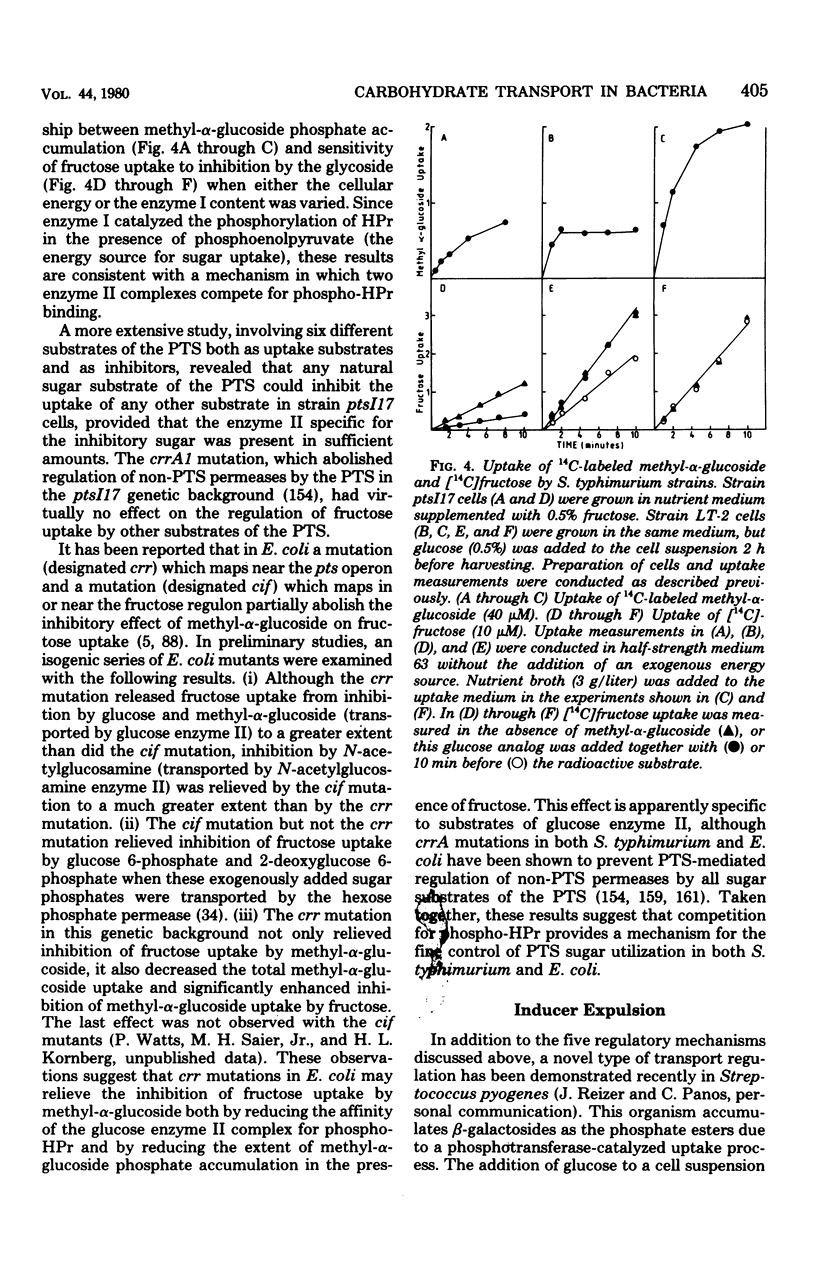
Full text
PDF
































Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adler J., Hazelbauer G. L., Dahl M. M. Chemotaxis toward sugars in Escherichia coli. J Bacteriol. 1973 Sep;115(3):824–847. doi: 10.1128/jb.115.3.824-847.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adler L. W., Rosen B. P. Functional mosaicism of membrane proteins in vesicles of Escherichia coli. J Bacteriol. 1977 Feb;129(2):959–966. doi: 10.1128/jb.129.2.959-966.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altendorf K., Lukas M., Kohl B., Müller C. R., Sandermann H., Jr Isolation and purification of bacterial membrane proteins by the use of organic solvents: the lactose permease and the carbodiimide-reactive protein of the adenosinetriphosphatase complex of Escherichia coli. J Supramol Struct. 1977;6(2):229–238. doi: 10.1002/jss.400060208. [DOI] [PubMed] [Google Scholar]
- Amaral D., Kornberg H. L. Regulation of fructose uptake by glucose in Escherichia coli. J Gen Microbiol. 1975 Sep;90(1):157–168. doi: 10.1099/00221287-90-1-157. [DOI] [PubMed] [Google Scholar]
- Anderson B., Weigel N., Kundig W., Roseman S. Sugar transport. 3. Purification and properties of a phosphocarrier protein (HPr) of the phosphoenolpyruvate-dependent phosphotransferase system of Escherichia coli. J Biol Chem. 1971 Nov 25;246(22):7023–7033. [PubMed] [Google Scholar]
- Anraku Y. Transport of sugars and amino acids in bacteria. I. Purification and specificity of the galactose- and leucine-binding proteins. J Biol Chem. 1968 Jun 10;243(11):3116–3122. [PubMed] [Google Scholar]
- BUTTIN G., COHEN G. N., MONOD J., RICKENBERG H. V. La galactoside-perméase d'Escherichia coli. Ann Inst Pasteur (Paris) 1956 Dec;91(6):829–857. [PubMed] [Google Scholar]
- Bachmann B. J., Low K. B., Taylor A. L. Recalibrated linkage map of Escherichia coli K-12. Bacteriol Rev. 1976 Mar;40(1):116–167. doi: 10.1128/br.40.1.116-167.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumann L., Baumann P. Catabolism of D-fructose and D-ribose by Pseudomonas doudoroffii. II. Properties of 1-phosphofructokinase and 6-phosphofructokinase. Arch Microbiol. 1975 Nov 7;105(3):241–248. doi: 10.1007/BF00447142. [DOI] [PubMed] [Google Scholar]
- Baumann P., Baumann L. Catabolism of D-fructose and D-ribose by Pseudomonas doudoroffii. I. Physiological studies and mutant analysis. Arch Microbiol. 1975 Nov 7;105(3):225–240. doi: 10.1007/BF00447141. [DOI] [PubMed] [Google Scholar]
- Belaich A., Simonpietri P., Belaich J. P. (Study on the beta-galactoside permease of Escherichia coli). Solubilization of a thiodigalactoside-binding protein from E.coli membrane containing beta-galactoside permease. Biochem Biophys Res Commun. 1978 Jun 14;82(3):847–852. doi: 10.1016/0006-291x(78)90860-4. [DOI] [PubMed] [Google Scholar]
- Bentaboulet M., Kepes A. Counter-transport mediated by the lactose permease of Escherichia coli. Biochim Biophys Acta. 1977 Nov 15;471(1):125–134. doi: 10.1016/0005-2736(77)90400-x. [DOI] [PubMed] [Google Scholar]
- Berger E. A. Different mechanisms of energy coupling for the active transport of proline and glutamine in Escherichia coli. Proc Natl Acad Sci U S A. 1973 May;70(5):1514–1518. doi: 10.1073/pnas.70.5.1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berger E. A., Heppel L. A. Different mechanisms of energy coupling for the shock-sensitive and shock-resistant amino acid permeases of Escherichia coli. J Biol Chem. 1974 Dec 25;249(24):7747–7755. [PubMed] [Google Scholar]
- Berman-Kurtz M., Lin E. C., Richey D. P. Promoter-like mutant with increased expression of the glycerol kinase operon of Escherichia coli. J Bacteriol. 1971 Jun;106(3):724–731. doi: 10.1128/jb.106.3.724-731.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berman M., Zwaig N., Lin E. C. Suppression of a pleiotropic mutant affecting glycerol dissimilation. Biochem Biophys Res Commun. 1970 Jan 23;38(2):272–278. doi: 10.1016/0006-291x(70)90708-4. [DOI] [PubMed] [Google Scholar]
- COHEN G. N., RICKENBERG H. V. Etude directe de la fixation d'un inducteur de la beta-galactosidase par les cellules d'Escherichia coli. C R Hebd Seances Acad Sci. 1955 Jan 24;240(4):466–468. [PubMed] [Google Scholar]
- Calmes R., Deal S. J. Glycerol transport by Nocardia asteroides. Can J Microbiol. 1972 Nov;18(11):1703–1708. doi: 10.1139/m72-264. [DOI] [PubMed] [Google Scholar]
- Carter J. R., Fox C. F., Kennedy E. P. Interaction of sugars with the membrane protein component of the lactose transport system of Escherichia coli. Proc Natl Acad Sci U S A. 1968 Jun;60(2):725–732. doi: 10.1073/pnas.60.2.725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cecchini G., Koch A. L. Effect of uncouplers on "downhill" beta-galactoside transport in energy-depleted cells of Escherichia coli. J Bacteriol. 1975 Jul;123(1):187–195. doi: 10.1128/jb.123.1.187-195.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cirillo V. P., Razin S. Distribution of a phosphoenolypyruvate-dependent sugar phosphotransferase system in mycoplasms. J Bacteriol. 1973 Jan;113(1):212–217. doi: 10.1128/jb.113.1.212-217.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins S. H., Jarvis A. W., Lindsay R. J., Hamilton W. A. Proton movements coupled to lactate and alanine transport in Escherichia coli: isolation of mutants with altered stoichiometry in alanine transport. J Bacteriol. 1976 Jun;126(3):1232–1244. doi: 10.1128/jb.126.3.1232-1244.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conrad R., Schlegel H. G. Different pathways for fructose and glucose utilization in Rhodopseudomonas capsulata and demonstration of 1-phosphofructokinase in phototrophic bacteria. Biochim Biophys Acta. 1974 Jul 17;358(1):221–225. doi: 10.1016/0005-2744(74)90273-3. [DOI] [PubMed] [Google Scholar]
- Conrad R., Schlegel H. G. Metabolism of fructose in Thiocapsa roseopersicina. Z Allg Mikrobiol. 1978;18(5):309–320. doi: 10.1002/jobm.3630180502. [DOI] [PubMed] [Google Scholar]
- Cordaro C. Genetics of the bacterial phosphoenolpyruvate: glycose phosphotransferase system. Annu Rev Genet. 1976;10:341–359. doi: 10.1146/annurev.ge.10.120176.002013. [DOI] [PubMed] [Google Scholar]
- Cordaro J. C., Anderson R. P., Grogan E. W., Jr, Wenzel D. J., Engler M., Roseman S. Promoter-like mutation affecting HPr and enzyme I of the phosphoenolpyruvate: sugar phosphotransferase system in Salmonella typhimurium. J Bacteriol. 1974 Oct;120(1):245–252. doi: 10.1128/jb.120.1.245-252.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cozzarelli N. R., Freedberg W. B., Lin E. C. Genetic control of L-alpha-glycerophosphate system in Escherichia coli. J Mol Biol. 1968 Feb 14;31(3):371–387. doi: 10.1016/0022-2836(68)90415-4. [DOI] [PubMed] [Google Scholar]
- Curtis S. J. Mechanism of energy coupling for transport of D-ribose in Escherichia coli. J Bacteriol. 1974 Oct;120(1):295–303. doi: 10.1128/jb.120.1.295-303.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das M. Mitogenic hormone-induced intracellular message: assay and partial characterization of an activator of DNA replication induced by epidermal growth factor. Proc Natl Acad Sci U S A. 1980 Jan;77(1):112–116. doi: 10.1073/pnas.77.1.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delobbe A., Chalumeau H., Claverie J. M., Gay P. Phosphorylation of intracellular fructose in Bacillus subtilis mediated by phosphoenolpyruvate-1-fructose phosphotransferase. Eur J Biochem. 1976 Jul 15;66(3):485–491. doi: 10.1111/j.1432-1033.1976.tb10573.x. [DOI] [PubMed] [Google Scholar]
- Dietz G. W., Heppel L. A. Studies on the uptake of hexose phosphates. 3. Mechanism of uptake of glucose 1-phosphate in Escherichia coli. J Biol Chem. 1971 May 10;246(9):2891–2897. [PubMed] [Google Scholar]
- Dietz G. W., Jr Growth of Escherichia coli on glucosamine 6-phosphate: selection of a constitutive hexose phosphate transport system mutant. Can J Microbiol. 1978 Mar;24(3):203–208. doi: 10.1139/m78-037. [DOI] [PubMed] [Google Scholar]
- Dietz G. W., Jr The hexose phosphate transport system of Escherichia coli. Adv Enzymol Relat Areas Mol Biol. 1976;44:237–259. doi: 10.1002/9780470122891.ch7. [DOI] [PubMed] [Google Scholar]
- Drapeau G. R., Matula T. I., MacLeod R. A. Nutrition and metabolism of marine bacteria. XV. Relation of Na+-activated transport to the Na+ requirement of a marine pseudomonad for growth. J Bacteriol. 1966 Jul;92(1):63–71. doi: 10.1128/jb.92.1.63-71.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EGAN J. B., MORSE M. L. CARBOHYDRATE TRANSPORT IN STAPHYLOCOCCUS AUREUS I. GENETIC AND BIOCHEMICAL ANALYSIS OF A PLEIOTROPIC TRANSPORT MUTANT. Biochim Biophys Acta. 1965 Feb 15;97:310–319. doi: 10.1016/0304-4165(65)90096-6. [DOI] [PubMed] [Google Scholar]
- Eidels L., Rick P. D., Stimler N. P., Osborn M. J. Transport of D-arabinose-5-phosphate and D-sedoheptulose-7-phosphate by the hexose phosphate transport system of Salmonella typhimurium. J Bacteriol. 1974 Jul;119(1):138–143. doi: 10.1128/jb.119.1.138-143.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Englesberg E., Wilcox G. Regulation: positive control. Annu Rev Genet. 1974;8:219–242. doi: 10.1146/annurev.ge.08.120174.001251. [DOI] [PubMed] [Google Scholar]
- Essenberg R. C., Kornberg H. L. Energy coupling in the uptake of hexose phosphates by Escherichia coli. J Biol Chem. 1975 Feb 10;250(3):939–945. [PubMed] [Google Scholar]
- Essenberg R. C., Kornberg H. L. Location of the gene specifying hexose phosphate transport (uhp) on the chromosome of Escherichia coli. J Gen Microbiol. 1977 Mar;99(1):157–169. doi: 10.1099/00221287-99-1-157. [DOI] [PubMed] [Google Scholar]
- Ferenci T., Boos W., Schwartz M., Szmelcman S. Energy-coupling of the transport system of Escherichia coli dependent on maltose-binding protein. Eur J Biochem. 1977 May 2;75(1):187–193. doi: 10.1111/j.1432-1033.1977.tb11516.x. [DOI] [PubMed] [Google Scholar]
- Ferenci T., Kornberg H. L., Smith Janet. Isolation and properties of a regulatory mutant in the hexose phosphate transport system of Escherichia coli. FEBS Lett. 1971 Mar 5;13(3):133–136. doi: 10.1016/0014-5793(71)80218-1. [DOI] [PubMed] [Google Scholar]
- Flagg J. L., Wilson T. H. A protonmotive force as the source of energy for galactoside transport in energy depleted Escherichia coli. J Membr Biol. 1977 Mar 8;31(3):233–255. doi: 10.1007/BF01869407. [DOI] [PubMed] [Google Scholar]
- Fox C. F., Carter J. R., Kennedy E. P. GENETIC CONTROL OF THE MEMBRANE PROTEIN COMPONENT OF THE LACTOSE TRANSPORT SYSTEM OF Escherichia coli. Proc Natl Acad Sci U S A. 1967 Mar;57(3):698–705. doi: 10.1073/pnas.57.3.698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fox C. F., Kennedy E. P. Specific labeling and partial purification of the M protein, a component of the beta-galactoside transport system of Escherichia coli. Proc Natl Acad Sci U S A. 1965 Sep;54(3):891–899. doi: 10.1073/pnas.54.3.891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fried V. A. A novel mutant of the lac transport system of Escherichia coli. J Mol Biol. 1977 Aug 25;114(4):477–490. doi: 10.1016/0022-2836(77)90173-5. [DOI] [PubMed] [Google Scholar]
- Friedman S. A., Hays J. B. Initial characterization of hexose and hexitol phosphoenolpyruvate-dependent phosphotransferases of Staphylococcus aureus. J Bacteriol. 1977 Jun;130(3):991–999. doi: 10.1128/jb.130.3.991-999.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gee D. L., Baumann P., Baumann L. Enzymes of D-fructose catabolism in species of Beneckea and Photobacterium. Arch Microbiol. 1975 Apr 7;103(2):205–207. doi: 10.1007/BF00436351. [DOI] [PubMed] [Google Scholar]
- HAYASHI S., LIN E. C. CAPTURE OF GLYCEROL BY CELLS OF ESCHERICHIA COLI. Biochim Biophys Acta. 1965 Mar 29;94:479–487. doi: 10.1016/0926-6585(65)90056-7. [DOI] [PubMed] [Google Scholar]
- Harold F. M. Conservation and transformation of energy by bacterial membranes. Bacteriol Rev. 1972 Jun;36(2):172–230. doi: 10.1128/br.36.2.172-230.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harris P., Kornberg H. L. The uptake of glucose by a thermophilic Bacillus sp. Proc R Soc Lond B Biol Sci. 1972 Sep 19;182(1067):159–170. doi: 10.1098/rspb.1972.0072. [DOI] [PubMed] [Google Scholar]
- Hayashi S. I., Lin E. C. Product induction of glycerol kinase in Escherichia coli. J Mol Biol. 1965 Dec;14(2):515–521. doi: 10.1016/s0022-2836(65)80200-5. [DOI] [PubMed] [Google Scholar]
- Henderson P. J., Giddens R. A., Jones-Mortimer M. C. Transport of galactose, glucose and their molecular analogues by Escherichia coli K12. Biochem J. 1977 Feb 15;162(2):309–320. doi: 10.1042/bj1620309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hengstenberg W. Enzymology of carbohydrate transport in bacteria. Curr Top Microbiol Immunol. 1977;77:97–126. doi: 10.1007/978-3-642-66740-4_4. [DOI] [PubMed] [Google Scholar]
- Hernandez-Asensio M., Ramirez J. M., Del Campo F. F. The control by respiration of the uptake of alpha-methyl glucoside in Escherichia coli K12. Arch Microbiol. 1975 Apr 7;103(2):155–162. doi: 10.1007/BF00436343. [DOI] [PubMed] [Google Scholar]
- Hobson A. C., Gho D., Müller-Hill B. Isolation, genetic analysis, and characterization of Escherichia coli mutants with defects in the lacY gene. J Bacteriol. 1977 Sep;131(3):830–838. doi: 10.1128/jb.131.3.830-838.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hofnung M. Divergent operons and the genetic structure of the maltose B region in Escherichia coli K12. Genetics. 1974 Feb;76(2):169–184. doi: 10.1093/genetics/76.2.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hogg R. W., Englesberg E. L-arabinose binding protein from Escherichia coli B-r. J Bacteriol. 1969 Oct;100(1):423–432. doi: 10.1128/jb.100.1.423-432.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong J. S. An ecf mutation in Escherichia coli pleiotropically affecting energy coupling in active transport but not generation or maintenance of membrane potential. J Biol Chem. 1977 Dec 10;252(23):8582–8588. [PubMed] [Google Scholar]
- Hong J. S., Hunt A. G., Masters P. S., Lieberman M. A. Requirements of acetyl phosphate for the binding protein-dependent transport systems in Escherichia coli. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1213–1217. doi: 10.1073/pnas.76.3.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsie A. W., Rickenberg H. V., Schulz D. W., Kirsch W. M. Steady-state concentrations of glucose-6-phosphate, 6-phosphogluconate, and reduced nicotinamide adenine dinucleotide phosphate in strains of Escherichia coli sensitive and resistant to catabolite repression. J Bacteriol. 1969 Jun;98(3):1407–1408. doi: 10.1128/jb.98.3.1407-1408.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hylemon P. B., Young J. L., Roadcap R. F., Phibbs P. V., Jr Uptake and incorporation of glucose and mannose by whole cells of Bacteroides thetaiotaomicron. Appl Environ Microbiol. 1977 Nov;34(5):488–494. doi: 10.1128/aem.34.5.488-494.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jacobson G. R., Lee C. A., Saier M. H., Jr Purification of the mannitol-specific enzyme II of the Escherichia coli phosphoenolpyruvate:sugar phosphotransferase system. J Biol Chem. 1979 Jan 25;254(2):249–252. [PubMed] [Google Scholar]
- KEPES A. [Kinetic studies on galactoside permease of Escherichia coli]. Biochim Biophys Acta. 1960 May 6;40:70–84. doi: 10.1016/0006-3002(60)91316-0. [DOI] [PubMed] [Google Scholar]
- KOCH A. L. THE ROLE OF PERMEASE IN TRANSPORT. Biochim Biophys Acta. 1964 Jan 27;79:177–200. doi: 10.1016/0926-6577(64)90050-6. [DOI] [PubMed] [Google Scholar]
- KUNDIG W., GHOSH S., ROSEMAN S. PHOSPHATE BOUND TO HISTIDINE IN A PROTEIN AS AN INTERMEDIATE IN A NOVEL PHOSPHO-TRANSFERASE SYSTEM. Proc Natl Acad Sci U S A. 1964 Oct;52:1067–1074. doi: 10.1073/pnas.52.4.1067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaback H. R. Molecular biology and energetics of membrane transport. J Cell Physiol. 1976 Dec;89(4):575–593. doi: 10.1002/jcp.1040890414. [DOI] [PubMed] [Google Scholar]
- Kaczorowski G. J., Kaback H. R. Mechanism of lactose translocation in membrane vesicles from Escherichia coli. 1. Effect of pH on efflux, exchange, and counterflow. Biochemistry. 1979 Aug 21;18(17):3691–3697. doi: 10.1021/bi00584a009. [DOI] [PubMed] [Google Scholar]
- Kaczorowski G. J., Robertson D. E., Kaback H. R. Mechanism of lactose translocation in membrane vesicles from Escherichia coli. 2. Effect of imposed delata psi, delta pH, and Delta mu H+. Biochemistry. 1979 Aug 21;18(17):3697–3704. doi: 10.1021/bi00584a010. [DOI] [PubMed] [Google Scholar]
- Kadner R. J. Genetic Control of the Transport of Hexose Phosphates in Escherichia coli: Mapping of the uhp Locus. J Bacteriol. 1973 Nov;116(2):764–770. doi: 10.1128/jb.116.2.764-770.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadner R. J., Winkler H. H. Isolation and characterization of mutations affecting the transport of hexose phosphates in Escherichia coli. J Bacteriol. 1973 Feb;113(2):895–900. doi: 10.1128/jb.113.2.895-900.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kay W. W., Cameron M. Citrate transport in Salmonella typhimurium. Arch Biochem Biophys. 1978 Sep;190(1):270–280. doi: 10.1016/0003-9861(78)90276-x. [DOI] [PubMed] [Google Scholar]
- Kellermann O., Szmelcman S. Active transport of maltose in Escherichia coli K12. Involvement of a "periplasmic" maltose binding protein. Eur J Biochem. 1974 Aug 15;47(1):139–149. doi: 10.1111/j.1432-1033.1974.tb03677.x. [DOI] [PubMed] [Google Scholar]
- Kennedy E. P., Rumley M. K., Armstrong J. B. Dierect measurement of the binding of labeled sugars to the lactose permease M protein. J Biol Chem. 1974 Jan 10;249(1):33–37. [PubMed] [Google Scholar]
- Klein W. L., Boyer P. D. Energization of active transport by Escherichia coli. J Biol Chem. 1972 Nov 25;247(22):7257–7265. [PubMed] [Google Scholar]
- Kobayashi H., Kin E., Anraku Y. Transport of sugars and amino acids in bacteria. X. Sources of energy and energy coupling reactions of the active transport systems for isoleucine and proline in E. coli. J Biochem. 1974 Aug;76(2):251–261. doi: 10.1093/oxfordjournals.jbchem.a130567. [DOI] [PubMed] [Google Scholar]
- Kornberg H. L. Fine control of sugar uptake by Escherichia coli. Symp Soc Exp Biol. 1973;27:175–193. [PubMed] [Google Scholar]
- Kornberg H. L., Riordan C. Uptake of galactose into Escherichia coli by facilitated diffusion. J Gen Microbiol. 1976 May;94(1):75–89. doi: 10.1099/00221287-94-1-75. [DOI] [PubMed] [Google Scholar]
- Korte T., Hengstenberg W. Purification and characterization of the inducible lactose-specific membrane-bound component of the staphylococcal phosphenolpyruvate-dependent phosphotransferase system. Eur J Biochem. 1971 Nov 11;23(2):295–302. doi: 10.1111/j.1432-1033.1971.tb01621.x. [DOI] [PubMed] [Google Scholar]
- Kozlov I. A., Skulachev V. P. H+-Adenosine triphosphatase and membrane energy coupling. Biochim Biophys Acta. 1977 Jun 21;463(1):29–89. doi: 10.1016/0304-4173(77)90003-9. [DOI] [PubMed] [Google Scholar]
- Kundig W. Molecular interactions in the bacterial phosphoenolpyruvate-phosphotransferase system (PTS). J Supramol Struct. 1974;2(5-6):695–814. doi: 10.1002/jss.400020514. [DOI] [PubMed] [Google Scholar]
- Kundig W., Roseman S. Sugar transport. I. Isolation of a phosphotransferase system from Escherichia coli. J Biol Chem. 1971 Mar 10;246(5):1393–1406. [PubMed] [Google Scholar]
- Lancaster J. R., Jr, Hill R. J., Struve W. G. The characterization of energized and partially de-energized (respiration-independent) beta-galactoside transport into Escherichia coli. Biochim Biophys Acta. 1975 Aug 20;401(2):285–298. doi: 10.1016/0005-2736(75)90312-0. [DOI] [PubMed] [Google Scholar]
- Lancaster J. R., Jr, Hinkle P. C. Studies of the beta-galactoside transporter in inverted membrane vesicles of Escherichia coli. II. Symmetrical binding of a dansylgalactoside induced by an electrochemical proton gradient and by lactose efflux. J Biol Chem. 1977 Nov 10;252(21):7662–7666. [PubMed] [Google Scholar]
- Langridge J. Characterization and intragenic position of mutations in the gene for galactoside permease of Escherichia coli. Aust J Biol Sci. 1974 Jun;27(3):331–340. doi: 10.1071/bi9740331. [DOI] [PubMed] [Google Scholar]
- Lanyi J. K. Coupling of aspartate and serine transport to the transmembrane electrochemical gradient for sodium ions in Halobacterium halobium. Translocation stoichiometries and apparent cooperativity. Biochemistry. 1978 Jul 25;17(15):3011–3018. doi: 10.1021/bi00608a012. [DOI] [PubMed] [Google Scholar]
- Lanyi J. K. Light energy conversion in Halobacterium halobium. Microbiol Rev. 1978 Dec;42(4):682–706. doi: 10.1128/mr.42.4.682-706.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levinson S. L., Krulwich T. A. Metabolism of L-rhamnose in Arthrobacter pyridinolis. J Gen Microbiol. 1976 Aug;96(2):277–286. doi: 10.1099/00221287-95-2-277. [DOI] [PubMed] [Google Scholar]
- Lieberman M. A., Hong J. S. A mutant of Escherichia coli defective in the coupling of metabolic energy to active transport. Proc Natl Acad Sci U S A. 1974 Nov;71(11):4395–4399. doi: 10.1073/pnas.71.11.4395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lieberman M. A., Hong J. S. Energization of osmotic shock-sensitive transport systems in Escherichia coli requires more than ATP. Arch Biochem Biophys. 1976 Jan;172(1):312–315. doi: 10.1016/0003-9861(76)90080-1. [DOI] [PubMed] [Google Scholar]
- Lieberman M. A., Simon M., Hong J. S. Characterization of Escherichia coli mutant incapable of maintaining a transmembrane potential. MetC ecfts mutations. J Biol Chem. 1977 Jun 25;252(12):4056–4067. [PubMed] [Google Scholar]
- Lin E. C. Glycerol dissimilation and its regulation in bacteria. Annu Rev Microbiol. 1976;30:535–578. doi: 10.1146/annurev.mi.30.100176.002535. [DOI] [PubMed] [Google Scholar]
- Lo T. C. The molecular mechanism of dicarboxylic acid transport in Escherichia coli K 12. J Supramol Struct. 1977;7(3-4):463–480. doi: 10.1002/jss.400070316. [DOI] [PubMed] [Google Scholar]
- London J., Chace N. M. New pathway for the metabolism of pentitols. Proc Natl Acad Sci U S A. 1977 Oct;74(10):4296–4300. doi: 10.1073/pnas.74.10.4296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lopilato J., Tsuchiya T., Wilson T. H. Role of Na+ and Li+ in thiomethylgalactoside transport by the melibiose transport system of Escherichia coli. J Bacteriol. 1978 Apr;134(1):147–156. doi: 10.1128/jb.134.1.147-156.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MAKMAN R. S., SUTHERLAND E. W. ADENOSINE 3',5'-PHOSPHATE IN ESCHERICHIA COLI. J Biol Chem. 1965 Mar;240:1309–1314. [PubMed] [Google Scholar]
- MITCHELL P. Coupling of phosphorylation to electron and hydrogen transfer by a chemi-osmotic type of mechanism. Nature. 1961 Jul 8;191:144–148. doi: 10.1038/191144a0. [DOI] [PubMed] [Google Scholar]
- MITCHELL P. Metabolism, transport, and morphogenesis: which drives which? J Gen Microbiol. 1962 Sep;29:25–37. doi: 10.1099/00221287-29-1-25. [DOI] [PubMed] [Google Scholar]
- MacDonald R. E., Greene R. V., Lanyi J. K. Light-activated amino acid transport systems in Halobacterium halobium envelope vesicles: role of chemical and electrical gradients. Biochemistry. 1977 Jul 12;16(14):3227–3235. doi: 10.1021/bi00633a029. [DOI] [PubMed] [Google Scholar]
- Maloney P. C. Obligatory coupling between proton entry and the synthesis of adenosine 5'-triphosphate in Streptococcus lactis. J Bacteriol. 1977 Nov;132(2):564–575. doi: 10.1128/jb.132.2.564-575.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maloney P. C., Wilson T. H. Metabolic control of lactose entry in Escherichia coli. Biochim Biophys Acta. 1978 Aug 17;511(3):487–498. doi: 10.1016/0005-2736(78)90283-3. [DOI] [PubMed] [Google Scholar]
- Maryanski J. H., Wittenberger C. L. Mannitol transport in Streptococcus mutans. J Bacteriol. 1975 Dec;124(3):1475–1481. doi: 10.1128/jb.124.3.1475-1481.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGinnis J. F., Paigen K. Catabolite inhibition: a general phenomenon in the control of carbohydrate utilization. J Bacteriol. 1969 Nov;100(2):902–913. doi: 10.1128/jb.100.2.902-913.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGinnis J. F., Paigen K. Site of catabolite inhibition of carbohydrate metabolism. J Bacteriol. 1973 May;114(2):885–887. doi: 10.1128/jb.114.2.885-887.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKay L. L., Walter L. A., Sandine W. E., Elliker P. R. Involvement of phosphoenolpyruvate in lactose utilization by group N streptococci. J Bacteriol. 1969 Aug;99(2):603–610. doi: 10.1128/jb.99.2.603-610.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell P. Chemiosmotic coupling in oxidative and photosynthetic phosphorylation. Biol Rev Camb Philos Soc. 1966 Aug;41(3):445–502. doi: 10.1111/j.1469-185x.1966.tb01501.x. [DOI] [PubMed] [Google Scholar]
- Mitchell P., Moyle J. Acid-base titration across the membrane system of rat-liver mitochondria. Catalysis by uncouplers. Biochem J. 1967 Aug;104(2):588–600. doi: 10.1042/bj1040588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell P., Moyle J. Respiration-driven proton translocation in rat liver mitochondria. Biochem J. 1967 Dec;105(3):1147–1162. doi: 10.1042/bj1051147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchell P. Translocations through natural membranes. Adv Enzymol Relat Areas Mol Biol. 1967;29:33–87. doi: 10.1002/9780470122747.ch2. [DOI] [PubMed] [Google Scholar]
- Neu H. C., Heppel L. A. The release of enzymes from Escherichia coli by osmotic shock and during the formation of spheroplasts. J Biol Chem. 1965 Sep;240(9):3685–3692. [PubMed] [Google Scholar]
- Overath P., Teather R. M., Simoni R. D., Aichele G., Wilhelm U. Lactose carrier protein of Escherichia coli. Transport and binding of 2'-(N-dansyl)aminoethyl beta-D-thiogalactopyranoside and p-nitrophenyl alpha-d-galactopyranoside. Biochemistry. 1979 Jan 9;18(1):1–11. doi: 10.1021/bi00568a001. [DOI] [PubMed] [Google Scholar]
- Parnes J. R., Boos W. Energy coupling of the -methylgalactoside transport system of Escherichia coli. J Biol Chem. 1973 Jun 25;248(12):4429–4435. [PubMed] [Google Scholar]
- Pastan I., Adhya S. Cyclic adenosine 5'-monophosphate in Escherichia coli. Bacteriol Rev. 1976 Sep;40(3):527–551. doi: 10.1128/br.40.3.527-551.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pastan I., Perlman R. L. Repression of beta-galactosidase synthesis by glucose in phosphotransferase mutants of Escherichia coli. Repression in the absence of glucose phosphorylation. J Biol Chem. 1969 Nov 10;244(21):5836–5842. [PubMed] [Google Scholar]
- Patni N. J., Alexander J. K. Catabolism of fructose and mannitol in Clostridium thermocellum: presence of phosphoenolpyruvate: fructose phosphotransferase, fructose 1-phosphate kinase, phosphoenolpyruvate: mannitol phosphotransferase, and mannitol 1-phosphate dehydrogenase in cell extracts. J Bacteriol. 1971 Jan;105(1):226–231. doi: 10.1128/jb.105.1.226-231.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plate C. A., Suit J. L., Jetten A. M., Luria S. E. Effects of colicin K on a mutant of Escherichia coli deficient in Ca 2+, Mg 2+-activated adenosine triphosphatase. J Biol Chem. 1974 Oct 10;249(19):6138–6143. [PubMed] [Google Scholar]
- Postma P. W., Roseman S. The bacterial phosphoenolpyruvate: sugar phosphotransferase system. Biochim Biophys Acta. 1976 Dec 14;457(3-4):213–257. doi: 10.1016/0304-4157(76)90001-0. [DOI] [PubMed] [Google Scholar]
- Ramos S., Kaback H. R. The electrochemical proton gradient in Escherichia coli membrane vesicles. Biochemistry. 1977 Mar 8;16(5):848–854. doi: 10.1021/bi00624a006. [DOI] [PubMed] [Google Scholar]
- Ramos S., Kaback H. R. The relationship between the electrochemical proton gradient and active transport in Escherichia coli membrane vesicles. Biochemistry. 1977 Mar 8;16(5):854–859. doi: 10.1021/bi00624a007. [DOI] [PubMed] [Google Scholar]
- Ramos S., Kaback H. R. pH-dependent changes in proton:substrate stoichiometries during active transport in Escherichia coli membrane vesicles. Biochemistry. 1977 Sep 20;16(19):4270–4275. doi: 10.1021/bi00638a022. [DOI] [PubMed] [Google Scholar]
- Randall-Hazelbauer L., Schwartz M. Isolation of the bacteriophage lambda receptor from Escherichia coli. J Bacteriol. 1973 Dec;116(3):1436–1446. doi: 10.1128/jb.116.3.1436-1446.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves R. E., Warren L. G., Hsu D. S. 1-Phosphofructokinase from an anaerobe. J Biol Chem. 1966 Mar 25;241(6):1257–1261. [PubMed] [Google Scholar]
- Reider E., Wagner E. F., Schweiger M. Control of phosphoenolpyruvate-dependent phosphotransferase-mediated sugar transport in Escherichia coli by energization of the cell membrane. Proc Natl Acad Sci U S A. 1979 Nov;76(11):5529–5533. doi: 10.1073/pnas.76.11.5529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rephaeli A. W., Saier M. H., Jr Kinetic analyses of the sugar phosphate:sugar transphosphorylation reaction catalyzed by the glucose enzyme II complex of the bacterial phosphotransferase system. J Biol Chem. 1978 Nov 10;253(21):7595–7597. [PubMed] [Google Scholar]
- Rephaeli A. W., Saier M. H., Jr Regulation of genes coding for enzyme constituents of the bacterial phosphotransferase system. J Bacteriol. 1980 Feb;141(2):658–663. doi: 10.1128/jb.141.2.658-663.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richey D. P., Lin E. C. Importance of facilitated diffusion for effective utilization of glycerol by Escherichia coli. J Bacteriol. 1972 Nov;112(2):784–790. doi: 10.1128/jb.112.2.784-790.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robbie J. P., Wilson T. H. Transmembrane effects of beta-galactosides on thiomethyl-beta-galactoside transport in Escherichia coli. Biochim Biophys Acta. 1969 Mar 11;173(2):234–244. doi: 10.1016/0005-2736(69)90107-2. [DOI] [PubMed] [Google Scholar]
- Romano A. H., Eberhard S. J., Dingle S. L., McDowell T. D. Distribution of the phosphoenolpyruvate: glucose phosphotransferase system in bacteria. J Bacteriol. 1970 Nov;104(2):808–813. doi: 10.1128/jb.104.2.808-813.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudnick G., Schildiner S., Kaback H. R. Equilibrium between two forms of the lac carrier protein in energized and nonenergized membrane vesicles from Escherichia coli. Biochemistry. 1976 Nov 16;15(23):5126–5131. doi: 10.1021/bi00668a028. [DOI] [PubMed] [Google Scholar]
- Saheb S. A. Perméation du glycérol et sporulation chez Bacillus subtilis. Can J Microbiol. 1972 Aug;18(8):1307–1313. [PubMed] [Google Scholar]
- Saier M. H., Jr Bacterial phosphoenolpyruvate: sugar phosphotransferase systems: structural, functional, and evolutionary interrelationships. Bacteriol Rev. 1977 Dec;41(4):856–871. doi: 10.1128/br.41.4.856-871.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U. Coordinate regulation of adenylate cyclase and carbohydrate permeases by the phosphoenolpyruvate:sugar phosphotransferase system in Salmonella typhimurium. J Biol Chem. 1975 Sep 10;250(17):7078–7080. [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U., Hofstadter L. J. Regulation of carbohydrate uptake and adenylate cyclase activity mediated by the enzymes II of the phosphoenolpyruvate: sugar phosphotransferase system in Escherichia coli. J Biol Chem. 1976 Feb 10;251(3):883–892. [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U., McCaman M. T. Regulation of intracellular adenosine cyclic 3':5'-monophosphate levels in Escherichia coli and Salmonella typhimurium. Evidence for energy-dependent excretion of the cyclic nucleotide. J Biol Chem. 1975 Oct 10;250(19):7593–7601. [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U. Regulation of carbohydrate transport activities in Salmonella typhimurium: use of the phosphoglycerate transport system to energize solute uptake. J Bacteriol. 1980 Feb;141(2):611–617. doi: 10.1128/jb.141.2.611-617.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U., Roseman S. Phosphoenolpyruvate-dependent fructose phosphorylation in photosynthetic bacteria. J Biol Chem. 1971 Dec 25;246(24):7819–7821. [PubMed] [Google Scholar]
- Saier M. H., Jr, Newman M. J. Direct transfer of the phosphoryl moiety of mannitol 1-phosphate to [14C]mannitol catalyzed by the enzyme II complexes of the phosphoenolpyruvate: mannitol phosphotransferase systems in Spirochaeta aurantia and Salmonella typhimurium. J Biol Chem. 1976 Jun 25;251(12):3834–3837. [PubMed] [Google Scholar]
- Saier M. H., Jr, Roseman S. Sugar transport. The crr mutation: its effect on repression of enzyme synthesis. J Biol Chem. 1976 Nov 10;251(21):6598–6605. [PubMed] [Google Scholar]
- Saier M. H., Jr, Simoni R. D. Regulation of carbohydrate uptake in gram-positive bacteria. J Biol Chem. 1976 Feb 10;251(3):893–894. [PubMed] [Google Scholar]
- Saier M. H., Jr, Simoni R. D., Roseman S. The physiological behavior of enzyme I and heat-stable protein mutants of a bacterial phosphotransferase system. J Biol Chem. 1970 Nov 10;245(21):5870–5873. [PubMed] [Google Scholar]
- Saier M. H., Jr, Staley J. T. Phosphoenolpyruvate:sugar phosphotransferase system in Ancalomicrobium adetum. J Bacteriol. 1977 Aug;131(2):716–718. doi: 10.1128/jb.131.2.716-718.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr, Straud H., Massman L. S., Judice J. J., Newman M. J., Feucht B. U. Permease-specific mutations in Salmonella typhimurium and Escherichia coli that release the glycerol, maltose, melibiose, and lactose transport systems from regulation by the phosphoenolpyruvate:sugar phosphotransferase system. J Bacteriol. 1978 Mar;133(3):1358–1367. doi: 10.1128/jb.133.3.1358-1367.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr, Wentzel D. L., Feucht B. U., Judice J. J. A transport system for phosphoenolpyruvate, 2-phosphoglycerate, and 3-phosphoglycerate in Salmonella typhimurium. J Biol Chem. 1975 Jul 10;250(13):5089–5096. [PubMed] [Google Scholar]
- Sanno Y., Wilson T. H., Lin E. C. Control of permeation to glycerol in cells of Escherichia coli. Biochem Biophys Res Commun. 1968 Jul 26;32(2):344–349. doi: 10.1016/0006-291x(68)90392-6. [DOI] [PubMed] [Google Scholar]
- Sawyer M. H., Baumann P., Baumann L., Berman S. M., Cánovas J. L., Berman R. H. Pathways of D-fructose catabolism in species of Pseudomonas. Arch Microbiol. 1977 Feb 4;112(1):49–55. doi: 10.1007/BF00446653. [DOI] [PubMed] [Google Scholar]
- Sawyer M. H., Baumann P., Baumann L. Pathways of D-fructose and D-glucose catabolism in marine species of Alcaligenes, Pseudomonas marina, and Alteromonas communis. Arch Microbiol. 1977 Mar 1;112(2):169–172. doi: 10.1007/BF00429331. [DOI] [PubMed] [Google Scholar]
- Schachtele C. F., Mayo J. A. Phosphoenolpyruvate-dependent glucose transport in oral streptococci. J Dent Res. 1973 Nov-Dec;52(6):1209–1215. doi: 10.1177/00220345730520060801. [DOI] [PubMed] [Google Scholar]
- Schindler H., Rosenbusch J. P. Matrix protein from Escherichia coli outer membranes forms voltage-controlled channels in lipid bilayers. Proc Natl Acad Sci U S A. 1978 Aug;75(8):3751–3755. doi: 10.1073/pnas.75.8.3751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schleif R. An L-arabinose binding protein and arabinose permeation in Escherichia coli. J Mol Biol. 1969 Nov 28;46(1):185–196. doi: 10.1016/0022-2836(69)90065-5. [DOI] [PubMed] [Google Scholar]
- Schuldiner S., Kaback H. R. Membrane potential and active transport in membrane vesicles from Escherichia coli. Biochemistry. 1975 Dec 16;14(25):5451–5461. doi: 10.1021/bi00696a011. [DOI] [PubMed] [Google Scholar]
- Schultz S. G., Curran P. F. Coupled transport of sodium and organic solutes. Physiol Rev. 1970 Oct;50(4):637–718. doi: 10.1152/physrev.1970.50.4.637. [DOI] [PubMed] [Google Scholar]
- Seals J. R., Jarett L. Activation of pyruvate dehydrogenase by direct addition of insulin to an isolated plasma membrane/mitochondria mixture: evidence for generated of insulin's second messenger in a subcellular system. Proc Natl Acad Sci U S A. 1980 Jan;77(1):77–81. doi: 10.1073/pnas.77.1.77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shamanna D. K., Sanderson K. E. Genetics and regulation of D-xylose utilization in Salmonella typhimurium LT2. J Bacteriol. 1979 Jul;139(1):71–79. doi: 10.1128/jb.139.1.71-79.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shamanna D. K., Sanderson K. E. Uptake and catabolism of D-xylose in Salmonella typhimurium LT2. J Bacteriol. 1979 Jul;139(1):64–70. doi: 10.1128/jb.139.1.64-70.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silhavy T. J., Szmelcman S., Boos W., Schwartz M. On the significance of the retention of ligand by protein. Proc Natl Acad Sci U S A. 1975 Jun;72(6):2120–2124. doi: 10.1073/pnas.72.6.2120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silva D. O., Dobrogosz W. J. Proton efflux associated with melibiose permease activity in Salmonella typhimurium. Biochem Biophys Res Commun. 1978 Apr 14;81(3):750–755. doi: 10.1016/0006-291x(78)91415-8. [DOI] [PubMed] [Google Scholar]
- Simoni R. D., Postma P. W. The energetics of bacterial active transport. Annu Rev Biochem. 1975;44:523–554. doi: 10.1146/annurev.bi.44.070175.002515. [DOI] [PubMed] [Google Scholar]
- Sobel M. E., Krulwich T. A. Metabolism of D-fructose by Arthrobacter pyridinolis. J Bacteriol. 1973 Feb;113(2):907–913. doi: 10.1128/jb.113.2.907-913.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprott G. D., Drozdowski J. P., Martin E. L., MacLeod R. A. Kinetics of Naplus-dependent amino acid transport using cells and membrane vesicles of a marine pseudomonad. Can J Microbiol. 1975 Jan;21(1):43–50. doi: 10.1139/m75-006. [DOI] [PubMed] [Google Scholar]
- Stock J., Roseman S. A sodium-dependent sugar co-transport system in bacteria. Biochem Biophys Res Commun. 1971 Jul 2;44(1):132–138. doi: 10.1016/s0006-291x(71)80168-7. [DOI] [PubMed] [Google Scholar]
- Szmelcman S., Hofnung M. Maltose transport in Escherichia coli K-12: involvement of the bacteriophage lambda receptor. J Bacteriol. 1975 Oct;124(1):112–118. doi: 10.1128/jb.124.1.112-118.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szmelcman S., Schwartz M., Silhavy T. J., Boos W. Maltose transport in Escherichia coli K12. A comparison of transport kinetics in wild-type and lambda-resistant mutants as measured by fluorescence quenching. Eur J Biochem. 1976 May 17;65(1):13–19. doi: 10.1111/j.1432-1033.1976.tb10383.x. [DOI] [PubMed] [Google Scholar]
- Tanaka S., Lin E. C. Two classes of pleiotropic mutants of Aerobacter aerogenes lacking components of a phosphoenolpyruvate-dependent phosphotransferase system. Proc Natl Acad Sci U S A. 1967 Apr;57(4):913–919. doi: 10.1073/pnas.57.4.913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson J., Turner K. W., Thomas T. D. Catabolite inhibition and sequential metabolism of sugars by Streptococcus lactis. J Bacteriol. 1978 Mar;133(3):1163–1174. doi: 10.1128/jb.133.3.1163-1174.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokuda H., Kaback H. R. Sodium-dependent binding of p-nitrophenyl alpha-D-galactopyranoside to membrane vesicles isolated from Salmonella typhimurium. Biochemistry. 1978 Feb 21;17(4):698–705. doi: 10.1021/bi00597a022. [DOI] [PubMed] [Google Scholar]
- Tokuda H., Kaback H. R. Sodium-dependent methyl 1-thio-beta-D-galactopyranoside transport in membrane vesicles isolated from Salmonella typhimurium. Biochemistry. 1977 May 17;16(10):2130–2136. doi: 10.1021/bi00629a013. [DOI] [PubMed] [Google Scholar]
- Tomchika K. I., Hong J. S. Transport-defective Escherichia coli ecf mutant permeable to protons and nucleotides. J Bacteriol. 1978 Feb;133(2):1008–1014. doi: 10.1128/jb.133.2.1008-1014.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsay S. S., Brown K. K., Gaudy E. T. Transport of glycerol by Pseudomonas aeruginosa. J Bacteriol. 1971 Oct;108(1):82–88. doi: 10.1128/jb.108.1.82-88.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuchiya T., Raven J., Wilson T. H. Co-transport of Na+ and methul-beta-D-thiogalactopyranoside mediated by the melibiose transport system of Escherichia coli. Biochem Biophys Res Commun. 1977 May 9;76(1):26–31. doi: 10.1016/0006-291x(77)91663-1. [DOI] [PubMed] [Google Scholar]
- Van Dijken J. P., Quayle J. R. Fructose metabolism in four Pseudomonas species. Arch Microbiol. 1977 Sep 28;114(3):281–286. doi: 10.1007/BF00446874. [DOI] [PubMed] [Google Scholar]
- Villarejo M., Ping C. Localization of the lactose permease protein(s) in the E. coli envelope. Biochem Biophys Res Commun. 1978 Jun 14;82(3):935–942. doi: 10.1016/0006-291x(78)90873-2. [DOI] [PubMed] [Google Scholar]
- West I. C. Lactose transport coupled to proton movements in Escherichia coli. Biochem Biophys Res Commun. 1970 Nov 9;41(3):655–661. doi: 10.1016/0006-291x(70)90063-x. [DOI] [PubMed] [Google Scholar]
- West I. C., Mitchell P. Stoicheiometry of lactose-H+ symport across the plasma membrane of Escherichia coli. Biochem J. 1973 Mar;132(3):587–592. doi: 10.1042/bj1320587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willecke K., Pardee A. B. Inducible transport of citrate in a Gram-positive bacterium, Bacillus subtilis. J Biol Chem. 1971 Feb 25;246(4):1032–1040. [PubMed] [Google Scholar]
- Willecke K., Pardee A. B. Inducible transport of citrate in a Gram-positive bacterium, Bacillus subtilis. J Biol Chem. 1971 Feb 25;246(4):1032–1040. [PubMed] [Google Scholar]
- Willis R. C., Furlong C. E. Purification and properties of a ribose-binding protein from Escherichia coli. J Biol Chem. 1974 Nov 10;249(21):6926–6929. [PubMed] [Google Scholar]
- Wilson D. B. Cellular transport mechanisms. Annu Rev Biochem. 1978;47:933–965. doi: 10.1146/annurev.bi.47.070178.004441. [DOI] [PubMed] [Google Scholar]
- Wilson D. B. Properties of the entry and exit reactions of the beta-methyl galactoside transport system in Escherichia coli. J Bacteriol. 1976 Jun;126(3):1156–1165. doi: 10.1128/jb.126.3.1156-1165.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson D. B. Source of energy for the Escherichia coli galactose transport systems induced by galactose. J Bacteriol. 1974 Nov;120(2):866–871. doi: 10.1128/jb.120.2.866-871.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson D. M., Alderette J. F., Maloney P. C., Wilson T. H. Protonmotive force as the source of energy for adenosine 5'-triphosphate synthesis in Escherichia coli. J Bacteriol. 1976 Apr;126(1):327–337. doi: 10.1128/jb.126.1.327-337.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson T. H., Kusch M. A mutant of Escherichia coli K 12 energy-uncoupled for lactose transport. Biochim Biophys Acta. 1972 Mar 17;255(3):786–797. doi: 10.1016/0005-2736(72)90391-4. [DOI] [PubMed] [Google Scholar]
- Wilson T. H., Kusch M., Kashket E. R. A mutant in Escherichia coli energy-uncoupled for lactose transporta defect in the lactose-operon. Biochem Biophys Res Commun. 1970 Sep 30;40(6):1409–1414. doi: 10.1016/0006-291x(70)90024-0. [DOI] [PubMed] [Google Scholar]
- Winkler H. H. A hexose-phosphate transport system in Escherichia coli. Biochim Biophys Acta. 1966 Mar 28;117(1):231–240. doi: 10.1016/0304-4165(66)90170-x. [DOI] [PubMed] [Google Scholar]
- Winkler H. H. A hexose-phosphate transport system in Escherichia coli. Biochim Biophys Acta. 1966 Mar 28;117(1):231–240. doi: 10.1016/0304-4165(66)90170-x. [DOI] [PubMed] [Google Scholar]
- Winkler H. H. Compartmentation in the induction of the hexose-6-phosphate transport system of Escherichia coli. J Bacteriol. 1970 Feb;101(2):470–475. doi: 10.1128/jb.101.2.470-475.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler H. H. Energy coupling of the hexose phosphate transport system in Escherichia coli. J Bacteriol. 1973 Oct;116(1):203–209. doi: 10.1128/jb.116.1.203-209.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler H. H. Kinetics of exogenous induction of the hexose-6-phosphate transport system of Escherichia coli. J Bacteriol. 1971 Jul;107(1):74–78. doi: 10.1128/jb.107.1.74-78.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler H. H., Wilson T. H. The role of energy coupling in the transport of beta-galactosides by Escherichia coli. J Biol Chem. 1966 May 25;241(10):2200–2211. [PubMed] [Google Scholar]
- Zilberstein D., Schuldiner S., Padan E. Proton electrochemical gradient in Escherichia coli cells and its relation to active transport of lactose. Biochemistry. 1979 Feb 20;18(4):669–673. doi: 10.1021/bi00571a018. [DOI] [PubMed] [Google Scholar]
- van Thienen G. M., Postma P. W., van Dam K. Na+-dependent methyl beta-thiogalactoside transport in Salmonella typhimurium. Biochim Biophys Acta. 1978 Nov 16;513(3):395–400. doi: 10.1016/0005-2736(78)90207-9. [DOI] [PubMed] [Google Scholar]
- von Hugo H., Gottschalk G. Distribution of 1-phosphofructokinase and PEP:fructose phosphotransferase activity in Clostridia. FEBS Lett. 1974 Sep 15;46(1):106–108. doi: 10.1016/0014-5793(74)80345-5. [DOI] [PubMed] [Google Scholar]



