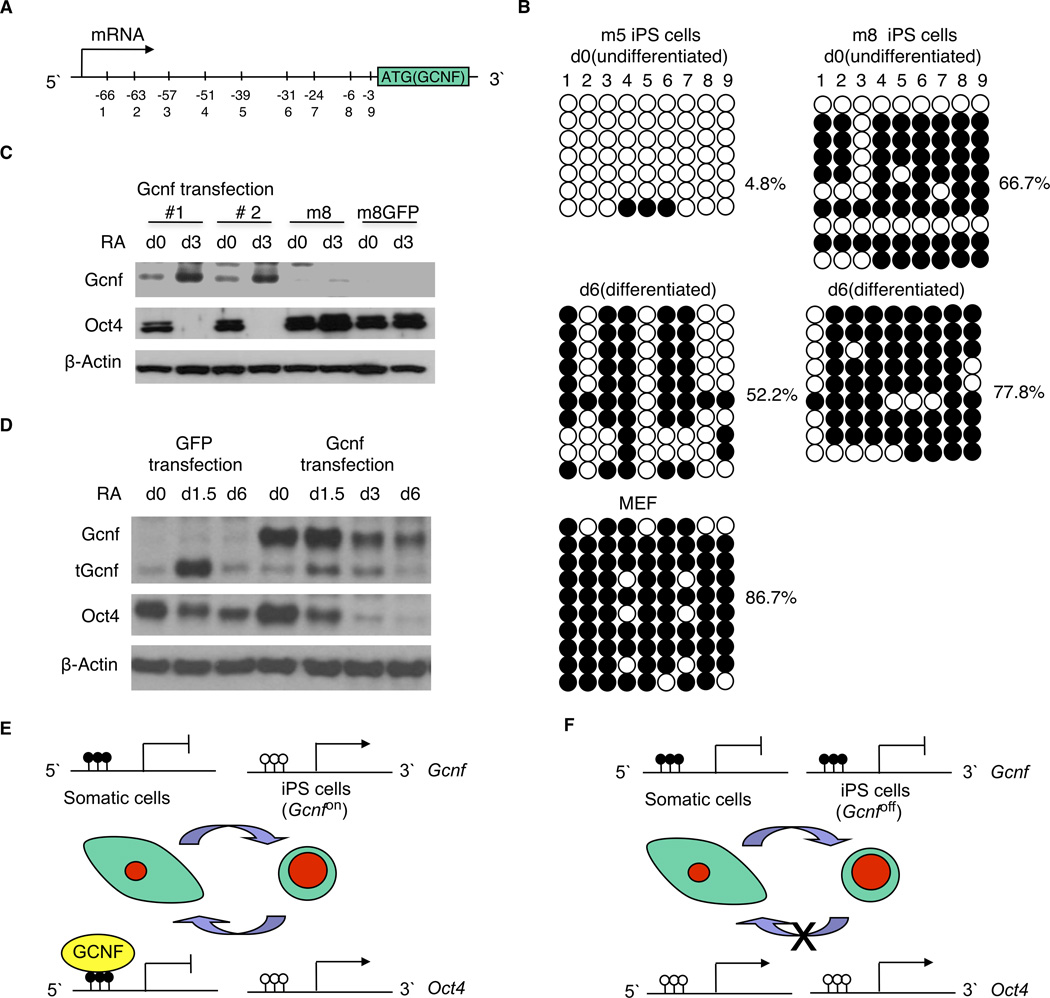
Figure 4.
Analysis of the DNA methylation status of the Gcnf promoter, and rescue of the m8 iPS cells. (A) The CpG island located in the Gcnf gene is close to the ATG and downstream of the transcriptional start site. (B) Methylation status of the Gcnf CpG island was determined by bisulphate sequencing in undifferentiated and differentiated iPS cells in both Gcnfon (m5) and Gcnfoff (m8) iPS cells, as well as the starting MEFs used for the reprogramming. White circles represent unmethylated CpG dinucleotides; black circles represent methylated CpG dinucleotides. (C) Oct4 repression was rescued in Gcnfoff (m8) iPS cells after retroviral re-expression of Gcnf. (D) Oct4 repression was rescued in Gcnf−/− ES cells after retroviral re-expression of Gcnf. The lower band was that of a truncated form of Gcnf (tGcnf) produced in this Gcnf mutant ES cell model. (E) Model showing that in good iPS cells DNA methylation of the Gcnf promoter has been removed during reprogramming allowing expression of Gcnf and repression of Oct4 during differentiation. (F) Model showing that in bad iPS cells DNA methylation of the Gcnf promoter has not been removed during reprogramming thus preventing expression of Gcnf and maintenance of Oct4 expression during subsequent iPS cell differentiation. White lollipops represent unmethylated CpG dinucleotides; black lollipops represent methylated CpG dinucleotides.
Gcnf rescues the repression of Oct4 in Gcnfoff iPS cells
In order to determine if the loss of Oct4 repression in the Gcnfoff iPS cells and Gcnf−/− ES cells can be rescued, mscv-Gcnf retrovirus (supplemental information Fig. S5A) and mscv-GFP control retrovirus (supplemental information Fig. S5B) were transfected into the Gcnfoff iPS cells and Gcnf−/− ES cells. After puromycin selection from transfected Gcnfoff iPS cells, M8 #1 and M8 #2 cell lines were used to analyze the expression of Gcnf and Oct4 during RA induced differentiation. Gcnf was expressed in both M8 #1 and M8 #2 cell lines, concomitantly Oct4 was repressed at day 3 of differentiation. In contrast, non-transfected Gcnfoff iPS cells and the control GFP transfected iPS cells both maintained Oct4 expression at day 3 because no Gcnf expression was detectable (Fig. 4C). The same results were observed after Gcnf was transfected into Gcnf−/− ES cells (Fig. 4D). Thus, Re-introduction of Gcnf can rescue the repression of Oct4 in the epigenetic KO of Gcnf m8 iPS cells. This result is the first demonstration in a gain-of-function experiment that Gcnf is required for inhibition of Oct4 expression during pluripotent cell differentiation.
Discussion
Our study demonstrates that re-activation of the silenced Gcnf gene during somatic cell reprogramming is required to maintain optimum pluripotency of iPS cells. Not all of the iPS clones selected reactivated the Gcnf gene. Only Gcnfon iPS clones can efficiently inhibit Oct4 expression after differentiation. Although the Gcnfoff iPS cells behave like self-renewing pluripotent cells in undifferentiated conditions, i.e. have the ability to form EBs and teratomas, and to differentiate into three germ layers, the capacity of differentiation into all cell types is significantly decreased similar to Gcnf−/− ES cells. However, like wt ES cells, the Gcnfon iPS cells can differentiate into the three germ layers in vitro and in vivo and give raise to chimaeras capable of germ line transmission.
Gcnf, as a silencer of pluripotency genes, represses not only Oct4 and Nanog [21] but also Cripto [32], and the level of Oct4 expression ultimately affects the direction ES cell differentiation [33]. During somatic cell reprogramming there is variable reactivation of the silenced Gcnf gene in iPS clones. The iPS clones that have demethylated the Gcnf gene can reactivate Gcnf expression during subsequent iPS cell differentiation (Fig. 4E). In contrast iPS clones in which the Gcnf gene was not demethylated Gcnf expression is not induced by RA and thus Oct4 expression is maintained during differentiation (Fig. 4F) [21]. Aberrant epigenetics often occur between different iPS clones during somatic cell reprogramming, which will affect the degree of pluripotency observed after differentiation [11]. This is an important finding because aberrant epigenetic reprogramming of the Gcnf gene was observed in nearly 50% of the clones analyzed. Maintenance of the epigenetic silencing of Gcnf appears to have a selective advantage during iPS formation as stable pluripotency gene expression is maintained. The aberrant re-programming of the Gcnf gene during iPS formation can account for ‘good looking’ iPS colonies that are not functional because of stable pluripotency gene expression [1]. However, stable maintenance of pluripotency gene expression comes at an expense, as these cells do not differentiate correctly.
Since the first patient-specific iPS cell lines have been developed [9,10], the derivation of patient tissue-specific cell types have been demonstrated, such as motoneurons [9]; thus the safety and quality of iPS cells is considered important and a limitation to their therapeutic application. Significant reprogramming variability in iPS cells compared to both ES cells and other iPS cells has been observed, especially differential iPS cell DNA methylation [34,35]. The methylation status of the Gcnf gene should be considered one of the markers of high quality and safe iPS cells, as it is the silencer of pluripotency genes. In addition, as an orphan member of the nuclear receptor gene family of ligand activated transcription factors these findings identify Gcnf as a small molecule target for the manipulation of iPS re-programming. Based on the results an antagonist that inhibits Gcnf transcriptional repression function should promote iPS formation in a stable but reversible manner.
Conclusion
Somatic cell reprogramming is a complicated stochastic process with variable degrees of re-writing the epigenetic code of important regulatory genes, which can subsequently affect iPS cell self-renew or differentiation. Our results show that correct reprogramming of the Gcnf gene is indispensible for the ability of iPS cells to differentiate correctly. Only those iPS clones where Gcnf expression was re-activated have the potential to differentiate into all three germ layers in vivo and in vitro. Clones that do not re-program the Gcnf gene maintain stable expression of pluripotency factors such as Oct4 and Nanog in the face of strong differentiation cues. The epigenetic status of the Gcnf gene should be considered one of the markers of high quality and safe iPS cells, as it is a repressor of pluripotency genes. The ability to rescue this defect by virally re-expressing Gcnf in these cells establishes Gcnf as a target to manipulate pluripotency gene expression. This is significant as Gcnf is an orphan member of the nuclear receptor gene family and thus has the potential to manipulated by small molecules.
Supplementary Material
Figure S1. iPS colonies was generated by introduction of retroviruses expressing-Oct4, Sox2, c-myc and Klf4. (A) Morphology of MEF cells, which were used to generate the iPS cells. Bars=20 µm. (B) iPS cell colony expressing GFP. (C) iPS colonies were stained with alkaline phosphatase and (D) mock. (E) The expression of total (T) and endogenous (E) Oct4 gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. (F) The expression of T and E Sox2 gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. (G) The expression of T and E c-Myc gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. (H) The expression of T and E Klf4 gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. Bars=20 µm.
Figure S2. Loss of Gcnf reprogramming resulted in defective activation of endoderm genes during iPS cell differentiation via EBs formation in vitro. (A) The cells were trypsinized into single cells and were plate into Petri dishes with differentiation media. All the type of cells could aggregate and form EBs. Bars=100 µm. (B) The EBs at day 4 were plated on the tissue culture dishes coated with 0.1% gelatin. Different morphological types of differentiated cells were observed after the EBs were placed on 0.1% gelatin coated plates. Bars=50 µm.
Figure S3. Histological analysis of teratoma sections after Haematoxylin and Eosin (H&E) staining. Teratomas derived from Gcnfon, Gcnfoff, wt and Gcnf−/− cells were sectioned, stained with H&E. Top bars=50 µm. Bottom bars=20 µm.
Figure S4. Diagram of production of iPS cell chimeras and germ line transmission of the chimeras. Gcnfon or Gcnfoff iPS cells were injected into the blastocysts of C57 Albino white genetic background. Chimeric blastocysts were subsequently transferred to day 2.5 pseudopregnant recipient CD-1 females. To obtain germ line transmission of iPS cells, chimeric males were bred with C57BL/6 females.
Figure S5. The map of Gcnf retroviral plasmid and GFP control plasmid used for transfection. (A) Mouse Gcnf (mGcnf) was cloned into vector under control of the mscv promotor. (B) GFP was cloned into a vector under the control of the mscv promotor.
ACKNOWLEDGEMENTS
We thank Dr. Ana Crane for technical help with the teratoma assays. We thank Dr. Francesco DeMayo for the blastocyst injections and generation of the chimeric mice. We thank Dr. Thomas Zwaka for critical reading of the manuscript. This work was supported a grant NIH P01 GM081627 to AJC and TPZ.
Footnotes
Author contributions:
H.W.: conception and design, Collection and/or assembly of data, data analysis and interpretation, Manuscript writing; X.W.; Collection and/or assembly of data, data analysis and interpretation; X.X: Collection and/or assembly of data; T.P.Z. Data analysis and interpretation; A. J. C.: conception and design, data analysis and interpretation, manuscript writing, financial support, final approval of manuscript
The authors indicate no potential conflicts of interest.
References
- 1.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 2.Aoi T, Yae K, Nakagawa M, et al. Generation of pluripotent stem cells from adult mouse liver and stomach cells. Science. 2008;321:699–702. doi: 10.1126/science.1154884. [DOI] [PubMed] [Google Scholar]
- 3.Stadtfeld M, Brennand K, Hochedlinger K. Reprogramming of Pancreatic β Cells into Induced Pluripotent Stem Cells. Curr Biol. 2008;18:890–894. doi: 10.1016/j.cub.2008.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim JB, Zaehres H, Wu G, et al. Pluripotent stem cells induced from adult neural stem cells by reprogramming with two factors. Nature. 2008;454:646–650. doi: 10.1038/nature07061. [DOI] [PubMed] [Google Scholar]
- 5.Hanna J, Markoulaki S, Schorderet P, et al. Direct reprogramming of terminally differentiated mature B lymphocytes to pluripotency. Cell. 2008;133:250–264. doi: 10.1016/j.cell.2008.03.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Park IH, Zhao R, West JA, et al. Reprogramming of human somatic cells to pluripotency with defined factors. Nature. 2008;451:141–146. doi: 10.1038/nature06534. [DOI] [PubMed] [Google Scholar]
- 7.Aasen T, Raya A, Barrero MJ, et al. Efficient and rapid generation of induced pluripotent stem cells from human keratinocytes. Nat Biotechnol. 2008;26:1276–1284. doi: 10.1038/nbt.1503. [DOI] [PubMed] [Google Scholar]
- 8.Loh YH, Agarwal S, Park IH, et al. Generation of induced pluripotent stem cells from human blood. Blood. 2009;113:5476–5479. doi: 10.1182/blood-2009-02-204800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Park IH, Arora N, Huo H, et al. Disease-specific induced pluripotent stem cells. Cell. 2008;134:877–886. doi: 10.1016/j.cell.2008.07.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Soldner F, Hockemeyer D, Beard C, et al. Parkinson’s disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell. 2009;136:964–977. doi: 10.1016/j.cell.2009.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stadtfeld M, Apostolou E, Akutsu H, et al. Aberrant silencing of imprinted genes on chromosome 12qF1 in mouse induced pluripotent stem cells. Nature. 2010;465:175–181. doi: 10.1038/nature09017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Takahashi K, Tanabe K, Ohnuki M, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 13.Chin MH, Mason Mike J, 1, et al. Induced pluripotent stem cells and embryonic stem cells are distinguished by gene expression signatures. Cell Stem Cell. 2009;5:111–123. doi: 10.1016/j.stem.2009.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marchetto MC, Yeo GW, Kainohana O, et al. Transcriptional signature and memory retention of human induced pluripotent stem cells. PloS One. 2009;4:e7076. doi: 10.1371/journal.pone.0007076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wilson KD, Venkatasubrahmanyam S, Jia F, et al. MicroRNA profiling of human-induced pluripotent stem cells. Stem Cells Del. 2009;18:749–758. doi: 10.1089/scd.2008.0247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pick M, Stelzer Y, Bar-Nur O, et al. Clone- and gene-specific aberrations of parental imprinting in human induced pluripotent stem cells. Stem Cells. 2009;27:2686–2690. doi: 10.1002/stem.205. [DOI] [PubMed] [Google Scholar]
- 17.Doi A, Park IH, Wen B, et al. Differential methylation of tissue- and cancer-specific CpG island shores distinguishes human induced pluripotent stem cells, embryonic stem cells and fibroblasts. Nature Genet. 2009;41:1350–1353. doi: 10.1038/ng.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Deng J, Shoemaker R, Xie B, et al. Targeted bisulfite sequencing reveals changes in DNA methylation associated with nuclear reprogramming. Nature Biotechnol. 2009;27:353–360. doi: 10.1038/nbt.1530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Boyer LA, Lee TI, Cole MF, et al. Core transcriptional regulatory circuitry in human embryonic stem cells. Cell. 2005;122:947–956. doi: 10.1016/j.cell.2005.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Johanasson H, Simonsson S. Core transcription factor, Oct4, Sox2 and nanog, individually from complexes with nucleophosmin(Npm1) to control embryonic stem (ES) cell fate determination. Aging. 2010;2:815–822. doi: 10.18632/aging.100222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gu P, LeMenuet D, Chung AC, et al. Orphan nuclear receptor GCNF is required for the repression of pluripotency genes during retinoic acid-induced embryonic stem cell differentiation. Mol Cell Biol. 2005;25:8507–8519. doi: 10.1128/MCB.25.19.8507-8519.2005. [DOI] [PMC free article] [PubMed] [Google Scholar] [Research Misconduct Found]
- 22.Sato N, Kondo M, Arai K. The orphan nuclear receptor GCNF recruits DNA methyltransferase for Oct-3/4 silencing. BBRC. 2006;344:845–851. doi: 10.1016/j.bbrc.2006.04.007. [DOI] [PubMed] [Google Scholar]
- 23.Fuhrmann G, Chung AC, Jackson KJ, et al. Mouse germline restriction of Oct4 expression by germ cell nuclear factor. Dev Cell. 2001;1:377–387. doi: 10.1016/s1534-5807(01)00038-7. [DOI] [PubMed] [Google Scholar]
- 24.Chung ACK, Katz D, Pereira FA, et al. Loss of Orphan Receptor Germ Cell Nuclear Factor Function Results in Ectopic Development of the Tail Bud and a Novel Posterior Truncation. Mol Cell Biol. 2001;21:663–677. doi: 10.1128/MCB.21.2.663-677.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Meissner A, Wernig M, Jaenisch R. Direct reprogramming of genetically unmodified fibroblasts into pluripotent cells. Nat Biotechnol. 2007;25:1177–1181. doi: 10.1038/nbt1335. [DOI] [PubMed] [Google Scholar]
- 26.Hromas R, Costa R. The hepatocyte nuclear factor-3/forkhead transcription regulatory family in development, inflammation, and neoplasia. Crit Rev Oncol Hematol. 1995;20:129–140. doi: 10.1016/1040-8428(94)00151-i. [DOI] [PubMed] [Google Scholar]
- 27.Guo Y, Costa R, Ramsey H, et al. The embryonic stem cell transcription factors Oct-4 and FoxD3 interact to regulate endodermal-specific promoter expression. Proc Natl Acad Sci USA. 2002;99:3663–3667. doi: 10.1073/pnas.062041099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Sattler U, Samochocki M, Maelicke A, et al. The expression level of the orphan nuclear receptor GCNF (germ cell nuclear factor) is critical for neuronal differentiation. Mol Endocrinol. 2004;18:2714–2726. doi: 10.1210/me.2004-0251. [DOI] [PubMed] [Google Scholar]
- 29.Akamatsu W, DeVeale B, Okano H, et al. Suppression of Oct4 by germ cell nuclear factor restricts pluripotency and promotes neural stem cell development in the early neural lineage. J Neurosci. 2009;29:2113–2124. doi: 10.1523/JNEUROSCI.4527-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Boland MJ, Hazen JL, Nazor KL, et al. Adult mice generated from induced pluripotent stem cells. Nature. 2009;461:91–94. doi: 10.1038/nature08310. [DOI] [PubMed] [Google Scholar]
- 31.Kang L, Wang J, Zhang Y, et al. iPS cells can support full-term development of tetraploid blastocyst-complemented embryos. Cell Stem Cell. 2009;5:135–138. doi: 10.1016/j.stem.2009.07.001. [DOI] [PubMed] [Google Scholar]
- 32.Hentschke M, Kurth I, Borgmeyer U, et al. Germ Cell Nuclear Factor Is a Repressor of CRIPTO-1 and CRIPTO-3. J Biol Chem. 2006;28:33497–33504. doi: 10.1074/jbc.M606975200. [DOI] [PubMed] [Google Scholar]
- 33.Niwa H, Miyazaki J, Smith AG. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat Genet. 2000;24:372–376. doi: 10.1038/74199. [DOI] [PubMed] [Google Scholar]
- 34.Baker M. Reprogramming: Faithful reporters. Nature Methods. 2012;9:231–234. [Google Scholar]
- 35.Lister R, Pelizzola M, Kida YS, et al. Hotspots of aberrant epigenomic reprogramming in human induced pluripotent stem cells. Nature. 2011;471:68–73. doi: 10.1038/nature09798. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. iPS colonies was generated by introduction of retroviruses expressing-Oct4, Sox2, c-myc and Klf4. (A) Morphology of MEF cells, which were used to generate the iPS cells. Bars=20 µm. (B) iPS cell colony expressing GFP. (C) iPS colonies were stained with alkaline phosphatase and (D) mock. (E) The expression of total (T) and endogenous (E) Oct4 gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. (F) The expression of T and E Sox2 gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. (G) The expression of T and E c-Myc gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. (H) The expression of T and E Klf4 gene in all undifferentiated iPS clones and wt ES cells was detected with Q-RT-PCR. Bars=20 µm.
Figure S2. Loss of Gcnf reprogramming resulted in defective activation of endoderm genes during iPS cell differentiation via EBs formation in vitro. (A) The cells were trypsinized into single cells and were plate into Petri dishes with differentiation media. All the type of cells could aggregate and form EBs. Bars=100 µm. (B) The EBs at day 4 were plated on the tissue culture dishes coated with 0.1% gelatin. Different morphological types of differentiated cells were observed after the EBs were placed on 0.1% gelatin coated plates. Bars=50 µm.
Figure S3. Histological analysis of teratoma sections after Haematoxylin and Eosin (H&E) staining. Teratomas derived from Gcnfon, Gcnfoff, wt and Gcnf−/− cells were sectioned, stained with H&E. Top bars=50 µm. Bottom bars=20 µm.
Figure S4. Diagram of production of iPS cell chimeras and germ line transmission of the chimeras. Gcnfon or Gcnfoff iPS cells were injected into the blastocysts of C57 Albino white genetic background. Chimeric blastocysts were subsequently transferred to day 2.5 pseudopregnant recipient CD-1 females. To obtain germ line transmission of iPS cells, chimeric males were bred with C57BL/6 females.
Figure S5. The map of Gcnf retroviral plasmid and GFP control plasmid used for transfection. (A) Mouse Gcnf (mGcnf) was cloned into vector under control of the mscv promotor. (B) GFP was cloned into a vector under the control of the mscv promotor.