Abstract
Cholangiocarcinoma is the second most common primary malignant tumor of the liver. Perihilar cholangiocarcinoma or Klatskin tumor represents more than 50% of all biliary tract cholangiocarcinomas. A wide range of risk factors have been identified among patients with Perihilar cholangiocarcinoma including advanced age, male gender, primary sclerosing cholangitis, choledochal cysts, cholelithiasis, cholecystitis, parasitic infection (Opisthorchis viverrini and Clonorchis sinensis), inflammatory bowel disease, alcoholic cirrhosis, nonalcoholic cirrhosis, chronic pancreatitis and metabolic syndrome. Various classifications have been used to describe the pathologic and radiologic appearance of cholangiocarcinoma. The three systems most commonly used to evaluate Perihilar cholangiocarcinoma are the Bismuth-Corlette (BC) system, the Memorial Sloan-Kettering Cancer Center and the TNM classification. The BC classification provides preoperative assessment of local spread. The Memorial Sloan-Kettering cancer center proposes a staging system according to three factors related to local tumor extent: the location and extent of bile duct involvement, the presence or absence of portal venous invasion, and the presence or absence of hepatic lobar atrophy. The TNM classification, besides the usual descriptors, tumor, node and metastases, provides additional information concerning the possibility for the residual tumor (R) and the histological grade (G). Recently, in 2011, a new consensus classification for the Perihilar cholangiocarcinoma had been published. The consensus was organised by the European Hepato-Pancreato-Biliary Association which identified the need for a new staging system for this type of tumors. The classification includes information concerning biliary or vascular (portal or arterial) involvement, lymph node status or metastases, but also other essential aspects related to the surgical risk, such as remnant hepatic volume or the possibility of underlying disease.
Keywords: Hilar cholangiocarcinoma, Klatskin tumor, Perihilar cholangiocarcinoma, Bile duct cancer
Core tip: The terminology and classification of Perihilar cholangiocarcinoma (Klatskin tumors) are sometime confusing. In the present revision, we analyze some of the risk factors identified as preneoplastic conditions, and the different systems used for staging these tumors, including the most recent consensus classification promoted by the European Hepato-Pancreato-Biliary Association.
RISK FACTORS FOR HILAR CHOLANGIOCARCINOMA
Cholangiocarcinoma (CCA) is the second most common primary malignant tumor of the liver after hepatocellular carcinoma. Depending on their location is generally divided into intrahepatic or extrahepatic (distal tumours of the common bile duct and perihilar or Klatskin tumor). The classical description of this tumor corresponds to William Altemeier (1957) and Gerald Klatskin (1965) and is a subtype of CCA that stems from aberrant growth of the ductal epithelium in the extrahepatic biliary tree. Perihilar cholangiocarcinoma (PHC) or Klatskin tumor represents more than 50% of all biliary tract cholangiocarcinomas[1].
In the United States, the incidence of this disease is rare with approximately 3000 cases diagnosed annually, and the age-adjusted incidence of extrahepatic CCA has decreased from 1.08 per 100000 to 0.82 per 100000 individuals over a 20-year period[2]. Some observed variations in the incidence of CCA and PHC may be due to a coding misclassification of these tumors in the International Classification of Diseases for Oncology (ICD-O), having been proposed a revision in order to ensure that all PHC are coded topographically to extrahepatic tumours only, rather than as currently to intra- or extrahepatic[3,4].
A wide range of risk factors have been identified among patients with PHC including advanced age, male gender, primary sclerosing cholangitis (PSC), choledochal cysts, cholelithiasis, cholecystitis, parasitic infection, inflammatory bowel disease, alcoholic cirrhosis, nonalcoholic cirrhosis and chronic pancreatitis[5].
One of the most influential and well-established risk factors is PSC. The prevalence of cholangiocarcinoma in patients who have PSC is 5%-15%, with an annual incidence rate of 0.6%-1.5%. In contrast to patients with ulcerative colitis, the time since diagnosis seems to have no importance. In most cases, cholangiocarcinomas are diagnosed within the first 2.5 years after the diagnosis of PSC, and prospective studies have reported that 37% of patients developing cholangiocarcinoma will do so within the first year following the diagnosis of PSC. The median age of diagnosis is in the 5th decade of life. At autopsy, CCA has been identified in as many as 40% of patients with PSC[6].
Cholelithiasis is also a known risk factor for both intrahepatic CCA and extrahepatic CCA. In a recent large retrospective review, patients with gallstones who did not have a cholecystectomy performed had a twofold increased incidence of CCA. This increased risk subsides to the equivalent of the normal population 10 years after cholecystectomy[7].
Bile-duct cysts are an established risk factor for CCA. Type I (solitary, extrahepatic) and IV (extrahepatic and intrahepatic) bile-duct cysts have the higher incidence. The lifetime incidence of CCA in these patients ranges from 6% to 30%. The average age at malignancy detection has been reported to be 32 years, which is younger than the age at presentation of CCA in the general population[8]. The risk of malignancy decreases after complete choledochal cyst excision; however, these patients are still at a increased risk of developing CC compared with the general population.
The hepatobiliary flukes Opisthorchis viverrini and Clonorchis sinensis are associated with the development of CCA, particularly in Southeast Asia (fivefold increased risk of CCA and an annual incidence of 87 per 100000). They are flat worms that inhabit the bile ducts and, occasionally, the gallbladder and pancreatic duct of mammals. Both parasites increase the susceptibility of cholangiocytes to endogenous and exogenous carcinogens via chronic irritation and increased cellular turnover. Nevertheless, a recent study from Thailand, found that despite the endemicity of Opisthorchis viverrini (24.5% prevalence among the adult population), the lifetime risk of CCA is only 5%, which suggest other co-factor must exists, such as role of lifestyle, diet and certain polymorphisms[9].
Another risk factor for CCA that is more common in Asian than Western countries is hepatolithiasis. It has been postulated that prolonged irritation and inflammation of the biliary epithelium by the calculi, bile stasis, and bacterial infections predispose to malignancy. CCA incidence rates of 10% in patients who have hepatolithiasis have been reported[10].
A recent study from China had stressed the importance of metabolic syndrome as potential risk factor for the development of biliary tract cancer. Cholelithiasis, triglycerides, LDL, diabetes, Apolipoprotein A and Apolipoprotein B were significantly associated with extrahepatic cholangiocarcinoma[11].
CLASSIFICATION OF HILAR CHOLANGIOCARCINOMA
Various terminology and classifications have been used to describe the pathologic and radiologic appearance of cholangiocarcinoma, and each describes a specific aspect of the tumor. However, some of the terminology and classifications are ambiguous and therefore confusing. In 1901, Eggel classified cholangiocarcinomas as nodular, massive and diffuse, like hepatocellular carcinoma. In 1983, Weinbren and Mutum classified cholangiocarcinoma into three types: nodular, sclerosing and papillary. Rosai, in 1996, distinguishes between polypoid and sclerosing forms. In the radiologic literature, hilar and extrahepatic cholangiocarcinomas have been classified as exophytic, infiltrating and polypoid (or papillary)[12].
The Liver Cancer Study Group of Japan proposed in 2000 a new classification based on growth characteristics, with tumors being identified as mass-forming, periductal-infiltrating and intraductal-growing types (Figure 1). This classification describes the gross appearance, growing characteristics, and biologic behavior, and it is helpful for radiologic interpretation. According to this classification, the exophytic or nodular type matches the mass-forming type, the infiltrating or sclerosing type matches the periductal-infiltrating, and the polypoid or papillary type matches the intraductal-growing type. The prognosis for mass-forming and periductal-infiltrating cholangiocarcinomas is generally unfavorable, whereas the prognosis for intraductal-growing types is much better after surgical resection[13].
Figure 1.

Morphologic classification of cholangiocarcinoma. A: Mass-forming; B: Periductal-infiltrating; C: Intraductal-growing.
CCAs can be classified anatomically as intrahepatic (peripheral), perihilar (Klatskin tumor), or extrahepatic. Perihilar cholangiocarcinoma arises at the bifurcation of the hepatic ducts, whereas intrahepatic cholangiocarcinoma arises from beyond second-order bile ducts. The extrahepatic bile ducts can be further divided into proximal, middle, and distal bile ducts. The proximal extrahepatic bile duct extends from the confluence of the right and left hepatic bile ducts to the level of the cystic duct. The middle portion of the extrahepatic bile ducts extends from the cystic duct to the level of the duodenum. The distal ducts are composed of the bile duct that extends to the level of the ampulla[14].
As in any other type of cancer, a staging system must ideally provide information about the prognosis and natural history of the disease, serve as a guide for therapy, and enable convincing comparisons of therapies among various institutions and over time. In so-called surgical diseases, a staging system is crucial for deciding between an aggressive approach (i.e., chance for cure) and only palliative alternatives. Another criteria for a good staging system is its ability to identify patients for the best type of surgery (e.g., local resection vs extensive resection or even liver transplantation).
The three systems most commonly used to evaluate PHC in most parts of the world are the Bismuth-Corlette (BC) system, the Memorial Sloan-Kettering Cancer Center (MSKCC) classification, and the American Joint Committee on Cancer (AJCC)/Union for International Cancer Control (UICC) TNM classification.
BC classification
The BC classification[15,16] provides preoperative assessment of local spread, and classifies Klatskin tumors as Type I (proximal bile duct tumors that do not extend to the bifurcation), Type II (tumors extend to the bifurcation without extension into the intrahepatic bile ducts), Types IIIa and IIIb (occluding the common hepatic duct and the right or left hepatic ducts, respectively), and Type IV (involving the confluence and both the right and left hepatic ducts) (Figure 2).
Figure 2.
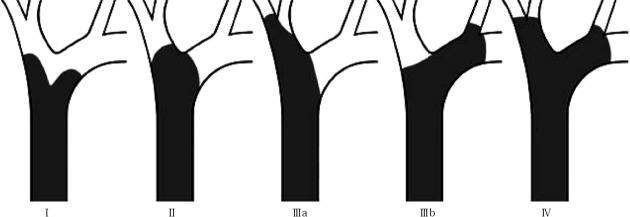
Bismuth-Corlette classification of Perihilar (Klatskin) tumors. TypeI: Proximal bile duct tumor that do not extend to the bifurcation; TypeII: Tumor extend to the bifurcation without extension into the intrahepatic bile ducts; TypeIIIa: Tumoral occlusion of the common hepatic duct and the right hepatic duct; TypeIIIb: Tumoral occlusion of the common hepatic duct and the left hepatic duct; Type IV: Tumor involving the confluence and both the right and left hepatic ducts.
In a recent study addressed to evaluate the accuracy, sensitivity, prognostic value and impact on the management of patients with Klatskin tumors, Paul et al[17], analyzing data of two centers of excellence and a meta-analysis of the literature, found that BC classification had an accuracy rate < 50%, with a low sensitivity for Type IIIA/IIIB tumors (in the 30% range), and it is not indicative of survival. Although the BC classification provides the first preoperative assessment of the possibility and extent of surgical resection, decision for laparotomy cannot be based on it, however, because does not include crucial information such as vascular encasement and distant metastases and further preoperative workup has to be made.
Another aspect to consider is that longitudinal spread pattern of a tumor can be related to gross morphology. Papillary tumors frequently present with long-range mucosal spread, while infiltrating tumors tend to show subepithelial extension.
MSKCC classification
In 1998, the MSKCC, proposed a different stage criteria for hilar cholangiocarcinoma according to three factors related to local tumor extent: the location and extent of bile duct involvement (in agreement to the BC system), the presence or absence of portal venous invasion, and the presence or absence of hepatic lobar atrophy. The initial system comprises four staging groups[18], but was simplified three years later to the definite model which comprises three stage groupings rather than four and represents a simple combining of two stages from the earlier format (Table 1). By taking full account of local tumor extent, the proposed staging system for hilar cholangiocarcinoma accurately predicts, in author’s opinions, resectability, the likelihood of metastatic disease, and survival[19].
Table 1.
Memorial Sloan-Kettering Cancer Center classification
| Stage | Criteria |
| T1 | The tumor involves the biliary confluence with unilateral involvement up to secondary biliary radicles. There is no portal vein involvement or liver atrophy |
| T2 | The tumor involves the biliary confluence with unilateral involvement up to secondary biliary radicles. There is ipsilateral portal vein involvement or ipsilateral hepatic lobar atrophy |
| T3 | The tumor involves the biliary confluence with bilateral involvement up to secondary biliary radicles, unilateral extension to secondary biliary radicles with contralateral portal vein involvement, unilateral involvement up to secondary biliary radicles with contralateral hepatic lobar atrophy, or main/bilateral portal vein involvement |
TNM classification
The AJCC has recently published new staging criteria for extrahepatic bile duct tumors (Table 2)[20]. These tumors were previously grouped into proximal, middle and distal tumors but were considered as a single entity and had single TNM classification. Now, the middle group of extrahepatic bile duct tumors have been removed as the treatment of this group is similar to either proximal or distal group. Currently, extrahepatic bile duct tumors are simply classified as perihilar and distal bile duct tumors. Further, these two subgroups have different TNM staging as their pathology, treatment and prognosis is variable.
Table 2.
Perihilar bile duct tumors (American Joint Commission on Cancer Staging 7th edition)
| T1 | Tumor confined to bile duct histologically |
| T2a | Tumor beyond the wall of bile duct into adjacent fat |
| T2b | Tumor beyond the wall of bile duct into liver parenchyma |
| T3 | Tumor invades ipsilateral portal vein (R or L) or hepatic artery (R or L) |
| T4 | Tumor invades |
| (1) Main portal vein or its branches bilaterally (or) | |
| (2) Common hepatic artery (or) | |
| (3) The second-order biliary radicals bilaterally | |
| (4) Unilateral second-order biliary radicals with contralateral portal vein or hepatic artery involvement | |
| Node | |
| Nx | Regional lymph nodes cannot be assessed. |
| N0 | No regional lymph node metastasis |
| N1 | Regional lymph node metastasis (including nodes along the cystic duct, common bile duct, hepatic artery, and portal vein) |
| N2 | Metastasis to periaortic, pericaval, superior mesenteric artery, and/or celiac artery lymph nodes |
| Metastasis | |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| Tumor stage AJCC staging 6th edition | |
| 0 | Tis, N0, M0 |
| I | T1, N0, M0 |
| II | T2a-b, N0, M0 |
| IIIa | T3, N0, M0 |
| IIIb | T1 or T2 |
| T3, N1, M0 | |
| IVa | T4, N0 |
| N1, M0 | |
| IVb | Any T, N2, M0 |
| Any T, any N, M1 | |
AJCC: American Joint Committee on Cancer; R: Right; L: Left.
Perihilar tumors refer to those located in the extrahepatic biliary tree proximal to the origin of the cystic duct. The early stage (T1) tumor for the extrahepatic bile duct cancers is described as tumor confined to the bile duct wall. On imaging this tumor presents as wall thickening of the bile duct. The low (fat) attenuation of the periductal fat is preserved. The T2 tumors are cancers that invade the periductal fat (T2a) or the liver (T2b). The proximal extrahepatic bile duct tumors may extend to the portal vein or hepatic artery.
The unilateral vascular extension is considered T3, whereas more advanced extension is considered T4. The latter (T4) includes extension into the main portal vein, common hepatic artery, contralateral vascular extension, and involvement of secondary biliary radical. Hepatic parenchymal involvement is now classified as T2 instead of T3, as patients with hepatic parenchymal involvement alone have a better prognosis compared to those with unilateral vascular involvement[21]. Distal bile duct tumors refer to those located between the junction of the cystic duct-bile duct and the ampulla of Vater. Previously these had the same AJCC classification as the proximal tumors but it has been recognized that these tumors have significant differences in the anatomy compared to the proximal lesions, which affect their resectability. Hence, these lesions have a separate TNM classification.
The nodal staging of bile ducts tumors is also different for the proximal and distal bile duct tumors. The proximal bile duct tumors have three classifications (N0, N1 and N2). N1 nodes refer to regional nodes such as hilar, cystic, pericholedochal, hepatic artery, portal and posterior pancreaticoduodenal. The N2 nodes refer to distant nodes such as celiac, superior mesenteric artery, and para-aortic nodes. The presence of N2 nodes may disqualify the patient from potential curative surgery. On imaging, there are no definite criteria for the diagnosis of malignant nodes[22] and for this reason the presence of equivocal lymph nodes cannot be used as a criterion for unresectability. A node that is larger than 1 cm in minimum diameter, round in morphology and heterogeneous in attenuation or with central necrosis, is likely to be malignant. Proximity to the primary mass also increases the likelihood of malignancy. The MR diffusion weighted images provide optimum contrast between lymph nodes and background anatomy. The M-staging for the extrahepatic biliary tumors is the similar for proximal and distal bile duct tumors. Metastases may be seen on computed tomography and magnetic resonance as soft tissue masses in the peritoneum, lungs, adrenals, liver and other sites. It is generally accepted that a fluorodeoxyglucose positron emission tomography (FDG-PET) is useful to detect distant metastases and may lead to change in management in up to 30% of patients.
Besides the stage grouping shown in Table 1, the TNM classification has additional descriptors for the residual tumor (which is labeled ‘‘R’’): Rx means that the presence of the residual tumor cannot be assessed, R0 represents no residual tumor, R1 reveals a microscopic residual tumor, and R2 denotes a macroscopic residual tumor. In addition, the histological grade (‘‘G’’) is expressed as Gx (no assessment), G1 (well differentiated), G2 (moderately differentiated), G3 (poorly differentiated), or G4 (undifferentiated).
The BC classification system is possibly the system most commonly used worldwide to stage PHC, although it fails to provide other key information such as vascular encasement, lymph node involvement, distant metastases and atrophy of a part of the liver. The MSKCC system does not evaluate the presence of nodal or distant metastases or the involvement of the artery. And the TNM staging is mostly used postoperatively and therefore fails to distinguish between the various surgical options, so that its usefulness in the preoperative setting is thus limited.
A consensus conference organized by the European Hepato-Pancreato-Biliary Association in 2007, identified the need for a new staging system for perihilar cholangiocarcinoma[23]. For this reason, an international working group was constituted with the aim to design a new staging system and registry for these tumors. The results of this project were published in 2011, proposing a new classification for Klatskin tumors using some parameters from previous staging systems[24] (Table 3). The BC classification is kept for the assessment of the bile duct (which is labeled ‘‘B’’ for bile duct or Bismuth); the letters ‘‘a’’ and ‘‘b’’ are omitted and are replaced by ‘‘R’’ (for right hepatic duct) and ‘‘L’’ (for left hepatic duct; Figure 3A). Thus, the label indicating one of the four types (depending on the localization of the tumor) will follow ‘‘B’’; for example, B2 indicates invasion of the bile duct confluence by the tumor. Additionally, the tumor size should be labeled as T1 (1 cm), T2 (1-3 cm), or T3 (3 cm). The choice of a 3-cm cutoff for T3 is based on accumulating data indicating a better prognosis for smaller tumors; this includes excellent outcomes after liver transplantation in the absence of any extrahepatic spread. The macroscopic form (which is labeled ‘‘F’’) will also be recorded as the periductal or sclerosing type (sclerosing), the nodular or mass-forming type (mass), or the polypoid or intraductal type (polypoid). Often, a distinction between the sclerosing type and the mass forming type is difficult, and therefore, a mixed type of tumor is added (mixed).
Table 3.
Consensus classification (European Hepato-Pancreato-Biliary Association)
| Label | Side location | Description |
| Bile duct (B) | ||
| B1 | Common bile duct | |
| B2 | Hepatic duct confluence | |
| B3 | R | Right hepatic duct |
| B3 | L | Left hepatic duct |
| B4 | Right and left hepatic duct | |
| Tumor size (T) | ||
| T1 | < 1 cm | |
| T2 | 1-3 cm | |
| T3 | ≥ 3 cm | |
| Tumor form (F) | ||
| Sclerosing | Sclerosing (or periductal) | |
| Mass | Mass-forming (or nodular) | |
| Mixed | Sclerosing and mass-forming | |
| Polypoid | Polypoid (or intraductal) | |
| Involvement (> 180°) of the portal vein (PV) | ||
| PV0 | No portal involvement | |
| PV1 | Main portal vein | |
| PV2 | Portal vein bifurcation | |
| PV3 | R | Right portal vein |
| PV3 | L | Left portal vein |
| PV4 | Right and left portal veins | |
| Involvement (> 180°) of the hepatic artery (HA) | ||
| HA0 | No portal involvement | |
| HA1 | Proper hepatic artery | |
| HA2 | Hepatic artery bifurcation | |
| HA3 | R | Right hepatic artery |
| HA3 | L | Left hepatic artery |
| HA4 | Right and left hepatic artery | |
| Liver remnant volume (V) | ||
| V0 | No information on the volume needed (liver resection not foreseen) | |
| V% | Indicate segments | Percentage of the total volume of a putative rem-nant liver after resection |
| Underlying liver disease (D) | Fibrosis | |
| Nonalcoholic steatohepatitis | ||
| Primary sclerosing cholangitis | ||
| Lymph nodes (N) | ||
| N0 | No lymph node involvement | |
| N1 | Hilar and/or hepatic artery lymph node involvement | |
| N2 | Periaortic lymph node involvement | |
| Metastases (M) | ||
| M0 | No distant metastases | |
| M1 | Distant metastases (including liver and peritoneal metastases) | |
R: Right; L: Left.
Figure 3.
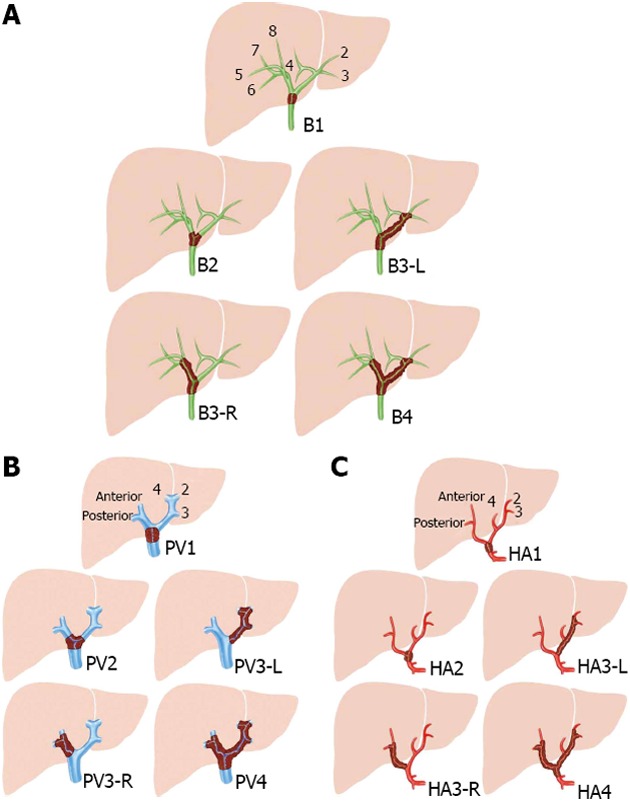
Consensus classification from the European Hepato-Pancreato-Biliary Association. Involvement of the portal vein or hepatic artery is considered when the tumor encompasses more than 180º of the circumference. A: Biliary involvement (B), based on the Bismuth-Corlette classification; B: Portal involvement; C: Arterial involvement. Adapted from Deoliveira et al [24].
The next factors providing information about the natural history and the choice of therapy include involvement of the vessels. In this regard, the portal vein is labeled ‘‘PV’’, and the hepatic artery is labeled ‘‘HA’’. The addition of “R” or “L” describes the side, right or left, with tumor involvement. It is also important to highlight when both the vein and the artery are free (HA0 and PV0, respectively).
In order to provide information related to the possibility to achieve a R0 resection in cases requiring en bloc resection of the bile duct and major hepatectomy, the staging system include a “V” (remnant hepatic volume) and a “D” (indicate the presence of an underlying disease such as fibrosis, nonalcoholic steatohepatitis, or PSC) labels, both identified as risk factors for surgery.
Lymph nodes are labeled ‘‘N’’, and classified as N1 (positive periportal or hepatic artery lymph nodes) and N2 for positive para-aortic lymph nodes. Metastases, including liver and peritoneal metastases, are marked as ‘‘M’’.
The staging should ideally be performed before and after surgery, and it should include all intraoperative information and results from macroscopic and microscopic examinations. In order to promote the use of this new complete, but complex, classification, an on line registry has been implemented and available at www.cholangioca.org (Figure 3).
Footnotes
P- Reviewers Aoyagi K, Stanojevic GZ, Sugimura H S- Editor Zhai HH L- Editor A E- Editor Yan JL
References
- 1.Jarnagin WR, Shoup M. Surgical management of cholangiocarcinoma. Semin Liver Dis. 2004;24:189–199. doi: 10.1055/s-2004-828895. [DOI] [PubMed] [Google Scholar]
- 2.Friman S. Cholangiocarcinoma--current treatment options. Scand J Surg. 2011;100:30–34. doi: 10.1177/145749691110000106. [DOI] [PubMed] [Google Scholar]
- 3.Shin HR, Oh JK, Masuyer E, Curado MP, Bouvard V, Fang Y, Wiangnon S, Sripa B, Hong ST. Comparison of incidence of intrahepatic and extrahepatic cholangiocarcinoma--focus on East and South-Eastern Asia. Asian Pac J Cancer Prev. 2010;11:1159–1166. [PubMed] [Google Scholar]
- 4.Khan SA, Emadossadaty S, Ladep NG, Thomas HC, Elliott P, Taylor-Robinson SD, Toledano MB. Rising trends in cholangiocarcinoma: is the ICD classification system misleading us? J Hepatol. 2012;56:848–854. doi: 10.1016/j.jhep.2011.11.015. [DOI] [PubMed] [Google Scholar]
- 5.Welzel TM, Graubard BI, El-Serag HB, Shaib YH, Hsing AW, Davila JA, McGlynn KA. Risk factors for intrahepatic and extrahepatic cholangiocarcinoma in the United States: a population-based case-control study. Clin Gastroenterol Hepatol. 2007;5:1221–1228. doi: 10.1016/j.cgh.2007.05.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tyson GL, El-Serag HB. Risk factors for cholangiocarcinoma. Hepatology. 2011;54:173–184. doi: 10.1002/hep.24351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nordenstedt H, Mattsson F, El-Serag H, Lagergren J. Gallstones and cholecystectomy in relation to risk of intra- and extrahepatic cholangiocarcinoma. Br J Cancer. 2012;106:1011–1015. doi: 10.1038/bjc.2011.607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Valero V, Cosgrove D, Herman JM, Pawlik TM. Management of perihilar cholangiocarcinoma in the era of multimodal therapy. Expert Rev Gastroenterol Hepatol. 2012;6:481–495. doi: 10.1586/egh.12.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Songserm N, Promthet S, Sithithaworn P, Pientong C, Ekalaksananan T, Chopjitt P, Parkin DM. Risk factors for cholangiocarcinoma in high-risk area of Thailand: role of lifestyle, diet and methylenetetrahydrofolate reductase polymorphisms. Cancer Epidemiol. 2012;36:e89–e94. doi: 10.1016/j.canep.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 10.Blechacz BR, Gores GJ. Cholangiocarcinoma. Clin Liver Dis. 2008;12:131–50, ix. doi: 10.1016/j.cld.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 11.Wu Q, He XD, Yu L, Liu W, Tao LY. The metabolic syndrome and risk factors for biliary tract cancer: a case-control study in China. Asian Pac J Cancer Prev. 2012;13:1963–1969. doi: 10.7314/apjcp.2012.13.5.1963. [DOI] [PubMed] [Google Scholar]
- 12.Lim JH. Cholangiocarcinoma: morphologic classification according to growth pattern and imaging findings. AJR Am J Roentgenol. 2003;181:819–827. doi: 10.2214/ajr.181.3.1810819. [DOI] [PubMed] [Google Scholar]
- 13.Liver Cancer Study Group of Japan. The general rules for the clinical and pathological study of primary liver cancer, 4th ed. Tokyo: Kanehara; 2000. [Google Scholar]
- 14.Sainani NI, Catalano OA, Holalkere NS, Zhu AX, Hahn PF, Sahani DV. Cholangiocarcinoma: current and novel imaging techniques. Radiographics. 2008;28:1263–1287. doi: 10.1148/rg.285075183. [DOI] [PubMed] [Google Scholar]
- 15.Bismuth H, Corlette MB. Intrahepatic cholangioenteric anastomosis in carcinoma of the hilus of the liver. Surg Gynecol Obstet. 1975;140:170–178. [PubMed] [Google Scholar]
- 16.Bismuth H, Nakache R, Diamond T. Management strategies in resection for hilar cholangiocarcinoma. Ann Surg. 1992;215:31–38. doi: 10.1097/00000658-199201000-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Paul A, Kaiser GM, Molmenti EP, Schroeder T, Vernadakis S, Oezcelik A, Baba HA, Cicinnati VR, Sotiropoulos GC. Klatskin tumors and the accuracy of the Bismuth-Corlette classification. Am Surg. 2011;77:1695–1699. [PubMed] [Google Scholar]
- 18.Burke EC, Jarnagin WR, Hochwald SN, Pisters PW, Fong Y, Blumgart LH. Hilar Cholangiocarcinoma: patterns of spread, the importance of hepatic resection for curative operation, and a presurgical clinical staging system. Ann Surg. 1998;228:385–394. doi: 10.1097/00000658-199809000-00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Jarnagin WR, Fong Y, DeMatteo RP, Gonen M, Burke EC, Bodniewicz BS J, Youssef BA M, Klimstra D, Blumgart LH. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann Surg. 2001;234:507–517; discussion 517-519. doi: 10.1097/00000658-200110000-00010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.American Joint Committee on Cancer. AJCC cancer staging manual. 7th ed. New York/London: Springer; 2011. [Google Scholar]
- 21.Ganeshan D, Moron FE, Szklaruk J. Extrahepatic biliary cancer: New staging classification. World J Radiol. 2012;4:345–352. doi: 10.4329/wjr.v4.i8.345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ruys AT, Kate FJ, Busch OR, Engelbrecht MR, Gouma DJ, van Gulik TM. Metastatic lymph nodes in hilar cholangiocarcinoma: does size matter? HPB (Oxford) 2011;13:881–886. doi: 10.1111/j.1477-2574.2011.00389.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Toouli J, Gigot JF, Gouma D, Belghiti J, Clavien PA. Editorial comment. Vol. 10. Oxford: HPB; 2008. p. 71. [Google Scholar]
- 24.Deoliveira ML, Schulick RD, Nimura Y, Rosen C, Gores G, Neuhaus P, Clavien PA. New staging system and a registry for perihilar cholangiocarcinoma. Hepatology. 2011;53:1363–1371. doi: 10.1002/hep.24227. [DOI] [PubMed] [Google Scholar]


