Summary
Background and objectives
Poor physical performance is common in patients with kidney failure on dialysis (CKD-5D). Whether lung congestion, a predictable consequence of cardiomyopathy and fluid overload, may contribute to the low physical performance of CKD-5D patients has not been investigated in hemodialysis patients.
Design, setting, participants, & measurements
This study investigated the relationship between the physical functioning scale of the Kidney Disease Quality of Life Short Form and a validated ultrasonographic measure of lung water in a multicenter survey of 270 hemodialysis patients studied between 2009 and 2010.
Results
Moderate to severe lung congestion by lung ultrasonography was observed in 156 (58%) patients; among these, 60 (38%) were asymptomatic (New York Heart Association [NYHA] class I). On univariate analysis, physical functioning was inversely associated with lung water in the whole group (r=−0.22; P<0.001) and in the subgroup of asymptomatic patients (r=−0.40; P=0.002). Age (r=−0.45; P<0.001) and past cardiovascular events (r=−0.22; P=0.002) were also inversely associated with physical functioning, whereas albumin (r=0.23; P<0.001) was directly associated with the same parameter. NYHA class correlated strongly with physical functioning (r=−0.52; P<0.001). In a multiple regression analysis, both NYHA class and lung water maintained an independent association with physical functioning, whereas albumin and background cardiovascular events failed to independently relate with the same outcome.
Conclusions
Symptomatic and asymptomatic lung congestion is associated with poor physical functioning in hemodialysis patients. This association is independent of NYHA, suggesting that this measurement and NYHA may have complementary value to explain the variability in physical performance in hemodialysis patients.
Introduction
Physical functioning, one of the most important dimensions of quality of life, is markedly compromised in patients with kidney failure on dialysis (stage CKD-5D) (1–3). Anemia, mineral and bone disorders, the inflammation-muscular-wasting complex, cardiomyopathy, neuropathy, and depression, all disturbances that frequently coexist in CKD-5D, may be involved in the poor physical functioning of this population (3–6). Pulmonary congestion detected and quantified by lung ultrasonography recently emerged as a powerful correlate of poor physical functioning in a multicenter study in peritoneal dialysis (PD) patients (7), a population with an exceedingly high prevalence of fluid overload and left ventricular dysfunction (8–10), suggesting that volume expansion and cardiomyopathy, two potentially reversible risk factors, play a major role in the poor physical performance of this population.
The dynamics of fluid removal (continuous versus intermittent) and the equilibrium between cardiopulmonary fluid compartments (stable versus variable) differ in hemodialysis and in PD; therefore, observations in PD patients do not necessarily apply to hemodialysis patients. To further explore this issue, we investigated the relationship between pulmonary congestion and physical functioning in a large multicenter survey in hemodialysis patients. Because fluid accumulation in the lung, per se or as an expression of cardiomyopathy, may lead to dyspnea, the association between pulmonary congestion and physical functioning was analyzed also considering the New York Heart Association (NYHA) classification, a scoring system of heart failure in which dyspnea represents a dominant component.
Materials and Methods
The study protocol was in conformity with the Declaration of Helsinki and was approved by the local ethics committee. Informed consent was obtained by all participants.
Study Population
All prevalent and incident patients on regular hemodialysis treatment between 2009 and 2010 in the dialysis units participating in this study were invited to enter into this survey. After exclusion of patients unable to complete the Kidney Disease Quality of Life Short Form (KDQOL-SF) (Rand Corporation, Santa Monica, CA) and those with previous amputations, severe neurologic deficit or orthopedic disease impairing physical activity, 270 patients, treated in 10 renal units (105 women, 165 men), were enrolled (Figure 1).
Figure 1.
Flow-diagram showing enrollment of hemodialysis patients. KDQOL-SF, Kidney Disease Quality of Life Short Form.
Physical Functioning Measurement
We used the subscale physical functioning of the KDQOL-SF, which was specifically validated on CKD patients in the Italian translation (11). This is a 10-question scale (items 3–12) that captures self-reported abilities to deal with the physical requirements of life, such as attending to personal needs and walking. The score of this scale can range from 100 (no limitation at all in activities) to 0 (limited a lot in activities). The median value of physical functioning (SF-36) in normative data of the Italian population, of age similar to that of our study sample, was 95. We adopted the cut-off value of 70 (corresponding to the 75th percentile of the physical functioning distribution in our study population) because this value approaches the 25th percentile of the same score in the healthy, Italian general population (12).
Lung Ultrasonography
Lung water was estimated by chest ultrasonography (13) before hemodialysis. We described in detail the technique and tested its validity in hemodialysis and PD patients (14,15). The rationale of the method is that in the presence of extravascular lung water, the ultrasound beam is reflected by subpleural interlobular septa thickened by lung edema. The reflection of the beam generates comet-tail reverberation artifacts, called ultrasound B lines (16). The sum of ultrasound B lines observed in well identified chest areas (see below) produces a score reflecting the extent of lung water accumulation (zero being no detectable ultrasound B lines).
As described (14), ultrasound scanning of the anterior and lateral chest was performed on the right and left hemi-thorax, from the second to the fourth (on the right side to the fifth) intercostal spaces, and from the parasternal to the axillary line. The severity of lung congestion was categorized in three classes, as described elsewhere (14), and patients with an ultrasound B line score between 15 and 30 and those with a score >30 were considered as having moderate and severe lung congestion, respectively. A detailed description of the technique is given in a YouTube film (http://www.youtube.com/watch?v=7y_hUFBHStM).
One operator was responsible for lung ultrasonography in each center. As described in detail elsewhere, the interoperator reproducibility of ultrasound B line determination in kidney failure patients on dialysis is excellent (14). All sonographers of participating units in this study were preliminarily trained at CNR-IBIM, Reggio Calabria, and the agreement of ultrasound B line measurements between them and the trainer sonographer was confirmed to be excellent (i.e., ±10% the reference measurements made by the sonographer-trainer). Ultrasound B lines and physical functioning were assessed in the same day and all sonographers were blinded to the clinical and laboratory data.
NYHA Scores
NYHA assessors were blind to detailed clinical information and were unaware of the ultrasound B line results. Categories of increasing severity in the NYHA classification show a progressively higher mortality risk in dialysis patients (17) and we documented a fairly good agreement between independent NYHA assessors in these patients (17).
Other Covariates Studied
We collected data on demographics (age, sex), treatment parameters [dialysis vintage, Kt/V estimated by the Daugirdas equation (18)], laboratory parameters (serum creatinine, calcium, phosphate, albumin, hemoglobin), body size as body mass index (BMI) (kg/m2), predialysis BP, and comorbidities including diabetes, chronic obstructive pulmonary disease, and previous cardiovascular events (electrocardiography-documented angina and myocardial infarction, stroke, transient ischemic attacks, heart failure, arrhythmias, or peripheral vascular disease). Treatment with antihypertensive and erythropoiesis-stimulating agents (ESAs) was also recorded. We also evaluated patients by the subscale mental health of the KDQOL-SF validated in the Italian translation (11). This five-question scale (items 24–26, 28, and 30) scores feelings pertaining to anxiety and depression and is considered as a useful screening instrument for depressive symptoms in dialysis patients (19). A score of 100 represents the best state, whereas 0 is the worst. Laboratory variables were measured by routine auto-analyzer methods at laboratories linked to each renal unit.
Statistical Analyses
Data are presented as the mean ± SD, median (interquartile range), or percent frequency and comparison between groups were made by t test, Mann–Whitney test, or chi-squared test, as appropriate. Variables that were not normally distributed were log transformed.
The independent association between ultrasound B lines and physical functioning by the KDQOL-SF was analyzed by simple and multiple linear regression analyses adjusting for covariates that were associated (P≤0.10) with physical functioning and/or ultrasound B lines at univariate analysis. In multiple linear regression analysis, multicollinearity was tested by investigating the variance inflation factor and tolerance. To further explore the risk for poor physical functioning (score <75 percentile) in relationship to lung congestion, we performed a logistic regression based on the same variables included in the multiple regression model of the same outcome variable. Data were expressed as standardized regression coefficients (β) and P values in the multiple linear regression analysis and as odds ratios, 95% confidence intervals, and P values in the multiple logistic regression analysis. All calculations were made using a standard statistical package (SPSS for Windows, version 9; SPSS Inc, Chicago, IL).
Results
The mean age of the study population was 65.9 years (SD 14.9), and median dialysis vintage was 51.2 months (interquartile range, 24–101). There were 235 patients on bicarbonate dialysis with standard or high-flux membranes, whereas 35 were being treated with hemodiafiltration. Of the patients, 248 were dialyzed thrice weekly, 19 were dialyzed four times per week, and 3 were dialyzed twice weekly. The cause of chronic renal disease was nephroangiosclerosis in 80, polycystic kidneys in 27, GN in 26, tubulointerstitial nephritis in 20, vasculitis in 3, hemolytic-uremic syndrome in 2, other diseases in 10, and unknown causes in 53 patients. Forty-nine patients had diabetic nephropathy but diabetes as a comorbidity was present in 25 additional patients.
The median KDQOL-SF physical functioning score was 40 (interquartile range, 15–70) and the vast majority of patients (Figure 2) scored <25th percentile of the distribution of the same score in the normal Italian population of similar age (12). Table 1 shows the main baseline characteristics of the patients divided into three groups on the basis of physical functioning tertiles. Patients in lower functional categories were older and more frequently were women; they had lower serum albumin, diastolic pressure, phosphate, and creatinine levels and more frequently had a history of diabetes, cardiovascular events, and chronic obstructive pulmonary disease.
Figure 2.
Bar graph of the physical functioning score distribution. The vertical arrow indicates the 75th percentile (corresponding to the 25th percentile of the distribution of the same score in the Italian general population) [see Apolone and Mosconi (12)].
Table 1.
Main demographic, clinical, and biochemical data of hemodialysis patients
| Variable | KDQOL-SF Score (Physical Functioning) | ||||
|---|---|---|---|---|---|
| I tertile <20 (n=91) | II tertile 20–55 (n=90) | III tertile >55 (n=89) | P | r (P) | |
| Age (yr) | 72±12 | 69±11 | 57±17 | <0.001a | −0.45 (<0.001)a |
| Male sex (%) | 46 | 71 | 66 | 0.001a | 0.15 (0.02)a |
| Diabetes (%) | 37 | 28 | 19 | 0.02a | −0.17 (0.01)a |
| Previous cardiovascular events (%)b | 57 | 40 | 32 | 0.002a | −0.22 (0.002)a |
| Dialysis vintage (mo) | 57 (24–108) | 51 (27–109) | 41 (20–82) | 0.18 | −0.06 (0.31) |
| COPD | 19 | 17 | 6 | 0.05a | −0.13 (0.04)a |
| Antihypertensive treatment (%) | 53 | 49 | 58 | 0.44 | 0.03 (0.67) |
| ESA treatment | 85 | 74 | 73 | 0.09 | −0.05 (0.41) |
| Predialysis systolic BP (mmHg) | 136±26 | 139±21 | 131±20 | 0.12 | −0.10 (0.12) |
| Predialysis diastolic BP (mmHg) | 69±12 | 75±11 | 73±13 | 0.04a | 0.13 (0.03)a |
| Body mass index (kg/m2) | 24.9±5.0 | 25.0±5.0 | 24.8±4.8 | 0.91 | −0.01 (0.93) |
| Serum creatinine (mg/dl) | 8.2±2.6 | 9.3±2.5 | 10.1±3.1 | <0.001a | 0.29 (<0.001)a |
| Hemoglobin (g/dl) | 11.1±1.2 | 11.5±1.4 | 11.3±1.4 | 0.19 | 0.03 (0.63) |
| Calcium (mg/dl) | 9.0±0.8 | 9.0±0.9 | 9.0±0.8 | 0.83 | 0.01 (0.84) |
| Phosphate (mg/dl) | 4.5±1.4 | 4.9±1.6 | 5.2±1.7 | 0.003a | 0.19 (0.002)a |
| Serum albumin (g/dl) | 3.8±0.4 | 4.0±0.3 | 4.0±0.4 | 0.001a | 0.23 (<0.001)a |
| Kt/V | 1.44±0.32 | 1.40±0.37 | 1.38±0.25 | 0.33 | −0.07 (0.36) |
| Predialysis ultrasound B lines (n) | 25 (13–55) | 18 (10–36) | 16 (8–30) | 0.10a | −0.22 (<0.001)a |
| NYHA class | <0.001a | −0.52 (<0.001)a | |||
| I | 15 | 8 | 46 | ||
| II | 28 | 62 | 48 | ||
| III | 36 | 27 | 6 | ||
| IV | 21 | 3 | 0 | ||
| Mental health score | 46±19 | 54±18 | 62±19 | <0.001a | 0.38 (<0.001)a |
Data are expressed as mean ± SD, median (interquartile range), or as percent frequency, as appropriate. Patients are divided into three groups on the basis of tertiles of KDQOL-SF physical functioning score. P tests the differences among the groups. r is the Pearson coefficient of correlation between KDQOL-SF physical functioning score and the variables. KDQOL-SF, Kidney Disease Quality of Life Short Form; COPD, chronic obstructive pulmonary disease; ESA, erythropoiesis-stimulating agent.
Indicates significant differences between groups and significant correlations.
Previous cardiovascular events such as electrocardiography-documented angina and myocardial infarction, stroke, transient ischemic attacks, heart failure, arrhythmias, or peripheral vascular disease.
Correlates of Physical Functioning
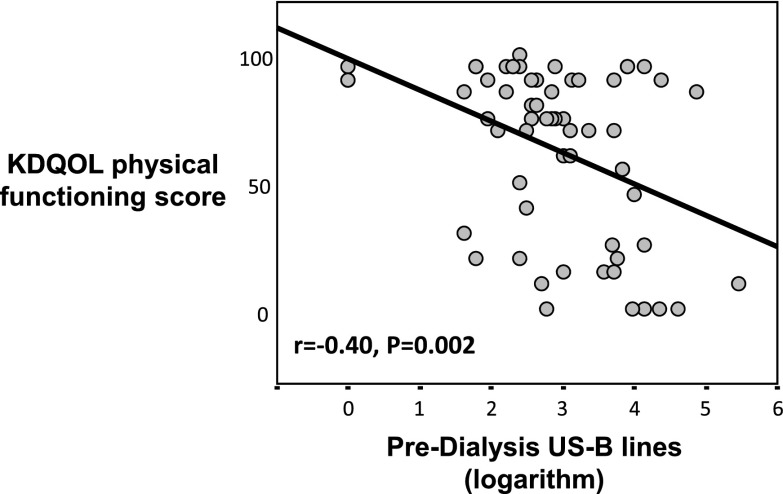
The number of ultrasound B lines was higher in patients with lower physical performance and this score correlated inversely (r=−0.22; P<0.001) with the same parameter (Table 1) and tended to be directly related to interdialysis weight gain (r=0.11; P=0.06). The association was even stronger (r=−0.40; P=0.002) in the subgroup of 60 asymptomatic patients (i.e., classified in NYHA class I) (Figure 3). Physical functioning was also inversely associated with age (r=−0.45; P<0.001), diabetes (r=−0.17; P=0.01), and background cardiovascular events (r=−0.22; P=0.002) and was directly associated with serum albumin (r=0.23; P<0.001), creatinine (r=0.29; P<0.001), phosphate (r=0.19; P=0.002), and diastolic BP (r=0.13; P=0.03) (Table 1). NYHA class (r=−0.52; P<0.001) and mental health (r=0.38; P<0.001) were strongly associated with physical functioning (Figure 4).
Figure 3.
Relationship between ultrasound B line score and physical functioning in patients scored as NYHA class I. NYHA, New York Heart Association; KDQOL-SF, Kidney Disease Quality of Life Short Form; USB, ultrasound B line.
Figure 4.
Median and interquartile range of physical functioning according to NYHA class and mental health score tertiles. The relationship between variables is also expressed as the correlation coefficient and P value. NYHA, New York Heart Association; KDQOL-SF, Kidney Disease Quality of Life Short Form.
Multivariate Analyses of Physical Functioning
To define the factors that explain the variability in physical functioning, we built a multiple regression model including all variables that were associated with physical functioning and/or with ultrasound B lines at univariate analysis. In this model, NYHA ranked as the strongest independent correlate of the outcome variable followed by mental health, age, and ultrasound B lines (Table 2). Forcing BMI into the model, as well as the full set of variables listed in Table 1, did not materially affect the strength of the relationship between ultrasound B lines and physical functioning. In the same model, BMI failed to be independently related to physical performance (P=0.42).
Table 2.
Multiple regression analysis of physical functioning
| Variable | β | P |
|---|---|---|
| NYHA class | −0.30 | <0.001 |
| Mental health | 0.26 | <0.001 |
| Age | −0.23 | <0.001 |
| Predialysis ultrasound B lines | −0.11 | 0.03 |
| Male sex | 0.08 | 0.15 |
| Serum albumin | 0.05 | 0.34 |
| Phosphate | 0.05 | 0.35 |
| Diabetes | −0.04 | 0.44 |
| Serum creatinine | 0.04 | 0.45 |
| COPD | −0.02 | 0.63 |
| Predialysis diastolic BP | −0.02 | 0.78 |
| Hemoglobin | −0.01 | 0.81 |
| ESA treatment | −0.01 | 0.91 |
| Previous cardiovascular events | −0.01 | 0.91 |
Data are expressed as standardized regression coefficient (β) and P value. NYHA, New York Heart Association; COPD, chronic obstructive pulmonary disease; ESA, erythropoiesis-stimulating agent.
Multivariate logistic regression analysis showed that for each 10-unit increase in ultrasound B lines, the probability of poor physical performance increased by 23% (odds ratio, 1.23; 95% confidence interval, 1.03 to 1.48; P=0.03) and the excess risk was again independent of NYHA class (Table 3).
Table 3.
Logistic regression analysis of physical functioning <75th percentile as the dependent variable
| Variable (Units of Increase) | Odds Ratio | 95% Confidence Interval | P |
|---|---|---|---|
| NYHA class (1 unit) | 2.76 | 1.48 to 5.13 | 0.001 |
| Age (1 yr) | 1.05 | 1.01 to 1.08 | 0.004 |
| Predialysis ultrasound B lines (10 units) | 1.23 | 1.03 to 1.48 | 0.03 |
| Mental health (1 unit) | 0.98 | 0.96 to 1.00 | 0.05 |
| Male sex | 0.46 | 0.19 to 1.09 | 0.08 |
| Predialysis diastolic BP (1 mmHg) | 1.03 | 0.99 to 1.07 | 0.09 |
| ESA treatmenta | 0.49 | 0.18 to 1.30 | 0.15 |
| COPDa | 2.55 | 0.53 to 12.21 | 0.24 |
| Phosphate (1 mg/dl) | 0.88 | 0.67 to 1.17 | 0.39 |
| Previous cardiovascular eventsa | 1.32 | 0.57 to 3.04 | 0.51 |
| Diabetesa | 1.14 | 0.56 to 2.87 | 0.78 |
| Serum albumin (1 g/dl) | 0.87 | 0.29 to 2.62 | 0.81 |
| Hemoglobin (1 g/dl) | 1.01 | 0.75 to 1.36 | 0.94 |
| Serum creatinine (1 mg/dl) | 1.00 | 0.87 to 1.16 | 0.95 |
NYHA, New York Heart Association; ESA, erythropoiesis-stimulating agent; COPD, chronic obstructive pulmonary disease.
0 = no; 1 = yes.
Discussion
This survey documents that lung water by chest ultrasonography (13–15) is associated with physical functioning in stage 5 CKD patients on hemodialysis treatment. Such an association is largely independent of other risk factors, including the severity of heart failure as assessed by NYHA, traditional, and CKD-related risk factors.
In keeping with previous surveys (1–3), we found that physical functioning is impaired in hemodialysis patients and that age, diabetes, and cardiovascular comorbidities represent the strongest correlates of poor physical functioning in this population. We also observed that nutrition biomarkers (namely, serum albumin, creatinine, and phosphate) were closely associated with physical functioning at univariate analysis but these parameters were no longer significantly related to this outcome variable in multiple regression analyses. Selective multivariate modeling showed that data adjustment by age was the critical factor abrogating the link between nutritional biomarkers and physical functioning, suggesting that this link is largely attributable to confounding by the aging process. Confirming observations in PD patients (7), physical performance was independent of hemoglobin, a parameter considered as a fundamental determinant of physical health. Such a lack of association most likely depends on the fact that anemia was adequately treated in most patients (81% of hemodialysis patients had hemoglobin ≥10.5 g/dl). NYHA classification was the strongest predictor of physical functioning score explaining more than a quarter (27%) of the variance in this parameter.
Pulmonary congestion frequently occurs in hemodialysis patients (14). This phenomenon more likely reflects impaired cardiac function than true volume overload because a previous study has demonstrated stronger correlations between ultrasound B lines and left ventricular systolic function than total body water as measured by body impedance analysis (14). The novel key finding of this study in hemodialysis patients is the independent association between physical functioning and pulmonary congestion as assessed by lung ultrasonography. Notably, this association was particularly strong in the subgroup of asymptomatic patients (classified as NYHA class I). Indeed, in this subgroup, 16% of the variance in physical functioning was explained by the simultaneous variability in lung water, suggesting that lung congestion contributes to physical impairment in apparently asymptomatic hemodialysis patients. Multivariate analysis attenuated but did not abolish the relationship between lung water and physical functioning and both lung water and NYHA class remained independent predictors of physical functioning, suggesting that the two scoring systems have complementary value to explain physical functioning in hemodialysis patients. Ultrasound B lines are tightly associated with fundamental functional parameters of the left ventricle such as ejection fraction, left atrial volume, and pulmonary pressure (13–15); therefore, the additional explanatory power of ultrasound B lines over and above the severity of dyspnea by NYHA likely depends on the fact that pulmonary congestion by this technique captures explanatory power conveyed by low ejection fraction and other left ventricular disorders underlying water accumulation in the lungs.
This study has several limitations. The main limitation depends on the cross-sectional design, which prevents us from making a causal interpretation of our findings. Another limitation is the lack of an objective indicator of muscular strength and physical performance. We used a well validated but self-reported scale of physical functioning. However, the subjective perception by patients of their physical functioning is one of the most important aspects of quality of life; in a previous study in CKD patients, we documented that physical functioning by the KDQOL-SF has a fairly good agreement with actual physical activity as measured by a step counter (7). A proportion of screened patients did not qualify for inclusion and our study population was racially and ethnically homogenous, with a proportion of patients with diabetes less than in the US Renal Data System. Ours was a typical sample of patients being treated in European centers. Indeed, patients considered into the study are fully comparable with those included in the European Renal Association – European Dialysis and Transplant Association registry for relevant prognostic factors such as age (66 years versus 64 years), sex (61% versus 64%), and diabetes (28% versus 27%).
In conclusion, our study generates the hypothesis that symptomatic as well as asymptomatic lung congestion contributes to poor physical performance in stage 5 CKD patients on hemodialysis. Because lung congestion is a modifiable risk factor, intervention studies are warranted to test whether treating asymptomatic pulmonary congestion may translate into improved physical functioning in hemodialysis patients.
Disclosures
None.
Acknowledgments
Collaborators of the Lung Comets Cohort Working Group are as follows: Giovanni Alati, Rosalia Boito, Graziella Bonanno, Simonetta Cassani, Antonio Chippari, Teresa Cicchetti, Anna Clementi, Maurizio Garozzo, Domenico Logozzo, Rosita Lucà, Domenico Mancuso, Francesco Mollica, Giuseppe Natale, and Arcangelo Sellaro.
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
See related editorial, “Pulmonary Congestion in Hemodialysis: An Old Chestnut Worth Screening For?,” on pages 1279–1281.
References
- 1.Kaysen GA, Larive B, Painter P, Craig A, Lindsay RM, Rocco MV, Daugirdas JT, Schulman G, Chertow GM, FHN Trial Group : Baseline physical performance, health, and functioning of participants in the Frequent Hemodialysis Network (FHN) trial. Am J Kidney Dis 57: 101–112, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Procaccini DA, Angelini P, Aucella F, Avanzi C, Brusasco S, Carta G, Cataldi G, Cogoni G, D’Agostino F, d’Elia F, Del Mastro G, Dell’aquila R, De Min AM, Feliciangeli G, Freddi P, Gallucci M, Giannico G, Gigante B, Infante B, Ktena M, Manno C, Marseglia CD, Navarra A, Pappani A, Pompa G, Querques M, Russo D, Sacchetti A, Sechi MA, Specchio A, Stallone C, Virgilio M, Apulia Nephrology Quality Of Life Study : [Health-related quality of life in patients with chronic kidney disease]. G Ital Nefrol 25: 694–701, 2008 [PubMed] [Google Scholar]
- 3.Merkus MP, Jager KJ, Dekker FW, Boeschoten EW, Stevens P, Krediet RT, The Necosad Study Group : Quality of life in patients on chronic dialysis: Self-assessment 3 months after the start of treatment. Am J Kidney Dis 29: 584–592, 1997 [DOI] [PubMed] [Google Scholar]
- 4.Feroze U, Noori N, Kovesdy CP, Molnar MZ, Martin DJ, Reina-Patton A, Benner D, Bross R, Norris KC, Kopple JD, Kalantar-Zadeh K: Quality-of-life and mortality in hemodialysis patients: Roles of race and nutritional status. Clin J Am Soc Nephrol 6: 1100–1111, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Anand S, Chertow GM, Johansen KL, Grimes B, Kurella Tamura M, Dalrymple LS, Kaysen GA: Association of self-reported physical activity with laboratory markers of nutrition and inflammation: The Comprehensive Dialysis Study. J Ren Nutr 21: 429–437, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Anand S, Kaysen GA, Chertow GM, Johansen KL, Grimes B, Dalrymple LS, Kurella Tamura M: Vitamin D deficiency, self-reported physical activity and health-related quality of life: The Comprehensive Dialysis Study. Nephrol Dial Transplant 26: 3683–3688, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Enia G, Tripepi R, Panuccio V, Torino C, Garozzo M, Battaglia GG, Zoccali C: Pulmonary congestion and physical functioning in peritoneal dialysis patients. Perit Dial Int 32: 531–536, 2012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Van Biesen W, Williams JD, Covic AC, Fan S, Claes K, Lichodziejewska-Niemierko M, Verger C, Steiger J, Schoder V, Wabel P, Gauly A, Himmele R; EuroBCM Study Group: Fluid status in peritoneal dialysis patients: The European Body Composition Monitoring (EuroBCM) study cohort. PLoS One 6: e17148, 2011 [DOI] [PMC free article] [PubMed]
- 9.Enia G, Mallamaci F, Benedetto FA, Panuccio V, Parlongo S, Cutrupi S, Giacone G, Cottini E, Tripepi G, Malatino LS, Zoccali C: Long-term CAPD patients are volume expanded and display more severe left ventricular hypertrophy than haemodialysis patients. Nephrol Dial Transplant 16: 1459–1464, 2001 [DOI] [PubMed] [Google Scholar]
- 10.Zoccali C, Benedetto FA, Mallamaci F, Tripepi G, Giacone G, Cataliotti A, Seminara G, Stancanelli B, Malatino LS: Prognostic value of echocardiographic indicators of left ventricular systolic function in asymptomatic dialysis patients. J Am Soc Nephrol 15: 1029–1037, 2004 [DOI] [PubMed] [Google Scholar]
- 11.Klersy C, Callegari A, Giorgi I, Sepe V, Efficace E, Politi P, Pavia Working Group on QoL in Organ Transplant : Italian translation, cultural adaptation and validation of KDQOL-SF, version 1.3, in patients with severe renal failure. J Nephrol 20: 43–51, 2007 [PubMed] [Google Scholar]
- 12.Apolone G, Mosconi P: The Italian SF-36 Health Survey: Translation, validation and norming. J Clin Epidemiol 51: 1025–1036, 1998 [DOI] [PubMed] [Google Scholar]
- 13.Gargani L: Lung ultrasound: A new tool for the cardiologist. Cardiovasc Ultrasound 9: 6, 2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mallamaci F, Benedetto FA, Tripepi R, Rastelli S, Castellino P, Tripepi G, Picano E, Zoccali C: Detection of pulmonary congestion by chest ultrasound in dialysis patients. JACC Cardiovasc Imaging 3: 586–594, 2010 [DOI] [PubMed] [Google Scholar]
- 15.Panuccio V, Enia G, Tripepi R, Torino C, Garozzo M, Battaglia GG, Marcantoni C, Infantone L, Giordano G, De Giorgi ML, Lupia M, Bruzzese V, Zoccali C: Chest ultrasound and hidden lung congestion in peritoneal dialysis patients. Nephrol Dial Transplant 27: 3601–3605, 2012 [DOI] [PubMed] [Google Scholar]
- 16.Volpicelli G, Elbarbary M, Blaivas M, Lichtenstein DA, Mathis G, Kirkpatrick AW, Melniker L, Gargani L, Noble VE, Via G, Dean A, Tsung JW, Soldati G, Copetti R, Bouhemad B, Reissig A, Agricola E, Rouby JJ, Arbelot C, Liteplo A, Sargsyan A, Silva F, Hoppmann R, Breitkreutz R, Seibel A, Neri L, Storti E, Petrovic T, International Liaison Committee on Lung Ultrasound (ILC-LUS) for International Consensus Conference on Lung Ultrasound (ICC-LUS) : International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med 38: 577–591, 2012 [DOI] [PubMed] [Google Scholar]
- 17.Postorino M, Marino C, Tripepi G, Zoccali C, Calabrian Registry of Dialysis and Transplantation : Prognostic value of the New York Heart Association classification in end-stage renal disease. Nephrol Dial Transplant 22: 1377–1382, 2007 [DOI] [PubMed] [Google Scholar]
- 18.Daugirdas JT: Second generation logarithmic estimates of single-pool variable volume Kt/V: An analysis of error. J Am Soc Nephrol 4: 1205–1213, 1993 [DOI] [PubMed] [Google Scholar]
- 19.van den Beukel TO, Siegert CE, van Dijk S, Ter Wee PM, Dekker FW, Honig A: Comparison of the SF-36 Five-item Mental Health Inventory and Beck Depression Inventory for the screening of depressive symptoms in chronic dialysis patients. Nephrol Dial Transplant 27: 4453–4457, 2012 [DOI] [PubMed] [Google Scholar]